ABSTRACT
The pandemic caused by the SARS-CoV-2 virus has triggered great interest in the search for the pathophysiological mechanisms of COVID-19 and its associated hyperinflammatory state. The presence of prognostic factors such as diabetes, cardiovascular disease, hypertension, obesity, and age influence the expression of the disease’s clinical severity. Other elements, such as 25-hydroxyvitamin D (25(OH)D3) concentrations, are currently being studied. Various studies, mostly observational, have sought to demonstrate whether there is truly a relationship between 25(OH)D3 levels and the acquisition and/or severity of the disease. The objective of this study was to carry out a review of the current data that associate vitamin D status with the acquisition, evolution, and/or severity of infection by the SARS-CoV-2 virus and to assess whether prevention through vitamin D supplementation can prevent infection and/or improve the evolution once acquired. Vitamin D system has an immunomodulatory function and plays a significant role in various bacterial and viral infections. The immune function of vitamin D is explained in part by the presence of its receptor (VDR) and its activating enzyme 25-hydroxyvitamin D-1alpha-hydroxylase (CYP27B1) in immune cells. The vitamin D, VDR, and Retinoid X Receptor complex allows the transcription of genes with antimicrobial activities, such as cathelicidins and defensins. COVID-19 characteristically presents a marked hyperimmune state, with the release of proinflammatory cytokines such as IL-6, TNF-α, and IL-1β. Thus, there are biological factors linking vitamin D to the cytokine storm, which can herald some of the most severe consequences of COVID-19, such as acute respiratory distress syndrome. Hypovitaminosis D is widespread worldwide, so the prevention of COVID-19 through vitamin D supplementation is being considered as a possible therapeutic strategy easy to implement. However, more-quality studies and well-designed randomized clinical trials are needed to address this relevant question.
KEYWORDS: Vitamin D, 25-hydroxyvitamin D, vitamin D receptor (VDR), 25-hydroxyvitamin D-1alpha-hydroxylase (CYP27B1), SARS-CoV-2, COVID-19
1. Introduction
The pandemic caused by the SARS-CoV-2 virus has triggered great interest in the search for the pathophysiological mechanisms of COVID-19 (the infectious disease caused by SARS-CoV-2) and the associated hyperinflammatory state. Several studies have noted that factors such as age, sex, race, obesity, diabetes and hypertension act as risk factors in establishing an uncontrolled cytokine release that leads to an exacerbated and unbalanced immune response [1–6].
In the immune response to SARS-CoV-2, the point at which the immune response is no longer regulated and becomes an exacerbated response appears to be at the transitional phase between innate and adaptive immunity. A correct protective response involves CD4 T cells, which influence antibody-producing B cells, as well as cytotoxic CD8 T cells, which are responsible for the elimination of infected cells. If these cells are not eliminated, and viral replication is not inhibited, the immune response will be perpetuated and exacerbated, resulting in acute respiratory distress syndrome (ARDS) or disseminated intravascular coagulation [7,8].
Some studies have shown that low 25-hydroxyvitamin D (25(OH)D3) concentrations are related to pathological conditions such as bacterial and/or viral infections [9–13]. In fact, vitamin D deficiency is a documented risk factor for the development of exaggerated and persistent inflammation, which is a precursor to ARDS [14]. The rapid spread of COVID-19 in the northern hemisphere during the winter season has been linked to vitamin D deficiency [15]. Adequate 25(OH)D3 levels in patients with COVID-19 could be considered a possible protective factor and may have prognostic value for the susceptibility and evolution of the disease.
It has been described that the active form of vitamin D exerts an immunomodulatory and antimicrobial effect, which supports the use of vitamin D supplementation for the prevention of infections [12,13]. In COVID-19, different studies suggest different mechanisms. Although the exact mechanism by which vitamin D could prevent Sars-Cov-2 infection or improve COVID-19 prognosis is unclear several studies have shown that vitamin D and its metabolites may be involved both in the viral replication process and in the binding of the virus to the host, so that supplementation with vitamin D metabolites as well as with its biologically active hydroxyderivatives could be a promising therapeutic strategy [16,17].
Based on these premises, the aim of this review was to contemplate whether 25(OH)D3 levels and its derivatives can influence the acquisition, evolution, and/or severity of COVID-19 through its possible mechanisms of action. In addition, we also consider whether vitamin D supplementation may prevent infection and/or improve the evolution of the infection once acquired analyzing delivery routes and doses.
2. Materials and methods
A search of the scientific literature published in PubMed through July 2021 was conducted to identify peer-reviewed articles on COVID-19 -especially about the immune functions of vitamin D and COVID-19 acquisition- as well as the evolution and its resolution or prevention through the presentation of optimal 25(OH)D3 levels. Original human research articles published in English, prospective and retrospective observational studies and randomized controlled trials were included. Also, single case reports, case series, editorials, letters to the editor, comments (to an article or from the editor), responses (to a comment, letter or article), short reports and communications, corrections, opinions perspectives and debates were considered. The largest studies and the most recent and strongest available evidence were prioritized.
3. COVID-19 and biological activity of vitamin D
The term vitamin D includes vitamin D3 (cholecalciferol) and vitamin D2 (ergocalciferol). Vitamin D3 is synthetized mainly in the skin, from its precursor 7-dehydrocholesterol by ultraviolet irradiation. Other sources of vitamin D3 include the diet. Circulating vitamin D3 is bound to vitamin D binding protein (DBP), which transports it to the liver where is hydroxylated by vitamin D25-hydroxylase to 25-hydroxyvitamin D3 (25(OH)D33 or calcidiol). This hydroxylated form is metabolized to its active metabolite 1,25-dihydroxyvitamin D3 (1,25(OH)2D3 or calcitriol), preferentially in the kidney by the enzyme CYP27B1. Vitamin D2 is obtained mainly from dietary. It is excreted mainly in the bile, but a small amount is reabsorbed in the small intestine [18].
The fact that CYP27B1 is expressed in several tissues that also express the vitamin D receptor (VDR), linked to about 3% of the human genome is regulated by the hormone 1,25(OH)2D3 [18–21],, suggests that vitamin D could have additional functions different than bone metabolism [22,23].
Although the best known activation pathway for vitamin D is through the sequence previously described, several evidences have shown an additional non-canonical vitamin D activation pathway through steroidogenic enzyme cytochrome P450scc (CYP11A1) produced by placenta, adrenal glands, and epidermal keratinocytes that generates novel vitamin D-hydroxyderivatives different from 25(OH)D3 and 1,25(OH)2D3. Although the importance and physiological role of the CYP11A1-derived vitamin D metabolites remain to be established, some studies suggested that they could act as hormones in vivo playing immunomodulatory roles, participating in the induction of intracellular free radical scavenging and attenuating DNA damage through the activation of the NRF2-regulated antioxidant response, p53-phosphorylation and its translocation to the nucleus [24–28].
VDR is the best known receptor of vitamin D belonging to the family of nuclear receptors involved in DNA transcription [29,30]. The VDR acts mainly as a nuclear transcription factor, however, non-genomic actions of vitamin D have been postulated that involve the rapid binding of 1,25(OH)2D3 to the cytosolic and membrane VDR activating numerous second messenger systems [31]. Some of the vitamin D hydroxyderivatives, can act as biased agonists of the VDR as well as inverse agonists of retinoic acid orphan receptors (ROR) α and γ [32,33].
Retinoid X Receptor (RXR) is the heterodimeric partner of VDR, and is found widely distributed in tissues and cells, and including those that do not express the VDR [34]. The VDR–RXR heterodimer binds to VDRs located in the promoter regions of target genes. VDR can either positively or negatively regulate the expression of certain genes by binding to the VDRs present in their promoter regions [35–37] or inhibit the expression of some genes by antagonizing the action of certain transcription factors, such as nuclear factor (NF)-AT and NF-κB [38–40]. 1,25(OH)2D3 leads to the activation of VDRs and acts by forming a complex with RXR that, in turn, influences the proteins of the innate and adaptive immune system. Additionally, liver X receptor (LXR) has been identified as the second nuclear receptor pathway for several D3-hydroxyderivatives, including 1,25(OH)2D3 suggesting a new mechanism of action for these compounds [41].
On the other hand, glutathione plays an important role in a multitude of cellular processes, and disturbances in glutathione homeostasis are involved in the etiology and/or progression of several diseases, including diseases of aging, cardiovascular, inflammatory, immune, metabolic and neurodegenerative diseases, and cancer. Moreover, low levels of intracellular glutathione are associated with decreased cellular antioxidant capacity [42]. The glutathione level also is involved in the regulation of vitamin D levels being essential for the conversion of provided vitamin D into active vitamin D metabolites: 25(OH)D3 and 1,25(OH)2D3 and regulating positively the bioavailability of 25(OH)D3 [43,44]. Moreover, vitamin D also increases glutathione level thus contributing to the reduction of the oxidative stress [45]. SARS-CoV2 infection triggers massive ROS production and oxidative damage. Jain et al have proposed that overproduction of ROS and excessive oxidative damage are responsible for impaired immunity, cytokine storm secretion and the onset of lung dysfunction in response to COVID-19 infection [46].
Vitamin D is a pluripotent hormone that modulates innate and adaptive immune responses [47]. Immunocytes such as macrophages, B and T lymphocytes, neutrophils, and dendritic cells express VDR, which enables the actions of vitamin D [48]. The role of vitamin D in immune functions appears to be mainly related to the bioavailability of the 25(OH)D3 form, the induction of the enzyme CYP27B1, and the peripheral tissue action of the active form [49].
