Introduction
The increasing prevalence of noncommunicable diseases in parallel with a high prevalence of communicable diseases threatens the lives of many in subSaharan Africa (SSA) and will overwhelm the already inadequate health budgets of African countries. CKD is common in SSA, with an estimated average prevalence of 14% (1). Patients with CKD are at increased risk of developing ESKD, cardiovascular disease, and death. The estimated prevalence of ESKD in SSA from diabetes and hypertension is 239 per million population, and of these only 2% of individuals who develop CKD stage 5 have access to maintenance dialysis or kidney transplantation (2). In a systematic review from Africa, 95% of adults and children with ESKD who could not access dialysis died or were presumed to have died, and 84% of adults with new-onset ESKD discontinued dialysis (3). The four- to five-fold increased risk of ESKD in Black people compared to European Americans is now attributed largely to variants in the Apolipoprotein L1 (APOL1) gene in the chromosome 22q12 locus, termed G1 and G2 (4,5). Recent studies show that the APOL1 gene variants are also associated with CKD in Africans (6). The APOL1 gene encodes circulating APOL1, which functions as a trypanolytic factor (4). The APOL1 renal risk variants evolved some 10,000 years ago in SSA, where trypanosomiasis was endemic, and have risen to a high prevalence in the population because they provided protection against Trypanosoma brucei rhodesiense (4). These variants are associated with an increased risk of hypertension-attributed ESKD, FSGS, HIV-associated nephropathy, and other nondiabetic kidney diseases (7). Given that ESKD afflicts an estimated 239,000 (per million population) individuals in SSA annually, there is a compelling need for identification of early modifiable risk markers of CKD, and a thorough investigation of the genetics of kidney disease causation and progression in African populations to promote the identification of new therapeutic targets and the formulation of effective treatment and prevention strategies.
Methods
The H3Africa Kidney Disease Research Network (H3A-KDRN) studies the genetic epidemiology and mechanisms of APOL1-associated nephropathy in individuals with CKD, using both general population controls and individuals with hypertension, diabetes, HIV infection, and sickle cell disease without CKD. The H3A-KDRN is a research consortium composed of 16 academic medical centers/teaching hospitals in five African countries (Cameroon, Ghana, Nigeria, South Africa, and Tanzania) (Figure 1). The H3A-KDRN Principal Investigators initially met over phone conferences, but over the last 4 years have met on web conferences every month. There was active collaboration with nine North American academic medical institutions and one in Israel. The H3A-KDRN is conducting three studies on Africans living in Africa: (1) a completed case control study of 4000 patients with CKD and 4000 controls that has been described (8); (2) an ongoing prospective cohort study of 4000 CKD participants that aims to identify the genetic determinants and characterize the phenotype for kidney disease progression in SSA; and (3) an ongoing study in two populations with CKD due to childhood-onset nephrotic syndrome and sickle cell anemia. The prospective cohort study is evaluating the independent contribution of risk variants in the APOL1 genes to the progression of clinically defined nephropathies among 2500 HIV-negative African Blacks and the effect gene-environment interactions between APOL1 variants and infections (hepatitis B, hepatitis C, and polyoma virus) on CKD. It will also evaluate the incidence, histopathologic spectrum, and the natural history of biopsy-confirmed glomerular diseases in 1500 HIV-negative African Blacks (Figure 2).
Figure 1.
