Abstract
A 12-year-old male was presented to the hospital with acute encephalopathy, headache, vomiting, diarrhea, and elevated troponin after recent COVID-19 vaccination. Two days prior to admission and before symptom onset, he received the second dose of the Pfizer-BioNTech COVID-19 vaccine. Symptoms developed within 24 h with worsening neurologic symptoms, necessitating admission to the pediatric intensive care unit. Brain magnetic resonance imaging within 16 h of admission revealed a cytotoxic splenial lesion of the corpus callosum (CLOCC). Nineteen days prior to admission, he developed erythema migrans, and completed an amoxicillin treatment course for clinical Lyme disease. However, Lyme antibody titers were negative on admission and nine days later, making active Lyme disease an unlikely explanation for his presentation to hospital. An extensive workup for other etiologies on cerebrospinal fluid and blood samples was negative, including infectious and autoimmune causes and known immune deficiencies. Three weeks after hospital discharge, all of his symptoms had dissipated, and he had a normal neurologic exam. Our report highlights a potential role of mRNA vaccine-induced immunity leading to MIS-C-like symptoms with cardiac involvement and a CLOCC in a recently vaccinated child and the complexity of establishing a causal association with vaccination. The child recovered without receipt of immune modulatory treatment.
Keywords: cytotoxic lesion of the corpus callosum, severe acute respiratory syndrome coronavirus 2, multisystem inflammatory syndrome in children, COVID-19 mRNA vaccine
1. Introduction
Encephalopathy with cytotoxic lesions of the corpus callosum (CLOCC) is increasingly being recognized in association with COVID-19 in children and adolescents [1,2,3]. CLOCCs are non-specific, non-enhancing areas of reduced diffusivity, indicating cytotoxic edema of non-vascular origin that have been observed in many pediatric infectious and non-infectious diseases [4,5,6]. CLOCCs have also been detected following mumps vaccination, which is an attenuated live virus [7,8].
Multisystem inflammatory syndrome in children (MIS-C) is a severe hyperinflammatory syndrome that temporally follows SARS-CoV-2 infection by 3–6 weeks and is presumably post-infectious [9]. As of 1 November 2021, there have been 5973 US cases of MIS-C associated with SARS-CoV-2 and 52 MIS-C-related deaths [10]. The Brighton Collaboration Working group recently proposed a case definition for MIS-C following COVID-19 vaccination [11]. MIS-C-like symptoms following COVID-19 mRNA vaccination have been reported in pediatric patients [12,13], neither had neurologic involvement. We conducted an extensive evaluation of a 12-year-old boy who developed neurologic and cardiac involvement following his second COVID-19 vaccination.
2. Case Report
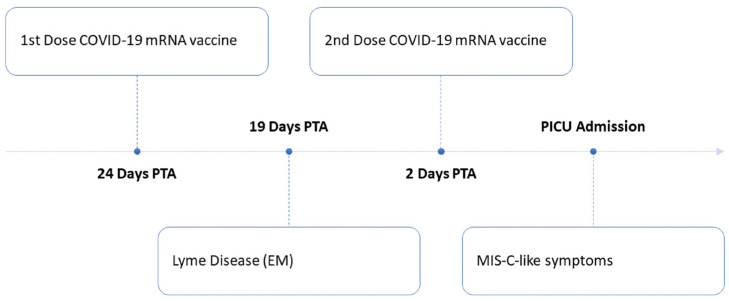
We report a MIS-C-like illness in a 12-year-old boy naïve to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) who developed encephalopathy and myocarditis following COVID-19 mRNA vaccination and was found to have an isolated CLOCC. The child lives in an endemic area for Lyme disease. Nineteen days prior to presentation (five days after his first COVID-19 vaccine), he developed a lesion consistent with erythema migrans (EM), and completed amoxicillin treatment for a clinical diagnosis of Lyme disease. He received the second dose of the Pfizer-BioNTech COVID-19 vaccine two days prior to hospital presentation. That night, he developed severe headache, and over the next 48 h had persistent headache, emesis, visual hallucinations, worsening encephalopathy and was noted to have an elevated troponin indicating cardiac involvement. A timeline of the development of MIS-C-like symptoms after Lyme disease and COVID-19 vaccination is shown in Figure 1. He was admitted to the pediatric intensive care unit for close neurologic and cardiac monitoring and underwent brain magnetic resonance imaging (MRI).
