Abstract
Multisystem inflammatory syndrome (MIS) is a severe complication described in a minority of patients with COVID-19. Myocarditis has been reported in patients with COVID-19, including MIS. In this study, we compared the clinical characteristics and cardiac magnetic resonance (CMR) findings of COVID-19 myocarditis in patients with and without MIS. In the 330 patients with COVID-19 who were referred for CMR at our institution between July 24, 2020, to March 31, 2021, 40 patients were identified as having myocarditis, MIS myocarditis (n = 21) and non-MIS myocarditis (n = 19). MIS myocarditis was characterized by global myocardial inflammation/edema with significantly elevated native T1, whereas only regional inflammation, and edema were noted in the non-MIS group. Distinct late gadolinium enhancement (LGE) patterns—inferior myocardial involvement in non-MIS myocarditis and septal involvement in MIS myocarditis—were identified. The LGE burden was comparable between the 2 groups (5.9% vs 6.6%, MIS vs non-MIS group, p = 0.83). Myocarditis was diagnosed more frequently by CMR in the MIS group (70% vs 6.3%, MIS vs non-MIS, p <0.001). In the 20 patients with a sequential CMR study at a median 102-day follow-up, 25% had persistent myocardial edema. The LGE burden improved over time, from a median of 5.0% (interquartile range 3.4% to 7.3%) to 3.2% (interquartile range 2.0% to 3.8%; p <0.001). In conclusion, MIS and non-MIS myocarditis exhibit distinct characteristics by CMR. Persistent LGE and edema were common at follow-up CMR examination in both groups.
Since the initial outbreak in December 2019, COVID-19 has quickly become a major global health threat.1 , 2 Myocardial injury has been reported in 7% to 28% of patients during acute hospitalization,1 , 3 , 4 and has been associated with increased mortality.3 , 4 Multisystem inflammatory syndrome (MIS) in children (MIS-C) or adults (MIS-A) is a serious condition that occurs in a small subset of patients diagnosed with COVID-19. It presents with a hyperinflammatory state and extrapulmonary organ dysfunction but minimal respiratory symptoms.5 , 6 The reported incidence of MIS-C in the United States was 0.14% in children with COVID-19 with a 1.4% mortality.7 Cardiovascular involvement has been documented in most patients,8 and acute cardiac failure is commonly observed.9 COVID-19–related myocarditis has been reported in patients with and without MIS.5 , 6 , 10, 11, 12, 13 Viral myocarditis is a frequent cause of dilated cardiomyopathy and sudden cardiac death.14 Therefore, the potential long-term cardiac effects of COVID-19 related myocarditis are of great concern. Given that cardiac magnetic resonance (CMR) imaging with parametric mapping plays a substantial role in the diagnosis and prognosis of myocarditis,15 the short-term and long-term CMR findings are of crucial importance. Although MIS has been considered a distinct entity from SARS-CoV-2 infection,1 , 6 it is unclear whether myocarditis related to MIS exhibits different CMR imaging patterns or clinical features when compared with non-MIS myocarditis. In this study, we aimed to compare the clinical characteristics and CMR features in patients with MIS and non-MIS myocarditis after recovery from COVID-19.
The study was approved by the Vanderbilt University Medical Center Institutional Review Board. A total of 330 patients diagnosed with COVID-19 were referred for a CMR at our institution between July 24, 2020, and March 31, 2021. The referral reasons included: (1) cardiac clearance for athletes before return to training; (2) patients recovering from COVID-19 with persistent or delayed (>2 weeks) cardiovascular symptoms, including chest discomfort, dyspnea, palpitations, presyncope, or syncope; (3) patients presenting with acute myocardial injury related to COVID-19 and/or MIS, despite lack of clinical symptoms: including new cardiac dysfunction, abnormal electrocardiogram (ECG), cardiac enzyme elevation. All patients who underwent CMR were included. Patients with CMR features of myocarditis based on the modified Lake Louise Criteria15 were divided based on clinical diagnosis into 2 groups, MIS myocarditis, and non-MIS myocarditis. MIS was diagnosed by the care team during acute hospitalization according to current recommendations.6
The baseline clinical data were collected through a review of the electronic medical records. Data extracted included demographic characteristics, medical co-morbidities including hypertension, diabetes mellitus, chronic kidney disease, chronic obstructive lung disease or asthma, chronic liver disease, cerebral vascular accident, coronary artery disease, and known history of structural heart disease. COVID-19 was diagnosed by either positive SARS-CoV-2 on real-time reverse transcriptase–polymerase chain reaction assay from the nasopharyngeal swab, and/or serum immunoglobulin M or immunoglobulin G positivity if the polymerase chain reaction result was negative. Troponin I level, and ECGs were collected during clinical care for cardiac symptoms, or at the time of COVID-19 diagnosis for asymptomatic athletic patients who were universally screened with CMR for myopericarditis following COVID-19 per our institutional protocol.
