Summary
Background
Data on emergency and critical care (ECC) capacity in low-income countries (LICs) are needed to improve outcomes and make progress towards realizing the goal of Universal Health Coverage.
Methods
We developed a novel research instrument to assess public sector ECC capacity and service readiness in LICs. From January 20th to February 18th, 2020 we administered the instrument at all four central hospitals and a simple random sample of nine of 24 district hospitals in Malawi, a landlocked and predominantly rural LIC of 19·1 million people in Southern Africa. The instrument contained questions on the availability of key resources across three domains and was administered to hospital administrators and clinicians from outpatient departments, emergency departments, and inpatient units. Results were used to generate an ECC Readiness Score, with a possible range of 0 to 1, for each facility.
Findings
A total of 114 staff members across 13 hospitals completed interviews for this study. Three (33%) district hospitals and all four central hospitals had ECC Readiness Scores above 0·5 (p-value 0·070). Absent equipment was identified as the most common barrier to ECC Readiness. Central hospitals had higher median ECC Readiness Scores with less variability 0·82 (interquartile range: 0·80–0·89) than district hospitals (0·33, 0·23 to 0·50, p-value 0·021).
Interpretation
This is the first study to employ a systematic approach to assessing ECC capacity and service readiness at both district and central hospitals in Malawi and provides a framework for measuring ECC capacity in other LICs. Prior ECC assessments potentially overestimated equipment availability and our methodology may provide a more accurate approach. There is an urgent need for investments in ECC services, particularly at district hospitals which are more accessible to Malawi's predominantly rural population. These findings highlight the need for long-term investments in health systems strengthening and underscore the importance of understanding capacity in LIC settings to inform these efforts.
Funding
Division of Pulmonary and Critical Care Medicine, Brigham and Women's Hospital and Department of Emergency Medicine, Brigham and Women's Hospital.
Keywords: Emergency and critical care, Intensive care, Health systems strengthening, Malawi, Service readiness, Universal health coverage
Research in context.
Evidence before this study
We identified seven studies reporting facility-level data on emergency and critical care (ECC) services from 244 low-income country (LIC) hospitals by searching PubMed for English language publications between Jan 1, 2000 and May 24, 2021 using terms related to ECC service delivery (“emergency care”, “critical care”, “intensive care unit”, and “high dependency unit”), critical illness (“diabetic ketoacidosis”, “respiratory failure”, “shock”, and “sepsis”), and the country setting (full list of LIC names and standard terms for LIC settings). All studies employed convenience samples and tertiary referral centres accounted for 92% of facilities in the four studies with hospital referral level information. Studies assessing critical care capacity outside of ICUs reported that critically ill patients were often managed in emergency departments and general medical wards.
Added value of this study
To our knowledge this is the first systematic national assessment of facility-level ECC capacity in an LIC. Our inclusion of secondary hospitals expands the evidence base, with increased relevance to predominantly rural LIC populations. Additionally, this is one of the few studies to assess ECC across multiple locations in the hospital, including the emergency department (or outpatient department if there was no emergency department) and inpatient wards.
Implications of all the available evidence
The available evidence shows that LICs hospitals provide treatment for critically ill patients in multiple areas, not just ICUs. There are significant gaps in ECC services, particularly at secondary hospitals, including lack of designated space, inadequate availability of equipment, and absence of clinical protocols. These findings have the potential to guide action by national governments as well as the international community to improve the availability and quality of ECC services in LICs.
Alt-text: Unlabelled box
Introduction
In the 40 years since the Alma Ata Declaration, there have been significant gains worldwide in the provision of primary care services as a cornerstone of Universal Health Coverage (UHC).1 In contrast, the development of hospital-based services has remained limited, particularly in low-income countries (LICs).2
Emergency and critical care (ECC) is the care of patients “who are critically-ill at arrival, or who were stable and subsequently deteriorated, and can be provided anywhere in the hospital: in the emergency department [ED], the intensive care unit (ICU), medical wards, post-operative recovery units, and high-dependency units [HDUs].”.3 Recent efforts have been directed at defining the most basic, core ECC capabilities that should be provided in all hospitals.4 These services have potential to provide significant benefit in LICs where critically ill patients tend to be younger with fewer comorbidities.5
In lower-middle income countries (LMICs), an estimated 54% of annual deaths are from conditions treatable by prehospital and facility-based emergency care.6 Despite this high burden of illness, information on the status of ECC services in LICs is lacking, limiting development of interventions to improve care7 as well as efforts to progress towards the objectives of UHC.
The World Health Organization (WHO) defines health service-specific readiness as “the capacity of health facilities to provide a specific service, measured through the presence of tracer items”8 organized into domains such as staff and training, equipment, and diagnostics. Standardized sets of tracer items exist for measuring readiness to deliver a number of specific services including emergency obstetric care, HIV counselling and testing, malaria diagnosis and treatment, and chronic disease management.8 However, there is no established methodology for measuring and reporting ECC service readiness.
Data from standardized assessments of ECC service readiness from LICs are needed to improve outcomes and advance towards the goal of UHC. The Malawi Emergency and Critical Care (MECC) survey is a cross-sectional study that aims to measure ECC service readiness in district and central hospitals in Malawi.
Methods
Study design and setting
Malawi is a landlocked LIC of 19·1 million people9 with 83% of the population living in rural areas.10 The national health system is organized into four tiers.11 The first two tiers are the community and primary levels, providing health services at health posts, dispensaries, village clinics, health centres, and community hospitals. The next tier comprises district hospitals, located in predominantly rural settings. The top tier consists of four central hospitals and one central psychiatric hospital. Malawi's health system faces many challenges including inadequate funding and shortages of staff, essential medicines, and equipment.11 While there are no prospective nationwide evaluations of ECC, one study showed that only 3·5% of the country's facilities are fully equipped to provide basic pediatric emergency care.12 These challenges translate to poor outcomes with inpatient pediatric mortality above 7%13 and adult sepsis mortality as high as 75%.14
The MECC survey measured cross-sectional ECC service readiness at a sample of Malawian public sector hospitals. From January 20th to February 18th, 2020, the research team visited all four full service public sector central (tertiary) hospitals and a simple random sample of nine of 24 public sector district (secondary) hospitals in Malawi. All hospitals included in the study sample granted permission for an on-site visit. Ethics approval was obtained from the Partners Healthcare Institutional Review Board in Boston, USA (2019P003457) and the National Health Science Research Committee in Malawi (Protocol #19/05/2346, approval number 2346). The Malawi Ministry of Health also approved the study. Recognizing the urgent nature of the COVID-19 pandemic, a post-hoc analysis of relevant unit-level data from the MECC survey was rapidly published in early 2020.15
Instrument
The MECC survey instrument measures unit- and facility-level ECC capacity and service readiness in LICs. The instrument contains questions drawn from three sources: the WHO Hospital Emergency Unit Assessment Tool (HEAT),16 novel questions developed and piloted by the research team to assess ECC capacity in LICs, and three expert-developed questions specific to the Malawian context.