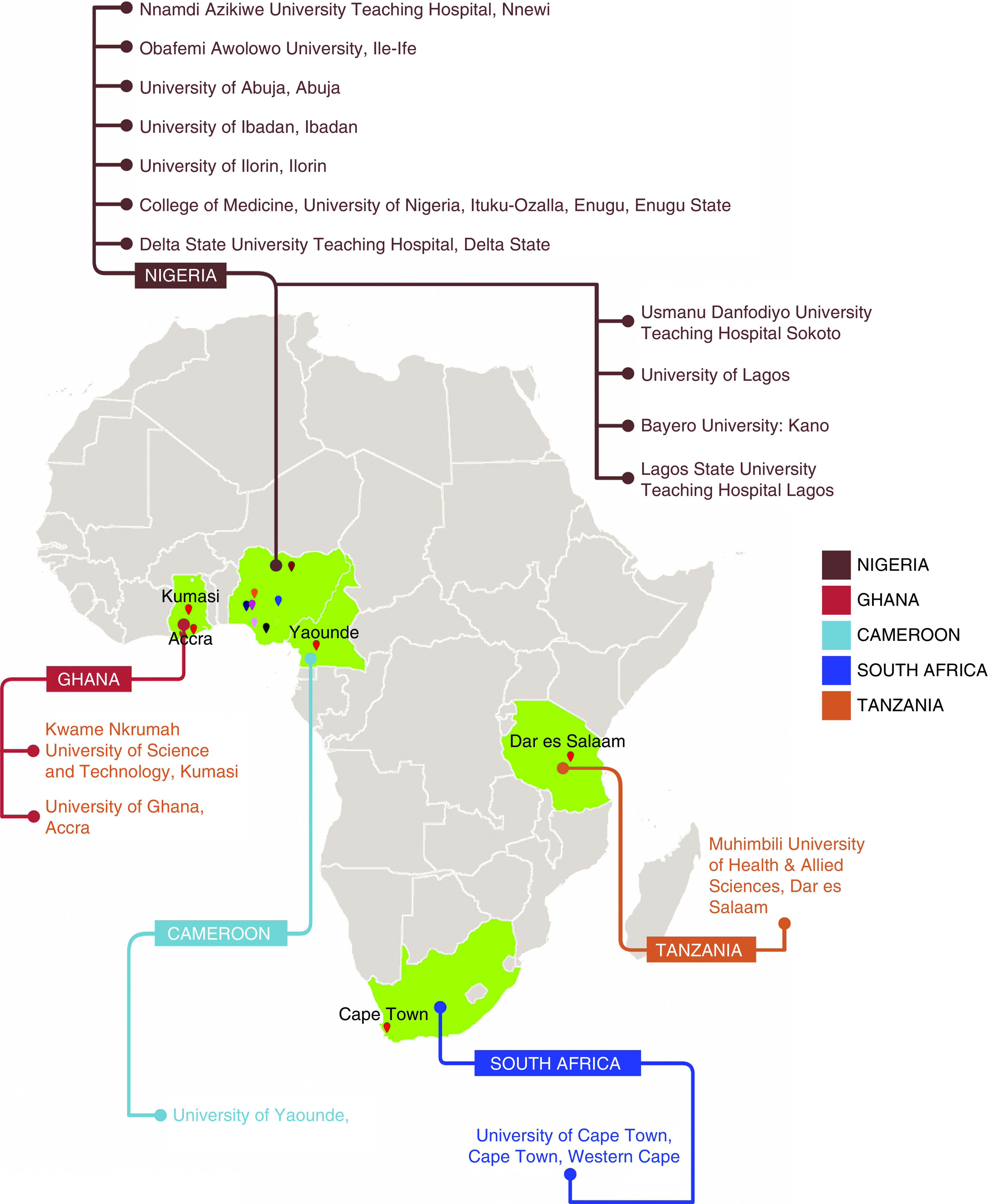
H3Africa Kidney Disease Research Network Participating Clinical Centers (Academic Medical Centers).
Figure 2.

H3Africa Kidney Disease Research Network summary image.
Ethics
The H3A-KDRN together with the H3Africa Consortium has developed a robust ethical framework for genomic research. A key element of this is that potential research participants are given clear information in language that was understood, using videos if necessary, and are given time to consider whether they wish to enroll in the study. The consenting process was three pronged with the giving of information followed by answering questions, and finally allowing potential research subjects time typically in excess of a week to decide whether to consent for the research. A careful explanation was given about broad consent that allows future use of samples and data as this would enable the best use of the research data and samples acquired. All investigators involved in recruitment are required to have mandatory training and certification in human subject protections. All studies received prior Institutional Review Board/Research Ethics Committee approval. A key part of the ethics approval process has been to engage with national research ethics committees to agree to policies specific to ethical issues attendant to genomic research (9).
Training
We are training clinical investigators and laboratory-based scientists and have developed infrastructure at two sites in Africa to conduct genomic research. Three Africa-based trainees have been trained at the University of Michigan as part of the H3A-KDRN program. A strategy of “mini-sabbaticals” has been implemented with Dr. Burke (University of Michigan) visiting H3A-KDRN laboratory groups in the University of Ghana and University of Ibadan, Nigeria. The visits have resulted in the establishment of research laboratories at both sites. Dr. Burke has developed a short course of didactic lectures and a guided laboratory procedure, targeted for students in Africa. The laboratory training is linked to an “open-resource” research-quality genetic typing system. Training included a 2-year training program in clinical research methods and biostatistics with the D43 Fogarty International Center Award (five investigators); Masters of Science in Human Genetics at the University of Michigan (two clinical investigators), MPhil in Molecular Biology at the University of Ghana (four scientists); and National Institutes of Health Biomedical/Biobehavioral Research Administrator Development Residency Training Program (two administrators). Three clinical investigators and four scientists are doing PhDs in clinical epidemiology and human genetics respectively. We also hosted over 11 sessions for training investigators and clinical research coordinators for more than 420 attendees.
