Abstract
The impressive technological advances in recent years have rapidly translated into the shift of endoscopic ultrasound (EUS) from diagnostic modality into an interventional and therapeutic tool. Despite the great advance in its diagnosis, the majority of pancreatic adenocarcinoma cases are inoperable when diagnosed, thus demanding alternative optional therapies. EUS has emerged as an easy, minimally invasive modality targeting this carcinoma with different interventions that have been reported recently. In this review we summarize the evolving role of interventional therapeutic EUS in pancreatic adenocarcinoma management.
Keywords: Endoscopic ultrasound, Pancreas, Cancer, Management, Palliative
Core Tip: The prognosis of pancreatic adenocarcinoma is poor in advanced stages. Several studies were conducted recently to assess the effect of different treatment options provided through endoscopic ultrasound (EUS). We present a comprehensive review on the role of EUS in unresectable pancreatic adenocarcinoma treatment while exploring its effect on survival and palliation. We found that EUS-guided intervention is feasible with excellent technical success, limited adverse events, a beneficial effect on cancer-associated pain and an as-yet unknown effect on survival. For EUS-assisted therapies there are still many unknowns and unanswered questions, prompting the need for additional prospective randomized controlled studies comparing the different treatment approaches combined with chemo +/- radiotherapy with respect to success, efficacy, safety and survival.
INTRODUCTION
Pancreatic adenocarcinoma is the seventh leading cause of cancer death worldwide with poor prognosis according to the 2020 GLOBOCAN cancer estimates[1]. About half of patients are diagnosed with metastatic disease and 30% with locally advanced disease and are deprived from the only potential cure of surgical intervention[2]. The median overall survival for stages IV and III is of 2-3 and 7-11 mo, respectively[3]. As a result, those patients are usually offered supportive care, palliative chemotherapy and radiology, and palliative surgical interventions. Endoscopic ultrasound (EUS), first introduced about 40 years ago as a diagnostic tool, has quickly gained popularity as an interventional therapeutic tool in a broad range of gastrointestinal, pancreato-biliary and liver diseases due to its high spatial resolution. There are several characteristics of EUS that improve its utility as an interventional therapeutic instrument. The first and most crucial property is its high spatial resolution and the proximity of its transducer to the target lesion, allowing it to access small lesions while avoiding intervening structures, blood vessels and air[4]. The second advantage lies in its minimal invasiveness and high safety profile in targeting pancreatic lesions; these have advanced this modality over interventional radiology and surgery in diverse pancreatic tumor treatment applications[5]. The third advantage is its ability to obtain contrast-enhancement images which seems to improve diagnostic performance in pancreatic masses[2]. The final benefit is the technical advancement in developing devices designed specifically to allow minimally invasive therapeutic interventions[6]. An increasing number of articles reporting these new EUS applications in pancreatic adenocarcinoma have been published, including EUS guided thermal ablation, ethanol ablation, delivery of antitumor agents, brachytherapy, fiducial marker placement (FMP), and EUS-guided celiac plexus neurolysis/block (CPN/B). In this review we summarize the literature dealing with interventional therapeutic EUS in pancreatic adenocarcinoma, aiming to present an updated comprehensive review on this topic.
LITERATURE SEARCH
A search for studies published before August 2021 was performed in the PubMed databases with the keywords EUS or endoscopic ultrasound and any of the following: Carcinoma or adenocarcinoma of pancreas, pancreatic tumor, treatment or therapeutic, intervention, ablation, injection, brachytherapy, fiducial markers and CPN. The search was restricted to articles in the English language and included prospective, retrospective, case series and randomized controlled studies. Review articles and case reports were not included. Subsequently, we generated a state-of-the-art comprehensive review by summarizing the most updated data on EUS-guided intervention published in the last several years and focusing on feasibility, technical success, safety and effect on overall survival and palliation when the data were available.
EUS-GUIDED INTRA-TUMORAL INJECTIONS
Intra-tumoral EUS fine needle injection (EUS-FNI), is a relatively new treat-to-target modality aiming to deliver and potentially achieve high intra-tumor drug concentration while minimizing systemic exposure and toxicity from those drugs[7,8]. This method allows tumor reduction prior to surgery or serves as a palliative treatment in unresectable tumors with mass effect including obstructive symptoms[8]. EUS-FNI enables performance of several therapeutic interventions including chemotherapy, immunotherapy, gene therapy and intra-tumoral implantation. Table 1 demonstrates all studies of EUS-guided intra-tumoral injections.
Table 1.
Human studies reporting endoscopic ultrasound-guided intra-tumoral injection therapies
|
Ref.
|
Study design
|
Cancer stage
|
EUS-guided intervention
|
Patients No.
|
Technical success (%)
|
Median overall survival (mo)
|
Pain palliation
|
Serious adverse events, n
|
| Levy et al[9], 2017 | Prospective | II (n = 3); III (n = 20); IV (n = 13) | Chemotherapy | 36 | 100 | 10.4 | Not reported | 0 |
| Chang et al[10], 2000 | Prospective | II (n = 4); III (n = 3); IV (n = 1) | Immunotherapy | 8 | 100 | 13.2 | Not reported | 0 |
| Irisawa et al[11], 2007 | Prospective | IV (n = 7) | Immunotherapy | 7 | 100 | 9.9 | Not reported | 0 |
| Endo et al[12], 2012 | Prospective | II (n = 1); III (n = 5); IV (n = 3) | Immunotherapy | 9 | 100 | 18 | Not reported | 31 |
| Buscail et al[23], 2015 | Prospective | III (n = 13); IV (n = 9) | Gene therapy | 22 | 100 | 12.6 | Not reported | 0 |
| Hecht et al[14], 2003 | Prospective | III (n = 9); IV (n = 21) | Gene therapy | 21 | 100 | 7.5 | Not reported | 42 |
| Hecht et al[16], 2012 | Prospective | III (n = 27) | Gene therapy | 27/50 | 100 | 9.9 | Not reported | 403 |
| Herman et al[17], 2013 | Prospective | III (n = 95) | Gene therapy | 95/187 | 100 | 11.5 | Not reported | 484 |
| Hanna et al[18], 2012 | Prospective | Unresectable | Gene therapy | 6 | 100 | 6 | Not reported | 15 |
| Hirooka et al[20], 2018 | Prospective | III (n = 9) | Gene therapy | 9 | 100 | 15.5 | Not reported | 26 |
| Nishimura et al[22], 2018 | Prospective | III (n = 5); IV (n = 1) | Gene therapy | 6 | 100 | 5.8 | Not reported | 0 |
| Golan et al[25], 2015 | Prospective | III (n = 15) | Intra-tumoral implantation | 15 | 100 | 15.1 | Not reported | 47 |
Pancreatic fistula (2 patients) and superior mesenteric artery pseudoaneurysm (1 patient).
Sepsis (2 patients), duodenal perforations (2 patients).
The authors did not state whether these adverse events were in the endoscopic ultrasound (EUS) group or in the percutaneous group: Gastrointestinal bleeding (6 patients), deep vein thrombosis (6 patients), pulmonary embolism (2 patients), pancreatitis (2 patients), cholecystitis (1 patient), biliary obstruction (8 patients), cholangitis (6 patients), hypotension (2 patients), bradycardia (1 patient), supraventricular tachycardia (1 patient), splenic artery thrombosis (1 patient), intestinal ischemia (1 patient), staphylococcus infection (1 patient), cerebrovascular accident (1 patient), cardio-pulmonary arrest (1 patient).
The authors did not state what are the serious adverse events and whether these adverse events were in the EUS group or in the percutaneous group.
Hypoglycemia (1 patient).
Perforation of duodenum (1 patient) and hepatic dysfunction 1 patient), but these events were considered not to be related.
Colonic obstruction (1 patient), pancreatitis (1 patient), cholangitis (1 patient), renal failure (1 patient).
CHEMOTHERAPY
A prospective study from Mayo Clinic evaluated EUS-FNI of gemcitabine in 36 patients (long-term data were available in 28 patients) with unresectable pancreatic adenocarcinoma (3 patients with stage II, 20 with stage III and 13 with stage IV). They reported no adverse events, partial response in 25% of patients, stable disease in 57% of patients and down-staging in 20% of stage III patients who underwent surgical resection, with a median of an overall survival of 10.4 mo (95% confidence interval, 2.7-68), and an overall survival of 78%, 44%, and 3% at 6 mo, 12 mo and 5 years, respectively, leading the authors to conclude that this treatment option is feasible, safe, and potentially effective[9].
