Abstract
Objective
This study aimed to decipher the bioactive compounds and potential mechanism of traditional Chinese medicine (TCM) formula Fuzi Lizhong Decoction (FLD) for nonalcoholic fatty liver disease (NAFLD) treatment via an integrative network pharmacology approach.
Methods
The candidate compounds of FLD and its relative targets were obtained from the TCMSP and PharmMapper web server, and the intersection genes for NAFLD were discerned using OMIM, GeneCards, and DisGeNET. Then, the PPI and component-target-pathway networks were constructed. Moreover, GO enrichment and KEGG pathway analysis were performed to investigate the potential signaling pathways associated with FLD's effect on NAFLD. Eventually, molecular docking simulation was carried out to validate the binding affinity between potential core components and key targets.
Results
A total of 143 candidate active compounds and 129 relative drug targets were obtained, in which 61 targets were overlapped with NAFLD. The PPI network analysis identified ALB, MAPK1, CASP3, MARK8, and AR as key targets, mainly focusing on cellular response to organic cyclic compound, steroid metabolic process, and response to steroid hormone in the biological processes. The KEGG pathway analysis demonstrated that 16 signaling pathways were closely correlated with FLD's effect on NALFD with cancer pathways, Th17 cell differentiation, and IL-17 signaling pathways as the most significant ones. In addition, the molecular docking analysis revealed that the core active compounds of FLD, such as 3′-methoxyglabridin, chrysanthemaxanthin, and Gancaonin H, had a high binding activity with such key targets as ALB, MAPK1, and CASP3.
Conclusions
This study suggested that FLD exerted its effect on NAFLD via modulating multitargets with multicompounds through multipathways. It also demonstrated that the network pharmacology-based approach might provide insights for understanding the interrelationship between complex diseases and interventions of the TCM formula.
1. Introduction
Nonalcoholic fatty liver disease (NAFLD) is a metabolic disorder with excessive hepatic fat deposition in the absence of significant alcoholic consumption, application of susceptible medication, or other preexisting liver conditions [1]. Epidemiological surveys indicate that its global prevalence reaches approximately 25%, with an increasing annual incidence [2]. As the most common chronic liver disease, NAFLD encompasses a broad spectrum of liver damage and has been considered a cause of end-stage liver disease. It is assumed to be associated with increased rates of hepatocellular carcinoma and death and is projected to become the leading cause of cirrhosis that requires liver transplantation within the next ten years [3]. This progression is potentially reversible with proper management. Although insulin resistance, metabolic complications, and genetic variants, including PNPLA3 and TM6SF2, may play a role [4], the pathogenesis of NAFLD remains incompletely decoded, and no pharmacological interventions have been officially approved. Furthermore, the treatment options available for NAFLD patients are still limited and mainly based on nutrition and exercise [5]. Therefore, there is an urgent need for the research and development of novel medical interventions.
Traditional Chinese medicine (TCM) has a long history of treating liver disease in China, and compelling lines of preclinical and clinical evidence have demonstrated that the TCM formula may yield effective outcomes for the management of NAFLD. Fuzi Lizhong Decoction (FLD) is a classic TCM formula from Volume 2 of San Yin Ji Yi Bing Zheng Fang Lun (Treatise on Diseases, Patterns, and Formulas Related to the Unification of the Three Etiologies, 1174 A.D.) written by Dr. Wu-ze Chen. It has been widely utilized to treat NAFLD clinically. Previous studies have shown that FLD can promote the proliferation and inhibit the apoptosis of NAFLD cells via upregulating the expression of cyclin-associated and antiapoptotic proteins and downregulating the expression of proapoptotic proteins and apoptotic actors [6]. It can also effectively reduce blood lipid content, improve liver function, and decrease liver index, thus alleviating the progression of NAFLD in rats. In addition, its pharmacological effects are closely related to activating AMPK, inhibiting the liver fat synthesis, and inhibiting the NF-κB signaling pathway and the secretion of inflammatory response factors TNF-α and IL-6 in the liver [7]. Moreover, it substantially reduces the fat deposition capacity of NAFLD cells and alleviates the inflammatory response [8]. These studies partially decipher the molecular mechanism of FLD's action on NAFLD. However, TCM formulas often contain complex constituents with multiple pharmacological activities at a variety of targets. From this perspective, the action mechanism of FLD on NAFLD may involve a complex of multicomponents, multitargets, and multipathways and requires a further study following the holistic strategy.
Network pharmacology integrates systematic medicine and bioinformation science and is consistent with the holistic concept of TCM treatment. It systematically decodes the therapeutic effects of complex TCM formula on disease via analyzing the interactions between herbal components, targets, diseases, and pathways. Hence, it has been considered as a frontier in the field of drug research and development. A growing body of research has currently verified its potential in investigating the possible molecular mechanisms of the TCM formula [9]. In this study, network pharmacology was utilized to reveal the components, targets, and pathways interaction relationships between FLD intervention and NAFLD management: (1) the candidate compounds and intersection targets were obtained; (2) the protein-protein interaction (PPI) and component-target-pathway-disease networks were constructed; (3) Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis were conducted; and (4) molecular docking simulation was performed to validate the binding affinity between potential core components and key targets. The detailed flowchart of the study design is shown in Figure 1.
Figure 1.
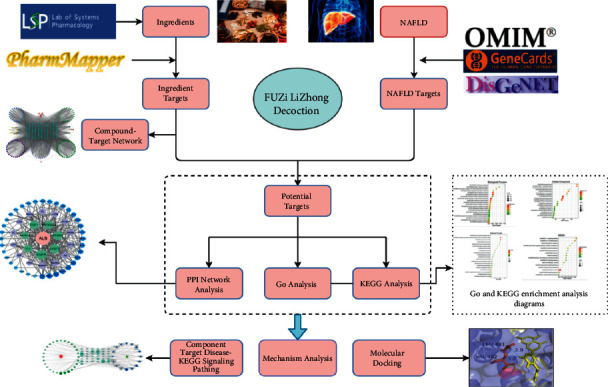
Detailed flowchart of the study design.
2. Material and Methods
2.1. Collection of FLD Active Components and Corresponding Targets
FLD comprises five herbs, namely FuZi (Radix Saposhnikoviae divaricate, FZ), GanJiang (Rhizoma Zingiberis, GJ), RenShen (Panax ginseng, RS), BaiZhu (Rhizoma Atractylodis Macrocephalae, BZ), and GanCao (Radix et Rhizoma Glycyrrhizae, GC). The active compounds of FLD were extracted from the TCMSP data platform (https://tcmspw.com/tcmsp.php) [10], which contains 499 Chinese herbs registered in Chines Pharmacopoeia (2010 edition) with 29,384 ingredients, 3,311targets, and 837 related diseases. The thresholds further filtering the obtained active compounds were set as Oral bioavailability (OB) ≥ 30% and drug-likeness (DL) ≥ 0.18. The protein targets of the collected active compounds were screened with a standard of “Norm Fit≥0.90” using the PharmMapper web server (http://www.msftconnecttest.com/redirect) [11]. The protein targets corresponding to the active components of herbs were imported into the Uniport data platform (https://www.uniprot.org/) to acquire the gene names, IDs, and functions, respectively [12].
2.2. Construction of FLD-Compound-Target Network
The FLD-active compound-target network was constructed and visualized using Cytoscape 3.8.2 software [13], and the core active ingredients of FLD were identified.
2.3. Acquisition of NAFLD-Associated Targets and Candidate Genes
Keywords such as “nonacholic fat liver disease” or “NAFLD” were employed to search the disease-related targets from the Online Mendelian Inheritance in Man (OMIM) (http://www.omim.org), Human Gene Database (GeneCards) (https://www.genecards.org), and DisGeNET (http://www.disgenet.org/) databases. After removing the duplicate targets, the protein targets related to NAFLD were imported to the Uniprot data platform. Then, the acquired genes were input to draw Venn diagrams using Venny2.1(https://bioinfogp.cnb.csic.es/tools/venny/), and the intersection genes were obtained as candidate genes.
