Abstract
Simple Summary
DNA methylation is an epigenetic modification of genes which affects corresponding gene expression. During the developmental stage, embryonic stem cells undergo various epigenetic modifications to produce different specialized cells. DNA methylation appears as one of the important epigenetic modifications which not only potentiate neuronal development but also have been sought in various neurodegenerative diseases, such as Alzheimer’s disease. The present work focuses on the history of DNA methylation, its role in neurodevelopment functions, and how assessment of DNA hypermethylation and hypomethylation can be utilized for the prognosis of AD and other neurodegenerative diseases. This review also paves the way for the development of novel treatment strategies based on targeting DNA methylation in neurodegenerative diseases.
Abstract
DNA methylation, in the mammalian genome, is an epigenetic modification that involves the transfer of a methyl group on the C5 position of cytosine to derive 5-methylcytosine. The role of DNA methylation in the development of the nervous system and the progression of neurodegenerative diseases such as Alzheimer’s disease has been an interesting research area. Furthermore, mutations altering DNA methylation affect neurodevelopmental functions and may cause the progression of several neurodegenerative diseases. Epigenetic modifications in neurodegenerative diseases are widely studied in different populations to uncover the plausible mechanisms contributing to the development and progression of the disease and detect novel biomarkers for early prognosis and future pharmacotherapeutic targets. In this manuscript, we summarize the association of DNA methylation with the pathogenesis of the most common neurodegenerative diseases, such as, Alzheimer’s disease, Parkinson’s disease, Huntington diseases, and amyotrophic lateral sclerosis, and discuss the potential of DNA methylation as a potential biomarker and therapeutic tool for neurogenerative diseases.
Keywords: epigenetic regulation, DNA methylation, genetic markers, histone modification, Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, amyloid lateral sclerosis
1. Introduction
Epigenetic modification refers to heritable changes in gene expression that are not encoded by the DNA sequence [1]. DNA methylation is an inherent epigenetic process in which DNA methyltransferases move a CH3 group covalently to the C-5 of the cytosine ring of DNA [2]. DNA methylation occurs at the cytosine in mammalian genetic material. In embryonic cells, around 25 percent of methylation has been identified in a non-CpG context.
Even though the brain contains a higher level of DNA methylation than any other tissue, a low level of 5mC in human genomes is recorded. In general, the methylation of DNA is mostly found at the CpG dinucleotide; however, in somatic cells, the methylation of DNA is highly prevalent on the non-CpG dinucleotide framework. Non-CpG methylation is highly enriched in brain tissue and embryonic stem cells, due to methylation taking place in a non-CpG context [3]. During zygote development, DNA methylation is normally detached and then again established in the embryo approximately during the time of installation [4]. Although several researchers hypothesized that DNA methylation could affect gene function, it was not until the 1980s that researchers showed that methylation in DNA has an impact on gene function and mitosis. This epigenetic modification is now generally accepted as a significant epigenetic factor affecting gene activities [5,6].
DNA methyltransferases (DNMTs) catalyze DNA methylation by transferring a methyl group from S-adenyl methionine (SAM) to the fifth carbon of a cytosine residue, resulting in 5mC. Whenever replication of DNA happens, DNA methyltransferase-1 retrieves the DNA methylation prototype from the parent strands of DNA and transfers it to the newly synthesized daughter strand [7,8]. Although postmitotic neurons in full-grown mammalian brains still demonstrate high levels of DNA methyltransferases, this finding indicates that DNA methyltransferases and epigenetic modification such as methylation in DNA may participate in new functions in the brain [9].
This is more common in species with heavy DNA methylation patterns. However, there can also be no repeat methylation, as investigated in the case of the invertebrate chordate Ciona intestinalis [10]. Despite its long evolutionary history, gene methylation is still inadequately understood. Epigenetic regulation also helps to mitigate chromatin disturbance induced by elongating RNA polymerase such as nucleosome displacement [11]. Cytosine-guanine dinucleotides in mammals are methylated on cytosine residues, but cytosine-guanine dinucleotides in promoters are largely unmethylated. Defects in DNA methylation become the cause of many diseases. An increased or decreased level of methylation can result in gene instability [7]. A heritable mechanism for controlling gene expression is a covalent alteration in the genome and proteins present in histone, which is mainly present in chromatin. Histone ends are exposed to an amount of covalent alteration such as acetylation, methylation, phosphorylation, ubiquitination, and sumoylation, all of which control key cellular processes including gene transcription, replication, as well as repair [12]. The addition of three methyl groups in histone has been planned as a condition following DNA methylation, which can be due to interactions between the parts of these histone methylation systems [7]. Histone lysine methyltransferases, suppressor of variegation 3-9 homolog-1 and enhancer of Zeste 2 polycomb repressive complex 2 subunits all perform together and promote their interaction with target promoters [13,14].
CpG islands are stretch of DNA with 500–1500 base pairs with elevated 5′-C-phosphate-G-3′ density compared to other parts of the gene but are mostly unmethylated. CpG islands are home to many gene promoters [15]. CpG islands seem to have evolved to facilitate transcription by controlling the chromatin structure and transcription factor binding. Nucleosomes are small, packaged sections of DNA that are wrapped around histone proteins regularly. DNA becomes less permissive for gene expression as it becomes more closely aligned with histone proteins. CpG islands have fewer nucleosomes than other DNA stretches, which is one of their most distinguishing characteristics [16,17]. Even though 50% of CpG islands have established transcription start sites, they are frequently vacant of common promoter essentials such as TATA boxes [18].
2. History and Development of DNA Methylation
DNA methylation, mainly in the cytosine and adenine positions, is responsible for the chromatin structure and dynamics. Out of all epigenetic modifications, methylation, thiouridylation, and pseudo-uridylation of bases in rRNAs and tRNAs are essential for survival. The importance of these modifications is in maintaining double-helical pairing in DNA and preventing mutagenic effects of base modification. They also allow RNA to recall biochemical diversity which is required for their role by protecting genetic material in an unmodified state. Only a small set of DNA alterations have entered evolution and been used to determine biological functions [19].
Mammalian DNA methylation was found almost as soon as DNA was identified as a source of genetic material [20]. Gerlach et al., 1965, reported that modified cytosine was identified in the calf thymus [21]. He proposed that such a proportion was 5-methylcytosine (5mC), since it segregated out cytosine in the same way as thymine segregated from uracil. He also believed that this modified cytosine occurred spontaneously in DNA. Even though many researchers hypothesized that methylation of DNA may affect the expression of genes, it was not until the 1980s that multiple studies showed that methylation of DNA was involved in the regulation of gene and cell division. DNA methylation is now widely recognized as an important epigenetic process influencing gene activity when combined with other promoters. [22].
A number of in silico methods have been developed to determine DNA modification according to the number of enzymes. X-ray crystallography and biochemical studies were used to identify such changes [23]. 5hmC and 5-hydroxymethyluracil synthases along with other enzymes, i.e., DNA base glycosyltransferases, alpha-glucosyltransferase, beta-glucosyltransferase, protein hydroxylases, and histone demethylases, modify bases in DNA and are responsible for moiety transfer from one DNA to another. All DNA methylases belong to monophyletic assemblage, and mainly contain bases in the nucleic acid or amino acid side chain. An examination of the origins of DNA N6A methylases (and related N4C methylases) and their predecessors overlooked their existence in eukaryotes [19].
3. Methylation Detection Method
Sodium bisulfite converting and sequencing, the cleavage of DNA by different enzymes, and methylated DNA capturing affinity are the methods used to identify DNA methylation. Methylated DNA immunoprecipitation (Me-DIP), which utilizes a DNA methyl-specific antibody, and methyl encapsulate, which uses methyl-CpG binding domain (MBD) proteins, are the two most widely reported DNA affinity capture methods [24].
The high-performance liquid chromatography-ultraviolet (HPLC-UV) technique, which helps in determining the concentration of deoxycytidine (dC) and methylated cytosines (5mC) contained in a hydrolyzed sample of DNA, is still used today. However, the method’s utility is restricted by the necessity for specialist laboratory apparatus and the need for relatively large amounts of DNA material (3–10 g) to be analyzed [25].
Liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS) is another method used to determine methylation status in DNA. In LC-MS/MS, a small number of samples are required. LC-MS/MS has been verified for identifying methylation levels ranging from 0.05% to 10% in mammalian DNA, and it can confidently identify variations among samples. It can even identify samples which have about 0.25 percent of the total cytosine residues, which equates to 5% differences in global DNA methylation [26].
Because ELISA-based assays have a high risk of considerable changes, they are only useful for estimating DNA methylation in a crude manner. Nonetheless, they are quick and simple to use approaches that work well for detecting substantial changes in global DNA methylation [25]. LINE-1 methylation levels can also be determined using a method that involves bisulfite conversion of DNA followed by PCR amplification of LINE-1 conservative regions. Pyrosequencing is used to determine the methylation status of the amplified fragments, which can resolve discrepancies among samples of DNA as small as 5 percent. Even though this method only focusses on LINE-1 components and hence only on lean CpG sites, it has been proven to accurately reflect overall DNA methylation modification. The method is particularly well-adapted to the examination of high-throughput cancer samples because hypomethylation is frequently linked to a poor prognosis. This approach works best with human DNA, although there are also variations that work with rat and mouse genomes [27].
Differently methylated DNA can also be detected by amplification fragment length polymorphism (AFLP) and restriction fragment length polymorphism (RFLP). These methods have now been replaced by other powerful and more accurate methods. AFLP and RFLP are inexpensive and quickly access the methylation alteration in DNA samples [28].
The luminometric methylation assay is a technique which combines two DNA restriction digest operations that are run in parallel, followed by pyrosequencing to fill in the gaps between the digested DNA strands’ projecting ends. The CpG-methylation-sensitive enzyme HpaII is used in one digesting step, whereas the methylation-insensitive enzyme MspI is used in the other, cutting at all CCGG sites [29]. Bisulfite sequencing is the “gold standard” technology in DNA methylation research. Recent DNA sequencing technology cannot tell the difference between methyl-cytosine and cytosine. The deamination of cytosine into uracil is mediated by bisulfite treatment of DNA, and these transformed residues are read as thymine by PCR amplification and subsequent Sanger sequencing analysis. Five methylated cytosine residues, on the other hand, are independent of this change and remain as cytosine. By comparing the “Sanger sequencing” reads from DNA samples that remain untreated to the cloned sample after bisulfite treatment, the 5mC can be detected. This approach may now be expanded to DNA methylation analysis over a complete genome because of the advent of next-generation sequencing (NGS) technology [30]. Apart from this array or bead hybridization [31], methylation-specific PCR [32], bead array [33], and pyrosequencing can be used to determine the methylation status of the gene of interest [25].
4. DNA Methylation in Premature and Mature Brain
The precise timing of de novo methylation and de-methylation in the developing brain is crucial. Multipotent neural progenitor cells (NPCs) go through neurogenesis and astrogliogenesis [34]. The addition and removal of a methyl group in the promoter gene follow the change in neural progenitor cells from neurogenesis to astrogliogenesis and neuron proliferation and development in the adult brain. [35]. The DNMT family of enzymes includes DNMT1 and DNMT3A/DNMT3B enzymes. DNMT1 preferentially leads to methylation of hemi-methylated DNA and further maintains DNA methylation after the replication of DNA, whilst DNMT3A and DNMT3B cause methylation on non-methylated and hemi-methylated DNA equally and are considered as de novo methyltransferase. DNMTs go through significant conformational changes, are capable of oligomerization, and can self-inhibit, all of which could help to regulate their activity [36].
A double knockout study (lacking both DNMT1 and DNMT3A) revealed their importance in the regulation of synaptic plasticity and memory formation through maintaining DNA methylation in neurons [37]. Furthermore, aberrant bdnf gene expression (possibly through epigenetic modifications) has been observed in several neurological disorders; however, the inhibition of DNMT resulted in blocking, which was reported to alter bdnf DNA methylation status in the hippocampus [38,39] and modulate learning and memory [40,41].
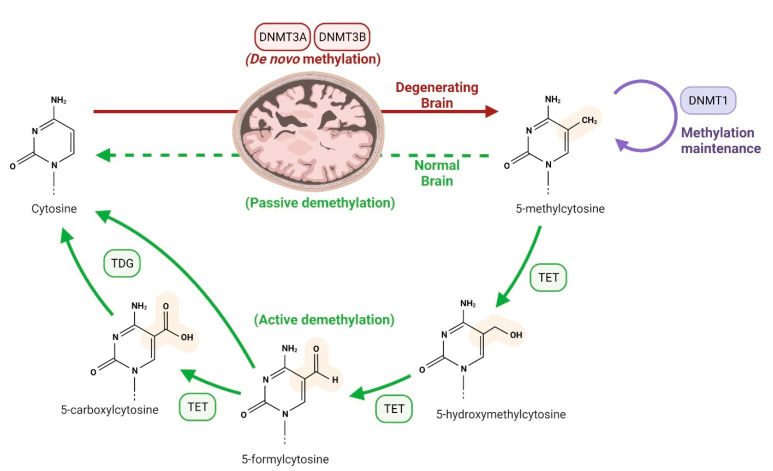
DNMT1 in neural progenitor cells are critical for maintaining the methylated sequence on the glial fibrillary acidic protein promoter throughout mitosis. During the early embryonic stage, Gfap gene is methylated; however, at a later stage, it gets demethylated [42]. Methyl CpG binding protein 2 is present in the brainstem and thalamus, which are the brain’s oldest areas. Methyl CpG binding protein 2 interacts with several protein synthesis inhibitors, including DNMTs, and regulates gene expression [43,44,45]. Methyl CpG binding protein 2 is a protein that plays a role in neuronal development, and its loss of phosphorylation causes an abnormal dendritic arborescent figure, a synaptic role, and smoothness [46,47]. The schematic representation of DNA methylation and demethylation is illustrated in Figure 1.