IMMUNOTHERAPY
Intra-tumoral immunotherapy, including mixed lymphocyte culture and immature dendritic cells, have the ability to induce a tumor-specific immune response which can be effective, not only locally but also on metastatic lesions[7]. The first clinical trial of immunotherapy was published about 20 years ago and enrolled 8 patients with unresectable pancreatic adenocarcinoma who were treated by EUS-FNI of mixed lymphocyte culture (cytoimplant). They showed this treatment option to be feasible without procedure-related complications and with no substantial toxicity. Notably, the median overall survival was 13.2 mo, with tumor response ranging from 'minor' until 'no change'; however, there were no cases of significant or complete tumor response[10]. Later, Irisawa et al[11] reported their experience with seven patients suffering from stage IV gemcitabine non-responsive pancreatic adenocarcinoma who underwent EUS-FNI of immature dendritic cells with radiation therapy administered first in five patients. They showed clinical response in three of the patients with no procedure-related adverse event nor dendritic cell-related toxicity, and with an overall median survival rate of 9.9 mo[11]. Another study from Japan evaluated the feasibility, safety and histological change of preoperative EUS fine-needle injection of immature dendritic cells with OK-432 (immune-potentiating agent) in pancreatic cancer patients. In their study, nine patients were enrolled and compared to a group of 15 patients who were operated without dendritic cell injection. They reported no adverse reaction following injection in the nine patients except for one with transient fever, and no significant difference in postoperative complication incidence between both groups or in the overall median survival. Interestingly, two patients in the injection group survived for more than 5 years without disease recurrence. Analysis of resected specimens in the injection group showed that CD83 + cells significantly accumulated in the regional lymph nodes, as well as Foxp3 + cells in the regional and distant lymph nodes[12].
GENE THERAPY
Gene therapy takes advantage of the preference of oncolytic attenuated adenovirus [ONYX-015 (Onyx Pharmaceuticals, United States)] to selectively replicate in malignant cells, leading to their lysis and death[13]. The first study was performed by Hecht et al[14] who enrolled 21 patients with locally advanced pancreatic adenocarcinoma in a trial of intra-tumoral ONYX-015 injection via EUS in combination with gemcitabine. They demonstrated the feasibility, safety and tolerability of this treatment modality when EUS-FNI was performed through a trans-gastric route with prophylactic antibiotic. However, no convincing evidence of efficacy was shown as only two patients showed partial regression, two showed minor response, and six had stable disease, while 11 had progressive disease, with a median overall survival of 7.5 mo[14]. Furthermore, a subsequent study by Senzer et al[15] demonstrated the safety and efficacy of intra-tumoral injection of TNFerade (GenVec Inc, United States), an adenovirus vector with replication deficiency that carries the human tumor necrosis factor-alpha gene regulated by a radiation-inducible promoter, followed by radiation has been demonstrated in a phase I clinical trial of 30 patients with solid tumors, 21 of 30 patients (70%) demonstrated objective tumor response (five complete, nine partial, and seven minimal responses), with only mild toxicities reported as the most common adverse event, including fever (22%), injection site pain (19%) and chills (19%)[15]. A phase I/II non-randomized study enrolled 50 patients for intra-tumoral TNFerade treatment with 5-fluorouracil and radiotherapy for locally advanced pancreatic cancer (27 patients were administered under EUS guidance and 23 patients through the percutaneous route). Their results showed promise with intra-tumoral TNFerade injection, with an overall median survival of 9.9 mo, and median time-to-tumor progression of 3.6 mo, as one patient had complete response, three had partial response, and twelve had stable disease, while 19 patients had progressive disease. Notably, there was a high safety signal in this study, as 40 serious adverse events were recorded, however we were unable to extract whether these adverse events were in the EUS group or in the percutaneous group, as this information was not supplied by the authors[16]. In a randomized phase III multi-institutional study enrolling 304 patients, 187 were treated with standard of care and TNFerade (95 patients under EUS-guidance and 91 percutaneously) vs 117 who received only standard of care therapy. Although the method was shown to be safe, it did not lead to prolonged survival, as the median overall survival was 10 mo in both the 'standard of care and TNFerade' and the 'standard of care alone' groups. Notably in that study, serious related adverse events occurred in 48 patients (25.7%) of the 'standard of care and TNFerade' group, as compared to 20 patients (16.7%) in the 'standard of care' group (P = 0.13); however, the serious adverse events were not detailed and the authors did not report whether those adverse events occurred in the EUS or the percutaneous sub-group of the 'standard of care and TNFerade' group[17]. BC-819 is a double-stranded DNA plasmid designated to target the expression of diphtheria-toxin gene under the control of H19 regulatory sequences, and thus have the potential to treat cancer with H19 overexpression. The pharmacokinetics, tolerability and safety and preliminary efficacy of intra-tumoral-injected BC-19 were assessed in a phase 1/2a study of nine patients with unresectable pancreatic adenocarcinoma. The authors reported no increase in tumor size 4 wk after receipt of first treatment, down staging and conversion into resectable cancer in two patients and partial response in three patients after 3 mo. Remarkably, only one spontaneously-resolving asymptomatic lipase elevation considered to be an adverse event, occurred. BC-819 combined with systemic chemotherapy may have additive therapeutic benefit in these patients[18]. Another oncolytic virus is HF10 that enjoys the unique property of being a spontaneous mutation product of herpes simplex virus-1 without artificial modification. It has a high affinity to tumor cells and high replication leading to antitumor immune response[19]. A phase I clinical trial of EUS-guided intra-tumoral injection of HF10 in combination with erlotinib and gemcitabine in 10 patients with unresectable locally-advanced pancreatic cancer reported three partial responses, four stable disease and two progressive diseases in the nine subjects who completed the treatment. However, five patients showed Grade III myelosuppression and two patients developed serious adverse events (perforation of duodenum, hepatic dysfunction), though these events were considered to be unrelated to HF10. Two patients underwent R0 surgical resection after down staging. The median progression-free survival was 6.3 mo and the overall survival 15.5 mo[20]. The effect of the synthetic double stranded RNA oligonucleotide, STNM01, known to selectively inhibit the expression of carbohydrate sulfotransferase-15 (CHST-15)[21], was explored by Nishimura et al[22], who injected STNM01 intra-tumorally with EUS-guidance in six patients with unresectable pancreatic cancer. They reported tumor necrosis in biopsy in four patients and significant reduction of CHST15 in two patients, with an overall survival of 15 mo in these two patients, but only 5.7 mo in the other four patients. The authors concluded that EUS-FNI of STNM01 in these patients is safe and feasible[22]. A previous interesting study with 22 patients aimed to assess the effect of CYL-02, a non-viral gene therapy targeted to sensitize pancreatic cells to chemotherapy, reported promising results. Nine patients showed stable disease up to 6 mo following treatment and two of these patients experienced long-term survival, with a median overall survival of 12.6 mo, and without serious adverse events[23].
INTRA-TUMORAL IMPLANTATION
Zorde Khvalevsky et al[24] developed a local prolonged siRNA delivery system (Local Drug EluteR, LODER) releasing siRNA against the mutated KRAS (siG12D LODER), enabling siRNA protection from degradation and prolonged periods of intra-tumoral slow release with proved therapeutic efficacy[24]. The tolerability, efficacy and safety of EUS-guided intra-tumoral injection of miniature biodegradable implant siG12D-LODER, releasing a specific silencing RNA against K-RAS mutations in combination with chemotherapy for locally advanced pancreatic cancer patients was shown in a study by Golan et al[25]. Their open-label Phase 1/2a study included 15 patients; of the 12 patients analyzed by computed tomography (CT) scans, 10 demonstrated stable disease and two showed partial response. Seven patients had a decrease in tumor marker CA19-9. The median overall survival was 15.12 mo. Serious adverse events were reported in four patients[25].
COMBINATION’S INJECTION
A recent phase 1 study by Lee et al[26] evaluating the safety and tolerability of Ad5-yCD/mutTK(SR39)rep-ADP (Ad5-DS), a replication-competent adenovirus-mediated double-suicide gene therapy in combination with gemcitabine, demonstrated the good tolerability and safety of this combination[26]. Five of their nine patients with inoperable locally-advanced pancreatic cancer treated with the combination of intravenous gemcitabine and EUS-FNI of dendritic cell followed by intravenous infusion of lymphokine-activated killer cells, showed response without treatment-related severe adverse events[27]. Another study evaluated the feasibility, safety and efficacy of EUS-FNI of zoledronate-pulsed dendritic cell combined with intravenous administration of αβT cells and gemcitabine in 15 patients with locally-advanced pancreatic adenocarcinoma. Most of these patients had clinical response and seven had stable disease; the authors concluded that this combination may have a therapeutic benefit. Adverse events were reported in four patients, two of which were related to gemcitabine[28].