2.4. Construction of PPI Network
The candidate genes were imported into the Search Tool for the Retrieval of Interacting Genes online analysis software (STRING, https://www.string-db.org/) to construct a PPI network. The visual network graphs were created by Cytoscape 3.8.2 software. Degree, betweenness, and closeness were three major topological parameters used to identify the key genes.
2.5. GO and KEGG Pathway Enrichment Analysis
The functional annotation of GO and KEGG pathways was performed using the Metascape platform (http://metascape.org/gp/index.html) to explore the relevant biological processes (BPs), cellular components (CCs), molecular functions (MFs), and signal pathways of potential anti-NAFLD targets. The results were saved and sorted with the False Discovery Rate algorithm for each term. The bioinformatics platform (http://www.bioinformatics.com.cn/) was applied to visualize GO and KEGG enrichment results by bubble diagrams.
2.6. Construction of Compound-Target-Pathway Network Construction
To further investigate the therapeutic mechanisms of FLD for NAFLD, the compound-target-pathway (C-T-P) network was constructed using Cytoscape 3.8.2. In this network, the compounds, target genes, and pathways are symbolized with nodes in different colors and shapes, and the association between nodes is represented by the edge.
2.7. Molecular Docking Simulation
A molecular docking simulation was conducted to assess the binding energy of the core compounds with the key targets. Autodock Vina 1.5.6 software developed by Olson's research group in Scripps Research Institute was adopted to assess molecular docking [14]. The top three targets of the PPI network and the top ten core compounds of FLD were selected, and their chemical structures were downloaded from the RCSB PDB database (http://www.rcsb.org/) and TCMSP data platform, respectively. The AutoDockTools were utilized to convert the candidate compounds into PDB format. The proteins were virtually dehydrated and hydrogenated, and the core compounds were hydrogenated. The original ligands were extracted and stored separately. AutoDockTool was utilized to convert compounds, ligands, and proteins into the “pdbqt” format and to define if the location of each protein or its ligands was the active pocket of the protein. When a binding energy value < 0, the molecular proteins were considered spontaneously binding and interacting. Accordingly, the lower the binding energy required for docking, the more stable the molecular conformation.
3. Results
3.1. Active Ingredients and Target Genes
Initially, 738 compounds were retrieved from the five herbs in FLD. Among them, 65, 55, 190, 148, and 280 compounds were from FZ, BZ, RS, GJ, and GC, respectively. A total of 147 active compounds were acquired upon OB ≥ 30% and DL ≥ 0.18. Specifically, 21, 7, 22, 5, and 92 compounds were from FZ, BZ, RS, GJ, and GC, respectively. In addition, some compounds were overlapped across different herbs, including sitosterol (ID: MOL000359) in FZ, GJ, and GC, beta-sitosterol (ID: MOL000358) in GJ and RS, and kaempferol (ID: MOL000422) in RS and GC. A total of 143 active compounds were identified after deleting duplicated entries. After the protein targets corresponding to the active components of herbs were imported into the Uniport data platform to acquire the gene names and eliminate the duplications, 129 gene targets with those putative components were acquired. Among which, 45 were associated with FZ, 7 with BZ, 30 with RS, 4 with GJ, and 43 with GC. The information of each candidate compound is shown in Table 1.
Table 1.
Active compounds of FLD in NAFLD treatment.
| Molecule ID | Molecule name | OB (%) | DL (%) | Source |
|---|---|---|---|---|
| MOL002211 | 11,14-Eicosadienoic acid | 39.99 | 0.2 | FZ1 |
| MOL002388 | Delphin_qt | 57.76 | 0.28 | FZ2 |
| MOL002392 | Deltoin | 46.69 | 0.37 | FZ3 |
| MOL002393 | Demethyldelavaine A | 34.52 | 0.18 | FZ4 |
| MOL002394 | Demethyldelavaine B | 34.52 | 0.18 | FZ5 |
| MOL002395 | Deoxyandrographolide | 56.3 | 0.31 | FZ6 |
| MOL002397 | Karakoline | 51.73 | 0.73 | FZ7 |
| MOL002398 | Karanjin | 69.56 | 0.34 | FZ8 |
| MOL002401 | Neokadsuranic acid B | 43.1 | 0.85 | FZ9 |
| MOL002406 | 2,7-Dideacetyl-2,7-dibenzoyl-taxayunnanine F | 39.43 | 0.38 | FZ10 |
| MOL002410 | Benzoylnapelline | 34.06 | 0.53 | FZ11 |
| MOL002415 | 6-Demethyldesoline | 51.87 | 0.66 | FZ12 |
| MOL002416 | Deoxyaconitine | 30.96 | 0.24 | FZ13 |
| MOL002419 | (R)-Norcoclaurine | 82.54 | 0.21 | FZ14 |
| MOL002421 | Ignavine | 84.08 | 0.25 | FZ15 |
| MOL002422 | Isotalatizidine | 50.82 | 0.73 | FZ16 |
| MOL002423 | Jesaconitine | 33.41 | 0.19 | FZ17 |
| MOL002433 | (3R,8S,9R,10 R,13R,14S,17R)-3-Hydroxy-4,4,9,13,14-pentamethyl-17-[(E,2R)-6-methyl-7-[(2R,3 R,4S,5S,6R)-3,4,5-trihydroxy-6-[[(2R,3 R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxymethyl]oxan-2-yl]oxyhept-5-en-2-yl]-1,2,3,7,8,10,12,15,16,17-decahydr | 41.52 | 0.22 | FZ18 |