Epithelia are the main barrier separating the environment from the body and are the first to respond to invading pathogens that lead to the activation of the innate immune system. These activate macrophages and dendritic cells and recruit neutrophils and T lymphocytes to the infection site. Epithelia express the CYP27B1 enzyme, and vitamin D stimulates the expression of gap protein and tight junction protein in the epithelia, which helps maintain the integrity of the epithelium, preventing virus penetration. In addition, vitamin D acts indirectly by stimulating autophagy and facilitating the death of epithelial virus-infected cells through the modulation of the mTOR pathway [49].
In addition, through the VDR-RXR complex, the production of peptides such as cathelicidin, defensin, and nucleotide binding oligomerization domain-containing protein 2 is stimulated, promoting the microorganisms destruction [49]. Furthermore, 1,25(OH)2D3 increases the synthesis of hepcidin, which contributes to the accumulation of iron intracellularly, thus preventing its use by microorganisms, and stimulates the production of nitric oxide and superoxide. The stimulation of innate immunity associated to vitamin D results in the synthesis of these proteins that exert antiviral activity [50].
A potential role for vitamin D in reducing the effects of the cytokine storm associated to severe COVID-19 is under study. In these severe cases, the initial infection of the airway epithelium leads to rapid viral replication linked to a delayed expression of class 1 interferon (IFNα/β) in dendritic cells due to viral infection [51]. IFNα/β normally stops viral replication allowing viral clearance by CD8 T cells, processes that in these severe cases, are inhibited [52]. On the other hand, the delayed expression of IFNα/β increases recruitment of proinflammatory cells, which secrete several proinflammatory cytokines/chemokines which attract other inflammatory cells such as monocytes, neutrophils and macrophages while sensitizing T cells to apoptosis [53]. The T cell response required for viral clearance is damped, and their role in buffering the cytokine storm is decreased [54].
Airway epithelia constitutively express both CYP27B1, 1,25(OH)2D3, and the VDR. Furthermore, several respiratory pathogens induce the expression of CYP27B1 and VDR in pulmonary alveolar macrophages, leading the activation of innate immunity and increasing local 1,25(OH)2D3 that has been shown to enhance viral neutralization and clearance while modulating the subsequent proinflammatory response. This hypothesis has to be confirmed for coronaviruses, such as SARS-CoV-2.
The role of vitamin D in the acquired immune response focuses on inhibition of the proliferation and differentiation of B and T cells, a reduction of the stimuli that give rise to the synthesis and secretion of antibodies and proinflammatory cytokines by these cells, and the proapoptotic effects of these cells, thus increasing immunotolerance in the organism. Thus, 1,25(OH)2D regulates adaptive immunity by limiting the maturation of dendritic cells and its capacity to present antigen to the T cells, as well as stimulating conversion of Th1 and Th17 proinflammatory subsets to Th2 and Treg subsets [55]. The result is a decrease in proinflammatory cytokines and an increase in anti-inflammatory cytokines, which can be mediated through the NFkβ pathway [55].
Moreover, non-calcemic CYP11A1-generated vitamin D3-hydroxyderivatives have anti-inflammatory properties, induce antioxidative responses, and stimulate innate immunity against infectious agents. These compounds are involved in the decrease of proinflammatory cytokines by downregulating nuclear-NFkβ-p65 activity and through an inverse agonism on ROR γ. They can also counteract the oxidative stress through activation of NRF-2 and p53-dependent pathways and induces the DNA repair system [28,56–58].
Although 25(OH)D3 is an important component of the antimicrobial response through its transformation to 1,25(OH)2D3, its role as a circulating lipophilic molecule bound to DBP is also noteworthy [59]. The ability of 25(OH)D3 to access immune target cells is limited by its binding to DBP, being the ‘free’ fraction of 25(OH)D3 which is acquired by immune cells to drive the intracrine induction of antimicrobial responses [60]. This suggests that, it is important to assess both the level of 25(OH)D3 and the associated concentration of DBP when considering the impact of vitamin D system on antimicrobial immune responses. In addition, it remains possible that infections and associated inflammatory immune responses lead to disturbances in vitamin D metabolism. Thus, 25(OH)D3 deficiency could be a possible consequence of an infectious disease and may, therefore, be considered a negative acute phase reactant [55].
Since vitamin D deficiency is a documented risk factor for the development of exaggerated and persistent inflammation, which is the prelude to ARDS, there may be a relationship between 25(OH)D3 levels and susceptibility to COVID-19.
COVID-19 begins when S protein binds to angiotensin-converting enzyme-2 (ACE2) [61]. ACE2’s cellular overexpression facilitates viral entry and replication. In the renin-angiotensin-aldosterone system (RAAS), the ACE converts angiotensin-I to angiotensin-II, a vasoactive peptide responsible for systemic vasoconstriction [62]. By contrast, ACE2 acts by inhibiting the effects of angiotensin-II, thereby catalyzing angiotensin-I and II by producing metabolites with vasodilatory action [63]. This makes it possible to control the occurrence of deleterious effects on the cardiovascular and immune system [64]. The down-regulation of ACE2 in COVID-19 leads to an accumulation of angiotensin-II and both pulmonary and cardiac damage, expressed as respiratory distress, myocarditis and other vascular complications [65,66]. ACE2 is less strongly expressed in males and the elderly, who are at the highest risk of severe COVID-19 infection [67]. Oxidative stress that is increased by viruses activating toll-like receptors (TLR) with subsequent release of cytokines constitutes an additional factor in the development of ARDS [68].
Vitamin D appears to have a potential effect on the RAAS. Calcitriol modulates the expression of angiotensin-I, renin, and angiotensin-II and appears to stimulate ACE2 expression. Thus, vitamin D may exert protective effects in the lung [69,70]. Moreover, 1,25(OH)2D3 has been shown to have a beneficial outcome in acute lung injury by improving lipopolysaccharide-induced lung permeability. The vitamin D/VDR signaling pathway may provide beneficial effects in lipopolysaccharide-mediated respiratory distress syndrome by decreasing cytokine storm and by regulating the RAAS [71] (Figure 1). Similarly, the anti-inflammatory and antioxidant properties of vitamin D hydroxyderivatives originating through the non-canonical vitamin D activation pathway, as well as their stimulatory action on innate immunity against infectious agents, may decrease the cytokine storm associated with ARDS. In this context, some metabolites of vitamin D have been shown to bind with high affinity to some enzymes of the Sars-Cov-2 virus replication machinery involved in the stabilization of the infection, inhibiting their activity. In addition, both vitamin D3 and its biologically active hydroxyderivatives can inhibit the binding of ACE2 to the RBD of SARS-CoV-2 thereby preventing the entry of SARS-CoV-2. Thus, treatment based on supplementation of these compounds appears to be a promising therapeutic strategy for COVID-19 [16,17,56,72].
Figure 1.
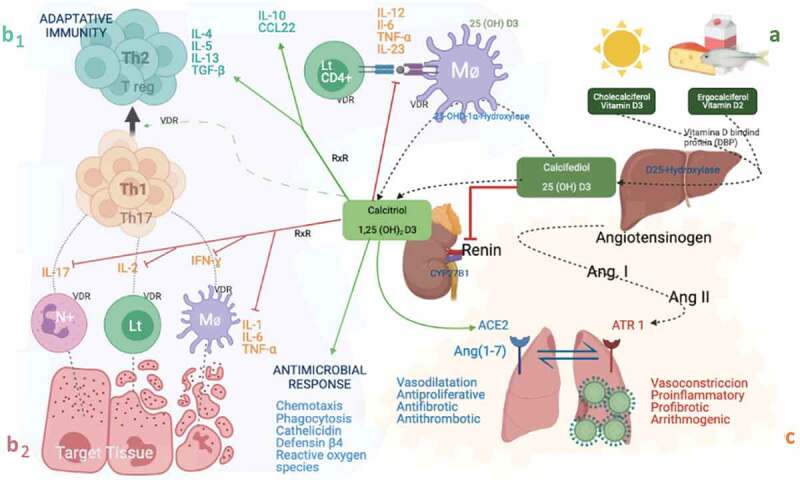
Schematic representation of the paracrine and intracrine functions of vitamin D and its metabolites and actions in the innate and adaptive immune systems. A: Vitamin D synthesis. B1: Adaptive immunity. B2: Innate immunity. C: Inflammatory response mediated by SARS-CoV-2
Therefore, the activation of innate immunity leading to increased local production of 1,25(OH)2D3 and its hydroxyderivatives that has been shown to improve the neutralization and clearance of virus while modulating the downstream proinflammatory response observed in some viruses and respiratory pathogens [55], is likely to be similar in SARS-CoV-2 based on recent findings.
Green and red arrows indicate activation and inhibition respectively. Proinflammatory cytokines in orange. Anti-inflammatory cytokines in greenish blue. Calcidiol or 25-hydroxyvitamin D: (25(OH)D3). Calcitriol or 1,25-dihydroxyvitamin D3: 1,25(OH)2D3. VDR: Vitamin D receptor, RXR: Retinoid X Receptor. CYP27B1: 25-hydroxyvitamin D-1alpha-hydroxylase. Lt: T Limfocite. Mo: Macrophages. N+: Neutrophil. Th: T Limfocite helper. LT CD4: T Limfocite CD4 + . IFN-α, IFN-γ: Interferon α and γ; IL: Interleukin. TNF-α: Tumor Necrosis Factor-α. TGFβ: Transforming growth factor α and β. CCL22: Chemokine (C-C motif) ligand 2. Ang: angiotensin. ACE2: Angiotensin Converting Enzyme 2.
