Abstract
Predator-prey interactions influence prey traits through both consumptive and non-consumptive effects, and variation in these traits can shape vector-borne disease dynamics. Meta-analysis methods were employed to generate predation effect sizes by different categories of predators and mosquito prey. This analysis showed that multiple families of aquatic predators are effective in consumptively reducing mosquito survival, and that the survival of Aedes, Anopheles, and Culex mosquitoes is negatively impacted by consumptive effects of predators. Mosquito larval size was found to play a more important role in explaining the heterogeneity of consumptive effects from predators than mosquito genus. Mosquito survival and body size were reduced by non-consumptive effects of predators, but development time was not significantly impacted. In addition, Culex vectors demonstrated predator avoidance behavior during oviposition. The results of this meta-analysis suggest that predators limit disease transmission by reducing both vector survival and vector size, and that associations between drought and human West Nile virus cases could be driven by the vector behavior of predator avoidance during oviposition. These findings are likely to be useful to infectious disease modelers who rely on vector traits as predictors of transmission.
Research organism: Mosquito
eLife digest
Mosquitoes are often referred to as the deadliest animals on earth because some species spread malaria, West Nile virus or other dangerous diseases when they bite humans and other animals. Adult mosquitoes fly to streams, ponds and other freshwater environments to lay their eggs. When the eggs hatch, the young mosquitoes live in the water until they are ready to grow wings and transform into adults.
In the water, the young mosquitoes are particularly vulnerable to being eaten by dragonfly larvae, fish and other predators. When adult females are choosing where to lay their eggs, they can use their sense of smell to detect these predators and attempt to avoid them. Along with eating the mosquitoes, the predators may also reduce mosquito populations in other ways. For example, predators can disrupt feeding among young mosquitoes, which may affect the time that it takes for them to grow into adults or the size of their bodies once they reach the adult stage. Although the impacts of different predators have been tested separately in multiple settings, the overall effects of predators on the ability of mosquitoes to spread diseases to humans remain unclear.
To address this question, Russell, Herzog et al. used an approach called meta-analysis on data from previous studies. The analysis found that along with increasing the death rates of mosquitoes, the presence of predators also leads to a reduction in the body size of those mosquitoes that survive, causing them to have shorter lifespans and fewer offspring.
Russell, Herzog et al. found that one type of mosquito known as Culex – which carries West Nile virus – avoided laying its eggs near predators. During droughts, increased predation in streams, ponds and other aquatic environments may lead adult female Culex mosquitoes to lay their eggs closer to residential areas with fewer predators. Russell, Herzog et al. propose that this may be one reason why outbreaks of West Nile virus in humans are more likely to occur during droughts.
In the future, these findings may help researchers to predict outbreaks of West Nile virus, malaria and other diseases carried by mosquitoes more accurately. Furthermore, the work of Russell, Herzog et al. provides examples of mosquito predators that could be used as biocontrol agents to decrease numbers of mosquitoes in certain regions.
Introduction
While it is well known that predation reduces vector populations through consumptive effects, non-consumptive effects of predators can also greatly impact prey demographics (Preisser et al., 2005). Mosquitoes are vectors of a variety of debilitating and deadly diseases, including malaria, lymphatic filariasis, and arboviruses, such as chikungunya, Zika, and dengue (Weaver and Reisen, 2010; WORLD HEALTH ORGANIZATION, 2020). Consequently, there is motivation from a public health perspective to better understand the different drivers of variation in mosquito traits that can ultimately impact vector population growth and disease transmission. In addition, recent work has suggested that incorporation of vector trait variation into disease models can improve the reliability of their predictions (Cator et al., 2020). In this study, systematic review and meta-analysis methods are used to synthesize a clearer understanding of the consumptive and non-consumptive effects of predators on mosquito traits, including survival, oviposition, development, and size.
Mosquito insecticide resistance is recognized as a growing problem (Hancock et al., 2018; Hemingway and Ranson, 2000; Liu, 2015) leading some to suggest that control efforts should rely more heavily on ‘non-insecticide based strategies’ (Benelli et al., 2016). The consumptive effects of predators on mosquitoes have previously been harnessed for biocontrol purposes. Past biocontrol efforts have used predators such as cyclopoid copepods (Kay et al., 2002; Marten, 1990; Russell et al., 1996; Veronesi et al., 2015) and mosquitofish (Pyke, 2008, Seale, 1917) to target the mosquito’s aquatic larval stage. The strength of the consumptive effects of these predators on mosquitoes can be influenced by multiple factors, including predator-prey size ratio and temperature. Predator-prey body size ratios tend to be higher in freshwater habitats than other types of habitats (Brose et al., 2006), and attack rate tends to increase with temperature (Kalinoski and DeLong, 2016; Dam and Peterson, 1988), although other studies suggest a unimodal response to temperature (Uiterwaal and Delong, 2020; Englund et al., 2011).
Predators can also have non-consumptive effects on prey (Peacor and Werner, 2001), and these effects are thought to be more pronounced in aquatic ecosystems than in terrestrial ecosystems (Preisser et al., 2005). Non-consumptive effects of predators are the result of the prey initiating anti-predator behavioral and/or physiological trait changes that can aid in predator avoidance (Hermann and Landis, 2017; Lima and Dill, 1990). Such plasticity in certain prey traits may also result in energetic costs (Lima, 1998). Predator detection is key for these trait changes to occur and can be mediated by chemical, tactile, and visual cues (Hermann and Thaler, 2014). In mosquitoes, exposure to predators is known to affect a variety of traits including behavior, size, development, and survival (Arav and Blaustein, 2006; Bond et al., 2005; Roberts, 2012; Roux et al., 2015, Zuharah et al., 2013). Experimental observations of predator effects on mosquito size and development are inconsistent and results sometimes vary by mosquito sex. For example, exposure to predation was found to increase the size of Culex pipiens mosquitoes (Alcalay et al., 2018) but decrease the size of Culiseta longiareolata (Stav et al., 2005). In addition, female Aedes triseriatus exhibited shorter development times when exposed to predation at high nutrient availability (Ower and Juliano, 2019), but male C. longiareolata had longer development times in the presence of predators (Stav et al., 2005). In some cases, a shared evolutionary history between predator and prey organisms can strengthen the non-consumptive effects of predators on mosquitoes (Buchanan et al., 2017; Sih, 1986).
This investigation assesses the consumptive and non-consumptive effects of predators on mosquito traits and describes how these effects could impact disease transmission. The roles of vector genus, predator family, mosquito larval instar (an indicator of prey size), and temperature are also examined as potential moderators of predator effects. Non-consumptive effects of predators are expected to cause a smaller reduction in mosquito survival than consumptive effects because, in practice, measures of consumptive effects always include both consumptive and non-consumptive effects. Based on previous findings, larger predators are more likely to consumptively reduce mosquito survival (Kumar et al., 2008). In addition, Aedes mosquito larvae may be more vulnerable to consumption than other genera because of the high degree of motility observed in this genus (Dieng et al., 2003; Marten and Reid, 2007; Soumare and Cilek, 2011). The oviposition response to predation is expected to be weakest among Aedes species that oviposit above the water line, due in part to their delayed-hatching eggs (Vonesh and Blaustein, 2010). Predation is predicted to reduce mosquito size and lengthen development time, consistent with the reduced growth response observed in other insect systems (Hermann and Landis, 2017). Certain non-consumptive effects of predation, particularly oviposition site selection and decreased vector size, are likely to play important roles in the dynamics of mosquito-borne disease.
Materials and methods
Literature screening
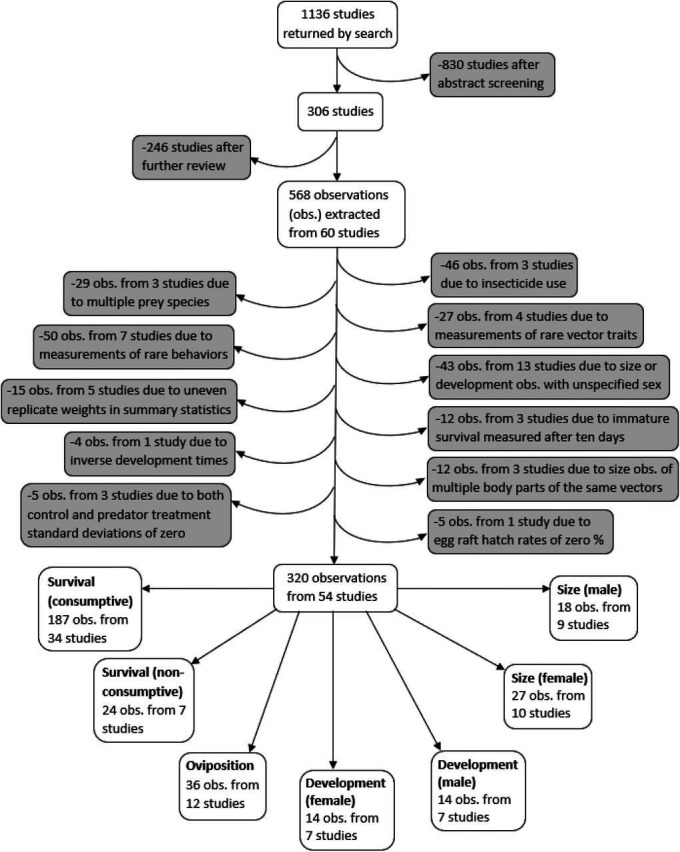
A systematic search was conducted for studies on predation of mosquitoes that were published between 1970 and July 1, 2019 using both PubMed and Web of Science search engines, according to the PRISMA protocol (Moher et al., 2009). Mosquito vectors of the Anopheles and Aedes genera were specifically highlighted in our search terms because these genera contain the vector species that transmit malaria, yellow fever, and dengue – the three most deadly mosquito-borne diseases worldwide (Hill et al., 2005). Searches included 18 combinations of three vector predation terms (mosquito predat*, Anopheles predat*, Aedes predat*) and six trait terms (survival, mortality, development, fecundity, dispers*, host preference). Abstracts from the 1136 studies were each screened by two different co-authors, using the ‘metagear’ package in R (Lajeunesse, 2016, R Development Core Team, 2020). If either screener thought the study had information relevant to predation of mosquitoes, or both screeners thought the abstract was ambiguous, the study was read in full. This resulted in 306 studies that were fully reviewed to determine if any predation data could be extracted (Figure 1).
Figure 1. Flowchart demonstrating the literature search, screening process, data exclusions, and the resulting seven different vector trait data subsets.
Study exclusion criteria
Data were extracted from studies that collected data on non-consumptive and/or consumptive effects of predators on mosquitoes. Studies were required to have a mean, error measurement, and at least two replicates for both control and predator treatments. The control treatment was required to have all the same conditions as the predator treatment, such as prey density and type of water, without the predators. Studies that were not published in English and studies that did not differentiate between predators of multiple families were excluded. Studies were also excluded if oviposition by free-flying female mosquitoes could have interfered with observing the consumptive effects of predators on vector survival. The final database comprised data extracted from 60 studies (Supplementary file 1). The data included observations from laboratory experiments, as well as semi-field experiments, in which mesocosms of different treatments were observed in outdoor settings.
Data extraction
Variables related to the publication, the vector, the predator, and the effect size (Table 1) were extracted from each study. Data from tables and text were recorded as they were published, and data from figures were extracted using WebPlotDigitizer (Rohatgi, 2020). Error measurements that were not originally presented as standard deviations were converted to standard deviations prior to the effect size calculation.
