Abstract
Background
Urine culturing practices are highly variable in long-term care and contribute to overprescribing of antibiotics for presumed urinary tract infections. The purpose of this study was to evaluate the use of virtual learning collaboratives to support long-term care homes in implementing a quality improvement programme focused on reducing unnecessary urine culturing and antibiotic overprescribing.
Methods
Over a 4-month period (May 2018–August 2018), 45 long-term care homes were self-selected from five regions to participate in virtual learning collaborative sessions, which provided an orientation to a quality improvement programme and guidance for implementation. A process evaluation complemented the use of a controlled before-and-after study with a propensity score matched control group (n=127) and a difference-in-difference analysis. Primary outcomes included rates of urine cultures performed and urinary antibiotic prescriptions. Secondary outcomes included rates of emergency department visits, hospital admission and mortality. An 18-month baseline period was compared with a 16-month postimplementation period with the use of administrative data sources.
Results
Rates of urine culturing and urinary antibiotic prescriptions per 1000 resident days decreased significantly more among long-term care homes that participated in learning collaboratives compared with matched controls (differential reductions of 19% and 13%, respectively, p<0.0001). There was no statistically significant changes to rates of emergency department visits, hospital admissions or mortality. These outcomes were observed with moderate adherence to the programme model.
Conclusions
Rates of urine culturing and urinary antibiotic prescriptions declined among long-term care homes that participated in a virtual learning collaborative to support implementation of a quality improvement programme. The results of this study have refined a model to scale this programme in long-term care.
Keywords: nursing homes, antibiotic management, implementation science, quality improvement
Background
Phased implementation efforts are designed to extend the benefits of evidence-based interventions to impact system-level outcomes. As there can be a drop in effectiveness when interventions move from controlled research protocols into real-world settings,1 there is a need to closely examine what supports contribute to successful implementation and how they can be replicated at scale. Intermediary organisations that scale evidence-based interventions are faced with the challenge of finding ways to package interventions and provide implementation support to extend benefits beyond a pilot stage.
The Urinary Tract Infection (UTI) Programme includes a focus on de-implementing low value practices that contribute to antibiotic overprescribing.2 In long-term care, prescribing is often driven by positive urine culture results in the absence of indicated signs and symptoms of a UTI, also known as asymptomatic bacteriuria.3 It has been estimated that a substantial proportion of antibiotics prescribed in long-term care are unnecessary and the most common reason for this is the treatment of asymptomatic bacteriuria.4–8 Ultimately, efforts to reduce inappropriate use of antibiotics can reduce antibiotic-related harms and improve resident health outcomes.4 9–11 A systematic review of antimicrobial stewardship strategies in long-term care found that UTIs were the most common area targeted to improve the quality of antibiotic use.12 However, the review emphasised the need to assess lower resource-intensive sustainable interventions in the long-term care setting.
Previous studies of the UTI Programme and similar multimodal programmes have described experts delivering on-site support.13 14 These types of supports present an opportunity to build the capacity of organisations to effectively implement practice change; however, tailored onsite visits may not be feasible beyond a pilot stage due to resource intensity. To help scale the UTI Programme in Ontario, Canada, virtual learning collaboratives were selected as a means to maintain support for implementation while also bringing together multiple long-term care homes (LTCHs) to facilitate shared learning.
A learning collaborative has been defined as the facilitation of groups of providers or organisations and fostering a collaborative learning environment to improve implementation of a clinical innovation.15 Originally described by the Institute for Healthcare Improvement with a focus on quality improvement initiatives in healthcare systems,16 this strategy brings together teams, practitioners or organisations with common components including didactic learning sessions combined with group discussion and skill building activities.17 18 While learning collaboratives have been widely used, they have not been well studied,17 and as a result, there has been a call for studies to verify the effectiveness of learning collaboratives for improving implementation.18 There is also a need for studies examining virtual sessions, as the majority of previous studies on this strategy have described the use of face-to-face meetings.16–18 Public health measures in response to coronavirus disease 2019 (COVID-19) have placed restrictions on in-person gatherings and access to LTCHs. To continue efforts to support the implementation of best practices, it is timely to examine the effectiveness of virtual options that can increase access to external supports and create connections between professionals working in this setting.
Considering past results demonstrating the effectiveness of multimodal interventions to improve practices in this area13 14 and what is known about the importance of providing implementation support, we postulated the following hypotheses: (i) participation in learning collaborative sessions would help maintain fidelity to the programme model and result in practice change and (ii) level of participation in the sessions and programme would be associated with better outcomes.
Accordingly, this study set out to evaluate the implementation and outcomes of a virtual learning collaborative to build programme and implementation-specific capacity among stakeholders in long-term care.
Methods
Reporting for this study followed the Transparent Reporting of Evaluations with Non-randomised Designs (TREND) statement (see online supplemental file 1).19
bmjqs-2020-012226supp001.pdf (238.5KB, pdf)
Design and setting
A quasi-experimental study design (longitudinal controlled before-and-after study) was used to assess the relationship between participating in the learning collaborative and subsequent impacts on practice change. Intervention facilities were self-selected and compared with matched control facilities that were passively exposed to provincial dissemination of electronic programme materials.
Participants
All LTCHs in Ontario were eligible for this intervention unless they had previously participated in a pilot study of the programme. Provincial administrative data sources provided the opportunity to establish the control group retrospectively.
A recruitment email was distributed in April 2018 to over 1600 contacts representing 620 LTCHs in Ontario. The email included electronic versions of programme materials. Contacts were prompted to contact the project team to further discuss interest and eligibility to receive additional implementation support.
