
Keywords: brain-derived neurotrophic factor, differentiation, electroacupuncture, motor function, neural/glial antigen 2, perilesional striatum, stroke, survival
Abstract
Neural/glial antigen 2 (NG2)-expressing cells has multipotent stem cell activity under cerebral ischemia. Our study examined the effects of electroacupuncture (EA) therapy (2 Hz, 1 or 3 mA, 20 minutes) at the Sishencong acupoint on motor function after ischemic insult in the brain by investigating the rehabilitative potential of NG2-derived cells in a mouse model of ischemic stroke. EA stimulation alleviated motor deficits caused by ischemic stroke, and 1 mA EA stimulation was more efficacious than 3 mA EA stimulation or positive control treatment with edaravone, a free radical scavenger. The properties of NG2-expressing cells were altered with 1 mA EA stimulation, enhancing their survival in perilesional brain tissue via reduction of tumor necrosis factor alpha expression. EA stimulation robustly activated signaling pathways related to proliferation and survival of NG2-expressing cells and increased the expression of neurotrophic factors such as brain-derived neurotrophic factor, tumor growth factor beta, and neurotrophin 3. In the perilesional striatum, EA stimulation greatly increased the number of NG2-expressing cells double-positive for oligodendrocyte, endothelial cell, and microglia/macrophage markers (CC1, CD31, and CD68). EA therapy also greatly activated brain-derived neurotrophic factor/tropomyosin receptor kinase B and glycogen synthase kinase 3 beta signaling. Our results indicate that EA therapy may prevent functional loss at the perilesional site by enhancing survival and differentiation of NG2-expressing cells via the activation of brain-derived neurotrophic factor -induced signaling, subsequently ameliorating motor dysfunction. The animal experiments were approved by the Animal Ethics Committee of Pusan National University (approval Nos. PNU2019-2199 and PNU2019-2884) on April 8, 2019 and June 19, 2019.
Chinese Library Classification No. R459.9; R364; R741
Introduction
Most stroke survivors suffer from long-term disabilities such as motor impairments, and there is no novel treatment to optimize neural functional recovery; thus, stroke survivors generally rely on rehabilitation therapy (Lindvall and Kokaia, 2011). In ischemic stroke, almost all neurons in the ischemic core, where the blood flow declines precipitously, undergo degeneration within a few days after injury (Dirnagl et al., 1999). The ischemic tissue in the perilesional zone, adjacent to the core, is not yet irreversibly injured; therefore, this region is a therapeutic target for optimizing functional recovery (Sugimoto et al., 2014).
Neural/glial antigen 2 (NG2)-expressing cells are characterized by the expression of chondroitin sulfate proteoglycan 4, an integral membrane glycoprotein (Levine et al., 2001). Although NG2-expressing cells have been recognized as a fourth glial cell type that retains its proliferative ability in the central nervous system (CNS) throughout an organism's life (Nishiyama et al., 2009; Dimou and Gallo., 2015; Jin et al., 2018), this cell type is highly heterogeneous with diverse properties and functional phenotypes. This suggests these cells play a more complex role rather than merely acting as oligodendrocyte precursors or NG2 glial cells (Levine et al., 2001; Dimou and Gallo., 2015; Nakano et al., 2017). NG2 is expressed in a variety of cells derived from non-neural lineages, vasculature-associated cells, including pericyte/smooth muscle and endothelial cells, and microglia/macrophages in the CNS (Smirkin et al., 2010; Sugimoto et al., 2014; Stallcup et al., 2016; Kyyriainen et al., 2017; Jin et al., 2018).
NG2-expressing cells play crucial roles in cerebral ischemia, displaying the potential to proliferate and differentiate in response to ischemic injuries (Zhang et al., 2013; Honsa et al., 2016). The number of NG2-expressing cells is markedly decreased in the infarct core, but their numbers increase in perilesional brain tissue (penumbra). Therefore, the decline of these cells contributes to susceptibility to damage following ischemic brain injuries (Wang et al., 2011). NG2-expressing glia act as a major component of oligodendrocyte generation by proliferation around the lesion site (Levine et al., 2001; Hackett et al., 2016; Gotoh et al., 2018). Expression of NG2 in neurovascular cells and microglia/macrophages enhances proliferation and induces morphological changes, exhibiting a close spatiotemporal relationship with changes in the lesion core and within perilesional brain tissue (Matsumoto et al., 2008; Smirkin et al., 2010; Jin et al., 2018). Furthermore, NG2 proteoglycan regulates diverse cellular functions that include cell proliferation, migration, and differentiation in response to stimulation by growth factors (Biname, 2014).
Acupuncture has beneficial effects in the treatment of stroke patients by improving specific functional impairments without serious adverse events (Yang et al., 2016). Electroacupuncture (EA) involves repeated stimulations of sensory systems by electrical pulses instead of traditional manual manipulation (Soligo et al., 2013, 2017). The underlying therapeutic mechanism of EA may involve activity-dependent regulation of neurotransmitters, cytokines, and neurotrophic factors via the modulation of sensory and autonomic nervous system activity (Manni et al., 2010; Soligo et al., 2013). Because NG2-expressing cells display multipotent stem cell activity under cerebral ischemia (Nishiyama et al., 2009; Zhang et al., 2013; Honsa et al., 2016), EA stimulation may promote the proliferation and differentiation of these cells via the activation of neurotrophic factors (Kim et al., 2014, 2018; Ahn et al., 2019). We hypothesized that EA therapy would enhance the activation potential of NG2-expressing cells in the perilesional zone of the brain following ischemic stroke via modulation of the expression of neurotrophic factors and related signaling, accordingly leading to the recovery of motor functional deficits.
Materials and Methods
Animals
Forty-two 8-week-old male C57BL/6 mice, weighing 18–22 g, were purchased from Dooyeol Biotech (Seoul, Korea). Six-week-old male (n = 2) and female (n = 4) NG2-membrane-anchored enhanced green fluorescent protein (mEGFP) (FVB. Cg-Tg[Cspg4-EGFP*]HDbe/J) mice, weighing 16–18 g were purchased from Jackson Laboratory (#022735, Bar Harbor, ME, USA). The room where the mice lived was controlled at 22°C with a 12-hour dark/light cycle. Breeding and generation conditions of NG2-mEGFP mice were maintained, and genotyping was performed by reverse transcription polymerase chain reaction with the following primers (5′–3′) to determine hemizygosity of the NG2 gene: F-CGT GTG TGA GAA CTG CCA TT; R-GTT GTG GCG GAT CTT GAA GT. The following internal, positive control was used for the mouse primers: F-CTA GGC CAC AGA ATT GAA AGA TCT; R-GTA GGT GGA AAT TCT AGC ATC C. The sample size was determined based on our previous results of post-stroke motor function test performance after experimental stroke from the G-power program (G*Power 3.1 software; http://www.gpower.hhu.de/). For each of the five groups, the expected sample size for this study was expected to be n = 5 per group (a fixed effect, omnibus, one-way analysis of variance (ANOVA) with five groups was performed, with an effect size f = 0.8, at α = 0.05 and β = 0.2). Consequently, group sizes of ≥ 5 (range, 5–6) were used in this study. After randomization, the five groups consisted of a control group without middle cerebral artery occlusion (MCAO), an MCAO group (MCAO surgery with sham stimulation), an MCAO + EA1 group (MCAO surgery with 1 mA EA stimulation), an MCAO + EA3 group (MCAO surgery with 3 mA EA stimulation), and an MCAO + Edaravone group (positive control, MCAO surgery with edaravone treatment). In total, 52 mice (C57BL/6J mice: n = 42, NG2-mEGFP mice: n = 10) were used in this study. All experiments were approved by the Animal Ethics Committee of Pusan National University (Approval Nos. PNU2019-2199 and PNU2019-2884) on April 8, 2019 and June 19, 2019.
MCAO model
A silicon coated 7-0 monofilament (Docol Corporation, Sharon, MA, USA) was inserted into the middle cerebral artery through the internal carotid artery in C57BL/6J and NG2-mEGFP mice. After 40 minutes, the filament was removed, and reperfusion was induced. The wounds were then carefully sutured. The regional cerebral blood flow was checked using the PeriFlux Laser Doppler System 5000 (Perimed, Stockholm, Sweden) attached to the skulls. Using a calibrated vaporizer (Midmark NIP 3000, Orchard, OH, USA), the mice were anesthetized under 20% O2 and 80% N2O with 2% isoflurane (200 mL/minute O2, 800 mL/minute N2O; VSP corporation, Choongwae, Seoul, Korea) during surgery.
EA stimulation
Four needles (0.18 mm in diameter, Dongbang Medical Co., Ltd., Boryeong, Korea) were inserted into the Sishencong (EX-HN1) acupoint of each mouse at a depth of 2 mm. The four points of Sishencong were located anteriorly, posteriorly, and laterally, 2 mm from Baihui (GV20, at the intersection point of the median and the line connecting the ends of the two ears). The mice received a frequency of 2 Hz and an intensity of either 1 mA (MCAO + EA1) or 3 mA (MCAO + EA3) for 20 minutes with an electrical stimulator (Pulsemaster Multichannel Stimulator SYS-A300, World Precision Instruments, Berlin, Germany) while they were under isoflurane anesthesia. The control and non-EA groups (i.e., the MCAO and MCAO + Edaravone groups) were administered isoflurane for equivalent amounts of time without EA stimulation. Mice were randomized to receive either three or six sessions of EA stimulation once every 2 days before the behavioral test.
Bromodeoxyuridine labeling and Edaravone treatment
To label proliferating cells incorporated into the DNA of dividing cells during the S-phase of the cell cycle, mice received an intraperitoneal injection of bromodeoxyuridine (BrdU, 50 mg/kg, Sigma-Aldrich, Merck KGaA, Darmstadt, Germany). The mice received the injection once a day for 6 successive days, starting 5 days after the MCAO surgery. As a positive control, the mice in the MCAO + Edaravone group were also intraperitoneally injected edaravone (3 mg/kg, Tocris Bioscience, Bristol, UK) once daily for 11 successive days, starting 5 days after the MCAO surgery. BrdU and Edaravone were dissolved into distilled water and used after being filtered through 0.4 and 0.2 μm syringe filters, respectively.
