Abstract
Non-typhoidal salmonella (NTS) is most commonly associated with self-limiting enterocolitis. However, in extremes of age and immune compromised patients, it can cause invasive and disseminated infection, including bacteremia and localized extraintestinal infections. Pneumonia and vertebral osteomyelitis are rare manifestation of NTS, with only a few cases reported worldwide. Our case describes a young man with sickle cell anemia, who developed multifocal invasive NTS infection with pulmonary involvement, vertebral osteomyelitis, and epidural abscess. He was treated with a six-week course of antibiotics and surgical drainage of the abscess. This case report highlights an atypical presentation of NTS infection that is associated with high morbidity and mortality, that can be reduced with early diagnosis and appropriate treatment.
Keywords: Salmonella, Pneumonia, Vertebral osteomyelitis, Sickle cell disease
Introduction
Globally, non-typhoidal salmonella (NTS) is one of the leading bacterial causes of diarrhea, with approximately 153 million cases of gastroenteritis and 57,000 deaths annually [1]. Although NTS most often manifests as a self-limiting diarrheal illness, in rare cases, it can cause invasive infection, such as bacteremia, and focal infections, such as meningitis and septic arthritis [2]. Invasive NTS infections usually present with non-specific febrile illnesses, rather than gastrointestinal symptoms, and have higher morbidity and mortality than non-invasive infections [3]. Multi-organ involvement is extremely rare in NTS, with only a few cases of pulmonary infection and vertebral osteomyelitis reported worldwide.
Case presentation
A 27-year-old Middle Eastern man, with a history of sickle cell disease, presented to the emergency department with a three-week history of worsening low back pain. He also reported cough with greenish sputum for one week, associated with fever. He denied shortness of breath, chills, night sweats or weight loss. There was no history of nausea, vomiting, abdominal pain or diarrhea. He also denied urinary or bowel incontinence, lower limb weakness or sensory deficits. There was no history of trauma and no sick contacts.
The patient’s past medical history was significant for sickle cell disease, with multiple hospitalizations for vaso-occlusive crisis, history of acute chest syndrome in 2017, avascular necrosis of the left tibia and fibula in 2019, and recurrent episodes of priapism. The patient was enrolled in a red blood cell exchange transfusion program every 8 weeks, but had poor compliance due to financial issues. His home medications included hydroxyurea 1500 mg daily, folic acid 5 mg a day, pregabalin 75 mg twice daily, and transdermal fentanyl and oxycodone as needed for pain. He was a nonsmoker and denied alcohol or illicit drug use. Past surgical history included a splenectomy in 2003 and appendectomy in 2005. The patient was a high school graduate and was not currently employed.
On examination, he appeared pale and in mild distress. He was afebrile with a temperature of 36.5 °C. Initial vital signs included blood pressure of 131/67 mmHg without orthostatic changes, heart rate of 114 beats per minute, and oxygen saturation of 99% on room air. Lungs were clear on auscultation. Abdomen was soft and non-tender. Back examination revealed severe tenderness over the thoracic and lumbar spine. Neurologic examination revealed 5/5 motor strength in upper and lower limbs bilaterally, with normal reflexes and no sensory deficits. There were no mucosal ulcers or skin rashes.
Initial laboratory evaluation revealed a microcytic anemia with hemoglobin of 64 g/dL (117–155 g/dL), MCV 57 fL (81–100 fL), MCH 17.5 pg (27–34 pg), WBC count of 21.6 × 10^9/L (4.5–11 ×10^9/L) with predominant neutrophils of 14.59 × 10^9/L (1.8–7.7 ×10^9/L), and platelets of 1098 × 10^9/L (140–400 ×10^9/L). Hemolysis panel was unremarkable, with an LDH of 184 IU/L (135–214 IU/L) and haptoglobin of 2.35 g/L (0.3–2 g/L). Inflammatory markers showed elevated C-reactive protein (CRP) of 138 mg/L (<5 mg/L) and normal procalcitonin 0.17 ng/ml (<0.5 ng/ml). Liver and kidney function tests were normal. COVID-19 PCR was negative.
A high-resolution chest CT scan showed advanced vertebral collapse of the T5 vertebra and bony fusion with T4, significant end plate lysis of a block of T4, T5 and T6 vertebrae, with mild posterior retropulsion into spinal canal at T5 level. There was no gross spinal canal stenosis. There was a large bilateral paraspinal soft tissue thickening with foci of calcification extending from the level of T3 down to T7, suggestive of spondylodiscitis and possible paraspinal abscess. The CT also showed bilateral interstitial thickening and pulmonary congestive changes.
