Abstract
Objective
To compare the severity of pulmonary embolism (PE) between patients with and without COVID, and to assess the association between severity and in-hospital-mortality.
Methods
We performed an analysis of 549 COVID (71.3% PCR-confirmed) and 439 non-COVID patients with PE consecutively included by 62 Spanish and 16 French emergency departments. PE-severity was assessed by size, the presence of right ventricular dysfunction (RVD), and the sPESI. The association of PE-severity and in-hospital-mortality was assessed both in COVID and non-COVID patients, and the interaction of COVID status and PE severity/outcome associations was also evaluated.
Results
COVID patients had PEs of smaller size (43% vs 56% lobar or larger, 42% vs. 35% segmental and 13% vs. 9% subsegmental, respectively; p = 0.01 for trend), less RVD (22% vs. 16%, p =0.02) and lower sPESI (p =0.03 for trend). Risk of in-hospital death was higher in COVID patients (12.8% vs. 5.3%, p < 0.001). PE-severity assessed by RVD and sPESI was independently associated with in-hospital-mortality in COVID patients, while PE size and sPESI were significantly associated with in-hospital-mortality in non-COVID. COVID status showed a significant interaction in the association of PE size and outcome (p =0.01), with OR for in-hospital mortality in COVID and non-COVID patients with lobar or larger PE of 0.92 (95%CI=0.19–4.47) and 4.47 (95%CI=1.60–12.5), respectively. Sensitivity analyses using only PCR-confirmed COVID cases confirmed these results.
Conclusion
COVID patients present a differential clinical picture, with PE of less severity than in non-COVID patients. An increased sPESI was associated with the risk of mortality in both groups but, PE size did not seem to be associated with in-hospital mortality in COVID patients.
Key words: Pulmonary embolism, Mortality, COVID, Risk factors, Severity, PESI
1. Introduction
Infection by SARS-Cov-2 is mainly characterized by fever and respiratory symptoms, with dyspnea and lung infiltrates in more severe cases[1,2]. Many patients also present a procoagulant state, which is biochemically detected by increased D-dimer levels and is related to complications and a worse prognosis[1]. Accordingly, some authors have suggested that pulmonary embolism (PE) is more common in patients with COVID-19 than in the uninfected population[3], [4], [5], [6], [7]. A recent meta-analysis of 66 studies that included 23,117 COVID patients that had been hospitalized reported a PE prevalence rate of 7.8% (95% confidence interval [CI]=6.2–9.4)[8].
Nonetheless, the severity of PE in COVID patients remains to be established. On one hand, the size of the pulmonary arteries in which PE occurs in COVID patients does not seem to be as large as in PE in non COVID patients, although this has not been properly assessed. Although only a small fraction of PEs (8%) was reported as subsegmental in the previously commented meta-analysis [8], other case series reported that in more than half of PEs in COVID patients the size of the involved arteries was segmental at most [9,10]. PEs associated with COVID may result from a hyperinflammatory state that leads to a pro-coagulant state, whereas in non-COVID patients PEs are often subsequent to a deep venous thrombosis (DVT), which may be of larger size. On the other hand, the frequency of right ventricular dysfunction (RVD) in large series of COVID patients with PE has not been previously described, and classification using classical indexes, such as the simplified Pulmonary Embolism Severity Index (sPESI) score, has seldom been reported. Finally, it is not yet known how all these PE severity markers correlate with COVID patient outcome. In non-COVID patients, it is well described that patients with subsegmental PEs have a very low mortality risk, while the presence of a RVD is associated with worse prognosis[11]. Bearing in mind all these gaps in the current knowledge, the present large, retrospective international, multicenter study aimed to describe differences in PE severity between COVID and non-COVID patients. The secondary objective was to investigate whether there are significant relationships between the estimated PE severity and mortality in COVID and non-COVID patients and if these relationships differ between these two groups of patients.
