Abstract
Psychometric validity and reliability of widely used atopic dermatitis (AD) outcome measures across different race and ethnicity is unclear. We describe rates of reporting race, ethnicity and skin tone in studies testing psychometric properties of AD outcome measures and compare psychometric analyses across race, ethnicity, and skin tone. We systematically reviewed MEDLINE and EMBASE for studies reporting psychometric properties of clinician reported (ClinROM) or patient reported outcome measures (PROM) in AD (PROSPERO: CRD42021239614). Overall, 16,100 non-duplicate articles were screened; 165 met inclusion criteria. Race and/or ethnicity were reported in 55 (33.3%) studies; of those, race was assessed by self-report in 10 (6.1%) or was unspecified in 45 (27.3%). Sixteen studies (9.7%) evaluated psychometric property differences by race and only 5 (4.4%) of those that did not recognized it as a limitation. Properties assessed across race, ethnicity or skin tone were differential item functioning, convergent validity feasibility, inter-rater reliability, intra-rater reliability, test-retest reliability, and known-groups validity. Multiple instruments demonstrated performance differences across ethno-racial groups. This review highlights the paucity of race/ethnicity consideration for psychometric property testing in AD outcome measurement instruments. More AD outcomes instruments should be validated in diverse populations.
INTRODUCTION
Atopic dermatitis (AD) is characterized by heterogeneous lesional morphology, distribution, symptom complex and comorbidities.(Silverberg et al., 2020g) Certain AD features vary between ethnoracial groups, including lesional distribution, frequency and degree of follicular and lichenoid lesions, lichenification and prurigo nodules.(Brunner and Guttman-Yassky, 2019, Kaufman et al., 2018) These differences may introduce challenges for standardized diagnostic criteria as well as disease severity assessments in research and clinical settings.(Zhao et al., 2017) For example, the Hanifin-Rajka and abridged United Kingdom Working Party criteria perform inadequately in certain non-white races and ethnicities.(Thyssen et al., 2020) Understanding psychometric properties of relevant measurement domains and outcome instruments across different races, ethnicities, and skin tones is therefore essential.
For clinical trials, Harmonizing Outcome Measures for Eczema (HOME) selected by international consensus preferred outcome measurements for AD symptoms (Patient-Oriented Eczema Measure [POEM]), signs (Eczema Area and Severity Index [EASI]), and quality of life (QoL) (Dermatology Life Quality Index [DLQI]) in adults.(Silverberg et al., 2020a) For clinical practice, HOME recommended POEM and Patient-Oriented SCORing Atopic Dermatitis (PO-SCORAD) to assess patient-reported symptoms.
However, little is known about differences in psychometric properties of commonly used patient-reported and clinician-reported outcome measures (PROMs and ClinROMs) by race, ethnicity or skin tone. Some signs evaluated (e.g. erythema, lichenification, and xerosis) and symptoms (e.g. pruritus) may present differently or in different degrees in skin of color (SOC).(Kaufman et al., 2018, Zhao et al., 2017) Some instruments were adapted for darker skin tones (e.g. PO-SCORAD)(Faye et al., 2020a), though most have not. Concerns remain regarding the potential of overestimating(Zhao et al., 2017) or underestimating(Ben-Gashir and Hay, 2002, Kaufman et al., 2018) AD severity in SOC patients depending on the instrument used.
It is imperative to assess AD signs and symptoms with instruments that are valid and reliable across all races, ethnicities and skin types in clinical care and research. Tools that perform differently across these groups may lead to inadequate clinical management, systematic bias and potentially spurious trial results. In this systematic review, we examined reporting of race, ethnicity and skin tone and compared results across these groups from studies of psychometric properties for outcome measures in AD.
RESULTS
Literature search
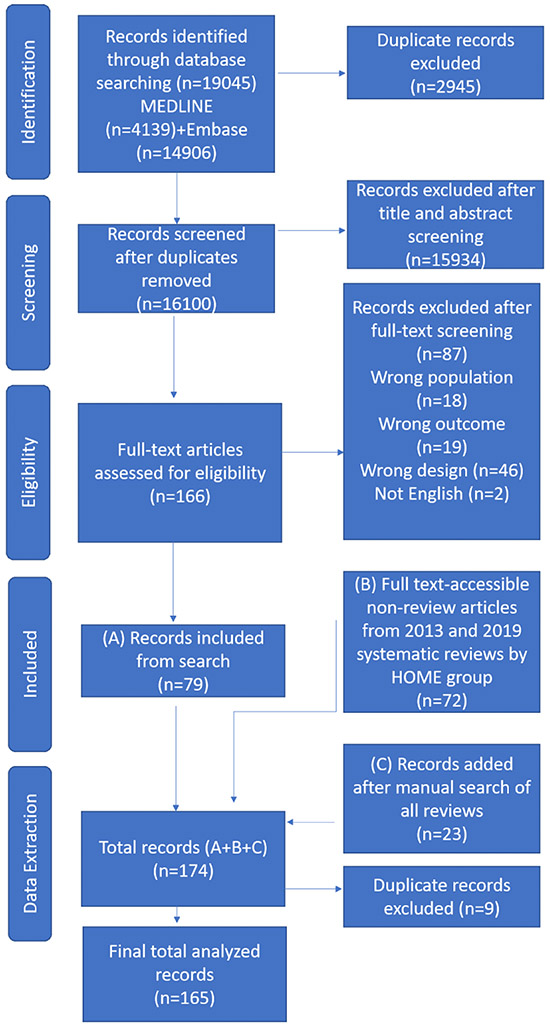
Overall, 16100 non-duplicate records underwent title and abstract screening and 166 full text review; 79 studies met eligibility criteria (Figure 1). Seventy-nine additional references were identified by snowballing from the 2013 and 2019 HOME group systematic reviews, of which 72 were non-review articles and available in full-text. An additional 23 articles were identified by snowballing from references of other included articles. In total, 165 non-duplicate articles were included in the review (Appendix 4). There was inconsistent use of terminologies for race and ethnicity in included papers. Results are reported according to the authors’ definitions, not consensus opinion (Appendix 5). The 165 studies included 41,146 persons (white: n=7,433 [18.1%]; black: n=1,003 [2.4%]; Hispanic: n=731 [1.8%]; Asian: n=1,630 [4.0%]; American Indian/Alaskan Native [n=6; 0.02%]; multiracial/other: n=430 [1.1%]; unspecified: n=29,913 [72.7 %]). Additional study and cohort characteristics and risk of bias analyses are presented in Supplemental Results.
Figure 1.
PRISMA flow diagram.
Reporting of race and ethnicity
Ethnoracial demographics of study participants were reported in only 55 (33.3%) studies; race was assessed by self-report in 10 (6.1%) or unspecified methods (n=45, 27.3%). Four (2.4%) studies described participant race, 9 (5.5%) ethnicity, 25 (15.2%) race and ethnicity combined, 7 (4.2%) race and ethnicity separately, and 10 (6.1%) unspecified.
Only 16 (9.7%) studies evaluated differences of psychometric properties by race, ethnicity, or skin tone; 113 (68.5%) did not, of which only 5 acknowledged absence of stratification by race as a study limitation or direction for future research. Race or ethnicity were assessed by self-report in 2 (1.2%) or unspecified in 14 (8.5%). The median (minimum-maximum) proportions of participants were 61% (36-71.9%) white, 11.88% (0.6-13.8%) black, 7.1% (0-18.8%) Hispanic, 16.7% (0-48%) for Asian, 0.0% (0-1.5%) American Indian/Alaskan native, and 3.9% (0-17.2%) multiracial/other (Table 1). The 16 studies included 5,900 persons (white: n=3,732 [63.3%]; black: n=330 [5.6%]; Hispanic: n=254 [4.3%]; Asian: n=436 [7.4%]; American Indian/Alaskan Native [n=2; 0.03%]; multiracial/other: n=135 [2.13%]; unspecified: n=1,011 [17.1%]).
Table 1.
Number of studies that investigated psychometric properties of clinician-reported and patient-reported outcomes by skin tone, race, or ethnicity by AD Instrument.
| Clinician-reported Outcome Measure |
Convergent Validity | DIF | Interrater Reliability | Intrarater Reliability | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||||
| Study | N | Findings^ | Study | N | Findings# | Study | N | Findings | Study | N | Findings | |
| EASI | 1. (Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | 1.(Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | − | 1.(Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | − | ||||
| 2. (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | 2.(Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | − | 2.(Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | − | |||||
| 3. (Vakharia P. P. et al., 2018) Prospective Cross-Sectional study* |
388 | − | ||||||||||
| 4. (Leshem et al., 2015a) Retrospective Cross-Sectional study* | 170 | − | ||||||||||
| (Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | (Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | − | (Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | − | |||||
| oSCORAD | (Zhao et al., 2017) Prospective Cross-Sectional study * |
25 | (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | − | (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | − | ||||
| (Vakharia P. P. et al., 2018) Prospective Cross-Sectional study * |
388 | − | ||||||||||
| IGA | (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | − | (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | − | ||||
| TIS | (Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | (Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | + | (Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | − | ||||
| Greyscale | (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | − | (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | + | ||||||
| SGAxBSA | 5. (Topham et al., 2020) Retrospective Cross-Sectional study* | 138 | ||||||||||
| SASSAD | (Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | (Zhao et al., 2015) Prospective Cross-Sectional study* |
18 | − | (Zhao et al., 2015) Prospective Cross-Sectional study* | 18 | − | ||||
| (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | |||||||||||
|
| ||||||||||||
|
Patient-Reported Outcome Measure
| ||||||||||||
| DLQI | 1. (Patel et al., 2019) Prospective Cross-Sectional study | 340 | + | |||||||||
| 2.(Silverberg et al., 2019b) Prospective Cross Sectional Study ~ |
602 | + | ||||||||||
| POSCORAD | 6. (Silverberg et al., 2020c) Prospective Cross-Sectional Study* | 291 | − | 3.(Silverberg et al., 2020c) Prospective Cross-Sectional Study | 291 | − | ||||||
| 4.(Silverberg et al., 2020h) Prospective Cross Sectional Study | 602 | + | ||||||||||
| POEM | (Zhao et al., 2017) Prospective Cross-Sectional study* | 25 | (Silverberg et al., 2020c) Prospective Cross-Sectional Study |
291 | − | |||||||
| (Silverberg et al., 2020c) Prospective Cross-Sectional Study* | 291 | (Silverberg et al., 2020h) Prospective Cross Sectional Study ~ |
602 | + | ||||||||
| NRS Itch | (Silverberg et al., 2020h) Prospective Cross Sectional Study ~ |
602 | + | |||||||||
| Self-reported Global AD Severity |
7. (Silverberg et al., 2018b) Prospective Cross Sectional Study~ | 602 | − | 5.(Silverberg et al., 2018b) Prospective Cross Sectional Study ~ |
602 | − | ||||||
| (Vakharia P. P. et al., 2018) Prospective Cross-Sectional Study * |
266 | − | ||||||||||
| Itchy QoL | (Patel et al., 2019) Prospective Cross-Sectional Study* | 340 | + | |||||||||
| 5D Itch | (Patel et al., 2019) Prospective Cross-Sectional Study* | 340 | + | |||||||||
| SF12 | (Silverberg et al., 2019b) Prospective Cross Sectional Study ~ |
602 | + | |||||||||
| PROMIS PIQ | 6. (Lei et al., 2020a) Prospective Cross-Sectional Study | 611 | − | |||||||||
| PROMIS SD | (Lei et al, 2020a) Prospective Cross-Sectional Study* | 611 | − | |||||||||
| 7.(Lei et al., 2020b) Prospective Cross-Sectional Study* | 420 | − | ||||||||||
| PROMIS SRI | (Lei et al., 2020a) Prospective Cross-Sectional Study* | 611 | ||||||||||
| (Lei et al., 2020b) Prospective Cross-Sectional Study* | 420 | − | ||||||||||
| ESS | (Lei et al., 2020a) Prospective Cross-Sectional Study* | 611 | − | |||||||||
| PIQ Itch Triggers | ||||||||||||
| PIQ Short Form | 8.(Silverberg et al., 2020b) Prospective Cross-Sectional Study* | 239 | + | |||||||||
| HADS | 9.(Silverberg et al., 2019a) Prospective Cross Sectional Study~ | 602 | + | |||||||||
|
| ||||||||||||
| Total studies | 7 | 9 | 2 | 2 | ||||||||
| Clinician-Reported Outcome Measure |
Test-retest reliability | Known groups validity | Feasibility | ||||||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| Study | N | Findings | Study | N | Findings | Study | N | Findings | |
| EASI | 1.(Leshem et al., 2015a) Retrospective Cross-Sectional Study* | 170 | − | ||||||
| oSCORAD | |||||||||
| IGA | |||||||||
| TIS | |||||||||
| Greyscale | |||||||||
| SGAxBSA | |||||||||
| SASSAD | |||||||||
|
| |||||||||
|
Patient-Reported Outcome Measure
| |||||||||
| DLQI | |||||||||
| POSCORAD | 1.(Silverberg et al., 2020c) Prospective Cross-Sectional Study* | 291 | + | 2.(Silverberg et al., 2020c) Prospective Cross-Sectional Study* | 291 | − | |||
| POEM | (Silverberg et al., 2020c) Prospective Cross-Sectional Study* | 291 | − | (Silverberg et al., 2020c) Prospective Cross-Sectional Study* | 291 | − | |||
| NRS Itch | |||||||||
| Self-reported Global AD Severity | 1.(Vakharia P. P. et al., 2018) Prospective Cross-Sectional Study* | 265 | |||||||
| Itchy QoL | |||||||||
| 5D Itch | |||||||||
| SF12 | |||||||||
| PROMIS PIQ | |||||||||
| PROMIS SD | 2.(Lei et al., 2020b) Prospective Cross-Sectional Study* | 420 | |||||||
| PROMIS SRI | (Lei et al., 2020b) Prospective Cross-Sectional Study* | 420 | |||||||
| ESS | |||||||||
| PIQ Itch Triggers | 3.(Silverberg et al., 2020g) Prospective Cross-Sectional Study* | 587 | − | ||||||
| PIQ Short Form | 4.(Silverberg et al., 2020b) Prospective Cross-Sectional Study* | 239 | − | ||||||
| HADS | |||||||||
|
| |||||||||
| Total studies | 1 | 2 | 4 | ||||||
N=sample size
+ Significant between-group differences found by race, ethnicity, or skin tone
− No significant between-group differences found upon assessment
Blank No assessment of between-group significance performed when reporting differences
Indication of significance for correlation coefficients of specific subgroups is coded in Table 2
Clinical Practice setting
Clinical Practice setting (Inpatient)
Analysis of web-based survey
Differences of psychometric properties by race, ethnicity or skin tone
Cross-cultural validity, reliability and construct validity were examined by race, ethnicity or skin tone: differential item functioning (DIF) (n=9, 56.3%), convergent validity (n=7, 50%), feasibility (n=4, 25%), inter-rater reliability (n=2, 12.5%), intra-rater reliability (n=2, 12.5%), test-retest reliability (n=1, 6.2%), and known-groups validity (n=2, 12.5%) (Table 1). No studies examined differences of content validity, floor and ceiling effects, measurement error, criterion validity, responsiveness, internal consistency, criterion validity, content validity, dimensionality, interpretability, or meaningful change by race, ethnicity or skin tone.