Table 1. Variables extracted from included studies.
| Variable | Description |
|---|---|
| Publication data: | |
| Title | Full study title |
| Journal | Name of journal that published the study |
| Year | Year of publication |
| Study environment | Environment where the experiment took place: lab or semi-field |
| Vector data: | |
| Order, Family, Genus, Species | Taxonomic identification |
| Trait | Outcome that was measured (e.g. survival, development, etc.) |
| Stage | Life stage: egg, larva, pupa, or adult |
| Larval instar | Early (1st and 2nd instars), late (3rd and 4th instars), both, or NA (eggs, pupae, or adults) |
| Sex | Male or female |
| Predator data: | |
| Phylum, Class, Order, Family, Genus, Species | Taxonomic identification |
| Starved | Whether the predator was starved: yes or no |
| Time starved | Amount of time that the predator was starved (in minutes) |
| Predation effect | Consumptive or non-consumptive |
| Effect size data: | |
| Units | Units of extracted data |
| Control mean | Average of the outcome measured among the controls |
| Control standard deviation | Standard deviation of the outcome measured in the controls |
| Control number of replicates | Number of control replicates |
| Predation mean | Average of the outcome measured in the predator treatment |
| Predation standard deviation | Standard deviation of the outcome measured in the predator treatment |
| Predation number of replicates | Number of predation replicates |
| Experiment ID | Alphabetic assignment to mark observations sharing a control group or representing the same prey individuals as originating from the same experiment |
| Additional data: | |
| Experiment time (days) | Duration of the experiment in days |
| Data source | Graph or text |
| Number of predators | Number of predators with access to prey, or ‘cue’ if there are no predators with direct access to prey |
| Number of prey (vectors) | Number of mosquito prey that are exposed to predation |
| Arena volume (mL) | Volume of the arena where prey encounter predators |
| Time exposed to predator(s) | Amount of time (in days) when the predator has direct access to the mosquito prey |
| Temperature (°C) | Temperature during the predation interaction |
| Type of predator cue | Predator cues, or cues from both predator(s) and dying conspecifics; NA for observations with a consumptive predation effect |
Data exclusions
A PRISMA plot of literature inclusion and exclusion is provided in Figure 1. Observations where insecticide was used were excluded because insecticides are known to interfere with consumptive and non-consumptive effects of predators (Delnat et al., 2019; Janssens and Stoks, 2012). In addition, observations from experiments with mosquito prey of two or more species were excluded because it was not possible to account for effects from apparent competition or prey-switching. Observations of vector fecundity, vector competence, behavioral traits other than oviposition, as well as observations where the vector trait was marked as ‘other’ were not analyzed because each of these traits were only recorded from three or fewer studies.
Due to protandry, the earlier emergence of males to maximize their reproductive success, mosquitoes respond to sex-specific selective forces that influence their development time and body size (Kleckner et al., 1995). Under low resource conditions, female mosquitoes are likely to maximize body mass by extending their development time, whereas males tend to minimize their development time at the expense of lower body mass (Kleckner et al., 1995). Observations of mosquito development time and body size in our database that were not sex-specific were excluded so that these vector traits could be analyzed while controlling for sex. In addition, among the observations of development time and body size, some predator means did not necessarily represent an evenly weighted average of the replicates. For example, if a total of 20 mosquitoes from three different predator replicates survived to adulthood, the mean development time and size of those 20 individuals may have been reported. To represent an evenly weighted average of the replicates, it is necessary to first calculate summary statistics among multiple individuals that emerge from the same replicate, and then report the average of the replicate-specific means. Observations that might have been influenced by uneven representation of replicates were excluded to prevent pseudo-replication from altering later meta-analyses.
For consumptive observations where life stage-specific survival was reported after more than 10 days of predator exposure, only data on survival marked by adult emergence were included for analysis. Effects observed among immature vector stages after such a long period of predator exposure were not analyzed because they could have resulted from a combination of non-consumptive effects on development, and consumptive effects on survival. Development time observations that were reported as the inverse of development time (units of days–1) were excluded because although their means could be converted to units of days, their standard deviations could not be converted to match units of days. In cases where multiple body sections of the same mosquitoes were measured to produce multiple size observations, only the wing measurement was included in the analysis to prevent pseudo-replication. Observations in which both the control and the predator treatments had standard deviations of zero were excluded because the meta-analysis methods did not support non-positive sampling variances.
Exclusions and data substitutions for predator treatment means of zero
One study that was included in our database reported egg survival data as the hatch rate of field collected Culex pervigilans rafts (Zuharah et al., 2013). However, mosquitoes have been shown to lay eggs independent of mating (O’Meara, 1979), and hatch rates of zero have previously been observed in rafts laid by Culex females that were held separately from males (Su and Mulla, 1997). Thus, hatch rates of zero were excluded from further analysis because these values may represent unfertilized egg rafts, rather than a strong impact of predators on survival. Twenty of the 187 consumptive survival observations had a predation mean of zero, and each of these zeros resulted from experiments that began with a specified number of live larvae. Consumptive survival zeros were each replaced with 0.5% of the starting number of mosquito prey to avoid undefined effect sizes. In addition, there was one zero out of the 36 oviposition predation means; this value had units of ‘number of egg rafts laid’ and was replaced with 0.5 rafts. Similar methods for replacing zero values in the treatment mean with small non-zero values have previously been employed (Thapa et al., 2018).
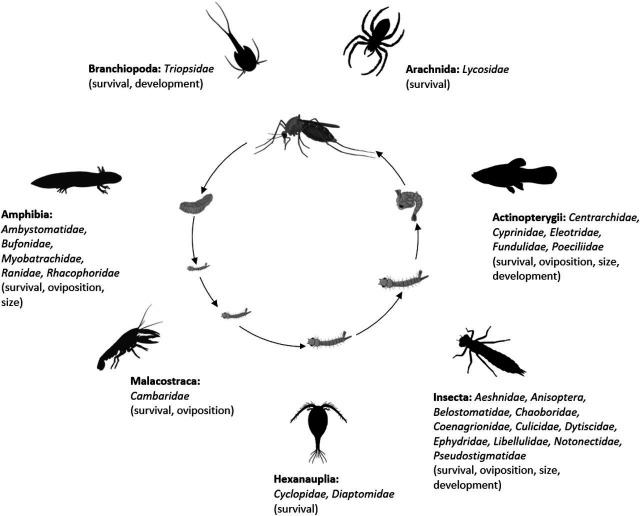
The final analysis dataset included seven subsets: consumptive effects on survival, non-consumptive effects on survival, oviposition, development (female and male), and size (female and male). The data included 187 observations from 34 studies of consumptive survival, 24 observations from seven studies of non-consumptive survival, 36 observations from 12 studies of oviposition, 14 observations from seven studies of female development, 14 observations from seven studies of male development, 27 observations from 10 studies of female size, and 18 observations from nine studies of male size (Figure 1). These observations covered seven different classes of predator families (Figure 2).
Figure 2. Mosquito predator classes (bold font) and families (italicized font) included in the database and the vector traits that they may influence (in parentheses); predator images not to scale, and placed randomly with respect to the different mosquito life stages.
Image sources: phylopic.org (CC BY 3.0 or public domain): Actinopterygii (creator: Milton Tan), Arachnida (creators: Sidney Frederic Harmer & Arthur Everett Shipley, vectorized by Maxime Dahirel), Branchiopoda (creator: Africa Gomez), and Insecta (creator: Marie Russell). BioRender.com: Amphibia, Hexanauplia, and Malacostraca class silhouettes; mosquito larval instars, pupa, and blood-feeding adult. Trishna Desai: mosquito egg raft.
Data analysis
Measuring effect sizes and heterogeneity
All analyses were conducted in R version 4.0.2 (R Development Core Team, 2020). For each subset of trait data (Figure 1), the ratio of means (ROM) measure of effect size was calculated using the ‘escalc’ function from the ‘metafor’ package; this effect measure is equal to a log-transformed fraction, where predation mean is the numerator and control mean is the denominator (Viechtbauer, 2010). Random effects models, using the ‘rma.uni’ function, were run with the ROM effect sizes as response variables; each model had a normal error distribution and a restricted maximum likelihood (REML) estimator for τ2, the variance of the distribution of true effect sizes (Viechtbauer, 2010). Although these random effects models could not account for multiple random effects or moderators, they provided overall estimates of the ROM effect sizes and estimates of the I2 statistics. Each I2 statistic represented the percentage of total variation across studies due to heterogeneity (Higgins et al., 2003). If the I2 statistic was equal to or greater than 75%, the heterogeneity was considered to be high (Higgins et al., 2003), and high heterogeneity has previously motivated further testing of moderators (Vincze et al., 2017).
Assessing publication bias
Publication bias was assessed by visually inspecting funnel plots and conducting Egger’s regression test (‘regtest’ function) with standard error as the predictor (Sterne and Egger, 2001; Viechtbauer, 2010). If the Egger’s regression test showed significant evidence of publication bias based on funnel plot asymmetry, the ‘trim and fill’ method (‘trimfill’ function) was used to estimate how the predation effect size might change after imputing values from missing studies (Duval and Tweedie, 2000a, Duval and Tweedie, 2000b; Viechtbauer, 2010). The trim and fill method has previously been recommended for testing the robustness of conclusions related to topics in ecology and evolution (Jennions and Møller, 2002). Of the two trim and fill estimators, R0 and L0, that were originally recommended (Duval and Tweedie, 2000a, Duval and Tweedie, 2000b), the L0 estimator was used in this study because it is more appropriate for smaller datasets (Shi and Lin, 2019).
Testing moderators
Data subsets that had high heterogeneity, observations from at least 10 studies, and no evidence of publication bias according to Egger’s regression results were analyzed further using multilevel mixed effects models with the ‘rma.mv’ function (Viechtbauer, 2010; Higgins et al., 2020). All multilevel mixed effects models had normal error distributions, REML estimators for τ2, and accounted for two random factors: effect size ID, and experiment ID nested within study ID. Moderators, such as predator family, vector genus, larval instar (directly correlated to prey size), and temperature, were tested within each data subset to determine if they affected the observed heterogeneity in ROM effect sizes. For categorical moderators, the intercept of the multilevel mixed effects model was removed, allowing an analysis of variance (ANOVA) referred to as the ‘test of moderators’ to indicate if any of the categories had an effect size different than zero. For data subsets with observations from 10 to 29 studies, only one moderator was tested at a time to account for sample size constraints. For subsets with observations from a higher number of studies (30 or more), up to two moderators were tested at once, and interaction between moderators was also tested. The small sample corrected Akaike Information Criterion (AICc) was used to compare multilevel mixed effects models and to select the model of best fit within each data subset; differences in AICc greater than two were considered meaningful (Burnham and Anderson, 2004).
Results
Random effects models
Each data subset (Figure 1) had an I2 statistic of greater than 75%, indicating high heterogeneity (Higgins et al., 2003). Random effects model results showed that predators consumptively decreased mosquito survival with an effect size of –1.23 (95% CI −1.43,–1.03), p-value < 0.0001, and non-consumptively reduced survival with a smaller effect size of –0.11 (95% CI −0.17,–0.04), p-value = 0.0016. In addition, predators non-consumptively reduced oviposition behavior with an effect size of –0.87 (95% CI −1.31,–0.42), p-value = 0.0001, and mosquito body size was non-consumptively reduced by predators in both males and females; the female effect size was –0.13 (95% CI −0.19,–0.06), p-value = 0.0002, and the male effect size was –0.03 (95% CI −0.06,–0.01), p-value = 0.0184. There was not a significant non-consumptive effect of predators on either male or female development time; the female effect size was –0.01 (95% CI –0.09, 0.07), p-value = 0.7901, and the male effect size was –0.04 (95% CI –0.12, 0.04), p-value = 0.3273.
The Egger’s regression test results showed that the non-consumptive survival subset, both development time subsets (male and female), and the female size subset exhibited funnel plot asymmetry indicative of publication bias. The ‘trim and fill’ procedure identified missing studies in the non-consumptive survival subset and the female size subset, but the procedure did not identify any missing studies in either of the development time subsets. Three studies were estimated to be missing from the non-consumptive survival data, and accounting for imputed values from missing studies resulted in a shift in the predation effect size from –0.11 (95% CI −0.17,–0.04), p-value = 0.0016, to -0.13 (95% CI −0.20,–0.07), p-value < 0.0001. Two studies were estimated to be missing from the female size data, and accounting for imputed values from these missing studies shifted the predation effect size from –0.13 (95% CI −0.19,–0.06), p-value = 0.0002, to -0.10 (95% CI −0.17,–0.03), p-value = 0.0083. Shifts in effect size estimates due to the trim and fill procedure were minor and did not cause any of the observed effects of predators to change direction or become insignificant.