Two CMR platforms were used in the exams: 1.5 T Siemens Avanto Fit scanner (Siemens Healthcare Sector, Erlangen, Germany) or 3.0 T Philips Ingenia Elition scanner (Phillips Healthcare, Amsterdam, The Netherlands). Balanced steady-state free precession cine imaging was used to evaluate cardiac function, volume, and mass. Gadolinium contrast (gadobutrol [Gadavist Bayer Healthcare Pharmaceuticals, Wayne, New Jersey] at a dose of 0.15 mmol/kg for 1.5-T scanner studies, and 0.2 mmol/kg for 3.0-T scanner studies) was administered through a peripheral intravenous line unless the patient had significantly impaired renal function with estimated glomerular filtration rate <30 ml/min (1 patient did not receive contrast). Late gadolinium enhancement (LGE) imaging was performed using segmented inversion recovery and single-shot phase-sensitive inversion recovery imaging in standard long-axis planes and a short-axis stack. Myocardial T1, T2, and after contrast T1 maps were acquired in the basal short-axis, mid short-axis, and four-chamber long-axis orientations. T1 maps were obtained using a modified Look-Locker inversion recovery sequence acquired using a 5(3s)3 protocol before contrast and a 4(1)3(1)2 protocol 15 minutes after contrast administration. Modified Look-Locker inversion recovery sequences were motion-corrected (only on the 1.5-T scanner), ECG-triggered, and obtained in diastole with typical imaging parameters: nonselective inversion with a 35° flip angle, single-shot steady-state free precession imaging, initial inversion time of 120 ms with 80 ms increments, field of view 340 × 272 mm2, matrix size 256 × 144, slice thickness 8 mm, voxel size 1.3 × 1.9 × 8.0 mm3, TR/TE 2.6 ms/1.1 ms, parallel imaging factor of 2. The matrix size was decreased to 192 × 128 for heart rates >90 (approximate voxel size 1.8 × 2.1 × 8 mm3). T2 mapping was performed using a breath-held, ECG-triggered, balanced steady-state free precession sequence with motion correction. Typical imaging parameters were as follows: adiabatic T2 preparation with 35° flip angle, field of view 340 × 272 mm2, matrix size 192 × 144, slice thickness 8 mm, voxel size 1.8 × 1.9 × 8.0 mm3, TR/TE 2.5 ms/1.1 ms, parallel imaging factor of 2.
CMR postprocessing was performed using Medis QMass and QMaps (MedisSuite 3.2, Medis, Leiden, The Netherlands). Left and right ventricular function parameters were automatically calculated from the endocardial and epicardial contours. The assessment of LGE was performed by cardiologists with extensive CMR reading experience (S.G.H., J.M.D., D.E.C., D.A.P., J.D.C.). Global T1, T2, and ECV values were obtained automatically after delineation within the LV mesocardium. Internal control data of native T1 and T2 were obtained from prospectively enrolled healthy controls for each magnet and were used to generate internal normal ranges. Normal ranges of global myocardial T1 by 1.5 and 3.0-T platforms were 930 to 1,010 ms, and 1,190 to 1,290 ms, respectively. The normal ranges of global myocardial T2 at 1.5 and 3.0 T were <50 and <49 ms, respectively. To compare the T1 and T2 between the non-MIS and MIS groups across field strengths, T1 and T2 Z-scores were calculated based on the Formula Z-score = (subject mean − control mean)/(control SD) using the control data from each CMR platform. Areas of LGE determined not to be because of myocardial infarction were quantified using the full width at half maximum method, as this has previously been shown to have the best intra-observer and inter-observer reproducibility and the strongest association with major adverse cardiac events.16 , 17 Acute myocarditis was diagnosed using the modified Lake Louise criteria.15 A combination of subepicardial and/or intramural myocardial LGE without myocardial edema was considered healed myocarditis after exclusion of other explanations.
Data are presented as count and frequencies for categorical variables, and median and interquartile range (IQR) for continuous variables. Comparisons between 2 independent groups were made by two-sided Fisher's exact test for categorical variables, and Mann-Whitney test for continuous variables. Comparisons between paired observations were made with Wilcoxon signed-rank test. To explore the correlation of inflammatory biomarkers with cardiac biomarkers and CMR findings, ordinal regression models were fit in which the dependent variable was the cardiac biomarkers or CMR findings, and the independent variable (predictor) was C-reactive protein (CRP). A p value <0.05 was considered statistically significant. Statistical analyses were performed with STATA software version 16.1 (StataCorp LLC, College Station, Texas) and R software version 4.02 (R Foundation for Statistical Computing, Vienna, Austria).
A total of 330 patients with evidence of previous SARS-CoV-2 infection were referred for CMR evaluation, including 30 patients with MIS (6 MIS-A and 24 MIS-C: age 5 to 20 years). A subset of pediatric patients (age <16 years) was imaged on a 3.0 T platform (n = 29, including 19 MIS-C cases), whereas all other patients were imaged at 1.5 T (n = 301). Forty patients had CMR features diagnostic for myocarditis: 21 patients with MIS and 19 patients without MIS. The rate of myocarditis was significantly higher in patients with MIS (70%) than without (6.3%, p <0.001).
We further analyzed clinical data from patients with COVID-19-related myocarditis. The median age was 20 years and 65% were male. The prevalence of medical co-morbidities was low. (Table 1 ) The time interval between COVID-19 diagnosis and CMR study was 36 (IQR 20 to 60) days and was comparable between MIS and non-MIS. At the time of CMR, all patients had recovered from the acute phase and were stable without the need for respiratory or hemodynamic support. During the acute phase of COVID-19, patients in the MIS group were more likely to require intensive care, to undergo treatment with corticosteroids and immunoglobins, and to exhibit troponin elevation above the 99% of upper reference limit. CRP and B-type natriuretic peptide were significantly elevated acutely in patients in the MIS group and were largely not tested in the non-MIS group (Table 1). Two patients from the MIS group had new-onset atrial fibrillation. During the convalescent phase, persistent cardiac symptoms were present in a subset of patients in both groups (29% vs 58%, MIS vs non-MIS, p = 0.11). At the time of CMR, patients in the MIS group had a significantly higher heart rate and lower systolic blood pressure (Table 1).
Table 1.
