Abstract
Diabetic nephropathy (DN) is one of the most devastating complications of diabetes mellitus. Although cadmium (Cd) exposure might be involved in the pathogenesis of DN, the underlying mechanism is still unclear. In this study, we explored the protective effects and possible mechanism of proanthocyanidins (OPC) from grape seed using a mouse model of Cd-induced DN. The successful establishment of this model was verified by analyzing the physiological and biochemical indices of mice, including their body weight and tissue ratio; levels of blood glucose, creatinine, microalbumin, total cholesterol, triglycerides, high-density lipoprotein-cholesterol and low-density lipoprotein-cholesterol; and was based on histopathological examination. Oxidative-antioxidative status, elemental analysis, and key signaling pathway analysis were performed to explore the possible protective mechanism of OPC. The protective effects of OPC and its possible mechanism in preventing the progression of DN were investigated using a multidimensional approach, including its ability in regulating oxidative-antioxidative status (lipid peroxidation, protein carbonyl, superoxide dismutase, and glutathione GST, GSH-Px), metal-binding ability (Cd levels in the kidneys and urine and MT content) and mediation of essential elements (Zn, Ca, Cu, and Fe levels in the kidneys), and activation of the p38 MAPK and Keap1/Nrf2 signaling pathways. OPC exhibited a significant renoprotective effect, attributed to the metal-chelating ability, anti-oxidative effect, and mediation of oxidative stress-related signaling pathway. These results highlight the potential of OPC in preventing or treating DN in humans and suggest the dietary intake of grapes, which are rich in polyphenols, for the prevention of type 2 diabetes mellitus and its complications.
Keywords: cadmium, diabetic nephropathy, proanthocyanidins, Keap1/Nrf2 signaling pathway, p38 MAPK signaling pathway
Introduction
Diabetic nephropathy (DN) is the most devastating complication of diabetes mellitus that causes significant morbidity and mortality (Zoja et al., 2020). It is gradually becoming a leading cause of end-stage renal disease (Duran-Salgado and Rubio-Guerra, 2014; Hansrivijit et al., 2021). Epidemiological studies reveal that more than 25% of patients with diabetes suffer from DN (Reidy et al., 2014), which is characterized by a series of renal abnormalities including mesangial expansion, basement membrane thickening, tubulointerstitial fibrosis, and glomerulosclerosis (Reidy et al., 2014; Li et al., 2021; Medina Rangel et al., 2021). These changes result in a clinical presentation characterized by hypertension, proteinuria, and a progressive reduction in kidney function (Reidy et al., 2014).
Although several studies have shed light on the pathogenesis of DN, several aspects of its condition remain unclear; thus, further comprehensive investigations and evidence-based findings are needed to elucidate the mechanism underlying disease progression (Mantovani and Zusi, 2020; Miao et al., 2021a). Cadmium (Cd), a transition metal that is, toxic by nature and an environmental pollutant, is generated from industrial sources and found in air, water, food, and cigarette smoke. The primary target organs for Cd toxicity are the liver and kidneys. Epidemiological and experimental studies suggest that Cd may act as a risk factor in the pathogenesis of diabetes mellitus and renal disease (Edwards and Ackerman, 2016). A dose-response relationship has been reported between urinary Cd and albuminuria in Torres Strait subjects with type 2 diabetes. It has been suggested that Cd-mediated toxicity may intensify the effects of diabetes on the kidneys. Exposure to Cd could lead to hyperglycemia and accelerate the progression of diabetes mellitus (Tangvarasittichai et al., 2015). Cd accumulation in the kidneys results in generalized dysfunction accompanied by polyuria, glucosuria, and low-molecular-weight proteinuria (Madrigal et al., 2019). Previous studies suggest that Cd can accelerate the occurrence of DN in male Kunming mice treated with a high fat and sugar diet (HFSD) (Gong et al., 2017).
The molecular pathogenesis of DN is poorly understood and no new drugs have been approved for the therapy of DN in almost 20 years. Thus, there is an urgent need for the screening and identification of novel and efficacious drug candidates for the management of DN. Natural products has long been used to be an alternative therapy for the prevention and treatment of various renal diseases including DN (Chen et al., 2018a; Wang et al., 2018a; Chen et al., 2018b; Yang and Wu, 2021). Therefore, Natural products were considered as an important source of new drugs over past 4 decades (Izzo et al., 2020; Newman and Cragg, 2020; Miao et al., 2021b; Zhou et al., 2021). Proanthocyanidins (OPC) are condensed tannins that are derivatives of catechin and gallic acid that are composed of propelargonidins, procyanidins, and prodelphinidins. These compounds are widely distributed in grapes, green and black teas, grape seeds, and wine (Yokozawa et al., 2012). Of late, OPC have been receiving increased attention owing to their potential health benefits, such as their antioxidant properties with strong radical-scavenging, blood glucose lowering, anti-apoptotic, and anti-inflammatory properties among other biological activities (Yokozawa et al., 2012; Ruan et al., 2020). As OPC are known to reduce blood glucose levels, balance oxidative stress, and ameliorate inflammatory responses, we wanted to further explore if they could play a role in protecting against Cd-induced DN. To our knowledge, there lack studies that have elucidated the protective effects on Cd-induced DN in mice. Therefore, we hypothesized that if OPC could effectively protect against Cd-induced DN, a diet comprising this natural compound may serve as potential therapeutic agent and prevent Cd-induced renal damage.
One of the challenges in drug discovery, especially in the case of diabetes, is the lack of robust animal models that can mimic key features of human DN. In this study, we established a mouse model of DN using Cd exposure and HFSD treatment. The successful establishment of this model was verified based on physiological and histopathological investigations and determining the biochemical indices of mice. We investigated the protective effect and potential mechanism of OPC against Cd-induced DN. This study may provide useful clues for better understanding the etiology of DN and highlight novel insights regarding the use of OPC in a clinical setting.
Methods
Materials and Reagents
OPC (purity > 99.50%) were purchased from Yifang S&T Co. Ltd. (Tianjin, China). Cadmium chloride and β-actin (sc-4778, Santa Cruz, CA) were purchased from Sigma-Aldrich Chemical Company (St. Louis, MO, United States) and stored at 4°C away from sunlight. Anti-MAPK (sc-393609, Santa Cruz, CA), anti-p38 (sc-81621, Santa Cruz, CA), anti-nuclear factor-erythroid-2-related factor 2 (Nrf2, sc-365949, Santa Cruz, CA), anti-Keap1 (sc-515432, Santa Cruz, CA), and horseradish peroxidase (HRP)-conjugated secondary antibodies (anti-rabbit) were obtained from Santa Cruz Biotechnology (CA, United States).
Animals Handing and Sample Preparation
Five-week-old male Kunming mice (weighing 20 ± 2 g) were supplied by Central Animal House, Xi’an JiaoTong University Health Science Center (Xi’an, China). The animals were housed in a specific pathogen free environment at a temperature of 22 ± 1°C and relative humidity of 50 ± 1%, and subjected to a 12/12 h light/dark cycle. All animals were provided free access to food and water and were allowed to acclimatize for 7 days prior to dosing. All animal studies complied with the regulations and guidelines of the Shaanxi University of Science and Technology Institutional Animal Care and were in accordance with the IACUC and AAALAC guidelines.
For experimental studies, the mice were randomly divided into the following four groups with 10 mice per group: control, DN, OPC, and HFSD. The mice in the control and HFSD groups were fed a standard pellet diet or HFSD and intraperitoneally (i.p.) administered physiological saline (0.9%) during the entire procedure. For the initial 12 weeks, mice in the DN and OPC groups were fed HFSD and received a daily injection of 1 mg/kg CdCl2 i.p. After treatment for 12 weeks, mice in the OPC group were administered OPC i.p. at a dose of 5 mg/kg body weight for consecutive 4 weeks. Mice in the DN group received 0.9% saline i.p. A detailed experimental protocol is listed in Table 1 and was used in our study (Gong et al., 2017).
TABLE 1.
Detail protocols for model construction.
| Group | Diet | Treatment | |
|---|---|---|---|
| 12 weeks | 4 weeks | ||
| Control Group (Con) | standard pellet diet | 0.9% saline i.p. | 0.9% saline i.p. |
| DN group (DN) | High sugar and Fat diet | 1 mg/kg CdCl2 i.p. | 0.9% saline i.p. |
| OPC treatment Group (DN + OPC) | High sugar and Fat diet | 1 mg/kg CdCl2 i.p. | 5 mg/kg bw OPC |
| High Fat and sugar Group (HFSD) | High sugar and Fat diet | 0.9% saline i.p. | 0.9% saline i.p. |
During the entire experimental period, the body weight and fasting blood glucose (FBG) levels of mice were recorded at regular intervals. Using metabolism cages, 24 h urine samples were collected for analysis. Mice were anesthetized by diethyl ether inhalation and sacrificed using cervical dislocation (Pavel et al., 2020). Blood samples were collected from the eyeballs of mice, centrifuged at 4°C to obtain serum, and frozen at −80°C. Kidneys were dissected and weighed immediately, washed clean, and either stored at −80°C or fixed in 10% formalin and stored until further use.