Behavioral testing
Behavioral tests were performed 10 and 17 days after MCAO. Motor coordination and equilibrium, as well as short-term memory, were measured. For the corner test, the mice were placed between two pieces of cardboard, with dimensions of 30 cm × 20 cm at an angle of 30°. The mice were allowed to turn either right or left at the corners. The result was calculated as the percentage of left-side turns. Each mouse performed 20 trials.
For the wire grip test, a 40 cm-long elevated wire mesh (0.3 cm) 45 cm above the ground was utilized. The mice needed to grip the wire using their forelimbs and tail, and they were assigned a score based on how well they could accomplish this. The result was calculated as the average score of five trials as follows; 0, if the mouse dropped from the wire; 1, if the mouse was holding the wire using the contralateral forelimb; 2, if the mouse used both forelimbs; 3, if the mouse used both forelimbs and its tail.
For the rotarod test, the mice were placed on a rotating rod (Panlab S.L.U., Barcelona, Spain), and the average latency to fall was determined for each mouse. After adaptation trials, the result was calculated as the average latency on the rotating-rod with the speed accelerating gradually from 4 r/min to 20 r/min. Each mouse performed three trials.
For the passive avoidance test, the mice were placed in a chamber consisting of one illuminated compartment and one dark compartment separated by an automatic door (Med-Associates, Inc., St. Albans, VT, USA). During the trial, if the mice moved into the dark compartment from the light compartment, they received a 0.5 mA electric foot shock for 3 seconds. After 24 hours, the result was calculated as the latency to enter the dark compartment. The result was recorded using MED-PC software (Med-Associates, Inc.) interfacing with the testing apparatus.
Immunohistochemistry
Mice were intraperitoneally injected with 50 mg/kg of sodium pentobarbital (#051100, SCI Pharmtech Inc., Taoyunan, Taiwan, China) as anesthetics. Then they received saline and 4% paraformaldehyde in phosphate-buffered saline (PBS) intracardially. Their brains were isolated from skulls, fixed again with the same fixative solution for 24 hours, and soaked in 30% sucrose for 48 hours at 4°C for cryosection. Coronal sites at coordinates + 0.37 mm to + 0.97 mm anterior to bregma were selected as the locations from which the perilesional brain tissue (Paxinos and Franklin, 2013) was obtained. Using a cryostat (CM3050, Leica Microsystems, Wetzlar, Germany), the frozen tissues were prepared at a thickness of 20 μm and incubated with buffer solution (1× PBS/5% normal horse serum/0.3% Triton X-100) to block non-specific labeling. The tissue was then incubated with primary antibodies at 4°C overnight in primary antibody solution (1× PBS, 1% bovine serum albumin, 0.3% Triton X-100): mouse-anti-NG2 (1:500, Cat# MAB5384, Merk Millipore, Burlington, MA, USA), rabbit-anti-NG2 (1:500, Cat# ab129051, Abcam, Cambridge, UK), mouse-anti-BrdU (1:500, Cat# MCA2483, AbD Serotec, Oxford, UK), rabbit-anti-Ki67 (1:500, Cat# ab15580, Abcam), rabbit-anti-platelet-derived growth factor receptor α (PDGFRα, 1:500; Cat# ab5460, Abcam), mouse-anti-CC1 (an oligodendrocyte marker, 1:500, Cat# GTX16794, Genetex, Irvine, CA, USA), mouse-anti-cluster of differentiation (CD) 31 (an endothelial cell marker; CD31, 1:500, Cat# ab24590, Abcam), mouse-anti-CD68 (a microgila/macrophage marker; 1:500Cat# MCA1957, AbD Serotec), rabbit anti-tumor necrosis factor alpha (TNFα, Cat# ab66579, dilution 1:500, Abcam), DeadEnd Fluorometric terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) system (Cat# G3250, Promega, Madison, WI, USA), rabbit anti-cleaved caspase 3 (cCaspase 3, 1:500, Cat# 9661, Cell Signaling, Danvers, MA, USA), rabbit anti-brain-derived neurotrophic factor (BDNF; 1:500, Cat# ab101747, Abcam), rabbit anti-phosphorylated tyrosine receptor kinase B (pTrkB, 1:500; Cat# ab131483, Abcam), mouse anti-glycogen synthase kinase 3 beta (GSK3β, 1:500; Cat# ab93926, Abcam), and rabbitanti-phosphorylated GSK3β (pGSK3β; 1:500, Cat# ab75745, Abcam). The tissue was washed in PBS and incubated in a fluorescent-conjugated goat anti-mouse IgG (Cat# A11001, Invitrogen, Crlsbad, CA, USA), goat anti-mouse IgG (Cat# A11008, Invitrogen) and Texas red-conjugated goat anti-mouse IgG (Cat# A11005, Invitrogen), goat anti-rabbit IgG (Cat# A11037, Invitrogen) secondary antibody solution at room temperature for 2 hours, then mounted with mounting medium (Cat# H-1200, Vector Laboratories Inc. Burlingame, CA, USA). Using a fluorescence microscope (Carl Zeiss Imager M1, Carl Zeiss Inc., Gottingen, Germany) and confocal microscope (LSM 510, Carl ZeissInc.), the images were captured. Ipsilateral perilesional striatum and corpus callosum were focused for analysis. The immunofluorescent images were analyzed with image analysis software (IMT i-Solution Inc. 10.1, 17TH-5989 Walter Gage Rd., Vancouver, BC, Canada). Adobe photoshop CS6 software was used to count the number of positive cells, and the average nmber of positive cells in each slice was calculated.
Western blotting
Perilesional striatal tissue samples adjacent to the ischemic core were homogenized in lysis buffer (250 mM NaCl, 5 mM ethylenediaminetetraacetic acid, 25 mM Tris-HCl (pH 7.5), 1% NP40, 1 mM phenylmethanesulfonyl fluoride, 5 mM dithiothreitol, 0.1 mM Na3VO4, 10 mM NaF, leupeptin, and protein inhibitor). To quantify the protein concentration, the supernatant was extracted after centrifugation at 10,000 × g for 20 minutes at 4°C. 20 μg/mL of protein was separated by 7.5%, 10%, and 12% sodium dodecyl sulphate-polyacrylamide gel electrophoresis, and then transferred to a nitrocellulose membrane (Whatman Piscataway, NJ, USA) or polyvinylidene fluoride membrane. The membrane was incubated for 1 hour in 5% skimmed milk in PBS with 0.3% Triton-X (PBST) to block non-specific labeling. The membranes were incubated with primary antibodies at 4°C overnight on a shaker: mouse-anti-β-actin (Cat# CSB-MA000091M0m, Cusabio, Wuhan, China), rabbit-anti-NG2 (1:200, Cat# ab129051, Abcam), rabbit anti-β-catenin (1:1000, Cat# 9562, Cell Signaling), rabbit anti-cyclin D1 (Cat# 2922, Cell Signaling), rabbit anti-extracellular signal-regulated kinase (ERK, 1:200; Cat# sc-94, Santa Cruz Biotechnology, Dallas, TX, USA), rabbit anti-phosphorylated ERK (pERK; 1:1000, Cat# 9101, Cell Signaling), rabbit anti-Akt (1:200, Cat# sc-1618, Santa Cruz Biotechnology), rabbit anti-phosphorylated Akt (pAkt; 1:1000, Cat# 4058, Cell Signaling), mouse anti-GSK3β (1:500, Cat# ab93926, Abcam), rabbit anti-phosphorylated GSK3β (pGSK3β; 1:1000, Cat# ab75745, Abcam), rabbit anti-BDNF (1:500, Cat# ab101747, Abcam), rabbit anti-transforming growth factor beta (TGFβ; 1:1000, Cat# ab92486, Abcam), rabbit anti-neurotrophin 3 (NT3; 1:1000, Cat# ANT-003, Alomone Labs), rabbit anti-nerve growth factor (NGF; 1:1000, Cat# ab6199, Abcam), or mouse anti-glial cell-derived neurotrophic factor (GDNF; 1:200, Cat# sc-13147, Santa Cruz Biotechnology). The membranes were washed three times for 5 minutes in PBST and incubated with the horseradish peroxidase-conjugated anti-mouse or anti-rabbit immunoglobulin G secondary antibodies (1:1000, Cat# ADI-SAB-100 and ADI-SAB-300, Enzo Life Sciences, Farmingdale, NY, USA) at room temperature for 1 hour. Using an enhanced chemiluminescent substrate (34580, Pierce Biotech, Rockford, IL, USA), specific bands were visualized on the membrane. Chemiluminescence was quantified by an ImageQuant LAS 4000 system (Fujifilm, Tokyo, Japan). Target proteins were nomalized against β-actin expression.
Cytokine array analysis
Mouse cytokine arrays were performed using tissues in the perilesional striatum of the brains. Using the Proteome Profiler Mouse XL cytokine array kit (ARY028, R&D systems, Minneapolis, MN, USA), cytokine profiles were performed as instructed in the manufacturer directions.