Baseline clinical characteristics
| MIS myocarditis(n = 21) | Non-MIS myocarditis(n = 19) | p Value | |
|---|---|---|---|
| Age (years) | 14 (8-20) | 24 (20-50) | <0.001 |
| Male | 15 (71%) | 11 (58%) | 0.51 |
| Heart rate (beats/minute) | 101 (83-116) | 76 (71-81) | <0.001 |
| Systolic blood pressure (mm Hg) | 111 (102-123) | 118 (115-133) | 0.024 |
| Diastolic blood pressure (mm Hg) | 71 (60-78) | 77 (68-84) | 0.11 |
| Diagnosis-to-CMR duration (day) | 35 (22-48) | 42 (15-135) | 0.24 |
| Hypertension | 1 (5%) | 2 (11%) | |
| Diabetes | 1 (5%) | 1 (5%) | |
| Coronary artery disease | 0 | 0 | |
| Chronic lung disease/asthma | 0 | 0 | |
| Cerebral vascular accident | 1 (5%) | 1 (5%) | |
| Chronic kidney disease | 1 (5%) | 0 | |
| Chronic liver disease | 0 | 0 | |
| Structural heart disease | 0 | 4 (21%) | |
| Diagnosis by PCR | 8 (38%) | 16 (84%) | |
| Diagnosis by antibody | 13 (62%) | 3 (16%) | |
| COVID-19 acute symptoms | 20 (95%) | 17 (89%) | 0.6 |
| Cardiac symptoms | 6 (29%) | 11 (58%) | 0.11 |
| Chest pain | 3 (14%) | 7 (37%) | 0.15 |
| Dyspnea | 1 (5%) | 5 (26%) | 0.08 |
| Palpitations | 2 (10%) | 3 (16%) | 0.65 |
| Presyncope/syncope | 0 | 1 (5%) | 0.47 |
| Hospitalization | 20 (95%) | 5 (26%) | <0.001 |
| None (Home recovery) | 1 (5%) | 14 (74%) | |
| Floor admission | 7 (33%) | 3 (16%) | |
| Intensive care | 13 (62%) | 2 (11%) | |
| Treatment: corticosteroids | 18 (86%) | 1 (5%) | |
| Treatment: immunoglobulin | 18 (86%) | 0 | |
| Treatment: remdesivir | 1 (5%) | 1 (5%) | |
| Respiratory support | 7 (33%) | 2 (11%) | 0.27 |
| Nasal cannula oxygen | 5 (24%) | 2 (11%) | |
| Non-invasive ventilation | 1 (5%) | 0 | |
| Mechanical ventilation | 1 (5%) | 0 | |
| Hemodynamic support | 6 (29%) | 0 | 0.032 |
| Pressor | 5 (24%) | 0 | |
| Mechanical circulatory support | 1 (5%) | 0 | |
| Troponin I* | 0.3 (0.04–1.6) | 0.01 (0.005–0.02) | 0.016 |
| Troponin elevation* | 16 (76.2%) | 2 (16.7%) | 0.001 |
| CRP† | 216 (87.6–325) | ||
| BNP† | 1,113 (460.8–1,927) | ||
| Abnormal ECG‡ | 12 (57%) | 5 (29%) | 0.11 |
*Troponin laboratory values were available in 12 of 19 patients in group B.
There is significant missing data of BNP and CRP in the non-MIS myocarditis group and therefore not listed.
ECG data was available in 18 of 19 patients in the non-MIS group.
BNP = B-type natriuretic peptide; BSA = body surface area; ECG = electrocardiogram; ECV = extracellular volume; HR = heart rate; LGE = late gadolinium enhancement.
p <0.05 is considered significant.
Of the 40 patients with CMR findings of myocarditis, 13 pediatric patients (age 5 to 15 years) were imaged at 3.0 T, whereas 27 patients (age 16 to 76 years) were imaged using a 1.5-T magnet. CMR data are listed in Table 2 . Patients in the MIS group had lower left ventricular ejection fraction (median 55% vs 60%, MIS vs non-MIS, p = 0.04) and stroke volume index. Cardiac index was not different between the 2 groups, because of the higher heart rate in the MIS group. Of the 40 patients, 37 (21 from MIS and 16 from non-MIS) had CMR findings diagnostic of acute myocarditis according to the modified Lake Louise criteria, whereas 3 patients (from the non-MIS group) demonstrated a subepicardial LGE pattern without active myocardial edema, suggestive of healed myocarditis.
Table 2.
CMR findings
| Myocarditis |
|||
|---|---|---|---|
| MIS(n = 21) | Non-MIS(n = 19) | p Value | |
| LVEF (%) | 55 (51-60) | 60 (58-62) | 0.04 |
| LVEDVi (ml/m2) | 78 (66-88) | 84 (82-103) | 0.07 |
| LVESVi (ml/m2) | 33 (28-38) | 32 (24-45) | 0.80 |
| LV mass/BSA (g/m2) | 55 (50-57) | 64 (47-73) | 0.14 |
| RVEF (%) | 55 (50-57) | 51 (46-54) | 0.14 |
| RVEDVi (ml/m2) | 78 (69-88) | 92 (84-119) | 0.03 |
| RVESVi (ml/m2) | 37 (30-40) | 48 (35-57) | 0.02 |
| SV index (ml/m2) | 38 (34-49) | 50 (47-62) | 0.04 |
| Cardiac index (L/min/m2) | 4.0 (3.2-4.5) | 3.4 (3.0-3.9) | 0.13 |
| Global T1 Z-score | 3.3 (2.2-5.9) | 1.5 (0.4-3.6) | 0.02 |
| Global T2 Z-score | 2.1 (0.2-3.8) | 1.9 (0.8-2.6) | 0.78 |
| Global ECV (%) | 33 (28-35) | 31 (27-33) | 0.73 |
| LGE burden (%)* | 5.9 (3.1-11.6) | 6.6 (3.8-8.0) | 0.83 |
Two participants in the MIS group were not administered gadolinium because of acute kidney injury.