Only 2 (11.8%) studies analyzed differences of psychometric properties by skin pigmentation or tone.(Zhao et al., 2017, Zhao et al., 2015) Skin tone was assessed via melanin index determined by machine readings of gluteal skin(Zhao et al., 2017) or clinician assessment of skin pigmentation (0 [no pigment] to 10 [darkest pigment]).(Zhao et al., 2015) One study validated AD instruments in an exclusively African population who were presumed to all be Fitzpatrick Group 6.(Faye et al., 2020a) Psychometric properties assessed by race and skin tone-based were summarized for each PROM and ClinROM (Appendix 5).
PROMS
Differential Item Functioning (DIF)
Nine studies investigated DIF by race or ethnicity.(Lei et al., 2020a, Lei et al., 2020b, Patel et al., 2019, Silverberg et al., 2018b, Silverberg et al., 2019a, Silverberg et al., 2019b, Silverberg et al., 2020b, Silverberg et al., 2020c, Silverberg et al., 2020h) A study of 291 adults found that no items from PO-SCORAD and POEM had significant DIF by race (white vs. non-white).(Silverberg et al., 2020c) In a study of 611adults, no items from the Patient-reported Outcome Measures Information System (PROMIS©) Itch Questionnaire (PIQ) Mood and Sleep, PROMIS Sleep Disturbance, PROMIS Sleep-Related Impairment, or ESS had significant uniform or nonuniform DIF by race/ethnicity (white vs. other).(Lei et al., 2020a) Three and four items from the Hospital Anxiety and Depression scales (HADS), respectively, had significant uniform and nonuniform DIF by race (white vs. black vs. Asian vs. other).(Silverberg et al., 2019a) A study of 239 adults with AD found that two items from PIQ short-forms met criteria for uniform or nonuniform DIF by race (white vs. non-white), but the magnitudes were low.(Silverberg et al., 2020b). A study of 340 adults with AD found 1 item from DLQI had uniform DIF and 1 item had nonuniform DIF by race; 4 items from ItchyQoL had uniform DIF and 5 items had nonuniform DIF; 1 item from 5-D itch had uniform DIF and 1 item had nonuniform DIF (white vs. non-white).(Patel et al., 2019) A study of 602 adults found no significant differences of self-reported global atopic dermatitis severity by race (white vs. non-white).(Silverberg et al., 2018b)
A study of 602 adults with AD found 1 and 0 items from SF-12 with uniform and nonuniform DIF; 5 and 2 items from DLQI displayed uniform and nonuniform DIF (Silverberg et al., 2019b); multiple items of PO-SCORAD (1 item with uniform; 2 items with nonuniform) and POEM (5 items with uniform DIF; 2 items with nonuniform DIF) by race (white vs black vs Asian vs other)(Silverberg et al., 2020h); Finally, no items in PROMIS SD or PROMIS SRI had significant uniform or nonuniform DIF by race (white vs. other).(Lei et al., 2020b)
DIF was assessed for DLQI, POSCORAD, POEM, PROMIS SD, PROMIS SRI in multiple studies. Two studies examined DIF using regression analysis. One showed that DLQI had one item with uniform DIF (Q10) and one with non-uniform DIF (Q2).(Patel et al., 2019) Another found 5 items (Q1, Q2, Q6, Q9, Q10) with uniform and 2 (Q2, Q6) with nonuniform DIF. Q10 and Q2 of DLQI showed uniform and nonuniform DIF, respectively, in both studies.(Patel et al., 2019, Silverberg et al., 2019b) One study used Item Response Theory (IRT) and also showed Q2 had uniform DIF.(Patel et al., 2019)
Two studies assessed DIF for PO-SCORAD and POEM and had contradictory findings. One found 4 items of PO-SCORAD with significant uniform (dry, NRS-itch, NRS-sleep) or nonuniform DIF (dry, red areas, NRS-itch).(Silverberg et al., 2020h) The same study found 5 items of POEM with significant uniform (sleep, bleeding, weeping, cracking, dry or rough) or nonuniform DIF (sleep, dry or rough). Another reported no significant DIF on any item for POSCORAD or POEM.(Silverberg et al., 2020c)
Convergent validity
Four studies identified correlations between PROMs by race, without significant inter-racial differences observed.(Silverberg et al., 2018b, Silverberg et al., 2020c, Vakharia P. P. et al., 2018, Zhao et al., 2017) Self-reported global AD severity showed significant associations with PO-SCORAD total and objective scores, NRS-itch, NRS-sleep, NRS-pain, POEM, and HADS in both white and non-white subgroups.(Silverberg et al., 2018b) Associations of self-reported global AD severity with objective-SCORAD, SCORAD, EASI, BSA, NRS-itch, POEM and DLQI showed similar effect-sizes in white vs. non-white patients.(Vakharia P. P. et al., 2018)
Correlational effect size data was available for eleven PROMs (POEM, POSCORAD, NRS worst itch, NRS average itch, DLQI, ItchyQoL, PHQ9, PROMIS SD, PROMIS SRI, INRS, PtGA) for both white/nonwhite patients and SOC/light skin patients.(Silverberg et al., 2020c, Zhao et al., 2017) The range of correlations for POEM in white patients was 0.41 (PROMIS SRI) to 0.65 (DLQI and ItchyQoL); in non-white patients this range was 0.48 (NRS worst) to 0.74 (ItchyQoL). The range of correlations for PO-SCORAD in white patients was 0.41 (PROMIS SD) to 0.68 (ItchyQoL); in non-white patients this range was from 0.17 (EASI) to 0.6 (ItchyQoL). All studies evaluating convergent validity of PROMs by race and skin tone using correlation coefficients are presented in Table 2.
Table 2.
Correlation between outcome measure in studies that stratified analyses by race, ethnicity or skin tone.
| Outcome Measure | POEM | PO-SCORAD | EASI | oSCORAD | TIS | SGAxBSA |
|---|---|---|---|---|---|---|
| POEM | (Silverberg et al., 2020c) white (r=0.51) non-white (r=0.57) |
|||||
| PO-SCORAD | ||||||
| EASI | (Zhao et al., 2017) SOC (r=0.53), light skin (r=0.48) |
(Silverberg et al., 2020c) white (r=0.43) non-white(r=0.17) |
||||
| (Silverberg et al., 2020c) white (p=0.48) non-white (r=0.54) | ||||||
| oSCORAD | (Zhao et al., 2017) SOC (r=0.42) light skin (r=0.31) |
(Zhao et al., 2015) non-pigmented (r=1.00) mildly-pigmented (r=0.857) non-pigmented (r=0.867) |
||||
| TIS | (Zhao et al., 2015) non-pigmented (r=0.919) mildly-pigmented (r=0.829) non-pigmented (r=0.645) |
(Zhao et al., 2015) non-pigmented (r=0.919) mildly-pigmented (r=0.919) |
||||
| NRS worst-itch | (Silverberg et al., 2020c) white (r=0.42), non-white(r=0.48) |
(Silverberg et al., 2020c) white (r=0.57) non-white (r=0.34) |
||||
| NRS average-itch | (Silverberg et al., 2020c) white (r=0.48) non-white(r=0.51) |
(Silverberg et al., 2020c) white (r=0.58) non-white(r=0.35) |
(Topham et al., 2020 white (r=0.74) non-white (r=0.62) |
|||
| DLQI | (Silverberg et al., 2020c) white (r=0.65) non-white(r=0.72) |
(Silverberg et al., 2020c) white (r=0.57) non-white(r=0.48) |
||||
| ItchyQoL | (Silverberg et al., 2020c) white (r=0.65) non-white(r=0.74) |
(Silverberg et al., 2020c) white (r=0.68) non-white(r=0.60) |
||||
| PHQ9 | (Silverberg et al., 2020c) white (r=0.44) non-white(r=0.68) |
(Silverberg et al., 2020c) white (r=0.50) non-white(r=0.52) |
||||
| Promis SD | (Silverberg et al., 2020c) white (r=0.46) non-white(r=0.50) |
(Silverberg et al., 2020c) white (r=0.41) non-white(r=0.48) |
||||
| Promis SRI | (Silverberg et al., 2020c) white (r=0.41), non-white(r=0.56) |
(Silverberg et al., 2020c) white (r=0.52) non-white(r=0.49) |
||||
| Patient global assessment of severity | (Topham et al., 2020) white (r=0.80) non-white (r=0.63) |
|||||
| IGA | (Zhao et al., 2017) SOC (r=0.64) light skin (r=0.30) |
|||||
| Static physician global assessment | (Topham et al., 2020) white (r=0.77) non-white (r=0.73) |
|||||
| SASSAD | (Zhao et al., 2015) non-pigmented (r=0.786) * highly pigmented (p=0.051) |
(Zhao et al., 2015) non-pigmented (r=0.786)* non-pigmented (r=0.872) |
(Zhao et al., 2015) non-pigmented (r=0.793)* non-pigmented (r=0.648) |
** |
nonsignificant, not reported for highly pigmented
nonsignificant, not reported for mildly pigmented
Italic=significant difference between r(POEM and instrument) and r(POSCORAD and same instrument)
Bolded=same study as split cell above, but ratings were performed overseas by a different dermatologist (original study Australian, overseas=South African)
Feasibility
Three studies examined feasibility of PROMs by race.(Silverberg et al., 2020b, Silverberg et al., 2020c, Silverberg et al., 2020g) The mean time required to complete PIQ Itch-Triggers was 0.8 minutes, without significant difference by race.(Silverberg et al., 2020g) Mean time required to complete PO-SCORAD and POEM also did not significantly differ by race.(Silverberg et al., 2020c) Mean time required to complete PIQ short forms (for 4 banks) was 1.8 minutes, without significant difference by race.(Silverberg et al., 2020b)
Test-retest Reliability
One study compared test-retest reliability in whites vs. non-whites.(Silverberg et al., 2020c) Significantly higher ICC was observed for the original PO-SCORAD in whites (0.86, 95% CI 0.78-0.92) than non-whites (0.58, 95% CI 0.34-0.75). Non-significant differences were identified in the ICC for POEM in white (0.90, 95% CI 0.83-0.95) versus non-white patients (0.79, 95% CI 0.63-0.89).
Known-groups Validity
Two studies analyzed known groups validity by race.(Lei et al., 2020b, Vakharia P. P. et al., 2018) One study found that both whites and non-whites had significant associations between patient-reported AD severity and severity strata for objective-SCORAD, SCORAD, EASI, NRS-itch, POEM, and DLQI.(Vakharia P. P. et al., 2018) A study found significant and stepwise differences in PROMIS SD and SRI scores across severity groups of sleep frequency, self-reported global AD severity, and VRS worst and average itch even after adjusting for race (white/black/non-white Hispanic/Asian) in models.(Lei et al., 2020b)
ClinROMs
Convergent validity
Six studies collected data regarding convergent validity in ClinROMs.(Leshem et al., 2015a, Silverberg et al., 2020c, Topham et al., 2020, Vakharia P. P. et al., 2018, Zhao et al., 2017, Zhao et al., 2015) In a study of 291 adults with AD, the strong and significant correlation of EASI with objective-SCORAD (r>0.90) did not significantly differ across race (white, black, Hispanic, multiracial/other).(Silverberg et al., 2020c) Similarly, in another study of 170 AD patients strong and significant correlation of EASI with investigator global assessment (IGA) (r>0.90) was consistent in whites, Asians, and other race, and both Hispanic and non-Hispanic ethnicity.(Leshem et al., 2015a) A study of 388 AD patients found no significant correlations of EASI with oSCORAD in white, black, Hispanic, or multiracial individuals (p>0.05).(Vakharia P. P. et al., 2018)
Correlational effect size data was available for seven ClinROMs (EASI, oSCORAD, SGAxBSA, TIS, IGA, SPGA, SASSAD) for both white/nonwhite patients and SOC/light skin patients.(Silverberg et al., 2020c, Topham et al., 2020, Zhao et al., 2017, Zhao et al., 2015) EASI correlated weakly in white patients with both POEM (0.48) and POSCORAD (0.43). In nonwhite patients, EASI correlated moderately and weakly with POEM (0.54) and POSCORAD (0.17), respectively. All studies evaluating convergent validity of ClinROMs by race and skin tone using correlation coefficients are presented in Table 2.