Novel questions included in the MECC survey instrument were developed by first generating a list of concepts and topics from review of previously published instruments,16, 17, 18, 19, 20 standards established by international organizations,21 and expert opinion. Questions were then formulated within the following three primary domains: (1) systems and space; (2) essential equipment, diagnostic tests, and medications; and (3) staff. Question response structures were selected to match the WHO HEAT survey tool. Questions were iteratively refined and reduced through a modified Nominal Group Technique including content experts in emergency medicine, pulmonary and critical care medicine, global health implementation, and survey methodology. Questions were then pre-tested by outside clinical experts from LICs. In November 2018, the questions were piloted using a convenience sample of staff at an LIC district hospital. We also administered an established clinical sensibility tool for survey development to gather information on comprehensiveness, clarity, and face validity.22 The questions were further refined based on feedback from the pilot and clinical sensibility testing.
The finalized instrument collected participant-level data using WHO HEAT response structures.16 Signal function questions asked participants to describe the availability of a given resource or ability to perform an intervention on a scale of 1 to 3, with 1 indicating generally unavailable, 2 some availability, and 3 adequate availability. For signal function questions we instructed participants to consider how often all patients in their unit who need the service or resource are able get it within the timeframe needed for emergency care or critical care. Questions asking the frequency of an activity were reported on a scale of 1 to 5, with 1 indicating almost never, 2 infrequently, 3 sometimes, 4 frequently, and 5 almost always.
For each signal function rated generally unavailable or some availability, participants were asked to identify all relevant barriers. There was no limit to the number of barriers participants could identify per signal function. Barriers were coded by participants and data collectors into WHO HEAT categories of infrastructure, absent equipment, broken equipment, stockout, personnel, training, user fees, and opening hours.
Participants
Eligible participants were 18 years or older and self-reported working as a nurse or clinician (ie, clinical officer, medical assistant, or physician) for at least one month in the selected hospital area (listed below). Potential participants were identified through discussions with local hospital leadership, personal introduction during facility visits, and announcements during hospital lectures and meetings. All participants provided written informed consent.
Data collection
Using the instrument, we interviewed staff working in the following hospital areas: administration, EDs, outpatient departments (OPDs), medical wards, ICUs, and HDUs. Survey content for each unit was similar with adjustments based on varying anticipated care activities at each location. At each facility, the research team administered the survey in person to a hospital administrator and three clinicians in each of the ED, medical ward, and ICU and/or HDU, if present. For hospitals without a designated ED, the OPD was substituted as the site most likely to receive new patients arriving to the facility. Data were collected from three clinicians per unit to address discrepancies among participant responses when generating unit-level data. The survey took approximately 45 min to complete. To reduce question burden, questions on availability of protocols, electronic cardiac monitoring, crash trolley, social work, security, dietician, physiotherapist, and spiritual support were only asked to one designated clinical lead out of the three respondents from each unit.
Variables
The primary outcome was the ECC Readiness Score, which is a measure of service readiness to provide ECC. The research team developed the ECC Readiness Score using the WHO's recommended methodology for measuring service-specific readiness as “the cumulative availability of components required in health facilities to deliver specific services, expressed as percentage.”.8 Tracer items for the ECC Readiness Score were divided into three domains: (1) systems and space; (2) essential equipment, diagnostic tests, and medications; and (3) staff. The research team selected tracer items essential to basic ECC practice and analysed their availability at a facility-level by combining unit-level data from the ED/OPD and inpatient units (ie, medical ward, ICU, and HDU). To reflect the entire care continuum, a tracer item was considered present (or adequately available) at the facility-level if it was reported as available in the ED/OPD and at least one inpatient unit, with the exceptions of tracer items only applicable to specific units (ie, triage, method of identifying critically ill patients on medical wards, and twice daily reassessment of critically ill inpatients). This facility level approach was selected because ECC, by definition, occurs across multiple areas of the hospital,3 and is consistent with prior assessments of ECC capacity.19,23
To generate facility-level tracer item data, unit-level data were first calculated by averaging the three participant responses within each unit. For signal function tracer items, we considered there to be “adequate availability” within a unit if the average score was >2·5 (out of 3). For yes/no questions, the tracer item was considered present if at least two staff members answered affirmatively. For tracer items reporting frequencies, an item was considered present if the average unit score was >4 out of 5. Using emergency obstetric care service readiness as a model,17 we first calculated domain sub-scores as the unweighted proportion of adequately available tracer items and then calculated ECC Readiness Score as the unweighted mean of the three domain sub-scores so that each domain contributed 33·3% to the ECC Readiness Score.
Facility-level barrier data are reported as a percentage calculated using the frequency with which each barrier category was identified relative to the total number of times participants at the facility were asked to identify barriers (ie, frequency with which participants rated a signal function as generally unavailable or some availability).
Missing data
It is unlikely a service or resource is rapidly and reliably accessible if a clinician is unaware of its availability. Therefore, for the purposes of data analysis, a response of “don't know” was considered equivalent to “generally unavailable” or “no” for signal functions and yes/no questions, respectively. For tracer items reporting frequencies, we treated an answer of “don't know” as missing/incomplete data. Frequency variables were treated differently than other tracer items because an event may still occur even if a respondent is unaware of how often it occurs. If data were only available from two respondents at a unit, we used the same methods as above. If data were available for less than two respondents in a unit, we considered the tracer item to be missing/incomplete for that unit.
Statistical analysis
Study data were collected and managed using REDCap electronic data capture tools.24 Data were analysed in Stata (Release 16). Continuous and ordinal variables were summarized by medians and interquartile ranges. Categorical variables were described using frequencies, proportions, and 95% confidence intervals. Individual hospital data were not reported to maintain anonymity.