Intervention
We described this intervention using the template for intervention description and replication checklist (see online supplemental file 2).20
bmjqs-2020-012226supp002.pdf (273.3KB, pdf)
The UTI Programme draws attention to specific practices that contribute to antibiotic overprescribing. This includes two practices that need to be de-implemented: discontinuing the use of dipsticks to diagnose UTIs21 22 and discontinuing routine annual or admission screening, if residents do not have indicated clinical signs and symptoms.23 This also includes three practices that need to be followed: obtaining urine cultures only when residents have indicated clinical signs and symptoms of a UTI24 25; promoting proper collection and storage of urine cultures and prescribing antibiotics only when specified criteria have been met and reassessing therapy when urine culture and sensitivity results are received.25 26
The UTI Programme also includes recommended strategies and resources to support the five practice changes described above.2 13 Materials include a clinical decision tool, designed for use with medically stable non-catheterised residents.27 The programme promotes strategies that can be used early during the change process to build readiness for change.28 Education and distribution of resources to frontline staff is a foundational strategy. Additional strategies are recommended to target known barriers to practice change including: coaching support; providing information to residents and families; improving documentation and communication of resident symptoms; monitoring progress with practice changes and delivering reminders. At this time of this study, all LTCHs in Ontario had access to electronic programme materials through Public Health Ontario’s (PHO) public facing website.29
PHO made a commitment to scale this quality improvement programme within the province through the delivery of support for implementation. Virtual learning collaboratives were selected to promote and support LTCHs with implementation. PHO is an intermediary organisation that provides scientific evidence and expert guidance to support partners in government, public health and healthcare. PHO was well positioned to promote and support implementation of this programme with internal expertise in antimicrobial stewardship and the presence of five Regional Infection Prevention an Control (IPAC) Support Teams situated across the province. These teams support a diverse range of stakeholders in the healthcare setting including long-term care.
All LTCHs interested in receiving implementation support selected an implementation lead from their home who participated in a readiness discussion with staff from the Regional IPAC Support Team to confirm the need for the programme, consider timing and available human resources and to discuss next steps (eg, securing senior leadership support).
PHO hosted five separate online learning collaboratives between May 2018 and August 2018, bringing together groups of LTCHs from the same geographic region. Regional learning collaboratives included three sessions that were 1–1.5 hours in length and were facilitated by staff from the Regional IPAC Support Team. A lead from each LTCH, typically a director or associate director of care with infection control responsibilities was asked to participate in the sessions.
Session 1 included a presentation on the UTI Programme and an overview of the importance of implementation planning and forming an implementation team. There was also a facilitated discussion around steps implementation leads were taking at their LTCH to establish readiness for the programme and plan for implementation. Implementation leads shared who they would be including on an implementation team. Session 2 included a facilitated discussion to identify anticipated barriers that would be most important at their LTCH. Facilitators then provided guidance on which recommended strategies would help overcome these types of barriers. The final session included a facilitated discussion on the status and details of action plans for each LTCH. There was also a focused discussion on planning for sustainability and approaches to monitoring practice changes. For additional details and materials, see online supplemental file 2.
Process evaluation
We measured participation in the learning collaborative through attendance tracking to document whether or not at least one representative from each LTCH was present. Programme withdrawal was also documented. Withdrawal was defined as situations where a LTCH decided not to implement the programme after attending a learning collaborative session or where the implementation lead from a LTCH only attended one session with no further contact.
The 45 implementation leads from each LTCH who attended the learning collaborative sessions were asked to provide feedback on the learning collaborative and to report on how they implemented the programme. Implementation leads were directors of care, associate directors of care or registered nurses with infection control responsibilities. Online surveys were used to collect this information. Following the delivery of the three learning collaborative sessions, an online survey was administered in October 2018 (online supplemental file 3). A second follow-up survey was administered 7 months after the learning collaborative sessions (March 2019) to learn how each LTCH implemented the programme (online supplemental file 3). Invitations to complete the survey and two reminders were sent to implementation leads by email from the Regional IPAC Support Team who delivered the learning collaborative sessions.
bmjqs-2020-012226supp003.pdf (331.5KB, pdf)
Outcomes
Primary outcomes included the number of urine cultures performed and number of urinary antibiotic prescriptions per 1000 resident days. These outcomes were selected as the programme ultimately aims to reduce antibiotic prescribing for asymptomatic bacteriuria and places a strong focus on reducing unnecessary urine culturing. For the purposes of this study, urinary antibiotics were defined as: ciprofloxacin, norfloxacin, nitrofurantoin, fosfomycin or trimethoprim with or without sulfonamides/sulfamethoxazole. A secondary outcome was total antibiotic prescriptions per 1000 resident days to detect whether there was a shift from one class of antibiotics to another. We incorporated additional balancing measures to monitor any signal of undertreatment of UTIs including rates of emergency department visits, hospital admissions and mortality.14
Data sources
Data to support the outcome assessment was available from existing administrative databases at ICES (formerly, the Institute for Clinical Evaluative Sciences). ICES is an independent, non-profit institute whose legal status under the Ontario’s health information privacy law allows it to collect and analyse healthcare demographic data, without consent, for health system evaluation and improvement. These datasets are linked using unique encoded identifiers and analysed at ICES (see online supplemental file 4 for all databases used in this study). ICES databases go through routine data quality assessments based on a data quality framework.30 The source of antibiotic prescribing data was from the Ontario Drug Benefit (ODB) database, which collects antibiotic dispensing data for patients over the age of 65 years and/or residents of LTCHs. Compared with manual chart review, ODB is associated with a 99.3% accuracy in determining drug dispensing.31
bmjqs-2020-012226supp004.pdf (171.6KB, pdf)
Sample size
We determined that a minimum of 32 LTCHs would be required to detect a statistically significant difference between treatment and control groups with 80% power and 5% significance for rates of urine cultures performed. This estimate was based on the following assumptions: an event rate difference of −1.0 per 1000 resident days, control event rate of 3.2 per 1000 resident days, an average home bed size of 16013 and an assumption that the coefficient of variation was 0.25. Using similar methods but with an event rate difference of −0.6 and control event rate of 1.5 per 1000 resident days, we determined that 22 LTCHs would be required for rates of urinary antibiotic prescriptions.13
Assignment method
We used propensity score matching to establish controls for this study using 1:3 nearest neighbour matching with a calliper size of 0.2. A logistic regression model was used to calculate propensity scores for all LTCHs in Ontario that met eligibility criteria. Scores were calculated based on predicted probability of being in the intervention group based on baseline rates of urine culturing and total antibiotic prescriptions in addition to bed size. LTCHs were eligible for inclusion in the control group if they had not previously participated in a pilot of the programme; had not closed during the study period or did not have counts of zero urine cultures over the full baseline period.
