Abstract
Background
Nuezhenide (NZD), an iridoid glycoside isolated from Ilex pubescens Hook. & Arn. var. kwangsiensis Hand.-Mazz., used as a traditional Chinese medicine for clearing away heat and toxic materials, displays a variety of biological activities such as anti-tumor, antioxidant, and other life-protecting activities. However, a few studies involving anti-inflammatory activity and the mechanism of NZD have also been reported. In the present study, the anti-inflammatory and antioxidative effects of NZD are illustrated.
Objective
This study aims to test the hypothesis that NZD suppresses LPS-induced inflammation by targeting the NF-κB pathway in RAW264.7 cells.
Methods
LPS-stimulated RAW264.7 cells were employed to detect the effect of NZD on the release of cytokines by ELISA. Protein expression levels of related molecular markers were quantitated by western blot analysis. The levels of ROS, NO, and Ca2+ were detected by flow cytometry. The changes in mitochondrial reactive oxygen species (ROS) and mitochondrial membrane potential (MMP) were observed and verified by fluorescence microscopy. Using immunofluorescence assay, the translocation of NF-κB/p65 from the cytoplasm into the nucleus was determined by confocal microscopy.
Results
NZD exhibited anti-inflammatory activity and reduced the release of inflammatory cytokines such as nitrite, TNF-α, and IL-6. NZD suppressed the expression of the phosphorylated proteins like IKKα/β, IκBα, and p65. Besides, the flow cytometry results indicated that NZD inhibited the levels of ROS, NO, and Ca2+ in LPS-stimulated RAW264.7 cells. JC-1 assay data showed that NZD reversed LPS-induced MMP loss. Furthermore, NZD suppressed LPS-induced NF-B/p65 translocation from the cytoplasm into the nucleus.
Conclusion
NZD exhibits anti-inflammatory effects through the NF-κB pathway on RAW264.7 cells.
Keywords: Nuezhenide, RAW264.7 cells, LPS, anti-inflammation, NF-κB pathway, cytotoxicity
1. INTRODUCTION
The inflammatory response is a common pathological reaction, which occurs in all tissues and organs of the body [1-3]. It is the defensive response of the body to various external stimuli [4]. Most of the inflammatory reactions are transduced through the NF-κB signaling pathway [5, 6]. When human immune cells are affected by inflammatory factors, they secrete some proteins or peptides with specific immunoregulatory functions, thereby performing the auto-immune regulation of the body, and thus further protecting the body [4, 7, 8]. However, an excessive inflammatory response can lead to a range of diseases [9, 10]. During the inflammatory process, the NF-κB pathway always plays an important role. NF-κB is composed of IκBs, p65, and p50 and in the cytoplasm, it is inactive [11]. When IκBs are degraded, the active heterodimers p65 and p50 translocate into the nucleus, resulting in pro-inflammatory cytokines release [12, 13]. Therefore, to seek out an anti-inflammatory agent targeting the NF-κB pathway is an effective strategy.
Ilex pubescens Hook. et Arn. var. kwangsiensis Hand.- Mazz. (Chinese name called Guangxi Maodongqing), used as a traditional Chinese medicine for clearing away heat and toxic materials, belongs to genus IIex. The previous studies indicated that Guangxi Maodongqing contains many ingredients such as flavonoids, iridoid glycosides, and triterpenoid saponins, which display a variety of biological activities [14-16]. Nuezhenide (NZD), an iridoid glycoside isolated from Guangxi Maodongqing, displays a variety of biological activities such as anti-tumor, antioxidant, and life-protecting, lipid-lowering, antivirus, and antibacterial activities [17]. However, there are a few studies about the anti-inflammatory activity and mechanism of exploration. The present study, using the LPS-stimulated RAW264.7 cells model, mainly illustrates the anti-inflammatory effects and mechanism of NZD, which provided the fundamental and scientific basis for the research and development of an anti-inflammatory agent.
2. MATERIALS AND METHODS
2.1. Chemicals and Reagents
NZD was isolated and identified by our laboratory. The purity of NZD was determined by HPLC (over 98%). Griess reagent (modified-G4410), Lipopolysaccharides from Escherichia coli O111:B4. 2’,7’-Dichlorodihydrofluorescein diacetate (DCFH2-DA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Fetal bovine serum (FBS) was obtained from Life Technologies/Gibco Laboratories (Grand Island, NY, USA). IL-6 and TNF-α ELISA kits were obtained from Neonbioscience (Shenzhen, China). Antibodies: the NF-κB pathway sampler kit (#8242T), iNOS (#13120), COX-2 (#4842), and GAPDH (#5174) were obtained from Cell Signaling Technologies (Beverly, MA, USA). Dulbecco's modified eagle medium (DMEM), Fluo-3/AM, NO detector DAF-FM, fetal bovine serum (FBS) were purchased from Life Technologies/Gibco Laboratories (Grand Island, NY, USA).
2.2. Cell Cultures
RAW264.7 cells were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). They were cultured in DMEM with 10% FBS,100U/mL penicillin and 100 μg/mL streptomycin. Cells were maintained in an incubator at 37°C in a humidified atmosphere of 5% CO2.
2.3. Cell Viability Assay
RAW264.7 cells were cultured in 96-well plates at a density of 5×104 cells/well overnight. Cells were treated with NZD (0, 10, 20, 40 μM) for 24 h, and subsequently, MTT (5 mg/mL) was added to each well and cultured for 4 h. After that, the culture medium was removed and replaced by DMSO (100 μL/well) to dissolve the crystals. The absorbance was read at 570 nm with a microplate reader (BioTek, Winowski, VT, USA).
2.4. Griess Reagent Assay
RAW264.7 cells were cultured in 24-well plates at a density of 1×105 cells/well overnight. The cells pretreated with NZD (0, 10, 20, 40 μM) for 1 h were co-cultured with or without LPS (1 μg/mL) for another 18 h. The medium was collected to determine the nitrite level by Griess reagent.
2.5. Determination of IL-6 and TNF-α
RAW264.7 cells were cultured in 24-well plates at a density of 1×105 cells/well overnight. The cells pretreated with NZD (0, 10, 20, 40 μM) for 1 h were co-cultured with or without LPS (1 μg/mL) for another 24 h. The medium was collected and IL-6 and TNF-α were determined by ELISA according to the manufacturers’ instructions.
2.6. Flow Cytometry Assay
RAW264.7 cells were cultured in 12-well plates at a density of 2×105 cells/well overnight. Subsequently, cells were pretreated with NZD (0, 10, 20, 40 μM) for 1 h, and then treated with or without LPS (1 μg/mL) for 8 h. DCFH2-DA (1 μM), DAF-FM (1 μM), and Fluo-3/AM (1 μM) were employed to label cells for 30 min, 1 h, and 1 h, to detect ROS level, NO level, and Ca2+ level, respectively. After probes incubation, cells were collected and tested by the flow cytometry at the FITC channel (Becton-Dickinson, Franklin Lakes, NJ, USA).
2.7. Western Blot Analysis
RAW264.7 cells were cultured in 6-well plates at a density of 3×105 cells/well overnight. The cells were pretreated with NZD (0, 10, 20, 40 μM) for 1 h, and subsequently, LPS induction was done for a certain time. Total cell proteins were extracted using RIPA (1% PMSF and 1% cocktail). BCA protein kit (Waltham, MA, USA) was employed to determine protein concentrations. The denatured proteins were separated by 8% or 10% SDS-PAGE gels and were transferred to the PVDF membrane (Millipore, Billerica, MA, USA). After blocking the PVDF membrane with 5% nonfat milk for 1 h, the PVDF membrane was incubated with primary antibodies (1:1000) for more than 12 h at 4 °C. After the membrane was washed with TBST and incubated with secondary antibody (1:5000) for 2 h at room temperature, the membrane was exposed to ChemiDoc™ MP Imaging System (Bio-Rad, Hercules, CA, USA).