EUS-GUIDED ABLATION THERAPIES
Dedicated ablation devices are designed to perform specific ablative procedures in patients with inoperable pancreatic cancer, or who are at high surgical risk or refuse surgery. The procedures include ethanol ablation, thermal ablation including hybrid cryothermal ablation, radiofrequency ablation (RFA), Photodynamic ablation (PDT) and laser ablation[6]. Table 2 shows all studies of EUS-guided ablation therapies.
Table 2.
Human studies reporting endoscopic ultrasound-guided ablation therapies
|
Ref.
|
Study design
|
Cancer stage
|
EUS-guided intervention
|
Patients No.
|
Technical success (%)
|
Median overall survival (mo)
|
Pain palliation (patients %)
|
Serious adverse events, n
|
| Facciorusso et al[31], 2017 | Prospective | III (n = 50); IV (n = 15) | Ethanol ablation | 65 | 100 | 8.3 | 90.7 at week 2 | 0 |
| Song et al[35], 2016 | Prospective | III (n = 4); IV (n = 2) | RFA | 6 | 100 | -1 | Not reported | 0 |
| Crinò et al[36], 2018 | Prospective | III (n = 8) | RFA | 8 | 100 | -1 | Not reported | 0 |
| Scopelliti et al[37], 2018 | Prospective | III (n = 10) | RFA | 10 | 100 | -1 | Not reported | 0 |
| Paiella et al[38], 2018 | Retrospective | Not reported | RFA | 30 | 100 | 15 | Not reported | 0 |
| Bang et al[39], 2019 | Prospective | II (n = 2); III (n = 3); IV (n = 7) | RFA | 12 | 100 | Not reported | Significant | 0 |
| Arcidiacono et al[41], 2012 | Prospective | III (n = 22) | HCA | 22 | 72.8 | 6 | Not reported | 12 |
| DeWitt et al[45], 2019 | Prospective | III (n = 12) | PDA | 12 | 100 | 11.5 | Not reported | 0 |
| Di Matteo et al[46], 2018 | Prospective | III (n = 9) | Laser ablation | 9 | 100 | 7.4 | Not reported | 0 |
Feasibility studies not aimed to assess impact on overall survival.
Minor bleeding in duodenal lumen successfully stopped by hemoclips (1 patient). RFA: Radiofrequency ablation; HCA: Hybrid cryothermal ablation; PDA: Photodynamic ablation; EUS: Endoscopic ultrasound.
ETHANOL ABLATION
Ethanol is an attractive ablative agent due to its wide availability, low cost and efficacy. Once injected it causes rapid coagulation necrosis resulting from protein denaturation, cell membrane lysis and vascular occlusion[29]. Its superiority over the percutaneous route resides in its proximity to the pancreas, allowing precise localization and measurement of the lesion with real-time imaging, thus minimizing damage to surrounding normal tissue[30]. To date, we could identify only one study that reported the effect of EUS-guided ethanol injection in pancreatic adenocarcinoma: Facciorusso et al[31] evaluated pain management in 123 patients with pancreatic adenocarcinoma, as well as the treatment's effect on overall survival. That study compared the efficacy and safety of EUS-guided tumor ethanol ablation in combination with CPN (65 patients) vs CPN alone (58 patients). The combination therapy was shown to be significantly superior to CPN alone in terms of pain relief (P = 0.005) and complete pain response (P = 0.003), with additional survival benefit (8.3 mo vs 6.5 mo, respectively). The median duration of pain relief lasted for 18 d (range 13-20) in the combined group, as compared to 10 d (range 7-14) in the CPN group (P = 0.004)[31].
RFA
The high temperature, ranging between 60-100 °C induced by RFA results in irreversible cellular damage, apoptosis and coagulative necrosis[32]. Additionally, it is believed that RFA induces immunomodulatory activity, with anticancer effect[33]. EUS-guided RFA is a minimally invasive, feasible, easy and safe ablative modality that constitutes the ablative modality of choice for several solid tumors[34]. Several small-case series recently assessed this modality in pancreatic cancer. Three feasibility studies were performed in this field; the first was by Song et al[35] in which six patients with unresectable pancreatic ductal adenocarcinoma were enrolled to assess feasibility and safety of this modality. This study demonstrated an ablation area within the tumor by contrast- enhanced EUS, with no major side effects (two patients suffered from mild abdominal pain) and with complete technical success[35]. The second study by Crinò et al[36] evaluated the technical success, feasibility and safety of EUS-guided RFA in eight patients with pancreatic adenocarcinoma and one patient with renal cell metastasis; they reported feasibility in eight patients, with no major side effects. One- and 30-d' CT demonstrated necrosis of about 30% of the tumor. Three patients reported mild abdominal pain. One of the nine patients was excluded due to a large necrotic portion[36]. The third study, by Scopelliti et al[37] enrolled 10 patients with pancreatic ductal adenocarcinoma, and reported success in all patients, with no major adverse events, and with scan-documented area of necrosis within tumor at 30 d post-ablation[37]. A study of 30 patients examined whether SMAD4 status affects post-RFA disease-specific survival in patients with locally advanced pancreatic adenocarcinoma. Results showed that patients with wild-type SMAD4 survived significantly longer than patients with mutant type SMAD4 (22 mo vs 12 mo, respectively) with an overall estimated post-RFA disease-specific survival of 15 mo, probably indicating that this gene may help in selecting patients for RFA[38]. Moreover, a recent study by Bang et al[39] assessed the role of EUS-guided RFA for pain relief in pancreatic cancer as compared to EUS-guided CPN, and revealed that the EUS-guided RFA was associated with significant improvement in pain associated with pancreatic cancer (P < 0.05), in addition to less-severe gastrointestinal symptoms, with better quality of life and emotional functioning[39].
HYBRID CRYOTHERMAL ABLATION
Using a flexible hybrid bipolar cryotherm probe, it is possible to combine radiofrequency with cryotechnology. Cryo is believed to induce a systemic inflammatory response with an antitumor response in addition to the thermal ablation induced by RFA[40]. Only one prospective clinical trial of this type was conducted in 22 patients with locally-advanced pancreatic cancer. Treating them with this hybrid intervention was technically successful in 72.8% of patients, with median post-ablation survival of 6 mo. The few late complications were mainly related to tumor progression, and the single immediate complication of duodenal bleeding was resolved by placing of hemo-clips[41]. However, more data are needed to assess this treatment modality.
PDT
PDT is a tumor-specific ablative treatment performed through a combination of photosensitizing drug administration with EUS-guided light irradiation, resulting in cell death by generating oxygen free radicals[42,43]. EUS-guided PDT was first published by Choi et al[44], who reported the first preliminary feasibility data for EUS-PDT in patients suffering from locally advanced pancreaticobiliary malignancies. They enrolled four patients, the first with pancreatic tail carcinoma, the second with distal CBD carcinoma and two patients with carcinoma of the caudate lobe of the liver. The treatment was effective and safe, as it induced a necrotic area of 4 cm3 without side effects. Notably, disease remained stable for a mean of 5 mo[44]. Recently, a prospective, dose-escalation phase 1 study of 12 patients with locally-advanced pancreatic cancer, treated with EUS-PDT and subsequent gemcitabine therapy 25 d later, showed tumor necrosis in 50% of patients, median progression-free and overall survival were 2.6 and 11.5 mo, respectively. Two patients were operated on, one of them had a complete response and the other one had a residual 2-mm tumor. Notably, there were eight serious adverse events but none related to EUS or EUS-PDT[45]. More data are needed to assess EUS-guided PDT on survival and palliation.
LASER ABLATION
To date, EUS-guided laser ablation has been reported by a single clinical human study that enrolled nine patients with unresectable pancreatic ductal adenocarcinoma who were unresponsive to previous chemotherapy. These patients were treated by laser ablation suing neodymium-yttrium aluminum garnet (Nd:YAG) laser light with different power settings, by flexible fiber, introduced through 22-gauge fine needle aspiration. The coagulative necrotic ablation area was demonstrated by CT scans at 24 h, 7 and 30 d, and was shown to be optimal with power setting of 4 W/1000J with the largest ablation area without adverse events. The median overall survival was 7.4 mo[46]. However, no data regarding palliative effect was reported, thus more data are warranted.
