Abstract
Reports of COVID-19-associated mucormycosis have been increasing in frequency since early 2021, particularly among patients with uncontrolled diabetes. Patients with diabetes and hyperglycaemia often have an inflammatory state that could be potentiated by the activation of antiviral immunity to SARS-CoV2, which might favour secondary infections. In this Review, we analysed 80 published and unpublished cases of COVID-19-associated mucormycosis. Uncontrolled diabetes, as well as systemic corticosteroid treatment, were present in most patients with COVID-19-associated mucormycosis, and rhino-orbital cerebral mucormycosis was the most frequent disease. Mortality was high at 49%, which was particularly due to patients with pulmonary or disseminated mucormycosis or cerebral involvement. Furthermore, a substantial proportion of patients who survived had life-changing morbidities (eg, loss of vision in 46% of survivors). Our Review indicates that COVID-19-associated mucormycosis is associated with high morbidity and mortality. Diagnosis of pulmonary mucormycosis is particularly challenging, and might be frequently missed in India.
Introduction
The world is facing a devastating pandemic, caused by SARS-CoV-2. Systemic corticosteroid treatment can reduce mortality in people with the most severe courses of disease but, together with immunological and other clinical factors,1 this treatment can also predispose patients to secondary fungal disease. Although COVID-19-associated pulmonary aspergillosis has been the primary focus in the literature of COVID-19 secondary infections,2, 3 other fungal superinfections, including Candida infections,4 rare mould infections (fusariosis),5, 6 and COVID-19-associated mucormycosis7, 8, 9 are likely to be under-reported.
The risk factors predisposing patients to mucormycosis are uncontrolled diabetes, neutropenia, haematological malignancies, organ transplantation, trauma and burn, and use of immunosuppressants such as corticosteroids.7, 10, 11 Furthermore, diabetes is also a particularly important comorbidity complicating COVID-19 management.12 Although dexamethasone has proven beneficial in some groups of patients with COVID-19 who have been hospitalised, it could increase the risk of developing invasive mould infections.13 Hyperglycaemia occurs in people with undiagnosed or uncontrolled diabetes, but it can also be induced by corticosteroids. Patients with diabetes and hyperglycaemia often have an inflammatory state that elicits constant recruitment and local activation of immune cells, including macrophages and neutrophils, which secrete proinflammatory cytokines and generate persistent inflammation.14 In these patients, the activation of antiviral immunity to SARS-CoV2 could potentiate this inflammatory phenotype, which could favour secondary infections.15
Diagnosis of COVID-19-associated mucormycosis is challenging as the clinical and radiological features of pulmonary and disseminated mucormycosis are non-specific and could overlap with findings thought to be associated with COVID-19, which results in missed or late diagnoses.7 Detailed guidance on how to diagnose mucormycosis7 and when to suspect COVID-19-associated mucormycosis in patients have been published.9 COVID-19-associated mucormycosis can also be mistaken for other angioinvasive fungal infections, particularly with COVID-19-associated pulmonary aspergillosis being the predominant mould disease in COVID-19-associated acute respiratory distress syndrome. The reversed halo sign, predominantly in the peripheral locations of the lung, has been considered to be suggestive of pulmonary mucormycosis in patients with immunodeficiency and useful for the initiation of pre-emptive antifungal therapy;16 however, in patients with COVID-19, it is less specific as it has also been described as one of the potential radiological features of COVID-19.17 Although cavitary lung lesions might be more specific for mould disease in COVID-19 than the reversed halo sign, these lesions are frequently observed in both COVID-19-associated pulmonary aspergillosis and pulmonary COVID-19-associated mucormycosis.2 In the absence of serum antigenic biomarkers and because the availability of PCR testing is low, particularly in low-income and middle-income countries, COVID-19-associated mucormycosis diagnosis is also challenging, with conventional culture and histopathological demonstration of Mucorales being the mainstay of diagnosis, albeit with low sensitivity.
Although case reports on COVID-19-associated mucormycosis continue to emerge, there are still too few reviews and analyses of large case series that compare the disease in different areas of the world.18 The objective of this Review was to describe the epidemiology, risk factors, treatment, and outcome of COVID-19-associated mucormycosis by analysing published and unpublished cases of COVID-19-associated mucormycosis.
Methods
Search strategy and selection criteria
The analysis was initiated by leadership of the European Confederation of Medical Mycology and International Society of Human and Animal Mycoses.
We searched PubMed using the search terms: “(zygomyco* OR mucormycos* OR Mucorales) AND (COVID-19 OR corona OR SARS-CoV-2 OR pandemic)” for articles published between Oct 1, 2019 and April 12, 2021. We also searched the reference lists of all relevant publications for additional case reports. Information on epidemiology (age at diagnosis, sex, and country), underlying conditions, COVID-19 on admission or previously, intensive care unit procedures, corticosteroid use, diagnosis of mucormycosis (radiological findings and mycological evidence), sites of infection (rhino-orbital, pulmonary, and CNS involvement), coinfections, time between COVID-19 and mucormycosis, clinical management (systemic antifungal therapy and surgical procedures), and outcome (overall response, survival, causes of death [where applicable], and follow-up times) were extracted from the literature by DS or PLW and verified in a second review of the articles by the other reviewer. Reports of new cases were provided via secure data transfer or through documentation in the FungiScope registry (a global emerging fungal infection registry). Cases were reviewed by the FungiScope team and questions were resolved with the respective contributor.
We identified patients with mucormycosis during or within 12 weeks after diagnosis of SARS-CoV-2 infection from the literature and through a global reach-out via email and social media within FungiScope, European Confederation of Medical Mycology, and International Society for Human and Animal Mycology networks during March and April, 2020.
Cases were classified according to the updated criteria of the European Organisation for Research and Treatment of Cancer and the Mycoses Study Group.19 However, the European Organisation for Research and Treatment of Cancer and Mycoses Study Group have stated that those criteria are only applicable to patients with cancer, transplant recipients, and other patients who are severely immunocompromised, and are not suitable for patients in intensive care units (ICUs).20 Therefore, criteria for probable disease were modified to include COVID-19 as host factor and a positive PCR test for Mucorales as mycological evidence, but criteria for proven disease remained unchanged.
All probable or proven cases reported or published up until April 12, 2021, were included in the analysis. Statistical analyses were done with SPSS 25. We estimated survival probability using the Kaplan-Meier method. Hazard ratios and 95% CIs were calculated using the univariate Cox regression model. Patients were grouped according to the site of infection—namely, rhino-orbital cerebral mucormycosis with CNS involvement, rhino-orbital cerebral mucormycosis without proof of CNS involvement, and others (eg, pulmonary, gastrointestinal, and disseminated mucormycosis)—and survival was compared using the log-rank test. The Fisher's exact test was used to compare demographic characteristics, clinical factors, and outcomes. For all tests, p values of less than or equal to 0·05 defined statistical significance.
Results
Case descriptions
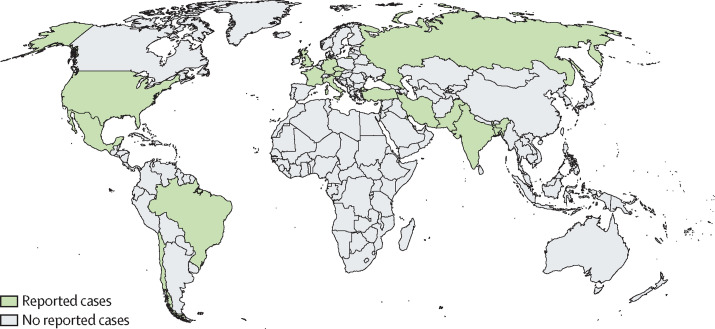
We identified 80 cases of COVID-19-associated mucormycosis (63 [79%] proven and 17 [21%] probable), including 29 (36%) unpublished cases. Cases were reported from 18 countries, the majority from India (42, ten of which were unpublished), the USA (eight), Pakistan (five unpublished from a single centre), France (four, three of which were unpublished from two centres), Iran (four, one of which was unpublished), and Mexico (four, three of which were unpublished from a single centre), Russia (two unpublished from two centres; figure 1 ). Single cases were reported from Austria, Bangladesh (unpublished), Brazil, Chile, Czech Republic (unpublished), Germany (unpublished), Italy, Kuwait (unpublished), Lebanon (unpublished), Turkey, and the UK.
Figure 1.
Global distribution of 80 COVID-19-associated mucormycosis cases
Cases were identified from the literature8, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45 and through experts within the European Confederation of Medical Mycology, International Society for Human and Animal Mycology, and FungiScope networks.