2.8. Fluorescence Assay
RAW264.7 cells were seeded in 96-well plates with a density of 5×104 cells/well overnight. Cells were pretreated with NZD (40 μM) for 1 h and then incubated with or without LPS for another 8 h. Staining was performed JC-1 (10 μg/mL) and DCFH2-DA (100 μM) for 30 min in dark. Fluorescence images were captured by fluorescence microscopy (Leica, Wetzlar, Germany).
2.9. Immunofluorescence Assay
RAW264.7 cells were cultured on confocal dishes (SPL, Pocheon, Korea) at a density of 2×105 cells/well overnight and then pretreated with NZD (40 μM) for 1 h. After stimulation with LPS (1 μg/mL) for another 1 h, cells were fixed, permeabilized, blocked, and incubated with rabbit anti-mouse NF-κB p65 antibody (1:200) for 1 h at room temperature. Cells were then incubated with Alexa Fluor 488-conjugated secondary antibody for 1 h. Hoechst 33342 was used to stain the nuclei for 10 min. The image was captured by a confocal laser scanning microscopy (Leica, Wetzlar, Germany) at an excitation/emission wavelength of 588/615-690 nm.
2.10. Statistical Analysis
All results were independently repeated at least thrice, and presented as mean ±SD. The significance of the intergroup differences was analyzed with the one-way analysis of variance (one-way-ANOVA) and Dunn’s multiple comparison tests using GraphPad Prism 6.0 software. *p<0.05 was considered a significant difference.
3. RESULTS
3.1. NZD Shows no Significant Cytotoxicity on RAW264.7 Cells
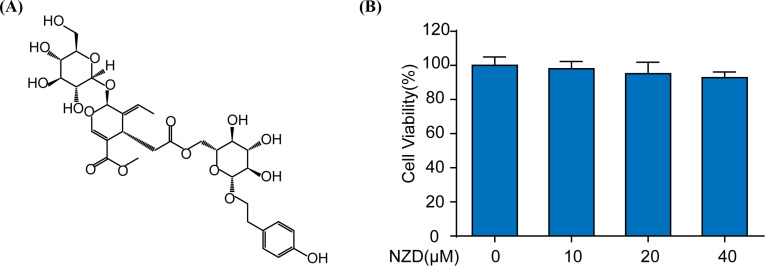
Nuezhenide (Fig. 1A), a natural product, was isolated from Guangxi Maodongqing. The cytotoxicity of NZD (0, 10, 20, 40 μM) was investigated by MTT assay in RAW264.7 cells. According to the results of this study, no significant cytotoxicity was observed (Fig. 1B).
Fig. (1).
NZD shows no significant cytotoxicity on RAW264.7 cells. (A) The chemical structure of NZD. (B) The cytotoxicity of NZD was detected by MTT assay after 24 h of treatment.
3.2. NZD Suppresses LPS-stimulated Inflammatory Response in RAW264.7 Cells
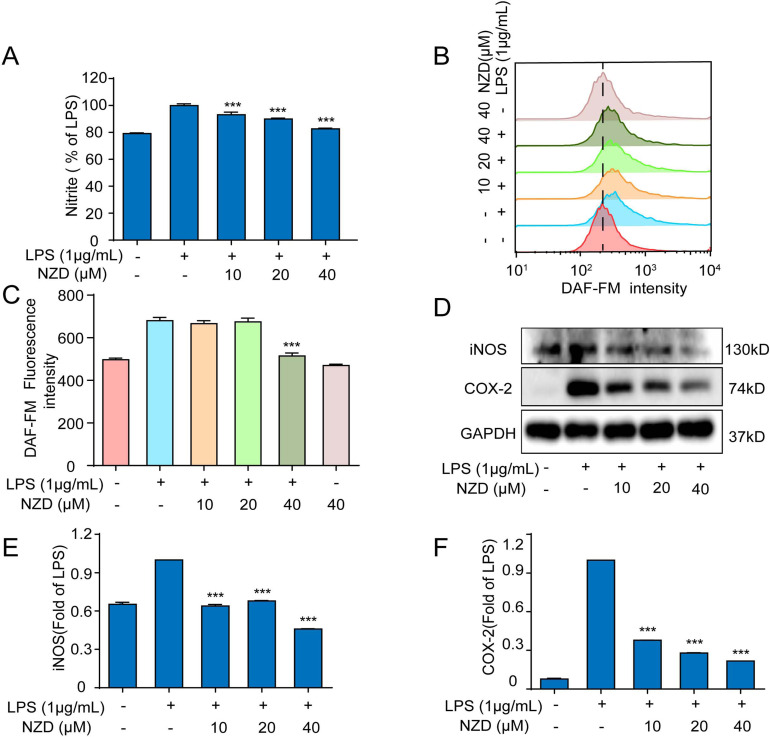
The expression of COX-2, an enzyme whose degree of expression is related to the severity of inflammation, can be induced [18-20]. Reducing or inhibiting the activation of COX-2, the synthesis and release of inflammatory mediators iNOS and NO can be regulated, which can effectively control the occurrence of inflammation [21, 22]. In order to detect and evaluate whether NZD could reduce the nitrite level and NO generation in LPS-stimulated RAW264.7 cells, we adopted the Griess reagent and flow cytometer. The results showed that the levels of nitrite level and NO release were significantly increased after the stimulation of LPS in RAW264.7 cells, and this phenomenon was reversed after NZD pretreatment (Fig. 2A-C). Besides, we further investigated whether NZD could reduce the expressions of proteins, iNOS and COX-2 in LPS-stimulated RAW264.7 cells. The results of this study indicate that the expressions of proteins, iNOS and COX-2 were significantly increased in the LPS-treated group, and pretreatment with NZD for another 18 h inhibited the expressions of proteins (Fig. 2D-F). Collectively, these results suggested that NZD displayed anti-inflammatory activities in RAW264.7 cells.
Fig. (2).
NZD suppresses LPS-stimulated inflammatory response in RAW264.7 Cells. RAW264.7 cells were pretreated with NZD for 1 h and then stimulated with LPS (1 μg/mL) for 18 h. (A) The level of nitrite was determined by Griess reagent. (B, C) RAW264.7 cells were pretreated with NZD for 1 h and then stimulated with LPS (1 μg/mL) for 8 h. The level of NO was measured by a flow cytometer with a fluorescence probe DAF-FM. (D, E, F) The expressions of iNOS and COX-2 were detected by Western blotting. * p < 0.05, ** p <0.01, ***p < 0.001 vs. LPS group.
