Abstract
During female lifetime and pregnancy, inflammation and cellular senescence are implicated in physiological processes, from ovulation and menstruation, to placental homeostasis and delivery. Several lifestyles, nutritional, and environmental insults, as well as long-lasting pregestational inflammatory diseases may lead to detrimental effects in promoting and sustaining a chronic excessive inflammatory response and inflammaging, which finally contribute to the decay of fertility and pregnancy outcome, with a negative effect on placental function, fetal development, and future health risk profile in the offspring. Maladaptation to pregnancy and obstetric disease may in turn increase maternal inflammaging in a feedback loop, speeding up aging processes and outbreak of chronic diseases. Maternal inflammaging may also impact, through transgenerational effects, on future adult health. Hence, efficacious interventions should be implemented by physicians and healthcare professionals involved in prevention activities to reduce the modifiable factors contributing to the inflammaging process in order to improve public health.
Keywords: Inflammaging, Senescence, Aging, Maternal low-grade chronic inflammation, Fetal programming, Fertility
Introduction
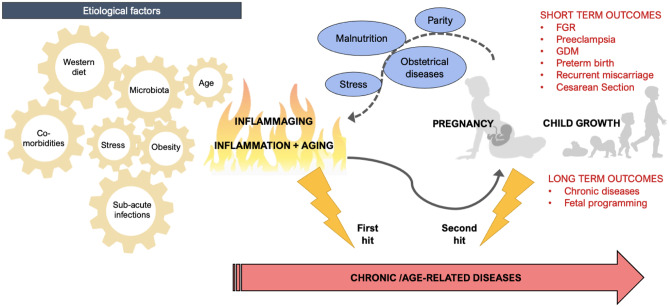
Inflammaging can negatively predispose the woman to reproductive dysfunction and obstetrical diseases during pregnancy. On the other hand, pregnancy itself can boost the onset of latent chronic pathologies finally exacerbated by maternal adaptations to pregnancy (Fig. 1).
Fig. 1.
Inflammaging effects on pregnancy, mother and offspring: On the left etiological factors contributing to the pathogenesis of inflammaging; on the right effects of inflammaging on pregnancy, maternal, and offspring outcomes and potential feedback loop of pregnancy effects on inflammaging and chronic age-related diseases onset; FGR fetal growth restriction, GDM gestational diabetes mellitus
Inflammaging encompasses the concept of a feedback loop between two triggering factors, namely inflammation and aging [1], leading to a range of short- and long-term effects both on the woman during the pre-conceptional and gestational period, and on the fetus [2–4]. In particular, inflammaging leads to an accelerated decline of ovarian and uterine functions, maternal maladaptive processes, and placental dysfunction during pregnancy, resulting in a disrupted intra uterine environment for the developing fetus [5–8]. Such a proinflammatory chronic status may also induce epigenetic modification and metabolic reprogramming both in the fetus and the placenta, making the offspring more susceptible to future non-communicable diseases [8, 9].
The present review aims to (1) examine the impact of inflammaging on fertility and pregnancy outcomes, by focusing both on maternal and fetal effects, and, in turn, (2) explore possible impacts of pregnancy on maternal inflammaging itself, hypothesizing pregnancy as a potential trigger of inflammaging. Moreover, the present work aims to highlight the role of both inflammaging and pregnancy on chronic, age-related diseases, where the former acts as a first hit and the latter as a potential second hit.
Etiological Factors
Long-term exposure to inflammatory triggers and stressors, such as infections, chemical irritation, and several lifestyle and nutritional habits, with the contribution of systemic dysregulation and prolonged insults induced by aging, synergically magnify the inflammatory status and level of inflammaging during the reproductive period and pregnancy [10] (Fig. 2). The source of inflammaging represents a complex network of many cofactors affecting this condition to varying degrees (Table 1).
Fig. 2.

Inflammaging as a synergic result of the interaction among many co-factors
Table 1.
Detailed summary of the principal studies about inflammaging and related risk factors
| Risk factor | Reference | Type of study | Topic | Population | Principal findings/key points |
|---|---|---|---|---|---|
| Obesity | Weisberg et al. 2003 [13] | Research article | Obesity and adipose tissue dysfunction | Animal | Obesity increases macrophage infiltration in the adipose tissue and local expression of TNF-α and IL-6, thus promoting local inflammatory response. |
| Chawla et al. 2011 [14] | Review | Obesity, inflammation and insulin resistance. | General | Chronic overnutrition triggers adaptive immune responses, which synergizes with macrophage-mediated inflammation to promote insulin resistance. | |
| Gregor and Hotamisligi 2011 [12] | Review | Obesity, meta-inflammation and metabolic dysfunction. | General |
Meta-inflammation—a low-grade inflammatory response to the excess of nutrients—induces: - activation of specialized immune cells - insulin resistance - disruption of nutrient and energy metabolism. |
|
| Parisi et al. 2021 [11] | Review | Obesity, meta-inflammation and feto-maternal outcomes. | Pregnant |
Obesity-related maternal inflammation affects reproductive and pregnancy outcomes at several levels: - Periconceptional period - Embryo-fetal development - Placental function - Neonatal long-term outcomes A pro-inflammatory environment leads to: - Intrauterine derangements - Compensatory adaptations - Metabolic reprogramming |
|
| Diet and microbiome | Lee et al. 2001 [17] | Research article | Effects of Saturated Fatty Acids (SFAs) and Unsaturated Fatty Acids (UFAs) on LPS-induced signaling pathways and Cyclooxygenase (COX-2) expression in monocyte/macrophage cells. | Animal |
SFAs, but not unsaturated fatty acids (UFAs), induce Nuclear Factor κB (NF-κB) activation and expression of COX-2 and other inflammatory markers. UFAs inhibit COX-2 expression induced by SFAs, TLR4, or LPS. |
| Raetz and Whitfield 2002 [16] | Review | Biosynthesis of LPS and Lipid A activation of Toll Like Receptor 4 (TLR4). | General | Lipid A—the hydrophobic anchoring part of bacterial lipopolysaccharide (LPS)—activates TLR4 exposed on macrophages, mononuclear and endothelial cells, triggering the biosynthesis of inflammatory mediators and costimulatory molecules. | |
| Cani et al. 2007 [15] | Research article | Bacterial lipopolysaccharide (LPS) as a trigger of low-grade chronic inflammation and metabolic derangement. | Animal | LPS-associated metabolic endotoxemia dysregulates the inflammatory tone and triggers body weight gain, insulin resistance, obesity and diabetes. | |
| Bailey and Holscher 2018 [18] | Review | Mediterranean diet contribution to metabolic health. | General | Mediterranean dietary pattern modulates gastrointestinal microbiota, lowering endotoxemia and inflammation. | |
| Environment | Doherty et al. 2009 [21] | Review | Intrauterine exposure to cigarette smoke (CS) | Pregnant | CS leads to cardiovascular, cancer and respiratory disease in the exposed offspring. |
| Bolten et al 2011 [25] | Research article | Maternal distress, cortisol level and fetal growth. | Pregnant | Maternal cortisol levels in pregnancy influence intrauterine fetal development and birth weight. | |
| Cetin et al. 2013 [20] | Review | Role of maternal characteristics and behaviors in altering placental function and fetal growth. | Pregnant |
Maternal nutritional and environmental exposures in association with - Fetal nutrient availability - Placental gene expression and function - Fetal growth trajectory. |
|
| Alavian–Ghavanini and Rüegg 2018 [23] | Review | Endocrine-disrupting chemicals (EDCs) and fetal development. | Pregnant | Early-life exposure to EDCs leads to long-term increased risk of obesity, diabetes and cancer in the offspring by means of epigenetic changes. | |
| Franzago et al. 2019 [19] | Review | Role of diet-related genetic variants and epigenetic modifications on feto-maternal outcomes. | Pregnant | Epigenetic modifications induced by nutritional and environmental exposures may lead to maternal hyperglycemia in pregnancy and fetal metabolic programming. | |
| Hu et al. 2019 [24] | Research article | Maternal psychological state and offspring microbiome. | Pregnant | High pregnancy-related anxiety is significantly associated with a less assorted meconium microbiota community. | |
| Comorbidities | Baltacıoğlu et al. 2014 [27] | Research article | Immuno-inflammatory response and oxidative stress in periodontal disease. | General | Bone resorption in periodontal disease is associated with both local and systemic oxidative stress. |
| Zambon et al. 2018 [28] | Research article | Maternal obesity, periodontal disease and systemic and local inflammatory status during pregnancy. | Pregnant | Obesity, GDM and periodontal disease may synergistically amplify the inflammatory and oxidative status in pregnancy | |
| Xu et al. 2021 [26] | Review | Inflammaging and severe hyper-inflammatory state during COVID-19 and other severe infections (sepsis). | General | Inflammaging and coagulating determine adverse course of viral and bacterial severe infections. |
TNF-α tumor necrosis factor α, IL-6 interleukin-6, SFAs saturated fatty acids, UFAs unsaturated fatty acids, NF-κB nuclear factor κB, COX-2 cyclo-oxigenase 2, TLR4 toll-like receptor 4, LPS lipopolysaccharide, CS cigarette smoke, GDM gestational diabetes mellitus, EDCs endocrine-disrupting chemicals
Obesity
Obesity plays a pivotal role in the pathogenesis of inflammaging, since the massive expansion of the adipose tissue, as a highly active endocrine organ, affects immune cell profile, deranges inflammatory cytokines production, and finally impairs metabolic function [11]. Such a metabolic dependent pro inflammatory background, the so-called meta-inflammation, in turn, influences the metabolism itself leading to increased oxidative stress and insulin resistance [12]. More specifically, obesity alters immune cell profile in visceral adipose tissue by increasing local macrophages, neutrophils, T cells, B cells and mast cells, and reducing T-helper cells, regulatory T cells and eosinophils [13]. The loss of anti-inflammatory mediators leads to macrophage pro inflammatory response, resulting in high expression of Interleukin-6 (IL-6), tumor necrosis factor- (TNF-), interferon- (IFN-, and endogenous toll-like receptor (TLR) ligands [14]. Macrophage infiltration in the adipose tissue is determinant to the dysregulated and vicious circle of systemic inflammatory cytokines production, insulin resistance, oxidative stress, endothelial dysfunction, and further increase of obesity [13].