| MOL002434 | Carnosifloside I_qt | 38.16 | 0.8 | FZ19 |
| MOL000359 | Sitosterol | 36.91 | 0.75 | FZ20 |
| MOL000538 | Hypaconitine | 31.39 | 0.26 | FZ21 |
| MOL002464 | 1-Monolinolein | 37.18 | 0.3 | GJ1 |
| MOL002501 | [(1S)-3-[(E)-but-2-enyl]-2-Methyl-4-oxo-1-cyclopent-2-enyl] (1r,3 R)-3-[(E)-3-methoxy-2-methyl-3-oxoprop-1-enyl]-2,2-dimethylcyclopropane-1-carboxylate | 62.52 | 0.31 | GJ2 |
| MOL002514 | Sexangularetin | 62.86 | 0.3 | GJ3 |
| MOL000358 | Beta-sitosterol | 36.91 | 0.75 | GJ4 |
| MOL000359 | Sitosterol | 36.91 | 0.75 | GJ5 |
| MOL000020 | 12-Senecioyl-2E,8 E,10E-atractylentriol | 62.4 | 0.22 | BZ1 |
| MOL000021 | 14-Acetyl-12-senecioyl-2E,8 E,10E-atractylentriol | 60.31 | 0.31 | BZ2 |
| MOL000022 | 14-Acetyl-12-senecioyl-2E,8Z,10E-atractylentriol | 63.37 | 0.3 | BZ3 |
| MOL000028 | α-Amyrin | 39.51 | 0.76 | BZ4 |
| MOL000033 | (3S,8S,9S,10R,13R,14S,17R)-10,13-Dimethyl-17-[(2R,5S)-5-propan-2-yloctan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-3-ol | 36.23 | 0.78 | BZ5 |
| MOL000049 | 3β-Acetoxyatractylone | 54.07 | 0.22 | BZ6 |
| MOL000072 | 8β-Ethoxy atractylenolide III | 35.95 | 0.21 | BZ7 |
| MOL002879 | Diop | 43.59 | 0.39 | RS1 |
| MOL000449 | Stigmasterol | 43.83 | 0.76 | RS2 |
| MOL000358 | Beta-sitosterol | 36.91 | 0.75 | RS3 |
| MOL003648 | Inermin | 65.83 | 0.54 | RS4 |
| MOL000422 | Kaempferol | 41.88 | 0.24 | RS5 |
| MOL004492 | Chrysanthemaxanthin | 38.72 | 0.58 | RS6 |
| MOL005308 | Aposiopolamine | 66.65 | 0.22 | RS7 |
| MOL005314 | Celabenzine | 101.88 | 0.49 | RS8 |
| MOL005317 | Deoxyharringtonine | 39.27 | 0.81 | RS9 |
| MOL005318 | Dianthramine | 40.45 | 0.2 | RS10 |
| MOL005320 | Arachidonate | 45.57 | 0.2 | RS11 |
| MOL005321 | Frutinone A | 65.9 | 0.34 | RS12 |
| MOL005344 | Ginsenoside rh2 | 36.32 | 0.56 | RS13 |
| MOL005348 | Ginsenoside-Rh4_qt | 31.11 | 0.78 | RS14 |
| MOL005356 | Girinimbin | 61.22 | 0.31 | RS15 |
| MOL005357 | Gomisin B | 31.99 | 0.83 | RS16 |
| MOL005360 | Malkangunin | 57.71 | 0.63 | RS17 |
| MOL005376 | Panaxadiol | 33.09 | 0.79 | RS18 |
| MOL005384 | Suchilactone | 57.52 | 0.56 | RS19 |
| MOL005399 | Alexandrin_qt | 36.91 | 0.75 | RS20 |
| MOL005401 | Ginsenoside Rg5_qt | 39.56 | 0.79 | RS21 |
| MOL000787 | Fumarine | 59.26 | 0.83 | RS22 |
| MOL001484 | Inermine | 75.18 | 0.54 | GC1 |
| MOL001792 | DFV | 32.76 | 0.18 | GC2 |
| MOL000211 | Mairin | 55.38 | 0.78 | GC3 |
| MOL002311 | Glycyrol | 90.78 | 0.67 | GC4 |
| MOL000239 | Jaranol | 50.83 | 0.29 | GC5 |
| MOL002565 | Medicarpin | 49.22 | 0.34 | GC6 |
| MOL000354 | Isorhamnetin | 49.6 | 0.31 | GC7 |
| MOL000359 | Sitosterol | 36.91 | 0.75 | GC8 |
| MOL003656 | Lupiwighteone | 51.64 | 0.37 | GC9 |
| MOL003896 | 7-Methoxy-2-methyl isoflavone | 42.56 | 0.2 | GC10 |
| MOL000392 | Formononetin | 69.67 | 0.21 | GC11 |
| MOL000417 | Calycosin | 47.75 | 0.24 | GC12 |
| MOL000422 | Kaempferol | 41.88 | 0.24 | GC13 |
| MOL004328 | Naringenin | 59.29 | 0.21 | GC14 |
| MOL004805 | (2S)-2-[4-Hydroxy-3-(3-methylbut-2-enyl)phenyl]-8,8-dimethyl-2,3-dihydropyrano[2,3-f]chromen-4-one | 31.79 | 0.72 | GC15 |
| MOL004806 | Euchrenone | 30.29 | 0.57 | GC16 |
| MOL004808 | Glyasperin B | 65.22 | 0.44 | GC17 |
| MOL004810 | Glyasperin F | 75.84 | 0.54 | GC18 |
| MOL004811 | Glyasperin C | 45.56 | 0.4 | GC19 |
| MOL004814 | Isotrifoliol | 31.94 | 0.42 | GC20 |
| MOL004815 | (E)-1-(2,4-Dihydroxyphenyl)-3-(2,2-dimethylchromen-6-yl)prop-2-en-1-one | 39.62 | 0.35 | GC21 |
| MOL004820 | Kanzonols W | 50.48 | 0.52 | GC22 |
| MOL004824 | (2S)-6-(2,4-Dihydroxyphenyl)-2-(2-hydroxypropan-2-yl)-4-methoxy-2,3-dihydrofuro[3,2-g]chromen-7-one | 60.25 | 0.63 | GC23 |
| MOL004827 | Semilicoisoflavone B | 48.78 | 0.55 | GC24 |
| MOL004828 | Glepidotin A | 44.72 | 0.35 | GC25 |
| MOL004829 | Glepidotin B | 64.46 | 0.34 | GC26 |
| MOL004833 | Phaseolinisoflavan | 32.01 | 0.45 | GC27 |
| MOL004835 | Glypallichalcone | 61.6 | 0.19 | GC28 |
| MOL004838 | 8-(6-Hydroxy-2-benzofuranyl)-2,2-dimethyl-5-chromenol | 58.44 | 0.38 | GC29 |
| MOL004841 | Licochalcone B | 76.76 | 0.19 | GC30 |
| MOL004848 | Licochalcone G | 49.25 | 0.32 | GC31 |
| MOL004849 | 3-(2,4-Dihydroxyphenyl)-8-(1,1-dimethylprop-2-enyl)-7-hydroxy-5-methoxy-coumarin | 59.62 | 0.43 | GC32 |
| MOL004855 | Licoricone | 63.58 | 0.47 | GC33 |
| MOL004856 | Gancaonin A | 51.08 | 0.4 | GC34 |
| MOL004857 | Gancaonin B | 48.79 | 0.45 | GC35 |
| MOL004860 | Licorice glycoside E | 32.89 | 0.27 | GC36 |
| MOL004863 | 3-(3,4-Dihydroxyphenyl)-5,7-dihydroxy-8-(3-methylbut-2-enyl)chromone | 66.37 | 0.41 | GC37 |
| MOL004864 | 5,7-Dihydroxy-3-(4-methoxyphenyl)-8-(3-methylbut-2-enyl)chromone | 30.49 | 0.41 | GC38 |
| MOL004866 | 2-(3,4-Dihydroxyphenyl)-5,7-dihydroxy-6-(3-methylbut-2-enyl)chromone | 44.15 | 0.41 | GC39 |
| MOL004879 | Glycyrin | 52.61 | 0.47 | GC40 |
| MOL004882 | Licocoumarone | 33.21 | 0.36 | GC41 |
| MOL004883 | Licoisoflavone | 41.61 | 0.42 | GC42 |
