Abstract
The nervous system has been increasingly recognized as a novel and accessible target in the regulation of inflammation. The use of implantable and invasive devices targeting neural circuits has yielded successful results in clinical settings but does have some risk or adverse effects. Recent advances in technology and understanding of mechanistic pathways have opened new avenues of non-invasive neuromodulation. Through this review we discuss the novel research and outcomes of major modalities of non-invasive neuromodulation in the context of inflammation including transcutaneous electrical, magnetic and ultrasound neuromodulation. In addition to highlighting the scientific observations and breakthroughs, we discuss the underlying mechanisms and pathways for neural regulation of inflammation.
Keywords: auricular, inflammatory reflex, ultrasound, vagus
Non-invasive neuromodulation to control inflammation
Introduction
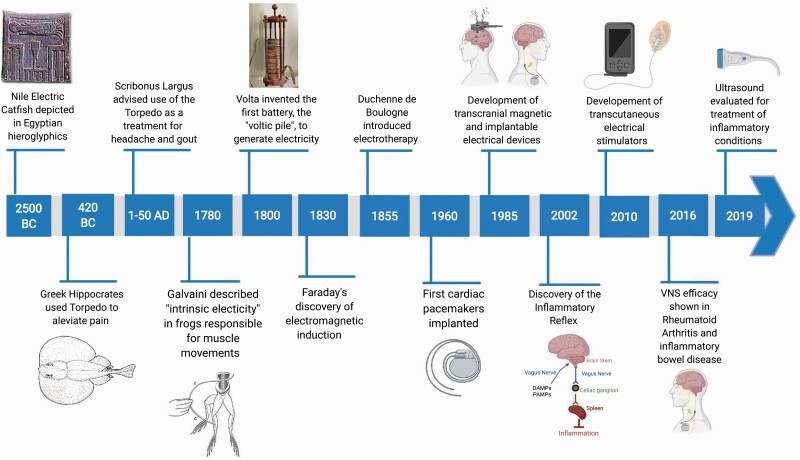
Reducing inflammation and pain with non-invasive electrical neuromodulation can be traced back for about 5000 years, as Egyptian tomb reliefs (ca B.C.E. 2500) prominently feature the Nile electric catfish (Malopturus) and hint at its use for painful conditions (1) (Fig. 1). The effects of ‘natural electricity’ from contact with the electric ray (Torpedo) were noted by Hippocrates and Aristotle to cause a numbing effect (2, 3). The first person known to have been cured of an inflammatory condition by electricity was Anteros, a court official of Emperor Tiberius (42 B. C. E. to 37 C.E.). While walking on a seashore, he accidentally stepped on a Torpedo, and received a strong electric shock (Torpedo voltages range up to 225 volts). After the numbing effect of the shock subsided, Anteros realized that he has been freed from his gout, a type of arthritis (4).
Fig. 1.
Timeline indicating the important developments in neuromodulation therapy (Created with BioRender.com).
Referring to this incident, Scribonius Largus (1–50 C.E.), the court physician to Roman emperor Claudius, suggested the use of electric shocks from the Torpedo as a therapy for arthritis (4), making the electric fish the first non-invasive neuromodulation device employed by humans to treat inflammatory diseases. Although Torpedo were in use for a long time for the regulation of inflammatory conditions, recent studies at the intersection of immunology, neuroscience and bioelectronic devices have provided important insights into the molecular mechanisms of neuro-immune communication that underlie its efficacy (5, 6).
Inflammation is a dynamic protective immune response mechanism against endogenous cell damage, toxic/metabolic insults and pathogens. While essential for eliminating the inciting stimulus, inflammation promotes tissue healing and, in the case of infection, establishes immunological memory. However, unresolved or excessive inflammation can be deleterious, resulting in autoimmune or autoinflammatory disorders (7). Immune dysregulation and aberrant inflammation also play a major role in obesity, type 2 diabetes, metabolic syndrome and cancer pathogenesis affecting millions of people worldwide (8–10).
Identifying the molecular mechanisms of inflammatory responses has resulted in the development of an array of pharmaceutical and biological agents to treat inflammation, accounting for a multi-billion dollar drug industry (11, 12). Standard therapies include glucocorticoids, methotrexate, monoclonal antibodies and other pharmacological agents targeting inflammatory pathways (12). In spite of the availability of these therapies, a significant number of patients are either unresponsive or become resistant (13). Even more concerning are the adverse side-effects of treatment, including increased susceptibility to infections, elevated risk of malignancy and off-target inflammatory responses (14–16). Accordingly, there is an unmet clinical need for alternative therapies that can directly treat inflammatory conditions.
Neuromodulation as therapy
Bioelectronic devices that harness electrical neuromodulation have been used to treat diverse diseases for decades. Based on the sequential discoveries of bioelectricity by Galvani (1780), the battery by Volta (1799) and electromagnetic induction by Faraday (1831), a variety of invasive and non-invasive electrical stimulators have been developed for the treatment of specific diseases (Fig. 1). For example, deep-brain stimulation improves quality of life for patients with depression or Parkinson’s disease (17, 18); sacral-nerve stimulation helps people with bowel and bladder problems (19); pacemakers and defibrillators have revolutionized the treatment of patients with cardiac arrhythmias (20); and vagus-nerve stimulation (VNS) has been used to treat pharmaco-resistant seizures for more than 30 years (21).
Recently, VNS has shown efficacy in modulating inflammatory diseases such as rheumatoid arthritis (RA) and Crohn’s disease (22, 23), following the discovery of a physiological mechanism accounting for its efficacy: the inflammatory reflex (Fig. 1) (24–26). The inflammatory reflex is a vagus nerve-based neural circuit in which afferent vagus nerve signaling activated by inflammatory mediators, such as cytokines or pathogen-derived molecular signals, functionally culminates into efferent vagus nerve activation that dampens pro-inflammatory cytokine production. To accomplish this, the motor (efferent) signals in the vagus nerve activate the splenic nerve (27), which culminates in the release of acetylcholine (ACh) by a subset of T lymphocytes expressing choline acetyltransferase (ChAT) (28). ACh interacts with the α7 nicotinic ACh receptor (α7nAChR) expressed by macrophages to inhibit cytokine production (29).
In addition to control of inflammation in peripheral tissues and organs, the inflammatory reflex may have therapeutic potential for central nervous system (CNS) inflammatory diseases, e.g. multiple sclerosis. Notably, ‘gateway reflexes’ have been discovered in which a variety of stimuli (gravity, pain, electricity, stress, light) activate neural circuits that result in focal inflammation of blood vessels of the blood–brain barrier (30). This inflammatory reaction allows for reactive lymphocytes and other immunocompetent cells to pass through the blood vessels and into the CNS, activating inflammatory disease. Modulation of this neuronal circuit reduces localized chemokine expression and suppresses the entry of pathogenic cells. Although not yet specifically explored, activation of the inflammatory reflex may be useful for therapeutic attenuation of gateway reflex pathways.
The discovery of the inflammatory reflex has prompted a substantial interest in developing strategies to target the nervous system as a key regulator of inflammatory responses and has been translated into therapeutic devices which have significantly improved disease-related end-points in clinical trials of patients with chronic inflammatory conditions (22, 23). In addition to these innovative programs, it is interesting to note that the traditional therapeutic technique of acupuncture is a form of neuromodulation and, as such, has been demonstrated to activate anti-inflammatory activity (31). For example, needle stimulation of nerve endings in the traditional ST36 Zusanli acupoint (i.e. in the anterior tibial muscle 2 mm lateral to the anterior tubercle of the tibia and 4 mm distal to the knee joint lower point, adjacent to the common peroneal and tibial branches of the sciatic nerve) has been shown to potently reduce inflammation and increase survival following endotoxin administration (32, 33); and using electroacupuncture reduces polymicrobial peritonitis (32) in the mouse. These effects depend upon an intact inflammatory reflex, as transection of the vagus nerve abolishes the anti-inflammatory effects of acupuncture.