Diet and Microbiome
Metabolic and microbial factors cooperate in developing a state of metabolic endotoxemia, described as a condition of two- to three-fold increased level of bacterial endotoxin Lipopolysaccharides (LPS) [15], which activates TLR-4 and then upregulates the proinflammatory cytokine expressions [16]. Gastrointestinal (GI) microbiota represents the main source of endotoxin and in turn diet is the primary modulator of the GI microbiota. In fact, diet rich in saturated fat enhances endotoxemia and inflammation both increasing the Gram-negative microbiome component and directly activating TLR-4 receptors [15, 17]. Moreover, high-fat diet boosts liver, adipose, and skeletal muscle expression of proinflammatory cytokines with a consequential metabolic dysfunction such as insulin resistance and dyslipidemia.
On the contrary, Mediterranean diet which is higher in fiber decreases endotoxemia levels by lowering the Gram-negative bacteria prevalence and improving microbial richness both in taxonomy and gene expression [18].
Environment
Further environmental factors can also affect inflammaging. Among them, smoking, alcohol and drug abuse, psychological stress, sedentary life, and endocrine disruptors. The latter are environmental contaminants interfering with hormone systems that can be carried by the atmosphere (for example, clorurate), or by food and drinks (for example, Bisphenol-A, and Ftalate). These environmental factors can act through alterations in cell metabolism and epigenetic modifications, with effects on the reproductive capacity, fetal development and long term effects on both the mother and offspring [19–25].
Comorbidities
Several chronic comorbidities, including cardiovascular, metabolic, neoplastic, and autoimmune diseases, are strongly associated with inflammaging in a self-feeding vicious cycle where inflammaging may increase the risk of developing diseases and the disease itself exasperates the underlying age-related inflammation and immunosenescence. Inflammaging may also play a role in contributing to severe hyperinflammatory state and cytokine storm in case of superimposed infections, as in case of sepsis in elderly or SARS-CoV-2 infection [26].
Chronic infective inflammatory disease including periodontal disease are known to dysregulate the antioxidant reactivity and increase a proinflammatory cytokine expression [27]. Moreover, a strong interaction has been described between periodontal disease and obesity in the exacerbation of the maternal systemic inflammatory status [28].
Inflammaging as a Cumulative Effect of Inflammation and Aging on Fertility and Pregnancy Outcomes
Inflammaging is associated with a modulation of the innate and acquired immune system gradually occurring in the aging process [1]. The assessment of inflammatory mediators (C-reactive protein (CRP), IL-6, TNF-α,) in the absence of acute infections is a useful marker in order to evaluate the rate of progression of inflammaging [29]. Such a persistent low-grade inflammatory environment becomes harmful over time, increasing vulnerability and dysfunctional processes which are the basis of several chronic and age-related diseases.
During female lifetime, inflammation is involved in several functional processes starting from fertility to pregnancy and finally arriving to menopause. In non-pregnant women, inflammation is implicated in ovulation and menstrual cycle, while during conception it is effective and functional for proper implantation, decidualization and pregnancy maintenance [30].
On the other hand, aging consists in a complex biological process influenced by the interaction of multiple intrinsic and extrinsic factors, which leads to natural degeneration of structures, function decline, and progressive loss of adaptability and resilience.
Aging is additionally associated with progressive modifications in the immune reactivity, the so-called immunosenescence. It consists in a decreased activity of macrophages and neutrophils with a concomitant increased production of pro-inflammatory cytokines [31]. Overall, these changes lead to a decline in the antibacterial defense and increased risk of viral infections such as influenza [32]. Immunosenescence plays a key role in the process of inflammaging, since aging and inflammation impact on each other in a vicious circle. First of all, chronic low-grade inflammatory status triggered by a persistent long-term exposure to antigens may have both a direct effect on cells, with a chronic insult concurring in the degenerative process of elderly, and an indirect effect, by means of chronic and latent infection which can periodically reactivate and lead to an immune response against infected cells. Moreover, chronic infection is able to escape immune response, induces DNA mutations, and enhances cellular degenerative process [33].
Conversely, aging promotes inflammation through epigenetic modification of genes involved in the inflammation process such as loss of methylation of TNF promoter which leads to an increased expression and production of the pro-inflammatory cytokines [34].
Telomeres shortening is recognized as a marker of cellular aging, providing a protective function for genomic integrity [35]. In fact, when telomere length becomes critically short, the cell arrests its proliferation and undergoes the process of cellular senescence and apoptosis [36]. Oxidative stress and oncogenic mutations, both increasing in the elderly due to the higher production of reactive oxygen species and the progressive loss of ability to DNA repair, play a pivotal role in the telomeres shortening and senescence process [37]. Likewise, telomeres loss enhances cells susceptibility to oxidative damage [38] and altogether they stimulate a proinflammatory response, by activating transcription and expression of nuclear factor-κ gene binding (NF-κB), cytokines (TNF-α, IL-6, and IFN-γ), growth factors, and matrix metalloproteinases, resulting in a chronic inflammation [39]. Such a proinflammatory reaction triggered by senescent cells has been described as senescence-associated secretory phenotype (SASP) [40] and it is known to foster age-related and degenerative pathologies [41].
Effects of Inflammaging on Fertility
Fertility declines as women age: females are born with a non-renewable oocyte pool, which progressively reduces, leading to loss of fertility, cycle irregularity, and, eventually, menopause. This is a natural process since the continuous activation of primordial follicles leads to the depletion of the follicular ovarian reserve. There are many factors that contribute to accelerate the ovarian aging. Many of them are still not known.
Shortened telomeres and senescent cells impact on aging of several organs and may additionally lead to premature aging of the ovaries. Animal models demonstrated that telomerase deficiency reduce mice eggs chance of getting fertilized and progress through the blastocyst phase, with an increment in pregnancy loss [42, 43]. In humans, compromised telomerase function plays a crucial role in infertility, indeed low telomerase activity and short telomeres have been observed in the granulosa cells of women with poor ovarian reserve [44]. Moreover, telomeres length in cumulus cells, which are specialized granulosa cells, correlates with oocyte quality and chance for embryo development and it can be used as a biomarker for selective embryo transfer [45].
Inflammaging may play a relevant role in ovarian senescence [46]. In fact, it may have a role also in premature ovarian insufficiency (POI), in which inflammation may be one of the causes [2]. To date, available data is still limited. In mice, it has been observed that ovarian aging is associated with a pro-inflammatory microenvironment [47]. Gene transcribing for inflammatory cytokines are upregulated in older ovaries. Specifically, TNF-α, IL-6, IL-10, and IL-18 show significative higher levels [48]. Moreover, it has been shown that nucleotide-binding domain (NOD)-like receptor protein 3 (NLRP3) inflammasome, which is considered to be a major factor of inflammaging, has an increased age-associated expression in mice ovaries [47]. In human studies, it has been observed that inflammatory cytokines and chemokines levels were higher in patients with POI than other women [2]. In addition to age-associated pro-inflammatory status, modification in immune cells has been described in mice aging ovary. Gene expression of colony-stimulating factor 1 (CSF-1) and CSF-2, involved in the proliferation and maturation of macrophages and granulocytes respectively, is significatively increased in 18-month-old mice compared to 2- and 6-month-old ones [2], suggesting a modification into ovary-resident innate immunity. Generally, macrophage is the most abundant type of immune cell, playing an important role in the ovarian tissue organization. Through the reproductive cycle, the ovary undergoes constant cycles of extracellular matrix (ECM) deposition, remodeling and wound healing [49]. In aging ovaries, fibrosis in the ovarian stroma increases. The mechanisms by which it occurs is still unknown. On the one hand, it may be associated with an imbalance between collagen deposition and matrix metalloproteinase (MMP)-associated remodeling [49]. On the other hand, it has been shown an increased M2 macrophage activation, while M1 polarization declines progressively. Interestingly, macrophage shift has been observed also in mouse retinal tissue. M2 macrophages activated by IL-4 and IL-13 produce proline, which is a critical building block of collagen. Thus, it can contribute to increase ovarian fibrosis [48]. Moreover, it has been described the presence of multinucleated macrophage giant cells, which express the α-smooth muscle actin marker, typical of fibroblast activation [49].
Ovarian aging is accompanied by other immune changes. The elevation of IL-4 and IL-13 not only leads to M2 activation but also to the elevation of eosinophil recruitment [48]. Moreover, it has been shown that B cells are increased in senescent ovaries. They are known to produce CSF-2, which may rebound the pro-inflammatory environment. CD4+ T cells infiltration in ovarian tissue increases with age, while CD8+ cell population does not seem to change [47].
In light of the fact that cytokine production and fibrosis may reduce ovarian reserve and function, it will be important to better understand the mechanisms of inflammaging on ovarian tissue, which may lead to a premature impairment of female fertility. The age-associated reduction of fertility is thought to be a combined effect of both the depletion of ovarian reserve and the reduced sensitivity of gonadotropin-releasing hormone, which leads to a deregulation of estrogen signaling. Since macrophages expression of estrogen receptors are influenced by estrogen signaling, it is likely that these alterations at a certain point of reproductive age contribute to alter macrophages functions in ovaries, facilitating the creation of fibrosis and a pro-inflammatory status [48].
Further studies will be useful to determine and describe the pathogenetic pathway involved in ovarian senescence.
Limited data is available about effect of inflammaging on uterus and fallopian tubes. In bovine models, it has been observed that oviduct epithelial cells (OECs) undergo age-dependent changes, which comprehend mainly the creation of a pro-inflammatory microenvironment. Aged OECs secrete higher levels of IL-1β, which stimulates fallopian tubes senescence. In contrast, ECM components in fallopian tubes diminish, so it may impair the function of bovine oviduct [4].
Less is known about the aging impact on the uterus. In bovine models, endometrial cells of older animals showed spontaneously higher levels of inflammatory signaling, IFN signaling, and DNA damage [50].
