Abstract
The phase III MAX clinical trial randomised patients with metastatic colorectal cancer (mCRC) to receive first-line capecitabine chemotherapy alone or in combination with the anti-VEGF-A antibody bevacizumab (± mitomycin C). We utilised this cohort to examine whether single nucleotide polymorphisms (SNPs) in VEGF-A, VEGFR1, and VEGFR2 are predictive of efficacy outcomes with bevacizumab or the development of hypertension. Genomic DNA extracted from archival FFPE tissue for 325 patients (69% of the MAX trial population) was used to genotype 16 candidate SNPs in VEGF-A, VEGFR1, and VEGFR2, which were analysed for associations with efficacy outcomes and hypertension. The VEGF-A rs25648 ‘CC’ genotype was prognostic for improved PFS (HR 0.65, 95% CI 0.49 to 0.85; P = 0.002) and OS (HR 0.70, 95% CI 0.52 to 0.94; P = 0.019). The VEGF-A rs699947 ‘AA’ genotype was prognostic for shorter PFS (HR 1.32, 95% CI 1.002 to 1.74; P = 0.048). None of the analysed SNPs were predictive of bevacizumab efficacy outcomes. VEGFR2 rs11133360 ‘TT’ was associated with a lower risk of grade ≥ 3 hypertension (P = 0.028). SNPs in VEGF-A, VEGFR1 and VEGFR2 did not predict bevacizumab benefit. However, VEGF-A rs25648 and rs699947 were identified as novel prognostic biomarkers and VEGFR2 rs11133360 was associated with less grade ≥ 3 hypertension.
Subject terms: Oncology, Genetic markers, Gastrointestinal cancer, Colorectal cancer, Tumour biomarkers, Predictive markers, Prognostic markers, Translational research, Targeted therapies, Cancer, Tumour angiogenesis
Introduction
Bevacizumab, a recombinant humanised anti-vascular endothelial growth factor-A (VEGF-A) monoclonal antibody, is an approved anti-angiogenic drug which is used in combination with chemotherapy for the treatment of metastatic colorectal cancer (mCRC). Although it is an active and effective drug in this setting, the survival benefit in unselected populations is modest1–3 and it is associated with potential side effects. There is a need to define predictive biomarkers that can identify the patients who are most likely to derive benefit.
Bevacizumab exerts its effects by targeting host-mediated angiogenesis4. Its target, VEGF-A, is the most well-characterised pro-angiogenic ligand in the VEGF family5. Effects of VEGF-A on endothelial cell differentiation, proliferation, migration and vasculogenesis are primarily mediated by the receptor tyrosine kinase VEGFR2 (KDR)6, however VEGF-A also binds to VEGFR1 (FLT-1) with high affinity7 and induces weak kinase activity of this receptor8. Therefore, common germline genetic variability in VEGF-A, VEGFR1, and VEGFR2 due to functional single nucleotide polymorphisms (SNPs) may explain heterogeneity in efficacy and toxicity amongst patients treated with bevacizumab.
Previous biomarker studies have identified a range of different single nucleotide polymorphisms (SNPs) in VEGF-A, VEGFR1 and VEGFR2 that were associated with efficacy outcomes (including objective response rate (ORR), progression-free survival (PFS) and overall survival (OS)) in studies of bevacizumab in mCRC. These include rs30250399, rs20109639–11, rs157036011, rs69994710, rs83306110,12, rs2564813 (VEGF-A); rs958203614, rs951307012 (VEGFR1); rs230594815, and rs1250575816 (VEGFR2). Other SNPs in VEGF family genes have also been associated with efficacy outcomes in studies of bevacizumab in non-CRC tumours, including rs69994617,18 (VEGF-A); rs955431619,20, rs951307019, rs955432019, rs799341819 (VEGFR1); rs207155921 and rs1113336018,22 (VEGFR2). However, whilst some of these associations were independently verified in one or more additional studies, other studies showed no association or conflicting results. Notably, only one of the mCRC studies (ITACa)9 was a Phase III randomised controlled trial with a non-bevacizumab control arm, enabling distinction between prognostic and predictive effects. Recently, SNPs in multiple non-VEGF family genes have been found to be associated with efficacy outcomes in studies of bevacizumab in mCRC, with the results replicated in validation cohorts. The AA genotype for the rs8602 SNP in MKNK1, which is associated with upregulation of angiogenic factors and stimulation of angiogenesis, was associated with a shorter PFS compared to those with any C allele in patients with KRAS exon 2 wild type (wt) mCRC treated with FOLFIRI/Bevacizumab23. The AA genotype for the rs4588 SNP in GC, a gene encoding a vitamin D-binding protein, was associated with worse OS compared to those with any C allele in patients with mCRC treated with the same combination24. The G allele for the rs555008 SNP in RSPO2, one of the genes encoding R-spondin proteins which stimulate Wnt signalling, was associated with longer OS compared to the TT genotype in patients with RAS and KRAS wt mCRC25. P values for SNP by treatment interaction were not reported in these studies.
Hypertension is a common side effect which is observed in approximately a quarter of bevacizumab-treated patients26. Potential mechanisms include vasoconstriction due to decreased nitrous oxide (NO) production27, increased vascular tone secondary to inhibition of VEGF-mediated vasodilation28, microvascular rarefaction29, and renal dysfunction as a direct consequence of VEGF inhibition30 or indirectly through associated NO downregulation31.
Previous studies conducted in a range of tumour types have found associations between SNPs in VEGF-A (rs201096332,33, rs83306132,34, rs69994734, rs302503934,35) and VEGFR2 (rs230594936, rs187037737) and bevacizumab-related hypertension. However, no association was replicated consistently across multiple studies and findings in several studies were discordant. SNPs in non-VEGF family genes were also recently reported to be associated with hypertension in studies of bevacizumab in mCRC. rs9381299 and rs834576 in the genomic region between SLC29A1 and HSP90AB1 were associated with early grade ≥ 3 hypertension, potentially through effects on adenosine signalling38. rs1129660 in FIP200, an autophagy-related gene, was associated with grade 2–3 hypertension in patients treated with FOLFIRI/bevacizumab39.
The Australian Gastro-intestinal Trials Group (AGITG) MAX clinical trial (NCT00294359) was an investigator-initiated phase III randomised controlled trial of first-line treatment for mCRC which met its primary end-point of improved PFS with the addition of bevacizumab to capecitabine-based chemotherapy3. The inclusion of a non-bevacizumab control arm provides an ideal dataset for retrospective predictive biomarker analysis. In this exploratory study, we examined the association of SNPs in VEGF-A, VEGFR1, and VEGFR2 with prognosis, benefit from bevacizumab treatment and hypertension in patients receiving capecitabine alone and in combination with bevacizumab (± mitomycin C) in the MAX clinical trial.
Material and methods
Patients and study design
The study design and eligibility criteria for the MAX clinical trial have been reported previously3. The primary objective of the trial was to determine the effect on PFS of adding bevacizumab, with or without mitomycin, to first-line capecitabine monotherapy in patients with unresectable mCRC. Overall survival (OS) was a secondary end-point. Patients were enrolled from July 2005 to June 2007 and randomly assigned 1:1:1 to capecitabine (C), capecitabine plus bevacizumab (CB), or capecitabine, bevacizumab and mitomycin (CBM). Participants were evaluated for tumour response every 6 weeks on study. In the absence of significant toxicity, treatment was continued until confirmed disease progression.
Ethics approval and consent to participate
Ethics approval for this study was obtained centrally from the Austin Health Human Research Ethics Committee. All participants in the current study provided written informed consent to participate in the main MAX clinical trial and additional informed consent for their tissue samples to be used in biomarker studies. The study was performed in accordance with the Declaration of Helsinki.
Archival tissue collection and processing
Archived formalin-fixed paraffin-embedded (FFPE) tissue specimens collected at the time of diagnosis and associated histopathology reports were retrieved from the pathology departments where the diagnosis was made, and stored centrally. Personnel who performed histopathology reporting and scientific assays for biomarker analysis were blinded to patient identification and clinical outcomes.
Where available, a core of normal, colorectal adenoma, or colorectal tumour tissue (listed in order of preference) was extracted using a Beecher Mark II Tissue Arrayer (Beecher Instruments, Sun Prairie, WI, USA). Haematoxylin and eosin stained slides were used to guide the coring procedure.