BSA = body surface index; LVEDVi = left ventricular end-diastolic volume index; LVEF = left ventricular ejection fraction; LVESVi = left ventricular end-systolic volume index; RVEDVi = right ventricular end-diastolic volume index; RVEF = right ventricular ejection fraction; RVESVi = right ventricular end-systolic volume index; SV = stroke volume index.
p <0.05 is considered significant.
Global myocardial inflammation and edema were noted in all patients with MIS myocarditis, whereas patients in the non-MIS group had regional inflammation and edema (Figures 1 and 2) . Native T1 and T2 times were converted to Z-scores to facilitate the comparison of studies from 1.5 and 3.0-T scanners. The global myocardial T1 Z-score was significantly elevated in the MIS group, whereas myocardial T2 Z-scores were comparable between the 2 groups (Table 2). We explored the association between the acute inflammatory marker CRP and myocardial involvement in patients with MIS myocarditis. Although CRP was associated with B-type natriuretic peptide (p = 0.004) during the acute phase, CRP was not associated with CMR findings, including left ventricular ejection fraction (p = 0.70), global T1 (p = 0.59), global T2 (p = 0.76), or LGE (p = 0.08).
Figure 1.
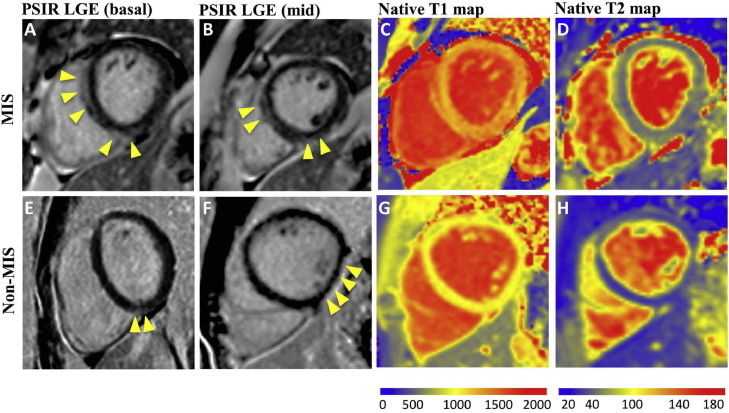
CMR features of COVID-19-related myocarditis (1.5-T platform). A 25-year-old man presented with MIS-A myocarditis and had elevated troponin I (peaked at 9 ng/ml) and non-specific ST-T changes. CMR showed septal and inferior subepicardial LGE (A, B) and global elevation of T1 (1,245 ms, Z-score 15.6) (C) and T2 values (63 ms, Z-score 11.2) (D). A 24-year-old man with non-MIS myocarditis had dyspnea on exertion after COVID-19. CMR showed basal inferoseptal and mid-inferolateral LGE (E, F), normal global myocardial T1 (968 ms, Z-score 0.2), (G) and mild regional T2 elevation (51 ms, Z-score 3.8) at the location of LGE (H). Yellow arrows point to the locations of LGE. The normal ranges of T1 and T2 were 930 to 1,010 ms, and <50 ms, respectively. The normal range of Z-score was −2 to 2.
Figure 2.
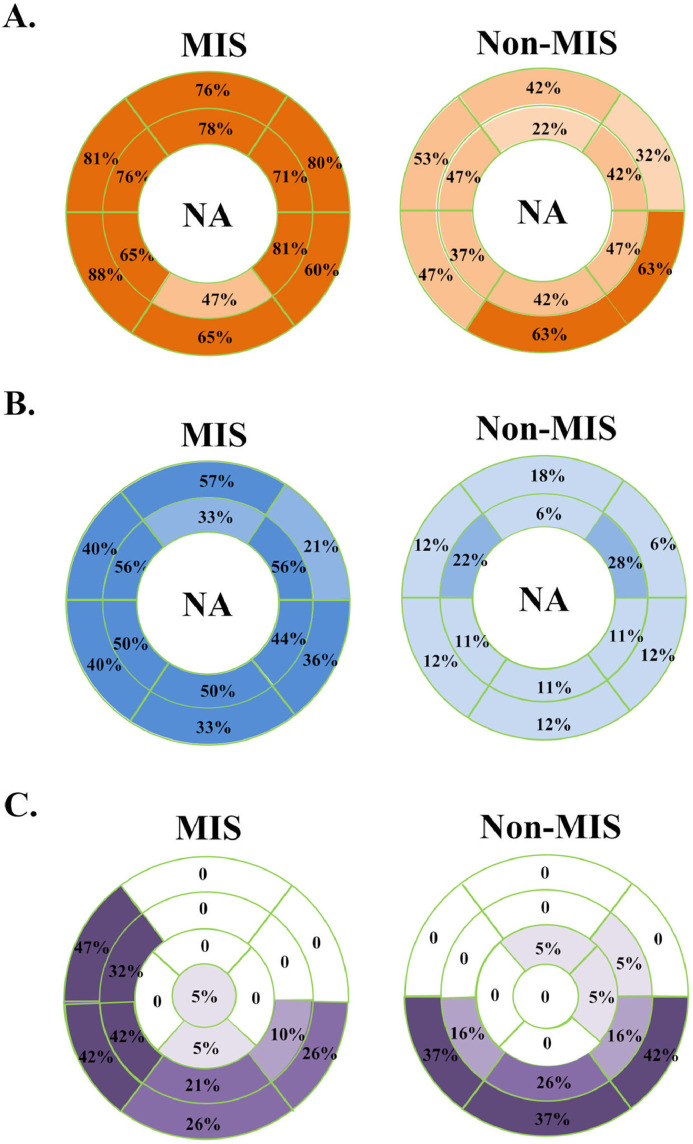
Myocardial segments demonstrating T1 elevation, T2 elevation, and LGE in the AHA 17-segment model. (A) The rate of myocardial T1 elevation by segment, in MIS and non-MIS patients with myocarditis. (B) The rate of myocardial T2 elevation by segment, in MIS and non-MIS patients with myocarditis. (C) Distribution of LGE in the MIS and non-MIS patients with myocarditis. The rate of occurrence within each segment was marked.