Multilevel mixed effects models
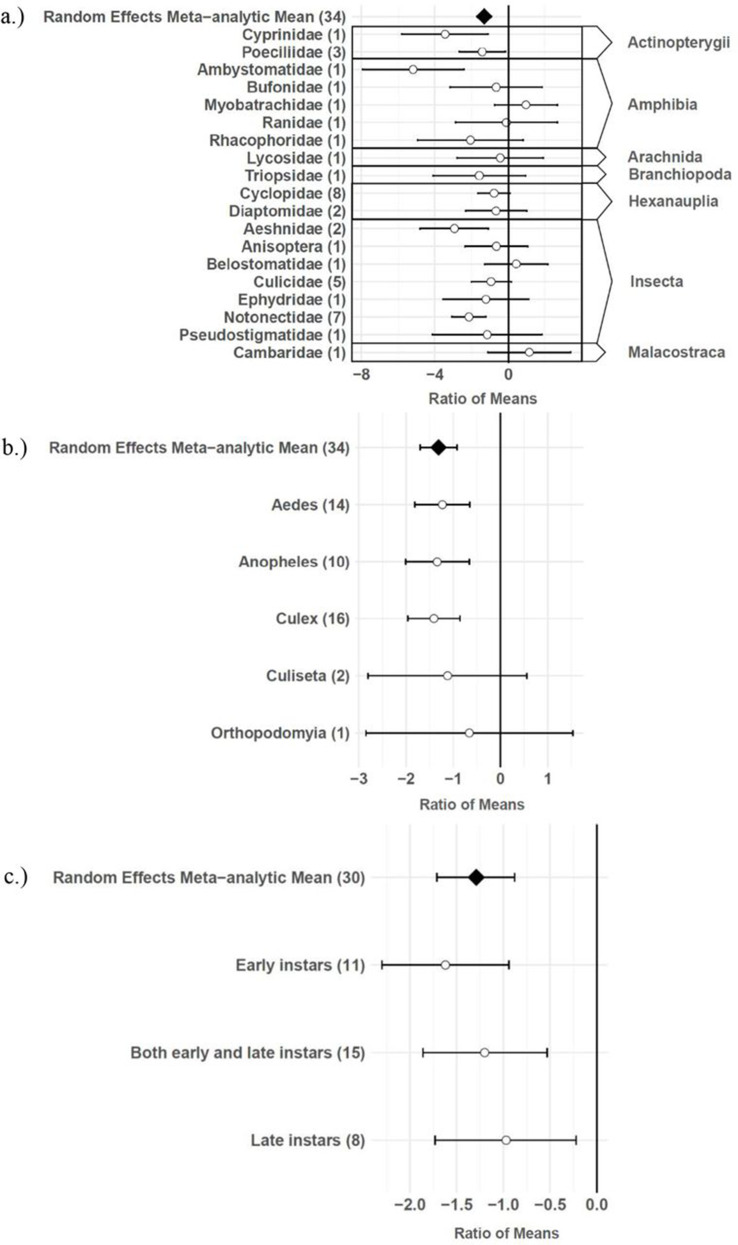
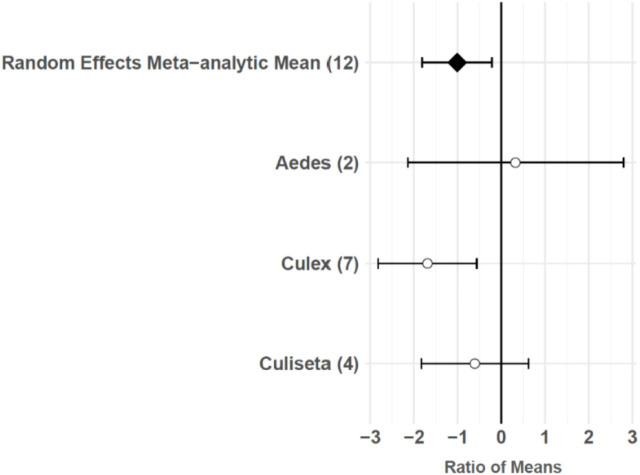
The consumptive survival and oviposition data subsets met the criteria of high heterogeneity, observations from at least 10 studies, and no evidence of publication bias. Therefore, these data subsets were tested for moderators using multilevel mixed effects models. Predator families that decreased mosquito survival included Cyprinidae: –3.44 (95% CI −5.79,–1.09), p-value = 0.0042; Poeciliidae: –1.42 (95% CI −2.67,–0.16), p-value = 0.0270; Ambystomatidae: –5.18 (95% CI −7.94,–2.42), p-value = 0.0002; Aeshnidae: –2.93 (95% CI −4.80,–1.07), p-value = 0.0020; and Notonectidae: –2.14 (95% CI −3.07,–1.21), p-value < 0.0001 (Figure 3a). Vector genera that experienced significant decreases in survival due to consumptive effects of predators included Aedes: –1.23 (95% CI −1.81,–0.65), p-value < 0.0001; Anopheles: –1.34 (95% CI −2.01,–0.66), p-value = 0.0001; and Culex: –1.41 (95% CI −1.96,–0.86), p-value < 0.0001 (Figure 3b). Among all 187 consumptive survival observations from 34 studies, the best model fit, according to AICc value, was achieved when an interaction between predator family and vector genus was included in the model (Table 2). However, among the 163 larval stage consumptive survival observations from 30 studies, adding an interactive term between larval instar (an indicator of prey size) and predator family had a greater improvement on model fit than adding an interactive term between vector genus and predator family (Figure 3c, Table 3). Temperature did not affect the heterogeneity of consumptive survival data, either as a linear moderator: –0.01 (95% CI –0.10, 0.07), p-value = 0.7559, or a quadratic moderator: 0.00 (95% CI 0.00, 0.00), p-value = 0.8184. The best oviposition model fit, according to AICc value, was achieved when vector genus was added as a moderator (Table 4). The mean oviposition effect size was not significantly different than zero for Aedes: 0.32 (95% CI –2.14, 2.79), p-value = 0.7970, or Culiseta: –0.61 (95% CI –1.83, 0.62), p-value = 0.3329, but for Culex mosquitoes, oviposition was significantly decreased by predator presence: –1.69 (95% CI −2.82,–0.56), p-value = 0.0033 (Figure 4).
Figure 3. Effect sizes and 95 % confidence intervals for consumptive effects of predators, for different categories of moderators (with number of studies in parentheses).
(a) predator family with predator class in the right-hand column, (b) vector genus, and (c) larval instar.
Table 2. Candidate multilevel mixed effects models of consumptive effects from predators on mosquito survival, fitted to dataset of effect sizes (n = 187 from 34 studies), and ranked by corrected Akaike’s information criterion (AICc).
| Moderator(s) | Test of moderators(degrees of freedom, p-value) | AICc | ΔAICc |
|---|---|---|---|
| Predator family x vector genus | 28, < 0.0001 | 500.5 | 0 |
| Predator family | 19, < 0.0001 | 507.0 | 6.5 |
| Predator family + vector genus | 23, < 0.0001 | 508.1 | 7.6 |
| Vector genus | 5, < 0.0001 | 573.0 | 72.5 |
| None | ---- | 576.5 | 76.0 |
Table 3. Candidate multilevel mixed effects models of consumptive effects from predators, fitted to dataset of effect sizes where larval instar is not missing (n = 163 from 30 studies), and ranked by corrected Akaike’s information criterion (AICc).
| Moderator(s) | Test of moderators(degrees of freedom, p-value) | AICc | ΔAICc |
|---|---|---|---|
| Predator family x larval instar | 25, < 0.0001 | 429.2 | 0 |
| Predator family + larval instar | 19, < 0.0001 | 443.5 | 14.3 |
| Predator family x vector genus | 25, < 0.0001 | 455.0 | 25.8 |
| Predator family | 17, < 0.0001 | 456.8 | 27.6 |
| Predator family + vector genus | 21, < 0.0001 | 458.4 | 29.2 |
| Larval instar | 3, < 0.0001 | 503.1 | 73.9 |
| Vector genus | 5, < 0.0001 | 504.7 | 75.5 |
| None | ---- | 508.5 | 79.3 |
Table 4. Candidate multilevel mixed effects models of non-consumptive effects of predators on mosquito oviposition behavior, fitted to dataset of effect sizes (n = 36 from 12 studies), and ranked by corrected Akaike’s information criterion (AICc).
| Moderator(s) | Test of moderators(degrees of freedom, p-value) | AICc | ΔAICc |
|---|---|---|---|
| Vector genus | 3, 0.0149 | 122.1 | 0 |
| None | ---- | 125.2 | 3.1 |
| Predator family | 12, 0.8855 | 167.9 | 45.8 |
Figure 4. Oviposition effect sizes and 95 % confidence intervals for different categories of vector genus (with number of studies in parentheses).
Discussion
In this study, laboratory and semi-field empirical data were obtained through a systematic literature review and used to conduct a meta-analysis that assessed consumptive and non-consumptive effects of predators on mosquito prey. Some results agree with previously observed trends, such as greater consumptive effects from larger predators (Kumar et al., 2008, Peters, 1983) and no oviposition response to predator cues among container-breeding Aedes mosquitoes (Vonesh and Blaustein, 2010). However, this meta-analysis revealed additional trends. Mosquito larval instar had an important role in moderating consumptive effects of predators, likely because of its direct correlation to prey size. Furthermore, a small, but significant, decrease in mosquito survival due to non-consumptive effects of predators was observed, suggesting that mosquitoes can be ‘scared to death’ by predators (Preisser et al., 2005). Both male and female body sizes were also reduced among mosquitoes that had been exposed to predators, and predator avoidance during oviposition was observed among female Culex mosquitoes. Effects of predators on different vector traits, particularly survival, body size, and oviposition behavior, have the potential to influence infectious disease dynamics.
Consumptive effects of predators on survival
Several larger predators reduced mosquito survival, including freshwater fish (Cyprinidae and Poeciliidae), salamander larvae (Ambystomatidae), dragonfly larvae (Aeshnidae), and backswimmers (Notonectidae) (Figure 3a). This finding is consistent with a previous analysis which showed a positive linear relationship between predator body mass and ingestion rate across taxa (Peters, 1983). In addition, more effect size heterogeneity in the consumptive survival data was explained by an interaction between predator family and larval instar than was explained by an interaction between predator family and vector genus (Table 3). This result suggests that the relative sizes of predator and prey groups could play a more important role in determining consumptive mosquito survival than variations in predator responses to different behaviors of prey genera, which are likely to be shaped by the degree of shared evolutionary history between trophic levels (Buchanan et al., 2017). Larval instar is an indicator of mosquito size, and previous modeling work has provided evidence of prey size selection by predators to maximize energetic gain (Mittelbach, 1981). While smaller cyclopoid copepods are more effective against early instar mosquito larvae (Dieng et al., 2002), larger predators including tadpoles, giant water bugs, dragonfly larvae, fish, and backswimmers are more effective against late instar larvae (Kweka et al., 2011).
Non-consumptive effects of predators on survival
Exposure to predation cues significantly lowered mosquito survival, and this non-consumptive effect has also been observed in dragonfly larvae prey (Leucorrhinia intacta) that were exposed to caged predators (McCauley et al., 2011). The reduction in mosquito survival from non-consumptive effects of predators was significantly smaller than the reduction that was observed from consumptive effects. This is partially due to the practical constraints of most experimental designs, which cause consumptive and non-consumptive effects of predators on survival to be grouped together and reported as consumptive effects. The greater impact of combined consumptive and non-consumptive effects, in comparison to only non-consumptive effects, has previously been observed in pea aphids (Acyrthosiphon pisum) (Nelson et al., 2004).
