Abstract
Zizyphus lotus L. is a perennial shrub particularly used in Algerian folk medicine, but little is known concerning the lipophilic compounds in the most frequently used parts, namely, root bark, pulp, leaves and seeds, which are associated with health benefits. In this vein, the lipophilic fractions of these morphological parts of Z. lotus from Morocco were studied by gas chromatography–mass spectrometry (GC–MS), and their antiproliferative and antimicrobial activities were evaluated. GC–MS analysis allowed the identification and quantification of 99 lipophilic compounds, including fatty acids, long-chain aliphatic alcohols, pentacyclic triterpenic compounds, sterols, monoglycerides, aromatic compounds and other minor components. Lipophilic extracts of pulp, leaves and seeds were revealed to be mainly composed of fatty acids, representing 54.3–88.6% of the total compounds detected. The leaves and seeds were particularly rich in unsaturated fatty acids, namely, (9Z,12Z)-octadeca-9,12-dienoic acid (2431 mg kg−1 of dry weight) and (9Z)-octadec-9-enoic acid (6255 mg kg−1 of dry weight). In contrast, root bark contained a high content of pentacyclic triterpenic compounds, particularly betulinic acid, accounting for 9838 mg kg−1 of dry weight. Root bark extract showed promising antiproliferative activity against a triple-negative breast cancer cell line, MDA-MB-231, with a half-maximal inhibitory concentration (IC50) = 4.23 ± 0.18 µg mL−1 of extract. Leaf extract displayed interesting antimicrobial activity against Escherichia coli, methicillin-sensitive Staphylococcus aureus and Staphylococcus epidermis, presenting minimum inhibitory concentration (MIC) values from 1024 to 2048 µg mL−1 of extract. Our results demonstrate that Zizyphus lotus L. is a source of promising bioactive components, which can be exploited as natural ingredients in pharmaceutical formulations.
Keywords: Zizyphus lotus L., lipophilic extracts, gas chromatography–mass spectrometry, pentacyclic triterpenic compounds, antimicrobial activity, triple-negative breast cancer
1. Introduction
Zizyphus, a plant genus belonging to the angiosperm Rhamnaceae family, order Rhamnales, includes about 135–170 species worldwide [1], of which Zizyphus jujuba Mill. and Ziziphus mauritiana Lam. are the most important in terms of distribution and economic significance [2]. Zizyphus lotus (Z. lotus), also known as “Sedra”, is indigenous to Morocco and has a wide ecological and geographical distribution in arid and semiarid plateau regions and along sandy riverbeds in the Saharan region [3], whereas, in Europe, this plant is restricted to certain semiarid areas, namely, in the southeast of Spain and in Sicily, Italy [4]. This wild shrub is mainly appreciated for its brown, flavorful and nutritive small fruits, which have contributed to the spread of these species. Moreover, Z. lotus, particularly its root bark, seeds, fruit pulp and leaves, have been associated with a wide range of health benefits, including in traditional medicine for the treatment of a variety of diseases and disorders, such as liver and urinary complaints, diabetes, skin infections, insomnia, inflammation and peptic ulcers, among others [4,5,6].
An increasing number of studies have identified a variety of bioactive secondary metabolites in Z. lotus, particularly phenolic compounds, which have been verified to be present in leaves, branches, and root and stem barks [7,8]. In addition, a vast number of biological properties have been attributed to Z. lotus polar extracts from different morphological parts, namely, antioxidant [8,9,10], antibacterial [8], anti-ulcerogenic [10], anti-inflammatory [5,8], analgesic [5], antidiabetic [9] and antispasmodic [11] activities. The less polar fractions of this shrub, however, remain poorly exploited and are most often limited to fatty acid composition [4,6,12,13,14], with a few other compounds, such as seven cyclopeptide alkaloids termed lotusines [15,16,17], four dammarane saponins [18] and one pentacyclic triterpenic compound [19], being previously isolated from leaves, root bark or pulp. However, other interesting families of natural compounds (from a biological activity point of view), such as sterols or tocopherols, have only been evaluated in Z. lotus seed oils [12,20] or whole fruit [14] and, in some cases, without quantification [14].
A few biological activities have also been exploited in lipophilic extracts of Z. lotus, including antispasmodic [21], anti-ulcerogenic [22], anti-inflammatory and analgesic activities [5]. In this vein, there is a lack of information about the different families of lipophilic compounds present in the different morphological parts of Z. lotus, including long-chain aliphatic alcohols, monoglycerides or terpenes. Triterpenic acids have been identified in the genus Zizyphus [23,24], but they have not yet been exploited in this shrub species despite their valuable properties, such as antitumor and antiangiogenic activities [25]. The objective of this work is to characterize the lipophilic composition of the morphological parts of Z. lotus that are commonly associated with promising biological activities and health benefits, namely, root bark, leaves, seeds and pulp, using gas chromatography–mass spectrometry (GC–MS). The different families of compounds present in these fractions, namely, fatty acids, long-chain aliphatic alcohols, sterols and triterpenic compounds, are qualitatively and quantitatively evaluated here for the first time. In addition, their antiproliferative activity against a triple-negative breast cancer cell line (MDA-MB-231 cells) and their antibacterial activity against Escherichia coli, methicillin-sensitive Staphylococcus aureus (MSSA) and Staphylococcus epidermidis are also evaluated. The characterization and profile comparison of these fractions between morphological parts of Z. lotus, together with the evaluation of their respective biological activities, may foster the development of strategies to exploit this shrub for pharmaceutical applications.
2. Results and Discussion
2.1. Extraction Yield
Dichloromethane (DCM) extracts from the four morphological parts of Z. lotus presented very distinct contents of extractives, with seeds showing the highest yield (9.4%), followed by leaves (4.1%), root bark (2.0%) and pulp (1.7%). A similar DCM extraction yield (1.6%) was reported before for Z. lotus pulp but was preceded by petroleum ether extraction [26]. DCM lipophilic extractive yields from leaves, seeds and root bark are described herein for the first time.
2.2. Lipophilic Composition
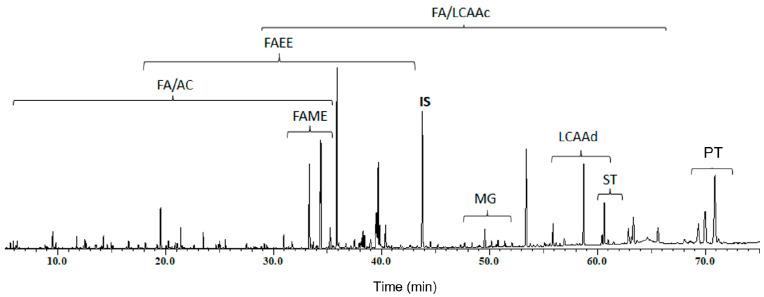
The lipophilic fractions of four morphological parts of Z. lotus, i.e., root bark, leaves, pulp and seeds obtained by DCM Soxhlet extraction, were characterized in detail using GC–MS analysis. The extract composition is shown in Table 1. As an example, the GC–MS chromatogram of the derivatized DCM extract of pulp is presented in Figure 1.
Table 1.
Quantitative analysis (mg kg−1 of dry weight) of lipophilic extracts derived from four morphological parts of Zizyphus lotus L.