These pre-clinical and clinical studies focusing on the anti-inflammatory effects of the vagus nerve resulted in the current development of bioelectronic medicine. This growing field combines new insights into neurally mediated regulation of the immune system and advances in bioelectronic technology to offer novel approaches to both diagnosis and treatment of diseases (5). Studies are identifying novel targets for neuro-immune regulation, including the sciatic/vagus pathway that regulates catecholamine production in the adrenal gland (32), central neural circuits maintaining glucose and energy homeostasis (34) and intestinal nerve pathways modulating gut immune responses to bacterial infection (35). Moreover, the scope of the neuromodulation as a therapeutic approach has been extended into a number of chronic inflammatory conditions from asthma (36), Alzheimer’s disease (37), diabetes (38) and digestive disorders (39) to cardiovascular diseases (40).
Although initial studies targeting neuromodulation have focused on using implantable bioelectronic devices, the development of non-invasive technologies could ultimately lead to conceptually personalized treatment for disease management. In this review, we briefly summarize key aspects of some of the non-invasive neuromodulation platforms, leading to current clinical translation for the treatment of inflammatory conditions.
Non-invasive VNS
VNS at the cervical region has been established as a non-pharmacologic therapeutic approach for control of inflammation in a number of pre-clinical disease models (5, 10, 41–43). Earlier clinical studies of VNS using implantable bioelectronic devices have shown the efficacy of VNS for the treatment of RA (22), Crohn’s disease (23) and fibromyalgia (44), validating the translational applicability of pre-clinical findings. The current bioelectronic device requires surgical implantation of a fine-wire electrode wrapped around the left cervical vagus nerve in the neck, which is associated with technical and surgical challenges including electrode fracture, dislocation, generator malfunction, wound infection, recurrent laryngeal nerve palsy and cardiac arrhythmias under test stimulation (45). It is also associated with potential adverse side-effects including syncope, asystole, bradycardia, late-onset bradyarrhythmia, paranesthesia, pain, sleep apnea, cough, hoarseness, dysphagia, dyspnea and thermal injury to the vagus nerve and adjacent area because of the radiofrequency exposure (46–48).
Given the number of conditions that VNS has the potential to benefit, two types of transcutaneous VNS have been developed: transcutaneous cervical VNS (TC-VNS) and transcutaneous auricular VNS (TA-VNS). Both treatments do not require surgical implantation of the device; therefore, exhibiting a much broader therapeutic potential. Functional magnetic resonance imaging demonstrated that both mechanisms of non-invasive VNS activate known brain projections of the vagus nerve, including the nucleus tractus solitarius (NTS), parabrachial area, hypothalamus, amygdala, nucleus accumbens, anterior insula and locus coeruleus (49–53).
Transcutaneous cervical VNS
TC-VNS can be accomplished by delivering a low-voltage electrical signal to the cervical vagus nerve with electrodes placed over the sternocleidomastoid muscle (54). Although implanted electrodes for VNS are positioned at a similar location, the position of the vagus nerve beneath the skin within the carotid sheath, superficial fascia and sternocleidomastoid muscle makes the transcutaneous electrical stimulation of the vagus fibers difficult; with current bioelectronic devices most likely stimulating both afferent and efferent fibers in the vagus nerve bundle (55).
GammaCore® transcutaneous device has been approved by the Food and Drugs Administration (FDA) for acute treatment of migraine and acute or prophylactic treatment of cluster headaches (56). It uses high- and low-frequency stimulation to penetrate deep into the neck as a clinical application for cluster headaches or migraines. The anti-inflammatory effects of TC-VNS have been explored using this device. TC-VNS (up to three times in a day) in healthy subjects resulted in a significant decrease in whole-blood cytokine and chemokine levels (57), and improved fatigue and immune responses in patients with primary Sjögren’s syndrome (58). Preliminary clinical data also demonstrated beneficial effects in hemicrania continua (59), asthma (60), asthma patients with bronchoconstriction (60) and respiratory distress associated with chronic obstructive pulmonary disease (COPD) (ClinicalTrials.gov Identifier: NCT01679314). These findings paved the way for the gammaCore® device to receive emergency use authorization from the FDA as an additional therapy for coronavirus disease 2019 (COVID-19) patients experiencing asthma-related breathing difficulties and reduced airflow (61). Computational modeling analysis indicated the utility of TC-VNS in models of spinal-cord stimulation, deep-brain stimulation and stimulation of other peripheral nerves; however, more work is needed to elucidate the clinical benefit in these disease conditions (62).
A number of clinical studies of TC-VNS are in progress including pancreatitis (ClinicalTrials.gov Identifier: NCT03357029), pain perception (ClinicalTrials.gov Identifier: NCT01174498), dyspepsia and irritable bowel syndrome (ClinicalTrials.gov Identifier: NCT02388269), depression (ClinicalTrials.gov Identifier: NCT04037111) and Raynaud’s phenomena (ClinicalTrials.gov Identifier: NCT03869008).
RA is a debilitating chronic autoimmune and inflammatory disease that affects more than 1.3 million people in the USA alone and is expensive to treat. Current therapy (e.g., anti-tumor necrosis factor (TNF), anti-interleukin (IL)-6 receptor, and anti-CD20 biologics, T-cell co-stimulation inhibitors, or methotrexate) is associated with significant toxicity and is not effective in all patients. Previous studies using implantable bioelectronic vagus-nerve stimulators demonstrated significant improvement in disease activity in RA patients for up to 84 days (22). A recent study targeted the cervical vagus nerve transcutaneously in RA patients using a portable electrical stimulator with two steel contact electrodes (63). TC-VNS performed three times per day for 4 days significantly decreased the disease activity score 28-C-reactive protein (DAS28-CRP) values in RA patients, with a concurrent decrease in circulating CRP levels and swollen-joints scores (63).
Pre-clinical studies have also shown the efficacy of TC-VNS in alleviating CNS inflammation and brain derangements by attenuating cerebral ischemic injury and altering microglial activation toward a neuroprotective phenotype (37, 64, 65). Zhao et al. stimulated the right cervical vagus nerve in a mouse model of focal cerebral ischemia (64) and identified microglial M2 polarization as an important mechanism underlying the TC-VNS-mediated neuroprotection against cerebral ischemia–reperfusion injury. This effect of TC-VNS was dependent on the inhibition of IL-17A production (64). Similarly, a single TC-VNS treatment in a mouse model of Alzheimer’s disease resulted in significantly altered morphological changes in microglia from a neuroinflammatory to a neuroprotective phenotype (37). Interestingly, only the older animals in this study showed the morphological changes following TC-VNS, indicating that primed or activated microglia, and not healthy microglia, are more susceptible to morphological changes as a result of TC-VNS. These studies suggest a stabilizing effect of TC-VNS on microglia morphology to a neuroprotective phenotype.
Transcutaneous auricular VNS
TA-VNS is an emerging therapeutic approach in the field of bioelectronic medicine. The external ear is the only location on the body where afferent fibers of the vagus nerve innervate the skin, specifically the antihelix, tragus, cymba concha and concha (66, 67). The proportion of vagus fibers varies in different areas of the ear and the cymba concha is the only distinct region with 100% innervation by the vagus nerve (67, 68). In brief, the auricular branch of the vagus nerve allows for an accessible external site for electrical stimulation, which then connects and transmits signals to the brainstem. In particular, the auricular branch of the vagus nerve within the cymba concha sends signals to nearby brainstem nuclei, including the NTS, the locus coeruleus and the dorsal raphe nucleus (66), and regulates the heart rate and sympathetic nerve activity (69).
A number of pre-clinical studies have highlighted the efficacy of TA-VNS in reducing inflammation. Zhao et al. originally demonstrated the protective effects of TA-VNS against endotoxemia in rats by reducing TNF levels after administration of lipopolysaccharide (LPS; also known as endotoxin) (70). These data were recently recapitulated in a mouse model of endotoxemia, with TA-VNS reducing a number of pro-inflammatory cytokines, including TNF, IL-6 and IL-1β (71). In a pre-clinical model of postoperative ileus, TA-VNS significantly attenuated intestinal cytokine expression, improved gastrointestinal transit and lowered leukocyte recruitment to the area of manipulation (71). TA-VNS has also been used to elicit neuroprotective effects in cerebral ischemia–reperfusion injury, significantly attenuating the volume of the infarct and inducing angiogenesis in rats (72).