4. Observational studies
4.1. Vitamin D status and COVID-19
D’Avolio et al. investigated the relationship between plasma 25(OH)D3 and the results of SARS-CoV-2 tests (polymerase chain reaction: PCR) showing significantly lower 25(OH)D3 levels in individuals with positive PCR compared to individuals with negative PCR (11 vs 24.6 ng/mL). Additionally, stratifying by age, people older than 70 years with positive PCR also showed significantly lower 25(OH)D3 compared to younger [73]. A study involving elderly showed that the majority had hypovitaminosis D (10–20 ng/mL) and more than half showed an important vitamin D deficiency (<10 ng/mL) that was correlated with inflammatory parameters [74]. In a study involving 56 patients admitted with COVID-19, in which all participants had data on 25(OH)D3 status in the 6 months prior to COVID-19 infection, it was found lower lymphocyte counts and higher C-reactive protein levels in patients with vitamin D deficiency (<20 ng/mL). In addition, the vitamin D status of patients requiring oxygen therapy was found to be lower than those not requiring oxygen therapy, and even patients un-supplemented with vitamin D in the 6 months prior to COVID-19 infection were more likely to be diagnosed with pneumonia [75]. In this line, Baktash et al. found lower 25(OH)D3 levels in patients over 65 years with COVID-19 compared to a control group of the same age. In the group of patients with vitamin D deficiency (<12 ng/mL), a higher incidence of the need for endotracheal intubation and admission to the ICU was found but not higher rates of mortality [76]. In a United Kingdom study of hospitalized COVID-19-patients, a lower percentage of patients with 25(OH)D3 levels above 20 ng/mL were observed in the group that required endotracheal intubation than in the group that did not (19% vs. 39%, p = 0.02), suggesting that low 25(OH)D3 levels may be associated with an increased risk of mechanical ventilation [77]. Another study showed an association between vitamin D deficiency (<12 ng/mL) and an increased risk of serious COVID-19 outcomes such as intubation or death after adjusting for sex, age, and comorbidities [78].
Additionally, a study in South Korea, showed higher proportion of hospitalized COVID-19 patients presenting 25(OH)D3 deficiency (≤20 ng/mL) compared to the control group (74% vs 43.3%, p = 0.003) [79]. Likewise, 24% of hospitalized patients showed severe 25(OH)D3 deficiency (≤10 ng/mL) compared to 7.3% of the controls. Accordingly, in a study including COVID-19 hospitalized patients, a rate of vitamin D deficiency (<30 ng/mL) was described in 75% of the patients, among which 85% required ICU care. A study including 216 patients and 197 controls, demonstrated that COVID-19 hospitalized patients had lower 25(OH)D3 (13.8 ± 7.2 ng/mL vs 20.9 ± 7.4 ng/mL) and a higher prevalence of vitamin D deficiency (<20 ng/ml) (82.2% vs 47.2%) [80]. However, the authors found no relationship between vitamin D concentrations or deficiency and the severity of infection.
An ecological study strengthened the hypothesis that 25(OH)D3 levels could correlate with the acquisition and severity of COVID-19. A negative correlation was observed between 25(OH)D3 levels (deficiency <10 ng/mL) in each of the 20 European countries studied (mean 22 ng/mL ± 4.24 ng/mL SD) and the number of COVID-19 cases (mean 295.5 ± 298.7 SD) and mortality per million population (mean 5.96 ± 15.13 SD). At the same time, 25(OH)D3 levels were markedly lower in older population, especially in Spain, Italy, and Switzerland [81]. The analysis of Lanham-New et al. assessing the relationship between COVID-19 severity and vitamin D status in Europe showed the highest rates of infection and death in the most vitamin D-deficient countries (<10 ng/mL) [82]. In the same line, Marik et al. found a higher COVID-19 case fatality rate in northern USA compared with the southern states [83]. Similarly, in a study involving 88 countries, a correlation analysis revealed that countries close to the equator had fewer COVID-19 deaths compared to distant countries, suggesting a possible relationship between vitamin D and COVID-19 mortality related to approximation to sunlight exposure and latitude [84].
By contrast, Alguwaihes and colleagues found no relationship between 25(OH)D3 deficiency (<20 ng/mL) the risk of SARS-CoV-2 infection in COVID19 hospitalized patients (222 SARS-CoV-2 (+) vs 72 SARS-CoV-2 (-)). However, they found that severe 25(OH)D3 deficiency (<5 ng/mL) appears to increase the risk of mortality [85]. Data from a British biobank found no differences between 25(OH)D3 concentrations (deficiency <10 ng/mL) and COVID-19 after adjustment for confounders, suggesting no association between 25(OH)D3 levels and the risk SARS-CoV-2 infection [86]. In this line, a recent study evaluating 148 COVID-19 infected patients found no significant differences in serum concentrations of 25(OH)D33, 25(OH)D32, 24,25(OH)2D33 or 25,26(OH)2D3 between 23% of patients who died within 30 days of admission and survivors [87].
The meta-analysis by Pereira M et al. is partially consistent with the results described above. It included studies that measured serum 25(OH)D3 levels in adults with COVID-19, with the main variable being the prevalence of 25(OH)D3 deficiency (<20 ng/mL) in severe cases. The results found that vitamin D was not related to the probability of COVID-19 but was related to the severity of the disease, especially in the elderly subpopulation. As a limitation, the included studies did not stratify by sex, and many of the data collected from the hospital samples were taken from secondary sources [88]. Accordingly, a recent systematic review and meta-analysis concluded that there may be a trend toward an association between low serum 25(OH)D3 levels and COVID-19-related health outcomes (ICU admission, invasive and noninvasive ventilation, mortality, hospitalization, hospital stay duration and disease severity), although no statistical significance was found [89].
All these findings are summarized in Table 1.
Table 1.
Summary of studies that have assessed the relationship between vitamin D and COVID-19
| Study/Year/Country | Study design | Sample size | Main findings |
|---|---|---|---|
| Meltzer et al., 2021, USA [3] | Retrospective cohort study | 4638 | COVID-19 risk increased among Black individuals with vitamin D level less than 40 ng/mL compared with those with 40 ng/mL or greater and decreased with increasing levels among individuals with levels greater than 30 ng/mL. No significant associations were noted for White individuals. |
| Cozier et al, 2021, USA [6] | Retrospective cohort study | 1974 | U.S. Black women with lower levels of 25(OH)D3 are at increased risk of infection with COVID-19. |
| D’Avolio et al., 2020, Switzerland [73] | Retrospective cohort study | 107 | Lower levels of 25(OH)D3 were found in SARS-CoV2 positive individuals. |
| Boccardi et al., 2019, Italy [74] | Retrospective cohort study | 237 | In very elderly hospitalized subjects, lower serum 25(OH)D3 levels were found to be associated with a higher comorbidity burden. In addition, vitamin D deficiency correlated with a higher inflammatory status which, together with comorbidity burden, negatively influenced the length of hospital stay. |
| Ünsal et al., 2021,Turkey [75] | Retrospective cohort study | 56 | Significantly lower lymphocyte counts and higher levels of inflammatory markers were found in patients with COVID-19 and vitamin D deficiency. |
| Panagiotou et al., 2020, UK [77] | Retrospective cohort study | 134 | When comparing patients who required admission to ICU versus those who were treated in medical wards, a higher prevalence of vitamin D deficiency was observed in the former. |
| Radujkovic et al.,2020, Germany [78] | Retrospective cohort study | 185 | An increased risk of invasive mechanical ventilation or death was associated with vitamin D deficiency. |
| Im et al, 2020, South Korea [79] | Retrospective cohort study | 50 | Selenium or vitamin D deficiency has been linked to a possible decrease in immune defenses against COVID-19 and to the progression of severe disease. |
| Hernández et al., 2021, Spain [80] | Retrospective case-control study | 413 | 25(OH)D3 levels are lower in hospitalized COVID-19 patients than in population-based controls and these patients had a higher prevalence of deficiency. They did not find any relationship between vitamin D concentrations or vitamin deficiency and the severity of the disease. |
| Alguwaihes et al., 2021, Saudi Arabia. [85]. | Retrospective cohort study | 222 | In hospital settings, 25(OH)D3 deficiency is not associated with SARS-CoV2 infection but may increase risk for mortality in severely deficient cases. |
| Hastie et al, 2020, UK [86]. | Retrospective cohort study | 502,624 | There is no potential relationship between the risk of COVID-19 infection and circulating vitamin D levels nor can ethnic differences in COVID-19 infection be explained by vitamin D concentrations. |
| Zelzer at, 2021, Austria [87] | Retrospective cohort study | 148 | Vitamin D levels did not influence the course and outcome of COVID-19. |
| Baktash et al., 2021,UK [76] | Cohort study | 105 | The occurrence of cytokine storms and the need for more care was associated with vitamin D deficiency. |
| Fasano et al., 2020,Italy [101] | Cohort study | 1486 | The incidence of COVID-19 in patients with Parkinson’s disease was higher in those younger, obese and COPD. In patients taking vitamin D supplements it was less frequent. |
| Ilie et al., 2019, Europe [81] | Ecological study | Vitamin D levels are severely low in the aging population especially in Spain, Italy and Switzerland. This is also the most vulnerable group of the population in relation to COVID-19. | |
| Lanham-New et al.,2020, UK, Ireland, USA [82] | It assessed the relationship between COVID-19 severity and vitamin D status. In Europe, it showed the highest rates of infection and death in the most vitamin D-deficient countries. | ||
| Marik et al., 2020, USA [83] | Higher COVID-19 case fatality rate in northern USA compared with the southern states. | ||
| Whittemore et al.,2020 [84] | Correlation analysis of data | 88 countries | Fewer COVID-19 deaths were observed in countries closer to the equator than those further away, with 16% of this effect attributed to latitude. |
| Mukherjee et al., 2021 [90] | Review | 61 studies | Male patients tend to have higher severity and mortality rates, although current evidence does not suggest a sex or gender difference in SARS-CoV-2 infection. |
| Grant et al., 2020 [93] | Review | 157 studies | Any comorbidity or circumstance that directly or indirectly decreases 25(OH)D3 could increase susceptibility and severity of COVID-19, summarizing that the presence of diseases associated with vitamin D deficiency, such as diabetes or hypertension, would significantly increase the case fatality rate of SARS-CoV2. |
