Abstract
Concomitant type 2 diabetes and chronic kidney disease increases the risk of heart failure. Recent studies demonstrate beneficial effects of sodium-glucose cotransporter 2 (SGLT2) inhibitors on chronic kidney disease progression and heart failure hospitalization in patients with and without diabetes. In addition to inhibiting glucose reabsorption, SGLT2 inhibitors decrease proximal tubular sodium reabsorption, possibly leading to transient natriuresis. We review the hypothesis that SGLT2 inhibitor’s natriuretic and osmotic diuretic effects mediate their cardioprotective effects. The degree to which these benefits are related to changes in sodium, independent of the kidney, is currently unknown. Aside from effects on osmotically active sodium, we explore the intriguing possibility that SGLT2 inhibitors could also modulate nonosmotic sodium storage. This alternative hypothesis is based on emerging literature that challenges the traditional 2-compartment model of sodium balance to provide support for a 3-compartment model that includes the binding of sodium to glycosaminoglycans, such as those in muscles and skin. This recent research on nonosmotic sodium storage, as well as direct cardiac effects of SGLT2 inhibitors, provides possibilities for other ways in which SGLT2 inhibitors might mitigate heart failure risk. Overall, we review the effects of SGLT2 inhibitors on sodium balance and sensitivity, cardiac tissue, interstitial fluid and plasma volume, and nonosmotic sodium storage.
Keywords: Heart failure, natriuresis, nonosmotic sodium, SGLT2 inhibitors
Lay summary
SGLT2 inhibitors have cardiovascular benefits that include HF outcomes in patients with and without diabetes. Because the underlying mechanisms are only partly explained by improvements in BP, body weight, or glucose control, other mechanisms have been proposed. We focus here on a central role for effects on sodium as underlying the positive benefits of SGLT2 inhibitors in HF. We explore the new (although still unconfirmed) idea that SGLT2 inhibitors exert some of their positive effects by affecting nonosmotic sodium (ie, sodium bound to muscles and skin and not dissolved in the blood).
SGLT2 inhibitors have emerged as a class of drugs, previously prescribed for patients with T2D, that have in more recent years been shown to have substantial heart and kidney clinical benefits in patients with and without T2D.
The degree to which these benefits are related to kidney-independent changes in sodium homeostasis is currently unknown.
A better understanding of the nonosmotic mechanisms underpinning the benefits of SGLT2 inhibition on HF (with reduced or preserved left ventricular ejection fraction) may allow researchers to assess the effects of SGLT2 inhibitors in combination with other treatments that affect sodium balance.
Type 2 diabetes (T2D) is an established risk factor for ischemic cardiovascular disease (CVD) and heart failure (HF).1 The risks of ischemic CVD and HF are increased with albuminuria and/or impaired kidney function. Although in recent decades cardiovascular outcomes have improved for adults with or without T2D, decreasing the burden associated with HF by treating classical cardiovascular risk factors has proven to be difficult and thus remains a major public health priority.2 Accordingly, the introduction of sodium-glucose cotransporter 2 (SGLT2) inhibitors offers promise to mitigate cardiorenal disease in people with or without T2D. However, to better understand the role of these drugs in the cardiovascular system, it is important to define their mechanism of action on the cardiorenal axis.
The kidney contributes to glucose homeostasis by actively reabsorbing nearly all of the filtered glucose in the proximal tubule. Although the kinetics of renal glucose reabsorption were first described nearly 90 years ago, it took until the early 1970s to demonstrate that glucose reabsorption occurs in the proximal tubule through 2 distinct sodium-glucose cotransport systems. Shortly thereafter, 2 SGLTs (SGLT1 and SGLT2) were discovered.3 SGLT1 is a high-affinity, low-capacity transporter responsible for approximately 10% of the renal glucose reabsorption; SGLT2 is a low-affinity, high-capacity transporter responsible for approximately 90% of the renal glucose reabsorption.4,5 Together, these transporters are thought to be responsible for total renal glucose reabsorption. In addition to glucose reabsorption, SGLT1 and SGLT2 also facilitate concomitant sodium reabsorption. Approximately two-thirds of the total kidney sodium reabsorption occurs in the proximal tubule, although the extent to which this reabsorption is mediated by SGLT1 and SGLT2 presently remains unknown.6
SGLT2 inhibitors were granted marketing authorization in 2014 as glucose-lowering drugs, and work by inducing glucosuria. Through their mechanism of action, the glucose-lowering effects of SGLT2 inhibitors in patients with chronic kidney disease (CKD) are modest.7 However, these drugs have recently received considerable attention in large cardiovascular safety trials owing to favorable HF and renal benefits. For example, in patients with T2D and high CVD risk, the EMPA-REG OUTCOME trial demonstrated a 35% relative risk reduction in hospitalization for HF for empagliflozin vs placebo,8 and the CANVAS Program with canagliflozin demonstrated beneficial cardiovascular and renal outcomes.9 The CREDENCE trial, which studied the effects of canagliflozin in patients with T2D and diabetic kidney disease,10 reported a decrease in HF hospitalization by 39% (95% confidence interval 20%–53%), in addition to attenuating the loss of kidney function. By comparison, studies assessing the cardiovascular effects of glucose lowering per se, when mediated by other agents, had not demonstrated similar benefits, while HF outcomes may even be worsened by some glucose-lowering drugs.11,12
Since the publication of the results of these and other cardiovascular safety trials (Fig. 1), as well as real-world effectiveness studies (eg, CVD-REAL13), investigators and clinicians have considered a variety of potential mechanisms underlying the cardiorenal benefits of SGLT2 inhibition. It is generally agreed that (modest) decreases in blood pressure (BP), glucose concentrations, body weight, and serum urate concentrations do not fully explain the observed cardiovascular benefits.14,15 Although the exact pathways are not fully understood, the purpose of this article is to describe the effects of SGLT2 inhibitors on sodium handling beyond diuresis and natriuresis per se, and to discuss the proposed cardiovascular consequences of changes in sodium sensitivity and balance, including direct sodium-related cardiac effects, effects on interstitial fluid and plasma volume, and changes in nonosmotic sodium storage.
Fig. 1.