Recently, two pilot open-label studies evaluated the effects of TA-VNS in normal subjects and RA patients. First, we demonstrated the efficacy of TA-VNS using a vibrotactile device at the cymba concha in attenuating endotoxin-induced inflammatory responses in healthy subjects (73). The therapeutic efficacy of this neuromodulation was also demonstrated in RA patients. Stimulation of the auricular branch of the vagus nerve at the cymba concha twice per day for 2 days significantly improved DAS28-CRP disease activity scores in patients with RA (73). In addition, a persistent improvement in visual analogue scale scores—a patient-derived measure of global health assessment—was observed following TA-VNS treatment (73). Initial results of a 12-week proof-of-concept pilot study of TA-VNS using a wearable device showed that the device is well-tolerated in RA patients, with significant reductions in the disease severity. Out of 30 RA patients receiving TA-VNS, 11 attained low disease activity and 7 achieved remission. The TA-VNS treatment was well-tolerated over the study period with American College of Rheumatology 20 (ACR20), ACR50 and ACR70 response rates of 53%, 33% and 17%, respectively, at 3 months (74). Follow-up studies demonstrated sustained, long-term benefits of TA-VNS in RA patients for up to 1 year (75). Fifteen out of 30 patients continued the study for another 9 months and used the wearable device for up to 30 min daily as in the first 12 weeks of the study. A significant reduction in the disease activity (DAS28-CRP) was observed, without significant adverse effects (75).
The use of TA-VNS as a treatment for postoperative ileus has also been suggested (71, 76). TA-VNS activated efferent vagus nerve signaling to the viscera and increased gastrin levels (a surrogate marker for vagus nerve activation) in 14 patients requiring open laparotomy. TA-VNS led to suppression of the action potential frequency and an increase in action potential amplitude, as analyzed by a free-running electromyography in the stomach of these patients (76). The protective effects of TA-VNS have been successfully explored in depression, epilepsy and cardiovascular diseases (77–80). Interestingly, these clinical studies have highlighted the anti-inflammatory effects of TA-VNS, including suppression of inflammatory cytokines, and attenuation of heart-rate irregularities in patients undergoing cardiac surgery or with paroxysmal atrial fibrillation (80, 81).
A recent randomized, double-blind, placebo-controlled pilot study demonstrated TA-VNS reduces pain and fatigue in patients with systemic lupus erythematosus (82). The same device was recently used in a clinical trial for stroke patients to enhance muscle recovery. Although results have yet to be published, it is reported that patients who received TA-VNS exhibited enhanced muscle recovery after 3 weeks of stimulation (ClinicalTrials.gov Identifier: NCT03592745). In light of the most recent COVID-19 pandemic, studies have highlighted the possible effects of TA-VNS in an acute respiratory distress syndrome with compelling evidence for this hypothesis yet to be tested (83).
The Cerbomed device NEMOS (Erlangen, Germany) utilizes a special earphone-like electrode for TA-VNS to ensure correct placement of electrodes for at-home use (84), and has received European clearance for the treatment of epilepsy, depression and pain relief. Its use in drug-resistant epilepsy patients has been associated with significant reductions in seizure frequency and severity, with corresponding improvements in quality of life (85). TA-VNS using NEMOS in recovering stroke patients demonstrated increased motor recovery, but no mechanistic insight has been evaluated (86).
Other clinical trials using the NEMOS device are investigating the effects of TA-VNS in juvenile inflammatory arthritis (ClinicalTrials.gov Identifier: NCT01924780) and on peripheral glucose metabolism (ClinicalTrials.gov Identifier: NCT03615209). An individualized approach is utilized for NEMOS, with the stimulation intensity chosen by the individuals on the basis of the intensity needed to elicit a non-painful stinging sensation, with a recommended stimulation duration up to 4 h per day. This non-standard individualized approach, however, impedes the establishment of a standardized protocol. Other TA-VNS devices, NET-1000 and NET-2000, developed by Auri-Stim, have been approved by the FDA for the treatment of depression, anxiety and insomnia (87), but the effects on inflammation have yet to be established.
Transcranial magnetic stimulation
Transcranial magnetic stimulation (TMS) transcutaneously delivers a rapidly pulsed, high-intensity magnetic field to cause an electric current at a specific area of the brain via electromagnetic induction (88). With a variable magnetic field, a voltage difference between two points is induced resulting in current flow which subsequently stimulates the neural circuits. As cell bodies have higher stimulation thresholds, TMS preferentially stimulates axons (89, 90).
In clinical practice, the non-invasive nature of TMS has several advantages: the magnetic field can pass through any medium without attenuation, and the field decreases inversely proportionally to the distance from the generator coil. TMS has an excellent safety profile, with patients rarely reporting pain due to stimulation; no charged particles are injected into the skin; and magnetic stimulation has only a weak recruitment ability for cutaneous sensory afferent fibers. TMS is delivered using stimulation coils, with the focal ability and depth of stimulation established by changing the type of coil attached to a high-current pulse generator. Several stimulation parameters have been proposed with TMS, with variations in duty cycle, frequency and intensity being actively explored.
TMS has seen ample clinical applications including depression, pain management, neural reinforcement after trauma, spasticity reduction, increased muscle strength after surgery and reduction of dysphagia. Repetitive TMS has been shown to reduce apoptotic cell death and neuroinflammation after hemicerebellectomy-induced focal brain injury in rats (91). Activation of the inflammatory reflex via TMS has yet to be explored; however, targeting brain areas associated with the modulation of immune function, including the dorsal motor nucleus (the efferent outflow of the vagus nerve), NTS (the afferent inflow of the vagus nerve), insular cortex and the hypothalamic–pituitary–adrenal axis, is achievable. Also of interest is the use of peripheral magnetic stimulation on sites other than the brain. Early studies have shown success but are mainly limited to pain-management models (92–95).
Ultrasound technology
Since the first report of ultrasound as a therapeutic tool in the 1920s, it has been widely used in clinical practice and clinical/translational research for the treatment of various human malignancies (96–98) and pathologies including Parkinson’s disease (99), stroke (100), prostatic hyperplasia (101), renal masses (102), treatment of abdominal subcutaneous adipose tissue (103), bone repair (104), osteoarthritis (105) and carpal tunnel syndrome (106). Ultrasound waves are sound waves generated by cyclic mechanical vibrations with frequencies higher than the upper audible range for the human (>20 kHz). Whereas diagnostic ultrasound uses frequencies in the MHz range, therapeutic ultrasound uses frequencies in the kHz range, leading to focused beams of ultrasound energy with higher levels of precision that target deeper tissues compared with existing non-invasive neuromodulatory approaches.
Several modalities of action of focused ultrasound have been proposed; including mechanical force, local heating and bubble cavitation, described in detail elsewhere (107). High-intensity focused ultrasound is currently approved by the FDA for thermal ablation in many pathologies, including atrial fibrillation (108), uterine fibroids (109) and visceral tumors (110). Although in clinical interest for more than half a century (111), the interest for focused ultrasound as a non-invasive neuromodulation approach for regulating inflammatory responses has increased recently (112–114).
Ultrasound stimulation targeted to the spleen in mice reduced antibody responses to sheep erythrocytes in a manner dependent on the dose of ultrasound energy, whereas ultrasound delivered to an area devoid of major lymphoid tissue was not immunosuppressive (115). Additionally, exposure to ultrasound impaired the phagocytic and bactericidal activity of peritoneal macrophages (116). Recently, our knowledge about the immunomodulatory functions of ultrasound was considerably advanced when ultrasound energy was characterized as a major regulator of inflammation (112, 113). Delivery of pulsed ultrasound to the spleen using a non-invasive clinical ultrasound machine diminished inflammation and tissue damage during renal ischemic–reperfusion injury (112, 113). While attempting to image the kidney vasculature before reperfusion, Gigliotti et al. demonstrated that ultrasound conferred a significant protection from renal ischemia–reperfusion (112). The protective effect of a single ultrasound stimulation lasted for 2 days and waned in a time-dependent manner when ultrasound was applied up to 7 days before kidney injury (112). Moreover, ultrasound treatment was also protective in reducing acute kidney injury in the cecal ligation–puncture model of induced sepsis (113).