3.3. NZD Inhibits LPS-induced Release of Pro-Inflammatory Cytokines in RAW264.7 Cells
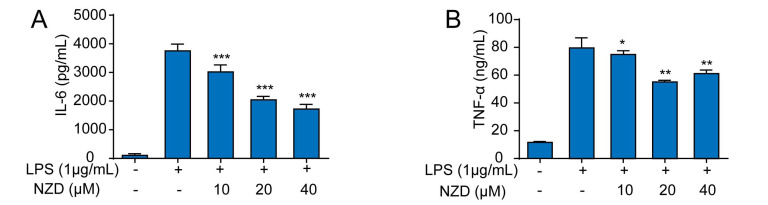
Inflammation is a complicated process, which is regulated by a series of inflammatory mediators and cytokines [23-25]. A great deal of evidence has confirmed that pro-inflammatory cytokines, such as TNF-α and IL-6, are able to induce inflammatory responses and aggravate the development of inflammation [26-28]. To further investigate the effect of NZD on the release of pro-inflammatory cytokines in LPS-stimulated RAW264.7 cells, we detected the secretion levels of IL-6 and TNF-α by ELISA. The results suggested that NZD decreased the levels of TNF-α and IL-6 levels, in LPS-stimulated RAW264.7 cells (Fig. 3A-B). Taken together, the results of this study prove that NZD suppresses LPS-induced release of pro-inflammatory cytokines in RAW264.7 cells.
Fig. (3).
NZD inhibits LPS-induced release of pro-inflammatory cytokines in RAW264.7 cells. RAW264.7 cells were pretreated with NZD for 1 h and then stimulated with LPS (1 μg/mL) for 18 h. (A, B) IL-6 and TNF-α, in the culture medium, were determined by ELISA kits. * p < 0.05, ** p <0.01, ***p < 0.001 vs. LPS group.
3.4. NZD Reverses LPS-Induced ROS Generation in RAW264.7 Cells
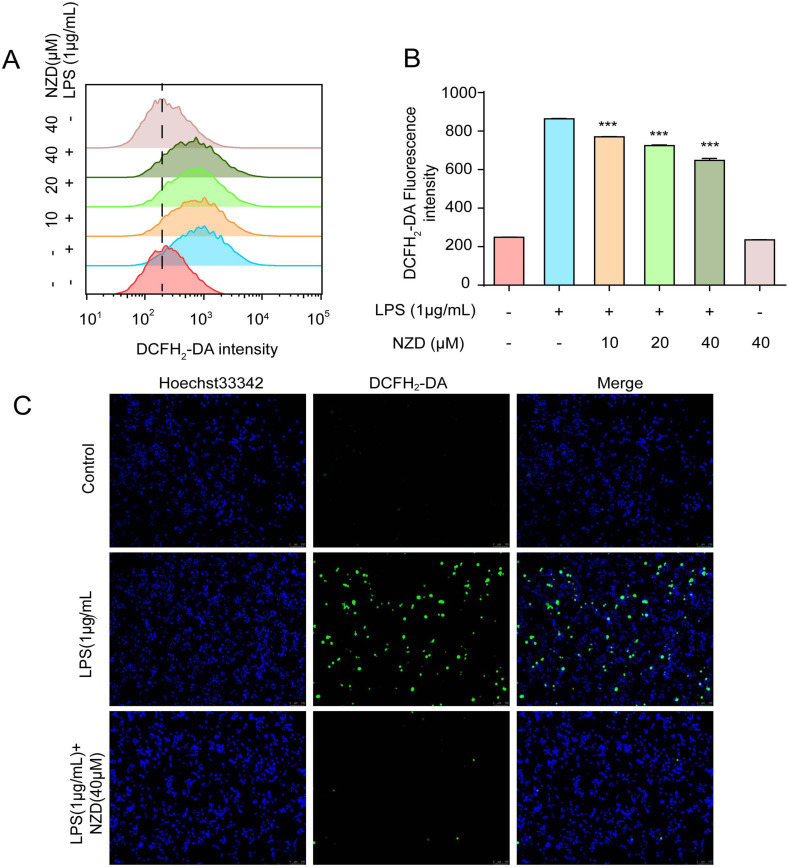
Generally, the term reactive oxygen species (ROS) refers to the oxygen in the body or natural environment, containing the properties of oxygen and active substances [29-31]. A large number of studies have shown that when the cell membranes of phagocytes in the body were stimulated, a deluge of ROS is produced through the respiratory outbreak mechanism [32-34]. Therefore, in an inflammatory response, ROS were greatly increased in cells. We used the flow cytometry to test the effects of NZD on the production of ROS. The results showed that LPS induced ROS production. NZD effectively inhibited LPS-induced ROS generation (Fig. 4A-B). Furthermore, we observed the production of ROS by fluorescence microscopy and the results were consistent with those obtained by flow cytometry (Fig. 4C). Thus, NZD decreased the LPS-induced ROS level in RAW264.7 cells.
Fig. (4).
NZD decreases LPS-induced ROS generation in RAW264.7 Cells. RAW264.7 cells were stimulated with LPS (1 μg/mL) for 8 h with or without NZD pretreatment for 1 h. (A, B) The ROS levels were measured by a flow cytometer with a fluorescence probe DCFH2-DA. (C) The ROS were detected by fluorescence microscopy. * p < 0.05, ** p <0.01, ***p < 0.001 vs. LPS group.
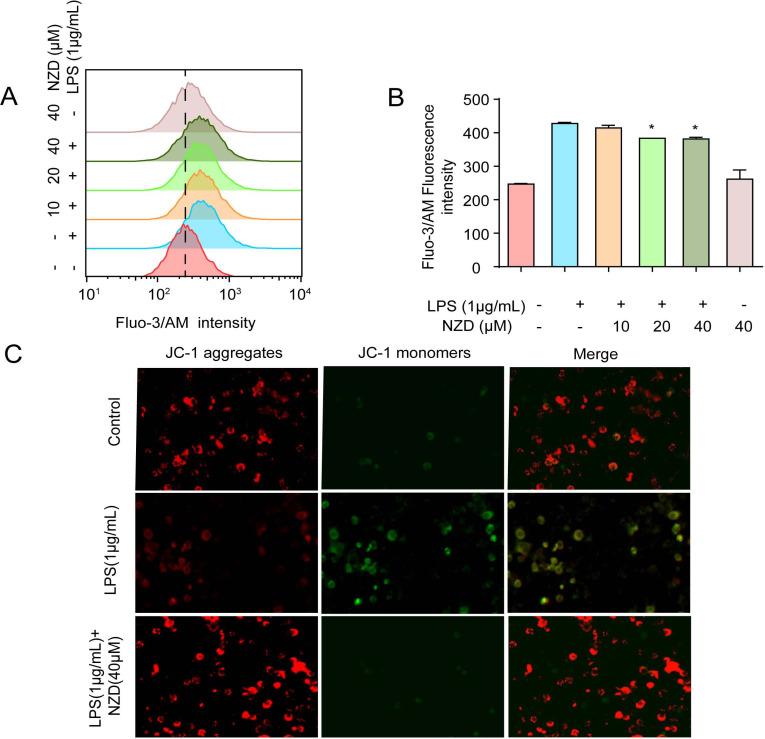
3.5. NZD Ameliorates LPS-Induced Calcium Level and MMP Loss in RAW264.7 Cells
The change in mitochondrial membrane potential and calcium concentration is closely related to inflammatory diseases [35, 36]. In this study, flow cytometry was employed to assess the effects of NZD on the level of calcium. The results showed that NZD inhibited LPS-induced calcium influx. (Fig. 5A-B). Mitochondrial membrane potential (MMP) is generated by the asymmetric distribution of protons and other ions on both sides of the inner membrane during respiratory oxidation, the stabilization of which is beneficial to maintain the normal physiological function of cells [37, 38]. LPS could disrupt the stability of MMP, which is not conducive to maintain normal physiological functions of the cell. Using the JC-1 assay and fluorescence microscopy, it was observed that NZD reversed MMP loss in LPS-stimulated RAW264.7 cells (Fig. 5C). Taken together, NZD reversed MMP loss and inhibited calcium influx.