For the first fifty cases (ordered consecutively by patient ID) where both colorectal tumour tissue and adjacent normal colorectal epithelial tissue were available, matching cores of tumour and normal tissue were extracted.
SNP genotyping of tissue samples
Genomic DNA was extracted from the tissue cores using the QIAamp DNA FFPE Tissue Kit (Qiagen, Hilden, Germany), as per the manufacturer’s instructions. DNA concentration was determined using the NanoDrop 2000 Spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA).
All of the tested candidate SNPs in VEGF-A, VEGFR1, and VEGFR2 were previously reported to have associations with clinical outcomes and/or to have an effect on corresponding VEGF ligand or receptor expression or function10,17,40–47. SNP genotyping was performed using Taqman SNP Genotyping Assays (Applied Biosystems, Waltham, MA, USA). Context sequences and primer sequences for the Taqman SNP Genotyping Assays used are provided in Supplementary Table S1. SNPs were required to have a minor allele frequency (MAF) of 10% or more to provide reassurance that they would have sufficient impact as a potential biomarker at the population level.
For each SNP assay, 5 ng of genomic DNA from each trial participant was diluted in 2.25 μL nuclease-free water, and mixed with 2.75 μL of reaction mix consisting of 2X Taqman GTXpress Master Mix (Applied Biosystems) and 20X TaqMan Genotyping Assay mix (Applied Biosystems). Samples were loaded into MicroAmp Optical 384-Well Reaction Plates with Barcode (Applied Biosystems), with each plate including at least one non-template control of nuclease-free water. Reaction plates were vortexed, centrifuged and PCR reactions performed on the ViiA 7 Real-Time PCR System (Applied Biosystems) with the following conditions: (1) a pre-read stage at 60 °C for 30 s, (2) a hold stage with an initial DNA polymerase activation step (95 °C for 20 s), (3) a PCR stage with 40 to 60 amplification cycles using a two-step cycling protocol of denaturing at 95 °C for 1 s and annealing and extension at 60 °C for 20 s, and (4) a post-read stage at 60 °C for 30 s.
Experiments were analysed using the Applied Biosystems ViiA 7 Real-Time PCR System software (Applied Biosystems). The genotyping optimisation tool was used to reveal traces to determine the ideal cycle for genotyping. Genotyping experiments were performed in duplicate as a minimum.
Each SNP was tested for departure from Hardy Weinberg Equilibrium (HWE) using the exact test in Haploview 4.2 (Broad Institute, Cambridge, MA, USA), the null hypothesis being that the genotype is in HWE in the population. A Bonferroni correction was applied to a significance level of P > 0.05, giving a significance level of P > 0.0024. SNPs with significant departure from HWE (P < 0.0024) were excluded from further analyses. The MAF for each SNP was calculated using Haploview 4.2.
VEGF family gene and protein expression analysis
To examine whether specific VEGF-A SNP genotypes were associated with VEGF-A expression, we interrogated corresponding gene expression and pro-angiogenic protein expression data we had previously generated for this patient cohort48,49.
In brief, for gene expression analysis H&E stained sections were reviewed by a pathologist and macro-dissected to obtain tumour-rich tissue. RNA was extracted and converted into cDNA and gene expression profiling performed using the Almac Xcel Array (Almac, Craigavon, UK), utilising Affymetrix GeneChip technology optimised for use with FFPE tissue.
For analysis of protein expression levels, protein lysates were prepared from the FFPE tumour sections and quantified using the EZQ Protein Quantitation Kit (Life Technologies, Carlsbad, CA, USA). Protein lysates were assayed for expression of multiple proteins including VEGF-A, using a custom multiplex Bio-Plex array on the Bio-Plex 200 System instrument (Bio-Rad, Hercules, CA, USA).
Statistical methods
Statistical analyses were performed using the statistical package ACCoRD (Analysis of Censored and Correlated Data) version 8.58 (Boffin Software, NSW, Australia) and Stata 13.1 (StataCorp, College Station, TX, USA). All reported P values are two-sided.
For the fifty consecutive cases where SNP genotyping was performed on matched tumour tissue and adjacent normal tissue, statistical analysis of concordance in the frequencies of each genotype between the tumour and normal tissue groups was calculated using Kendall’s τ. Concordance was analysed in cases where the full genotypes for both tumour and normal tissue had been determined.
Statistical analysis for PFS and OS was performed using the proportional hazards model. PFS was defined as time from randomisation until documented evidence of disease progression according to Response Evaluation Criteria in Solid Tumours (RECIST) version 1.0 or death as a result of any cause. OS was defined as the time from randomisation until death from any cause.
For each SNP, we first compared survival outcomes in participants with and without a homozygous major allele genotype. Survival outcomes in participants with and without a homozygous minor allele genotype were subsequently compared.
To determine whether SNP genotype was a predictive biomarker for resistance to bevacizumab, SNPs which showed a univariate association with survival outcomes were included in a model which included the allocated treatment covariate (‘CB and CBM’ versus ‘C’) and a ‘SNP by treatment’ interaction. These SNPs were also tested in a multivariate model which included baseline variables which were found to be associated with survival outcomes in the main MAX study (treatment received, Eastern Cooperative Oncology Group (ECOG) performance status, neutrophils, bilirubin and alkaline phosphatase levels, prior radiotherapy, whether the primary tumour was resected and the number of metastatic sites)3. Variables significantly associated with the survival outcomes in the multivariate model were retained in the final model.
In additional analyses, a multivariate model which included KRAS and BRAF mutation status along with the baseline prognostic variables was tested. Mutation status was only retained as a variable in the final model if it was significantly associated with survival outcomes.
A logistic regression model with SNP genotype status, treatment group covariate, and a ‘SNP-by-treatment’ interaction term was used to determine whether SNP genotype was predictive of an effect of bevacizumab on objective response rate (ORR) (defined as a best response of complete response (CR) or partial response (PR)).
For SNPs which were found to be associated with survival outcomes, the association between SNP genotype and corresponding (1) log2 transformed gene expression data from the Almac Xcel microarray and (2) protein expression data from Bioplex suspension array analyses were analysed using two-sided unpaired t-tests.
Univariate and multivariate associations between the SNPs and on-study grade ≥ 3 hypertension according to the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE) version 3.0 were examined using logistic regression models. Models with SNP genotype alone, and with both the SNP genotype and allocated treatment covariates (‘CB and CBM’ versus ‘C’) were used to explore potential associations with hypertension, and both unadjusted and adjusted odds ratios (ORs) were reported. If the ORs were not estimable due to zero events in one cell of the 2 × 2 contingency table, the P values for the Conditional Binomial Exact Test (CBET) for independence between SNP and hypertension were reported50.
For each SNP, only participants with the full genotype identified were included in analyses of associations between SNP genotype and efficacy and hypertension outcomes.
Due to the exploratory nature of this analysis, no formal adjustment for multiple comparisons was made.
Results
Patient characteristics
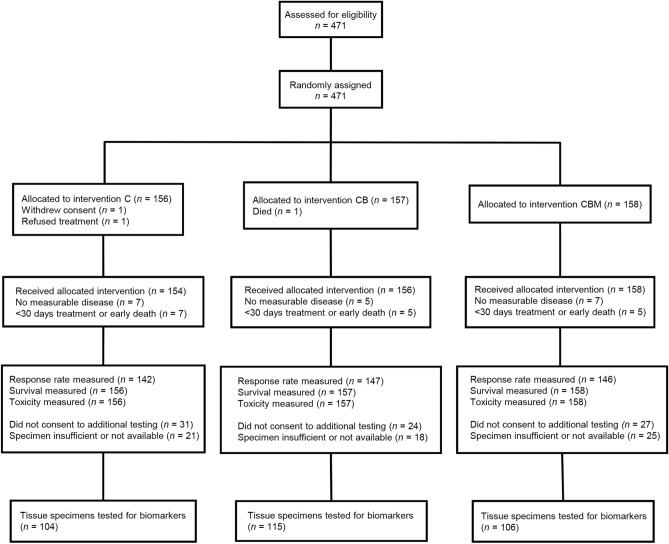
389 of the 471 randomised participants in the MAX clinical trial consented to participation in biological sub-studies. Of these, archived FFPE specimens were available from 325 participants (69% of the total study population) for analysis of VEGF family SNPs (Fig. 1).
Figure 1.
CONSORT flow diagram. Flow diagram of progress through the MAX clinical trial and VEGF SNP biomarker sub-study.