Feasibility
Mean time for investigators to perform EASI was 6.0 minutes, which did not significantly differ based on patient race or Hispanic ethnicity.(Leshem et al., 2015a)
Intrarater reliability
Two studies investigated intrarater reliability across different skin pigmentation.(Zhao et al., 2017, Zhao et al., 2015) In a study of photographic assessment of 3 patients, multiple instruments had differential performance as measured by intra-class correlation coefficients (ICC) in patients with different clinician-determined skin pigmentation levels.(Zhao et al., 2015) SASSAD showed best reliability in highly (0.787) and mildly (0.458) pigmented patients while TIS (−0.434) and oSCORAD (−0.537) had the poorest reliability in these patient groups, respectively. EASI and TIS had the highest (0.83) and lowest (−0.76) ICCs in nonpigmented patients, respectively. There were no significant between or within pigmentation group differences.(Zhao et al., 2015)
Another small study (n=6) analyzed EASI, objective-SCORAD, and IGA.(Zhao et al., 2017) Patients were categorized into light skinned or skin of color (SOC) based on a machine measuring melanin content of gluteal skin.(Zhao et al., 2017) SOC patients were further stratified into intermediate or very dark skinned for certain analyses. All instruments showed excellent reliability with the lowest correlation in SOC seen for IGA (0.89) and lowest correlation in lighter skin for objective-SCORAD (0.87). No significant between-group or within-group differences were observed by pigment level. This study also reported psychometric findings for an ad-hoc scale examining greyness in SOC patients. Replacing greyscale with erythema in EASI or adding greyscale to EASI maintained comparable reliability metrics to the unmodified EASI. When SOC was stratified into intermediate- and dark-skinned subgroups and greyscale was independently evaluated, it performed best with very dark patient (0.88) and poorly in intermediate-skinned patients (0.33).(Zhao et al., 2017)
Interrater reliability
The same two studies investigated interrater reliability across different skin pigmentation.(Zhao et al., 2017, Zhao et al., 2015) In a sample of 18 patients, multiple instruments had differential performance as measured by ICC in patients with different clinician-assessed skin pigmentation levels. In highly pigmented individuals, EASI showed best ICC (−0.054) and TIS showed poorest ICC (−0.21). In mildly pigmented patients, oSCORAD showed best ICC (0.588) and SASSAD showed worst ICC (0.341). In non-pigmented patients, SASSAD showed best ICC (0.667) and TIS showed worst ICC (0.403). TIS, SASSAD, EASI, and oSCORAD had poor ICC in highly pigmented fair ICC in mildly pigmented patients. In non-pigmented patients, TIS and oSCORAD had fair correlations; EASI and SASSAD had good ICC.(Zhao et al., 2015)
EASI, objective-SCORAD, IGA and Greyscale from 11 lighter skin and 14 SOC patients were evaluated for inter-rater reliability.(Zhao et al., 2017) SOC was further stratified into intermediate and very dark patients. In lighter skin patients, EASI (0.83) and objective-SCORAD (0.68) were the best and worst performing instruments, respectively. EASI was also the best performing instrument for all categories of SOC, displaying best performance in the darkest skinned patients (SOC overall: 0.77, intermediate: 0.67, very dark 0.84).
Greyscale alone was the poorest performing instrument for all SOC categories, but in SOC patients when it replaced erythema in EASI (0.78) or was added to EASI (0.79) it did not produce significantly better results compared to original EASI. Otherwise, IGA was the poorest performer in all categories of SOC, especially for intermediate patients (0.56).(Zhao et al., 2017)
Although each instrument performed differently for each pigmentation level, these cross-racial differences were not statistically significant, likely due to the very small study sample sizes. EASI items assessing components other than erythema contributed significantly greater variability (assessed by coefficient of variance) to EASI inter-rater reliability scores than erythema itself in lighter skin patients. In SOC, variability in EASI inter-rater reliability was not significantly influenced by presence of erythema nor greyness items of EASI. Variability in oSCORAD inter-rater reliability was not explained by erythema in any pigmentation group.(Zhao et al., 2017)
DISCUSSION
This systematic review reveals significant reporting and knowledge gaps with respect to psychometric properties of outcome measures by race, ethnicity or skin tone in AD. The included studies ranged in quality, and only 47% or 18% of measurement properties evaluated using COSMIN and NOS scales were rated as very good or good, respectively (Tables 3 and 4). Most validation studies did not even mention the distribution of race, ethnicity or skin tone in their studies. The few studies examining psychometrics by race, ethnicity or skin tone used disparate methodologies to assess race, ethnicity and skin tone. Some studies of ClinROMs suffered from very small samples. Additionally, many studies that assessed psychometrics of outcome measures in non-white populations were performed in homogeneous patient populations in different countries. However, little is known about how well these measures perform, particularly ClinROMs, in diverse patient populations. There is insufficient evidence to determine optimal PROM and ClinROMs for SOC. Since AD may manifest differently in SOC, potential shortcomings of outcome measures can impact clinical care and research.(Brunner and Guttman-Yassky, 2019)
Table 3:
COSMIN Risk of Bias Scores
| Last name | Year published |
Box 1 |
Box 2 |
Box 3 |
Box 4 |
Box 5 |
Box 6 |
Box 7 |
Box 8 |
Box 9 |
Box 10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Dauden(Daudén et al.) | 2011 | - | - | 1 | 1 | 2 | 1 | - | - | - | - |
| Giovannini(Giovannini et al., 2020) | 2020 | 1 | - | - | - | - | 1 | - | 1 | - | - |
| Zhao(Zhao et al., 2017) | 2016 | - | - | - | - | 2 | 1 | - | 3 | 1 | - |
| Whalley(Whalley et al.) | 2004 | 2 | - | 1 | 1 | - | 2 | - | - | 1 | 1 |
| Zhao(Zhao et al., 2015) | 2015 | - | - | - | - | - | 1 | - | - | 0 | - |
| Borzutsky(Borzutzky et al., 2014) | 2014 | - | - | - | - | - | 3 | - | - | - | - |
| Beattie(Beattie and Lewis-Jones) | 2006 | - | - | - | - | - | - | - | - | 2 | - |
| Coutanceau(Coutanceau and Stalder, 2014) | 2014 | - | - | - | - | - | - | - | - | 1 | - |
| Marinello(Marinello et al., 2016) | 2016 | - | 2 | - | - | - | 2 | - | - | 2 | - |
| Bozek(Bożek and Reich, 2017) | 2017 | - | - | - | - | - | 2 | - | - | - | - |
| Silverberg(Silverberg et al., 2020g) | 2020 | - | - | - | - | - | - | - | - | - | - |
| Taieb(Taïeb et al., 2015) | 2015 | 2 | 3 | - | 3 | 2 | 2 | - | - | 2 | - |
| Meni(Méni et al., 2013) | 2013 | 2 | 3 | - | 2 | 2 | 2 | - | 3 | 3 | - |
| Darrigade(Darrigade et al., 2020) | 2018 | - | - | - | - | - | - | - | 1 | 1 | - |
| Arents(Arents et al., 2019) | 2019 | 2 | - | 1 | 1 | - | 1 | - | - | 1 | - |
| Twiss(Twiss et al.) | 2012 | - | - | 2 | - | - | - | - | - | - | - |
| Mitchell(Mitchell et al., 2017) | 2016 | 2 | - | 3 | 3 | - | - | - | - | 2 | - |
| Chamlin(Chamlin et al.) | 2007 | - | - | - | - | - | 2 | - | 1 | - | 3 |
| Aziah(Aziah et al.) | 2002 | - | - | - | - | - | 2 | - | - | - | - |
| Kunz(Kunz et al., 1997) | 1997 | - | - | - | - | - | 4 | - | - | - | - |
| Silverberg(Silverberg et al., 2020c) | 2020 | - | - | - | - | - | 2 | - | 2 | 2 | 2 |
| Chernyshov(Chernyshov et al., 2015) | 2015 | - | - | - | - | - | - | - | - | 3 | - |
| Silverberg(Silverberg et al., 2018a) | 2018 | 2 | - | - | 3 | 2 | - | - | 2 | 2 | - |
| Chernyshov(Chernyshov et al.) | 2018 | - | - | - | - | - | - | - | - | - | - |
| Bruscky(Bruscky et al., 2017) | 2017 | - | - | - | 4 | - | 2 | - | - | 1 | - |
| Alzolibani(Alzolibani) | 2013 | - | - | - | 3 | - | 2 | - | - | 1 | - |
| Finlay(Finlay and Khan) | 1994 | 3 | - | - | 4 | - | 2 | - | - | - | - |
| Willemsen(Willemsen et al., 2009) | 2009 | - | - | - | - | - | 1 | - | - | 2 | - |
| Foley(Foley et al.) | 2019 | 2 | 1 | - | - | - | 0 | - | - | - | - |
| Heckman(Heckman C.J. et al., 2020) | 2020 | - | - | - | 3 | - | 3 | - | 3 | 2 | - |
| He(He et al.) | 2014 | 3 | - | 2 | 2 | - | 2 | - | 2 | 2 | - |
| Heckman(Heckman CJ et al., 2020) | 2020 | 3 | - | - | - | - | 3 | - | 2 | 3 | 2 |
| Pereyra-Rodriguez(Pereyra-Rodriguez et al., 2019) | 2019 | 4 | - | - | 1 | - | 3 | - | - | 3 | - |
| Cheng(Cheng et al., 2020) | 2020 | - | - | - | - | - | - | - | 1 | 1 | 1 |
| Yamaguchi(Yamaguchi et al.) | 2016 | 4 | - | 4 | 1 | - | 3 | - | 1 | - | - |
| Gabes(Gabes et al., 2020a) | 2020 | 1 | 3 | 1 | 1 | - | 2 | 2 | - | - | - |
| Chamlin(Chamlin et al.) | 2005 | 1 | 3 | 1 | 1 | - | - | - | 1 | - | - |
| Schram(Schram et al.) | 2011 | - | - | - | - | - | - | 3 | - | - | - |
| Tremp(Tremp et al., 2011) | 2011 | - | - | - | - | - | - | 3 | - | - | - |
| Gabes(Gabes et al., 2020b) | 2019 | - | - | - | - | - | - | - | - | - | - |
| Newton(Newton et al., 2019) | 2019 | - | 1 | 1 | - | - | - | 3 | - | - | - |
| Augustin(Augustin et al., 2004) | 2004 | - | - | 1 | - | - | - | 3 | - | - | - |
| Charman(Charman and Varigos, 1999) | 1999 | - | - | 1 | - | - | - | - | - | - | - |
| Chernyshov(Chernyshov, 2016) | 2016 | 1 | - | 2 | 1 | - | 1 | - | - | - | - |
| Howells(Howells et al., 2018) | 2018 | - | - | 1 | - | - | - | 3 | - | - | - |
| Gabes(Gabes and Apfelbacher, 2020) | 2020 | - | 1 | - | - | - | - | - | - | - | - |
| Chren(Chren et al.) | 1997 | - | 1 | 2 | 1 | - | 2 | 1 | 2 | 2 | 2 |
| Topham(Topham et al., 2020) | 2020 | 1 | - | 2 | - | 2 | - | - | - | 2 | 2 |
| McKenna(McKenna et al.) | 2005 | 1 | 2 | 2 | 3 | - | 3 | - | 3 | 4 | - |
| Neri(Neri et al.) | 2012 | - | - | 2 | 3 | - | - | 1 | - | - | - |
| Hon(Hon et al.) | 2006 | - | - | - | - | - | - | - | 2 | - | - |
| Holm(Holm et al.) | 2006 | - | - | - | - | - | - | 3 | - | - | - |
| Charman(Charman et al.) | 1999 | - | - | - | - | - | 2 | - | - | - | - |