Fig. (5).
NZD Ameliorates LPS-Induced Calcium Level and MMP Loss in RAW264.7 cells. (A, B) RAW264.7 cells were treated with LPS (1 μg/mL) and the calcium levels were measured by cytometer with a fluorescence probe Fluo-3/AM. (C) RAW264.7 cells were stimulated with LPS (1 μg/mL) for 6 h with or without NZD (40 μM) pretreatment for 1 h. The MMP was detected by fluorescence microscopy. * p < 0.05, ** p < 0.01, *** p < 0.001 vs. LPS group.
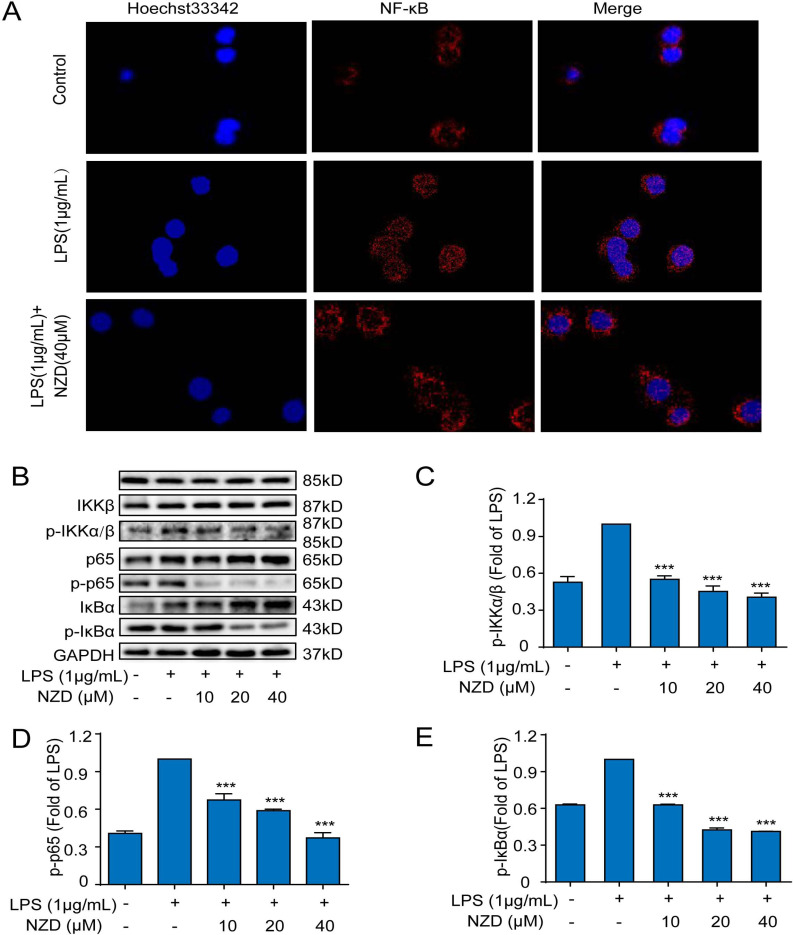
3.6. The NF-κB Pathway Participates in the Anti-Inflammatory Effect of NZD in RAW264.7 Cells
LPS could activate the NF-κB signaling pathway, spawning a pathogen-specific innate immune response through the release of pro-inflammatory cytokines [39, 40]. To further study the mechanisms of the anti-inflammatory effect of NZD, the NF-κB signaling pathway was investigated. As shown in Fig. (6A), LPS led to the translocation of NF-κB/p65 from the cytoplasm into the nucleus, which was reversed by NZD pretreatment. Besides, the results of this study showed that NZD suppressed the phosphorylation expression of IKKα/β, p65, and IκBα without altering the total expression of IKKα/β, p65, and IκBα. (Fig. 6B-E). Thus, the activity of the NF-κB pathway was suppressed by NZD in LPS-stimulated RAW264.7 cells.
Fig. (6).
The NF-κB pathway participates in the anti-inflammatory effect of NZD in RAW264.7 cells. RAW264.7 cells were stimulated with LPS (1 μg/mL) for 8 h after NZD pretreatment for 1 h. (A) The localization of p65 in the cytoplasm and nucleus was measured by immunofluorescence staining. The expressions of proteins involved in NF-κB (B) pathway, including p-IKKα/β (C), p-p65 (D), p-IκBα (E) proteins, were measured by Western blotting. * p < 0.05, ** p <0.01, ***p < 0.001 vs. LPS group.
4. DISCUSSION
Inflammation is a defensive response of the body to various stimuli. However, long chronic inflammation damages the body. Thus, the homeostasis of inflammation in the body should be maintained [41, 42]. Macrophages,an important effector cells of human innate immune response,can secrete a variety of pro-inflammatory cytokines such as IL-6, IL-12, IL-23, TNF-α, iNOS, and COX-2 [43, 44]. TNF-α,a pro-inflammatory factor, is a marker of early inflammation, mediating immune response and tissue damage [45, 46]. Excessive TNF-α can induce the production of other cytokines, thus aggravating inflammatory damage, and various inflammation-related diseases that represent high levels of TNF-α [47, 48]. IL-6 is both a pro-inflammatory factor and an anti-inflammatory factor [49]. When the concentration of IL-6 in the body is low, it can enhance the immune defense response of the body [50]. When the concentration of IL-6 in the body is high, it will lead to a series of inflammatory reactions, causing certain damage to tissues and organs [27, 51, 52]. Furthermore, IL-6 is one of the most important mediators of various chronic and acute inflammatory reactions [53]. NO is an important inflammatory mediator, which can remove pathogens in the early stage of inflammation and provide protective measures for the body [54]. However, excessive NO reacts with superoxide anion to generate peroxide nitrite, leading to local tissue damage and promoting the occurrence of inflammatory diseases [55]. In this study, it was found that the secretion of NO, TNF-α, and IL-6 was significantly inhibited after the pretreatment of NZD, suggesting that NZD exerts its anti-inflammatory effect via inhibiting the release of NO, TNF-α, and IL-6 in RAW264.7 cells. As already known, SARS-CoV-2-induced COVID-19 has outbreak in the world has lead to over one million pneumonia patients and 50,000 deaths, and no effective drugs to treat COVID-19 have been reported until now. Previous studies indicated that a significant inflammatory storm was discovered in COVID-19 patients. More specifically, IL-6 was found to be significantly increased in most COVID-19 patients [56]. Thus, a drug that inhibits IL-6 release may ameliorate lung injury induced by SARS-Cov-2. Accoring to the results of the present study, it can be speculated that NZD can not directly kill the virus due to the absence of cytotoxicity. However, NZD can significantly suppress IL-6 release, suggesting that NZD can indirectly effectively treat COVID through anti-inflammatory effects.
COX has three isozymes: COX-1, COX-2, and COX-3 [57]. COX-2, an inducible expression enzyme, is closely related to inflammatory diseases [58, 59]. Its expression is related to the severity of inflammation [60]. The synthesis and release of inflammatory mediator COX-2 affected the occurrence of inflammation [61]. In this study, we found that NZD inhibited the expression of COX-2 in LPS-stimulated RAW264.7 cells, suggesting that NZD could be used as an inhibitor of COX-2 and considered for further study.