EUS-FMP
Chemoradiation is offered as adjuvant or neoadjuvant to patients with pancreatic adenocarcinoma; however, one of the major challenges with radiation is the proximity of the pancreas to several vital organs. Intensity-modulated radiation therapy was shown to reduce radiation-induced toxicity in these organs in patients with pancreatic and ampullary cancers[47]. Intra-tumoral FMP serves as a landmark enabling accurate radiation targeting of the tumor with minimal harm to neighboring structures. To date, only several feasibility studies were reported addressing safety and technical success, without reporting the effect on overall survival. The first report of EUS-FMP was published in 2006 by Pishvaian et al[48] who successfully placed fiducial markers in six of seven pancreatic cancer patients, with no observed complications[48]. After that, several feasibility studies on FMP under EUS-guidance were reported, showing this to be an easy and safe modality with excellent technical success, enabling accurate radiation targeting and without procedure related adverse events in patients with pancreatic cancer[49-54]. As mentioned earlier, to date, the studies on EUS-guided FMP have reported only technical success and adverse events, with no data on survival and palliative benefit, necessitating further studies to assess their therapeutic effect (Table 3).
Table 3.
Human studies reporting endoscopic ultrasound-guided fiducial markers placement, -brachytherapy and -celiac plexus neurolysis
|
Ref.
|
Study design
|
Cancer stage
|
EUS-guided intervention
|
Patients No.
|
Technical success (%)
|
Median overall survival (mo)
|
Pain palliation (patients %)
|
Serious adverse events, n
|
| Pishvaian et al[48], 2006 | Prospective | Unresectable | FMP | 7 | 85.7 | -1 | Not reported | 0 |
| Choi et al[49], 2014 | Prospective | Unresectable | FMP | 29 | 100 | -1 | Not reported | 12 |
| Varadarajulu et al[50], 2010 | Prospective | III (n = 9) | FMP | 9 | 100 | Not reported | Not reported | 0 |
| Park et al[51], 2010 | Prospective | III (n = 57) | FMP | 57 | 94 | -1 | Not reported | 0 |
| Sanders et al[52], 2010 | Prospective | III (n = 36); Recurrent (n = 15) | FMP | 51 | 90 | -1 | Not reported | 12 |
| Dávila et al[53], 2014 | Prospective | II (n = 1); III (n = 22) | FMP | 23 | 100 | -1 | Not reported | 0 |
| Khashab et al[54], 2012 | Retrospective | III (n = 39) | FMP | 39 | 100 | -1 | Not reported | 0 |
| Sun et al[57], 2012 | Prospective | III (n = 8) | Brachytherapy | 8 | 100 | 8.3 | 50 at week 33 | 0 |
| Sun et al[58], 2006 | Prospective | III (n = 8); IV (n = 7) | Brachytherapy | 15 | 100 | 10.6 | 30 at week 42 | 33 |
| Jin et al[59], 2008 | Prospective | II (n = 4); III (n = 10); IV (n = 8) | Brachytherapy | 22 | 100 | 9 | 81.8 at week 1 | 0 |
| Sun et al[60], 2017 | Retrospective | III (n = 18); IV (n = 24) | Brachytherapy | 42 | 100 | 9 | Not reported | 0 |
| Wiersema et al[62], 1996 | Prospective | Unresectable | CPN | 29 | 100 | Not reported | 86 at week 2; 84 at week 4; 79 at week 8; 88 at week 12 | 0 |
| Levy et al[64], 2019 | Prospective | II (n = 2); III (n = 27); IV (n = 31) | CPN | 60 | 100 | 10.46 | 40.4 at week 12 | 0 |
| Seicean et al[65], 2013 | Prospective | Unresectable | CPN | 32 | 100 | Not reported | 75 at week 2 | 0 |
| Facciorusso et al[31], 2017 | Prospective | III (n = 48); IV (n = 10) | CPN | 58 | 100 | 6.5 | 70.6 at week 2 | 0 |
Feasibility studies not aimed to assess impact on overall survival.
Pancreatitis supportively treated (1 patient).
Pancreatitis complicated with pseudocyst formation (3 patients).
FMP: Fiducial markers placements; CPN: Celiac plexus neurolysis; EUS: Endoscopic ultrasound.
EUS-GUIDED BRACHYTHERAPY
EUS-guided brachytherapy is defined as the implantation of radioactive seeds near the pancreatic tumorous tissue, followed by exposure of the seeds to steady emissions of gamma rays which lead to localized ablative effect. About two decades ago, Sun et al[55] showed that EUS-guided radioactive seeds into pancreatic tissue in a porcine model is a feasible and safe modality for brachytherapy[55]. The favored radioactive seeds in brachytherapy of the rapidly growing pancreatic cancer are iodine-125 due to their long halftime of 59.7 d, which is appropriate in targeting such rapidly-growing tumors. Importantly, the dose rate of these radioactive seeds is low and their penetration depth does not exceed 1.7 cm, thus minimizing radiation exposure and injury to the neighboring organs[56]. Only a few human studies have been conducted with EUS-guided brachytherapy for pancreatic adenocarcinoma. Sun et al[57] reported eight patients with locally advanced pancreatic adenocarcinoma who underwent EUS-guided brachytherapy and showed a favorable effect of this modality on pain severity which was ameliorated in four of the eight patients. The pain decrease lasted for 3.5 mo and the patients had a median overall survival time of 8.3 mo with no procedure- or treatment-related adverse events[57]. Another study by Sun et al[58] reported this treatment modality in 15 patients, with 5 of 15 patients (33.3%) experiencing clinical benefit as assessed by pain reduction and improved Karnofsky performance status score, with a median time-to-achieve clinical benefit of 2.2 mo. Notably, the median overall survival was 10.6 mo, with only three cases of serious complications of pancreatitis complicated with pseudocysts, and no life-threatening adverse events[58]. Similar results were reported by Jin et al[59] in 22 patients who showed partial remission in 13.6% of the patients and stable disease in 45.5% during a 4-wk period. Cancer-related pain improved in 18 patients (81.8%) at 1 wk after the intervention, with an estimated median overall survival of 9 mo and no treatment-related adverse events[59]. Finally, the most recent study of this modality performed by Sun et al[60] was in 2017 and included 42 patients; once again the research group demonstrated its safety and efficacy, a median overall survival of 9 mo, and no serious adverse events reported[60] (Table 3).
EUS-CPN/B
Abdominal and back pain is a common complaint in pancreatic adenocarcinoma, occurring in about 80% of patients, and it is severe in the majority of patients[61]. Because most patients are diagnosed at an advanced stage, the treatment is mostly palliative, including pain control. The WHO recommends a step-up approach for the control of pancreatic cancer pain, beginning with non-opioid analgesics and progressing to opioid analgesics with increasing dose according to need. Unresponsive patients, those with intolerable side effects, may be candidates for EUS-CPN/B (October 14, 2008. WHO Steering Group on Pain Guidelines). The first description of EUS-CPN/B was by Wiersema et al[62] who reported the first human study on EUS-guided brachytherapy in 1996, injection of bupivacaine and 98% dehydrated absolute alcohol in 29 patients with pancreatic adenocarcinoma. Pain score improved in 86%, 84%, 79% and 88% at weeks 2, 4, 8 and 12 post intervention, respectively[62]. Consequently, this therapy rapidly gained popularity as safe and minimally-invasive with the advantage of real-time imaging of blood vessels, compared to the percutaneous route. CPN/B is achieved by alcohol or phenol injection into or around the celiac plexus/ganglion, resulting in its permanent chemical ablation, while CPB is achieved by injecting a corticosteroid in combination with long-acting anesthetic, thus inhibiting pain transmission to the brain[63]. A recent study by Levy et al[64] reported the efficacy of EUS-guided CPN/B on the pain score of 60 patients with a pain response rate in 40.4% at 12 wk after intervention, and with an overall survival rate of 10.46 mo[64]. Similarly, another recent study by Facciorusso et al[31] reported efficacy in 58 patients, among them 41 patients (70.6%) who achieved pain relief within a median time of 5 d and median pain duration relief of 10 wk, with an overall survival rate of 6.5 mo[31]. The beneficial effect of this modality was shown in a previous study by Seicean et al[65] who reported significant pain improvement in 24 (75%) out of 32 patients, and without significant adverse events[65]. Minor side effects of CPN/B including abdominal pain, diarrhea and hypotension due to autonomic nervous system disruption are usually self-limiting. Rare serious adverse events were reported in case reports, including fatal celiac artery thrombosis causing infarction[66], paralysis from anterior spinal cord infection[67] and necrotic gastric perforation[68]. Given the high efficacy of EUS-guided EUS-CPN/B and rarity of adverse events, the latest (1/2020) version of the National Comprehensive Cancer Network (NCCN) guidelines, recommends EUS-CPN for pain palliation in severe pain unresponsive to around-the-clock analgesics or undesirable analgesics side effects[69] (Table 3).