| Griffin et al., 2020 [96] | Review | 91 studies | Evidence linking vitamin D deficiency with COVID-19 severity is circumstantial but considerable, including associations with age, obesity, ethnicity and institutionalization; latitudinal association; preliminary reports of associations with COVID-19 severity in hospitalized patients; and basic biological studies reporting the impact that vitamin D exert on the immune system. |
| Pereira et al.,2020 [88] | Systematic review and meta-analysis | 21 studies | The correlation between vitamin D deficiency and severely ill COVID-19 patients is possibly explained by the fact that 25(OH)D3 is inversely associated with pro-inflammatory cytokines, such as IL-6, increased CRP and heart failure, which are related to the severity of COVID-19 and to its unfavorable outcome. Despite the correlation between higher vitamin D levels, immune defenses and favorable prognosis in other viral infections, the use of vitamin D blood tests and analysis in SARS-CoV2 patients has not shown a clear causal relationship between vulnerability to COVID-19 infection and 25(OH)D3 deficiency. |
| Bassatne et al., 2021 [89] | Systematic review and meta-analysis | 31 studies | There appears to be a trend toward an association between vitamin D deficiency and COVID-19-related health outcomes although not statistically significant. |
| Munshi et al.,2021 [97] | Systematic review and meta-analysis | 6 retrospective articles | Vitamin D levels could act as an useful prognostic estimator for COVID-19 outcomes. |
| Ohaegbulam et al.,2020, USA [104] | Clinical case series | 4 | Faster recovery, based on reduced inflammatory markers, comparatively reduced oxygen requirements and reduced length of hospital stay, was observed in patients receiving high doses of cholecalciferol or ergocalciferol supplementation. |
| Oristrell et al., 2021, Catalonia-Spain [105] | Population based cohort study | 108,343 | An increased incidence of SARS-CoV2 infection was associated with reduced serum 25(OH)D3 levels but not with mortality due to disease severity in un-supplemented patients with vitamin D deficiency compared to patients with sufficient vitamin D supplementation. |
| Nogues et al., 2021, Spain [106] | Observational study | 838 | Treatment with calcifediol significantly reduced mortality and ICU admission in COVID-19 hospitalized patients. |
| Tan et al., 2020, Singapore [102] | Cohort Study | 43 | On admission, patients with 1000 IU/d oral vitamin D3, 150 mg/d oral magnesium and 500 mcg/d oral vitamin B12 not required oxygen therapy. Patients receiving DMB showed significantly less deterioration to the point of requiring oxygen therapy or intensive care support after correction for demographics and hypertension. |
| Ye et al. 2020, China [113] | Case-control study | 142 | Elderly and people with comorbidities were susceptible to severe COVID-19 infection. Vitamin D deficiency was a risk factor for COVID-19, especially for severe/critical cases. |
| Alcala-Diaz et al.,2021, Spain [107] | Retrospective, multicentre, non-randomized cohort study | 537 | After diagnosis of COVID-19, treatment with calcifediol was significantly associated with a reduction in 30-day mortality. |
| Annweiler et al.,2020, France [100] | Quasi-experimental study | 77 | At three months follow-up, an improvement in mortality was found in elderly patients with regular vitamin D supplementation prior to COVID-19. |
| Lakkireddy et al., 2021, India [118] | Prospective randomized, open-label study | 87 | Levels of C-reactive protein, lactate dehydrogenase, ferritin, IL-6 and neutrophil-to-lymphocyte ratio showed significant improvement in patients who received a daily 60,000 IU vitamin D supplement for eight days compared to patients who did not receive supplementation. |
| Entrenas-Castillo et al., 2020, Spain [103] | Parallel pilot randomized open-label trial | 76 | A reduction in the severity of COVID-19 in patients requiring ICU admission was found with the administration of high-dose calcifediol. |
| Murai et al., 2021, Brazil [119] | Multi-center parallel double-blind RCT | 240 | There was no evidence of a reduction in hospital stay, mortality, ICU admission or need for ventilation in the group receiving a single dose of cholecalciferol compared to the group receiving placebo. |
4.2. Common risk factors for hypovitaminosis D and COVID-19 severity
Certain studies have supported the hypothesis of a relationship between race and COVID-19. Meltzer et al. found an inverse association between the percentage of COVID-19 positivity and 25(OH)D3 levels only in black subjects suggesting that lower 25(OH)D3 levels are related to a higher risk of infection in this population (insufficiency 20 to <30 ng/mL and deficiency <20 ng/mL) [3]. Consistently, a recent study including 1,974 COVID-19 women suggested that US black women with lower levels of 25(OH)D3 (<20 ng/mL) are at increased risk of COVID-19 infection [6]. However, the description of gender differences in COVID-19 as a result of ACE2 or vitamin D could be simplistic. Individual differences could be attributed to fewer morbidities, associated with the protective effect that estrogens confer in cardiovascular and immunological health [90,91]. Certain polymorphisms in the VDR have also been associated with increased susceptibility to viral infections [51]. However, polymorphisms have not only been found in the VDR but also in DBP. A study aimed to evaluate whether there was any association between the DBP gene polymorphism at loci rs7041 and rs4588 and the prevalence of COVID-19 and its mortality rates among populations from 10 countries revealed a positive association between COVID-19 prevalence and mortality rate with the GT genotype along with a negative correlation related to the TT genotype. These findings suggested that the GT genotype can confer increased susceptibility to COVID-19 in certain populations, such as Germans, Mexicans, Czechs, and Turks [92].
In the review by Grant et al. concluded that any comorbidity or circumstance that directly or indirectly decreases 25(OH)D3 could increase susceptibility and severity of COVID-19, summarizing that the presence of diseases associated with vitamin D deficiency, such as diabetes or hypertension, would significantly increase the case fatality rate of SARS-CoV-2 [93]. This would explain, at least partially, why mortality from COVID-19 is higher in hypertensive, diabetic, and heart-failure patients [94,95]. The reason for the increase in COVID-19 case fatality rate with age could be doubly explained by the increased incidence of chronic diseases related to aging and by the fact that advanced age generally leads to a decrease in the active form of vitamin D [93].
In a review by Griffin et al. concluded that the evidence linking vitamin D deficiency (<12 ng/mL) with COVID-19 severity is circumstantial but considerable, including associations with age, obesity, ethnicity and institutionalization; latitudinal association; preliminary reports of associations with COVID-19 severity in hospitalized patients; and basic biological studies reporting the impact that vitamin D exert on the immune system [96].
A meta-analysis attempted to demonstrate that COVID-19 severity is not related to the presence of comorbidities or age but instead to 25(OH)D3 levels. The results demonstrated that patients with a worse prognosis (defined by ICU admission, ARDS, need for mechanical ventilation, or death) presented lower 25(OH)D3 levels. Conclusions were drawn while considering the confounding factors of age, sex, and comorbidities (diabetes, heart failure, hypertension, and COPD), none of which had a statistically significant causal relationship with COVID-19 prognosis. The authors concluded that vitamin D appears to have an independent causal role in the disease’s severity [97]. However, since most of the studies analyzed were retrospective, it remains difficult to demonstrate that vitamin D deficiency in ICU patients due to COVID-19 is the real cause of patient admission.
Nevertheless, the clinical data available on observational studies have been merely preliminary, and many articles are retrospective, descriptive, or only associative. Thus, the information in such studies should be interpreted with caution. This information, although not unanimous, suggests that vitamin D may have an influential role in the likelihood of COVID-19. The putative mechanisms underlying the role of vitamin D and of its non-calcemic hydroxyderivatives in immunity and actions non-skeletal- related such as antiinflamatory and antioxidant functions, would provide support for the hypothesis that vitamin D deficiency is a risk factor for the disease. What is clearer, however, is the higher prevalence of vitamin D deficiency among the more severe forms of COVID-19 based on the most recent and consistent studies [88].
Although the current evidence suggests an inverse relationship between 25(OH)D3 levels and COVID-19 severity, further research is needed to better understand the relationship between vitamin D and COVID-19 (Table 1).
5. Interventional studies
5.1. Vitamin D supplementation studies in COVID-19
Studies in critically ill patients without COVID-19 support the potential therapeutic applications of vitamin D, so that vitamin D supplementation has been shown to attenuate acute organ dysfunction secondary to infections, including acute lung injury in children and adults [12,13] and in adults [98,99]. It is therefore thought that vitamin D supplementation may play a role in the acquisition and/or evolution of COVID-19.
Several quasi-experimental case/control studies and a pilot study of a cohort of COVID-19 patients with pneumonia have been published. Although the sample sizes in these studies were small (except for one), some favorable results were obtained [67,80,100–104]. However, these studies have limitations. These studies include no data on the baseline or final 25(OH)D3 values, although all of them assess important outcome variables, such as disease incidence and mortality. In a case-control study by Hernandez JL et al, patients with COVID-19 supplemented with different doses of vitamin D (cholecalciferol, 25,000 IU/monthly or 5600 IU/weekly, and calcifediol, 266 ug/monthly) had a slightly less unfavorable outcome than those not supplemented [80]. However, the low number of supplemented patients (n = 19) compared to unsupplemented (n = 197) and the lack of extrapolation of the results to other ethnicities or countries prevented the authors from drawing solid conclusions in this regard.
A small study conducted in a nursing home showed a reduction in the COVID-19 mortality rate among supplemented patients (80,000 IU vitamin D3) during the week following suspicion or diagnosis of COVID-19 or in the preceding month in comparison with un-supplemented group (17.5% vs. 55.6%; p = 0.023) [100].
