Abstract
Compound blue nevus had clinical and histological similarities with other heavily pigmented melanocytic tumor, like the pigmented epithelioid melanocytoma. Distinctive genomic aberrations have allowed differentiating it. The defining characteristic of blue nevi family is the presence of activating mutations in the G protein α‐subunits, GNAQ and GNA11.
Keywords: dermatology, genetics
The particular clinical, histologic, and molecular features allow to differentiate compound blue nevus from pigmented epithelioid melanocytoma suggest that the latter does not belong to the BN family despite the microscopic resemblance.

1. INTRODUCTION
Blue nevus (BN) is a benign melanocytic lesion that owes its name to its blue color due to Tyndall effect. 1 Moreover, blue nevi are a heterogeneous group of lesions that can display a variety of clinicopathological characteristics. In fact, they occur mainly in childhood, adolescence, or early adulthood but they can occur at any age. Females are more frequently affected than males. In fact, the most common presentation is the isolated blue or blue‐black dome‐shaped well‐demarcated macules or papules, which are most common on the face, the scalp, the sacral area, and the dorsal face of extremities. The dermoscopic appearance of BN is commonly characterized by global patterns, such as homogeneous bluish or steel‐blue grayish pigmentation. However, vascular structures are not commonly seen in blue nevi, but in some case series, polymorphic, dotted, comma, linear irregular, and arborizing vessels have been reported. Moreover, a dermoscopic rainbow pattern was also described. 2 For their part, the blue nevi include different histological types, such as common, cellular, desmoplastic cellular, combined, compound, and malignant. 3
We report a challenging case of compound BN (CBN) with clinical and histological similarities with other heavily pigmented melanocytic tumor‐like “pigmented epithelioid melanocytoma.” We completed with a molecular testing which revealed a mutation for GNAQ leading to the final diagnosis of CBN.
2. CASE REPORT
A 43‐year‐old man, with no medical history, presented with a 4‐yearhistory of solitary painless nodule of the scalp that had slightly increased in size in the last 4 months; however, he had no personal or familial history of melanoma. In fact, a physical examination revealed a blue‐gray nodular lesion measuring 15 mm in diameter, firm, well‐demarcated symmetrical on the occipital region of the scalp (Figure 1). Dermoscopy showed a blue‐steel pigmentation with hypopigmented yellowish areas and linear irregular vessels (Figure 2). A marginal excision was performed. Histopathological examination revealed an epithelioid and spindle‐shaped cell proliferation in the dermis, composed of mildly atypical melanocytes with a heavy pigmentation (Figure 3A). Then, the nuclei were vesicular, with prominent nucleoli and rare mitoses (1/10HPF). Melanophages were abundant between fibrotic bundles. An additional junctional dendritic component was associated (Figure 3B). However, no perineural invasion, ulceration, vascular emboli, satellites, or in transit metastases were noted. On the contrary, immunohistochemical staining showed an expression of HMB45 and a low proliferation index (Ki67<1%). From the above findings, we can say that the proposed diagnosis is a heavily pigmented melanocytic tumor evoking pigmented epithelioid melanocytoma (PEM) while additional immunohistochemistry did not identify any inactivating mutation for PRKAR1A. Moreover, molecular testing revealed a mutation for GNAQ but not for BRAF, NRAS, and CKIT.
FIGURE 1.
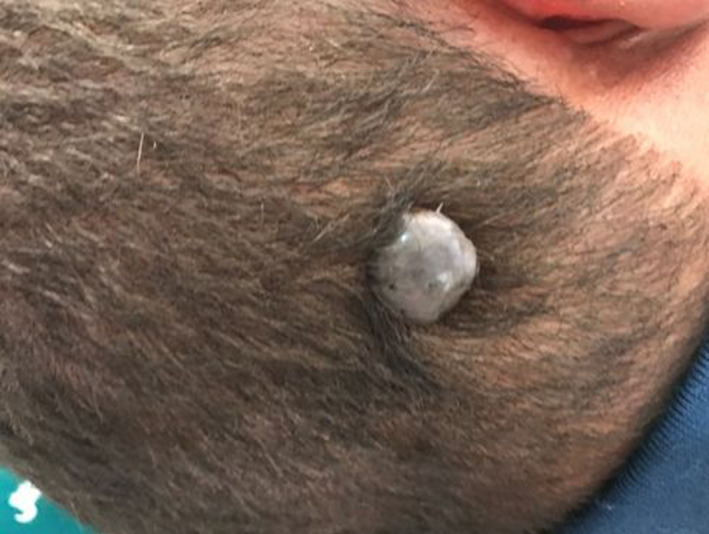
Solitary, well‐demarcated and symmetrical blue‐gray nodular lesion, measuring 15 mm in diameter
FIGURE 2.