Renal tissues were accurately weighed, cut into pieces, and homogenized in a pre-cooled Teflon homogenizer with an appropriate amount of ice-cold Tris–HCl buffer (100 mM; pH 7.4) [sample weight (g):buffer volume (ml) = 1:9] to obtain the tissue homogenate. The supernatant was centrifuged at 12,000 rpm for 20 min at 4°C to obtain samples for subsequent assays.
Assessment of Blood and Urine Parameters
Fasting blood glucose (FBG) levels were determined using a glucometer and mice with FBG levels of more than 11.1 mmol/L were considered diabetic. Creatinine and microalbumin (mALB) levels in the urine were analyzed using the corresponding assay kits. Total cholesterol (TC), triglyceride (TG), high-density lipoprotein-cholesterol (HDL-C), and low-density lipoprotein-cholesterol (LDL-C) levels in the blood were determined based on enzymatic assays using commercial kits (Jiangchen Co., Nanjing, China) and following the manufacturer’s instructions.
Renal Histopathology
For renal histopathological studies, the kidney sections from each mouse were fixed in 10% formalin immediately after dissection and washed using a gradient concentration of ethanol for dehydration, as previously described (Miao et al., 2020). The slides were transparentized with xylene, dipped in wax, and embedded in paraffin. The blocks were sectioned and stained with hematoxylin-eosin (H&E) to observe the normal cellular structure. Periodic acid-Schiff (PAS) staining was used to determine the glycogen content and Masson’s trichrome staining was used to analyze collagen deposition. The stained sections were covered with a glass slide, dried, and observed using an optical microscope (Leica DM750).
Assessment of Antioxidative Status
Proteins in the kidney tissue homogenates were determined using a bicinchoninic acid (BCA) assay. The oxidative-antioxidative status in mouse kidneys was determined based on the malondialdehyde (MDA), protein carbonyl (PCO), superoxide dismutase (SOD), glutathione (GSH), glutathione S transferase (GST), and glutathione peroxidase (GSH-Px) levels using commercially procured kits. Renal nitric oxide (NO) levels were determined using a colorimetric method based on the Griess reaction.
Determination of Essential Elements and Metallothionein in Kidney
To quantify the content of essential elements, renal samples were accurately weighed and dry-ashed using a muffle furnace. The ash was solubilized in 3M HCl and appropriately diluted. The serum and urine samples were also pre-deposited, respectively. Tissue, serum, and urine samples were tested for Zn (213.9 nm), Fe (248.5 nm), Cu (324.8 nm), Ca (422.7 nm), and Cd (228.8 nm) using atomic absorption spectrometry (Hitachi Z-2000). The element levels are expressed as micrograms of the element per gram of wet tissue weight (μg/g w.t.w.) or micrograms of the element per milliliter sample (μg/ml) (Gong et al., 2008). MT content in mouse kidney was determined following the methods published by Gong et al. (2012).
Western Blotting Analysis
Western blotting analysis was performed following previously published studies (Chen et al., 2019a). RIPA buffer was used for the extraction of total protein. Renal tissues were first weighed, mixed with RIPA buffer supplemented with protease and phosphatase inhibitors (Roche Applied HEART), and homogenized at 4°C. The supernatant was centrifuged at 4°C and 13,000 × g for 30 min and transferred to a new enzyme-free tube. Proteins were quantified using a BCA kit.
Western blotting was performed using anti-MAPK (Santa Cruz, CA), anti-p38 (Santa Cruz, CA), anti-Nrf2 (Santa Cruz, CA), anti-Keap1 (Santa Cruz, CA), anti-actin (Santa Cruz, CA), and HRP-conjugated secondary antibodies (anti-rabbit) from Santa Cruz. Proteins were separated using electrophoresis on an SDS-polyacrylamide gel and the bands were electrophoretically transferred onto a polyvinylidene difluoride membrane. The membranes were blocked for 1 h at 37°C and incubated with primary antibodies overnight at 4°C followed by hybridization with HRP-conjugated secondary antibodies for 1 h. Proteins were detected using the enhanced chemiluminescence (ECL) system and ECL Hyperfilm (Amersham Pharmacia Biotech, UK Ltd., Little Chalfont, Buckinghamshire, UK). The intensities of relative bands were quantified using densitometry.
Statistical Analysis
All values are expressed as mean ± standard deviation. All statistical tests were performed using SPSS version 20.0. Significant differences among the different groups were evaluated using one-way analysis of variance (ANOVA) followed by Dunnett’s post hoc test. p < 0.05 was regarded significant differences.
Results
Effects of OPC on Metabolic Dysfunction in Cd-Induced DN Mice
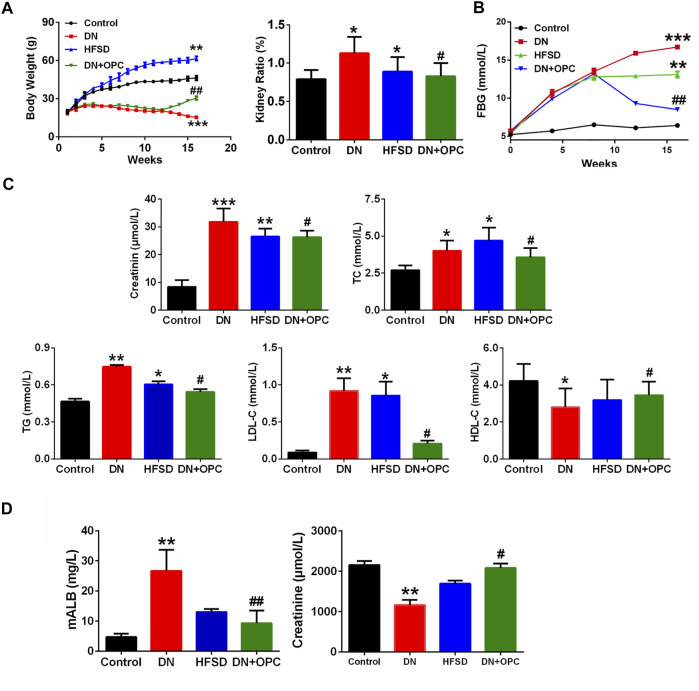
The body weight gain of DN mice was significantly decreased compared with the control mice. We found that the administration of OPC for 12–16 weeks could recover this decline (p < 0.05). When mice were treated only with HFSD, significant increases in body weight were observed (Figure 1A). Compared with the control group, the tissue ratio was significantly increased by 43.1% in the DN group and treatment with OPC could decrease this ratio. Moreover, the tissue ratio in the HFSD group also showed a significant increase of 12.7% compared with the control mice (Figure 1A).
FIGURE 1.
Physiological and biochemical indexes (A) The body weight and kidney ratio in the different groups at week 16. kidney ratio equal to kidney weight/body weight. (B) FBG in the different groups. (C) Serum parameters in the different groups. Including serum creatine, TC contents, TG contents, LDL-C contents, and HDL-C contents in the different groups. (D) mALB contents and Urine creatinine in the different groups. *p < 0.05, **p < 0.01, ***p < 0.001 compared with control group; # p < 0.05, ## p < 0.01, compared with DN group.
Effects of OPC on Glucose and Lipid Metabolism in Cd-Induced DN Mice
FBG levels were also measured during the entire study. As shown in Figure 1B, in the initial 12 weeks, when treated with HFSD and/or Cd, the FBG levels of mice in the DN, OPC, and HFSD groups were all above 11.1 mmol/L, indicating that the animals were diabetic. The administration with OPC caused a significant decrease in FBG (p < 0.01), suggesting its possible role in lowering FBG. To determine the protective effects of OPC in mice with Cd-induced DN, the serum biochemical parameters, such as plasma TG, TC, HDL-C, and LDL-C, related to in vivo glucose and lipid metabolism were evaluated, and the results are presented in Figure 1C. The levels of TG, TC, and LDL-C of mice in the DN group were markedly increased, whereas HDL-C levels were significantly decreased compared with the control group. On the other hand, TG, TC, and LDL-C levels were found to be significantly decreased and HDL-C level was significantly increased in mice treated with OPC compared with those in the DN group. Additionally, the changes in TC, TG, HDL-C, and LDL-C levels of mice in the HFSD group were moderate, probably because Cd exposure accelerated the progression of DN.
Effects of OPC on the Renal Function in Cd-Induced DN Mice
Next, we determined renal function and evaluated the protective effects of OPC against Cd- and HFSD-induced DN. The combined effect of Cd and HFSD led to remarkable renal dysfunction as evidenced by serum/urine creatinine and urine mALB levels (Figures 1C,D). Plasma and urine creatinine levels were determined to assess the degree of renal injury. As shown in Figure 1C, plasma creatinine levels were remarkably elevated in mice in the DN group compared with the control mice. Creatinine levels of mice in the OPC group were 17.5% lower than those in the DN group. Of note, the change in urine creatinine levels showed an opposite trend; creatinine levels of mice in the DN group were remarkably decreased to 54% vs. those in the control group. However, OPC treatment effectively ameliorated this change and restored to near normal levels.