For the remaining participants, tissue either could not be retrieved or was insufficient for extracting cores. The baseline characteristics and survival outcomes in the total study population and the SNP biomarker study population were comparable (Supplementary Table S2). The median follow-up time for the included participants was 30 months (range, 0.4 to 42.4 months).
SNP genotyping
Of the available archived FFPE specimens from 325 participants, normal tissue was available for 164 participants (51%). Colorectal adenoma tissue was available for seven participants (2%). Colorectal carcinoma tissue was available for the remaining 154 participants (47%), of which 90% (n = 137) were from primary tumour (Supplementary Fig. S1).
Twenty-one SNPs were genotyped—7 in VEGF-A (rs699946, rs699947, rs833061, rs1570360, rs2010963, rs25648, rs3025039), 5 in VEGFR1 (rs9513070, rs9554316, rs7993418, rs9582036, rs9554320) and 9 in VEGFR2 (rs12505758, rs7655964, rs1870377, rs2305948, rs2305949, rs11133360, rs7667298, rs2071559, rs1551641) (Supplementary Table S1). Of these, 16 SNPs were found to be in HWE using Haploview and were used in subsequent analyses (Supplementary Table S3). The calculated MAF for each of these analysed SNPs was 10% or greater (Supplementary Table S4).
The full genotype was successfully identified in ≥ 84% of cases for each of the SNPs genotyped. The proportion of cases where the partial genotype was known (presence of the major allele or minor allele known, but only one allele determined) was not more than 7% for each SNP. The proportion of cases with an undetermined genotype (due to discordant genotyping calls between replicates, failure to amplify, or insufficient genomic DNA template) was not more than 9% for each SNP (Supplementary Table S5).
In the fifty cases of matched normal tissue and adjacent tumour tissue which we examined, concordance of genotype calls was strong (Kendall’s τ > 0.70; 15/16 SNPs) or moderate (Kendall’s τ > 0.40; 1/16 SNPs—rs12505758) for all SNPs analysed (Supplementary Table S6).
Progression-free survival
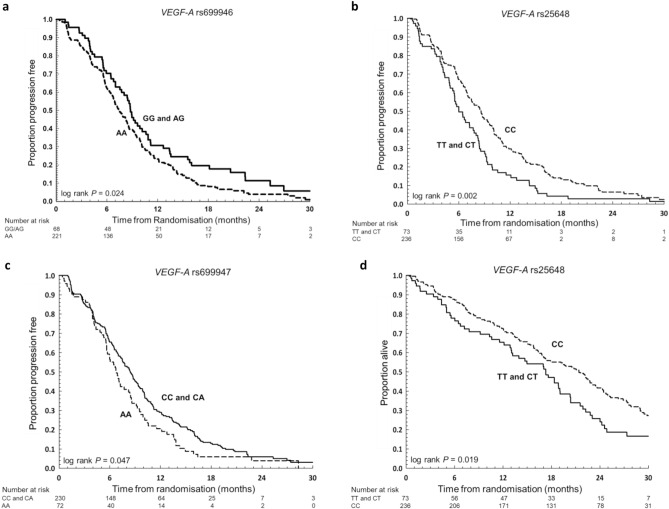
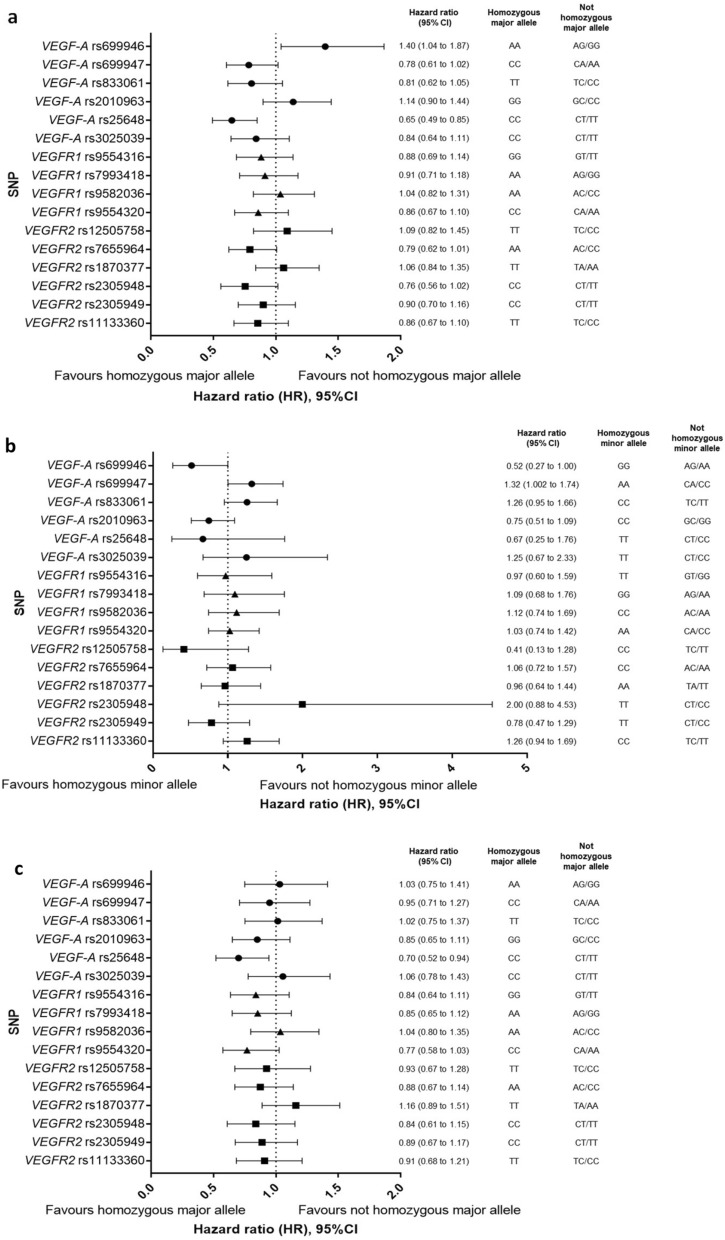
There was a significantly lower PFS in participants with the VEGF-A rs699946 ‘AA’ genotype (homozygous major allele) compared to ‘GG and AG’ genotypes (median 7.5 months versus 8.8 months, log rank P = 0.024) (Fig. 2A). The hazard ratio (HR) for PFS in VEGF-A rs699946 ‘AA’ versus ‘GG and AG’ was 1.40 (95% CI 1.04 to 1.87, P = 0.025) (Fig. 3A, Table 1, Supplementary Table S7). A model including VEGF-A rs699946 genotype and allocated treatment (‘CB/CBM’ versus ‘C’) together with an interaction term (SNPxTreatment) did not show any statistically significant interaction effect between VEGF-A rs699946 genotype and bevacizumab treatment (interaction P value = 0.93).
Figure 2.
Survival outcomes by SNP genotypes. Progression free survival for patients with (a) VEGF-A rs 699946 ‘AA’ compared to ‘GG and AG’ genotypes, (b) VEGF-A rs25648 ‘CC’ compared to ‘TT and CT’ genotypes and (c) VEGF-A rs699947 ‘AA’ compared to ‘CC and AC’ genotypes. (d) Overall survival for patients with VEGF-A rs25648 ‘CC’ compared to ‘TT and CT’ genotypes. The P-value shown is for the log rank test of equality across the strata for the genotypes being compared.
Figure 3.
Forest plots of survival outcomes by SNP genotype. Hazard Ratios with 95% confidence intervals for the comparisons of (a) Progression free survival in patients with homozygous major allele genotype versus without a homozygous major allele genotype, (b) Progression free survival in patients with homozygous minor allele genotype versus without a homozygous minor allele genotype, and (c) Overall survival in patients with a homozygous major allele genotype versus without a homozygous major allele genotype.
Table 1.