In a retrospective study, the aim was to analyze the associations between cholecalciferol or calcifediol supplementation, serum 25(OH)D3 levels and COVID-19 scores in a large population in Barcelona (Spain). Cholecalciferol supplementation (dose equivalent to 400 IU/day) was associated with a slight protection against SARS-CoV2 infection compared to controls. However, the use of calcifediol (bolus >250 μg) was not associated with a lower risk of SARS-CoV2 infection or mortality. Nevertheless, patients supplemented with cholecalciferol or calcifediol who achieved 25(OH)D3 levels ≥30 ng/mL had lower risk of SARS-CoV2 infection, lower risk of severe COVID-19 and lower mortality than non-supplemented 25(OH)D3-deficient patients (insufficiency 20 to <30 ng/mL and deficiency <20 ng/mL) [105].
The study of Ohaegbulam KC et al. observed better outcomes associated with higher doses of vitamin D (50,000 IU ergocalciferol daily for 5 days vs 1000 IU cholecalciferol daily). Supplemented patients who were able to normalize their 25(OH)D3 levels (>30 ng/mL) improved their clinical recovery: shorter hospital stays, lower oxygen requirements, and a reduction in inflammatory markers [104]. Likewise, an observational study including COVID-19 patients in a hospital in Spain has attempted to elucidate the effect of calcifediol treatment on COVID-19-related outcomes. The results have shown the beneficial role of supplementation with calcifediol (532ug on day 1, plus 266ug on days 3,7,15 and 30) at the beginning of hospital admission both in terms of severity and mortality associated with COVID-19. The authors reported a decrease in the percentage of patients who required ICU care (4.7% vs. 15.9%) and in the COVID-19 mortality risk (OR = 0.21 vs 0.52) in supplemented patients vs unsupplemented [106]. In agreement with these results, another retrospective study concluded that the supplementation with calcifediol immediately after admission (2 capsules of 0.266 mg on entry and one capsule on day 3, 7, 14, 21, and 28; n = 79) is associated with significant lower mortality rate in COVID-19 hospitalized patients during the first 30 days compared with those unsupplemented (5% vs 20%) [107]. In the meta-analysis and systematic review by Bassatne et al, it was concluded that the supplementation with calcifediol could have a protective effect on COVID-19-related ICU admissions. However, they infer that the current use of high-dose vitamin D in patients with COVID-19 is not based on solid evidence [89].
On the other hand, SARS-CoV2 infection triggers massive ROS production and oxidative damage. Jain et al. have proposed that co-optimizing the redox status of impaired glutathione and excess 25(OH)D3 deficiencies has the potential to reduce oxidative stress, boost immunity and reduce the adverse clinical effects of COVID-19 infection [46]. Studies in mice revealed that the administration of vitamin D alone compared to co-supplementation of vitamin D and L-cysteine (a glutathione precursor) showed greater efficacy in improving glutathione levels and vitamin D regulatory genes at the cellular/tissue level, increasing 25(OH)D3 levels, and reducing biomarkers of inflammation in the blood. Thus, Jain et al. suggest that combined vitamin D and glutathione administration could be useful for COVID-19 treatment [108]. Accordingly, several studies have shown that the supplementation with vitamin D combined with L-cysteine may be more successful in increasing both glutathione and vitamin D metabolism genes compared to vitamin D supplementation alone. Therefore, this supplementation form could constitute an effective strategy for the treatment of vitamin D deficiency related to conditions at risk for severe COVID-19, such as diseases associated with insulin resistance like type 2 diabetes [44,45,109,110]. Based on these evidences, the combined supplementation with vitamin D and L-cysteine seems to be more potent and promising in providing hope for treating COVID-19.
These findings agree with the hypothesis that in a state of hypovitaminosis D, there is an increase in inflammatory parameters with a determining influence on the prognosis, severity, and development of ARDS and the appearance of disseminated intravascular coagulation associated with COVID-19 [111,112]. On the other hand, the reported stimulatory effect attributed to CYP11A1-generated hydroxyderivatives of vitamin D3 besides to their anti-inflammatory and antioxidant effects that are shared by calcitriol suggests that a direct delivery of these metabolites deserves consideration as a therapeutic strategy for ARDS associated with COVID-19 [56,72].
To date, the optimal vitamin D threshold for the prevention or treatment of COVID-19 and the doses that should be used remain unknown. The study performed in China, placed this threshold at 16.5 ng/mL [113]. It has also been postulated that levels above 20 ng/mL and preferably above 30 ng/mL seem acceptable [114]. Furthermore, there is speculation on the best dose to achieve vitamin D sufficiency levels. A high amount of 300,000 IU of vitamin D3 in a single dose may be adequate and was already shown in clinical practice to be safe [115]. However, a review by Griffin et al. concluded that daily supplementation is a better alternative to high-dose intermittent boluses, suggesting 800–1000 IU of vitamin D3 per day as the optimal dose to prevent vitamin D deficiency [96]. However, the upper daily limit established by the Endocrine Society for healthy adults is 10,000 IU/day. This dose could be administered orally preventively to improve the prognosis of COVID-19. However, for COVID-19 hospitalized patients this dose is probably not sufficient and higher doses are required as reported by several past studies treating different pathologies [116]. Similarly, recent studies have shown that 200,000 IU of orally delivered vitamin D3 was an effective strategy to attenuate proinflammatory responses without signs of toxicity [117]. Although these data suggest that high doses of vitamin D could provide a benefit for COVID-19 hospitalized patients [56], further studies are needed to establish the optimal dose of vitamin D as well as the type, form and strategy of vitamin D supplementation to achieve beneficial effects on the infection and course of COVID-19 since delivery routes could influence final outcomes [72]. (Table 1).
5.2. Clinical trials: vitamin D COVID-19
Despite the evidence supported by these findings, the lack of controlled trials to determine the role of vitamin D supplementation in protection against COVID-19 has been noted. Currently, more than 30 relevant clinical trials are registered on clinicaltrials.gov.
A randomized clinical trial by Entrenas M et al. demonstrated that administration of high-dose calcifediol or cholecalciferol reduced the need for ICU admission among hospitalized COVID-19 patients compared to unsupplemented patients. This result could be because the treatment improved the severity of the disease [103]. A prospective, randomized, open-label study involving COVID-19 87 patients having hypovitaminosis D found that besides to standard care, vitamin D supplementation significantly improved inflammatory markers [118]. Levels of C-reactive protein, lactate dehydrogenase, ferritin, IL-6 and neutrophil-to-lymphocyte ratio showed significant improvement in patients who received a daily 60,000 IU vitamin D supplement for eight days compared to patients who did not receive supplementation. By contrast, the multicenter, double-blind, randomized, placebo-controlled trial conducted by Murai et al. in Brazil found no association between increased 25(OH)D3 levels derived from the administration of a single high dose of vitamin D3 (200,000 IU) and reduction in hospital stay in COVID-19 hospitalized patients compared to placebo group. Although, these findings do not support the use of a high dose of vitamin D3 for the treatment of moderate to severe COVID-19, it should be taken into account that the supplemented patients were moderately to severely ill in whom the cytokine storm could have been triggered [119]. Therefore, the results should be interpreted with caution considering the possibility that earlier supplementation strategies could be associated with better outcomes in COVID-19 outcomes as described in recent observational studies [106,107]. (Table 1).
The CARED-TRIAL (NCT04411446), a multicenter, randomized, double-blind, sequential clinical trial comparing the administration of high-dose vitamin D with placebo, has begun. They will evaluate the effects of vitamin D supplementation on blood oxygenation (physiological effects), the effects on clinical outcome and to determine whether vitamin D administration is useful in the prevention of COVID-19 [120]. A clinical trial is also underway in the UK: CORONAVIT (NCT04579640) comparing a dose of 800 IU/day with 3200 IU/day, currently being the recommendation of the UK government of 400 IU/day. In addition, more clinical trials are underway that will allow us to understand more precisely the role of vitamin D supplementation in COVID-19.
However, current literature reports that clinical trials of vitamin D supplementation are not entirely successful and show discordant results both in COVID-19 and in non-COVID-19 subjects [121,122]. Although randomized controlled trials are commonly accepted as a ‘gold standard’ for assessing the efficacy of new forms of treatment, have often failed to provide supporting evidence for the expected health benefits of vitamin D supplementation. This may be because clinical trials have used designs developed to test drugs, whereas vitamin D is a nutrient. In addition, vitamin D absorption varies with factors such as age or excess weight, which could also affect the results of clinical trials. Furthermore, it should be stressed that there are no controlled trials of sufficient duration to test whether achieving long-term repletion can and will reduce the risks of the very chronic conditions that it can aggravate, such as cardiovascular disease, as leaving controls deficient in the long term is unethical. Finally, there is no consensus on which vitamin D levels are recommended to be achieved, so this could be another factor causing heterogeneity in clinical trials [123]. All these insights are essential to identify health benefits from existing randomized controlled trial data and to improve the design of future randomized controlled trials.
5.3. Guidelines: vitamin D supplementation and COVID-19
An update was published by the National Institute for Health and Care Excellence (NICE): ‘COVID-19 rapid guideline: vitamin D’ that recommended vitamin D supplementation for people in confined spaces, those living in residences, with low sun exposure, or spending more time at home due to the COVID-19 pandemic [124]. The recommended dose is 400 IU/day of cholecalciferol. The recommended period is between October and March (indicated all year round for people without sun exposure during spring and summer). The objective proposed by the authors was to achieve levels of 25(OH)D3 > 10 ng/mL to maintain optimal bone health. The authors indicated that low 25(OH)D3 levels are associated with more severe COVID-19 outcomes. However, they also cautioned that vitamin D supplementation should not be administered solely for the purpose of preventing or treating COVID-19, as clinical trials are necessary to give this recommendation. The US National Academy of Medicine and the European Food Safety Authority recommend achieving 25(OH)D3 levels at least 20 ng/mL. For this purpose, it was indicated that supplementation should be done at 800 IU/day. In addition, in the case of hospital admissions, it is recommended that 25(OH)D3 levels be determined on admission [125].