Analyses
The balance of baseline facility and resident level characteristics between intervention and control LTCHs was assessed with standardised differences.32 A threshold of ≤0.1 is recommended to consider differences between groups.32
Outcome evaluation
We defined an 18-month baseline period (November 2016–April 2018), a 4-month implementation planning phase (wash-in period) (May 2018–August 2018) and a 16-month postmplementation phase (September 2018–December 2019). In the study protocol, the point of intervention was defined as the start of the postimplementation phase. It was expected that LTCHs would need time to participate in the learning collaborative and plan for implementation before seeing changes in practice.
A difference-in-difference analysis was used to examine changes in outcome variables.33 34 This estimate is the difference in average outcome in the intervention homes before and after implementation minus the difference in average outcome in the control homes before and after implementation. This method requires that baseline temporal trends are equivalent with the idea that trends in both groups would have continued to be similar in the absence of receiving support to implement this programme. A Poisson regression model was used with the unit of analysis as monthly outcome data for each facility over the 38-month study period, and random-intercepts corresponding to each home. Resulting effect estimates from the difference-in-difference analysis were used to calculate the relative difference in the decline.34
The primary analysis focused on LTCHs that were recruited irrespective of whether they withdrew during the study period (an intention-to-treat analysis). There was no missing data for the outcome assessment. We conducted additional sensitivity analyses to examine how level of engagement with the programme could be diluting the intervention effect. We examined outcomes excluding LTCHs that withdrew (per-protocol analysis). The analysis was also replicated focusing only on the LTCHs that withdrew. We also separately examined intervention effects among those LTCHs that attended all three learning collaborative sessions. All analyses were performed using a commercial statistical software (Statistical Analysis System for Windows, V.9.4 using the SAS Enterprise Guide V.7.15, SAS Institute, Cary, North Carolina, USA).
Results
Recruitment
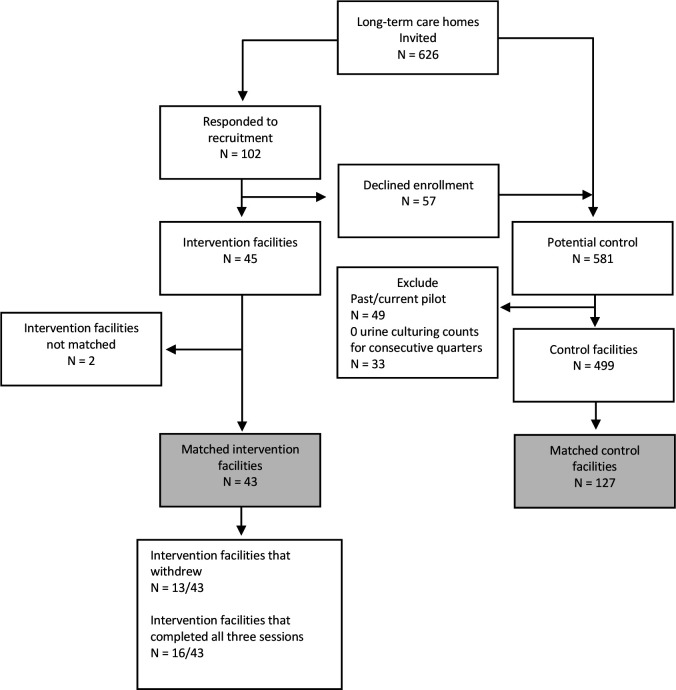
Following the recruitment notice, of the 102 LTCHs that responded, 45 agreed to attend the learning collaborative after participating in a readiness assessment and 57 LTCHs declined enrolment (figure 1). Twenty-five LTCHs that declined enrolment expressed interest in a future wave. The remaining 32 LTCHs did not enrol due to either: competing priorities, decided to implement without the learning collaborative or there was no response following an initial email of interest. LTCHs that declined enrollment were eligible for the control group.
Figure 1.
Description of sample, recruitment and allocation results.
Process evaluation
Of the 45 LTCHs that signed up for the programme, 13 withdrew. Only 16 LTCHs (36%) attended all three sessions. Eighty-four per cent of LTCHs attended the first session, 64% attended session two and 49% attended session three. Among LTCHs that submitted the first survey (n=27/45), 89% (n=24/27) indicated they felt more prepared for implementation after participating in the learning collaborative. Thirty-seven per cent (n=10/27) felt that LTCHs would need additional support after the sessions. While the majority agreed (n=20/27) that the peer support component was valuable, 26% disagreed or felt neutral about this component (online supplemental file 2).
There were 17 comments shared by implementation leads who had attended the learning collaborative sessions (see online supplemental file 3). Positive feedback was received on the quality of programme resources, facilitation support and the peer support component. Recommendations for improvement focused on timing of the sessions considering LTCHs may need to implement the programme at a different pace. For example, one participant noted: “The issue was that our home rolled out the programme fairly quickly then the collaborative was still discussing steps we had already completed”. Participants also noted that additional interactions between PHO staff and LTCH staff may have been beneficial to increase engagement and buy-in.
Eleven implementation strategies are described in the UTI Programme. Table 1 includes a summary of implementation strategies that were reported as adopted by implementation leads representing the 32 LTCHs that participated in the learning collaborative and implemented the programme (ie, did not withdraw). Twenty-nine of the 30 LTCHs that submitted the final survey reported using at least one recommended strategy and on average reported using seven strategies. Six LTCHs reported using all of the 11 recommended strategies. The most common types of strategies selected were educational including providing education to staff (84%), coaching (78%) and delivering education to families and residents (78%) (table 1). There was lower uptake of strategies designed to build readiness for implementation and notably use of implementation teams (47%) (table 1).
Table 1.