We also found that compared with the control group, urine mALB levels of mice in the DN group were significantly increased. OPC treatment greatly reduced mALB levels to 65.1% compared with those observed in the DN group (Figure 1D). These results indicated the protective effects of OPC against the development of DN.
Effects of OPC on the Renal Histology in Cd-Induced DN Mice
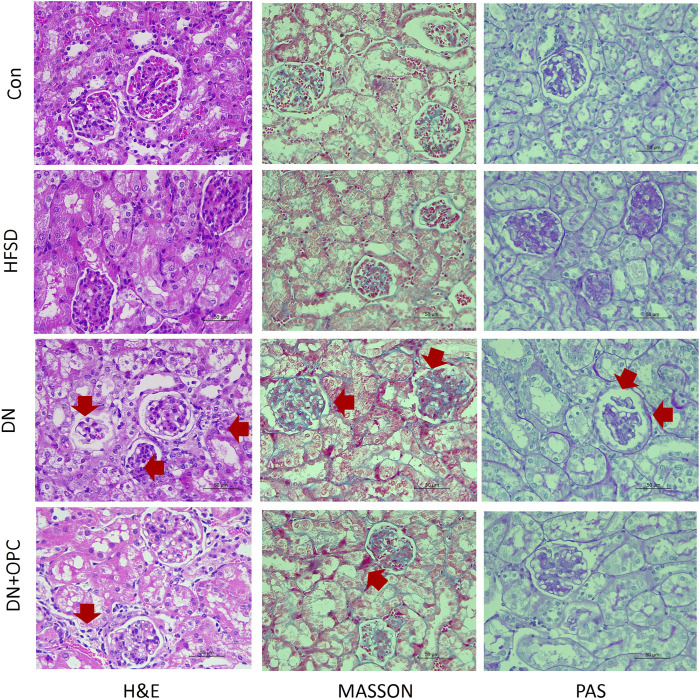
Kidney histology was studied using H&E staining, which revealed significant changes as glomerular thickening in the tissue samples of mice from the DN group (Figure 2). Interstitial and glomerular changes were noticeably ameliorated after OPC treatment (H&E staining, Figure 2). A diffuse expansion of the mesangial matrix and prominent thickening of the glomerular basement membrane to a mild extent was observed in samples from the DN group after PAS staining, which was greatly reduced after OPC treatment.
FIGURE 2.
Pathologic analysis of the kidneys. H&E staining in the different groups, including control, HFSD, DN, and DN + OPC groups, (*) represent glomerulus atrophy, (→) represent renal tubular necrosis. PAS staining in the different groups, including control, HFSD, DN, and DN + OPC groups, (→) represent mesangial expansion. MASSON staining in the different groups, including control, HFSD, DN, and DN + OPC groups. (→) represent inflammatory infiltrates, areas of tubulointerstitial fibrosis. Magnification times × 400 and scale bar in 50 μm.
Moreover, renal histology was studied using Masson’s staining (Figure 2), which demonstrated visible changes in the tissues from the DN groups, as evidenced by mild tubulointerstitial fibrosis, accumulation of extracellular matrix (ECM), tubular dilatation, and atrophy. OPC treatment was found to partly reduce the tubulointerstitial fibrosis index in the renal cortex and medulla. Collectively, these results indicated the protective role of OPC in Cd-induced DN by the amelioration of morphological changes.
Effects of OPC on Renal Oxidative Stress Parameters and NO Levels in Cd-Induced DN Mice
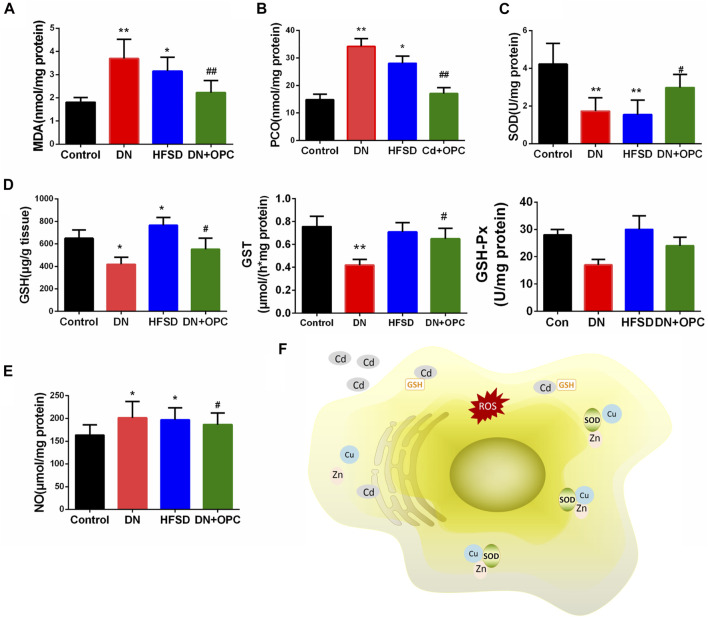
As the pathology of DN is related to the overproduction of ROS and an imbalance in oxidative stress, we determined the oxidative stress parameters. As shown in Figures 3A,B, the MDA and PCO levels showed a 2.1-fold and 2.3-fold increase in the DN group, respectively, after exposure to Cd and HFSD. OPC treatment could attenuate oxidative stress by significantly decreasing MDA and PCO levels. As expected, the synergistic effect of Cd and HFSD resulted in a 59.1% decrease in SOD activities in mice with DN (p < 0.01), whereas OPC treatment could effectively reverse this decrease (Figure 3C).
FIGURE 3.
Anti-oxidative parameters of the kidneys. (A) MDA levels. (B) PCO levels. (C) SOD activities. (D) GSH, GST, and GSH-Px content. (E) NO levels. (F) Possible mechanism of cadimium on oxidative stress. *p < 0.05, **p < 0.01, ***p < 0.001 compared with control group; and # p < 0.05, ## p < 0.01, compared with DN group.
We also found that compared to that in the control animals, the combined effect of Cd and HFSD could reduce GSH, GST and GSH-Px levels by 35.5, 44.4, and 39.2% in mice in the DN group, respectively, whereas OPC treatment significantly increased GSH, GST and GSH-Px levels (Figure 3D). Moreover, NO levels in the tissues of mice from the DN group were significantly increased co-treatment with Cd and HFSD (Figure 3E). However, NO levels were found to be reduced in tissues in the OPC group. Treatment with only HFSD led to significant changes in the oxidative and antioxidant parameters except for the NO levels.
Effects of OPC on p38 MAPK and Keap1/Nrf2 Pathway-Mediated Oxidative Stress
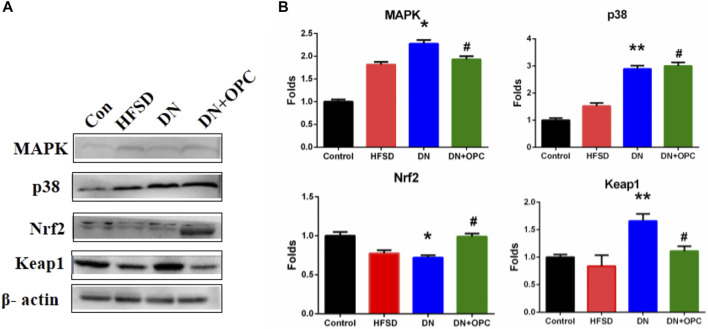
Western blotting was used to further confirm the role of oxidative stress in the progression of DN and to determine the protective mechanism of OPC in oxidative stress-related protein expression. Accordingly, we examined the protein expression of the p38 MAPK and Keap1/Nrf2 pathways. Our findings indicated that p38 and MAPK expression significantly increased in DN kidney samples; however, this increase was prevented after treatment with OPC (Figure 4).
FIGURE 4.
Western blot analysis. (A) Protein Bands. (B) Quantitative analysis. Data were repeated three times. *p < 0.05, **p < 0.01, compared with control group and # p < 0.05 compared with DN group.
There was a significant decrease in Nrf2 expression and an increased expression of Keap1 in the samples obtained from the DN group. However, treatment with OPC significantly increased Nrf2 and decreased Keap1 expression in diabetic conditions (Figure 4). These results suggested that OPC may exert protective effects in DN, which might be associated with p38 MAPK and Keap1/Nrf2 signaling pathways.