| Herd(Herd et al.) | 1997 | - | - | - | - | - | - | - | 3 | - | - |
| Lei(Lei et al., 2020a) | 2020 | - | - | - | - | 2 | 2 | - | 2 | 2 | - |
| Silverberg(Silverberg et al., 2019a) | 2019 | - | - | - | 2 | 2 | - | - | - | 3 | - |
| Silverberg(Silverberg et al., 2020a) | 2020 | - | 2 | - | 2 | 2 | 2 | 2 | 2 | 3 | - |
| Silverberg(Silverberg et al., 2020b) | 2020 | - | 2 | - | 2 | 2 | - | - | 2 | 3 | - |
| Silverberg(Silverberg et al., 2020d) | 2020 | - | - | - | - | - | 1 | 2 | 1 | 3 | 1 |
| Silverberg(Silverberg et al., 2020e) | 2020 | - | - | - | - | 3 | 1 | 2 | 1 | 2 | 2 |
| Patel(Patel et al., 2019) | 2019 | - | 2 | - | 2 | 1 | 1 | - | - | 2 | 1 |
| Charman(Charman et al.) | 2005 | - | - | - | - | - | - | - | 2 | - | - |
| Badia(Badia et al.) | 1999 | - | 3 | - | 3 | - | 2 | - | - | - | 1 |
| Cheng(Cheng et al., 2019) | 2019 | - | - | - | 3 | - | 2 | - | - | 3 | - |
| Darsow(Darsow et al.) | 2001 | - | - | 2 | - | - | - | - | - | - | - |
| Kido-Nakahara(Kido-Nakahara et al., 2020) | 2020 | - | - | - | - | - | 2 | - | - | 3 | - |
| Stalder(Stalder et al.) | 2011 | - | - | - | - | 4 | - | - | - | 1 | - |
| Vourch'h-Jourdain(Vourc'h-Jourdain et al., 2009) | 2009 | 3 | 2 | - | - | - | - | - | - | 1 | - |
| Fishbein(Fishbein et al., 2020) | 2020 | - | - | - | - | - | 4 | - | - | 1 | - |
| Yosipovitch(Yosipovitch et al., 2019) | 2019 | - | 1 | - | - | - | 1 | 1 | - | 1 | 1 |
| Deon(Deon et al.) | 2011 | - | - | - | 1 | - | 1 | - | - | 1 | - |
| Suh(Suh et al., 2020) | 2020 | - | - | - | - | - | - | - | - | 1 | - |
| Van Valburg(van Valburg et al.) | 2011 | - | - | - | - | - | 1 | - | - | 1 | - |
| Bahmer(Bahmer et al.) | 1991 | - | - | - | - | - | - | - | - | - | - |
| Liu(Liu et al.) | 2016 | - | - | 1 | - | - | - | - | - | - | - |
| Chopra(Chopra et al.) | 2017 | - | - | - | - | - | - | - | - | - | - |
| Lee(Lee et al., 2018) | 2018 | - | - | - | 3 | - | 3 | 4 | 1 | - | 1 |
| Charman(Charman et al.) | 2002 | - | - | - | - | - | 2 | - | - | - | - |
| Mazzotti(Mazzotti et al., 2008) | 2008 | - | - | - | - | - | 3 | - | - | 1 | - |
| Simpson(Simpson et al., 2019) | 2019 | - | - | - | - | - | - | - | - | - | - |
| Pucci(Pucci et al.) | 2005 | - | - | - | 3 | - | - | - | - | - | - |
| Costa(Costa et al., 1989) | 1989 | - | - | - | - | - | 4 | - | - | 2 | - |
| Oranje(Oranje et al.) | 1997 | - | - | - | - | - | - | - | - | - | - |
| Wolkerstorfer(Wolkerstorfer et al.) | 1999 | - | - | - | - | - | 1 | - | - | 1 | - |
| Murao(Murao et al.) | 2004 | 3 | - | - | - | - | 1 | - | - | - | - |
| Sprikkelman(Sprikkelman et al.) | 1997 | - | - | - | - | - | - | 1 | - | - | - |
| Rullo(Rullo et al.) | 2008 | - | - | - | 1 | - | 3 | - | - | - | 1 |
| European Task Force on Atopic Dermatitis(1993) | 1993 | 3 | - | - | - | - | - | - | - | - | - |
| Chopra(Chopra et al., 2017) | 2017 | - | - | - | - | - | - | - | - | - | - |
| Vakharia(Vakharia PP et al., 2018b) | 2018 | - | - | - | - | - | - | - | - | - | - |
| Silverberg(Silverberg et al., 2018c) | 2018 | - | - | - | - | - | - | - | - | - | - |
| Berth-Jones(Berth-Jones) | 1996 | - | - | - | - | - | - | 3 | - | - | - |
| Baek(Baek et al., 2015) | 2015 | - | - | - | - | - | 2 | - | - | 1 | 1 |
| Martin(Martin et al., 2020) | 2020 | 1 | 1 | - | - | - | - | - | - | - | - |
| Carel(Carel et al.) | 2008 | - | - | - | - | - | - | - | - | 1 | - |
| Lewis-Jones(Lewis-Jones and Finlay) | 1995 | 3 | - | - | - | - | 2 | - | - | - | - |
| Charman(Charman et al., 1999) | 1999 | - | - | - | - | - | 1 | - | - | - | - |
| Hanifin(Hanifin et al.) | 2001 | - | - | - | - | - | 1 | - | - | - | - |
| Baars(Baars et al.) | 2005 | 3 | 1 | 1 | 1 | - | 1 | - | - | - | - |
| Lewis-Jones(Lewis-Jones et al.) | 2001 | 3 | - | - | - | - | 1 | - | - | 1 | 1 |
| Silny(Silny et al., 2005) | 2005 | 4 | - | - | - | - | - | - | - | - | - |
| Emerson(Emerson et al.) | 2000 | - | - | - | - | - | - | - | - | 1 | - |
| Angelova- Fischer(Angelova-Fischer et al.) |
2005 | - | - | - | - | - | - | - | - | - | - |
| Sugarman(Sugarman et al.) | 2003 | - | - | - | - | - | - | - | - | - | - |
| Gaunt(Gaunt et al., 2016) | 2016 | - | - | - | - | - | - | - | - | - | 1 |
| Charman(Charman et al.) | 2004 | 1 | 1 | - | 1 | - | 1 | 1 | - | 1 | - |
| Yew(Yew et al., 2020) | 2020 | - | - | - | - | - | - | - | - | - | - |
| Zhao(Zhao et al., 2015) | 2015 | - | - | - | - | 1 | 1 | - | - | 1 | - |
| Van Velsen(van Velsen et al.) | 2010 | - | - | - | - | - | - | - | - | 1 | - |
| Simpson(Simpson et al., 2020) | 2020 | - | 1 | - | - | - | 1 | - | - | - | - |
| Boleira(M et al., 2014) | 2014 | - | 3 | 4 | 4 | - | - | - | - | 1 | - |
| Housman(Housman et al.) | 2002 | - | - | - | - | - | - | - | - | - | - |
| Ganemo(Gånemo et al., 2016) | 2016 | - | - | - | 1 | - | - | - | - | 1 | 1 |
| Hon(Hon et al., 2019) | 2019 | - | - | - | - | - | - | - | - | 1 | - |
| Silverberg(Silverberg et al., 2019b) | 2019 | - | - | - | 1 | 1 | - | - | - | 1 | - |
| Ramirez-Anaya(Ramírez-Anaya et al.) | 2010 | - | 4 | - | 1 | - | 2 | - | 1 | - | - |
| Hon(Hon et al.) | 2003 | - | - | - | - | - | - | - | - | - | - |
| Silverberg(Silverberg et al., 2020h) | 2020 | - | - | - | 1 | 1 | - | - | 1 | 1 | - |
| Lei(Lei et al., 2020b) | 2019 | - | - | - | - | - | - | - | 1 | 1 | 1 |
| Vakharia(Vakharia PP et al., 2018a) | 2018 | - | - | - | - | - | - | - | - | 1 | 1 |
| Lei(Lei et al., 2020b) | 2020 | 2 | 1 | 2 | 1 | 2 | 1 | - | 1 | 1 | 1 |
| Udkoff(Udkoff and Silverberg, 2018) | 2018 | 1 | 1 | 2 | 1 | 2 | - | - | - | 1 | 1 |
| Chernyshov(Chernyshov et al., 2019) | 2019 | 1 | - | 1 | 1 | 1 | 1 | - | - | 1 | 1 |
| Barbier(Barbier et al.) | 2003 | - | - | - | 1 | - | 1 | 3 | 1 | 1 | 1 |
| Faye(Faye et al., 2020a) | 2019 | - | - | - | - | - | - | - | - | - | - |
| Mohammadi(Mohammadi et al., 2019) | 2019 | - | - | - | 2 | - | 3 | - | - | - | - |
| Silverberg(Silverberg et al., 2020f) | 2020 | - | - | - | - | - | - | - | - | - | - |
| Leshem(Leshem et al., 2015b) | 2015 | - | - | - | - | - | - | - | - | - | - |
| Charman(Charman et al., 2013) | 2013 | - | - | - | - | - | - | - | - | - | - |
| Hurault(Hurault et al., 2018) | 2018 | - | - | - | - | - | - | - | - | - | - |
-, not assessed in paper
1, very good
2, adequate
3, doubtful
4, inadequate
Table 4:
Newcastle Ottawa Scale Scores
| First Author | Year | Elements of Newcastle-Ottawa Scale | |||
|---|---|---|---|---|---|
| Selection (0-5) | Comparability (0-1) |
Outcome (0-3) | Total stars (0-9) | ||
| Cherynyshov(Chernyshov et al., 2013) | 2013 | 2 | 1 | 3 | 6 |
| Holm(Holm et al.) | 2006 | 3 | 1 | 3 | 7 |
| Hassab-el-Naby(Hassab-El-Naby et al.) | 2009 | 2 | 0 | 2 | 4 |
| Rea(Rea et al., 2018) | 2018 | 2 | 0 | 3 | 5 |
| El-Mongy(El-Mong et al.) | 2006 | 2 | 1 | 3 | 6 |
| Ring(Ring et al.) | 2019 | 1 | 0 | 3 | 4 |
| Hon(Hon et al., 2006) | 2006 | 2 | 0 | 3 | 5 |
| Cosickic(Cosickic et al., 2010) | 2010 | 2 | 0 | 3 | 5 |
| Al-buanain(Al-Buainain et al.) | 2009 | 2 | 0 | 3 | 5 |
| Hon(Hon et al.) | 2008 | 2 | 0 | 3 | 5 |
| Shim(Shim et al.) | 2011 | 2 | 0 | 3 | 5 |
| Yang(Yang et al.) | 2010 | 2 | 0 | 3 | 5 |
| Schafer(Schäfer et al.) | 1997 | 2 | 1 | 3 | 6 |
| Djurovic(Ražnatović Djurović et al., 2015) | 2015 | 2 | 0 | 3 | 5 |
| Maksimovic(Maksimović et al.) | 2012 | 0 | 0 | 3 | 3 |
| Andersen(Andersen et al., 2020) | 2020 | 3 | 1 | 3 | 7 |
| Campos(Campos et al., 2017) | 2017 | 1 | 0 | 3 | 4 |
| Alzolibani(Alzolibani, 2014) | 2014 | 2 | 0 | 3 | 5 |
| Weisshaar(Weisshaar et al., 2008) | 2008 | 0 | 1 | 3 | 4 |
| Vilsboll(Vilsbøll et al., 2020) | 2020 | 5 | 0 | 3 | 8 |
| Haeck(Haeck et al.) | 2012 | 0 | 0 | 3 | 3 |
| Ben-Gashir(Ben-Gashir et al.) | 2004 | 3 | 1 | 3 | 7 |
| Kim(Kim et al.) | 2012 | 2 | 0 | 3 | 5 |
| Lam(Lam) | 2010 | 3 | 1 | 3 | 7 |
| Mozaffari(Mozaffari et al.) | 2007 | 0 | 1 | 3 | 4 |
| Amaral(Amaral et al.) | 2012 | 2 | 0 | 3 | 5 |
| Pusitsek(Pustišek et al., 2016) | 2016 | 2 | 1 | 3 | 6 |
| Djurovic(Djurović et al., 2020) | 2020 | 2 | 0 | 3 | 5 |
| Ganemo(Gånemo et al., 2007) | 2007 | 2 | 0 | 3 | 5 |
| Monti(Monti et al.) | 2011 | 2 | 0 | 3 | 5 |
| Hachisuka(Hachisuka et al.) | 2009 | 2 | 0 | 3 | 5 |
| Verwimp(Verwimp et al.) | 1995 | 0 | 0 | 1 | 1 |
| Al Shobaili(Al Shobaili) | 2010 | 4 | 0 | 3 | 7 |
7-9 stars=high quality, 4-6 stars=fair quality, 0-3 stars=poor quality.
Significant DIF by race was found for one or more item of PO-SCORAD, PIQ-Short Forms, POEM, DLQI, HADS, ItchyQoL, 5D-itch, SF-12, NRS-itch. For DLQI, in particular, item 2, “…how embarrassed or self-conscious have you been because of your skin?” and item 10, “…how much of a problem has the treatment for your skin been, for example by making your home messy, or by taking up time?” exhibited significant DIF by race in two studies.(Patel et al., 2019, Silverberg et al., 2019b) Nonuniform DIF may be corrected by removal of the item in question.(Pallant and Tennant, 2007) Alternatively, DLQI scores should be examined separately for patients in different racial/ethnic subgroups and should not be pooled together. This is concerning because DLQI was recently selected as the preferred outcome measure to assess QoL in AD. Though, it may not be optimal for use in diverse populations and non-white patients. PROMIS-SD and SRI were investigated in two studies; both showed no DIF for any items of either instrument.(Lei et al., 2020a, Lei et al., 2020b) These newer scales appear well suited to measure sleep specific impairment across races/ethnicities in AD.
Correlations of POEM with PHQ9 showed greatest differences between white and nonwhite patients (Δrho=0.24); correlations were moderate for non-whites and weak for whites.(Silverberg et al., 2020c) Subjective disease severity (POEM) may be more closely associated with the construct of depression (PHQ9) in nonwhite vs. white patients. Correlations of POEM with IGA differed the most between SOC and light skin (Δrho=0.34); the correlation was moderate for SOC and weak for light skinned patients.(Zhao et al., 2017) This suggest that IGA may better align with symptoms in SOC patients. Though, these observations should be interpreted with caution as they are from a single small-scale study.