Adoption of recommended implementation strategies among participating long-term care homes (n=32)
| Implementation strategy description | Complete n (%) |
Incomplete* n (%) |
No response† n (%) |
| Readiness strategies | |||
| Consensus: ensuring agreement on the practices among prescribers | 24 (75) | 5 (16) | 3 (9) |
| Policies and procedures: ensuring policies and procedures are aligned with five practice changes | 23 (72) | 6 (19) | 3 (9) |
| Champion: selecting a champion and involving them in implementation | 21 (66) | 8 (25) | 3 (9) |
| Local opinion leaders: finding way for prescribers to be involved in delivering strategies | 19 (59) | 10 (31) | 3 (9) |
| Implementation team: having at least three people that are part of an implementation team | 15 (47) | 14 (44) | 3 (9) |
| Education and support | |||
| Delivering education to staff | 27 (84) | 1 (3) | 4 (13) |
| Coaching: ensuring there is someone to help with questions/concerns after education | 25 (78) | 3 (9) | 4 (13) |
| Providing information/education to residents and families | 25 (78) | 3 (9) | 4 (13) |
| Monitoring and communication | |||
| Resident symptoms: addressing improvement in how resident symptoms are documented and communicated | 26 (81) | 1 (3) | 5 (16) |
| Reminders: looking for opportunities to remind staff about the practice changes | 23 (72) | 4 (13) | 5 (16) |
| Process surveillance: checking records of urine cultures and antibiotics prescribed for UTIs to see how they are aligned with the programme algorithm | 22 (69) | 5 (16) | 5 (16) |
*Incomplete refers to LTCHs that indicated they did not complete this strategy.
†No response refers to LTCHs that did not complete the survey or question. Proportions reflect the total number of participating LTCHs and not the total number of LTCHs that responded to the survey.
LTCHs, long-term care homes; UTI, urinary tract infection.
Matching results
A total of 43 intervention LTCHs were matched to 127 controls (figure 1). Two intervention facilities could not be matched due to their very large bed size. The resulting sample provided diversity in facility-level characteristics including a mix of urban and rural locations and variation in size (table 2). Resident-level characteristics including demographics, functional status and device use were similar between intervention LTCHs and matched controls (table 2). Baseline rates for the primary outcomes were also similar between the two groups; however, there was a small difference in the baseline rate of urine cultures performed (table 2).
Table 2.
Baseline characteristics of residents and long-term care homes (LTCHs)
| Characteristics | Matched intervention LTCHs (n=43) |
Matched control LTCHs (n=127) |
Standardised differences* |
| Bed size†, mean (SD) | 128.3 (70.8) | 136.2 (76.8) | −0.11 |
| Region | |||
| Urban (%) | 33 (77) | 106 (84) | −0.17 |
| Rural (%) | 10 (23) | 21 (17) | |
| Mean rate at baseline per 1000 resident days | |||
| Urine culturing† (IQR) | 4.0 (2.5–5.5) | 3.4 (2.2–4.9) | 0.15 |
| Total antibiotic prescriptions† (IQR) | 4.2 (3.7–5.1) | 4.4 (3.5–5.2) | 0.05 |
| Urinary antibiotic prescriptions (IQR) | 1.5 (1.2–2.0) | 1.4 (1.1–1.8) | 0.08 |
| Hospital admissions (IQR) | 1.7 (1.4–2.3) | 2.0 (1.6–2.5) | −0.40 |
| No. unique residents | 7960 | 24 827 | |
| Mean resident age (SD) | 85.17 (8.1) | 84.79 (8) | 0.05 |
| Female proportion (%) | 5515 (69) | 17 023 (69) | 0.02 |
| Comorbidities, n (%): | |||
| Residents with severe cognitive impairment‡ | 2857 (36) | 8818 (36) | 0.01 |
| Dementia and Alzheimer’s disease | 5423 (68) | 16 713 (67) | 0.02 |
| Paralysis | 14 (0.2) | 110 (0.4) | −0.05 |
| Parkinson’s disease | 550 (7) | 1649 (7) | 0.01 |
| Obstructive lung disease | 1510 (19) | 4475 (18) | 0.02 |
| Cancer | 768 (10) | 2812 (11) | −0.06 |
| Gastrointestinal disease | 2546 (32) | 7055 (28) | 0.08 |
| Liver disease | 94 (1) | 365 (2) | −0.03 |
| Diabetes mellitus | 2229 (28) | 7173 (29) | −0.02 |
| Renal disease | 775 (10) | 2815 (11) | −0.05 |
| Atherosclerotic heart disease | 1367 (17) | 4172 (17) | 0.01 |
| Congestive heart failure | 1156 (15) | 3210 (13) | 0.05 |
| Peripheral vascular disease | 555 (7) | 1521 (6) | 0.03 |
| Stroke | 2055 (26) | 6392 (26) | 0.002 |
| Functional status, n (%) | |||
| Bladder incontinence | 7337 (92) | 22 412 (90) | 0.07 |
| Bowel incontinence | 6371 (80) | 19 513 (79) | 0.04 |
| Devices, n (%) | |||
| Indwelling catheter | 582 (7) | 1526 (6) | 0.05 |
| Other urinary catheter | 40 (0.5) | 212 (0.9) | −0.04 |
| Feeding tube | 56 (0.7) | 241 (1.0) | −0.03 |
*A threshold of ≤0.1 is recommended to consider differences between groups.
†Variables used to match intervention to control LTCHs.
‡Cognitive Performance Scale ≥4.