We compared proportions of central and district hospitals with ECC scores above 0·5. The use of 0·5 for dichotomization was selected for consistency with prior studies employing service readiness methodology.25, 26, 27 In additional analyses we compared median ECC Readiness Scores, domain sub-scores, and barrier frequencies between district and central hospitals using Fisher's Exact test. A nominal level of 5% for statistical significance (two-tailed) was used for comparisons.
We also assessed instrument reliability (see Supplement). Interrater reliability of newly developed questions was assessed by calculating the intraclass correlation coefficients for one-way random-effects models measuring average absolute agreement of participants within each hospital unit. Internal consistency was assessed by calculating Kuder-Richardson coefficients for groups of questions tapping into the same construct.
Sample size
We hypothesized that less than 20% of district hospitals surveyed would have ECC Readiness Scores above 0·5. This determination was made based on prior studies employing readiness score methodology, literature on availability of critical care resources in similar settings,17, 18, 19, 20,23,28,29 review of pilot data, and local expert opinion. The district hospital sample size of 9 was calculated using methodology recommended by the WHO Service Availability and Readiness Assessment (SARA),17 assuming a confidence level of 95 and a 15% margin of error (Supplement). A simple random sample of the 9 district hospitals included in the MECC survey was generated from a list of all 24 Malawian public sector district hospitals using a random number generator.
Study reporting
The research presented in this manuscript adheres to the reporting standards of the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) Statement and STROBE checklist for cross-sectional studies.
Role of funding source
The funders had no role in study design; collection, analysis, and interpretation of data; decision to publish; or preparation of the manuscript.
Results
Respondent and hospital characteristics
A total of 114 staff members across nine district and four central hospitals participated (Table 1); 54% were nurses. All staff members who were approached by the research team agreed to participate. We interviewed three staff members from 33 (97%) units. At the one remaining unit, we interviewed the only two staff members present during data collection and data were treated as missing for one staff member. The median facility catchment area populations were 601,000 (Interquartile range (IQR): 451,000 to 681,000) for district hospitals and 4,492,000 (3,351,000 to 6,048,000) for central hospitals, with median annual inpatient admissions of 13,300 (10,000 to 21,000) and 35,100 (27,900 to 51,600), respectively.
Table 1.
Respondent and facility characteristics.
| District hospitals | Central hospitals | |
|---|---|---|
| Respondents | ||
| Total n | 71 | 43 |
| Administrator n (%) | 9 (13%) | 4 (9%) |
| Nurse n (%) | 36 (51%) | 25 (58%) |
| Clinical officer n (%) | 19 (27%) | 5 (12%) |
| Doctor (with or without subspecialty training) n (%) | 2 (3%) | 7 (16%) |
| Medical assistant n (%) | 4 (6%) | 0 |
| Other n (%) | 0 | 2 (5%) |
| Missing/unknown n (%) | 1 (1%) | 0 |
| Number of days per week spent working in targeted unit*median (IQR) | 5 (5 to 5) | 5 (5 to 5) |
| Facilities | ||
| Total n | 9 | 4 |
| Population served (thousands) median (IQR) | 601 (451 to 681) | 4492 (3351 to 6048) |
| Inpatient beds median (IQR) | 296 (250 to 340) † | 911 (487 to 1239) |
| Annual inpatient admissions (thousands) median (IQR) | 13·3 (10·0 to 21·0) | 35·1 (27·9 to 51·6) |
| Running water n (%) | 1 (11%) | 4 (100%) |
| Electricity n (%) | 4 (44%) | 3 (75%) |
| Areas of hospital where critically ill patients are managed | ||
| HDU or ICU n (%) | 3 (33%) | 4 (100%) |
| Post-operative recovery area n (%) | 2 (22%) | 3 (75%) |
| Emergency unit^n (%) | 6 (67%) | 4 (100%) |
| Cohorted areas within general inpatient wards n (%) | 6 (67%) | 3 (75%) |
| Interspersed throughout general inpatient wards n (%) | 1 (11%) | 3 (75%) |
| Operating theater n (%) | 7 (78%) | 3 (75%) |
Does not include administrators.
Data available for 8 hospitals.
Emergency unit includes emergency departments and designated areas within outpatient departments.
Care of critically ill patients occurred across multiple hospital areas. Emergency units (defined as EDs and designated areas within OPDs) and operating theatres were the most common sites of critical care, used by 77% of all hospitals. Three (33%) district hospitals and all four central hospitals had a designated ICU and/or HDU. In addition to designated units, 3 (75%) central and 6 (67%) district hospitals also cohorted critically ill patients in general medical wards.
ECC readiness
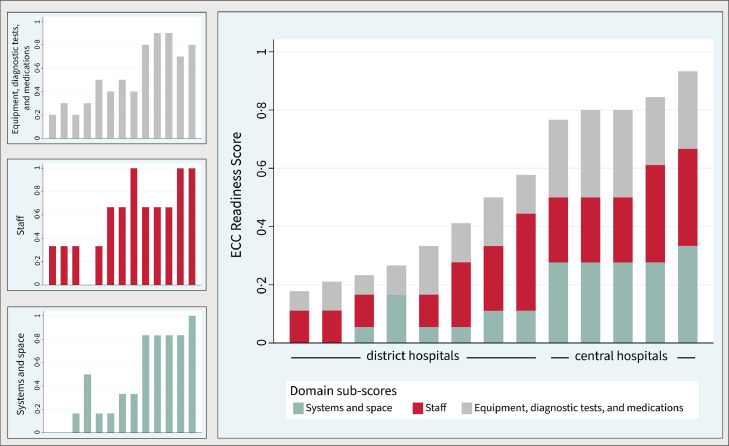
Only 3 (33%) district hospitals, compared to all 4 central hospitals (Table 2), had ECC Readiness Scores above 0·5 (p-value 0·070). Of the 3 district hospitals with HDUs, only 1 had an ECC Readiness Score above 0·5. Figure 1 shows the distribution of ECC Readiness Scores by hospital. Central hospitals had higher median ECC Readiness Scores with less variability (0·82, 0·80 to 0·89) than district hospitals (0·33, 0·23 to 0·50, p-value 0·021). Domain scores were higher for central hospitals in two of the three measured domains: (1) systems and space, and (2) essential equipment, diagnostic tests, and medications. In addition to data counted as missing for the unit with two participants, there were two tracer items within the ECC score with missing data from a single participant, representing 0·6% of responses used to calculate the ECC scores.
Table 2.