Community Engagement
Our success in recruiting over 12,000 participants for our research studies over the last 9 years is largely due to active community engagement. Community engagement is an important component in increasing research participants’ understanding of genomic research and improving recruitment (10). The aim has been to increase genomics and clinical research understanding among our key stakeholders that can support the successful implementation of our research projects. We developed community engagement strategies that helped us to engage patient-support groups, faith-based institutions (including Christian and Moslem communities), the kidney patient community, and urban communities. We have interviewed H3A-KDRN research participants and their relatives for their views on return of individual genetic findings. The views of genetic research scientists have also been sought, as have the views of Ethical Review Committee members in deliberative workshops.
Biorepositories
As part of the H3Africa program, three biorepositories were established in Nigeria, Uganda, and South Africa for DNA and other biosamples. DNA from the H3A-KDRN has been deposited in the Institute of Human Virology of Nigeria Abuja biorepository (11). In addition, an H3Africa data and biospecimen access committee was set up to ensure responsible access by scientists from outside the consortium.
Results
Currently 8000 participants have been recruited into the case control study, and of these 3000 of these have had a GWAS using the new H3Africa Illumina chip with 2.5 million single nucleotide polymorphisms, enriched for African single nucleotide polymorphisms (https://www.h3abionet.org/resources/h3africa-chip).
Out of the 3500 participants enrolled so far into the prospective cohort study, 750 have undergone renal biopsies with light, immunofluorescence, electron microscopy, and transcriptional studies in the glomerular and interstitial compartments. A repository of DNA, plasma, serum, urine, and kidney tissue provides a rich resource for the study of kidney disease in Africa. Recruitment of participants with HIV CKD, childhood nephrotic syndrome, and sickle cell anemia is ongoing.
Discussion
The lamentable paucity of epidemiologic, genomics, and translational studies of kidney disease among Africans represents a major obstacle to preventative and cost-effective interventions by health care providers, governments, and individual citizens on the African continent. We are now poised to further enhance collaborative efforts in clinical, genomic, and translational research across the African continent and these studies will positively affect the understanding of health and disease in Africa. Our training programs for biomedical scientists, laboratory technicians, research coordinators, grant managers, and administrators within Africa have provided a platform for sustainable research into kidney diseases. The knowledge gained from these studies could have a major effect on kidney disease in Black patients by: (1) serving as basis for the development of screening programs for cardiovascular disease and APOL1-associated nephropathy in high-risk populations; (2) generating data for prognostic stratification of CKD and their risk factors; (3) informing public health policy and planning; and (4) identifying mechanistic pathways that could ultimately yield new therapeutic targets.
Ancillary Studies
The H3-KDRN actively encourages ancillary studies that will complement the aims and objectives of understanding the genomics and clinical aspects of kidney disease in Africa. In particular, the framework established would facilitate treatment and prevention studies.
Disclosures
All authors have nothing to disclose.
Funding
This work was supported by National Institutes of Health/National Human Genome Research Institute/National Institute of Diabetes and Digestive and Kidney Diseases H3Africa Kidney Disease Research Network grant 5U54 HG006939, H3Africa Kidney Disease Cohort Study grant 7U01 DK107131-05), and H3Africa Kidney Disease Research Network-Collaborative Centers grant 5U54 DK116913-10.
Acknowledgments
The content of this article reflects the personal experience and views of the author(s) and should not be considered medical advice or recommendations. The content does not reflect the views or opinions of the American Society of Nephrology (ASN) or Kidney360. Responsibility for the information and views expressed herein lies entirely with the author(s).
Author Contributions
A. Ojo was responsible for the conceptualization, funding acquisition, methodology, and writing review and editing. D. Adu was responsible for the conceptualization, methodology, project administration and writing the original draft.