A growing body of experimental evidence in recent years indicates that targeting the spleen with focused ultrasound controls peripheral immune responses and inflammation (114, 117). When applied either prior to or at the time of endotoxin challenge, focused ultrasound treatment was found equally effective in TNF reduction as compared with traditional VNS using implanted electrodes (114). In this study, an ultrasound transducer was focused directly to the center of the spleen, using a second imaging transducer to align the ultrasound delivery, and pulsed ultrasound energy was delivered to the spleen prior to and after endotoxin administration (114). A single session of ultrasound stimulation suppressed TNF in rodent models. In addition, ablating the ACh-producing T cells or blocking α7nAChR suppressed the immunomodulatory effect of ultrasound stimulation (114), confirming the role of the inflammatory reflex. Although ultrasound stimulation at several distinct locations within the spleen provided similar modulation of the TNF response, stimulation at the off-target sites (i.e. liver) did not modulate the LPS-induced inflammatory response (114). Interestingly, splenic ultrasound stimulation showed no effect on the heart rate, a known side-effect of stimulation of the vagus nerve. This study also demonstrated the ability of site-specific effects of ultrasound stimulation that cannot be achieved with traditional cervical VNS. Cotero and colleagues demonstrated that targeting the ultrasound energy to the porta hepatis region of the liver, which contains glucose-sensitive neurons, but not at the liver lobes or the spleen, reduced LPS-induced hyperglycemia.
In line with the effects seen in clinical trials studying efficacy of VNS in RA (22), Zachs et al. demonstrated that focused splenic ultrasound significantly attenuates the disease severity in a model of inflammatory arthritis (117). Importantly, using single-cell RNA sequencing, their study showed ultrasound stimulation-induced changes in gene expression in splenic lymphocytes from arthritic but not from non-arthritic mice, suggesting a unique therapeutic effect in the setting of inflammation (117). A clinical study is in progress to study the effects of focused splenic ultrasound in RA (ClinicalTrials.gov Identifier: NCT03690466).
The mechanism of this splenic ultrasound-mediated immunomodulation is unknown, but several findings suggest the protective effect is mediated via activation of the inflammatory reflex circuit. First, the immunomodulatory effect of ultrasound is dependent on the spleen, as splenectomized animals fail to respond to ultrasound treatment (112). Second, targeting the spleen is crucial in achieving these protective effects, since ultrasound stimulation of other body locations is ineffective (114, 115). Third, catecholamine depletion by reserpine (114) or chemical sympathectomy by using splenic administration of 6-hydroxydopamine (a neurotoxin that destroys catecholaminergic neurons) (113) abolishes the protective effect of ultrasound, indicating a requirement for innervation of the spleen. Fourth, the protective effect of ultrasound is absent in mice lacking T and or B cells, but could be reconstituted by adoptive transfer of CD4+ T cells (112). Fifth, mice lacking expression of α7nAChR or with knockout of CD4-ChAT cells (CD4+ T cells that express ChAT) fail to respond to ultrasound (114); α7nAChR and CD4-ChAT cells are the key regulators of the inflammatory reflex pathway (28, 29). Blocking of α7nAChR with α-bungarotoxin abrogates the protective effect of splenic ultrasound stimulation (114). Finally, splenic ultrasound stimulation drives neurotransmitter and cytokine changes within the spleen consistent with modulation of the inflammatory reflex (114). Both norepinephrine and ACh concentrations increase in the spleen following splenic ultrasound stimulation. In addition, splenic ultrasound reduces levels of pro-inflammatory cytokines, such as TNF and IL-1 in the spleen from endotoxemic animals (114). Taken together, these studies indicate, similar to VNS, splenic ultrasound-mediated immunomodulation is due to activation of the inflammatory reflex pathway.
Focused ultrasound modulation of neural signaling has also been evaluated for other disease models. Attenuation of post-myocardial infarction ventricular arrhythmias and inflammation can be achieved in a canine model by modulating the sympathetic neural activity (118). As focused ultrasound technologies continue to advance the ability to penetrate deeper into the body while maintaining specificity, the idea of this invasive modulation to translate to a non-invasive focused ultrasound is not a far-fetched concept. Similar to electrical VNS, a single focused ultrasound stimulation on the cervical vagus nerve was protective in endotoxemic animals in a dose-dependent manner (119). In addition, ultrasound has been explored as a therapy for inflammation induced by soft-tissue injury. Compared with placebo, ultrasound stimulation in 76 patients with lateral epicondylitis lowered inflammation and pain (120). It was shown to reduce swelling and pain, and accelerate tissue repair (121). In addition, anti-inflammatory effects of ultrasound are closely related to the decrease of inflammatory cell infiltration in the synovium and attenuation of hyperplasia (122).
Ultrasound stimulation targeted at the porta hepatis region of the liver (a region that is highly innervated by glucose-sensitive neurons (34)) provided protection against LPS-induced hyperglycemia (114). Hepatic ultrasound stimulation limited the increase in blood glucose levels. Furthermore, this protective effect was anatomically specific, as targeting the stimulation toward the right or left lobe of the liver reduced the glucose-lowering effect of hepatic ultrasound stimulation. In addition, ultrasound stimulation of the porta hepatis did not change concentrations of signaling molecules associated with hepatic glycolysis/gluconeogenesis within the liver; instead, resulted in increased insulin receptor substrate 1 and protein kinase B activation and reduced concentrations of neuropeptide Y and pro-opiomelanocortin in the hypothalamus (114). Interestingly, hypothalamic neuronal activation was accompanied by increased c-Fos expression within the NTS, suggesting ultrasound-mediated modulation via signaling through afferent pathways.
Obesity increases the risk of cardiovascular disease, type 2 diabetes and other diseases (123). Chronic low-grade inflammation mediated by immune and metabolic dysregulation is a characteristic feature in patients with obesity and is causally linked with insulin resistance and other metabolic complications (124, 125). It is increasingly recognized that the brain and the nervous system are involved in the regulation of obesity and obesity-associated complications (26). Accordingly, therapeutic strategies targeting chronic inflammation and improving autonomic function have been proposed (9, 126).
To study the effect of hepatic ultrasound stimulation on the long-term management of obesity and obesity-associated complications, our group has also performed hepatic stimulation experiments in obese mice that were fed a western diet (127). Obese mice were treated with daily ultrasound stimulation targeted to the porta hepatis for 4 weeks. At the time of the treatment initiation, mice on the western diet had already increased weight, which reached a difference of ~10 g when compared with mice fed a low-fat control diet. Ultrasound stimulation at the porta hepatis gradually attenuated the body weight gain, reaching a significant difference with the sham-stimulated group by week 12. In addition, hepatic ultrasound reduced food intake and moderated abdominal fat accumulation in obese mice. Interestingly, this reduction in weight occurred concurrently with decreases in circulating inflammatory cytokines, adipokines, lipids and hepatic leukocyte infiltration, indicating that hepatic ultrasound attenuated inflammatory responses in western-diet-fed obese mice (127). Together, these studies suggest that ultrasound stimulation focused on peripheral organs is an increasingly attractive target to develop organ-specific non-invasive therapeutic strategies for a range of inflammatory conditions.
Concluding remarks
The recent viral COVID-19 pandemic has alarmingly added to the urgent need of utilizing non-invasive therapeutic strategies for treatment of inflammation. Ongoing studies have provided mechanistic insight into neuro-immune communication, and in controlling inflammation by targeting neural circuits using bioelectronic devices. A number of pre-clinical and clinical studies have established the efficacy of non-invasive neural stimulation in the regulation of inflammatory conditions. Specifically, these studies have indicated that targeting the vagus nerve-mediated inflammatory reflex pathway by non-invasive transcutaneous VNS or focused ultrasound stimulation as promising new approaches for treating inflammatory and autoimmune conditions.
Several inter-related issues will need to be clarified for each of these potential new treatment modalities. A primary issue is to define the specificity of stimulation, i.e. to what extent are off-target tissues modulated? Because non-invasive modalities offer the potential to more selectively activate tissue-specific neural pathways and/or specific anatomical locations in contrast to invasive neuromodulation, fewer adverse and unintended effects may result. In this regard, results of clinical trials conducted in a number of diseases have not shown serious adverse effects. For example, TA-VNS treatment for drug-resistant epilepsy carried out in 10 separate trials (350 patients) has resulted in only minor adverse effects related to electrode placement: headache (9%) being most common, followed by skin irritation at the stimulation site (7%) and nasopharyngitis (5%) (85). Another pressing issue is to define specific treatment parameters (stimulation parameters such as waveform, amplitude, timing, duration, etc.) which will likely depend both upon studies of the disease as well as the device.