Urine cultures
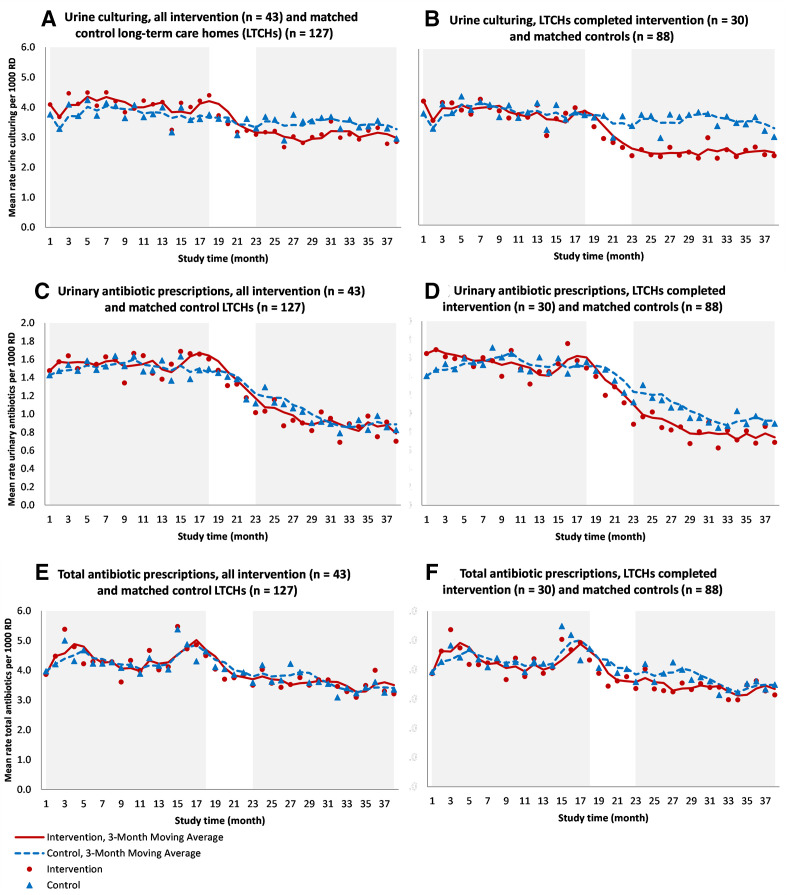
Figure 2A graphs 3-month moving averages for rates of urine cultures performed per 1000 resident days over the study period for matched intervention and control LTCHs. Rates of urine cultures performed per 1000 resident days started to decline early during the implementation planning phase when learning collaboratives were hosted by PHO. For the full cohort of LTCHs recruited (intention-to-treat analysis) (n=45), between the 18-month baseline period and the 16-month postimplementation period, the difference-in-difference analysis showed that the change in the rate of urine cultures performed was 19% lower [(1–exp (−0.21)] among LTCHs that participated in the learning collaboratives than the control group (p<0.0001) (table 3).
Figure 2.
Rates of urine culturing, urinary antibiotic prescriptions and total antibiotic prescriptions per 1000 resident days (RD) for intervention and matched control long-term care homes (LTCHs), November 2016–December 2019.
Table 3.
Baseline and postimplementation rates in the intervention and control facilities and difference-in-difference estimates for the intention-to-treat analysis
| Outcome | Control (n=127) | Intervention (n=43) | Difference-in-difference | ||||
| Before mean (SD) | After mean (SD) | Before mean (SD) | After mean (SD) | Estimate† | SE | 95% CI | |
| Urine culturing | 3.8 (2.2) | 3.4 (2.0) | 4.1 (1.9) | 3.1 (2.1) | −0.21** | 0.02 | −0.24 to to 0.17 |
| Urinary antibiotics | 1.5 (0.7) | 1.0 (0.5) | 1.6 (0.7) | 0.9 (0.5) | −0.13** | 0.03 | −0.19 to to 0.07 |
| Total antibiotics | 4.4 (1.1) | 3.6 (1.2) | 4.4 (1.2) | 3.5 (1.1) | −0.05* | 0.02 | −0.08 to to 0.01 |
| Acute care admission | 2.1 (0.8) | 2.0 (0.7) | 1.8 (0.6) | 1.8 (0.7) | 0.01 | 0.02 | −0.03 to 0.06 |
| Mortality | 1.8 (0.6) | 1.9 (0.7) | 1.9 (0.4) | 2.1 (0.6) | 0.01 | 0.04 | −0.06 to 0.09 |
*P<0.01; **p<0.0001.
†From the difference-in-difference estimate (Poisson random-effects model). Before=baseline period from November 2016–April 2018. After=postimplementation period from September 2018–December 2019.
Prescriptions
Figure 2 also graphs 3-month moving averages for urinary antibiotic prescriptions (figure 2C) and total antibiotic prescriptions (figure 2E) per 1000 resident days over the study period for matched intervention and control LTCHs. There was a steady decline in rates of urinary antibiotic prescriptions in both groups during the study period; however, the decline was more pronounced for LTCHs in the intervention group. For the full cohort of LTCHs recruited (intention-to-treat analysis) (n=45), the change in the rate of urinary antibiotics prescribed was 13% lower [(1–exp (−0.13)] among LTCHs that participated in the learning collaborative than the control group (p<0.0001) (table 3). The change in the rate of total antibiotic prescriptions was 5% lower [(1–exp (−0.05)] among intervention LTCHs (p<0.01) (table 3).
Secondary outcomes
There were no signs of undertreatment of UTIs with no significant changes in rates of mortality or rates of emergency department visits and hospital admissions (table 3).
Sensitivity analysis
For the per-protocol analysis that excluded LTCHs that withdrew (did not go on to implement the programme) (n=30), reductions were even more substantial (figure 2B, D, F). The change in the rate of urine cultures performed was 28% lower [1–exp (−0.33)] among LTCHs that participated in the learning collaborative and went on to implement the programme compared with LTCHs in the control group (p<0.0001) (online supplemental file 5). The change in rate of urinary antibiotics prescribed was 20% lower [1–exp (−0.22)] (p<0.0001) and 6% lower [1–exp (−0.06)] for total antibiotic prescriptions (p<0.01) (online supplemental file 5). LTCHs that withdrew during the study period (n=13), did not see significant change in primary and secondary outcomes (online supplemental file 4). Finally, restricting the sample to LTCHs that attended all scheduled learning collaboratives (n=16) produced similar outcomes as the per-protocol analysis for rates of urine culturing and urinary antibiotic prescriptions (differential reductions of 34% and 19%, respectively, p<0.0001) (online supplemental file 5).