ECC readiness score and domain sub-scores.
| All hospitals (n = 13) | District hospitals (n = 9) | Central hospitals (n = 4) | p-value | |
|---|---|---|---|---|
| ECC Readiness Score ≥ 0·5 n (%) | 7 (54%) | 3 (33%) | 4 (100%) | 0·070+ |
| ECC Readiness Score median (IQR) | 0·50 (0·27 to 0·80) | 0·33 (0·23 to 0·50) | 0·82 (0·80 to 0·89) | 0·021+ |
| Staff domain | ||||
| Nurse present at all times n (%) | 8 (62%) | 4 (44%) | 4 (100%) | |
| Clinician present at all times n (%) | 12 (92%) | 8 (89%) | 4 (100%) | |
| Increased nursing to patient ratios for critically ill patients*n (%) | 3 (23%) | 1 (11%) | 2 (50%) | |
| Staff domain sub-score median (IQR) | 0·67 (0·33 to 0·67) | 0·67 (0·33 to 0·67) | 0·67 (0·67 to 1·0) | 0·20+ |
| Systems and space domain | ||||
| Available critical care protocol†n (%) | 9 (69%) | 5 (56%) | 4 (100%) | |
| Formal triage system in OPD or ED n (%) | 8 (62%) | 5 (56%) | 3 (75%) | |
| Method of identifying critically ill patients on general ward n (%) | 2 (15%) | 0 | 2 (50%) | |
| Twice daily reassessment of critically ill inpatients n (%) | 6 (46%) | 2 (22%) | 4 (100%) | |
| Ability to perform basic signal functions‡n (%) | 5 (38%) | 1 (11%) | 4 (100%) | |
| Dedicated area for critically ill patients n (%) | 6 (46%) | 2 (22%) | 4 (100%) | |
| Systems and space domain sub-score median (IQR) | 0·33 (0·17 to 0·83) | 0·17 (0·17 to 0·33) | 0·83 (0·83 to 0·92) | 0·021+ |
| Essential equipment, diagnostic tests, and medications domain | ||||
| Xray or ultrasound n (%) | 12 (92%) | 8 (89%) | 4 (100%) | |
| Basic laboratory studies^n (%) | 2 (15%) | 0 | 2 (50%) | |
| Oxygen n (%) | 5 (38%) | 1 (11%) | 4 (100%) | |
| Suction n (%) | 5 (38%) | 1 (11%) | 4 (100%) | |
| Blood pressure cuff n (%) | 13 (100%) | 9 (100%) | 4 (100%) | |
| Red blood cell transfusion n (%) | 7 (54%) | 4 (44%) | 3 (75%) | |
| Intravenous fluids and equipment to administer n (%) | 11 (85%) | 7 (78%) | 4 (100%) | |
| Any intravenous or intramuscular antibiotic n (%) | 9 (69%) | 5 (56%) | 4 (100%) | |
| Continuous pulse oximetry n (%) | 4 (31%) | 1 (11%) | 3 (75%) | |
| Any intravenous vasopressor n (%) | 1 (8%) | 0 | 1 (25%) | |
| Essential equipment, diagnostic tests, and medications domain sub-score median (IQR) | 0·50 (0·30 to 0·80) | 0·40 (0·30 to 0·50) | 0·85 (0·75 to 0·90) | 0·007+ |
Two-sided Fisher's Exact test comparing district and central hospitals.
Not applicable to ICUs or HDUs.
Initial approach to airway, breathing, circulation, and basic neurologic function; medical resuscitation; sepsis; or trauma.
Administration of therapies for reactive airway disease, place peripheral intravenous access, place urinary catheter, administration of adrenaline, diagnose and treat hypoglycemia.
basic chemistry, creatinine/blood urea nitrogen, and hemoglobin.
Figure 1.
Domain composition of ECC readiness score by facility. The bar charts represent ECC Readiness Scores and domain sub-scores by hospital. Each bar represents one of the 13 facilities included in the MECC survey. The larger figure to the right shows each facility’s ECC Readiness Score broken down by colors representing relative contributions from each of the three domain sub-scores. The three smaller figures on the left show each domain sub-score by hospital.
Staff domain
All central hospitals had nurses and clinicians present 24 h a day to respond to the acute needs of critically ill patients. However, district hospital nurses and clinicians were continuously present at only 4 (44%) and 8 (89%) facilities, respectively. Less than a quarter of hospitals adjusted general ward nursing ratios for critically ill patients.
Systems and space domain
District hospitals’ lowest sub-score was in the systems and space domain (0·17, 0·17 to 0·33). All 4 central and 2 (22%) district hospitals had designated space in both inpatient areas and the OPD/ED for critically ill patients. Formal triage systems were used at 3 (75%) central and 5 (56%) district hospitals. Only 2 (15%) hospitals had a system for identifying critically ill patients on the inpatient ward. All 4 central and 2 (22%) district hospitals regularly reassessed critically ill inpatients at least twice daily. Although not included as an ECC Readiness Score tracer item, most hospitals reported taking vital signs more frequently for critically ill patients.
Essential equipment, diagnostic tests, and medications domain
All 4 central and most district hospitals reported adequate availability of imaging (ie, xray or ultrasound), blood pressure cuffs, and adequate ability to administer antibiotics and intravenous fluids. All 4 central hospitals were able to provide oxygen and suction compared to only 1 (11%) district hospital. Laboratory capabilities were limited: only 2 (50%) central and no district hospitals reported adequate availability of the essential laboratory studies included in the ECC Readiness Score (basic chemistry, creatinine/blood urea nitrogen, and hemoglobin).
Barriers to ECC readiness
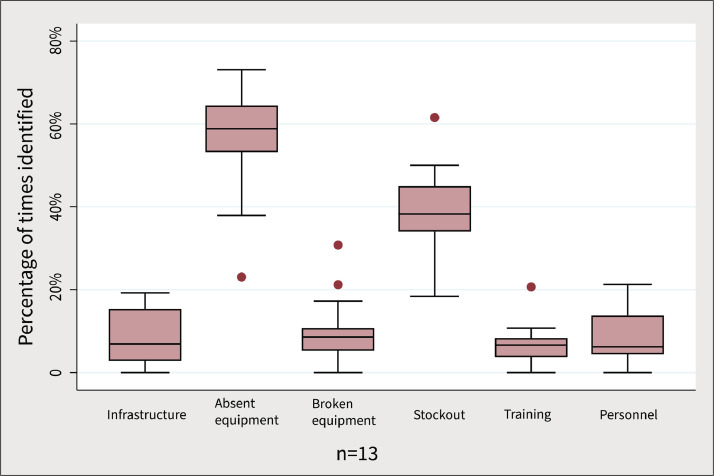
Participants identified absent equipment (56% [95% confidence interval: 48 to 65%]) and stockouts (39% [32 to 46%]) most often as the barriers to performing signal functions included as ECC Readiness Score tracer items (Figure 2). There was no clear evidence of differences in reported barriers between district and central hospitals.