2. Methods
2.1. Study design and setting
This is an ancillary analysis of two retrospective large cohorts. On one hand, the SIESTA cohort is a multipurpose Spanish cohort generated by 62 EDs that included all COVID patients diagnosed with 1 of the 10 unusual manifestations subject to investigation as well as randomly selected non-COVID patients with the same manifestation included as controls. One of these manifestations was PE, and all COVID and non-COVID patients diagnosed with PE by a computed tomographic pulmonary angiogram (CTPA) form this cohort were included in the present study, irrespective of the severity of PE and the patient hemodynamic status. The COVID patients with PE included in the present analysis were recruited in the 62 EDs during March-April 2020 (during the first wave of the COVID pandemic), while and non-COVID patients with PE were recruited during the same period (first wave) as well as during March-April 2019 (one year before the COVID pandemic). Extensive details of the SIESTA protocol have been extensively described elsewhere[12], [13], [14]. On the other hand, the PEPCOV cohort is an international cohort (with the participation of 26 centers from France, Spain, Italy, Belgium, Chile and Canada) that included all patients in whom a CTPA was performed during patient evaluation in the Emergency Department (ED) between February 1 to April 10, 2020. All COVID and non-COVID patients included in the PEPCOV registry coming from the 16 French EDs with a final diagnosis of PE based on CTPA findings were included in the present analysis, irrespective of the severity of PE and the patient hemodynamic status. Extensive details of the PEPCOV protocol and have been extensively described elsewhere[7,15,16].
COVID diagnosis was made based on SARS-CoV-2 RNA detection in a nasopharyngeal swab by reverse transcriptase polymerase chain reaction (RT-PCR). Spanish and French hospitals experienced a huge shortage of tests to confirm SARS-CoV-2 infection during several weeks of the first wave of the COVID-19 pandemic[17,18], and therefore, COVID diagnosis was also accepted in the epidemiological context of the first COVID pandemic wave by the presence of a clinically compatible clinical picture of SARS-CoV-2 infection (including at least malaise, fever and cough) and a CTPA with typical findings (i.e. bilateral interstitial lung ground-glass infiltrates, peripheral consolidations, or crazy-paving).
2.2. PE diagnosis and severity assessment
All CTPA were requested by ED physicians. Every diagnosis of PE was confirmed by a senior radiologist at a local level.
The severity of PE was estimated in three different ways. First, according to PE size, that was classified by the localization (or "size") of the most proximal artery involved: (1) lobar, defined when lobar or larger arteries were involved; (2) segmental, defined when only segmental with or without subsegemental arteries were involved, and (3) subsegmental, when PE was limited to subsegmental arteries. Second, according to the presence of RVD in the CTPA, defined as a right ventricle / left ventricle diameter ratio ≥ 1. And third, the sPESI score was calculated in every patient based on retrospective chart review[19]. We also recorded a concomitant diagnosis of deep venous thrombosis (DVT) in patients coming from the SIESTA cohort (as this data had not been recorded in patients coming from the PEPCOV cohort).
2.3. Independent variables
The following data were collected from patients in the two registries: nine baseline characteristics (demographic: age, sex; comorbidity: hypertension, chronic heart failure, chronic renal disease; risk factors for PE: active cancer, previous DVT, immobilization or surgery the previous 30 days and treatment with estrogens) and 12 clinical characteristics of the index episode (clinical manifestations: shortness of breath, chest pain, leg pain/edema, hemoptysis, length of symptoms; vitals at ED arrival: systolic blood pressure, heart rate, pulse oxymetry, temperature; analytical findings: D-dimer, C- reactive protein –CRP-, leukocytes).
2.4. Outcome
Patients were followed until hospital discharge. The outcome considered in the present study was in-hospital all-cause mortality that was adjudicated at a local level. At the time of performing the present analysis, all patients had finished the index episode (i.e., they had been discharged home or had died during hospital admission).