bmjqs-2020-012226supp005.pdf (248KB, pdf)
Discussion
The results of the difference-in-difference analysis provided support to the study hypothesis that participation in the learning collaborative sessions would result in practice changes targeted by the UTI Programme. There was a statistically significant decline in both rates of urine cultures performed and urinary antibiotics prescribed between the baseline and postimplementation period among LTCHs that participated in the intervention, after accounting for declines in the control group. The rate of total antibiotic prescriptions also declined during the study period. These findings are further supported by a per-protocol analysis that showed more substantial changes among LTCHs that did not withdraw. The results also support the hypothesis that the uptake of evidence-based practices to reduce antibiotic overprescribing for asymptomatic bacteriuria would not result in signs of undertreatment including changes in rates of emergency department visits, hospital admissions and mortality.14 35
To some extent, this study demonstrates an association between outcomes and the level of participation in the sessions and programme. There were no significant changes in primary outcomes among LTCHs that did not go on to implement the programme; however, LTCHs reduced rates of urine culturing and urinary antibiotic use through variable adoption of recommended strategies. This aligns with an important assumption made in the development of this programme where LTCHs may encounter different barriers to practice change and thus may require different types of implementation strategies.2 A sensitivity analysis showed that those attending all scheduled sessions did not necessarily have better outcomes. This further supports the idea that different LTCHs may require tailored support, dependent on their needs.
Overall trends show declines in both primary outcomes over the observation period for both intervention and control groups. It is important to note that an email promoting the UTI Programme went out to all LTCHs in Ontario during the recruitment phase for this intervention with links to programme materials. Incorporating the difference-in-difference analysis was an important component of the study design to measure the added value of learning collaborative sessions in the context of many other regional and national antimicrobial stewardship campaigns that were launched during the study period.
Implications
Results are consistent with studies on multimodal improvement programmes in this area.13 14 However, in previous studies, external experts came on-site to support implementation. The findings from this study have demonstrated the value of virtual sessions with peer organisations for knowledge exchange and implementation support in long-term care. The fact that these sessions were virtual made the scale-up of this model more feasible. This study aligns with promising findings from a few emerging studies looking at the value of learning collaboratives for supporting practice change across disciplines and areas of clinical practice improvement.36–40
Based on the results of this study, a refined virtual implementation support model for the scale-up of this programme was developed including more tailored support for LTCHs. Engaging LTCHs in multisession learning collaboratives could be better facilitated through shorter sessions; more flexible scheduling; encouraging homes to plan ahead for staff transitions and providing additional resources for antibiotic stewardship.
Generalisability
Sustained outcomes were achieved with virtual learning collaboratives that were delivered without added travel or additional funding and that relied on existing resources. Outcomes were also observed with moderate adherence to recommended implementation strategies. Considering generalisability of the findings, a large number of LTCHs with diverse facility-level characteristics (eg, urban and rural locations and variation in size) were involved in this study. However, the recruitment process, documentation of challenges to implementation and reasons for having to withdraw during the study period demonstrate the importance of using strategies that strengthen readiness for implementation in order to replicate outcomes of this study.
Limitations
These results need to be interpreted in light of a few limitations. Use of the clinical decision-making tool and process surveillance forms were not assessed which limited opportunities to examine more precisely changes in the proportion of urine cultures performed and antibiotics prescribed in the absence of indicated signs and symptoms of a UTI. Efforts were made to capture antibiotic prescriptions commonly used to treat UTIs; however, some prescriptions meant for other indications may have been included. While the outcomes were assessed with knowledge of LTCH’s group allocation, the use of administrative data sources for the primary and secondary outcomes were objective.
The overall impact of the programme was modest and the sensitivity analyses demonstrated that impact could have been enhanced if fewer LTCHs had to withdraw from the programme. There are a number of challenges to implementing antibiotic stewardship in LTCH considering available resources in this area.
This study used a non-randomised study design for operational reasons and while efforts were made to match intervention facilities and compare across a number of facility and resident-level characteristics, there may be additional unmeasured differences between intervention and control facilities. Notably, there could be differences in baseline readiness and other contextual factors that contributed to LTCHs ability to improve practices in this area. However, it is important to point out that the UTI Programme was designed for LTCHs that need to build readiness and overcome barriers to practice change. LTCHs with more resources, experience with implementing quality improvement initiatives and with fewer barriers may be more likely to implement practice changes independently from the learning collaboratives. To further investigate the contribution of virtual learning collaboratives in improving antibiotic use in long-term care, future research could potentially randomise LTCHs to either learning collaboratives or a less intensive support strategy or randomise the start of learning collaboratives to different time intervals.41
While efforts were made to measure components of implementation quality for both the learning collaborative sessions and UTI Programme through attendance tracking and surveys, additional interviews with more staff from each LTCH could provide more information about how well the learning collaborative and UTI Programme were implemented.
Conclusion
Rates of urine culturing and urinary antibiotic prescriptions declined among LTCHs that participated in a virtual learning collaborative to support implementation of a quality improvement programme. There was positive feedback on the sessions in addition to the intervention package. However, it was evident that not all LTCHs required the same level of support and faced challenges attending multiple group sessions. The scale-up of this programme would benefit from more tailored support and guidance on building readiness for practice change.
Acknowledgments
We would like to thank members of the UTI Program team at Public Health Ontario that supported the delivery of the learning collaborative sessions. We would also like to thank all the leads from participating long-term care homes that provided feedback on the programme. Finally, we would like to thank Dr Lennon Li for advising on the study protocol and power calculations.
Footnotes
Contributors: AC led the design of the study and contributed to analysis and interpretation of data. GG provided overall supervision, contributed to the study design and interpretation of data. CC conducted the statistical analysis of the outcome data. KA contributed to the design of the study, provided coordination support to access the data and contributed to analysis and interpretation of data. KB, ND, BL, VL, KS, JEM, JQ, SM and TC contributed expertise to the study design and interpretation of data. All authors contributed to drafting and revising the article, and final approval of the version for submission.