Pre-clinical studies have unraveled the molecular and cellular mechanisms underlying the effects of neuromodulation using non-invasive bioelectronic devices and provided a rationale for clinical translation. Non-invasive neuromodulation as a therapy for inflammatory conditions is not yet in prime time, but is gaining momentum as a novel therapeutic approach by harnessing the body’s own protective neural circuits. Arguably, various modalities of non-invasive neuromodulation are at the forefront of the technological revolution and warrant multidisciplinary collaborative research efforts to advance bioelectronic medicine and create novel therapeutic strategies.
Although bioelectricity derived from electric fish was used as immunotherapy in antiquity, an understanding of the underlying biophysics and development of therapeutic technology awaited the discoveries that electricity stimulated excitable tissue by Galvani and electromagnetic induction by Faraday, as well as the invention of a portable electricity source by Volta around the turn of the 19th century. Following a long gestational period, implanted electrical stimulators were developed to regulate cardiac rhythm (128, 129). Thereafter, the vagus-nerve stimulator was developed to control drug-resistant epilepsy (130) and activate the inflammatory reflex (22, 23). The first widespread non-invasive neuromodulation utilized TMS (131), which depends upon electromagnetic induction. Further developments in non-invasive neuromodulation have evolved into transcutaneous vagus stimulation (57, 132). Very recently, focused non-invasive mechanical nerve stimulation using ultrasound has been employed as a therapeutic method of activating the inflammatory reflex (114, 117) (please see text for details).
Acknowledgements
The authors apologize to colleagues whose work was not cited because of space limitations. The figure was created using Biorender.
Funding
This work was supported by the National Institutes of Health (NIH), National Institute of General Medical Sciences (NIGMS) Grant: R01GM132672 (to S.S.C.).
Conflicts of interest statement: the authors declared no conflicts of interest.
References
- 1. Brewer, D. J. and Friedman, R. 1990. Fish and Fishing in Ancient Egypt. American University in Cairo Press, Cairo, Egypt. [Google Scholar]
- 2. Heidland, A., Fazeli, G., Klassen, A.et al. 2013. Neuromuscular electrostimulation techniques: historical aspects and current possibilities in treatment of pain and muscle waisting. Clin. Nephrol. 79(Suppl. 1):S12. [PubMed] [Google Scholar]
- 3. Kane, K. and Taub, A. 1975. A history of local electrical analgesia. Pain 1:125. [DOI] [PubMed] [Google Scholar]
- 4. Tsoucalas, G. and Sgantzos, M. 2016. Electric current to cure arthritis and cephalaea in ancient Greek medicine. Mediterr. J. Rheumatol. 27:198. [Google Scholar]
- 5. Pavlov, V. A., Chavan, S. S. and Tracey, K. J. 2020. Bioelectronic medicine: from preclinical studies on the inflammatory reflex to new approaches in disease diagnosis and treatment. Cold Spring Harb. Perspect. Med. 10:a034140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chavan, S. S., Ma, P. and Chiu, I. M. 2018. Neuro-immune interactions in inflammation and host defense: implications for transplantation. Am. J. Transplant. 18:556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Serhan, C. N. and Savill, J. 2005. Resolution of inflammation: the beginning programs the end. Nat. Immunol. 6:1191. [DOI] [PubMed] [Google Scholar]
- 8. Balkwill, F. and Mantovani, A. 2001. Inflammation and cancer: back to Virchow? Lancet 357:539. [DOI] [PubMed] [Google Scholar]
- 9. Pavlov, V. A. and Tracey, K. J. 2012. The vagus nerve and the inflammatory reflex—linking immunity and metabolism. Nat. Rev. Endocrinol. 8:743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Chavan, S. S., Pavlov, V. A. and Tracey, K. J. 2017. Mechanisms and therapeutic relevance of neuro-immune communication. Immunity 46:927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Dinarello, C. A. 2010. Anti-inflammatory agents: present and future. Cell 140:935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Anonymous. 2020. Anti-Inflammatory Biologics Market Size, Share & Global Report, 2016–2020. Available at: https://www.fortunebusinessinsights.com/anti-inflammatory-biologics-market-102733.
- 13. Bluestone, J. A. and Anderson, M. 2020. Tolerance in the age of immunotherapy. N. Engl. J. Med. 383:1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Bongartz, T., Sutton, A. J., Sweeting, M. J.et al. 2006. Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA 295:2275. [DOI] [PubMed] [Google Scholar]
- 15. Wallis, R. S. 2007. Reactivation of latent tuberculosis by TNF blockade: the role of interferon γ. J. Investig. Dermatol. Symp. Proc. 12:16. [DOI] [PubMed] [Google Scholar]
- 16. Guerra, I., Algaba, A., Pérez-Calle, J. L.et al. 2012. Induction of psoriasis with anti-TNF agents in patients with inflammatory bowel disease: a report of 21 cases. J. Crohns Colitis 6:518. [DOI] [PubMed] [Google Scholar]
- 17. Mayberg, H. S., Lozano, A. M., Voon, V.et al. 2005. Deep brain stimulation for treatment-resistant depression. Neuron 45:651. [DOI] [PubMed] [Google Scholar]
- 18. Deuschl, G., Schade-Brittinger, C., Krack, P.et al. ; German Parkinson Study Group, Neurostimulation Section. 2006. A randomized trial of deep-brain stimulation for Parkinson’s disease. N. Engl. J. Med. 355:896. [DOI] [PubMed] [Google Scholar]
- 19. Thaha, M. A., Abukar, A. A., Thin, N. N.et al. 2015. Sacral nerve stimulation for faecal incontinence and constipation in adults. Cochrane Database Syst. Rev. 24:CD004464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Pai, R. G., Varadarajan, P. and Pai, S. M. 2018. Pacemakers and defibrillators. Cardiology Board Review, 1st edn, p. 463. John Wiley & Sons, Hoboken, NJ. [Google Scholar]
- 21. Toffa, D. H., Touma, L., El Meskine, T.et al. 2020. Learnings from 30 years of reported efficacy and safety of vagus nerve stimulation (VNS) for epilepsy treatment: a critical review. Seizure 83:104. [DOI] [PubMed] [Google Scholar]
- 22. Koopman, F. A., Chavan, S. S., Miljko, S.et al. 2016. Vagus nerve stimulation inhibits cytokine production and attenuates disease severity in rheumatoid arthritis. Proc. Natl Acad. Sci. USA 113:8284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Bonaz, B., Sinniger, V., Hoffmann, D.et al. 2016. Chronic vagus nerve stimulation in Crohn’s disease: a 6-month follow-up pilot study. Neurogastroenterol. Motil. 28:948. [DOI] [PubMed] [Google Scholar]
- 24. Tracey, K. J. 2002. The inflammatory reflex. Nature 420:853. [DOI] [PubMed] [Google Scholar]
- 25. Chavan, S. S. and Tracey, K. J. 2017. Essential neuroscience in immunology. J. Immunol. 198:3389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Pavlov, V. A., Chavan, S. S. and Tracey, K. J. 2018. Molecular and functional neuroscience in immunity. Annu. Rev. Immunol. 36:783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kressel, A. M., Tsaava, T., Levine, Y. A.et al. 2020. Identification of a brainstem locus that inhibits tumor necrosis factor. Proc. Natl Acad. Sci. USA 117:29803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Rosas-Ballina, M., Olofsson, P. S., Ochani, M.et al. 2011. Acetylcholine-synthesizing T cells relay neural signals in a vagus nerve circuit. Science 334:98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wang, H., Yu, M., Ochani, M.et al. 2003. Nicotinic acetylcholine receptor alpha7 subunit is an essential regulator of inflammation. Nature 421:384. [DOI] [PubMed] [Google Scholar]