Figure 1.
Timeline of development of MIS-C-like symptoms after Lyme disease and COVID-19 mRNA vaccination. Abbreviations: EM, erythema migrans; PICU, pediatric intensive care unit; PTA, prior to admission.
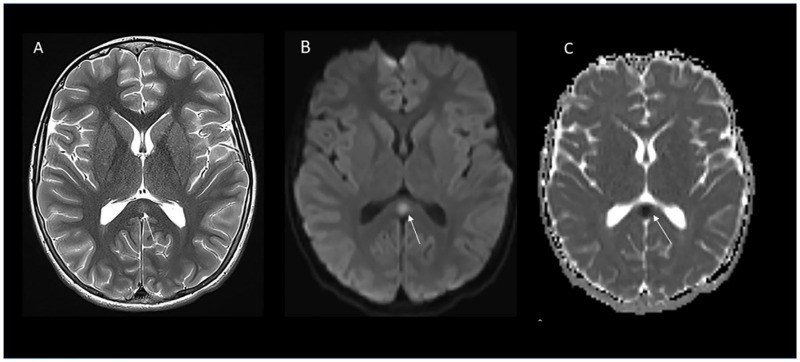
Pertinent vital signs and laboratory studies during admission were: temperature 38.3 degrees Centigrade, hyponatremia (125 mmol/L), elevated C-reactive protein (5.8 mg/dL), elevated troponin T (0.22 ng/mL), neutrophilia (9840, normal 4520–9170 cells/μL) and lymphopenia (1080, normal 1490–3110 cells/μL). He had no immunologic markers of acute or recent infection, as evidenced by no elevation of effector/memory populations within CD4+ or CD8+ T cells and a normal level of soluble CD25, a marker of T-cell activation that is increased in viral, bacterial, and tick-borne infections. On the second hospital day, BNP peaked at 190 pg/mL (100 pg/mL upper limit of normal). Neurologic examination on admission was focal with a positive Babinski response on the left. Cerebrospinal fluid (CSF) analysis was not suggestive of encephalitis. Brain MRI showed T2 prolongation and reduced diffusivity in the splenium of the corpus callosum in Figure 2. EEG showed intermittent right posterior slowing without epileptiform activity to suggest an underlying diagnosis of epilepsy. No specific immunomodulatory therapies were administered to our patient because of normal cardiac MRI and reassuring clinical trajectory and improving cardiac and inflammatory biomarkers. He was discharged on hospital day five. Diagnostic evaluation results are summarized in Table 1. Three weeks post discharge, he was asymptomatic with a normal neurologic exam. Whole-genome sequencing did not identify any coding variants indicative of an underlying immune disorder.
Figure 2.
Cytotoxic lesion of the corpus callosum. Axial T2 image (A) shows focus of T2 prolongation representing cytotoxic edema in the splenium of corpus callosum (arrow) with reduced diffusivity on trace diffusion image (B) (arrow) and apparent diffusion coefficient map (C).
Table 1.
Diagnostic studies and results during hospital admission.