Prevalence of mucormycosis among people with COVID-19 who had been admitted to an ICU was reported from four centres (in France, Germany, Mexico, and Turkey) and ranged between 0·3% and 0·8%.
Demographics, underlying diseases, and COVID-19 treatments
The majority of patients were male (62 [78%] of 80) and the median age was 55 years (range 10–86). The table provides a summary of clinical characteristics and outcomes of COVID-19-associated mucormycosis in 80 patients and separates data of patients with rhino-orbital cerebral disease from data of those with pulmonary disease. At the time of COVID-19-associated mucormycosis diagnosis, 74 (93%) patients were hospitalised with ongoing COVID-19 infection, and six (8%) patients had COVID-19 infection before hospitalisation for mucormycosis-associated symptoms, four of whom were hospitalised for treatment of COVID-19 1–3 months before mucormycosis diagnosis.
Table.
Summary of clinical characteristics, treatment, and outcome of 80 cases of patients with COVID-19-associated mucormycosis
| COVID-19-associated mucormycosis (n=80)* | Rhino-orbital cerebral disease (n=59) | Pulmonary disease†(n=20) | |
|---|---|---|---|
| Median age (range), years | 55 (10–86) | 54 (10–79) | 57 (22–86) |
| Male-to-female-patient ratio | 62:18 (78%:23%) | 43:16 (73%:27%) | 18:2 (90%:10%) |
| Country of case origin | India (42 [53%]), USA (8 [10%]), Pakistan (5 [6%]), France (4 [5%]), Mexico (4 [5%]), Iran (4 [5%]), Russia (2 [3%]), Austria (1 [1%]), Bangladesh (1 [1%]), Brazil (1 [1%]), Chile (1 [1%]), Czech Republic (1 [1%]), Germany (1 [1%]), Italy (1 [1%]), Kuwait (1 [1%]), Lebanon (1 [1%]), Turkey (1 [1%]), UK (1 [1%]) | India (41 [69%]), USA (4 [7%]), Pakistan (1 [2%]), France (2 [3%]), Mexico (3 [5%]), Iran (4 [7%]), Russia (2 [3%]), Bangladesh (1 [2%]), Turkey (1 [2%]) | India (1 [5%]), USA (4 [20%]), Pakistan (4 [20%]), France (2 [10%]), Mexico (1 [5%]), Austria (1 [5%]), Chile (1 [5%]), Czech Republic (1 [5%]), Germany (1 [5%]), Italy (1 [5%]), Kuwait (1 [5%]), Lebanon (1 [5%]), UK (1 [5%]) |
| Underlying conditions | Diabetes (66 [83%], 55 [83%] of which had uncontrolled diabetes‡), haematological malignancy (5 [6%]§), lymphopenia (2 [3%]), hypertension (1 [1%]), chronic kidney disease (1 [1%]), severe obesity (1[1%]), no known risk factor (4 [5%]) | Diabetes (55 [93%], 48 [87%] of which had uncontrolled diabetes), haematological malignancy (1 [2%]), hypertension (1 [2%]), no known risk factor (2 [3%]) | Diabetes (11 [55%], 7 [64%] of which had uncontrolled diabetes), haematological malignancy (4 [20%]), lymphopenia (1 [5%]), chronic kidney disease (1 [5%]), severe obesity (1 [5%]), no known risk factor (2 [10%]) |
| Comorbidities¶ | Hypertension (14 [18%]), kidney disease (6 [8%]), obesity (3 [4%]), cardiac disease (5 [6%]), asthma (2 [3%]), hyperlipidaemia (2 [3%]) | Hypertension (7 [12%]), kidney disease (4 [7%]), obesity (1 [2%]), cardiac disease (2 [3%]), asthma (2 [3%]), hyperlipidaemia (2 [3%]) | Hypertension (6 [30%]), kidney disease (2 [10%]), cardiac disease (3 [15%]), hypothyroidism (1 [5%]), pancreatitis (1 [5%]), obesity (1 [5%]) |
| COVID-19 diagnosis | On admission (58 [68%]), previous (10 [13%]), during admission (1 [1%]), unknown (14 [18%]) | On admission (37 [63%]), previous (9 [15%]), unknown (13 [22%]) | On admission (17 [85%]), previous (1 [5%]), during admission (1 [5%]), unknown (1 [5%]) |
| COVID-19 severity | Severe or critical (36 [45%]), mild or moderate (36 [45%]), asymptomatic (2 [3%]), unknown (6 [8%]) | Severe or critical (19 [32%]), mild or moderate (33 [56%]), asymptomatic (2 [3%]), unknown (5 [8%]) | Severe or critical (16 [80%]), mild or moderate (4 [20%]) |
| Intensive care unit admission | Yes (38 [48%]), no (34 [43%]), unknown (8 [10%]) | Yes (19 [32%]), no (33 [56%]), unknown (7 [12%]) | Yes (18 [90%]), no (1 [5%]), unknown (1 [5%]) |
| Corticosteroids administered | Yes (63 [79%])‖, no (14 [18%]), unknown (3 [4%]) | Yes (47 [80%]), no (10 [17%]), unknown (2 [3%]) | Yes (15 [75%]), no (4 [20%]), unknown (1 [5%]) |
| Causative Mucorales spp | Rhizopus spp (16 [20%]), Mucor spp (6 [8%])**, R arrhizus (11 [14%]), R microsporus (7 [9%]), Lichtheimia spp (1 [1%]), Rhizomucor pusillus (1 [1%]), not specified (39 [49%]) | Rhizopus spp (10 [17%]), Mucor spp (6 [10%])**, R arrhizus (8 [14%]), Lichtheimia spp (1 [2%]), R microsporus (1 [2%]), not specified (34 [58%]) | Rhizopus spp (6 [30%]), R microsporus (6 [30%]), R arrhizus (3 [15%]), R pusillus (1 [5%]), not specified (4 [20%]) |
| Antifungal therapy | Liposomal amphotericin B (54 [68%]), conventional amphotericin B (9 [11%]), unknown amphotericin B formulation (7 [9%]), amphotericin lipid complex (2 [3%]), voriconazole (5 [6%]) isavuconazole (5 [6%]), posaconazole (6 [8%]), caspofungin (2 [3%]), micafungin (1 [1%]), antifungal combination (ie, drugs simultaneously; 14 [18%]), none (2 [3%]), unknown (1 [1%]) | Liposomal amphotericin B (44 [75%]), conventional amphotericin B (5 [8%]), unknown amphotericin B formulation (6 [10%]), amphotericin lipid complex (2 [3%]), voriconazole (2 [3%]), isavuconazole (2 [3%]), posaconazole (6 [10%]), caspofungin (1 [2%]), antifungal combination (ie, drugs simultaneously; 10 [17%]), none (1 [2%]), unknown (1 [2%]) | Liposomal amphotericin B (10 [50%]), conventional amphotericin B (4 [20%]), unknown amphotericin B formulation (1 [5%]), voriconazole (3 [15%]), isavuconazole (3 [15%]), caspofungin (1 [5%]), micafungin (1 [5%]), antifungal combination (ie, drugs simultaneously; 4 [20%]), none (1 [5%]) |
| Surgical intervention | Yes (45 [56%]), no (29 [36%]), deferred or unknown (6 [8%]) | Yes (43 [72%]), no (12 [20%]), deferred or unknown (4 [7%]) | Yes (2 [10%]), no (16 [80%]), deferred or unknown (2 [10%]) |
| Therapeutic strategy | Systemic antifungals plus surgery (44 [55%]), systemic antifungals only (32 [40%]), surgery only (1 [1%]), none (2 [3%]), unknown (1 [1%]) | Systemic antifungals plus surgery (42 [71%]), systemic antifungals only (15 [25%]), surgery only (1 [2%]), unknown (1 [2%]) | Systemic antifungals plus surgery (2 [10%]), systemic antifungals only (17 [85%]), none (1 [5%]) |
| Fungal coinfections | Aspergillosis (9 [11%]), none (71 [89%]) | Aspergillosis (3 [5%]), none (56 [95%]) | Aspergillosis (6 [30%]), none (14 [70%]) |
| Outcome | Survived (37 [46%]), died (39 [49%]), unknown (4 [5%]) | Survived (33 [56%]), died (22 [37%]), unknown (4 [7%]) | Survived (4 [20%]), died (16 [80%]) |
| Life-changing morbidities in survivors | Loss of vision (19 [24%]) | Loss of vision (19 [32%]) | .. |
Data are n (%) unless otherwise specified; n is the number of patients and the percentage is the proportion of patients out of the total indicated at the top of the column.