| MOL004884 | Licoisoflavone B | 38.93 | 0.55 | GC43 |
| MOL004885 | Licoisoflavanone | 52.47 | 0.54 | GC44 |
| MOL004891 | Shinpterocarpin | 80.3 | 0.73 | GC45 |
| MOL004898 | (E)-3-[3,4-Dihydroxy-5-(3-methylbut-2-enyl)phenyl]-1-(2,4-dihydroxyphenyl)prop-2-en-1-one | 46.27 | 0.31 | GC46 |
| MOL004903 | Liquiritin | 65.69 | 0.74 | GC47 |
| MOL004904 | Licopyranocoumarin | 80.36 | 0.65 | GC48 |
| MOL004905 | 3,22-Dihydroxy-11-oxo-delta(12)-oleanene-27-alpha-methoxycarbonyl-29-oic acid | 34.32 | 0.55 | GC49 |
| MOL004907 | Glyzaglabrin | 61.07 | 0.35 | GC50 |
| MOL004908 | Glabridin | 53.25 | 0.47 | GC51 |
| MOL004910 | Glabranin | 52.9 | 0.31 | GC52 |
| MOL004911 | Glabrene | 46.27 | 0.44 | GC53 |
| MOL004912 | Glabrone | 52.51 | 0.5 | GC54 |
| MOL004913 | 1,3-Dihydroxy-9-methoxy-6-benzofurano [3,2-c]chromenone | 48.14 | 0.43 | GC55 |
| MOL004914 | 1,3-Dihydroxy-8,9-dimethoxy-6-benzofurano [3,2-c]chromenone | 62.9 | 0.53 | GC56 |
| MOL004915 | Eurycarpin A | 43.28 | 0.37 | GC57 |
| MOL004917 | Glycyroside | 37.25 | 0.79 | GC58 |
| MOL004924 | (-)-Medicocarpin | 40.99 | 0.95 | GC59 |
| MOL004935 | Sigmoidin-B | 34.88 | 0.41 | GC60 |
| MOL004941 | (2R)-7-Hydroxy-2-(4-hydroxyphenyl)chroman-4-one | 71.12 | 0.18 | GC61 |
| MOL004945 | (2S)-7-Hydroxy-2-(4-hydroxyphenyl)-8-(3-methylbut-2-enyl)chroman-4-one | 36.57 | 0.32 | GC62 |
| MOL004948 | Isoglycyrol | 44.7 | 0.84 | GC63 |
| MOL004949 | Isolicoflavonol | 45.17 | 0.42 | GC64 |
| MOL004957 | HMO | 38.37 | 0.21 | GC65 |
| MOL004959 | 1-Methoxyphaseollidin | 69.98 | 0.64 | GC66 |
| MOL004961 | Quercetin der. | 46.45 | 0.33 | GC67 |
| MOL004966 | 3'-Hydroxy-4'-O-Methylglabridin | 43.71 | 0.57 | GC68 |
| MOL000497 | Licochalcone a | 40.79 | 0.29 | GC69 |
| MOL004974 | 3'-Methoxyglabridin | 46.16 | 0.57 | GC70 |
| MOL004978 | 2-[(3R)-8,8-Dimethyl-3,4-dihydro-2h-pyrano [6,5-f]chromen-3-yl]-5-methoxyphenol | 36.21 | 0.52 | GC71 |
| MOL004980 | Inflacoumarin A | 39.71 | 0.33 | GC72 |
| MOL004985 | Icos-5-enoic acid | 30.7 | 0.2 | GC73 |
| MOL004988 | Kanzonol F | 32.47 | 0.89 | GC74 |
| MOL004989 | 6-Prenylated eriodictyol | 39.22 | 0.41 | GC75 |
| MOL004990 | 7,2',4'-Trihydroxy-5-methoxy-3-arylcoumarin | 83.71 | 0.27 | GC76 |
| MOL004991 | 7-Acetoxy-2-methylisoflavone | 38.92 | 0.26 | GC77 |
| MOL004993 | 8-Prenylated eriodictyol | 53.79 | 0.4 | GC78 |
| MOL004996 | Gadelaidic acid | 30.7 | 0.2 | GC79 |
| MOL000500 | Vestitol | 74.66 | 0.21 | GC80 |
| MOL005000 | Gancaonin G | 60.44 | 0.39 | GC81 |
| MOL005001 | Gancaonin H | 50.1 | 0.78 | GC82 |
| MOL005003 | Licoagrocarpin | 58.81 | 0.58 | GC83 |
| MOL005007 | Glyasperins M | 72.67 | 0.59 | GC84 |
| MOL005008 | Glycyrrhiza flavonol A | 41.28 | 0.6 | GC85 |
| MOL005012 | Licoagroisoflavone | 57.28 | 0.49 | GC86 |
| MOL005013 | 18α-Hydroxyglycyrrhetic acid | 41.16 | 0.71 | GC87 |
| MOL005016 | Odoratin | 49.95 | 0.3 | GC88 |
| MOL005017 | Phaseol | 78.77 | 0.58 | GC89 |
| MOL005018 | Xambioona | 54.85 | 0.87 | GC90 |
| MOL005020 | Dehydroglyasperins C | 53.82 | 0.37 | GC91 |
| MOL000098 | Quercetin | 46.43 | 0.28 | GC92 |
| MOL000359 | Sitosterol | 36.91 | 0.75 | FZ20, Gj5, GC8 (A1) |
| MOL000358 | Beta-sitosterol | 36.91 | 0.75 | GJ4, RS3(B1) |
| MOL000422 | Kaempferol | 41.88 | 0.24 | RS5,GC13(C1) |
3.2. FLD-Compound-Target Network
The FLD-C-T network contained 280 nodes (143 nodes for compound, 129 nodes for target, and 8 nodes for herbs) and 3,199 edges (Figure 2). Sitosterol showed the highest degree of connectivity in the network with 57 targets, followed by beta-sitosterol with 44 targets, and ginsenoside rh2 with 35 targets. The properties of this network were suitable for displaying complex compounds, multiple targets, and interactions between compounds and targets. The OB of the three compounds mentioned above was 36.91%, 36.91%, and 36.32%, respectively, indicating they were potential key active compounds. Detailed information about the compound-target network is presented in Supplement Table 1.
Figure 2.
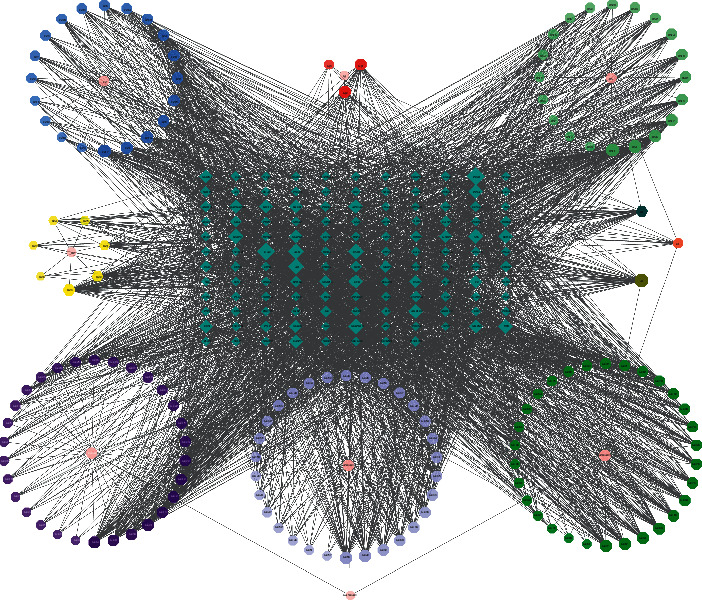
Compound-target network: pink circles represent the herbs in FLD; hexagons represent active compounds of each herb, and A1, B1, and C1 hexagons correspond to active compounds shared by different herbs; blue diamonds represent related targets (the IDs of the components are presented in Table 1).
3.3. NAFLD-Associated Targets and Candidate Genes
A total of 3,181 targets for NAFLD were integrated from multiple databases, including 512 targets from OMIM, 1,611 from GeneCards, and 1,058 from DisGeNET. The final list of 2,608 NAFLD-related targets was obtained after eliminating duplicates (Supplement Table 2). Among them, 61 intersection targets between FLD and NAFLD were identified and collected for further mechanism investigation (Figure 3 and Supplement Table 3).
Figure 3.
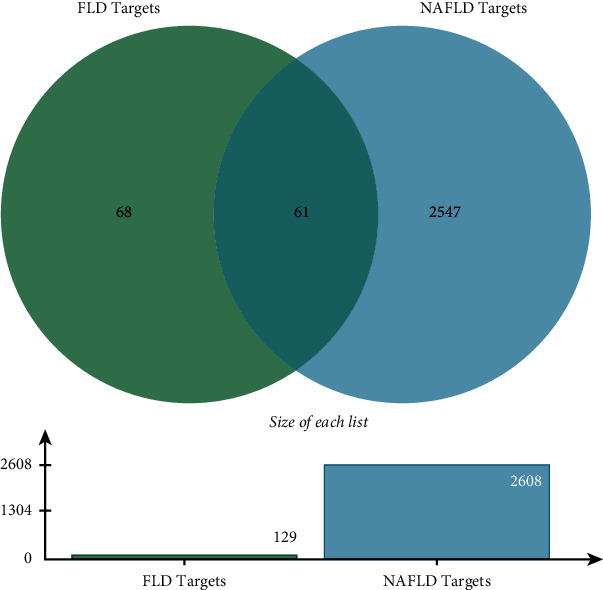
Venn's diagram of intersection targets of FLD and NAFLD.