Effects of OPC on Elemental Levels in Cd-Induced DN Mice Kidneys
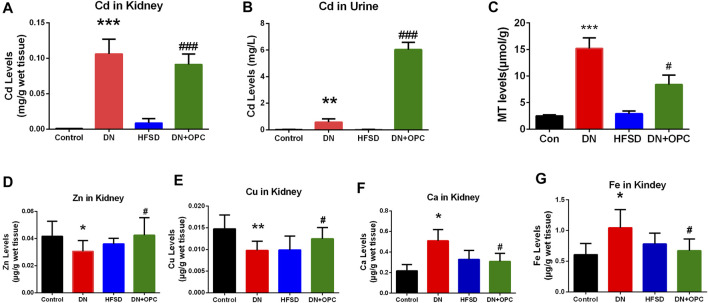
An imbalance in elemental levels is common in patients with DN. Thus, we investigated the changes in levels of the essential elements in DN and the effect of OPC on elemental levels. It can be seen in Figure 5 that Cd was accumulated in the kidneys of mice in the DN and OPC-treated groups. A significant elevation of Cd levels was observed in samples obtained from the DN group (111.1-fold higher than that in the Control group; p < 0.001, compared to the Control group). This accumulation was reduced after OPC treatment and Cd levels in the kidneys were found to be reduced to 85.9% compared with those in the DN group. Cd was excreted in the urine in a small amount. OPC treatment led to a significant increase in Cd excretion (10.4 folds higher than that in the DN group; p < 0.01, compared with the DN group). There was no significant increase in Cd levels in the kidneys or urine of samples obtained from the HFSD group.
FIGURE 5.
Elemental analysis and MT content determination. (A) Cadmium content in kidney. (B) Cadmium content in urine. (C) MT content. (D) Zinc content in kidney. (E) Copper content in kidney. (F) Calcium content in kidney. (G) Iron content in kidney. ***p < 0.001, **p < 0.01, *p < 0.05 compared with control group and # p < 0.05, ## p < 0.01, ### p < 0.001, compared with DN group.
As seen in Figure 5, levels of the essential elements were significantly different among groups. Zn levels in the kidney tissues of mice from the DN group were significantly decreased to 73.3%, and OPC could restore these levels.
Cu levels showed a 1.34-fold increase in the tissue samples obtained from mice in the DN group compared with those from the control group. OPC could ameliorate this increase and return to normalcy. Ca and Fe levels in the tissue samples in the DN group showed a 2.35-fold and 1.72-fold increase, respectively, whereas OPC could effectively decrease this tendency and reduce Ca and Fe levels to 60.5 and 64.1%, respectively. Changes in the elemental levels in the HFSD group were not significant.
To further elucidate the mechanism of OPC chelating with cadmium, the content of MT in kidney were assessed. As shown in Figure 5, an increase of MT content in DN group was observed (p < 0.001), whereas the treatment of OPC could lower the content of MT, suggesting the binding ability of OPC with cadmium.
Discussion
The progression of DN, one of the biggest problems in nephrology field, pointing to that patients progress end-stage renal disease and require dialysis (Aydin et al., 2019; Bacharaki et al., 2021) or kidney transplantation (Webster et al., 2017; Jain et al., 2019; Van Sandwijk et al., 2019). Recent epidemiological studies reveal a link between Cd exposure and the incidence of diabetes and its complications (Chen et al., 2006; Afridi et al., 2008; Akinloye et al., 2010; Satarug et al., 2010; Wallin et al., 2014). Several hypotheses have been proposed to explain the interplay between Cd exposure and incidence of diabetes and DN (Edwards and Prozialeck, 2009; Park et al., 2019). Gong et al. suggest that Cd might be a risk factor in DN pathology (Gong et al., 2017); however, the underlying mechanism of the role of Cd in the development and progression of DN is yet to be completely elucidated. In this study, we investigated the possible etiology of Cd-induced DN and studied OPC as novel and efficacious drug candidates in DN therapy.
A decrease in body weight in patients with diabetes may be due to the compensatory gluconeogenesis in cells to account for glucose deficit. Thus, weight reduction may be considered a marker of diabetes mellitus and its complications. The body weight reduction in DN mice is probably due to abnormal carbohydrate and lipid metabolism and increased protein catabolism (Ghosh et al., 1994). With the onset of DN, the kidney size and weight increased by an average of 15% and was accompanied by a progressive reduction in renal function. The increase in kidney weight was likely due to the development of renal hypertrophy. OPC are known to effectively alleviate kidney enlargement (Mansouri et al., 2011). Elevated FBG levels in mice with DN suggested the diabetogenic effect of Cd also revealed that the administration of OPC could effectively reduce the spike in blood glucose.
We also observed an increase of mALB levels in the DN group, which probably resulted from the damage to the renal glomeruli filtration barrier; however, the precise mechanism of this phenomenon is still unknown. A plausible explanation is that the severity of pro-oxidative stress in diabetes results in disturbed metabolism in the basement membrane (Ha and Kim, 1995). mALB is the predominant renal risk marker in patients with DN. The high mALB levels exacerbate renal damage. A decrease in mALB results in a proportional increase in renal function. Moreover, the creatinine in urine decreased as the concentration of creatinine in the blood was acutely increased 6–8 folds by creatinine infusion. This decrease was thought to be due to the saturation of the tubular secretory mechanism (Perrone et al., 1992). The renal glomeruli from DN mice showed a characteristic morphology that correlated with mesangial cell proliferation and excessive accumulation of ECM (Ahn et al., 2004). We found that OPC exerted a protective effect, prevented the increase in mALB and creatinine levels, and retained renal morphology. Our findings were in agreement with the results of the study by Stefanovic et al. wherein grape seeds, which are rich in antioxidative bioflavonoids, were found to bring about a significant morphologic improvement and amelioration in kidney function (Stefanovic et al., 2000).
The unusually high levels of serum lipids are probably due to the increased mobilization of free fatty acids from the peripheral fat deposits, as insulin suppresses the production of hormone-sensitive lipase (Sugano et al., 2006; Vaziri, 2006). Therefore, elevated TG, TC, and LDL-C levels that were found in the DN group may also constitute risk markers during the development of DN. We found that OPC could abate the susceptibility of lipids to oxidation and stabilize the membrane lipids, thereby relieving oxidative stress by exerting hypocholesterolemic, hypotriglyceridemic, and hypophospholipidemic effects.
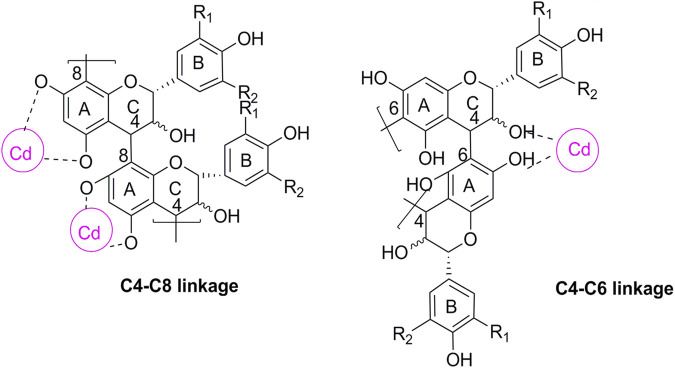
The significant increase in Cd levels in the tissue samples from the DN group suggested Cd accumulation, especially in the kidneys. The toxicity of Cd may be partly attributed to its long half-life and low excretion rate. The urine Cd levels were found to be remarkably elevated after OPC treatment, possibly suggesting the metal-chelating ability of OPC in reducing Cd levels in vivo. Grape seeds are a rich source of plant flavonoids and OPC oligomers. Proanthocyanidins from grape seed are polyphenolic compounds that are mainly concentrated in tree barks and outer skin of the seeds. OPC is of a diphenylpropane structure of C6 -C3 -C6. Most often found as a glycoside derivative, this compound class is composed of three monomer units of catechin, epicatechin, and epigallocatechin (Harborne and Mabry, 2013). OPC bind metals through complexation via their o-diphenol groups. Fine et al. found that OPC can chelate free iron molecules, and inhibit iron-induced lipid peroxidation (Fine, 2000). As seen in Figure 6, OPC can bind with Cd ions to form a covalent structure that can be excreted via urine.
FIGURE 6.
Chemical structure of proanthocyanidin-cadmium polymers. R1, R2 = H, propelargonidins; R1 = OH, R2 = H, procyanidins; R1, R2 = OH, prodelphinidins. The cycle in purple represent the possible binding site for Cd.
Enteric neuropathy and microvascular disease affect the in vivo absorption of essential minerals in diabetic individuals (Eliasson et al., 1995). Cd competes with essential elements such as Ca, Zn, Fe, and Cu, thereby resulting in an imbalance. Low Zn levels may interfere with the ability of the pancreatic islet cells to produce insulin, especially in individuals with type 2 diabetes. Moreover, a decrease in Zn is linked to the overproduction of free radicals and increased lipid oxidation in patients with diabetes (DiSilvestro, 2000). Moreover, Zn deficiency may exacerbate the toxicity of other metals such as Fe and Cu. During hyperglycemia and inflammation, excess Fe results in the overproduction of ROS and may affect glucose control, leading to the development and progression of DN (Al-Hafidh Khattab and Al-Youzbaki, 2018). Intracellular calcium levels are increased in patients with diabetes. Ca2+ release in the cytosol accelerates Cd-induced mitochondrial injury, thus mediating cytochrome-c release and caspase-9 activation. Increased Cu levels have been correlated with the oxidation of LDL-C and alterations in arterial wall structure, eventually leading to infection, stress, and diabetes mellitus (Beshgetoor and Hambidge, 1998; Tan et al., 1999). Since MT is belived to play a privotal role in the detoxication of heavy metals, such as cadmium, and scavenging of free radicals, the metal ability of OPC were assessed by measuring the content of MT. It is obvious that OPC treatment could lower the content of MT, suggesting the protective of OPC against Cd-induced nephrotoxicity probably due to its ability to chelating metal ions (Gong et al., 2012).