Flow cytometric analysis
For flow cytometric analysis, ipsilateral tissues from the perilesional striatum were dissected using a razor blade into buffer A (Cat# HA-LF, BrainBits, Springfield, IL, USA). The supernatant was extracted after centrifugation at 110 × g for 2 minutes at 4°C, and the tissue was exposed to an enzyme solution for 30 minutes for digestion of the proteins. After being centrifuged at 960 × g for 2 minutes at 4°C, the supernatant was removed, and the sample was fixed in cold ethanol. After cell fixation, the samples were filtered with 70 μm and 40 μm cell strainers. The samples were then incubated with following primary antibodies: mouse anti-green fluorescent protein (GFP; 1:100, Cat# 2955, Cell Signaling), rabbit anti-GFP (1:100, Cat# G10362, Invitrogen), rat anti-BrdU (1:100, Cat# ab6326, Abcam), rabbit anti-CC1 (1:100, Cat# ab40778, Abcam), rat anti-CD31 (1:100, Cat# 550274, BD Biosciences, San Jose, CA, USA), mouse anti-CD68 (1:100, Cat# MCA1957, AbD Serotec), rabbit anti-BDNF (1:100, Cat# ab108319, Abcam), or mouse anti-GSK3β (1:100, Cat# ab93926, Abcam), and counterstained with 4′,6-diamidino-2-phenylindole (DAPI, H3570, Invitrogen) to label cell nuclei. After incubation, the samples were washed three times in PBS and incubated with goat anti-mouse IgG-Alexa Fluor 488 (Cat# A11029, Invitrogen), goat anti-rabbit IgG-Alexa Fluor 488 (Cat# A11008, invitrogen), goat anti-rabbit IgG-Alexa Fluor 633 (Cat# A21070, Invitrogen), goat anti-rat IgG-Alexa Fluor 633 (Cat# A21070, Invitrogen), goat anti-mouse IgG-Alexa Fluor 633 (Cat# A21052, Invitrogen) secondary antibody solution at room temperature for 30 minutes, after which the samples were transferred to a round-bottomed flow cytometry sample tube with a cell strainer cap. The immunolabeled cells were kept on ice, and flow cytometric analysis was performed immediately (FACS CANTO TMII; Becton-Dickinson, San Jose, CA, USA).
Statistical analysis
Data are shown as mean ± standard error of the mean. The data were analyzed using SigmaStat version 11.2 (Systat Software Inc., San Jose, CA, USA)and compared them with a independent samples t-test or one-way analysis of variance (ANOVA) followed by Tukey's post hoc tests. A level of P < 0.05 was considered statistically significant.
Results
Effects of EA therapy on behavioral function improvement following ischemic stroke
The mice received EA stimulation six times at 2-day intervals beginning 5 days after the MCAO, and behavioral testing was performed at 10 and 17 days after the MCAO (Figure 1A). The MCAO group exhibited significant motor and memory dysfunction compared with those in the control group (corner test, day 10: F(4,22) = 12.008, P < 0.001; corner test, day 17: F(4,22) = 3.605, P = 0.015; wire grip test, day 10: F(4,22) = 33.039, P < 0.001; wire grip test, day 17: F(4,22) = 18.973, P < 0.001; rotarod test, day 10: F(4,22) = 22.140; rotarod test, day 17: P < 0.001, F(4,22) = 13.782, P < 0.001). However, the corner test evaluating motor function showed significant differences between the MCAO and MCAO + EA1 groups at 10 days post-MCAO (F(4,22) = 12.008, P < 0.001). In addition, the wire grip and rotarod tests also revealed significant differences between MCAO and MCAO + EA1 groups at 17 days post-MCAO (wire grip test: F(4,22) = 18.973, P = 0.016; rotarod test: F(4,22) = 13.782, P = 0.028). Moreover, the MCAO + EA1 group showed significantly better performance than did the MCAO + EA3 group and the MCAO + Edaravone positive control group in the corner and wire grip tests at 10 and 17 days after MCAO, respectively (corner test, day 10: F(4,22) = 12.008, P = 0.006 and P = 0.003; wire grip test, day 17: F(4,22) = 18.973, P < 0.001 and P = 0.002). The MCAO + EA1 group also exhibited significantly better performance in the rotarod test than did the positive control group 17 days after MCAO (F(4,22) = 13.782, P = 0.009) (Figure 1B–E). There were no significant differences in the passive avoidance tests between the MCAO group and the MCAO + EA or MCAO + Edaravone groups (data not shown). The results demonstrate that EA stimulation attenuated functional motor deficits after ischemic stroke, and 1 mA EA was more efficacious than 3 mA EA and the positive control treatment with edaravone.
Figure 1.
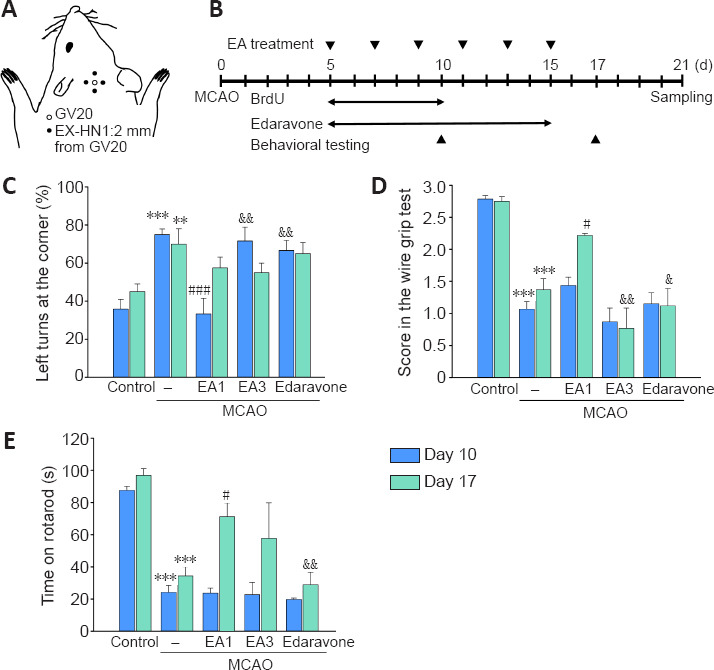
Experimental schematic diagram and the effects of EA stimulation on the improvement of motor functions ofmice following cerebral ischemia.
The Sishencong acupoint (A). Experimental timeline of EA treatment, BrdU labeling, edaravone administration, and behavioral testing at 10 and 17 days after MCAO (B). Quantification of the motor function test results of the corner test (C), wire grip test (D), and rotarod test (E). 1 mA EA stimulation was more efficacious than 3 mA EA or edaravone treatment in attenuating motor dysfunction in MCAO mice. n = 6. All data are shown as mean ± SEM. **P < 0.01, ***P < 0.001, vs. control group; #P < 0.05, ###P < 0.001, vs. MCAO group; &P < 0.05, &&P < 0.01, vs. MCAO + EA1 group (one-way analysis of variance with Tukey's post hoc test). EA1: Electroacupuncture at 1 mA; EA3: electroacupuncture at 3 mA; MCAO: middle cerebral artery occlusion.
Effects of EA therapy on the survival of NG2-expressing cells following ischemic stroke
Prominent increases in the numbers of NG2-expressing cells are commonly noted in perilesional brain tissue after ischemic stroke. Thus, the proliferation and survival of NG2-expressing cells 21 days after MCAO (Figure 1A) were assessed. In the perilesional striatum, the numbers of newly generated BrdU- or Ki67-positive cells were markedly increased in the MCAO + EA1 group compared to those in the MCAO group (BrdU: F(3,20) = 39.340, P < 0.001; Ki67: F(3,20) = 54.827, P = 0.04). However, there was no marked difference in the number of NG2-positive cells. Moreover, the number of newly generated NG2-expressing cells showing a double-labeling for BrdU was much lower in MCAO + EA1 group than in the MCAO group (F(3,20) = 32.751, P < 0.001) (Figure 2A and B). When we assessed two key cell surface proteins of NG2-expressing cells, we found that the number of cells double positive for NG2 and PDGFRαwas also markedly decreased in the EA-treated group (F(3,20) = 44.233, P = 0.002) (Figure 2D and E). However, EA stimulation markedly increased the number of newly generated NG2-expressing cells exhibiting double-positive for NG2 and BrdU in the corpus callosum (F(3,20) = 21.981, P < 0.001) (Figure 2A and C).
Figure 2.
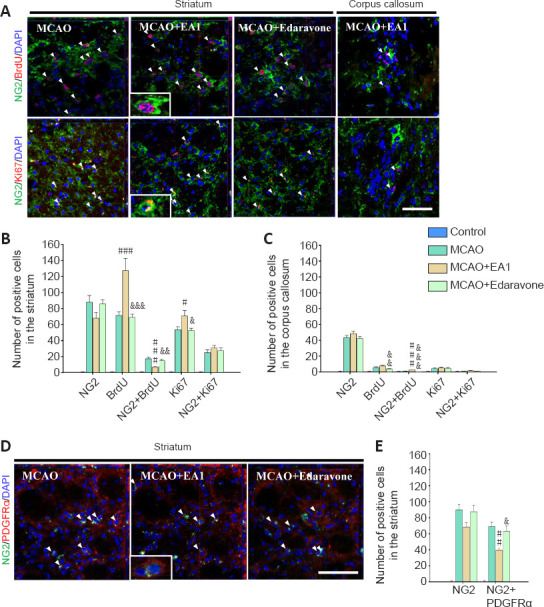
EA stimulation on the proliferation of NG2-expressing cells in the perilesional striatum and corpus callosum of mice at 21 days after MCAO.
Photomicrographs (A) and histograms showing the labeling and quantification of NG2- (green), BrdU- (red) and Ki67 (red) -positive cells in the perilesional striatum (B) and corpus callosum (C). Photomicrographs (D) and histogram showing the labeling and quantification of NG2 (green) and PDGFRα (red) double-positive cells in the perilesional striatum (E). EA stimulation significantly decreased the number of newly generated NG2-expressing cells in the perilesional striatum, relative to the number in the MCAO control group (without EA1 stimulation or edaravone treatment). All data are shown as mean ± SEM (n = 6). #P < 0.05, ##P < 0.01, ###P < 0.001, vs. MCAO group; &P < 0.05, &&P < 0.01, &&&P < 0.001, vs. MCAO + EA1 group (one-way analysis of variance with Tukey's post hoc test). Scale bars in A and D: 20 μm. BrdU: Bromodeoxyuridine; EA1: electroacupuncture at 1 mA; MCAO: middle cerebral artery occlusion; NG2: neural/glial antigen 2; PDGFRα: platelet-derived growth factor receptor alpha.