Funding: This study was conducted with Public Health Ontario operational funds. This study was also supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and Long-term Care (MOHLTC). Parts of this material are based on data and/or information compiled and provided by the Canadian Institute for Health Information (CIHI). The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES, CIHI or the Ontario MOHLTC is intended or should be inferred.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available on reasonable request. Data may be obtained from a third party and are not publicly available. Survey data for the process measures is available on reasonable request. The dataset from this study is held securely in coded form at ICES. While data sharing agreements prohibit ICES from making the dataset publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at www.ices.on.ca/DAS. The full dataset creation plan and underlying analytic code are available from the authors on request, understanding that the computer programs may rely on coding templates or macros that are unique to ICES and are therefore either inaccessible or may require modification.
Ethics statements
Patient consent for publication
Not required.
References
- 1. Kilbourne AM, Neumann MS, Pincus HA, et al. Implementing evidence-based interventions in health care: application of the replicating effective programs framework. Implement Sci 2007;2:42. 10.1186/1748-5908-2-42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Chambers A, MacFarlane S, Zvonar R, et al. A recipe for antimicrobial stewardship success: using intervention mapping to develop a program to reduce antibiotic overuse in long-term care. Infect Control Hosp Epidemiol 2019;40:24–31. 10.1017/ice.2018.281 [DOI] [PubMed] [Google Scholar]
- 3. Flokas ME, Andreatos N, Alevizakos M, et al. Inappropriate management of asymptomatic patients with positive urine cultures: a systematic review and meta-analysis. Open Forum Infect Dis 2017;4:ofx207. 10.1093/ofid/ofx207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Nicolle LE, Bentley DW, Garibaldi R, et al. Antimicrobial use in Long-Term–Care facilities. Infect. Control Hosp. Epidemiol. 2000;21:537–45. 10.1086/501798 [DOI] [PubMed] [Google Scholar]
- 5. Morrill HJ, Caffrey AR, Jump RLP, et al. Antimicrobial stewardship in long-term care facilities: a call to action. J Am Med Dir Assoc 2016;17:183.e1–183.e16. 10.1016/j.jamda.2015.11.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Peron EP, Hirsch AA, Jury LA, et al. Another setting for stewardship: high rate of unnecessary antimicrobial use in a Veterans Affairs long-term care facility. J Am Geriatr Soc 2013;61:289–90. 10.1111/jgs.12099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Daneman N, Gruneir A, Newman A, et al. Antibiotic use in long-term care facilities. J Antimicrob Chemother 2011;66:2856–63. 10.1093/jac/dkr395 [DOI] [PubMed] [Google Scholar]
- 8. Thornley T, Ashiru-Oredope D, Normington A, et al. Antibiotic prescribing for residents in long-term-care facilities across the UK. J Antimicrob Chemother 2019;74:1447–51. 10.1093/jac/dkz008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Daneman N, Bronskill SE, Gruneir A, et al. Variability in antibiotic use across nursing homes and the risk of antibiotic-related adverse outcomes for individual residents. JAMA Intern Med 2015;175:1331–9. 10.1001/jamainternmed.2015.2770 [DOI] [PubMed] [Google Scholar]
- 10. Shehab N, Patel PR, Srinivasan A, et al. Emergency department visits for antibiotic-associated adverse events. Clin Infect Dis 2008;47:735–43. 10.1086/591126 [DOI] [PubMed] [Google Scholar]
- 11. Daneman N, Gruneir A, Bronskill SE, et al. Prolonged antibiotic treatment in long-term care: role of the prescriber. JAMA Intern Med 2013;173:673–82. 10.1001/jamainternmed.2013.3029 [DOI] [PubMed] [Google Scholar]
- 12. Wu JH-C, Langford BJ, Daneman N, et al. Antimicrobial stewardship programs in long-term care settings: a meta-analysis and systematic review. J Am Geriatr Soc 2019;67:392–9. 10.1111/jgs.15675 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Brown KA, Chambers A, MacFarlane S, et al. Reducing unnecessary urine culturing and antibiotic overprescribing in long-term care: a before-and-after analysis. CMAJ Open 2019;7:E174–81. 10.9778/cmajo.20180064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Pasay DK, Guirguis MS, Shkrobot RC, et al. Antimicrobial stewardship in rural nursing homes: impact of interprofessional education and clinical decision tool implementation on urinary tract infection treatment in a cluster randomized trial. Infect Control Hosp Epidemiol 2019;40:432–7. 10.1017/ice.2019.9 [DOI] [PubMed] [Google Scholar]
- 15. Powell BJ, Waltz TJ, Chinman MJ, et al. A refined compilation of implementation strategies: results from the expert recommendations for implementing change (ERIC) project. Implement Sci 2015;10:21. 10.1186/s13012-015-0209-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Institute for Healthcare Improvement . The Breakthrough Series: IHI’s collaborative model for achieving breakthrough improvement (IHI Innovation Series white paper). Boston: Institute for Healthcare Improvement, 2003. http://www.ihi.org/resources/Pages/IHIWhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchievingBreakthroughImprovement.aspx [Google Scholar]
- 17. Nadeem E, Olin SS, Hill LC, et al. A literature review of learning Collaboratives in mental health care: used but untested. Psychiatr Serv 2014;65:1088–99. 10.1176/appi.ps.201300229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Bunger AC, Doogan N, Hanson RF, et al. Advice-seeking during implementation: a network study of clinicians participating in a learning collaborative. Implement Sci 2018;13:101. 10.1186/s13012-018-0797-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Des Jarlais DC, Lyles C, Crepaz N, et al. Improving the reporting quality of nonrandomized evaluations of behavioral and public health interventions: the trend statement. Am J Public Health 2004;94:361–6. 10.2105/AJPH.94.3.361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hoffmann TC, Glasziou PP, Boutron I, et al. Better reporting of interventions: template for intervention description and replication (TIDieR) checklist and guide. BMJ 2014;348:g1687. 10.1136/bmj.g1687 [DOI] [PubMed] [Google Scholar]