The patient was admitted to the general medical ward for management of sickle cell vaso-occlusive crisis, community acquired pneumonia and further work up and treatment of a suspected thoracic para-spinal abscess. He received 2 units of cross-matched red blood cells and was started on intravenous hydration with normal saline and pain control with morphine through patient-controlled analgesia. After blood, sputum and urine cultures were collected, the patient was started empirically on a broad spectrum antibiotic, Pipercillin-tazobactam.
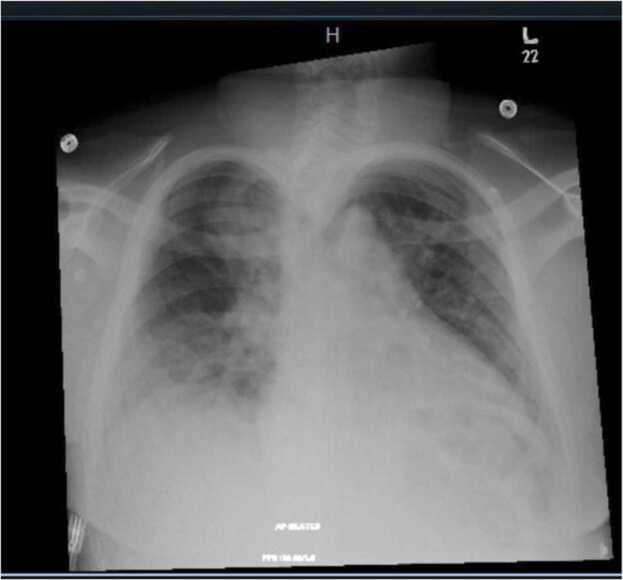
Over the first two days of admission, the patient continued to have back pain, but he was otherwise clinically stable, with decreasing inflammatory markers. Specifically, the CRP decreased from 138 to 96 and the WBC decreased from 24 to 17. On day 3 of hospitalization, the patient became febrile to 38 °C and developed tachypnea with worsening cough and blood-tinged sputum. Chest x ray revealed bilateral multifocal areas of opacification (Fig. 1). Repeat blood, sputum and urine cultures were collected and antibiotic coverage was expanded to meropenem and azithromycin. The hematology service was consulted, and the patient underwent a plasma exchange transfusion for possible acute chest syndrome.
Fig. 1.
Portable chest x-ray showing multifocal airspace opacifications noted bilaterally more in the right lower zone.
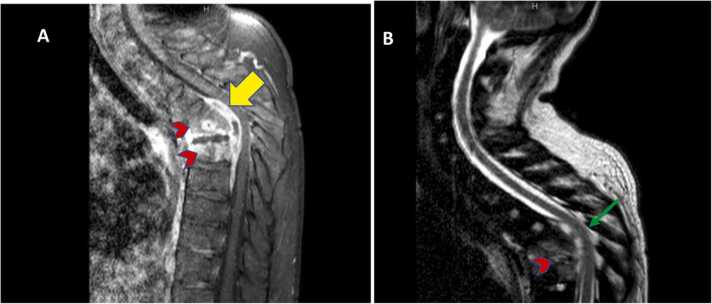
MRI Spine with contrast showed focal spondylodiscitis at the T5-T6 level with a multiloculated intraspinal epidural abscess, extending from the level of T4 to T7 below the intact posterior longitudinal ligament, compressing the cord with moderate spinal canal stenosis, vertebral destruction with prominent kyphosis, and a large bilateral paravertebral phlegmon with abscess formation (Fig. 2).
Fig. 2.
MRI spine with contrast T1 (A) and T2 (B) showing focal spondylodiscitis (arrowhead) at T5-T6 level with multiloculated intraspinal epidural abscesses (yellow arrow) extending from the level of T4 down to T7 below the intact posterior longitudinal ligament, compressing the cord (green arrow) with moderate spinal canal stenosis, vertebral destruction with prominent kyphosis.
The patient remained neurologically intact, but he was unable to ambulate due to severe back pain. A diagnostic aspiration of the paraspinal abscess was performed on the 6th day of hospitalization by the interventional radiologist, and a sample of pus was obtained. Cultures and Tuberculosis Polymerase Chain Reaction from the spinal abscess fluid were negative. Blood and sputum cultures both grew Salmonella species (non typhi). Antibiotics were changed to ceftriaxone, based on culture sensitivity. Further blood workup was negative for HIV and an echocardiogram did not reveal any vegetations.