COMBINED EUS AND ERCP IN PANCREATIC ADENOCARCINOMA TREATMENT
Bile duct obstruction with resultant obstructive jaundice and occasional-disabling pruritus is among the most common symptoms of pancreatic head adenocarcinoma. It is usually drained through bile duct stenting introduced via ERCP. Employing the beneficial effect of brachytherapy using the radioactive seeds iodine-125, Liu et al[70] reported that brachytherapy through a preloaded pancreatic stent with iodine-125 seeds, was feasible and safe in an animal experiment using pigs[70]. Two years later, the Liu et al[71] group reported the feasibility and tolerability, in a pilot study, of combined radioactive stents with metallic and/or plastic stent in peripancreatic head advanced carcinomas, with stable disease in 72.7% of patients[71]. A recent retrospective study evaluated the role of EUS and/or percutaneous ultrasound-guided iodine-125 seed implantation in 50 patients with unresectable pancreatic carcinoma combined with prior biliary stenting via ERCP vs biliary stenting alone in 51 patients. They reported longer survival, increased pain reduction with improved life quality, postponed gastric outlet obstruction and longer stent patency in the combination treatment group[72].
SUMMARY
Overall, we identified 12 prospective studies including 261 patients, most in stage III of disease, that utilized EUS-guided intra-tumoral injection therapies and mainly reported effect on patient survival. These studies reported complete technical success without significant effect on overall survival rate, but with several severe adverse events varying in occurrence among the studies. Similarly, in the EUS-guided ablation therapies, we identified one retrospective and eight prospective studies that included 174 patients and reported excellent technical success and minor adverse events, but an inconclusive effect on survival, as half of the studies were feasibility studies not reporting overall survival. Only two of those studies reported pain palliation, however the palliation was significant, thus leading to a hope for performing this treatment for palliative purposes. Finally, we identified seven studies on FMPs, most of them were feasibility studies showing high technical success and minor adverse events. Four studies on brachytherapy included 87 patients and four studies on CPN including 179 patients, with significant improvement in pain ranging from 33% to 90% of patients, no survival benefit and no serious procedure-related adverse events (Table 4).
Table 4.
Summary of efficacy and safety of endoscopic ultrasound-guided angio-therapy procedures
|
Procedure
|
Intra-tumoral injection therapies
|
Ablation therapies
|
Fiducial markers placement, brachytherapy and celiac plexus neurolysis
|
| Technical success | High | High | High |
| Safety (complications) | Uncertain1 | Minor | Minor |
| Efficacy | |||
| Survival | Modest | None | None |
| Palliation | Not reported | Encouraging | High |
| Mortality | None | None | None |
The two studies that had the highest adverse events rate did not state whether they were in the endoscopic ultrasound or in the percutaneous group. See Table 1.
CONCLUSIONS
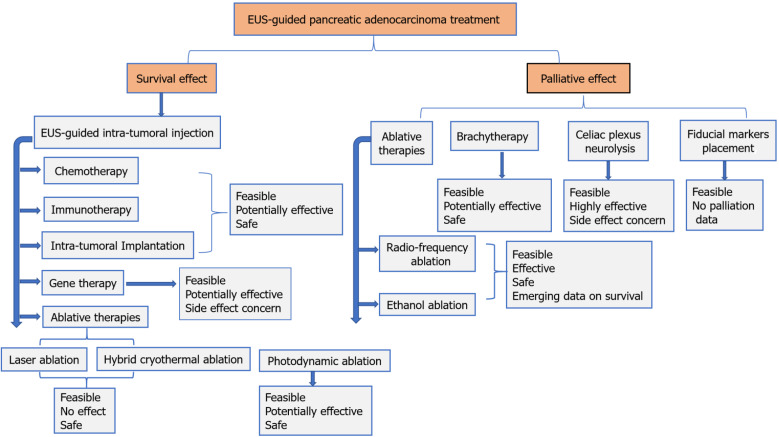
In recent years we have witnessed a great advance in interventional and therapeutic EUS. With these developments, EUS has become the preferred alternative for intra-tumoral injections, ablative therapies, implantation therapies, FMP, brachytherapy, and CPN/B in advanced pancreatic adenocarcinoma. Most EUS-FNI treatments are still far from optimal, and are still in their early stage with little available data, generated from small trials. Figure 1 summarizes the available treatment options for pancreatic adenocarcinoma. The EUS-guided ablation therapies, although encouraging, are far from being standardized. These techniques are in the midst of a long process, necessitating the performance of large prospective randomized controlled studies that compare the different treatment approaches combined with chemo +/- radiotherapy, with respect to success, efficacy, safety and survival. Finally, EUS-guided FMP and brachytherapy are easy, safe and promising modalities, but studies comparing them with the conventional approach of radiotherapy are lacking, while EUS-guided CPN/B is a feasible and accepted tool in pancreatic cancer-related pain control.
Figure 1.
Demonstrates the available endoscopic ultrasound-guided treatment options in pancreatic adenocarcinoma. EUS: Endoscopic ultrasound.
Footnotes
Conflict-of-interest statement: The authors declare no conflict of interest.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Peer-review started: September 28, 2021
First decision: November 18, 2021
Article in press: January 6, 2022
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Israel
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Jin ZD, Nakahodo J S-Editor: Fan JR L-Editor: Filipodia P-Editor: Fan JR
Contributor Information
Wisam Sbeit, Department of Gastroenterology, Galilee Medical Center, Faculty of Medicine in the Galilee, Bar-Ilan University, Nahariya 2221006, Israel.
Bertrand Napoléon, Department of Endoscopy Unit, Private Hospital Jean Mermoz, Ramsay Generale de Sante, Lyon 69008, France.
Tawfik Khoury, Department of Gastroenterology, Galilee Medical Center, Faculty of Medicine in the Galilee, Bar-Ilan University, Nahariya 2221006, Israel. tawfik.khoury.83@gmail.com.
References
- 1.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Werner J, Combs SE, Springfeld C, Hartwig W, Hackert T, Büchler MW. Advanced-stage pancreatic cancer: therapy options. Nat Rev Clin Oncol. 2013;10:323–333. doi: 10.1038/nrclinonc.2013.66. [DOI] [PubMed] [Google Scholar]
- 3.Worni M, Guller U, White RR, Castleberry AW, Pietrobon R, Cerny T, Gloor B, Koeberle D. Modest improvement in overall survival for patients with metastatic pancreatic cancer: a trend analysis using the surveillance, epidemiology, and end results registry from 1988 to 2008. Pancreas. 2013;42:1157–1163. doi: 10.1097/MPA.0b013e318291fbc5. [DOI] [PubMed] [Google Scholar]
- 4.Andanappa HK, Dai Q, Korimilli A, Panganamamula K, Friedenberg F, Miller L. Acoustic liver biopsy using endoscopic ultrasound. Dig Dis Sci. 2008;53:1078–1083. doi: 10.1007/s10620-008-0211-4. [DOI] [PubMed] [Google Scholar]
- 5.Venkatachalapathy S, Nayar MK. Therapeutic endoscopic ultrasound. Frontline Gastroenterol. 2017;8:119–123. doi: 10.1136/flgastro-2016-100774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rimbaș M, Rizzatti G, Larghi A. EUS-guided ablation of pancreatic neoplasms. Minerva Gastroenterol (Torino) 2021 doi: 10.23736/S2724-5985.21.02866-7. [DOI] [PubMed] [Google Scholar]
- 7.Larghi A, Rimbaș M, Rizzatti G, Carbone C, Gasbarrini A, Costamagna G, Alfieri S, Tortora G. Endoscopic ultrasound-guided therapies for pancreatic solid tumors: An overview. Semin Oncol. 2021;48:95–105. doi: 10.1053/j.seminoncol.2021.01.004. [DOI] [PubMed] [Google Scholar]