In addition, cellular senescence affects uterine function, by altering decidualization and impairing the uterine role of supporting pregnancy and embryos development until full term [4]. Studies on mice showed how telomerase deficiency was associated with reduced myometrial thickness, atrophic smooth muscle cells, and lower uterine size [42]. Furthermore, persistently elevated levels of senescent cells in the uterus were associated with downregulated expression of antioxidant enzymes such as tissue-specific Trp53 [51], and thus demonstrated the involvement of oxidative stress in the process of senescence and its negative influence on uterine environment required for a successful pregnancy.
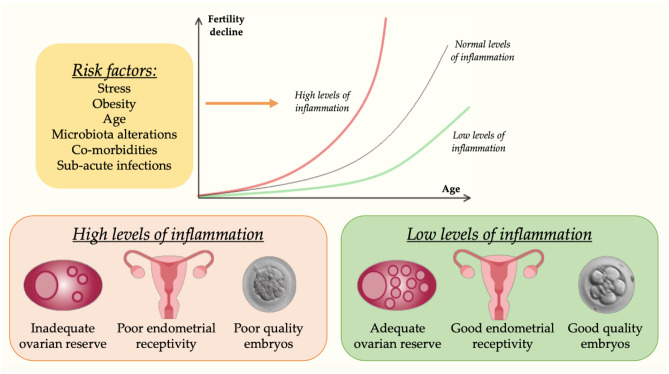
In summary, fertility is progressively reduced by age and negatively influenced by levels of inflammation (Fig. 3).
Fig. 3.
Effects of Inflammaging on fertility. Fertility spontaneously declines as women age. However, higher levels of inflammation lead to a faster reduction of the ovarian reserve, as well as are associated with poor quality embryos and inadequate endometrial receptivity in In-Vitro Fertilization (IVF) treatments. Reduction of risk factors has a positive impact on inflammation and, in turn, on female fertility
In Vitro Fertilization
High-grade chronic systemic inflammatory response has been shown to negatively impact on in vitro fertilization (IVF) success and embryo implantation [52, 53]. Several studies compared gene expression and transcriptomic profile of follicular cells and endometrial fluid of women who successfully got pregnant with those who failed IVF, aiming to understand the reason of IVF failure and to determine non-invasive molecular biomarkers of implantation success [54–57].
High pro-inflammatory mediator expression (e.g., chemokine C-C motif ligand 3 (CCL3), chemokine C-X-C motif ligand 8 (CXCL8) or IL8, CXCL2, IL1B, and CCL4) has been found both in follicular cells and endometrial samples, suggesting that a sustained pro-inflammatory response together with the downregulation of anti-inflammatory mediators may represent the key factor limiting IVF success and performance [52, 58]. Moreover, overexpression of pro-inflammatory mediators in ovarian tissue could also promote vascular endothelial growth factor (VEGF) transcription, as well as ovarian renin–angiotensin system activation [59, 60].
High concentrations of these mediators lead to increased vascular permeability and may trigger hemodynamic and endocrine disorders, finally resulting in the onset of the ovarian hyperstimulation syndrome [52, 54–58]. Many studies investigate the value of serum CRP concentration as a surrogate marker of chronic low-grade inflammation in predicting IVF success and adverse outcomes.
Available data support the association between high serum CRP values before embryo implantation and ART failure [53, 61]. However, many of the reported studies did not adjust for confounding factors, including age [62], thus erasing concerns about the real effect of inflammaging on reproductive performance [53, 61–67].
Effects of Inflammaging on Pregnancy Outcomes and Fetal Programming
Once pregnancy is established, the modulation of the maternal immune system is crucial to the embryo development and pregnancy maintenance [68]. Despite the historical idea that maternal systemic immune suppression is essential for fetal acceptance, a more complex feto–maternal immune interaction has been recently described, with both intrauterine and systemic inflammation as pivotal components of healthy pregnancies [11]. In this scenario, several pregestational maternal stressors (i.e., obesity, chronic comorbidities, advanced maternal age, malnutrition, smoking) have been associated with maladaptive changes in pregnancy and obstetric complications with disrupted intrauterine environment and fetal development. The common denominator of these stressors may be partially attributed to the excessive (prolonged or high-grade) pregestational inflammation, resulting in aberrant upregulation of systemic and placental inflammatory mediators, local and systemic oxidative stress, and increased short- and long-term morbidity for both mother and offspring [69].
In detail, both inflammation and premature aging have been related to placental dysfunction, leading to pregnancy pathologies and adverse pregnancy outcomes, with short- and long-term consequences in the offspring [6, 70–72]. Inflammation can generate oxidative stress and vice versa, in a negative vicious cycle leading to alterations in cell metabolism. In turn, oxidative stress and inflammation play a key role in cell aging, precipitating in cell senescence, mitochondrial dysfunction, and premature aging of the placenta. These conditions have been related to placental insufficiency [6, 73].
Maternal malnutrition, in terms of both nutrients excess and deficiency, has been reported as one of the most impacting factors on placental altered function and bioenergetics [70, 71, 74–76]. Indeed, it generates a lipotoxic intrauterine environment increasing inflammation, oxidative stress and cell senescence [76, 77].
The inflammatory response also plays a decisive role during labor enhancing myometrial contraction and cervical ripening [78, 79]. In addition, the gradual process of physiological senescence has a relevant role in the dynamic of delivery. The progressive loss of telomeres and the concomitant increase of cellular senescence inflammatory reaction which includes rising of p38, p53, p21, IL-1b, IL-6, IL-8, and senescence-associated beta-galactosidase in the decidua and fetal membranes take part in the complex phenomenon of labor onset [80]. However, excessive inflammation and its predisposing factors may accelerate the telomeres consumption and exacerbate SASP, with a consequent increase of oxidative stress damage and exacerbation of hypoxia, which turns to further deteriorate placental function and intrauterine environment [81, 82]. Therefore, such a systemic inflammatory response along with premature and accelerated aging of placenta and fetal membranes may induce pregnancy complications including recurrent pregnancy loss [83, 84], preeclampsia, fetal growth restriction (FGR), gestational diabetes mellitus (GDM), preterm birth, and premature preterm pre-labor rupture of membrane[85–89].
Many studies have investigated the association of recurrent pregnancy loss (RPL) with immune dysfunction and inflammatory imbalance. Chronic inflammatory response may play an important role in the pathogenesis of miscarriage and implantation failure [90]. Many studies have reported high level of pro-inflammatory cytokines and chemokines, such as IL-6 [91], IL-8 [92], IL-10 [93], IL-18 [94], and TNF-α [95], in blood and decidua of women with RPL. Chronic inflammation triggers Th1 polarization of immune cells and downregulation of Th2 response weakening the maternal immune tolerance to embryo implantation and increasing the risk of miscarriage [96]. Impaired regulatory T cells (Treg) function, together with low prevalence of natural killer (NK) and dendritic cells (DC) have been widely observed in the decidua of women with RPL [90, 97]. As a result of innate immune dysfunction, chronic senescent cells accumulate in the decidua leading to SASP response has deleterious effects on implantation [84].
Previous studies on placentas of pregnancies complicated by preeclampsia, FGR, GDM, and preterm delivery have demonstrated high telomere shortening, low telomerase activity, mitochondrial dysfunctions, raised DNA oxidation, and elevated expression of senescence markers including p53, p16, and p21 [70–72, 76, 89, 98–100]. Significant increased expression of cytokines has been found in pregnant women with SARS-CoV-2 infection [101] and recent findings reported a strong association between COVID-19 during pregnancy and preeclampsia [102].
All these detrimental events contribute to create an unfavorable intrauterine environment and predispose the fetus to future adult-onset diseases [103]. Several conditions during pregnancy may contribute to the telomeres shortening, including maternal stress, smoking, malnutrition, nutritional, and sleeping disorders [20, 104–106]. On the contrary, micronutrient intakes and in particular folate has been studied as a protective factor as directly proportional to fetal telomere length [107].
Effects of Pregnancy on Inflammaging
Pregnancy as a Potential Trigger for Inflammaging
Inflammaging can affect the course and outcome of pregnancy. On the other hand, pregnancy itself might result in a triggering condition predisposing to enhanced inflammaging.
Adaptation to Pregnancy
During pregnancy, numerous physiological changes in maternal organism occur, in order to create a finely balanced system allowing to achieve a successful pregnancy in terms of fetal development and maternal survival. Hemodynamic modifications lead to a reduction of peripheric vascular resistance, as well as to increased blood volume and cardiac output. Moreover, pregnancy generates structural and functional changes in the cardiovascular system, such as increased left ventricular mass and aortic compliance [108]. A great number of studies have demonstrated the critical function of these changes for the physiological course of gestation and the association between suboptimal adaptations and obstetrical complications, such as preeclampsia and FGR [109–111].
Aging leads as well to multisystemic structural and functional alterations which tend to conflict with the suitable gestational adaptations. In fact, biological aging manifestations include an increased arterial stiffness and sympathetic nervous system activity with a loss of vascular compliance, antioxidant activity, and vascular endothelial cell function [112, 113]. All these conditions make the woman more susceptible to obstetric complications and may further impact on future offspring health by injuring intrauterine full genetically defined development.
Pregnancy as a Stress Test on Women
In a more general perspective and as a consequence of extended systemic adaptative changes in maternal body, pregnancy represents a critical period during female life acting as a stress test and possibly unmasking latent underhanded pathological processes. This is particularly evident in case of underlying conditions requiring systemic pregestational adaptation and effort for maternal organism, whose precarious state may be deranged by the overlying pregnancy. Several conditions such as chronic pregestational diseases and metabolic dysfunctions, together with the cumulative effects of aging, play a pivotal role in reducing maternal reserve and ability to adapt to pregnancy, enhancing the vulnerability of the system. In this vision, pregnancy acts as a stress test and sub-optimal systemic modifications may compromise both gestational success and post-gestational woman health.
In facts, hemodynamic modifications, as well as the insulin resistance induced by pregnancy, represent a strong trigger for the onset of metabolic and cardiovascular diseases later in life. The effect of pregnancy induced modifications in the pathogenesis of chronical diseases depends on the pregestational maternal condition and ability to adapt to the enhanced requests. Moreover, preeclampsia and other obstetric diseases may magnify the detrimental impact of pregnancy on the maternal systemic balance as previously suggested by Redman and Sargent [85].