Includes 59 patients with rhino-orbital cerebral disease, 20 patients with pulmonary disease (three of whom had disseminated pulmonary disease), and one patient with gastrointestinal disease.
Includes 17 patients with pulmonary disease only and three patients with pulmonary disease and disseminated disease.
Well-controlled diabetes was reported in eight (12%) of 66 patients; the diabetes status of patients was unknown in three of 66 patients.
Two patients had lymphoma, two patients had acute myeloid leukaemia, and one patient had acute lymphoblastic leukaemia.
Numbers are super-additive.
Details available from 40 patients: 28 patients were given dexamethasone 6 mg for 10 days, and 12 were given methylprednisolone (40–120 mg for 5–28 days after hospital admission).
Rhizopus and Mucor spp was identified in one patient.
Most patients (76 [95%] of 80) had additional risk factors. Diabetes was the most prevalent condition overall (66 [83%] of 80), with type 2 diabetes being more common than type 1 diabetes (59 [89%] of 66 patients with diabetes had type 2 and 6 [9%] had type 1). Most of the 66 patients with diabetes had uncontrolled or poorly controlled diabetes (reported in 55 [83%] of 66), as characterised by diabetic ketoacidosis (27 [49%] of 55), hyperglycaemia (23 [42%] of 55), elevated HbA1c levels (available in 23 [77%] of 30 patients in whom HbA1c levels were available; median 11·0% [range 7·2–15·5%] and median 14·9 mmol/L [8·9–22·1 mmol/L]), or end-stage renal disease (6 [9%] of 66). Specifically, diabetes was a more predominant risk factor in patients from India than in patients from other countries (40 [95%] of 42 patients in India vs 26 [68%] of 38 from other countries, p=0·0024). Hypertension was the second most common underlying condition, identified in 15 (19%) of 80 patients, followed by chronic kidney diseases (five [6%]), and haematological malignancies (five [6%]). No apparent risk factor, aside from COVID-19, was noted in only four (5%) of 80 patients.
Treatment with systemic corticosteroids was reported in 63 (79%) of 80 cases, and 51 (81%) of 63 patients given systemic corticosteroids started treatment before the diagnosis of mucormycosis. Tocilizumab was given to six patients in addition to corticosteroids.
Clinical presentation and diagnosis
Patients with mucormycosis most commonly had rhino-orbital cerebral infection (59 [74%] of 80), with extension into the CNS confirmed in 22 (37%) of 59 patients. Rhino-orbital cerebral infection was particularly common in patients from India, where 41 (98%) of 42 patients had rhino-orbital cerebral infection (proof of cerebral involvement in 11 [27%] of 41 patients). Pulmonary disease was reported in 20 (25%) patients, three of whom had disseminated mucormycosis with sinus (one patient) or CNS (two patients) involvement, and one patient had gastrointestinal mucormycosis. Among patients with pulmonary disease, 18 (90%) of 20 patients were admitted to the ICU, mostly with severe or critical COVID-19 (15 [83%] of 18). Rhino-orbital cerebral mucormycosis was mostly seen in patients with diabetes (55 [93%] of 59 patients with rhino-orbital cerebral disease had diabetes, only four of whom had well-controlled blood sugar concentrations), whereas patients with other clinical manifestations of mucormycosis (20 with pulmonary and one with gastrointestinal COVID-19-associated mucormycosis) were less likely to have diabetes (11 [52%] of 21, four of whom had well-controlled blood sugar concentrations) than were those with rhino-orbital cerebral mucormycosis (p<0·0001). Four of the five patients with haematological malignancy had pulmonary mucormycosis during management of severe COVID-19 in the ICU and one patient had rhino-orbital cerebral infection 3 months after diagnosis of severe COVID-19. In the patients with pulmonary, gastrointestinal, or disseminated mucormycosis, the majority (17 [81%] of 21) had severe or critical COVID-19, whereas in the patients with rhino-orbital cerebral infection, COVID-19 was mainly mild or moderate (in 35 [59%] of 59). Mucormycosis was diagnosed at a median of 10 days (range 0–90 days) after diagnosis of COVID-19; 19 patients hospitalised with COVID-19 had clinical signs of mucormycosis at the time of admission (18 had rhino–orbital cerebral mucormycosis and 16 of these 18 had poorly controlled diabetes, and one patient with haematological malignancy presented with pulmonary infection). In the 61 patients who did not show signs of mucormycosis at the time of hospital admission, diagnosis was made at a median of 14·5 days (1–90 days) after admission. In the 55 patients with uncontrolled diabetes, COVID-19-associated mucormycosis was diagnosed in a median of 3·5 days (0–50 days) after COVID-19 (48 [87%] of 55 had rhino-orbital cerebral infection), versus a median of 20 days (0–60 days) in the 11 patients with controlled diabetes (7 [64%] of 11 had rhino-orbital cerebral infection). 25 (31%) patients required advanced critical care for at least 1 day before the onset of mucormycosis. In these patients, mucormycosis was diagnosed a median of 8·0 days (1–35 days) after admission to the ICU.
Proven diagnosis of mucormycosis, based on histology or culture of tissue or other samples from sterile sites, was obtained in 63 (79%) patients (five of those were obtained post mortem), with radiological abnormalities (eg, sinusitis, necrosis in patients with rhino-orbital cerebral mucormycosis, and nodules or cavities in patients with pulmonary disease) described in 40 (64%) of 63 patients. Probable pulmonary mucormycosis was confirmed by radiological findings and a positive culture or PCR from respiratory samples in 14 (82%) of 17 patients with rhino-orbital cerebral disease and by positive culture from swab or scraping samples in the other three (18%) patients with rhino-orbital cerebral disease.
Identification to the genus level was described in 25 [42%] of 59 patients with rhino-orbital cerebral infection and in 16 [76%] of 21 with pulmonary, gastrointestinal, or disseminated disease, and 19 of these infections were described to the species level. Rhizopus spp were the most common species identified, identified in 19 (32%) of 59 patients with rhino-orbital cerebral infection and 15 (71%) of 21 patients with pulmonary, gastrointestinal, or disseminated disease. Mucor spp were identified only in patients with rhino-orbital cerebral disease (six of 59 patients). Nine patients had mixed infection with Aspergillus spp, six of whom had pulmonary disease, and two of the three patients with rhino-orbital cerebral disease had proven cerebral involvement.
Treatment and outcome of COVID-19-associated mucormycosis
Systemic antifungal agents targeted against mucormycosis were used in all but three patients. Amphotericin B formulations were used in the vast majority of patients (71 [89%] of 80), and posaconazole was used in addition to amphotericin B in six patients with rhino-orbital cerebral mucormycosis. Isavuconazole was used in five patients, either in combination with amphotericin B (two patients), as salvage therapy (two patients), or as monotherapy (one patient). In addition, 45 (58%) patients underwent surgical resection (43 [96%] of 45 had rhino-orbital cerebral mucormycosis, of whom 16 had orbital exenteration).
All-cause mortality occurred in 39 (49%) of 80 patients. Mortality was reported in 22 (37%) of 59 patients who had rhino-orbital cerebral mucormycosis and in 17 (81%) of 21 patients who had pulmonary, gastrointestinal, or disseminated mucormycosis (p<0·0008). Mortality in patients with rhino-orbital cerebral disease with confirmed CNS involvement was higher (13 [59%] of 22 patients) than in those without signs of progression to the CNS (nine [24%] of 37 patients; p=0·012). Among survivors, loss of vision was reported in 19 (46%) of 41 patients (all with rhino-orbital cerebral mucormycosis).
Surgery was associated with improved outcomes in rhino-orbital cerebral mucormycosis in patients without proven CNS involvement, in whom mortality was higher in those who received systemic antifungals alone (five [63%] of eight) than in those who also had surgery (four [14%] of 29, of whom one had only surgery; p=0·012). Surgery did not significantly affect survival for patients with rhino-orbital cerebral mucormycosis who had proven CNS involvement (mortality in five [71%] of seven of patients who were given systemic antifungals alone vs eight [57%] of 14 who also had surgery; p=0·66).
14 (18%) patients required ICU treatment after the diagnosis of mucormycosis was made, 11 of whom were admitted with COVID-19 and clinical signs of rhino-orbital cerebral mucormycosis. Patients with severe or critical COVID-19 were more frequently treated in the ICU (30 [86%] of 36) than were patients with mild or moderate COVID-19 (seven [26%] of 37; p<0·0001). Overall, admission to the ICU was associated with worse outcomes, with overall mortality in 27 (71%) of 38 patients treated in the ICU versus 11 (32%) of 34 patients not treated in the ICU (p<0·0019).