Prognostic associations of VEGF-A family SNP genotypes for progression free survival and overall survival.
| Progression free survival (PFS) | |||||||
|---|---|---|---|---|---|---|---|
| SNP | Univariate analysis | Variables in multivariable model | Multivariate analysis | ||||
| HR (95% CI) | P value | HR (95% CI) | P value | ||||
| VEGF-A rs699946 | Homozygous major allele ‘AA’ vs ‘GG and AG’ | 1.40 (1.04 to 1.87) | P = 0.025 | ||||
| VEGF-A rs25648 | Homozygous major allele ‘CC’ vs ‘TT and CT’ | 0.65 (0.49 to 0.85) | P = 0.002 | VEGF-A rs25648 | Homozygous major allele ‘CC’ vs ‘TT and CT’ | 0.62 (0.47 to 0.82) | P = 0.001 |
| Treatment | ‘CB + CBM’ vs ‘C’ | 0.59 (0.46 to 0.77) | P < 0.001 | ||||
| ECOG PS | ≥ 1 vs 0 | 1.63 (1.34 to 1.99) | P < 0.001 | ||||
| Neutrophils ≥ 8 × 109/L | Yes vs no | 1.60 (1.14 to 2.26) | P = 0.007 | ||||
| Serum bilirubin ≥ 14 μmol/L | Yes vs no | 1.53 (1.13 to 2.06) | P = 0.006 | ||||
| VEGF-A rs699947 | Homozygous minor allele ‘AA’ vs ‘CC and CA’ | 1.32 (1.002 to 1.74) | P = 0.048 | VEGF-A rs699947 | Homozygous minor allele ‘AA’ vs ‘CC and CA’ | 1.50 (1.13 to 1.99) | P = 0.005 |
| Treatment | ‘CB + CBM’ vs ‘C’ | 0.62 (0.48 to 0.80) | P < 0.001 | ||||
| ECOG PS | ≥ 1 vs 0 | 1.69 (1.39 to 2.07) | P < 0.001 | ||||
| Neutrophils ≥ 8 × 109/L | Yes vs no | 1.60 (1.14 to 2.25) | P = 0.007 | ||||
| Serum bilirubin ≥ 14 μmol/L | Yes vs no | 1.53 (1.12—2.08) | P = 0.007 | ||||
| Overall survival (OS) | |||||||
|---|---|---|---|---|---|---|---|
| SNP | Univariate analysis | Variables in multivariable model | Multivariate analysis | ||||
| HR (95% CI) | P value | HR (95% CI) | P value | ||||
| VEGF-A rs25648 | Homozygous major allele ‘CC’ vs ‘TT and CT’ | 0.70 (0.52 to 0.94) | P = 0.019 | VEGF-A rs25648 | |||
| VEGF-A rs25648 | Homozygous major allele ‘CC’ vs ‘TT and CT’ | 0.68 (0.50 to 0.92) | P = 0.011 | ||||
| ECOG PS | ≥ 1 vs 0 | 2.08 (1.68 to 2.58) | P < 0.001 | ||||
| Neutrophils ≥ 8 × 109/L | Yes vs no | 1.85 (1.27 to 2.70) | P = 0.001 | ||||
| Serum ALP ≥ 140 U/L | Yes vs no | 1.002 (1.000 to 1.003) | P = 0.028 | ||||
| VEGF-A rs25648, ALP excluded from model | |||||||
| VEGF-A rs25648 | Homozygous major allele ‘CC’ vs ‘TT and CT’ | 0.67 (0.49 to 0.90) | P = 0.008 | ||||
| ECOG PS | ≥ 1 vs 0 | 2.07 (1.66 to 2.56) | P < 0.001 | ||||
| Neutrophils ≥ 8 × 109/L | Yes vs no | 1.84 (1.27 to 2.68) | P = 0.001 | ||||
| VEGF-A rs25648, BRAF included in model | |||||||
| VEGF-A rs25648 | Homozygous major allele ‘CC’ vs ‘TT and CT’ | 0.70 (0.52 to 0.96) | P = 0.024 | ||||
| ECOG PS | ≥ 1 vs 0 | 2.10 (1.68 to 2.61) | P < 0.001 | ||||
| Neutrophils ≥ 8 × 109/L | Yes vs no | 1.85 (1.26 to 2.72) | P = 0.002 | ||||
| BRAF mutation status | Wild type vs mutant | 0.49 (0.32 to 0.75) | P = 0.001 | ||||
VEGF-A = vascular endothelial growth factor-A; SNP = single nucleotide polymorphism; HR = hazard ratio; CI = confidence interval; CB = capecitabine and bevacizumab; CBM = capecitabine, bevacizumab and mitomycin; C = capecitabine; ECOG PS = Eastern Cooperative Oncology Group performance status; ALP: alkaline phosphatase.
Participants with the VEGF-A rs25648 ‘CC’ genotype (homozygous major allele) had a significantly longer PFS compared to the ‘TT and CT’ genotypes (median 8.6 months versus 6.0 months, log rank P = 0.002) (Fig. 2B). The HR for PFS in VEGF-A rs25648 ‘CC’ versus ‘TT and CT’ was 0.65 (95% CI 0.49 to 0.85, P = 0.002) (Fig. 3A, Table 1, Supplementary Table S7). A similar model to the analysis of VEGF-A rs699946 was fitted to the VEGF-A rs25648 genotype, and no statistically significant interaction was observed between this SNP and the addition of bevacizumab (interaction P value = 0.78).
Participants with the VEGF-A rs699947 ‘AA’ genotype (homozygous minor allele) had a significantly inferior PFS compared to the ‘CC and CA’ genotypes (median 6.8 months versus 8.4 months, log rank P = 0.047) (Fig. 2C). The HR for PFS in VEGF-A rs699947 ‘AA’ versus ‘CC and CA’ was 1.32 (95% CI 1.002 to 1.74, P = 0.048) (Fig. 3B, Table 1, Supplementary Table S7). As for VEGF-A rs699946 and VEGF-A rs25648, a model was fitted to examine interaction effects for VEGF-A rs699947 genotype and the addition of bevacizumab. No statistically significant interaction effect was observed (interaction P value = 0.70).
In a multivariable model adjusting for baseline prognostic factors, of the SNPs analysed, only VEGF-A rs25648 and VEGF-A rs699947 remained significant independent prognostic factors for PFS. The adjusted HR for VEGF-A rs25648 ‘CC’ versus ‘TT and CT’ was 0.62 (95% CI 0.47 to 0.82, P = 0.001) and the adjusted HR for VEGF-A rs699947 ‘AA’ versus ‘CC and CA’ was 1.50 (95% CI 1.13 to 1.99, P = 0.005) (Table 1). In an additional multivariable model which also included KRAS and BRAF mutation status, neither of these covariates was significantly associated with the PFS outcome.
Overall survival
There was a significantly longer OS in participants with the VEGF-A rs25648 ‘CC’ genotype (homozygous major allele) compared to the ‘TT and CT’ genotypes (median 21.4 months versus 17.3 months, log rank P = 0.019) (Fig. 2D). The HR for OS in VEGF-A rs25648 ‘CC’ versus ‘TT and CT’ was 0.70 (95% CI 0.52 to 0.94, P = 0.019) (Fig. 3C, Table 1, Supplementary Table S8). A model including VEGF-A rs25648 genotype and allocated treatment (‘CB/CBM’ versus ‘C’) together with an interaction term (SNPxTreatment) did not show any statistically significant interaction effect between VEGF-A rs25648 genotype and bevacizumab treatment (interaction P value = 0.54).
The VEGF-A rs25648 ‘CC’ genotype remained a significant prognostic factor for OS in a multivariable model adjusting for baseline prognostic factors. The adjusted HR for VEGF-A rs25648 ‘CC’ versus ‘TT and CT’ was 0.68 (95% CI 0.50 to 0.92, P = 0.011). In the multivariable model, the HR for serum alkaline phosphatase (ALP) was 1.002 (95% CI 1.000 to 1.003), which may not be considered clinically significant. When serum ALP was excluded from the multivariable model, the adjusted HR for OS was 0.67 (95% CI 0.49 to 0.90, P = 0.008) (Table 1).
In a further multivariable model which included KRAS and BRAF mutation status, BRAF but not KRAS mutation status was significantly associated with OS. When BRAF mutation status was additionally included in the final multivariable model, the adjusted HR for VEGF-A rs25648 ‘CC’ versus ‘TT and CT’ did not change appreciably (HR 0.70, 95% CI 0.52 to 0.96, P = 0.024) (Table 1).
Objective response
Participants with VEGF-A rs699946 ‘GG and AG’, rs25648 ‘CC’ or rs699947 ‘CC and CA’ genotypes did not have a greater likelihood of achieving an objective response with the addition of bevacizumab, compared to the alternative genotypes (P = 0.63, P = 0.77, and P = 0.70 for the interaction between SNP genotype and the assigned treatment, respectively) (Table 2). None of the other analysed SNPs was predictive of improved objective response with the addition of bevacizumab (Supplementary Table S9).