| Laboratory Studies | Source | Result (Normal Range) |
|---|---|---|
| Infectious Disease | ||
| SARS-CoV2 PCR | NP swab | Negative |
| Anti-SARS-CoV2 nucleocapsid antibodies | Blood | Negative |
| Culture and gram stain | CSF | Negative |
| Adenovirus PCR | Blood, NP swab | Negative |
| Enterovirus PCR | Blood, CSF | Negative |
| Herpes Simplex Virus type 1 and type 2 | CSF | Negative |
| Varicella Zoster Virus PCR | CSF | Negative |
| Epstein Barr Virus PCR | Blood | Negative |
| Coxsackie A9 antibodies | Blood | Negative |
| Ehrlichia and Anaplasma PCR | Blood | Negative |
| Rickettsia rickettsia serologies, IgM, IgG | Blood | Negative |
| Eastern Equine Encephalitis IgM | Blood | Negative |
| West Nile Virus IgM | Blood | Negative |
| Pneumococcus IgG | Blood | Negative |
| Tetanus IgG | Blood | Negative |
| RVP (Adenovirus PCR, hMPV PCR, Rhinovirus PCR, Influenza A and B PCR, RSV PCR) | NP swab | Negative |
| Legionella antigen | Urine | Negative |
| Lyme antibody index | CSF | Negative |
| Neurologic | ||
| Anti-MOG antibodies | Blood, CSF | Negative |
| Autoimmune encephalitis panel | Blood, CSF | Negative |
| CNS demyelinating disease panel # | Blood | Negative |
| Cardiac | ||
| Viral Respiratory Panel (Myocarditis) | NP Swab | Negative |
| Electrocardiogram (ECG) | N/A | Widespread repolarization abnormalities, with nonspecific ST-T wave changes |
| Echocardiogram | N/A | Normal systolic and diastolic ventricular function, normal coronary dimensions, no significant valvar dysfunction, and no pericardial effusion |
| Cardiac MRI | N/A | No evidence of active myocarditis or late gadolinium enhancement |
| Immunology * | ||
| Soluble CD25, units/mL | Blood | 690 (137–838) |
| Serum inflammatory markers ^,* | ||
| D-dimer, mcg/mL FEU | Blood | 0.43 |
| Erythrocyte sedimentation rate, mm/h | Blood | 5 |
| Platelet count, K cells/μL | Blood | 313 |
| Other * | ||
| White blood, cells/mm3 | CSF | 2 with 7% neutrophils, 35% lymphocytes, 58% macrophages |
| Red blood cells | CSF | 0 |
| Protein, mg/dL | CSF | 14.9 |
| Glucose, mg/dL | CSF | 85 |
| Opening pressure, cmH2O | CSF | 21 |
| Prothrombin time, seconds | Blood | 15.7 (12.1–14.6) |
| AST, unit/L | Blood | 24 (2–40) |
| ALT, unit/L | Blood | 35 (3–30) |
Abbreviations: SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; NP, nasopharyngeal; CSF, cerebrospinal fluid; PCR, polymerase chain reaction; RVP, respiratory viral panel; hMPV, human metapneumovirus; RSV, respiratory syncytial virus; MOG, myelin oligodendrocyte glycoprotein; N/A, not applicable. # Includes antibodies to NMO/AQP4 (Neuromyelitis Optica/aquaporin-4) and MOG, sent 3 weeks after hospital discharge. * Maximum values during hospital admission. ^ Serum levels of procalcitonin, ferritin, lactate dehydrogenase, fibrinogen and IL6 and CSF IL-6 and IL-10 were not measured.
On admission, COVID-19 IgG (anti-SARS-CoV-2 spike) index by semi-quantitative chemiluminescent assay (ARUP Laboratories, SLC, UT) was elevated (19.83 IV, reference interval <= 0.99) whereas SARS-CoV-2 anti-nucleocapsid antibodies and respiratory swab PCR were negative, suggesting that antibodies were from vaccination and not infection. First tier Whole Cell Sonicate Lyme enzyme immunoassay (EIA) index values at time of admission and 9 days after admission were negative (0.24 acute and 0.70 convalescent).
3. Discussion
We present the first published report of a complex multisystem inflammatory disorder with cardiac and neurologic involvement in a 12-year-old boy naïve to SARS-CoV2 associated with the second dose of the Pfizer BioNTech vaccine. Although he had recent clinical Lyme disease, we do not believe it explains his MIS-C-like syndrome. Our patient’s clinical presentation has similarities with previous reports of CLOCCs in children in the setting of COVID-19 or MIS-C [1], probable myocarditis after COVID-19 vaccination in children [14,15,16], and CLOCC in a previously healthy young male after initial COVID-19 mRNA vaccination [17].
Clinical features of children with CLOCCs in association with SARS-CoV-2 and other infectious diseases typically include fever, altered consciousness (acute encephalopathy, delirium or coma), visual hallucinations, hyponatremia, and seizures [5]. Cerebrospinal fluid analysis may be normal or reveal mild pleocytosis [7,18,19,20]. Magnetic resonance imaging shows T2 prolongation in the splenium and/or genu of the corpus callosum with reduced diffusivity with or without white matter abnormalities [1]. Most patients with CLOCCs secondary to acute infections are either asymptomatic [21], or have transient neurologic symptoms with complete recovery within a few weeks with or without immunotherapies [22,23]. The pathogenesis of encephalopathy with CLOCCs remains unclear. CLOCCs are thought to be associated with increased density of cytokine and glutamate receptors in the corpus callosum, particularly the splenium, leading to cytokinopathy, increased glutamate in the extracellular space, and cytotoxic edema in the corpus callosum [1,4,24,25]. This has been corroborated by measures of elevated IL-6 and IL-10 in CSF in children with splenial lesions [19,26,27].