INOS is closely related to inflammation, which can catalyze the continuous production of NO by L-arginine [62-64]. Excessive NO leads to cell damage and tissue necrosis, thus promoting the development of inflammatory diseases [65]. In this study, the data indicated that NZD suppresses NO release, nitrite level, and the expression of iNOS protein.
Reactive oxygen species (ROS), a by-product of aerobic metabolism, have strong chemical reactivity and play an important role in cell signaling transduction and body stability [66, 67]. Once stimulated, ROS increase sharply, and then induce inflammation through a series of signal transduction pathways influenced by oxidative stress of cells [68]. Moreover, excessive ROS cause damage to the mitochondria by decreasing MMP [69, 70]. Besides, calcium, as a second signal message, plays an important role in the activation of macrophages, such as transcriptional control, and the activation of kinases, and phosphatases [71, 72]. The results of this study indicate that NZD recused MMP loss and reversed ROS generation and Calcium influx in LPS-stimulated RAW264.7 cells.
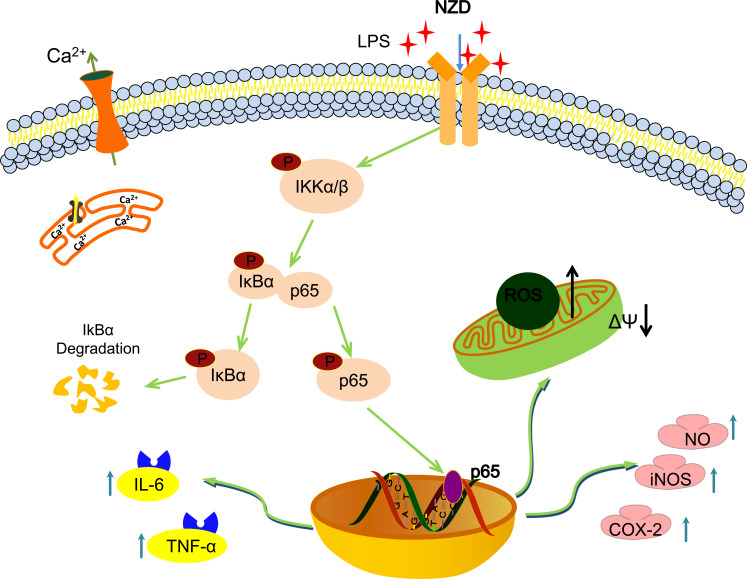
The NF-κB signaling pathway plays a vital role in the pathological process of inflammatory responses [73-75]. NF-κB is a key transcription factor in the inflammatory responses. It exists in the cytoplasm in the form of inactive NF-κB/IκBα complex at rest [76, 77]. Furthermore, the nucleus translocation of NF-κB/p65 into the nucleus can regulate the release of large amounts of inflammatory mediators such as TNF-α, IL-6, etc [78, 79]. Therefore, the changes in NF-κB/p65 can be a marker of inflammation. When cells are stimulated by LPS, IκBα is phosphorylated and degraded, and then NF-κB is translocated into the nucleus to regulate the release of cytokines like NO, ROS, IL-6, and TNF-α, chemokines and inflammatory response [75, 80]. As shown in Fig. (7), the results of this study suggest that NZD can inhibit the translocation of NF-κB/p65 from the cytoplasm into the nucleus and also inhibit the expression of the proteins of p-IKKα/β, p-p65, and p-IκBα proteins, indicating that NZD exerts anti-inflammatory activity through the NF-κB pathway.
Fig. (7).
The schematic representation of the anti-inflammatory effect and mechanisms of NZD. NZD showed an inhibitory effect on LPS-induced inflammation in RAW264.7 cells. The underlying mechanism is suppressing the NF-κB pathway.
CONCLUSION
In summary, this study shows that NZD displays anti-inflammatory effects through the NF-κB pathway. Thus, NZD can be considered a potential new drug for the clinical treatment of inflammation.
ACKNOWLEDGEMENTS
Declared none.
LIST OF ABBREVIATIONS
- COX-2
Cyclooxygenase-2
- DCFH2-DA 2'
7'-Dichlorodihydrofluorescein diacetate
- DAF-FM 4-amino-5-methylamino-2'
7'-difluorofluorescein diacetate
- NZD
Nuezhenide
- DMEM
Dulbecco's Modified Eagle Medium
- DMSO
Dimethyl Sulfoxide
- ELISA
Enzyme-linked Immunosorbent Assay
- Fluo-3AM
Fluo-3-pentaacetoxymethyl Ester
- FBS
Fetal Bovine Serum
- IL-6
Interleukin-6
- IKKα
Inhibitor of Nuclear Factor Kappa-B Kinase
- IKKβ
Inhibitor of Nuclear Factor kappa-B Kinase
- IκBα
Inhibitor of Kappa B α
- iNOS
Inducible Nitric Oxide Synthase
- LPS
Lipopolysaccharide
- MMP
Mitochondrial Membrane Potential; NO Nitric Oxide
- MTT
3-(4, 5-dimethylthiazol-2-yl)-2 5-diphenyl tetrazolium bromide
- NO
Nitric Oxide
- NF-κB
Nuclear Factor-kappa B
- NO
Nitric Oxide
- PVDF
Polyvinylidene Fluoride
- ROS
Reactive Oxygen Species
- TNF-α
Tumor Necrosis Factor-α
- SD
Standard Deviation
Funding Statement
We would like to appreciate the support from the National Natural Science Foundation of China (NSFC, 81803807), Guangxi Natural Science Foundation (2018JJB140265), Guangxi Science and Technology Base and Talent Special Project (2018AD19034), the Profect of China-ASEAN Traditional Chinese Medicine Development Research Center (05J1601602 and 0501801715), the Project of cultivating High-level Talent Teams in the Qi Huang Project of Guangxi University of Chinese Medicine (2018002), Study the effect and mechanism of YZZ4 405 in the treatment of nephritis based on the influence of P2X7/ROS signaling pathway 406 (YCSZ20190012) and Ph.D. Fund of Guangxi University of Chinese Medicine (B170023), and the project of Guangxi overseas “100 persons plan” high-level expert.
ETHICAL APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
We would like to appreciate the support from the National Natural Science Foundation of China (NSFC, 81803807), Guangxi Natural Science Foundation (2018JJB140265), Guangxi Science and Technology Base and Talent Special Project (2018AD19034), the Profect of China-ASEAN Traditional Chinese Medicine Development Research Center (05J1601602 and 0501801715), the Project of cultivating High-level Talent Teams in the Qi Huang Project of Guangxi University of Chinese Medicine (2018002), Study the effect and mechanism of YZZ4 405 in the treatment of nephritis based on the influence of P2X7/ROS signaling pathway 406 (YCSZ20190012) and Ph.D. Fund of Guangxi University of Chinese Medicine (B170023), and the project of Guangxi overseas “100 persons plan” high-level expert.
CONFLICT OF INTEREST
The authors have no conflicts of interest, financial or otherwise.