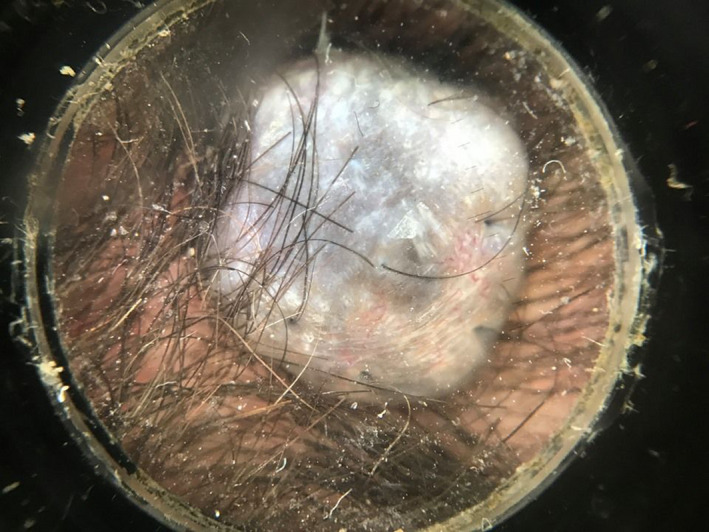
Dermoscopic findings: blue‐steel pigmentation with hypopigmented yellowish areas and linear irregular vessels
FIGURE 3.
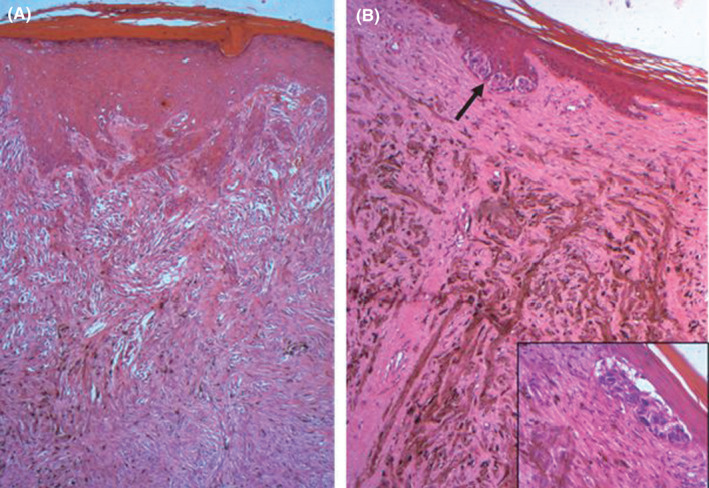
(A) Histology showing dermal proliferation of heavily pigmented epithelioid (HE× 400); (B) Spindle cells with an additional junctional dendritic component (arrow and inset; HE× 200)
2.1. Diagnosis
Compound blue nevus (CBN).
3. DISCUSSION
Common BN is a well‐defined dermal melanocytic proliferation characterized by the absence of the dermo‐epidermal junctional component. It frequently shows a heavily pigmented proliferation of fusiform melanocytes with elongated dendritic processes and a Grenz zone. Usually, the lesional cells are strongly immunoreactive for HMB45 and MART1, whereas S100 protein may be negative.