- 8.Kaplan J, Khalid A, Cosgrove N, Soomro A, Mazhar SM, Siddiqui AA. Endoscopic ultrasound-fine needle injection for oncological therapy. World J Gastrointest Oncol. 2015;7:466–472. doi: 10.4251/wjgo.v7.i12.466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Levy MJ, Alberts SR, Bamlet WR, Burch PA, Farnell MB, Gleeson FC, Haddock MG, Kendrick ML, Oberg AL, Petersen GM, Takahashi N, Chari ST. EUS-guided fine-needle injection of gemcitabine for locally advanced and metastatic pancreatic cancer. Gastrointest Endosc. 2017;86:161–169. doi: 10.1016/j.gie.2016.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chang KJ, Nguyen PT, Thompson JA, Kurosaki TT, Casey LR, Leung EC, Granger GA. Phase I clinical trial of allogeneic mixed lymphocyte culture (cytoimplant) delivered by endoscopic ultrasound-guided fine-needle injection in patients with advanced pancreatic carcinoma. Cancer. 2000;88:1325–1335. doi: 10.1002/(sici)1097-0142(20000315)88:6<1325::aid-cncr8>3.0.co;2-t. [DOI] [PubMed] [Google Scholar]
- 11.Irisawa A, Takagi T, Kanazawa M, Ogata T, Sato Y, Takenoshita S, Ohto H, Ohira H. Endoscopic ultrasound-guided fine-needle injection of immature dendritic cells into advanced pancreatic cancer refractory to gemcitabine: a pilot study. Pancreas. 2007;35:189–190. doi: 10.1097/01.mpa.0000250141.25639.e9. [DOI] [PubMed] [Google Scholar]
- 12.Endo H, Saito T, Kenjo A, Hoshino M, Terashima M, Sato T, Anazawa T, Kimura T, Tsuchiya T, Irisawa A, Ohira H, Hikichi T, Takagi T, Gotoh M. Phase I trial of preoperative intratumoral injection of immature dendritic cells and OK-432 for resectable pancreatic cancer patients. J Hepatobiliary Pancreat Sci. 2012;19:465–475. doi: 10.1007/s00534-011-0457-7. [DOI] [PubMed] [Google Scholar]
- 13.Ries SJ. Elucidation of the molecular mechanism underlying tumor-selective replication of the oncolytic adenovirus mutant ONYX-015. Future Oncol. 2005;1:763–766. doi: 10.2217/14796694.1.6.763. [DOI] [PubMed] [Google Scholar]
- 14.Hecht JR, Bedford R, Abbruzzese JL, Lahoti S, Reid TR, Soetikno RM, Kirn DH, Freeman SM. A phase I/II trial of intratumoral endoscopic ultrasound injection of ONYX-015 with intravenous gemcitabine in unresectable pancreatic carcinoma. Clin Cancer Res. 2003;9:555–561. [PubMed] [Google Scholar]
- 15.Senzer N, Mani S, Rosemurgy A, Nemunaitis J, Cunningham C, Guha C, Bayol N, Gillen M, Chu K, Rasmussen C, Rasmussen H, Kufe D, Weichselbaum R, Hanna N. TNFerade biologic, an adenovector with a radiation-inducible promoter, carrying the human tumor necrosis factor alpha gene: a phase I study in patients with solid tumors. J Clin Oncol. 2004;22:592–601. doi: 10.1200/JCO.2004.01.227. [DOI] [PubMed] [Google Scholar]
- 16.Hecht JR, Farrell JJ, Senzer N, Nemunaitis J, Rosemurgy A, Chung T, Hanna N, Chang KJ, Javle M, Posner M, Waxman I, Reid A, Erickson R, Canto M, Chak A, Blatner G, Kovacevic M, Thornton M. EUS or percutaneously guided intratumoral TNFerade biologic with 5-fluorouracil and radiotherapy for first-line treatment of locally advanced pancreatic cancer: a phase I/II study. Gastrointest Endosc. 2012;75:332–338. doi: 10.1016/j.gie.2011.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Herman JM, Wild AT, Wang H, Tran PT, Chang KJ, Taylor GE, Donehower RC, Pawlik TM, Ziegler MA, Cai H, Savage DT, Canto MI, Klapman J, Reid T, Shah RJ, Hoffe SE, Rosemurgy A, Wolfgang CL, Laheru DA. Randomized phase III multi-institutional study of TNFerade biologic with fluorouracil and radiotherapy for locally advanced pancreatic cancer: final results. J Clin Oncol. 2013;31:886–894. doi: 10.1200/JCO.2012.44.7516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hanna N, Ohana P, Konikoff FM, Leichtmann G, Hubert A, Appelbaum L, Kopelman Y, Czerniak A, Hochberg A. Phase 1/2a, dose-escalation, safety, pharmacokinetic and preliminary efficacy study of intratumoral administration of BC-819 in patients with unresectable pancreatic cancer. Cancer Gene Ther. 2012;19:374–381. doi: 10.1038/cgt.2012.10. [DOI] [PubMed] [Google Scholar]
- 19.Eissa IR, Naoe Y, Bustos-Villalobos I, Ichinose T, Tanaka M, Zhiwen W, Mukoyama N, Morimoto T, Miyajima N, Hitoki H, Sumigama S, Aleksic B, Kodera Y, Kasuya H. Genomic Signature of the Natural Oncolytic Herpes Simplex Virus HF10 and Its Therapeutic Role in Preclinical and Clinical Trials. Front Oncol. 2017;7:149. doi: 10.3389/fonc.2017.00149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hirooka Y, Kasuya H, Ishikawa T, Kawashima H, Ohno E, Villalobos IB, Naoe Y, Ichinose T, Koyama N, Tanaka M, Kodera Y, Goto H. A Phase I clinical trial of EUS-guided intratumoral injection of the oncolytic virus, HF10 for unresectable locally advanced pancreatic cancer. BMC Cancer. 2018;18:596. doi: 10.1186/s12885-018-4453-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Suzuki K, Yokoyama J, Kawauchi Y, Honda Y, Sato H, Aoyagi Y, Terai S, Okazaki K, Suzuki Y, Sameshima Y, Fukushima T, Sugahara K, Atreya R, Neurath MF, Watanabe K, Yoneyama H, Asakura H. Phase 1 Clinical Study of siRNA Targeting Carbohydrate Sulphotransferase 15 in Crohn's Disease Patients with Active Mucosal Lesions. J Crohns Colitis. 2017;11:221–228. doi: 10.1093/ecco-jcc/jjw143. [DOI] [PubMed] [Google Scholar]
- 22.Nishimura M, Matsukawa M, Fujii Y, Matsuda Y, Arai T, Ochiai Y, Itoi T, Yahagi N. Effects of EUS-guided intratumoral injection of oligonucleotide STNM01 on tumor growth, histology, and overall survival in patients with unresectable pancreatic cancer. Gastrointest Endosc. 2018;87:1126–1131. doi: 10.1016/j.gie.2017.10.030. [DOI] [PubMed] [Google Scholar]
- 23.Buscail L, Bournet B, Vernejoul F, Cambois G, Lulka H, Hanoun N, Dufresne M, Meulle A, Vignolle-Vidoni A, Ligat L, Saint-Laurent N, Pont F, Dejean S, Gayral M, Martins F, Torrisani J, Barbey O, Gross F, Guimbaud R, Otal P, Lopez F, Tiraby G, Cordelier P. First-in-man phase 1 clinical trial of gene therapy for advanced pancreatic cancer: safety, biodistribution, and preliminary clinical findings. Mol Ther. 2015;23:779–789. doi: 10.1038/mt.2015.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zorde Khvalevsky E, Gabai R, Rachmut IH, Horwitz E, Brunschwig Z, Orbach A, Shemi A, Golan T, Domb AJ, Yavin E, Giladi H, Rivkin L, Simerzin A, Eliakim R, Khalaileh A, Hubert A, Lahav M, Kopelman Y, Goldin E, Dancour A, Hants Y, Arbel-Alon S, Abramovitch R, Galun E. Mutant KRAS is a druggable target for pancreatic cancer. Proc Natl Acad Sci U S A. 2013;110:20723–20728. doi: 10.1073/pnas.1314307110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Golan T, Khvalevsky EZ, Hubert A, Gabai RM, Hen N, Segal A, Domb A, Harari G, David EB, Raskin S, Goldes Y, Goldin E, Eliakim R, Lahav M, Kopleman Y, Dancour A, Shemi A, Galun E. RNAi therapy targeting KRAS in combination with chemotherapy for locally advanced pancreatic cancer patients. Oncotarget. 2015;6:24560–24570. doi: 10.18632/oncotarget.4183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lee JC, Shin DW, Park H, Kim J, Youn Y, Kim JH, Hwang JH. Tolerability and safety of EUS-injected adenovirus-mediated double-suicide gene therapy with chemotherapy in locally advanced pancreatic cancer: a phase 1 trial. Gastrointest Endosc. 2020;92:1044–1052.e1. doi: 10.1016/j.gie.2020.02.012. [DOI] [PubMed] [Google Scholar]