Table 2.
Objective response rate by VEGF-A rs699946, rs25648 and rs699947 genotypes.
| VEGF-A 699946 | |||
|---|---|---|---|
| Treatment | AA genotype n (%) | GG and AG genotypes n (%) | P valuea |
| C | 26 (79) | 7 (21) | 0.63 |
| CB | 29 (78) | 8 (22) | |
| CBM | 31 (74) | 11 (26) | |
| VEGF-A 25648 | |||
|---|---|---|---|
| Treatment | CC genotype n (%) | TT and CT genotypes n (%) | P valuea |
| C | 29 (83) | 6 (17) | 0.77 |
| CB | 29 (73) | 11 (27) | |
| CBM | 40 (87) | 6 (13) | |
| VEGF-A 699947 | |||
|---|---|---|---|
| Treatment | AA genotype n (%) | CC and CA genotypes n (%) | P valuea |
| C | 9 (26) | 26 (74) | 0.70 |
| CB | 9 (24) | 29 (76) | |
| CBM | 8 (18) | 37 (82) | |
VEGF-A = vascular endothelial growth factor-A; C = capecitabine; CB = capecitabine and bevacizumab; CBM = capecitabine, bevacizumab and mitomycin.
aP value for interaction between SNP genotype biomarker status and the allocated treatment (‘CB + CBM’ vs ‘C’).
SNP genotype and VEGF-A expression
Data from gene expression profiling was available for 239 of 309 (77%) participants who had a full genotype known for VEGF-A rs25648, and 234 of 302 (77%) participants who had a full genotype known for VEGF-A rs699947.
However, no difference in VEGF-A gene expression level was observed between the better prognosis VEGF-A rs25648 ‘CC’ genotype group and the ‘TT and CT’ genotypes (mean 0.00091 versus − 0.020, P = 0.77). Similarly, there was no difference in VEGF-A gene expression level between the better prognosis VEGF-A rs699947 ‘CC and CA’ genotypes and the ‘AA’ genotype (mean 0.0012 vs. − 0.022, P = 0.74) (Fig. 4a,b).
Figure 4.
Box and whiskers plots of prognostic VEGF-A SNP genotypes and corresponding VEGF-A gene or protein expression. (a) VEGF-A rs25648 and (b) VEGF-A rs699947 genotypes with corresponding VEGF-A gene expression (log transformed, Almac Xcel microarray data). (c) VEGF-A rs25648 and (d) VEGF-A rs699947 genotypes with corresponding VEGF-A protein expression (Bioplex suspension array data). Boxes show the median with 25th (lower hinge) and 75th (upper hinge) percentiles, and whiskers show the upper and lower adjacent values. Solid circles represent the outliers.
Data from protein expression profiling was available for 193 of 309 (62%) participants who had a full genotype known for VEGF-A rs25648, and 193 of 302 (64%) participants who had a full genotype known for VEGF-A rs699947.
Consistent with the mRNA expression data, no difference in VEGF-A protein expression level was observed between the better prognosis VEGF-A rs25648 ‘CC’ genotype group and the ‘TT and CT’ genotypes (mean 6.26 versus 5.80, P = 0.38), and between the better prognosis VEGF-A rs699947 ‘CC and CA’ genotypes compared to the ‘AA’ genotype (mean 6.04 versus 6.26, P = 0.66) (Fig. 4c,d).
Grade ≥ 3 hypertension
Participants with the VEGFR2 rs11133360 ‘TT’ genotype (homozygous major allele) had a significantly lower odds of grade ≥ 3 hypertension than participants with the ‘CC and TC’ genotypes (P = 0.028, Table 3).
Table 3.
Association between VEGFR2 rs11133360 genotypes and grade ≥ 3 hypertension.
| VEGFR2 rs111333360 | Grade ≥ 3 hypertension (n) | ||
|---|---|---|---|
| No | Yes | ||
| Not homozygous TT | 180 | 9 | |
| Homozygous TT | 102 | 0 | P = 0.028 |
aConditional Binomial Exact Test (CBET) for independence between SNP and hypertension.
As there were no events in participants with the VEGFR2 rs11133360 ‘TT’ genotype, an OR was not estimable for both a univariate and multivariate analysis adjusting for allocated treatment. There was no association between any of the other SNPs and grade ≥ 3 hypertension (Supplementary Table S10).
Discussion
In this study we interrogated genomic DNA from mCRC patients who participated in the phase III MAX clinical trial to identify polymorphisms in the VEGF-A, VEGFR1 and VEGFR2 genes which were prognostic or predictive of outcome following first-line chemotherapy with or without bevacizumab treatment.
We observed that the VEGF-A rs25648 ‘CC’ genotype was associated with improved PFS and OS outcomes, and the VEGF-A rs699947 ‘AA’ genotype was associated with shorter PFS. However, neither of these SNPs was predictive of greater bevacizumab treatment effect on PFS and OS, nor ORR. The VEGFR2 rs11133360 ‘TT’ genotype was associated with a lower risk of grade ≥ 3 hypertension regardless of bevacizumab treatment.
The MAX study had the ideal study design for incorporation of a retrospective predictive biomarker study, as it was a Phase 3 randomised controlled trial which included a non-bevacizumab chemotherapy-only control arm. We identified only one other study which examined SNPs in VEGF-A, VEGFR1 and VEGFR2 as predictive biomarkers of bevacizumab outcome in patients with mCRC which also had this optimal study design (Ulivi et al.9). In contrast to our study, where SNP genotyping was performed using FFPE normal tissue and tumour tissue in 50% and 47% of participants respectively, Ulivi et al. used peripheral blood samples in 153/237 (65%) participants and FFPE tumour tissue in 84/237 (35%) participants. Their study also did not identify any SNPs which were predictive of bevacizumab response. However, they found that the VEGF-A rs3025039 ‘TT’ genotype was prognostic for poorer PFS and OS, and that the VEGF-A rs2010963 ‘GC’ genotype was prognostic for an inferior ORR. In comparison, there was no association between these SNP genotypes and efficacy outcomes in our study.
However, our study did have some limitations. As this was a post-hoc retrospective analysis of multiple genetic biomarkers, the findings are exploratory in nature. Therefore, there is a possibility that our findings may have been a chance finding. We did not make specific a priori hypotheses with respect to which SNPs would demonstrate clear signals, the magnitude of any signals observed, or the variability and the strength (P values) of these signals. Accordingly, due to the exploratory nature of the analysis, no formal adjustment for multiple comparisons was made. We have not confirmed our findings in a second validation cohort. While we considered such a test-validation approach, we were constrained by the distribution of events within SNP level, the distribution of treatment across the levels of SNPs, the lack of knowledge regarding which SNPs would hold the signals, and the relatively small sample size of the study.
This biomarker analysis is restricted to 69% of participants in the MAX study for whom FFPE tissue samples were available, and is therefore not necessarily generalisable to the general population. However, the baseline characteristics and survival outcomes in the total study population and the SNP biomarker study population were comparable. SNPs were genotyped using a candidate approach, based on previous demonstration of functional effects on corresponding gene and/or protein expression or associations with clinical outcomes. An alternative approach for selecting SNPs to genotype is SNP tagging. This is a more systematic method that may lead to the testing of SNPs which were not identified in previous studies19.
Genetic variants may be associated with different disease risk in different ethnic populations51, and race or ethnicity may be a confounder in SNP biomarker studies20. Heterogeneity of effect by race or ethnicity may be observed due to variation in linkage disequilibrium among populations, allelic heterogeneity, or gene–gene and gene-environment interactions51–54. Information on the race or ethnicity of participants was not collected in the MAX clinical trial, therefore, we were not able to restrict analyses to an ethnically homogenous population or adjust for ethnicity in the analysis, which are approaches which have been used in some previous genetic biomarker studies of bevacizumab17,19,20,55,56.
Our finding that the VEGF-A rs25648 ‘CC’ genotype was associated with improved PFS and OS is supported by the results of a phase II single-arm study of patients treated with first-line FOLFIRI and bevacizumab for mCRC (Becouarn et al.), where participants with VEGF-A rs25648 C-variants had a longer OS than those with alternative genotypes13.