Figure 1.
A systemic representation of DNA methylation and demethylation. By means of the action of DNA methyltransferases, the methyl group is transferred to the 5th carbon in cytosine. Generally, DNA methylation is initiated by DNMT3A/3B (de novo pathway) and is maintained by DNMT1. Most gene expression is suppressed by DNA methylation, which contributes to the development of neurodegeneration. On the other hand, methylated cytosine turned into cytosine through an active/passive demethylation process. This cycle regulates the gene expression, while under certain environmental conditions, abnormality in this cycle may contribute to the development of neurodegenerative diseases.
Methyl-binding proteins are another type of protein that works with DNA methylation to control transcription in the CNS [48]. Neuronal activity causes phosphorylation of MeCP2, which leads to changes in gene expression. Synapse development, synaptic flexibility, and learning and recall activity are all impaired when MeCP2 phosphorylation is inhibited. Since phosphorylation is typically a transient alteration, activity-dependent phosphorylation can, temporarily, release methyl CpG binding protein-2 from the enhancer, allowing demethylation of the genome pattern. Methyl group addition or deletion may be to blame for long-term changes in genome number, which is responsible for controlling synapse flexibility, learning, and memory [49,50,51]. The brain is mostly made up of post-mitotic neurons and glial cells that have small proliferative capacities. DNMT1 and DNMT3A are both asserted by mature neurons. In both development and illness, DNMT3B is required for the dynamic programming of epigenetic control. The amino acid sequences of DNMT3A and DNMT3B are very similar. Immunodeficiency, centromeric instability, and facial abnormalities syndrome are caused by DNMT3B mutations [52]. This unexpected finding prompted researchers to investigate further into the function of active gene methylation in adult brain post-mitotic neurons.
5. Role of DNA Methylation in Neurological Disorders
Alteration in the methylation pattern of certain genes can modulate neuronal survival and regeneration, which in turn leads to the progression of neuronal degeneration. A summary of recent evidence supporting the hyper/hypomethylation of genes in various neurodegenerative diseases is elaborated in Table 1 and illustrated in Figure 2.
Table 1.
DNA methylation status of different genes in neurodegenerative diseases.
| Disease | Sample | Methylation (Hyper/Hypo) | Experimental Method | Gene | Ref. |
|---|---|---|---|---|---|
| AD | Blood | Hypermethylation | Bisulphite sequencing PCR and methylation-specific PCR are used | SIRT1 | [53] |
| AD | Dorsolateral prefrontal cortex tissue | Differently methylated | CpG sites generated using a bead assay | SORL1, ABCA7, HLA-DRB5, SLC24A4, BIN1. | [54] |
| AD | Hippocampus | Hypomethylation | Bisulfite cloning sequencing of CpG sites in two promoter regions Prom1 and Prom2 | CREB-regulated transcription factor 1 | [55] |
| AD | Blood | Hypermethylation | Bisulfite treated DNA was analyzed by melting curve analysis-methylation assay | UQCRC1 | [56] |
| AD | Hippocampus | Hypermethylation | Bisulfite cloning sequencing and further measured by 5-hydroxymethycytosine (5hmC) | TREM2 | [57] |
| AD | Blood | Hypermethylation | Dual-luciferase assays | OPRM1, OPRL1 | [58] |
| AD | Blood | Hypomethylation | Quantitative bisulfite-PCR pyrosequencing | PICALM | [59] |
| AD | Brain | Hypermethylation | Bisulfite pro-sequencing | ANK1 gene | [60] |
| AD | Hippocampus | Hypermethylation | RT-qPCR | PLD3 gene | [61] |
| PD | Postmortem human brain samples (frontal cortex) | Hypermethylation | Illumina Infinium array | MRI1, TMEM9 | [62] |
| PD | Postmortem human brain samples (frontal cortex) | Hypomethylation | Illumina Infinium array | GSST1, TUBA3E, KCNH1 | [62] |
| PD | Brain tissue | Hypomethylation | Fluorescence-activated nuclei sorting and bisulfite pro-sequencing | CpGs located in SNCA intron 1 | [63] |
| PD | Blood | Differently methylated | Cross-sectional analysis of blood methylation | SRSF7, ADNP, GDNF, SYN3, CPLX1, SNCA, TREM2. | [64] |
| PD | Blood and saliva | Altered methylation | Illumina Infinium array | ABCB9, C1orf200, AZU1, LARS2, PARK2, LRRK2, APC, AXIN1 | [65] |
| PD | Brain | Differently methylated | Genome wide screening and RNA sequencing | ARFGAP1, DUSP22 promoter, SNCA | [66] |
| PD | Leukocytes | Hypomethylation | Methylation-specific PCR | NPAS2 | [67] |
| PD | Brain | Hypermethylation | Bisulfite sequencing and micro array gene expression analysis | PGC1-α | [68] |
| PD | Brain | Hypomethylation | Genome wide methylation | CYP2E1 | [69] |
| PD | Blood | Hypomethylation | - | NOS2 | [70] |
| PD | Leukocytes, Brain | Hypermethylation | Bisulfite pyrosequencing and MAPT promoter methylation assay | MAPT | [71] |
| PD | Brain | Hypermethylation | Illumina Infinium array | FANCC/TNKS2 | [72] |
| HD | Striatal cells carrying polyglutamine-expanded HTT (STHdhQ111/Q111) and wild-type cells (STHdhQ7/Q7) | Altered DNA methylation | mRNA-Seq, ChIP-Seq assay and Motif Scanning | Htt | [73] |
| HD | Prefrontal cortex | Differently methylated | Fluorescence-based nuclei sorting (FACS)-ChIP-seq | HES4 | [74] |
| HD | Putamen of HD patients and striatum of mice | Differently methylated | Bisulfite sequencing and TaqMan PCR | ADORA2A | [75] |
| HD | Blood | Differently methylated | Microarray methylation | CLDN16, NXT2, DDC. | [76] |
| HD | Blood | Differently methylated | mRNA-Seq, ChIP-Seq assay and motif scanning | FBXL5, S100P, PRDX1, COPS7B, SP1, SEC24C, PDIA6, USP5, GRAP, POP5, WRB, PCSK7. | [77] |
| ALS | Postmortem spinal cord tissue | Hypomethylation | Bisulfite pyrosequencing, genome-wide expression profiling, and RT-PCR | MLC1, CRB1, CTNND2, FURIN, SLC31A1, CMTM3, STAT5A, SRGAP1, LPXN, PLD4, OBFC2A, TXNIP, PSAP, SLC35E1, RBM38, CLEC4A, HMHA1, PLSCR1, AXL, PHYHD1. | [78] |
| ALS | Postmortem spinal cord tissue | Hypermethylation | Bisulfite pyrosequencing, genome wide expression profiling, and RT-PCR | LUM, SLC13A4, GJB2, TYRP1, CLDN19, LINGO2, PLEKHA4, NNAT, TSPAN18, PLCB4, TMEM139, PNMAL1, DMBT1, TNFSF10, NNAT, PCP4, MAB21L2, PEG10, TMEM139, KCNJ12, FGF18. | [78] |
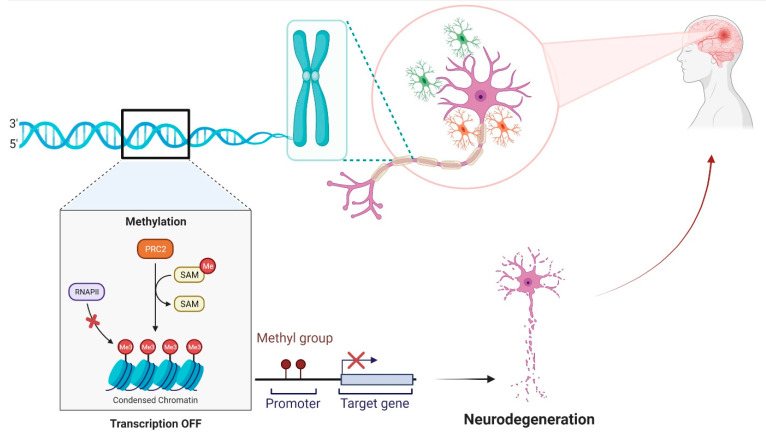
Figure 2.
Schematic representation of DNA methylation causing the inhibition of target gene expression and progression towards neurodegeneration.
5.1. Alzheimer’s Disease
Dementia is a progressive age-related neurodegenerative disease described by progressive cognitive impairment that affects 35.6 million people in the world and is becoming a more pressing issue as the population ages [79]. Extracellular amyloid plaques and intracellular neurofibrillary tangles are hallmarks of Alzheimer’s disease (AD). β-secretase and γ-secretase cleave amyloid precursor protein (APP) sequentially, generating amyloidogenic Aβ peptides that accumulate in the outer space of the cell to form insoluble Aβ plaques.
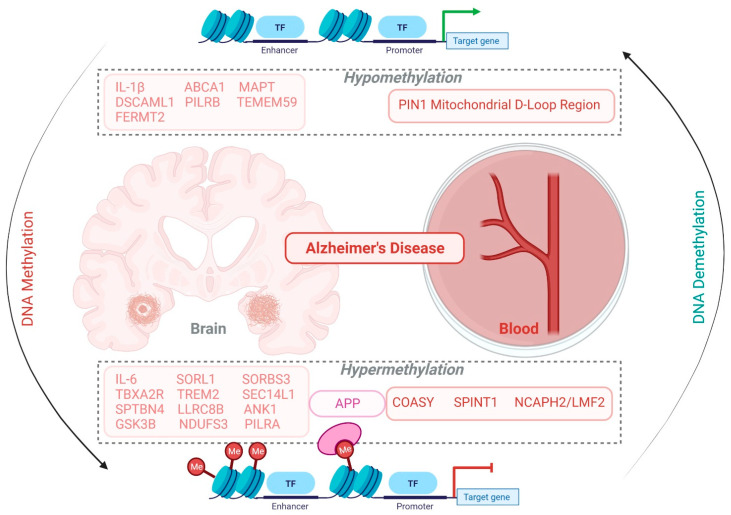
Decreased global DNMT1 and 5mC in the temporal cortex and reduced global 5mC and 5hmC in the hippocampus of AD patients have been reported [80]. In contrast, some studies support the elevated level of 5mC and 5hmC in the frontal lobe, temporal cortex, and hippocampus in AD [81,82]. Furthermore, reduced 5mC levels in APP, PSEN1, and SERT1 promotors in the brain and blood of AD patients have been recorded [83,84]. Meanwhile, a recent study supported a positive correlation between 5mC and 5hmC values in AD patients [85]. This could be helpful in the differential diagnosis of AD patients from PD (with lower 5hmC level and unchanged DNMT3A expression). The status of methylation and demethylation of various genes in the brains and blood of Alzheimer’s patients is illustrated in Figure 3.
Figure 3.
Differential methylation pattern of various genes in brain and peripheral blood in Alzheimer’s disease. This image illustrates the alteration in the expression of different genes in the AD brain and peripheral blood circulation due to the hyper/hypomethylation of DNA. The genes encoded in light red are specific to the brain region, while the genes encoded in red color are restricted to peripheral blood circulation. In addition, hypermethylation of the APP gene is reported as a common gene in the AD brain and peripheral blood circulation.
Important variations in DNA methylation profiles of the APP, microtubule-associated protein tau, and GSK3B were also discovered, along with PSEN1, beta-secretase 1 precursor, or apolipoprotein E. Many studies have reported genetic variants linked to enhanced AD susceptibility, but do not limit many other promoters. Almost 28 gene locations are linked with AD, and while little is known about bridging integrator-1 function in AD pathogenesis, it may have a considerable impact on tau pathology, amyloid precursor protein endocytosis, and inflammation in neurons [86,87,88,89].
Most of the AD studies performed to date have mostly considered a gene-directed analysis; thus, the methylation of promoter genes of AD (especially APP) has been widely explored. As mutant APP genes pose a risk factor for AD, it is apparent that epigenetic modifications of the APP promoter leading to enhanced gene expression are also risk factors for AD. West and colleagues reported hypomethylation of the APP promoter in AD patients [90]. Meanwhile, some contrasting studies rejected these findings, suggesting this as an outcome of larger epigenetic modification rather than a specific alteration in methylation [83].
Furthermore, the expression of cortical PSEN1 appeared stable during embryonic development, while upregulated levels have been reported in AD patients. The cortical PSEN1 expression in rodents appears to be tightly regulated by histone modification at various stages of neuronal development. Generally, the PSEN1 gene remains partly methylated and suppressed after development, while the hypomethylation of PSEN1 is reportedly associated with its elevated expression in the AD population [91].
In DNA, when hydrogen is replaced with a hydroxyl group, it is also related to regulating gene expression, although its mechanism is still unclear. In comparison to the moderately constant tissue distribution of 5mC, 5hmC has a wide range of tissue distribution. This is common in the brain, with the cerebral cortex having the largest proportion of 5hmC compared to other parts of the brain [92]. The thromboxane A2 receptor and Sorbin SH3 domain containing 3 (Sorbs3) genes were found to be highly methylated in Alzheimer’s disease. The thromboxane A2 receptor regulates protein synthesis factors such as cyclic adenosine monophosphate response-element binding protein (CREB), which are concerned with neuronal plasticity, protection, and long-term memory development. In the temporal cortex of Alzheimer’s patients, Sorbin SH3 domain containing 3 was found to be highly methylated [93].