- 21. Juthani-Mehta M, Tinetti M, Perrelli E, et al. Role of dipstick testing in the evaluation of urinary tract infection in nursing home residents. Infect Control Hosp Epidemiol 2007;28:889–91. 10.1086/518752 [DOI] [PubMed] [Google Scholar]
- 22. Nicolle LE, SHEA Long-Term-Care-Committee . Urinary tract infections in long-term-care facilities. Infect Control Hosp Epidemiol 2001;22:167–75. 10.1086/501886 [DOI] [PubMed] [Google Scholar]
- 23. Nicolle LE, Gupta K, Bradley SF, et al. Clinical practice guideline for the management of asymptomatic bacteriuria: 2019 update by the infectious diseases Society of America. Clin Infect Dis 2019;68:1611–5. 10.1093/cid/ciz021 [DOI] [PubMed] [Google Scholar]
- 24. Loeb M, Brazil K, Lohfeld L, et al. Effect of a multifaceted intervention on number of antimicrobial prescriptions for suspected urinary tract infections in residents of nursing homes: cluster randomised controlled trial. BMJ 2005;331:669. 10.1136/bmj.38602.586343.55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Nace DA, Perera SK, Hanlon JT, et al. The improving outcomes of UTI management in long-term care project (IOU) consensus guidelines for the diagnosis of uncomplicated cystitis in nursing home residents. J Am Med Dir Assoc 2018;19:765–9. 10.1016/j.jamda.2018.05.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Nicolle LE, Bradley S, Colgan R, et al. Infectious diseases Society of America guidelines for the diagnosis and treatment of asymptomatic bacteriuria in adults. Clin Infect Dis 2005;40:643–54. 10.1086/427507 [DOI] [PubMed] [Google Scholar]
- 27. Public Health Ontario . Urinary tract infection (UTI) program: when to obtain urine cultures in medically stable non-catheterized residents. Toronto, ON: Public Health Ontario, 2019. https://www.publichealthontario.ca/-/media/documents/U/2016/uti-assessment-algorithm.pdf [Google Scholar]
- 28. Leeman J, Birken SA, Powell BJ, et al. Beyond "implementation strategies": classifying the full range of strategies used in implementation science and practice. Implement Sci 2017;12:125. 10.1186/s13012-017-0657-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Public Health Ontario . Urinary tract infection (UTI) program. Toronto, ON: Public Health Ontario, 2019. https://www.publichealthontario.ca/en/health-topics/antimicrobial-stewardship/uti-program [Google Scholar]
- 30. Azimaee M, Kalappa G, Ji S. Datafit toolkit user guide: a practical approach to population data quality assessment Toronto, on: ICES, 2019. https://github.com/icescentral/DataFit-Toolkit [Google Scholar]
- 31. Levy AR, O'Brien BJ, Sellors C, et al. Coding accuracy of administrative drug claims in the Ontario drug benefit database. Can J Clin Pharmacol 2003;10:67–71. [PubMed] [Google Scholar]
- 32. Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res 2011;46:399–424. 10.1080/00273171.2011.568786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wing C, Simon K, Bello-Gomez RA. Designing difference in difference studies: best practices for public health policy research. Annu Rev Public Health 2018;39:453–69. 10.1146/annurev-publhealth-040617-013507 [DOI] [PubMed] [Google Scholar]
- 34. Zhou H, Taber C, Arcona S, et al. Difference-in-differences method in comparative effectiveness research: utility with unbalanced groups. Appl Health Econ Hea 2016;14:419–29. 10.1007/s40258-016-0249-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Brown KA, Daneman N, Schwartz KL, et al. The urine-culturing cascade: variation in nursing home urine culturing and association with antibiotic use and Clostridiodes difficile infection. Clin Infect Dis 2020;70:1620–7. 10.1093/cid/ciz482 [DOI] [PubMed] [Google Scholar]
- 36. Lang J, Connell C, Barrette K. Clinician and agency factors associated with implementation outcomes in learning Collaboratives. Presented at: 4th Biennial Conference of the Society for Implementation Research Collaboration (SIRC), 2017. [Google Scholar]
- 37. Johnson C, Mallard K, Carreño P. The effect of message board correspondence on therapist fidelity and adaptation in cognitive processing therapy for PTSD. Presented at: 4th Biennial Conference of the Society for Implementation Research Collaboration (SIRC), 2017. [Google Scholar]
- 38. Harris SB, Green ME, Brown JB, et al. Impact of a quality improvement program on primary healthcare in Canada: a mixed-method evaluation. Health Policy 2015;119:405–16. 10.1016/j.healthpol.2014.10.019 [DOI] [PubMed] [Google Scholar]
- 39. Zubkoff L, Neily J, Delanko V, Young-Xu Y, et al. How to prevent falls and fall-related injuries: a virtual breakthrough series collaborative in long term care. Phys Occup Ther Geriatr 2019;37:234–46. 10.1080/02703181.2019.1636923 [DOI] [Google Scholar]
- 40. Salem-Schatz S, Griswold P, Kandel R, et al. A statewide program to improve management of suspected urinary tract infection in long-term care. J Am Geriatr Soc 2020;68:62–9. 10.1111/jgs.16261 [DOI] [PubMed] [Google Scholar]
- 41. Hemming K, Haines TP, Chilton PJ, et al. The stepped wedge cluster randomised trial: rationale, design, analysis, and reporting. BMJ 2015;350:h391. 10.1136/bmj.h391 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjqs-2020-012226supp001.pdf (238.5KB, pdf)
bmjqs-2020-012226supp002.pdf (273.3KB, pdf)
bmjqs-2020-012226supp003.pdf (331.5KB, pdf)
bmjqs-2020-012226supp004.pdf (171.6KB, pdf)
bmjqs-2020-012226supp005.pdf (248KB, pdf)
Data Availability Statement
Data are available on reasonable request. Data may be obtained from a third party and are not publicly available. Survey data for the process measures is available on reasonable request. The dataset from this study is held securely in coded form at ICES. While data sharing agreements prohibit ICES from making the dataset publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at www.ices.on.ca/DAS. The full dataset creation plan and underlying analytic code are available from the authors on request, understanding that the computer programs may rely on coding templates or macros that are unique to ICES and are therefore either inaccessible or may require modification.