Our finding that the VEGF-A rs699947 ‘AA’ genotype was prognostic for shorter PFS than C-variants in the MAX study is supported by one phase III study of patients treated with first-line docetaxel with or without bevacizumab for metastatic breast cancer, in which the VEGF-A rs699947 C-allele was associated with longer PFS in the placebo arm55. However, the results of several other previous studies in the literature conflict with this finding. In a single-arm pilot study of 59 patients with mCRC who were treated with second-line gemcitabine, irinotecan and bevacizumab, Abajo et al. reported that participants with at least one of the VEGF-A rs699947 ‘AA’, rs833061 ‘CC’, or rs2010963 ‘GG’ genotypes had a significantly longer median TTP than those without these genotypes10. In studies of patients with glioblastoma (Galanis et al.)21 and metastatic breast cancer (Schneider et al.)32, the VEGF-A rs699947 AA genotype was associated with a superior 6-month PFS and median OS, respectively. The conflicting results amongst these studies of VEGF family SNPs and efficacy outcomes could be explained by the heterogeneous patient populations, or differences in tumour vasculature between different cancer types or following treatment with different regimens22.
In this study, no participant with VEGFR2 rs11133360 ‘TT’ recorded grade ≥ 3 hypertension. Due to no events in participants with the VEGFR2 rs11133360 ‘TT’ genotype, an OR was not estimable for multivariate analysis adjusting for bevacizumab treatment. Further research validating this finding in a larger cohort of patients would support our findings and may enable further analysis adjusting for bevacizumab. To our knowledge, this is the first study to demonstrate an association between VEGFR2 rs11133360 and hypertension, although a previous study showed an association between the rs11133360 T-allele and improved PFS in patients who received bevacizumab18,22. We did not find an association between other VEGF family SNPs and grade ≥ 3 hypertension.
Having identified associations between specific SNPs in VEGF family genes and efficacy outcomes, we considered how these SNPs may alter VEGF-A function to bring about this effect. One possibility is that, being located in the 5’UTR (rs25648) or near or within a proximal enhancer/promoter regulatory element (rs699947), these SNPs may impact levels of VEGF-A mRNA expression. However, no associations between these VEGF-A SNP genotypes and corresponding VEGF-A expression which could explain their links with prognosis were observed. Our group previously found that tumour expression of VEGF-D using immunohistochemistry may be a potential predictive biomarker for bevacizumab efficacy on PFS in patients with mCRC57. However, we did not analyse associations between SNPs in VEGF-A and its receptors and VEGF-D expression because they would not be expected to have an effect on VEGF-D expression or function. Notably, the FFPE tissue samples which we used to measure VEGF-A mRNA and protein expression were from participants’ primary tumours. The majority of patients did not have tissue samples from their metastatic tumour available for mRNA extraction—a common situation in biomarker studies in patients with metastatic disease58. As angiogenesis is a complex, dynamic and adaptive process which varies over time22,58, VEGF-A expression in the primary tumour may not reflect expression levels in the metastatic tumour at the time of study participation, when prognosis was examined. This may explain the observed lack of association between VEGF-A gene and protein expression and VEGF-A SNP genotypes. However, in 47 (10%) MAX study participants for whom matched primary and metastatic tumour samples were available, we have found a moderate positive correlation between VEGF-A mRNA expression in the primary and metastatic tumour (r = 0.40, P = 0.006). Studies examining the association between VEGF-A mRNA in matched primary and metastatic tumour from a larger number of patients are required to further address this.
Another possibility is that the SNPs which we identified as prognostic biomarkers exert their prognostic impact by affecting the expression of other genes, for example, by acting as a distal enhancer.
rs11133360, which was associated with hypertension in our study, is located in the third intron of the VEGFR2 gene, ~ 7 kb from the promoter and 5’UTR (Supplementary Fig. S2). Intronic SNPs may influence the transcriptional activity or splicing efficiency of host genes, or alter the expression of alternative transcripts59. Notably, our bioinformatic analysis revealed that rs11133360 is located within a GATA2 binding region and also coincides with a DNAase I hypersensitivity site (Supplementary Fig. S2). GATA2 has previously been demonstrated to be a bona fide transcription factor that regulates the expression of mouse isoforms of VEGFR2 (FLK-1) via an enhancer sequence60. Our in silico analysis indicated that the ‘not homozygous TT’ (i.e. ‘CC’ or ‘CT’) genotype is expected to result in the loss of GATA2 binding at the rs11133360-associated GATA2 binding site, which could lead to the reduction of GATA2-mediated VEGFR2 expression in endothelial cells. Blocking of VEGFR2 has been associated with hypertension in pre-clinical studies61, providing biological plausibility for our finding of increased hypertension in patients without the homozygous ‘TT’ genotype.
Conclusions
In conclusion, we did not find any grade ≥ 3 hypertension among participants having the VEGFR2 rs11133360 ‘TT’ genoptype. This is the first study to demonstrate this association and future research should focus on validating this finding in further cohorts. We also found that the VEGF-A rs25648 ‘CC’ genotype was a prognostic biomarker for improved PFS and OS outcomes, and the VEGF-A rs699947 ‘AA’ genotype was prognostic for a shorter PFS. However, neither of these SNPs were predictive biomarkers for bevacizumab treatment effect. Ongoing research to identify and validate predictive biomarkers for efficacy outcomes in patients with mCRC treated with bevacizumab is required, in order to better select patients for this treatment.
Supplementary Information
Acknowledgements
We thank Carmel Murone for providing pathology assistance. This research was presented in part at the 2014 American Society of Clinical Oncology Annual Meeting (Chicago, USA, 30 May to 3 June 2014; published conference abstract #3609).
Author contributions
Conception and design—F.C., V.G., J.S., J.M.M., N.C.T. Methodology—F.C., V.G., J.S., J.M.M., N.C.T. Formal analysis—F.C., V.G., CKL, ACC. Investigation—F.C., S.J.A., J.K.M., M.A.B., A.C.C., D.S.W. Resources—J.M.M., N.C.T.. Data curation—F.C., AJW, KW. Writing—original draft—F.C.. Writing—review & editing—V.G., S.J.A., J.K.M., M.A.B., C.K.L., A.C.C., D.S.W., A.J.W., K.W., A.M.S., J.S., J.E.H., T.J.P., J.M.M., N.C.T. Visualisation—F.C., V.G., C.K.L., A.C.C. Supervision—F.C., AMS, J.M.M., N.C.T. Project administration—KW. Funding acquisition—F.C., J.M.M., N.C.T.
Funding
This translational sub-study was supported by a 2012 National Health and Medical Research Council (NH&MRC) Project Grant (1048088). FC was supported by a 2011 NH&MRC Medical/Dental Postgraduate Research Scholarship (1017737), 2011 Pfizer Australia Cancer Research Grant and 2017 Brian Smith Memorial Award. CKL is supported by research funding from Astra Zeneca. AJW is supported by research funding from Merck and BMS. AMS is supported by an NH&MRC Investigator grant (1177837). JMM was supported by a NH&MRC Senior Research Fellowship (1046092). None of these funding sources had a role in the study design; collection, analysis or interpretation of data; writing of the report or the decision to submit the article for publication.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
FC: honorarium—Roche. CKL: honoraria—Astra Zeneca, Novartis, Pfizer, Roche and Boehringer Ingelheim, advisory role—Astra Zeneca, Novartis, Pfizer and Boehringer Ingelheim, and travel/accommodation/other expenses from Astra Zeneca and Boehringer Ingelheim. AJW: honoraria—Merck, Bristol Myers Squibb, advisory role—Merck, Pfizer, Bristol Myers Squibb, Ipsen. The other authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: John M. Mariadason and Niall C. Tebbutt.
Contributor Information
John M. Mariadason, Email: john.mariadason@onjcri.org.au
Niall C. Tebbutt, Email: niall.tebbutt@austin.org.au
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-03952-y.