This reduction in the methylation level of CREB-regulated transcription factor 1 was found in the hippocampus of AD patients when compared with controls, and this methylation within CREB-regulated transcription factor 1 was inversely connected with phosphorylation in tau expression [94]. Inflammation of neurons is linked to AD. Evidence also revealed that interleukin-1 and interleukin-6 expression levels increase in Alzheimer’s disease, but in the late stages of Alzheimer’s, their levels return to normal [95].
It is also understood that the peripheral blood is also an indicator for AD. Brain-derived neurotrophic factor is responsible for controlling neuronal endurance, division, and flexibility [96,97]. Brain-derived neurotrophic factor is also a key factor in AD. Its level is higher in the peripheral blood of AD patients than in normal controls. Importantly, the percent methylation of specific CpG sites within the brain-derived neurotrophic factor promoter suggests that methylation of the brain-derived neurotrophic factor promoter is associated with clinical manifestations of AD [98,99].
One study also suggested a role for gene expression in AD. H3K4me3 and H3K27ac are responsible for gene alteration. Increased expression of genes causes alterations in immune responses, while decreased expression of genes causes impairment in the learning process. The histone marker H4K16ac causes an alteration in the chromosome which connects it with DNA damage and aging in neurodegenerative diseases. The amount of H4K16a in the aged brain decreases due to the aged brain’s inability to regulate it, which results in changes in brain function [100,101]. An in vitro study on AD found that methylation levels in the 2′,5′-oligoadenylate (2-5A) synthetase gene were reduced [102].
Changes in the methylation levels of SORL1, ABCA7, HLA-DRB5, SLC24A4, and BIN1 also contribute to AD [103]. Hypermethylation of the UQCRC1 gene is responsible for inflammation and oxidative stress in AD [104]. An increase in DNA methylation in TREM2 was also reported, which causes alteration in AD biomarker TREM2 mRNA expression [105]. Hyper methylation of OPRM1 and OPRL1 genes in AD patients has also been reported in many studies that demonstrate the role of opioid receptors in the diagnosis of AD [106]. A decrease in the methylation level of the PICALM gene leads to an alteration in the cognitive behavior of AD patients [107]. The ANK1 gene’s elevated methylation profile demonstrated its role in AD [108]. OTX gene methylation changes also lead to AD [101].
Apart from this, DNA hydroxymethylation is also one of the epigenetic mechanisms that lead to the progression of the disease. As discussed above, hypermethylation of the ANK1 gene leads to AD. Hypo-hydroxymethylation of the ANK1 gene is also one of the factors related to AD. Hypermethylation of the PLD3 gene also plays a role in AD [109]. In the 5xFAD mouse model, as well as in the 3xTg-AD mouse model, changes in DNA methylation and hydroxymethylation patterns in genes were observed. These models proved that a reduction in hydroxymethylation also plays an important role in AD [110]. Hydroxy methylation of genes is also related to cognitive behavior and memory impairment. Hydroxy methylation of the DNA sequence gives rise to 5-hydroxymethycytosine, which is present in large amounts in the brain and plays a role in neurodevelopment. DNA hydroxymethylation in the TREM2 gene was found to be increased in AD patients. Different hydroxymethylated regions can be the reason for neuritic plaques and neurofibrillary tangles. Alterations in DNA patterns in the prefrontal cortex cause neurogenesis [111,112]. Levels of 5-hydroxymethylcytosine were also found to be elevated in mitochondria, which also suggested its role in AD [113].
5.2. Parkinson Disease
In the elderly population, Parkinson’s disease (PD) is the most widespread disease caused by the degeneration of neurons, mainly dopaminergic neurons. In post-mortem brains, Lewy bodies, irregular protein aggregates contained inside nerve cells, and gradual depletion of dopamine neurons present in the substantia nigra have been discovered [114]. In the last few years, complete hereditary screening of Parkinson’s disease families has sought to detect mutations linked to the disease, which will provide a better understanding of the actual mechanisms involved in the disease. Several gene locations that are associated with familial Parkinson’s disease, such as Parkinson’s disease (PARK 1-15) and other genes, have been identified through genetic studies. Other genes linked to sporadic PD have been identified, including leucine-rich repeat kinase-2, alpha-synuclein, microtubule-associated protein tau, and encoding for the lysosomal enzyme glucocerebrosidase [115,116].
Alpha-synuclein aggregation leads to Lewy body expansion, a characteristic of PD [117,118]. DNA methylation has been projected as a possible means for the deregulation of alpha-synuclein in PD [119]. The use of a DNA methylation inhibitor reduced CpG-2 methylation while significantly raising alpha-synuclein mRNA and protein levels. The addition of a methyl group, at least at the intrinsic CpG-2 island, has been shown in recent studies to regulate alpha-synuclein gene action [120].
One study also claimed that alterations in methyl group addition levels in alpha-synuclein take place in many parts of the brain. Both the addition and removal of methyl groups were found in the promoter gene region as well as in intron 1 of different Lewy body disease/PD levels [121]. Other studies would be required to overcome these doubts about the validity of DNA methyl group removal at the alpha-synuclein intron 1 concerning PD, given the inconsistencies. So, this DNA methylation modification may act as a biomarker for Lewy body-related diseases rather than a particular biomarker for PD [122,123].
Apart from SNCA, beta-synuclein (SNCB) also plays a major role in PD. In vitro, SNCB prevents the formation of alpha-synuclein fibril aggregation, suggesting that it may help in protecting neurons that are prone to degeneration [124]. The promoter of the beta-synuclein gene was originally found to be unmethylated in the brain. Bisulfite sequencing of the beta-synuclein promoter revealed no 5mC along the cytosine phosphate guanine island in four uncontaminated disperse Lewy body pathology cases [125].
Peptidyl arginine aminases (PADs) also play a role in PD, and their promoter is found to have a reduced methylation level in the brain, but it is the opposite in white matter. Other than alpha-synuclein, some genes such as ubiquitin carboxyl-terminal hydrolase isozyme L1 (UCHL1) promoter, ATP13A2 promoter, Parkin (PARK2) gene, and other clock genes such as period circadian regulator (PER1), period circadian regulator-2, cryptochrome circadian regulator-1, cryptochrome circadian Regulator-2, neuronal PAS domain protein 2 (NPAS2), and brain and muscle ARNT-like 1 (BMAL1) have also been measured in genomic DNA isolated from PD patients to check their role in PD. Mutations in the ATP13A2 promoter and PARK2 are related to PD. However, in the case of the UCHL1 promoter, ATP13A2 promoter, and PARK2, there were no variations in 5′-C-phosphate-G-3′ methylation percentages between PD cases and control groups [126,127,128,129,130]. DNA methylation was found in the cryptochrome circadian regulator 1 and neuronal PAS domain protein 2 promoters, but not in other gene promoters studied [131].
The microtubule-associated protein tau (MAPT) gene is associated genetically with PD. In MAPT, the H1 haplotype had more DNA methylation than the H2 haplotype (this haplotype was linked appreciably with PD). The MAPT gene was found to have increased methyl group levels in the cerebellum, but not in the putamen of PD patients [132,133,134].
In the CpG-1 and CpG-2 islands of PARK7, there is no methylation in both the PD and the control groups, where the PGC-1 promoter is highly methylated [135,136]. When investigated, single CpG sites of both Fanconi anemia complementation group C (FANCC) and tankyrase 2 (TNKS2) showed differences in methylation patterns in the PD and control groups. Another study revealed that about 20 genes were found to have DNA methylation differences in PD patients [137].
A comparative analysis of DNA methylation studies in the brain and blood of PD patients suggested alteration in their DNA expression. KCTD5, VAV2, MOG, TRIM10, HLA-DQA1, ARHGEF10, GFPT2, HLA-DRB5, TMEM9, MRI1, MAPT, HLA-DRB6, LASS3, GSTTP2, GSTTP were found to be hypermethylated, and DNAJA3, JAKMIP3, FRK, LRRC27, DMBX1, LGALS7, FOXK1, APBA1, MAGI2, SLC25A24, GSTT1, MYOM2, ME 886, TUBA3E, TMCO3 genes were hypomethylated in the brains and blood of PD patients [138].
Recent evidence elaborating DNA methylation of different genes in AD and PD is presented mentioned in Table 2.
Table 2.
Recent evidence supporting the role of DNA methylation in Alzheimer’s and Parkinson’s disease.
| Methylation of DNA | Gene/Target/Pathway Involved | Effect | Model | Experimental Method | Outcomes | Ref. |
|---|---|---|---|---|---|---|
| 5-mC | B3GALT4, ZADH2 | Decrease | AD and healthy patients | Rey Auditory Verbal Learning Test (RAVLT), Trail Making Test Part B (TMT-B), INNOTEST assays, and Triplex assay | Hypomethylation of B3GALT4, ZADH2 associated with the level of AB and tau in CSF | [139] |
| 5-mC | HOXA3, GSTP1, CXXC1-3, BIN1 | Increase | AD and healthy patients | Laser-assisted microdissection and Infinium DNA Methylation 450K analysis | 504 DMCs and 237 DMRs were identified and increased in the 5mC pyramidal layer, which is associated with oxidative stress | [140] |
| 5-mC | KIAA056 | Decrease | NFT pathology stages I-IV | Bisulfite sequencing and Infinium Human Methylation 450 BeadChip | Downregulation of 5mC in KIAA056 and in NFT pathology cases | [141] |
| 5-mC | ANKRD30B, ANK1, Cell adhesion | Increase | AD and neurotypical patients | Genome-wide DNA methylation, mRNA expression profiling, functional enrichment analysis, and differential methylation of genes | 856 DMCs were identified along with a correlation between 5-mC and gene expression | [142] |
| 5-mC | WNT5B, ANK1, ARD5B | Increase and decrease | AD patients | Illumina Infinium Human Methylation 450K microarray | Increased 5-mC level in WNT5B, ANK1, and decreased in ARD5Bz | [143] |
| 5-mC | Amyloid neuropathy and neurogenesis | Decrease | AD and healthy patients | RNA sequencing, aging analysis, gene annotation, and enrichment analysis | Identification of 1224 DMRs, enhancement in the DCSAML1 gene which targets BACE1 | [144] |
| 5-mC | - | Decrease | AD and healthy patients | Immunohistochemistry | Downregulation of 5-mC and negative correlation between 5mC and amyloid plaque level | [145] |
| 5-mC | - | Increase | AD patients and preclinical samples | Immunohistochemistry | Upregulation of 5-mC and hippocampus gyrus in both clinical and preclinical cases | [146] |
| 5-mC | - | Increase | Early and late-onset AD patients | Immunohistochemistry | Upregulation of 5mC in middle frontal gyrus and middle temporal gyrus in AD patients and shows a positive correlation with AD biomarkers | [147] |
| 5-mC | AS3MT, WTI, TBX15 | Decrease | AD with psychosis and without psychosis patients | Immunohistochemistry | Decrease level of AS3MT, WTI, TBX15 gene associated with AD patients | [148] |
| 5-mC | - | Decrease | Early and late AD patients | Immunohistochemistry | Genetic dysregulation may be occurring in astrocytes and NF-positive pyramidal neurons in AD | [149] |
| IL-1β Promoters | IL-1β | Decrease | BALB/c mice (3–4- and 18–20-month-old) |
LPS-induced neuroinflammation and Quantitative PCR (qPCR) | Microglial transferred to M1 phenotype which causes neuroinflammation and neuronal cell damages | [150] |
| SNCA Promoters | SNCA | Decrease | Healthy and PD patients | qPCR | Aggregation of a-syn, neuronal damage of DA, and neuroinflammation is triggered by activating glial cells | [151] |
| PGC-1α Promoters | PGC-1α | Increase | Human brain of PD and healthy patients | Bisulfite sequencing, Microarray gene expression analysis, ELISA analysis | Up-regulation of neuroinflammation, ER stress, epigenetic modification, and ROS production | [152] |
| TNF-α Promoters | TNF-alpha | Decrease | PD and healthy patients | Bisulfite PCR and sequencing | SNpc cells could underlie the increased susceptibility of dopaminergic neurons to TNF-alpha-mediated inflammatory reactions. | [153] |
| NOS2 Promoters | NOS2 | Decrease | PD and healthy patients | Qiagen’s Assay | Down-regulation of NO production to deactivate the microglial | [154] |
5.3. Huntington Disease
Huntington’s disease (HD) is caused by a CAG repeat mutation in the huntingtin gene. Huntingtin is a disease protein that has been established to affect a variety of epigenetic markers, such as histone modifications, and mainly DNA methylation [155]. Huntingtin (Htt), a protein with an enlarged glutamine domain, is prearranged by the mutant Htt gene. The specific mechanism by which the mutant huntingtin protein causes degeneration of neurons is unidentified [156]. Many interactions with certain transcription factors are involved, such as meddling with protein synthesis mechanisms and modifications of histones after protein synthesis. The mutant Htt has a large influence on gene expression, by pushing chromatin towards a thicker form [157,158]. Changes in DNA methylation have been associated with Htt protein expression in a variety of HD model systems, and in the human HD brain, according to numerous studies. Cedric Ng and his coworkers revealed differential methylation in DNA patterns in STHdhQ111 cells compared to WTQ7 cells of mice by using condensed representation bisulfite sequencing to check DNA methylation [77]. Transgenic mice had decreased levels of 5-hydroxymethylcytosine in the striatum and brain compared to controls [159]. One study claimed that the hairy enhancer of split-4 (HES4) gene promoter has a major site-specific DNA methylation modification in HD patients [160].