When chronically exposuring to Cd, approximately 50% of the absorbed Cd is accumulated in the kidneys and causes damage to renal microtubules (Satarug et al., 2017). However, the mechanism for cadmium nephrotoxicity remains uncertain. Using a variety of cell culture systems or animal models, ROS is thought to be associated with cadmium toxicity. Cadmium depletes glutathione and protein-bound sulfhydryl groups, leading to the production of reactive oxygen species, such as superoxide anions, hydrogen peroxide, and hydroxyl radicals. The depletion in renal GSH has been observed in response to oxidative stress by cadmium treatment, resulting in enhanced lipid peroxidation, and excessive lipid peroxidation caused increased GSH consumption. As GSH-dependent antioxidant enzymes, the decrease abilities of GST and GSH-Px probably due to reduced content of its substrate GSH. The detoxification effect of GST play a critical role for endogenous compounds such as peroxidised lipids, as well as the metabolism of xenobiotics. GSH-Px could eliminate hydrogen peroxide and lipid peroxides and thus interrupt the propagation of the lipid peroxidation reaction (Valavanidis et al., 2006). Moreover, the exposure to cadmium lead to the loss of Cu and Zn in the kidneys. Cu and Zn are antioxidant trace elements because they act as the cofactors of cytoplasmic superoxide dismutase (Pokusa and Kráľová Trančíková, 2017). The decreased SOD activity might be related to the loss of copper and zinc which are essential for the enzyme activity, resulting in the insufficient abilities to scavenge the superoxide anion produced during the normal metabolic process. Hamasaki et al. found that an imbalance between Zn and Cu is associated with renal dysfunction in humans, which is mediated by oxidative stress (Hamasaki et al., 2016). The increased production of different radical species accelerates the degradation of carbohydrates, lipids, and DNA, and further result in hyperglycemia and glucose auto-oxidation (Gong et al., 2017). As a result, oxidative stress induced by Cd may be an intervening factor in the toxic mechanism of Cd on renal tubules (Figure 3F).
Hyperglycemia-induced oxidative stress might also act as a common but key event in the development and progression of diabetes and its complications (Ceriello, 2000). Decreased SOD activity has been reported in diabetes (Mohora et al., 2006). Depletion of reduced GSH could significantly affect the overall redox potential of the cell. NO reacts with superoxide radicals and is converted to the harmful peroxynitrite, leading to increased levels of steady-state free radicals in the kidneys of diabetic individuals (Huie and Padmaja, 1993). Stadler et al. have demonstrated that NO production is increased in the kidneys of streptozocin-induced diabetic rats, which provides credible evidence for the involvement of ROS, NO, and peroxynitrite-derived species in the development and progression of early diabetic tissue damage (Stadler et al., 2003). OPC as free radical scavengers can increase free GSH levels to further detoxify the products of lipid peroxidation (Bagchi et al., 2000), and then restoring the renal antioxidant defense system. OPC also promote insulin release by stimulating the surviving pancreatic cells and regulating insulin release, thereby facilitating serum glucose levels to normalcy. In the present study, combined treatment of cadmium and HFSD resulted in significant increase in TBARS and PCO as well as NO, significant decrease in antioxidant content in renal tissue, however, treatment with OPC could ameliorate these changes, indicating the protection offered by OPC against renal injury.
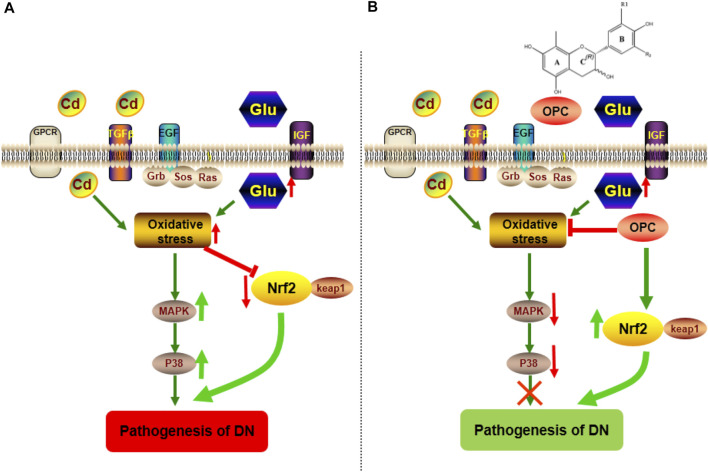
MAPK functions as a key point in several biochemical processes and participates in cellular processes such as proliferation, differentiation, and apoptosis. p38 MAPK is an important member of MAPK that plays a pathological role in diabetes and result in podocyte apoptosis (Susztak et al., 2006; Chuang et al., 2007). Hyperglycemia is known to activate p38 MAPK (Igarashi et al., 1999; Adhikary et al., 2004), which is associated with the apoptosis of pancreatic β-cells. The p38 pathway is also activated in the renal cortex following podocyte decrease, albuminuria, and glomerulosclerosis during early DN (Wang et al., 2018b). Nrf2 signaling pathway played a critical role in the pathogenesis of renal disease including DN (Chen et al., 2016; Chen et al., 2017; Chen et al., 2019a; Feng et al., 2019a; Wu et al., 2021). A decline in Nrf2 activity leads to the decreased transcription of several antioxidant enzymes and an increased accumulation of ROS. Nrf2 activation not only mediates glucose metabolism and glycogen formation but also plays a role in alleviating oxidative stress by neutralizing ROS and decreasing oxidative damage to the kidneys. Moreover, Nrf2 can negatively regulate TGF-β1 activity (Zheng et al., 2011). Increased evidence has indicated that a number of natural products improve renal injury by activating the impaired Keap1/Nrf2 signaling pathway (Chen et al., 2019b; Feng et al., 2019b; Chen et al., 2019c; Wang et al., 2020). OPC can activate Nrf2 expression associated with the MAPK pathway (Bak et al., 2012). We found that OPC could protect against Cd-induced toxicity by counteracting oxidative injury via regulation of the p38 MAPK pathway coupled with Keap1/Nrf2 pathway. The functional group at R2 moiety on OPC appeared important to the inhibitory action of MAPK-related signaling pathway, because prodelphinidin B2 3,3 di-O-gallate, having two galloyl moieties, showed strongest effect while prodelphinidin B2, having no galloyl moiety, failed to show such inhibitory effect (Hou et al., 2007). Our results indicated that targeting of the p38 MAPK and Keap1/Nrf2 signaling pathways might be involved in the pathogenesis of DN. Moreover, OPC triggered a complex crosstalk between p38 MAPK and Keap1/Nrf2 signaling pathway via modulating oxidative stress (Figure 7).
FIGURE 7.
Possible mechanism involved in the pathogenesis of DN and protective effect of OPC. (A) The combination of Cd and HFSD can induce the increase of oxidative stress, which activate the expression of p38 MAPK pathway, moreover, block the effect of Nrf2/Keap1 pathway. (B) The treatment of OPC can downregulate the expression of p38 MAPK pathway and activate Nrf2/Keap1pathway to exert its protective effect against the progressive of DN.
Conclusion
To summarize, we found that OPC from grape seeds exhibit morphological and functional protection in Cd-induced DN and its progression. The protective effects of OPC may be attributed to multidimensional aspects, including the regulation of oxidative-antioxidative status (LPO, PCO, SOD, and GSH), metal-binding ability (Cd in kidney and urine), mediation of the levels of essential elements (Zn, Ca, Cu, and Fe levels in the kidneys), and activation of the p38 MAPK and Keap1/Nrf2 signaling pathways. Based on our findings, it can be reasonably concluded that OPC from grapes can serve as functional foods in preventing the onset of type 2 diabetes mellitus and its complications. However, further studies on the mechanism of action and safe dose of grape OPC are required prior to its use in a clinical setting.
Acknowledgments
The authors would like to thank Shaanxi University of Science and Technology for providing lab and equipment to conduct this study.
Data Availability Statement
The raw data supporting the conclusion of this article will be made available by the authors, without undue reservation.
Ethics Statement
The animal study was reviewed and approved by the Shaanxi University of Science and Technology Institutional Animal Care.
Author Contributions
PW, SiP, and ShP performed the treatment and assays with DN animals, carried out the statistical analysis, and assisted in writing the manuscript. XC and WY participated in the detection of metal levels and the protein expression analysis. LW and FC contributed to the research design and data analysis, and assisted in writing the manuscript. PG, YG, and FC conceived and participated in the project design and coordination and corrected the manuscript. All authors contributed to writing the final manuscript and approved it.