3.4. PPI Network of FLD and NAFLD Targets Construction
A visualized PPI network composed of 61 nodes representing proteins with 277 edges representing the interactions between proteins was constructed (Figure 4 and Supplement Table 4). The average node degree value of the PPT network was 9.08, and the average local clustering coefficient was 0.604. The genes with higher values of degree, betweenness, and closeness above the median were acquired as the key targets of FLD for NAFLD. Consequently, the targets including ALB, MAPK1, CASP3, MARK8, AR, HSP90AA1, and EGFR were defined as key genes.
Figure 4.
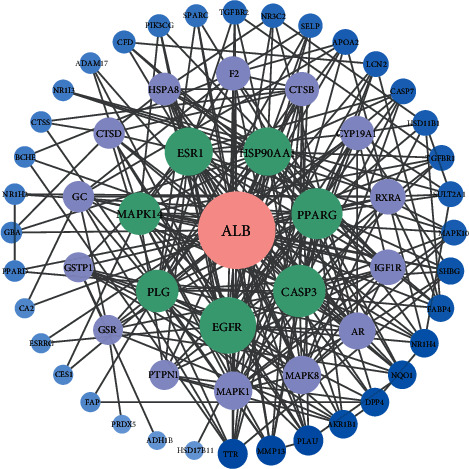
PPI network of FLD for NAFLD treatment. Each node represents a protein target, and each edge symbolizes the interaction between two nodes. The PPI network diagram is arranged according to the df. The greater the significance, the more central the node is.
3.5. GO and KEGG Pathway Enrichment Analysis
To further explore the biological functions of the 61 target genes of FLD for NAFLD, GO enrichment analysis was performed based on BP, CC, and MF and yielded 722 entries. BP enrichment analysis provided 617 entries, and the top 20 most enriched terms were presented in a bubble chart with cellular response to organic cyclic compound, steroid metabolic process, and response to steroid hormone as the top three (Figure 5(a)). CC analysis obtained 44 entries primarily involving the vesicle lumen, secretory granule lumen, and ficolin-1-rich granule lumen (Figure 5(b)). MF enrichment analysis revealed 61 entries with nuclear receptor activity, transcription factor activity, direct ligand regulated sequence-specific DNA binding, and lipid binding at the top ones (Figure 5(c)). To further investigate the biological processes of these targets, the KEGG pathway analysis was conducted and yielded 181 items (p<0.01), and the 16 pathways associated with NAFLD were displayed in Figure 5(d) with pathways in cancer, Th17 cell differentiation, and IL-17 signaling pathway as the top ones, indicating they might be essential pathways of FLD for NAFLD treatment and are worthy of further study. The pathway results were also intensively enriched in cancer, immune, bile secretion, substance metabolism, and apoptosis-related pathways. The detailed information of functional analysis is presented in Supplement Table 5.
Figure 5.
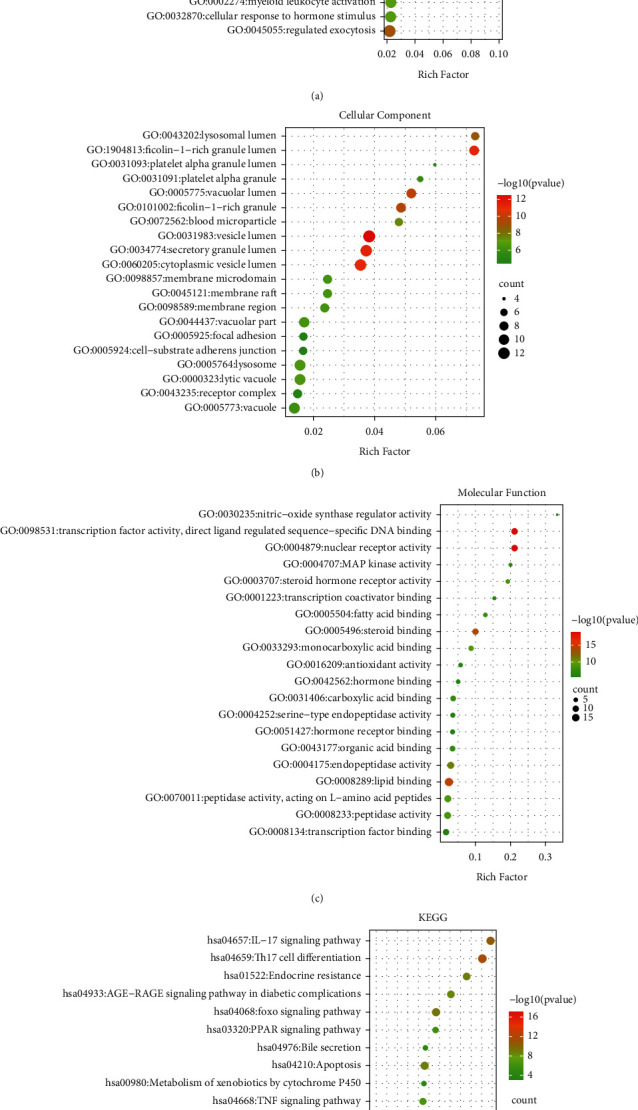
The 20 most significant of GO analysis (a) for BP, (b) for CC and (c) for MF and pathway enrichment (d) KEGG analysis of therapeutic target genes of FLD on NFALD.
3.6. Compound-Target-Pathway Network
The compound-target-pathway network contained 83 nodes (including 28 for compounds, 37 for targets, and 16 for pathways) and 268 edges (Figure 6). Analysis on the network revealed that multiple components from FLD targeted at least one gene, and formononetin (GC11) was considered the most potent compound that interacted with 17 genes. Most genes were regulated by at least two active compounds, and at least five genes were potentially involved in each pathway related to NAFLD. Moreover, MAPK8 and MAPK10 had the highest volume, followed by MAPK1, MAPK14, CASP3, and TGFBR1. The results revealed that FLD might exert therapeutic effects on NAFLD via modulating multiple targets and pathways with multiple compounds.
Figure 6.
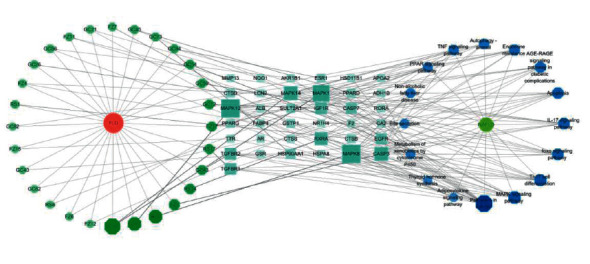
Component-target-pathway network. Green represents active ingredients of FLD; blue represents targets; dark blue represents signaling pathways of NAFLD.
3.7. Molecular Docking
The docking of the top ten active compounds obtained by the C-T network analysis with three potential core targets, CASP3, ALB, and MAPK1 acquired from the PPT network analysis, was performed. The information of compounds and targets is shown in Table 2 and Figure 7. The binding energies are presented in Table 3. A binding energy value less than 0 indicates that the ligand molecules can spontaneously bind to the receptor protein, while a value less than −5.0 kJ ·mol-1 suggests that the ligand molecules have a desirable binding affinity [15]. The results of the molecular docking analysis revealed that all compound-target pairs are lower than 0, indicating that every core compound has a good binding affinity to the top three targets. Among them, the 3′-methoxyglabridin and ALB presented the tightest binding energy (−9.5) with the LEU-481 and VAL-482 as active sites for hydrogen bond interaction (Figure 8). The docking prediction might provide a preliminary foundation for further investigation of drug targets.
Table 2.