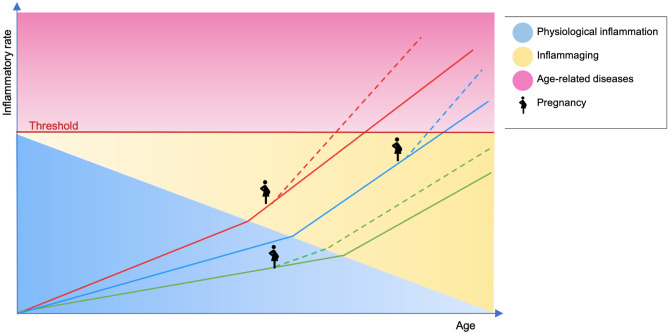
The process of inflammaging plays a leading role since it assesses the individual level of harmful conditions and available resources, characterizing baseline conditions and defining a specific risk for the onset of chronic diseases (“first hit”) [29]. Different levels of inflammaging provide a proportional risk for pathologies and, in this scenario, pregnancy and obstetric diseases may impact as a stress test or a “second hit” [85], resulting in early onset of chronic diseases [114] (Fig. 4).
Fig. 4.
Inflammaging and pregnancy effect on aging trajectories and age-related diseases outbreak. Solid lines (red, blue and green) represent the trajectories of aging of three women, diverging from the beginning as an effect of different intra-uterine environment and experiences in early life. Line slopes are affected by the occurrence of inflammaging acting as an accelerator toward the onset of age-related diseases. Dashed lines show the effect of pregnancy on the progression of aging trajectories, playing the role of a second hit accelerator to the pathological inflammatory threshold and disease outbreaks
Parity Effects on Inflammaging
The number of previous pregnancies may contribute to increased inflammaging. Previous studies revealed a correlation between the number of children and levels of inflammatory markers, including IL-6, TNF-α, and CRP, particularly strong in case of male offspring [115, 116]. This different impact may be explained by the Y-chromosome related antigens effect on cellular and humoral immunity which increase for each additional son and generates a greater immunological reaction increasing inflammaging [117]. Another effect of parity was shown by Pollack et al. who observed a reduction in telomeres length between nulliparous and parous women, despite adjustment for age, body mass index, and other contributing factors [118].
Inflammaging is also affected by birth sequence, timing of reproduction and inter-birth period length, since it is reasonable that advanced maternal age, shorter inter-pregnancy interval, and succession of sons altogether can impact negatively [115].
It has been supposed from a natural-history point of view that reproduction results in a significant effort which requires a considerable amount of resources and then impact on maternal post-reproductive health and longevity by means of inflammaging [119, 120].
Moreover, parity may result in an accumulation of maladaptive modifications induced by pregnancy such as weight gain, increment of abdominal fat, insulin resistance, elevation of proatherogenic lipid levels, endothelial dysfunction, and cardiometabolic alterations. The pregestational state of health and inflammatory status together with the number and outcome of pregnancies can therefore influence future levels of inflammaging and the consequent incidence of chronic diseases.
Fetal Microchimerism
During gestation, small DNA fragments derived from apoptotic and necrotic fetal-placental cells are disseminated into the maternal circulation [121]. The presence of non-self cells and DNA is defined as cellular fetal microchimerism (cFMC) [122]. Typically, the rate of cell-free fetal DNA (cffDNA) progressively rises with gestational age and then rapidly drops after delivery, even though it may persist in maternal tissues for a long time [123]. This fetal DNA generates an inflammatory cascade, by activating NF-κB and stimulator of inteferon genes (STING), with a consequent release of pro-inflammatory cytokines and chemokines such as IL-6, IFN-β and IFN-α, by means of TLR-9 [35, 124, 125].
Several epidemiological studies have investigated the association of a cFMC and pregnancy complications reporting conflicting evidence due to the vast heterogeneity of populations and methodologies [126]. However, the cffDNA rate appears to correlate with placental dysfunction, being detected in greater quantities in women with preeclampsia and HELLP, FGR, and preterm labor compared to gestational age-matched uncomplicated pregnancies [127]. cFMC impacts on maternal health with both beneficial and detrimental effects and, in this way, it can influence the inflammaging process. Indeed, some fetal cells act as a stem cells and can differentiate into endothelial cells, neurons, cardiomyocytes, and smooth muscle cells [122]. Thus, such stem cells supply may contrast tissue degeneration and cell loss related to aging [128]. cFMC seems to affect maternal health either in a positive and negative way, by means of several mechanisms which are mostly not completely understood. More specifically, cFMC exerts a negative effect on autoimmune diseases, contributing to the outbreak or the worsening in non-pregnant parous women of several conditions, such as Hashimoto’s thyroiditis, Grave’s disease, systemic sclerosis, systemic lupus erythematosus, Sjogren’s syndrome, rheumatoid arthritis, multiple sclerosis, and primary biliary cirrhosis. The pathogenic basis of this detrimental effect on autoimmunity is still not fully known, but it seems that cFMC may induce an alloimmune response against the fetal human leukocyte antigens peptides [129].
Moreover, studies on cFMC and cardiovascular diseases have reported controversial findings, with a trend toward elevate cFMC and women mortality for cardiovascular diseases. Role of cFMC and neurological decay has been not completely clarified, since research on women with Alzheimer’s disease has shown less presence of fetal cells compared to controls [130], whereas experimental data on animal models with Parkinson’s disease have found more fetal DNA compared to controls [131].
Effect of Obstetric Diseases
Numerous obstetric complications, including preterm delivery, hypertensive disorders of pregnancy, and gestational diabetes, are notoriously related to the pregnancy period, thus resolving by definition after delivery. Nevertheless, more and more evidence is demonstrating that a woman experiencing an adverse pregnancy outcome also presents a higher risk of developing long-term effects and chronic diseases in future life. In detail, a higher risk of long-term kidney disease and metabolic and cardiovascular disorders has been strongly associated with gestational complications, thus giving the unique opportunity to prevent future diseases by correctly managing pregnancy care [132, 133].
The pathogenesis of the association between adverse pregnancy outcome and long-term maternal health remains not fully understood, possibly involving (1) genetic predisposition and pregestational characteristics leading to deranged adaptation to pregnancy; (2) short-term obstetric complications caused by systemic endothelial dysfunction, intrauterine and systemic oxidative stress and excessive cumulative inflammation, as previously described; and (3) evident long-term disease as a result of long-lasting endothelial and metabolic dysfunction originating during pregnancy, later additional insults and aging.
Preventive Strategies and Therapeutic Options
No treatment exists against the passing of time and the aging process. However, many strategies provide chances to reach a healthy state in the elderly and to prevent the early onset of age-related diseases. The most effective treatment against the effect of inflammaging on the reproductive health is lifestyle improvement, which consists essentially in limiting the modifiable pro-inflammatory factors previously discussed, dropping down the individual basal inflammatory state. For instance, correction of BMI, a quality diet, and weight loss are primary goals to promote fertility and achieve a successful pregnancy [134]. Weight loss and lifestyle interventions during the periconceptional period may improve fertility and decrease the risk of adverse pregnancy and neonatal outcomes [135–137]. In order to get this endpoint, it is mandatory to address physical inactivity and eating disorders. Moreover, postpartum interventions promoting healthy diet and exercise may reduce the risk of weight retention and a negative rebound effect on future pregnancies. Physical activity acts as an efficacious and costless method to reduce the level of inflammation [138] and to improve psychological wellbeing, which may itself affect the inflammation status [139]. Increasing evidence shows that the Mediterranean diet (MedDiet), high in antioxidants, fiber, and mono- and polyunsaturated fatty acids, has an immunomodulant effect on the systemic inflammatory balance, leading to a boost of the anti-inflammatory response and contrasting the progressive increase of inflammaging [140]. Several studies disclosed a positive effect of MedDiet among subfertile couples, increasing the chance of spontaneous pregnancy and IVF success [141–143]. MedDiet also impacts on maternal and offspring health, improving embryonic and fetal growth [144], and decreasing the risk of GDM, pre-eclampsia, and preterm birth [145, 146]. Additionally, a correct dietary intake of essential micronutrients (zinc, copper, selenium) stimulates the immune system and antioxidant enzymes, with a strong epigenetic effect on many genes related to inflammation and oxidative stress, contributing to the rebalancing of the inflammatory state and the integrity of metabolic function [147]. A specific immunomodulatory role has also been demonstrated for vitamin D and many guidelines indicate supplementation with vitamin D during pregnancy for the prevention of pregnancy complications [148]
Individual nutritional assessment in the periconceptional period may prove useful in addressing specific nutritional deficiencies with simple scoring systems to provide advice during obstetrical assessment [149]. Besides this, it is mandatory to reduce all potential sources of increased inflammation, such as smoking [21] but also by implementing practices such as oral hygiene [28]
Another promising, simple, and safe strategy for reducing inflammaging is the use of probiotics, which provides many advantages during the periconceptional period, pregnancy and breastfeeding, lowering bacterial vaginosis, GDM, Streptococcus B haemolyticus colonization, postpartum depression, and atopic sensitization [150–153]. Probiotics play a pivotal role as an immunomodulant, downregulating local pro-inflammatory cytokines, and promoting anti-inflammatory signaling [153]. It also improves the protective effect of commensal microbiota community against the detrimental, proinflammatory impact of dysbiosis [154]. Maternal probiotics may also affect offspring future health by influencing the primary bacterial gut colonization, which is known to influence the immune system and the inflammatory status since the first days of life [153].
Future research needs to explore the effect of intervention strategies (i.e., nutritional, supplementations, anti-inflammatory molecules, i.e., ASA or heparin) on markers of inflammaging, systemic and local inflammatory status, and reproductive/pregnancy outcomes.