Similar strengths of correlation (convergent validity) were observed for POEM with DLQI between white and nonwhite patients(Silverberg et al., 2020c) and POEM with EASI between white and nonwhite(Silverberg et al., 2020c) and light skin and SOC(Zhao et al., 2017) patients. These preliminary findings support the recommendations of the HOME to include POEM, EASI, and DLQI in the core outcome set of clinical trials. However, there is currently insufficient evidence to be confident about how well any PROMs or ClinROMs perform across different races, ethnicities, or skin tones in AD.
Differences of test-retest reliability by race were only tested for POEM and PO-SCORAD.(Silverberg et al., 2020c) The original PO-SCORAD showed significant test-retest reliability differences between white and nonwhite patients; an observation not seen for POEM.(Silverberg et al., 2020c) Recently, an updated version of PO-SCORAD was developed for black skin to address these limitations in SOC.(Faye et al., 2020b) There were no test-retest evaluations on the basis of race or skin tone for any other instruments.
EASI, SCORAD and other ClinROMs assess erythema using a spectrum of pinks and reds. In order to address difficulties with pigment masking of erythema in SOC, it is recommended by the HOME group to upcode erythema scores by one-level in SOC.(2017) It is unclear whether this guidance was used in the included studies of ClinROMs or whether such guidance is sufficient for properly assessing SOC. Erythema often appears in shades of purples and browns in AD patients with SOC, and can be difficult to assess reliably when patients have profound xerosis and lichenification. Results from a single small sample study suggest the erythema component of EASI can be supplemented with or replaced by an identically assessed clinical sign of greyness without compromising reliability; however, such a modification did not significantly improve EASI’s reliability,(Zhao et al., 2017) and has not undergone comprehensive psychometric testing. Guidance on how to score erythema and other AD signs should be expanded to address AD manifestations in SOC.
Race and ethnicity were inconsistently defined in included papers, presenting a barrier to standardizing categories for between-group analysis. Clinician assumptions about racial and ethnic identity may also be spurious. More importantly, different non-white races and ethnicities span a wide range from light to very dark skin tones.(Jothishankar and Stein, 2019) Thus, demonstration of ClinROM validity and reliability across different races does not mean that those tools perform well across all skin tones. It also does not obviate the need for studies specifically examining the psychometrics of ClinROMs across different skin types. Some validation studies included patients from one specific region or nation (e.g. Saudi Arabian children, Chinese adults). It is unclear whether these cohorts were diverse with respect to race, ethnicity or skin tone, and consequently whether the results are generalizable to other regions.
This study has several strengths, including comprehensive search strategy across multiple databases. Limitations include the relative dearth of studies specifically examining psychometrics across different races, ethnicities or skin types and the analysis of full-texts only accessible in English. Some studies of ClinROMs were underpowered to detect meaningful differences between patient subgroups. There were insufficient data to perform meta-regression of differences between patient subgroups. More studies are sorely needed to address these limitations.
METHODS
This study was exempt from institutional review board (IRB) approval because it only included published literature. This review was registered with PROSPERO (CRD42021239614). The review was carried out in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.(Liberati et al., 2009)
Literature Search
The following databases were searched through October 31, 2020: MEDLINE via PubMed (1996-present) and EMBASE (1947-present) for AD terms combined with terms regarding psychometric properties. Search strategies were modified from those previously used HOME group 2013 and 2019 systematic reviews of clinical sign instruments and quality-of-life instruments, respectively(Gabes et al., 2020c, Schmitt et al., 2013) (Appendix 1 and 2). The clinical signs search dates were (10-02-2012 to 10-30-2020) and the quality of life search dates were (1-23-2019 to 10-30-2020). Inclusion criteria are presented in Appendix 3.
Data Extraction
At least two authors (A.G., D.A., E.C., T.K, U.R) independently screened title/abstract and full-text articles, performed data extraction in duplicate, and performed quality or risk of bias assessment. Inconsistencies were resolved by consensus discussion. Additional references were identified by snowballing of all included articles, as well as excluded systematic reviews.
Data extracted included study design, validation or non-validation (such as correlational) study, demographic information, instruments analyzed, study participant breakdown by race, ethnicity, and skin type; and psychometric properties including content validity, construct validity, convergent validity, concurrent validity, inter-rater reliability, intra-rater reliability, responsiveness, and interpretability.
Quality and Risk of Bias Assessment
COSMIN Risk of Bias checklist was used to assess the methodological quality of validation studies. Newcastle Ottawa Scale (NOS) cross sectional adaptation was used to assess quality of non-validation non-randomized studies.(Mokkink et al., 2018),(Wells) We determined, a priori, that an NOS score of 7-9 stars would be considered high quality, 4-6 stars fair quality, and 0-3 stars poor quality.
Supplementary Material
Funding:
Emily Croce’s effort on this research was supported by the National Institute of Nursing Research of the National Institutes of Health under Award Number T32NR019035. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Emily Croce’s research was also supported by a Pfizer Dermatology Research Fellowship.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflicts of Interest:
Dr. Thyssen has been an advisor, Investigator and Speaker for Abbvie, Pfizer, LEO Pharma, Sanofi-Genzyme, Eli Lilly & Co and Regeneron. He has received grants from Sanofi-Genzyme and Regeneron.
Dr. Alexis declares Grants (funds to institution) - Leo, Novartis, Almirall, Bristol-Myers-Squibb, Amgen, Menlo, Galderma, Valeant (Bausch Health), Cara, Arcutis. Consultant/Advisory Board - Leo, Galderma, Pfizer, Sanofi-Regeneron, Dermavant, Beiersdorf, Valeant, L’Oreal, BMS, Bausch health , UCB, Vyne , Arcutis, Janssen, Allergan, Almirall, Abbvie, Sol-Gel, Amgen. Speaker - Regeneron, SANOFI-Genzyme, Pfizer, Astra Zeneca
The remaining authors state no conflict of interest.
Data availability:
All data were collected from the published literature. No datasets were generated during the current study.
References
- Severity scoring of atopic dermatitis: the SCORAD index. Consensus Report of the European Task Force on Atopic Dermatitis. Dermatology 1993;186(1):23–31. [DOI] [PubMed] [Google Scholar]
- EASI for clinical signs. HOME For Eczema: HOME For Eczema; 2017. [Google Scholar]
- Al-Buainain H, Kholoud A, Mahmoud M, Allam M. Correlation between Quality of Life and Disease Severity in Children with Atopic Dermatitis in the State of Qatar. Panarab J;20. [Google Scholar]
- Al Shobaili HA. The impact of childhood atopic dermatitis on the patients' family. Pediatr Dermatol;27(6):618–23. [DOI] [PubMed] [Google Scholar]
- Alzolibani A Impact of atopic dermatitis on the quality of life of Saudi children. Saudi medical journal 2014;35(4):391–6. [PubMed] [Google Scholar]
- Alzolibani AA. Cultural adaptation of the Arabic version of the Infants Dermatitis Quality of Life Index. Saudi Med J;34(5):518–24. [PubMed] [Google Scholar]
- Amaral CS, March Mde F, Sant'Anna CC. Quality of life in children and teenagers with atopic dermatitis. An Bras Dermatol;87(5):717–23. [DOI] [PubMed] [Google Scholar]
- Andersen L, Nyel ME, Nyberg F. Higher self-reported severity of atopic dermatitis in adults is associated with poorer self-reported health-related quality of life in France, Germany, the U.K. and the U.S.A. Br J Dermatol 2020;182(5):1176–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angelova-Fischer I, Bauer A, Hipler UC, Petrov I, Kaz, jieva J, et al. The objective severity assessment of atopic dermatitis (OSAAD) score: validity, reliability and sensitivity in adult patients with atopic dermatitis. Br J Dermatol;153(4):767–73. [DOI] [PubMed] [Google Scholar]
- Arents BWM, Mensing U, Seitz IA, Wettemann N, Fink-Wagner AH, de Carlo G, et al. Atopic eczema score of emotional consequences—a questionnaire to assess emotional consequences of atopic eczema. Allergo J Int 2019;28(7):277–88. [Google Scholar]
- Augustin M, Wenninger K, Amon U, Schroth MJ, Küster W, Chren M, et al. German adaptation of the Skindex-29 questionnaire on quality of life in dermatology: validation and clinical results. Dermatology 2004;209(1):14–20. [DOI] [PubMed] [Google Scholar]
- Aziah MS, Rosnah T, Mardziah A, Norzila MZ. Childhood atopic dermatitis: a measurement of quality of life and family impact. Med J Malaysia;57(3):329–39. [PubMed] [Google Scholar]
- Baars RM, Atherton CI, Koopman HM, Bullinger M, Power M. The European DISABKIDS project: development of seven condition-specific modules to measure health related quality of life in children and adolescents. Health Qual Life Outcomes;3:70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badia X, Mascaró JM, Lozano R. Measuring health-related quality of life in patients with mild to moderate eczema and psoriasis: clinical validity, reliability and sensitivity to change of the DLQI. The Cavide Research Group. Br J Dermatol;141(4):698–702. [DOI] [PubMed] [Google Scholar]
- Baek J, Park C, Choi K, Yang Y, Lee S, Koh J, et al. The Atopic Dermatitis Antecubital Severity score: validity, reliability, and sensitivity to change in patients with atopic dermatitis. England 2015. 2015-December. 1382–9 p. [DOI] [PubMed] [Google Scholar]
- Bahmer FA, Schäfer J, Schubert HJ. Quantification of the extent and the severity of atopic dermatitis: the ADASI score. Arch Dermatol;127(8):1239–40. [PubMed] [Google Scholar]
- Barbier N, Paul C, Luger T, Allen R, De Prost Y, Papp K, et al. Validation of the Eczema Area and Severity Index for atopic dermatitis in a cohort of 1550 patients from the pimecrolimus cream 1% randomized controlled clinical trials programme. Br J Dermatol;150(1):96–102. [DOI] [PubMed] [Google Scholar]
- Beattie PE, Lewis-Jones MS. An audit of the impact of a consultation with a paediatric dermatology team on quality of life in infants with atopic eczema and their families: further validation of the Infants' Dermatitis Quality of Life Index and Dermatitis Family Impact score. Br J Dermatol;155(6):1249–55. [DOI] [PubMed] [Google Scholar]
- Ben-Gashir MA, Hay RJ. Reliance on erythema scores may mask severe atopic dermatitis in black children compared with their white counterparts. The British journal of dermatology 2002;147(5):920–5. [DOI] [PubMed] [Google Scholar]
- Ben-Gashir MA, Seed PT, Hay RJ. Quality of life and disease severity are correlated in children with atopic dermatitis. Br J Dermatol;150(2):284–90. [DOI] [PubMed] [Google Scholar]
- Berth-Jones J Six area, six sign atopic dermatitis (SASSAD) severity score: a simple system for monitoring disease activity in atopic dermatitis. Br J Dermatol;135:25–30. [DOI] [PubMed] [Google Scholar]
- Borzutzky A, Hoyos-Bachiloglu R, Navarrete-Dechent C, Vera-Kellet C, Silva-Valenzuela S, Cifuentes L, et al. Agreement between dermatologic and pediatric researchers in scoring of atopic dermatitis in children. Pediatric dermatology 2014;31(6):e118–9. [DOI] [PubMed] [Google Scholar]
- Bożek A, Reich A. Assessment of Intra- and Inter-Rater Reliability of Three Methods for Measuring Atopic Dermatitis Severity: EASI, Objective SCORAD, and IGA. Switzerland 2017. 2017. 16–22 p. [DOI] [PubMed] [Google Scholar]
- Brunner PM, Guttman-Yassky E. Racial differences in atopic dermatitis. Ann Allergy Asthma Immunol 2019;122(5):449–55. [DOI] [PubMed] [Google Scholar]