References
- 1.Hurwitz HI, et al. Bevacizumab in combination with fluorouracil and leucovorin: An active regimen for first-line metastatic colorectal cancer. J. Clin. Oncol. 2005;23:3502–3508. doi: 10.1200/JCO.2005.10.017. [DOI] [PubMed] [Google Scholar]
- 2.Saltz LB, et al. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: A randomized phase III study. J. Clin. Oncol. 2008;26:2013–2019. doi: 10.1200/JCO.2007.14.9930. [DOI] [PubMed] [Google Scholar]
- 3.Tebbutt NC, et al. Capecitabine, bevacizumab, and mitomycin in first-line treatment of metastatic colorectal cancer: Results of the Australasian Gastrointestinal Trials Group Randomized Phase III MAX Study. J. Clin. Oncol. 2010;28:3191–3198. doi: 10.1200/JCO.2009.27.7723. [DOI] [PubMed] [Google Scholar]
- 4.Schneider BP, Radovich M, Miller KD. The role of vascular endothelial growth factor genetic variability in cancer. Clin Cancer Res. 2009;15:5297–5302. doi: 10.1158/1078-0432.CCR-08-2576. [DOI] [PubMed] [Google Scholar]
- 5.Chung AS, Ferrara N. Developmental and pathological angiogenesis. Annu. Rev. Cell. Dev. Biol. 2011;27:563–584. doi: 10.1146/annurev-cellbio-092910-154002. [DOI] [PubMed] [Google Scholar]
- 6.Koch S, Tugues S, Li X, Gualandi L, Claesson-Welsh L. Signal transduction by vascular endothelial growth factor receptors. Biochem. J. 2011;437:169–183. doi: 10.1042/BJ20110301. [DOI] [PubMed] [Google Scholar]
- 7.Shinkai A, et al. Mapping of the sites involved in ligand association and dissociation at the extracellular domain of the kinase insert domain-containing receptor for vascular endothelial growth factor. J. Biol. Chem. 1998;273:31283–31288. doi: 10.1074/jbc.273.47.31283. [DOI] [PubMed] [Google Scholar]
- 8.Koch S, Claesson-Welsh L. Signal transduction by vascular endothelial growth factor receptors. Cold Spring Harb. Perspect. Med. 2012;2:a006502. doi: 10.1101/cshperspect.a006502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ulivi P, et al. eNOS polymorphisms as predictors of efficacy of bevacizumab-based chemotherapy in metastatic colorectal cancer: Data from a randomized clinical trial. J. Transl. Med. 2015;13:258. doi: 10.1186/s12967-015-0619-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Abajo A, et al. Dose-finding study and pharmacogenomic analysis of fixed-rate infusion of gemcitabine, irinotecan and bevacizumab in pretreated metastatic colorectal cancer patients. Br. J. Cancer. 2010;103:1529–1535. doi: 10.1038/sj.bjc.6605908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Formica V, et al. Predictive value of VEGF gene polymorphisms for metastatic colorectal cancer patients receiving first-line treatment including fluorouracil, irinotecan, and bevacizumab. Int. J. Colorectal. Dis. 2011;26:143–151. doi: 10.1007/s00384-010-1108-1. [DOI] [PubMed] [Google Scholar]
- 12.Sohn BS, et al. Single-nucleotide polymorphisms in the vascular endothelial growth factor pathway and outcomes of patients treated with first-line cytotoxic chemotherapy combined with bevacizumab for advanced colorectal cancer. Oncology. 2014;87:280–292. doi: 10.1159/000365593. [DOI] [PubMed] [Google Scholar]
- 13.Becouarn Y, et al. FOLFIRI(R) and bevacizumab in first-line treatment for colorectal cancer patients: Safety, efficacy and genetic polymorphisms. BMC Res. Notes. 2014;7:260. doi: 10.1186/1756-0500-7-260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hansen TF, et al. The predictive value of single nucleotide polymorphisms in the VEGF system to the efficacy of first-line treatment with bevacizumab plus chemotherapy in patients with metastatic colorectal cancer: Results from the Nordic ACT trial. Int. J. Colorectal. Dis. 2012;27:715–720. doi: 10.1007/s00384-011-1382-6. [DOI] [PubMed] [Google Scholar]
- 15.Gerger A, et al. Pharmacogenetic angiogenesis profiling for first-line Bevacizumab plus oxaliplatin-based chemotherapy in patients with metastatic colorectal cancer. Clin. Cancer Res. 2011;17:5783–5792. doi: 10.1158/1078-0432.CCR-11-1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Loupakis F, et al. Prospective validation of candidate SNPs of VEGF/VEGFR pathway in metastatic colorectal cancer patients treated with first-line FOLFIRI plus bevacizumab. PLoS ONE. 2013;8:e66774. doi: 10.1371/journal.pone.0066774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.de Haas S, et al. Genetic variability of VEGF pathway genes in six randomized phase III trials assessing the addition of bevacizumab to standard therapy. Angiogenesis. 2014;17:909–920. doi: 10.1007/s10456-014-9438-1. [DOI] [PubMed] [Google Scholar]
- 18.Lambrechts D, et al. Single nucleotide polymorphism analysis and outcome in advanced-stage cancer patients treated with bevacizumab. Eur J Cancer. 2011;47:S173–920. doi: 10.1016/S0959-8049(11)70907-8. [DOI] [Google Scholar]
- 19.Lambrechts D, et al. VEGF pathway genetic variants as biomarkers of treatment outcome with bevacizumab: An analysis of data from the AViTA and AVOREN randomised trials. Lancet Oncol. 2012;13:724–733. doi: 10.1016/S1470-2045(12)70231-0. [DOI] [PubMed] [Google Scholar]
- 20.Pallaud C, et al. Clinical genotyping and efficacy outcomes: Exploratory biomarker data from the phase II ABIGAIL study of first-line bevacizumab plus chemotherapy in non-squamous non-small-cell lung cancer. Lung Cancer. 2014;86:67–72. doi: 10.1016/j.lungcan.2014.07.019. [DOI] [PubMed] [Google Scholar]
- 21.Galanis E, et al. Phase II study of bevacizumab in combination with sorafenib in recurrent glioblastoma (N0776): A north central cancer treatment group trial. Clin. Cancer Res. 2013;19:4816–4823. doi: 10.1158/1078-0432.CCR-13-0708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lambrechts D, Lenz HJ, de Haas S, Carmeliet P, Scherer SJ. Markers of response for the antiangiogenic agent bevacizumab. J. Clin. Oncol. 2013;31:1219–1230. doi: 10.1200/JCO.2012.46.2762. [DOI] [PubMed] [Google Scholar]
- 23.Berger MD, et al. Impact of genetic variations in the MAPK signaling pathway on outcome in metastatic colorectal cancer patients treated with first-line FOLFIRI and bevacizumab: Data from FIRE-3 and TRIBE trials. Ann. Oncol. 2017;28:2780–2785. doi: 10.1093/annonc/mdx412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Berger MD, et al. A polymorphism within the vitamin D transporter gene predicts outcome in metastatic colorectal cancer patients treated with FOLFIRI/bevacizumab or FOLFIRI/cetuximab. Clin. Cancer Res. 2018;24:784–793. doi: 10.1158/1078-0432.CCR-17-1663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Berger MD, et al. A polymorphism within the R-spondin 2 gene predicts outcome in metastatic colorectal cancer patients treated with FOLFIRI/bevacizumab: Data from FIRE-3 and TRIBE trials. Eur. J. Cancer. 2020;131:89–97. doi: 10.1016/j.ejca.2020.02.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ranpura V, Pulipati B, Chu D, Zhu X, Wu S. Increased risk of high-grade hypertension with bevacizumab in cancer patients: A meta-analysis. Am. J. Hypertens. 2010;23:460–468. doi: 10.1038/ajh.2010.25. [DOI] [PubMed] [Google Scholar]
- 27.Wang Y, Fei D, Vanderlaan M, Song A. Biological activity of bevacizumab, a humanized anti-VEGF antibody in vitro. Angiogenesis. 2004;7:335–345. doi: 10.1007/s10456-004-8272-2. [DOI] [PubMed] [Google Scholar]
- 28.Thijs AM, et al. Role of endogenous vascular endothelial growth factor in endothelium-dependent vasodilation in humans. Hypertension. 2013;61:1060–1065. doi: 10.1161/HYPERTENSIONAHA.111.00841. [DOI] [PubMed] [Google Scholar]
- 29.Kamba T, et al. VEGF-dependent plasticity of fenestrated capillaries in the normal adult microvasculature. Am. J. Physiol. Heart Circ. Physiol. 2006;290:H560–576. doi: 10.1152/ajpheart.00133.2005. [DOI] [PubMed] [Google Scholar]