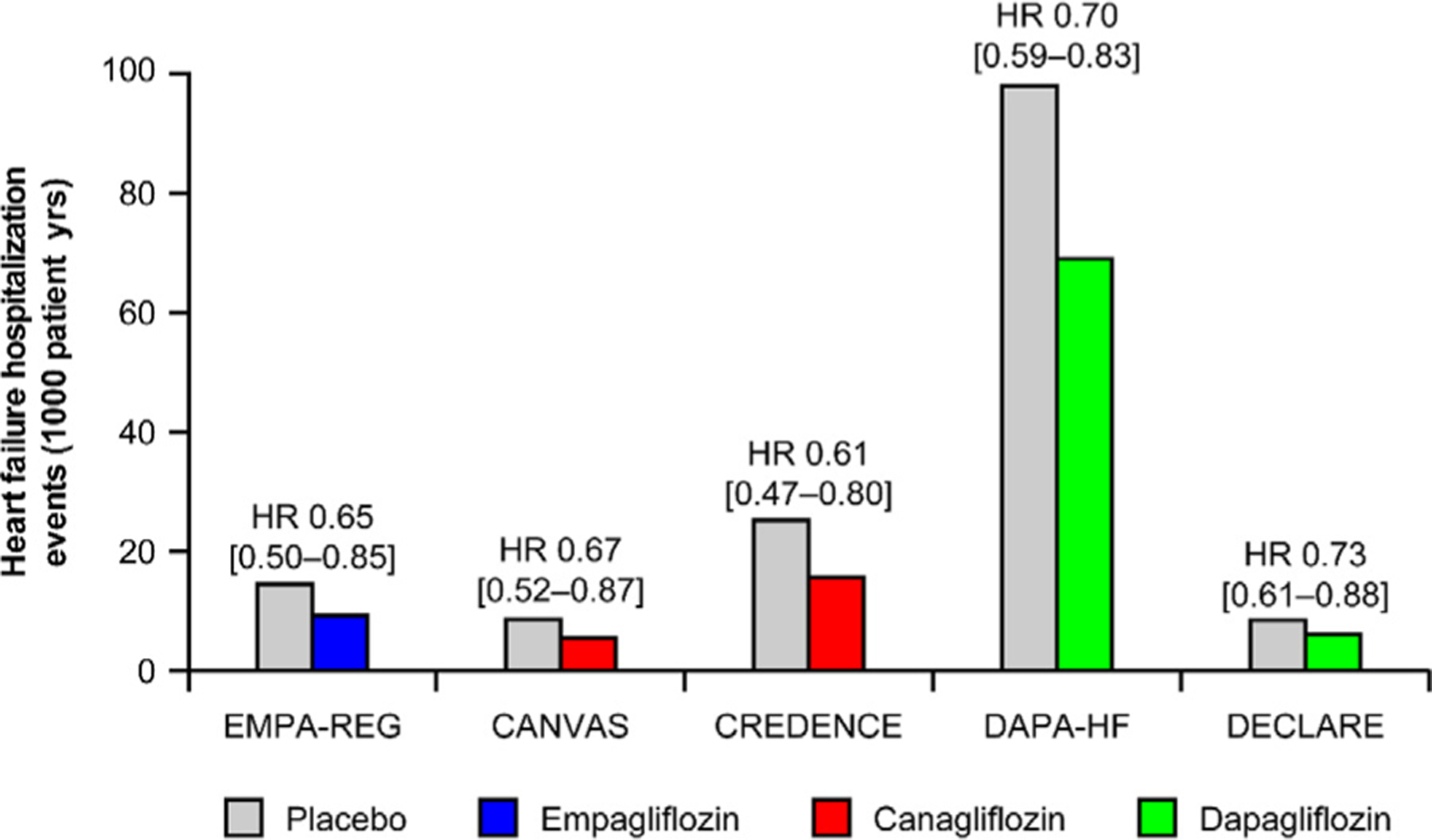
Summary of hospitalization for heart failure results from recent cardiovascular safety trials of sodium-glucose cotransporter 2 (SGLT2) inhibitors. Values in brackets are 95% confidence intervals. Values were derived from the trials’ publications; slightly different terminology was used in the trials to describe heart failure hospitalization: EMPA-REG and DECLARE used “rate per 1000 pt-yrs,” CANVAS used “number of participants per 1000 pt-yrs,” and CREDENCE and DAPA-HF used “events per 1000 [or 100 for DAPA-HF] pt-yrs.” CANVAS, CANagliflozin cardioVascular Assessment Study; CREDENCE, Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation; DAPA-HF, Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure; DECLARE, Dapagliflozin Effect on Cardiovascular Events—Thrombolysis in Myocardial Infarction 58; EMPA-REG, EMPAgliflozin cardiovascular outcome event trial in type 2 diabetes mellitus patients—Removing Excess Glucose; HR, hazard ratio.
Sodium Balance, Sodium Sensitivity, and Cardiovascular Health
Given the well-known role of sodium in cardiovascular health, we next address recent research that challenges traditional views about sodium homeostasis, with potential implications for the pathophysiology and treatment of HF, including the pathophysiological role of interstitial sodium in HF, as reviewed elsewhere.16 In most adult populations, the average salt intake well exceeds the approximately 5-g daily limit recommended by the World Health Organization.17 Excessive salt intake has been linked to hypertension, CVD,18 and CKD. Although the pathogenesis underlying the relationship between excessive salt intake and cardiorenal complications remains debated, the leading hypothesis for decades has been that—in so-called salt-sensitive individuals—excess sodium intake with concomitant impaired renal sodium excretion results in extracellular volume expansion and hypertension.19 In patients with CKD, a lower glomerular filtration rate and activation of the renin–angiotensin–aldosterone system result in an increased venous pressure, decreased renal perfusion, decreased cardiac output, and ultimately HF. The net results of these pathophysiological changes include further sodium and water retention with activation of the renin–angiotensin–aldosterone system and the sympathetic nervous system. However, carefully designed sodium-balance studies in so-called salt-resistant participants, that is, individuals in whom increased salt intake does not increase BP or body water/weight, show that much of the ingested sodium excess is in fact not excreted in the urine.20 Rather, these studies have proposed that sodium may be stored nonosmotically (ie, without altering the extracellular volume) at extrarenal locations, which serve to act as an osmotic sodium buffer. For example, daily rhythmic fluctuations in total body sodium content were found with large variations in 24-hour urinary sodium excretion, despite a fixed sodium intake, which suggests nonosmotic sodium accumulation and the storage of salt in a third body compartment.20 Osmotic excretion of significant amounts of sodium has also been shown in healthy people after hypertonic saline infusion.21 Using 23Na magnetic resonance imaging, muscle and skin were shown to contain considerable amounts of sodium without associated water retention.22 Another compartment that binds sodium in a nonosmotic manner and thus could influence extracellular volume and BP regulation is the endothelial surface layer, or glycocalyx, located on the luminal side of the vascular endothelium. The endothelial surface layer has abundant negatively charged glycosaminoglycans and is in direct contact with circulating blood sodium and glucose. These glycosaminoglycans have been shown to display avid sodium-binding capacity.23 Emerging literature has challenged the traditional 2-compartment model of sodium balance providing support to a 3-compartment model that includes the binding of sodium to glycosaminoglycans, such as those in the muscles and skin. Endothelial surface layer damage has been observed in patients with T2D24 and CKD,25 which could explain (at least in part) the salt sensitivity observed in this population. Other functions of the endothelial surface layer include the production of nitric oxide owing to shear stress, and the formation of a barrier to prevent circulating inflammatory cells from reaching underlying tissues. Consistent with these data, restoration of the endothelial surface layer by sulodexide, a mixture of endothelial surface layer constituents, has been shown to decrease BP.26
Although nonosmotic sodium storage seems beneficial in the short term, saturated sodium depots in the skin have been linked to both hypertension and left ventricular hypertrophy.27 Additionally, high vs low dietary sodium intake has been shown to increase the number of monocytes,28 which could trigger an inflammatory response. Furthermore, inflammation of the interstitium might drive microvascular and macrovascular stiffening and impair endothelial function.29
Nonosmotic buffering of sodium in tissues, such as glycocalyx, may decrease the adverse hemodynamic effects of sodium in the short term, although long-standing sodium overload may have deleterious consequences for the cardiovascular system. For example, some evidence suggests that sodium accumulation in the endothelial glycocalyx could lead to arterial stiffness.30 Accordingly, strategies to decrease tissue and interstitial sodium by facilitating renal sodium excretion may enhance cardiovascular health, although we stress that the concept of nonosmotic sodium storage remains theoretical; efforts are undertaken to more definetly determine its presence and role. We next discuss other mechanisms behind the putatively central role of sodium in mediating the favorable cardiorenal effects of SGLT2 inhibition.
Effects of SGLT2 Inhibitors on Sodium Balance
Although SGLT2 inhibitors were designed primarily to decrease plasma glucose concentrations, it is evident that there is concomitant inhibition of proximal tubular sodium uptake with the inhibition of glucose reabsorption. Data showing inhibition of lithium reabsorption, as a marker for proximal tubular function, support this notion.31 However, studies of proximal tubular sodium absorption have not been conducted in people with HF. Initial natriuresis is thought to contribute to the osmotic diuresis, which drives the increased urine output associated with acute SGLT2 inhibition, as shown in some32,33 but not all studies.31 The placebo-controlled RECEDE-CHF trial conducted in patients with T2D and HF demonstrated a significant increase in 24-hour urine volume without an increase in urinary sodium concentration when empagliflozin was used in combination with a loop diuretic.34 Although RECEDE-CHF did not find a significant increase in the fractional excretion of sodium with empagliflozin, another study did,32 a difference possibly explainable by the different time points studied between the 2 studies as well as differences in sodium intake at baseline. Notably, the major limitation of current studies showing natriuresis with SGLT2 inhibition is that study participants were not on a fixed sodium diet. The DAPASALT study, by contrast, was conducted in patients (N = 17) with T2D and preserved kidney function on a fixed sodium diet.31 The study participants received dapagliflozin and had 24-hour urine collected prior to treatment, after acute dosing, after 2 weeks of treatment, and 3 days after treatment cessation, but changes in natriuresis or plasma volume were not found.31 Whereas the bulk of sodium is reabsorbed in the proximal tubule, it is unclear to what extent SGLT2 transporters contribute to total sodium reabsorption in absolute terms. This phenomenon may be particularly relevant in people with T2D, in whom there is increased glucose flux through the SGLT2 transporters owing to chronic tubular hyperglycemia. It is likely that SGLT2 transporters also interact functionally with Na+/H+ exchanger isoform 3 in the proximal tubule (Fig. 2).