In the putamen of HD patients and the striatum of R6/1 and R6/2 mice, 5-methylcytidine-5′-monophosphate and 5-hydroxymethylcytosine contents were detected in the 5′ untranslated region (5′UTR) region of the adenosine receptor gene-A2A (ADORA2A). The pathological drop in adenosine A2a receptor expression levels seen in HD is linked to the abnormal methylation patterns of the adenosine receptor gene A2a [161]. Growth arrest and DNA-damage-45a (Gadd45a) expression were shown to be lower in the striatum of animals and muscle of transgenic mice, while growth arrest and the expression of DNA-damage-45g, a member of a group of genes whose transcript levels are increased following stressful growth arrest conditions, were found to be elevated in HD STHdhQ111 cells. Finally, one more investigation revealed that ring finger protein 4, another gene associated with DNA demethylation, was expressed differently in HD mice [162].
One study also suggested that altered DNA methylation profiles of CLDN16, DDC, and NXT2 also play a role in the progression of the disease [163]. Twelve genes—FBXL5, S100P, PRDX1, COPS7B, SP1, SEC24C, PDIA6, USP5, GRAP, POP5, WRB, and PCSK7—were also found to be differently methylated in HD patients’ blood. Sox2, Pax6, and Nes genes were found to have increased methylation levels, which tends to reduce their expression and result in impaired neurogenesis [73,164].
5.4. Amyotrophic Lateral Sclerosis
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease. Motor neurons in the motor cortex, brain stem, and spinal cord degenerate in this condition. Oxidative stress, glutamate excitotoxicity, impaired axonal transport, neurotrophic deprivation, neuroinflammation, apoptosis, altered protein turnover, and mitochondrial dysfunction are the mechanisms involved in the pathogenesis of ALS. Changes in the immune system, more physical activity, exposure to toxins, and dietary factors can also lead to the progression of this disease [165]. Alteration of DNA methylation sequences also plays a role in ALS. Elevated levels of 5-methylcytosine participate in disease pathology. This is due to the increased activity of DNMTs [166].
Increased levels of 5-hydroxymethylcytocise lead to aging and a rise in oxidative stress [167]. Methylation changes influence immune-related genes, i.e., TREM2, chemokine (C-C motif) receptor1/RANTES receptor (CCR1), SLC11A1, the transmembrane receptor C-type lectin domain family 4 member A isoform 1 (CLEC4A), and the IgE receptor (FCER1G). All these genes are elevated in ALS and cause an increase in the number of immune cells [168,169].
STAT5A and C/EBPB are transcription factors that activate other genes such as interleukin 6, which are responsible for the pathogenesis of ALS and neurodegeneration [170]. Changes in gene expression of Slit-Robo Rho GTPase activating protein 1 (SRGAP1), Crumbs homolog 1 (CRB1), MSX2, MLC1, CTNND2, AXL, RUNX3, NNAT, and NRN1 cause neurodegeneration. Crumbs homolog 1 was found to be hypomethylated and responsible for intellectual disability and neurodegeneration in ALS patients [171,172,173]. Scientists also reported a modest overlap of four concordant epigeneses; Purkinje cell protein 4 (PCP4), catenin (CTNNAL1), fibroblast growth factor 18 (FGF18), and flavin-containing monooxygenase 1 (FMO1) [164]. Mutations in SOD1, FUS, TARDBP, and C9orf72 cause oxidative stress in ALS [173].
Mitochondria are the powerhouses of the cell. Alterations in mitochondria can also be related to ALS. Mito-epigenetics assesses the DNA methylation and DNA hydroxymethylation levels in mitochondria. One study also proved the role of mitochondria in ALS by demonstrating that mitochondrial DNA methylation patterns are altered in the skeletal muscles and spinal cord of ALS patients [173]. D-loop hypomethylation in SOD1 carriers could be related to a rise in mtDNA methylation, which gives rise to an increase in mtDNA methylation that causes an increase in oxidative stress. SODI is an antioxidant enzyme. A mutation in this enzyme causes ALS disease.
6. Targeting DNA Methylation in Management of AD and Other Neurodegenerative Diseases
Identifying DNA methylation in peripheral blood and brain samples could be a promising biomarker for diagnosing AD. In this regard, the hypermethylated APP gene appears as a promising biomarker for AD prognosis. Based upon these observations, several researchers have carried out some preliminary studies which support the assertion that targeting DNMT could be beneficial in halting amyloid pathology and other neurodegenerative diseases. Some of the corresponding studies are presented in Table 3 and are elaborated herewith.
Table 3.
Recent advances in the management of AD and other neurogenerative diseases by targeting DNA methylation.
| Neurodegenerative Disease | Drug | Class of Drug | Inference | Reference |
|---|---|---|---|---|
| AD | Epigallocatechin gallate, epigallocatechin 3-gallate, tea catechin, tea vigo, catechin deriv., | DNMT inhibitors | Improve memory, prevent cell death in Aβ-treated neurons, Aβ aggregation. | [174] |
| AD | Vitamin B6, folate, Folacin; Pteroylglutamic acid | SAMe methyl donors | Attenuate homocystine level | [174] |
| PD | 5-Aza-2′-Deoxycytidine | DNMTs inhibitor | Upregulate tyrosine hydroxylase, dopamine production, and alpha-synuclein expression | [175] |
| HD | decitabine and FdCyd | DNMTs inhibitors | Restore expression of Bndf | [176] |
| ALS | RG108 | DNMTs inhibitors | Block DNA methylation accumulation in motor neurons | [177] |
Epigallocatechin gallate, epigallocatechin 3-gallate, tea catechin, and catechin derivatives are example DNMTs inhibitors that have proven their role in the treatment of AD. Epigallocatechin-3-gallate (EGCG) prevents misfolded proteins from undergoing fibrillation and protects from cell death in Aβ-treated neurons. Etanercept also helps to treat AD by modulating the immune system. Neurodegeneration medications include 5-aza-2′-deoxycytidine (decitabine) and 5-azacytidine (azacitidine), as well as the small molecules hydralazine and procainamide [174].
Wang et al., 2013, tested 5-aza-2′-deoxycytidine (5-aza-dC), a DNMT inhibitor, in the treatment of Parkinson’s disease. The scientist reported that 5-aza-dC induced CpG demethylation in the promoter and upregulated transcriptional levels of the α-synuclein gene. 5-aza-dC has been demonstrated to increase the expression of tyrosine hydroxylase dopamine production and alpha-synuclein expression. [175]. In general, if levodopa is shown to work via an epigenetic pathway, existing treatments should be reassessed to elucidate fresh epigenetic characteristics and develop innovative and more targeted drugs [178]. Vitamin B, folic acid, and SAMe are the main methylation storage compounds submitted to clinical trials for the treatment of neurodegenerative diseases [174]. Folate and vitamin B6 are also prescribed by a doctor to treat elevated levels of homocysteine, which is a known risk factor for AD [174].
Pan et al., 2016, proved that decitabine and FdCyd, DNMT inhibitors, can attenuate neurotoxicity in HD patients. DNMT inhibition leads to the restoration of the expression of Bndf and can be used as a therapeutic target for treatment [176].
5-aza-cytidine (5-azaC), 5-aza-2-deoxycytidine (5-azadC, decitabine), zebularine, and RG108 are drugs that inhibit DNA methylation and can be used in the improvement of ALS. RG108 blocks the methylation in motor neurons and causes improvement in disease [177].
7. Conclusions
This review summarizes those epigenetic modifications that are responsible for many genes’ functions in the body. Studies have investigated the relationship between DNA methylation of genes and their level in the pathology of neurodegenerative diseases. DNA methylation influences the pathophysiology of age-related disease, aging, and dementia [179,180], Modifications in chromatin organization, transcriptional changes, and a variety of neurological illnesses and diseases are all linked to abnormal methylation changes [181]. In AD, the DNA methylation of genes such as amyloid precursor protein, PSEN1, MAPT, apolipoprotein E, presenilin-1, beta-secretase 1 precursor, or apolipoprotein E, Sorbin SH3 domain containing 3 (Sorbs3) and BDNF was found to be altered in three parts of the brain. Based on the above-mentioned points, it may be proposed the identification of DNA methylation in peripheral blood and brain samples could provide valuable insights for AD diagnosis. Furthermore, designing suitable DNA methylation promotors or inhibitors could provide a novel target for the management of AD and other neurodegenerative diseases.
Acknowledgments
The author (AM) would like to acknowledge the infrastructure and facility provided by the National Institute of Pharmaceutical Education and Research (NIPER), Guwahati and Department of Pharmaceuticals, Ministry of Chemicals, and Fertilizers, Government of India. The images in the manuscript are “Created with BioRender.com”.
Author Contributions
Conceptualization, G.K. and A.M.; methodology, G.K. and A.M.; software, A.M.; validation, A.M. and J.A.; formal analysis, G.K. and S.S.S.R.; investigation, G.K., S.S.S.R., M.M.G., S.A., J.A. and N.A.A.; resources, J.A.; writing—original draft preparation, G.K., S.S.S.R., M.M.G. and S.A.; writing—review and editing, J.A., A.M. and N.A.A.; visualization, J.A.; supervision, A.M.; funding acquisition, N.A.A. All authors have read and agreed to the published version of the manuscript.
Funding
The Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, Saudi Arabia has funded this project, under grant no. (FP-017-43).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing does not apply to this article.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Jin B., Li Y., Robertson K.D. DNA Methylation: Superior or Subordinate in the Epigenetic Hierarchy? Genes Cancer. 2011;2:607–617. doi: 10.1177/1947601910393957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Robertson K.D. DNA methylation and human disease. Nat. Rev. Genet. 2005;6:597–610. doi: 10.1038/nrg1655. [DOI] [PubMed] [Google Scholar]
- 3.Lister R., Pelizzola M., Dowen R.H., Hawkins R.D., Hon G., Tonti-Filippini J., Nery J.R., Lee L., Ye Z., Ngo Q.-M., et al. Human DNA methylomes at base resolution show widespread epigenomic differences. Nature. 2009;462:315–322. doi: 10.1038/nature08514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhu J.-K. Active DNA Demethylation Mediated by DNA Glycosylases. Annu. Rev. Genet. 2009;43:143–166. doi: 10.1146/annurev-genet-102108-134205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Holliday R., Pugh J.E. DNA Modification Mechanisms and Gene Activity during Development. Science. 1975;187:226–232. doi: 10.1126/science.187.4173.226. [DOI] [PubMed] [Google Scholar]
- 6.Compere S.J., Palmiter R.D. DNA methylation controls the inducibility of the mouse metallothionein-I gene in lymphoid cells. Cell. 1981;25:233–240. doi: 10.1016/0092-8674(81)90248-8. [DOI] [PubMed] [Google Scholar]
- 7.Moore L.D., Le T., Fan G. DNA Methylation and Its Basic Function. Neuropsychopharmacology. 2013;38:23–38. doi: 10.1038/npp.2012.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Deng J., Szyf M. Downregulation of DNA (cytosine-5-) methyltransferase is a late event in NGF-induced PC12 cell differen-tiation. Brain Res. Mol. Brain Res. 1999;71:23–31. doi: 10.1016/S0169-328X(99)00147-3. [DOI] [PubMed] [Google Scholar]
- 9.Goto K., Numata M., Komura J.-I., Ono T., Bestor T.H., Kondo H. Expression of DNA methyltransferase gene in mature and immature neurons as well as proliferating cells in mice. Differentiation. 1994;56:39–44. doi: 10.1046/j.1432-0436.1994.56120039.x. [DOI] [PubMed] [Google Scholar]