Contributor Information
Collaborators: H3A-KDRN Investigators, Babatunde L. Salako, Ifeoma Ulasi, Yemi Raji, Olukemi Amodu, Manmak Mamven, Fatiu Arogundade, Samuel Ajayi, Timothy Olanrewaju, Rasheed Gbadegesin, Bamidele Tayo, Richard Cooper, Santosh Saraf, Victor Gordeuk, Rulan Parekh, Vincent Boima, Charlotte Osafo, Charles Odenigbo, Aliyu Abdu, Ogochukwu Okoye, Sampson Antwi, Adaobi Solarin, Toyin Amira, David Burke, Matthias Kretzler, Jeffrey Hodgin, Christopher Esezobor, Karl Skorecki, Ademola Adebowale, Adebowale Adeyemo, Cheryl Winkler, Muhammad Makusidi, Gloria Ashuntantang, Mignon McCulloch, Peter Nourse, Michael Matekole, Anita Ghansah, Alexander Nyarko, Titilayo Ilori, Francis Furia, and Jacob Plange-Rhule
References
- 1.Stanifer JW, Jing B, Tolan S, Helmke N, Mukerjee R, Naicker S, Patel U: The epidemiology of chronic kidney disease in sub-Saharan Africa: A systematic review and meta-analysis. Lancet Glob Health 2: e174–e181, 2014. 10.1016/S2214-109X(14)70002-6 [DOI] [PubMed] [Google Scholar]
- 2.Anand S, Bitton A, Gaziano T: The gap between estimated incidence of end-stage renal disease and use of therapy. PLoS One 8: e72860, 2013. 10.1371/journal.pone.0072860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ashuntantang G, Osafo C, Olowu WA, Arogundade F, Niang A, Porter J, Naicker S, Luyckx VA: Outcomes in adults and children with end-stage kidney disease requiring dialysis in sub-Saharan Africa: A systematic review. Lancet Glob Health 5: e408–e417, 2017. 10.1016/S2214-109X(17)30057-8 [DOI] [PubMed] [Google Scholar]
- 4.Genovese G, Friedman DJ, Ross MD, Lecordier L, Uzureau P, Freedman BI, Bowden DW, Langefeld CD, Oleksyk TK, Uscinski Knob AL, Bernhardy AJ, Hicks PJ, Nelson GW, Vanhollebeke B, Winkler CA, Kopp JB, Pays E, Pollak MR: Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science 329: 841–845, 2010. 10.1126/science.1193032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tzur S, Rosset S, Shemer R, Yudkovsky G, Selig S, Tarekegn A, Bekele E, Bradman N, Wasser WG, Behar DM, Skorecki K: Missense mutations in the APOL1 gene are highly associated with end stage kidney disease risk previously attributed to the MYH9 gene. Hum Genet 128: 345–350, 2010. 10.1007/s00439-010-0861-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ulasi II, Tzur S, Wasser WG, Shemer R, Kruzel E, Feigin E, Ijoma CK, Onodugo OD, Okoye JU, Arodiwe EB, Ifebunandu NA, Chukwuka CJ, Onyedum CC, Ijoma UN, Nna E, Onuigbo M, Rosset S, Skorecki K: High population frequencies of APOL1 risk variants are associated with increased prevalence of non-diabetic chronic kidney disease in the Igbo people from south-eastern Nigeria. Nephron Clin Pract 123: 123–128, 2013. 10.1159/000353223 [DOI] [PubMed] [Google Scholar]
- 7.Freedman BI, Limou S, Ma L, Kopp JB: APOL1-Associated Nephropathy: A key contributor to racial disparities in CKD. Am J Kidney Dis 72[Suppl 1]: S8–S16, 2018. 10.1053/j.ajkd.2018.06.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Osafo C, Raji YR, Burke D, Tayo BO, Tiffin N, Moxey-Mims MM, Rasooly RS, Kimmel PL, Ojo A, Adu D, Parekh RS; H3Africa Kidney Disease Research Network Investigators as members of The H3Africa Consortium: Human Heredity and Health (H3) in Africa kidney disease research network: A focus on methods in sub-Saharan Africa. Clin J Am Soc Nephrol 10: 2279–2287, 2015. 10.2215/CJN.11951214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.de Vries J, Abayomi A, Littler K, Madden E, McCurdy S, Ouwe Missi Oukem-Boyer O, Seeley J, Staunton C, Tangwa G, Tindana P, Troyer J; The H3Africa Working Group on Ethics: Addressing ethical issues in H3Africa research – the views of research ethics committee members. HUGO J 9: 1, 2015. 10.1186/s11568-015-0006-6 [DOI] [Google Scholar]
- 10.Tindana P, Campbell M, Marshall P, Littler K, Vincent R, Seeley J, de Vries J, Kamuya D; H3Africa Community Engagement Working Group: Developing the science and methods of community engagement for genomic research and biobanking in Africa. Glob Health Epidemiol Genom 2: e13, 2017. 10.1017/gheg.2017.9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Croxton T, Agala N, Jonathan E, Balogun O, Ozumba PJ, Onyemata E, Lawal S, Mamven M, Ajayi S, Melikam SE, Owolabi M, Ovbiagele B, Adu D, Ojo A, Beiswanger CM, Abimiku A: H3Africa partnerships to empower clinical research sites to generate high-quality biological samples. Afr J Lab Med 9: 935, 2020. 10.4102/ajlm.v9i1.935 [DOI] [PMC free article] [PubMed] [Google Scholar]