- 27.Hirooka Y, Itoh A, Kawashima H, Hara K, Nonogaki K, Kasugai T, Ohno E, Ishikawa T, Matsubara H, Ishigami M, Katano Y, Ohmiya N, Niwa Y, Yamamoto K, Kaneko T, Nieda M, Yokokawa K, Goto H. A combination therapy of gemcitabine with immunotherapy for patients with inoperable locally advanced pancreatic cancer. Pancreas. 2009;38:e69–e74. doi: 10.1097/MPA.0b013e318197a9e3. [DOI] [PubMed] [Google Scholar]
- 28.Hirooka Y, Kawashima H, Ohno E, Ishikawa T, Kamigaki T, Goto S, Takahara M, Goto H. Comprehensive immunotherapy combined with intratumoral injection of zoledronate-pulsed dendritic cells, intravenous adoptive activated T lymphocyte and gemcitabine in unresectable locally advanced pancreatic carcinoma: a phase I/II trial. Oncotarget. 2018;9:2838–2847. doi: 10.18632/oncotarget.22974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gelczer RK, Charboneau JW, Hussain S, Brown DL. Complications of percutaneous ethanol ablation. J Ultrasound Med. 1998;17:531–533. doi: 10.7863/jum.1998.17.8.531. [DOI] [PubMed] [Google Scholar]
- 30.Zhang WY, Li ZS, Jin ZD. Endoscopic ultrasound-guided ethanol ablation therapy for tumors. World J Gastroenterol. 2013;19:3397–3403. doi: 10.3748/wjg.v19.i22.3397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Facciorusso A, Di Maso M, Serviddio G, Larghi A, Costamagna G, Muscatiello N. Echoendoscopic ethanol ablation of tumor combined with celiac plexus neurolysis in patients with pancreatic adenocarcinoma. J Gastroenterol Hepatol. 2017;32:439–445. doi: 10.1111/jgh.13478. [DOI] [PubMed] [Google Scholar]
- 32.Paiella S, Salvia R, Ramera M, Girelli R, Frigerio I, Giardino A, Allegrini V, Bassi C. Local Ablative Strategies for Ductal Pancreatic Cancer (Radiofrequency Ablation, Irreversible Electroporation): A Review. Gastroenterol Res Pract. 2016;2016:4508376. doi: 10.1155/2016/4508376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Haen SP, Pereira PL, Salih HR, Rammensee HG, Gouttefangeas C. More than just tumor destruction: immunomodulation by thermal ablation of cancer. Clin Dev Immunol. 2011;2011:160250. doi: 10.1155/2011/160250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tatli S, Tapan U, Morrison PR, Silverman SG. Radiofrequency ablation: technique and clinical applications. Diagn Interv Radiol. 2012;18:508–516. doi: 10.4261/1305-3825.DIR.5168-11.1. [DOI] [PubMed] [Google Scholar]
- 35.Song TJ, Seo DW, Lakhtakia S, Reddy N, Oh DW, Park DH, Lee SS, Lee SK, Kim MH. Initial experience of EUS-guided radiofrequency ablation of unresectable pancreatic cancer. Gastrointest Endosc. 2016;83:440–443. doi: 10.1016/j.gie.2015.08.048. [DOI] [PubMed] [Google Scholar]
- 36.Crinò SF, D'Onofrio M, Bernardoni L, Frulloni L, Iannelli M, Malleo G, Paiella S, Larghi A, Gabbrielli A. EUS-guided Radiofrequency Ablation (EUS-RFA) of Solid Pancreatic Neoplasm Using an 18-gauge Needle Electrode: Feasibility, Safety, and Technical Success. J Gastrointestin Liver Dis. 2018;27:67–72. doi: 10.15403/jgld.2014.1121.271.eus. [DOI] [PubMed] [Google Scholar]
- 37.Scopelliti F, Pea A, Conigliaro R, Butturini G, Frigerio I, Regi P, Giardino A, Bertani H, Paini M, Pederzoli P, Girelli R. Technique, safety, and feasibility of EUS-guided radiofrequency ablation in unresectable pancreatic cancer. Surg Endosc. 2018;32:4022–4028. doi: 10.1007/s00464-018-6217-x. [DOI] [PubMed] [Google Scholar]
- 38.Paiella S, Malleo G, Cataldo I, Gasparini C, De Pastena M, De Marchi G, Marchegiani G, Rusev B, Scarpa A, Girelli R, Giardino A, Frigerio I, D'Onofrio M, Secchettin E, Bassi C, Salvia R. Radiofrequency ablation for locally advanced pancreatic cancer: SMAD4 analysis segregates a responsive subgroup of patients. Langenbecks Arch Surg. 2018;403:213–220. doi: 10.1007/s00423-017-1627-0. [DOI] [PubMed] [Google Scholar]
- 39.Bang JY, Sutton B, Hawes RH, Varadarajulu S. EUS-guided celiac ganglion radiofrequency ablation vs celiac plexus neurolysis for palliation of pain in pancreatic cancer: a randomized controlled trial (with videos) Gastrointest Endosc. 2019;89:58–66.e3. doi: 10.1016/j.gie.2018.08.005. [DOI] [PubMed] [Google Scholar]
- 40.Joosten JJ, van Muijen GN, Wobbes T, Ruers TJ. Cryosurgery of tumor tissue causes endotoxin tolerance through an inflammatory response. Anticancer Res. 2003;23:427–432. [PubMed] [Google Scholar]
- 41.Arcidiacono PG, Carrara S, Reni M, Petrone MC, Cappio S, Balzano G, Boemo C, Cereda S, Nicoletti R, Enderle MD, Neugebauer A, von Renteln D, Eickhoff A, Testoni PA. Feasibility and safety of EUS-guided cryothermal ablation in patients with locally advanced pancreatic cancer. Gastrointest Endosc. 2012;76:1142–1151. doi: 10.1016/j.gie.2012.08.006. [DOI] [PubMed] [Google Scholar]
- 42.Nelson JS, Liaw LH, Orenstein A, Roberts WG, Berns MW. Mechanism of tumor destruction following photodynamic therapy with hematoporphyrin derivative, chlorin, and phthalocyanine. J Natl Cancer Inst. 1988;80:1599–1605. doi: 10.1093/jnci/80.20.1599. [DOI] [PubMed] [Google Scholar]
- 43.Henderson BW, Dougherty TJ. How does photodynamic therapy work? Photochem Photobiol. 1992;55:145–157. doi: 10.1111/j.1751-1097.1992.tb04222.x. [DOI] [PubMed] [Google Scholar]
- 44.Choi JH, Oh D, Lee JH, Park JH, Kim KP, Lee SS, Lee YJ, Lim YS, Song TJ, Seo DW, Lee SK, Kim MH, Park DH. Initial human experience of endoscopic ultrasound-guided photodynamic therapy with a novel photosensitizer and a flexible laser-light catheter. Endoscopy. 2015;47:1035–1038. doi: 10.1055/s-0034-1392150. [DOI] [PubMed] [Google Scholar]
- 45.DeWitt JM, Sandrasegaran K, O'Neil B, House MG, Zyromski NJ, Sehdev A, Perkins SM, Flynn J, McCranor L, Shahda S. Phase 1 study of EUS-guided photodynamic therapy for locally advanced pancreatic cancer. Gastrointest Endosc. 2019;89:390–398. doi: 10.1016/j.gie.2018.09.007. [DOI] [PubMed] [Google Scholar]
- 46.Di Matteo FM, Saccomandi P, Martino M, Pandolfi M, Pizzicannella M, Balassone V, Schena E, Pacella CM, Silvestri S, Costamagna G. Feasibility of EUS-guided Nd:YAG laser ablation of unresectable pancreatic adenocarcinoma. Gastrointest Endosc. 2018;88:168–174.e1. doi: 10.1016/j.gie.2018.02.007. [DOI] [PubMed] [Google Scholar]
- 47.Yovino S, Poppe M, Jabbour S, David V, Garofalo M, Pandya N, Alexander R, Hanna N, Regine WF. Intensity-modulated radiation therapy significantly improves acute gastrointestinal toxicity in pancreatic and ampullary cancers. Int J Radiat Oncol Biol Phys. 2011;79:158–162. doi: 10.1016/j.ijrobp.2009.10.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pishvaian AC, Collins B, Gagnon G, Ahlawat S, Haddad NG. EUS-guided fiducial placement for CyberKnife radiotherapy of mediastinal and abdominal malignancies. Gastrointest Endosc. 2006;64:412–417. doi: 10.1016/j.gie.2006.01.048. [DOI] [PubMed] [Google Scholar]