- 10.Suzuki M.M., Kerr A.R., De Sousa D., Bird A. CpG methylation is targeted to transcription units in an invertebrate genome. Genome Res. 2007;17:625–631. doi: 10.1101/gr.6163007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schübeler D. Function and information content of DNA methylation. Nature. 2015;517:321–326. doi: 10.1038/nature14192. [DOI] [PubMed] [Google Scholar]
- 12.Cedar H., Bergman Y. Linking DNA methylation and histone modification: Patterns and paradigms. Nat. Rev. Genet. 2009;10:295–304. doi: 10.1038/nrg2540. [DOI] [PubMed] [Google Scholar]
- 13.Lehnertz B., Ueda Y., Derijck A.A., Braunschweig U., Perez-Burgos L., Kubicek S., Chen T., Li E., Jenuwein T., Peters A.H. Suv39h-Mediated Histone H3 Lysine 9 Methylation Directs DNA Methylation to Major Satellite Repeats at Pericentric Heterochromatin. Curr. Biol. 2003;13:1192–1200. doi: 10.1016/S0960-9822(03)00432-9. [DOI] [PubMed] [Google Scholar]
- 14.Vire E., Brenner C., Deplus R., Blanchon L., Fraga M., Didelot C.M., Morey L., Van Eynde A., Bernard D., Vanderwinden J.-M., et al. The Polycomb group protein EZH2 directly controls DNA methylation. Nature. 2005;439:871–874. doi: 10.1038/nature04431. [DOI] [PubMed] [Google Scholar]
- 15.Saxonov S., Berg P., Brutlag D.L. A genome-wide analysis of CpG dinucleotides in the human genome distinguishes two distinct classes of promoters. Proc. Natl. Acad. Sci. USA. 2006;103:1412–1417. doi: 10.1073/pnas.0510310103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tazi J., Bird A. Alternative chromatin structure at CpG islands. Cell. 1990;60:909–920. doi: 10.1016/0092-8674(90)90339-G. [DOI] [PubMed] [Google Scholar]
- 17.Ramirez-Carrozzi V.R., Braas D., Bhatt D.M., Cheng C.S., Hong C., Doty K.R., Black J., Hoffmann A., Carey M., Smale S.T. A Unifying Model for the Selective Regulation of Inducible Transcription by CpG Islands and Nucleosome Remodeling. Cell. 2009;138:114–128. doi: 10.1016/j.cell.2009.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Carninci P., Sandelin A., Lenhard B., Katayama S., Shimokawa K., Ponjavic J., Semple C., Taylor M., Engström P., Frith M., et al. Genome-wide analysis of mammalian promoter architecture and evolution. Nat. Genet. 2006;38:626–635. doi: 10.1038/ng1789. [DOI] [PubMed] [Google Scholar]
- 19.Iyer L.M., Abhiman S., Aravind L. Natural History of Eukaryotic DNA Methylation Systems. Prog. Mol. Biol. Transl. Sci. 2011;101:25–104. doi: 10.1016/b978-0-12-387685-0.00002-0. [DOI] [PubMed] [Google Scholar]
- 20.Avery O.T., MacLeod C.M., Mccarty M. Studies on the chemical nature of the substance inducing transformation of pneumococcal types: Induction of transformation by a desoxyribonucleic acid fraction isolated from pneumococcus type III. J. Exp. Med. 1944;79:137–158. doi: 10.1084/jem.79.2.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gerlach E., Dreisbach R., Deuticke B. Paper chromatographic separation of nucleotides, nucleosides, purines, and pyrimidines. J. Chromatogr. A. 1965;18:81–85. doi: 10.1016/S0021-9673(01)80323-X. [DOI] [PubMed] [Google Scholar]
- 22.MacDonald J.L., Roskams A.J. Epigenetic regulation of nervous system development by DNA methylation and histone deacetylation. Prog. Neurobiol. 2009;88:170–183. doi: 10.1016/j.pneurobio.2009.04.002. [DOI] [PubMed] [Google Scholar]
- 23.Iyer L.M., Abhiman S., De Souza R.F., Aravind L. Origin and evolution of peptide-modifying dioxygenases and identification of the wybutosine hydroxylase/hydroperoxidase. Nucleic Acids Res. 2010;38:5261–5279. doi: 10.1093/nar/gkq265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Laird P.W. Principles and challenges of genome-wide DNA methylation analysis. Nat. Rev. Genet. 2010;11:191–203. doi: 10.1038/nrg2732. [DOI] [PubMed] [Google Scholar]
- 25.Kurdyukov S., Bullock M. DNA Methylation Analysis: Choosing the Right Method. Biology. 2016;5:3. doi: 10.3390/biology5010003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Song L., James S.R., Kazim L., Karpf A.R. Specific Method for the Determination of Genomic DNA Methylation by Liquid Chromatography-Electrospray Ionization Tandem Mass Spectrometry. Anal. Chem. 2004;77:504–510. doi: 10.1021/ac0489420. [DOI] [PubMed] [Google Scholar]
- 27.Hur K., Cejas P., Feliu J., Moreno-Rubio J., Burgos E., Boland C.R., Goel A. Hypomethylation of long interspersed nuclear element-1 (LINE-1) leads to activation of proto-oncogenes in human colorectal cancer metastasis. Gut. 2013;63:635–646. doi: 10.1136/gutjnl-2012-304219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jaligot E., Beulé T., Rival A. Methylation-sensitive RFLPs: Characterisation of two oil palm markers showing somaclonal variation-associated polymorphism. Theor. Appl. Genet. 2002;104:1263–1269. doi: 10.1007/s00122-002-0906-4. [DOI] [PubMed] [Google Scholar]
- 29.Karimi M., Johansson S., Stach D., Corcoran M., Grandér D., Schalling M., Bakalkin G., Lyko F., Larsson C., Ekström T.J. LUMA (LUminometric Methylation Assay)—A high throughput method to the analysis of genomic DNA methylation. Exp. Cell Res. 2006;312:1989–1995. doi: 10.1016/j.yexcr.2006.03.006. [DOI] [PubMed] [Google Scholar]
- 30.Miura F., Fumihito M. Highly sensitive targeted methylome sequencing by post-bisulfite adaptor tagging. DNA Res. 2014;22:13–18. doi: 10.1093/dnares/dsu034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bibikova M., Fan J.-B. GoldenGate® Assay for DNA Methylation Profiling. Methods Mol. Biol. 2009;507:149–163. doi: 10.1007/978-1-59745-522-0_12. [DOI] [PubMed] [Google Scholar]
- 32.Herman J.G., Graff J.R., Myohanen S., Nelkin B.D., Baylin S.B. Methylation-specific PCR: A novel PCR assay for methylation status of CpG islands. Proc. Natl. Acad. Sci. USA. 1996;93:9821–9826. doi: 10.1073/pnas.93.18.9821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mahapatra S., Klee E.W., Young C.Y., Sun Z., Jimenez R.E., Klee G.G., Tindall D.J., Donkena K.V. Global Methylation Profiling for Risk Prediction of Prostate Cancer. Clin. Cancer Res. 2012;18:2882–2895. doi: 10.1158/1078-0432.CCR-11-2090. [DOI] [PubMed] [Google Scholar]
- 34.Qian X., Shen Q., Goderie S.K., He W., Capela A., Davis A.A., Temple S. Timing of CNS Cell Generation. Neuron. 2000;28:69–80. doi: 10.1016/S0896-6273(00)00086-6. [DOI] [PubMed] [Google Scholar]
- 35.Teter B., Osterburg H.H., Anderson C.P., Finch C.E. Methylation of the rat glial fibrillary acidic protein gene shows tissue-specific domains. J. Neurosci. Res. 1994;39:680–693. doi: 10.1002/jnr.490390609. [DOI] [PubMed] [Google Scholar]
- 36.Yokoyama A.S., Rutledge J.C., Medici V. DNA methylation alterations in Alzheimer’s disease. Environ. Epigenet. 2017;3:dvx008. doi: 10.1093/eep/dvx008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Feng J., Zhou Y., Campbell S.L., Le T., Li E., Sweatt J.D., Silva A., Fan G. Dnmt1 and Dnmt3a maintain DNA methylation and regulate synaptic function in adult forebrain neurons. Nat. Neurosci. 2010;13:423–430. doi: 10.1038/nn.2514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lubin F.D., Roth T.L., Sweatt J.D. Epigenetic Regulation of bdnf Gene Transcription in the Consolidation of Fear Memory. J. Neurosci. 2008;28:10576–10586. doi: 10.1523/JNEUROSCI.1786-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Martinowich K., Hattori D., Wu H., Fouse S., He F., Hu Y., Fan G., Sun Y.E. DNA Methylation-Related Chromatin Remodeling in Activity-Dependent Bdnf Gene Regulation. Science. 2003;302:890–893. doi: 10.1126/science.1090842. [DOI] [PubMed] [Google Scholar]
- 40.Guo J.U., Ma D.K., Mo H., Ball M., Jang M.-H., Bonaguidi M.A., Balazer J.A., Eaves H.L., Xie B., Ford E., et al. Neuronal activity modifies the DNA methylation landscape in the adult brain. Nat. Neurosci. 2011;14:1345–1351. doi: 10.1038/nn.2900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nguyen S., Meletis K., Fu D., Jhaveri S., Jaenisch R. Ablation of de novo DNA methyltransferase Dnmt3a in the nervous system leads to neuromuscular defects and shortened lifespan. Dev. Dyn. 2007;236:1663–1676. doi: 10.1002/dvdy.21176. [DOI] [PubMed] [Google Scholar]
- 42.LaSalle J.M., Goldstine J., Balmer D., Greco C.M. Quantitative localization of heterogeneous methyl-CpG-binding protein 2 (MeCP2) expression phenotypes in normal and Rett syndrome brain by laser scanning cytometry. Hum. Mol. Genet. 2001;10:1729–1740. doi: 10.1093/hmg/10.17.1729. [DOI] [PubMed] [Google Scholar]
- 43.Nan X., Ng H.-H., Johnson C.A., Laherty C.D., Turner B.M., Eisenman R.N., Bird A. Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature. 1998;393:386–389. doi: 10.1038/30764. [DOI] [PubMed] [Google Scholar]
- 44.Sarraf S.A., Stancheva I. RETRACTED: Methyl-CpG Binding Protein MBD1 Couples Histone H3 Methylation at Lysine 9 by SETDB1 to DNA Replication and Chromatin Assembly. Mol. Cell. 2004;15:595–605. doi: 10.1016/j.molcel.2004.06.043. [DOI] [PubMed] [Google Scholar]
- 45.Chen R.Z., Akbarian S., Tudor M., Jaenisch R. Deficiency of methyl-CpG binding protein-2 in CNS neurons results in a Rett-like phenotype in mice. Nat. Genet. 2001;27:327–331. doi: 10.1038/85906. [DOI] [PubMed] [Google Scholar]
- 46.Moretti P., Levenson J.M., Battaglia F., Atkinson R., Teague R., Antalffy B., Armstrong D., Arancio O., Sweatt J.D., Zoghbi H. Learning and Memory and Synaptic Plasticity Are Impaired in a Mouse Model of Rett Syndrome. J. Neurosci. 2006;26:319–327. doi: 10.1523/JNEUROSCI.2623-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Nelson E.D., Kavalali E.T., Monteggia L.M. MeCP2-Dependent Transcriptional Repression Regulates Excitatory Neurotransmission. Curr. Biol. 2006;16:710–716. doi: 10.1016/j.cub.2006.02.062. [DOI] [PubMed] [Google Scholar]
- 48.Ng H.-H., Zhang Y., Hendrich B., Johnson C.A., Turner B.M., Erdjument-Bromage H., Tempst P., Reinberg D., Bird A. MBD2 is a transcriptional repressor belonging to the MeCP1 histone deacetylase complex. Nat. Genet. 1999;23:58–61. doi: 10.1038/12659. [DOI] [PubMed] [Google Scholar]
- 49.Cohen S., Gabel H.W., Hemberg M., Hutchinson A.N., Sadacca L.A., Ebert D.H., Harmin D.A., Greenberg R.S., Verdine V.K., Zhou Z., et al. Genome-Wide Activity-Dependent MeCP2 Phosphorylation Regulates Nervous System Development and Function. Neuron. 2011;72:72–85. doi: 10.1016/j.neuron.2011.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Li B.-Z., Huang Z., Cui Q., Song X.-H., Du L., Jeltsch A., Chen P., Li G., Li E., Xu G.-L. Histone tails regulate DNA methylation by allosterically activating de novo methyltransferase. Cell Res. 2011;21:1172–1181. doi: 10.1038/cr.2011.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Li H., Zhong X., Chau K.F., Williams E.C., Chang Q. Loss of activity-induced phosphorylation of MeCP2 enhances synaptogenesis, LTP and spatial memory. Nat. Neurosci. 2011;14:1001–1008. doi: 10.1038/nn.2866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gao L., Emperle M., Guo Y., Grimm S.A., Ren W., Adam S., Uryu H., Zhang Z.M., Chen D., Yin J., et al. Comprehensive struc-ture-function characterization of DNMT3B and DNMT3A reveals distinctive de novo DNA methylation mechanisms. Nat. Commun. 2020;11:3355. doi: 10.1038/s41467-020-17109-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Vignini A., Morganti S., Salvolini E., Sartini D., Luzzi S., Fiorini R., Provinciali L., Di Primio R., Mazzanti L., Emanuelli M. Amyloid precursor protein expression is enhanced in human platelets from subjects with Alzheimer’s disease and frontotemporal lobar degeneration: A real-time PCR study. Exp. Gerontol. 2013;48:1505–1508. doi: 10.1016/j.exger.2013.10.008. [DOI] [PubMed] [Google Scholar]
