Abstract
A 60-year-old woman, who experienced progressive right visual loss, was diagnosed with an unruptured large cerebral aneurysm. Magnetic resonance imaging (MRI) and angiography revealed a large partially thrombosed anterior communicating artery (Acom) aneurysm. The aneurysmal neck was located at the junction of the left A1–A2 segments, and the aneurysmal dome communicated with the right A1–A2 junction by the Acom. Endovascular treatment using the flow alteration technique was selected. Following an oral antiplatelet therapy for 9 days, balloon test occlusion (BTO) of the medial portion of the left A1 segment was performed under local anesthesia. After confirming the tolerance of the BTO, internal trapping of the medial portion of the left A1 segment by detachable coils was performed following intra-aneurysmal coil embolization. Oral antiplatelet treatment was continued for 19 days postoperatively. Within 3 months following the operation, her right visual acuity dramatically improved to the original level. Owing to aneurysmal recanalization and the disappearance of the thrombus, the second and third embolization was performed through the Acom route, 4 months and 3 years following the first embolization, respectively, and followed up for an additional 7 years by MRI; no deterioration of her visual acuity and no aneurysmal recanalization was observed. Thus, endosaccular embolization combined with flow alteration is considered a useful alternative treatment for large and partially thrombosed Acom aneurysms.
Keywords: flow alteration, visual acuity, visual field, balloon test occlusion, antiplatelet therapy
Introduction
Although unruptured anterior communicating artery (Acom) aneurysms, especially those manifesting in a way that compresses the optic nerves, are rare (1–2%), large Acom aneurysms are frequently reported to compress the optic nerves.1–3) Partially thrombosed large Acom aneurysms are reportedly difficult to treat by direct open surgery and endovascular treatment. Direct open surgery often requires thrombectomy, aneurysmal reconstruction, and bypass surgery.4) Simple endosaccular embolization of partially thrombosed aneurysms frequently results in recanalization.5,6) On the other hand, treatment utilizing flow alteration by parent-artery occlusion and bypass surgeries has been reported for partially thrombosed large aneurysms at various locations.7–10) We presumed that the application of flow alteration to partially thrombosed large Acom aneurysms by endovascular procedures would be effective.
Herein, we report a case of a partially thrombosed large Acom aneurysm treated using flow alteration by internal trapping of one of the A1 segments, resulting in the recovery of visual loss and the long-term prevention of aneurysmal recanalization.
Case Report
A 60-year-old woman experienced blurry vision of her right eye 3 months prior to her referral to our hospital. She visited an eye clinic 1 month prior, and her visual acuity in both the eyes was normal with 1.2. She complained of progressive visual loss of her right eye and was diagnosed with right visual acuity of 0.1 without any abnormality of her lens, vitreous body, or retina, 2 weeks prior to her referral to our hospital. She underwent magnetic resonance imaging (MRI) and was diagnosed with an unruptured large aneurysm.
On admission, she had no history of hypertension or other diseases, and her routine preoperative evaluation results were within normal ranges, including blood coagulation data.
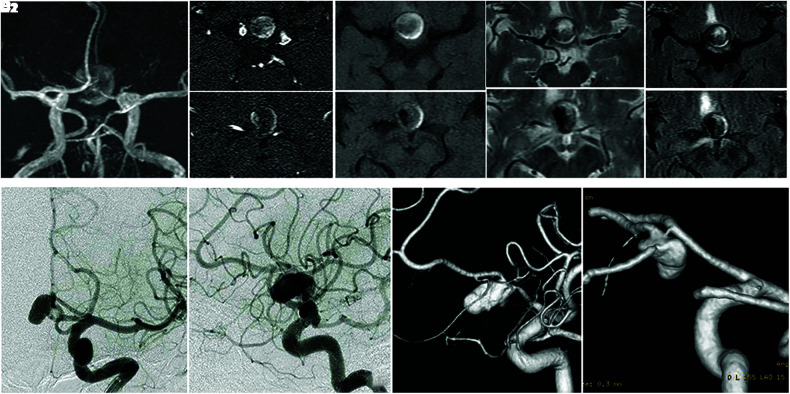
The MRI scans (Fig. 1A–1E) revealed a round mass, indicating a patent cavity on the right side surrounded by an intra-aneurysmal thrombus.11,12) An angiogram revealed a large Acom aneurysm (maximum diameter, 19 mm; Fig. 1F and 1G). The aneurysmal neck was located at the junction of the left A1–A2 segments, and the aneurysmal dome was communicated with the right A1–A2 junction by the Acom (Fig. 1H and 1I).