| RT (min) | Compound | Pulp | Seeds | Leaves | Root Bark |
|---|---|---|---|---|---|
| Fatty acids | 1469 | 8512 | 4643 | 434 | |
| Saturated fatty acids | 799 | 1260 | 1461 | 265 | |
| Decanoic acid | 19.50 | 67 | 5 | 5 | 1 |
| Undecanoic acid | 22.58 | 9 | n.d. | n.d. | n.d. |
| Dodecanoic acid | 25.49 | 17 | 2 | 4 | 2 |
| Tridecanoic acid | 28.25 | 4 | n.d. | n.d. | n.d. |
| Tetradecanoic acid | 30.89 | 28 | 10 | 52 | n.d. |
| Pentadecanoic acid | 33.40 | 9 | 4 | 5 | 2 |
| Hexadecanoic acid | 35.82 | 366 | 594 | 877 | 152 |
| Heptadecanoic acid | 38.10 | 22 | 7 | 8 | 7 |
| Octadecanoic acid | 40.32 | 42 | 570 | 276 | 49 |
| Nonadecanoic acid | 42.43 | 2 | n.d. | n.d. | 1 |
| Eicosanoic acid | 44.48 | 16 | 48 | 40 | 7 |
| Heneicosanoic acid | 46.48 | 5 | n.d. | 3 | 3 |
| Docosanoic acid | 48.34 | 10 | 19 | 25 | 13 |
| Tricosanoic acid | 50.16 | n.d. | n.d. | n.d. | 9 |
| Tetracosanoic acid | 52.04 | 10 | n.d. | 26 | 14 |
| Pentacosanoic acid | 54.02 | 3 | n.d. | 9 | 5 |
| Hexacosanoic acid | 56.10 | 10 | n.d. | 52 | n.d. |
| Heptacosanoic acid | 58.30 | 11 | n.d. | n.d. | n.d. |
| Octacosanoic acid | 60.56 | 114 | n.d. | 79 | n.d. |
| Triacontanoic acid | 65.51 | 54 | n.d. | tr | n.d. |
| Unsaturated fatty acids | 421 | 7222 | 3175 | 159 | |
| Tetradecenoic acid | 30.19 | 2 | n.d. | n.d. | n.d. |
| Hexadecenoic acid isomer a | 35.05 | 4 | 10 | 4 | 2 |
| Hexadecenoic acid isomer b | 35.19 | 40 | 11 | 23 | 2 |
| Hexadecenoic acid isomer c | 35.44 | 7 | n.d. | n.d. | 2 |
| Heptadecenoic acid isomer a | 37.44 | 24 | 7 | n.d. | n.d. |
| Heptadecenoic acid isomer b | 37.52 | n.d. | n.d. | n.d. | 1 |
| Heptadecenoic acid isomer c | 37.60 | n.d. | n.d. | n.d. | 1 |
| (9Z,12Z)-Octadeca-9,12-dienoic acid | 39.42 | 60 | 737 | 544 | 50 |
| (9Z,12Z,15Z)-Octadeca-9,12,15-trienoic acid | 39.50 | 45 | n.d. | 2431 | 9 |
| (9Z)-Octadec-9-enoic acid | 39.62 | 179 | 6255 | 120 | 59 |
| (9E)-Octadec-9-enoic acid | 39.78 | 50 | 135 | 54 | 18 |
| Nonadecenoic acid | 41.72 | 7 | n.d. | n.d. | n.d. |
| Eicos-11-enoic acid | 43.83 | 2 | 66 | <0.5 | 14 |
| Diacids | 7 | n.d. | n.d. | n.d. | |
| Hexadecanedioic acid | 45.19 | 7 | n.d. | n.d. | n.d. |
| ω-Hydroxy fatty acids | 10 | n.d. | 7 | 9 | |
| 22-Hydroxydocosanoic acid | 55.04 | 5 | n.d. | 7 | 9 |
| 2-Hydroxytetracosanoic acid | 55.24 | 5 | n.d. | n.d. | n.d. |
| Fatty acid ethyl esters | 228 | 16 | n.d. | n.d. | |
| Ethyl decanoate | 17.08 | 1 | n.d. | n.d. | n.d. |
| Ethyl tetradecanoate | 29.11 | 11 | n.d. | n.d. | n.d. |
| Ethyl pentadecanoate | 31.74 | 4 | n.d. | n.d. | n.d. |
| Ethyl hexadec-9-enoate isomer a | 33.62 | 12 | n.d. | n.d. | n.d. |
| Ethyl hexadec-9-enoate isomer b | 33.86 | 3 | n.d. | n.d. | n.d. |
| Ethyl hexadecanoate | 34.26 | 104 | 5 | n.d. | n.d. |
| Ethyl (9Z)-octadec-9-enoate | 38.24 | 33 | 11 | n.d. | n.d. |
| Ethyl (9E)-octadec-9-enoate | 38.39 | 25 | n.d. | n.d. | n.d. |
| Ethyl octadecanoate | 38.95 | 29 | n.d. | n.d. | n.d. |
| Ethyl eicosanoate | 43.27 | 6 | n.d. | n.d. | n.d. |
| Fatty acid methyl esters | 5 | 15 | n.d. | 1 | |
| Methyl hexadecanoate | 32.53 | 5 | n.d. | n.d. | n.d. |
| Methyl (9Z)-octadec-9-enoate | 36.67 | n.d. | 15 | n.d. | 1 |
| Monoglycerides | 27 | 255 | 189 | 24 | |
| 2-Palmitoylglycerol | 47.05 | n.d. | 3 | 5 | n.d. |
| 1-Palmitoylglycerol | 47.67 | 13 | 44 | 47 | 12 |
| 1-Linoleoylglycerol | 50.61 | n.d. | 35 | 30 | 3 |
| 1-Linolenoylglycerol | 50.72 | n.d. | n.d. | 84 | n.d. |
| 1-Oleoylglycerol | 50.73 | 14 | 155 | n.d. | 4 |
| 1-Stearoylglycerol | 51.28 | n.d. | 17 | 24 | 4 |
| Long chain aliphatic alcohols | 340 | 2 | 438 | 51 | |
| Tetradecan-1-ol | 28.89 | n.d. | n.d. | n.d. | 2 |
| Hexadecan-1-ol | 33.96 | 4 | 2 | 4 | 9 |
| (9Z)-Octadec-9-en-1-ol | 37.87 | 9 | n.d. | 11 | 14 |
| Octadecan-1-ol | 38.60 | 4 | n.d. | 2 | 6 |
| Docosan-1-ol | 46.83 | 3 | n.d. | n.d. | 3 |
| Tetracosan-1-ol | 50.51 | n.d. | n.d. | 5 | 3 |
| Hexacosan-1-ol | 54.36 | 7 | n.d. | 118 | 3 |
| Heptacosan-1-ol | 56.46 | 8 | n.d. | 23 | n.d. |
| Octacosan-1-ol | 58.65 | 207 | n.d. | 230 | 11 |
| Nonacosan-1-ol | 60.91 | 19 | n.d. | 13 | n.d. |
| Triacontan-1-ol | 63.24 | 79 | n.d. | 31 | n.d. |
| Pentacyclic triterpenic compounds | 608 | 483 | 248 | 10230 | |
| Lupeol | 63.86 | n.d. | n.d. | 78 | 105 |
| Oleanolic acid | 69.21 | 103 | 164 | 51 | 287 |
| Betulinic acid | 69.82 | 160 | 238 | 119 | 9838 |
| Ursolic acid | 70.72 | 345 | 81 | n.d. | n.d. |
| Sterols | 81 | 96 | 355 | 257 | |
| Campesterol | 60.70 | n.d. | n.d. | 28 | 4 |
| Stigmasterol | 61.41 | 13 | n.d. | 119 | 126 |
| β-Sitosterol | 62.79 | 68 | 96 | 208 | 127 |
| Aromatic compounds | 29 | 31 | 21 | 11 | |
| Benzoic acid | 11.71 | 23 | 2 | 5 | n.d. |
| Vanillin | 21.00 | n.d. | 20 | n.d. | 3 |
| Salicylic acid | 21.02 | n.d. | n.d. | 4 | n.d. |
| Vanillyl alcohol | 24.99 | n.d. | 5 | n.d. | 1 |
| Syringaldehyde | 25.93 | n.d. | n.d. | n.d. | 1 |
| Homovanillyl alcohol | 27.00 | n.d. | n.d. | n.d. | 2 |
| Vanillic acid | 28.39 | 4 | 4 | n.d. | 2 |
| Hydroxytyrosol | 28.96 | n.d. | n.d. | n.d. | 2 |
| Protocatechuic acid | 30.42 | n.d. | n.d. | n.d. | <0.5 |
| Syringic acid | 31.86 | n.d. | n.d. | n.d. | 1 |
| p-Coumaric acid | 32.88 | 2 | n.d. | 5 | n.d. |
| E-Ferulic acid | 36.54 | n.d. | n.d. | 7 | n.d. |
| Others | 151 | 228 | 832 | 9 | |
| Solerol | 13.45 | 6 | n.d. | n.d. | n.d. |
| Glycerol | 14.21 | 41 | 148 | 251 | 9 |
| Loliolide | 28.26 | n.d. | n.d. | 39 | n.d. |
| Neophytadiene isomer a | 30.76 | tr | n.d. | 141 | n.d. |
| Neophytadiene isomer b | 31.29 | n.d. | n.d. | 29 | n.d. |
| Neophytadiene isomer c | 31.75 | n.d. | n.d. | 49 | n.d. |
| Inositol | 36.94 | n.d. | n.d. | 13 | n.d. |
| Phytol | 39.04 | n.d. | n.d. | 117 | n.d. |
| Squalene | 51.53 | n.d. | 79 | 39 | n.d. |
| γ-Tocopherol | 55.21 | n.d. | n.d. | 54 | n.d. |
| Tetracosyl acetate | 55.33 | n.d. | n.d. | 26 | n.d. |
| Octacosanal | 55.82 | 52 | n.d. | n.d. | n.d. |
| Nonacosan-10-one | 56.86 | 24 | n.d. | n.d. | n.d. |
| α-Tocopherol | 58.19 | n.d. | n.d. | 74 | n.d. |
| Triacontanal | 60.35 | 27 | n.d. | n.d. | n.d. |
| Total | 2704 | 9607 | 6726 | 11016 | |
Results represent the average of the concordant values obtained for six aliquots of each sample, with less than 5% variation between samples. Abbreviations: n.d. not detected; tr, traces.
Figure 1.
GC−MS chromatogram of the trimethylsilyl-derivatized dichloromethane extract from Z. lotus pulp. Abbreviations: AC, aromatic compounds; IS, internal standard (tetracosane); FA, fatty acids; FAEE, fatty acid ethyl ester; FAME, fatty acid methyl esters; LCAAc, long-chain aliphatic alcohols; LCAAd, long-chain aliphatic aldehydes; MG, monoglycerides; PT, pentacyclic triterpenic compounds; ST, sterols.