- 54.Traynor B.J., Renton A.E. Exploring the epigenetics of Alzheimer disease. JAMA Neurol. 2015;72:8–9. doi: 10.1001/jamaneurol.2014.3057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Altuna M., Urdánoz-Casado A., Sánchez-Ruiz de Gordoa J., Zelaya M.V., Labarga A., Lepesant J., Roldán M., Blanco-Luquin I., Per-dones Á., Larumbe R., et al. DNA methylation signature of human hippocampus in Alzheimer’s disease is linked to neurogenesis. Clin. Epigenet. 2019;11:91. doi: 10.1186/s13148-019-0672-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Huo Z., Zhu Y., Yu L., Yang J., De Jager P., Bennett D.A., Zhao J. DNA methylation variability in Alzheimer’s disease. Neurobiol. Aging. 2019;76:35–44. doi: 10.1016/j.neurobiolaging.2018.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tan Y.J., Ng A., Vipin A., Lim J., Chander R.J., Ji F., Qiu Y., Ting S., Hameed S., Lee T.S., et al. Higher Pe-ripheral TREM2 mRNA Levels Relate to Cognitive Deficits and Hippocampal Atrophy in Alzheimer’s Disease and Amnestic Mild Cognitive Impairment. J. Alzheimers Dis. 2017;58:413–423. doi: 10.3233/JAD-161277. [DOI] [PubMed] [Google Scholar]
- 58.Liu G., Ji H., Liu J., Xu C., Chang L., Cui W., Ye C., Hu H., Chen Y., Zhou X., et al. Association of OPRK1 and OPRM1 methylation with mild cognitive impairment in Xinjiang Han and Uygur populations. Neurosci. Lett. 2017;636:170–176. doi: 10.1016/j.neulet.2016.11.018. [DOI] [PubMed] [Google Scholar]
- 59.Mitsumori R., Sakaguchi K., Shigemizu D., Mori T., Akiyama S., Ozaki K., Niida S., Shimoda N. Lower DNA methylation levels in CpG island shores of CR1, CLU, and PICALM in the blood of Japanese Alzheimer’s disease patients. PLoS ONE. 2020;15:e0239196. doi: 10.1371/journal.pone.0239196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Smith A.R., Smith R.G., Macdonald R., Marzi S.J., Burrage J., Troakes C., Al-Sarraj S., Mill J., Lunnon K. The histone modification H3K4me3 is al-tered at the ANK1 locus in Alzheimer’s disease brain. Future Sci. OA. 2021;7:FSO665. doi: 10.2144/fsoa-2020-0161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wang J., Yu J.T., Tan L. PLD3 in Alzheimer’s disease. Mol. Neurobiol. 2015;51:480–486. doi: 10.1007/s12035-014-8779-5. [DOI] [PubMed] [Google Scholar]
- 62.Masliah E., Dumaop W., Galasko U., Desplats P. Distinctive patterns of DNA methylation associated with Parkinson disease: Identification of concordant epigenetic changes in brain and peripheral blood leukocytes. Epigenetics. 2013;8:1030–1038. doi: 10.4161/epi.25865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gu J., Barrera J., Yun Y., Murphy S.K., Beach T.G., Woltjer R.L., Serrano G.E., Kantor B., Chiba-Falek O. Cell-Type Specific Changes in DNA Methylation of SNCA Intron 1 in Synucleinopathy Brains. Front. Neurosci. 2021;15:652226. doi: 10.3389/fnins.2021.652226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nasamran C.A., Sachan A.N.S., Mott J., Kuras Y.I., Scherzer C.R., Harvard Biomarkers Study. Ricciardelli E., Jepsen K., Edland S.D., Fisch K.M., et al. Differential blood DNA methylation across Lewy body dementias. Alzheimer’s Dement. Diagn. Assess. Dis. Monit. 2021;13:e12156. doi: 10.1002/dad2.12156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chuang Y.-H., Paul K.C., Bronstein J.M., Bordelon Y., Horvath S., Ritz B. Parkinson’s disease is associated with DNA methylation levels in human blood and saliva. Genome Med. 2017;9:76. doi: 10.1186/s13073-017-0466-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Young J.I., Sivasankaran S.K., Wang L., Ali A., Mehta A., Davis D.A., Dykxhoorn D.M., Petito C.K., Beecham G.W., Martin E.R., et al. Genome-wide brain DNA methylation analysis suggests epigenetic reprogramming in Parkinson disease. Neurol. Genet. 2019;5:e342. doi: 10.1212/NXG.0000000000000342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mao W., Zhao C., Ding H., Liang K., Xue J., Chan P., Cai Y. Pyrosequencing analysis of methylation levels of clock genes in leukocytes from Parkinson’s disease patients. Neurosci. Lett. 2018;668:115–119. doi: 10.1016/j.neulet.2018.01.027. [DOI] [PubMed] [Google Scholar]
- 68.Mudò G., Mäkelä J., Di Liberto V., Tselykh T.V., Olivieri M., Piepponen P., Eriksson O., Mälkiä A., Bonomo A., Kairisalo M., et al. Transgenic expression and activation of PGC-1α protect dopaminergic neurons in the MPTP mouse model of Parkinson’s disease. Cell. Mol. Life Sci. 2012;69:1153–1165. doi: 10.1007/s00018-011-0850-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kaut O., Schmitt I., Wüllner U. Genome-scale methylation analysis of Parkinson’s disease patients’ brains reveals DNA hypomethylation and increased mRNA expression of cytochrome P450 2E1. Neurogenetics. 2012;13:87–91. doi: 10.1007/s10048-011-0308-3. [DOI] [PubMed] [Google Scholar]
- 70.Racette B.A., Searles Nielsen S., Criswell S.R., Sheppard L., Seixas N., Warden M.N., Checkoway H. Dose-dependent progression of parkinsonism in manganese-exposed welders. Neurology. 2017;88:344–351. doi: 10.1212/WNL.0000000000003533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Coupland K.G., Kim W.S., Halliday G.M., Hallupp M., Dobson-Stone C., Kwok J.B. Role of the Long Non-Coding RNA MAPT-AS1 in Regulation of Microtubule Associated Protein Tau (MAPT) Expression in Parkinson’s Disease. PLoS ONE. 2016;11:e0157924. doi: 10.1371/journal.pone.0157924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Go R., Corley M.J., Ross G.W., Petrovitch H., Masaki K.H., Maunakea A.K., He Q., Tiirikainen M.I. Ge-nome-wide epigenetic analyses in Japanese immigrant plantation workers with Parkinson’s disease and exposure to orga-nochlorines reveal possible involvement of glial genes and pathways involved in neurotoxicity. BMC Neurosci. 2020;21:31. doi: 10.1186/s12868-020-00582-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Wood H. Neurodegenerative disease: Altered DNA methylation and RNA splicing could be key mechanisms in Huntington dis-ease. Nature reviews. Neurology. 2013;9:119. doi: 10.1038/nrneurol.2013.23. [DOI] [PubMed] [Google Scholar]
- 74.Dong X., Tsuji J., Labadorf A., Roussos P., Chen J.F., Myers R.H., Akbarian S., Weng Z. The Role of H3K4me3 in Transcrip-tional Regulation Is Altered in Huntington’s Disease. PLoS ONE. 2015;10:e0144398. doi: 10.1371/journal.pone.0144398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Villar-Menéndez I., Nuñez F., Díaz-Sánchez S., Albasanz J.L., Taura J., Fernández-Dueñas V., Ferrer I., Martín M., Ciruela F., Barrachina M. Striatal adenosine A2A receptor expression is controlled by S-adenosyl-L-methionine-mediated methylation. Purinergic Signal. 2014;10:523–528. doi: 10.1007/s11302-014-9417-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zsindely N., Siági F., Bodai L. DNA Methylation in Huntington’s Disease. Int. J. Mol. Sci. 2021;22:12736. doi: 10.3390/ijms222312736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ng C.W., Yildirim F., Yap Y.S., Dalin S., Matthews B.J., Velez P.J., Labadorf A., Housman D.E., Fraenkel E. Extensive changes in DNA methylation are associated with expression of mutant huntingtin. Proc. Natl. Acad. Sci. USA. 2013;110:2354–2359. doi: 10.1073/pnas.1221292110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lederer C.W., Torrisi A., Pantelidou M., Santama N., Cavallaro S. Pathways and genes differentially expressed in the motor cortex of patients with sporadic amyotrophic lateral sclerosis. BMC Genom. 2007;8:26. doi: 10.1186/1471-2164-8-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hurd M.D., Martorell P., Delavande A., Mullen K.J., Langa K. Monetary Costs of Dementia in the United States. N. Engl. J. Med. 2013;368:1326–1334. doi: 10.1056/NEJMsa1204629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wang J., Yu J.T., Tan M.S., Jiang T., Tan L. Epigenetic mechanisms in Alzheimer’s disease: Implications for pathogenesis and therapy. Ageing Res. Rev. 2013;12:1024–1041. doi: 10.1016/j.arr.2013.05.003. [DOI] [PubMed] [Google Scholar]
- 81.Lybartseva G., Smith J.L., Markesbery W.R., Lovell M.A. Alterations of zinc transporter proteins ZnT-1, ZnT-4 and ZnT-6 in preclinical Alzheimer’s disease brain. Brain Pathol. 2010;20:343–350. doi: 10.1111/j.1750-3639.2009.00283.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lashley T., Gami P., Valizadeh N., Li A., Revesz T., Balazs R. Alterations in global DNA methylation and hydroxymethylation are not detected in Alzheimer’s disease. Neuropathol. Appl. Neurobiol. 2015;41:497–506. doi: 10.1111/nan.12183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Wang S.-C., Oelze B., Schumacher A. Age-Specific Epigenetic Drift in Late-Onset Alzheimer’s Disease. PLoS ONE. 2008;3:e2698. doi: 10.1371/journal.pone.0002698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hou Y., Chen H., He Q., Jiang W., Luo T., Duan J., Mu N., He Y., Wang H. Changes in methylation patterns of multiple genes from peripheral blood leucocytes of Alzheimer’s disease patients. Acta Neuropsychiatr. 2013;25:66–76. doi: 10.1111/j.1601-5215.2012.00662.x. [DOI] [PubMed] [Google Scholar]
- 85.Martínez-Iglesias O., Carrera I., Carril J.C., Fernández-Novoa L., Cacabelos N., Cacabelos R. DNA Methylation in Neurodegenerative and Cerebrovascular Disorders. Int. J. Mol. Sci. 2020;21:2220. doi: 10.3390/ijms21062220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.LaFerla F.M., Green K.N., Oddo S. Intracellular amyloid-β in Alzheimer’s disease. Nat. Rev. Neurosci. 2007;8:499–509. doi: 10.1038/nrn2168. [DOI] [PubMed] [Google Scholar]
- 87.Rogaeva E., Meng Y., Lee J.H., Gu Y., Kawarai T., Zou F., Katayama T., Baldwin C.T., Cheng R., Hasegawa H., et al. The neuronal sortilin-related receptor SORL1 is genetically associated with Alzheimer disease. Nat. Genet. 2007;39:168–177. doi: 10.1038/ng1943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Li H., Karl T., Garner B. Understanding the function of ABCA7 in Alzheimer’s disease. Biochem. Soc. Trans. 2015;43:920–923. doi: 10.1042/BST20150105. [DOI] [PubMed] [Google Scholar]
- 89.Tan M.-S., Yu J.-T., Tan L. Bridging integrator 1 (BIN1): Form, function, and Alzheimer’s disease. Trends Mol. Med. 2013;19:594–603. doi: 10.1016/j.molmed.2013.06.004. [DOI] [PubMed] [Google Scholar]
- 90.West R.L., Lee J.M., Maroun L.E. Hypomethylation of the amyloid precursor protein gene in the brain of an alzheimer’s disease patient. J. Mol. Neurosci. 1995;6:141–146. doi: 10.1007/BF02736773. [DOI] [PubMed] [Google Scholar]
- 91.Monti N., Cavallaro R.A., Stoccoro A., Nicolia V., Scarpa S., Kovacs G.G., Fiorenza M.T., Lucarelli M., Aronica E., Ferrer I., et al. CpG and non-CpG Presenilin1 methylation pattern in course of neurodevelopment and neurodegeneration is associated with gene expression in human and murine brain. Epigenetics. 2020;15:781–799. doi: 10.1080/15592294.2020.1722917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Globisch D., Münzel M., Müller M., Michalakis S., Wagner M., Koch S., Brückl T., Biel M., Carell T. Tissue Distribution of 5-Hydroxymethylcytosine and Search for Active Demethylation Intermediates. PLoS ONE. 2010;5:e15367. doi: 10.1371/journal.pone.0015367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lonze B., Ginty D.D. Function and Regulation of CREB Family Transcription Factors in the Nervous System. Neuron. 2002;35:605–623. doi: 10.1016/S0896-6273(02)00828-0. [DOI] [PubMed] [Google Scholar]
- 94.Mendioroz M., Celarain N., Altuna M., De Gordoa J.S.-R., Zelaya M.V., Roldán M., Rubio I., Larumbe R., Erro M.E., Méndez I., et al. CRTC1 gene is differentially methylated in the human hippocampus in Alzheimer’s disease. Alzheimer’s Res. Ther. 2016;8:15. doi: 10.1186/s13195-016-0183-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Tekpli X., Landvik N.E., Anmarkud K.H., Skaug V., Haugen A., Zienolddiny S. DNA methylation at promoter regions of interleukin 1B, interleukin 6, and interleukin 8 in non-small cell lung cancer. Cancer Immunol. Immunother. 2012;62:337–345. doi: 10.1007/s00262-012-1340-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Connor B., Young D., Yan Q., Faull R., Synek B., Dragunow M. Brain-derived neurotrophic factor is reduced in Alzheimer’s disease. Mol. Brain Res. 1997;49:71–81. doi: 10.1016/S0169-328X(97)00125-3. [DOI] [PubMed] [Google Scholar]
- 97.Tanila H. The role of BDNF in Alzheimer’s disease. Neurobiol. Dis. 2017;97:114–118. doi: 10.1016/j.nbd.2016.05.008. [DOI] [PubMed] [Google Scholar]