Fig. 2.
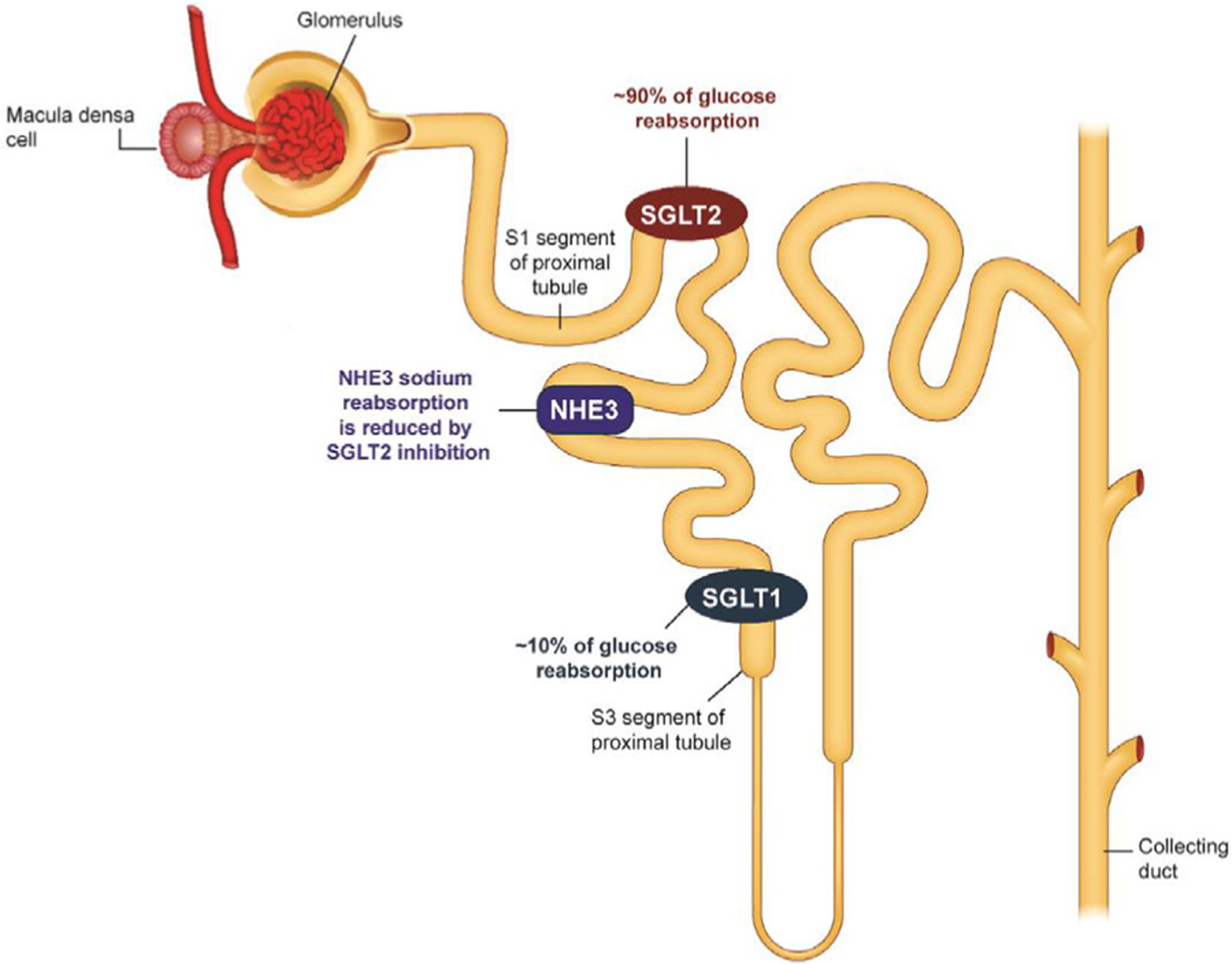
Schematic of a nephron and sodium-glucose cotransporter (SGLT) inhibitors. NHE3, Na+/H+ exchanger isoform 3; SGLT2, sodium-glucose cotransporter 2.
As such, SGLT2 inhibition is associated with marked inhibition of Na+/H+ exchanger isoform 3, even in the absence of glucose, which is likely to account for a significant proportion of the natriuresis observed with agents of this class.35 Another study (conducted in various animal models and human cells) did not find that SGLT2 inhibition with empagliflozin inhibited the ubiquitously expressed plasma membrane Na+/H+ exchanger Na+/H+ exchanger 1, however.36 Limited data exist describing proximal sodium reabsorption in humans with T2D before and during SGLT2 inhibitor therapy.31 The kidneys rapidly adapt to the initial natriuresis by matching sodium excretion to sodium intake, maintaining a neutral sodium balance. Therefore, sodium excretion is usually not altered with prolonged treatment,37,38 likely because of compensatory sodium reabsorption at more distal tubular segments. Where the additional sodium is being absorbed remains unclear. Any increase in renal sodium absorption induced by SGLT2 inhibition is likely distal to the macula densa, because the decrease in the estimated glomerular filtration rate induced by SGLT2 inhibition through tubuloglomerular feedback is thought to be driven by increased sodium and chloride concentrations detected by the macula densa. Gene expression analyses of key sodium transporters located in the distal tubule could expand knowledge of these compensatory pathways in humans.
The natriuresis and osmotic diuresis associated with SGLT2 inhibition has been shown in some studies to be associated with a modest decrease in plasma volume,39–41 although a study of canagliflozin treatment found this decrease to be attenuated at week 12.42 The decrease in plasma volume is reflected by an increase in hematocrit and radioactive-labeled albumin,43 which is sustained during prolonged treatment but is reversed after cessation of therapy. A mathematical model-based analysis to assess the fluid effects of dapagliflozin and the loop diuretic bumetanide was recently reported, based on data acquired in a healthy volunteer study of these 2 drugs.40,41 A key finding of this analysis was that a similar decrease in the interstitial volume occurred in response to dapagliflozin as compared with that observed with bumetanide, but a smaller decrease was observed in plasma volume, which may result in improved tissue perfusion and less acute kidney injury incidence with SGLT2 inhibition compared with loop diuretics. Such studies highlight the differences between SGLT2 inhibitors and loop diuretics. However, the hypothesis that SGLT2 inhibitors decrease the interstitial volume is based on modeling assumptions and not on direct measurements. Further, although the mechanism by which SGLT2 inhibitors decrease the interstitial volume is not known, osmotic diuresis resulting from increased urinary glucose excretion might lead to more electrolyte-free water clearance.32,44 Possibly because of their different site of action in the tubular system, these drug classes have markedly different effects on potassium, uric acid, glucose, renal hemodynamics, and markers of the renin–angiotensin–aldosterone–system (Fig. 3).40,41 SGLT2 inhibitors have consistently been shown to modestly increase renin levels owing to their diuretic/natriuretic effect.45 Decreases in interstitial fluid volume may contribute to the cardiovascular benefits observed in recent cardiovascular safety trials, particularly vis-à-vis HF.46 A mediation analysis of the EMPA-REG OUTCOME trial found that change in hematocrit explained 51.8% of the effect of empagliflozin vs placebo on the risk of cardiovascular death.14 Increased hematocrit has also been observed in patients with T2D without HF47,48 and in patients with HF with reduced ejection fraction (HFrEF) (in which 42% had a history of diabetes at baseline).49
Fig. 3.