Non-consumptive effects of predators on body size
While predators did not significantly impact mosquito development time through non-consumptive effects in either sex, mosquito body size was decreased by the non-consumptive effects of predators in both sexes. Smaller body size is associated with lower reproductive success in mosquitoes because smaller females lay fewer eggs (Blackmore and Lord, 2000; Lyimo and Takken, 1993; Oliver and Howard, 2011; Styer et al., 2007; Tsunoda et al., 2010), and smaller males produce less sperm (Hatala et al., 2018; Ponlawat and Harrington, 2007). These effects suggest that predation could non-consumptively reduce mosquito population growth. The smaller size of mosquitoes exposed to predators could also limit disease transmission. Vector lifespan contributes disproportionately to disease transmission because older vectors are more likely to have been exposed to pathogens, more likely to already be infectious after having survived the extrinsic incubation period, and more likely to survive long enough to bite subsequent hosts (Cator et al., 2020). It is well-established that smaller mosquito body size is associated with shorter mosquito lifespan (Araújo et al., 2012; Hawley, 1985, Reisen et al., 1984; Reiskind and Lounibos, 2009; Xue et al., 2010). Therefore, non-consumptive effects of predators may limit the transmission of mosquito-borne diseases.
Non-consumptive effects of predators on oviposition behavior
Predator presence also non-consumptively reduced oviposition behavior in adult female mosquitoes. Meta-regression results showed that Culex females significantly avoid oviposition sites that contain predators or predator cues, but Aedes and Culiseta females do not avoid these sites, despite a slight non-significant trend toward predator avoidance in Culiseta (Figure 4). Both Culex and Culiseta mosquitoes have an ‘all-or-none’ oviposition strategy (Johnson and Fonseca, 2014), in which they lay hundreds of rapidly hatching eggs in rafts on the water’s surface (Day, 2016). Such an oviposition strategy is conducive to evolving predator avoidance behaviors, and a previous meta-analysis showed significant predator avoidance in both Culex and Culiseta during oviposition (Vonesh and Blaustein, 2010). Conversely, it is likely that an oviposition response to predation is not particularly advantageous for Aedes because the delayed hatching of their eggs (Day, 2016) can prevent the level of predation risk at the time of oviposition from matching the level of predation risk present in the eventual larval environment (Vonesh and Blaustein, 2010). The predator avoidance response in Aedes species that lay their eggs above the water’s edge in containers has previously been described as ‘non-existent’ (Vonesh and Blaustein, 2010). Both Aedes species included in this study’s oviposition data subset, Ae. albopictus and Ae. aegypti, meet the criterion of ovipositing above water in containers (Juliano, 2009). Predator avoidance during oviposition has previously been found to increase the mosquito population size at equilibrium (Spencer et al., 2002). However, this study’s results and those of a previous meta-analysis (Vonesh and Blaustein, 2010) suggest that models of oviposition site selection, such as those using parameters from Notonectidae predators and Culiseta prey (Kershenbaum et al., 2012), are not generalizable to Aedes vectors.
Implications for West Nile Virus disease dynamics
Predator avoidance during oviposition by Culex mosquitoes (Figure 4) may be of particular importance to West Nile virus (WNV) disease dynamics. Previous work has shown that Cx. pipiens, Cx. restuans, and Cx. tarsalis all avoid predator habitats (Vonesh and Blaustein, 2010), and that Cx. pipiens is the primary bridge vector of WNV responsible for spill-over transmission from avian reservoir hosts to humans (Fonseca et al., 2004; Hamer et al., 2008a, Kramer et al., 2008; Andreadis, 2012). Cx. pipiens mosquitoes can live in permanent aquatic environments, such as ground pools (Amini et al., 2020; Barr, 1967; Dida et al., 2018; Sulesco et al., 2015), ponds (Lühken et al., 2015), stream edges (Amini et al., 2020), and lake edges (Vinogradova, 2000) that are more common in rural areas, but Cx. pipiens are also found in urban and suburban residential areas, where they typically breed in artificial containers (Sulesco et al., 2015), including tires (Lühken et al., 2015; Nikookar et al., 2017; Verna, 2015), rainwater tanks (Townroe and Callaghan, 2014), and catch basins (Gardner et al., 2012). Small artificial containers, such as discarded tires, are generally unlikely to harbor larger predators, including freshwater fish (Cyprinidae and Poeciliidae), salamander larvae (Ambystomatidae), dragonfly larvae (Aeshnidae), and backswimmers (Notonectidae), because temporary aquatic environments cannot support the relatively long development times of these organisms. The mean dispersal distance of adult Culex mosquitoes is greater than one kilometer (Ciota et al., 2012; Hamer et al., 2014), and female Cx. pipiens have exhibited longer dispersal distances after developing in the presence of a fish predator (Alcalay et al., 2018). Therefore, predator avoidance during oviposition may cause Cx. pipiens populations to disperse from permanent aquatic environments in more rural areas to artificial container environments in urbanized areas, where the risk of human WNV infection is higher (Brown et al., 2008).
Predator cue levels may be altered by climate conditions, and these changes in cue levels can impact WNV transmission to humans. Drought has previously been associated with human WNV cases (Johnson and Sukhdeo, 2013; Marcantonio et al., 2015; Roehr, 2012; Shaman et al., 2005; Epstein and Defilippo, 2001; Paull et al., 2017), but the association has thus far lacked a clear underlying mechanism. Under drought conditions, the density of aquatic organisms increases and predation pressures can intensify due to compressed space and high encounter rates (Amundrud et al., 2019). A previous study of a stream ecosystem found that impacts of fish predation are more severe during the dry season (Dudgeon, 1993). In addition, reductions in water volume can facilitate consumption of mosquito larvae by crane fly larvae (Tipulidae), whereas mosquito consumption by tipulids was not observed at a higher water level (Amundrud et al., 2019). Laboratory and semi-field studies have shown that mosquitoes respond to a gradient of predator cues (Roux et al., 2014; Silberbush and Blaustein, 2011). The frequency of larval anti-predator behavior is correlated with the concentration of predator cues (Roux et al., 2014), and adult female mosquitoes prefer oviposition sites with lower predator densities (Silberbush and Blaustein, 2011). Therefore, as predator cue levels increase due to drought, permanent aquatic habitats are likely to transition from suitable oviposition sites for one generation of female mosquitoes, to unsuitable oviposition sites for the next generation.
When suitable oviposition sites are absent, females retain their eggs until sites become available (Bentley and Day, 1989). Cx. pipiens females can retain their eggs for up to five weeks, allowing them enough time to find container sites with low predation risk, often located in residential areas (Johnson and Fonseca, 2014). The movement of gravid female Cx. pipiens to residential areas increases the risk of WNV spill-over to humans because these vectors are likely to have already blood-fed at least once (Clements, 1992), suggesting that they have a higher risk of WNV infection, relative to non-gravid mosquitoes. This is consistent with studies that have reported associations between drought and WNV-infected mosquitoes in urban and residential areas (Johnson and Sukhdeo, 2013; Paull et al., 2017). In addition, vertical transmission of WNV from gravid females to their progeny may occur during oviposition (Rosen, 1988), when the virus is transmitted by an accessory gland fluid that attaches eggs to one another (Nelms et al., 2013). Because the rate of vertical transmission in Cx. pipiens increases with the number of days following WNV infection (Anderson et al., 2008), extended searches for oviposition sites due to drought could increase the frequency of vertical transmission. However, the impact of vertical transmission on WNV epidemics is thought to be minimal because when transmission to an egg raft did occur, only 4.7% of the progeny were found to be infected as adults (Anderson et al., 2008), and only about half of those infected adults are estimated to be female. In summary, the movement of Cx. pipiens females toward more residential areas, combined with potential limited WNV amplification from increased vertical transmission, suggests that the vector trait of predator avoidance during oviposition can serve as a plausible explanation for associations between drought and human WNV cases.
Another theory for the association between drought and human WNV cases is based on the hypothesis that increased contact between mosquito vectors and passerine reservoir hosts occurs during drought conditions (Paull et al., 2017; Shaman et al., 2005). The proposed aggregation of bird and mosquito populations during drought was originally thought to occur in humid, densely vegetated hammocks – a type of habitat that is specific to southern Florida (Shaman et al., 2005), but WNV incidence is more consistently clustered in other regions of the US, particularly the Northern Great Plains (CENTERS FOR DISEASE CONTROL AND PREVENTION, 2021; Sugumaran et al., 2009). Northern cardinals (Cardinalis cardinalis), American robins (Turdus migratorius), and house sparrows (Passer domesticus) were among the bird species that most frequently tested seropositive for WNV antibodies in 2005 and 2006 in Chicago, where high numbers of human cases were reported (Hamer et al., 2008b), and these passerine species are more abundant in residential areas, regardless of precipitation patterns (Anderson, 2006b; Beddall, 1963; Lepczyk et al., 2008). Apart from drought, landowners’ participation in supplemental bird feeding, providing bird houses, gardening, and maintaining vegetation can strongly influence passerine abundance in residential areas (Lepczyk et al., 2004). Furthermore, as terrestrial foragers that can obtain hydration from their diet of insects, fruits, and other plant material (Anderson, 2006a; Brzek et al., 2009; Malmborg and Willson, 1988; Renne et al., 2000), passerine reservoir hosts of WNV are less likely to move in response to drought than the mosquito vectors of WNV, which have obligate aquatic life stages.
While hatch-year birds are more vulnerable to mosquito biting, and thus contribute to the amplification of WNV (Hamer et al., 2008b), it is illogical to expect an increased abundance of hatch-year birds during drought conditions. However, some have argued that in cases where drought decreases the abundance of juvenile birds, the ratio of mosquitoes to birds increases, and this could lead to higher WNV prevalence in the mosquito population (Paull et al., 2017). Although reductions in both hatching success (George et al., 1992) and survival of recently fledged birds (Yackel Adams et al., 2006) have been observed during drought conditions, the impact of drought on avian abundance varies widely by species (Verner and Purcell, 1999). In particular, synanthropic species, such as those likely to harbor WNV, are less negatively affected by drought (Albright et al., 2009). Additionally, the droughts that impact avian abundance often occur over much longer periods of time than the seasonal droughts that predict WNV transmission to humans. For example, avian abundance has been modeled based on precipitation metrics spanning 32 weeks, and house wren (Troglodytes aedon) abundance has been predicted by precipitation averages spanning four years (Verner and Purcell, 1999). Finally, birds with higher levels of stress hormones are more likely to be fed on by mosquitoes, and certain factors associated with residential areas, such as road noise, light pollution, and pesticide exposure, can cause avian stress (Gervasi et al., 2016). Therefore, elevated avian stress hormones in these habitats may contribute to WNV prevalence in the mosquito population, independent of drought conditions.
Implications for mosquito-borne disease modeling
Although the aquatic phase of the mosquito life cycle is often overlooked in mathematical models of mosquito-borne pathogen transmission (Reiner et al., 2013), vector survival at immature stages plays an important role in determining mosquito population abundance, which is an essential factor for predicting disease transmission (Beck-Johnson et al., 2013). The results of this study show that mosquito survival decreases among the Aedes, Anopheles, and Culex genera due to consumptive effects of predators (Figure 3b), and that there is also a reduction in mosquito survival due to non-consumptive effects. Other studies have demonstrated that aquatic predators dramatically impact mosquito survival and abundance. For example, a biocontrol intervention relying on the application of copepod predators eliminated Aedes albopictus from three communes in Nam Dinh, Vietnam, where dengue transmission was previously detected, and reduced vector abundance by 86–98% in three other communes (Kay et al., 2002). Conversely, the annual abundance of Culex and Anopheles mosquitoes was observed to increase 15-fold in semi-permanent wetlands in the year following a drought, likely because the drought eliminated aquatic predators from wetlands that dried completely, and mosquitoes were able to re-colonize newly formed aquatic habitats more quickly than their most effective predators (Chase and Knight, 2003).