- 98.Chang L., Wang Y., Danjie J., Dai D., Xu X., Jiang D., Zhongming C., Ye H., Zhang X., Zhou X., et al. Elevation of Peripheral BDNF Promoter Methylation Links to the Risk of Alzheimer’s Disease. PLoS ONE. 2014;9:e110773. doi: 10.1371/journal.pone.0110773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Nagata T., Kobayashi N., Ishii J., Shinagawa S., Nakayama R., Shibata N., Kuerban B., Ohnuma T., Kondo K., Arai H., et al. Association between DNA Methylation of the BDNF Promoter Region and Clinical Presentation in Alzheimer’s Disease. Dement. Geriatr. Cogn. Disord. Extra. 2015;5:64–73. doi: 10.1159/000375367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Berson A., Nativio R., Berger S.L., Bonini N.M. Epigenetic Regulation in Neurodegenerative Diseases. Trends Neurosci. 2018;41:587–598. doi: 10.1016/j.tins.2018.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Xiao X., Liu X., Jiao B. Epigenetics: Recent Advances and Its Role in the Treatment of Alzheimer’s Disease. Front. Neurol. 2020;11:538301. doi: 10.3389/fneur.2020.538301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.An S., Khanna K.K., Wu J.M. mRNA levels and methylation patterns of the 2-5A synthetase gene in control and Alzheimer’s disease (AD) fibroblasts. Biochem. Mol. Biol. Int. 1994;33:835–840. [PubMed] [Google Scholar]
- 103.JingYun Y., Chibnik L.B., Srivastava G.P., Pochet N., Yang J., Xu J., Kozubek J., Obholzer N., Leurgans S.E., Schneider J.A., et al. Association of Brain DNA Methylation in SORL1, ABCA7, HLA-DRB5, SLC24A4, and BIN1 With Pathological Diagnosis of Alzheimer Disease. JAMA Neurol. 2015;72:15–24. doi: 10.1001/jamaneurol.2014.3049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ma S.L., Tang N.L.S., Lam L.C.W. Association of gene expression and methylation of UQCRC1 to the predisposition of Alzheimer’s disease in a Chinese population. J. Psychiatr. Res. 2016;76:143–147. doi: 10.1016/j.jpsychires.2016.02.010. [DOI] [PubMed] [Google Scholar]
- 105.Celarain N., De Gordoa J.S.-R., Zelaya M.V., Roldán M., Larumbe R., Pulido L., Echavarri C., Mendioroz M. TREM2 upregulation correlates with 5-hydroxymethycytosine enrichment in Alzheimer’s disease hippocampus. Clin. Epigenet. 2016;8:37. doi: 10.1186/s13148-016-0202-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Xu C., Liu G., Ji H., Chen W., Dai D., Chen Z., Zhou D., Xu L., Hu H., Cui W., et al. Elevated methylation of OPRM1 and OPRL1 genes in Alzheimer’s disease. Mol. Med. Rep. 2018;18:4297–4302. doi: 10.3892/mmr.2018.9424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Mercorio R., Pergoli L., Galimberti D., Favero C., Carugno M., Valle E.D., Barretta F., Cortini F., Scarpini E., Valentina V.B., et al. PICALM Gene Methylation in Blood of Alzheimer’s Disease Patients Is Associated with Cognitive Decline. J. Alzheimer’s Dis. 2018;65:283–292. doi: 10.3233/JAD-180242. [DOI] [PubMed] [Google Scholar]
- 108.Smith A.R., Smith R.G., Burrage J., Troakes C., Al-Sarraj S., Kalaria R.N., Sloan C., Robinson A., Mill J., Lunnon K. A cross-brain regions study of ANK1 DNA methylation in different neurodegenerative diseases. Neurobiol. Aging. 2018;74:70–76. doi: 10.1016/j.neurobiolaging.2018.09.024. [DOI] [PubMed] [Google Scholar]
- 109.Blanco-Luquin I., Altuna M., De Gordoa J.S.-R., Casado A.U., Roldán M., Cámara M., Zelaya V., Erro M.E., Echavarri C., Mendioroz M. PLD3 epigenetic changes in the hippocampus of Alzheimer’s disease. Clin. Epigenet. 2018;10:116. doi: 10.1186/s13148-018-0547-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Smith R.G., Hannon E., De Jager P.L., Chibnik L., Lott S.J., Condliffe D., Smith A.R., Haroutunian V., Troakes C., Al-Sarraj S., et al. Elevated DNA methylation across a 48-kb region spanning the HOXA gene cluster is associated with Alzheimer’s disease neuropathology. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2018;14:1580–1588. doi: 10.1016/j.jalz.2018.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Bernstein A., Lin Y., Street R.C., Lin L., Dai Q., Yu L., Bao H., Gearing M., Lah J.J., Nelson P.T., et al. 5-Hydroxymethylation-associated epigenetic modifiers of Alzheimer’s disease modulate Tau-induced neurotoxicity. Hum. Mol. Genet. 2016;25:2437–2450. doi: 10.1093/hmg/ddw109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Zhao J., Zhu Y., Yang J., Li L., Wu H., De Jager P.L., Jin P., Bennett D.A. A genome-wide profiling of brain DNA hydroxymethylation in Alzheimer’s disease. Alzheimer’s Dement. 2017;13:674–688. doi: 10.1016/j.jalz.2016.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Blanch M., Mosquera J.L., Ansoleaga B., Ferrer I., Barrachina M. Altered Mitochondrial DNA Methylation Pattern in Alzheimer Disease–Related Pathology and in Parkinson Disease. Am. J. Pathol. 2016;186:385–397. doi: 10.1016/j.ajpath.2015.10.004. [DOI] [PubMed] [Google Scholar]
- 114.Miranda-Morales E., Meier K., Carrillo A.S., Pacheco J.S., Vazquez P., Arias-Carrión O. Implications of DNA Methylation in Parkinson’s Disease. Front. Mol. Neurosci. 2017;10:225. doi: 10.3389/fnmol.2017.00225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Mishra A., Mishra P.S., Bandopadhyay R., Khurana N., Angelopoulou E., Paudel Y.N., Piperi C. Neuroprotective Potential of Chrysin: Mechanistic Insights and Therapeutic Potential for Neurological Disorders. Molecules. 2021;26:6456. doi: 10.3390/molecules26216456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Angelopoulou E., Paudel Y.N., Piperi C., Mishra A. Neuroprotective potential of cinnamon and its metabolites in Parkinson’s disease: Mechanistic insights, limitations, and novel therapeutic opportunities. J. Biochem. Mol. Toxicol. 2021;35:e22711. doi: 10.1002/jbt.22720. [DOI] [PubMed] [Google Scholar]
- 117.Singleton A.B., Farrer M., Johnson J., Singleton A., Hague S., Kachergus J., Hulihan M., Peuralinna T., Dutra A., Nussbaum R., et al. alpha-Synuclein Locus Triplication Causes Parkinson’s Disease. Science. 2003;302:841. doi: 10.1126/science.1090278. [DOI] [PubMed] [Google Scholar]
- 118.Chartier-Harlin M.-C., Kachergus J., Roumier C., Mouroux V., Douay X., Lincoln S., Levecque C., Larvor L., Andrieux J., Hulihan M., et al. α-synuclein locus duplication as a cause of familial Parkinson’s disease. Lancet. 2004;364:1167–1169. doi: 10.1016/S0140-6736(04)17103-1. [DOI] [PubMed] [Google Scholar]
- 119.Matsumoto L., Takuma H., Tamaoka A., Kurisaki H., Date H., Tsuji S., Iwata A. CpG Demethylation Enhances Alpha-Synuclein Expression and Affects the Pathogenesis of Parkinson’s Disease. PLoS ONE. 2010;5:e15522. doi: 10.1371/journal.pone.0015522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Guhathakurta S., Evangelista B.A., Ghosh S., Basu S., Kim Y.S. Hypomethylation of intron1 of α-synuclein gene does not correlate with Parkinson’s disease. Mol. Brain. 2017;10:6. doi: 10.1186/s13041-017-0285-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.De Boni L., Tierling S., Roeber S., Walter J., Giese A., Kretzschmar H.A. Next-Generation Sequencing Reveals Regional Differences of the α-Synuclein Methylation State Independent of Lewy Body Disease. Neuromol. Med. 2011;13:310–320. doi: 10.1007/s12017-011-8163-9. [DOI] [PubMed] [Google Scholar]
- 122.Funahashi Y., Yoshino Y., Yamazaki K., Mori Y., Mori T., Ozaki Y., Sao T., Ochi S., Iga J.-I., Ueno S.-I. DNA methylation changes atSNCAintron 1 in patients with dementia with Lewy bodies. Psychiatry Clin. Neurosci. 2016;71:28–35. doi: 10.1111/pcn.12462. [DOI] [PubMed] [Google Scholar]
- 123.Yoshino Y., Mori T., Yoshida T., Yamazaki K., Ozaki Y., Sao T., Funahashi Y., Iga J.-I., Ueno S.-I. Elevated mRNA Expression and Low Methylation of SNCA in Japanese Alzheimer’s Disease Subjects. J. Alzheimer’s Dis. 2016;54:1349–1357. doi: 10.3233/JAD-160430. [DOI] [PubMed] [Google Scholar]
- 124.Park J.-Y., Lansbury J.P.T. β-Synuclein Inhibits Formation of α-Synuclein Protofibrils: A Possible Therapeutic Strategy against Parkinson’s Disease. Biochemistry. 2003;42:3696–3700. doi: 10.1021/bi020604a. [DOI] [PubMed] [Google Scholar]
- 125.Beyer K., Domingo-Sàbat M., Santos C., Tolosa E., Ferrer I., Ariza A. The decrease of β-synuclein in cortical brain areas defines a molecular subgroup of dementia with Lewy bodies. Brain. 2010;133:3724–3733. doi: 10.1093/brain/awq275. [DOI] [PubMed] [Google Scholar]
- 126.Jang B., Ishigami A., Maruyama N., Carp R.I., Kim Y.-S., Choi E.-K. Peptidylarginine deiminase and protein citrullination in prion diseases. Prion. 2013;7:42–46. doi: 10.4161/pri.22380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Barrachina M., Ferrer I. DNA Methylation of Alzheimer Disease and Tauopathy-Related Genes in Postmortem Brain. J. Neuropathol. Exp. Neurol. 2009;68:880–891. doi: 10.1097/NEN.0b013e3181af2e46. [DOI] [PubMed] [Google Scholar]
- 128.Behrens M.I., Brüggemann N., Chana P., Venegas P., Kägi M., Parrao T., Orellana P., Garrido C., Rojas C.V., Hauke J., et al. Clinical spectrum of Kufor-Rakeb syndrome in the Chilean kindred with ATP13A2 mutations. Mov. Disord. 2010;25:1929–1937. doi: 10.1002/mds.22996. [DOI] [PubMed] [Google Scholar]
- 129.Cai M., Tian J., Zhao G.-H., Luo W., Zhang B.-R. Study of Methylation Levels of Parkin Gene Promoter in Parkinson’s Disease Patients. Int. J. Neurosci. 2011;121:497–502. doi: 10.3109/00207454.2011.580866. [DOI] [PubMed] [Google Scholar]
- 130.Cai Y., Liu S., Sothern R.B., Xu S., Chan P. Expression of clock genesPer1andBmal1in total leukocytes in health and Parkinson’s disease. Eur. J. Neurol. 2009;17:550–554. doi: 10.1111/j.1468-1331.2009.02848.x. [DOI] [PubMed] [Google Scholar]
- 131.Lin Q., Ding H., Zheng Z., Gu Z., Ma J., Chen L., Chan P., Cai Y. Promoter methylation analysis of seven clock genes in Parkinson’s disease. Neurosci. Lett. 2012;507:147–150. doi: 10.1016/j.neulet.2011.12.007. [DOI] [PubMed] [Google Scholar]
- 132.Simón-Sánchez J., Schulte C., Bras J.M., Sharma M., Gibbs J.R., Berg D., Paisan-Ruiz C., Lichtner P., Scholz S.W., Hernandez D.G., et al. Genome-wide association study reveals genetic risk underlying Parkinson’s disease. Nat. Genet. 2009;41:1308–1312. doi: 10.1038/ng.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Bsc K.G.C., Mellick G., Silburn P.A., Mather K., Armstrong N.J., Sachdev P., Brodaty H., Huang Y., Halliday G., Hallupp M., et al. DNA methylation of theMAPTgene in Parkinson’s disease cohorts and modulation by vitamin EIn Vitro. Mov. Disord. 2013;29:1606–1614. doi: 10.1002/mds.25784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Kwok J.B.J., Teber E.T., Loy C., Hallupp M., Nicholson G., Mellick G.D., Buchanan D.D., Silburn P.A., Schofield P.R. Tau haplotypes regulate transcription and are associated with Parkinson’s disease. Ann. Neurol. 2004;55:329–334. doi: 10.1002/ana.10826. [DOI] [PubMed] [Google Scholar]
- 135.Tsunemi T., La Spada A.R. PGC-1α at the intersection of bioenergetics regulation and neuron function: From Huntington’s disease to Parkinson’s disease and beyond. Prog. Neurobiol. 2012;97:142–151. doi: 10.1016/j.pneurobio.2011.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Tan Y., Wu L., Li D., Liu X., Ding J., Chen S. Methylation status of DJ-1 in leukocyte DNA of Parkinson’s disease patients. Transl. Neurodegener. 2016;5:5. doi: 10.1186/s40035-016-0052-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Moore K., McKnight A.J., Craig D., O’Neill F.A. Epigenome-Wide Association Study for Parkinson’s Disease. Neuromol. Med. 2014;16:845–855. doi: 10.1007/s12017-014-8332-8. [DOI] [PubMed] [Google Scholar]
- 138.Wüllner U., Kaut O., Deboni L., Piston D., Schmitt I. DNA methylation in Parkinson’s disease. J. Neurochem. 2016;139:108–120. doi: 10.1111/jnc.13646. [DOI] [PubMed] [Google Scholar]
- 139.Madrid A., Hogan K.J., Papale L.A., Clark L.R., Asthana S., Johnson S.C., Alisch R.S. DNA Hypomethylation in Blood Links B3GALT4 and ZADH2 to Alzheimer’s Disease. J. Alzheimer’s Dis. 2018;66:927–934. doi: 10.3233/JAD-180592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Hernández H.G., Sandoval-Hernández A.G., Garrido-Gil P., Labandeira-Garcia J.L., Zelaya M.V., Bayon G.F., Fernández A.F., Fraga M.F., Arboleda G., Arboleda H. Alzheimer’s disease DNA methylome of pyramidal layers in frontal cortex: Laser-assisted microdissection study. Epigenomics. 2018;10:1365–1382. doi: 10.2217/epi-2017-0160. [DOI] [PubMed] [Google Scholar]