Following the FDA expansion of the Emergency Use Authorization of 2 Pfizer-BioNTech mRNA COVID-19 vaccines to include adolescents 12 to 15 years of age in May 2021, cases of post COVID-19 vaccination myocarditis and pericarditis have been reported to the U.S. Centers for Disease Control and Federal Drug Administration in this age group [28,29]. In addition to myocarditis in our patient, the diagnosis of probable post-vaccine induced MIS-C may be supported by the following features according to the Brighton criteria (Level 2b) [11]: fever for at least one day prior to admission; acute neurologic and gastrointestinal symptoms; laboratory markers of inflammation (CRP elevated); neutrophilia, lymphopenia; and elevated cardiac biomarkers (troponin and BNP levels). In the present case, SARS-CoV-2 PCR and nucleocapsid antibodies were negative, highlighting lack of SARS-CoV-2 exposure. Our patient with encephalopathy and focal neurological signs on admission, focal EEG slowing and normal CSF analysis is similar to recently reported cases of acute encephalitis in the setting of MIS-C [30].
Toll-like receptors, which are activated by mRNA vaccines, cause significant changes in cytokine and gene expression beginning within hours after receptor activation, peak within 6 to 24 h, and subside over the following 48 to 168 h [31], consistent with this patient’s disease timeline. It is unlikely that Borrelia burgdorferi infection alone caused the MIS-like illness. The patient’s Lyme disease diagnosis was based on the history of an EM lesion with negative acute and convalescent serology. In endemic areas, the rash alone is diagnostic of Lyme disease [32] as the sensitivity of serology is low with cutaneous disease [33,34,35] and patients may remain seronegative after appropriate treatment [36]. However, infectious pathogens may cause reversible, epigenetic reprogramming of the innate immune response prompting an atypical response to a subsequent immune challenge [37].
4. Conclusions
Our report highlights a potential role of vaccine-induced immunity leading to MIS-C-like symptoms with cardiac involvement and CLOCC in a recently vaccinated child who had an antecedent Lyme infection. Given the rarity and atypical nature of his clinical presentation, his symptoms likely arose from a complex interplay amongst multiple factors: his genetic background, recent Lyme infection, and the second dose of the COVID-19 mRNA vaccine. Importantly, the self-limited and brief course of our patient’s symptoms contrast with the sequelae of acute COVID-19, MIS-C, and post-acute sequelae of SARS-CoV-2 infection. Thus, while this case report of a rare complication expands knowledge of possible vaccine-associated events in children with recent Lyme disease, in isolation it should not be interpreted as a contraindication to SARS-CoV-2 vaccination in children.
Author Contributions
Conceptualization and data curation, T.Y.P., K.L.L., J.W.N., J.C., L.E.N., T.N. and A.G.R. Writing—original draft preparation, K.L.L., A.G.R. and T.Y.P. Writing—review and editing, T.Y.P., K.L.L., J.W.N., J.C., L.E.N., T.N. and A.G.R. All authors have read and agreed to the published version of the manuscript.
Funding
The U.S. Centers for Disease Control and Prevention funded the Overcoming COVID-19 Immunobiology Study (contract #75D30121C10297 to A.G.R.) that this patient consented to participate in contributing data and samples. This work represents the findings and conclusions of the authors and not the U.S. Centers for Disease Control and Prevention.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of Boston Children’s Hospital (IRB-P00033157 with approval date 27 May 2020).
Informed Consent Statement
Informed consent was obtained from the parents of the patient involved in the study. Written informed consent has been obtained from the parents to publish this paper.
Data Availability Statement
All available supportive data are reported in this case report.