The GC–MS analysis revealed remarkable diversity of lipophilic constituents of Z. lotus extracts, allowing the identification of compounds from six families, namely, fatty acids (including fatty acid ethyl esters (FAEEs) and fatty acid methyl esters (FAMEs)), long-chain aliphatic alcohols, pentacyclic triterpenic compounds, sterols, monoglycerides and aromatic compounds, among other minor compounds.
2.2.1. Fatty Acids
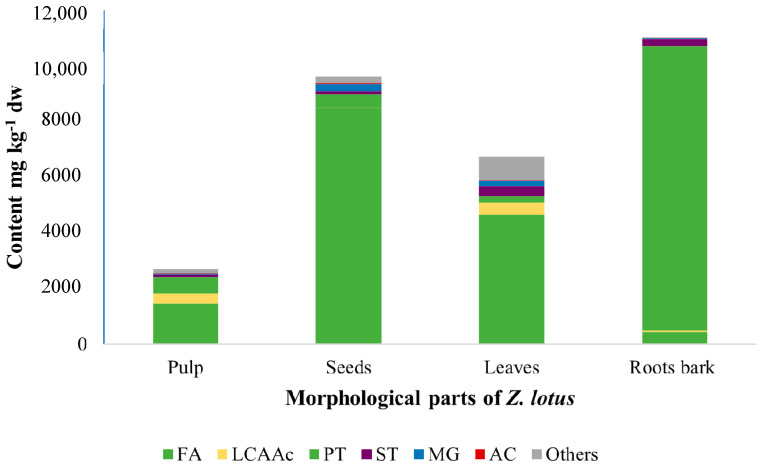
Fatty acids represented the major family of lipophilic components identified in all Z. lotus extracts, except for root bark (Table 1 and Figure 2). This family mainly includes saturated and unsaturated fatty acids, a diacid and two ω-hydroxy fatty acids that were observed in the four morphological parts of Z. lotus. To the best of our knowledge, this family is described for the first time in root bark, while some saturated and unsaturated fatty acids have been previously reported in extracts of leaves, seeds, pulp or whole fruit [4,12,13,14].
Figure 2.
Major families of lipophilic compounds identified by GC–MS in dichloromethane extracts of Z. lotus. Abbreviations: AC, aromatic compounds; FA, fatty acids; LCAAc, long-chain aliphatic alcohols; LCAAd, long-chain aliphatic aldehydes; MG, monoglycerides; PT, pentacyclic triterpenic compounds; ST, sterols.
A wide diversity of saturated fatty acids (SFAs) (C10–C30) was identified in this shrub species, with hexadecanoic acid (palmitic acid) as the most abundant SFA in all Z. lotus extracts, ranging from 152 mg kg−1 in root bark to 877 mg kg−1 dry weight (dw) in leaves. Significant amounts of octadecanoic acid were also observed in leaves (276 mg kg−1 dw) and seeds (570 mg kg−1 dw). Other SFAs were detected in the studied extracts in lower amounts, seven of which (C21, C23, C25–C28 and C30) were identified for the first time in the present work as Z. lotus L. constituents [4,6,12,13,14]. Additionally, to the best of our knowledge, four SFAs in some of the studied morphological parts of Z. lotus L. are reported herein for the first time, namely, eicosanoic and docosanoic acids in pulp and leaves and nonadecanoic and tetracosanoic acids in pulp [4,6,12,13].
Unsaturated fatty acids (UFAs) accounted for 28.7–84.8% of the total identified fatty acids of Z. lotus, with (9Z)-octadec-9-enoic acid (oleic acid) being the most abundant UFA in Z. lotus pulp, root bark and seeds (59–6255 mg kg−1 dw). The abundance of UFAs observed in the leaves is mainly due to the presence of (9Z,12Z)-octadeca-9,12-dienoic (linoleic acid; ω-6) and (9Z,12Z,15Z)-octadeca-9,12,15-trienoic (linolenic acid; ω-3) acids, which accounted for 544 and 2431 mg kg−1, respectively. In fact, linolenic acid was the major lipophilic compound detected in leaves, corresponding to 36.1% of all identified compounds in this fraction. Regarding Z. lotus pulp, to the best of our knowledge, six of the UFAs detected in this study are novel components of this fraction, namely, tetradecenoic acid, three heptadecenoic acid isomers, (9Z,12Z,15Z)-octadecatri-9,12,15-enoic and eicos-11-enoic acids [4,12,13,14]. Hexadecenoic and heptadecenoic acids were found to be present in three positional isomer forms each; however, their exact configurations were not possible to determine.
Omega-3 and ω-6 polyunsaturated fatty acids (PUFAs) are known to be essential fatty acids in the human diet. Both types offer health benefits; however, the importance of a balanced intake of ω-6 and ω-3 PUFAs is necessary to prevent and manage many diseases [27]. The ideal ω-6/ω-3 ratio is between 1 and 5, which has been related to a significant decrease in inflammatory, cancer, cardiovascular and autoimmune diseases [27]. In the case of Z. lotus pulp extract, this ratio, which corresponds to the linoleic/linolenic acid ratio, is approximately 1.33, highlighting the potential of Z. lotus pulp to be exploited in nutraceutical applications. Moreover, minor amounts of (9E)-otadec-9-enoic acid (18–135 mg kg−1 dw) and eicos-11-enoic acid (<0.5–66 mg kg−1 dw) were also detected in the four morphological parts studied.
A diacid, namely, hexadecanedioic acid, was detected among the minor components in Z. lotus L. pulp lipophilic extracts. Additionally, two ω-hydroxy fatty acids were mainly found in the pulp fraction, as shown in Table 1, with a value of 10 mg kg−1 dw.
Finally, a wide range of fatty acid esters were identified predominantly as components of Z. lotus pulp, with ethyl hexadecanoate as the major component of this subfamily, accounting for up to 44.8% of the total FAEE content identified in the extract of this fraction (Table 1). Minor amounts of ethyl hexadecanoate and ethyl (9Z)-octadec-9-enoate were also detected in seeds, accounting for 5 and 11 mg kg−1 dw, respectively. Other fatty acid esters, namely, ethyl decanoate and ethyl eicosanoate, were also detected, although in considerably lower amounts (Table 1). Ethyl hexadec-9-enoate and ethyl octadec-9-enoate were both found in cis and trans configurations, but it was not possible to unambiguously differentiate between stereoisomers. Two fatty acid methyl esters were also identified, namely, methyl (9Z)-octadec-9-enoate detected in seeds and root bark extracts and methyl hexadecanoate identified only in the pulp extract. Methyl hexadecanoate was also previously reported in Z. lotus fruit essential oil, while methyl (9Z)-octadec-9-enoate is reported here for the first time as a component of Z. lotus. FAEEs and FAMEs have been reported to naturally occur in different plants and microalgae [28,29]. In fact, the presence of FAEEs and FAMEs in Z. lotus was previously reported, namely, in the Z. lotus fruit and in its essential oil, although without quantification [14,30]. Due to their promising biological activities [31], the investigation and quantification of these components in natural sources have become important.
2.2.2. Monoglycerides
Particular attention should be paid to the values observed for several monoglycerides detected in all morphological parts of Z. lotus (Table 1). This family was concentrated in the leaves and seeds (189 and 255 mg kg−1 dw, respectively) due to the presence of 1-linolenoylglycerol in leaves and 1-oleoylglycerol in the seed extract, representing 44.4% and 60.8% of the total monoglyceride content, respectively. To our knowledge, the six monoglycerides are described here for the first time as components of Z. lotus.
2.2.3. Long-Chain Aliphatic Alcohols
The profile of long-chain aliphatic alcohols (LCAAc) from the morphological parts of Z. lotus is reported here for the first time, as, in previous studies, only octacosanol has been identified in Z. lotus fruit (without quantification) [14]. Trace LCAAc accounted for 11.9% of the total amount of detected compounds (Table 1). This family is mainly concentrated in the pulp (340 mg kg−1 dw) and leaf extracts (438 mg kg−1 dw) (Figure 2), and octacosan-1-ol remains the predominant LCAAc (11–230 mg kg−1 dw), being present in all Z. lotus fractions, except in the seeds. Other LCAAc from C14 (tetradecan-1-ol) to C30 (triacontan-1-ol) were detected in root bark, leaf, and pulp extracts of Z. lotus, whereas, in the seeds, hexadecan-1-ol (2 mg kg−1 dw) was the LCAAc found.