Conclusions
Women future health and longevity are strongly related to the rate of progression of inflammaging, which can affect female reproductive function in manyfold ways. In fact, inflammaging combines the effect of all processes involved in chronic inflammation and in aging course, with a multifaced consequence on fertility and chance to achieve a successful pregnancy. Furthermore, maternal inflammaging has a transgenerational impact on the future adult fetal life and for all these effects, it should be considered as a crucial factor for healthy life span both of the mother and the fetus. Therefore, as a matter of public health improvement, clinicians and healthcare providers should offer strategies to adjust women lifestyle and behavior before and during pregnancy, in order to decrease inflammaging status and then reduce the rate of adverse short- and long-term outcomes for the woman and her offspring. Interventions should focus on the reduction of risk factors leading to inflammaging, such as obesity, western diet, stress, and microbiota dysregulation. As aging is a unidirectional process and cannot be reversed, therapeutic interventions should also be focused on reducing the causes of aging acceleration, such as oxidative stress and mitochondrial dysfunctions.
Declarations
Competing Interests
The authors declared no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Franceschi C, Bonafè M, Valensin S, et al. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann N Y Acad Sci. 2000;908:244–254. doi: 10.1111/j.1749-6632.2000.tb06651.x. [DOI] [PubMed] [Google Scholar]
- 2.Huang Y, Hu C, Ye H, et al. Inflamm-aging: a new mechanism affecting premature ovarian insufficiency. J Immunol Res. 2019;2019:8069898. doi: 10.1155/2019/8069898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Weiss G, Goldsmith LT, Taylor RN, et al. Inflammation in reproductive disorders. Reprod Sci. 2009;16:216–229. doi: 10.1177/1933719108330087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shirasuna K, Iwata H. Effect of aging on the female reproductive function. Contracept Reprod Med. 2017;2:23. doi: 10.1186/s40834-017-0050-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Storer M, Mas A, Robert-Moreno A, et al. Senescence is a developmental mechanism that contributes to embryonic growth and patterning. Cell. 2013;155:1119–1130. doi: 10.1016/j.cell.2013.10.041. [DOI] [PubMed] [Google Scholar]
- 6.Manna S, McCarthy C, McCarthy FP. Placental ageing in adverse pregnancy outcomes: telomere shortening, cell senescence, and mitochondrial dysfunction. Oxid Med Cell Longev. 2019;2019:3095383. doi: 10.1155/2019/3095383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pantham P, Aye ILMH, Powell TL. Inflammation in maternal obesity and gestational diabetes mellitus. Placenta. 2015;36:709–715. doi: 10.1016/j.placenta.2015.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ingvorsen C, Brix S, Ozanne SE, et al. The effect of maternal Inflammation on foetal programming of metabolic disease. Acta Physiol (Oxf) 2015;214:440–449. doi: 10.1111/apha.12533. [DOI] [PubMed] [Google Scholar]
- 9.Segovia SA, Vickers MH, Gray C, et al. Maternal obesity, inflammation, and developmental programming. Biomed Res Int. 2014;2014:418975. doi: 10.1155/2014/418975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Larbi A, Franceschi C, Mazzatti D, et al. Aging of the immune system as a prognostic factor for human longevity. Physiology (Bethesda) 2008;23:64–74. doi: 10.1152/physiol.00040.2007. [DOI] [PubMed] [Google Scholar]
- 11.Parisi F, Milazzo R, Savasi VM et al (2021) Maternal low-grade chronic inflammation and intrauterine programming of health and disease. Int J Mol Sci 22. 10.3390/ijms22041732 [DOI] [PMC free article] [PubMed]
- 12.Gregor MF, Hotamisligil GS. Inflammatory mechanisms in obesity. Annu Rev Immunol. 2011;29:415–445. doi: 10.1146/annurev-immunol-031210-101322. [DOI] [PubMed] [Google Scholar]
- 13.Weisberg SP, McCann D, Desai M, et al. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112:1796–1808. doi: 10.1172/JCI19246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chawla A, Nguyen KD, Goh YPS. Macrophage-mediated inflammation in metabolic disease. Nat Rev Immunol. 2011;11:738–749. doi: 10.1038/nri3071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cani PD, Amar J, Iglesias MA, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56:1761–1772. doi: 10.2337/db06-1491. [DOI] [PubMed] [Google Scholar]
- 16.Raetz CRH, Whitfield C. Lipopolysaccharide endotoxins. Annu Rev Biochem. 2002;71:635–700. doi: 10.1146/annurev.biochem.71.110601.135414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee JY, Sohn KH, Rhee SH, et al. Saturated fatty acids, but not unsaturated fatty acids, induce the expression of cyclooxygenase-2 mediated through Toll-like receptor 4. J Biol Chem. 2001;276:16683–16689. doi: 10.1074/jbc.M011695200. [DOI] [PubMed] [Google Scholar]
- 18.Bailey MA, Holscher HD. Microbiome-mediated effects of the mediterranean diet on inflammation. Adv Nutr. 2018;9:193–206. doi: 10.1093/advances/nmy013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Franzago M, Fraticelli F, Stuppia L, et al. Nutrigenetics, epigenetics and gestational diabetes: consequences in mother and child. Epigenetics. 2019;14:215–235. doi: 10.1080/15592294.2019.1582277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cetin I, Mandò C, Calabrese S. Maternal predictors of intrauterine growth restriction. Curr Opin Clin Nutr Metab Care. 2013;16:310–319. doi: 10.1097/MCO.0b013e32835e8d9c. [DOI] [PubMed] [Google Scholar]
- 21.Doherty SP, Grabowski J, Hoffman C, et al. Early life insult from cigarette smoke may be predictive of chronic diseases later in life. Biomarkers. 2009;14(Suppl 1):97–101. doi: 10.1080/13547500902965898. [DOI] [PubMed] [Google Scholar]
- 22.World Health Organization (2012) Possible developmental early effects of endocrine disrupters on child health. World Health Organization. https://apps.who.int/iris/handle/10665/75342
- 23.Alavian-Ghavanini A, Rüegg J. Understanding epigenetic effects of endocrine disrupting chemicals: from mechanisms to novel test methods. Basic Clin Pharmacol Toxicol. 2018;122:38–45. doi: 10.1111/bcpt.12878. [DOI] [PubMed] [Google Scholar]
- 24.Hu J, Ly J, Zhang W, et al. Microbiota of newborn meconium is associated with maternal anxiety experienced during pregnancy. Dev Psychobiol. 2019;61:640–649. doi: 10.1002/dev.21837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bolten MI, Wurmser H, Buske-Kirschbaum A, et al. Cortisol levels in pregnancy as a psychobiological predictor for birth weight. Arch Womens Ment Health. 2011;14:33–41. doi: 10.1007/s00737-010-0183-1. [DOI] [PubMed] [Google Scholar]
- 26.Xu K, Wei Y, Giunta S, et al. Do inflammaging and coagul-aging play a role as conditions contributing to the co-occurrence of the severe hyper-inflammatory state and deadly coagulopathy during COVID-19 in older people? Exp Gerontol. 2021;151:111423. doi: 10.1016/j.exger.2021.111423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Baltacıoğlu E, Kehribar MA, Yuva P, et al. Total oxidant status and bone resorption biomarkers in serum and gingival crevicular fluid of patients with periodontitis. J Periodontol. 2014;85:317–326. doi: 10.1902/jop.2013.130012. [DOI] [PubMed] [Google Scholar]
- 28.Zambon M, Mandò C, Lissoni A, et al. Inflammatory and oxidative responses in pregnancies with obesity and periodontal disease. Reprod Sci. 2018;25:1474–1484. doi: 10.1177/1933719117749758. [DOI] [PubMed] [Google Scholar]
- 29.Franceschi C, Garagnani P, Parini P, et al. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat Rev Endocrinol. 2018;14:576–590. doi: 10.1038/s41574-018-0059-4. [DOI] [PubMed] [Google Scholar]
- 30.Mor G, Cardenas I, Abrahams V, et al. Inflammation and pregnancy: the role of the immune system at the implantation site. Ann N Y Acad Sci. 2011;1221:80–87. doi: 10.1111/j.1749-6632.2010.05938.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gomez CR, Boehmer ED, Kovacs EJ. The aging innate immune system. Curr Opin Immunol. 2005;17:457–462. doi: 10.1016/j.coi.2005.07.013. [DOI] [PubMed] [Google Scholar]
- 32.Castle SC. Clinical relevance of age-related immune dysfunction. Clin Infect Dis. 2000;31:578–585. doi: 10.1086/313947. [DOI] [PubMed] [Google Scholar]
- 33.Gavazzi G, Krause K-H. Ageing and infection. The Lancet Infectious Diseases. 2002;2:659–666. doi: 10.1016/s1473-3099(02)00437-1. [DOI] [PubMed] [Google Scholar]
- 34.Bjornsson HT, Sigurdsson MI, Fallin MD, et al. Intra-individual change over time in DNA methylation with familial clustering. JAMA. 2008;299:2877–2883. doi: 10.1001/jama.299.24.2877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Phillippe M. Cell-Free Fetal DNA, Telomeres, and the spontaneous onset of parturition. Reprod Sci. 2015;22:1186–1201. doi: 10.1177/1933719115592714. [DOI] [PubMed] [Google Scholar]
- 36.Wang Y, Wang X, Flores ER, et al. Dysfunctional telomeres induce p53-dependent and independent apoptosis to compromise cellular proliferation and inhibit tumor formation. Aging Cell. 2016;15:646–660. doi: 10.1111/acel.12476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen J-H, Hales CN, Ozanne SE. DNA damage, cellular senescence and organismal ageing: causal or correlative? Nucleic Acids Res. 2007;35:7417–7428. doi: 10.1093/nar/gkm681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hemann MT, Strong MA, Hao L-Y, et al. The shortest telomere, not average telomere length, is critical for cell viability and chromosome stability. Cell. 2001;107:67–77. doi: 10.1016/s0092-8674(01)00504-9. [DOI] [PubMed] [Google Scholar]