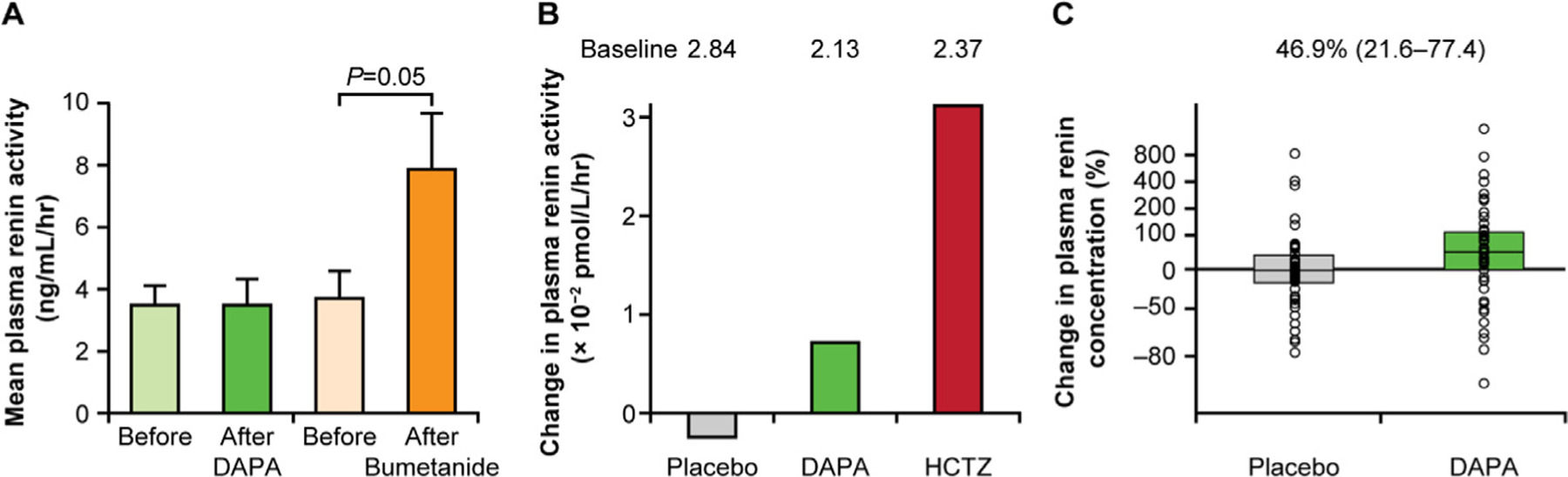
Plasma renin activity with sodium-glucose cotransporter 2 (SGLT2) inhibition. (A) Increase in plasma renin activity by 117% after 1 week of bumetanide treatment in healthy volunteers (adapted from Wilcox et al. 201841; see Supplementary Fig. S6; available at: https://www.ahajournals.org/doi/10.1161/JAHA.117.007046; used under CC BY-NC 4.0; text slightly updated, parts of panels A and B combined, layout altered, and color added). (B) Week 12 change from baseline in plasma renin activity in patients with type 2 diabetes (T2D) treated with dapagliflozin (DAPA) or hydrochlorothiazide (HCTZ) (based on data from: Heerspink et al. 201343, Table 2). (C) Changes in plasma volume markers during DAPA treatment vs placebo treatment in patients with T2D (P < .01) (adapted from: Eickhoff et al 2019,50 Fig. 2, fourth panel from the left; available at: https://www.mdpi.com/2077-0383/8/6/779; used under CC BY 4.0; text slightly updated, layout altered, and color added).
Changes in hematocrit could reflect hemodynamic changes related to plasma volume contraction, which may decrease ventricular filling pressures and cardiac workload.14,43 It is unclear whether the beneficial HF outcomes are partly because of a direct increase in hematocrit or attributable to factors underlying the increase in hematocrit, although the latter is more likely. Changes in hematocrit are unlikely to be explained by changes in plasma volume alone. The natriuretic response induced by SGLT2 inhibitors might restore the physiologic tubuloglomerular feedback, thus decreasing the intraglomerular pressure, as stated elsewhere in this article.51 Changes in kidney physiology could then lead to changes in renal oxygen metabolism that affect erythropoietin production, although this notion remains speculative. An increase in erythropoiesis, resulting from a decrease in distal tubular oxygen content secondary to increased workload, could contribute to the increase in hematocrit and could be a marker of a beneficial action of these agents, given that increased erythropoietin levels may contribute to improved myocardial oxygen delivery.47 It is partly through this possible sequence of events that SGLT2 inhibitors could benefit the heart. Treatment with dapagliflozin has been shown to suppress hepcidin levels.52 Given that SGLT2 inhibition has been shown to decrease adipose tissue inflammation in a murine model of obesity,53 this point also raises the intriguing possibility that SGLT2 inhibition might decrease hepcidin levels via an anti-inflammatory effect, subsequently improving anemia associated with HFrEF. The effects of SGLT2 inhibitors on interstitial fluid and circulatory volumes should be confirmed, and downstream effects explored, to better understand their cardioprotective mechanisms.
Aside from inducing changes in osmotically active sodium, SGLT2 inhibitors could also modulate nonosmotic sodium storage, although less evidence is available to support this hypothesis.40 In a porcine model of HF, empagliflozin decreased skin sodium content and interstitial fluid volume to a greater extent than did furosemide.54 In a study in which 51 participants with T2D were treated with dapagliflozin or placebo for 6 weeks, sodium content in the skin and muscles of the lower leg was measured by 23Na-magnetic resonance imaging.55 Serum sodium, 24-hour urinary sodium excretion, and muscle sodium content were not significantly changed at 6 weeks with dapagliflozin treatment; however, skin sodium content was decreased. Similar studies in patients with CKD, who commonly manifest extracellular volume overload, should be conducted.
In summary, SGLT2 inhibitors were initially designed to reduce tubular glucose reabsorption, thereby lowering serum glucose and glycosylated hemoglobin. However, SGLT2 inhibitors might also induce a natriuretic response associated with diuresis, with a more pronounced effect on interstitial fluid compared with plasma volume. Natriuresis is unlikely to completely explain the benefits of SGLT2 inhibition, because it is transient (ie, likely present in the first 24 hours after first dosing) and modest when compared with diuretics,33 and recent findings may even call for a reexamination of this hypothesis.31 Accordingly, other consequences of SGLT2 inhibitor induced alterations in sodium handling, that is, changes in systemic hemodynamics and the vascular system, as well as potential nonosmotic sodium storage, may also contribute to the observed cardiovascular benefit (Fig. 4).
Fig. 4.