2.2.4. Pentacyclic Triterpenic Compounds
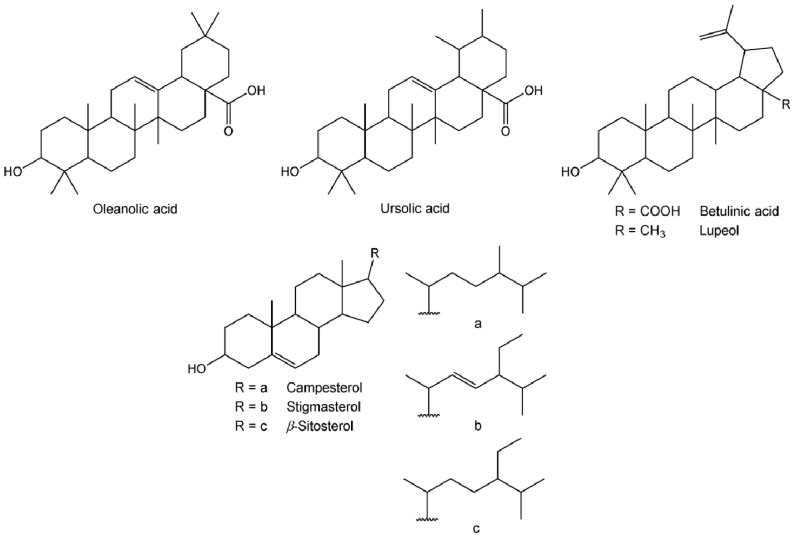
Pentacyclic triterpenic compounds were the most abundant family of lipophilic compounds detected in root bark (Figure 2), accounting for 10,230 mg kg−1 dw. Their chemical structures are presented in Figure 3. Considerable amounts of triterpenes were also observed in the other morphological parts of Z. lotus, ranging from 248 mg kg−1 dw in leaves to 608 mg kg−1 dw in pulp. Betulinic acid (BA) was the major compound identified in root bark, accounting for 9838 mg kg−1 dw (492 mg g−1 of extract), which corresponds to 89.3% of the total lipophilic compounds identified in this extract. Given the broad range of pharmacological activities already known for BA [32], its abundance in root bark highlights the value of this shrub species as a promising source of high-value ingredients for nutraceutical and pharmaceutical applications. In fact, the BA concentration in Z. lotus L. root bark is in the range of that found in other natural rich sources, namely, the outer bark of Eucalyptus nitens (Myrtaceae) (6621 mg kg−1 dw) [33] and the bark of Betula platyphylla suk. (Betulaceae) (10,800 mg kg−1 dw) [32], using the same extraction solvent. Additionally, the presented BA content is 2.9-fold higher than in Betula pendula bark and up to 3.5-fold lower than in Platanus acerifolia (Platanaceae) cork [34]. Considerable amounts of BA were also observed in the other morphological parts of Z. lotus. In addition, all of the studied extracts were shown to be composed of oleanolic acid (OA), with contents from 51 mg kg−1 dw in leaves to 287 mg kg−1 dw in root bark. Lupeol was also identified in leaves and root bark, while ursolic acid was exclusively detected in the Z. lotus fruit, namely, in pulp and seeds.
Figure 3.
Structures of pentacyclic triterpenic compounds and sterols identified in Z. lotus.
Lupeol, oleanolic, betulinic and ursolic acids (Figure 3) are common triterpenic compounds in other Zizyphus species [35,36]; however, as far as our literature survey could ascertain, lupeol, betulinic and ursolic acids are identified for the first time as constituents of Z. lotus, while oleanolic acid has been previously identified in Z. lotus L. pulp [19].
2.2.5. Sterols
Three sterols, the chemical structures of which are present in Figure 3, were also detected in Z. lotus extracts, particularly in root bark and leaves, with total contents of 257 and 355 mg kg−1 dw, respectively (Table 1). Beta-sitosterol was the main sterol observed in all morphological parts of Z. lotus, ranging from 68 mg kg−1 in pulp to 208 mg kg−1 in leaves. Stigmasterol was also found in pulp, leaves and root bark, while campesterol was only detected in root bark and leaves and was present in small amounts (4 and 28 mg kg−1 dw, respectively). To the best of our knowledge, this is the first study reporting the sterol profile of different Z. lotus morphological parts, although these compounds have been previously reported in Z. lotus fruit [14] and Z. lotus seed oil [12]. The presence of these sterols, particularly known for their various beneficial health effects [37] as Z. lotus components, increase the value of this shrub as a promising source of bioactive compounds.
2.2.6. Aromatic and Other Compounds
Apart from the major families reported above, aromatic compounds are represented by 12 compounds, which were unequally distributed in the four morphological parts of Z. lotus in quite low amounts, ranging from 11 mg kg−1 dw in the root bark to 31 mg kg−1 dw in the seed extract (Table 1). Benzoic acid is the major aromatic compound detected in the pulp, while vanillin was mainly observed in the seed extract. Of all of the aromatic compounds detected, only benzoic and p-coumaric acids have been previously identified as components of Z. lotus fruits [14].
Finally, leaf extract revealed the presence of other minor but still interesting compounds, which distinguishes it from the remaining studied fractions (Table 1). Three positional isomers of neophytadiene were identified, as well as phytol, tetracosyl acetate, inositol and squalene, which were also found in significant amounts in the seed extract. Two long-chain aliphatic aldehydes were detected in pulp extracts, namely, octacosanal (52 mg kg−1 dw) and triacontanal (27 mg kg−1 dw), which were also previously detected in Z. lotus fruits, although their contents remained unknown [14].
Two isomers of vitamin E, namely, α-tocopherol and γ-tocopherol, were detected in the lipophilic leaf extract with a total amount of 128 mg kg−1 dw (Table 1). The contents of α-tocopherols in the four morphological parts of Z. lotus were mentioned in a previous report [4].
Other minor components, such as loliolide and solerol, were also detected in leaf and pulp extracts, respectively.
2.3. Biological Activities of Lipophilic Z. lotus Extracts
2.3.1. Antiproliferative Activity
The antiproliferative effect of lipophilic extracts of different morphological parts of Z. lotus, as shown in Table 2, was evaluated for the first time against the MDA-MB-231 cell line. Cell viability was estimated by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Lipophilic root bark extract showed a significant antiproliferative effect (half-maximal inhibitory concentration (IC50) of 4.23 µg mL−1) in vitro against the triple-negative breast cancer (TNBC) MDA-MB-231 cell line (Table 2), whereas lipophilic extracts of pulp, seeds and leaves had a lower inhibitory activity (IC50 higher than 50 µg mL−1 at 48 h) even compared with the IC50 reported for the ethanolic extract of Tunisian Z. lotus leaves (45.5 µg mL−1) [38].
Table 2.
IC50 values of lipophilic extracts of Z. lotus on TNBC cell line MDA-MB-231, obtained by using the MTT assay.
| Lipophilic Z. lotus Extract | MDA-MB-231 (IC50 µg mL−1) |
|---|---|
| Pulp | >50 |
| Seeds | >50 |
| Leaves | >50 |
| Root bark | 4.23 ± 0.18 a |
| Synthetic root bark mixture * | 15.27 ± 1.79 b |
Data are expressed as mean ± standard error (n = 3). Means with different letters are statistically different at p < 0.05 using Student’s t-test. * BA + OA (97.1%:2.9%).
The strong antiproliferative effect of lipophilic root bark extract is clearly related to its particular composition, as root bark revealed the highest content of lipophilic compounds, particularly pentacyclic triterpenic compounds (and BA in particular), which represent 10,230 mg kg−1 dw, corresponding to 92.9% of the total detected compounds (Table 1). In fact, these compounds can be responsible for the suppression of MDA-MB-231 cellular growth, and BA, which is the major compound of root bark extract (9838 mg kg−1 dw, 89.3% of the total detected compounds), is known to be a promising agent against different cancer types [39]. Several reports have demonstrated the efficacy of BA against TNBC, in which it causes cell cycle arrest and, ultimately, apoptosis [40,41]. Considering the BA concentration in root bark extract (491.9 mg g−1 extract), the determined IC50 value would be 2.1 µg of BA mL−1, which is clearly lower than the toxicity described in the literature for BA (IC50 within the 10–31 µg mL−1 concentration range) [39,41], suggesting the presence of a synergistic interaction between BA and other components of the extract, namely, oleanolic acid. In fact, BA, together with lupeol, was previously suggested to be responsible for the significant antiproliferative activity of a DCM bark extract from Ziziphus mauritiana against MCF-7 cells (IC50 = 5 µg mL−1) [42]. A synthetic mixture of BA plus OA in the same proportion as that in the root bark extract (97.1%BA:2.9%OA) presented a lower antiproliferative effect (IC50 of 15.27 µg mL−1) in the studied MDA-MB-231 cell line, underlining the synergetic contributions of other presented compounds in addition to OA.
Sterols are another family of bioactive compounds characterized by their anticancer potential [37]. Beta-sitosterol was found to inhibit MDA-MB-231 cell growth by inducing cell cycle arrest at the G2/M phase and as an anti-metastatic agent [43], while stigmasterol has antiproliferative activity, in contrast to cholesterol and campesterol, which were found to have no cytotoxic effect on MDA-MB-231 cell growth [44]. Considering the significant content of sterols in leaves and root bark (355 and 257 mg kg−1 dw, respectively), these compounds could also be involved in the obtained results. Therefore, a more in-depth analysis is necessary to better understand the suppressing effect of lipophilic root bark extracts on MDA-MB-231 cell viability and the possible synergistic actions between extract components.
2.3.2. Antibacterial Activity
The antibacterial activity of the different lipophilic extracts of Z. lotus was evaluated against the bacterial strains E. coli, MSSA and S. epidermidis. The results obtained using the Resazurin assay (Table 3) demonstrated that the leaf extract exerted the highest activity among all fractions, as it had an inhibitory effect on all strains studied, especially for E. coli and S. epidermidis, with a minimum inhibitory concentration (MIC) of 1024 µg mL−1. In contrast, lipophilic pulp extract did not demonstrate antibacterial activity against any of the studied bacterial strains in the concentration range used. Moreover, the lipophilic extracts of leaves and root bark showed a slightly inhibitory effect on MSSA (MIC of 2048 μg mL−1). S. epidermidis showed susceptibility to Z. lotus extracts between 1024 (for seed and leaf extracts) and 2048 µg mL−1 (for root bark extract).