Conflicts of Interest
Adrienne Randolph, Janet Chou, Tanya Novak, Tina Young Poussaint and Jane Newburger receive funding from the Centers for Disease Control and Prevention as a contract to the institution for work related to COVID-19 vaccine effectiveness and COVID-19-related complications in children and adolescents including neurologic sequelae of MIS-C. Jane Newburger has grant funding from the NHLBI for the MUSIC study: Long-TerM OUtcomes after the Multisystem Inflammatory Syndrome In Children. Janet Chou has grant funding from NIAID. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.LaRovere K.L., Riggs B.J., Poussaint T.Y., Young C.C., Newhams M.M., Maamari M., Walker T.C., Singh A.R., Dapul H., Hobbs C.V., et al. Neurologic Involvement in Children and Adolescents Hospitalized in the United States for COVID-19 or Multisystem Inflammatory Syndrome. JAMA Neurol. 2021;78:536–547. doi: 10.1001/jamaneurol.2021.0504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lindan C.E., Mankad K., Ram D., Kociolek L.K., Silvera V.M., Boddaert N., Stivaros S.M., Palasis S., Group A.P.C. Neuroimaging manifestations in children with SARS-CoV-2 infection: A multinational, multicentre collaborative study. Lancet Child. Adolesc. Health. 2021;5:167–177. doi: 10.1016/S2352-4642(20)30362-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Abdel-Mannan O., Eyre M., Lobel U., Bamford A., Eltze C., Hameed B., Hemingway C., Hacohen Y. Neurologic and Radiographic Findings Associated With COVID-19 Infection in Children. JAMA Neurol. 2020;77:1440–1445. doi: 10.1001/jamaneurol.2020.2687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Starkey J., Kobayashi N., Numaguchi Y., Moritani T. Cytotoxic Lesions of the Corpus Callosum That Show Restricted Diffusion: Mechanisms, Causes, and Manifestations. Radiographics. 2017;37:562–576. doi: 10.1148/rg.2017160085. [DOI] [PubMed] [Google Scholar]
- 5.Kashiwagi M., Tanabe T., Shimakawa S., Nakamura M., Murata S., Shabana K., Shinohara J., Odanaka Y., Matsumura H., Maki K., et al. Clinico-radiological spectrum of reversible splenial lesions in children. Brain Dev. 2014;36:330–336. doi: 10.1016/j.braindev.2013.05.007. [DOI] [PubMed] [Google Scholar]
- 6.Chen W.X., Liu H.S., Yang S.D., Zeng S.H., Gao Y.Y., Du Z.H., Li X.J., Lin H.S., Liang H.C., Mai J.N. Reversible splenial lesion syndrome in children: Retrospective study and summary of case series. Brain Dev. 2016;38:915–927. doi: 10.1016/j.braindev.2016.04.011. [DOI] [PubMed] [Google Scholar]
- 7.Takanashi J., Shiihara T., Hasegawa T., Takayanagi M., Hara M., Okumura A., Mizuguchi M. Clinically mild encephalitis with a reversible splenial lesion (MERS) after mumps vaccination. J. Neurol. Sci. 2015;349:226–228. doi: 10.1016/j.jns.2014.12.019. [DOI] [PubMed] [Google Scholar]
- 8.Hara M., Mizuochi T., Kawano G., Koike T., Shibuya I., Ohya T., Ohbu K., Nagai K., Nagamitsu S., Yamashita Y., et al. A case of clinically mild encephalitis with a reversible splenial lesion (MERS) after mumps vaccination. Brain Dev. 2011;33:842–844. doi: 10.1016/j.braindev.2010.12.013. [DOI] [PubMed] [Google Scholar]
- 9.Centers for Disease Control and Prevention Information for Healthcare Providers about Multisystem Inflammatory Syndrome in Children (MIS-C) [(accessed on 26 December 2021)]; Available online: https://www.cdc.gov/mis-c/hcp/
- 10.Centers for Disease Control and Prevention Multisystem Inflammatory Syndrome (MIS) [(accessed on 26 December 2021)]; Available online: https://covid.cdc.gov/covid-data-tracker/#mis-national-surveillance.