The patient was transferred to another facility for surgical intervention and underwent a right thoracoscopy and pneumolysis, as well as a posterior T4 to T6 laminectomy and decompression, with drainage of the epidural abscess. The patient’s post-op course was unremarkable. He received a total of four weeks of ceftriaxone and was discharged on oral trimethoprim/sulfamethoxazole for an additional two weeks. He progressed well with physiotherapy, and ultimately returned to his baseline level of mobility.
Discussion
Salmonellae are gram-negative bacteria that belong to the family of Enterobacteriaceae. Over 2500 Salmonella serotypes have been reported. Nontyphoidal salmonella (NTS) refers to all serotypes of Salmonella, excluding Typhi and Paratyphi A, B and C [4]. Risk factors for salmonella infection include extremes of age and immune compromised conditions, primarily HIV infection, malignancy and reticuloendothelial blockade, such as in sickle-cell disease (SCD) [6]. NTS often occurs in children with SCD, and is a significant cause of morbidity and mortality in developing countries. However, there are limited reports on the clinical presentation, complications and outcomes of NTS in adults with SCD. A chart review of 3500 adults with SCD diagnosed with NTS in 3 referral centers in France, conducted between 2006 and 2016, found that the clinical presentation of NTS in adults with SCD was often non-specific at admission. A triad of bacteremia, secondary foci of infection and localized bone infections was observed in up to 30% of cases [7]. Localization of infection may occur at any site after salmonella bacteremia. Ramos et al. noted that the most frequent site of extra-intestinal infection was the urinary tract (30%), followed by the respiratory tract (20%) and the skeletal system (19%) [8].
Vertebral osteomyelitis is an extremely rare complication of NTS, with unknown global incidence [9]. In the largest review of Salmonella vertebral osteomyelitis, which included 44 patients, Santos and Sapico reported a higher incidence of salmonella vertebral osteomyelitis in men than women, with a ratio of 1.7:1. Low back pain (92%) and fever (87%) were the most common presenting symptoms. Blood cultures were positive for salmonella in 48% of cases, whereas 36% had positive stool cultures, and 23% had positive urine cultures. Paravertebral abscesses were diagnosed in 39% of the patients, while 4% of cases had epidural abscesses. In total, 72% of the patients were treated with medical management alone and 28% required surgical intervention [10].
Pulmonary infections secondary to NTS are extremely rare, with only a few cases reported in the literature [11]. The mechanism by which NTS causes lung infection is unclear, but may be due to direct extension from a nearby infection, aspiration of gastric secretions, or hematogenous spread from the gastrointestinal tract [12]. In a longitudinal observational study in Spain, eleven cases with NTS pleuro-pulmonary disease were noted over a period of twenty-seven years. Pneumonia occurred in eight patients, lung abscesses in two, and empyema in one. All patients with pneumonia had positive blood cultures. In this study, immune compromised patients with salmonella pneumonia had a high mortality rate, approaching 63% [13]. Pre-existing lung disease is likely a risk factor for salmonella lung involvement. Cohen et al. reviewed thirty-six patients with salmonella pneumonia, empyema, or lung abscess and found that thirteen (36%) had prior pulmonary or pleural disease [14].
Our patient was initially managed for community acquired pneumonia and possible acute chest syndrome, due to his symptoms of dyspnea, cough and opacities on chest x-ray. He was started empirically on piperacillin-tazobactam and exchange transfusion. His first sputum culture showed no growth. When his respiratory status worsened, antibiotic coverage was extended, and repeated blood and sputum cultures both grew non-typhoidal salmonella. We believe that the epidural abscess culture was negative because the aspiration was performed after the patient was started on empiric broad-spectrum antibiotics. He required surgical intervention for vertebral osteomyelitis and epidural abscess, and he received total of four weeks of IV ceftriaxone and two weeks of oral trimethoprim/sulfamethoxazole. Our patient was known to have SCD, which put him at a higher risk for complicated salmonella infection. Yet, salmonella was not initially considered to be in the differential diagnosis given the multifocal involvement and absence of gastrointestinal symptoms, emphasizing the need to have a high clinical suspicion to consider and treat disseminated salmonella in high-risk patients.