- Bruscky D, Melo A, Sarinho E. CROSS-CULTURAL ADAPTATION AND VALIDATION OF THE ITCHING SEVERITY SCALE IN CHILDREN AND ADOLESCENTS WITH ATOPIC DERMATITIS. Revista paulista de pediatria : orgao oficial da Sociedade de Pediatria de Sao Paulo 2017;35(3):244–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campos A, Araújo F, Santos M, Santos A, Pires C. IMPACT OF ATOPIC DERMATITIS ON THE QUALITY OF LIFE OF PEDIATRIC PATIENTS AND THEIR GUARDIANS. Revista paulista de pediatria : orgao oficial da Sociedade de Pediatria de Sao Paulo 2017;35(1):5–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carel K, Bratton DL, Miyazawa N, Gyorkos E, Kelsay K, Bender B, et al. The Atopic Dermatitis Quickscore (ADQ): validation of a new parent-administered atopic dermatitis scoring tool. Ann Allergy Asthma Immunol;101(5):500–7. [DOI] [PubMed] [Google Scholar]
- Chamlin SL, Cella D, Frieden IJ, Williams ML, Mancini AJ, Lai JS, et al. Development of the Childhood Atopic Dermatitis Impact Scale: initial validation of a quality-of-life measure for young children with atopic dermatitis and their families. J Invest Dermatol;125(6):1106–11. [DOI] [PubMed] [Google Scholar]
- Chamlin SL, Lai JS, Cella D, Frieden IJ, Williams ML, Mancini AJ, et al. Childhood Atopic Dermatitis Impact Scale: reliability, discriminative and concurrent validity, and responsiveness. Arch Dermatol;143(6):768–72. [DOI] [PubMed] [Google Scholar]
- Charman C, Venn A, Ravenscroft J, Williams H. Translating Patient-Oriented Eczema Measure (POEM) scores into clinical practice by suggesting severity strata derived using anchor-based methods. The British journal of dermatology 2013;169(6):1326–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Charman CR, Venn AJ, Williams H. Measuring atopic eczema severity visually: which variables are most important to patients? Arch Dermatol;141(9):1146–51; discussion 51. [DOI] [PubMed] [Google Scholar]
- Charman CR, Venn AJ, Williams HC. Measurement of body surface area involvement in atopic eczema: an impossible task? Br J Dermatol;140(1):109–11. [DOI] [PubMed] [Google Scholar]
- Charman CR, Venn AJ, Williams HC. The patient-oriented eczema measure: development and initial validation of a new tool for measuring atopic eczema severity from the patients' perspective. Arch Dermatol;140(12):1513–9. [DOI] [PubMed] [Google Scholar]
- Charman CR, Venn AJ, Williams HC. Reliability testing of the Six Area, Six Sign Atopic Dermatitis severity score. Br J Dermatol;146(6):1057–60. [DOI] [PubMed] [Google Scholar]
- Charman D, Varigos G, Horne DJ, Oberklaid F. The development of a practical and reliable assessment measure for atopic dermatitis (ADAM). J Outcome Meas 1999;3(1):21–34. [PubMed] [Google Scholar]
- Charman DP, Varigos GA. Grades of severity and the validation of an atopic dermatitis assessment measure (ADAM). J Outcome Meas 1999;3(2):162–75. [PubMed] [Google Scholar]
- Cheng NS, Chau PCJ, Hon KLE, Choi KC, Kung JSC, Ng WG, et al. Measuring the quality of life of the families of children with eczema in Hong Kong. Asia Pacific Allergy 2019;9(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng R, Zhang H, Zong W, Tang J, Han X, Zhang L, et al. Development and validation of new diagnostic criteria for atopic dermatitis in children of China. J Eur Acad Dermatol Venereol 2020;34(3):542–8. [DOI] [PubMed] [Google Scholar]
- Chernyshov P Health-related quality of life in adult atopic dermatitis and psoriatic patients matched by disease severity. Giornale italiano di dermatologia e venereologia : organo ufficiale, Societa italiana di dermatologia e sifilografia 2016;151(1):37–43. [PubMed] [Google Scholar]
- Chernyshov P, Jirakova A, Ho R, Moed H, Caldeira A, Alvarenga T, et al. An international multicenter study on quality of life and family quality of life in children with atopic dermatitis. Indian journal of dermatology, venereology and leprology 2013;79(1):52–8. [DOI] [PubMed] [Google Scholar]
- Chernyshov P, Kaliuzhna L, Reznikova A, Basra M. Comparison of the impairment of family quality of life assessed by disease-specific and dermatology-specific instruments in children with atopic dermatitis. England 2015. 2015-June. 1221–4 p. [DOI] [PubMed] [Google Scholar]
- Chernyshov PV, Boffa MJ, Corso R, Pustišek N, Marinovic B, Manolache L, et al. Creation and pilot test results of the dermatology-specific proxy instrument: the Infants and Toddlers Dermatology Quality of Life. J Eur Acad Dermatol Venereol;32(12):2288–94. [DOI] [PubMed] [Google Scholar]
- Chernyshov PV, Sampogna F, Pustišek N, Marinovic B, Manolache L, Suru A, et al. Validation of the dermatology-specific proxy instrument the Infants and Toddlers Dermatology Quality of Life. J Eur Acad Dermatol Venereol 2019;33(7):1405–11. [DOI] [PubMed] [Google Scholar]
- Chopra R, Vakharia P, Sacotte R, Patel N, Immaneni S, White T, et al. Severity strata for Eczema Area and Severity Index (EASI), modified EASI, Scoring Atopic Dermatitis (SCORAD), objective SCORAD, Atopic Dermatitis Severity Index and body surface area in adolescents and adults with atopic dermatitis. The British journal of dermatology 2017;177(5):1316–21. [DOI] [PubMed] [Google Scholar]
- Chopra R, Vakharia PP, Sacotte R, Patel N, Immaneni S, White T, et al. Relationship between EASI and SCORAD severity assessments for atopic dermatitis. J Allergy Clin Immunol;140(6):1708–10.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chren MM, Lasek RJ, Flocke SA, Zyzanski SJ. Improved discriminative and evaluative capability of a refined version of Skindex, a quality-of-life instrument for patients with skin diseases. Arch Dermatol;133(11):1433–40. [PubMed] [Google Scholar]
- Cosickic A, Skokic F, Colic-Hadzic B, Jahic M. Clinical characteristics and estimation severity of the atopic dermatitis in children. Med Arh 2010;64(3):178–82. [PubMed] [Google Scholar]
- Costa C, Rilliet A, Nicolet M, Saurat JH. Scoring atopic dermatitis: the simpler the better? Acta Derm Venereol 1989;69(1):41–5. [PubMed] [Google Scholar]
- Coutanceau C, Stalder JF. Analysis of correlations between patient-oriented SCORAD (PO-SCORAD) and other assessment scores of atopic dermatitis severity and quality of life. Dermatology 2014;229(3):248–55. [DOI] [PubMed] [Google Scholar]
- Darrigade A-S, Colmant C, de Montjoye L, Herman A, Bugli C, Tromme I, et al. Atopic Dermatitis Score 7 (ADS7): A promising tool for daily clinical assessment of atopic dermatitis. Allergy Eur J Allergy Clin Immunol 2020;75(5):1264–6. [DOI] [PubMed] [Google Scholar]
- Darsow U, Scharein E, Simon D, Walter G, Bromm B, Ring J. New aspects of itch pathophysiology: component analysis of atopic itch using the 'Eppendorf Itch Questionnaire'. Int Arch Allergy Immunol;124(1):326–31. [DOI] [PubMed] [Google Scholar]
- Daudén E, Sánchez-Perez J, Prieto M, Roset M. [Validation of the Spanish Version of the Itch Severity Scale: the PSEDA study]. Actas Dermosifiliogr;102(7):527–36. [DOI] [PubMed] [Google Scholar]
- Deon KC, Santos DM, Bullinger M, Santos CB. Preliminary psycometric assessment of the Brazilian version of the DISABKIDS Atopic Dermatitis Module. Rev Saude Publica;45(6):1072–8. [DOI] [PubMed] [Google Scholar]
- Djurović MR, Janković J, Ćirković A, Spirić VT, Maksimović N, Timotijević ZS, et al. Quality of life in infants with atopic dermatitis and their families. Postepy Dermatol Alergol 2020;37(1):66–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Mong S, Ahmed E-S, el-bahaey W. Atopic Dermatitis: Relation Between Disease Severity, Anxiety and Quality of Life in Children and Adults. Egyptian Dermatol Online J;2. [Google Scholar]
- Emerson RM, Charman CR, Williams HC. The Nottingham Eczema Severity Score: preliminary refinement of the Rajka and Langeland grading. Br J Dermatol;142(2):288–97. [DOI] [PubMed] [Google Scholar]
- Faye O, Meledie N'Djong AP, Diadie S, Coniquet S, Niamba PA, Atadokpede F, et al. Validation of the Patient-Oriented SCORing for Atopic Dermatitis tool for black skin. J Eur Acad Dermatol Venereol 2020a;34(4):795–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faye O, Meledie N'Djong AP, Diadie S, Coniquet S, Niamba PA, Atadokpede F, et al. Validation of the Patient-Oriented SCORing for Atopic Dermatitis tool for black skin. Journal of the European Academy of Dermatology and Venereology 2020b;34(4):795–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finlay AY, Khan GK. Dermatology Life Quality Index (DLQI)--a simple practical measure for routine clinical use. Clin Exp Dermatol;19(3):210–6. [DOI] [PubMed] [Google Scholar]
- Fishbein A, Lor J, Penedo F, Forrest C, Griffith J, Paller A. Patient-Reported Outcomes for Measuring Sleep Disturbance in Pediatric Atopic Dermatitis: cross sectional study of PROMIS Pediatric Sleep Measures and Actigraphy. Journal of the American Academy of Dermatology 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foley C, Tundia N, Simpson E, Teixeira HD, Litcher-Kelly L, Bodhani A. Development and content validity of new patient-reported outcome questionnaires to assess the signs and symptoms and impact of atopic dermatitis: the Atopic Dermatitis Symptom Scale (ADerm-SS) and the Atopic Dermatitis Impact Scale (ADerm-IS). Curr Med Res Opin;35(7):1139–48. [DOI] [PubMed] [Google Scholar]
- Gabes M, Apfelbacher C. IDQoL, CDLQI and the 45-item CADIS received a sufficient content validity rating during the HOME VII meeting in Japan: a group discussion study. J Eur Acad Dermatol Venereol 2020. [DOI] [PubMed] [Google Scholar]
- Gabes M, Chamlin SL, Lai J-S, Cella D, Mancini AJ, Apfelbacher CJ. Development of a validated short-form of the Childhood Atopic Dermatitis Impact Scale, the CADIS-SF15. J Eur Acad Dermatol Venereol 2020a;34(8):1773–8. [DOI] [PubMed] [Google Scholar]
- Gabes M, Chamlin SL, Lai J-S, Cella D, Mancini AJ, Apfelbacher CJ. Evaluation of responsiveness and estimation of smallest detectable change and minimal important change scores for the Childhood Atopic Dermatitis Impact Scale. Br J Dermatol 2020b;182(2):348–54. [DOI] [PubMed] [Google Scholar]
- Gabes M, Tischer C, Apfelbacher C. Measurement properties of quality-of-life outcome measures for children and adults with eczema: An updated systematic review. Pediatr Allergy Immunol 2020c;31(1):66–77. [DOI] [PubMed] [Google Scholar]
- Gånemo A, Svensson A, Lindberg M, Wahlgren CF. Quality of life in Swedish children with eczema. Acta Derm Venereol 2007;87(4):345–9. [DOI] [PubMed] [Google Scholar]
- Gånemo A, Svensson A, Svedman C, Grönberg B, Johansson A, Wahlgren C. Usefulness of Rajka & Langeland Eczema Severity Score in Clinical Practice. Acta dermato-venereologica 2016;96(4):521–4. [DOI] [PubMed] [Google Scholar]
- Gaunt D, Metcalfe C, Ridd M. The Patient-Oriented Eczema Measure in young children: responsiveness and minimal clinically important difference. Allergy 2016;71(11):1620–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giovannini M, Geat D, Barlocco G, Pertile R, Mori F, Filippeschi C, et al. A caregiver-reported global severity assessment in pediatric atopic eczema: The Comano score. Ital J Pediatr 2020;46(1). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hachisuka J, Takeuchi S, Kido M, Fukiwake N, Furue M. Severity of disease, rather than xerosis, correlates with pruritus in patients with atopic dermatitis. Int J Dermatol;48(4):374–8. [DOI] [PubMed] [Google Scholar]
- Haeck IM, ten Berge O, van Velsen SG, de Bruin-Weller MS, Bruijnzeel-Koomen CA, Knol MJ. Moderate correlation between quality of life and disease activity in adult patients with atopic dermatitis. J Eur Acad Dermatol Venereol;26(2):236–41. [DOI] [PubMed] [Google Scholar]
- Hanifin JM, Thurston M, Omoto M, Cherill R, Tofte SJ, Graeber M. The eczema area and severity index (EASI): assessment of reliability in atopic dermatitis. EASI Evaluator Group. Exp Dermatol;10(1):11–8. [DOI] [PubMed] [Google Scholar]
- Hassab-El-Naby H, Mohamed Y, Ammar M, Mostafa A. Assessment of quality of life among school children with atopic dermatitis in a locality in Cairo. Journal of the Egyptian Women’s Dermatologic Society;8:7–10. [Google Scholar]
- He Z, Lu C, Chren MM, Zhang Z, Li Y, Ni X, et al. Development and psychometric validation of the Chinese version of Skindex-29 and Skindex-16. Health Qual Life Outcomes;12:190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heckman C, Schut C, Riley M, Ehlers A, Valdes-Rodriguez R, Kupfer J, et al. Development and psychometrics of the English version of the Itch Cognitions Questionnaire. PloS one 2020;15(3):e0230585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heckman CJ, Riley M, Valdes-Rodriguez R, Yosipovitch G. Development and Initial Psychometric Properties of Two Itch-Related Measures: Scratch Intensity and Impact, Sleep-Related Itch and Scratch. J Invest Dermatol 2020;140(11):2138–45.e1. [DOI] [PubMed] [Google Scholar]