- 11.Vogel T.P., Top K.A., Karatzios C., Hilmers D.C., Tapia L.I., Moceri P., Giovannini-Chami L., Wood N., Chandler R.E., Klein N.P., et al. Multisystem inflammatory syndrome in children and adults (MIS-C/A): Case definition & guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine. 2021;39:3037–3049. doi: 10.1016/j.vaccine.2021.01.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chai Q., Nygaard U., Schmidt R.C., Zaremba T., Moller A.M., Thorvig C.M. Multisystem inflammatory syndrome in a male adolescent after his second Pfizer-BioNTech COVID-19 vaccine. Acta Paediatr. 2022;111:125. doi: 10.1111/apa.16141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Buchhorn R., Meyer C., Schulze-Forster K., Junker J., Heidecke H. Autoantibody Release in Children after Corona Virus mRNA Vaccination: A Risk Factor of Multisystem Inflammatory Syndrome? Vaccines. 2021;9:1353. doi: 10.3390/vaccines9111353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dionne A., Sperotto F., Chamberlain S., Baker A.L., Powell A.J., Prakash A., Castellanos D.A., Saleeb S.F., de Ferranti S.D., Newburger J.W., et al. Association of Myocarditis With BNT162b2 Messenger RNA COVID-19 Vaccine in a Case Series of Children. JAMA Cardiol. 2021;6:1446–1450. doi: 10.1001/jamacardio.2021.3471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gargano J.W., Wallace M., Hadler S.C., Langley G., Su J.R., Oster M.E., Broder K.R., Gee J., Weintraub E., Shimabukuro T., et al. Use of mRNA COVID-19 Vaccine After Reports of Myocarditis Among Vaccine Recipients: Update from the Advisory Committee on Immunization Practices-United States, June 2021. MMWR Morb. Mortal. Wkly. Rep. 2021;70:977–982. doi: 10.15585/mmwr.mm7027e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bozkurt B., Kamat I., Hotez P.J. Myocarditis With COVID-19 mRNA Vaccines. Circulation. 2021;144:471–484. doi: 10.1161/CIRCULATIONAHA.121.056135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Youn T., Yang H. Cytotoxic Lesion of the Corpus Callosum (CLOCCs) after SARS-CoV-2 mRNA Vaccination. J. Korean Med. Sci. 2021;36:e228. doi: 10.3346/jkms.2021.36.e228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ka A., Britton P., Troedson C., Webster R., Procopis P., Ging J., Chua Y.W., Buckmaster A., Wood N., Jones C., et al. Mild encephalopathy with reversible splenial lesion: An important differential of encephalitis. Eur. J. Paediatr. Neurol. 2015;19:377–382. doi: 10.1016/j.ejpn.2015.01.011. [DOI] [PubMed] [Google Scholar]
- 19.Yuan Z.F., Shen J., Mao S.S., Yu Y.L., Xu L., Jiang P.F., Gao F., Xia Z.Z. Clinically mild encephalitis/encephalopathy with a reversible splenial lesion associated with Mycoplasma pneumoniae infection. BMC Infect. Dis. 2016;16:230. doi: 10.1186/s12879-016-1556-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fluss J., Ferey S., Menache-Starobinski C., Delavelle J., Van Bogaert P., Vargas M.I. Mild influenza-associated encephalopathy/encephalitis with a reversible splenial lesion in a Caucasian child with additional cerebellar features. Eur. J. Paediatr. Neurol. 2010;14:97–100. doi: 10.1016/j.ejpn.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 21.Tsuji M., Yoshida T., Miyakoshi C., Haruta T. Is a reversible splenial lesion a sign of encephalopathy? Pediatr. Neurol. 2009;41:143–145. doi: 10.1016/j.pediatrneurol.2009.02.019. [DOI] [PubMed] [Google Scholar]
- 22.Bulakbasi N., Kocaoglu M., Tayfun C., Ucoz T. Transient splenial lesion of the corpus callosum in clinically mild influenza-associated encephalitis/encephalopathy. Am. J. Neuroradiol. 2006;27:1983–1986. [PMC free article] [PubMed] [Google Scholar]
- 23.Pan J.J., Zhao Y.Y., Lu C., Hu Y.H., Yang Y. Mild encephalitis/encephalopathy with a reversible splenial lesion: Five cases and a literature review. Neurol. Sci. 2015;36:2043–2051. doi: 10.1007/s10072-015-2302-2. [DOI] [PubMed] [Google Scholar]