The LGE burden was similar in both groups (5.9% vs 6.6%, MIS vs non-MIS, p = 0.83). However, the 2 groups exhibited different LGE distributions: the MIS group had LGE frequently located at the septum, whereas the non-MIS group often had LGE in the inferior half of the myocardium (Figure 2). Of note, 31 of the 49 segments (63.3%) with LGE in the MIS myocarditis group were either anteroseptal or inferoseptal segments; in contrast, 10 of the 36 segments (27.8%) with LGE in the non-MIS myocarditis group were septal segments (p = 0.001).
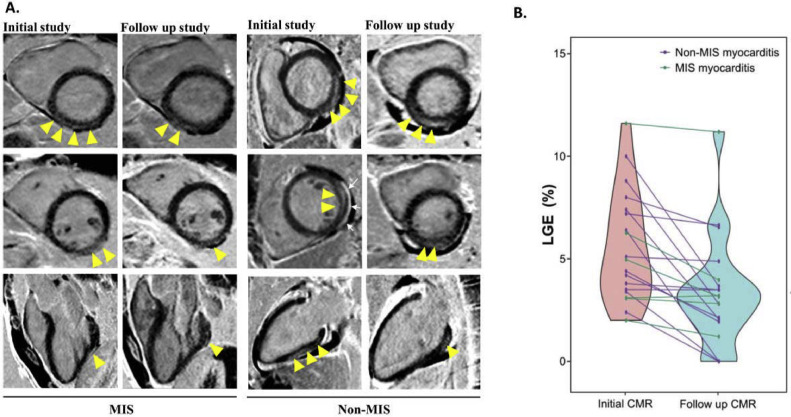
Of the 40 patients with myocarditis, 20 patients had a follow-up CMR (7 from the MIS group and 13 from the non-MIS group) at a median follow-up of 102 (IQR 85 to 150) days after the first CMR. One case from the MIS group was performed without gadolinium because of the estimated glomerular filtration rate <30 ml/min/1.73 m2. Five patients (25%, 3 from MIS, and 2 from the non-MIS group) continued to demonstrate regional myocardial inflammation/edema (T1 and T2 elevations), and 1 of them (from the MIS group) had persistent dyspnea on exertion; all other patients had complete resolution of myocardial inflammation/edema. The LGE burden improved over time, from a median of 5.0% (IQR 3.4% to 7.3%) to 3.2% (IQR 2.0% to 3.8%) (p <0.001), at a variable rate (Figure 3 ).
Figure 3.
(A) Follow-up CMR imaging showing reduction of myocardial LGE in both MIS and non-MIS patients. Arrowheads point to the locations of LGE. White arrows point to the pericardial effusion. (B) Resolution of myocardial LGE in follow-up CMR in patients with MIS and non-MIS myocarditis.
This study demonstrates distinct differences in myocardial tissue characterization between MIS myocarditis and non-MIS myocarditis after COVID-19. This suggests divergent underlying pathogenesis of myocarditis and adds to our understanding of complications. MIS myocarditis is characterized by global myocardial inflammation with more significant elevations in T1, compared with regional involvement in non-MIS myocarditis. Furthermore, distinct LGE localizations were found in MIS and non-MIS myocarditis, despite a comparable LGE burden. Patients with MIS were found to have a septal LGE distribution, a finding previously reported as prognostic of worse long-term outcomes.18 Although this study was not set up to evaluate the incidence of myocarditis, our observations agree with a recent study showing that MIS patients were more likely to have cardiac involvement compared with severe acute COVID-19.5
The mechanism of non-MIS COVID-19–related myocarditis is unclear and likely multifactorial. SARS-CoV-2 has been detected in heart tissue,19 , 20 with the possibility of direct myocardial invasion. Endomyocardial biopsies in patients with severe myocarditis have also shown lymphocytic infiltration without focal necrosis or structural disruption, suggesting a critical role of hyperinflammation.11 In constrast, MIS is recognized as secondary to cytokine dysregulation in response to SARS-CoV-2 infection and is associated with a high incidence of myocardial dysfunction and circulatory shock. The CMR findings from our study support the important role of a global hyperinflammatory milieu in MIS-related myocarditis. Non-MIS COVID-19 myocarditis, in contrast, commonly demonstrates regional involvement similar to other viral myocarditis, but with a unique predilection for the inferior wall. The distinct parametric mapping and LGE patterns between the 2 groups merit consideration of different underlying pathophysiology of MIS versus non-MIS myocarditis.