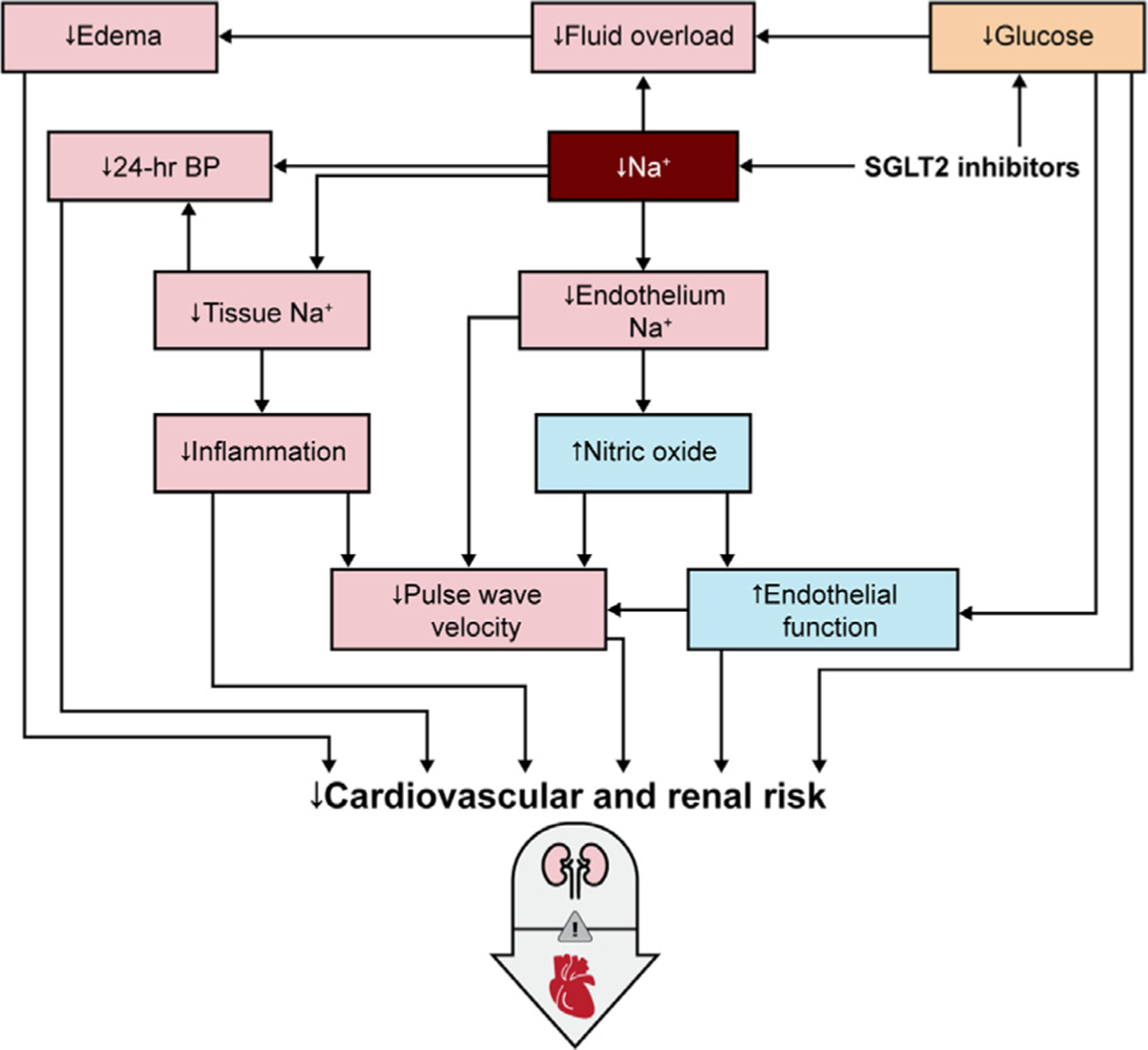
Putative sodium-centric mechanisms of benefit of sodium-glucose cotransporter 2 (SGLT2) inhibitors in people with type 2 diabetes (T2D) and cardiorenal disease. BP, blood pressure.
Effects of SGLT2 Inhibition on Arterial Stiffness and Endothelial Function
SGLT2 inhibition demonstrates durable BP reduction, which may partly account for SGLT2 inhibitors’ cardiorenal benefits and may be partly driven by reductions in body sodium content.8,9,56 SGLT2 inhibitors have also been shown to decrease arterial stiffness and improve endothelial function. Arterial stiffness, determined largely by the elastin-to-collagen ratio in the vessel wall, is associated with the risk for cardiovascular events. Accordingly, noninvasive measures of central and peripheral arterial stiffness can serve as useful surrogate markers to determine the effectiveness of pharmacotherapies in improving cardiovascular health.57
Arterial stiffness, measured by pulse wave velocity, decreased in response to 8 weeks of empagliflozin 25 mg in an open-label, prospective clinical trial in young adults with type 1 diabetes mellitus.58 Consistent with these data, a post hoc analysis from phase III trials in adults with T2D demonstrated decreased arterial stiffness, as assessed by pulse pressure and ambulatory arterial stiffness index, and arterial resistance as measured by mean arterial pressure, in response to empagliflozin.59 In a pilot study of 16 adults with T2D, 2 days of dapagliflozin 12.5 mg was shown to increase flow-mediated dilatation and to decrease the pulse wave velocity and renal resistive index, independent of decreases in the BP.60 These data suggest that the effects of SGLT2 inhibition on systemic and renal vascular stiffness and on endothelial dysfunction are acute and persistent. Empagliflozin or dapagliflozin also restore nitric oxide production by human endothelial cells, which may contribute to the beneficial effects of SGLT2 inhibition on endothelial function, although such results were not in vivo and used high empagliflozin concentrations (1 µM).61,62 In vivo data from a porcine model of patients with HF without diabetes indicate that empagliflozin improves nitric oxide signaling and diastolic function.63 A post hoc analysis of pooled data from 4 phase III studies demonstrated that canagliflozin attenuated pulse pressure and mean arterial pressure in adults with T2D.64 Finally, trials have also demonstrated improvement in endothelial function by reactive hyperemia peripheral arterial tonometry65 and flow-mediated dilatation66 in response to dapagliflozin in adults with T2D. The mechanisms by which SGLT2 inhibition decreases arterial stiffness and improves endothelial dysfunction are not fully understood but may be related to changes in sodium exposure.67,68 The effect of SGLT2 inhibition on arterial stiffness is particularly relevant to the glycocalyx, which, as noted elsewhere in this article, functions as a nonosmotic sodium buffer and can be damaged by sodium and glucose overload.
Direct Sodium-Related Cardiac Effects of SGLT2 Inhibition
The full mechanisms of action of SGLT2 inhibition remain incompletely understood. Given the potentiating effect of increased myocardial intracellular sodium concentrations in HF, various lines of research have investigated the direct effects of SGLT2 inhibitors on the heart in in vitro model systems. Direct effects of SGLT2 inhibition on sodium concentrations in cardiomyocytes have been identified, independent of systemic effects produced via the kidney, which is remarkable given the absence of SGLT2 receptors in the heart. For example, empagliflozin was shown to decrease cardiac cytoplasmic sodium concentration via cardiac Na+/H+ exchanger inhibition.69 Apart from sodium-mediated effects, other direct effects, reviewed elsewhere,70 are possibly also involved in the long-term cardioprotective effects of SGLT2 inhibitors but are beyond the scope of the present review.
Moving From Mechanisms to Clinical Outcomes
As briefly stated elsewhere in this article, SGLT2 inhibition has yielded remarkable effects on cardiovascular (particularly HF) and kidney outcomes in large clinical trials (Fig. 1).8–10,56,71,72 These results were confirmed in cardiovascular safety trials in patients with T2D and established CVD or at high risk for cardiovascular events in the DECLARE-TIMI 5856 and CANVAS9 studies, which also demonstrated improvement in HF outcomes without previous documented HF or established CVD at baseline. Because echocardiography was not performed routinely to assess the ejection fraction in these cardiovascular safety trials, any differential effect of SGLT2 inhibitors on patients with HF with preserved or reduced ejection fraction merits further study. Based on the beneficial HF findings in cardiovascular safety trials, dedicated HF trials were designed to characterize the effects of SGLT2 inhibition in preventing adverse outcomes in patients with HF. In the DEFINE-HF study, treatment with dapagliflozin indeed decreased symptoms and improved the quality of life in 263 patients with HFrEF with or without T2D, despite no significant decrease in levels of N-terminal pro B-type natriuretic peptide.73 In the DAPA-HF trial, which recruited 4744 patients with or without T2D with New York Heart Association functional class II, III, or IV HF and an ejection fraction of 40% or less, dapagliflozin decreased the primary end point of a composite of cardiovascular death or worsening HF (hospitalization or an urgent visit resulting in intravenous therapy for HF) by 26% (95% confidence interval 15%–35%).72 No differences were observed between individuals with or without diabetes.72 Confirmation of beneficial effects in patients with HFrEF has come from the EMPEROR-Reduced study, which investigated the effects of empagliflozin on HF outcomes in patients with or without diabetes.74 Reverse left ventricular remodeling with SGLT2 inhibition has also been demonstrated in patients with HFrEF with75 or without T2D.76
The limited effect of SGLT2 inhibitors on atherothrombotic disease (11% reduction in MACE in a recent meta-analysis77), and the strong effects on HF and kidney outcomes with early divergence of group outcomes (benefits seen after 3 months), strongly point to a hemodynamic effect that relates to sodium balance as postulated here. The fact that SGLT2 inhibitors improve HF outcomes to the same extent in patients with HFrEF with and without diabetes is the strongest argument to date that these beneficial effects are completely glucose independent.