Figure 2.
Facility level barriers. Box plot of facility level barrier data displaying percentage the six most common barrier categories were identified. Horizontal whisker lines represent upper and lower adjacent values. Upper adjacent values, nu, defined as nu< 75th percentile + 1.5*interquartile range and nu+1 >75th percentile + 1.5*interquartile range. Lower adjacent values, nl, defined as nl > 25th percentile - 1.5*interquartile range and nl-1 < 25th percentile - 1.5*interquartile range. The numerator for a given facility level barrier category is calculated as the number of times the barrier category was identified by a participant at the facility for signal functions included in the ECC Readiness Score. The denominator is the number of times participants at the facility were asked to identify barriers for those signal functions (ie, the number of times participants rated any of the signal functions as “generally unavailable” or “somewhat available”). (Signal functions included in the ECC Readiness Score: Administration of therapies for reactive airway disease, place peripheral intravenous access, place urinary catheter, administration of adrenaline, diagnose and treat hypoglycemia, perform portable xray, perform non-portable xray, availability of ultrasound machine, check electrolytes, check blood urea nitrogen/creatinine, check hemoglobin, perform transfusion, administration of intravenous fluids, administration of intravenous or intramuscular antibiotics, and administration of intravenous vasopressors.)
Facility data
Radiologists were available to interpret imaging results at 3 (75%) central and 2 (22%) district hospitals (Table 3). Social work staff were adequately available at half of central and a third of district hospitals. Dedicated spiritual support staff were adequately available at 1 (25%) central and no district hospitals. The most commonly available clinical protocol was for diabetic ketoacidosis, at all 4 central and 7 (78%) district hospitals. At least half of central and a third of district hospitals had clinical protocols for volume resuscitation, asthma, pneumonia, and sepsis. No hospitals had protocols for end-of-life care.
Table 3.
Additional facility data.
| All hospitals (n = 13) | District hospitals (n = 9) | Central hospitals (n = 4) | |
|---|---|---|---|
| Staff | |||
| Social work n (%) | 5 (38%) | 3 (33%) | 2 (50%) |
| Security n (%) | 8 (62%) | 5 (56%) | 3 (75%) |
| Spiritual support n (%) | 1 (8%) | 0 | 1 (25%) |
| Radiology results interpreted by radiologist n (%) | 5 (38%) | 2 (22%) | 3 (75%) |
| Dietician n (%)* | 11 (85%) | 8 (89%) | 3 (75%) |
| Physiotherapist n (%)* | 12 (92%) | 8 (89%) | 4 (100%) |
| Protocols | |||
| Initial approach to airway, breathing, circulation, and basic neurologic function n (%) | 5 (38%) | 4 (44%) | 1 (25%) |
| Medical resuscitation n (%) | 5 (38%) | 3 (33%) | 2 (50%) |
| Volume resuscitation n (%) | 6 (46%) | 3 (33%) | 3 (75%) |
| Adjust fluid resuscitation for malnourished or anemic patients n (%) | 3 (23%) | 2 (22%) | 1 (25%) |
| Asthma treatment n (%) | 6 (46%) | 3 (33%) | 3 (75%) |
| Pneumonia treatment n (%) | 6 (46%) | 3 (33%) | 3 (75%) |
| Sepsis treatment n (%) | 5 (38%) | 3 (33%) | 2 (50%) |
| Diabetic ketoacidosis n (%) | 11 (85%) | 7 (78%) | 4 (100%) |
| End of life care n (%) | 0 | 0 | 0 |
| Equipment | |||
| Electronic cardiac monitoring n (%) | 1 (8%) | 1 (11%) | 0 |
| Crash trolley or code cart n (%) | 2 (15%) | 1 (11%) | 1 (25%) |
| Oxygen sources | |||
| Central piped system n (%) | 1 (8%) | 1 (11%) | 0 |
| Concentrator stored on unit n (%) | 4 (31%) | 0 | 4 (100%) |
| Call for concentrator from central location if needed n (%) | 4 (31%) | 1 (11%) | 3 (75%) |
| Tanks stored on unit n (%) | 3 (23%) | 0 | 3 (75%) |
| Call for tank from central location if needed n (%) | 5 (38%) | 1 (11%) | 4 (100%) |
| None n (%) | 8 (62%) | 8 (89%) | 0 |
Inpatient areas only.
Signal functions
The ability to perform non-portable xray was adequately available at 8 (89%) district and 3 (75%) central hospitals (Table 4). Neither electrocardiograms nor cardiac markers were available at any hospitals. Most facilities were able to perform bag-valve-mask ventilation. All 4 central hospitals were able to administer insulin and treat hypoglycemia as opposed to 5 (56%) and 3 (33%) district hospitals, respectively. Only 1 (11%) district and 3 (75%) central hospitals reported adequate ability to communicate poor prognoses with patients and their families and no hospitals had the ability to de-escalate care (ie, transition to comfort measures).
Table 4.