- 49.Choi JH, Seo DW, Park DH, Lee SK, Kim MH. Fiducial placement for stereotactic body radiation therapy under only endoscopic ultrasonography guidance in pancreatic and hepatic malignancy: practical feasibility and safety. Gut Liver. 2014;8:88–93. doi: 10.5009/gnl.2014.8.1.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Varadarajulu S, Trevino JM, Shen S, Jacob R. The use of endoscopic ultrasound-guided gold markers in image-guided radiation therapy of pancreatic cancers: a case series. Endoscopy. 2010;42:423–425. doi: 10.1055/s-0029-1243989. [DOI] [PubMed] [Google Scholar]
- 51.Park WG, Yan BM, Schellenberg D, Kim J, Chang DT, Koong A, Patalano C, Van Dam J. EUS-guided gold fiducial insertion for image-guided radiation therapy of pancreatic cancer: 50 successful cases without fluoroscopy. Gastrointest Endosc. 2010;71:513–518. doi: 10.1016/j.gie.2009.10.030. [DOI] [PubMed] [Google Scholar]
- 52.Sanders MK, Moser AJ, Khalid A, Fasanella KE, Zeh HJ, Burton S, McGrath K. EUS-guided fiducial placement for stereotactic body radiotherapy in locally advanced and recurrent pancreatic cancer. Gastrointest Endosc. 2010;71:1178–1184. doi: 10.1016/j.gie.2009.12.020. [DOI] [PubMed] [Google Scholar]
- 53.Dávila Fajardo R, Lekkerkerker SJ, van der Horst A, Lens E, Bergman JJ, Fockens P, Bel A, van Hooft JE. EUS-guided fiducial markers placement with a 22-gauge needle for image-guided radiation therapy in pancreatic cancer. Gastrointest Endosc. 2014;79:851–855. doi: 10.1016/j.gie.2013.12.027. [DOI] [PubMed] [Google Scholar]
- 54.Khashab MA, Kim KJ, Tryggestad EJ, Wild AT, Roland T, Singh VK, Lennon AM, Shin EJ, Ziegler MA, Sharaiha RZ, Canto MI, Herman JM. Comparative analysis of traditional and coiled fiducials implanted during EUS for pancreatic cancer patients receiving stereotactic body radiation therapy. Gastrointest Endosc. 2012;76:962–971. doi: 10.1016/j.gie.2012.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sun S, Qingjie L, Qiyong G, Mengchun W, Bo Q, Hong X. EUS-guided interstitial brachytherapy of the pancreas: a feasibility study. Gastrointest Endosc. 2005;62:775–779. doi: 10.1016/j.gie.2005.06.050. [DOI] [PubMed] [Google Scholar]
- 56.Jin Z, Chang KJ. Endoscopic ultrasound-guided fiducial markers and brachytherapy. Gastrointest Endosc Clin N Am. 2012;22:325–331, x. doi: 10.1016/j.giec.2012.04.012. [DOI] [PubMed] [Google Scholar]
- 57.Sun S, Ge N, Wang S, Liu X, Wang G, Guo J. Pilot trial of endoscopic ultrasound-guided interstitial chemoradiation of UICC-T4 pancreatic cancer. Endosc Ultrasound. 2012;1:41–47. doi: 10.7178/eus.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Sun S, Xu H, Xin J, Liu J, Guo Q, Li S. Endoscopic ultrasound-guided interstitial brachytherapy of unresectable pancreatic cancer: results of a pilot trial. Endoscopy. 2006;38:399–403. doi: 10.1055/s-2006-925253. [DOI] [PubMed] [Google Scholar]
- 59.Jin Z, Du Y, Li Z, Jiang Y, Chen J, Liu Y. Endoscopic ultrasonography-guided interstitial implantation of iodine 125-seeds combined with chemotherapy in the treatment of unresectable pancreatic carcinoma: a prospective pilot study. Endoscopy. 2008;40:314–320. doi: 10.1055/s-2007-995476. [DOI] [PubMed] [Google Scholar]
- 60.Sun X, Lu Z, Wu Y, Min M, Bi Y, Shen W, Xu Y, Li Z, Jin Z, Liu Y. An endoscopic ultrasonography-guided interstitial brachytherapy based special treatment-planning system for unresectable pancreatic cancer. Oncotarget. 2017;8:79099–79110. doi: 10.18632/oncotarget.15763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Minaga K, Takenaka M, Kamata K, Yoshikawa T, Nakai A, Omoto S, Miyata T, Yamao K, Imai H, Sakamoto H, Kitano M, Kudo M. Alleviating Pancreatic Cancer-Associated Pain Using Endoscopic Ultrasound-Guided Neurolysis. Cancers (Basel) 2018;10 doi: 10.3390/cancers10020050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wiersema MJ, Wiersema LM. Endosonography-guided celiac plexus neurolysis. Gastrointest Endosc. 1996;44:656–662. doi: 10.1016/s0016-5107(96)70047-0. [DOI] [PubMed] [Google Scholar]
- 63.Oh SY, Irani S, Kozarek RA. What are the current and potential future roles for endoscopic ultrasound in the treatment of pancreatic cancer? World J Gastrointest Endosc. 2016;8:319–329. doi: 10.4253/wjge.v8.i7.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Levy MJ, Gleeson FC, Topazian MD, Fujii-Lau LL, Enders FT, Larson JJ, Mara K, Abu Dayyeh BK, Alberts SR, Hallemeier CL, Iyer PG, Kendrick ML, Mauck WD, Pearson RK, Petersen BT, Rajan E, Takahashi N, Vege SS, Wang KK, Chari ST. Combined Celiac Ganglia and Plexus Neurolysis Shortens Survival, Without Benefit, vs Plexus Neurolysis Alone. Clin Gastroenterol Hepatol. 2019;17:728–738.e9. doi: 10.1016/j.cgh.2018.08.040. [DOI] [PubMed] [Google Scholar]
- 65.Seicean A, Cainap C, Gulei I, Tantau M, Seicean R. Pain palliation by endoscopic ultrasound-guided celiac plexus neurolysis in patients with unresectable pancreatic cancer. J Gastrointestin Liver Dis. 2013;22:59–64. [PubMed] [Google Scholar]
- 66.Gimeno-García AZ, Elwassief A, Paquin SC, Sahai AV. Fatal complication after endoscopic ultrasound-guided celiac plexus neurolysis. Endoscopy. 2012;44 Suppl 2 UCTN:E267. doi: 10.1055/s-0032-1309709. [DOI] [PubMed] [Google Scholar]
- 67.Fujii L, Clain JE, Morris JM, Levy MJ. Anterior spinal cord infarction with permanent paralysis following endoscopic ultrasound celiac plexus neurolysis. Endoscopy. 2012;44 Suppl 2 UCTN:E265–E266. doi: 10.1055/s-0032-1309708. [DOI] [PubMed] [Google Scholar]
- 68.Loeve US, Mortensen MB. Lethal necrosis and perforation of the stomach and the aorta after multiple EUS-guided celiac plexus neurolysis procedures in a patient with chronic pancreatitis. Gastrointest Endosc. 2013;77:151–152. doi: 10.1016/j.gie.2012.03.005. [DOI] [PubMed] [Google Scholar]
- 69.Tempero MA, Malafa MP, Al-Hawary M, Behrman SW, Benson AB, Cardin DB, Chiorean EG, Chung V, Czito B, Del Chiaro M, Dillhoff M, Donahue TR, Dotan E, Ferrone CR, Fountzilas C, Hardacre J, Hawkins WG, Klute K, Ko AH, Kunstman JW, LoConte N, Lowy AM, Moravek C, Nakakura EK, Narang AK, Obando J, Polanco PM, Reddy S, Reyngold M, Scaife C, Shen J, Vollmer C, Wolff RA, Wolpin BM, Lynn B, George GV. Pancreatic Adenocarcinoma, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2021;19:439–457. doi: 10.6004/jnccn.2021.0017. [DOI] [PubMed] [Google Scholar]
- 70.Liu Y, Liu JL, Cai ZZ, Lu Z, Dong YH, Li ZS, Gong YF, Man XH. A novel approach for treatment of unresectable pancreatic cancer: design of radioactive stents and trial studies on normal pigs. Clin Cancer Res. 2007;13:3326–3332. doi: 10.1158/1078-0432.CCR-07-0154. [DOI] [PubMed] [Google Scholar]
- 71.Liu Y, Lu Z, Zou DW, Jin ZD, Liu F, Li SD, Zhan XB, Zhang WJ, Wu RP, Yao YZ, Yang L, Li Z. Intraluminal implantation of radioactive stents for treatment of primary carcinomas of the peripancreatic-head region: a pilot study. Gastrointest Endosc. 2009;69:1067–1073. doi: 10.1016/j.gie.2008.08.033. [DOI] [PubMed] [Google Scholar]
- 72.Li W, Wang X, Wang Z, Zhang T, Cai F, Tang P, Meng J, Du H, Wang H, Li M, Li S. The role of seed implantation in patients with unresectable pancreatic carcinoma after relief of obstructive jaundice using ERCP. Brachytherapy. 2020;19:97–103. doi: 10.1016/j.brachy.2019.08.010. [DOI] [PubMed] [Google Scholar]