Fig. 1. (A–E) The MRI scans 20 days prior to the first coil embolization indicate a signal void on the right side of the mass (A, B1, B2; TOF-MRA), surrounded by iso-intensity of the center and high-intensity peripherally of the T1-weighted images (C1, C2), heterogeneity intensity of the T2-weighted images (D1, D2), and fluid-attenuated inversion recovery (E1, E2). (F, G) Right CAG performed 15 days prior to the first coil embolization. (F) anteroposterior view, (G) left lateral view, (H) left CAG (3D DSA), left anterior-oblique view, (I) Right CAG (3D DSA), left posterior-oblique view. Acom: anterior communicating artery, CAG: carotid angiography, MRI: magnetic resonance imaging, TOF-MRA: time-of-flight magnetic resonance angiography, 3D DSA: three-dimensional digital subtraction angiography.
The patient preferred to receive endovascular treatment at first and consented to open direct surgery if the first treatment failed. Preoperatively, oral clopidogrel (75 mg/day) was administered for 9 days, and the first endovascular treatment was performed under local anesthesia. Following femoral sheath insertion, systemic heparinization was initiated until an activated clotting time longer than twice that of pre-heparinization was achieved. A HyperForm occlusion balloon catheter (7 mm × 4 mm, Medtronic, Irvine, CA, USA) was introduced to the medial portion of the left A1 segment, and balloon test occlusion (BTO) was performed for 15 min. We confirmed the absence of motor-sensory or verbal dysfunction, and the perfusion of the left anterior cerebral artery through the Acom route by the right carotid angiogram during BTO, which also confirmed the absence of a perforator originating from the medial portion of the left A1 segment.
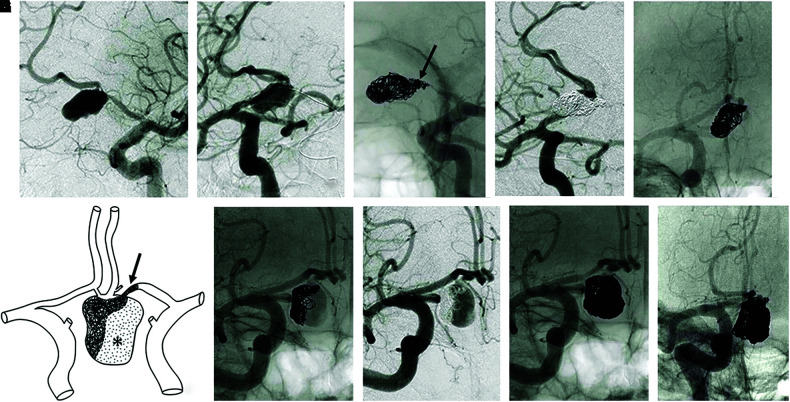
Subsequently, endosaccular embolization and internal trapping of the medial portion of the left A1 segment were performed using Matrix2 Detachable Coils (total length, 75 cm; Stryker, Fremont, CA, USA) and GDC Detachable Coils (total length, 30 cm; Stryker, Fremont, CA, USA) (Fig. 2A–2E). Figure 2F shows the image of the first embolization. Oral clopidogrel (75 mg/day) was continued for 19 days postoperatively.
Fig. 2. (A–E) CAGs showing the first endovascular treatment. (A, C) Left CAG; (B, D, E) Right CAG. (B) Balloon test occlusion of the left A1 segment, (C) endosaccular embolization and the internal trapping of the medial portion of the left A1 segment (black arrow). (D, E) Post-first embolization. (A–D), left-anterior oblique view; (E) antero-posterior view. (F) Illustration of the image of the first embolization. Black arrow, internal trapping of the medial portion of the left A1 segment; *, intra-aneurysmal thrombus. (G–J) Right CAGs 4 months after the first treatment. G (non-subtracted) and H (subtracted) revealed complete disappearance of the aneurysmal thrombus and remarkable recanalization. I and J show post-second embolization images. (G–I) Right anterior oblique view, (J) antero-posterior view. CAG: carotid angiography.