- Herd RM, Tidman MJ, Ruta DA, Hunter JA. Measurement of quality of life in atopic dermatitis: correlation and validation of two different methods. Br J Dermatol;136(4):502–7. [PubMed] [Google Scholar]
- Holm EA, Wulf HC, Stegmann H, Jemec GB. Life quality assessment among patients with atopic eczema. Br J Dermatol;154(4):719–25. [DOI] [PubMed] [Google Scholar]
- Holm EA, Wulf HC, Thomassen L, Jemec GB. Assessment of atopic eczema: clinical scoring and noninvasive measurements. Br J Dermatol;157(4):674–80. [DOI] [PubMed] [Google Scholar]
- Hon KL, Leung TF, Wong KY, Chow CM, Chuh A, Ng PC. Does age or gender influence quality of life in children with atopic dermatitis? Clin Exp Dermatol;33(6):705–9. [DOI] [PubMed] [Google Scholar]
- Hon KL, Leung TF, Wong Y, Fok TF. Lesson from performing SCORADs in children with atopic dermatitis: subjective symptoms do not correlate well with disease extent or intensity. Int J Dermatol;45(6):728–30. [DOI] [PubMed] [Google Scholar]
- Hon KL, Ma KC, Wong E, Leung TF, Wong Y, Fok TF. Validation of a self-administered questionnaire in Chinese in the assessment of eczema severity. Pediatr Dermatol;20(6):465–9. [DOI] [PubMed] [Google Scholar]
- Hon KL, Ng WGG, Kung JSC, Cheng NS, Ng DKK, Leung TF. Utility of the Pediatric Allergic Disease Quality of Life Questionnaire for childhood eczema. Br J Dermatol 2019;181(2):290–5. [DOI] [PubMed] [Google Scholar]
- Hon KLE, Kam WYC, Lam MCA, Leung TF, Ng PC. CDLQI, Scorad and Ness: Are they Correlated? Quality of Life Research 2006;15(10):1551. [DOI] [PubMed] [Google Scholar]
- Housman TS, Patel MJ, Camacho F, Feldman SR, Fleischer AB Jr., Balkrishnan R. Use of the Self-Administered Eczema Area and Severity Index by parent caregivers: results of a validation study. Br J Dermatol;147(6):1192–8. [DOI] [PubMed] [Google Scholar]
- Howells L, Ratib S, Chalmers J, Bradshaw L, Thomas K. How should minimally important change scores for the Patient-Oriented Eczema Measure be interpreted? A validation using varied methods. The British journal of dermatology 2018;178(5):1135–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurault G, Schram ME, Roekevisch E, Spuls PI, Tanaka RJ. Relationship and probabilistic stratification of Eczema Area and Severity Index and objective Scoring Atopic Dermatitis severity scores for atopic dermatitis. Br J Dermatol 2018;179(4):1003–5. [DOI] [PubMed] [Google Scholar]
- Jothishankar B, Stein SL. Impact of skin color and ethnicity. Clin Dermatol 2019;37(5):418–29. [DOI] [PubMed] [Google Scholar]
- Kaufman BP, Guttman-Yassky E, Alexis AF. Atopic dermatitis in diverse racial and ethnic groups-Variations in epidemiology, genetics, clinical presentation and treatment. Exp Dermatol 2018;27(4):340–57. [DOI] [PubMed] [Google Scholar]
- Kido-Nakahara M, Nakahara T, Yasukochi Y, Ulzii D, Furue M. Patient-oriented eczema measure score: A useful tool for web-based surveys in patients with atopic dermatitis. Acta Derm-Venereol 2020;100(10):1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim DH, Li K, Seo SJ, Jo SJ, Yim HW, Kim CM, et al. Quality of life and disease severity are correlated in patients with atopic dermatitis. J Korean Med Sci;27(11):1327–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunz B, Oranje AP, Labrèze L, Stalder JF, Ring J, Taïeb A. Clinical validation and guidelines for the SCORAD index: consensus report of the European Task Force on Atopic Dermatitis. Dermatology 1997;195(1):10–9. [DOI] [PubMed] [Google Scholar]
- Lam K Quality of life assessment among patients with atopic eczema attending dermatology clinics in Hong Kong. Hong Kong J Dermatol Venereol;18. [Google Scholar]
- Lee J, Kim MB, Yang H, Kim H, Cho J, Kim Y, et al. Reliability and validity of the Atopic Dermatitis Symptom Score (ADSS). Pediatric allergy and immunology : official publication of the European Society of Pediatric Allergy and Immunology 2018;29(3):290–5. [DOI] [PubMed] [Google Scholar]
- Lei DK, Yousaf M, Janmohamed SR, Vakharia PP, Chopra R, Chavda R, et al. Measurement Properties of 4 Patient-Reported Outcome Measures to Assess Sleep Disturbance in Adults With Atopic Dermatitis. Dermatitis : contact, atopic, occupational, drug 2020a;31(5):321–7. [DOI] [PubMed] [Google Scholar]
- Lei DK, Yousaf M, Janmohamed SR, Vakharia PP, Chopra R, Sacotte R, et al. Validation of Patient-Reported Outcomes Information System Sleep Disturbance and Sleep-Related Impairment in adults with atopic dermatitis. Br J Dermatol 2020b. [DOI] [PubMed] [Google Scholar]
- Leshem Y, Hajar T, Hanifin J, Simpson E. What the Eczema Area and Severity Index score tells us about the severity of atopic dermatitis: an interpretability study. England 2015a. 2015. 1353–7 p. [DOI] [PubMed] [Google Scholar]
- Leshem Y, T H, JM H, EL S. What the Eczema Area and Severity Index score tells us about the severity of atopic dermatitis: an interpretability study. England 2015b. 2015. 1353–7 p. [DOI] [PubMed] [Google Scholar]
- Lewis-Jones MS, Finlay AY. The Children's Dermatology Life Quality Index (CDLQI): initial validation and practical use. Br J Dermatol;132(6):942–9. [DOI] [PubMed] [Google Scholar]
- Lewis-Jones MS, Finlay AY, Dykes PJ. The Infants' Dermatitis Quality of Life Index. Br J Dermatol;144(1):104–10. [DOI] [PubMed] [Google Scholar]
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JPA, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 2009;339:b2700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Li T, An J, Zeng W, Xiao S. Rasch analysis holds no brief for the use of the Dermatology Life Quality Index (DLQI) in Chinese neurodermatitis patients. Health Qual Life Outcomes;14:17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- M B, O L, GV P, G D, AJ S, DB G. Translation and validation of Portuguese of a questionnaire for evaluation of psychosomatic symptoms in adults with atopic dermatitis. Anais brasileiros de dermatologia 2014;89(5):763–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maksimović N, Janković S, Marinković J, Sekulović LK, Zivković Z, Spirić VT. Health-related quality of life in patients with atopic dermatitis. J Dermatol;39(1):42–7. [DOI] [PubMed] [Google Scholar]
- Marinello E, Linder D, Spoto A, Palmer K, Rohrer P, Büchi S, et al. Assessing the Impact of Atopic Dermatitis on the Patients' Parents with the Visual Instrument 'Caregiver-PRISM'. Dermatology (Basel, Switzerland) 2016;232(4):490–5. [DOI] [PubMed] [Google Scholar]
- Martin SA, Brown TM, Fehnel S, Deal LS, Katz EG, Chiou C-F. The atopic dermatitis itch scale: development of a new measure to assess pruritus in patients with atopic dermatitis. J Dermatol Treat 2020;31(5):484–90. [DOI] [PubMed] [Google Scholar]
- Mazzotti E, Paradisi M, Provini A, Tabolli S. Reproducibility of severity rating for atopic dermatitis. Acta Derm Venereol 2008;88(1):60–1. [DOI] [PubMed] [Google Scholar]
- McKenna SP, Whalley D, Dewar AL, Erdman RA, Kohlmann T, Niero M, et al. International development of the Parents' Index of Quality of Life in Atopic Dermatitis (PIQoL-AD). Qual Life Res;14(1):231–41. [DOI] [PubMed] [Google Scholar]
- Méni C, Bodemer C, Toulon A, Merh, Perez-Cullell N, Branchoux S, et al. Atopic dermatitis burden scale: creation of a specific burden questionnaire for families. Journal of the European Academy of Dermatology and Venereology : JEADV 2013;27(11):1426–32. [DOI] [PubMed] [Google Scholar]
- Mitchell A, Morawska A, Fraser J, Sillar K. Child behaviour problems and childhood illness: development of the Eczema Behaviour Checklist. Child: care, health and development 2017;43(1):67–74. [DOI] [PubMed] [Google Scholar]
- Mohammadi S, Farajzadeh S, Safizadeh H, Khalili M, Aflatoonian M, Amiri R, et al. Validity and reliability of persian version of infants’ dermatitis quality of life index (IDQOL) questionnaire. Iran J Dermatol 2019;22(4):139–44. [Google Scholar]
- Mokkink LB, de Vet HCW, Prinsen CAC, Patrick DL, Alonso J, Bouter LM, et al. COSMIN Risk of Bias checklist for systematic reviews of Patient-Reported Outcome Measures. Qual Life Res 2018;27(5):1171–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monti F, Agostini F, Gobbi F, Neri E, Schianchi S, Arcangeli F. Quality of life measures in Italian children with atopic dermatitis and their families. Ital J Pediatr;37:59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mozaffari H, Pourpak Z, Pourseyed S, Farhoodi A, Aghamohammadi A, Movahadi M, et al. Quality of life in atopic dermatitis patients. J Microbiol Immunol Infect;40(3):260–4. [PubMed] [Google Scholar]
- Murao Y, Mae H, Konishi T, Gao X, Tanizawa T. Semiquantitative scoring in children under treatment for atopic dermatitis. Pediatr Int;46(3):330–6. [DOI] [PubMed] [Google Scholar]
- Neri E, Agostini F, Gremigni P, Gobbi F, Casu G, Chamlin SL, et al. Italian validation of the Childhood Atopic Dermatitis Impact Scale: a contribution to its clinical application. J Invest Dermatol;132(11):2534–43. [DOI] [PubMed] [Google Scholar]
- Newton L, DeLozier A, Griffiths P, Hill J, Hudgens S, Symonds T, et al. Exploring content and psychometric validity of newly developed assessment tools for itch and skin pain in atopic dermatitis. Journal of patient-reported outcomes 2019;3(1):42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oranje AP, Stalder JF, Taïeb A, Tasset C, de Longueville M. Scoring of atopic dermatitis by SCORAD using a training atlas by investigators from different disciplines. ETAC Study Group. Early Treatment of the Atopic Child. Pediatr Allergy Immunol;8(1):28–34. [DOI] [PubMed] [Google Scholar]
- Pallant JF, Tennant A. An introduction to the Rasch measurement model: an example using the Hospital Anxiety and Depression Scale (HADS). Br J Clin Psychol 2007;46(Pt 1):1–18. [DOI] [PubMed] [Google Scholar]
- Patel KR, Singam V, Vakharia PP, Chopra R, Sacotte R, Patel N, et al. Measurement properties of three assessments of burden used in atopic dermatitis in adults. Br J Dermatol 2019;180(5):1083–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pereyra-Rodriguez JJ, Dominguez-Cruz JJ, Hern, ez-Montoya C, Galan-Gutiérrez M, Carrasco PM, et al. Development and validation of a questionnaire to measure empowerment in adult patients with atopic dermatitis. The DATEMP questionnaire. JDDG J German Soc Dermatol 2019;17(9):923–31. [DOI] [PubMed] [Google Scholar]
- Pucci N, Novembre E, Cammarata MG, Bernardini R, Monaco MG, Calogero C, et al. Scoring atopic dermatitis in infants and young children: distinctive features of the SCORAD index. Allergy;60(1):113–6. [DOI] [PubMed] [Google Scholar]
- Pustišek N, Vurnek Živković M, Šitum M. Quality of Life in Families with Children with Atopic Dermatitis. Pediatric dermatology 2016;33(1):28–32. [DOI] [PubMed] [Google Scholar]
- Ramírez-Anaya M, Macías ME, Velázquez-González E. Validation of a Mexican Spanish version of the Children's Dermatology Life Quality Index. Pediatr Dermatol;27(2):143–7. [DOI] [PubMed] [Google Scholar]
- Ražnatović Djurović M, Janković J, Tomić Spirić V, Janković S. Health-related Quality of Life in Children with Moderate to Severe Atopic Dermatitis. Acta Dermatovenerol Croat 2015;23(3):178–84. [PubMed] [Google Scholar]
- Rea CJ, Tran KD, Jorina M, Wenren LM, Hawryluk EB, Toomey SL. Associations of Eczema Severity and Parent Knowledge With Child Quality of Life in a Pediatric Primary Care Population. Clin Pediatr (Phila) 2018;57(13):1506–14. [DOI] [PubMed] [Google Scholar]
- Ring J, Zink A, Arents BWM, Seitz IA, Mensing U, Schielein MC, et al. Atopic eczema: burden of disease and individual suffering - results from a large EU study in adults. J Eur Acad Dermatol Venereol;33(7):1331–40. [DOI] [PubMed] [Google Scholar]
- Rullo VE, Segato A, Kirsh A, Sole D. Severity scoring of atopic dermatitis: a comparison of two scoring systems. Allergol Immunopathol (Madr);36(4):205–11. [PubMed] [Google Scholar]
- Schäfer T, Dockery D, Krämer U, Behrendt H, Ring J. Experiences with the severity scoring of atopic dermatitis in a population of German pre-school children. Br J Dermatol;137(4):558–62. [DOI] [PubMed] [Google Scholar]
- Schmitt J, Langan S, Deckert S, Svensson A, von Kobyletzki L, Thomas K, et al. Assessment of clinical signs of atopic dermatitis: a systematic review and recommendation. J Allergy Clin Immunol 2013;132(6):1337–47. [DOI] [PubMed] [Google Scholar]
- Schram ME, Spuls PI, Leeflang MM, Lindeboom R, Bos JD, Schmitt J. EASI, (objective) SCORAD and POEM for atopic eczema: responsiveness and minimal clinically important difference. Allergy;67(1):99–106. [DOI] [PubMed] [Google Scholar]