6. Conclusions and perspectives
Currently, the relationship between vitamin D and COVID 19 is not fully established although it has biological plausibility. The studies available to date are not conclusive. The presence of biases in several studies such as the lack of stratification by sex, or the lack 25(OH)D3 levels assessment at the time of COVID-19 diagnosis, makes it difficult to detect causality, and if causality is found, its reliability must still be questioned until it is confirmed via randomized controlled clinical trials with a large sample size featuring different age groups, population groups, and climatological/geographical environments. This reality precludes drawing population-based conclusions beyond those found in the samples. However, there is clearly biological plausibility and consistency in the findings to date, and the immunomodulatory function of vitamin D and its hydroxymetabolites has already been demonstrated on many occasions in the scientific literature. Therefore, vitamin D could influence the acquisition of COVID-19, and, in some cases, a positive association between vitamin D deficiency and SARS-CoV-2 infection has been detected. In addition, vitamin D has been related to factors that molecularly, genetically, and immunologically influence the likelihood of becoming infected with SARS-CoV-2, including sex, country of origin, DBP, VDR, and ACE2 levels. Vitamin D deficiency is a common problem, many times related to unhealthy lifestyles, and vitamin D seems clearly to intervene favorably in the evolution and decrease in severity of COVID-19. So, vitamin D has been shown to play a prognostic role in COVID-19 infected patients, especially in severe cases and the elderly. Therefore, vitamin D administration could be considered as a measure to be implemented in countries with a high prevalence of vitamin D deficiency to improve the clinical outcomes of patients with severe COVID-19. Also, it could be useful to promote healthy lifestyles as stimulation of physical activity in high risk population as preventive tool against COVID-19 infection or in order to reduce the severity of symptomatology in case of infection.
In addition, 25(OH)D3 levels should be tested in people admitted with COVID-19, and supplementation should be considered in case of deficiency based on studies clarifying the optimal form of supplementation and establishing the levels of 25OD that should be achieved to improve COVID-19 outcomes.
Further studies and clinical trials should be conducted to confirm or rule out the relationship between vitamin D and the likelihood of acquiring COVID-19 infection, the disease’s severity, and the possibility of preventing or improving the disease through vitamin D supplementation as well as to clarify the optimal form of supplementation and establish the 25OD levels that should be achieved to improve COVID-19 outcomes.
Funding Statement
This research was funded by Instituto de Salud Carlos III grants (PI18-00803 and PI18-01235), co-funded by the European Regional Development Fund (FEDER) and by Junta de Andalucía grant (PI-0268-2019). In addition, V. C-B and C.G-F are funded by postdoctoral fellowships from the Junta de Andalucía and Instituto de Salud Carlos III respectively (RH-0141-2020; CD20/00022)
Disclosure statement
The authors have no relevant conflicts of interest to disclose. Peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
References
- 1.Costa de Lucena TMC, Da Silva Santos AF, de Lima BR, et al. Mechanism of inflammatory response in associated comorbidities in COVID-19. Diabetes Metab Syndr Clin Res Rev. 2020;14(4):597–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mathur R, Rentsch CT, Morton CE, et al. Ethnic differences in SARS-CoV-2 infection and COVID-19-related hospitalisation, intensive care unit admission, and death in 17 million adults in England: an observational cohort study using the OpenSAFELY platform. Lancet. 2021;397(10286):1711–1724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Meltzer DO, Best TJ, Zhang H, et al. Association of vitamin D levels, race/ ethnicity, and clinical characteristics with COVID-19 test results. AMA Netw Open. 2021;4(3):1–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mauvais-Jarvis F. Aging, male sex, obesity, and metabolic inflammation create the perfect storm for COVID-19. Diabetes. 2020;69(9):1857–1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tal Y, Adini A, Eran A, et al. Racial disparity in Covid-19 mortality rates - A plausible explanation. Clin Immunol. 2020;217:108481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cozier YC, Castro-Webb N, Hochberg NS, et al. Lower serum 25(OH)D levels associated with higher risk of COVID-19 infection in U.S. Black women. Laganà AS, editor. PLOS ONE. 2021;16(7):e0255132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.García LF. Immune response, inflammation, and the clinical spectrum of COVID-19. Front Immunol. 2020;11:1441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Weiskopf D, Schmitz KS, Raadsen MP, et al. Phenotype and kinetics of SARS-CoV-2-specific T cells in COVID-19 patients with acute respiratory distress syndrome. Sci Immunol. 2020;5(48):eabd2071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cannell JJ, Vieth R, Umhau JC, et al. Epidemic influenza and vitamin D. Epidemiol Infect. 2006;134(6):1129–1140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vo P, Koppel C, Espinola JA, et al. Vitamin D status at the time of hospitalization for bronchiolitis and its association with disease severity. J Pediatr. 2018;203:416–422.e1. [DOI] [PubMed] [Google Scholar]
- 11.Monlezun D, Bittner E, Christopher K, et al. Vitamin D status and acute respiratory infection: cross sectional results from the United States National Health And Nutrition Examination Survey, 2001–2006. Nutrients. 2015;7(3):1933–1944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Martineau AR, Jolliffe DA, Greenberg L, et al. Vitamin D supplementation to prevent acute respiratory infections: individual participant data meta-analysis. Health Technol Assess. 2019;23(2):1–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jolliffe DA, Camargo CA, Sluyter JD, et al. Vitamin D supplementation to prevent acute respiratory infections: a systematic review and meta-analysis of aggregate data from randomised controlled trials. Lancet Diabetes Endocrinol. 2021;9(5):276–292. [DOI] [PubMed] [Google Scholar]
- 14.Dancer RCA, Parekh D, Lax S, et al. Vitamin D deficiency contributes directly to the acute respiratory distress syndrome (ARDS). Thorax. 2015;70(7):617–624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Qu G, Li X, Hu L, et al. An imperative need for research on the role of environmental factors in transmission of novel Coronavirus (COVID-19). Environ Sci Technol. 2020;54(7):3730–3732. [DOI] [PubMed] [Google Scholar]
- 16.Qayyum S, Mohammad T, Slominski RM, et al. Vitamin D and lumisterol novel metabolites can inhibit SARS-CoV-2 replication machinery enzymes. Am J Physiol - Endocrinol Metab. 2021;321(2):E246–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Song Y, Qayyum S, Greer RA, et al. Vitamin D3 and its hydroxyderivatives as promising drugs against COVID-19: a computational study. J Biomol Struct Dyn. 2021;1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bouillon R. Vitamin D: from photosynthesis, metabolism and action to clinical applications. In: Jameson JL, De Groot LJ, editors. Endocrinology. 6th ed. Philadelphia: PA: Saunders Elsevier; 2010. p. 1089–1110. [Google Scholar]
- 19.Holick MF. Vitamin D: a millenium perspective. J Cell Biochem. 2003;88(2):296–307. [DOI] [PubMed] [Google Scholar]
- 20.Holick MF. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am J Clin Nutr. 2004;80(6):1678S–1688S. [DOI] [PubMed] [Google Scholar]
- 21.Rosen CJ, Adams JS, Bikle DD, et al. The Nonskeletal effects of vitamin D: an endocrine society scientific statement. Endocr Rev. 2012;33(3):456–492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014;21(3):319–329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hossein-nezhad A, Holick MF. Vitamin D for health: a global perspective. Mayo Clin Proc. 2013;88(7):720–755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Slominski AT, Kim T-K, Shehabi HZ, et al. In vivo evidence for a novel pathway of vitamin D₃ metabolism initiated by P450scc and modified by CYP27B1. FASEB J Off Publ Fed Am Soc Exp Biol. 2012;26(9):3901–3915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Slominski AT, Kim T-K, Shehabi HZ, et al. In vivo production of novel vitamin D2 hydroxy-derivatives by human placentas, epidermal keratinocytes, Caco-2 colon cells and the adrenal gland. Mol Cell Endocrinol. 2014;383(1–2):181–192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Slominski AT, Li W, Kim T-K, et al. Novel activities of CYP11A1 and their potential physiological significance. J Steroid Biochem Mol Biol. 2015;151:25–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Slominski AT, Kim T-K, Li W, et al. Detection of novel CYP11A1-derived secosteroids in the human epidermis and serum and pig adrenal gland. Sci Rep. 2015;5:14875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Slominski AT, Chaiprasongsuk A, Janjetovic Z, et al. Photoprotective properties of vitamin D and Lumisterol Hydroxyderivatives. Cell Biochem Biophys. 2020;78(2):165–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pike JW, Meyer MB. The vitamin D receptor: new paradigms for the regulation of gene expression by 1,25-Dihydroxyvitamin D3. Endocrinol Metab Clin North Am. 2010;39(2):255–269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kato S. The function of vitamin D receptor in vitamin D action. J Biochem (Tokyo). 2000;127(5):717–722. [DOI] [PubMed] [Google Scholar]
- 31.Norman AW. From vitamin D to hormone D: fundamentals of the vitamin D endocrine system essential for good health. Am J Clin Nutr. 2008;88(2):491S–499S. [DOI] [PubMed] [Google Scholar]