While relationships between temperature and different vector traits, such as fecundity and lifespan, have been incorporated into models of temperature effects on mosquito population density (El Moustaid and Johnson, 2019), models of predator effects on vector borne disease transmission have focused primarily on the impacts of predation on vector survival. Previous models have shown that predators of vector species can decrease or eliminate pathogen infection in host populations as vector fecundity increases (Moore et al., 2010). The findings of this meta-analysis suggest that predators also decrease vector fecundity through non-consumptive effects on vector body size. In addition, the entomological inoculation rate (EIR) is likely to be reduced by effects of predators on mosquito fecundity and lifespan, as well as effects of predators on mosquito survival. The EIR has been defined as the product of three variables: (m) the number of mosquitoes per host, (a) the daily rate of mosquito biting, and (s) the proportion of mosquitoes that are infectious (Beck-Johnson et al., 2013). Based on this study’s findings, predators are likely to decrease the number of mosquitoes per host by reducing mosquito survival through both consumptive and non-consumptive effects, and by reducing mosquito fecundity through non-consumptive effects on body size. In addition, predators are likely to decrease the proportion of mosquitoes that are infectious by shortening the vector lifespan through non-consumptive effects on body size. The relationship between mosquito body size and biting rate is unclear, with some studies showing higher biting rates among larger mosquitoes (Araújo et al., 2012; Gunathilaka et al., 2019), and others reporting higher biting rates among smaller mosquitoes (Farjana and Tuno, 2013; Leisnham et al., 2008). The links between factors that influence the EIR and observed effects of predators on mosquito prey demonstrate the necessity of including both consumptive and non-consumptive effects of predators in models of mosquito-borne disease.
Conclusion
This meta-analysis on mosquito predation demonstrates that predators not only play an important role in directly reducing mosquito populations, but also have non-consumptive effects on surviving mosquitoes that may ultimately reduce further population growth and decrease disease transmission. While families of larger sized predators were effective in reducing mosquito survival, other factors, such as impacts on native species, as well as the economic cost of mass-rearing and field applications (Kumar and Hwang, 2006; Pyke, 2008), should be carefully considered before selecting a predator as a suitable biocontrol agent. Predictive disease models are likely to be more reliable when the non-consumptive effects of predation are incorporated. Although exposure of mosquito larvae to predators is commonplace in outdoor field settings, it remains rare in most laboratory-based assessments of vector traits. Therefore, mosquitoes observed in nature are likely to have smaller body sizes than those observed under optimal laboratory conditions. It is important for disease modelers to recognize these impacts of predation on vector traits as they can reduce mosquito population growth and limit disease transmission due to shorter vector lifespans. Within the WNV disease system, consideration of the oviposition behavioral response to predation cues by Culex vectors can improve current understanding of the association between drought and human cases. This study provides general estimates of the effects of predators on selected mosquito traits for use in predictive disease models.
Future directions
Modeling efforts that aim to optimize the application of biocontrol predators should also consider incorporating predator effects on vector survival, fecundity, and lifespan. These additions to predictive models of various biocontrol interventions are likely to help public health officials choose the most cost-effective strategies for limiting disease transmission. In the 60-study database that was compiled, only one study was designed to directly measure the effect of larval-stage predation on vector competence (Roux et al., 2015). Therefore, future efforts to assess the impact of predators on mosquito-borne disease transmission should prioritize experimental studies in which infected mosquito larvae are observed throughout an initial period of aquatic exposure to predators, followed by a period of blood-feeding in the adult stage.
Two studies from the compiled database examined the compatibility of predators with Bacillus thuringiensis var. israelensis (Bti), a commonly used bacterial biocontrol agent (Chansang et al., 2004; Op de Beeck et al., 2016). Previous studies have supported the simultaneous application of cyclopoid copepod predators and Bti (Marten et al., 1993; Tietze et al., 1994), but additional analyses are needed on the use of Bti with other families of mosquito predators. Populations of other insect pests, such as the southern green stink bug (Nezara viridula), are known to be regulated by both predators and parasites (Ehler, 2002). The literature search conducted for this meta-analysis returned studies on water mite parasites (Rajendran and Prasad, 1994) and nematode parasitoids (de Valdez, 2006) of mosquitoes, and ascogregarine parasites have previously been evaluated as biocontrol agents against Aedes mosquitoes (Tseng, 2007). A more thorough review of the impacts of parasites and parasitoids on vector traits, such as survival, fecundity, and lifespan, is needed before incorporating these potential biocontrol agents into integrated vector control plans.
Three studies in the 60-study database included experiments where two mosquito prey species were made available to the predator species (Grill and Juliano, 1996; Griswold and Lounibos, 2005, Micieli et al., 2002). In these cases, the effect size measurement for each mosquito species could be influenced by interspecific competition, or a preference of the predator species for a certain prey species. Hetero-specific prey observations were excluded from this meta-analysis, but future analyses centered on the concepts of interspecific competition or predator preferences might further evaluate these data. In addition, this meta-analysis investigated consumptive and non-consumptive effects of predators separately. More research is needed to determine how models should combine these different types of predator effects to accurately reflect predation interactions as they occur in natural environments.
Acknowledgements
We are grateful to Dr. Lauren Cator for facilitating the early stages of this project and helping to edit this manuscript. We also thank Dr. Peter Hudson for his helpful advice in early discussions about the project’s aims. In addition, we thank all members of the VectorBiTE RCN (https://vectorbite.org/) for taking the initiative to forge productive collaborations between empiricists and modelers in vector ecology. This work was funded by NIH grant 1R01AI122284-01 and BBSRC grant BB/N013573/1 as part of the joint (NIH-NSF-USDA-BBSRC) Ecology and Evolution of Infectious Diseases program. It was also funded by a President’s PhD Scholarship from Imperial College London awarded to Marie C Russell.
Funding Statement
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Contributor Information
Marie C Russell, Email: marie.clare.russell@gmail.com.
Sarah E Cobey, University of Chicago, United States.
Christian Rutz, University of St Andrews, United Kingdom.
Funding Information
This paper was supported by the following grants:
National Institutes of Health 1R01AI122284-01 to Zachary Gajewski, Fadoua El Moustaid.
Biotechnology and Biological Sciences Research Council BB/N013573/1 to Zachary Gajewski, Fadoua El Moustaid.
Imperial College London President's PhD Scholarship to Marie C Russell.
Additional information
Competing interests
No competing interests declared.
No competing interests declared.
Author contributions
Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Validation, Visualization, Writing – original draft, Writing – review and editing.
Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing – review and editing.
Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Validation, Writing – review and editing.
Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Writing – review and editing.
Conceptualization, Data curation, Writing – review and editing.
Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Validation, Writing – review and editing.
Conceptualization, Data curation, Visualization, Writing – review and editing.
Conceptualization, Data curation, Supervision, Writing – review and editing.
Conceptualization, Data curation, Supervision, Writing – review and editing.
Conceptualization, Supervision, Writing – review and editing.
Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing – review and editing.
Additional files
Data availability
The database can be accessed here: https://doi.org/10.5061/dryad.4qrfj6q9x. The R code file, showing all analyses, can be accessed here: https://doi.org/10.5281/zenodo.5790092.
The following dataset was generated:
Russell MC, Herzog CM, Gajewski Z, Ramsay C, El Moustaid F, Evans M, Desai T, Gottdenker N, Hermann S, Power A, McCall A. 2021. Both consumptive and non-consumptive effects of predators impact mosquito populations and have implications for disease transmission. Dryad Digital Repository.
References
- Albright TP, Pidgeon AM, Rittenhouse CD, Clayton MK, Flather CH, Culbert PD, Wardlow BD, Radeloff VC. Effects of drought on avian community structure. Global Change Biology. 2009;16:2158–2170. doi: 10.1111/j.1365-2486.2009.02120.x. [DOI] [Google Scholar]
- Alcalay Y, Tsurim I, Ovadia O. Female mosquitoes disperse further when they develop under predation risk. Behavioral Ecology. 2018;29:1402–1408. doi: 10.1093/beheco/ary113. [DOI] [Google Scholar]
- Amini M, Hanafi-Bojd AA, Aghapour AA, Chavshin AR. Larval habitats and species diversity of mosquitoes (Diptera: Culicidae) in West Azerbaijan Province, Northwestern Iran. BMC ecology. 2020;20:60. doi: 10.1186/s12898-020-00328-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amundrud SL, Clay-Smith SA, Flynn BL, Higgins KE, Reich MS, Wiens DRH, Srivastava DS. Drought alters the trophic role of an opportunistic generalist in an aquatic ecosystem. Oecologia. 2019;189:733–744. doi: 10.1007/s00442-019-04343-x. [DOI] [PubMed] [Google Scholar]
- Anderson TR. Chapter 6: Foraging Behavior and Food. Biology of the Ubiquitous House Sparrow: From Genes to Populations. New York: Oxford University Press; 2006a. [DOI] [Google Scholar]
- Anderson TR. Chapter 10: Human Commensalism and Pest Management. Biology of the Ubiquitous House Sparrow: From Genes to Populations. New York: Oxford University Press; 2006b. [DOI] [Google Scholar]
- Anderson JF, Main AJ, Delroux K, Fikrig E. Extrinsic incubation periods for horizontal and vertical transmission of West Nile virus by Culex pipiens pipiens (Diptera: Culicidae) Journal of medical entomology. 2008;45:445–451. doi: 10.1603/0022-2585(2008)45[445:eipfha]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- Andreadis TG. The contribution of Culex pipiens complex mosquitoes to transmission and persistence of West Nile virus in North America. Journal of the American Mosquito Control Association. 2012;28:137–151. doi: 10.2987/8756-971X-28.4s.137. [DOI] [PubMed] [Google Scholar]