REFERENCE
- 1.Jiang W-Y. Therapeutic wisdom in traditional chinese medicine: A perspective from modern science. Trends Pharmacol. Sci. 2005;26(11):558–563. doi: 10.1016/j.tips.2005.09.006. [DOI] [PubMed] [Google Scholar]
- 2.Mohan H. Textbook of pathology. Jaypee Brothers. Medical Publishers Pvt. Limited; 2018. [Google Scholar]
- 3.Gieseck R.L., III, Wilson M.S., Wynn T.A. Type 2 immunity in tissue repair and fibrosis. Nat. Rev. Immunol. 2018;8(1):62–76. doi: 10.1038/nri.2017.90. [DOI] [PubMed] [Google Scholar]
- 4.Valesini G., Gerardi M.C., Iannuccelli C., Pacucci V.A., Pendolino M., Shoenfeld Y. Citrullination and autoimmunity.Mosaic of autoimmunity. Elsevier; 2019. pp. 117–126. [DOI] [PubMed] [Google Scholar]
- 5.May M.J., Ghosh S. Signal transduction through nf-κb. Immunol. Today. 1998;19(2):80–88. doi: 10.1016/s0167-5699(97)01197-3. [DOI] [PubMed] [Google Scholar]
- 6.Haque M.A., Jantan I., Harikrishnan H., Ghazalee S. Standardized extract of Zingiber zerumbet suppresses lps-induced pro-inflammatory responses through nf-κb, mapk and pi3k-akt signaling pathways in u937 macrophages. Phytomedicine. 2019;54:195–205. doi: 10.1016/j.phymed.2018.09.183. [DOI] [PubMed] [Google Scholar]
- 7.Pearson J.A., Wong F.S., Li W. The importance of the non obese diabetic (NOD) mouse model in autoimmune diabetes. J. Autoimmun. 2016;66:76–88. doi: 10.1016/j.jaut.2015.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dwivedi M., Kumar P., Laddha N.C., Kemp E.H. Induction of regulatory t cells: A role for probiotics and prebiotics to suppress autoimmunity. Autoimmun. Rev. 2016;15(4):379–392. doi: 10.1016/j.autrev.2016.01.002. [DOI] [PubMed] [Google Scholar]
- 9.Chen L., Deng H., Cui H., Fang J., Zuo Z., Deng J., Li Y., Wang X., Zhao L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2018;9(6):7204–7218. doi: 10.18632/oncotarget.23208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang H., Tsao R. In: Current opinion in food science. Marangoni A.G., Sant'Ana Anderson, editors. Vol. 8. Elsevier Science B. V; Amsterdam: 2016. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. ; pp. 33–42. [Google Scholar]
- 11.Wu X., Gao H., Hou Y., Yu J., Sun W., Wang Y., Chen X., Feng Y., Xu Q-M., Chen X. Dihydronortanshinone, a natural product, alleviates lps-induced inflammatory response through nf-κb, mitochondrial ros, and mapk pathways. Toxicol. Appl. Pharmacol. 2018;355:1–8. doi: 10.1016/j.taap.2018.06.007. [DOI] [PubMed] [Google Scholar]
- 12.Herrington F.D., Carmody R.J., Goodyear C.S. Modulation of nf-κb signaling as a therapeutic target in autoimmunity. J. Biomol. Screen. 2016;21(3):223–242. doi: 10.1177/1087057115617456. [DOI] [PubMed] [Google Scholar]
- 13.Ning S., Friedrich T., Guo L. Nfκb and kidney injury. Front. Immunol. 2019;10:815. doi: 10.3389/fimmu.2019.00815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sparg S., Light M., Staden J.V. Biological activities and distribution of plant saponins. J. Ethnopharmacol. 2004;94(2-3):219–243. doi: 10.1016/j.jep.2004.05.016. [DOI] [PubMed] [Google Scholar]
- 15.Thring T.S., Hili P., Naughton D.P. Anti-collagenase, anti-elastase and anti-oxidant activities of extracts from 21 plants. BMC Complement. Altern. Med. 2009;9(1):27. doi: 10.1186/1472-6882-9-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bai J., Kwok W.C., Thiery J-P. Traditional Chinese medicine and regulatory roles on epithelial–mesenchymal transitions. Chin. Med. 2019;14(1):1–13. doi: 10.1186/s13020-019-0257-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhao Y-C., Xue C-H., Zhang T-T., Wang Y-M. Saponins from sea cucumber and their biological activities. J. Agric. Food Chem. 2018;66(28):7222–7237. doi: 10.1021/acs.jafc.8b01770. [DOI] [PubMed] [Google Scholar]
- 18.Minghetti L. Cyclooxygenase-2 (cox-2) in inflammatory and degenerative brain diseases. J. Neuropathol. Exp. Neurol. 2004;63(9):901–910. doi: 10.1093/jnen/63.9.901. [DOI] [PubMed] [Google Scholar]
- 19.Dhir A. An update of cyclooxygenase (cox)-inhibitors in epilepsy disorders. Expert Opin. Investig. Drugs. 2019;28(2):191–205. doi: 10.1080/13543784.2019.1557147. [DOI] [PubMed] [Google Scholar]
- 20.Zhong Z., Zhang Q., Tao H., Sang W., Cui L., Qiang W., San Cheang W., Hu Y., Yu H., Wang Y. Anti-inflammatory activities of sigesbeckia glabrescens makino: Combined in vitro and in silico investigations. Chin. Med. 2019;14(1):35. doi: 10.1186/s13020-019-0260-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shin W-B., Dong X., Kim Y-S., Park J-S., Kim S-J., Go E-A., Kim E-K., Park P-J. Springer; 2019. Anti-inflammatory effects of batillaria multiformis water extracts via nf-кb and mapk signaling pathways in lps-induced raw 264.7 cells Taurine 11. pp. 1001–1014. [DOI] [PubMed] [Google Scholar]
- 22.Wu C-T., Deng J-S., Huang W-C., Shieh P-C., Chung M-I., Huang G-J., Longevity C. Salvianolic acid c against acetaminophen-induced acute liver injury by attenuating inflammation, oxidative stress, and apoptosis through inhibition of the keap1/nrf2/ho-1 signaling. Oxid. Med. Cell. Longev. 2019;2019:1–13. doi: 10.1155/2019/9056845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sugimoto M.A., Vago J.P., Perretti M., Teixeira M. Mediators of the resolution of the inflammatory response. Trends Immunol. 2019;40(3):212–227. doi: 10.1016/j.it.2019.01.007. [DOI] [PubMed] [Google Scholar]
- 24.Mantovani A., Dinarello C.A., Molgora M., Garlanda C. Interleukin-1 and related cytokines in the regulation of inflammation and immunity. Immunity. 2019;50(4):778–795. doi: 10.1016/j.immuni.2019.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ahn C-B., Cho Y-S., Je J-Y. Purification and anti-inflammatory action of tripeptide from salmon pectoral fin byproduct protein hydrolysate. Food Chem. 2015;168:151–156. doi: 10.1016/j.foodchem.2014.05.112. [DOI] [PubMed] [Google Scholar]
- 26.Alam Q., Zubair Alam M., Mushtaq G., Haque A. Inflammatory process in alzheimer’s and parkinson’s diseases: Central role of cytokines. Curr. Pharm. Des. 2016;22(5):541–548. doi: 10.2174/1381612822666151125000300. [DOI] [PubMed] [Google Scholar]
- 27.Del Giudice M., Gangestad S. Behavior, immunity: Rethinking il-6 and crp: Why they are more than inflammatory biomarkers, and why it matters. Brain Behav. Immun. 2018;70:61–75. doi: 10.1016/j.bbi.2018.02.013. [DOI] [PubMed] [Google Scholar]