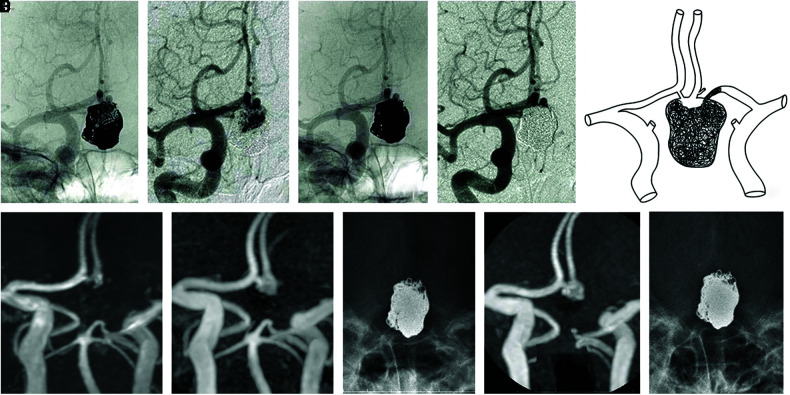
The patient’s right visual acuity dramatically improved from 0.1 to 0.4 at 18 days, 0.9 at 43 days, and 1.2 at 3 months following the embolization. However, coil transformation was detected, and the angiogram at 4 months following the first embolization revealed the complete disappearance of the intra-aneurysmal thrombus and marked aneurysmal recanalization (Fig. 2G and 2H). Subsequently, we performed a second coil embolization through the Acom route (Fig. 2I and 2J). We also performed a third embolization 3 years following the first embolization (Fig. 3A–3D). Figure 3E indicates the aneurysmal image following the last embolization. MRI (Fig. 3F, 3G, and 3I) and plane X-ray imaging (3H, 3J) were performed during the follow-up of 7 years following the last embolization. Although we observed gradual neck recanalization following the third embolization, we did not opt for an additional coiling owing to the small remnant space and good clinical signs, and the absence of recanalization progress for 3 years (Fig. 3F–3J).
Fig. 3. (A–D) Right CAGs showing the third embolization 3 years after the first embolization, antero-posterior view. A (non-subtracted) and B (subtracted) showing the aneurysmal recanalization. C (non-subtracted) and D (subtracted) showing the post-third embolization. (E) Illustration showing a recent image of the aneurysm. (F, G, I) TOF-MRA images of the next day (F), 4 years (G), and 7 years (I) following the third embolization. (H, J) Plane X-ray of 4 years (H) and 7 years (J) following the third embolization. CAG: carotid angiography, TOF-MRA: time-of-flight magnetic resonance angiography.
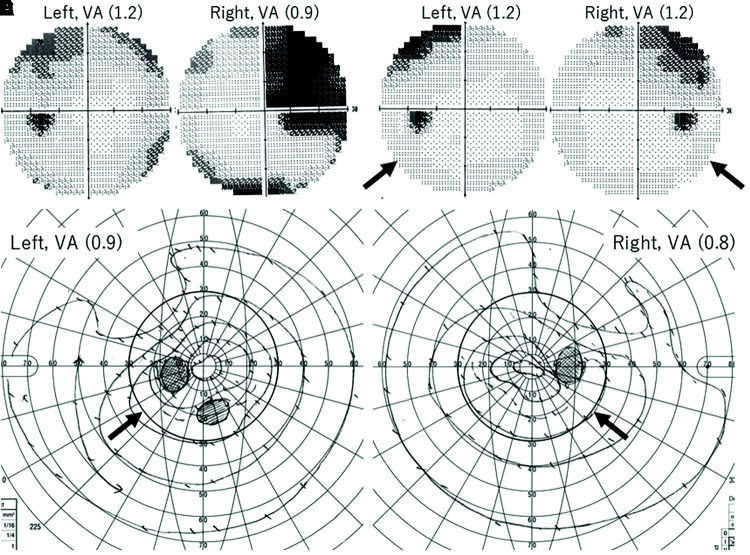
Figure 4 shows the time course of visual acuity and perimetry. Preoperative perimetry could not be performed owing to disturbed visual acuity. Figure 4A and 4B (Humphrey), at 43 days following the first embolization, indicates bitemporal hemianopsia predominantly in the right eye. Figure 4C and 4D (Humphrey), at 2 years, and 4E and 4F (Goldmann) at 10 years, indicates that favorable visual function results maintained the improved bitemporal hemianopsia in the range of 30 degree.