Table 3.
MIC values of lipophilic Z. lotus extracts against E. coli, MSSA and S. epidermidis, determined through Resazurin assay.
| Z. lotus Extract | MIC (μg mL−1) | ||
|---|---|---|---|
| E. coli | Methicillin-Sensitive Staphylococcus aureus (MSSA) | S. epidermidis | |
| Pulp | >2048 | >2048 | >2048 |
| Seeds | >2048 | >2048 | 1024 |
| Leaves | 1024 | 2048 | 1024 |
| Root bark | >2048 | 2048 | 2048 |
According to the literature, our results revealed that Z. lotus leaf extract had a stronger effect on E. coli when compared to the leaf methanolic extract analyzed by Ghazghazi et al. [13] (MIC of 12,500 µg mL−1) but similar to the methanolic extracts of Z. lotus studied by Naili et al. (2010) [45] (MIC of 1000 µg mL−1). Moreover, Ghazghazi et al. (2014) [13] also studied anti-S. aureus activity, which was shown to be less effective (MIC of 25,000 µg mL−1). However, other Z. lotus leaf extracts showed higher antibacterial activities, such as the acetone-derived extracts reported by Tlili et al. [46], which presented MIC values of 1000 µg mL−1 and 250 µg mL−1 against MSSA and S. epidermidis ATCC 35984, respectively, as well as the aqueous extract of leaves obtained by Rached et al. [8], which was shown to be effective against MSSA (MIC of 1250 µg mL−1).
UFAs were identified as the major components of the lipophilic extract of Z. lotus leaves, which suggests that they may be responsible for the antibacterial activity of this extract. In fact, these components are known to have promising antibacterial activities against both Gram-positive and Gram-negative bacteria by destabilizing bacterial cell membranes [47]. However, a significantly higher content of UFA was detected in seed extract, which only presented antibacterial capacity against S. epidermidis, and in an amount similar to that in leaf extract. This highlights that other minor components can be responsible or promote synergisms, leading to the reported leaf extract’s antibacterial activity. This can be attributed to long-chain aliphatic alcohols, which have been described as potent antibacterial agents [48,49].
In contrast, the antibacterial activity of root bark against MSSA and S. epidermidis may be attributed to their major compounds, namely, pentacyclic triterpenic compounds, particularly BA. This lipophilic compound is known for having antibacterial activity against Staphylococcus spp. (MIC of 64 µg mL−1 against MSSA), with evidence indicating that the cell membrane is the main target via interference with peptidoglycan biosynthesis [50].
Based on our findings, lipophilic Z. lotus extracts, particularly root bark and leaves, show interesting biological potential due to their favorable chemical composition. Z. lotus is a source of valuable bioactive compounds, and as a widespread indigenous plant occurring in arid and semiarid plateau regions of North Africa, this plant can integrate additional economic valorization with health-promoting solutions, such as human nutrition or pharmaceutics.
3. Materials and Methods
3.1. Reagents
Nonadecan-1-ol (99% purity), hexadecanoic acid (≥99% purity), β-stigmasterol (95% purity), vanillin (99%), anhydrous pyridine (99.8% purity), dichloromethane (99% purity), N,O-bis(trimethylsilyl)trifluoroacetamide (99% purity), trimethylchlorosilane (99% purity) and tetracosane (99% purity) were obtained from Sigma Chemicals Co. (Madrid, Spain). Ursolic acid (98% purity) was purchased from Aktin Chemicals (Chengdu, China). Dimethyl sulfoxide (DMSO), cell culture grade, was obtained from PanReac Applichem (Gatersleben, Germany). Acetone (≥99% purity) was supplied by VWR (Radnor, OA, USA). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide was obtained from Calbiochem (San Diego, CA, USA), and Mueller–Hinton agar or broth was purchased from Liofilchem (Roseto degli Abruzzi, Italy). Brucella Broth was purchased from Fluka Analytical, and Resazurin was obtained from Sigma-Aldrich (St. Louis, MI, USA).
3.2. Samples
Wild Z. lotus was collected from the regions of Beni Mellal, Morocco (32°20′21.998″ N; 6°21′38.999″ W), between September and October 2016. Species identification was performed based on the botanical criteria of the authors and authenticated by Professors of Botany at the University of Sultan Moulay Slimane, Morocco. The shrub was separated manually into four different morphological parts, namely, root bark, leaves, pulp and seeds; each fraction was shade-dried (15 days) and milled to granularity lower than 2 mm prior to extraction.
3.3. Extraction
Adequate mass (15 g of dw) from each Z. lotus part, i.e., root bark, leaves, pulp and seeds, were Soxhlet-extracted with DCM (150 mL) for 8 h in order to obtain the lipophilic extractives. DCM was chosen since it is known to be a specific solvent to extract lipophilic compounds from plants [51]. The solvent was evaporated to dryness under vacuum using a rotary evaporator, and the extracts were weighed. Extractions were performed in triplicate, and the results are expressed as percentage of dw material (% w/w).
3.4. GC–MS Analysis
3.4.1. Derivatization
Before GC–MS analysis, approximately 20 mg of each dried DCM extract was dissolved in 250 μL of pyridine containing 1 mg of tetracosane used as an internal standard. The compounds with carboxylic and hydroxyl groups were converted into their trimethylsilyl derivatives by adding 250 µL of N,O-bis-(trimethylsilyl)trifluoroacetamide, 50 µL of trimethylchlorosilane and 250 µL of pyridine. The mixture was heated at 70 °C for 30 min. The trimethylsilyl derivatives were analyzed by GC–MS.
3.4.2. GC–MS Conditions
GC–MS-QP2010 Ultra (Shimadzu, Kyoto, Japan) was used to analyze the derivatized extracts. A DB-1 J&W capillary column (30 m × 0.32 mm i.d., 0.25 μm film thickness, Santa Clara, CA, USA) was used to separate lipophilic compounds, using helium (at a flow rate of 35 cm s−1) as the carrier gas. The chromatographic conditions were as follows: initial temperature, 80 °C for 5 min; first temperature gradient, 4 °C min−1 up to 260 °C; second temperature gradient, 2 °C min−1 up to 285 °C for 8 min; injector temperature, 250 °C; transfer-line temperature, 290 °C; split ratio, 1:50. The injected volume was 1 µL.
Lipophilic compounds were identified by comparing their mass spectra with the GC–MS spectral libraries (Wiley 275 and U.S. National Institute of Science and Technology (NIST14)), with their retention time obtained under the same conditions [52,53,54], and comparing their MS fragmentation profiles with the literature [55,56,57,58,59,60].
3.4.3. Quantitative Analysis
Lipophilic compounds were quantified by their peak areas, with GC–MS being calibrated with pure reference compounds (after derivatization as described above) representative of each family, namely, hexadecanoic acid, nonadecan-1-ol, vanillin, β-stigmasterol and ursolic acid, relative to tetracosane (the internal standard). The respective response factors were calculated as the average of six GC−MS runs, with less than 5% variation between injections. The triplicates of each lipophilic extract were injected in duplicate, and the results, expressed in milligrams per kilogram of dw, represent the average of the concordant values obtained for the six runs.
3.5. Antiproliferative Activity
3.5.1. Cell Culture
The human triple-negative breast cancer (TNBC) MDA-MB-231 cell line was obtained from American Type Cell Culture (Manassas, VA, USA). MDA-MB-231 cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) (Biowest, Nuaillé, France) supplemented with 10% (v/v) heat-inactivated fetal bovine serum (FBSi) (Sigma-Aldrich, St. Louis, MI, USA) and 1% penicillin–streptomycin mixture (Biowest, Nuaillé, France). The cells were maintained at 37 °C in a 5% CO2 humidified atmosphere (C150, Binder GmbH, Tuttlingen, Germany). Before confluence, the cells were washed with phosphate-buffered saline (PBS), harvested with the addition of a trypsin solution (0.5 gL−1)/EDTA (0.2 gL−1) (Biowest, Nuaillé, France) and suspended in fresh growth medium before plating. All experiments were performed during the linear phase of cellular growth.
3.5.2. Cell Viability Assay
MDA-MB-231 cells were seeded in 96-well plates at 2 × 105 cells/mL and allowed to adhere for 24 h at 37 °C. Cells were then incubated with different lipophilic extracts of Z. lotus (root bark, leaves, seeds and pulp) at different concentrations (0.1, 5, 10, 20, 50 and 100 μg mL−1) for 48 h. Under the same conditions, cells were also incubated with a synthetic root bark mixture in the proportion of 2.9% OA plus 97.1% BA. Vehicle solvent control cells received dimethyl sulfoxide (DMSO) (<1% (v/v)), cell culture grade (Applichem, Germany). Cell viability was estimated by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) (Calbiochem, San Diego, CA, USA) assay as previously described [61]. Briefly, 20 μL of MTT stock solution was added to each well (final concentration 0.5 mg mL−1), followed by an incubation period of 4 h. A DMSO/ethanol (1:1) solution was then added to dissolve the formed formazan crystals, followed by a spectrophotometric determination at 570 nm (MultiSkan FC, Thermo Scientific, Rochester, NY, USA). Results are expressed as the percentage of cell viability relative to the control (cells with vehicle solvent). IC50, defined as the concentration necessary to cause 50% inhibition of cell viability, was calculated using GraphPad Prism 5.0 (GraphPad Prism Software Inc., San Diego, CA, USA) by plotting the percentage of cell viability as a function of sample concentration logarithm. Triplicates were performed in three independent experiments for each treatment.