- 28.Du H., Kuang T-T., Qiu S., Xu T., Huan C-L.G., Fan G., Zhang Y. Fecal medicines used in traditional medical system of china: A systematic review of their names, original species, traditional uses, and modern investigations. Chin. Med. 2019;14(1):31. doi: 10.1186/s13020-019-0253-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tafani M., Sansone L., Limana F., Arcangeli T., De Santis E., Polese M., Fini M., Russo M.A. The interplay of reactive oxygen species, hypoxia, inflammation, and sirtuins in cancer initiation and progression. Oxid. Med. Cell. Longev. 2016;2016:3907147. doi: 10.1155/2016/3907147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang Y., Dai M., Yuan Z. Methods for the detection of reactive oxygen species. Anal. Methods. 2018;10(38):4625–4638. doi: 10.1039/C8AY01339J. [DOI] [Google Scholar]
- 31.Garcia A.J., III, Viemari J. C. Khuu M.A. Neurobiology: Respiratory rhythm generation, hypoxia, and oxidative stress-implications for development. Respir. Physiol. Neurobiol. 2019:103259. doi: 10.1016/j.resp.2019.103259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dominguez-Andres J., Netea M. Long-term reprogramming of the innate immune system. J. Leukoc. Biol. 2019;105(2):329–338. doi: 10.1002/JLB.MR0318-104R. [DOI] [PubMed] [Google Scholar]
- 33.Bols N.C., Brubacher J.L., Ganassin R.C., Lee L. Ecotoxicology and innate immunity in fish. Dev. Comp. Immunol. 2001;25(8-9):853–873. doi: 10.1016/S0145-305X(01)00040-4. [DOI] [PubMed] [Google Scholar]
- 34.Yuan R., Huang L., Du L-J., Feng J-F., Li J., Luo Y-Y., Xu Q-M., Yang S-L., Gao H., Feng Y-L. Dihydrotanshinone exhibits an anti-inflammatory effect in vitro and in-vivo through blocking tlr4 dimerization. Pharmacol. Res. 2019;142:102–114. doi: 10.1016/j.phrs.2019.02.017. [DOI] [PubMed] [Google Scholar]
- 35.Perry S.W., Norman J.P., Barbieri J., Brown E.B., Gelbard H.A. Mitochondrial membrane potential probes and the proton gradient: A practical usage guide. Biotechniques. 2011;50(2):98–115. doi: 10.2144/000113610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sena L.A., Chandel N. Physiological roles of mitochondrial reactive oxygen species. Mol. Cell. 2012;48(2):158–167. doi: 10.1016/j.molcel.2012.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Poli G., Schaur R.J., Leonarduzzi GJMrr. 4-hydroxynonenal: A membrane lipid oxidation product of medicinal interest. Med. Res. Rev. 2008;28(4):569–631. doi: 10.1002/med.20117. [DOI] [PubMed] [Google Scholar]
- 38.Lecaille F., Brömme D., Lalmanach G. Biochemical properties and regulation of cathepsin k activity. Biochimie. 2008;90(2):208–226. doi: 10.1016/j.biochi.2007.08.011. [DOI] [PubMed] [Google Scholar]
- 39.Jault C., Pichon L., Chluba J. Toll-like receptor gene family and tir-domain adapters in danio rerio. Mol. Immunol. 2004;40(11):759–771. doi: 10.1016/j.molimm.2003.10.001. [DOI] [PubMed] [Google Scholar]
- 40.He J., Han S., Li X-X., Wang Q-Q., Cui Y., Chen Y., Gao H., Huang L., Yang S. Diethyl blechnic exhibits anti-inflammatory and antioxidative activity via the tlr4/myd88 signaling pathway in lps-stimulated raw264.7 cells. Molecules. 2019;24(24):4502. doi: 10.3390/molecules24244502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008;454(7203):428. doi: 10.1038/nature07201. [DOI] [PubMed] [Google Scholar]
- 42.Bäck M., Yurdagul A., Tabas I., Öörni K., Kovanen P.T. Inflammation and its resolution in atherosclerosis: Mediators and therapeutic opportunities. Nat. Rev. Cardiol. 2019;16(7):389–406. doi: 10.1038/s41569-019-0169-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Perkins D.J., Were T., Davenport G.C., Kempaiah P., Hittner J.B., Ong'echa J.M. Severe malarial anemia: Innate immunity and pathogenesis. Int. J. Biol. Sci. 2011;7(9):1427. doi: 10.7150/ijbs.7.1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shapouri-Moghaddam A., Mohammadian S., Vazini H., Taghadosi M., Esmaeili S.A., Mardani F., Seifi B., Mohammadi A., Afshari J.T., Sahebkar A. Macrophage plasticity, polarization, and function in health and disease. J. Cell. Physiol. 2018;233(9):6425–6440. doi: 10.1002/jcp.26429. [DOI] [PubMed] [Google Scholar]
- 45.Munford R.S., Pugin J. Normal responses to injury prevent systemic inflammation and can be immunosuppressive. Am. J. Respir. Crit. Care Med. 2001;163(2):316–321. doi: 10.1164/ajrccm.163.2.2007102. [DOI] [PubMed] [Google Scholar]
- 46.Lenz A., Franklin G.A., Cheadle W.G. Systemic inflammation after trauma. Injury. 2007;38(12):1336–1345. doi: 10.1016/j.injury.2007.10.003. [DOI] [PubMed] [Google Scholar]
- 47.Mauriz J.L., Collado P.S., Veneroso C., Reiter R.J. A review of the molecular aspects of melatonin’s anti-inflammatory actions: Recent insights and new perspectives. J. Pineal Res. 2013;54(1):1–14. doi: 10.1111/j.1600-079X.2012.01014.x. [DOI] [PubMed] [Google Scholar]
- 48.Tian R., Hou G., Li D., Yuan T-F. A possible change process of inflammatory cytokines in the prolonged chronic stress and its ultimate implications for health. Scientific World Journal. 2014;2014:780616. doi: 10.1155/2014/780616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kunz M., Ceresér K.M., Goi P.D., Fries G.R., Teixeira A.L., Fernandes B.S., Belmonte-de-Abreu P.S. Serum levels of il-6, il-10 and tnf-Α in patients with bipolar disorder and schizophrenia: Differences in pro-and anti-inflammatory balance. Br. J. Psychiatry. 2011;33(3):268–274. doi: 10.1590/s1516-44462011000300010. [DOI] [PubMed] [Google Scholar]
- 50.Steensberg A., Fischer C.P., Keller C., Møller K. Metabolism: Il-6 enhances plasma il-1ra, il-10, and cortisol in humans. 2003;285(2):E433–E437. doi: 10.1152/ajpendo.00074.2003. [DOI] [PubMed] [Google Scholar]
- 51.Ferrucci L., Fabbri E. Inflammageing: Chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 2018;15(9):505–522. doi: 10.1038/s41569-018-0064-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hu X., Liu S., Zhu J., Ni H. Dachengqi decoction alleviates acute lung injury and inhibits inflammatory cytokines production through tlr4/nf-κb signaling pathway in vivo and in vitro. J. Cell. Biochem. 2019;120(6):8956–8964. doi: 10.1002/jcb.27615. [DOI] [PubMed] [Google Scholar]
- 53.Alcorn J.L. Translational inflammation. Elsevier; 2019. Innate immunity and pulmonary inflammation: A balance between protection and disease. ; pp. 153–175. [Google Scholar]