- 39.Zhang J, Rane G, Dai X, et al. Ageing and the telomere connection: an intimate relationship with inflammation. Ageing Res Rev. 2016;25:55–69. doi: 10.1016/j.arr.2015.11.006. [DOI] [PubMed] [Google Scholar]
- 40.Coppé J-P, Patil CK, Rodier F, et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6:2853–2868. doi: 10.1371/journal.pbio.0060301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kirkland JL, Tchkonia T. Cellular senescence: a translational perspective. EBioMedicine. 2017;21:21–28. doi: 10.1016/j.ebiom.2017.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lee HW, Blasco MA, Gottlieb GJ, et al. Essential role of mouse telomerase in highly proliferative organs. Nature. 1998;392:569–574. doi: 10.1038/33345. [DOI] [PubMed] [Google Scholar]
- 43.Liu L, Franco S, Spyropoulos B, et al. Irregular telomeres impair meiotic synapsis and recombination in mice. Proc Natl Acad Sci U S A. 2004;101:6496–6501. doi: 10.1073/pnas.0400755101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Butts S, Riethman H, Ratcliffe S, et al. Correlation of telomere length and telomerase activity with occult ovarian insufficiency. J Clin Endocrinol Metab. 2009;94:4835–4843. doi: 10.1210/jc.2008-2269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cheng E-H, Chen S-U, Lee T-H, et al. Evaluation of telomere length in cumulus cells as a potential biomarker of oocyte and embryo quality. Hum Reprod. 2013;28:929–936. doi: 10.1093/humrep/det004. [DOI] [PubMed] [Google Scholar]
- 46.Broekmans FJ, Soules MR, Fauser BC. Ovarian aging: mechanisms and clinical consequences. Endocr Rev. 2009;30:465–493. doi: 10.1210/er.2009-0006. [DOI] [PubMed] [Google Scholar]
- 47.Lliberos C, Liew SH, Zareie P, et al. Evaluation of inflammation and follicle depletion during ovarian ageing in mice. Sci Rep. 2021;11:278. doi: 10.1038/s41598-020-79488-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhang Z, Schlamp F, Huang L, et al. Inflammaging is associated with shifted macrophage ontogeny and polarization in the aging mouse ovary. Reproduction. 2020;159:325–337. doi: 10.1530/REP-19-0330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Briley SM, Jasti S, McCracken JM, et al. Reproductive age-associated fibrosis in the stroma of the mammalian ovary. Reproduction. 2016;152:245–260. doi: 10.1530/REP-16-0129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Tanikawa N, Ohtsu A, Kawahara-Miki R, et al. Age-associated mRNA expression changes in bovine endometrial cells in vitro. Reprod Biol Endocrinol. 2017;15:63. doi: 10.1186/s12958-017-0284-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Burnum KE, Hirota Y, Baker ES, et al. Uterine deletion of Trp53 compromises antioxidant responses in the mouse decidua. Endocrinology. 2012;153:4568–4579. doi: 10.1210/en.2012-1335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fortin CS, Leader A, Mahutte N, et al. Gene expression analysis of follicular cells revealed inflammation as a potential IVF failure cause. J Assist Reprod Genet. 2019;36:1195–1210. doi: 10.1007/s10815-019-01447-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Brouillet S, Boursier G, Anav M, et al. C-reactive protein and ART outcomes: a systematic review. Hum Reprod Update. 2020;26:753–773. doi: 10.1093/humupd/dmaa012. [DOI] [PubMed] [Google Scholar]
- 54.Burnik Papler T, Vrtačnik Bokal E, Maver A, et al. Specific gene expression differences in cumulus cells as potential biomarkers of pregnancy. Reprod Biomed Online. 2015;30:426–433. doi: 10.1016/j.rbmo.2014.12.011. [DOI] [PubMed] [Google Scholar]
- 55.Hamel M, Dufort I, Robert C, et al. Identification of differentially expressed markers in human follicular cells associated with competent oocytes. Hum Reprod. 2008;23:1118–1127. doi: 10.1093/humrep/den048. [DOI] [PubMed] [Google Scholar]
- 56.Hamel M, Dufort I, Robert C, et al. Identification of follicular marker genes as pregnancy predictors for human IVF: new evidence for the involvement of luteinization process. Mol Hum Reprod. 2010;16:548–556. doi: 10.1093/molehr/gaq051. [DOI] [PubMed] [Google Scholar]
- 57.Gebhardt KM, Feil DK, Dunning KR, et al. Human cumulus cell gene expression as a biomarker of pregnancy outcome after single embryo transfer. Fertil Steril. 2011;96:47–52.e2. doi: 10.1016/j.fertnstert.2011.04.033. [DOI] [PubMed] [Google Scholar]
- 58.Azkargorta M, Escobes I, Iloro I, et al. Differential proteomic analysis of endometrial fluid suggests increased inflammation and impaired glucose metabolism in non-implantative IVF cycles and pinpoints PYGB as a putative implantation marker. Hum Reprod. 2018;33:1898–1906. doi: 10.1093/humrep/dey274. [DOI] [PubMed] [Google Scholar]
- 59.Palumbo A, Ávila J, Naftolin F. The ovarian renin-angiotensin system (OVRAS): a major factor in ovarian function and disease. Reprod Sci. 2016;23:1644–1655. doi: 10.1177/1933719116672588. [DOI] [PubMed] [Google Scholar]
- 60.Vloeberghs V, Peeraer K, Pexsters A, et al. Ovarian hyperstimulation syndrome and complications of ART. Best Pract Res Clin Obstet Gynaecol. 2009;23:691–709. doi: 10.1016/j.bpobgyn.2009.02.006. [DOI] [PubMed] [Google Scholar]
- 61.Sahin A, Engin-Ustun Y, Tokmak A, et al. Serum levels of transforming growth factor β1 and C-reactive protein as possible markers of intra uterine insemination outcome. Eur Cytokine Netw. 2018;29:121–126. doi: 10.1684/ecn.2018.0418. [DOI] [PubMed] [Google Scholar]
- 62.Robinson S, Pemberton P, Laing I, et al. Low grade inflammation, as evidenced by basal high sensitivity CRP, is not correlated to outcome measures in IVF. J Assist Reprod Genet. 2008;25:383–388. doi: 10.1007/s10815-008-9253-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Almagor M, Hazav A, Yaffe H. The levels of C-reactive protein in women treated by IVF. Hum Reprod. 2004;19:104–106. doi: 10.1093/humrep/deh036. [DOI] [PubMed] [Google Scholar]
- 64.Sacks GP, Trew G, Lavery S (2004) C-reactive protein in women treated by IVF. Hum Reprod 19:1677; author reply 1677-8. 10.1093/humrep/deh242 [DOI] [PubMed]
- 65.Wunder DM, Kretschmer R, Bersinger NA. Concentrations of leptin and C-reactive protein in serum and follicular fluid during assisted reproductive cycles. Hum Reprod. 2005;20:1266–1271. doi: 10.1093/humrep/deh767. [DOI] [PubMed] [Google Scholar]
- 66.Yildizfer F, Donma O, Yen M et al (2015) In vitro fertilization, levels of pro-inflammatory factors and lipid peroxidation. Int J Fertil Steril 9:277–284. 10.22074/ijfs.2015.4541 [DOI] [PMC free article] [PubMed]
- 67.Buyuk E, Asemota OA, Merhi Z, et al. Serum and follicular fluid monocyte chemotactic protein-1 levels are elevated in obese women and are associated with poorer clinical pregnancy rate after in vitro fertilization: a pilot study. Fertil Steril. 2017;107:632–640.e3. doi: 10.1016/j.fertnstert.2016.12.023. [DOI] [PubMed] [Google Scholar]
- 68.Thiele K, Diao L, Arck PC. Immunometabolism, pregnancy, and nutrition. Semin Immunopathol. 2018;40:157–174. doi: 10.1007/s00281-017-0660-y. [DOI] [PubMed] [Google Scholar]
- 69.Romero R, Gotsch F, Pineles B, et al. Inflammation in pregnancy: its roles in reproductive physiology, obstetrical complications, and fetal injury. Nutr Rev. 2007;65:S194–202. doi: 10.1111/j.1753-4887.2007.tb00362.x. [DOI] [PubMed] [Google Scholar]
- 70.Diceglie C, Anelli GM, Martelli C et al (2021) Placental antioxidant defenses and autophagy-related genes in maternal obesity and gestational diabetes mellitus. Nutrients 13. 10.3390/nu13041303 [DOI] [PMC free article] [PubMed]
- 71.Mandò C, Anelli GM, Novielli C, et al. Impact of obesity and hyperglycemia on placental mitochondria. Oxid Med Cell Longev. 2018;2018:2378189. doi: 10.1155/2018/2378189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Mandò C, de Palma C, Stampalija T, et al. Placental mitochondrial content and function in intrauterine growth restriction and preeclampsia. Am J Physiol Endocrinol Metab. 2014;306:E404–13. doi: 10.1152/ajpendo.00426.2013. [DOI] [PubMed] [Google Scholar]
- 73.Menon R. Oxidative stress damage as a detrimental factor in preterm birth pathology. Front Immunol. 2014;5:567. doi: 10.3389/fimmu.2014.00567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Bianchi C, Taricco E, Cardellicchio M, et al. The role of obesity and gestational diabetes on placental size and fetal oxygenation. Placenta. 2021;103:59–63. doi: 10.1016/j.placenta.2020.10.013. [DOI] [PubMed] [Google Scholar]
- 75.Mandò C, Calabrese S, Mazzocco MI, et al. Sex specific adaptations in placental biometry of overweight and obese women. Placenta. 2016;38:1–7. doi: 10.1016/j.placenta.2015.12.008. [DOI] [PubMed] [Google Scholar]
- 76.Fattuoni C, Mandò C, Palmas F, et al. Preliminary metabolomics analysis of placenta in maternal obesity. Placenta. 2018;61:89–95. doi: 10.1016/j.placenta.2017.11.014. [DOI] [PubMed] [Google Scholar]
- 77.Robker RL, Wu LL-Y, Yang X. Inflammatory pathways linking obesity and ovarian dysfunction. J Reprod Immunol. 2011;88:142–148. doi: 10.1016/j.jri.2011.01.008. [DOI] [PubMed] [Google Scholar]