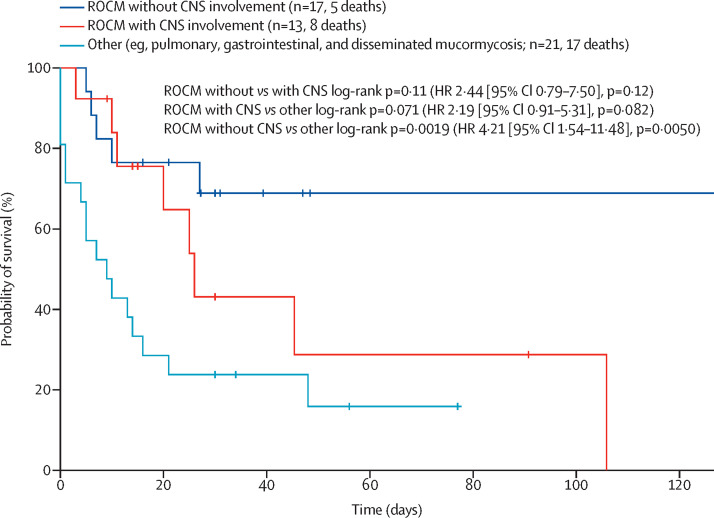
For patients with a fatal outcome, median survival time from the day of diagnosis of mucormycosis was 106 days (95% CI 9·4–202·3) for patients with rhino-orbital cerebral disease versus 9 days (2·4–15·6) for patients with pulmonary mucormycosis. Kaplan-Meier survival analysis showed a higher survival probability in patients with rhino-orbital cerebral disease without proven CNS infection than in those with other forms of the disease (17 with pulmonary disease, three with disseminated disease [including pulmonary mucormycosis], and one with gastrointestinal disease; log-rank p=0·0019; hazard ratio 4·210 [95% CI 1·54–11·48]; figure 2 ). Median survival time for patients with proven CNS involvement during rhino-orbital cerebral disease was 26 days (17·4–34·6), and survival over the disease course did not significantly differ from that in patients without proven CNS involvement and those with other forms of COVID-19-associated mucormycosis disease (compared with rhino-orbital cerebral diseases without proven CNS involvement [p=0·11], and compared with other forms of the disease [p=0·071]).
Figure 2.
Survival estimation of COVID-19-associated mucormycosis cases
Kaplan-Meier survival estimation for patients with rhino-orbital cerebral disease without CNS involvement, rhino-orbital cerebral mucormycosis with CNS involvement, and other forms of mucormycosis (eg, pulmonary, gastrointestinal, and disseminated). HR=hazard ratio.
Discussion
In this Review, we describe 80 cases of COVID-19-associated mucormycosis, with a predominance (42 of 80) of cases from India. There were three major findings: uncontrolled diabetes and systemic corticosteroid treatment were present in large proportions of patients; rhino-orbital cerebral mucormycosis was the most frequent presentation of disease (74%) and was present in 41 of 42 patients from India, whereas pulmonary disease almost exclusively occurred in the ICU setting and was present in 50% of patients reported from outside India; and amphotericin B formulations and surgery were the predominant treatment strategies, but mortality was high at 49%, mostly because of the high mortality of patients with pulmonary or disseminated mucormycosis and those with cerebral involvement. Furthermore, a substantial proportion of surviving patients had life-changing morbidities (eg, loss of vision in 46% of survivors).
Uncontrolled diabetes, and specifically ketoacidosis (in 27 [49%] of 55 patients) and systemic corticosteroid treatment (in 79% of patients), was present in a large proportion of our COVID-19-associated mucormycosis cohort, suggesting that patients with diabetes and COVID-19 receiving corticosteroids might be particularly susceptible to the development of COVID-19-associated mucormycosis. In particular, diabetes was almost ubiquitous in patients from India (present in 95% of patients). However, in pulmonary cases, most of which were reported in patients from Europe and the USA, diabetes was observed in 55% of patients with COVID-19-associated mucormycosis, which was higher than in a previous multinational cohort of patients in ICUs with COVID-19 acute respiratory failure without COVID-19-associated mucormycosis (27% of patients had diabetes).3 Although diabetes was frequently observed as an underlying disease, poor glycaemic control might be a specific predictor of rhino-orbital cerebral COVID-19-associated mucormycosis. Patients with uncontrolled diabetes developed mucormycosis (87% had rhino-orbital cerebral) a median of 2–3 weeks earlier after diagnosis of COVID-19 than patients with well-controlled diabetes, in whom 64% had COVID-19-associated mucormycosis that was rhino–orbital cerebral. The most probable mechanism behind this observation is the hyperglycaemia-induced inflammatory state that might be potentiated once antiviral immunity to SARS-CoV-2 activates. Moreover, Mucorales induce an augmented secretion of proinflammatory cytokines by immune cells.46 Therefore, even in the absence of immunosuppressive therapies, several divergent inflammatory pathways in patients with COVID-19 might act together to establish an inflammatory environment that is highly permissive to the development of COVID-19-associated mucormycosis, potentiating the expression of specific virulence factors and associated host damages. In a large randomised controlled trial in patients who were critically ill with COVID-19, treatment with IL-6 receptor antagonists improved outcomes, including survival.47 However, it is not known whether this therapeutic strategy of dampening an important cytokine in antifungal immunity could predispose patients to COVID-19-associated mucormycosis.
78% of COVID-19-associated mucormycosis cases reported in this Review occurred in male patients. Whether this gender imbalance might be the result of differing susceptibility—confirming what was recently observed in a diabetic ketoacidosis mouse model (in which female mice were more resistant to Mucorales infection than male mice)48—needs to be investigated in future studies.
The invasion of host cells during mucormycosis is elicited by the spore coat protein homologue, CotH3, which has been shown to mediate the interaction between spores and GRP78 on nasal epithelial cells to promote invasion and damage.49 GRP78 can also be upregulated in patients with COVID-19, and experimental studies have shown that GRP78 can form a complex with the spike protein and ACE2 to act as a host auxiliary factor for SARS-CoV2 entry and infection.50 The relevance of these mechanisms to human disease is supported by the induction of GRP78 by host factors characteristic of diabetic ketoacidosis, namely high glucose concentration, high iron concentration, and ketone bodies. Additionally, Mucorales produce the toxin mucoricin, which displays structural and functional features of the plant toxin ricin, and is endowed with the ability to induce inflammation, vascular permeability in vitro, and hypovolaemic shock and organ necrosis in mouse models of mucormycosis.51 Therefore, vascular endothelial injury, potentiated by COVID-19-associated inflammation, might also represent an important susceptibility feature of mucormycosis. On a cellular level, COVID-19 and its management might augment development of COVID-19-associated mucormycosis. First, the ability to restrict intracellular iron availability and inhibit fungal persistence and growth is an essential mechanism required to counter infection with Mucorales.52 With increased free iron concentrations observed in most patients with COVID-1953 and the Mucorales species' need for free iron for their biological processes, iron availability might represent a crucial mechanism involved in the pathogenesis of COVID-19-associated mucormycosis.54 Serum concentration of free iron is also increased in people with COVID-19 and ketoacidosis due to the release of iron from binding proteins during acidosis.55 In this context, the renal tropism of SARS-CoV2 might also become relevant to COVID-19-associated mucormycosis, as patients with renal failure undergoing deferoxamine chelation have increased concentrations of circulating free iron (since deferoxamine acts as a siderophore for Mucorales), and are more likely to develop mucormycosis than are patients not undergoing deferoxamine chelation.54 Second, the impairment of neutrophil migration, ingestion, and phagolysosome fusion caused by corticosteroids (reported in 79% of patients in this Review) might amplify the risk for mucormycosis. Third, the dysbiosis exerted by the intensive use of antibiotic treatment, along with the severe injuries of the epithelium, provide a permissive environment for developing invasive fungal infections,1, 4 including mucormycosis. Fourth, severe lymphocytopenia and lymphocyte dysfunction might also influence the development of COVID-19-associated mucormycosis as the decreased naive T-cell responses in patients with COVID-19 are aggravated with diabetes;56 Mucorales-specific T cells are detected in patients with mucormycosis and display important direct fungicidal activity.57
There might be several explanations for the observed differences between COVID-19-associated mucormycosis cases in India, where rhino-orbital cerebral disease was present in 41 of 42 patients compared with 18 of 38 patients from outside India. First, the distribution of Mucorales species varies between geographical regions, with Rhizopus arrhizus being the most common species in India58 and Cunninghamella spp being the most common species in Spain.59 Second, there might be differences in environmental exposure of pathogens, as ecological studies from India indicate the presence of many species of Mucorales in the soil and a large number of spores in indoor environments, such as air conditioning filters and hospital air, and in outdoor air.60 16 (80%) of 20 patients with pulmonary involvement had severe or critical COVID-19, and 18 (90%) required treatment in an ICU for acute respiratory failure, whereas only 19 (32%) of 59 patients with rhino-orbital cerebral mucormycosis had severe COVID-19 and 20 (34%) of 59 patients were admitted to an ICU. This finding might support the notion that the rhino-orbital cerebral form of COVID-19-associated mucormycosis occurs more frequently outside the ICU setting and, possibly, in patients with less severe COVID-19, than other forms of COVID-19-associated mucormycosis.