- Araújo M, Gil LHS, e-Silva A. Larval food quantity affects development time, survival and adult biological traits that influence the vectorial capacity of Anopheles darlingi under laboratory conditions. Malaria Journal. 2012;11:261. doi: 10.1186/1475-2875-11-261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arav D, Blaustein L. Effects of pool depth and risk of predation on oviposition habitat selection by temporary pool dipterans. Journal of Medical Entomology. 2006;43:493–497. doi: 10.1603/0022-2585(2006)43[493:eopdar]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- Barr AR. Occurrence and distribution of the Culex pipiens complex. Bulletin of the World Health Organization. 1967;37:293–296. [PMC free article] [PubMed] [Google Scholar]
- Beck-Johnson LM, Nelson WA, Paaijmans KP, Read AF, Thomas MB, Bjørnstad ON. The effect of temperature on Anopheles mosquito population dynamics and the potential for malaria transmission. PLOS ONE. 2013;8:e79276. doi: 10.1371/journal.pone.0079276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beddall BG. Range expansion of the cardinal and other birds in the northeastern states. The Wilson Bulletin. 1963;75:140–158. [Google Scholar]
- Benelli G, Jeffries CL, Walker T. Biological Control of Mosquito Vectors: Past, Present, and Future. Insects. 2016;7:E52. doi: 10.3390/insects7040052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bentley MD, Day JF. Chemical ecology and behavioral aspects of mosquito oviposition. Annual review of entomology. 1989;34:401–421. doi: 10.1146/annurev.en.34.010189.002153. [DOI] [PubMed] [Google Scholar]
- Blackmore MS, Lord CC. The relationship between size and fecundity in Aedes albopictus. Journal of Vector Ecology. 2000;25:212–217. [PubMed] [Google Scholar]
- Bond JG, Arredondo-Jimenez JI, Rodriguez MH, Quiroz-Martinez H, Williams T. Oviposition habitat selection for a predator refuge and food source in a mosquito. Ecological Entomology. 2005;30:255–263. doi: 10.1111/j.0307-6946.2005.00704.x. [DOI] [Google Scholar]
- Brose U, Jonsson T, Berlow EL, Warren P, Banasek-Richter C, Bersier L-F, Blanchard JL, Brey T, Carpenter SR, Blandenier M-FC, Cushing L, Dawah HA, Dell T, Edwards F, Harper-Smith S, Jacob U, Ledger ME, Martinez ND, Memmott J, Mintenbeck K, Pinnegar JK, Rall BC, Rayner TS, Reuman DC, Ruess L, Ulrich W, Williams RJ, Woodward G, Cohen JE. Consumer-resource body-size relationships in natural food webs. Ecology. 2006;87:2411–2417. doi: 10.1890/0012-9658(2006)87[2411:cbrinf]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- Brown HE, Childs JE, Diuk-Wasser MA, Fish D. Ecological factors associated with West Nile virus transmission, northeastern United States. Emerging Infectious Diseases. 2008;14:1539–1545. doi: 10.3201/eid1410.071396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brzek P, Kohl K, Caviedes-Vidal E, Karasov WH. Developmental adjustments of house sparrow (Passer domesticus) nestlings to diet composition. The Journal of experimental biology. 2009;212:1284–1293. doi: 10.1242/jeb.023911. [DOI] [PubMed] [Google Scholar]
- Buchanan AL, Hermann SL, Lund M, Szendrei Z. A meta-analysis of non-consumptive predator effects in arthropods: the influence of organismal and environmental characteristics. Oikos. 2017;126:04384. doi: 10.1111/oik.04384. [DOI] [Google Scholar]
- Burnham KP, Anderson DR. Multimodel Inference: Understanding AIC and BIC in Model Selection. Sociological Methods & Research. 2004;33:261–304. doi: 10.1177/0049124104268644. [DOI] [Google Scholar]
- Cator LJ, Johnson LR, Mordecai EA, Moustaid FE, Smallwood TRC, LaDeau SL, Johansson MA, Hudson PJ, Boots M, Thomas MB, Power AG, Pawar S. The Role of Vector Trait Variation in Vector-Borne Disease Dynamics. Frontiers in ecology and evolution. 2020;8:189. doi: 10.3389/fevo.2020.00189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CENTERS FOR DISEASE CONTROL AND PREVENTION West Nile virus: Final Cumulative Maps & Data for 1999-2019. CDC. 2021 https://www.cdc.gov/westnile/statsmaps/cumMapsData.html
- Chansang UR, Bhumiratana A, Kittayapong P. Combination of Mesocyclops thermocyclopoides and Bacillus thuringiensis var. israelensis: a better approach for the control of Aedes aegypti larvae in water containers. Journal of Vector Ecology. 2004;29:218–226. [PubMed] [Google Scholar]
- Chase JM, Knight TM. Drought-induced mosquito outbreaks in wetlands. Ecology Letters. 2003;6:1017–1024. doi: 10.1046/j.1461-0248.2003.00533.x. [DOI] [Google Scholar]
- Ciota AT, Drummond CL, Ruby MA, Drobnack J, Ebel GD, Kramer LD. Dispersal of Culex mosquitoes (Diptera: Culicidae) from a wastewater treatment facility. Journal of medical entomology. 2012;49:35–42. doi: 10.1603/me11077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clements AN. The Biology of Mosquitoes: Development, Nutrition and Reproduction. Chapman & Hall; 1992. [Google Scholar]
- Dam HG, Peterson WT. The effect of temperature on the gut clearance rate constant of planktonic copepods. Journal of Experimental Marine Biology and Ecology. 1988;10:90105. doi: 10.1016/0022-0981(88)90105-0. [DOI] [Google Scholar]
- Day JF. Mosquito Oviposition Behavior and Vector Control. Insects. 2016;7:E65. doi: 10.3390/insects7040065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Valdez MRW. Parasitoid-induced behavioral alterations of Aedes aegypti mosquito larvae infected with mermithid nematodes (Nematoda: Mermithidae) Journal of vector ecology. 2006;31:344–354. doi: 10.3376/1081-1710(2006)31[344:pbaoaa]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- Delnat V, Tran TT, Janssens L, Stoks R. Resistance to a chemical pesticide increases vulnerability to a biopesticide: Effects on direct mortality and mortality by predation. Aquatic toxicology. 2019;216:105310. doi: 10.1016/j.aquatox.2019.105310. [DOI] [PubMed] [Google Scholar]
- Dida GO, Anyona DN, Abuom PO, Akoko D, Adoka SO, Matano A-S, Owuor PO, Ouma C. Spatial distribution and habitat characterization of mosquito species during the dry season along the Mara River and its tributaries, in Kenya and Tanzania. Infectious diseases of poverty. 2018;7:2. doi: 10.1186/s40249-017-0385-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dieng H, Boots M, Tuno N, Tsuda Y, Takagi M. A laboratory and field evaluation of Macrocyclops distinctus, Megacyclops viridis and Mesocyclops pehpeiensis as control agents of the dengue vector Aedes albopictus in a peridomestic area in Nagasaki, Japan. Medical and veterinary entomology. 2002;16:285–291. doi: 10.1046/j.1365-2915.2002.00377.x. [DOI] [PubMed] [Google Scholar]
- Dieng H, Boots M, Tuno N, Tsuda Y, Takagi M. Life history effects of prey choice by copepods: implications for biocontrol of vector mosquitoes. Journal of the American Mosquito Control Association. 2003;19:67–73. [PubMed] [Google Scholar]
- Dudgeon D. The effects of spate-induced disturbance, predation and environmental complexity on macroinvertebrates in a tropical stream. Freshwater Biology. 1993;30:189–197. doi: 10.1111/j.1365-2427.1993.tb00801.x. [DOI] [Google Scholar]
- Duval S, Tweedie R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000a;56:455–463. doi: 10.1111/j.0006-341x.2000.00455.x. [DOI] [PubMed] [Google Scholar]
- Duval S, Tweedie R. A Nonparametric “Trim and Fill” Method of Accounting for Publication Bias in Meta-Analysis. Journal of the American Statistical Association. 2000b;95:89–98. doi: 10.1080/01621459.2000.10473905. [DOI] [Google Scholar]
- Ehler LE. An evaluation of some natural enemies of >Nezara viridula in northern California. BioControl. 2002;47:309–325. doi: 10.1023/A:1014895028451. [DOI] [Google Scholar]
- El Moustaid F, Johnson LR. Modeling Temperature Effects on Population Density of the Dengue Mosquito Aedes aegypti. Insects. 2019;10:E393. doi: 10.3390/insects10110393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Englund G, Ohlund G, Hein CL, Diehl S. Temperature dependence of the functional response. Ecology letters. 2011;14:914–921. doi: 10.1111/j.1461-0248.2011.01661.x. [DOI] [PubMed] [Google Scholar]
- Epstein PR, Defilippo C. West Nile Virus and Drought. Global Change and Human Health. 2001;2:105–107. doi: 10.1023/A:1015089901425. [DOI] [Google Scholar]
- Farjana T, Tuno N. Multiple blood feeding and host-seeking behavior in Aedes aegypti and Aedes albopictus (Diptera: Culicidae) Journal of medical entomology. 2013;50:838–846. doi: 10.1603/me12146. [DOI] [PubMed] [Google Scholar]
- Fonseca DM, Keyghobadi N, Malcolm CA, Mehmet C, Schaffner F, Mogi M, Fleischer RC, Wilkerson RC. Emerging vectors in the Culex pipiens complex. Science. 2004;303:1535–1538. doi: 10.1126/science.1094247. [DOI] [PubMed] [Google Scholar]
- Gardner AM, Hamer GL, Hines AM, Newman CM, Walker ED, Ruiz MO. Weather variability affects abundance of larval Culex (Diptera: Culicidae) in storm water catch basins in suburban Chicago. Journal of medical entomology. 2012;49:270–276. doi: 10.1603/me11073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- George TL, Fowler AC, Knight RL, McEwen LC. Impacts of a Severe Drought on Grassland Birds in Western North Dakota. Ecological applications. 1992;2:275–284. doi: 10.2307/1941861. [DOI] [PubMed] [Google Scholar]
- Gervasi SS, Burkett-Cadena N, Burgan SC, Schrey AW, Hassan HK, Unnasch TR, Martin LB. Host stress hormones alter vector feeding preferences, success, and productivity. Proceedings. Biological Sciences. 2016;283:1278. doi: 10.1098/rspb.2016.1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grill CP, Juliano SA. Predicting Species Interactions Based on Behaviour: Predation and Competition in Container-Dwelling Mosquitoes. The Journal of Animal Ecology. 1996;65:63. doi: 10.2307/5700. [DOI] [Google Scholar]
- Griswold MW, Lounibos LP. Does differential predation permit invasive and native mosquito larvae to coexist in Florida? Ecological Entomology. 2005;30:122–127. doi: 10.1111/j.0307-6946.2005.00671.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunathilaka N, Upulika H, Udayanga L, Amarasinghe D. Effect of Larval Nutritional Regimes on Morphometry and Vectorial Capacity of Aedes aegypti for Dengue Transmission. BioMed Research International. 2019;2019:1–11. doi: 10.1155/2019/3607342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamer GL, Kitron UD, Brawn JD, Loss SR, Ruiz MO, Goldberg TL, Walker ED. Culex pipiens (Diptera: Culicidae): a bridge vector of West Nile virus to humans. Journal of Medical Entomology. 2008a;45:125–128. doi: 10.1603/0022-2585(2008)45[125:cpdcab]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- Hamer GL, Walker ED, Brawn JD, Loss SR, Ruiz MO, Goldberg TL, Schotthoefer AM, Brown WM, Wheeler E, Kitron UD. Rapid amplification of West Nile virus: the role of hatch-year birds. Vector Borne and Zoonotic Diseases. 2008b;8:57–67. doi: 10.1089/vbz.2007.0123. [DOI] [PubMed] [Google Scholar]
- Hamer GL, Anderson TK, Donovan DJ, Brawn JD, Krebs BL, Gardner AM, Ruiz MO, Brown WM, Kitron UD, Newman CM, Goldberg TL, Walker ED. Dispersal of adult culex mosquitoes in an urban west nile virus hotspot: a mark-capture study incorporating stable isotope enrichment of natural larval habitats. PLOS Neglected Tropical Diseases. 2014;8:e2768. doi: 10.1371/journal.pntd.0002768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hancock PA, Wiebe A, Gleave KA, Bhatt S, Cameron E, Trett A, Weetman D, Smith DL, Hemingway J, Coleman M, Gething PW, Moyes CL. Associated patterns of insecticide resistance in field populations of malaria vectors across Africa. PNAS. 2018;115:5938–5943. doi: 10.1073/pnas.1801826115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatala AJ, Harrington LC, Degner EC. Age and Body Size Influence Sperm Quantity in Male Aedes albopictus (Diptera: Culicidae) Mosquitoes. Journal of medical entomology. 2018;55:1051–1054. doi: 10.1093/jme/tjy040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hawley WA. The Effect of Larval Density on Adult Longevity of a Mosquito, Aedes sierrensis: Epidemiological Consequences. The Journal of Animal Ecology. 1985;54:955. doi: 10.2307/4389. [DOI] [Google Scholar]