- 78.Abrahams VM. Thirty years of reproductive immunology: an introduction. Am J Reprod Immunol. 2010;63:411–412. doi: 10.1111/j.1600-0897.2010.00849.x. [DOI] [PubMed] [Google Scholar]
- 79.Nadeau-Vallée M, Obari D, Palacios J, et al. Sterile inflammation and pregnancy complications: a review. Reproduction. 2016;152:R277–R292. doi: 10.1530/REP-16-0453. [DOI] [PubMed] [Google Scholar]
- 80.Menon R, Behnia F, Polettini J et al (2016) Placental membrane aging and HMGB1 signaling associated with human parturition. Aging (Albany NY) 8:216–230. 10.18632/aging.100891 [DOI] [PMC free article] [PubMed]
- 81.Hirata Y, Katsukura Y, Henmi Y, et al. Advanced maternal age induces fetal growth restriction through decreased placental inflammatory cytokine expression and immune cell accumulation in mice. J Reprod Dev. 2021 doi: 10.1262/jrd.2021-034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cetin I, Taricco E, Mandò C, et al. Fetal oxygen and glucose consumption in human pregnancy complicated by fetal growth restriction. Hypertension. 2020;75:748–754. doi: 10.1161/HYPERTENSIONAHA.119.13727. [DOI] [PubMed] [Google Scholar]
- 83.Laird SM, Tuckerman EM, Cork BA, et al. A review of immune cells and molecules in women with recurrent miscarriage. Hum Reprod Update. 2003;9:163–174. doi: 10.1093/humupd/dmg013. [DOI] [PubMed] [Google Scholar]
- 84.Lucas ES, Vrljicak P, Muter J, et al. Recurrent pregnancy loss is associated with a pro-senescent decidual response during the peri-implantation window. Commun Biol. 2020;3:37. doi: 10.1038/s42003-020-0763-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Redman CWG, Sargent IL. Immunology of pre-eclampsia. Am J Reprod Immunol. 2010;63:534–543. doi: 10.1111/j.1600-0897.2010.00831.x. [DOI] [PubMed] [Google Scholar]
- 86.Cox LS, Redman C. The role of cellular senescence in ageing of the placenta. Placenta. 2017;52:139–145. doi: 10.1016/j.placenta.2017.01.116. [DOI] [PubMed] [Google Scholar]
- 87.Menon R, Boldogh I, Hawkins HK, et al. Histological evidence of oxidative stress and premature senescence in preterm premature rupture of the human fetal membranes recapitulated in vitro. Am J Pathol. 2014;184:1740–1751. doi: 10.1016/j.ajpath.2014.02.011. [DOI] [PubMed] [Google Scholar]
- 88.Dutta EH, Behnia F, Boldogh I, et al. Oxidative stress damage-associated molecular signaling pathways differentiate spontaneous preterm birth and preterm premature rupture of the membranes. Mol Hum Reprod. 2016;22:143–157. doi: 10.1093/molehr/gav074. [DOI] [PubMed] [Google Scholar]
- 89.Sultana Z, Maiti K, Aitken J et al (2017) Oxidative stress, placental ageing-related pathologies and adverse pregnancy outcomes. Am J Reprod Immunol 77. 10.1111/aji.12653 [DOI] [PubMed]
- 90.Negishi Y, Shima Y, Takeshita T, et al. Harmful and beneficial effects of inflammatory response on reproduction: sterile and pathogen-associated inflammation. Immunol Med. 2021;44:98–115. doi: 10.1080/25785826.2020.1809951. [DOI] [PubMed] [Google Scholar]
- 91.Zenclussen AC, Kortebani G, Mazzolli A, et al. Interleukin-6 and soluble interleukin-6 receptor serum levels in recurrent spontaneous abortion women immunized with paternal white cells. Am J Reprod Immunol. 2000;44:22–29. doi: 10.1111/j.8755-8920.2000.440104.x. [DOI] [PubMed] [Google Scholar]
- 92.Hattori Y, Nakanishi T, Ozaki Y, et al. Uterine cervical inflammatory cytokines, interleukin-6 and -8, as predictors of miscarriage in recurrent cases. Am J Reprod Immunol. 2007;58:350–357. doi: 10.1111/j.1600-0897.2007.00516.x. [DOI] [PubMed] [Google Scholar]
- 93.Jianchun S, Dachun H, Jun C, Yundong L, Zhongmi W (2012) The correlation between unexplained recurrent spontaneous abortion and cytokine immune balance. Lab Med Clin 568–569
- 94.Yan S, Lin S, Pei C, Jiaquan L, Jinchun Y (2012) The expression of interleukin-6 and interleukin-18 mRNA in villi of patients with recurrent spontaneous abortion. Guangdong Med J 33
- 95.Zhang C, Deng X, Zhang X, et al. Association between Serum TNF-α levels and recurrent spontaneous miscarriage: a meta-analysis. Am J Reprod Immunol. 2016;75:86–93. doi: 10.1111/aji.12447. [DOI] [PubMed] [Google Scholar]
- 96.Kwak-Kim JYH, Chung-Bang HS, Ng SC, et al. Increased T helper 1 cytokine responses by circulating T cells are present in women with recurrent pregnancy losses and in infertile women with multiple implantation failures after IVF. Hum Reprod. 2003;18:767–773. doi: 10.1093/humrep/deg156. [DOI] [PubMed] [Google Scholar]
- 97.Huang N, Chi H, Qiao J. Role of regulatory T cells in regulating fetal-maternal immune tolerance in healthy pregnancies and reproductive diseases. Front Immunol. 2020;11:1023. doi: 10.3389/fimmu.2020.01023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Menon R, Yu J, Basanta-Henry P, et al. Short fetal leukocyte telomere length and preterm prelabor rupture of the membranes. PLoS One. 2012;7:e31136. doi: 10.1371/journal.pone.0031136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Biron-Shental T, Sukenik Halevy R, Goldberg-Bittman L, et al. Telomeres are shorter in placental trophoblasts of pregnancies complicated with intrauterine growth restriction (IUGR) Early Hum Dev. 2010;86:451–456. doi: 10.1016/j.earlhumdev.2010.06.002. [DOI] [PubMed] [Google Scholar]
- 100.Assi E, D'Addio F, Mandò C et al (2020) Placental proteome abnormalities in women with gestational diabetes and large-for-gestational-age newborns. BMJ Open Diabetes Res Care 8. 10.1136/bmjdrc-2020-001586 [DOI] [PMC free article] [PubMed]
- 101.Fenizia C, Biasin M, Cetin I, et al. Analysis of SARS-CoV-2 vertical transmission during pregnancy. Nat Commun. 2020;11:5128. doi: 10.1038/s41467-020-18933-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Papageorghiou AT, Deruelle P, Gunier RB, et al. Preeclampsia and COVID-19: results from the INTERCOVID prospective longitudinal study. Am J Obstet Gynecol. 2021 doi: 10.1016/j.ajog.2021.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Barker DJ. In utero programming of chronic disease. Clin Sci (Lond) 1998;95:115–128. doi: 10.1042/CS19980019. [DOI] [PubMed] [Google Scholar]
- 104.Send TS, Gilles M, Codd V, et al. Telomere length in newborns is related to maternal stress during pregnancy. Neuropsychopharmacology. 2017;42:2407–2413. doi: 10.1038/npp.2017.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Menon R, Mesiano S, Taylor RN. Programmed fetal membrane senescence and exosome-mediated signaling: a mechanism associated with timing of human parturition. Front Endocrinol (Lausanne) 2017;8:196. doi: 10.3389/fendo.2017.00196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Whiteman VE, Goswami A, Salihu HM (2017) Telomere length and fetal programming: a review of recent scientific advances. Am J Reprod Immunol 77. 10.1111/aji.12661 [DOI] [PubMed]
- 107.Louis-Jacques AF, Salihu HM, King LM, et al. A positive association between umbilical cord RBC folate and fetal TL at birth supports a potential for fetal reprogramming. Nutr Res. 2016;36:703–709. doi: 10.1016/j.nutres.2016.01.009. [DOI] [PubMed] [Google Scholar]
- 108.May L. Cardiac Physiology of Pregnancy. Compr Physiol. 2015;5:1325–1344. doi: 10.1002/cphy.c140043. [DOI] [PubMed] [Google Scholar]
- 109.Mecacci F, Avagliano L, Lisi F, et al. Fetal growth restriction: does an integrated maternal hemodynamic-placental model fit better? Reprod Sci. 2020 doi: 10.1007/s43032-020-00393-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sibai B, Dekker G, Kupferminc M. Pre-eclampsia. The Lancet. 2005;365:785–799. doi: 10.1016/S0140-6736(05)17987-2. [DOI] [PubMed] [Google Scholar]
- 111.Bird IM, Zhang L, Magness RR. Possible mechanisms underlying pregnancy-induced changes in uterine artery endothelial function. Am J Physiol Regul Integr Comp Physiol. 2003;284:R245–58. doi: 10.1152/ajpregu.00108.2002. [DOI] [PubMed] [Google Scholar]
- 112.Okada Y, Galbreath MM, Shibata S, et al. Relationship between sympathetic baroreflex sensitivity and arterial stiffness in elderly men and women. Hypertension. 2012;59:98–104. doi: 10.1161/HYPERTENSIONAHA.111.176560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.El Assar M, Angulo J, Rodríguez-Mañas L. Oxidative stress and vascular inflammation in aging. Free Radic Biol Med. 2013;65:380–401. doi: 10.1016/j.freeradbiomed.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 114.Sattar N, Greer IA. Pregnancy complications and maternal cardiovascular risk: opportunities for intervention and screening? BMJ. 2002;325:157–160. doi: 10.1136/bmj.325.7356.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Galbarczyk A, Klimek M, Blukacz M, et al. Inflammaging: blame the sons. Relationships between the number of sons and the level of inflammatory mediators among post-reproductive women. Am J Phys Anthropol. 2021;175:656–664. doi: 10.1002/ajpa.24295. [DOI] [PubMed] [Google Scholar]
- 116.Marttila S, Nevalainen T, Kananen L, et al. Number of sons contributes to ageing-associated inflammation. Sci Rep. 2015;5:8631. doi: 10.1038/srep08631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Piper KP, McLarnon A, Arrazi J, et al. Functional HY-specific CD8+ T cells are found in a high proportion of women following pregnancy with a male fetus. Biol Reprod. 2007;76:96–101. doi: 10.1095/biolreprod.106.055426. [DOI] [PubMed] [Google Scholar]