Several studies that might support or refute these hypotheses are currently ongoing. The DELIVER (dapagliflozin; NCT03619213) and EMPEROR-Preserved (empagliflozin; NCT03057951) studies investigate whether the decrease in HF hospitalizations extends to patients with HF with preserved ejection fraction and with normal or near normal kidney function (without albuminuria). The phase III DAPA-CKD trial, which was stopped early based on overwhelming efficacy, reported a hazard ratio for the composite of death from cardiovascular causes or hospitalization for HF of 0.71 (95% confidence interval 0.55−0.92, P = .009) as well as benefit on kidney outcomes, in patients with CKD with and without diabetes.71 Additionally, studies such as DAPACARD (NCT03387683) and ERADICATE-HF (NCT03416270) investigate the underlying mechanisms, focusing on myocardial substrate metabolism, sodium reabsorption, and plasma volumes.
Conclusion
This review has summarized data on the salutary effects of SGLT2 inhibition unrelated to glucose metabolism, including changes in sodium balance, sodium sensitivity, and direct sodium effects, on the heart and nonosmotic sodium stores, and hypothesized that these may partly contribute to improved HF outcomes. Importantly, however, data correlating either changes in extracellular sodium stores or direct cardiac effects with clinical outcomes are not available yet. Moreover, despite some support, the nonosmotic sodium hypothesis is novel and remains to be validated fully. A better understanding of the nonosmotic mechanisms underpinning the cardiorenal benefits of SGLT2 inhibition may allow researchers to assess the effects of SGLT2 inhibitors in combination with other drugs that affect sodium.
Acknowledgments
Editorial assistance was provided by Steven Tresker and Varun Kumar Pandey, PhD, both of Cactus Life Sciences (part of Cactus Communications), which was supported by AstraZeneca.
Supported by the Dutch Diabetes Foundation and EU Marie Curie program [research fellowship to DHvR]. This work was also supported by the National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases [grant number K23 DK116720–01, R01 DK129211, R21 DK129720; salary and research support to PB] and additionally, by the Thrasher Research Fund; the Juvenile Diabetes Research Foundation; the National Institute of Diabetes and Digestive and Kidney Diseases/Diabetic Complications Consortium; the International Society of Pediatric and Adolescent Diabetes; the Colorado Clinical & Translational Sciences Institute; and the Center for Women’s Health Research at University of Colorado [research support to PB].
Footnotes
Visual Take-Home Graphic
Sodium may occupy a central role in the many mechanisms by which SGLT2 inhibitors might benefit people with HF.
Disclosures
HJLH serves as a consultant for AbbVie, AstraZeneca, Boehringer Ingelheim, CSL Pharma, Fresenius, Gilead, Janssen, Merck, Mitsubishi Tanabe, Mundipharma, and Retrophin. PB has acted as a consultant for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Horizon Pharma, Novo Nordisk, and Sanofi and serves on the advisory board of XORTX. DHvR has acted as a consultant and received honoraria from Boehringer Ingelheim and Eli Lilly, Merck, Novo Nordisk, and Sanofi and has received research operating funds from AstraZeneca, Boehringer Ingelheim-Eli Lilly Diabetes Alliance, MSD, and Novo Nordisk. GMC has served as a consultant to AstraZeneca and has received research support from Janssen. DCW has acted as a consultant and/or received honoraria from Astellas, AstraZeneca, Boehringer Ingelheim, GlaxoSmithK-line, Janssen, Napp, Mitsubishi Tanabe, Mundipharma, and Vifor Fresenius. PJG and AML are employees and shareholders of AstraZeneca.
References
- 1.Kannel WB, Hjortland M, Castelli WP. Role of diabetes in congestive heart failure: the Framingham study. Am J Cardiol 1974;34:29–34. [DOI] [PubMed] [Google Scholar]
- 2.Rawshani A, Rawshani A, Franzén S, Sattar N, Eliasson B, Svensson AM, et al. Risk factors, mortality, and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2018;379:633–44. [DOI] [PubMed] [Google Scholar]
- 3.Hediger MA, Coady MJ, Ikeda TS, Wright EM. Expression cloning and cDNA sequencing of the Na+/glucose co-transporter. Nature 1987;330:379–81. [DOI] [PubMed] [Google Scholar]
- 4.DeFronzo RA, Norton L, Abdul-Ghani M. Renal, metabolic and cardiovascular considerations of SGLT2 inhibition. Nat Rev Nephrol 2017;13:11–26. [DOI] [PubMed] [Google Scholar]
- 5.Wright EM, Loo DD, Hirayama BA. Biology of human sodium glucose transporters. Physiol Rev 2011;91:733–94. [DOI] [PubMed] [Google Scholar]
- 6.Layton AT, Layton HE. A computational model of epithelial solute and water transport along a human nephron. PLoS Comput Biol 2019;15:e1006108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kelly MS, Lewis J, Huntsberry AM, Dea L, Portillo I. Efficacy and renal outcomes of SGLT2 inhibitors in patients with type 2 diabetes and chronic kidney disease. Postgrad Med 2019;131:31–42. [DOI] [PubMed] [Google Scholar]
- 8.Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015;373:2117–28. [DOI] [PubMed] [Google Scholar]
- 9.Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017;377:644–57. [DOI] [PubMed] [Google Scholar]
- 10.Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink HJL, Charytan DM, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med 2019;380:2295–306. [DOI] [PubMed] [Google Scholar]
- 11.Arnold SV, Inzucchi SE, Echouffo-Tcheugui JB, Tang F, Lam CSP, Sperling LS, et al. Understanding contemporary use of thiazolidinediones. Circ Heart Fail 2019;12:e005855. [DOI] [PubMed] [Google Scholar]
- 12.Scirica BM, Bhatt DL, Braunwald E, Steg PG, Davidson J, Hirshberg B, et al. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med 2013;369:1317–26. [DOI] [PubMed] [Google Scholar]
- 13.Kosiborod M, Cavender MA, Fu AZ, Wilding JP, Khunti K, Holl RW, et al. Lower risk of heart failure and death in patients initiated on sodium-glucose cotransporter-2 inhibitors versus other glucose-lowering drugs. Circulation 2017;136: 249–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Inzucchi SE, Zinman B, Fitchett D, Wanner C, Ferrannini E, Schumacher M, et al. How does empagliflozin reduce cardiovascular mortality? Insights from a mediation analysis of the EMPA-REG OUTCOME trial. Diabetes Care 2018;41:356–63. [DOI] [PubMed] [Google Scholar]
- 15.van Baar MJB, van Ruiten CC, Muskiet MHA, van Bloemendaal daal L, IJzerman RG, van Raalte DH. SGLT2 inhibitors in combination therapy: from mechanisms to clinical considerations in type 2 diabetes management. Diabetes Care 2018;41:1543–56. [DOI] [PubMed] [Google Scholar]
- 16.Nijst P, Verbrugge FH, Grieten L, Dupont M, Steels P, Tang WHW, et al. The pathophysiological role of intersitial sodium in heart failure. J Am Coll Cardiol 2015;65:378–88. [DOI] [PubMed] [Google Scholar]
- 17.Brown IJ, Tzoulaki I, Candeias V, Elliott P. Salt intakes around the world: implications for public health. Int J Epidemiol 2009; 38; 791–813. [DOI] [PubMed] [Google Scholar]
- 18.Strazzullo P, D’Elia L, Kandala NB, Cappuccio FP. Salt intake, stroke, and cardiovascular disease: meta-analysis of prospective studies. BMJ 2009;339:b4567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Choi HY, Park HC, Ha SK. Salt sensitivity and hypertension: a paradigm shift from kidney malfunction to vascular endothelial dysfunction. Electrolyte Blood Press 2015;13: 7–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rakova N, jüttner K, Dahlmann A, Schröder A, Linz P, Kopp C, et al. Long-term space flight simulation reveals infradian rhythmicity in human Na(+) balance. Cell Metab 2013;17:125–31. [DOI] [PubMed] [Google Scholar]