2.5. Statistical analysis
Continuous variables were presented as median (interquartile range –IQR-) and discrete variables as absolute values and percentages. The characteristics of COVID and non-COVID patients were compared with the non-parametric Mann-Whitney test and the chi-square test for continuous and discrete variables, respectively. When discrete variables represented an ordinal variable, chi-square for trend was used.
Associations with in-hospital mortality were tested for PE size (dichotomized as lobar vs. segmental/subsegmental), the presence of RVD, and sPESI (dichotomized as 0 vs. ≥1 points) and expressed as odds ratio (OR) with 95% CI, first unadjusted, and then progressively adjusting the OR (aOR) for baseline patient characteristics (model A), clinical characteristics of the index episode (model B) and by both types of characteristics (model C, fully adjusted). For the adjusted models, we created 10 datasets in which missing values in the covariates were replaced by imputed values using the multiple imputation technique provided by SPSS software, which is based on random drawings of imputed data from a Bayesian posterior distribution, and we used Mersenne twister as pseudorandom number generator and 2000,000 as seed. In the fully adjusted model, we checked the existence of a first-order interaction of COVID status in the associations between PE severity markers and outcomes. Analyses regarding the sPESI score and its association with COVID status and outcomes were only reported unadjusted, because most of the items used for adjustment are comprised in the sPESI score itself. As sensitivity analysis, we repeated all calculations using only including in the COVID group those patients with PCR confirmation of -SARS-CoV-2 infection. Statistical significance of differences between groups was accepted if p <p<0.05 or the 95%CI of the OR excluded the value 1. The SPSS v.25 (IMB, Armonk, NY, USA) and InStat v 3.0 (GraphPad Software, San Diego, CA, USA) packages were used for statistical calculations.
2.6. Ethics
The SIESTA cohort was approved by the Ethics Committee of the Hospital Clínic of Barcelona (Spain; reference number HCB/2020/0534) and the PEPCOV cohort by the Steering Committee of Assistance Publique–Hôpitaux de Paris. Due to the retrospective, non-interventional nature of the cohorts, and the urgent need for information during the first wave of the COVID pandemic, informed consent was waived in all the participating centers of the two cohorts. The present study was carried out in strict compliance with the principles of the Declaration of Helsinki.
3. Results
The present analysis included 988 patients with PE, with the SIESTA cohort providing 677 patients from 62 Spanish EDs, and the PEPCOV cohort providing 311 patients from 16 French EDs. The median age was 67 years (IQR 54–78), and 46.5% were females. Of these, 549 were COVID patients (SIESTA: 339; PEPCOV: 210), and 439 were non-COVID patients (SIESTA: 338; PEPCOV: 101). SARS-CoV-2 infection was microbiologically confirmed by RT-PCR in 71.3% of COVID patients (SIESTA: 72.8%, PEPCOV: 66.0%; p =0.196). Compared to non-COVID patients with PE, COVID patients with PE were younger, more frequently males, risk factors for PE were less frequent, and symptom duration was longer before ED consultation, and they had less leg pain/edema, lower systolic blood pressure and pulse oxymetry and a higher temperature and CRP values (Table 1 ). A concomitant diagnosis of DVT was made in 38.1% of COVID patients with PE and in 17.5% of non-COVID patients with PE (p <0.001).
Table 1.
Clinical characteristics of patients with pulmonary embolism included in the present study.