Conclusion
This case highlights the importance of recognizing salmonella as the potential cause of multifocal infections in high-risk patients. As the incidence of invasive and disseminated NTS infections increases worldwide, it is important to recognize salmonella as a causative agent of a wide range of infections, other than gastrointestinal disease. Despite pulmonary involvement and vertebral osteomyelitis being a rare manifestation of NTS infection, they should be considered in any immune compromised patient. This, in return, will promote early diagnosis and administration of appropriate treatment to reduce the morbidity and mortality associated with inappropriately treated NTS infection.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Ethical approval
Ethical approval is not required at our institution to publish an anonymous case report.
Consent for publication
Written consent was obtained from the patient.
CRediT authorship contribution statement
Salma Elnour: Conceptualization, Writing – original draft, Writing – review & editing. Maram Hashim: Conceptualization, Writing – original draft, Writing – review & editing. Halah Ibrahim: Conceptualization, Writing – original draft, Writing – review & editing.
Declaration of Competing Interest
The authors declare no conflicts of interest.
Acknowledgments
None.
References
- 1.Gal-Mor O., Healy J.M. et al.: Travel-related infectious diseases: salmonellosis (nontyphoidal). In: CDC Yellow Book 2020: Health Information for International Travel.
- 2.Acheson D., Hohmann E.L. Nontyphoidal salmonellosis. Clin Infect Dis. 2001;32(2):263–269. doi: 10.1086/318457. [DOI] [PubMed] [Google Scholar]
- 3.Crump J.A., Sjölund-Karlsson M., Gordon M.A., Parry C.M. Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive salmonella infections. Clin Microbiol Rev. 2015;28(4):901–937. doi: 10.1128/CMR.00002-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gal-Mor O., Boyle E.C., Grassl G.A. Same species, different diseases: how and why typhoidal and non-typhoidal Salmonella enterica serovars differ. Front Microbiol. 2014;5:391. doi: 10.3389/fmicb.2014.00391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Acheson David, Hohmann Elizabeth L. Nontyphoidal Salmonellosis. Clin Infect Dis. 2001;32(2):263–269. doi: 10.1086/318457. [DOI] [PubMed] [Google Scholar]
- 7.Guery Romain, Habibi Anoosha, Arlet Jean-Benoît, Lionnet François, Lastours Victoire de, Decousser Jean-Winoc, et al. Severe, non specific symptoms in non-typhoidal Salmonella infections in adult patients with sickle cell disease: a retrospective multicentre study. Infect Dis. 2018;50(11–12):822–830. doi: 10.1080/23744235.2018.1500706. [DOI] [PubMed] [Google Scholar]
- 8.Ramos J.M., García-Corbeira P., Aguado J.M., Alés J.M., Soriano F. Classifying extraintestinal non-typhoid Salmonella infections. QJM. 1996;89(2):123–126. doi: 10.1093/qjmed/89.2.123. [DOI] [PubMed] [Google Scholar]
- 9.Librianto D., Suwarto S., Imran D., Pramukti H., Saleh I., Ipang F., et al. An extremely rare case of upper thoracic Salmonella infection. Orthop Res Rev. 2021;13:107–112. doi: 10.2147/ORR.S319616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Santos E.M., Sapico F.L. Vertebral osteomyelitis due to salmonellae: report of two cases and review. Clin Infect Dis. 1998;27(2):287–295. doi: 10.1086/514668. [DOI] [PubMed] [Google Scholar]
- 11.Kam J.C., Abdul-Jawad S., Modi C., Abdeen Y., Asslo F., Doraiswamy V., et al. Pleural empyema due to Group D Salmonella. Case Rep Gastrointest Med. 2012;2012 doi: 10.1155/2012/524561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Persson K., Mörgelin M., Lindbom L., Alm P., Björck L., Herwald H. Severe lung lesions caused by Salmonella are prevented by inhibition of the contact system. J Exp Med. 2000;192(10):1415–1424. doi: 10.1084/jem.192.10.1415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Aguado J.M., Obeso G., Cabanillas J.J., Fernández-Guerrero M., Alés J. Pleuropulmonary infections due to nontyphoid strains of Salmonella. Arch Intern Med. 1990;150(1):54–56. [PubMed] [Google Scholar]
- 14.Cohen J.I., Bartlett J.A., Corey G.R. Extra-intestinal manifestations of salmonella infections. Medicine. 1987;66(5):349–388. doi: 10.1097/00005792-198709000-00003. [DOI] [PubMed] [Google Scholar]