Core active compounds of FLD.
| Molecule ID | Molecule name | Degree | Source |
|---|---|---|---|
| MOL000022 | 14-Acetyl-12-senecioyl-2E,8Z,10E-atractylentriol | 51 | BZ3 |
| MOL005320 | Arachidonate | 45 | RS11 |
| MOL002423 | Jesaconitine | 45 | FZ17 |
| MOL002879 | Diop | 44 | RS1 |
| MOL004974 | 3′-Methoxyglabridin | 44 | GC56 |
| MOL004898 | (E)-3-[3,4-Dihydroxy-5-(3-methylbut-2-enyl)phenyl]-1-(2,4-dihydroxyphenyl)prop-2-en-1-one | 40 | GC42 |
| MOL004492 | Chrysanthemaxanthin | 39 | RS6 |
| MOL002211 | 11,14-Eicosadienoic acid | 38 | FZ1 |
| MOL005360 | Malkangunin | 38 | RS17 |
| MOL005001 | Gancaonin H | 38 | GC77 |
Figure 7.
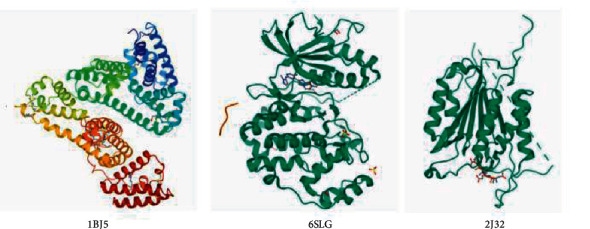
3D structure diagram of the top three target proteins.
Table 3.
The binding energy values of core compounds of FLD and core targets.
| Molecule name | Target | PDB ID | Binding affinity (KJ·mol−1) |
|---|---|---|---|
| 14-Acetyl-12-senecioyl-2E,8Z,10E-atractylentriol | ALB | 1BJ5 | −6.9 |
| MAPK1 | 6SLG | −5.3 | |
| CASP3 | 2J32 | −5.0 | |
|
| |||
| Arachidonate | ALB | 1BJ5 | −7.3 |
| MAPK1 | 6SLG | −5.0 | |
| CASP3 | 2J32 | −3.5 | |
|
| |||
| Jesaconitine | ALB | 1BJ5 | −7.0 |
| MAPK1 | 6SLG | −6.5 | |
| CASP3 | 2J32 | −5.3 | |
|
| |||
| Diop | ALB | 1BJ5 | −7.5 |
| MAPK1 | 6SLG | −6.0 | |
| CASP3 | 2J32 | −4.3 | |
|
| |||
| 3′-Methoxyglabridin | ALB | 1BJ5 | −9.5 |
| MAPK1 | 6SLG | −7.5 | |
| CASP3 | 2J32 | −7.4 | |
|
| |||
| (E)-3-[3,4-Dihydroxy-5-(3-methylbut-2-enyl)phenyl]-1-(2,4-dihydroxyphenyl)prop-2-en-1-one | ALB | 1BJ5 | −7.9 |
| MAPK1 | 6SLG | −7.1 | |
| CASP3 | 2J32 | −6.4 | |
|
| |||
| Chrysanthemaxanthin | ALB | 1BJ5 | −6.1 |
| MAPK1 | 6SLG | −9.1 | |
| CASP3 | 2J32 | −8.2 | |
|
| |||
| 11,14-Eicosadienoic acid | ALB | 1BJ5 | −6.3 |
| MAPK1 | 6SLG | −4.4 | |
| CASP3 | 2J32 | −4.4 | |
|
| |||
| Malkangunin | ALB | 1BJ5 | −6.8 |
| MAPK1 | 6SLG | −6.5 | |
| CASP3 | 2J32 | −7.1 | |
|
| |||
| Gancaonin H | ALB | 1BJ5 | −9.2 |
| MAPK1 | 6SLG | −7.6 | |
| CASP3 | 2J32 | −7.6 | |
ALB: albumin; MAPK1: mitogen-activated protein kinase 1; CASP3: caspase-3.
Figure 8.
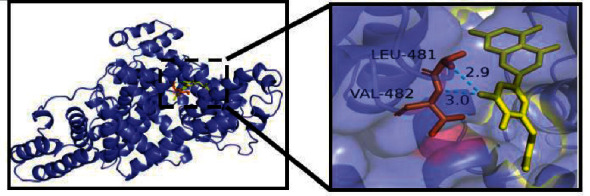
Molecular docking of 3′-methoxyglabridin and ALB. The dashed blue lines represent hydrogen bonds.
4. Discussion
The treatment of NAFLD remains a major public health challenge globally. Increasing lines of evidence indicate that FLD can treat NALFD effectively, but its mechanism of action remains obscure, especially from a perspective of holistic review. Network pharmacology can reveal the interaction relationships between multicompounds, multitargets, and multipathways of complex formula and therefore and therefore may play a guiding role in drug research and development [16]. Moreover, it is consistent with the holistic view of TCM. Therefore, this study utilized the network pharmacology approach with molecular docking to explore the potential molecular mechanism of FLZ's therapeutic effect on NAFLD to provide further insight.
In this study, 143 active compounds and 61 relative intersection targets were acquired. The topological analysis on the C-T network indicated the core compounds included sitosterol, beta-sitosterol, and ginsenoside rh2. Intriguingly, previous studies have shown that both sitosterol and beta-sitosterol can reduce hepatic lipid accumulation and regulate the hepatic lipid metabolism of NAFLD. In addition, sitosterol has been reported to reduce inflammation caused by FLD [17, 18]. Ginsenoside rh2 can reduce lipid deposition and improve glucose tolerance [19]. These previous investigations may imply that FLD is prone to exert therapeutic effects on NAFLD with the compound of sitosterol, beta-sitosterol, and ginsenoside rh2, which may provide a new idea and direction for further study.
The PPI network was constructed with 61 potential intersection targets. The results indicated that the active compounds in FLD might play pivotal roles in anti-NAFLD through the core targets such as albumin (ALB), mitogen-activated protein kinase1 (MAPKI), caspase-3 (CASP3), mitogen-activated protein kinase 8 (MAPK8), and androgen receptor (AR). ALB binding function has been reported as a novel biomarker to evaluate early liver damage and disease progression of NAFLD [20]. ALB binding function is strongly associated with NAFLD. Such key mediators of NAFLD as metabolism, reactive oxygen species, oxidative stress, and inflammation interfere with posttranslational modifications of ALB, which may partially explain why ALB binding function decreases earlier than other liver enzymes in NAFLD patients. Reduced ALB binding capacity may lead to toxic metabolite accumulation and poor antioxidant capacity, thus resulting in exacerbation of NAFLD [20, 21]. In addition, ALB could readily bind to Ca2+, Na+ and fatty acids and promote the occurrence and development of NAFLD through the interaction between Yes-associated protein and TGF-β signaling pathways [22]. Previous studies have also shown that MAPK1 can regulate liver lipid metabolism, and CASP3 plays a vital role in the initiation and propagation of apoptosis, which is involved in various disorders, such as neurodegenerative and inflammatory diseases [23, 24]. Moreover, MAPK8 was confirmed to be related to liver regeneration in mice and might contribute to a preventive effect on NAFLD [25]. In addition, extensive investigations have demonstrated that androgen plays an essential role in the process of glycolipid metabolism. As a member of the nuclear receptor superfamily distributed in many tissues and organs of the body, AR can increase the release of androgens, thus strengthening the regulation of liver lipid metabolism and maintaining the homeostasis of fat synthesis and decomposition [26]. It can also decrease the expression levels of TNF-α, IL-6, and IL-1β and reduce the damage of inflammatory factors to liver cells [27]. The above research results suggest that FLD may exert anti-NAFLD activity through modulating such core targets as ALB, MAPKI, CASP3, MAPK8, and AR.