| Total(N = 988)n (%) | Missing valuesn (%) | COVID(N =439)n (%) | Non-COVID(N =549)n (%) | p | |
|---|---|---|---|---|---|
| Baseline characteristics | |||||
| Demographic data | |||||
| Age (years) [median (IQR)] | 67 (54–78) | 0 (0) | 65 (54–77) | 65 (54–80) | 0.046 |
| Sex female | 459 (46.5) | 0 (0) | 177 (40.3) | 282 (48.6) | 0.001 |
| Comorbidity | |||||
| Hypertension | 463 (46.9) | 1 (0.1) | 199 (45.3) | 264 (48.2) | 0.373 |
| Chronic heart failure | 66 (6.7) | 1 (0.1) | 24 (5.5) | 42 (7.7) | 0.170 |
| Chronic renal disease | 41 (4.3) | 1 (0.1) | 19 (4.3) | 22 (4.0) | 0.806 |
| Risk factors for pulmonary embolism | |||||
| Active cancer | 171 (17.3) | 1 (0.1) | 49 (11.2) | 122 (22.3) | <0.001 |
| Previous deep venous thrombosis | 137 (13.9) | 2 (0.2) | 28 (6.4) | 109 (19.9) | <0.001 |
| Immobilization/Surgery the previous month | 100 (10.1) | 2 (0.2) | 40 (9.1) | 60 (10.9) | 0.348 |
| On estrogen treatment | 31 (3.1) | 1 (0.1) | 6 (1.4) | 25 (4.6) | 0.004 |
| Clinical characteristics of the index episode | |||||
| Clinical manifestations | |||||
| Shortness of breath | 701 (71.0) | 0 (0) | 323 (73.6) | 378 (68.9) | 0.104 |
| Chest pain | 342 (34.7) | 1 (0.1) | 140 (32.0) | 202 (36.8) | 0.113 |
| Leg pain/edema | 218 (22.1) | 1 (0.1) | 56 (12.8) | 162 (29.5) | <0.001 |
| Hemoptysis | 29 (2.9) | 2 (0.2) | 12 (2.7) | 17 (3.1) | 0.738 |
| Length of symptoms (days) [median (IQR)] | 4 (1–10) | 31 (3.1) | 7 (3–13) | 3 (1–7) | <0.001 |
| First vitals at emergency department arrival | |||||
| Systolic blood pressure (mmHg) [median (IQR)] | 130 (116–147) | 2 (0.2) | 129 (113–142) | 133 (118–150) | <0.001 |
| Heart rate (bpm) [median (IQR)] | 93 (80–110) | 2 (0.2) | 93 (82–110) | 93 (79–110) | 0.262 |
| Pulse oxymetry (%)[median (IQR)] | 95 (92–97) | 11 (1.1) | 95 (91–97) | 96 (93–98) | <0.001 |
| Temperature ( °C) [median (IQR)] | 36.5 (36.0–37.1) | 4 (0.4) | 36.6 (36.0–37.3) | 36.5 (36.0–37.0) | 0.010 |
| Analytical findings | |||||
| D-dimer (ng/mL)[median (IQR)] | 4526 (1765–10,000) | 156 (15.8) | 5024 (1760–13,237) | 4340 (1771–8890) | 0.114 |
| CRP (mg/dL) [median (IQR)] | 53 (16–122) | 148 (15.0) | 84 (29–164) | 31 (11–86) | <0.001 |
| Leucocytes (cells/µL) [median (IQR)] | 9.2 (7.0–11.9) | 11 (1.1) | 9.5 (6.9–12.1) | 9.0 (7.2–11.5) | 0.409 |
*P calculated by chi-square for trend for qualitative variables, and by linear regression for quantitative variables
Bold numbers denote statistical significance (p <0.05)
RVD: right ventricular dysfunction; CRP: C-reactive protein; IQR: interquartile range
In patients with COVID, PEs were of more distal topography compared to non-COVID patients: 43% lobar, 44% segmental and 13% sub-segmental vs. 56%, 35% and 9%, respectively (p <0.001 for trend, Table 2 ). PEs were lobar or larger in 189 COVID patients (43%) and in 241 non-COVID patients (56%) (difference 13%, 95%CI 7% to 19%, p <0.001, Table 2). The presence of RVD on CTPA was less frequent in COVID patients compared to non-COVID patients: 22% vs. 16% (difference 6%, 95%CI 1% to 10%, p =0.026). COVID patients had a significantly lower sPESI score than non-COVID patients (p =0.033 for trend, Table 2). Similar results were obtained in the sensitivity analysis including only COVID cases confirmed by PCR, although the lower sPESI score in COVID group did not reach statistical significance (Table 2).