- 30. Uchida, M., Yamamoto, R., Matsuyama, S.et al. 2021. Gateway reflexes, neuronal circuits that regulate the autoreactive T cells in organs having blood barriers. Int. Immunol. 2021. [DOI] [PubMed] [Google Scholar]
- 31. Chavan, S. S. and Tracey, K. J. 2014. Regulating innate immunity with dopamine and electroacupuncture. Nat. Med. 20:239. [DOI] [PubMed] [Google Scholar]
- 32. Torres-Rosas, R., Yehia, G., Peña, G.et al. 2014. Dopamine mediates vagal modulation of the immune system by electroacupuncture. Nat. Med. 20:291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Lim, H. D., Kim, M. H., Lee, C. Y.et al. 2016. Anti-inflammatory effects of acupuncture stimulation via the vagus nerve. PLoS One 11:e0151882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Roh, E., Song, D. K. and Kim, M. S. 2016. Emerging role of the brain in the homeostatic regulation of energy and glucose metabolism. Exp. Mol. Med. 48:e216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Gabanyi, I., Muller, P. A., Feighery, L.et al. 2016. Neuro-immune interactions drive tissue programming in intestinal macrophages. Cell 164:378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Staats, P., Emala, C., Simon, B.et al. 2018. Neurostimulation for asthma. In Krames E. S., Hunter Peckham P., and Rezai A. R. eds., Neuromodulation, 2nd edn, p. 1339. Elsevier, Amsterdam, The Netherlands. [Google Scholar]
- 37. Kaczmarczyk, R., Tejera, D., Simon, B.et al. 2018. Microglia modulation through external vagus nerve stimulation in a murine model of Alzheimer’s disease. J. Neurochem. 146:76. [DOI] [PubMed] [Google Scholar]
- 38. Masi, E. B., Levy, T., Tsaava, T.et al. 2019. Identification of hypoglycemia-specific neural signals by decoding murine vagus nerve activity. Bioelectron. Med. 5:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Payne, S. C., Furness, J. B., Burns, O.et al. 2019. Anti-inflammatory effects of abdominal vagus nerve stimulation on experimental intestinal inflammation. Front. Neurosci. 13:418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Hou, Y., Zhou, Q. and Po, S. S. 2016. Neuromodulation for cardiac arrhythmia. Heart Rhythm 13:584. [DOI] [PubMed] [Google Scholar]
- 41. Borovikova, L. V., Ivanova, S., Zhang, M.et al. 2000. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 405:458. [DOI] [PubMed] [Google Scholar]
- 42. Levine, Y. A., Koopman, F. A., Faltys, M.et al. 2014. Neurostimulation of the cholinergic anti-inflammatory pathway ameliorates disease in rat collagen-induced arthritis. PLoS One 9:e104530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. de Jonge, W. J., van der Zanden, E. P., The, F. O.et al. 2005. Stimulation of the vagus nerve attenuates macrophage activation by activating the Jak2-STAT3 signaling pathway. Nat. Immunol. 6:844. [DOI] [PubMed] [Google Scholar]
- 44. Lange, G., Janal, M. N., Maniker, A.et al. 2011. Safety and efficacy of vagus nerve stimulation in fibromyalgia: a phase I/II proof of concept trial. Pain Med. 12:1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Spuck, S., Tronnier, V., Orosz, I.et al. 2010. Operative and technical complications of vagus nerve stimulator implantation. Neurosurgery 67(2 Suppl. Operative):489. [DOI] [PubMed] [Google Scholar]
- 46. Asconapé, J. J., Moore, D. D., Zipes, D. P.et al. 1999. Bradycardia and asystole with the use of vagus nerve stimulation for the treatment of epilepsy: a rare complication of intraoperative device testing. Epilepsia 40:1452. [DOI] [PubMed] [Google Scholar]
- 47. Fahy, B. G. 2010. Intraoperative and perioperative complications with a vagus nerve stimulation device. J. Clin. Anesth. 22:213. [DOI] [PubMed] [Google Scholar]
- 48. Marzec, M., Edwards, J., Sagher, O.et al. 2003. Effects of vagus nerve stimulation on sleep-related breathing in epilepsy patients. Epilepsia 44:930. [DOI] [PubMed] [Google Scholar]
- 49. Yakunina, N., Kim, S. S. and Nam, E. C. 2017. Optimization of transcutaneous vagus nerve stimulation using functional MRI. Neuromodulation 20:290. [DOI] [PubMed] [Google Scholar]
- 50. Kraus, T., Hösl, K., Kiess, O.et al. 2007. BOLD fMRI deactivation of limbic and temporal brain structures and mood enhancing effect by transcutaneous vagus nerve stimulation. J. Neural. Transm. 114:1485. [DOI] [PubMed] [Google Scholar]
- 51. Dietrich, S., Smith, J., Scherzinger, C.et al. 2008. A novel transcutaneous vagus nerve stimulation leads to brainstem and cerebral activations measured by functional MRI. Biomed. Tech. 53:104. [DOI] [PubMed] [Google Scholar]
- 52. Kraus, T., Kiess, O., Hösl, K.et al. 2013. CNS BOLD fMRI effects of sham-controlled transcutaneous electrical nerve stimulation in the left outer auditory canal—a pilot study. Brain Stimul. 6:798. [DOI] [PubMed] [Google Scholar]
- 53. Frangos, E. and Komisaruk, B. R. 2017. Access to vagal projections via cutaneous electrical stimulation of the neck: fMRI evidence in healthy humans. Brain Stimul. 10:19. [DOI] [PubMed] [Google Scholar]
- 54. Ben-Menachem, E., Revesz, D., Simon, B. J.et al. 2015. Surgically implanted and non-invasive vagus nerve stimulation: a review of efficacy, safety and tolerability. Eur. J. Neurol. 22:1260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Yuan, H. and Silberstein, S. D. 2016. Vagus nerve and vagus nerve stimulation, a comprehensive review: part II. Headache 56:259. [DOI] [PubMed] [Google Scholar]
- 56. Mertens, A., Raedt, R., Gadeyne, S.et al. 2018. Recent advances in devices for vagus nerve stimulation. Expert Rev. Med. Devices 15:527. [DOI] [PubMed] [Google Scholar]
- 57. Lerman, I., Hauger, R., Sorkin, L.et al. 2016. Noninvasive transcutaneous vagus nerve stimulation decreases whole blood culture-derived cytokines and chemokines: a randomized, blinded, healthy control pilot trial. Neuromodulation 19:283. [DOI] [PubMed] [Google Scholar]
- 58. Tarn, J., Legg, S., Mitchell, S.et al. 2019. The effects of noninvasive vagus nerve stimulation on fatigue and immune responses in patients with primary Sjögren’s syndrome. Neuromodulation 22:580. [DOI] [PubMed] [Google Scholar]
- 59. Nesbitt, A. D., Marin, J., Tomkins, E.et al. 2031. Non-invasive vagus nerve stimulation for the treatment of cluster headache: a case series. Headache Pain 14(Suppl. 1):P231. [Google Scholar]
- 60. Steyn, E., Mohamed, Z. and Husselman, C. 2013. Non-invasive vagus nerve stimulation for the treatment of acute asthma exacerbations—results from an initial case series. Int. J. Emerg. Med. 6:7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Staats, P., Giannakopoulos, G., Blake, J.et al. 2020. The use of non-invasive vagus nerve stimulation to treat respiratory symptoms associated with COVID-19: a theoretical hypothesis and early clinical experience. Neuromodulation 23:784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Mourdoukoutas, A. P., Truong, D. Q., Adair, D. K.et al. 2018. High-resolution multi-scale computational model for non-invasive cervical vagus nerve stimulation. Neuromodulation 21:261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Drewes, A. M., Brock, C., Rasmussen, S. E.et al. 2021. Short-term transcutaneous non-invasive vagus nerve stimulation may reduce disease activity and pro-inflammatory cytokines in rheumatoid arthritis: results of a pilot study. Scand. J. Rheumatol. 50:20. [DOI] [PubMed] [Google Scholar]