- 21.Olde Engberink RH, Rorije NM, van den Born BH, Vogt L. Quantification of nonosmotic sodium storage capacity following acute hypertonic saline infusion in healthy individuals. Kidney Int 2017;91:738–45. [DOI] [PubMed] [Google Scholar]
- 22.Kopp C, Linz P, Dahlmann A, Hammon M, Jantsch J, Müller DN, et al. 23Na magnetic resonance imaging-determined tissue sodium in healthy subjects and hypertensive patients. Hypertension 2013;61:635–40. [DOI] [PubMed] [Google Scholar]
- 23.Oberleithner H Two barriers for sodium in vascular endothelium? Ann Med 2012;44(suppl. 1):S143–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Broekhuizen LN, Lemkes BA, Mooij HL, Meuwese MC, Verberne H, Holleman F, et al. Effect of sulodexide on endothelial glycocalyx and vascular permeability in patients with type 2 diabetes mellitus. Diabetologia 2010;53:2646–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vlahu CA, Lemkes BA, Struijk DG, Koopman MG, Krediet RT, Vink H. Damage of the endothelial glycocalyx in dialysis patients. J Am Soc Nephrol 2012;23:1900–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Olde Engberink RH, Rorije NM, Homan van der Heide JJ, van den Born BJ, Vogt L. Role of the vascular wall in sodium homeostasis and salt sensitivity. J Am Soc Nephrol 2015;26:777–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schneider MP, Raff U, Kopp C, Scheppach JB, Toncar S, Wanner C, et al. Skin sodium concentration correlates with left ventricular hypertrophy in CKD. J Am Soc Nephrol 2017;28:1867–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yi B, Titze J, Rykova M, Feuerecker M, Vassilieva G, Nichiporuk I, et al. Effects of dietary salt levels on monocytic cells and immune responses in healthy human subjects: a longitudinal study. Transl Res 2015;166:103–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wiig H, Luft FC, Titze JM. The interstitium conducts extrarenal storage of sodium and represents a third compartment essential for extracellular volume and blood pressure homeostasis. Acta Physiol (Oxf) 2018:222. [DOI] [PubMed] [Google Scholar]
- 30.Oberleithner H, Peters W, Kusche-Vihrog K, Korte S, Schillers H, Kliche K, et al. Salt overload damages the glycocalyx sodium barrier of vascular endothelium. Pflugers Arch 2011;462:519–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Scholtes RA, Muskiet MHA, van Baar MJB, Greasley PJ, Karlsson C, Hammarstedt A, et al. Natriuretic effect of 2 weeks of dapagliflozin treatment in patients with type 2 diabetes and preserved kidney function during standardized sodium intake: results of the DAPASALT trial. Presented at the European Society of Cardiology Congress, August 29 – September 2, 2020, Amsterdam, the Netherlands. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Griffin M, Rao VS, Ivey-Miranda J, Fleming J, Mahoney D, Maulion C, et al. Empagliflozin in heart failure: diuretic and cardiorenal effects. Circulation 2020;142:1028–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tanaka H, Takano K, Iijima H, Kubo H, Maruyama N, Hashimoto T, et al. Factors affecting canagliflozin-induced transient urine volume increase in patients with type 2 diabetes mellitus. Adv Ther 2017;34:436–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mordi NA, Mordi IR, Singh JS, McCrimmon RJ, Struthers AD, Lang CC. Renal and cardiovascular effects of SGLT2 inhibition in combination with loop diuretics in patients with type 2 diabetes and chronic heart failure: the RECEDE-CHF trial. Circulation 2020;142:1713–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pessoa TD, Campos LC, Carraro-Lacroix L, Girardi AC, Malnic G. Functional role of glucose metabolism, osmotic stress, and sodium-glucose cotransporter isoform-mediated transport on Na+/H+ exchanger isoform 3 activity in the renal proximal tubule. J Am Soc Nephrol 2014;25:2028–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chung YJ, Park KC, Tokar S, Eykyn TR, Fuller W, Pavlovic D, et al. Off-target effects of sodium-glucose co-transporter 2 blockers: empagliflozin does not inhibit Na+/H+ exchanger-1 or lower [Na+]i in the heart. Cardiovascular Research 2020. Nov 2;cvaa323 [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Komoroski B, Vachharajani N, Feng Y, Li L, Kornhauser D, Pfister M. Dapagliflozin, a novel, selective SGLT2 inhibitor, improved glycemic control over 2 weeks in patients with type 2 diabetes mellitus. Clin Pharmacol Ther 2009;85:513–9. [DOI] [PubMed] [Google Scholar]
- 38.Heise T, Jordan J, Wanner C, Heer M, Macha S, Mattheus M, et al. Pharmacodynamic effects of single and multiple doses of empagliflozin in patients with type 2 diabetes. Clin Ther 2016;38:2265–76. [DOI] [PubMed] [Google Scholar]
- 39.Chen L, LaRocque LM, Efe O, Wang J, Sands JM, Klein JD. Effect of dapagliflozin treatment on fluid and electrolyte balance in diabetic rats. Am J Med Sci 2016;352:517–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hallow KM, Greasley PJ, Helmlinger G, Chu L, Heerspink HJL, Boulton DW. Evaluation of renal and cardiovascular protection mechanisms of SGLT2 inhibitors: model-based analysis of clinical data. Am J Physiol Renal Physiol 2018;315:F1295–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wilcox CS, Shen W, Boulton DW, Leslie BR, Griffen SC. Interaction between the sodium-glucose-linked transporter 2 inhibitor dapagliflozin and the loop diuretic bumetanide in normal human subjects. J Am Heart Assoc 2018;7:e007046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sha S, Polidori D, Heise T, Natarajan J, Farrell K, Wang SS, et al. Effect of the sodium glucose co-transporter 2 inhibitor canagliflozin on plasma volume in patients with type 2 diabetes mellitus. Diabetes Obes Metab 2014;16:1087–95. [DOI] [PubMed] [Google Scholar]
- 43.Lambers Heerspink HJ, de Zeeuw D, Wie L, Leslie B, List J. Dapagliflozin a glucose-regulating drug with diuretic properties in subjects with type 2 diabetes. Diabetes Obes Metab 2013;15:853–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dekkers CCJ, Sjöström CD, Greasley PJ, Cain V, Boulton DW, Heerspink HJL. Effects of the sodium-glucose co-transporter-2 inhibitor dapagliflozin on estimated plasma volume in patients with type 2 diabetes. Diabetes Obes Metab 2019;21:2667–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.van Bommel EJM, Muskiet MHA, van Baar MJB, Tonneijck L, Smits MM, Emanuel AL, et al. The renal hemodynamic effects of the SGLT2 inhibitor dapagliflozin are caused by post-glomerular vasodilatation rather than pre-glomerular vasoconstriction in metformin-treated patients with type 2 diabetes in the randomized, double-blind RED trial. Kidney Int 2020;97:202–12. [DOI] [PubMed] [Google Scholar]
- 46.Hallow KM, Helmlinger G, Greasley PJ, McMurray JJV, Boulton DW. Why do SGLT2 inhibitors reduce heart failure hospitalization? A differential volume regulation hypothesis. Diabetes Obes Metab 2018;20:479–87. [DOI] [PubMed] [Google Scholar]
- 47.Mazer CD, Hare GMT, Connelly PW, Gilbert RE, Shehata N, Quan A, et al. Effect of empagliflozin on erythropoietin levels, iron stores, and red blood cell morphology in patients with type 2 diabetes mellitus and coronary artery disease. Circulation 2020;141:704–7. [DOI] [PubMed] [Google Scholar]
- 48.Verma S, Mazer CD, Yan AT, Mason T, Garg V, Teoh H, et al. Effect of empagliflozin on left ventricular mass in patients with type 2 diabetes mellitus and coronary artery disease. Circulation 2019;140:1693–702. [DOI] [PubMed] [Google Scholar]