Since TNFα is a cytokine that is important for the proliferation and subsequent differentiation of NG2-expressing cells, we evaluated its expression and associated cell survival. Lower integrated optical density (IOD) of TNFα expression was determined in the perilesional striatum of rats in the MCAO + EA1 group than MCAO group (F(3,20) = 469.899, P < 0.001) (Figure 3A and B). Although the number of cells double-positive for BrdU and TNFα was significantly higher, the number of cells double-positive for NG2 and TNFα was significantly lower in the MCAO + EA1 group than in the MCAO group (BrdU + TNFα: F(3,20) = 32.999, P < 0.001; NG2 + TNFα: F(3,20) = 30.810, P < 0.001, respectively) (Figure 3A and C). Moreover, the number of cells double-positive for NG2 + TUNEL and NG2 + cCaspase 3 was also significantly decreased after EA treatment (NG2 + TUNEL: F(3,20) = 18.580, P = 0.014; NG2 + cCaspase 3: F(3,20) = 31.061, P = 0.004) (Figure 3A, D and E). These results suggest that EA stimulation reduced neuroinflammatory cytokine TNFα expression in the perilesional striatum and improved the survival of NG2-expressing cells from death after ischemic stroke.
Figure 3.
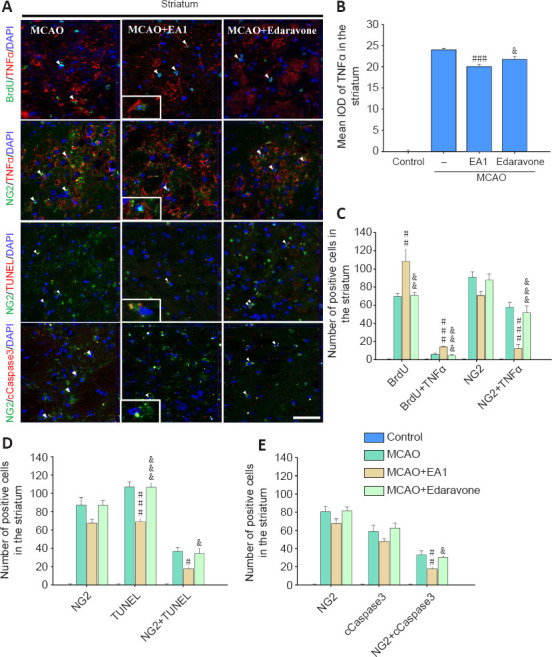
EA stimulation on the cell death of NG2-expressing cells in the perilesional striatum of mice at 21 days after MCAO.
Photomicrographs (A) and histograms showing the labeling and quantification of TNFα (red; B and C), TUNEL (red; D), and cleaved caspase 3 (cCaspase-3, red; E)-positive cells in the perilesional striatum. EA stimulation significantly decreased the cell death of NG2 (green) -expressing cells. All data are shown as mean ± SEM (n = 6). #P < 0.05, ##P < 0.01, ###P < 0.01, vs. MCAO group; &P < 0.05, &&P < 0.01, &&&P < 0.001, vs. MCAO + EA1 group (one-way analysis of variance with Tukey's post hoc test). Scale bar in A = 20 μm. DAPI: 4′,6-Diamidino-2-phenylindole; EA1: electroacupuncture at 1 mA; IOD: integrated optical density; MCAO: middle cerebral artery occlusion; NG2: neural/glial antigen 2; TNFα: tumor necrosis factor alpha; TUNEL: terminal deoxynucleotidyl transferase dUTP nick end labeling.
Effects of EA therapy on signaling pathways related to the activation of NG2-expressing cells and neurotrophic factors following ischemic stroke
Western blotting of the perilesional striatum was performed in two groups, MCAO and MCAO + EA1, to investigate the mechanisms underlying the activation of NG2-expressing cells and neurotrophic factors. EA stimulation was performed three times at 2-day intervals starting from the fifth day after the MCAO, and western blot analysis was performed on tissue collected 10 days post-MCAO (Figure 4A). Expression of NG2 was significantly more upregulated in the MCAO + EA1 group than in the MCAO group (F(1,10) = 7.367, P = 0.022), and this was accompanied by a marked increase in β-catenin and cyclin D1 expression, and an increase in ERK and Akt (β-catenin: F(1,10) = 7.276, P = 0.023; cyclin D1: F(1,10) = 47.057, P < 0.001; pERK/ERK: F(1,10) = 8.541, P = 0.015; pAkt/Akt: F(1,10) = 9.173, P = 0.013). Moreover, activation of GSK3β, a molecule that modulates cross-talk between other common signaling pathways, was markedly increased relative to that in the MCAO group (F(1,10) = 7.372, P = 0.022) (Figure 4B and C). For the neurotrophic factors, BDNF, TGFβ, and NT3 expressions in the perilesional striatum were significantly increased after EA stimulation (BDNF: F(1,10) = 9.844, P = 0.011; TGFβ: F(1,10) = 7.518, P = 0.021; NT3: F(1,10) = 9.445, P = 0.012) (Figure 4D and E). Western blot full image is shown in Additional Figure 1 (1MB, tif) . As activation of NG2-expressing cells can be attributed, in part, to the induction of various cytokines and chemokines, cytokine array analysis was performed for comparison betweenthe MCAO and MCAO + EA1 groups (Additional Figure 2 (952.8KB, tif) ). EA stimulation induced remarkable increases in the expression levels of matrix metallopeptidase (MMP) 3, neutrophil gelatinase-associated lipocalin (NGAL), MMP9, chemokine ligand (CCL) 6, endostatin, CXC chemokine (Lix), and CCL12, along with marked decreases in low density lipoprotein receptor (LDLR), C-reactive protein (CRP), and CX3C chemokine receptor 1 (CX3CL1). These results show that EA stimulation upregulated the expression of signaling molecules related to NG2 cell activation, such as β-catenin, ERK, and Akt, as well as specific growth factors, such as BDNF, TGFβ and NT3, in the perilesional striatum.
Figure 4.
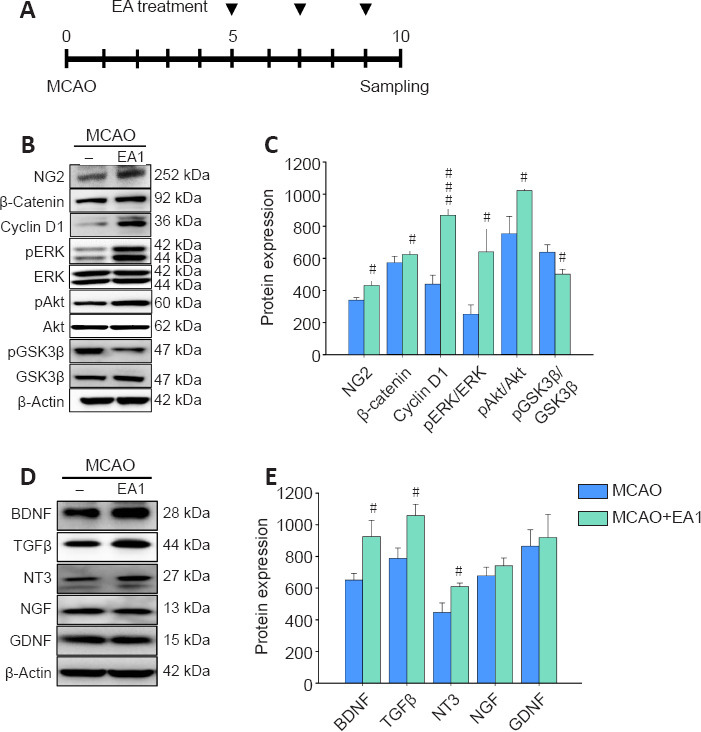
Effects of EA stimulation on the expression of signaling markers related to the activation of NG2-expressing cells and on the upregulation of neurotrophic factors in the perilesional striatum of mice at 21 days after MCAO.
Experimental timeline of MCAO generation, EA treatment, and sampling (A). Western blots of signaling markers related to NG2 activation (B) and neurotrophic factor expression (D), and their quantification by densitometric analysis (C and E). EA stimulation upregulated signaling pathways related to NG2 cell activation and neurotrophic factor expression involving BDNF, TGFβ, and NT3. All data are shown as mean ± SEM (n = 6). #P < 0.05 and ###P < 0.001, vs. MCAO (independent samples t-test). EA1: Electroacupuncture at 1 mA; GDNF: glial cell-derived neurotrophic factor; MCAO: middle cerebral artery occlusion; NG2: neural/glial antigen 2; NGF: nerve growth factor; NT-3: neurotrophin-3; pAKt: phosphorylated Akt; pERK: phosphorylated ERK; pGSK3β: phosphorylated GSK3β; TGFβ: transforming growth factor-β. Uncropped images of western blot are shown in Additional Figure 1 (1MB, tif) .