3.5.3. Statistical Analysis
The statistical treatment of the antiproliferative activity data was carried out through one-way analysis of variance (ANOVA) using IBM® SPSS ® Statistics version 25 (IBM Corporation, New York, NY, USA). The source of the differences was identified through the Student’s t-test with p < 0.05 as the significance level.
3.6. Antibacterial Activity
The antibacterial activity of lipophilic Z. lotus extracts (root bark, leaves, pulp and seeds) was determined using the minimal inhibitory concentration (MIC) through the microbroth dilution method. Extracts were tested against the bacterial strains E. coli ATCC 25922, S. aureus ATCC 6538 (methicillin-sensitive Staphylococcus aureus—MSSA) and S. epidermis (clinical isolate), which were kindly provided by Portuguese Catholic University (Porto, Portugal). These bacterial strains were maintained at −80 °C in Brucella Broth supplemented with 5% DMSO until use. Assays were performed with bacterial cultures grown in Mueller–Hinton agar (MHA) plates incubated overnight. Briefly, bacterial strains in the exponential growth phase were suspended in Mueller–Hinton broth (MHB) to obtain a final inoculum concentration of 1 × 105 CFU mL−1 according to Clinical and Laboratory Standards Institute guidelines [62].
Serial dilutions of lipophilic Z. lotus extracts in 96-well plates were performed using concentrations between 8 and 2048 μg mL−1. The following controls were also performed: (i) solvent control: bacterial cultures with 4% (v/v) DMSO or acetone; (ii) growth control: bacterial inoculum; and (iii) sterility control: culture media. Three independent experiments were performed for each extract, each one in triplicate. The MIC values were determined after 24 h of incubation at 37 °C by using the Resazurin assay adapted from Sarker et al. (2007) [63]. MIC was considered to be the minimum concentration of the tested sample at which the color did not change from blue to pink and did not fluoresce when reduced to resorufin by oxidoreductases within viable cells.
4. Conclusions
The lipophilic fractions of Z. lotus pulp, seeds, leaves and root bark were characterized in detail by gas chromatography–mass spectrometry, allowing the identification and quantification of 99 compounds, including fatty acids, long-chain aliphatic alcohols, pentacyclic triterpenic compounds, sterols, monoglycerides, aromatic compounds and other minor components. The four studied morphological parts of Z. lotus were determined to be composed of valuable bioactive lipophilic compounds. Root bark, in particular, was observed to be a source of betulinic acid (9838 mg kg−1 dw). The extract of this morphological part showed promising antiproliferative activity against a triple-negative breast cancer cell line, MDA-MB-231, while leaf extract revealed interesting antimicrobial activity against Escherichia coli, methicillin-sensitive Staphylococcus aureus and Staphylococcus epidermis. This study highlights the potential of Zizyphus lotus L., promoting its economic exploitation as a natural ingredient in pharmaceutical formulations, which can only be implemented after the development of sustainable extraction methodologies, a careful evaluation of technical and economic aspects and, finally, an analysis to ensure that their exploitation does not have an ecological impact. Ultimately, this means that the sustainable exploitation of the plant (integrated with fruit exploitation) or, ultimately, its cultivation as a dedicated crop can also be considered. Thus, this study represents an important step by identifying the potential of this plant as a source of bioactive compounds, but further studies encompassing the sustainable exploitation of the plant will be necessary.
Author Contributions
Conceptualization, S.A.O.S., M.F.D. and A.J.D.S.; methodology and formal analysis, S.Z., P.A.B.R., D.R. and M.M.C.; investigation, S.Z., P.A.B.R., D.R. and M.M.C.; writing—original draft preparation, S.Z.; writing—review and editing, S.A.O.S., M.F.D. and P.A.B.R.; visualization, M.C. and A.J.; supervision, S.A.O.S., M.F.D. and A.J.D.S. All authors have read and agreed to the published version of the manuscript.
Funding
This work was developed within the scope of the project CICECO-Aveiro Institute of Materials (UIDB/50011/2020 and UIDP/50011/2020), LAQV-REQUIMTE (UIDB/50006/2020 and UIDP/50006/2020), as well within the project MED—Mediterranean Institute for Agriculture, Environment and Development FCT Ref. UIDP/05183/2020, both financed by national funds through the FCT/MCTES and, where applicable, co-financed by the FEDER within the PT2020 Partnership Agreement. S.A.O. Santos thanks the project AgroForWealth: Biorefining of agricultural and forest by-products and wastes: integrated strategic for valorization of resources towards society wealth and sustainability (CENTRO-01-0145-FEDER-000001), funded by Centro 2020 through FEDER and PT2020, for financial support. Patrícia A. B. Ramos acknowledges the “MultiBiorefinery” project (POCI-01-0145-FEDER-016403) for her postdoctoral grant. Sofia Zazouli gratefully acknowledges the financial support from University Sultan Moulay Slimane, Faculty of Sciences and Technology of Beni Mellal, Morocco.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of the compounds are not available from the authors.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Maraghni M., Gorai M., Neffati M. Seed germination at different temperatures and water stress levels, and seedling emergence from different depths of Ziziphus lotus. S. Afr. J. Bot. 2010;76:453–459. doi: 10.1016/j.sajb.2010.02.092. [DOI] [Google Scholar]
- 2.Sheng J.P., Shen L. Postharvest Biology and Technology of Tropical and Subtropical Fruits. Woodhead Publishing Limited; Oxford, UK: 2011. 13-Chinese jujube (Ziziphus jujuba Mill.) and Indian jujube (Ziziphus mauritiana Lam.) pp. 299–326. [Google Scholar]
- 3.Regehr D.L., El Brahli A. Wild Jujube (Ziziphus lotus) Control in Morocco. Weed Technol. 1995;9:326–330. doi: 10.1017/S0890037X00023423. [DOI] [Google Scholar]
- 4.Benammar C., Hichami A., Yessoufou A., Simonin A.M., Belarbi M., Allali H., Khan N.A. Zizyphus lotus L. (Desf.) modulates antioxidant activity and human T-cell proliferation. BMC Complement. Altern. Med. 2010;10:1–9. doi: 10.1186/1472-6882-10-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Borgi W., Ghedira K., Chouchane N. Antiinflammatory and analgesic activities of Zizyphus lotus root barks. Fitoterapia. 2007;78:16–19. doi: 10.1016/j.fitote.2006.09.010. [DOI] [PubMed] [Google Scholar]
- 6.Abdeddaim M., Lombarkia O., Bacha A., Fahloul D., Abdeddaim D., Farhat R., Saadoudi M., Noui Y., Lekbir A. Biochemical characterization and nutritional properties of Zizyphus lotus L. fruits in Aures region, northeastern of Algeria. Food Sci. Technol. 2014;15:75–81. [Google Scholar]
- 7.Maciuk A., Ghedira K., Thepenier P., Lavaud C., Zeches-Hanrot M. A new flavonol glycoside from leaves of Zizyphus Lotus. Die Pharm.-Int. J. Pharm. Sci. 2003;58:158–159. doi: 10.1002/chin.200323205. [DOI] [PubMed] [Google Scholar]
- 8.Rached W., Barros L., Ziani B.E.C., Bennaceur M., Calhelha R.C., Heleno S.A., Alves M.J., Marouf A., Ferreira I.C.F.R. HPLC-DAD-ESI-MS/MS screening of phytochemical compounds and the bioactive properties of different plant parts of Zizyphus lotus (L.) Desf. Food Funct. 2019;10:5898–5909. doi: 10.1039/C9FO01423C. [DOI] [PubMed] [Google Scholar]
- 9.Benammar C., Baghdad C. Antidiabetic and Antioxidant Activities of Zizyphus lotus L. Aqueous Extracts in Wistar Rats. J. Nutr. Food Sci. 2014;s8:8–13. doi: 10.4172/2155-9600.S8-004. [DOI] [Google Scholar]