GO and KEGG analyses were conducted using the Metascape data platform. The Go results suggested that putative targets were mainly enriched in cellular response to organic cyclic compound, steroid metabolic process, and response to steroid hormones in BP, vesicle lumen, secretory granule lumen, and ficolin-1-rich granule lumen in CC, and nuclear receptor activity, transcription factor activity, direct ligand regulated sequence-specific DNA binding, and lipid binding in MF. Additionally, the KEGG analysis yielded 181 entries, 16 of which were directly related to the NAFLD, and three pathways that significantly enriched, including pathways in cancer, Th17 cell differentiation, and IL-17 signaling pathway, were retrieved as core pathways for FLD to treat NAFLD. Specifically, the most aggregated targets were pathways in cancer, such as PPAR signaling, MAPK signaling pathway, and Estrogen signaling pathway (Figure 9). Their roles in regulating fat metabolism, improving liver fibrosis and cell apoptosis, and reducing oxidative damage are well-documented [28–30]. It is worth noting that GC and FZ in the FLD have 11 and 4 compounds that act on the pathways in cancer, respectively, indicating that modifying these two herbs may significantly influence the ratio of active compounds in FLD and provide references for conducting further experiments. Previous studies have also revealed that Th17 is involved in the inflammatory response of NAFLD progression to nonalcoholic steatohepatitis [31]. Moreover, IL-17 is classic proinflammatory cytokines and has proinflammatory effects that can accelerate NAFLD progression in mice [32]. Hence, it is rational to presume that various mechanisms may get involved in the FLD's actions on NAFLD.
Figure 9.
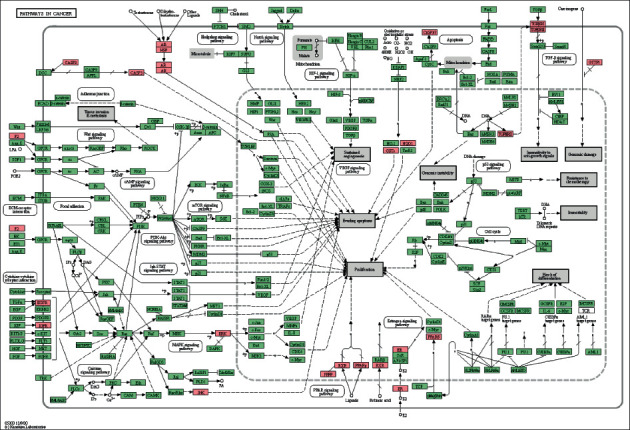
Potential target proteins of FLD regulating NAFLD on the predicted pathways (the pink nodes are potential target proteins of FLD, and the green nodes are relevant targets in the pathway).
Further, the molecular docking analysis results revealed that all compound-target pairs were lower than 0, indicating that all ten core compounds have good binding affinity to each of the three key targets, namely CASP3, ALB, and MAPK1. The docking pair of 3′-methoxyglabridin-ALB presented the tightest binding (−9.5), followed by chrysanthemaxanthin-MAPK1(−9.2) and Gancaonin H-ALB (−9.1), suggesting that the core compounds of FLD might relieve NAFLD through binding ALB, MAPK1, and H-ALB.
5. Conclusion
In conclusion, this network pharmacology-based study revealed the active compounds and potential mechanism through which FLD is effective on NAFLD by modulating various pathways, such as pathways in cancer, Th17 cell differentiation, and IL-17 signaling pathway. Subsequent molecular docking demonstrated that the top ten compounds of FLD presented desirable binding energy with CASP3, ALB, and MAPK1, further revealing the potentional mechanism of FLD's action on NAFLD. Thus, this study preliminarily reflected the multicomponent, multitarget, and multipathway characteristics of FLD and may provide some insights for future research and development of new anti-NAFLD drugs. However, further experiments are still needed to validate these findings. Special attention will be paid to interpret the mutual interactions between various compounds (or compound compatibility) that may attenuate potential toxicity, decode a structure-activity relationship between the compounds that suggests a chemical nucleus and implies a possibility of structural modification to increase activity, and identify the potential drug “cocktail” - a small collection of drug molecules ideally binding all desired variants [33].
Acknowledgments
This study was supported by the Key Research and Development Program of Science and Technology Department of Sichuan Province (grant nos. 2018SZ0078 and 2020JDZH0018), The fourth National TCM Doctor (Clinical And Basic) Excellent Talents Research and Study Program (grant number CMM no. 24 [2017]), the International Cooperation and Exchange Project of Sichuan Provincial Science and Technology Department (grant no. 2017HH0004), the Sichuan Provincial Administration of Traditional Chinese Medicine (grant no. 2021MS464), the National Natural Science Foundation of China (grant no. 81603537), the Youth Scholar Project of Chengdu University of Traditional Chinese Medicine (grant no. QNXZ2019043), and the “Double First-class” construction project of Chengdu University of Traditional Chinese Medicine (030041043).
Abbreviations
- FLD:
Fuzi Lizhong Decoction
- NAFLD:
Nonalcoholic fatty liver disease
- TCMSP:
Traditional Chinese Medicine Systems Pharmacology
- DL:
Drug-likeness
- OB:
Oral bioavailability
- GO:
Gene ontology
- BP:
Biological process
- KEGG:
Kyoto Encyclopedia of Genes and Genomes
- ALB:
Albumin
- MAPK1:
Mitogen-activated protein kinase 1
- CASP3:
Caspase-3
- MAPK8:
Mitogen-activated protein kinase 8
- AR:
Androgen receptor
- HSP90AA1:
Heat shock protein HSP 90-alpha
- EGFR:
Epidermal growth factor receptor
- CC:
Cellular component
- MF:
Molecular Function
- MAPK14:
Mitogen-activated protein kinase 14
- TGFBR1:
TGF-beta receptor type-1
- TCM:
Traditional Chinese medicine.
Contributor Information
Yun-Hui Chen, Email: chenyunhui@cdutcm.edu.cn.
Tian-E. Zhang, Email: zhte2003@cdutcm.edu.cn.
Data Availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on request.
Ethical Approval
As a study based on network pharmacology, ethical approval is not applicable.
Consent
Informed consent of patients is not applicable.
Conflicts of Interest
The authors declare that there are no conflicts of interest regarding the publication of this study.
Authors' Contributions
TEZ, YHC, and ZLconceptualized the study. ZL, LYX, YHC, and LLH were responsible for methodology and visualization. WYH, CJZ, DL, and YQW were responsible for software. YHC and ZL prepared the original draft. YHC and TEZ reviewed and edited the manuscripe. TEZ, YMX, and QZY were involved in supervision and project administration. All authors have read and agreed to the published version of the manuscript.
Supplementary Materials
Supplement Table 1: detailed information about the compound-target network. Supplement Table 2: information of 2,608 NAFLD-related targets. Supplement Table 3: information of 61 intersection targets between FLD and NAFLD. Supplement Table 4: detailed information about the protein-protein network. Supplement Table 5: detailed information about functional analysis.