Funding
This study was funded by the grants from the Natural Science foundation of China (Nos. 21407104, 81803698, 31760016) which respond for the design and the study and collection, analysis. Moreover, Weiyang Project of Xi’an (Nos. 202036, 202131), Project from Xi’an City innovation plan- Agricultural Field (21NYYF0022), Project from Ningxia Zhong ning Goji Industry Innovation Research Institute (ZNGQCX-A-2020003) and General Plan of Shaanxi Province-Agricultural Field (No. 2021NY-161), Key industrial chain projects of Shaanxi Province-Agricultural Field (2021ZDLNY04-01) are responsible for the interpretation of data and in writing the manuscript.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
ANOVA, one-way analysis of variance; BCA, bicinchoninic acid; Cd, cadmium; DN, diabetic nephropathy; ECL, chemiluminescence; GSH, glutathione; H&E, hematoxylin-eosin; HDL-C, high-density lipoprotein-cholesterol; HFSD, high fat and sugar diet; i.p.,intraperitoneally; LDL-C, low-density lipoprotein-cholesterol; mALB, microalbumin; MDA, malondialdehyde; NO, nitric oxide; OPC, proanthocyanidins; PAS, periodic acid-Schiff; PCO, protein carbonyl; PVDF, polyvinylidene difluoride; SOD, superoxide dismutase; TC, total cholesterol; TG, triglyceride.
References
- Adhikary L., Chow F., Nikolic-Paterson D. J., Stambe C., Dowling J., Atkins R. C., et al. (2004). Abnormal P38 Mitogen-Activated Protein Kinase Signalling in Human and Experimental Diabetic Nephropathy. Diabetologia 47, 1210–1222. %@ 0012-186X. 10.1007/s00125-004-1437-0 [DOI] [PubMed] [Google Scholar]
- Afridi H. I., Kazi T. G., Kazi N., Jamali M. K., Arain M. B., Jalbani N., et al. (2008). Evaluation of Status of Toxic Metals in Biological Samples of Diabetes Mellitus Patients. Diabetes Res. Clin. Pract. 80, 280–288. %@ 0168-8227. 10.1016/j.diabres.2007.12.021 [DOI] [PubMed] [Google Scholar]
- Ahn J. D., Morishita R., Kaneda Y., Kim H. J., Kim Y. D., Lee H. J., et al. (2004). Transcription Factor Decoy for AP-1 Reduces Mesangial Cell Proliferation and Extracellular Matrix Production In Vitro and In Vivo . Gene Ther. 11, 916–923. %@ 1476-5462. 10.1038/sj.gt.3302236 [DOI] [PubMed] [Google Scholar]
- Akinloye O., Ogunleye K., Oguntibeju O. O. (2010). Cadmium, lead, Arsenic and Selenium Levels in Patients with Type 2 Diabetes Mellitus. Afr. J. Biotechnol. 9, 5189–5195. %@ 1684-5315. [Google Scholar]
- Al-Hafidh Khattab M. A., Al-Youzbaki W. B. (2018). THE RELATIONSHIP BETWEEN SERUM FERRITIN AND INSULIN RESISTANCE IN TYPE 2 DIABETIC PATIENTS TREATED BY METFORMIN. Int. J. Pharm. Chem. Biol. Sci. 8, 18–26. [Google Scholar]
- Aydin Z., Karadag S., Ozturk S., Gursu M., Uzun S., Cebeci E., et al. (2019). Evaluation of the Relationship between Advanced Oxidation End Products and Inflammatory Markers in Maintenance Hemodialysis Patients. Jna 1, 24–30. 10.14302/issn.2574-4488.jna-19-3112 [DOI] [Google Scholar]
- Bacharaki D., Chrysanthopoulou E., Grigoropoulou S., Giannakopoulos P., Simitsis P., Frantzeskaki F., et al. (2021). Siblings with Coronavirus Disease 2019 Infection and Opposite Outcome-The Hemodialysis's Better Outcome Paradox: Two Case Reports. World J. Nephrol. 10, 21–28. 10.5527/wjn.v10.i2.21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bagchi D., Bagchi M., Stohs S. J., Das D. K., Ray S. D., Kuszynski C. A., et al. (2000). Free Radicals and Grape Seed Proanthocyanidin Extract: Importance in Human Health and Disease Prevention. Toxicology 148, 187–197. %@ 0300-483X. 10.1016/s0300-483x(00)00210-9 [DOI] [PubMed] [Google Scholar]
- Bak M.-J., Jun M., Jeong W.-S. (2012). Procyanidins from Wild Grape (Vitis Amurensis) Seeds Regulate ARE-Mediated Enzyme Expression via Nrf2 Coupled with P38 and PI3K/Akt Pathway in HepG2 Cells. Int. J. Mol. Sci. 13, 801–818. 10.3390/ijms13010801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beshgetoor D., Hambidge M. (1998). Clinical Conditions Altering Copper Metabolism in Humans. Am. J. Clin. Nutr. 67, 1017S–1021S. %@ 0002-9165. 10.1093/ajcn/67.5.1017S [DOI] [PubMed] [Google Scholar]
- Ceriello A. (2000). Oxidative Stress and Glycemic Regulation. Metabolism 49, 27–29. %@ 0026-0495. 10.1016/s0026-0495(00)80082-7 [DOI] [PubMed] [Google Scholar]
- Chen D. Q., Cao G., Chen H., Argyopoulos C. P., Yu H., Su W., et al. (2019). Identification of Serum Metabolites Associating with Chronic Kidney Disease Progression and Anti-fibrotic Effect of 5-methoxytryptophan. Nat. Commun. 10, 1476. 10.1038/s41467-019-09329-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen D. Q., Feng Y. L., Cao G., Zhao Y. Y. (2018). Natural Products as a Source for Antifibrosis Therapy. Trends Pharmacol. Sci. 39, 937–952. 10.1016/j.tips.2018.09.002 [DOI] [PubMed] [Google Scholar]
- Chen D. Q., Hu H. H., Wang Y. N., Feng Y. L., Cao G., Zhao Y. Y. (2018). Natural Products for the Prevention and Treatment of Kidney Disease. Phytomedicine 50, 50–60. 10.1016/j.phymed.2018.09.182 [DOI] [PubMed] [Google Scholar]
- Chen D. Q., Cao G., Chen H., Liu D., Su W., Yu X. Y., et al. (2017). Gene and Protein Expressions and Metabolomics Exhibit Activated Redox Signaling and Wnt/β-Catenin Pathway Are Associated with Metabolite Dysfunction in Patients with Chronic Kidney Disease. Redox Biol. 12, 505–521. 10.1016/j.redox.2017.03.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen D. Q., Feng Y. L., Chen L., Liu J. R., Wang M., Vaziri N. D., et al. (2019). Poricoic Acid A Enhances Melatonin Inhibition of AKI-To-CKD Transition by Regulating Gas6/Axl-NF-κB/Nrf2 axis. Free Radic. Biol. Med. 134, 484–497. 10.1016/j.freeradbiomed.2019.01.046 [DOI] [PubMed] [Google Scholar]
- Chen H., Cao G., Chen D. Q., Wang M., Vaziri N. D., Zhang Z. H., et al. (2016). Metabolomics Insights into Activated Redox Signaling and Lipid Metabolism Dysfunction in Chronic Kidney Disease Progression. Redox Biol. 10, 168–178. 10.1016/j.redox.2016.09.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L., Lei L., Jin T., Nordberg M., Nordberg G. F. (2006). Plasma Metallothionein Antibody, Urinary Cadmium, and Renal Dysfunction in a Chinese Type 2 Diabetic Population. Diabetes care 29, 2682–2687. %@ 0149-5992. 10.2337/dc06-1003 [DOI] [PubMed] [Google Scholar]
- Chen L., Chen D. Q., Liu J. R., Zhang J., Vaziri N. D., Zhuang S., et al. (2019). Unilateral Ureteral Obstruction Causes Gut Microbial Dysbiosis and Metabolome Disorders Contributing to Tubulointerstitial Fibrosis. Exp. Mol. Med. 51, 38. 10.1038/s12276-019-0234-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chuang P. Y., Yu Q., Fang W., Uribarri J., He J. C. (2007). Advanced Glycation Endproducts Induce Podocyte Apoptosis by Activation of the FOXO4 Transcription Factor. Kidney Int. 72, 965–976. %@ 0085-2538. 10.1038/sj.ki.5002456 [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiSilvestro R. A. (2000). Zinc in Relation to Diabetes and Oxidative Disease. J. Nutr. 130, 1509S–1511S. %@ 0022-3166. 10.1093/jn/130.5.1509S [DOI] [PubMed] [Google Scholar]