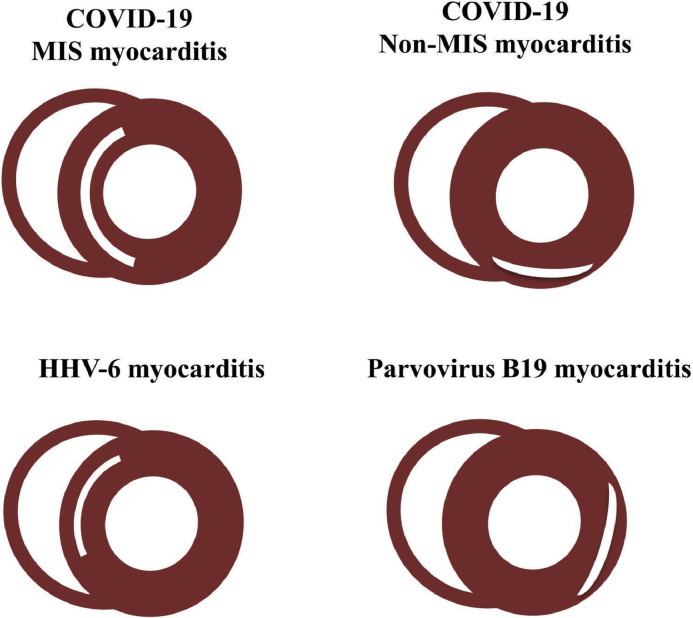
The long-term prognosis after COVID-19-related myocarditis remains to be determined. Both the presence and localization of LGE are known major predictors of long-term cardiovascular events in patients with acute viral myocarditis.21 Different patterns of LGE localization have been shown in acute viral myocarditis: left ventricular inferior and lateral wall LGE is commonly seen in parvovirus B19 myocarditis, whereas septal LGE is more frequently observed in human herpes virus-6 myocarditis.22 Myocarditis with septal LGE is reported to have a worse prognosis.18 Recently, Huang et al23 showed a predilection of LGE localized to the basal-to-mid inferior wall in 15 patients with COVID-19 (non-MIS) myocarditis. By comparison, we demonstrate different LGE patterns of MIS and non-MIS myocarditis: the anteroseptal and inferoseptal segments were most frequently involved in MIS myocarditis, whereas inferoseptal, inferior, and inferolateral LGE was commonly observed in non-MIS myocarditis (Figure 4 ). The reason for differential segmental LGE predilections and prognostic implications remains unclear, although multiple physiological perturbations have been described because of COVID-19, including ongoing inflammation, endothelial dysfunction, and microthrombi.24 , 25 Whether the LGE presence and pattern forecast long-term cardiovascular outcomes in MIS versus non-MIS myocarditis warrants further study.
Figure 4.
Comparison of LGE localization patterns in MIS myocarditis, non-MIS myocarditis, and common viral myocarditis including HHV-6 and parvovirus B16 myocarditis.
Rapid and continuous recovery was previously reported in acute viral myocarditis, with largely resolved myocardial inflammation/edema after 8 weeks.26 In our study, sequential CMR in 20 patients demonstrated myocardial recovery at a median follow-up of 102 days since the first CMR, with an overall improvement of LGE burden with individual variations. Future investigation of both MIS and non-MIS myocarditis is needed to elucidate the evolution of COVID-19-related myocarditis and its clinical significance.
Our study has several limitations. First, it is a single-center study, with selection bias. Most of the patients in the study cohort were healthy at baseline and few non-MIS patients suffered severe COVID-19. Furthermore, many patients were referred based on clinical suspicion for myocarditis. Second, there are age disparities between the groups because of the inclusion of MIS-C patients in the MIS group. Third, there was a bias toward obtaining CMR in patients with a clinical suspicion for myocarditis. MIS, particularly MIS-A, may be under-reported in the population and more likely to be diagnosed in critically ill patients with troponin elevation, as was common in our MIS cohort.8 Fourth, delay of CMR to the convalescent phase—because of local COVID-19 protocols to limit exposure to actively infected individuals—may have led to an underestimation of myocardial injury. Finally, limited follow-up CMR data are currently available to study myocardial recovery in patients with MIS myocarditis.
In conclusion, the CMR features of COVID-19–related myocarditis differ in patients with and without MIS. In patients with myocarditis after COVID-19, MIS is associated with both an increased degree of myocardial inflammation and more global involvement relative to patients without MIS. LGE in non-MIS COVID-19 myocarditis is frequently located at the inferior myocardial segments, whereas septal involvement is more common in MIS myocarditis. Recovery is noted in all forms of COVID-19 myocarditis, albeit residual LGE is common at a median follow-up of 102 days after the diagnosis of myocarditis by CMR.
Disclosures
The authors have no conflicts of interest to declare.
Footnotes
This work was supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health under award number T32HL007411. The publication described was supported by Clinical and Translational Sciences Award (CTSA) award number UL1 TR002243 from the National Center for Advancing Translational Sciences. Its contents are solely the responsibility of the authors and do not necessarily represent official views of the National Center for Advancing Translational Sciences or the National Institutes of Health.