Table 2.
Severity of patients with pulmonary embolism included in the present study.
| Total(N =988)n (%) | COVID(all clinically diagnosed)(N =439)n (%) | COVID(only PCR-confirmed)(N =308)n (%) | Non-COVID(N =549)n (%) | p value* | p value⁎⁎ | |
|---|---|---|---|---|---|---|
| According to the size of the pulmonary embolism | <0.001⁎⁎⁎ | <0.001 | ||||
| Only subsegmental | 105 (10.6) | 56 (12.8) | 39 (12.7) | 49 (8.9) | ||
| Segmental at most | 386 (39.1) | 194 (44.2) | 142 (46.1) | 192 (35.0) | ||
| Lobar or larger | 497 (50.3) | 189 (43.1) | 127 (41.2) | 308 (56.1) | ||
| According to the presence of right ventricular dysfunction | 0.026 | 0.12 | ||||
| No | 795 (80.5) | 367 (83.6) | 262 (85.1) | 428 (78.0) | ||
| Yes | 193 (19.5) | 72 (16.4) | 46 (14.9) | 121 (22.0) | ||
| According to the simplified PESI score | 0.033⁎⁎⁎ | 0.144 | ||||
| 0 points | 388 (39.3) | 178 (40.5) | 124 (40.3) | 210 (38.3) | ||
| 1 point | 372 (37.7) | 178 (40.5) | 120 (39.0) | 194 (35.3) | ||
| 2 points | 173 (17.5) | 64 (14.6) | 49 (15.9) | 109 (19.9) | ||
| >2 points | 55 (5.6) | 19 (4.3) | 15 (4.9) | 36 (6.6) |
Bold numbers denote statistical significance (p <0.05)
p value refereed to comparison between COVID patients (all clinically diagnosed) and non-COVID patients.
p value refereed to comparison between COVID patients (only PCR-confirmed) and non-COVID patients.
p value calculated by chi-square for trend for qualitative variables
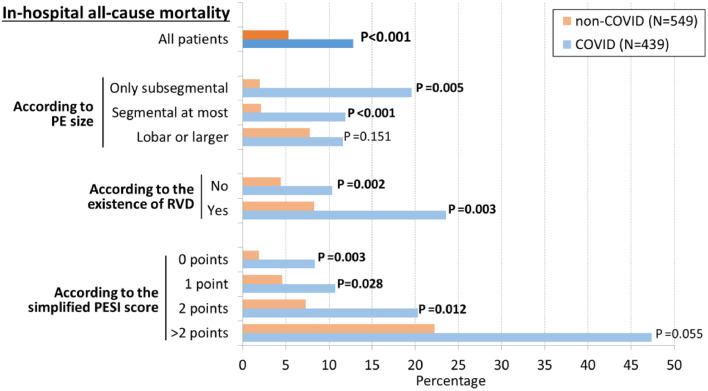
Eighty-five patients died during hospitalization (8.6%), and the risk of in-hospital death was higher in COVID patients (56 deaths, 12.8%) than in non-COVID patients (29 deaths, 5.3%; difference 7%, 95%CI 4% to 11%, p <0.001; Fig. 1 ). In-hospital mortality was always higher in COVID patients in all subgroup analyses based on PE size, DVD and sPESI score, and these differences were always statistically significant, with the exception of patients with PE size that was lobar or larger (p =0.151) and patients with a sPESI score of >2 points (p =0.055, Fig. 1).
Fig. 1.
Comparison of outcomes between COVID and non-COVID patients with pulmonary embolism, overall and according to the severity of pulmonary embolism (assessed by pulmonary embolism size, the existence of right ventricular dysfunction and the simplified Pulmonary Embolism Severity Index [PESI] score)
PE: pulmonary embolism; RVD: right ventricular dysfunction
Bold numbers denote statistical significance (p <0.05).