Signal functions.
| All hospitals (n = 13) | District hospitals (n = 9) | Central hospitals (n = 4) | |
|---|---|---|---|
| Imaging | |||
| Non-portable xray n (%) | 11 (85%) | 8 (89%) | 3 (75%) |
| Perform and interpret point of care ultrasound n (%) | 2 (15%) | 0 | 2 (50%) |
| Respiratory care | |||
| Administration of therapies for reactive airway disease n (%) | 5 (38%) | 1 (11%) | 4 (100%) |
| Arterial blood gas n (%) | 1 (8%) | 0 | 1 (25%) |
| Placement of chest tube n (%) | 4 (31%) | 1 (11%) | 3 (75%) |
| Manual maneuvers to open the airway n (%) | 6 (46%) | 3 (33%) | 3 (75%) |
| Bag-valve-mask ventilation n (%) | 7 (54%) | 3 (33%) | 4 (100%) |
| Endotracheal intubation n (%) | 0 | 0 | 0 |
| Non-invasive ventilation n (%) | 0 | 0 | 0 |
| Invasive mechanical ventilation n (%) | 0 | 0 | 0 |
| Fluid resuscitation | |||
| Administer oral rehydration n (%) | 10 (77%) | 6 (67%) | 4 (100%) |
| Place peripheral intravenous access n (%) | 12 (92%) | 8 (89%) | 4 (100%) |
| Establish central venous access n (%) | 1 (8%) | 0 | 1 (25%) |
| Cardiac | |||
| Cardiac marker (eg, troponin) n (%) | 0 | 0 | 0 |
| Perform and interpret electrocardiogram n (%) | 0 | 0 | 0 |
| Administer aspirin for ischemia n (%) | 9 (69%) | 6 (67%) | 3 (75%) |
| Perform external defibrillation and/or cardioversion n (%) | 2 (15%) | 0 | 2 (50%) |
| Administration of adrenaline n (%) | 11 (85%) | 7 (78%) | 4 (100%) |
| Neurologic | |||
| Protect unconscious patient from secondary injury n (%) | 8 (62%) | 4 (44%) | 4 (100%) |
| Perform mental status exam n (%) | 9 (69%) | 6 (67%) | 3 (75%) |
| Administer benzodiazepine n (%) | 11 (85%) | 7 (78%) | 4 (100%) |
| Perform lumbar puncture n (%) | 9 (69%) | 6 (67%) | 3 (75%) |
| Supportive care | |||
| Place urinary catheter n (%) | 11 (85%) | 7 (78%) | 4 (100%) |
| Diagnose and treat hypoglycemia n (%) | 9 (69%) | 5 (56%) | 4 (100%) |
| Administer insulin for hyperglycemia n (%) | 7 (54%) | 3 (33%) | 4 (100%) |
| Management of extreme temperatures n (%) | 11 (85%) | 7 (78%) | 4 (100%) |
| Communicate with patient and/or families, including sharing poor prognoses n (%) | 4 (31%) | 1 (11%) | 3 (75%) |
| De-escalate care (eg, stop treatments or remove life support) for patients with poor prognoses based on the expressed goals and wishes of the patient or their families n (%) | 0 | 0 | 0 |
Discussion
ECC is an essential component of high-quality health systems but there is limited information about facility-level ECC capacity from LICs. Applying the WHO methodology for assessing service readiness, we have provided a detailed description of ECC capacity and service readiness across district and central hospitals in an LIC health system. We found that critically ill patients were treated across multiple hospital units, even in facilities with designated critical care areas, highlighting that the provision of ECC in Malawi is not limited to ICUs or HDUs, supporting a facility-wide approach to understanding these services. Although most hospitals had ECC Readiness Scores above 0·5, significant gaps exist.
Effective care of critically ill patients requires timely collaboration across multiple areas of the hospital from initial triage, recognition, and stabilization to ongoing monitoring, treatment, and supportive care. An alternative interpretation of ECC Readiness Score is as a measure of the extent of improvement and increased resources required for optimal care of critically ill patients. In other words, what inputs are needed to achieve an ECC Readiness Score of 100%.
Protocols and standardized procedures for ECC were notably absent at many hospitals. Only 62% of facilities used triage systems, which ensure rapid care for the sickest patients.30 On the inpatient wards, less than half of facilities regularly reassessed critically ill patients and only 15% had a method of identifying critically ill patients, a known practical and effective intervention in LIC hospitals.31
Of particular concern is the large disparity in ECC readiness between levels of Malawi's health system—district hospitals had lower and more variable capacity to provide ECC services compared to central hospitals. The population of Malawi, like most LICs, is predominantly rural with only 17% residing in the urban areas10 where central hospitals are located. Due to their geographic distribution throughout the country, most critically ill patients are likely to receive at least initial care at district hospitals32. Although district and central hospitals are designed and resourced to provide different levels of care, the ECC Readiness Score measures only basic essential elements of ECC which should be available at all secondary and tertiary facilities. Accordingly, there is significant overlap between the tracer items included in the ECC Readiness Score and a recent consensus statement on ECC processes that should be available to critically ill patients in all hospitals.4
When probed, participants identified absent equipment as the most common barrier to performing signal functions. This includes situations where a single item of equipment is shared across multiple units, limiting timely access. This suggests that even when an item is technically “present” it may not be immediately available for patients when needed, thus limiting care delivery. This is particularly relevant for critically ill patients who require interventions in a timely manner. Interestingly, a different instrument used in two prior assessments of ECC in Tanzania and Sierra Leone reported availability of equipment and commodities among the highest scoring domains.19,23 This discrepancy between prior work and this study may be attributable to methodology: our survey asked about the ability to deliver an intervention or perform a test when needed, considering equipment as just one component of a clinical process. In other words, our question-response structure accounted for the possibility that the mere presence of equipment within a hospital does not guarantee it is always available when needed. Using signal function questions with follow-up barrier probes and interviewing multiple participants from each unit may have improved the accuracy of our estimates.
To our knowledge, this is one of the first assessments to analyze ECC at a facility level in an LIC setting by considering capabilities across multiple locations in the hospital, including the ED (or OPD if there was no ED) and inpatient units. By including district hospitals, these findings expand existing evidence on ECC in LICs, which is biased towards academic and central referral hospitals in large cities.33
The Malawi Ministry of Health, recognizing the importance of long-term health system investment for ECC services, recently developed a 10-year ECC national strategy. These results provide the first national baseline for ECC capacity at district and central hospitals in Malawi and have the potential to influence policy interventions across the health system. The MECC survey also affords an opportunity to measure impact of these interventions through re-administration. More broadly, the MECC survey provides a standard approach for harmonized multi-country assessments of ECC capacity in LICs, emphasizing inclusion of district hospitals and multiple care areas within hospitals.
These findings highlight the need for interventions to improve ECC service readiness in LICs. Although ECC health systems strengthening requires a combination of equipment, commodities, clinical protocols, dedicated space, and trained staff, these results provide valuable insight into specific areas of immediate need. To ensure universal access to quality ECC, particular attention should be paid to district hospitals, where most patients initially seek care and ECC Readiness is low. These data also suggest a need for designated critical care areas and proven interventions such as early warning systems,31 critical care training,34 and triage.30
Adequate access to proper equipment when caring for critically ill patients is essential. Our findings support a more nuanced approach to understanding equipment availability in the context of ECC. When planning health system strengthening interventions, it should not be assumed that the mere presence of equipment equates with adequate availability and usability. Procurement should be linked to burden of disease by adapting tools such as the Partners In Health UHC Monitoring and Planning Tool35 to include ECC services. Finally, the capacity to care for patients at the end of life, provide spiritual support, and transition to comfort-focused care when appropriate must not be overlooked as an essential aspect of ECC.