- 54.Bannenberg G., Serhan C.N. Specialized pro-resolving lipid mediators in the inflammatory response.An update. Biochim. Biophys. Acta. 2010;1801(12):1260–1273. doi: 10.1016/j.bbalip.2010.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Salvemini D., Cuzzocrea S.J.Ccm. Therapeutic potential of superoxide dismutase mimetics as therapeutic agents in critical care medicine. 2003;31(1):S29–S38. doi: 10.1097/00003246-200301001-00005. [DOI] [PubMed] [Google Scholar]
- 56.Epidemiology Working Group for NCIP Epidemic Response, Chinese Center for Disease Control and Preventio. The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (covid-19) in China. Zhonghua Liu Xing Bing Xue Za Zhi. 2020;41(2):145–151. doi: 10.3760/cma.j.issn.0254-6450.2020.02.003. [DOI] [PubMed] [Google Scholar]
- 57.Supuran C.T., Casini A., Mastrolorenzo A., Scozzafava A. Cox-2 selective inhibitors, carbonic anhydrase inhibition and anticancer properties of sulfonamides belonging to this class of pharmacological agents. Mini Rev. Med. Chem. 2004;4(6):625–632. doi: 10.2174/1389557043403792. [DOI] [PubMed] [Google Scholar]
- 58.Kawanishi S., Ohnishi S., Ma N., Hiraku Y., Murata M. Crosstalk between DNA damage and inflammation in the multiple steps of carcinogenesis. Int. J. Mol. Sci. 2017;18(8):1808. doi: 10.3390/ijms18081808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Jia Z., He J. Paeoniflorin ameliorates rheumatoid arthritis in rat models through oxidative stress, inflammation and cyclooxygenase 2. Exp. Ther. Med. 2016;11(2):655–659. doi: 10.3892/etm.2015.2908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lee S-B., Lee W.S., Shin J-S., Jang D.S., Lee K.T. Xanthotoxin suppresses lps-induced expression of inos, cox-2, tnf-α, and il-6 via ap-1, nf-κb, and jak-stat inactivation in raw 264.7 macrophages. Int. Immunopharmacol. 2017;49:21–29. doi: 10.1016/j.intimp.2017.05.021. [DOI] [PubMed] [Google Scholar]
- 61.Gilroy D.W., Colville-Nash P., Willis D., Chivers J., Paul-Clark M., Willoughby D. Inducible cyclooxygenase may have anti-inflammatory properties. Nat. Med. 1999;5(6):698. doi: 10.1038/9550. [DOI] [PubMed] [Google Scholar]
- 62.Chen J-Y., Ye Z-X., Wang X-F., Chang J., Yang M-W., Zhong H-H., Hong F-F., Yang S-L. Nitric oxide bioavailability dysfunction involves in atherosclerosis. Biomed. Pharmacother. 2018;97:423–428. doi: 10.1016/j.biopha.2017.10.122. [DOI] [PubMed] [Google Scholar]
- 63.Yu X., Ge L., Niu L. longevity c: The dual role of inducible nitric oxide synthase in myocardial ischemia/reperfusion injury: Friend or foe? Oxid. Med. Cell. Longev. 2018;7 doi: 10.1155/2018/8364848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kapoor Y., Kumar K. Medicinal and chemical perspectives of nitric oxide: An overview. SF J. Pharm. Anal. Chem. 2019;2(1):1015. [Google Scholar]
- 65.Sasaki M., Kodama Y., Shimoyama Y., Ishikawa T., Kimura S. Aciduricity and acid tolerance mechanisms of streptococcus anginosus. J. Gen. Appl. Microbiol. 2018;64(4):174–179. doi: 10.2323/jgam.2017.11.005. [DOI] [PubMed] [Google Scholar]
- 66.Nafees M., Fahad S., Shah A.N., Bukhari M.A., Ahmed I., Ahmad S., Hussain S. Plant abiotic stress tolerance. Springer; 2019. Reactive oxygen species signaling in plants. ; pp. 259–272. [Google Scholar]
- 67.Ozcan A., Ogun M. Biochemistry of reactive oxygen and nitrogen species. . Basic principles and clinical significance of oxidative stress. 2015;3:37–58. [Google Scholar]
- 68.Forrester S.J., Kikuchi D.S., Hernandes M.S., Xu Q. Reactive oxygen species in metabolic and inflammatory signaling. Circ. Res. 2018;122(6):877–902. doi: 10.1161/CIRCRESAHA.117.311401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Grimm A., Friedland K., Eckert A.J. Mitochondrial dysfunction: The missing link between aging and sporadic alzheimer’s disease. Biogerontology. 2016;17(2):281–296. doi: 10.1007/s10522-015-9618-4. [DOI] [PubMed] [Google Scholar]
- 70.Han S., Gao H., Chen S., Wang Q., Li X., Du L-J., Li J., Luo Y-Y., Li J-X., Zhao L.C., Feng J., Yang S. Procyanidin a1 alleviates inflammatory response induced by lps through nf-κb, mapk, and nrf2/ho-1 pathways in raw264.7 cells. Sci. Rep. 2019;9(1):1–13. doi: 10.1038/s41598-019-51614-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Afonina I.S., Zhong Z., Karin M., Beyaert R. Limiting inflammation-the negative regulation of nf-κb and the nlrp3 inflammasome. Nat. Immunol. 2017;18(8):861–869. doi: 10.1038/ni.3772. [DOI] [PubMed] [Google Scholar]
- 72.Cantrell D. Signaling in lymphocyte activation. Cold Spring Harb. Perspect. Biol. 2015;7(6):a018788. doi: 10.1101/cshperspect.a018788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Yang Y., Wang J.K. The functional analysis of micrornas involved in nf-κb signaling. Eur. Rev. Med. Pharmacol. Sci. 2016;20(9):1764–1774. [PubMed] [Google Scholar]
- 74.Shi J., Hu H., Harnett J., Zheng X., Liang Z., Wang Y-T., Ung C.O.L. An evaluation of randomized controlled trials on nutraceuticals containing traditional Chinese medicines for diabetes management: a systematic review. Chin. Med. 2019;14(1):54. doi: 10.1186/s13020-019-0276-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Dedic N., Chen A., Deussing J.M. The crf family of neuropeptides and their receptors - mediators of the central stress response. Curr. Mol. Pharmacol. 2018;11(1):4–31. doi: 10.2174/1874467210666170302104053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wu B., Wang R., Li S., Wang Y., Song F., Gu Y., Yuan Y.J.P.R. Antifibrotic effects of fraxetin on carbon tetrachloride-induced liver fibrosis by targeting nf-κb/iκbα, mapks and bcl-2/bax pathways. Pharmacol. Rep. 2019;71(3):409–416. doi: 10.1016/j.pharep.2019.01.008. [DOI] [PubMed] [Google Scholar]
- 77.Chandra S., Stanford D., Fletcher E., Walker L.A. The science and regulations of naturally derived complex drugs. Springer; 2019. Raw materials production and manufacturing process control strategies. ; pp. 175–190. [Google Scholar]
- 78.Chen Z., Zhang C., Gao F., Fu Q., Fu C., He Y., Zhang J.J.F. A systematic review on the rhizome of ligusticum chuanxiong hort.(chuanxiong). Food Chem. Toxicol. 2018;119:309–325. doi: 10.1016/j.fct.2018.02.050. [DOI] [PubMed] [Google Scholar]
- 79.Ye L., Cao Z., Wang W., Zhou N. New insights in cannabinoid receptor structure and signaling. Curr. Mol. Pharmacol. 2019;12(3):239–248. doi: 10.2174/1874467212666190215112036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Mothes J., Busse D., Kofahl B., Wolf J.J.B. Sources of dynamic variability in nf-κb signal transduction: A mechanistic model. BioEssays. 2015;37(4):452–462. doi: 10.1002/bies.201400113. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.