Regarding the relationship between PE severity and outcome, independent predictors of in-hospital mortality were RVD in COVID patients (aOR=3.4, 95%CI=1.5–7.5) and lobar PE size in non-COVID patients (aOR for PE lobar or larger =5.0, 95%CI=1.6–16.4) (Table 3 ). Additionally, a sPESI score >0 was associated with increased in-hospital mortality in both COVID and non-COVID patients (OR=2.03, 95%CI=1.08–3.78, and OR=4.10, 95%CI=1.41–11.95; respectively) (Table 3). Results obtained in the sensitivity analysis confirmed all these findings (Table 3). COVID status exhibited significant interaction in the relationship between PE size and outcome (p =0.01), but not in the relationship between RVD or sPESI and outcome (p =0.641 and p =0.265, respectively, Fig. 2 ). Similar results were obtained in the sensitivity analysis, with p values for interaction of 0.041, 0.505 and 0.255, respectively.
Table 3.
Unadjusted and adjusted in-hospital all-cause mortality according to the severity of pulmonary embolism (assessed by the size of the pulmonary embolism, the presence of right ventricular dysfunction and the simplified Pulmonary Embolism Severity Index (PESI) score in COVID and non-COVID patients.
| COVID patients(all clinically diagnosed)Odds Ratio (95% CI) | COVID patients(only PCR-confirmed)Odds Ratio (95% CI) | Non-COVID patientsOdds Ratio (95% CI) | |
|---|---|---|---|
| According to the size of pulmonary embolism | |||
| Segmental/subsegmental | 1 (Ref.) | 1 (Ref.) | 1 (Ref.) |
| Lobar or larger | |||
| Unadjusted | 1.182 (0.757–1.847) | 1.327 (0.827–2.128) | 3.989 (1.499–10.616) |
| Adjusted (model A, adjusted by baseline characteristics) | 1.101 (0.701–1.728) | 1.275 (0.793–2.052) | 4.459 (1.627–12.219) |
| Adjusted (model B, adjusted by clinical characteristics of episode) | 1.265 (0.787–2.033) | 1.475 (0.885–2.458) | 3.516 (1.213–10.198) |
| Adjusted (model C, fully adjusted) | 1.224 (0.758–1.979) | 1.471 (0.879–2.463) | 5.042 (1.551–16.384) |
| According to the existence of right ventricular dysfunction in CTPA | |||
| No | 1 (Ref.) | 1 (Ref.) | 1 (Ref.) |
| Yes | |||
| Unadjusted | 2.600 1.375–4.916) | 2.017 (1.205–3.377) | 1.939 (0.877–4.290) |
| Adjusted (model A, adjusted by baseline characteristics) | 3.346 (1.670–6.704) | 2.059 (1.223–3.468) | 2.227 (0.972–5.106) |
| Adjusted (model B, adjusted by clinical characteristics of episode) | 2.438 (1.174–5.064) | 1.895 (1.096–3.275) | 1.518 (0.643–3.873) |
| Adjusted (model C, fully adjusted) | 3.378 (1.519–7.513) | 1.961 (1.123–3.424) | 1.861 (0.723–4.791) |
| According to the simplified PESI score | |||
| 0 points | 1 (Ref.) | 1 (Ref.) | 1 (Ref.) |
| ≥1 points | |||
| Unadjusted* | 2.025 (1.084–3.784) | 2.232 (1.292–3.857) | 4.100 (1.406–11.954) |
CTPA: computerized tomography pulmonary angiogram, ICU: intensive care unit; Ref.: reference
Bold numbers denote statistical significance (p <0.05)
Evaluation of in-hospital all-cause mortality according to simplified PESI score was not adjusted as many of the covariates used for adjustments are already included in the simplified PESI
Fig. 2.
Analysis of interaction of COVID status on the relationship between pulmonary embolism severity (assessed by pulmonary embolism size, existence of right ventricular dysfunction and simplified Pulmonary Embolism Severity Index [PESI] score) and adjusted in-hospital all-cause mortality.