Only 6% of cases were diagnosed at autopsy, highlighting the challenges of diagnosing mucormycosis and indicating that the prevalence of COVID-19-associated mucormycosis might be underestimated, considering the rareness of autopsies, particularly among patients dying with COVID-19 in high numbers in resource-limited countries.61 Although bronchoalveolar lavage and biopsy samples can be difficult to obtain in patients with COVID-19, there is hope for more specific diagnostic tests. Several studies have shown that Mucorales PCR on blood samples, and in other matrices such as bronchoalveolar lavage or tissue specimens, is an early, sensitive, and specific non-invasive tool for diagnosis and monitoring of treatment efficacy.62, 63 In-house assays are designed to detect the most relevant Mucorales, including Mucor spp, Rhizopus spp, Lichtheimia spp, and Rhizomucor spp.62, 63 A Mucorales multiplex real-time PCR assay (MucorGenius, Pathonostics, Maastricht, Netherlands) that is easy to use adds Cunninghamella spp to the main Mucorales and was shown to be effective both in bronchoalveolar lavage and serum.64, 65 Molecular techniques to detect Mucorales are now considered new pivotal strategies for the diagnosis of mucormycosis and should be more broadly used in patients with COVID-19-associated mucormycosis (PCR results were only reported in one patient in this Review). Although PCR can provide species-level identification, a positive culture is required for in-vitro susceptibility testing. Indeed, a decrease in susceptibility to lipid-based amphotericin B or isavuconazole has been reported for some genera; therefore, species-level identification might be helpful to the clinician.7
The treatment of mucormycosis in patients with COVID-19 is similar to the treatment of patients with this invasive mycosis in other risk groups. However, treatment of mucormycosis in patients with COVID-19 is generally associated with fewer issues regarding neutropenia management than in patients with haematological malignancies, for example, but it is more focused on diabetes and corticosteroid exposure than in other risk groups. The three pivotal treatment principles of mucormycosis are control of the underlying disease or risk factor, surgical debridement of necrotic infected tissue, and specific antifungal therapy.7 First, it is important to carefully control glucose intolerance and rapidly treat any ketoacidosis. The reduction and elimination of long-term high-dose corticosteroids should be considered. Second, necrotic tissue resulting from major invasive mould infection should be carefully debrided to control the infection. This strategy needs to be done at the bedside by skilled surgeons in cooperation with the medical team. A common mistake is to delay crucial surgical debridement and assume antifungal agents alone will successfully treat the infection. In this group of patients with COVID-19-associated mucormycosis, we did see a positive outcome with surgery for rhino-orbital disease without proven CNS disease. Although surgical intervention is often essential in the successful management of mucormycosis, specific robust guidelines are difficult to develop that allow precision and the correct technical outcomes. Third, the use of antifungal agents is an essential feature of treatment. Initial antifungal therapy of serious life-threatening mucormycosis is generally centred around the most potent and broad-spectrum class of drugs, the polyenes, including amphotericin B.66 If available, lipid formulations of amphotericin B treatment are favoured because they have less nephrotoxicity than other formulations, especially when given in high daily doses.67 Two extended-spectrum azoles, isavuconazole and posaconazole, have emerged as viable alternatives to liposomal amphotericin B in the treatment of mucormycosis, particularly in patients with impaired renal function.7, 68 However, it is important to recognise that mucormycosis is caused by various genera and species, and some of the Mucorales respond poorly to azoles in vitro. Therefore, lipid amphotericin B formulations remain the primary treatment, which was also shown in this Review with 89% of patients receiving amphotericin B formulations. Following the guidelines, over half of the patients received additional surgical debridement. The observed all-cause mortality of 49%, which was highest for patients with pulmonary or disseminated mucormycosis and for patients with cerebral involvement, suggests that the prognosis of mucormycosis with SARS-CoV-2 is dismal, even with amphotericin B treatment and surgery. In this Review, the mortality rate was higher than it was in a large review of patients with diabetes and mucormycosis (44%).66
The currently available therapeutic interventions are not sufficient in reducing the high mortality associated with mucormycosis. Fortunately, genetic studies have identified new genes and widespread genome duplications in mucormycetes, allowing researchers to characterise virulence factors, such as versatility, robust growth, and multidrug resistance. These new insights might expose promising targets for future antifungal therapies.69 Many antifungal agents are less effective for the treatment of mucormycosis than for the treatment of aspergillosis. At present, new antifungal drugs in development have some in-vitro activity against Mucorales, but robust clinical studies are difficult and are not likely to be done in the short-term. Treatment of mucormycosis will generally be empirical for the near future. Other alternative strategies against mucormycosis should be considered in serious cases.70 Current challenges in diagnosis and management of COVID-19-associated mucormycosis are outlined in the panel .
Panel. Current challenges in diagnosis and management of COVID-19-associated mucormycosis.
Little awareness of COVID-19-associated mucormycosis 71
The countries with a high incidence of diabetes and COVID-19 (eg, Bangladesh, India, Pakistan, Iran, Mexico, China, Russia, Indonesia, and the USA) should monitor patients closely for a month after recovery from COVID-19, as most cases of COVID-19-associated mucormycosis are detected within this time period. COVID-19-induced immune dysregulation is seen late in the COVID-19 clinical course. Patients can be advised on discharge from hospital after COVID-19 recovery to look for nasal blockage, increased nasal discharge, and eye swelling and to seek urgent medical attention if these symptoms occur. This advice can help in the early diagnosis of cases. Mucormycosis is a rare disease: the literature contains only case reports without much knowledge of the prevalence of COVID-19-associated mucormycosis. There is a need for a multicentre epidemiology study to understand the impact of the disease.
Diagnosis of COVID-19-associated mucormycosis
Diagnosis of pulmonary mucormycosis is a major challenge in patients with COVID-19 due to avoidance of respiratory samples or bronchoscopy. Pulmonary mucormycosis should be suspected not only in patients in classical risk groups (eg, haematological malignancy, solid organ transplantation, and autoimmune diseases) but also in other patient groups, including those with COVID-19. Pulmonary cavities or multiple nodules on chest CT would suggest that a closer assessment is needed of other characteristic imaging signs (including bronchoscopy, biopsy, and molecular testing from obtained respiratory specimens), not to be confused with COVID-19 lesions. Other than rhino-orbital cerebral sites, diagnosis of mucormycosis at other sites is a challenge, potentially explaining the near absence of pulmonary COVID-19-associated mucormycosis diagnoses in India. Broader global availability of standardised molecular diagnostics of mucormycosis in the blood of patients are needed.72
Treatment of COVID-19-associated mucormycosis
Steroids are given indiscriminately in patients who are in hospital with COVID-19. Simultaneously, close glycaemic control is required in patients with diabetes as uncontrolled diabetes is the major risk factor for rhino-orbital cerebral mucormycosis. COVID-19 can damage β cells of the pancreas, resulting in metabolic derangement and eventually diabetes.73, 74 The cost of treating mucormycosis is a serious challenge in low-income and middle-income countries, and a considerable number of patients cannot afford therapy. Knowledge of managing mucormycosis is also a challenge among clinicians in low-income and middle-income countries.75
Limitations of our study include that not all data were available for all cases; the absence of a control cohort did not allow us to draw firm conclusions regarding risk factors for COVID-19-associated mucormycosis; and underlying conditions and risk factors might have been under-reported, particularly at resource-limited centres.