- Hemingway J, Ranson H. Insecticide resistance in insect vectors of human disease. Annual review of entomology. 2000;45:371–391. doi: 10.1146/annurev.ento.45.1.371. [DOI] [PubMed] [Google Scholar]
- Hermann SL, Thaler JS. Prey perception of predation risk: volatile chemical cues mediate non-consumptive effects of a predator on a herbivorous insect. Oecologia. 2014;176:669–676. doi: 10.1007/s00442-014-3069-5. [DOI] [PubMed] [Google Scholar]
- Hermann SL, Landis DA. Scaling up our understanding of non-consumptive effects in insect systems. Current opinion in insect science. 2017;20:54–60. doi: 10.1016/j.cois.2017.03.010. [DOI] [PubMed] [Google Scholar]
- Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA. Cochrane Handbook for Systematic Reviews of Interventions. John Wiley & Sons, Inc; 2020. [DOI] [Google Scholar]
- Hill CA, Kafatos FC, Stansfield SK, Collins FH. Arthropod-borne diseases: vector control in the genomics era. Nature reviews. Microbiology. 2005;3:262–268. doi: 10.1038/nrmicro1101. [DOI] [PubMed] [Google Scholar]
- Janssens L, Stoks R. How does a pesticide pulse increase vulnerability to predation? Combined effects on behavioral antipredator traits and escape swimming. Aquatic toxicology. 2012;110–111:91–98. doi: 10.1016/j.aquatox.2011.12.019. [DOI] [PubMed] [Google Scholar]
- Jennions MD, Møller AP. Publication bias in ecology and evolution: an empirical assessment using the “trim and fill” method. Biological Reviews of the Cambridge Philosophical Society. 2002;77:211–222. doi: 10.1017/s1464793101005875. [DOI] [PubMed] [Google Scholar]
- Johnson BJ, Sukhdeo MVK. Drought-induced amplification of local and regional West Nile virus infection rates in New Jersey. Journal of Medical Entomology. 2013;50:195–204. doi: 10.1603/me12035. [DOI] [PubMed] [Google Scholar]
- Johnson B, Fonseca DM. The effects of forced-egg retention on the blood-feeding behavior and reproductive potential of Culex pipiens (Diptera: Culicidae) Journal of insect physiology. 2014;66:53–58. doi: 10.1016/j.jinsphys.2014.05.014. [DOI] [PubMed] [Google Scholar]
- Juliano SA. Species interactions among larval mosquitoes: context dependence across habitat gradients. Annual review of entomology. 2009;54:37–56. doi: 10.1146/annurev.ento.54.110807.090611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalinoski RM, DeLong JP. Beyond body mass: how prey traits improve predictions of functional response parameters. Oecologia. 2016;180:543–550. doi: 10.1007/s00442-015-3487-z. [DOI] [PubMed] [Google Scholar]
- Kay BH, Nam VS, Tien TV, Yen NT, Phong TV, Diep VTB, Ninh TU, Bektas A, Aaskov JG. Control of aedes vectors of dengue in three provinces of Vietnam by use of Mesocyclops (Copepoda) and community-based methods validated by entomologic, clinical, and serological surveillance. The American journal of tropical medicine and hygiene. 2002;66:40–48. doi: 10.4269/ajtmh.2002.66.40. [DOI] [PubMed] [Google Scholar]
- Kershenbaum A, Spencer M, Blaustein L, Cohen JE. Modelling evolutionarily stable strategies in oviposition site selection, with varying risks of predation and intraspecific competition. Evolutionary Ecology. 2012;26:955–974. doi: 10.1007/s10682-011-9548-9. [DOI] [Google Scholar]
- Kleckner CA, Hawley WA, Bradshaw WE, Holzapfel CM, Fisher IJ. Protandry in aedes sierrensis - the significance of temporal variation in female fecundityROTANDRY IN AEDES SIERRENSIS - THE SIGNIFICANCE OF TEMPORAL VARIATION IN FEMALE FECUNDITY. Ecology. 1995;76:1242–1250. [Google Scholar]
- Kramer LD, Styer LM, Ebel GD. A global perspective on the epidemiology of West Nile virus. Annual review of entomology. 2008;53:61–81. doi: 10.1146/annurev.ento.53.103106.093258. [DOI] [PubMed] [Google Scholar]
- Kumar R, Hwang JS. Larvicidal efficiency of aquatic predators: A perspective for mosquito biocontrol. Zoological Studies. 2006;45:447–466. [Google Scholar]
- Kumar R, Muhid P, Dahms HU, Tseng LC, Hwang JS. Potential of three aquatic predators to control mosquitoes in the presence of alternative prey: a comparative experimental assessment. Marine and Freshwater Research. 2008;59:817. doi: 10.1071/MF07143. [DOI] [Google Scholar]
- Kweka EJ, Zhou G, Gilbreath TM, Afrane Y, Nyindo M, Githeko AK, Yan G. Predation efficiency of Anopheles gambiae larvae by aquatic predators in western Kenya highlands. Parasites & Vectors. 2011;4:128. doi: 10.1186/1756-3305-4-128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lajeunesse MJ. Facilitating systematic reviews, data extraction and meta‐analysis with the metagear package for R. Methods in Ecology and Evolution. 2016;7:323–330. doi: 10.1111/2041-210X.12472. [DOI] [Google Scholar]
- Leisnham PT, Sala LM, Juliano SA. Geographic variation in adult survival and reproductive tactics of the mosquito Aedes albopictus. Journal of Medical Entomology. 2008;45:210–221. doi: 10.1603/0022-2585(2008)45[210:gviasa]2.0.co;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lepczyk CA, Mertig AG, Liu J. Assessing landowner activities related to birds across rural-to-urban landscapes. Environmental management. 2004;33:110–125. doi: 10.1007/s00267-003-0036-z. [DOI] [PubMed] [Google Scholar]
- Lepczyk CA, Flather CH, Radeloff VC, Pidgeon AM, Hammer RB, Liu J. Human impacts on regional avian diversity and abundance. Conservation biology. 2008;22:405–416. doi: 10.1111/j.1523-1739.2008.00881.x. [DOI] [PubMed] [Google Scholar]
- Lima SL, Dill LM. Behavioral decisions made under the risk of predation: a review and prospectus. Canadian Journal of Zoology. 1990;68:619–640. doi: 10.1139/z90-092. [DOI] [Google Scholar]
- Lima SL. Nonlethal Effects in the Ecology of Predator-Prey Interactions: What are the ecological effects of anti-predator decision-making. Bioscience. 1998;48:25–34. doi: 10.2307/1313225. [DOI] [Google Scholar]
- Liu N. Insecticide resistance in mosquitoes: impact, mechanisms, and research directions. Annual Review of Entomology. 2015;60:537–559. doi: 10.1146/annurev-ento-010814-020828. [DOI] [PubMed] [Google Scholar]
- Lühken R, Steinke S, Leggewie M, Tannich E, Krüger A, Becker S, Kiel E. Physico-Chemical Characteristics of Culex pipiens sensu lato and Culex torrentium (Diptera: Culicidae) Breeding Sites in Germany. Journal of medical entomology. 2015;52:932–936. doi: 10.1093/jme/tjv070. [DOI] [PubMed] [Google Scholar]
- Lyimo EO, Takken W. Effects of adult body size on fecundity and the pre-gravid rate of Anopheles gambiae females in Tanzania. Medical and veterinary entomology. 1993;7:328–332. doi: 10.1111/j.1365-2915.1993.tb00700.x. [DOI] [PubMed] [Google Scholar]
- Malmborg PK, Willson MF. Foraging ecology of avian frugivores and some consequences for seed dispersal in an illinois woodlot. The Condor. 1988;90:173–186. doi: 10.2307/1368446. [DOI] [Google Scholar]
- Marcantonio M, Rizzoli A, Metz M, Rosà R, Marini G, Chadwick E, Neteler M. Identifying the environmental conditions favouring West Nile Virus outbreaks in Europe. PLOS ONE. 2015;10:e0121158. doi: 10.1371/journal.pone.0121158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marten GG. Elimination of Aedes albopictus from tire piles by introducing Macrocyclops albidus (Copepoda, Cyclopidae) Journal of the American Mosquito Control Association. 1990;6:689–693. [PubMed] [Google Scholar]
- Marten GG, Che W, Bordes ES. Compatibility of cyclopoid copepods with mosquito insecticides. Journal of the American Mosquito Control Association. 1993;9:150–154. [PubMed] [Google Scholar]
- Marten GG, Reid JW. Cyclopoid copepods. Journal of the American Mosquito Control Association. 2007;23:65–92. doi: 10.2987/8756-971X(2007)23[65:CC]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- McCauley SJ, Rowe L, Fortin M-J. The deadly effects of “nonlethal” predators. Ecology. 2011;92:2043–2048. doi: 10.1890/11-0455.1. [DOI] [PubMed] [Google Scholar]
- Micieli MV, Marti G, García JJ. Laboratory evaluation of Mesocyclops annulatus (Wierzejski, 1892) (Copepoda: Cyclopidea) as a predator of container-breeding mosquitoes in Argentina. Memorias do Instituto Oswaldo Cruz. 2002;97:835–838. doi: 10.1590/s0074-02762002000600014. [DOI] [PubMed] [Google Scholar]
- Mittelbach GG. Foraging Efficiency and Body Size: A Study of Optimal Diet and Habitat Use by Bluegills. Ecology. 1981;62:1370–1386. doi: 10.2307/1937300. [DOI] [Google Scholar]
- Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535. doi: 10.1136/bmj.b2535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore SM, Borer ET, Hosseini PR. Predators indirectly control vector-borne disease: linking predator-prey and host-pathogen models. Journal of the Royal Society, Interface. 2010;7:161–176. doi: 10.1098/rsif.2009.0131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelms BM, Fechter-Leggett E, Carroll BD, Macedo P, Kluh S, Reisen WK. Experimental and natural vertical transmission of West Nile virus by California Culex (Diptera: Culicidae) mosquitoes. Journal of medical entomology. 2013;50:371–378. doi: 10.1603/me12264. [DOI] [PubMed] [Google Scholar]
- Nelson EH, Matthews CE, Rosenheim JA. Predators reduce prey population growth by inducing changes in prey behaviorREDATORS REDUCE PREY POPULATION GROWTH BY INDUCING CHANGES IN PREY BEHAVIOR. Ecology. 2004;85:1853–1858. [Google Scholar]
- Nikookar SH, Fazeli-Dinan M, Azari-Hamidian S, Mousavinasab SN, Arabi M, Ziapour SP, Shojaee J, Enayati A. Species composition and abundance of mosquito larvae in relation with their habitat characteristics in Mazandaran Province, northern Iran. Bulletin of entomological research. 2017;107:598–610. doi: 10.1017/S0007485317000074. [DOI] [PubMed] [Google Scholar]
- Oliver J, Howard JJ. Fecundity of wild-caught gravid Culiseta morsitans (Diptera: Culicidae) Journal of medical entomology. 2011;48:196–201. doi: 10.1603/me10095. [DOI] [PubMed] [Google Scholar]
- Op de Beeck L, Janssens L, Stoks R. Synthetic predator cues impair immune function and make the biological pesticide Bti more lethal for vector mosquitoes. Ecological Applications. 2016;26:355–366. doi: 10.1890/15-0326. [DOI] [PubMed] [Google Scholar]
- Ower GD, Juliano SA. The demographic and life-history costs of fear: Trait-mediated effects of threat of predation on Aedes triseriatus. Ecology and evolution. 2019;9:3794–3806. doi: 10.1002/ece3.5003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Meara GF. Variable expressions of autogeny in three mosquito species. International Journal of Invertebrate Reproduction. 1979;1:253–261. doi: 10.1080/01651269.1979.10553321. [DOI] [Google Scholar]
- Paull SH, Horton DE, Ashfaq M, Rastogi D, Kramer LD, Diffenbaugh NS, Kilpatrick AM. Drought and immunity determine the intensity of West Nile virus epidemics and climate change impacts. Proceedings. Biological Sciences. 2017;284:284. doi: 10.1098/rspb.2016.2078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peacor SD, Werner EE. The contribution of trait-mediated indirect effects to the net effects of a predator. PNAS. 2001;98:3904–3908. doi: 10.1073/pnas.071061998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters RH. The Ecological Implications of Body Size. Cambridge University Press; 1983. [DOI] [Google Scholar]