Immunohistochemical analysis of the effects of EA therapy on NG2-expressing cell types and the activation of BDNF signaling in ischemic stroke
Since NG2 is widely expressed in oligodendrocytes, neurovascular cells, and microglia/macrophages depending on differentiation after brain injury, the phenotypes of NG2-expressing cells after six EA stimulations on day 21 post-MCAO were characterized (Figure 1A). In the perilesional striatum, a higher number of cells with double-positive for NG2 + CC1, NG2 + CD31, and NG2 + CD68 were observed in the MCAO + EA group than in the MCAO group, but only the number of NG2 and CD68 double-positive cells significantly differed between groups (F(3,20) = 28.731, P = 0.004), and no differences were observed in the corpus callosum. Of these double-labeled cells, more NG2 and CC1 double-positive cellswere observed than the other cell types in the perilesional striatum, and NG2+CD31 double-positive cells accounted for the majority of the cell types in the corpus callosum (Figure 5A–C). The western blot analysis revealed that EA stimulation robustly activated NTFs, such as BDNF, TGFβ, and NT3, in the perilesional striatum, as well as signals related to the proliferation and survival of NG2-expressing cells. Therefore, the activation of BDNF, its receptor, TrkB, and GSK3β were evaluated. The numbers of BDNF- and pTrkB-positive cells were markedly enhanced in the MCAO + EA group compared to those in the MCAO group (BDNF: F(3,20) = 152.541, P = 0.008; pTrkB: F(3,20) = 272.468, P = 0.02), and the numbers of cells double-positive for NG2 + BDNF and NG2 + pTrkB were also markedly increased (NG2 + BDNF: F(3,20) = 38.042, P = 0.048; NG2 + pTrkB: F(3,20) = 89.536, P = 0.023). The number of GSK3β-positive cells was markedly increased in the MCAO + EA group compared to that in the MCAO group (F(3,20) = 69.016, P = 0.019). However, no significant difference in NG2 + GSK3β double-positive cells was observed (Figure 6A–C). These results demonstrate that EA treatment induced the differentiation of various NG2-expressing cell types through modulation of signaling mechanisms involving neurotrophic factors, BDNF.
Figure 5.
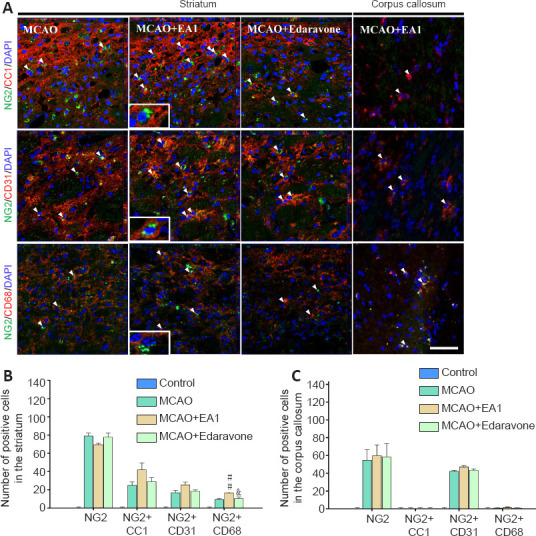
Effects of EA stimulation on NG2-expressing cell types in the perilesional striatum and the corpus callosum of mice at 21 days after MCAO.
Photomicrographs (A) and histograms (B and C) showing labeling and quantification of NG2 (green) -, CC1 (red) -, CD31 (red) - and CD68 (red) -positive cells in the perilesional striatum and the corpus callosum of MCAO mice. EA stimulation significantly increased the NG2 and CD68double positive cells in the perilesional striatum. n = 6. All data are shown as mean ± SEM. ##P < 0.01, vs. MCAO group; &P < 0.05, vs. MCAO + EA1 group (one-way analysis of variance with Tukey's post hoc tests). Scale bar in A: 20 μm. DAPI: 4′,6-Diamidino-2-phenylindole; EA1: electroacupuncture at 1 mA; MCAO: middle cerebral artery occlusion; NG2: neural/glial antigen 2.
Figure 6.

Effects of EA stimulation on the expression of BDNF, pTrkB, and GSK3β in the perilesional striatum of mice at 21 days after MCAO.
Photomicrograph (A) and histograms (B and C) showing labeling of BDNF (red) -, pTrkB (red) - and GSK3β(red) -positive cells in the perilesional striatum of MCAO mice. The number of NG2+/BDNF+ and NG2+/pTrkB+ cells was significantly increased after EA1 stimulation. n = 6. All data are shown as mean ± SEM. #P < 0.05 and ##P < 0.01, vs. MCAO group; &P < 0.05, vs. MCAO + EA1 group (one-way analysis of variance with Tukey's post hoc test). Scale bar in A: 20 μm. BDNF: Brain-derived neurotrophic factor; DAPI: 4′,6-Diamidino-2-phenylindole; EA1: electroacupuncture at 1 mA; MCAO: middle cerebral artery occlusion; NG2: neural/glial antigen 2; (p)GSK3β: (phosphorylated) glycogen synthase kinase 3 beta; pTrkB: phosphorylated tyrosine receptor kinase B.
Flow cytometric analysis regarding the effects of EA therapy on NG2-expressing cell types and BDNF expression following ischemic stroke in NG2-mEGFP mice
Since the expression of NG2 gradually declines in mature cells, it was necessary to confirm the results of immunohistochemical analysis using other techniques. Therefore, flow cytometric analysis was performed to compare the MCAO and MCAO + EA1 groups using NG2-mEGFP mice. The numbers of cells double-positive for BrdU + GFP, CC1 + GFP, CD31 + GFP, and GSK3β + GFP were significantly increased in the MCAO + EA group (BrdU + GFP: F(1,8) = 27.870, P < 0.001; CC1 + GFP: F(1,8) = 6.776, P = 0.032; CD31 + GFP: F(1,8) = 6.346, P = 0.036; GSK3β + GFP: F(1,8) = 27.381, P < 0.001). There was no significant difference in the number of cells cells double-positive for BDNF and GFP. However, the number of BDNF-positive cells labeled with DAPI was significantly altered by EA stimulation (F(1,8) = 6.374, P = 0.036), suggesting that BDNF was secreted by other cerebral cells, not only by specific NG2-expressing cells (Figure 7A–D). Assessment of these cells in the contralateral striatum revealed a slight increase in the expression of these markers, but there was no significant difference between groups (Additional Figure 3 (900.6KB, tif) ). These results confirmed, through immunohistochemical analysis in NG2-mEGFP mice, that EA stimulation induced the differentiation of various cell types expressing NG2 through increasing neurotrophic factor-related signaling, such as those modulated by BDNF.
Figure 7.
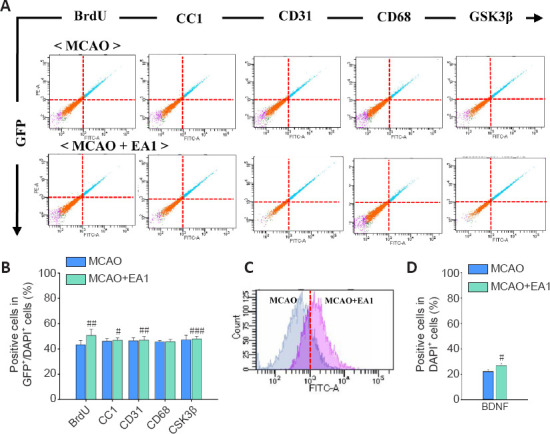
Effects of EA stimulation on cellular phenotype and the expression of BDNF and GSK3β in GFP+/DAPI+ NG2-expressing cells, or whole cells in ipsilateral perilesional striatum of NG2-mEGFP mice at 21 days after MCAO.
Quantitative analysis of the flow cytometry data (A and C) and histograms showing the percentage of GFP+/DAPI+ cells also expressing BrdU, CC1, CD31, CD68, and GSK3β, as well as the percentage of DAPI+ cells also expressing BDNF (B and D, respectively). The numbers of GFP+ cells that also express BrdU, CC1, CD31and GSKβ were significantly increased after EA1 stimulation. n = 5. All data are shown as mean ± SEM. #P < 0.05, ##P < 0.01, and ###P < 0.01, vs. MCAO group (independent samples t-test). BDNF: Brain-derived neurotrophic factor; BrdU: bromodeoxyuridine; DAPI: 4′,6-Diamidino-2-phenylindole; EA1: Electroacupuncture at 1 mAEA1: electroacupuncture at 1 mA; GFP: green fluorescent protein; GSK3β: glycogen synthase kinase 3 beta; MCAO: middle cerebral artery occlusion; mEGFP: mutated enhanced green fluorescent protein; NG2: neural/glial antigen 2.
Discussion
In the present study, we assessed functional changes in NG2-expressing cells in the perilesional tissue that were treated with EA after ischemic stroke. We also assessed the contribution of NG2-expressing cells in functional recovery. EA therapy robustly induced the survival and differentiation of multiple NG2-expressing cell types, including oligodendrocytes, microvascular endothelial cells, and microglia/macrophages in the perilesional brain tissue. These changes were associated with increased activation of GSK3β and NTF signaling, especially pathways involving BDNF, revealing strong evidence of the validity and therapeutic efficacy of EA therapy for treating stroke.
The Sishencong acupoint innervates the branches of the supraorbital, greater occipital, and auriculotemporal nerves, and it plays a functional role in refreshing the brain and restoring the mind (Chen, 1995). Manipulation of this acupoint is widely performed in the clinical treatment of mental disorders and cerebrovascular diseases, and those involving post-stroke treatment, and it can improve cerebral microcirculation and regulate nervous system excitability (Tan et al., 2017; Lu et al., 2019). When we applied EA stimulation at the Sishencong acupoint, it significantly alleviated motor deficits after ischemic stroke, and EA therapy was more efficacious than the positive control treatment with edaravone, providing further evidence that EA may indicate an adjuvant therapy for treating motor dysfunction in stroke patients. Noninvasive electrical stimulation such as ear-clip electrode and transcranial direct current also enhance functional recovery from stroke insults via enhancement of neurogenesis and expression of growth factors, respectively (Ahn et al, 2020; Balseanu et al., 2020). Because EA involves stimulation of electrical pulses, electrical stimulation may also be involved in ameliorating the effects of motor dysfunction along with stimulation of a specific acupoints.