- Duran-Salgado M. B., Rubio-Guerra A. F. (2014). Diabetic Nephropathy and Inflammation. World J. Diabetes 5, 393–398. 10.4239/wjd.v5.i3.393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards J., Ackerman C. (2016). A Review of Diabetes Mellitus and Exposure to the Environmental Toxicant Cadmium with an Emphasis on Likely Mechanisms of Action. Curr. Diabetes Rev. 12, 252–258. %@ 1573-3998. 10.2174/1573399811666150812142922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards J. R., Prozialeck W. C. (2009). Cadmium, Diabetes and Chronic Kidney Disease. Toxicol. Appl. Pharmacol. 238, 289–293. %@ 0041-008X. 10.1016/j.taap.2009.03.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eliasson B., Björnsson E., Urbanavicius V., Andersson H., Fowelin J., Attvall S., et al. (1995). Hyperinsulinaemia Impairs Gastrointestinal Motility and Slows Carbohydrate Absorption. Diabetologia 38, 79–85. %@ 0012-186X. 10.1007/BF02369356 [DOI] [PubMed] [Google Scholar]
- Feng Y. L., Cao G., Chen D. Q., Vaziri N. D., Chen L., Zhang J., et al. (2019). Microbiome-metabolomics Reveals Gut Microbiota Associated with Glycine-Conjugated Metabolites and Polyamine Metabolism in Chronic Kidney Disease. Cell Mol. Life Sci. : CMLS 76, 4961–4978. 10.1007/s00018-019-03155-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng Y. L., Chen H., Chen D. Q., Vaziri N. D., Su W., Ma S. X., et al. (2019). Activated NF-κB/Nrf2 and Wnt/β-Catenin Pathways Are Associated with Lipid Metabolism in CKD Patients with Microalbuminuria and Macroalbuminuria. Biochim. Biophys. Acta Mol. basis Dis. 1865, 2317–2332. 10.1016/j.bbadis.2019.05.010 [DOI] [PubMed] [Google Scholar]
- Fine A. M. (2000). Oligomeric Proanthocyanidin Complexes: History, Structure, and Phytopharmaceutical Applications. Altern. Med. Rev. a J. Clin. Ther. 5 (2), 144–151. [PubMed] [Google Scholar]
- Ghosh R., Mukherjee B., Chatterjee M. (1994). A Novel Effect of Selenium on Streptozotocin-Induced Diabetic Mice. Diabetes Res. (Edinburgh, Scotland) 25, 165–171. %@ 0265-5985. [PubMed] [Google Scholar]
- Gong P., Chang X., Chen X., Bai X., Wen H., Pi S., et al. (2017). Metabolomics Study of Cadmium-Induced Diabetic Nephropathy and Protective Effect of Caffeic Acid Phenethyl Ester Using UPLC-Q-TOF-MS Combined with Pattern Recognition. Environ. Toxicol. Pharmacol. 54, 80–92. %@ 1382-6689. 10.1016/j.etap.2017.06.021 [DOI] [PubMed] [Google Scholar]
- Gong P., Chen F., Liu X., Gong X., Wang J., Ma Y. (2012). Protective Effect of Caffeic Acid Phenethyl Ester against Cadmium-Induced Renal Damage in Mice. J. Toxicol. Sci. 37, 415–425. 10.2131/jts.37.415 [DOI] [PubMed] [Google Scholar]
- Gong P., Chen F. X., Ma G. F., Feng Y., Zhao Q., Wang R. (2008). Endomorphin 1 Effectively Protects Cadmium Chloride-Induced Hepatic Damage in Mice. Toxicology 251, 35–44. %@ 0300-483X. 10.1016/j.tox.2008.07.051 [DOI] [PubMed] [Google Scholar]
- Ha H., Kim K. H. (1995). Role of Oxidative Stress in the Development of Diabetic Nephropathy. Kidney Int. Supplement 51, S18–S21. [PubMed] [Google Scholar]
- Hamasaki H., Kawashima Y., Yanai H. (2016). Serum Zn/Cu Ratio Is Associated with Renal Function, Glycemic Control, and Metabolic Parameters in Japanese Patients with and without Type 2 Diabetes: a Cross-Sectional Study. Front. Endocrinol. 7, 147. 10.3389/fendo.2016.00147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansrivijit P., Chen Y.-J., Lnu K., Trongtorsak A., Puthenpura M. M., Thongprayoon C., et al. (2021). Prediction of Mortality Among Patients with Chronic Kidney Disease: A Systematic Review. Wjn 10, 59–75. 10.5527/wjn.v10.i4.59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harborne J. B., Mabry T. J. (2013). The Flavonoids: Advances in Research. Boston, MA: Springer. 10.1007/978-1-4899-2915-0 [DOI] [Google Scholar]
- Hou D.-X., Masuzaki S., Hashimoto F., Uto T., Tanigawa S., Fujii M., et al. (2007). Green tea Proanthocyanidins Inhibit Cyclooxygenase-2 Expression in LPS-Activated Mouse Macrophages: Molecular Mechanisms and Structure–Activity Relationship. Arch. Biochem. Biophys. 460, 67–74. 10.1016/j.abb.2007.01.009 [DOI] [PubMed] [Google Scholar]
- Huie R. E., Padmaja S. (1993). The Reaction of NO with Superoxide. Free Radic. Res. Commun. 18, 195–199. %@ 8755-0199. 10.3109/10715769309145868 [DOI] [PubMed] [Google Scholar]
- Igarashi M., Wakasaki H., Takahara N., Ishii H., Jiang Z.-Y., Yamauchi T., et al. (1999). Glucose or Diabetes Activates P38 Mitogen-Activated Protein Kinase via Different Pathways. J. Clin. Invest. 103, 185–195. %@ 0021-9738. 10.1172/JCI3326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Izzo A. A., Teixeira M., Alexander S. P. H., Cirino G., Docherty J. R., George C. H., et al. (2020). A Practical Guide for Transparent Reporting of Research on Natural Products in the British Journal of Pharmacology: Reproducibility of Natural Product Research. Br. J. Pharmacol. 177, 2169–2178. 10.1111/bph.15054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain D., Haddad D. B., Goel N. (2019). Choice of Dialysis Modality Prior to Kidney Transplantation: Does it Matter? World J. Nephrol. 8, 1–10. 10.5527/wjn.v8.i1.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li S. S., Sun Q., Hua M. R., Suo P., Chen J. R., Yu X. Y., et al. (2021). Targeting the Wnt/β-Catenin Signaling Pathway as a Potential Therapeutic Strategy in Renal Tubulointerstitial Fibrosis. Front. Pharmacol. 12, 719880. 10.3389/fphar.2021.719880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madrigal J. M., Ricardo A. C., Persky V., Turyk M. (2019). Associations between Blood Cadmium Concentration and Kidney Function in the U.S. Population: Impact of Sex, Diabetes and Hypertension. Environ. Res. 169, 180–188. %@ 0013-9351. 10.1016/j.envres.2018.11.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mansouri E., Panahi M., Ghaffari M. A., Ghorbani A. (2011). Effects of Grape Seed Proanthocyanidin Extract on Oxidative Stress Induced by Diabetes in Rat Kidney. Iranian Biomed. J. 15, 100. [PMC free article] [PubMed] [Google Scholar]
- Mantovani A., Zusi C. (2020). PNPLA3 Gene and Kidney Disease. Explor Med. 1, 42–50. 10.37349/emed.2020.00004 [DOI] [Google Scholar]
- Medina Rangel P. X., Priyadarshini A., Tian X. (2021). New Insights into the Immunity and Podocyte in Glomerular Health and Disease: From Pathogenesis to Therapy in Proteinuric Kidney Disease. Integr. Med. Nephrol. Androl. 8, 5. 10.4103/imna.imna_26_21 [DOI] [Google Scholar]
- Miao H., Cao G., Wu X. Q., Chen Y. Y., Chen D. Q., Chen L., et al. (2020). Identification of Endogenous 1-aminopyrene as a Novel Mediator of Progressive Chronic Kidney Disease via Aryl Hydrocarbon Receptor Activation. Br. J. Pharmacol. 177, 3415–3435. 10.1111/bph.15062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miao H., Wu X. Q., Zhang D. D., Wang Y. N., Guo Y., Li P., et al. (2021). Deciphering the Cellular Mechanisms Underlying Fibrosis-Associated Diseases and Therapeutic Avenues. Pharmacol. Res. 163, 105316. 10.1016/j.phrs.2020.105316 [DOI] [PubMed] [Google Scholar]
- Miao H., Wu X. Q., Wang Y. N., Chen D. Q., Chen L., Vaziri N. D., et al. (2021). 1-Hydroxypyrene Mediates Renal Fibrosis through Aryl Hydrocarbon Receptor Signalling Pathway. Br. J. Pharmacol. 10.1111/bph.15705 [DOI] [PubMed] [Google Scholar]
- Mohora M., Vîrgolici B., Paveliu F., Lixandru D., Muscurel C., Greabu M. (2006). Free Radical Activity in Obese Patients with Type 2 Diabetes Mellitus. Rom. J. Intern. medicine= Revue roumaine de médecine interne 44 (2), 69–78. [PubMed] [Google Scholar]
- Newman D. J., Cragg G. M. (2020). Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 83, 770–803. 10.1021/acs.jnatprod.9b01285 [DOI] [PubMed] [Google Scholar]
- Park C., Yook J., Lim H., Hong Y. (2019). Exposure to Cadmium Is Associated with Impaired Fasting Glucose. Environ. Epidemiol. 3, 303. 10.1097/01.EE9.0000609288.34911.56 [DOI] [Google Scholar]