Conclusion
COVID-19-associated mucormycosis can be a serious complication of severe COVID-19, particularly in patients with uncontrolled diabetes. The complex relationship between COVID-19 and mucormycosis is still mostly unknown. Diagnosis of pulmonary and gastrointestinal mucormycosis is challenging and might frequently be missed, particularly in India and other low-income and middle-income countries. Despite its reputation as a difficult-to-treat mould infection and its high mortality in patients with COVID-19, particularly those with pulmonary disease, a careful management plan can be successful for rhino-orbital cerebral disease if there is early diagnosis of infection and control of COVID-19.
Data sharing
Collected data from all cases are available on request.
Declaration of interests
MH received research funding from Gilead, Pfizer, Astellas, Scynexis, and US National Institutes of Health. J-PG received speaker and expert advice fees from Pfizer and Gilead. NK has received research grants or honoraria as a speaker or advisor from Astellas, Gilead, MSD, and Pfizer, outside the submitted work. KL received consultancy fees from SMB Laboratories Brussels, MSD, and Gilead; travel support from Pfizer; speaker fees from FUJIFILM Wako, Pfizer, and Gilead; and a service fee from Thermo Fisher Scientific. OAC reports grants or contracts from Amplyx, Basilea, BMBF, Cidara, DZIF, EU-DG RTD (101037867), F2G, Gilead, Matinas, MedPace, MSD, Mundipharma, Octapharma, Pfizer, and Scynexis; Consulting fees from Amplyx, Biocon, Biosys, Cidara, Da Volterra, Gilead, Matinas, MedPace, Menarini, Molecular Partners, MSG-ERC, Noxxon, Octapharma, PSI, Scynexis, and Seres; honoraria for lectures from Abbott, Al-Jazeera Pharmaceuticals, Astellas, Grupo Biotoscana/United Medical/Knight, Hikma, MedScape, MedUpdate, Merck/MSD, Mylan, and Pfizer; payment for expert testimony from Cidara; participation on a data safety monitoring board or advisory board from Actelion, Allecra, Cidara, Entasis, IQVIA, Jannsen, MedPace, Paratek, PSI, and Shionogi; a patent at the German Patent and Trade Mark Office (DE 10 2021 113 0077); and other interests from DGHO, DGI, ECMM, ISHAM, MSG-ERC, and Wiley. JRP received research support from Amplyx, Appili, and Astellas; and honoraria and consulting fees from Pfizer, Appili, and Matinas. PLW has done diagnostic evaluations and received meeting sponsorship from Bruker, Dynamiker, and Launch Diagnostics; speakers fees, expert advice fees, and meeting sponsorship from Gilead; speaker and expert advice fees from F2G; speaker fees MSD and Pfizer; and is a founding member of the European Aspergillus PCR Initiative. ACh received funding support from an educational grant from Pfizer, MSD, and Gilead.
Acknowledgments
Acknowledgments
MH received funding from Astellas for two investigator-initiated studies (ISR005824 and ISR005838), and was supported by the US National Institutes of Health (UL1TR001442). ACa was supported by the Fundação para a Ciência e a Tecnologia (UIDB/50026/2020 and UIDP/50026/2020), the Northern Portugal Regional Operational Programme (NORTE 2020), under the Portugal 2020 Partnership Agreement, through the European Regional Development Fund (NORTE-01-0145-FEDER-000039), the EU's Horizon 2020 research and innovation programme (847507), and the la Caixa Foundation (100010434) and Fundação para a Ciência e a Tecnologia (LCF/PR/HR17/52190003). We thank Lidiya Didenko, Mahboobeh Karimi-Galougahi, Nariman Khan, Ricardo Rabagliati, Hülya Sungurtekin, Amirreza Veisi, and Salomon Waizel-Haiat for providing additional information on their published cases.
Contributors
MH and ACa coordinated the work of the authors and guided the development of the Review. Literature review and data extraction were done by PLW and DS. DS, PLW, MH, and ACh collected unpublished cases through a global reach-out via email and social media within the FungiScope network, European Confederation of Medical Mycology, and International Society for Human and Animal Mycology networks. ACa contributed to the immunology sections. Statistical analysis was done by DS. MH, DS, ACa, SMR, AA, J-PG, KL, JFM, OAC, JRP, PLW, and ACh wrote the initial manuscript draft. NN, AB, JA, NK, and AS contributed unpublished cases, critically revised the draft, and contributed important intellectual content. European Confederation of Medical Mycology and International Society for Human and Animal Mycology collaborators also contributed unpublished cases. The table summarising cases was created by DS and PLW. The manuscript was drafted by MH, DS, PLW, JP, ACa, AA, JFM, J-PG, ACh, SMR, KL, and OAC. Other authors critically revised the draft and added important intellectual content. All authors participated in review and revisions, approved the final manuscript, and are accountable for all aspects of the work and for ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors had access to and verified the data presented here.
European Confederation of Medical Mycology and International Society for Human and Animal Mycology collaborators
Souha S Kanj (Department of Internal Medicine, American University of Beirut Medical Center, Beirut, Lebanon), Hossein Zarrinfar (Allergy Research Center, Mashhad University of Medical Sciences, Mashhad, Iran), Abdolmajid Fata (Department of Parasitology and Mycology, School of Medicine, University of Medical Sciences, Mashhad, Iran), Khaled Alobaid (Mubarak Al-Kabeer Hospital, Kuwait City, Kuwait), Philipp Enghard (Department of Nephrology and Medical Intensive Care, Charité University Medicine Berlin, Berlin, Germany), Pavlina Lyskova (Public Health Institute in Ústí nad Labem, Prague, Czech Republic), Ariful Basher (Critical Care Medicine, Bangabandhu Sheikh Mujib Medical University, Dhaka, Bangladesh), Anne-Lise Munier (Department of Infectious Diseases, St-Louis Lariboisiere Hospital, ECMM Excellence Center for Medical Mycology, University of Paris, Paris, France), Blandine Denis (Department of Infectious Diseases, St-Louis Lariboisiere Hospital, ECMM Excellence Center for Medical Mycology, University of Paris, Paris, France), Kausar Jabeen (Department of Pathology and Laboratory Medicine, Section of Microbiology, Aga Khan University Karachi, Karachi, Pakistan), Joveria Farooqi (Department of Pathology and Laboratory Medicine, Section of Microbiology, Aga Khan University Karachi, Karachi, Pakistan), Syed Faisal Mahmood (Department of Medicine, Section of Adult Infectious Diseases, Aga Khan University Karachi, Karachi, Pakistan), Sofya Khostelidi (Department of Clinical Mycology, Allergy and Immunology, North Western State Medical University named after II Mechnikov, St Petersburg, Russia)
References
- 1.Arastehfar A, Carvalho A, van de Veerdonk FL, et al. COVID-19 associated pulmonary Aspergillosis (CAPA)—from immunology to treatment. J Fungi (Basel) 2020;6:91. doi: 10.3390/jof6020091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Koehler P, Bassetti M, Chakrabarti A, et al. Defining and managing COVID-19-associated pulmonary aspergillosis: the 2020 ECMM/ISHAM consensus criteria for research and clinical guidance. Lancet Infect Dis. 2020;21:e149–e162. doi: 10.1016/S1473-3099(20)30847-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Prattes J, Wauters J, Giacobbe DR, et al. Diagnosis and treatment of COVID-19 associated pulmonary apergillosis in critically ill patients: results from a European confederation of medical mycology registry. Intensive Care Med. 2021;47:1158–1160. doi: 10.1007/s00134-021-06471-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Arastehfar A, Carvalho A, Nguyen MH, et al. COVID-19-associated candidiasis (CAC): an underestimated complication in the absence of immunological predispositions? J Fungi (Basel) 2020;6:e211. doi: 10.3390/jof6040211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Poignon C, Blaize M, Vezinet C, Lampros A, Monsel A, Fekkar A. Invasive pulmonary fusariosis in an immunocompetent critically ill patient with severe COVID-19. Clin Microbiol Infect. 2020;26:1582–1584. doi: 10.1016/j.cmi.2020.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hoenigl M, Salmanton-García J, Walsh TJ, et al. Global guideline for the diagnosis and management of rare mould infections: an initiative of the European Confederation of Medical Mycology in cooperation with the International Society for Human and Animal Mycology and the American Society for Microbiology. Lancet Infect Dis. 2021;21:e246–e257. doi: 10.1016/S1473-3099(20)30784-2. [DOI] [PubMed] [Google Scholar]