NG2-expressing cells, as well as NG2 glial lineage and non-NG2 lineage cells, including neurovascular cells and microglia/macrophages, react to brain injuries, such as those that occur following ischemic stroke, by proliferating and undergoing morphological changes around the lesion site (Matsumoto et al., 2008; Smirkin et al., 2010; Claus et al., 2013; Zhang et al., 2013; Hackett et al., 2016). NG2 is expressed by oligodendrocyte progenitor cells; these cells were initially known as NG2 glial cells or oligodendrocyte precursor cells, and they display wide differentiation potential in response to cerebral ischemia (Nishiyama et al., 2009; Zhang et al., 2013; Dimou and Gallo, 2015; Honsa et al., 2016). NG2 is upregulated in vasculogenic cells within the lesion area, which display multipotent stem cell activity under pathological states such as ischemia; this suggests that NG2 stimulates the proliferation, motility, and recruitment of endothelium during vascular remodeling and morphogenesis (Ozerdem et al., 2001; Ahlskog, 2011; Stallcup et al., 2016; Jin et al., 2018). NG2-expressing microglia/macrophage-like cells have been detected in the narrow demarcation area between the perilesional region and the ischemic core (Matsumoto et al., 2008; Sugimoto et al., 2014), and they play a beneficial role in recovery from ischemic brain lesions through the secretion of neuroprotective factors (Smirkin et al., 2010).
Appropriate control of NG2-expressing cells in perilesional brain tissue within recoverable injured regions might be a novel target for promoting functional recovery through EA therapy after ischemic stroke. Therefore, we aimed to investigate the activity of NG2-expressing cells in the perilesional brain tissue in the MCAO model in mice. Our results demonstrated a remarkable increase of proliferating cells in the perilesional area induced by EA stimulation; however, the number of newly generated NG2-expressing cells decreased, but not in the corpus callosum, a region distant from the inflammatory core. Upregulation of TNFα accompanies cerebral ischemia, and this pro-inflammatory cytokine is the most probable inducer in the proliferation/regeneration of NG2-expressing cells in the CNS (Arnett et al., 2001). A significant decrease in TNFα expression in the perilesional brain tissue was induced by EA stimulation similar to the results of a previous study that showed EA stimulation exerted a neuroprotective effect in cerebral ischemia/reperfusion injury, which was at least partially associated with anti-inflammatory mechanisms (Long et al., 2019). Subsequently, when we assessed the survival of NG2-expressing cells, we observed that EA treatment significantly increased their survival. Our results indicate that the proliferation of NG2-expressing cells is alleviated by the anti-inflammatory function of EA stimulation; this therapy showed a beneficial effect on the survival of NG2-expressing cells.
Functional and histological recoveries involve the activation of signaling that promotes cell survival and the upregulation of neurotrophic factors (Francardo et al., 2014). Moreover, the proteoglycan NG2 controls various cellular functions, involving proliferation, migration, and differentiation in response to growth factor stimulation (Biname, 2014). Increased proliferation and differentiation resulting from the activation of NG2-expressing cells are largely dependent on PI3K/Akt, MEK/ERK and Wnt/β-catenin signaling (Hill et al., 2013; Xie et al., 2016; Jiang et al., 2019). In neural development, these pathways are highly interconnected, with crosstalk and convergence points with GSK3. Therefore, GSK3 signaling acts as a mediator for the integration of multiple proliferation and differentiation signals (Kim et al., 2009; Gonzalez-Fernandez et al., 2018; Courchesne et al., 2019).
EA stimulation enhances the proliferation and differentiation of adult stem cells and improves neuronal function in ischemic stroke through the expression of neurotrophic factors and their related signaling pathways (Kim et al., 2014, 2018; Ahn et al., 2019). These neurotrophic factors promote progenitor cell survival and generation through the regulation of major intracellular mediators of ERK and Akt signaling (Barnabe-Heider and Miller, 2003; Islam et al., 2009). Higher concentrations of growth factors are associated with repair processes in ischemic brain injury, resulting in greater differentiation of NG2-expressing cells (Honsa et al., 2016). Therefore, essential signaling involving the activation of NG2-expressing cells and the expression of neurotrophic factors was compared between the MCAO and MCAO + EA1 groups. EA stimulation greatly enhanced the expression of ERK, Akt, β-catenin and GSK3β in the perilesional striatum, along with a marked increase in BDNF, TGFβ, and NT3 expression.
Subsequently, the cell types expressing NG2 were investigated by immunohistochemistry and flow cytometric analysis to reveal the relationship between activation of NG2-expressing cells and neurotrophic factor-mediated signaling pathways. EA stimulation significantly enhanced the number of NG2-expressing cells double-positive for oligodendrocyte, microvascular endothelial cell, and microglia/macrophage markers, suggesting that EA enhanced the survival and differentiation of these cell types. The relationship between the increase in NG2-expressing cells and the expression of BDNF and the mediator of functional integration, GSK3β, was also investigated by immunohistochemistry and flow cytometric analysis. BDNF/TrkB and GSK3β signaling were activated in perilesional brain tissue after MCAO; this activation also occurred in NG2-expressing cells. Interestingly, there was no change in BDNF level in NG2-expressing cells, but a remarkable whole cell-type increase was observed within the perilesional site, indicating that BDNF was also secreted by other type cells, not only by specific NG2-expressing cells.
NG2-expressing glia undergo proliferation and morphological changes, and they produce oligodendrocytes in response to cerebral ischemia (Zhang et al., 2013; Honsa et al., 2016). NG2 expression in neurovascular cells in the lesion core contributes to vascular remodeling following ischemic stroke (Stallcup et al., 2016; Jin et al., 2018; Yang et al., 2018), along with increased expression of markers of angiogenesis, such as Ki67, NG2, and MMP3 within the perilesional site (Yang et al., 2018). In NG2-expressing cells, the protein CD68, a marker of active phagocytosis, plays a role in the elimination of neuronal debris to accelerate tissue repair; thus, these microglia/macrophages are a therapeutic target for the control of secondary nerve degeneration in perilesional region following ischemic injury (Perego et al., 2011; Sugimoto et al., 2014). According to our results, the increased numbers of multiple phenotypes of NG2-expressing cells suggests the possibility that EA therapy could be efficacious for ischemic stroke through various neuroprotective mechanisms, including enhancing the production of oligodendrocytes, increasing vascular remodeling, and eliminating neuronal debris.
This study has some major limitations. First, other neurotrophic factors may play an important role in the therapeutic effects of EA. Our results also suggest a possible role for other neurotrophic factors, including the involvement of TGFβ and NT3, and the activation of their related signaling pathways may explain the efficacy of EA in the treatment of ischemic stroke. TGFβ in the ischemic core diffuses toward the perilesional area where it induces NG2-positive microglia that assist in the alleviation of damage within ischemic brain injuries (Sugimoto et al., 2014). NT3 increases the number and differentiation of endogenous oligodendrocyte precursor cells, resulting in functional improvements (Huang et al., 2011). Second, cytokine regulation by EA stimulation may have beneficial effects on NG2-expressing cells. Cytokines also play a pivotal role in the accumulation of NG2-expressing cells in the injured brain (Levine, 2016; Tei et al., 2013). In our cytokine array analysis, changes in inflammatory cytokine expression induced by EA stimulation, such as MMP3 and MMP9, Lix, CRP, and CX3CR1, may be involved in the functional activation of NG2-expressing cells (Li et al., 2016; Tei et al., 2013; Yang et al., 2018). Third, aging and comorbidities such as arterial hypertension and diabetes have a major impact on the incidence, consequences, and efficacy of therapy in stroke patients (Joseph et al., 2012; Buga et al., 2013). In the present study, we employed young animals; thus, there are some discrepancies between our encouraging results and clinical outcomes.
Ultimately, EA therapy may be a viable treatment option for restoring functional activity of perilesional brain tissue after ischemic injury. Our results indicate that EA therapy enhanced the survival and differentiation of multiple NG2-expressing cells types, suggesting the engagement of various neuroprotective mechanisms. The generation of oligodendrocytes, vascular remodeling, and elimination of degenerating debris contribute to the efficacy of EA therapy for ischemic stroke.
Additional files:
Additional Figure 1 (1MB, tif) : Western blot images of regions used for figure 4 marked with rectangles and molecular size marker (kDa).
Western blot images of regions used for figure 4 marked with rectangles and molecular size marker (kDa).
Additional Figure 2 (952.8KB, tif) : Effects of EA stimulation on cytokine expression in perilesional striatum of mice at 21 days after MCAO.
EA stimulation on cytokine expression in perilesional striatum of mice at 21 days after MCAO.
(A, B) Cytokine array results (A) and quantitative analysis of expression (B). The figure shows sample cytokine array data from the perilesional striatum of the MCAO and MCAO + EA1 groups. The red circles indicate upregulated cytokine and blue ones downregulated cytokine compared to MCAO and MCAO + EA1. All data are shown as mean ± SEM (n = 4). CCL6: Chemokine ligand 6; CCL12: chemokine ligand 12; CRP: C-reactive protein; CX3CL1: CX3C chemokine receptor 1; EA1: electroacupuncture at 1 mA; LDLR: low density lipoprotein receptor; Lix: CXC chemokine; MCAO: middle cerebral artery occlusion; MMP3: matrix metallopeptidase 3; MMP9: matrix metallopeptidase 9; NGAL: neutrophil gelatinase-associated lipocalin.
Additional Figure 3 (900.6KB, tif) : Effects of EA stimulation of NG2-mEGFP mice on cellular phenotypes of NG2-expressing cells and the expression of BDNF and GSK3β in the contralateral striatum of mice at 21 days after MCAO.
EA stimulation of NG2-mEGFP mice on cellular phenotypes of NG2-expressing cells and the expression of BDNF and GSK3β in the contralateral striatum of mice at 21 days after MCAO.