- Pavel M. A., Petersen E. N., Wang H., Lerner R. A., Hansen S. B. (2020). Studies on the Mechanism of General Anesthesia. Proc. Natl. Acad. Sci. 117 (24), 13757–13766. 10.1073/pnas.2004259117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perrone R. D., Madias N. E., Levey A. S. (1992). Serum Creatinine as an index of Renal Function: New Insights into Old Concepts. Clin. Chem. 38, 1933–1953. %@ 0009-9147. 10.1093/clinchem/38.10.1933 [DOI] [PubMed] [Google Scholar]
- Pokusa M., Kráľová Trančíková A. (2017). The central Role of Biometals Maintains Oxidative Balance in the Context of Metabolic and Neurodegenerative Disorders. Oxidative Med. Cell. longevity 2017, 8210734. 10.1155/2017/8210734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reidy K., Kang H. M., Hostetter T., Susztak K. (2014). Molecular Mechanisms of Diabetic Kidney Disease. J. Clin. Invest. 124, 2333–2340. 10.1172/JCI72271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruan Y., Jin Q., Zeng J., Ren F., Xie Z., Ji K., et al. (2020). Grape Seed Proanthocyanidin Extract Ameliorates Cardiac Remodelling after Myocardial Infarction through PI3K/AKT Pathway in Mice. Front. Pharmacol. 11, 585984. 10.3389/fphar.2020.585984 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satarug S., Garrett S. H., Sens M. A., Sens D. A. (2010). Cadmium, Environmental Exposure, and Health Outcomes. Environ. Health Perspect. 118, 182–190. %@ 0091-6765. 10.1289/ehp.0901234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satarug S., Vesey D. A., Gobe G. C. (2017). Kidney Cadmium Toxicity, Diabetes and High Blood Pressure: the Perfect Storm. Tohoku J. Exp. Med. 241, 65–87. %@ 0040-8727. 10.1620/tjem.241.65 [DOI] [PubMed] [Google Scholar]
- Stadler K., Jenei V., Von Bölcsházy G., Somogyi A., Jakus J. (2003). Increased Nitric Oxide Levels as an Early Sign of Premature Aging in Diabetes. Free Radic. Biol. Med. 35, 1240–1251. %@ 0891-5849. 10.1016/s0891-5849(03)00499-4 [DOI] [PubMed] [Google Scholar]
- Sugano M., Yamato H., Hayashi T., Ochiai H., Kakuchi J., Goto S., et al. (2006). High-fat Diet in Low-Dose-Streptozotocin-Treated Heminephrectomized Rats Induces All Features of Human Type 2 Diabetic Nephropathy: a New Rat Model of Diabetic Nephropathy. Nutr. Metab. Cardiovasc. Dis. 16, 477–484. %@ 0939-4753. 10.1016/j.numecd.2005.08.007 [DOI] [PubMed] [Google Scholar]
- Susztak K., Raff A. C., Schiffer M., Böttinger E. P. (2006). Glucose-induced Reactive Oxygen Species Cause Apoptosis of Podocytes and Podocyte Depletion at the Onset of Diabetic Nephropathy. Diabetes 55, 225–233. %@ 0012-1797. 10.2337/diabetes.55.01.06.db05-0894 [DOI] [PubMed] [Google Scholar]
- Tan K. C. B., Ai V. H. G., Chow W. S., Chau M. T., Leong L., Lam K. S. L. (1999). Influence of Low Density Lipoprotein (LDL) Subfraction Profile and LDL Oxidation on Endothelium-dependent and Independent Vasodilation in Patients with Type 2 Diabetes. J. Clin. Endocrinol. Metab. 84, 3212–3216. %@ 0021-972X. 10.1210/jcem.84.9.5959 [DOI] [PubMed] [Google Scholar]
- Tangvarasittichai S., Niyomtam S., Meemark S., Pingmuangkaew P., Nunthawarasilp P. (2015). Elevated Cadmium Exposure Associated with Hypertension, Diabetes and Chronic Kidney Disease, in the Population of Cadmium-Contaminated Area. Int. J. Toxicol. Pharmacol. Res. 7, 50–56. [Google Scholar]
- Valavanidis A., Vlahogianni T., Dassenakis M., Scoullos M. (2006). Molecular Biomarkers of Oxidative Stress in Aquatic Organisms in Relation to Toxic Environmental Pollutants. Ecotoxicology Environ. Saf. 64, 178–189. 10.1016/j.ecoenv.2005.03.013 [DOI] [PubMed] [Google Scholar]
- Van Sandwijk M. S., Klooster A., Ten Berge I. J., Diepstra A., Florquin S., Hoelbeek J. J., et al. (2019). Complement Activation and Long-Term Graft Function in ABO-Incompatible Kidney Transplantation. World J. Nephrol. 8, 95–108. 10.5527/wjn.v8.i6.95 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaziri N. D. (2006). Dyslipidemia of Chronic Renal Failure: the Nature, Mechanisms, and Potential Consequences. Am. J. Physiology-Renal Physiol. 290, F262–F272. %@ 1931-857X. 10.1152/ajprenal.00099.2005 [DOI] [PubMed] [Google Scholar]
- Stefanovic V., Savic V., Vlahovic P., Cvetkovic T., Najman S., Mitic-Zlatkovic M. (2000). Reversal of Experimental Myoglobinuric Acute Renal Failure with Bioflavonoids from Seeds of Grape. Ren. Fail. 22, 255–266. %@ 0886-022X. 10.1081/jdi-100100870 [DOI] [PubMed] [Google Scholar]
- Wallin M., Sallsten G., Lundh T., Barregard L. (2014). Low-level Cadmium Exposure and Effects on Kidney Function. Occup. Environ. Med. 71, 848–854. %@ 1351-0711. 10.1136/oemed-2014-102279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang M., Chen D. Q., Chen L., Cao G., Zhao H., Liu D., et al. (2018). Novel Inhibitors of the Cellular Renin-Angiotensin System Components, Poricoic Acids, Target Smad3 Phosphorylation and Wnt/β-Catenin Pathway against Renal Fibrosis. Br. J. Pharmacol. 175, 2689–2708. 10.1111/bph.14333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang M., Hu H. H., Chen Y. Y., Chen L., Wu X. Q., Zhao Y. Y. (2020). Novel Poricoic Acids Attenuate Renal Fibrosis through Regulating Redox Signalling and Aryl Hydrocarbon Receptor Activation. Phytomedicine 79, 153323. 10.1016/j.phymed.2020.153323 [DOI] [PubMed] [Google Scholar]
- Wang R.-M., Wang Z.-B., Wang Y., Liu W.-Y., Li Y., Tong L.-C., et al. (2018). Swiprosin-1 Promotes Mitochondria-dependent Apoptosis of Glomerular Podocytes via P38 MAPK Pathway in Early-Stage Diabetic Nephropathy. Cell Physiol. Biochem. 45, 899–916. %@ 1015-8987. 10.1159/000487285 [DOI] [PubMed] [Google Scholar]
- Webster A. C., Nagler E. V., Morton R. L., Masson P. (2017). Chronic Kidney Disease. Lancet 389, 1238–1252. 10.1016/S0140-6736(16)32064-5 [DOI] [PubMed] [Google Scholar]
- Wu X. Q., Zhang D. D., Wang Y. N., Tan Y. Q., Yu X. Y., Zhao Y. Y. (2021). AGE/RAGE in Diabetic Kidney Disease and Ageing Kidney. Free Radic. Biol. Med. 171, 260–271. 10.1016/j.freeradbiomed.2021.05.025 [DOI] [PubMed] [Google Scholar]
- Yang Y., Wu C. (2021). Traditional Chinese Medicine in Ameliorating Diabetic Kidney Disease via Modulating Gut Microbiota. Integr. Med. Nephrol. Androl. 8, 8. 10.4103/imna.imna_28_21 [DOI] [Google Scholar]
- Yokozawa T., Cho E. J., Park C. H., Kim J. H. (2012). Protective Effect of Proanthocyanidin against Diabetic Oxidative Stress. Evid Based. Complement. Altern. Med. 2012, 623879. 10.1155/2012/623879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng H., Whitman S. A., Wu W., Wondrak G. T., Wong P. K., Fang D., et al. (2011). Therapeutic Potential of Nrf2 Activators in Streptozotocin-Induced Diabetic Nephropathy. Diabetes 60, 3055–3066. %@ 0012-1797. 10.2337/db11-0807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou X. F., Wang Y., Luo M. J., Zhao T. T., Li P. (2021). Tangshen Formula Attenuates Renal Fibrosis by Downregulating Transforming Growth Factor β1/Smad3 and LncRNA-MEG3 in Rats with Diabetic Kidney Disease. Integr. Med. Nephrol. Androl. 8, 2. 10.4103/imna.imna_22_21 [DOI] [Google Scholar]
- Zoja C., Xinaris C., Macconi D. (2020). Diabetic Nephropathy: Novel Molecular Mechanisms and Therapeutic Targets. Front. Pharmacol. 11, 586892. 10.3389/fphar.2020.586892 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The raw data supporting the conclusion of this article will be made available by the authors, without undue reservation.