- 24.Rasmussen C., Niculescu I., Patel S., Krishnan A. COVID-19 and Involvement of the Corpus Callosum: Potential Effect of the Cytokine Storm? Am. J. Neuroradiol. 2020;41:1625–1628. doi: 10.3174/ajnr.A6680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Moonis G., Filippi C.G., Kirsch C.F.E., Mohan S., Stein E.G., Hirsch J.A., Mahajan A. The Spectrum of Neuroimaging findings on CT and MRI in Adults with Coronavirus Disease (COVID-19) Am. J. Roentgenol. 2020;217:959–974. doi: 10.2214/AJR.20.24839. [DOI] [PubMed] [Google Scholar]
- 26.Miyata R., Tanuma N., Hayashi M., Imamura T., Takanashi J., Nagata R., Okumura A., Kashii H., Tomita S., Kumada S., et al. Oxidative stress in patients with clinically mild encephalitis/encephalopathy with a reversible splenial lesion (MERS) Brain Dev. 2012;34:124–127. doi: 10.1016/j.braindev.2011.04.004. [DOI] [PubMed] [Google Scholar]
- 27.Kometani H., Kawatani M., Ohta G., Okazaki S., Ogura K., Yasutomi M., Tanizawa A., Ohshima Y. Marked elevation of interleukin-6 in mild encephalopathy with a reversible splenial lesion (MERS) associated with acute focal bacterial nephritis caused by Enterococcus faecalis. Brain Dev. 2014;36:551–553. doi: 10.1016/j.braindev.2013.07.012. [DOI] [PubMed] [Google Scholar]
- 28.Clinical Considerations: Myocarditis and Pericarditis after Receipt of mRNA COVID-19 Vaccines among Adolescents and Young Adults. [(accessed on 10 December 2021)]; Available online: https://www.cdc.gov/vaccines/covid-19/clinical-considerations/myocarditis.html.
- 29.Myocarditis and Pericarditis Following mRNA COVID-19 Vaccination. [(accessed on 10 December 2021)]; Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/myocarditis.html.
- 30.Olivotto S., Basso E., Lavatelli R., Previtali R., Parenti L., Fiori L., Dilillo D., Zuccotti G.V., Veggiotti P., Bova S.M. Acute encephalitis in pediatric multisystem inflammatory syndrome associated with COVID-19. Eur. J. Paediatr. Neurol. 2021;34:84–90. doi: 10.1016/j.ejpn.2021.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Teijaro J.R., Farber D.L. COVID-19 vaccines: Modes of immune activation and future challenges. Nat. Rev. Immunol. 2021;21:195–197. doi: 10.1038/s41577-021-00526-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Schwartz A.M., Hinckley A.F., Mead P.S., Hook S.A., Kugeler K.J. Surveillance for Lyme Disease-United States, 2008–2015. MMWR Surveill. Summ. 2017;66:1–12. doi: 10.15585/mmwr.ss6622a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Aguero-Rosenfeld M.E., Nowakowski J., McKenna D.F., Carbonaro C.A., Wormser G.P. Serodiagnosis in early Lyme disease. J. Clin. Microbiol. 1993;31:3090–3095. doi: 10.1128/jcm.31.12.3090-3095.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Steere A.C., McHugh G., Damle N., Sikand V.K. Prospective study of serologic tests for lyme disease. Clin. Infect. Dis. 2008;47:188–195. doi: 10.1086/589242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lantos P.M., Rumbaugh J., Bockenstedt L.K., Falck-Ytter Y.T., Aguero-Rosenfeld M.E., Auwaerter P.G., Baldwin K., Bannuru R.R., Belani K.K., Bowie W.R., et al. Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA), American Academy of Neurology (AAN), and American College of Rheumatology (ACR): 2020 Guidelines for the Prevention, Diagnosis and Treatment of Lyme Disease. Clin. Infect. Dis. 2021;72:e1–e48. doi: 10.1093/cid/ciaa1215. [DOI] [PubMed] [Google Scholar]
- 36.Rebman A.W., Crowder L.A., Kirkpatrick A., Aucott J.N. Characteristics of seroconversion and implications for diagnosis of post-treatment Lyme disease syndrome: Acute and convalescent serology among a prospective cohort of early Lyme disease patients. Clin. Rheumatol. 2015;34:585–589. doi: 10.1007/s10067-014-2706-z. [DOI] [PubMed] [Google Scholar]
- 37.Divangahi M., Aaby P., Khader S.A., Barreiro L.B., Bekkering S., Chavakis T., van Crevel R., Curtis N., DiNardo A.R., Dominguez-Andres J., et al. Trained immunity, tolerance, priming and differentiation: Distinct immunological processes. Nat. Immunol. 2021;22:2–6. doi: 10.1038/s41590-020-00845-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All available supportive data are reported in this case report.