Quantitative analysis of the flow cytometry and (A and C) and histograms showing the numbers of GFP + DAPI-positive cells also expressing BrdU, CC1, CD31, CD68, and GSK3β, and the number of DAPI-positive cells expressing BDNF (B and D, respectively). There were no significant differences between the MCAO and MCAO + EA1 groups. All data are shown as mean ± SEM (n = 5). BDNF: brain-derived neurotrophic factor; BrdU: Bromodeoxyuridine; DAPI: 4',6-diamidino-2-phenylindole; EA1: electroacupuncture at 1 mA; GFP: green fluorescent protein; GSK3β: glycogen synthase kinase 3 beta; MCAO: middle cerebral artery occlusion; mEGFP: membrane-anchored enhanced green fluorescent protein; NG2: neural/glial antigen 2.
Footnotes
Conflicts of interest: The authors declare that there are no financial competing interests.
Financial support: This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), which was funded by the Ministry of Science, ICT, and Future Planning (2018R1A2A2A05018926) (to BTC). The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Institutional review board statement: The animal experiments were approved by the Pusan National University Animal Care (approval nos. PNU2019-2199 and PNU2019-2884) on April 8, 2019 and June 19, 2019.
Copyright license agreement: The Copyright License Agreement has been signed by all authors before publication.
Data sharing statement: Datasets analyzed during the current study are available from the corresponding author on reasonable request.
Plagiarism check: Checked twice by iThenticate.
Peer review: Externally peer reviewed.
Funding: This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), which was funded by the Ministry of Science, ICT, and Future Planning (2018R1A2A2A05018926) (to BTC).
C-Editor: Zhao M; S-Editor: Li CH; L-Editor: Song LP; T-Editor: Jia Y.
References
- 1.Ahlskog JE. Does vigorous exercise have a neuroprotective effect in Parkinson disease? Neurology. 2011;77:288–294. doi: 10.1212/WNL.0b013e318225ab66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ahn SM, Jung DH, Lee HJ, Pak ME, Jung YJ, Shin YI, Shin HK, Choi BT. Contralesional application of transcranial direct current stimulation on functional improvement in ischemic stroke mice. Stroke. 2020;51:2208–2218. doi: 10.1161/STROKEAHA.120.029221. [DOI] [PubMed] [Google Scholar]
- 3.Ahn SM, Kim YR, Shin YI, Ha KT, Lee SY, Shin HK, Choi BT. Therapeutic potential of a combination of electroacupuncture and TrkB-expressing mesenchymal stem cells for ischemic stroke. Mol Neurobiol. 2019;56:157–173. doi: 10.1007/s12035-018-1067-z. [DOI] [PubMed] [Google Scholar]
- 4.Arnett HA, Mason J, Marino M, Suzuki K, Matsushima GK, Ting JP. TNF alpha promotes proliferation of oligodendrocyte progenitors and remyelination. Nat Neurosci. 2001;4:1116–1122. doi: 10.1038/nn738. [DOI] [PubMed] [Google Scholar]
- 5.Balseanu AT, Grigore M, Pinosanu LR, Slevin M, Hermann DM, Glavan D, Popa-Wagner A. Electric stimulation of neurogenesis improves behavioral recovery after focal ischemia in aged rats. Front Neurosci. 2020;14:732. doi: 10.3389/fnins.2020.00732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barnabe-Heider F, Miller FD. Endogenously produced neurotrophins regulate survival and differentiation of cortical progenitors via distinct signaling pathways. J Neurosci. 2003;23:5149–5160. doi: 10.1523/JNEUROSCI.23-12-05149.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Biname F. Transduction of extracellular cues into cell polarity: the role of the transmembrane proteoglycan NG2. Mol Neurobiol. 2014;50:482–493. doi: 10.1007/s12035-013-8610-8. [DOI] [PubMed] [Google Scholar]
- 8.Buga AM, Napoli MD, Popa-Wagner A. Preclinical models of stroke in aged animals with or without comorbidities: role of neuroinflammation. Biogerontology. 2013;14:651–662. doi: 10.1007/s10522-013-9465-0. [DOI] [PubMed] [Google Scholar]
- 9.Chen E. Edinburgh. New York: Churchill Livingstone; 1995. Cross-sectional anatomy of acupoints. [Google Scholar]
- 10.Claus HL, Walberer M, Simard ML, Emig B, Muesken SM, Rueger MA, Fink GR, Schroeter M. NG2 and NG2-positive cells delineate focal cerebral infarct demarcation in rats. Neuropathology. 2013;33:30–38. doi: 10.1111/j.1440-1789.2012.01322.x. [DOI] [PubMed] [Google Scholar]
- 11.Courchesne E, Pramparo T, Gazestani VH, Lombardo MV, Pierce K, Lewis NE. The ASD Living Biology: from cell proliferation to clinical phenotype. Mol Psychiatry. 2019;24:88–107. doi: 10.1038/s41380-018-0056-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dimou L, Gallo V. NG2-glia and their functions in the central nervous system. Glia. 2015;63:1429–1451. doi: 10.1002/glia.22859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dirnagl U, Iadecola C, Moskowitz MA. Pathobiology of ischemic stroke: an integrated view. Trends Neurosci. 1999;22:391–397. doi: 10.1016/s0166-2236(99)01401-0. [DOI] [PubMed] [Google Scholar]
- 14.Francardo V, Bez F, Wieloch T, Nissbrandt H, Ruscher K, Cenci MA. Pharmacological stimulation of sigma-1 receptors has neurorestorative effects in experimental parkinsonism. Brain. 2014;137:1998–2014. doi: 10.1093/brain/awu107. [DOI] [PubMed] [Google Scholar]
- 15.Gonzalez-Fernandez E, Jeong HK, Fukaya M, Kim H, Khawaja RR, Srivastava IN, Waisman A, Son YJ, Kang SH. PTEN negatively regulates the cell lineage progression from NG2(+) glial progenitor to oligodendrocyte via mTOR-independent signaling. Elife. 2018;7:e32021. doi: 10.7554/eLife.32021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gotoh H, Wood WM, Patel KD, Factor DC, Boshans LL, Nomura T, Tesar PJ, Ono K, Nishiyama A. NG2 expression in NG2 glia is regulated by binding of SoxE and bHLH transcription factors to a Cspg4 intronic enhancer. Glia. 2018;66:2684–2699. doi: 10.1002/glia.23521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hackett AR, Lee DH, Dawood A, Rodriguez M, Funk L, Tsoulfas P, Lee JK. STAT3 and SOCS3 regulate NG2 cell proliferation and differentiation after contusive spinal cord injury. Neurobiol Dis. 2016;89:10–22. doi: 10.1016/j.nbd.2016.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hill RA, Patel KD, Medved J, Reiss AM, Nishiyama A. NG2 cells in white matter but not gray matter proliferate in response to PDGF. J Neurosci. 2013;33:14558–14566. doi: 10.1523/JNEUROSCI.2001-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Honsa P, Valny M, Kriska J, Matuskova H, Harantova L, Kirdajova D, Valihrach L, Androvic P, Kubista M, Anderova M. Generation of reactive astrocytes from NG2 cells is regulated by sonic hedgehog. Glia. 2016;64:1518–1531. doi: 10.1002/glia.23019. [DOI] [PubMed] [Google Scholar]
- 20.Huang SF, Ding Y, Ruan JW, Zhang W, Wu JL, He B, Zhang YJ, Li Y, Zeng YS. An experimental electro-acupuncture study in treatment of the rat demyelinated spinal cord injury induced by ethidium bromide. Neurosci Res. 2011;70:294–304. doi: 10.1016/j.neures.2011.03.010. [DOI] [PubMed] [Google Scholar]
- 21.Islam O, Loo TX, Heese K. Brain-derived neurotrophic factor (BDNF) has proliferative effects on neural stem cells through the truncated TRK-B receptor, MAP kinase, AKT, and STAT-3 signaling pathways. Curr Neurovasc Res. 2009;6:42–53. doi: 10.2174/156720209787466028. [DOI] [PubMed] [Google Scholar]
- 22.Jiang R, Prell C, Lonnerdal B. Milk osteopontin promotes brain development by up-regulating osteopontin in the brain in early life. FASEB J. 2019;33:1681–1694. doi: 10.1096/fj.201701290RR. [DOI] [PubMed] [Google Scholar]
- 23.Jin X, Riew TR, Kim HL, Choi JH, Lee MY. Morphological characterization of NG2 glia and their association with neuroglial cells in the 3-nitropropionic acid-lesioned striatum of rat. Sci Rep. 2018;8:5942. doi: 10.1038/s41598-018-24385-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Joseph C, Buga AM, Vintilescu R, Balseanu AT, Moldovan M, Junker H, Walker L, Lotze M, Popa-Wagner A. Prolonged gaseous hypothermia prevents the upregulation of phagocytosis-specific protein annexin 1 and causes low-amplitude EEG activity in the aged rat brain after cerebral ischemia. J Cereb Blood Flow Metab. 2012;32:1632–1642. doi: 10.1038/jcbfm.2012.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kim WY, Wang X, Wu Y, Doble BW, Patel S, Woodgett JR, Snider WD. GSK-3 is a master regulator of neural progenitor homeostasis. Nat Neurosci. 2009;12:1390–1397. doi: 10.1038/nn.2408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kim YR, Ahn SM, Pak ME, Lee HJ, Jung DH, Shin YI, Shin HK, Choi BT. Potential benefits of mesenchymal stem cells and electroacupuncture on the trophic factors associated with neurogenesis in mice with ischemic stroke. Sci Rep. 2018;8:2044. doi: 10.1038/s41598-018-20481-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kim YR, Kim HN, Ahn SM, Choi YH, Shin HK, Choi BT. Electroacupuncture promotes post-stroke functional recovery via enhancing endogenous neurogenesis in mouse focal cerebral ischemia. PLoS One. 2014;9:e90000. doi: 10.1371/journal.pone.0090000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kyyriainen J, Ekolle Ndode-Ekane X, Pitkanen A. Dynamics of PDGFRbeta expression in different cell types after brain injury. Glia. 2017;65:322–341. doi: 10.1002/glia.23094. [DOI] [PubMed] [Google Scholar]