- 10.Bakhtaoui F.Z., Lakmichi H., Megraud F., Chait A., Gadhi C.E.A. Gastro-protective, anti-Helicobacter pylori and, antioxidant properties of Moroccan Zizyphus lotus L. J. Appl. Pharm. Sci. 2014;4:81–87. doi: 10.7324/JAPS.2014.401015. [DOI] [Google Scholar]
- 11.Borgi W., Chouchane N. Anti-spasmodic effects of Zizyphus lotus (L.) Desf. extracts on isolated rat duodenum. J. Ethnopharmacol. 2009;126:571–573. doi: 10.1016/j.jep.2009.09.022. [DOI] [PubMed] [Google Scholar]
- 12.Chouaibi M., Mahfoudhi N., Rezig L., Donsì F., Ferrari G., Hamdi S. Nutritional composition of Zizyphus lotus L. seeds. J. Sci. Food Agric. 2012;92:1171–1177. doi: 10.1002/jsfa.4659. [DOI] [PubMed] [Google Scholar]
- 13.Ghazghazi H., Aouadhi C., Riahi L., Maaroufi A., Hasnaoui B. Fatty acids composition of Tunisian Ziziphus lotus L. (Desf.) fruits and variation in biological activities between leaf and fruit extracts. Nat. Prod. Res. 2014;28:1106–1110. doi: 10.1080/14786419.2014.913244. [DOI] [PubMed] [Google Scholar]
- 14.Cadi H.E., Bouzidi H.E., Selama G., Cadi A.E., Ramdan B., Oulad El Majdoub Y., Alibrando F., Dugo P., Mondello L., Lanjri A.F., et al. Physico-Chemical and Phytochemical Characterization of Moroccan Wild Jujube “Zizyphus lotus (L.)” Fruit Crude Extract and Fractions. Molecules. 2020;25:5237. doi: 10.3390/molecules25225237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ghedira K., Chemli R., Richard B., Nuzillard J.-M., Zeches M., Men-Olivier L. Le Two cyclopeptide alkaloids from Zizyphus lotus. Phytochemistry. 1993;32:1591–1594. doi: 10.1016/0031-9422(93)85186-U. [DOI] [Google Scholar]
- 16.Ghedira K., Chemli R., Caron C., Nuzilard J.M., Zeches M., Le Men-Olivier L. Four cyclopeptide alkaloids from Zizyphus lotus. Phytochemistry. 1995;38:767–772. doi: 10.1016/0031-9422(94)00669-K. [DOI] [Google Scholar]
- 17.Le Crouéour G., Thépenier P., Richard B., Petermann C., Ghédira K., Zèches-Hanrot M. Lotusine G: A new cyclopeptide alkaloid from Zizyphus lotus. Fitoterapia. 2002;73:63–68. doi: 10.1016/S0367-326X(01)00363-X. [DOI] [PubMed] [Google Scholar]
- 18.Maciuk A., Lavaud C., Thépenier P., Jacquier M.J., Ghédira K., Zèches-Hanrot M. Four new dammarane saponins from Zizyphus lotus. J. Nat. Prod. 2004;67:1639–1643. doi: 10.1021/np0499362. [DOI] [PubMed] [Google Scholar]
- 19.Jmiai A., El Ibrahimi B., Tara A., Chadili M., El Issami S., Jbara O., Khallaayoun A., Bazzi L. Application of Zizyphus Lotuse—Pulp of Jujube extract as green and promising corrosion inhibitor for copper in acidic medium. J. Mol. Liq. 2018;268:102–113. doi: 10.1016/j.molliq.2018.06.091. [DOI] [Google Scholar]
- 20.El Aloui M., Mguis K., Laamouri A., Albouchi A., Cerny M., Mathieu C., Vilarem G., Hasnaoui B. Fatty acid and sterol oil composition of four Tunisian ecotypes of Ziziphus zizyphus (L.) H.Karst. Acta Bot. Gall. 2012;159:25–31. doi: 10.1080/12538078.2012.671633. [DOI] [Google Scholar]
- 21.Borgi W., Recio M.C., Ríos J.L., Chouchane N. Anti-inflammatory and analgesic activities of flavonoid and saponin fractions from Zizyphus lotus (L.) Lam. S. Afr. J. Bot. 2008;74:320–324. doi: 10.1016/j.sajb.2008.01.009. [DOI] [Google Scholar]
- 22.Wahida B., Abderrahman B., Nabil C. Antiulcerogenic activity of Zizyphus lotus (L.) extracts. J. Ethnopharmacol. 2007;112:228–231. doi: 10.1016/j.jep.2007.02.024. [DOI] [PubMed] [Google Scholar]
- 23.Guo S., Duan J.A., Tang Y.P., Yang N.Y., Qian D.W., Su S.L., Shang E.X. Characterization of triterpenic acids in fruits of ziziphus species by HPLC-ELSD-MS. J. Agric. Food Chem. 2010;58:6285–6289. doi: 10.1021/jf101022p. [DOI] [PubMed] [Google Scholar]
- 24.Masullo M., Montoro P., Autore G., Marzocco S., Pizza C., Piacente S. Quali-quantitative determination of triterpenic acids of Ziziphus jujuba fruits and evaluation of their capability to interfere in macrophages activation inhibiting NO release and iNOS expression. Food Res. Int. 2015;77:109–117. doi: 10.1016/j.foodres.2015.09.009. [DOI] [Google Scholar]
- 25.Domingues R.M.A., Guerra A.R., Duarte M., Freire C.S.R., Neto C.P., Silva C.M.S., Silvestre A.J.D. Bioactive triterpenic acids: From agroforestry biomass residues to promising therapeutic tools. Mini. Rev. Org. Chem. 2014;11:382–399. doi: 10.2174/1570193X113106660001. [DOI] [Google Scholar]
- 26.Rsaissi N., Kamili E.L., Bencharki B., Hillali L., Bouhache M. Antimicrobial activity of fruits extracts of the wild jujube “Ziziphus lotus” (L.) Desf. Int. J. Sci. Eng. Res. 2013;4:1521–1528. [Google Scholar]
- 27.Simopoulos A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharm. 2002;56:365–379. doi: 10.1016/S0753-3322(02)00253-6. [DOI] [PubMed] [Google Scholar]
- 28.Annarao S., Sidhu O.P., Roy R., Tuli R., Khetrapal C.L. Lipid profiling of developing Jatropha curcas L. seeds using (1)H NMR spectroscopy. Bioresour. Technol. 2008;99:9032–9035. doi: 10.1016/j.biortech.2008.04.023. [DOI] [PubMed] [Google Scholar]
- 29.Herrera-Valencia V.A., Us-Vázquez R.A., Larqué-Saavedra F.A., Barahona-Pérez L.F. Naturally occurring fatty acid methyl esters and ethyl esters in the green microalga Chlamydomonas reinhardtii. Ann. Microbiol. 2012;62:865–870. doi: 10.1007/s13213-011-0361-z. [DOI] [Google Scholar]
- 30.Widad O., Hamza F., Youcef M., Jean-claude C., Pierre C., Fadila B., Samir B., Université B., El A. Chemical Composition and Antioxidant Activity of the Fruit Essential Oil of Zizyphus lotus (L.) Desf (Rhamnaceae) Int. J. Pharmacogn. Phytochem. Res. 2017;9:228–232. doi: 10.25258/phyto.v9i2.8067. [DOI] [Google Scholar]
- 31.Pinto M.E.A., Araújo S.G., Morais M.I., Sá N.P., Lima C.M., Rosa C.A., Siqueira E.P., Johann S., Lima L.A.R.S. Antifungal and antioxidant activity of fatty acid methyl esters from vegetable oils. An. Acad. Bras. Cienc. 2017;89:1671–1681. doi: 10.1590/0001-3765201720160908. [DOI] [PubMed] [Google Scholar]
- 32.Amiri S., Dastghaib S., Ahmadi M., Mehrbod P., Khadem F., Behrouj H., Aghanoori M.-R., Machaj F., Ghamsari M., Rosik J., et al. Betulin and its derivatives as novel compounds with different pharmacological effects. Biotechnol. Adv. 2020;38:107409. doi: 10.1016/j.biotechadv.2019.06.008. [DOI] [PubMed] [Google Scholar]
- 33.Domingues R.M.A., Patinha D.J.S., Sousa G.D.A., Villaverde J.J., Silva C.M., Freire C.S.R., Silvestre A.J.D., Neto C.P. Eucalytpus biomass residues from agro-forest and pulping industries as sources of high-value triterpenic compounds. Cellullose Chem. Technol. 2011;45:475–481. [Google Scholar]
- 34.Galgon T., Höke D., Dräger B. Identification and quantification of betulinic acid. Phytochem. Anal. 1999;10:187–190. doi: 10.1002/(SICI)1099-1565(199907/08)10:4<187::AID-PCA443>3.0.CO;2-K. [DOI] [Google Scholar]
- 35.Yang B., Yang H., Chen F., Hua Y., Jiang Y. Phytochemical analyses of Ziziphus jujuba Mill. var. spinosa seed by ultrahigh performance liquid chromatography-tandem mass spectrometry and gas chromatography-mass spectrometry. Analyst. 2013;138:6881–6888. doi: 10.1039/c3an01478a. [DOI] [PubMed] [Google Scholar]
- 36.Hossain M.J., Sikder M.A.A., Kaisar M.A., Haque M.R., Chowdhury A.A., Rashid M.A. Phytochemical and Biological Investigations of Methanol Extract of Leaves of Ziziphus mauritiana Lam. Bol. Latinoam. Y Del Caribe Plantas Med. Y Aromat. 2015;14:179–189. [Google Scholar]