Fig. 4. (A–F) Perimetry charts of the Humphrey (A–D) and the Goldmann (E, F). (A, B) 1 month; (C, D) 2 years; (E, F) 10 years following the first embolization. Comparing between the perimetry C–D and E–F within the circle range of 30 degree (black arrows), the area of improved bitemporal hemianopsia was maintained for 8 years. VA: visual acuity.
Discussion
To the best of our knowledge, this report presents the first case of a partially thrombosed large Acom aneurysm successfully treated with flow alteration using an endovascular technique.
Direct clipping surgery of partially thrombosed large aneurysms to manage the vulnerable thrombus, remove the organized thrombus using ultrasonic aspirators, and construct in-situ anastomosis is technically demanding.4) The simple endovascular coil embolization of partially thrombosed aneurysms is considered to result in frequent recanalizations.5,6) Flow alteration treatments have reportedly been used to treat large and partially thrombosed anterior cerebral, internal carotid, and vertebra-basilar artery aneurysms using parent-artery occlusion and bypass surgery.7–10) Despite early recanalization being observed following the first embolization, our case suggested that flow alteration by internal trapping of the left A1 segment might decrease the inflow effect to the aneurysm resulting in successful obliteration by additional coil embolization.
Before parent-artery occlusion, it is crucial to completely understand the vascular anatomy and confirm the safety. The tolerance of the internal trapping was confirmed by BTO of the medial portion of the left A1 segment, where few perforators are reported to bifurcate.13)
The early disappearance of the intra-aneurysmal thrombus was an unexpected phenomenon. Although bioactive coils were used to prevent aneurysmal recanalization,14,15) marked recanalization occurred within 4 months. Therefore, the bioactive coils failed to play a role in the aneurysm. The mechanisms of thrombus resolution are thought to be the following: first, the age of the thrombus was premature and second, perioperative antiplatelet therapy protected further thrombus formation, followed by natural thrombolysis.
The MRI findings of the large Acom aneurysm in this patient revealed a characteristic signal void in the dome, indicating a partially thrombosed large aneurysm. The heterogeneous signal intensities of the thrombus mimic subacute intracerebral hematoma.16) However, the age of the intra-aneurysmal thrombus and the presence of thrombus organization are difficult to estimate.11,12) Since the thrombus disappeared within 4 months following the first embolization, we retrospectively assumed that this intra-aneurysmal thrombus did not contain an organized thrombus.
The mechanism of the optic nerve recovery is attributed to the decrease of the mass effect following the disappearance of intra-aneurysmal thrombus, and the reduction of the pulsatile blood flow, the so-called “water-hammer effect,”17) following flow alteration. Flow alteration by endovascular treatment could be a useful alternative to treat technically demanding large Acom aneurysms.
Acknowledgments
The authors thank Dr. Tomonori TAMURA (Eye Clinic) for his mindful follow-up and Ms. Yoko TONE for the illustrations.
Footnotes
Conflicts of Interest Disclosure
None of the authors have conflict of interest to declare.