- 141.Andrés-Benito P., Delgado-Morales R., Ferrer I. Altered Regulation of KIAA0566, and Katanin Signaling Expression in the Locus Coeruleus With Neurofibrillary Tangle Pathology. Front. Cell. Neurosci. 2018;12:131. doi: 10.3389/fncel.2018.00131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Miao L., Yin R.-X., Zhang Q.-H., Hu X.-J., Huang F., Chen W.-X., Cao X.-L., Wu J.-Z. Integrated DNA methylation and gene expression analysis in the pathogenesis of coronary artery disease. Aging. 2019;11:1486–1500. doi: 10.18632/aging.101847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Smith A., Smith R.G., Pishva E., Hannon E., Roubroeks J.A.Y., Burrage J., Troakes C., Al-Sarraj S., Sloan C., Mill J., et al. Parallel profiling of DNA methylation and hydroxymethylation highlights neuropathology-associated epigenetic variation in Alzheimer’s disease. Clin. Epigenet. 2019;11:52. doi: 10.1186/s13148-019-0636-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Li P., Marshall L., Oh G., Jakubowski J.L., Groot D., He Y., Wang T., Petronis A., Labrie V. Epigenetic dysregulation of enhancers in neurons is associated with Alzheimer’s disease pathology and cognitive symptoms. Nat. Commun. 2019;10:2246. doi: 10.1038/s41467-019-10101-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Chouliaras L., Mastroeni D., Delvaux E., Grover A., Kenis G., Hof P.R., Steinbusch H.W., Coleman P.D., Rutten B., Hove D.L.V.D. Consistent decrease in global DNA methylation and hydroxymethylation in the hippocampus of Alzheimer’s disease patients. Neurobiol. Aging. 2013;34:2091–2099. doi: 10.1016/j.neurobiolaging.2013.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Bradley-Whitman M., Lovell M. Epigenetic changes in the progression of Alzheimer’s disease. Mech. Ageing Dev. 2013;134:486–495. doi: 10.1016/j.mad.2013.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Coppieters N., Dieriks B., Lill C., Faull R., Curtis M., Dragunow M. Global changes in DNA methylation and hydroxymethylation in Alzheimer’s disease human brain. Neurobiol. Aging. 2014;35:1334–1344. doi: 10.1016/j.neurobiolaging.2013.11.031. [DOI] [PubMed] [Google Scholar]
- 148.Pishva E., Creese B., Smith A.R., Viechtbauer W., Proitsi P., Hove D.L.V.D., Ballard C., Mill J., Lunnon K. Psychosis-associated DNA methylomic variation in Alzheimer’s disease cortex. Neurobiol. Aging. 2020;89:83–88. doi: 10.1016/j.neurobiolaging.2020.01.001. [DOI] [PubMed] [Google Scholar]
- 149.Phipps A.J., Vickers J.C., Taberlay P.C., Woodhouse A. Neurofilament-labeled pyramidal neurons and astrocytes are deficient in DNA methylation marks in Alzheimer’s disease. Neurobiol. Aging. 2016;45:30–42. doi: 10.1016/j.neurobiolaging.2016.05.003. [DOI] [PubMed] [Google Scholar]
- 150.Henry C.J., Huang Y., Wynne A.M., Godbout J.P. Peripheral lipopolysaccharide (LPS) challenge promotes microglial hyperactivity in aged mice that is associated with exaggerated induction of both pro-inflammatory IL-1β and anti-inflammatory IL-10 cytokines. Brain Behav. Immun. 2009;23:309–317. doi: 10.1016/j.bbi.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Jowaed A., Schmitt I., Kaut O., Wüllner U. Methylation Regulates Alpha-Synuclein Expression and Is Decreased in Parkinson’s Disease Patients’ Brains. J. Neurosci. 2010;30:6355–6359. doi: 10.1523/JNEUROSCI.6119-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Su X., Chu Y., Kordower J.H., Li B., Cao H., Huang L., Nishida M., Song L., Wang D., Federoff H.J. PGC−1α Promoter Methylation in Parkinson’s Disease. PLoS ONE. 2015;10:e0134087. doi: 10.1371/journal.pone.0134087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Pieper H.C., Evert B.O., Kaut O., Riederer P.F., Waha A., Wüllner U. Different methylation of the TNF-alpha promoter in cortex and substantia nigra: Implications for selective neuronal vulnerability. Neurobiol. Dis. 2008;32:521–527. doi: 10.1016/j.nbd.2008.09.010. [DOI] [PubMed] [Google Scholar]
- 154.Nielsen S.S., Checkoway H., Criswell S.R., Farin F.M., Stapleton P.L., Sheppard L., Racette B.A. Inducible nitric oxide synthase gene methylation and parkinsonism in manganese-exposed welders. Park. Relat. Disord. 2015;21:355–360. doi: 10.1016/j.parkreldis.2015.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Lee J., Hwang Y.J., Kim K.Y., Kowall N.W., Ryu H. Epigenetic Mechanisms of Neurodegeneration in Huntington’s Disease. Neurotherapeutics. 2013;10:664–676. doi: 10.1007/s13311-013-0206-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Thomas E.A. DNA methylation in Huntington’s disease: Implications for transgenerational effects. Neurosci. Lett. 2015;625:34–39. doi: 10.1016/j.neulet.2015.10.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Sugars K.L., Rubinsztein D.C. Transcriptional abnormalities in Huntington disease. Trends Genet. 2003;19:233–238. doi: 10.1016/S0168-9525(03)00074-X. [DOI] [PubMed] [Google Scholar]
- 158.Stack E.C., Del Signore S.J., Luthi-Carter R., Soh B.Y., Goldstein D.R., Matson S., Goodrich S., Markey A.L., Cormier K., Hagerty S.W., et al. Modulation of nucleosome dynamics in Huntington’s disease. Hum. Mol. Genet. 2007;16:1164–1175. doi: 10.1093/hmg/ddm064. [DOI] [PubMed] [Google Scholar]
- 159.Wang F., Yang Y., Lin X., Wang J.-Q., Wu Y.-S., Xie W., Wang D., Zhu S., Liao Y.-Q., Sun Q., et al. Genome-wide loss of 5-hmC is a novel epigenetic feature of Huntington’s disease. Hum. Mol. Genet. 2013;22:3641–3653. doi: 10.1093/hmg/ddt214. [DOI] [PubMed] [Google Scholar]
- 160.Bai G., Cheung I., Shulha H.P., Coelho J., Li P., Dong X., Jakovcevski M., Wang Y., Grigorenko A., Jiang Y., et al. Epigenetic dysregulation of hairy and enhancer of split 4 (HES4) is associated with striatal degeneration in postmortem Huntington brains. Hum. Mol. Genet. 2014;24:1441–1456. doi: 10.1093/hmg/ddu561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Villar-Menéndez I., Blanch M., Tyebji S., Pereira-Veiga T., Albasanz J.L., Martín M., Ferrer I., Perez-Navarro E., Barrachina M. Increased 5-Methylcytosine and Decreased 5-Hydroxymethylcytosine Levels are Associated with Reduced Striatal A2AR Levels in Huntington’s Disease. Neuromol. Med. 2013;15:295–309. doi: 10.1007/s12017-013-8219-0. [DOI] [PubMed] [Google Scholar]
- 162.Tang B., Seredenina T., Coppola G., Kuhn A., Geschwind D.H., Luthi-Carter R., Thomas E.A. Gene expression profiling of R6/2 transgenic mice with different CAG repeat lengths reveals genes associated with disease onset and progression in Huntington’s disease. Neurobiol. Dis. 2011;42:459–467. doi: 10.1016/j.nbd.2011.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Zadel M., Maver A., Kovanda A., Peterlin B. DNA Methylation Profiles in Whole Blood of Huntington’s Disease Patients. Front. Neurol. 2018;9:655. doi: 10.3389/fneur.2018.00655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Figueroa-Romero C., Hur J., Bender D.E., Delaney C.E., Cataldo M.D., Smith A.L., Yung R., Ruden D.M., Callaghan B.C., Feldman E.L. Identification of Epigenetically Altered Genes in Sporadic Amyotrophic Lateral Sclerosis. PLoS ONE. 2012;7:e52672. doi: 10.1371/journal.pone.0052672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Chestnut B.A., Chang Q., Price A., Lesuisse C., Wong M., Martin L.J. Epigenetic Regulation of Motor Neuron Cell Death through DNA Methylation. J. Neurosci. 2011;31:16619–16636. doi: 10.1523/JNEUROSCI.1639-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Chen H., Dzitoyeva S., Manev H. Effect of aging on 5-hydroxymethylcytosine in the mouse hippocampus. Restor. Neurol. Neurosci. 2012;30:237–245. doi: 10.3233/RNN-2012-110223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Eklow C., Makrygiannakis D., Backdahl L., Padyukov L., Ulfgren A.-K., Lorentzen J.C., Malmstrom V. Cellular distribution of the C-type II lectin dendritic cell immunoreceptor (DCIR) and its expression in the rheumatic joint: Identification of a subpopulation of DCIR+ T cells. Ann. Rheum. Dis. 2008;67:1742–1749. doi: 10.1136/ard.2007.076976. [DOI] [PubMed] [Google Scholar]
- 168.Piccio L., Buonsanti C., Mariani M., Cella M., Gilfillan S., Cross A.H., Colonna M., Panina-Bordignon P. Blockade of TREM-2 exacerbates experimental autoimmune encephalomyelitis. Eur. J. Immunol. 2007;37:1290–1301. doi: 10.1002/eji.200636837. [DOI] [PubMed] [Google Scholar]
- 169.Ramji D., Vitelli A., Tronche F., Cortese R., Ciliberto G. The two C/EBP isoforms, IL6DBP/NFIL6 and CEBP6δ/NFIL63, are induced by IL6β to promote acute phase gene transcription via different mechanisms. Nucleic Acids Res. 1993;21:289–294. doi: 10.1093/nar/21.2.289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Bacon C., Endris V., Rappold G. Dynamic expression of the Slit-Robo GTPase activating protein genes during development of the murine nervous system. J. Comp. Neurol. 2009;513:224–236. doi: 10.1002/cne.21955. [DOI] [PubMed] [Google Scholar]
- 171.Pellikka M., Tanentzapf G., Pinto M., Smith C.T., McGlade C.J., Ready D.F., Tepass U. Crumbs, the Drosophila homologue of human CRB1/RP12, is essential for photoreceptor morphogenesis. Nature. 2002;416:143–149. doi: 10.1038/nature721. [DOI] [PubMed] [Google Scholar]
- 172.Inoue K.-I., Shiga T., Ito Y. Runx transcription factors in neuronal development. Neural Dev. 2008;3:20. doi: 10.1186/1749-8104-3-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Stoccoro A., Mosca L., Carnicelli V., Cavallari U., Lunetta C., Marocchi A., Migliore L., Coppedè F. Mitochondrial DNA copy number and D-loop region methylation in carriers of amyotrophic lateral sclerosis gene mutations. Epigenomics. 2018;10:1431–1443. doi: 10.2217/epi-2018-0072. [DOI] [PubMed] [Google Scholar]
- 174.Teijido O., Cacabelos R. Pharmacoepigenomic Interventions as Novel Potential Treatments for Alzheimer’s and Parkinson’s Diseases. Int. J. Mol. Sci. 2018;19:3199. doi: 10.3390/ijms19103199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Wang Y., Wang X., Li R., Yang Z.-F., Wang Y.-Z., Gong X.-L., Wang X.-M. A DNA Methyltransferase Inhibitor, 5-Aza-2′-Deoxycytidine, Exacerbates Neurotoxicity and Upregulates Parkinson’s Disease-Related Genes in Dopaminergic Neurons. CNS Neurosci. Ther. 2013;19:183–190. doi: 10.1111/cns.12059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Pan Y., Daito T., Sasaki Y., Chung Y.H., Xing X., Pondugula S., Swamidass S.J., Wang T., Kim A.H., Yano H. Inhibition of DNA Methyltransferases Blocks Mutant Huntingtin-Induced Neurotoxicity. Sci. Rep. 2016;6:31022. doi: 10.1038/srep31022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Martin L.J., Wong M. Aberrant Regulation of DNA Methylation in Amyotrophic Lateral Sclerosis: A New Target of Disease Mechanisms. Neurotherapeutics. 2013;10:722–733. doi: 10.1007/s13311-013-0205-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Fernández-Santiago R., Ezquerra M. Epigenetic Research of Neurodegenerative Disorders Using Patient iPSC-Based Models. Stem Cells Int. 2015;2016:9464591. doi: 10.1155/2016/9464591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Salameh Y., Bejaoui Y., El Hajj N. DNA Methylation Biomarkers in Aging and Age-Related Diseases. Front. Genet. 2020;11:171. doi: 10.3389/fgene.2020.00171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Delgado-Morales R., Esteller M. Opening up the DNA methylome of dementia. Mol. Psychiatry. 2017;22:485–496. doi: 10.1038/mp.2016.242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Armstrong M.J., Jin Y., Allen E.G., Jin P. Diverse and dynamic DNA modifications in brain and diseases. Hum. Mol. Genet. 2019;28:R241–R253. doi: 10.1093/hmg/ddz179. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing does not apply to this article.