- 64. Zhao, X. P., Zhao, Y., Qin, X. Y.et al. 2019. Non-invasive vagus nerve stimulation protects against cerebral ischemia/reperfusion injury and promotes microglial M2 polarization via interleukin-17A inhibition. J. Mol. Neurosci. 67:217. [DOI] [PubMed] [Google Scholar]
- 65. Ay, I., Nasser, R., Simon, B.et al. 2016. Transcutaneous cervical vagus nerve stimulation ameliorates acute ischemic injury in rats. Brain Stimul. 9:166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Mercante, B., Ginatempo, F., Manca, A.et al. 2018. Anatomo-physiologic basis for auricular stimulation. Med. Acupunct. 30:141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Peuker, E. T. and Filler, T. J. 2002. The nerve supply of the human auricle. Clin. Anat. 15:35. [DOI] [PubMed] [Google Scholar]
- 68. Kaniusas, E., Kampusch, S., Tittgemeyer, M.et al. 2019. Current directions in the auricular vagus nerve stimulation I—a physiological perspective. Front. Neurosci. 13:854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Clancy, J. A., Mary, D. A., Witte, K. K.et al. 2014. Non-invasive vagus nerve stimulation in healthy humans reduces sympathetic nerve activity. Brain Stimul. 7:871. [DOI] [PubMed] [Google Scholar]
- 70. Zhao, Y. X., He, W., Jing, X. H.et al. 2012. Transcutaneous auricular vagus nerve stimulation protects endotoxemic rat from lipopolysaccharide-induced inflammation. Evid. Based Complement. Alternat. Med. 2012:627023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Hong, G. S., Zillekens, A., Schneiker, B.et al. 2019. Non-invasive transcutaneous auricular vagus nerve stimulation prevents postoperative ileus and endotoxemia in mice. Neurogastroenterol. Motil. 31:e13501. [DOI] [PubMed] [Google Scholar]
- 72. Ma, J., Zhang, L., He, G.et al. 2016. Transcutaneous auricular vagus nerve stimulation regulates expression of growth differentiation factor 11 and activin-like kinase 5 in cerebral ischemia/reperfusion rats. J. Neurol. Sci. 369:27. [DOI] [PubMed] [Google Scholar]
- 73. Addorisio, M. E., Imperato, G. H., de Vos, A. F.et al. 2019. Investigational treatment of rheumatoid arthritis with a vibrotactile device applied to the external ear. Bioelectron. Med. 5:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Marsal, S., Corominas, H., De Agustin De Oro, J. J.et al. 2020. Non-invasive vagus nerve stimulation improves signs and symptoms of rheumatoid arthritis: results of a pilot study. ACR Convergence Meeting 2020, Abstract Number 1995. Available at: https://acrabstracts.org/abstract/non-invasive-vagus-nerve-stimulation-improves-signs-and-symptoms-of-rheumatoid-arthritis-results-of-a-pilot-study/. [Google Scholar]
- 75. Marsal, S., Corominas, H., De Agustin, J. J.et al. 2021. AB0264 1-year results of a non-invasive auricular vagus nerve stimulation device in patients with rheumatoid arthritis. Ann. Rheum. Dis. 80(Suppl. 1):1158.33926922 [Google Scholar]
- 76. Hong, G. S., Pintea, B., Lingohr, P.et al. 2019. Effect of transcutaneous vagus nerve stimulation on muscle activity in the gastrointestinal tract (transVaGa): a prospective clinical trial. Int. J. Colorectal Dis. 34:417. [DOI] [PubMed] [Google Scholar]
- 77. Rong, P., Liu, A., Zhang, J.et al. 2014. Transcutaneous vagus nerve stimulation for refractory epilepsy: a randomized controlled trial. Clin. Sci. 2014. [DOI] [PubMed] [Google Scholar]
- 78. Rong, P., Liu, J., Wang, L.et al. 2016. Effect of transcutaneous auricular vagus nerve stimulation on major depressive disorder: a nonrandomized controlled pilot study. J. Affect. Disord. 195:172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Yu, L., Scherlag, B. J., Li, S.et al. 2013. Low-level transcutaneous electrical stimulation of the auricular branch of the vagus nerve: a noninvasive approach to treat the initial phase of atrial fibrillation. Heart Rhythm. 10:428. [DOI] [PubMed] [Google Scholar]
- 80. Stavrakis, S., Humphrey, M. B., Scherlag, B. J.et al. 2015. Low-level transcutaneous electrical vagus nerve stimulation suppresses atrial fibrillation. J. Am. Coll. Cardiol. 65:867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Stavrakis, S., Humphrey, M. B., Scherlag, B.et al. 2017. Low-level vagus nerve stimulation suppresses post-operative atrial fibrillation and inflammation: a randomized study. JACC Clin. Electrophysiol. 3:929. [DOI] [PubMed] [Google Scholar]
- 82. Aranow, C., Atish-Fregoso, Y., Lesser, M.et al. 2021. Transcutaneous auricular vagus nerve stimulation reduces pain and fatigue in patients with systemic lupus erythematosus: a randomised, double-blind, sham-controlled pilot trial. Ann. Rheum. Dis. 80:203. [DOI] [PubMed] [Google Scholar]
- 83. Kaniusas, E., Szeles, J. C., Kampusch, S.et al. 2020. Non-invasive auricular vagus nerve stimulation as a potential treatment for Covid19-originated acute respiratory distress syndrome. Front. Physiol. 11:890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Ellrich, J. 2019. Transcutaneous auricular vagus nerve stimulation. J. Clin. Neurophysiol. 36:437. [DOI] [PubMed] [Google Scholar]
- 85. Lampros, M., Vlachos, N., Zigouris, A.et al. 2021. Transcutaneous vagus nerve stimulation (t-VNS) and epilepsy: a systematic review of the literature. Seizure 91:40. [DOI] [PubMed] [Google Scholar]
- 86. Redgrave, J. N., Moore, L., Oyekunle, T.et al. 2018. Transcutaneous auricular vagus nerve stimulation with concurrent upper limb repetitive task practice for poststroke motor recovery: a pilot study. J. Stroke Cerebrovasc. Dis. 27:1998. [DOI] [PubMed] [Google Scholar]
- 87. Ramkissoon, C. M., Güemes, A. and Vehi, J. 2021. Overview of therapeutic applications of non-invasive vagus nerve stimulation: a motivation for novel treatments for systemic lupus erythematosus. Bioelectron. Med. 7:8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Rossini, P. M., Rossini, L. and Ferreri, F. 2010. Transcranial magnetic stimulation: a review. IEEE Eng. Med. Biol. Mag. 29:84. [DOI] [PubMed] [Google Scholar]
- 89. Treister, R., Lang, M., Klein, M. M.et al. 2013. Non-invasive transcranial magnetic stimulation (TMS) of the motor cortex for neuropathic pain—at the tipping point? Rambam Maimonides Med. J. 4:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Chail, A., Saini, R. K., Bhat, P. S.et al. 2018. Transcranial magnetic stimulation: a review of its evolution and current applications. Ind. Psychiatry J. 27:172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Sasso, V., Bisicchia, E., Latini, L.et al. 2016. Repetitive transcranial magnetic stimulation reduces remote apoptotic cell death and inflammation after focal brain injury. J. Neuroinflammation. 13:150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Kanjanapanang, N., Munakomi, S. and Chang, K.-V. 2021. Peripheral Magnetic Stimulation. StatPearls Publishing, Treasure Island, FL. [PubMed] [Google Scholar]
- 93. Khedr, E. M., Ahmed, M. A., Alkady, E. A.et al. 2012. Therapeutic effects of peripheral magnetic stimulation on traumatic brachial plexopathy: clinical and neurophysiological study. Neurophysiol. Clin. 42:111. [DOI] [PubMed] [Google Scholar]
- 94. Massé-Alarie, H., Flamand, V. H., Moffet, H.et al. 2013. Peripheral neurostimulation and specific motor training of deep abdominal muscles improve posturomotor control in chronic low back pain. Clin. J. Pain 29:814. [DOI] [PubMed] [Google Scholar]
- 95. Smania, N., Corato, E., Fiaschi, A.et al. 2005. Repetitive magnetic stimulation a novel therapeutic approach for myofascial pain syndrome. J. Neurol. 252:307. [DOI] [PubMed] [Google Scholar]