Although this study provides important data from an LIC context, there are several limitations. Data were collected from a single country. Although similar gaps are likely present in other LICs, the degree of generalisability is unclear. Although the facility sample was relatively small, it included nearly half of all public sector secondary and tertiary hospitals in Malawi. The sample size was calculated using a formula recommended by the WHO SARA. However, the WHO SARA advises oversampling when variation is likely (ie, smaller strata),17 suggesting that the study was underpowered. Despite this limitation, we believe the MECC survey still provides valuable findings. Furthermore, the random selection of district hospitals helped reduce the likelihood that sampled hospitals differ significantly from non-sampled ones. Administration of the survey in person may have introduced some reporting bias which could result in overestimation of resource availability and capacity. Though our primary outcome, ECC Readiness Score, has not been validated, we followed the approach to quantifying service specific readiness used by the WHO SARA. We also ensured expert consultation and piloted the instrument prior to implementation. Future studies should explore the relationship between ECC Readiness Scores and patient outcomes.
In conclusion, this study provides a detailed description of ECC capacity and service readiness across district and central hospitals in an LIC health system and establishes a framework for measuring ECC in other LICs. Using signal function questions may provide a more accurate method of assessing availability of equipment and commodities. Future efforts to improve ECC services should include district hospitals where the need is greatest. This study underscores the need for long-term investments in health systems strengthening through proven interventions as well as the importance of assessing capacity and service-specific readiness in LIC settings to inform these efforts.
Contributors
PDS, MN, NK, RHM, EBW, KWS, AB, GBK, JSM, and SAR conceptualised the study. PDS, MN, NK, RHM, EBW, KWS, AB, EC, GBK, JSM, and SAR determined the methodological approach. PDS and SAR led funding acquisition and RHM, KWS, and AB contributed. PDS, MN, NK, EBW, EC, TM, and MK implemented the study. TM and MK enrolled patients and acquired data. PDS and SAR accessed and were responsible for raw data associated with the study. PDS and SAR investigated the data and conducted formal data analysis. PDS wrote the original draft with input from MN and SAR. All authors contributed to the editing and writing of the final version of the manuscript. All authors participated In the decision to submit and have read and agreed to the final version of the manuscript.
Funding
Division of Pulmonary and Critical Care Medicine, Brigham and Women's Hospital and Department of Emergency Medicine, Brigham and Women's Hospital.
Data sharing statement
De-identified data collected for this study will be made available via email request to the first author for non-commercial research purposes. Data collection tools will be made available upon email request to the first author with the exception of questions from the WHO HEAT, which can be requested from the WHO Emergency Care team via email (emergencycare@who.int).
Declaration of interests
PDS received support for this study from Brigham and Women's Hospital, Division of Pulmonary and Critical Care Medicine in the form of a faculty research fund and has received consulting fees from the University of California-San Francisco/Sustaining Technical and Analytic Resources as lead technical advisor for ventilator technical assistance in Haiti. SAR received support for this study in the form of a seed grant from Brigham and Women's Hospital Department of Emergency Medicine. JSM is the Chief Medical Officer at Partners In Health and sits on the boards of Village Health Works (Burundi/Muso and Mali), The Institute for Justice and Democracy in Haiti, and Free Speech for People.
Acknowledgements
We thank the Ministry of Health and Population of the Republic of Malawi, including district medical officers and the office of non-communicable diseases for their support and dedication to these important topics. We thank the staff at Abwenzi Pa Za Umoyo/Partners In Health for their support of survey implementation. Most of all, we wish to thank all the study participants.
Footnotes
Supplementary material associated with this article can be found in the online version at doi:10.1016/j.eclinm.2021.101245.
Appendix. Supplementary materials
References
- 1.World Health Organization and the United Nations Children's Fund (UNICEF) World Health Organization; Geneva: 2018. A vision for primary health care in the 21st century: towards universal health coverage and the sustainable development goals.https://www.who.int/docs/default-source/primary-health/vision.pdf (accessed Sept 27, 2021) [Google Scholar]
- 2.Firth P., Ttendo S. Intensive care in low-income countries–a critical need. N Engl J Med. 2012;367:1974–1976. doi: 10.1056/NEJMp1204957. [DOI] [PubMed] [Google Scholar]
- 3.Schell C.O., Gerdin Wärnberg M., Hvarfner A., et al. The global need for essential emergency and critical care. Crit Care. 2018;22:284. doi: 10.1186/s13054-018-2219-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Schell C.O., Khalid K., Wharton-Smith A., et al. Essential emergency and critical care: a consensus among global clinical experts. BMJ Glob Health. 2021;6 doi: 10.1136/bmjgh-2021-006585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Murthy S., Adhikari N.K. Global health care of the critically ill in low-resource settings. Ann Am Thorac Soc. 2013;10:509–513. doi: 10.1513/AnnalsATS.201307-246OT. [DOI] [PubMed] [Google Scholar]
- 6.Reynolds T.A., Sawe H., Rubiano A.M., et al. In: Disease control priorities: improving health and reducing poverty. 3rd edn. Jamison DT, Gelband H, Horton S, et al., editors. The International Bank for Reconstruction and Development /The World Bank; Washington (DC): 2017. Strengthening health systems to provide emergency care. [DOI] [Google Scholar]
- 7.Dondorp A.M., Iyer S.S., Schultz M.J. Critical care in resource-restricted settings. JAMA. 2016;315:753–754. doi: 10.1001/jama.2016.0976. [DOI] [PubMed] [Google Scholar]
- 8.World Health Organization . World Health Organization; Geneva: 2010. Monitoring the building blocks of health systems: a handbook of indicators and their measurement strategies.https://www.who.int/workforcealliance/knowledge/toolkit/26.pdf?ua=1 (accessed Sept 27, 2021) [Google Scholar]
- 9.World Bank Group. World Bank Country and Lending Groups [Internet]. Washington (DC): World Bank Group; 2021. https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups (accessed Sept 27, 2021).