- Shim WH, Park HJ, Kim HS, Kim SH, Ko HC, Kim MB, et al. Does the EASI score reflect itch severity? Ann Allergy Asthma Immunol;106(6):540–1. [DOI] [PubMed] [Google Scholar]
- Silny W, Czarnecka-Operacz M, Silny P. The new scoring system for evaluation of skin inflammation extent and severity in patients with atopic dermatitis. Acta Dermatovenerol Croat 2005;13(4):219–24. [PubMed] [Google Scholar]
- Silverberg J, Chiesa Fuxench Z, Gelf J, Margolis D, Boguniewicz M, Fonacier L, et al. Content and construct validity, predictors, and distribution of self-reported atopic dermatitis severity in US adults. Annals of allergy, asthma & immunology : official publication of the American College of Allergy, Asthma, & Immunology 2018a;121(6):729–34.e4. [DOI] [PubMed] [Google Scholar]
- Silverberg J, Chiesa Fuxench Z, Gelf J, Margolis J, Boguniewicz M, Fonacier L, et al. Content and construct validity, predictors, and distribution of self-reported atopic dermatitis severity in US adults. Annals of allergy, asthma & immunology : official publication of the American College of Allergy, Asthma, & Immunology 2018b;121(6):729–34.e4. [DOI] [PubMed] [Google Scholar]
- Silverberg J, Gelf J, Margolis D, Fonacier L, Boguniewicz M, Schwartz L, et al. Severity strata for POEM, PO-SCORAD, and DLQI in US adults with atopic dermatitis. Annals of allergy, asthma & immunology : official publication of the American College of Allergy, Asthma, & Immunology 2018c;121(4):464–8.e3. [DOI] [PubMed] [Google Scholar]
- Silverberg JI, Gelf J, Margolis D, Boguniewicz M, Fonacier LS, et al. Measurement properties of Hospital Anxiety and Depression Scale used in atopic dermatitis in adults. J Allergy Clin Immunol 2019a;143(2):AB130. [DOI] [PubMed] [Google Scholar]
- Silverberg JI, Gelf JM, Margolis DJ, Boguniewicz M, Fonacier L, et al. Validation and Interpretation of Short Form 12 and Comparison with Dermatology Life Quality Index in Atopic Dermatitis in Adults. J Invest Dermatol 2019b;139(10):2090–7.e3. [DOI] [PubMed] [Google Scholar]
- Silverberg JI, Lai J-S, Patel KR, Singam V, Vakharia PP, Chopra R, et al. Measurement properties of the Patient-Reported Outcomes Information System (PROMIS®) Itch Questionnaire: itch severity assessments in adults with atopic dermatitis. Br J Dermatol 2020a. [DOI] [PubMed] [Google Scholar]
- Silverberg JI, Lai J-S, Vakharia PP, Patel K, Singam V, Chopra R, et al. Measurement properties of the Patient-Reported Outcomes Measurement Information System Itch Questionnaire item banks in adults with atopic dermatitis. J Am Acad Dermatol 2020b;82(5):1174–80. [DOI] [PubMed] [Google Scholar]
- Silverberg JI, Lei D, Yousaf M, Janmohamed SR, Vakharia PP, Chopra R, et al. Comparison of Patient-Oriented Eczema Measure and Patient-Oriented Scoring Atopic Dermatitis vs Eczema Area and Severity Index and other measures of atopic dermatitis: A validation study. Ann Allergy Asthma Immunol 2020c;125(1):78–83. [DOI] [PubMed] [Google Scholar]
- Silverberg JI, Lei D, Yousaf M, Janmohamed SR, Vakharia PP, Chopra R, et al. Measurement properties of the product of investigator’s global assessment and body surface area in children and adults with atopic dermatitis. J Eur Acad Dermatol Venereol 2020d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverberg JI, Lei D, Yousaf M, Janmohamed SR, Vakharia PP, Chopra R, et al. Measurement properties of the Rajka–Langeland severity score in children and adults with atopic dermatitis. Br J Dermatol 2020e. [DOI] [PubMed] [Google Scholar]
- Silverberg JI, Lei D, Yousaf M, Janmohamed SR, Vakharia PP, Chopra R, et al. What are the best endpoints for Eczema Area and Severity Index and Scoring Atopic Dermatitis in clinical practice? A prospective observational study. Br J Dermatol 2020f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverberg JI, Lei D, Yousaf M, Janmohamed SR, Vakharia PP, Chopra R, et al. Association of itch triggers with atopic dermatitis severity and course in adults. Ann Allergy Asthma Immunol 2020g;125(5):552–9.e2. [DOI] [PubMed] [Google Scholar]
- Silverberg JI, Margolis DJ, Boguniewicz M, Fonacier L, Grayson MH, Ong PY, et al. Validation of five patient-reported outcomes for atopic dermatitis severity in adults. Br J Dermatol 2020h;182(1):104–11. [DOI] [PubMed] [Google Scholar]
- Simpson E, Bissonnette R, Eichenfield LF, Guttman-Yassky E, King B, Silverberg JI, et al. The Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD): The development and reliability testing of a novel clinical outcome measurement instrument for the severity of atopic dermatitis. J Am Acad Dermatol 2020;83(3):839–46. [DOI] [PubMed] [Google Scholar]
- Simpson EL, de Bruin-Weller M, Eckert L, Whalley D, Guillemin I, Reaney M, et al. Responder Threshold for Patient-Oriented Eczema Measure (POEM) and Children’s Dermatology Life Quality Index (CDLQI) in Adolescents with Atopic Dermatitis. Dermatol Ther 2019;9(4):799–805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sprikkelman AB, Tupker RA, Burgerhof H, Schouten JP, Br PL, et al. Severity scoring of atopic dermatitis: a comparison of three scoring systems. Allergy;52(9):944–9. [DOI] [PubMed] [Google Scholar]
- Stalder JF, Barbarot S, Wollenberg A, Holm EA, De Raeve L, Seidenari S, et al. Patient-Oriented SCORAD (PO-SCORAD): a new self-assessment scale in atopic dermatitis validated in Europe. Allergy;66(8):1114–21. [DOI] [PubMed] [Google Scholar]
- Sugarman JL, Fluhr JW, Fowler AJ, Bruckner T, Diepgen TL, Williams ML. The objective severity assessment of atopic dermatitis score: an objective measure using permeability barrier function and stratum corneum hydration with computer-assisted estimates for extent of disease. Arch Dermatol;139(11):1417–22. [DOI] [PubMed] [Google Scholar]
- Suh TP, Ramach, ran D, Patel V, Jackson KL, Rangel SM, et al. Product of Investigator Global Assessment and Body Surface Area (IGAxBSA): A practice-friendly alternative to the Eczema Area and Severity Index to assess atopic dermatitis severity in children. J Am Acad Dermatol 2020;82(5):1187–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taïeb A, Boralevi F, Seneschal J, Merh, Georgescu V, Taieb C, et al. Atopic Dermatitis Burden Scale for Adults: Development and Validation of a New Assessment Tool. Acta dermato-venereologica 2015;95(6):700–5. [DOI] [PubMed] [Google Scholar]
- Thyssen JP, Andersen Y, Halling AS, Williams HC, Egeberg A. Strengths and limitations of the United Kingdom Working Party criteria for atopic dermatitis in adults. J Eur Acad Dermatol Venereol 2020;34(8):1764–72. [DOI] [PubMed] [Google Scholar]
- Topham C, Haynes D, Brazil M, Simpson E. Initial validation of the product of the signs global assessment and body surface area in atopic dermatitis. J Am Acad Dermatol 2020. [DOI] [PubMed] [Google Scholar]
- Tremp M, Knafla I, Burg G, Wüthrich B, Schmid-Grendelmeier P. 'EASIdig'--a digital tool to document disease activity in atopic dermatitis. Dermatology 2011;223(1):68–73. [DOI] [PubMed] [Google Scholar]
- Twiss J, Meads DM, Preston EP, Crawford SR, McKenna SP. Can we rely on the Dermatology Life Quality Index as a measure of the impact of psoriasis or atopic dermatitis? J Invest Dermatol;132(1):76–84. [DOI] [PubMed] [Google Scholar]
- Udkoff J, Silverberg J. Validation of scratching severity as an objective assessment for itch. J Am Acad Dermatol 2018;79(3):AB311. [DOI] [PubMed] [Google Scholar]
- Vakharia P, Chopra R, Sacotte R, Patel N, Immaneni S, White T, et al. Validation of patient-reported global severity of atopic dermatitis in adults. Allergy 2018a;73(2):451–8. [DOI] [PubMed] [Google Scholar]
- Vakharia P, Chopra R, Sacotte R, Patel N, Immaneni S, White T, et al. Severity strata for five patient-reported outcomes in adults with atopic dermatitis. The British journal of dermatology 2018b;178(4):925–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vakharia PP, Chopra R, Sacotte R, Patel N, Immaneni S, White T, et al. Validation of patient-reported global severity of atopic dermatitis in adults. Allergy 2018;73(2):451–8. [DOI] [PubMed] [Google Scholar]
- van Valburg RW, Willemsen MG, Dirven-Meijer PC, Oranje AP, van der Wouden JC, Moed H. Quality of life measurement and its relationship to disease severity in children with atopic dermatitis in general practice. Acta Derm Venereol;91(2):147–51. [DOI] [PubMed] [Google Scholar]
- van Velsen SG, Knol MJ, Haeck IM, Bruijnzeel-Koomen CA, Pasmans SG. The Self-administered Eczema Area and Severity Index in children with moderate to severe atopic dermatitis: better estimation of AD body surface area than severity. Pediatr Dermatol;27(5):470–5. [DOI] [PubMed] [Google Scholar]
- Verwimp JJ, Bindels JG, Barents M, Heymans HS. Symptomatology and growth in infants with cow's milk protein intolerance using two different whey-protein hydrolysate based formulas in a Primary Health Care setting. Eur J Clin Nutr;49:S39–48. [PubMed] [Google Scholar]
- Vilsbøll AW, Kragh N, Hahn-Pedersen J, Jensen CE. Mapping Dermatology Life Quality Index (DLQI) scores to EQ-5D utility scores using data of patients with atopic dermatitis from the National Health and Wellness Study. Qual Life Res 2020;29(9):2529–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vourc'h-Jourdain M, Barbarot S, Taieb A, Diepgen T, Ambonati M, Durosier V, et al. Patient-oriented SCORAD: a self-assessment score in atopic dermatitis. A preliminary feasibility study. Dermatology 2009;218(3):246–51. [DOI] [PubMed] [Google Scholar]
- Weisshaar E, Diepgen TL, Bruckner T, Fartasch M, Kupfer J, Lob-Corzilius T, et al. Itch intensity evaluated in the German Atopic Dermatitis Intervention Study (GADIS): correlations with quality of life, coping behaviour and SCORAD severity in 823 children. Acta Derm Venereol 2008;88(3):234–9. [DOI] [PubMed] [Google Scholar]
- Wells G, Shea B, O'Connell D, et al. The Newcastle-Ottawa Scale (NOS) AQ5 for Assessing the Quality of Nonrandomised Studies in Meta-Analyses.: Ottawa Hospital Research Institute. [Google Scholar]
- Whalley D, McKenna SP, Dewar AL, Erdman RA, Kohlmann T, Niero M, et al. A new instrument for assessing quality of life in atopic dermatitis: international development of the Quality of Life Index for Atopic Dermatitis (QoLIAD). Br J Dermatol;150(2):274–83. [DOI] [PubMed] [Google Scholar]
- Willemsen MG, van Valburg RW, Dirven-Meijer PC, Oranje AP, van der Wouden JC, Moed H. Determining the severity of atopic dermatitis in children presenting in general practice: an easy and fast method. Dermatol Res Pract 2009;2009:357046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolkerstorfer A, de Waard van der Spek FB, Glazenburg EJ, Mulder PG, Oranje AP. Scoring the severity of atopic dermatitis: three item severity score as a rough system for daily practice and as a pre-screening tool for studies. Acta Derm Venereol;79(5):356–9. [DOI] [PubMed] [Google Scholar]
- Yamaguchi C, Futamura M, Chamlin SL, Ohya Y, Asano M. Development of a Japanese Culturally Modified Version of the Childhood Atopic Dermatitis Impact Scale (JCMV-CADIS). Allergol Int;65(3):312–9. [DOI] [PubMed] [Google Scholar]
- Yang HJ, Jeon YH, Pyun BY. Evaluation of patient's subjective severity using various scoring system in Korean children with atopic dermatitis. Asian Pac J Allergy Immunol;28(2):130–5. [PubMed] [Google Scholar]
- Yew YW, Zhao X, Apfelbacher CJ. The Patient-Oriented Eczema Measure: estimating the minimal important change in an outpatient clinic cohort. J Eur Acad Dermatol Venereol 2020;34(6):1273–9. [DOI] [PubMed] [Google Scholar]
- Yosipovitch G, Reaney M, Mastey V, Eckert L, Abbé A, Nelson L, et al. Peak Pruritus Numerical Rating Scale: psychometric validation and responder definition for assessing itch in moderate-to-severe atopic dermatitis. Br J Dermatol 2019;181(4):761–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao CY, Hao EY, Oh DD, Daniel BS, Martin LK, Su JC, et al. A comparison study of clinician-rated atopic dermatitis outcome measures for intermediate- to dark-skinned patients. England 2017. 2017-April. 985–92 p. [DOI] [PubMed] [Google Scholar]
- Zhao CY, Wijayanti A, Doria MC, Harris AG, Jain SV, Legaspi KN, et al. The reliability and validity of outcome measures for atopic dermatitis in patients with pigmented skin: A grey area. Int J Womens Dermatol 2015;1(3):150–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data were collected from the published literature. No datasets were generated during the current study.