- 32.Slominski AT, Kim T-K, Takeda Y, et al. RORα and ROR γ are expressed in human skin and serve as receptors for endogenously produced noncalcemic 20-hydroxy- and 20,23-dihydroxyvitamin D. FASEB J Off Publ Fed Am Soc Exp Biol. 2014;28(7):2775–2789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Slominski AT, Kim T-K, Janjetovic Z, et al. Differential and overlapping effects of 20,23(OH)₂D3 and 1,25(OH)₂D3 on gene expression in human epidermal keratinocytes: identification of AhR as an alternative receptor for 20,23(OH)₂D3. Int J Mol Sci. 2018;19(10):E3072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Feldman D, Krishnan A, Swami, S, et al. Vitamin D. Fourth. Osteoporosis; 2013. Available from: https://www.elsevier.com/books/osteoporosis/marcus/978-0-12-415853-5. [Google Scholar]
- 35.Dong X, Craig T, Xing N, et al. Direct transcriptional regulation of RelB by 1α,25-Dihydroxyvitamin D3 and its analogs. J Biol Chem. 2003;278(49):49378–49385. [DOI] [PubMed] [Google Scholar]
- 36.Griffin MD, Dong X, Kumar R. Vitamin D receptor-mediated suppression of RelB in antigen presenting cells: a paradigm for ligand-augmented negative transcriptional regulation. Arch Biochem Biophys. 2007;460(2):218–226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pinette K, Yee Y, Amegadzie B, et al. Vitamin D receptor as a drug discovery target. Mini-Rev Med Chem. 2003;3(3):193–204. [DOI] [PubMed] [Google Scholar]
- 38.Selvaraj P Vitamins and the immune system. Vitamin D Receptor, and Cathelicidin in the treatment of tuberculosis. Vitamins & Hormones; 2011. [DOI] [PubMed] [Google Scholar]
- 39.Alroy I, Towers TL, Freedman LP. Transcriptional repression of the interleukin-2 gene by vitamin D3: direct inhibition of NFATp/AP-1 complex formation by a nuclear hormone receptor. Mol Cell Biol. 1995;15(10):5789–5799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Harant H, Andrew PJ, Reddy GS, et al. 1alpha,25-Dihydroxyvitamin D3 and a variety of its natural metabolites transcriptionally repress nuclear-factor-kappaB-mediated Interleukin-8 gene expression. Eur J Biochem. 1997;250(1):63–71. [DOI] [PubMed] [Google Scholar]
- 41.Slominski AT, Kim T-K, Qayyum S, et al. Vitamin D and lumisterol derivatives can act on liver X receptors (LXRs). Sci Rep. 2021;11(1):8002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ballatori N, Krance SM, Notenboom S, et al. Glutathione dysregulation and the etiology and progression of human diseases. Biol Chem. 2009;390(3). DOI: 10.1515/BC.2009.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Parsanathan R, Jain SK. Glutathione deficiency induces epigenetic alterations of vitamin D metabolism genes in the livers of high-fat diet-fed obese mice. Sci Rep. 2019;9(1):14784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jain SK, Parsanathan R, Achari AE, et al. Glutathione stimulates vitamin D regulatory and glucose-metabolism genes, lowers oxidative stress and inflammation, and increases 25-hydroxy-vitamin D levels in blood: a novel approach to treat 25-hydroxyvitamin D deficiency. Antioxid Redox Signal. 2018;29(17):1792–1807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jain SK, Micinski D. Vitamin D upregulates glutamate cysteine ligase and glutathione reductase, and GSH formation, and decreases ROS and MCP-1 and IL-8 secretion in high-glucose exposed U937 monocytes. Biochem Biophys Res Commun. 2013;437(1):7–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jain SK, Parsanathan R, Levine SN, et al. The potential link between inherited G6PD deficiency, oxidative stress, and vitamin D deficiency and the racial inequities in mortality associated with COVID-19. Free Radic Biol Med. 2020;161:84–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rezaei R, Aslani S, Marashi M, et al. Immunomodulatory effects of vitamin D in influenza infection. Curr Immunol Rev. 2018;14(1):40–49. [Google Scholar]
- 48.Di Rosa M, Malaguarnera M, Nicoletti F, et al. Vitamin D3: a helpful immuno-modulator: vitamin D3 as immuno-modulator. Immunology. 2011;134(2):123–139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Prietl B, Treiber G, Pieber T, et al. Vitamin D and immune function. Nutrients. 2013;5(7):2502–2521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mohan M, Cherian JJ, Sharma A. Exploring links between vitamin D deficiency and COVID-19. Chowdhary A, editor. PLOS Pathog. 2020;16(9):e1008874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chu C-M. Initial viral load and the outcomes of SARS. Can Med Assoc J. 2004;171(11):1349–1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kohlmeier JE, Cookenham T, Roberts AD, et al. Type I interferons regulate cytolytic activity of memory CD8+ T cells in the lung airways during respiratory virus challenge. Immunity. 2010;33(1):96–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Channappanavar R, Fehr AR, Vijay R, et al. Dysregulated type I interferon and inflammatory monocyte-macrophage responses cause lethal pneumonia in SARS-CoV-infected mice. Cell Host Microbe. 2016;19(2):181–193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhao J, Zhao J, Perlman S. T cell responses are required for protection from clinical disease and for virus clearance in severe acute respiratory syndrome coronavirus-infected mice. J Virol. 2010;84(18):9318–9325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bilezikian JP, Bikle D, Hewison M, et al. Mechanisms in endocrinology: vitamin D and COVID-19. Eur J Endocrinol. 2020;183(5):R133–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Slominski AT, Slominski RM, Goepfert PA, et al. Reply to Jakovac and to Rocha et al.: can vitamin D prevent or manage COVID-19 illness? Am J Physiol - Endocrinol Metab. 2020;319(2):E455–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chaiprasongsuk A, Janjetovic Z, Kim T-K, et al. CYP11A1-derived vitamin D3 products protect against UVB-induced inflammation and promote keratinocytes differentiation. Free Radic Biol Med. 2020;155:87–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Chaiprasongsuk A, Janjetovic Z, Kim T-K, et al. Protective effects of novel derivatives of vitamin D3 and lumisterol against UVB-induced damage in human keratinocytes involve activation of Nrf2 and p53 defense mechanisms. Redox Biol. 2019;24:101206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Jassil NK, Sharma A, Bikle D, et al. Vitamin D binding protein and 25-hydroxyvitamin D levels: emerging clinical applications. Endocr Pract. 2017;23(5):605–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chun RF, Lauridsen AL, Suon L, et al. Vitamin D-binding protein directs monocyte responses to 25-hydroxy- and 1,25-dihydroxyvitamin D. J Clin Endocrinol Metab. 2010;95(7):3368–3376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Walls AC, Park Y-J, Tortorici MA, et al. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181(2):281–292.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Li XC, Zhang J, Zhuo JL. The vasoprotective axes of the renin-angiotensin system: physiological relevance and therapeutic implications in cardiovascular, hypertensive and kidney diseases. Pharmacol Res. 2017;125:21–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Patel VB, Zhong J-C, Grant MB, et al. Role of the ACE2/angiotensin 1–7 axis of the renin–angiotensin system in heart failure. Circ Res. 2016;118(8):1313–1326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Turner AJ, Hiscox JA, Hooper NM. ACE2: from vasopeptidase to SARS virus receptor. Trends Pharmacol Sci. 2004;25(6):291–294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Crackower MA, Sarao R, Oudit GY, et al. Angiotensin-converting enzyme 2 is an essential regulator of heart function. Nature. 2002;417(6891):822–828. [DOI] [PubMed] [Google Scholar]
- 66.Imai Y, Kuba K, Ohto-Nakanishi T, et al. Angiotensin-converting enzyme 2 (ACE2) in disease pathogenesis. Circ J. 2010;74(3):405–410. [DOI] [PubMed] [Google Scholar]
- 67.Annweiler G, Corvaisier M, Gautier J, et al. Vitamin D supplementation associated to better survival in hospitalized frail elderly COVID-19 patients: the GERIA-COVID Quasi-experimental study. Nutrients. 2020;12(11):3377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lee C. Therapeutic modulation of virus-induced oxidative stress via the Nrf2-dependent antioxidative pathway. Oxid Med Cell Longev. 2018;2018:6208067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hanff TC, Harhay MO, Brown TS, et al. Is there an association between COVID-19 mortality and the renin-angiotensin system? A call for epidemiologic investigations. Clin Infect Dis. 2020;71(15):870–874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Xu J, Yang J, Chen J, et al. Vitamin D alleviates lipopolysaccharide-induced acute lung injury via regulation of the renin-angiotensin system. Mol Med Rep. 2017;16(5):7432–7438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Quesada-Gomez JM, Entrenas-Castillo M, Bouillon R. Vitamin D receptor stimulation to reduce acute respiratory distress syndrome (ARDS) in patients with coronavirus SARS-CoV-2 infections. J Steroid Biochem Mol Biol. 2020;202:105719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Slominski RM, Stefan J, Athar M, et al. COVID-19 and Vitamin D: a lesson from the skin. Exp Dermatol. 2020;29(9):885–890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.D’Avolio A, Avataneo V, Manca A, et al. 25-hydroxyvitamin D concentrations are lower in patients with positive PCR for SARS-CoV-2. Nutrients. 2020;12(5):1359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Boccardi V, Lapenna M, Gaggi L, et al. Hypovitaminosis D: a disease marker in hospitalized very old persons at risk of malnutrition. Nutrients. 2019;11(1):128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ünsal YA, Öö G, Cander S, et al. Retrospective analysis of vitamin D status on ınflammatory markers and course of the disease in patients with COVID-19 infection. J Endocrinol Invest. 2021;44(12):2601–2607. s40618-021-01566-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Baktash V, Hosack T, Patel N, et al. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad Med J. 2020:postgradmedj-2020-138712. DOI: 10.1136/postgradmedj-2020-138712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Panagiotou G, Tee SA, Ihsan Y, et al. Low serum 25‐hydroxyvitamin D (25[OH]D) levels in patients hospitalized with COVID‐19 are associated with greater disease severity. Clin Endocrinol (Oxf). 2020;93(4):508–511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Radujkovic A, Hippchen T, Tiwari-Heckler S, et al. Vitamin D deficiency and outcome of COVID-19 patients. Nutrients. 2020;12(9):2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Im JH, Je YS, Baek J, et al. Nutritional status of patients with COVID-19. Int J Infect Dis. 2020;100:390–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hernández JL, Nan D, Fernandez-Ayala M, et al. Vitamin D status in hospitalized patients with SARS-CoV-2 infection. J Clin Endocrinol Metab. 2021;106(3):e1343–53. [DOI] [PubMed] [Google Scholar]