References
- 1). Date I, Asari S, Ohmoto T: Cerebral aneurysms causing visual symptoms: their features and surgical outcome. Clin Neurol Neurosurg 100: 259– 267, 1998 [DOI] [PubMed] [Google Scholar]
- 2). Park JH, Park SK, Kim TH, Shin JJ, Shin HS, Hwang YS: Anterior communicating artery aneurysm related to visual symptoms. J Korean Neurosurg Soc 46: 232– 238, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3). Kesani M, Pelluru PK, Bhattacharjee S, Alugolu R, Purohit AK: Junctional chiasmatic syndrome due to large anterior communicating artery aneurysm. J Neurosci Rural Pract 8: 455– 457, 2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4). Lawton MT, Quiñones-Hinojosa A, Chang EF, Yu T: Thrombotic intracranial aneurysms: classification scheme and management strategies in 68 patients. Neurosurgery 56: 441– 454; discussion 441–454, 2005 [DOI] [PubMed] [Google Scholar]
- 5). Roccatagliata L, Guédin P, Condette-Auliac S, et al. : Partially thrombosed intracranial aneurysms: symptoms, evolution, and therapeutic management. Acta Neurochir (Wien) 152: 2133– 2142, 2010 [DOI] [PubMed] [Google Scholar]
- 6). Yang K, Park JC, Ahn JS, Kwon DH, Kwun BD, Kim CJ: Characteristics and outcomes of varied treatment modalities for partially thrombosed intracranial aneurysms: a review of 35 cases. Acta Neurochir (Wien) 156: 1669– 1675, 2014 [DOI] [PubMed] [Google Scholar]
- 7). Miyamoto S, Funaki T, Iihara K, Takahashi JC: Successful obliteration and shrinkage of giant partially thrombosed basilar artery aneurysms through a tailored flow reduction strategy with bypass surgery. J Neurosurg 114: 1028– 1036, 2011 [DOI] [PubMed] [Google Scholar]
- 8). Sato K, Yamada M, Abe K, Oka H, Kurata A, Fujii K: Tailored flow alteration treatment for intracranial internal carotid artery aneurysms: strategy beyond parent artery occlusion with bypass. Case report. Neurol Med Chir (Tokyo) 52: 213– 216, 2012 [DOI] [PubMed] [Google Scholar]
- 9). Abla AA, Lawton MT: Anterior cerebral artery bypass for complex aneurysms: an experience with intracranial-intracranial reconstruction and review of bypass options. J Neurosurg 120: 1364– 1377, 2014 [DOI] [PubMed] [Google Scholar]
- 10). Labib MA, Gandhi S, Cavallo C, et al. : Anterior cerebral artery bypass for complex aneurysms: advances in intracranial-intracranial bypass techniques. World Neurosurg 141: e42– e54, 2020 [DOI] [PubMed] [Google Scholar]
- 11). Atlas SW, Grossman RI, Goldberg HI, Hackney DB, Bilaniuk LT, Zimmerman RA: Partially thrombosed giant intracranial aneurysms: correlation of MR and pathologic findings. Radiology 162: 111– 114, 1987 [DOI] [PubMed] [Google Scholar]
- 12). Martin AJ, Hetts SW, Dillon WP, et al. : MR imaging of partially thrombosed cerebral aneurysms: characteristics and evolution. AJNR Am J Neuroradiol 32: 346– 351, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13). Perlmutter D, Rhoton AL: Microsurgical anatomy of the anterior cerebral-anterior communicating-recurrent artery complex. J Neurosurg 45: 259– 272, 1976 [DOI] [PubMed] [Google Scholar]
- 14). Murayama Y, Viñuela F, Tateshima S, Song JK, Gonzalez NR, Wallace MP: Bioabsorbable polymeric material coils for embolization of intracranial aneurysms: a preliminary experimental study. J Neurosurg 94: 454– 463, 2001 [DOI] [PubMed] [Google Scholar]
- 15). Ansari SA, Dueweke EJ, Kanaan Y, et al. : Embolization of intracranial aneurysms with second-generation Matrix-2 detachable coils: mid-term and long-term results. J Neurointerv Surg 3: 324– 330, 2011 [DOI] [PubMed] [Google Scholar]
- 16). Bradley WG: MR appearance of hemorrhage in the brain. Radiology 189: 15– 26, 1993 [DOI] [PubMed] [Google Scholar]
- 17). Kwan ES, Heilman CB, Shucart WA, Klucznik RP: Enlargement of basilar artery aneurysms following balloon occlusion–“water-hammer effect”. Report of two cases. J Neurosurg 75: 963– 968, 1991 [DOI] [PubMed] [Google Scholar]