- 49.Docherty KF, Curtain JP, Anand IS, Bengtsson O, Inzucchi SE, Køber L, et al. Effect of dapagliflozin on anaemia in DAPA-HF. Eur J Heart Fail 2021;23:617–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Eickhoff MK, Dekkers CCJ, Kramers BJ, Laverman GD, Frimodt-Møller M, Jørgensen NR, et al. Effects of dapagliflozin on volume status when added to renin-angiotensin system inhibitors. J Clin Med 2019;8:779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Heerspink HJ, Perkins BA, Fitchett DH, Husain M, Cherney DZ. Sodium glucose cotransporter 2 inhibitors in the treatment of diabetes mellitus: cardiovascular and kidney effects, potential mechanisms, and clinical applications. Circulation 2016;134:752–72. [DOI] [PubMed] [Google Scholar]
- 52.Ghanim H, Abuaysheh S, Hejna J, Green K, Batra M, Makdissi A, et al. Dapagliflozin suppresses hepcidin and increases erythropoiesis. J Clin Endocrinol Metab 2020;105:e1056–63. [DOI] [PubMed] [Google Scholar]
- 53.Miyachi Y, Tsuchiya K, Shiba K, Mori K, Komiya C, Ogasawara N, et al. A reduced M1-like/M2-like ratio of macrophages in healthy adipose tissue expansion during SGLT2 inhibition. Sci Rep 2018;8:16113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Santos-Gallego CG, Requena-Ibanez JA, San Antonio R, Picatoste B, Ishikawa K, Vahl T, et al. SGLT2 inhibitor empagliflozin reduces skin sodium content and interstitial fluid volume more than loop diuretics in heart failure. Poster presented at the American College of Cardiology 2020 Virtual Meeting, March 28–30. Poster 1459–67. Available at: https://cslide-us.ctimeetingtech.com/acc2020_eposter/attendee/eposter/poster/3046?q=Santis-Gallego. [Google Scholar]
- 55.Karg MV, Bosch A, Kannenkeril D, Striepe K, Ott C, Schneider MP, et al. SGLT-2-inhibition with dapagliflozin reduces tissue sodium content: a randomised controlled trial. Cardiovasc Diabetol 2018;17:5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2019;380:347–57. [DOI] [PubMed] [Google Scholar]
- 57.Ben-Shlomo Y, Spears M, Boustred C, May M, Anderson SG, Benjamin EJ, et al. Aortic pulse wave velocity improves cardiovascular event prediction: an individual participant meta-analysis of prospective observational data from 17,635 subjects. J Am Coll Cardiol 2014;63:636–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cherney DZ, Perkins BA, Soleymanlou N, Har R, Fagan N, Johansen OE, et al. The effect of empagliflozin on arterial stiffness and heart rate variability in subjects with uncomplicated type 1 diabetes mellitus. Cardiovasc Diabetol 2014;13:28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Chilton R, Tikkanen I, Cannon CP, Crowe S, Woerle HJ, Broedl UC, et al. Effects of empagliflozin on blood pressure and markers of arterial stiffness and vascular resistance in patients with type 2 diabetes. Diabetes Obes Metab 2015;17:1180–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Solini A, Giannini L, Seghieri M, Vitolo E, Taddei S, Ghiadoni L, et al. Dapagliflozin acutely improves endothelial dysfunction, reduces aortic stiffness and renal resistive index in type 2 diabetic patients: a pilot study. Cardiovasc Diabetol 2017;16:138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Jüni RP, Kuster DWD, Goebel M, Helmes M, Musters RJP, van der Velden J, et al. Cardiac microvascular endothelial enhancement of cardiomyocyte function is impaired by inflammation and restored by empagliflozin. JACC Basic Transl Sci 2019;4:575–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Uthman L, Homayr A, Jüni RP, Spin EL, Kerindongo R, Boomsma M, et al. Empagliflozin and dapagliflozin reduce ROS generation and restore NO bioavailability in tumor necrosis factor α-stimulated human coronary arterial endothelial cells. Cell Physiol Biochem 2019;53:865–86. [DOI] [PubMed] [Google Scholar]
- 63.Santos-Gallego CG, Requena-Ibanez JA, San Antonio R, Garcia-Ropero A, Ishikawa K, Watanabe S, et al. Empagliflozin ameliorates diastolic dysfunction and left ventricular fibrosis/stiffness in nondiabetic heart failure: a multimodality study. JACC Cardiovasc Imaging 2021;14:393–407. [DOI] [PubMed] [Google Scholar]
- 64.Pfeifer M, Townsend RR, Davies MJ, Vijapurkar U, Ren J. Effects of canagliflozin, a sodium glucose co-transporter 2 inhibitor, on blood pressure and markers of arterial stiffness in patients with type 2 diabetes mellitus: a post hoc analysis. Cardiovasc Diabetol 2017;16:29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sugiyama S, Jinnouchi H, Kurinami N, Hieshima K, Yoshida A, Jinnouchi K, et al. The SGLT2 inhibitor dapagliflozin significantly improves the peripheral microvascular endothelial function in patients with uncontrolled type 2 diabetes mellitus. Intern Med 2018;57:2147–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Shigiyama F, Kumashiro N, Miyagi M, Ikehara K, Kanda E, Uchino H, et al. Effectiveness of dapagliflozin on vascular endothelial function and glycemic control in patients with early-stage type 2 diabetes mellitus: DEFENCE study. Cardiovasc Diabetol 2017;16:84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kusche-Vihrog K, Schmitz B, Brand E. Salt controls endothelial and vascular phenotype. Pflugers Arch 2015;467:499–512. [DOI] [PubMed] [Google Scholar]
- 68.Oberleithner H A physiological concept unmasking vascular salt sensitivity in man. Pflugers Arch 2012;464:287–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Baartscheer A, Schumacher CA, Wüst RC, Fiolet JW, Stienen GJ, Coronel R, et al. Empagliflozin decreases myocardial cytoplasmic Na+ through inhibition of the cardiac Na+/H+ exchanger in rats and rabbits. Diabetologia 2017;60:568–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Uthman L, Baartscheer A, Schumacher CA, Fiolet JWT, Kuschma MC, Hollmann MW, et al. Direct cardiac actions of sodium glucose cotransporter 2 inhibitors target pathogenic mechanisms underlying heart failure in diabetic patients. Front Physiol 2018;9:1575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Heerspink HJL, Stefansson BV, Correa-Rotter R, Chertow GM, Greene T, Hou FF, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med 2020;383:1436–46. [DOI] [PubMed] [Google Scholar]
- 72.McMurray JJV, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med 2019;381:1995–2008. [DOI] [PubMed] [Google Scholar]
- 73.Nassif ME, Windsor SL, Tang F, Khariton Y, Husain M, Inzucchi SE, et al. Dapagliflozin effects on biomarkers, symptoms, and functional status in patients with heart failure with reduced ejection fraction: the DEFINE-HF trial. Circulation 2019;140:1463–76. [DOI] [PubMed] [Google Scholar]
- 74.Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med 2020;383:1413–24. [DOI] [PubMed] [Google Scholar]
- 75.Lee MMY, Brooksbank KJM, Wetherall K, Mangion K, Roditi G, Campbell RT, et al. Effect of empagliflozin on left ventricular volumes in patients with type 2 diabetes, or prediabetes, and heart failure with reduced ejection fraction (SUGAR-DM-HF). Circulation 2021;143:516–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Santos-Gallego CG, Vargas-Delgado AP, Requena-Ibanez JA, Garcia-Ropero A, Mancini D, Pinney S, et al. Randomized trial of empagliflozin in nondiabetic patients with heart failure and reduced ejection fraction. J Am Coll Cardiol 2021;77:243–55. [DOI] [PubMed] [Google Scholar]
- 77.Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Bonaca MP, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet 2019;393:31–9. [DOI] [PubMed] [Google Scholar]