- 96. Wood, A. K. and Sehgal, C. M. 2015. A review of low-intensity ultrasound for cancer therapy. Ultrasound Med. Biol. 41:905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. McHale, A. P., Callan, J. F., Nomikou, N.et al. 2016. Sonodynamic therapy: concept, mechanism and application to cancer treatment. Adv. Exp. Med. Biol. 880:429. [DOI] [PubMed] [Google Scholar]
- 98. Liu, H. L., Hua, M. Y., Chen, P. Y.et al. 2010. Blood-brain barrier disruption with focused ultrasound enhances delivery of chemotherapeutic drugs for glioblastoma treatment. Radiology 255:415. [DOI] [PubMed] [Google Scholar]
- 99. Yang, H., Yuan, Y., Wang, X.et al. 2020. Closed-loop transcranial ultrasound stimulation for real-time non-invasive neuromodulation in vivo. Front. Neurosci. 14:445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Mijajlovic, M. D., Pavlovic, A. M. and Covickovic-Sternic, N. 2013. Is sonothrombolysis an effective stroke treatment? J. Ultrasound Med. 32:1117. [DOI] [PubMed] [Google Scholar]
- 101. Madersbacher, S., Schatzl, G., Djavan, B.et al. 2000. Long-term outcome of transrectal high-intensity focused ultrasound therapy for benign prostatic hyperplasia. Eur. Urol. 37:687. [DOI] [PubMed] [Google Scholar]
- 102. Hurwitz, M. D., Ghanouni, P., Kanaev, S. V.et al. 2014. Magnetic resonance–guided focused ultrasound for patients with painful bone metastases: phase III trial results. J. Natl Cancer Inst. 106:dju082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Friedmann, D. P. 2015. A review of the aesthetic treatment of abdominal subcutaneous adipose tissue: background, implications, and therapeutic options. Dermatol. Surg. 41:18. [DOI] [PubMed] [Google Scholar]
- 104. Padilla, F., Puts, R., Vico, L.et al. 2014. Stimulation of bone repair with ultrasound: a review of the possible mechanic effects. Ultrasonics 54:1125. [DOI] [PubMed] [Google Scholar]
- 105. Rutjes, A., Nüesch, E., Sterchi, R.et al. 2010. Therapeutic ultrasound for osteoarthritis of the knee or hip. Cochrane Database Syst. Rev. 20:CD003132. [DOI] [PubMed] [Google Scholar]
- 106. Page, M., O’Connor, D., Pitt, V.et al. 2012. Therapeutic ultrasound for carpal tunnel syndrome. Cochrane Database Syst. Rev. 18:CD009601. [DOI] [PubMed] [Google Scholar]
- 107. Blackmore, J., Shrivastava, S., Sallet, J.et al. 2019. Ultrasound neuromodulation: a review of results, mechanisms and safety. Ultrasound Med. Biol. 45:1509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Ninet, J., Roques, X., Seitelberger, R.et al. 2005. Surgical ablation of atrial fibrillation with off-pump, epicardial, high-intensity focused ultrasound: results of a multicenter trial. J. Thorac. Cardiovasc. Surg. 130:803. [DOI] [PubMed] [Google Scholar]
- 109. Tempany, C. M., Stewart, E. A., McDannold, N.et al. 2003. MR imaging-guided focused ultrasound surgery of uterine leiomyomas: a feasibility study. Radiology 226:897. [DOI] [PubMed] [Google Scholar]
- 110. Klingler, H. C., Susani, M., Seip, R.et al. 2008. A novel approach to energy ablative therapy of small renal tumours: laparoscopic high-intensity focused ultrasound. Eur. Urol. 53:810. [DOI] [PubMed] [Google Scholar]
- 111. Miller, D. L., Smith, N. B., Bailey, M. R.et al. 2012. Overview of therapeutic ultrasound applications and safety considerations. J. Ultrasound Med. 31:623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Gigliotti, J. C., Huang, L., Ye, H.et al. 2013. Ultrasound prevents renal ischemia-reperfusion injury by stimulating the splenic cholinergic anti-inflammatory pathway. J. Am. Soc. Nephrol. 24:1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Gigliotti, J. C., Huang, L., Bajwa, A.et al. 2015. Ultrasound modulates the splenic neuroimmune axis in attenuating AKI. J. Am. Soc. Nephrol. 26:2470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Cotero, V., Fan, Y., Tsaava, T.et al. 2019. Noninvasive sub-organ ultrasound stimulation for targeted neuromodulation. Nat. Commun. 10:952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Anderson, D. W. and Barrett, J. T. 1979. Ultrasound: a new immunosuppressant. Clin. Immunol. Immunopathol. 14:18. [DOI] [PubMed] [Google Scholar]
- 116. Anderson, D. W. and Barrett, J. T. 1981. Depression of phagocytosis by ultrasound. Ultrasound Med. Biol. 7:267. [DOI] [PubMed] [Google Scholar]
- 117. Zachs, D. P., Offutt, S. J., Graham, R. S.et al. 2019. Noninvasive ultrasound stimulation of the spleen to treat inflammatory arthritis. Nat. Commun. 10:951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Wang, S., Li, B., Li, X.et al. 2020. Low-intensity ultrasound modulation may prevent myocardial infarction-induced sympathetic neural activation and ventricular arrhythmia. J. Cardiovasc. Pharmacol. 75:432. [DOI] [PubMed] [Google Scholar]
- 119. Wasilczuk, K. M., Bayer, K. C., Somann, J. P.et al. 2019. Modulating the inflammatory reflex in rats using low-intensity focused ultrasound stimulation of the vagus nerve. Ultrasound Med. Biol. 45:481. [DOI] [PubMed] [Google Scholar]
- 120. Binder, A., Hodge, G., Greenwood, A. M.et al. 1985. Is therapeutic ultrasound effective in treating soft tissue lesions? Br. Med. J. 290:512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Nagao, M., Tanabe, N., Manaka, S.et al. 2017. LIPUS suppressed LPS-induced IL-1α through the inhibition of NF-κB nuclear translocation via AT1-PLCβ pathway in MC3T3-E1 cells. J. Cell. Physiol. 232:3337. [DOI] [PubMed] [Google Scholar]
- 122. Chung, J. I., Barua, S., Choi, B. H.et al. 2012. Anti-inflammatory effect of low intensity ultrasound (LIUS) on complete Freund’s adjuvant-induced arthritis synovium. Osteoarthr. Cartil. 20:314. [DOI] [PubMed] [Google Scholar]
- 123. Eckel, R. H., Grundy, S. M. and Zimmet, P. Z. 2005. The metabolic syndrome. Lancet 365:1415. [DOI] [PubMed] [Google Scholar]
- 124. Bastard, J. P., Maachi, M., Lagathu, C.et al. 2006. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur. Cytokine Netw. 17:4. [PubMed] [Google Scholar]
- 125. Hotamisligil, G. S. 2006. Inflammation and metabolic disorders. Nature 444:860. [DOI] [PubMed] [Google Scholar]
- 126. Esser, N., Paquot, N. and Scheen, A. J. 2015. Anti-inflammatory agents to treat or prevent type 2 diabetes, metabolic syndrome and cardiovascular disease. Expert Opin. Investig. Drugs. 24:283. [DOI] [PubMed] [Google Scholar]
- 127. Huerta, T. S., Devarajan, A., Tsaava, T.et al. 2021. Targeted peripheral focused ultrasound stimulation attenuates obesity-induced metabolic and inflammatory dysfunctions. Sci. Rep. 11:5083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Chardack, W. M., Gage, A. A. and Greatbatch, W. 1960. A transistorized, self-contained, implantable pacemaker for the long-term correction of complete heart block. Surgery 48:643. [PubMed] [Google Scholar]
- 129. Trummer, M. J., Walter, E. P., Volk, R.et al. 1962. The use of a self-contained, implantable pacemaker in the long-term management of complete heart block. Mil. Med. 127:647. [PubMed] [Google Scholar]
- 130. Penry, J. K. and Dean, J. C. 1990. Prevention of intractable partial seizures by intermittent vagal stimulation in humans: preliminary results. Epilepsia 31(Suppl. 2):S40. [DOI] [PubMed] [Google Scholar]
- 131. Barker, A. T., Jalinous, R. and Freeston, I. L. 1985. Non-invasive magnetic stimulation of human motor cortex. Lancet 1:1106. [DOI] [PubMed] [Google Scholar]
- 132. Napadow, V., Edwards, R. R., Cahalan, C. M.et al. 2012. Evoked pain analgesia in chronic pelvic pain patients using respiratory-gated auricular vagal afferent nerve stimulation. Pain Med. 13:777. [DOI] [PMC free article] [PubMed] [Google Scholar]