- 29.Levine J. The reactions and role of NG2 glia in spinal cord injury. Brain Res. 2016;1638:199–208. doi: 10.1016/j.brainres.2015.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Levine JM, Reynolds R, Fawcett JW. The oligodendrocyte precursor cell in health and disease. Trends Neurosci. 2001;24:39–47. doi: 10.1016/s0166-2236(00)01691-x. [DOI] [PubMed] [Google Scholar]
- 31.Li Y, Du XL, He BP. Lipopolysaccharide upregulates the expression of CINC-3 and LIX in primary NG2 cells. Neurochem Res. 2016;41:1448–1457. doi: 10.1007/s11064-016-1856-6. [DOI] [PubMed] [Google Scholar]
- 32.Lindvall O, Kokaia Z. Stem cell research in stroke: how far from the clinic? Stroke. 2011;42:2369–2375. doi: 10.1161/STROKEAHA.110.599654. [DOI] [PubMed] [Google Scholar]
- 33.Long M, Wang Z, Zheng D, Chen J, Tao W, Wang L, Yin N, Chen Z. Electroacupuncture pretreatment elicits neuroprotection against cerebral ischemia-reperfusion injury in rats associated with transient receptor potential vanilloid 1-mediated anti-oxidant stress and anti-inflammation. Inflammation. 2019;42:1777–1787. doi: 10.1007/s10753-019-01040-y. [DOI] [PubMed] [Google Scholar]
- 34.Lu J, Liu X, Tian Y, Li H, Ren Z, Liang S, Zhang G, Zhao C, Li X, Wang T, Chen D, Kuang W, Zhu M. Moxibustion exerts a neuroprotective effect through antiferroptosis in Parkinson's disease. Evid Based Complement Alternat Med 2019. 2019 doi: 10.1155/2019/2735492. 2735492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Manni L, Albanesi M, Guaragna M, Barbaro Paparo S, Aloe L. Neurotrophins and acupuncture. Auton Neurosci. 2010;157:9–17. doi: 10.1016/j.autneu.2010.03.020. [DOI] [PubMed] [Google Scholar]
- 36.Matsumoto H, Kumon Y, Watanabe H, Ohnishi T, Shudou M, Chuai M, Imai Y, Takahashi H, Tanaka J. Accumulation of macrophage-like cells expressing NG2 proteoglycan and Iba1 in ischemic core of rat brain after transient middle cerebral artery occlusion. J Cereb Blood Flow Metab. 2008;28:149–163. doi: 10.1038/sj.jcbfm.9600519. [DOI] [PubMed] [Google Scholar]
- 37.Nakano M, Tamura Y, Yamato M, Kume S, Eguchi A, Takata K, Watanabe Y, Kataoka Y. NG2 glial cells regulate neuroimmunological responses to maintain neuronal function and survival. Sci Rep. 2017;7:42041. doi: 10.1038/srep42041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nishiyama A, Komitova M, Suzuki R, Zhu X. Polydendrocytes (NG2 cells): multifunctional cells with lineage plasticity. Nat Rev Neurosci. 2009;10:9–22. doi: 10.1038/nrn2495. [DOI] [PubMed] [Google Scholar]
- 39.Ozerdem U, Grako KA, Dahlin-Huppe K, Monosov E, Stallcup WB. NG2 proteoglycan is expressed exclusively by mural cells during vascular morphogenesis. Dev Dyn. 2001;222:218–227. doi: 10.1002/dvdy.1200. [DOI] [PubMed] [Google Scholar]
- 40.Paxinos G, Franklin K. Paxinos and Franklin's the mouse brain in stereotaxic coordinates. London, Waltham, San Diego, Elsevier Inc. 2013 [Google Scholar]
- 41.Perego C, Fumagalli S, De Simoni MG. Temporal pattern of expression and colocalization of microglia/macrophage phenotype markers following brain ischemic injury in mice. J Neuroinflammation. 2011;8:174. doi: 10.1186/1742-2094-8-174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Smirkin A, Matsumoto H, Takahashi H, Inoue A, Tagawa M, Ohue S, Watanabe H, Yano H, Kumon Y, Ohnishi T, Tanaka J. Iba1(+)/NG2(+) macrophage-like cells expressing a variety of neuroprotective factors ameliorate ischemic damage of the brain. J Cereb Blood Flow Metab. 2010;30:603–615. doi: 10.1038/jcbfm.2009.233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Soligo M, Nori SL, Protto V, Florenzano F, Manni L. Acupuncture and neurotrophin modulation. Int Rev Neurobiol. 2013;111:91–124. doi: 10.1016/B978-0-12-411545-3.00005-5. [DOI] [PubMed] [Google Scholar]
- 44.Soligo M, Piccinin S, Protto V, Gelfo F, De Stefano ME, Florenzano F, Berretta E, Petrosini L, Nistico R, Manni L. Recovery of hippocampal functions and modulation of muscarinic response by electroacupuncture in young diabetic rats. Sci Rep. 2017;7:9077. doi: 10.1038/s41598-017-08556-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Stallcup WB, You WK, Kucharova K, Cejudo-Martin P, Yotsumoto F. NG2 proteoglycan-dependent contributions of pericytes and macrophages to brain tumor vascularization and progression. Microcirculation. 2016;23:122–133. doi: 10.1111/micc.12251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sugimoto K, Nishioka R, Ikeda A, Mise A, Takahashi H, Yano H, Kumon Y, Ohnishi T, Tanaka J. Activated microglia in a rat stroke model express NG2 proteoglycan in peri-infarct tissue through the involvement of TGF-beta1. Glia. 2014;62:185–198. doi: 10.1002/glia.22598. [DOI] [PubMed] [Google Scholar]
- 47.Tan TT, Wang D, Huang JK, Zhou XM, Yuan X, Liang JP, Yin L, Xie HL, Jia XY, Shi J, Wang F, Yang HB, Chen SJ. Modulatory effects of acupuncture on brain networks in mild cognitive impairment patients. Neural Regen Res. 2017;12:250–258. doi: 10.4103/1673-5374.200808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tei N, Tanaka J, Sugimoto K, Nishihara T, Nishioka R, Takahashi H, Yano H, Matsumoto S, Ohue S, Watanabe H, Kumon Y, Ohnishi T. Expression of MCP-1 and fractalkine on endothelial cells and astrocytes may contribute to the invasion and migration of brain macrophages in ischemic rat brain lesions. J Neurosci Res. 2013;91:681–693. doi: 10.1002/jnr.23202. [DOI] [PubMed] [Google Scholar]
- 49.Wang P, Tian WW, Song J, Guan YF, Miao CY. Deficiency of NG2+ cells contributes to the susceptibility of stroke-prone spontaneously hypertensive rats. CNS Neurosci Ther. 2011;17:327–332. doi: 10.1111/j.1755-5949.2011.00265.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Xie D, Shen F, He S, Chen M, Han Q, Fang M, Zeng H, Chen C, Deng Y. IL-1beta induces hypomyelination in the periventricular white matter through inhibition of oligodendrocyte progenitor cell maturation via FYN/MEK/ERK signaling pathway in septic neonatal rats. Glia. 2016;64:583–602. doi: 10.1002/glia.22950. [DOI] [PubMed] [Google Scholar]
- 51.Yang A, Wu HM, Tang JL, Xu L, Yang M, Liu GJ. Acupuncture for stroke rehabilitation. Cochrane Database Syst Rev. 2016 doi: 10.1002/14651858.CD004131.pub3. CD004131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yang Y, Kimura-Ohba S, Thompson JF, Salayandia VM, Cosse M, Raz L, Jalal FY, Rosenberg GA. Vascular tight junction disruption and angiogenesis in spontaneously hypertensive rat with neuroinflammatory white matter injury. Neurobiol Dis. 2018;114:95–110. doi: 10.1016/j.nbd.2018.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhang R, Chopp M, Zhang ZG. Oligodendrogenesis after cerebral ischemia. Front Cell Neurosci. 2013;7:201. doi: 10.3389/fncel.2013.00201. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Western blot images of regions used for figure 4 marked with rectangles and molecular size marker (kDa).
EA stimulation on cytokine expression in perilesional striatum of mice at 21 days after MCAO.
(A, B) Cytokine array results (A) and quantitative analysis of expression (B). The figure shows sample cytokine array data from the perilesional striatum of the MCAO and MCAO + EA1 groups. The red circles indicate upregulated cytokine and blue ones downregulated cytokine compared to MCAO and MCAO + EA1. All data are shown as mean ± SEM (n = 4). CCL6: Chemokine ligand 6; CCL12: chemokine ligand 12; CRP: C-reactive protein; CX3CL1: CX3C chemokine receptor 1; EA1: electroacupuncture at 1 mA; LDLR: low density lipoprotein receptor; Lix: CXC chemokine; MCAO: middle cerebral artery occlusion; MMP3: matrix metallopeptidase 3; MMP9: matrix metallopeptidase 9; NGAL: neutrophil gelatinase-associated lipocalin.
EA stimulation of NG2-mEGFP mice on cellular phenotypes of NG2-expressing cells and the expression of BDNF and GSK3β in the contralateral striatum of mice at 21 days after MCAO.
Quantitative analysis of the flow cytometry and (A and C) and histograms showing the numbers of GFP + DAPI-positive cells also expressing BrdU, CC1, CD31, CD68, and GSK3β, and the number of DAPI-positive cells expressing BDNF (B and D, respectively). There were no significant differences between the MCAO and MCAO + EA1 groups. All data are shown as mean ± SEM (n = 5). BDNF: brain-derived neurotrophic factor; BrdU: Bromodeoxyuridine; DAPI: 4',6-diamidino-2-phenylindole; EA1: electroacupuncture at 1 mA; GFP: green fluorescent protein; GSK3β: glycogen synthase kinase 3 beta; MCAO: middle cerebral artery occlusion; mEGFP: membrane-anchored enhanced green fluorescent protein; NG2: neural/glial antigen 2.