- Ponlawat A, Harrington LC. Age and body size influence male sperm capacity of the dengue vector Aedes aegypti (Diptera: Culicidae) Journal of medical entomology. 2007;44:422–426. doi: 10.1603/0022-2585(2007)44[422:aabsim]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- Preisser EL, Bolnick DI, Benard MF. Scared to death? the effects of intimidation and consumption in predator–prey interactions. Ecology. 2005;86:501–509. doi: 10.1890/04-0719. [DOI] [Google Scholar]
- Pyke GH. Plague Minnow or Mosquito Fish? A Review of the Biology and Impacts of Introduced Gambusia Species. Annual Review of Ecology, Evolution, and Systematics. 2008;39:171–191. doi: 10.1146/annurev.ecolsys.39.110707.173451. [DOI] [Google Scholar]
- R Development Core Team . Vienna, Austria: R Foundation for Statistical Computing; 2020. http://www.r-project.org [Google Scholar]
- Rajendran R, Prasad RS. A laboratory study on the life cycle and feeding behaviour of Arrenurus madaraszi (Acari: Arrenuridae) parasitizing Anopheles mosquitoes. Annals of tropical medicine and parasitology. 1994;88:169–174. doi: 10.1080/00034983.1994.11812855. [DOI] [PubMed] [Google Scholar]
- Reiner RC, Perkins TA, Barker CM, Niu T, Chaves LF, Ellis AM, George DB, Le Menach A, Pulliam JRC, Bisanzio D, Buckee C, Chiyaka C, Cummings DAT, Garcia AJ, Gatton ML, Gething PW, Hartley DM, Johnston G, Klein EY, Michael E, Lindsay SW, Lloyd AL, Pigott DM, Reisen WK, Ruktanonchai N, Singh BK, Tatem AJ, Kitron U, Hay SI, Scott TW, Smith DL. A systematic review of mathematical models of mosquito-borne pathogen transmission: 1970-2010. Journal of the Royal Society, Interface. 2013;10:20120921. doi: 10.1098/rsif.2012.0921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reisen WK, Milby MM, Bock ME. The effects of immature stress on selected events in the life-history of culex-tarsalisHE EFFECTS OF IMMATURE STRESS ON SELECTED EVENTS IN THE LIFE-HISTORY OF CULEX-TARSALIS. Mosquito News. 1984;44:385–395. [Google Scholar]
- Reiskind MH, Lounibos LP. Effects of intraspecific larval competition on adult longevity in the mosquitoes Aedes aegypti and Aedes albopictus. Medical and veterinary entomology. 2009;23:62–68. doi: 10.1111/j.1365-2915.2008.00782.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renne IJ, Gauthreaux SA, Gresham CA. Seed Dispersal of the Chinese Tallow Tree (Sapium sebiferum (L.) Roxb.) by Birds in Coastal South Carolina. The American Midland Naturalist. 2000;144:202–215. doi: 10.1674/0003-0031(2000)144[0202:SDOTCT]2.0.CO;2. [DOI] [Google Scholar]
- Roberts D. Responses of three species of mosquito larvae to the presence of predatory dragonfly and damselfly larvae. Entomologia Experimentalis et Applicata. 2012;145:23–29. doi: 10.1111/j.1570-7458.2012.01300.x. [DOI] [Google Scholar]
- Roehr B. US hit by massive West Nile virus outbreak centred around Texas. British Medical Journal. 2012;345:e5633. doi: 10.1136/bmj.e5633. [DOI] [PubMed] [Google Scholar]
- Rohatgi A. WebPlotDigitizer. 2020. [July 14, 2020]. https://automeris.io/WebPlotDigitizer
- Rosen L. Further observations on the mechanism of vertical transmission of flaviviruses by Aedes mosquitoes. The American journal of tropical medicine and hygiene. 1988;39:123–126. doi: 10.4269/ajtmh.1988.39.123. [DOI] [PubMed] [Google Scholar]
- Roux O, Diabaté A, Simard F. Divergence in threat sensitivity among aquatic larvae of cryptic mosquito species. The Journal of animal ecology. 2014;83:702–711. doi: 10.1111/1365-2656.12163. [DOI] [PubMed] [Google Scholar]
- Roux O, Vantaux A, Roche B, Yameogo KB, Dabiré KR, Diabaté A, Simard F, Lefèvre T. Evidence for carry-over effects of predator exposure on pathogen transmission potential. Proceedings. Biological Sciences. 2015;282:20152430. doi: 10.1098/rspb.2015.2430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell BM, Muir LE, Weinstein P, Kay BH. Surveillance of the mosquito Aedes aegypti and its biocontrol with the copepod Mesocyclops aspericornis in Australian wells and gold mines. Medical and veterinary entomology. 1996;10:155–160. doi: 10.1111/j.1365-2915.1996.tb00722.x. [DOI] [PubMed] [Google Scholar]
- Seale A. The Mosquito Fish, Gambusia affinis (Baird and Girard), in the Philippine Islands. Philippine Journal of Science. 1917;12:177–187. [Google Scholar]
- Shaman J, Day JF, Stieglitz M. Drought-induced amplification and epidemic transmission of West Nile virus in southern Florida. Journal of medical entomology. 2005;42:134–141. doi: 10.1093/jmedent/42.2.134. [DOI] [PubMed] [Google Scholar]
- Shi L, Lin L. The trim-and-fill method for publication bias: practical guidelines and recommendations based on a large database of meta-analyses. Medicine. 2019;98:e15987. doi: 10.1097/MD.0000000000015987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sih A. Antipredator Responses and the Perception of Danger by Mosquito Larvae. Ecology. 1986;67:434–441. doi: 10.2307/1938587. [DOI] [Google Scholar]
- Silberbush A, Blaustein L. Mosquito females quantify risk of predation to their progeny when selecting an oviposition site. Functional Ecology. 2011;25:1091–1095. doi: 10.1111/j.1365-2435.2011.01873.x. [DOI] [Google Scholar]
- Soumare MKF, Cilek JE. The effectiveness of Mesocyclops longisetus (Copepoda) for the control of container-inhabiting mosquitoes in residential environments. Journal of the American Mosquito Control Association. 2011;27:376–383. doi: 10.2987/11-6129.1. [DOI] [PubMed] [Google Scholar]
- Spencer M, Blaustein L, Cohen JE. Oviposition habitat selection by mosquitoes (culiseta longiareolata) and consequences for population size. Ecology. 2002;83:669–679. doi: 10.1890/0012-9658(2002)083[0669:OHSBMC]2.0.CO;2. [DOI] [Google Scholar]
- Stav G, Blaustein L, Margalit Y. Individual and interactive effects of a predator and controphic species on mosquito populations. Ecological Applications. 2005;15:587–598. doi: 10.1890/03-5191. [DOI] [Google Scholar]
- Sterne JA, Egger M. Funnel plots for detecting bias in meta-analysis: guidelines on choice of axis. Journal of clinical epidemiology. 2001;54:1046–1055. doi: 10.1016/s0895-4356(01)00377-8. [DOI] [PubMed] [Google Scholar]
- Styer LM, Meola MA, Kramer LD. West Nile virus infection decreases fecundity of Culex tarsalis females. Journal of medical entomology. 2007;44:1074–1085. doi: 10.1603/0022-2585(2007)44[1074:wnvidf]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- Su T, Mulla MS. Physiological aspects of autogeny in Culex tarsalis (Diptera: Culicidae): influences of sugar-feeding, mating, body weight, and wing length. Journal of Vector Ecology. 1997;22:115–121. [PubMed] [Google Scholar]
- Sugumaran R, Larson SR, Degroote JP. Spatio-temporal cluster analysis of county-based human West Nile virus incidence in the continental United States. International Journal of Health Geographics. 2009;8:43. doi: 10.1186/1476-072X-8-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sulesco TM, Toderas LG, Uspenskaia IG, Toderas IK. Larval Habitats Diversity and Distribution of the Mosquito (Diptera: Culicidae) Species in the Republic of Moldova. Journal of medical entomology. 2015;52:1299–1308. doi: 10.1093/jme/tjv142. [DOI] [PubMed] [Google Scholar]
- Thapa R, Mirsky SB, Tully KL. Cover Crops Reduce Nitrate Leaching in Agroecosystems:A Global Meta-Analysis. Journal of environmental quality. 2018;47:1400–1411. doi: 10.2134/jeq2018.03.0107. [DOI] [PubMed] [Google Scholar]
- Tietze NS, Hester PG, Shaffer KR, Prescott SJ, Schreiber ET. Integrated management of waste tire mosquitoes utilizing Mesocyclops longisetus (Copepoda: Cyclopidae), Bacillus thuringiensis var. israelensis, Bacillus sphaericus, and methoprene. Journal of the American Mosquito Control Association. 1994;10:363–373. [PubMed] [Google Scholar]
- Townroe S, Callaghan A. British container breeding mosquitoes: the impact of urbanisation and climate change on community composition and phenology. PLOS ONE. 2014;9:e95325. doi: 10.1371/journal.pone.0095325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tseng M. Ascogregarine parasites as possible biocontrol agents of mosquitoes. Journal of the American Mosquito Control Association. 2007;23:30–34. doi: 10.2987/8756-971X(2007)23[30:APAPBA]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- Tsunoda T, Fukuchi A, Nanbara S, Takagi M. Effect of body size and sugar meals on oviposition of the yellow fever mosquito, Aedes aegypti (Diptera: Culicidae) Journal of vector ecology. 2010;35:56–60. doi: 10.1111/j.1948-7134.2010.00028.x. [DOI] [PubMed] [Google Scholar]
- Uiterwaal SF, Delong JP. Functional responses are maximized at intermediate temperatures. Ecology. 2020;101:2975. doi: 10.1002/ecy.2975. [DOI] [PubMed] [Google Scholar]
- Verna TN. Species Composition and Seasonal Distribution of Mosquito Larvae (Diptera: Culicidae) in Southern New Jersey, Burlington County. Journal of medical entomology. 2015;52:1165–1169. doi: 10.1093/jme/tjv074. [DOI] [PubMed] [Google Scholar]
- Verner J, Purcell KL. Fluctuating Populations of House Wrens and Bewick’s Wrens in Foothills of the Western Sierra Nevada of California. The Condor. 1999;101:219–229. doi: 10.2307/1369985. [DOI] [Google Scholar]
- Veronesi R, Carrieri M, Maccagnani B, Maini S, Bellini R. Macrocyclops albidus (Copepoda: cyclopidae) for the Biocontrol of Aedes albopictus and Culex pipiens in Italy. Journal of the American Mosquito Control Association. 2015;31:32–43. doi: 10.2987/13-6381.1. [DOI] [PubMed] [Google Scholar]
- Viechtbauer W. Conducting Meta-Analyses in R with the metafor Package. Journal of Statistical Software. 2010;36:1–48. doi: 10.18637/jss.v036.i03. [DOI] [Google Scholar]
- Vincze E, Seress G, Lagisz M, Dinemanse N, Sprau P. Does Urbanization Affect Predation of Bird Nests? A Meta-Analysis. Frontiers in Ecology and Evolution. 2017;5:29. doi: 10.3389/fevo.2017.00029. [DOI] [Google Scholar]
- Vinogradova EB. Culex Pipiens Pipiens Mosquitoes: Taxonomy, Distribution, Ecology, Physiology, Genetics, Applied Importance and Control. Pensoft; 2000. [Google Scholar]
- Vonesh JR, Blaustein L. Predator-Induced Shifts in Mosquito Oviposition Site Selection: A Meta-Analysis and Implications for Vector Control. Israel Journal of Ecology and Evolution. 2010;56:263–279. doi: 10.1560/IJEE.56.3-4.263. [DOI] [Google Scholar]
- Weaver SC, Reisen WK. Present and future arboviral threats. Antiviral research. 2010;85:328–345. doi: 10.1016/j.antiviral.2009.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WORLD HEALTH ORGANIZATION Vector-borne diseases. 2020. [August 2, 2021]. https://www.who.int/news-room/fact-sheets/detail/vector-borne-diseases
- Xue R-D, Barnard DR, Muller GC. Effects of body size and nutritional regimen on survival in adult Aedes albopictus (Diptera: Culicidae) Journal of medical entomology. 2010;47:778–782. doi: 10.1603/me09222. [DOI] [PubMed] [Google Scholar]
- Yackel Adams AA, Skagen SK, Savidge JA. Modeling post-fledging survival of Lark Buntings in response to ecological and biological factors. Ecology. 2006;87:178–188. doi: 10.1890/04-1922. [DOI] [PubMed] [Google Scholar]
- Zuharah WF, Fadzly N, Lester PJ. Lethal and sublethal impacts of predaceous backswimmer Anisops wakefieldi (Hemiptera: Notonectidae) on the life-history traits of the New Zealand mosquito Culex pervigilans (Diptera: Culicidae) Journal of Medical Entomology. 2013;50:1014–1024. doi: 10.1603/me12136. [DOI] [PubMed] [Google Scholar]