- 81.Ilie PC, Stefanescu S, Smith L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin Exp Res. 2020;32(7):1195–1198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lanham-New SA, Webb AR, Cashman KD, et al. Vitamin D and SARS-CoV-2 virus/COVID-19 disease. BMJ Nutr Prev Health. 2020;3(1):106–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Marik PE, Kory P, Varon J. Does vitamin D status impact mortality from SARS-CoV-2 infection? Med Drug Discov. 2020;6:100041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Whittemore PB. COVID-19 fatalities, latitude, sunlight, and vitamin D. Am J Infect Control. 2020;48(9):1042–1044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Alguwaihes AM, Sabico S, Hasanato R, et al. Severe vitamin D deficiency is not related to SARS-CoV-2 infection but may increase mortality risk in hospitalized adults: a retrospective case–control study in an Arab Gulf country. Aging Clin Exp Res. 2021;33(5):1415–1422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hastie CE, Mackay DF, Ho F, et al. Corrigendum to “Vitamin D concentrations and COVID-19 infection in UK Biobank.” Diabetes Metab Syndr Clin Res Rev. septiembre de 2020;14(5):1315–1316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Zelzer S, Prüller F, Curcic P, et al. Vitamin D metabolites and clinical outcome in hospitalized COVID-19 patients. Nutrients. 2021;13(7):2129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Pereira M, Dantas Damascena A, Galvão Azevedo LM, et al. Vitamin D deficiency aggravates COVID-19: systematic review and meta-analysis. Crit Rev Food Sci Nutr. 2020:1–9. DOI: 10.1080/10408398.2020.1841090. [DOI] [PubMed] [Google Scholar]
- 89.Bassatne A, Basbous M, Chakhtoura M, et al. The link between COVID-19 and vitamin D (VIVID): a systematic review and meta-analysis. Metabolism. 2021;119:154753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Mukherjee S, Pahan K. Is COVID-19 Gender-sensitive? J Neuroimmune Pharmacol. 2021;16(1):38–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Jolliffe DA, Greiller CL, Mein CA, et al. Vitamin D receptor genotype influences risk of upper respiratory infection. Br J Nutr. 2018;120(8):891–900. [DOI] [PubMed] [Google Scholar]
- 92.Karcioglu Batur L, Hekim N. The role of DBP gene polymorphisms in the prevalence of new coronavirus disease 2019 infection and mortality rate. J Med Virol. 2021;93(3):1409–1413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Grant W, Lahore H, McDonnell S, et al. Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients. 2020;12(4):988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Formenti AM, Tecilazich F, Frara S, et al. Body mass index predicts resistance to active vitamin D in patients with hypoparathyroidism. Endocrine. 2019;66(3):699–700. [DOI] [PubMed] [Google Scholar]
- 95.Isaia G, Giorgino R, Rini GB, et al. Prevalence of hypovitaminosis D in elderly women in Italy: clinical consequences and risk factors. Osteoporos Int. 2003;14(7):577–582. [DOI] [PubMed] [Google Scholar]
- 96.Griffin G, Hewison M, Hopkin J, et al. Vitamin D and COVID-19: evidence and recommendations for supplementation. R Soc Open Sci. 2020;7(12):201912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Munshi R, Hussein MH, Toraih EA, et al. Vitamin D insufficiency as a potential culprit in critical COVID-19 patients. J Med Virol. 2021;93(2):733–740. [DOI] [PubMed] [Google Scholar]
- 98.Zhang Y-P, Wan Y-D, Sun T-W, et al. Association between vitamin D deficiency and mortality in critically ill adult patients: a meta-analysis of cohort studies. Crit Care. 2014;18(6):684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Amrein K, Schnedl C, Holl A, et al. Effect of high-dose vitamin D3 on hospital length of stay in critically ill patients with vitamin D deficiency: the VITdAL-ICU randomized clinical trial. JAMA. 2014;312(15):1520. [DOI] [PubMed] [Google Scholar]
- 100.Annweiler C, Hanotte B, Grandin de L’eprevier C, et al. Vitamin D and survival in COVID-19 patients: a quasi-experimental study. J Steroid Biochem Mol Biol. 2020;204:105771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Fasano A, Cereda E, Barichella M, et al. COVID-19 in Parkinson’s disease patients living in Lombardy, Italy. Mov Disord. 2020;35(7):1089–1093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Tan CW, Ho LP, Kalimuddin S, et al. Cohort study to evaluate the effect of vitamin D, magnesium, and vitamin B12 in combination on progression to severe outcomes in older patients with coronavirus (COVID-19). Nutrition. 2020;79-80:111017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Entrenas Castillo M, Entrenas Costa LM, Vaquero Barrios JM, et al. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: a pilot randomized clinical study. J Steroid Biochem Mol Biol. 2020;203:105751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ohaegbulam KC, Swalih M, Patel P, et al. Vitamin D Supplementation in COVID-19 patients: a clinical case series. Am J Ther. 2020;27(5):e485–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Oristrell J, Oliva JC, Casado E, et al. Vitamin D supplementation and COVID-19 risk: a population-based, cohort study. J Endocrinol Invest. 2021:s40618-021-01639-9. DOI: 10.1007/s40618-021-01639-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Nogues X, Ovejero D, Pineda-Moncusí M, et al. Calcifediol treatment and COVID-19–related outcomes. J Clin Endocrinol Metab. 2021;106(10):e4017–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Alcala-Diaz JF, Limia-Perez L, Gomez-Huelgas R, et al. Calcifediol treatment and hospital mortality due to COVID-19: a cohort study. Nutrients. 2021;13(6):1760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Jain SK, Parsanathan R. Can vitamin D and L-cysteine co-supplementation reduce 25(OH)-vitamin D deficiency and the mortality associated with COVID-19 in African Americans? J Am Coll Nutr. 2020;39(8):694–699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Jain SK, Micinski D, Parsanathan R. L-cysteine stimulates the effect of vitamin D on inhibition of oxidative stress, IL-8, and MCP- 1 secretion in high glucose treated monocytes. J Am Coll Nutr. 2021;40(4):327–332. [DOI] [PubMed] [Google Scholar]
- 110.Parsanathan R, Achari AE, Manna P, et al. l-cysteine and vitamin D co-supplementation alleviates markers of musculoskeletal disorders in vitamin D-deficient high-fat diet-fed mice. Nutrients. 2020;12(11):3406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Trovas G, Tournis S. Vitamin D and COVID-19. Hormones. 2021;20(1):207–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Mahmudpour M, Roozbeh J, Keshavarz M, et al. COVID-19 cytokine storm: the anger of inflammation. Cytokine. 2020;133:155151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Ye K, Tang F, Liao X, et al. Does serum vitamin D level affect COVID-19 infection and its severity?-A case-control study. J Am Coll Nutr. 2021;40(8):724–731. [DOI] [PubMed] [Google Scholar]
- 114.Pérez Castrillón J, Casado E, Corral Gudino L, et al. COVID-19 y vitamina D. Documento de posición de la Sociedad Española de Investigación Ósea y del Metabolismo Mineral (SEIOMM). Rev Osteoporos Metab Miner. 2020;12(4):155–159. [Google Scholar]
- 115.Liu G, Hong T, Yang J. A single large dose of vitamin D Could be used as a means of coronavirus disease 2019 prevention and treatment. Drug Des Devel Ther. 2020;14:3429–3434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357(3):266–281. [DOI] [PubMed] [Google Scholar]
- 117.Scott JF, Das LM, Ahsanuddin S, et al. Oral vitamin D rapidly attenuates inflammation from sunburn: an interventional study. J Invest Dermatol. 2017;137(10):2078–2086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Lakkireddy M, Gadiga SG, Malathi RD, et al. Impact of daily high dose oral vitamin D therapy on the inflammatory markers in patients with COVID 19 disease. Sci Rep. 2021;11(1):10641. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 119.Murai IH, Fernandes AL, Sales LP, et al. Effect of a single high dose of vitamin D3 on hospital length of stay in patients with moderate to severe COVID-19: a randomized clinical trial. JAMA. 2021;325(11):1053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Mariani J, Tajer C, Antonietti L, et al. High-dose vitamin D versus placebo to prevent complications in COVID-19 patients: a structured summary of a study protocol for a randomised controlled trial (CARED-TRIAL). Trials. 2021;22(1):111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Maretzke F, Bechthold A, Egert S, et al. Role of vitamin D in preventing and treating selected Extraskeletal diseases—an umbrella review. Nutrients. 2020;12(4):969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Mousa A, Naderpoor N, de Courten MP, et al. Vitamin D supplementation has no effect on insulin sensitivity or secretion in vitamin D–deficient, overweight or obese adults: a randomized placebo-controlled trial. Am J Clin Nutr. 2017:ajcn152736. DOI: 10.3945/ajcn.117.152736. [DOI] [PubMed] [Google Scholar]
- 123.Boucher BJ. Why do so many trials of vitamin D supplementation fail? Endocr Connect. 2020;9(9):R195–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.National Institute for Health and Care Excellence (UK) . COVID-19 rapid guideline: vitamin D. National Institute for Health and Care Excellence: Clinical Guidelines; 2020. [PubMed] [Google Scholar]
- 125.Authority EFS . Vitamin D: EFSA sets dietary reference values. 2016.