- 118.Pollack AZ, Rivers K, Ahrens KA. Parity associated with telomere length among US reproductive age women. Hum Reprod. 2018;33:736–744. doi: 10.1093/humrep/dey024. [DOI] [PubMed] [Google Scholar]
- 119.Jasienska G. Reproduction and lifespan: trade-offs, overall energy budgets, intergenerational costs, and costs neglected by research. Am J Hum Biol. 2009;21:524–532. doi: 10.1002/ajhb.20931. [DOI] [PubMed] [Google Scholar]
- 120.Jasienska G. Costs of reproduction and ageing in the human female. Philos Trans R Soc Lond B Biol Sci. 2020;375:20190615. doi: 10.1098/rstb.2019.0615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Lo YMD, Corbetta N, Chamberlain PF, et al. Presence of fetal DNA in maternal plasma and serum. The Lancet. 1997;350:485–487. doi: 10.1016/S0140-6736(97)02174-0. [DOI] [PubMed] [Google Scholar]
- 122.Boddy AM, Fortunato A, Wilson Sayres M, et al. Fetal microchimerism and maternal health: a review and evolutionary analysis of cooperation and conflict beyond the womb. Bioessays. 2015;37:1106–1118. doi: 10.1002/bies.201500059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lo YM, Tein MS, Lau TK, et al. Quantitative analysis of fetal DNA in maternal plasma and serum: implications for noninvasive prenatal diagnosis. Am J Hum Genet. 1998;62:768–775. doi: 10.1086/301800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Scharfe-Nugent A, Corr SC, Carpenter SB, et al. TLR9 provokes inflammation in response to fetal DNA: mechanism for fetal loss in preterm birth and preeclampsia. J Immunol. 2012;188:5706–5712. doi: 10.4049/jimmunol.1103454. [DOI] [PubMed] [Google Scholar]
- 125.Barber GN. STING: infection, inflammation and cancer. Nat Rev Immunol. 2015;15:760–770. doi: 10.1038/nri3921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Cheng S-B, Davis S, Sharma S. Maternal-fetal cross talk through cell-free fetal DNA, telomere shortening, microchimerism, and inflammation. Am J Reprod Immunol. 2018;79:e12851. doi: 10.1111/aji.12851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Fjeldstad HE, Johnsen GM, Staff AC. Fetal microchimerism and implications for maternal health. Obstet Med. 2020;13:112–119. doi: 10.1177/1753495X19884484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Sharpless NE, DePinho RA. How stem cells age and why this makes us grow old. Nat Rev Mol Cell Biol. 2007;8:703–713. doi: 10.1038/nrm2241. [DOI] [PubMed] [Google Scholar]
- 129.Adams Waldorf KM, Nelson JL. Autoimmune disease during pregnancy and the microchimerism legacy of pregnancy. Immunol Invest. 2008;37:631–644. doi: 10.1080/08820130802205886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Chan WFN, Gurnot C, Montine TJ, et al. Male microchimerism in the human female brain. PLoS One. 2012;7:e45592. doi: 10.1371/journal.pone.0045592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Zeng XX, Tan KH, Yeo A, et al. Pregnancy-associated progenitor cells differentiate and mature into neurons in the maternal brain. Stem Cells Dev. 2010;19:1819–1830. doi: 10.1089/scd.2010.0046. [DOI] [PubMed] [Google Scholar]
- 132.Barrett PM, McCarthy FP, Kublickiene K, et al. Adverse pregnancy outcomes and long-term maternal kidney disease: a systematic review and meta-analysis. JAMA Netw Open. 2020;3:e1920964. doi: 10.1001/jamanetworkopen.2019.20964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Parikh NI, Gonzalez JM, Anderson CAM, et al. Adverse pregnancy outcomes and cardiovascular disease risk: unique opportunities for cardiovascular disease prevention in women: a scientific statement from the American Heart Association. Circulation. 2021;143:e902–e916. doi: 10.1161/CIR.0000000000000961. [DOI] [PubMed] [Google Scholar]
- 134.Stang J, Huffman LG. Position of the academy of nutrition and dietetics: obesity, reproduction, and pregnancy outcomes. J Acad Nutr Diet. 2016;116:677–691. doi: 10.1016/j.jand.2016.01.008. [DOI] [PubMed] [Google Scholar]
- 135.Espinós JJ, Polo A, Sánchez-Hernández J, et al. Weight decrease improves live birth rates in obese women undergoing IVF: a pilot study. Reprod Biomed Online. 2017;35:417–424. doi: 10.1016/j.rbmo.2017.06.019. [DOI] [PubMed] [Google Scholar]
- 136.Sim KA, Partridge SR, Sainsbury A. Does weight loss in overweight or obese women improve fertility treatment outcomes? A systematic review. Obes Rev. 2014;15:839–850. doi: 10.1111/obr.12217. [DOI] [PubMed] [Google Scholar]
- 137.Palomba S, Falbo A, Valli B, et al. Physical activity before IVF and ICSI cycles in infertile obese women: an observational cohort study. Reprod Biomed Online. 2014;29:72–79. doi: 10.1016/j.rbmo.2014.03.006. [DOI] [PubMed] [Google Scholar]
- 138.Colbert LH, Visser M, Simonsick EM, et al. Physical activity, exercise, and inflammatory markers in older adults: findings from the Health, Aging and Body Composition Study. J Am Geriatr Soc. 2004;52:1098–1104. doi: 10.1111/j.1532-5415.2004.52307.x. [DOI] [PubMed] [Google Scholar]
- 139.Coussons-Read ME, Okun ML, Nettles CD. Psychosocial stress increases inflammatory markers and alters cytokine production across pregnancy. Brain Behav Immun. 2007;21:343–350. doi: 10.1016/j.bbi.2006.08.006. [DOI] [PubMed] [Google Scholar]
- 140.Martucci M, Ostan R, Biondi F, et al. Mediterranean diet and inflammaging within the hormesis paradigm. Nutr Rev. 2017;75:442–455. doi: 10.1093/nutrit/nux013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Parisi F, Rousian M, Huijgen NA, et al. Periconceptional maternal 'high fish and olive oil, low meat' dietary pattern is associated with increased embryonic growth: The Rotterdam Periconceptional Cohort (Predict) Study. Ultrasound Obstet Gynecol. 2017;50:709–716. doi: 10.1002/uog.17408. [DOI] [PubMed] [Google Scholar]
- 142.Vujkovic M, de Vries JH, Lindemans J, et al. The preconception Mediterranean dietary pattern in couples undergoing in vitro fertilization/intracytoplasmic sperm injection treatment increases the chance of pregnancy. Fertil Steril. 2010;94:2096–2101. doi: 10.1016/j.fertnstert.2009.12.079. [DOI] [PubMed] [Google Scholar]
- 143.Twigt JM, Bolhuis MEC, Steegers EAP, et al. The preconception diet is associated with the chance of ongoing pregnancy in women undergoing IVF/ICSI treatment. Hum Reprod. 2012;27:2526–2531. doi: 10.1093/humrep/des157. [DOI] [PubMed] [Google Scholar]
- 144.Thompson JMD, Wall C, Becroft DMO, et al. Maternal dietary patterns in pregnancy and the association with small-for-gestational-age infants. Br J Nutr. 2010;103:1665–1673. doi: 10.1017/S0007114509993606. [DOI] [PubMed] [Google Scholar]
- 145.Amati F, Hassounah S, Swaka A (2019) The impact of Mediterranean dietary patterns during pregnancy on maternal and offspring health. Nutrients 11. 10.3390/nu11051098 [DOI] [PMC free article] [PubMed]
- 146.Mikkelsen TB, Osterdal ML, Knudsen VK, et al. Association between a Mediterranean-type diet and risk of preterm birth among Danish women: a prospective cohort study. Acta Obstet Gynecol Scand. 2008;87:325–330. doi: 10.1080/00016340801899347. [DOI] [PubMed] [Google Scholar]
- 147.Mocchegiani E, Costarelli L, Giacconi R, et al. Micronutrient-gene interactions related to inflammatory/immune response and antioxidant activity in ageing and inflammation. A systematic review. Mech Ageing Dev. 2014;136–137:29–49. doi: 10.1016/j.mad.2013.12.007. [DOI] [PubMed] [Google Scholar]
- 148.Berti C, Agostoni C, Davanzo R, et al. Early-life nutritional exposures and lifelong health: immediate and long-lasting impacts of probiotics, vitamin D, and breastfeeding. Nutr Rev. 2017;75:83–97. doi: 10.1093/nutrit/nuw056. [DOI] [PubMed] [Google Scholar]
- 149.Parisi F, Savasi VM, di Bartolo I et al (2020) Associations between first trimester maternal nutritional score, early markers of placental function, and pregnancy outcome. Nutrients 12. 10.3390/nu12061799 [DOI] [PMC free article] [PubMed]
- 150.Abbasi A, Aghebati-Maleki A, Yousefi M, et al. Probiotic intervention as a potential therapeutic for managing gestational disorders and improving pregnancy outcomes. J Reprod Immunol. 2021;143:103244. doi: 10.1016/j.jri.2020.103244. [DOI] [PubMed] [Google Scholar]
- 151.Barbonetti A, Cinque B, Vassallo MRC, et al. Effect of vaginal probiotic lactobacilli on in vitro-induced sperm lipid peroxidation and its impact on sperm motility and viability. Fertil Steril. 2011;95:2485–2488. doi: 10.1016/j.fertnstert.2011.03.066. [DOI] [PubMed] [Google Scholar]
- 152.Dawe JP, McCowan LME, Wilson J, et al. Probiotics and maternal mental health: a randomised controlled trial among pregnant women with obesity. Sci Rep. 2020;10:1291. doi: 10.1038/s41598-020-58129-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Swartwout B, Luo XM. Implications of Probiotics on the Maternal-Neonatal Interface: Gut Microbiota, Immunomodulation, and Autoimmunity. Front Immunol. 2018;9:2840. doi: 10.3389/fimmu.2018.02840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Barthow C, Wickens K, Stanley T, et al. The Probiotics in Pregnancy Study (PiP Study): rationale and design of a double-blind randomised controlled trial to improve maternal health during pregnancy and prevent infant eczema and allergy. BMC Pregnancy Childbirth. 2016;16:133. doi: 10.1186/s12884-016-0923-y. [DOI] [PMC free article] [PubMed] [Google Scholar]