References
- 1.Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dong E, Du H, Gardner L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect Dis. 2020;20:533–534. doi: 10.1016/S1473-3099(20)30120-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guo T, Fan Y, Chen M, Wu X, Zhang L, He T, Wang H, Wan J, Wang X, Lu Z. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5:811–818. doi: 10.1001/jamacardio.2020.1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shi S, Qin M, Shen B, Cai Y, Liu T, Yang F, Gong W, Liu X, Liang J, Zhao Q, Huang H, Yang B, Huang C. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020;5:802–810. doi: 10.1001/jamacardio.2020.0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Feldstein LR, Tenforde MW, Friedman KG, Newhams M, Rose EB, Dapul H, Soma VL, Maddux AB, Mourani PM, Bowens C, Maamari M, Hall MW, Riggs BJ, Giuliano JS, Jr, Singh AR, Li S, Kong M, Schuster JE, McLaughlin GE, Schwartz SP, Walker TC, Loftis LL, Hobbs CV, Halasa NB, Doymaz S, Babbitt CJ, Hume JR, Gertz SJ, Irby K, Clouser KN, Cvijanovich NZ, Bradford TT, Smith LS, Heidemann SM, Zackai SP, Wellnitz K, Nofziger RA, Horwitz SM, Carroll RW, Rowan CM, Tarquinio KM, Mack EH, Fitzgerald JC, Coates BM, Jackson AM, Young CC, Son MBF, Patel MM, Newburger JW, Randolph AG. Overcoming COVID-19 Investigators. Characteristics and outcomes of US children and adolescents with multisystem inflammatory syndrome in children (MIS-C) compared with severe acute COVID-19. JAMA. 2021;325:1074–1087. doi: 10.1001/jama.2021.2091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Morris SB, Schwartz NG, Patel P, Abbo L, Beauchamps L, Balan S, Lee EH, Paneth-Pollak R, Geevarughese A, Lash MK, Dorsinville MS, Ballen V, Eiras DP, Newton-Cheh C, Smith E, Robinson S, Stogsdill P, Lim S, Fox SE, Richardson G, Hand J, Oliver NT, Kofman A, Bryant B, Ende Z, Datta D, Belay E. Godfred-Cato S. Case series of multisystem inflammatory syndrome in adults associated with SARS-CoV-2 infection - United Kingdom and United States, March-August 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1450–1456. doi: 10.15585/mmwr.mm6940e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rubens JH, Akindele NP, Tschudy MM. Sick-Samuels AC. Acute covid-19 and multisystem inflammatory syndrome in children. BMJ. 2021;372:n385. doi: 10.1136/bmj.n385. [DOI] [PubMed] [Google Scholar]
- 8.Davogustto GE, Clark DE, Hardison E, Yanis AH, Lowery BD, Halasa NB, Wells QS. Characteristics associated with multisystem inflammatory syndrome in adults with SARS-CoV-2 infection. JAMA Netw Open. 2021;4 doi: 10.1001/jamanetworkopen.2021.10323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Belhadjer Z, Méot M, Bajolle F, Khraiche D, Legendre A, Abakka S, Auriau J, Grimaud M, Oualha M, Beghetti M, Wacker J, Ovaert C, Hascoet S, Selegny M, Malekzadeh-Milani S, Maltret A, Bosser G, Giroux N, Bonnemains L, Bordet J, Di Filippo S, Mauran P, Falcon-Eicher S, Thambo JB, Lefort B, Moceri P, Houyel L, Renolleau S, Bonnet D. Acute heart failure in multisystem inflammatory syndrome in children in the context of global SARS-CoV-2 pandemic. Circulation. 2020;142:429–436. doi: 10.1161/CIRCULATIONAHA.120.048360. [DOI] [PubMed] [Google Scholar]
- 10.Clark DE, Parikh A, Dendy JM, Diamond AB, George-Durrett K, Fish FA, Slaughter JC, Fitch W, Hughes SG, Soslow JH. COVID-19 myocardial pathology evaluation in athletes with cardiac magnetic resonance (COMPETE CMR) Circulation. 2021;143:609–612. doi: 10.1161/CIRCULATIONAHA.120.052573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Puntmann VO, Carerj ML, Wieters I, Fahim M, Arendt C, Hoffmann J, Shchendrygina A, Escher F, Vasa-Nicotera M, Zeiher AM, Vehreschild M, Nagel E. Outcomes of cardiovascular magnetic resonance imaging in patients recently recovered from coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5:1265–1273. doi: 10.1001/jamacardio.2020.3557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rajpal S, Tong MS, Borchers J, Zareba KM, Obarski TP, Simonetti OP, Daniels CJ. Cardiovascular magnetic resonance findings in competitive athletes recovering from COVID-19 infection. JAMA Cardiol. 2021;6:116–118. doi: 10.1001/jamacardio.2020.4916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Valverde I, Singh Y, Sanchez-de-Toledo J, Theocharis P, Chikermane A, Di Filippo S, Kuciñska B, Mannarino S, Tamariz-Martel A, Gutierrez-Larraya F, Soda G, Vandekerckhove K, Gonzalez-Barlatay F, McMahon CJ, Marcora S, Napoleone CP, Duong P, Tuo G, Deri A, Nepali G, Ilina M, Ciliberti P, Miller O. AEPC COVID-19 Rapid Response Team*. Acute cardiovascular manifestations in 286 children with multisystem inflammatory syndrome associated with COVID-19 infection in Europe. Circulation. 2021;143:21–32. doi: 10.1161/CIRCULATIONAHA.120.050065. [DOI] [PubMed] [Google Scholar]