- 37.Jiang L., Zhao X., Xu J., Li C., Yu Y., Wang W., Zhu L. The protective effect of dietary phytosterols on cancer risk: A systematic meta-analysis. J. Oncol. 2019;2019:7479518. doi: 10.1155/2019/7479518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Souid S., Elsayed H.E., Ebrahim H.Y., Mohyeldin M.M., Siddique A.B., Karoui H., El Sayed K.A., Essafi-Benkhadir K. 131-Oxophorbine protopheophorbide A from Ziziphus lotus as a novel mesenchymal-epithelial transition factor receptor inhibitory lead for the control of breast tumor growth in vitro and in vivo. Mol. Carcinog. 2018;57:1507–1524. doi: 10.1002/mc.22874. [DOI] [PubMed] [Google Scholar]
- 39.Sousa J.L.C., Freire C.S.R., Silvestre A.J.D., Silva A.M.S. Recent Developments in the Functionalization of Betulinic Acid and Its Natural Analogues: A Route to New Bioactive Compounds. Molecules. 2019;24:355. doi: 10.3390/molecules24020355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Guerra A., Soares B., Guerreiro O., Ramos P., Oliveira H., Silvestre A., Freire C., Duarte M.F. Anti-tumoral activity of lipophilic Eucalyptus bark extracts, enriched on triterpenic acids, against breast cancer cells. Planta Med. 2014;80:SL41. doi: 10.1055/s-0034-1394529. [DOI] [Google Scholar]
- 41.Weber D., Zhang M., Zhuang P., Zhang Y., Wheat J., Currie G., Al-eisawi Z. The efficacy of betulinic acid in triple-negative breast cancer. SAGE Open Med. 2014;2:1–12. doi: 10.1177/2050312114551974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tagne R.S., Telefo B.P., Talla E., Nyemb J.N., Njina S.N., Asrar M.F.M., Kamdje A.H.N., Moundipa P.F., Ahsana Dar Farooq M.I.C. Bio-guided fractionation of methanol extract of Ziziphus mauritiana Lam. (bark) and effect of the most active fraction on cancer cell lines. Asian Pac. J. Trop. Dis. 2015;5:307–312. doi: 10.1016/S2222-1808(14)60788-X. [DOI] [Google Scholar]
- 43.Barber M.D., Fearon K.C.H., Tisdale M.J., Mcmillan D.C., Ross A., Ross J.A. Effect of a Fish Oil-Enriched Nutritional Supplement on Metabolic Mediators in Patients With Pancreatic Cancer Cachexia Effect of a Fish Oil-Enriched Nutritional Supplement on Metabolic Mediators in Patients With Pancreatic Cancer Cachexia. Nutr. Cancer. 2009;40:157–164. doi: 10.1207/S15327914NC402. [DOI] [PubMed] [Google Scholar]
- 44.Awad A.B., Fink C.S. Phytosterols as Anticancer Dietary Components: Evidence and Mechanism of Action. J. Nutr. 2000;130:2127–2130. doi: 10.1093/jn/130.9.2127. [DOI] [PubMed] [Google Scholar]
- 45.Naili M.B., Alghazeer R.O., Saleh N.A., Al-Najjar A.Y. Evaluation of antibacterial and antioxidant activities of Artemisia campestris (Astraceae) and Ziziphus lotus (Rhamnacea) Arab. J. Chem. 2010;3:79–84. doi: 10.1016/j.arabjc.2010.02.002. [DOI] [Google Scholar]
- 46.Tlili H., Marino A., Ginestra G., Cacciola F., Mondello L., Miceli N., Taviano M.F., Najjaa H., Nostro A. Polyphenolic profile, antibacterial activity and brine shrimp toxicity of leaf extracts from six Tunisian spontaneous species. Nat. Prod. Res. 2019;35:1–7. doi: 10.1080/14786419.2019.1616725. [DOI] [PubMed] [Google Scholar]
- 47.Yoon B.K., Jackman J.A., Valle-González E.R., Cho N.-J. Antibacterial free fatty acids and monoglycerides: Biological activities, experimental testing, and therapeutic applications. Int. J. Mol. Sci. 2018;19:1114. doi: 10.3390/ijms19041114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mukherjee K., Tribedi P., Mukhopadhyay B., Sil A.K. Antibacterial activity of long-chain fatty alcohols against mycobacteria. FEMS Microbiol. Lett. 2013;338:177–183. doi: 10.1111/1574-6968.12043. [DOI] [PubMed] [Google Scholar]
- 49.Togashi N., Shiraishi A., Nishizaka M., Matsuoka K., Endo K., Hamashima H., Inoue Y. Antibacterial activity of long-chain fatty alcohols against Staphylococcus aureus. Molecules. 2007;12:139–148. doi: 10.3390/12020139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Chung P.Y., Chung L.Y., Navaratnam P. Potential targets by pentacyclic triterpenoids from Callicarpa farinosa against methicillin-resistant and sensitive Staphylococcus aureus. Fitoterapia. 2014;94:48–54. doi: 10.1016/j.fitote.2014.01.026. [DOI] [PubMed] [Google Scholar]
- 51.Rabah S., Kouachi K., Ramos P.A.B., Gomes A.P., Almeida A., Haddadi-Guemghar H., Madani K., Silvestre A.J.D., Santos S.A.O. Unveiling the bioactivity of Allium triquetrum L. lipophilic fractions: Chemical characterization and in vitro antibacterial activity against methicillin-resistant Staphylococcus aureus. Food Funct. 2020;11:5257–5265. doi: 10.1039/D0FO00769B. [DOI] [PubMed] [Google Scholar]
- 52.Freire C.S.R., Silvestre A.J.D., Neto C.P. Identification of new hydroxy fatty acids and ferulic acid esters in the wood of Eucalyptus globulus. Holzforschung. 2002;56:143–149. doi: 10.1515/HF.2002.024. [DOI] [Google Scholar]
- 53.Villaverde J.J., Domingues R.M.A., Freire C.S.R., Silvestre A.J.D., Neto C.P., Ligero P., Vega A. Miscanthus x giganteus extractives: A source of valuable phenolic compounds and sterols. J. Agric. Food Chem. 2009;57:3626–3631. doi: 10.1021/jf900071t. [DOI] [PubMed] [Google Scholar]
- 54.Domingues R.M.A., Sousa G.D.A., Silva C.M., Freire C.S.R., Silvestre A.J.D., Neto C.P. High value triterpenic compounds from the outer barks of several Eucalyptus species cultivated in Brazil and in Portugal. Ind. Crops Prod. 2011;33:158–164. doi: 10.1016/j.indcrop.2010.10.006. [DOI] [Google Scholar]
- 55.Ramos P.A.B., Guerra Â.R., Guerreiro O., Freire C.S.R., Silva A.M.S., Duarte M.F., Silvestre A.J.D. Lipophilic extracts of Cynara cardunculus L. var. altilis (DC): A source of valuable bioactive terpenic compounds. J. Agric. Food Chem. 2013;61:8420–8429. doi: 10.1021/jf402253a. [DOI] [PubMed] [Google Scholar]
- 56.Silvério F.O., Barbosa L.C.A., Silvestre A.J.D., Piló-Veloso D., Gomide J.L. Comparative study on the chemical composition of lipophilic fractions from three wood tissues of Eucalyptus species by gas chromatography-mass spectrometry analysis. J. Wood Sci. 2007;53:533–540. doi: 10.1007/s10086-007-0901-0. [DOI] [Google Scholar]
- 57.Coelho D., Marques G., Gutiérrez A., Silvestre A.J.D., del Río J.C. Chemical characterization of the lipophilic fraction of giant reed (Arundo donax) fibres used for pulp and paper manufacturing. Ind. Crops Prod. 2007;26:229–236. doi: 10.1016/j.indcrop.2007.04.001. [DOI] [Google Scholar]
- 58.Saitta M., Salvo F., Di Bella G., Dugo G., La Torre G.L. Minor compounds in the phenolic fraction of virgin olive oils. Food Chem. 2009;112:525–532. doi: 10.1016/j.foodchem.2008.06.001. [DOI] [Google Scholar]
- 59.Canini A., Alesiani D., D’Arcangelo G., Tagliatesta P. Gas chromatography—Mass spectrometry analysis of phenolic compounds from Carica papaya L. leaf. J. Food Compos. Anal. 2007;20:584–590. doi: 10.1016/j.jfca.2007.03.009. [DOI] [Google Scholar]
- 60.Zhao H.-Y., Fan M.-X., Wu X., Wang H.-J., Yang J., Si N., Bian B.-L. Chemical profiling of the Chinese herb formula Xiao-Cheng-Qi Decoction using liquid chromatography coupled with electrospray ionization mass spectrometry. J. Chromatogr. Sci. 2013;51:273–285. doi: 10.1093/chromsci/bms138. [DOI] [PubMed] [Google Scholar]
- 61.Mosmann T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays. J. Immunol. Methods. 1983;65:55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- 62.Clinical and Laboratory Standards Institute . M07: Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically. 11th ed. Clinical and Laboratory Standards Institute; Wayne, PA, USA: 2018. [Google Scholar]
- 63.Sarker S.D., Nahar L., Kumarasamy Y. Microtitre plate-based antibacterial assay incorporating resazurin as an indicator of cell growth, and its application in the in vitro antibacterial screening of phytochemicals. Methods. 2007;42:321–324. doi: 10.1016/j.ymeth.2007.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.