References
- 1.Calzadilla Bertot L., Adams L. A. The natural course of nonalcoholic fatty liver disease. International Journal of Molecular Sciences . 2016;17(5) doi: 10.3390/ijms17050774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Younossi Z. M., Koenig A. B., Abdelatif D., Fazel Y., Henry L., Wymer M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology . 2016;64(1):73–84. doi: 10.1002/hep.28431. [DOI] [PubMed] [Google Scholar]
- 3.Maurice J., Manousou P. Nonalcoholic fatty liver disease. Clinical Medicine . 2018;18(3):245–250. doi: 10.7861/clinmedicine.18-3-245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cobbina E., Akhlaghi F. Nonalcoholic fatty liver disease (NAFLD) - pathogenesis, classification, and effect on drug metabolizing enzymes and transporters. Drug Metabolism Reviews . 2017;49(2):197–211. doi: 10.1080/03602532.2017.1293683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Parra-Vargas M., Rodriguez-Echevarria R., Jimenez-Chillaron J. C. Nutritional approaches for the management of nonalcoholic fatty liver disease: an evidence-based review. Nutrients . 2020;12(12) doi: 10.3390/nu12123860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jia-yao Y. A. N. G., Dong-qing T. A. O., Song L. I. U. Effects of three Wenyang Jianpi Tang on cell proliferation and apoptosis of nonalcoholic fatty liver cells. China Journal of Chinese Materia Medica . 2017;42(08):1591–1596. doi: 10.19540/j.cnki.cjcmm.20170222.010. [DOI] [PubMed] [Google Scholar]
- 7.Jia-yao Y. A. N. G., Zhao-hong S. H. I., Ma W. Effect of Fuzi Lizhong decoction in reducing liver injury of rats with nonalcoholic fatty liver via activating AMPK and suppressing NF-κBp65 pathway. China Journal of Chinese Materia Medica . 2018;43(15):3176–3183. doi: 10.19540/j.cnki.cjcmm.2018.0094. [DOI] [PubMed] [Google Scholar]
- 8.Jia-yao Y. A. N. G., Hong-feng Y. I., Liao Y. Effects of Fuzi Lizhong Decoction on fat deposition in nonalcoholic fatty liver cell model and related mechanism. Chinese Journal of Basic Medicine in Traditional Chinese Medicine . 2021;27(01):96–101. [Google Scholar]
- 9.Li S., Zhang B. Traditional Chinese medicine network pharmacology: theory, methodology and application. Chinese Journal of Natural Medicines . 2013;11(2):110–120. doi: 10.1016/s1875-5364(13)60037-0. [DOI] [PubMed] [Google Scholar]
- 10.Ru J., Li P., Wang J., et al. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. Journal of Cheminformatics . 2014;6(1):13. doi: 10.1186/1758-2946-6-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang X., Shen Y., Wang S., et al. PharmMapper 2017 update: a web server for potential drug target identification with a comprehensive target pharmacophore database. Nucleic Acids Research . 2017;45(W1):W356–w360. doi: 10.1093/nar/gkx374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Morgat A., Lombardot T., Coudert E. Enzyme annotation in UniProtKB using rhea. Bioinformatics . 2020;36(6):1896–1901. doi: 10.1093/bioinformatics/btz817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shannon P., Markiel A., Ozier O., et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Research . 2003;13(11):2498–2504. doi: 10.1101/gr.1239303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Trott O., Olson A. J. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. Journal of Computational Chemistry . 2010;31(2):455–461. doi: 10.1002/jcc.21334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhuang L., Yuan-yuan Z., Yao W.-F. The mechanism study of protecting kidney of Erzhi Pill based on network pharmacology. Acta Pharmaceutica Sinica . 2019;54(05):877–885. [Google Scholar]
- 16.Hopkins A. L. Network pharmacology: the next paradigm in drug discovery. Nature Chemical Biology . 2008;4(11):682–690. doi: 10.1038/nchembio.118. [DOI] [PubMed] [Google Scholar]
- 17.Feng S., Dai Z., Liu A. B., et al. Intake of stigmasterol and β-sitosterol alters lipid metabolism and alleviates NAFLD in mice fed a high-fat western-style diet. Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids . 2018;1863(10):1274–1284. doi: 10.1016/j.bbalip.2018.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Plat J., Baumgartner S., Houben T. Comment on Tauriainen et al: serum, liver and bile sitosterol and sitostanol in obese patients with and without NAFLD[J] Bioscience Reports . 2018;38(5) doi: 10.1042/bsr20180505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lu H., Yuan X., Zhang Y., et al. HCBP6 deficiency exacerbates glucose and lipid metabolism disorders in nonalcoholic fatty liver mice. Biomedicine & Pharmacotherapy . 2020;129 doi: 10.1016/j.biopha.2020.110347.110347 [DOI] [PubMed] [Google Scholar]
- 20.Sun L., Wang Q., Liu M., et al. Albumin binding function is a novel biomarker for early liver damage and disease progression in nonalcoholic fatty liver disease. Endocrine . 2020;69(2):294–302. doi: 10.1007/s12020-020-02319-z. [DOI] [PubMed] [Google Scholar]
- 21.Michal P., Philippe L., Bart S. Molecular mechanism of PPARα action and its impact on lipid metabolism, inflammation and fibrosis in nonalcoholic fatty liver disease. Journal of Hepatology . 2015;62(3):720–733. doi: 10.1016/j.jhep.2014.10.039. [DOI] [PubMed] [Google Scholar]
- 22.Chen P., Luo Q., Huang C., et al. Pathogenesis of nonalcoholic fatty liver disease mediated by YAP. Hepatology International . 2018;12(1):26–36. doi: 10.1007/s12072-017-9841-y. [DOI] [PubMed] [Google Scholar]
- 23.Xiao Y., Liu H., Yu J., et al. MAPK1/3 regulate hepatic lipid metabolism via ATG7-dependent autophagy. Autophagy . 2016;12(3):592–593. doi: 10.1080/15548627.2015.1135282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kong W. H., Sung D. K., Kim K. S., et al. Self-assembled complex of probe peptide - E. Coli RNA I conjugate and nano graphene oxide for apoptosis diagnosis. Biomaterials . 2012;33(30):7556–7564. doi: 10.1016/j.biomaterials.2012.06.086. [DOI] [PubMed] [Google Scholar]
- 25.Langiewicz M., Graf R., Humar B., Clavien P. A. JNK1 induces hedgehog signaling from stellate cells to accelerate liver regeneration in mice. Journal of Hepatology . 2018;69(3):666–675. doi: 10.1016/j.jhep.2018.04.017. [DOI] [PubMed] [Google Scholar]
- 26.Yu I.-C., Lin H.-Y., Sparks J. D., Yeh S., Chang C. Androgen receptor roles in insulin resistance and obesity in males: the linkage of androgen-deprivation therapy to metabolic syndrome. Diabetes . 2014;63(10):3180–3188. doi: 10.2337/db13-1505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chin K. Y., Ima-Nirwana S. The effects of testosterone deficiency and its replacement on inflammatory markers in rats: a pilot study. International Journal of Endocrinology and Metabolism . 2017;15(1) doi: 10.5812/ijem.43053.e43053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Besse-Patin A., Léveillé M., Oropeza D., Nguyen B. N., Prat A., Estall J. L. Estrogen signals through peroxisome proliferator-activated Receptor−γ coactivator 1α to reduce oxidative damage associated with diet-induced fatty liver disease. Gastroenterology . 2017;152(1):243–256. doi: 10.1053/j.gastro.2016.09.017. [DOI] [PubMed] [Google Scholar]
- 29.Shen X., Guo H., Xu J., Wang J. Inhibition of lncRNA HULC improves hepatic fibrosis and hepatocyte apoptosis by inhibiting the MAPK signaling pathway in rats with nonalcoholic fatty liver disease. Journal of Cellular Physiology . 2019;234(10):18169–18179. doi: 10.1002/jcp.28450. [DOI] [PubMed] [Google Scholar]
- 30.Wang Y., Nakajima T., Gonzalez F. J. PPARs as metabolic regulators in the liver: lessons from liver-specific PPAR-null mice. International Journal of Molecular Sciences . 2020;21(6) doi: 10.3390/ijms21062061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chackelevicius C. M., Gambaro S. E., Tiribelli C., Rosso N. Th17 involvement in nonalcoholic fatty liver disease progression to nonalcoholic steatohepatitis. World Journal of Gastroenterology . 2016;22(41):9096–9103. doi: 10.3748/wjg.v22.i41.9096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Harley I. T. W., Stankiewicz T. E., Giles D. A., et al. IL‐17 signaling accelerates the progression of nonalcoholic fatty liver disease in mice. Hepatology . 2014;59(5):1830–1839. doi: 10.1002/hep.26746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Radhakrishnan M. L., Tidor B. Optimal drug cocktail design: methods for targeting molecular ensembles and insights from theoretical model systems. Journal of Chemical Information and Modeling . 2008;48(5):1055–1073. doi: 10.1021/ci700452r. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplement Table 1: detailed information about the compound-target network. Supplement Table 2: information of 2,608 NAFLD-related targets. Supplement Table 3: information of 61 intersection targets between FLD and NAFLD. Supplement Table 4: detailed information about the protein-protein network. Supplement Table 5: detailed information about functional analysis.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on request.