- 7.Cornely OA, Alastruey-Izquierdo A, Arenz D, et al. Global guideline for the diagnosis and management of mucormycosis: an initiative of the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium. Lancet Infect Dis. 2019;19:e405–e421. doi: 10.1016/S1473-3099(19)30312-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zurl C, Hoenigl M, Schulz E, et al. Autopsy proven pulmonary mucormycosis due to Rhizopus microsporus in a critically ill COVID-19 patient with underlying hematological malignancy. J Fungi (Basel) 2021;7:88. doi: 10.3390/jof7020088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rudramurthy SM, Hoenigl M, Meis JF, et al. ECMM/ISHAM recommendations for clinical management of COVID-19 associated mucormycosis in low- and middle-income countries. Mycoses. 2021;64:1028–1037. doi: 10.1111/myc.13335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lamoth F, Chung SJ, Damonti L, Alexander BD. Changing epidemiology of invasive mold infections in patients receiving azole prophylaxis. Clin Infect Dis. 2017;64:1619–1621. doi: 10.1093/cid/cix130. [DOI] [PubMed] [Google Scholar]
- 11.Jenks JD, Reed SL, Seidel D, et al. Rare mould infections caused by Mucorales, Lomentospora prolificans and Fusarium, in San Diego, CA: the role of antifungal combination therapy. Int J Antimicrob Agents. 2018;52:706–712. doi: 10.1016/j.ijantimicag.2018.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in china: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323 doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 13.Horby P, Lim WS, Emberson JR, et al. Dexamethasone in hospitalized patients with COVID-19. N Engl J Med. 2021;384:693–704. doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Morales-Franco B, Nava-Villalba M, Medina-Guerrero EO, et al. Host-pathogen molecular factors contribute to the pathogenesis of Rhizopus spp in diabetes mellitus. Curr Trop Med Rep. 2021 doi: 10.1007/s40475-020-00222-1. published online Jan 22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tay MZ, Poh CM, Rénia L, MacAry PA, Ng LFP. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol. 2020;20:363–374. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Georgiadou SP, Sipsas NV, Marom EM, Kontoyiannis DP. The diagnostic value of halo and reversed halo signs for invasive mold infections in compromised hosts. Clin Infect Dis. 2011;52:1144–1155. doi: 10.1093/cid/cir122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Alexander BD, Lamoth F, Heussel CP, et al. Guidance on imaging for invasive pulmonary aspergillosis and mucormycosis: from the imaging working group for the revision and update of the Consensus Definitions of Fungal Disease from the EORTC/MSGERC. Clin Infect Dis. 2021;72(suppl 2):S79–S88. doi: 10.1093/cid/ciaa1855. [DOI] [PubMed] [Google Scholar]
- 18.John TM, Jacob CN, Kontoyiannis DP. When uncontrolled diabetes mellitus and severe covid-19 converge: the perfect storm for mucormycosis. J Fungi (Basel) 2021;7:298. doi: 10.3390/jof7040298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Donnelly JP, Chen SC, Kauffman CA, et al. Revision and update of the consensus definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2019;71:1367–1376. doi: 10.1093/cid/ciz1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bassetti M, Azoulay E, Kullberg BJ, et al. EORTC/MSGERC definitions of invasive fungal diseases: summary of activities of the Intensive Care Unit Working Group. Clin Infect Dis. 2021;72(suppl 2):S121–S127. doi: 10.1093/cid/ciaa1751. [DOI] [PubMed] [Google Scholar]
- 21.Alekseyev K, Didenko L, Chauhry B. Rhinocerebral mucormycosis and COVID-19 pneumonia. J Med Cases. 2021;12:85–89. doi: 10.14740/jmc3637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Karimi-Galougahi M, Arastou S, Haseli S. Fulminant mucormycosis complicating coronavirus disease 2019 (COVID-19) Int Forum Allergy Rhinol. 2021;11:1029–1030. doi: 10.1002/alr.22785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Khan NGC, Martinez DV, Proud KC. A case report of COVID-19 associated pulmonary mucormycosis. Arch Clin Cases. 2020;7:46–51. doi: 10.22551/2020.28.0703.10172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rabagliati R, Rodríguez N, Núñez C, Huete A, Bravo S, Garcia P. COVID-19-associated mold infection in critically ill patients, Chile. Emerg Infect Dis. 2021;27:1454–1456. doi: 10.3201/eid2705.204412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sargin F, Akbulut M, Karaduman S, Sungurtekin H. Severe rhinocerebral mucormycosis case developed after COVID-19. J Bacteriol Parasitol. 2021;12 [Google Scholar]
- 26.Veisi A, Bagheri A, Eshaghi M, Rikhtehgar MH, Rezaei Kanavi M, Farjad R. Rhino-orbital mucormycosis during steroid therapy in COVID-19 patients: a case report. Eur J Ophthalmol. 2021 doi: 10.1177/11206721211009450. published online April 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Waizel-Haiat S, Guerrero-Paz JA, Sanchez-Hurtado L, Calleja-Alarcon S, Romero-Gutierrez L. A case of fatal rhino-orbital mucormycosis associated with new onset diabetic ketoacidosis and COVID-19. Cureus. 2021;13 doi: 10.7759/cureus.13163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sen M, Lahane S, Lahane TP, Parekh R, Honavar SG. Mucor in a viral land: a tale of two pathogens. Indian J Ophthalmol. 2021;69:244–252. doi: 10.4103/ijo.IJO_3774_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Khatri A, Chang KM, Berlinrut I, Wallach F. Mucormycosis after coronavirus disease 2019 infection in a heart transplant recipient—case report and review of literature. J Mycol Med. 2021;31 doi: 10.1016/j.mycmed.2021.101125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kanwar A, Jordan A, Olewiler S, Wehberg K, Cortes M, Jackson BR. A fatal case of Rhizopus azygosporus pneumonia following COVID-19. J Fungi (Basel) 2021;7:174. doi: 10.3390/jof7030174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bellanger AP, Navellou JC, Lepiller Q, et al. Mixed mold infection with Aspergillus fumigatus and Rhizopus microsporus in a severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) patient. Infect Dis Now. 2021;51:633–635. doi: 10.1016/j.idnow.2021.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Werthman-Ehrenreich A. Mucormycosis with orbital compartment syndrome in a patient with COVID-19. Am J Emerg Med. 2021;42:264. doi: 10.1016/j.ajem.2020.09.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mekonnen ZK, Ashraf DC, Jankowski T, et al. Acute invasive rhino-orbital mucormycosis in a patient with COVID-19-associated acute respiratory distress syndrome. Ophthal Plast Reconstr Surg. 2021;37:e40–e80. doi: 10.1097/IOP.0000000000001889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Garg D, Muthu V, Sehgal IS, et al. Coronavirus disease (Covid-19) associated mucormycosis (CAM): case report and systematic review of literature. Mycopathologia. 2021;186:289–298. doi: 10.1007/s11046-021-00528-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dallalzadeh LO, Ozzello DJ, Liu CY, Kikkawa DO, Korn BS. Secondary infection with rhino-orbital cerebral mucormycosis associated with COVID-19. Orbit. 2021;12 doi: 10.1080/01676830.2021.1903044. [DOI] [PubMed] [Google Scholar]