- 10.United Nations. World Urbanization Prospects 2018 [Internet]. New York (NY): United Nations; 2018. https://population.un.org/wup/DataQuery/ (accessed Sept 27, 2021).
- 11.Government of the Republic of Malawi. Health Sector Strategic Plan II (2017-2022). Lilongwe: Government of the Republic of Malawi; 2017. https://extranet.who.int/countryplanningcycles/sites/default/files/planning_cycle_repository/malawi/health_sector_strategic_plan_ii_030417_smt_dps.pdf (accessed March 30, 2020).
- 12.Johansson E.W., Lindsjö C., Weiss D.J., et al. Accessibility of basic paediatric emergency care in Malawi: analysis of a national facility census. BMC Public Health. 2020;20:992. doi: 10.1186/s12889-020-09043-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Robison J.A., Ahmad Z.P., Nosek C.A., et al. Decreased pediatric hospital mortality after an intervention to improve emergency care in Lilongwe, Malawi. Pediatrics. 2012;130 doi: 10.1542/peds.2012-0026. [DOI] [PubMed] [Google Scholar]
- 14.Prin M., Onofrey L., Purcell L., Kadyaudzu C., Prevalence Charles A. Etiology, and outcome of sepsis among critically Ill patients in Malawi. Am J Trop Med Hyg. 2020;103:472–479. doi: 10.4269/ajtmh.19-0605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sonenthal P.D., Masiye J., Kasomekera N., et al. COVID-19 preparedness in Malawi. Lancet Glob Health. 2020;8:e890–e892. doi: 10.1016/S2214-109X(20)30250-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.World Health Organization. Emergency care [Internet]. Geneva: World Health Organization; 2020. https://www.who.int/health-topics/emergency-care#tab=tab_1 (accessed March 30, 2020).
- 17.World Health Organization . World Health Organization; Geneva: 2015. Service availability and readiness assessment (SARA) an annual monitoring system for service delivery: implementation guide, version 2.2. [DOI] [Google Scholar]
- 18.Vukoja M., Riviello E., Gavrilovic S., et al. A survey on critical care resources and practices in low- and middle-income countries. Glob Heart. 2014;9:335–337. doi: 10.1016/j.gheart.2014.08.002. [DOI] [PubMed] [Google Scholar]
- 19.Baker T., Lugazia E., Eriksen J., Mwafongo V., Irestedt L., Konrad D. Emergency and critical care services in Tanzania: a survey of ten hospitals. BMC Health Serv Res. 2013;13:140. doi: 10.1186/1472-6963-13-140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Leligdowicz A., Bhagwanjee S., Diaz J.V., et al. Development of an intensive care unit resource assessment survey for the care of critically ill patients in resource-limited settings. J Crit Care. 2017;38:172–176. doi: 10.1016/j.jcrc.2016.11.002. [DOI] [PubMed] [Google Scholar]
- 21.Marshall J.C., Bosco L., Adhikari N.K., et al. What is an intensive care unit? A report of the task force of the world federation of societies of intensive and critical care medicine. J Crit Care. 2017;37:270–276. doi: 10.1016/j.jcrc.2016.07.015. [DOI] [PubMed] [Google Scholar]
- 22.Burns K.E.A., Duffett M., Kho M.E., et al. A guide for the design and conduct of self-administered surveys of clinicians. CMAJ. 2008;179:245–252. doi: 10.1503/cmaj.080372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Coyle R.M., Harrison H.L. Emergency care capacity in Freetown, sierra Leone: a service evaluation. BMC Emerg Med. 2015;15:1–9. doi: 10.1186/s12873-015-0027-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Harris P.A., Taylor R., Thielke R., Payne J., Gonzalez N., Conde J.G. Research electronic data capture (REDCap)-A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–381. doi: 10.1016/j.jbi.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bintabara D., Ernest A., Mpondo B. Health facility service availability and readiness to provide basic emergency obstetric and newborn care in a low-resource setting: evidence from a Tanzania national survey. BMJ Open. 2019;9 doi: 10.1136/bmjopen-2017-020608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bintabara D., Mpondo B.C.T. Preparedness of lower-level health facilities and the associated factors for the outpatient primary care of hypertension: evidence from Tanzanian national survey. PLoS ONE. 2018;13 doi: 10.1371/journal.pone.0192942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bintabara D., Nakamura K., Seino K. Determinants of facility readiness for integration of family planning with HIV testing and counseling services: evidence from the Tanzania service provision assessment survey, 2014-2015. BMC Health Serv Res. 2017;17:844. doi: 10.1186/s12913-017-2809-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Losonczy L.I., Barnes S.L., Liu S., et al. Critical care capacity in Haiti: a nationwide cross-sectional survey. PLoS ONE. 2019;14 doi: 10.1371/journal.pone.0218141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Baelani I., Jochberger S., Laimer T., et al. Availability of critical care resources to treat patients with severe sepsis or septic shock in Africa: a self-reported, continent-wide survey of anaesthesia providers. Crit Care. 2011;15:R10. doi: 10.1186/cc9410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Marsh R.H., Chalmers K.D., Checkett K.A., et al. Emergency department design in low- and middle-income settings: lessons from a university hospital in Haiti. Ann Glob Health. 2020;86:6. doi: 10.5334/aogh.2568. –6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kruisselbrink R., Kwizera A., Crowther M., et al. Modified early warning score (MEWS) identifies critical illness among ward patients in a resource restricted setting in Kampala, Uganda: a prospective observational study. PLoS ONE. 2016;11 doi: 10.1371/journal.pone.0151408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Marsh R.H., Rouhani S.A. Gaps in physical access to emergency care in sub-Saharan Africa. Lancet Glob Health. 2018;6:e240–e241. doi: 10.1016/S2214-109X(18)30026-3. [DOI] [PubMed] [Google Scholar]
- 33.Murthy S., Leligdowicz A., Adhikari N.K.J. Intensive care unit capacity in low-income countries: a systematic review. PLoS ONE. 2015;10 doi: 10.1371/journal.pone.0116949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Haniffa R., Lubell Y., Cooper B.S., et al. Impact of a structured ICU training programme in resource-limited settings in Asia. PLoS ONE. 2017;12 doi: 10.1371/journal.pone.0173483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mukherjee J.S., Mugunga J.C., Shah A., et al. A practical approach to universal health coverage. Lancet Glob Health. 2019;7:e410–e411. doi: 10.1016/S2214-109X(19)30035-X. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.