- 14.Pollack A, Kontorovich AR, Fuster V, Dec GW. Viral myocarditis–diagnosis, treatment options, and current controversies. Nat Rev Cardiol. 2015;12:670–680. doi: 10.1038/nrcardio.2015.108. [DOI] [PubMed] [Google Scholar]
- 15.Ferreira VM, Schulz-Menger J, Holmvang G, Kramer CM, Carbone I, Sechtem U, Kindermann I, Gutberlet M, Cooper LT, Liu P, Friedrich MG. Cardiovascular magnetic resonance in nonischemic myocardial inflammation: expert recommendations. J Am Coll Cardiol. 2018;72:3158–3176. doi: 10.1016/j.jacc.2018.09.072. [DOI] [PubMed] [Google Scholar]
- 16.Moon JC, Messroghli DR, Kellman P, Piechnik SK, Robson MD, Ugander M, Gatehouse PD, Arai AE, Friedrich MG, Neubauer S, Schulz-Menger J, Schelbert EB. Society for Cardiovascular Magnetic Resonance Imaging, Cardiovascular Magnetic Resonance Working Group of the European Society of Cardiology. Myocardial T1 mapping and extracellular volume quantification: a Society for Cardiovascular Magnetic Resonance (SCMR) and CMR Working Group of the European Society of Cardiology consensus statement. J Cardiovasc Magn Reson. 2013;15:92. doi: 10.1186/1532-429X-15-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gräni C, Eichhorn C, Bière L, Kaneko K, Murthy VL, Agarwal V, Aghayev A, Steigner M, Blankstein R, Jerosch-Herold M, Kwong RY. Comparison of myocardial fibrosis quantification methods by cardiovascular magnetic resonance imaging for risk stratification of patients with suspected myocarditis. J Cardiovasc Magn Reson. 2019;21:14. doi: 10.1186/s12968-019-0520-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Aquaro GD, Perfetti M, Camastra G, Monti L, Dellegrottaglie S, Moro C, Pepe A, Todiere G, Lanzillo C, Scatteia A, Di Roma M, Pontone G, Perazzolo Marra M, Barison A, Di Bella G. Cardiac Magnetic Resonance Working Group of the Italian Society of Cardiology. Cardiac MR with late gadolinium enhancement in acute myocarditis with preserved systolic function: ITAMY study. J Am Coll Cardiol. 2017;70:1977–1987. doi: 10.1016/j.jacc.2017.08.044. [DOI] [PubMed] [Google Scholar]
- 19.Wenzel P, Kopp S, Göbel S, Jansen T, Geyer M, Hahn F, Kreitner KF, Escher F, Schultheiss HP, Münzel T. Evidence of SARS-CoV-2 mRNA in endomyocardial biopsies of patients with clinically suspected myocarditis tested negative for COVID-19 in nasopharyngeal swab. Cardiovasc Res. 2020;116:1661–1663. doi: 10.1093/cvr/cvaa160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tavazzi G, Pellegrini C, Maurelli M, Belliato M, Sciutti F, Bottazzi A, Sepe PA, Resasco T, Camporotondo R, Bruno R, Baldanti F, Paolucci S, Pelenghi S, Iotti GA, Mojoli F, Arbustini E. Myocardial localization of coronavirus in COVID-19 cardiogenic shock. Eur J Heart Fail. 2020;22:911–915. doi: 10.1002/ejhf.1828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Grün S, Schumm J, Greulich S, Wagner A, Schneider S, Bruder O, Kispert EM, Hill S, Ong P, Klingel K, Kandolf R, Sechtem U, Mahrholdt H. Long-term follow-up of biopsy-proven viral myocarditis: predictors of mortality and incomplete recovery. J Am Coll Cardiol. 2012;59:1604–1615. doi: 10.1016/j.jacc.2012.01.007. [DOI] [PubMed] [Google Scholar]
- 22.Mahrholdt H, Wagner A, Deluigi CC, Kispert E, Hager S, Meinhardt G, Vogelsberg H, Fritz P, Dippon J, Bock CT, Klingel K, Kandolf R, Sechtem U. Presentation, patterns of myocardial damage, and clinical course of viral myocarditis. Circulation. 2006;114:1581–1590. doi: 10.1161/CIRCULATIONAHA.105.606509. [DOI] [PubMed] [Google Scholar]
- 23.Huang L, Zhao P, Tang D, Zhu T, Han R, Zhan C, Liu W, Zeng H, Tao Q, Xia L. Cardiac involvement in patients recovered from COVID-2019 identified using magnetic resonance imaging. JACC Cardiovasc Imaging. 2020;13:2330–2339. doi: 10.1016/j.jcmg.2020.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Evans PC, Rainger GE, Mason JC, Guzik TJ, Osto E, Stamataki Z, Neil D, Hoefer IE, Fragiadaki M, Waltenberger J, Weber C, Bochaton-Piallat ML, Bäck M. Endothelial dysfunction in COVID-19: a position paper of the ESC Working Group for Atherosclerosis and Vascular Biology, and the ESC Council of Basic Cardiovascular Science. Cardiovasc Res. 2020;116:2177–2184. doi: 10.1093/cvr/cvaa230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pellegrini D, Kawakami R, Guagliumi G, Sakamoto A, Kawai K, Gianatti A, Nasr A, Kutys R, Guo L, Cornelissen A, Faggi L, Mori M, Sato Y, Pescetelli I, Brivio M, Romero M, Virmani R, Finn AV. Microthrombi as a major cause of cardiac injury in COVID-19: a pathologic study. Circulation. 2021;143:1031–1042. doi: 10.1161/CIRCULATIONAHA.120.051828. [DOI] [PubMed] [Google Scholar]
- 26.Luetkens JA, Homsi R, Dabir D, Kuetting DL, Marx C, Doerner J, Schlesinger-Irsch U, Andrié R, Sprinkart AM, Schmeel FC, Stehning C, Fimmers R, Gieseke J, Naehle CP, Schild HH, Thomas DK. Comprehensive cardiac magnetic resonance for short-term follow-up in acute myocarditis. J Am Heart Assoc. 2016;5 doi: 10.1161/JAHA.116.003603. [DOI] [PMC free article] [PubMed] [Google Scholar]