Unlike the BN, the PEM often has a junctional component and distinctive large epithelioid cells with vesicular nuclei and prominent nucleoli resembling Reed Sternberg cells. PEM is genetically linked to PRKAR1A mutations or PRKCA fusions, but not to GNAQ, GNA11, or CYSLTR2 mutations. 4 Then, the defining characteristic of the BN family is the presence of activating mutations in the G protein α‐subunits, GNAQ and GNA11, which can be identified in up to 90% of cases. 5 On the contrary, malignant blue nevi acquire additional mutations, including in BAP1 on chromosome. 3 These mutations are different from various kinase fusions or HRAS mutations found in Spitz neoplasms and from BRAF or NRAS mutations in conventional melanocytic nevi. 6
In fact, in 1990, Kamino and Tam described a rare variant of BN characterized by the presence of an additional junctional dendritic component, contiguous with the dermal component. They coined the term “compound blue nevus” (CBN). 7 However, very few papers have so far confirmed the existence of this kind of BN. In 2002, Ferrara et al. reported five other cases with similar histological findings. The authors concluded that CBN is a clinically, dermoscopically, and histologically distinctive variant of BN. 8
We report a case of challenging case of Gαq‐mutated CBN, which expands the morphological spectrum of the variant of BN and can mimic other compound lesions especially PEM. In fact, CBN has been the topic of limited publications with challenging diagnosis. Distinctive genomic aberrations can be differentiated from the other nevi in order to avoid unnecessary invasive procedure.
CONFLICT OF INTEREST
The authors have no potential conflict of interest to disclose.
AUTHOR CONTRIBUTIONS
MB and KS wrote the manuscript. NE revised the manuscript. IS, CK, NG, and TS wrote parts of the manuscript related to the histopathological aspects of the disease. HBM, FK, and MA contributed to the management of the patient and revised the article. HT critically reviewed the manuscript and gave final approval. All authors have read and approved the final manuscript and agree to take full responsibility for the integrity and accuracy of the work.
CONSENT
Written informed consent was obtained from the patient to publish this report in accordance with the journal's patient consent policy.
ACKNOWLEDGEMENT
None.
Baklouti M, Sellami K, Elleuch N, et al. Compound blue nevus: A misleading pigmented melanocytic tumor. Clin Case Rep. 2022;10:e05311. doi: 10.1002/ccr3.5311
Funding information
None
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Zembowicz A. Blue nevi and related tumors. Clin Lab Med. 2017;37(3):401‐415. [DOI] [PubMed] [Google Scholar]
- 2. Piccolo V, De Barros M, Corneli P, et al. Dermoscopy of blue naevus on acral volar skin: a review of the literature. Australas J Dermatol. 2019;60(4):336‐338. [DOI] [PubMed] [Google Scholar]
- 3. Caroline de Lorenzi C, Quenan S, Ibrahim YL. Atypical cellular blue nevus of the foot: a case report. Dermatopathology (Basel). 2019;6(1):20‐22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Goto K, Pissaloux D, Paindavoine S, et al. CYSLTR2‐mutant cutaneous melanocytic neoplasms frequently simulate “pigmented epithelioid melanocytoma”, expanding the morphologic spectrum of blue tumors: a clinicopathologic study of 7 cases. Am J Surg Pathol. 2019;43(10):1368‐1376. [DOI] [PubMed] [Google Scholar]
- 5. Jaquemus J, Perron E, Buisson A, et al. Compound blue nevus: a reappraisal of the concept in the genomic era. Virchows Arch. 2020;476(3):439‐443. [DOI] [PubMed] [Google Scholar]
- 6. Dika E, Fanti PA, Fiorentino M, et al. Spitzoid tumors in children and adults: a comparative clinical, pathological, and cytogenetic analysis. Melanoma Res. 2015;25(4):295‐301. [DOI] [PubMed] [Google Scholar]
- 7. Kamino H, Tam ST. Compound blue nevus: a variant of blue nevus with an additional junctional dendritic component: a clinical, histopathologic, and immunohistochemical study of six cases. Arch Dermatol. 1990;126(10):1330‐1333. [PubMed] [Google Scholar]
- 8. Ferrara G, Argenziano G, Zgavec B, et al. Compound blue nevus”: a reappraisal of “superficial blue nevus with prominent intraepidermal dendritic melanocytes” with emphasis on dermoscopic and histopathologic features. J Am Acad Dermatol. 2002;46(1):85‐89. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