- 36.Johnson AK, Ghazarian Z, Cendrowski KD, Persichino JG. Pulmonary aspergillosis and mucormycosis in a patient with COVID-19. Med Mycol Case Rep. 2021;32:64–67. doi: 10.1016/j.mmcr.2021.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pasero D, Sanna S, Liperi C, et al. A challenging complication following SARS-CoV-2 infection: a case of pulmonary mucormycosis. Infection. 2020;49:1055–1060. doi: 10.1007/s15010-020-01561-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Placik DA, Taylor WL, Wnuk NM. Bronchopleural fistula development in the setting of novel therapies for acute respiratory distress syndrome in SARS-CoV-2 pneumonia. Radiol Case Rep. 2020;15:2378–2381. doi: 10.1016/j.radcr.2020.09.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mehta S, Pandey A. Rhino-orbital mucormycosis associated with COVID-19. Cureus. 2020;12 doi: 10.7759/cureus.10726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Monte ESD, Junior, Santos MELD, Ribeiro IB, et al. Rare and fatal gastrointestinal mucormycosis (zygomycosis) in a COVID-19 patient: a case report. Clin Endosc. 2020;53:746–749. doi: 10.5946/ce.2020.180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hanley B, Naresh KN, Roufosse C, et al. Histopathological findings and viral tropism in UK patients with severe fatal COVID-19: a post-mortem study. Lancet Microbe. 2020;1:e245–e253. doi: 10.1016/S2666-5247(20)30115-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Moorthy A, Gaikwad R, Krishna S, et al. SARS-CoV-2, uncontrolled diabetes and corticosteroids—an unholy trinity in invasive fungal infections of the maxillofacial region? A retrospective, multi-centric analysis. J Maxillofac Oral Surg. 2021;20:1–8. doi: 10.1007/s12663-021-01532-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sarkar S, Gokhale T, Choudhury SS, Deb AK. COVID-19 and orbital mucormycosis. Indian J Ophthalmol. 2021;69:1002–1004. doi: 10.4103/ijo.IJO_3763_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bhavani P PS, Patil SD. Imaging findings of rhino-orbito-cerebral mucormycosis in a COVID-19 patient. Eurorad. https://www.eurorad.org/case/17205
- 45.Deshmukh RUK, Patwadkar R, Patil S. Mucor mycosis in COVID-19: case reports. J Adv Res Med. 2020;7:20–23. [Google Scholar]
- 46.Wurster S, Thielen V, Weis P, et al. Mucorales spores induce a proinflammatory cytokine response in human mononuclear phagocytes and harbor no rodlet hydrophobins. Virulence. 2017;8:1708–1718. doi: 10.1080/21505594.2017.1342920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gordon AC, Mouncey PR, Al-Beidh F, et al. Interleukin-6 receptor antagonists in critically ill patients with covid-19. N Engl J Med. 2021;384:1491–1502. doi: 10.1056/NEJMoa2100433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gebremariam T, Alkhazraji S, Alqarihi A, et al. Evaluation of sex differences in murine diabetic ketoacidosis and neutropenic models of invasive mucormycosis. J Fungi (Basel) 2021;7:313. doi: 10.3390/jof7040313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Alqarihi A, Gebremariam T, Gu Y, et al. GRP78 and integrins play different roles in host cell invasion during mucormycosis. MBio. 2020;11:e01087–e01120. doi: 10.1128/mBio.01087-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Carlos AJ, Ha DP, Yeh DW, et al. The chaperone GRP78 is a host auxiliary factor for SARS-CoV-2 and GRP78 depleting antibody blocks viral entry and infection. J Biol Chem. 2021;296 doi: 10.1016/j.jbc.2021.100759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Soliman SSM, Baldin C, Gu Y, et al. Mucoricin is a ricin-like toxin that is critical for the pathogenesis of mucormycosis. Nat Microbiol. 2021;6:313–326. doi: 10.1038/s41564-020-00837-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Andrianaki AM, Kyrmizi I, Thanopoulou K, et al. Iron restriction inside macrophages regulates pulmonary host defense against Rhizopus species. Nat Commun. 2018;9 doi: 10.1038/s41467-018-05820-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Reid G, Lynch JP, 3rd, Fishbein MC, Clark NM. Mucormycosis. Semin Respir Crit Care Med. 2020;41:99–114. doi: 10.1055/s-0039-3401992. [DOI] [PubMed] [Google Scholar]
- 55.Schell WA. Unusual fungal pathogens in fungal rhinosinusitis. Otolaryngol Clin North Am. 2000;33:367–373. doi: 10.1016/s0030-6665(00)80011-0. [DOI] [PubMed] [Google Scholar]
- 56.Schwartz MD, Emerson SG, Punt J, Goff WD. Decreased naive T-cell production leading to cytokine storm as cause of increased COVID-19 severity with comorbidities. Aging Dis. 2020;11:742–745. doi: 10.14336/AD.2020.0619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Potenza L, Vallerini D, Barozzi P, et al. Mucorales-specific T cells emerge in the course of invasive mucormycosis and may be used as a surrogate diagnostic marker in high-risk patients. Blood. 2011;118:5416–5419. doi: 10.1182/blood-2011-07-366526. [DOI] [PubMed] [Google Scholar]
- 58.Prakash H, Chakrabarti A. Global epidemiology of mucormycosis. J Fungi (Basel) 2019;5:e26. doi: 10.3390/jof5010026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Guinea J, Escribano P, Vena A, et al. Increasing incidence of mucormycosis in a large Spanish hospital from 2007 to 2015: epidemiology and microbiological characterization of the isolates. PLoS One. 2017;12 doi: 10.1371/journal.pone.0179136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Prakash H, Singh S, Rudramurthy SM, et al. An aero mycological analysis of Mucormycetes in indoor and outdoor environments of northern India. Med Mycol. 2020;58:118–123. doi: 10.1093/mmy/myz031. [DOI] [PubMed] [Google Scholar]
- 61.Salerno M, Sessa F, Piscopo A, et al. No autopsies on COVID-19 deaths: a missed opportunity and the lockdown of science. J Clin Med. 2020;9 doi: 10.3390/jcm9051472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Millon L, Scherer E, Rocchi S, Bellanger AP. Molecular strategies to diagnose mucormycosis. J Fungi (Basel) 2019;5:e24. doi: 10.3390/jof5010024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Millon L, Herbrecht R, Grenouillet F, et al. Early diagnosis and monitoring of mucormycosis by detection of circulating DNA in serum: retrospective analysis of 44 cases collected through the French Surveillance Network of Invasive Fungal Infections (RESSIF) Clin Microbiol Infect. 2016;22:810. doi: 10.1016/j.cmi.2015.12.006. [DOI] [PubMed] [Google Scholar]
- 64.Guegan H, Iriart X, Bougnoux ME, Berry A, Robert-Gangneux F, Gangneux JP. Evaluation of MucorGenius® mucorales PCR assay for the diagnosis of pulmonary mucormycosis. J Infect. 2020;81:311–317. doi: 10.1016/j.jinf.2020.05.051. [DOI] [PubMed] [Google Scholar]
- 65.Mercier T, Reynders M, Beuselinck K, Guldentops E, Maertens J, Lagrou K. Serial detection of circulating mucorales DNA in invasive mucormycosis: a retrospective multicenter evaluation. J Fungi (Basel) 2019;5:E113. doi: 10.3390/jof5040113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Roden MM, Zaoutis TE, Buchanan WL, et al. Epidemiology and outcome of zygomycosis: a review of 929 reported cases. Clin Infect Dis. 2005;41:634–653. doi: 10.1086/432579. [DOI] [PubMed] [Google Scholar]
- 67.Lanternier F, Lortholary O. AMBIZYGO: phase II study of high dose liposomal amphotericin B (AmBisome) [10 mg/kg/j] efficacy against zygomycosis. Med Mal Infect. 2008;38(suppl 2):S90–S91. doi: 10.1016/S0399-077X(08)73003-8. [DOI] [PubMed] [Google Scholar]
- 68.Perfect JR, Cornely OA, Heep M, et al. Isavuconazole treatment for rare fungal diseases and for invasive aspergillosis in patients with renal impairment: challenges and lessons of the VITAL trial. Mycoses. 2018;61:420–429. doi: 10.1111/myc.12769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Trieu TA, Navarro-Mendoza MI, Pérez-Arques C, et al. RNAi-Based functional genomics identifies new virulence determinants in mucormycosis. PLoS Pathog. 2017;13 doi: 10.1371/journal.ppat.1006150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Dannaoui E, Schwarz P, Lortholary O. In vitro interactions between antifungals and immunosuppressive drugs against zygomycetes. Antimicrob Agents Chemother. 2009;53:3549–3551. doi: 10.1128/AAC.00184-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tan BH, Chakrabarti A, Patel A, et al. Clinicians' challenges in managing patients with invasive fungal diseases in seven Asian countries: an Asia Fungal Working Group (AFWG) survey. Int J Infect Dis. 2020;95:471–480. doi: 10.1016/j.ijid.2020.01.007. [DOI] [PubMed] [Google Scholar]
- 72.Rocchi S, Scherer E, Mengoli C, et al. Interlaboratory evaluation of Mucorales PCR assays for testing serum specimens: a study by the fungal PCR Initiative and the Modimucor Study Group. Med Mycol. 2021;59:126–138. doi: 10.1093/mmy/myaa036. [DOI] [PubMed] [Google Scholar]
- 73.Müller JA, Groß R, Conzelmann C, et al. SARS-CoV-2 infects and replicates in cells of the human endocrine and exocrine pancreas. Nat Metab. 2021;3:149–165. doi: 10.1038/s42255-021-00347-1. [DOI] [PubMed] [Google Scholar]
- 74.Accili D. Can COVID-19 cause diabetes? Nat Metab. 2021;3:123–125. doi: 10.1038/s42255-020-00339-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Patel A, Kaur H, Xess I, et al. A multicentre observational study on the epidemiology, risk factors, management and outcomes of mucormycosis in India. Clin Microbiol Infect. 2020;26:944. doi: 10.1016/j.cmi.2019.11.021. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Collected data from all cases are available on request.