- 30.Eremina V, et al. VEGF inhibition and renal thrombotic microangiopathy. N. Engl. J. Med. 2008;358:1129–1136. doi: 10.1056/NEJMoa0707330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zou AP, Cowley AW., Jr Role of nitric oxide in the control of renal function and salt sensitivity. Curr. Hypertens. Rep. 1999;1:178–186. doi: 10.1007/s11906-999-0016-7. [DOI] [PubMed] [Google Scholar]
- 32.Schneider BP, et al. Association of vascular endothelial growth factor and vascular endothelial growth factor receptor-2 genetic polymorphisms with outcome in a trial of paclitaxel compared with paclitaxel plus bevacizumab in advanced breast cancer: ECOG 2100. J. Clin. Oncol. 2008;26:4672–4678. doi: 10.1200/JCO.2008.16.1612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Etienne-Grimaldi MC, et al. Prospective analysis of the impact of VEGF-A gene polymorphisms on the pharmacodynamics of bevacizumab-based therapy in metastatic breast cancer patients. Br. J. Clin. Pharmacol. 2011;71:921–928. doi: 10.1111/j.1365-2125.2010.03896.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Morita S, et al. Association between bevacizumab-related hypertension and vascular endothelial growth factor (VEGF) gene polymorphisms in Japanese patients with metastatic colorectal cancer. Cancer Chemother. Pharmacol. 2013;71:405–411. doi: 10.1007/s00280-012-2028-2. [DOI] [PubMed] [Google Scholar]
- 35.Sibertin-Blanc C, et al. Vascular Endothelial Growth Factor A c.*237C>T polymorphism is associated with bevacizumab efficacy and related hypertension in metastatic colorectal cancer. Dig. Liver Dis.. 2015;47:331–337. doi: 10.1016/j.dld.2014.12.013. [DOI] [PubMed] [Google Scholar]
- 36.Lambrechts D, et al. Genetic markers of bevacizumab-induced hypertension. Angiogenesis. 2014;17:685–694. doi: 10.1007/s10456-014-9424-7. [DOI] [PubMed] [Google Scholar]
- 37.Jain L, et al. Hypertension and hand-foot skin reactions related to VEGFR2 genotype and improved clinical outcome following bevacizumab and sorafenib. J. Exp. Clin. Cancer Res. 2010;29:95. doi: 10.1186/1756-9966-29-95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Li M, et al. Identification of a genomic region between SLC29A1 and HSP90AB1 associated with risk of bevacizumab-induced hypertension: CALGB 80405 (Alliance) Clin. Cancer Res. 2018;24:4734–4744. doi: 10.1158/1078-0432.CCR-17-1523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Berger MD, et al. Autophagy-related polymorphisms predict hypertension in patients with metastatic colorectal cancer treated with FOLFIRI and bevacizumab: Results from TRIBE and FIRE-3 trials. Eur. J. Cancer. 2017;77:13–20. doi: 10.1016/j.ejca.2017.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Glubb DM, et al. Novel functional germline variants in the VEGF receptor 2 gene and their effect on gene expression and microvessel density in lung cancer. Clin. Cancer Res. 2011;17:5257–5267. doi: 10.1158/1078-0432.CCR-11-0379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yamamori M, et al. Association of VEGF genotype with mRNA level in colorectal adenocarcinomas. Biochem. Biophys. Res. Commun. 2004;325:144–150. doi: 10.1016/j.bbrc.2004.10.005. [DOI] [PubMed] [Google Scholar]
- 42.Chen MH, et al. VEGF -460T –> C polymorphism and its association with VEGF expression and outcome to FOLFOX-4 treatment in patients with colorectal carcinoma. Pharmacogenomics J. 2011;11:227–236. doi: 10.1038/tpj.2010.48. [DOI] [PubMed] [Google Scholar]
- 43.Qin C, et al. Variants in angiogenesis-related genes and the risk of clear cell renal cell carcinoma. Mutagenesis. 2014;29:419–425. doi: 10.1093/mutage/geu046. [DOI] [PubMed] [Google Scholar]
- 44.Di Stefano AL, et al. VEGFA SNP rs2010963 is associated with vascular toxicity in recurrent glioblastomas and longer response to bevacizumab. J. Neurooncol. 2015;121:499–504. doi: 10.1007/s11060-014-1677-x. [DOI] [PubMed] [Google Scholar]
- 45.Watson CJ, Webb NJ, Bottomley MJ, Brenchley PE. Identification of polymorphisms within the vascular endothelial growth factor (VEGF) gene: Correlation with variation in VEGF protein production. Cytokine. 2000;12:1232–1235. doi: 10.1006/cyto.2000.0692. [DOI] [PubMed] [Google Scholar]
- 46.Ruggiero D, et al. Genetics of VEGF serum variation in human isolated populations of cilento: Importance of VEGF polymorphisms. PLoS ONE. 2011;6:e16982. doi: 10.1371/journal.pone.0016982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Fraga A, et al. Genetic polymorphisms in key hypoxia-regulated downstream molecules and phenotypic correlation in prostate cancer. BMC Urol. 2017;17:12. doi: 10.1186/s12894-017-0201-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mooi JK, et al. The prognostic impact of consensus molecular subtypes (CMS) and its predictive effects for bevacizumab benefit in metastatic colorectal cancer: Molecular analysis of the AGITG MAX clinical trial. Ann. Oncol. 2018;29:2240–2246. doi: 10.1093/annonc/mdy410. [DOI] [PubMed] [Google Scholar]
- 49.Bruhn MA, et al. Proangiogenic tumor proteins as potential predictive or prognostic biomarkers for bevacizumab therapy in metastatic colorectal cancer. Int. J. Cancer. 2014;135:731–741. doi: 10.1002/ijc.28698. [DOI] [PubMed] [Google Scholar]
- 50.Rice WR. A new probability model for determining exact P-values for 2 x 2 contingency tables when comparing binomial proportions. Biometrics. 1988;44:1–22. doi: 10.2307/2531892. [DOI] [Google Scholar]
- 51.Rosenberg NA, et al. Genome-wide association studies in diverse populations. Nat. Rev. Genet. 2010;11:356–366. doi: 10.1038/nrg2760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Thomas D. Gene–environment-wide association studies: Emerging approaches. Nat Rev Genet. 2010;11:259–272. doi: 10.1038/nrg2764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wood AR, et al. Allelic heterogeneity and more detailed analyses of known loci explain additional phenotypic variation and reveal complex patterns of association. Hum. Mol. Genet. 2011;20:4082–4092. doi: 10.1093/hmg/ddr328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Seyerle AA, et al. Evidence of heterogeneity by race/ethnicity in genetic determinants of QT interval. Epidemiology. 2014;25:790–798. doi: 10.1097/EDE.0000000000000168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Miles DW, et al. Biomarker results from the AVADO phase 3 trial of first-line bevacizumab plus docetaxel for HER2-negative metastatic breast cancer. Br. J. Cancer. 2013;108:1052–1060. doi: 10.1038/bjc.2013.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Makhoul I, et al. Germline Genetic Variants in TEK, ANGPT1, ANGPT2, MMP9, FGF2 and VEGFA Are Associated with Pathologic Complete Response to Bevacizumab in Breast Cancer Patients. PLoS ONE. 2017;12:e0168550. doi: 10.1371/journal.pone.0168550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Weickhardt AJ, et al. Vascular endothelial growth factor D expression is a potential biomarker of bevacizumab benefit in colorectal cancer. Br. J. Cancer. 2015;113:37–45. doi: 10.1038/bjc.2015.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Jubb AM, Harris AL. Biomarkers to predict the clinical efficacy of bevacizumab in cancer. Lancet Oncol. 2010;11:1172–1183. doi: 10.1016/S1470-2045(10)70232-1. [DOI] [PubMed] [Google Scholar]
- 59.Cooper DN. Functional intronic polymorphisms: Buried treasure awaiting discovery within our genes. Hum Genomics. 2010;4:284–288. doi: 10.1186/1479-7364-4-5-284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kappel A, et al. Role of SCL/Tal-1, GATA, and ets transcription factor binding sites for the regulation of flk-1 expression during murine vascular development. Blood. 2000;96:3078–3085. doi: 10.1182/blood.V96.9.3078. [DOI] [PubMed] [Google Scholar]
- 61.Facemire CS, Nixon AB, Griffiths R, Hurwitz H, Coffman TM. Vascular endothelial growth factor receptor 2 controls blood pressure by regulating nitric oxide synthase expression. Hypertension. 2009;54:652–658. doi: 10.1161/HYPERTENSIONAHA.109.129973. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.