*Evaluation of in-hospital all-cause mortality according to simplified PESI score was not adjusted as many of the covariates used for adjustments are already included in the simplified PESI
PE: pulmonary embolism; RVD: right ventricular dysfunction
Bold numbers denote statistical significance (p <0.05).
A significant increase of risk of in-hospital mortality in both COVID and non-COVID patients was observed as the number of severity markers (lobar or larger PE, RVD, sPESI score >0) increased, with mortality rising from 8.7% in COVID patients with no severity marker to 24.2% when the three markers were present (p =0.022; from 12.3% to 38.1% in the sensitivity analysis using only COVID patients confirmed by PCR; p =0.020), and from 0% to 11.7%, respectively, in non-COVID patients (p <0.001, Table 4 ). The p value for interaction of COVID status with the relationship between number of severity markers and outcome was 0.06 (0.10 in the sensitivity analysis).
Table 4.
Comparison of in-hospital mortality in COVID and non-COVID patients according to the number of markers of pulmonary embolism severity (PE size lobar or larger, right ventricular dysfunction and sPESI score >0).
| Number of severity markers being present in patients with pulmonary embolism(PE size lobar or larger; RVD; sPESI score >0) | |||||
|---|---|---|---|---|---|
| None | One(any marker) | Two(in any combination) | Three(all present) | p value(for trend) | |
| COVID patients (all clinically diagnosed) | |||||
| Number of cases (%) | 92 (21.0) | 205 (46.7) | 109 (24.8) | 33 (7.5) | |
| Number of deaths (% of in-hospital mortality) | 8 (8.7) | 24 (11.7) | 16 (14.7) | 8 (24.2) | 0.024 |
| Odds ratio (95% confidence interval) | 1 (Ref.) | 1.39 (0.60–3.23) | 1.81 (0.74–4.44) | 3.36 (1.15–9.86) | |
| COVID patients (only PCR-confirmed) | |||||
| Number of cases (%) | 65 (21.1) | 150 (48.7) | 72 (23.4) | 21 (6.8) | |
| Number of deaths (% of in-hospital mortality) | 8 (12.3) | 20 (13.3) | 12 (16.7) | 8 (38.1) | 0.020 |
| Odds ratio (95% confidence interval) | 1 (Ref.) | 1.10 (0.46–2.64) | 1.42 (0.54–3.74) | 4.39 (1.39–13.9) | |
| Non-COVID patients | |||||
| Number of cases (%) | 85 (15.5) | 220 (40.1) | 184 (33.5) | 60 (10.9) | |
| Number of deaths (% of in-hospital mortality) | 0 (0) | 6 (2.7) | 16 (8.7) | 7 (11.7) | <0.001 |
| Odds ratio (95% confidence interval)* | 1 (Ref.) | 5.18 (0.29–93.1) | 16.9 (0.99–282) | 24.0 (1.34–429) | |
| Subgroups comparison of in-hospital mortality between COVID and non-COVID patients | |||||
| p value (using all clinically diagnosed COVID patients) | 0.007 | <0.001 | 0.13 | 0.14 | - |
| p value (using only PCR-confirmed COVID patients) | 0.003 | <0.001 | 0.11 | 0.11 | - |
PE: pulmonary embolism; RVD: right ventricular dysfunction; sPESI: simplified Pulmonary Embolism Severity Index
Bold numbers denote statistical significance (p <0.05)
Odds ratio and confidence interval was calculated using the approximation of Woolf. Since at least one value was zero, 0.5 was added to each value to make calculations possible.
4. Discussion
In this retrospective analysis of Spanish and French cohorts of patients diagnosed with PE in the ED during the first wave of the COVID-19 pandemic, PE severity differed between COVID and non-COVID patients, with the former presenting PEs of lower severity. This study confirms that a higher sPESI score was associated with a higher risk of in-hospital mortality in both groups. However, PE topography did not seem to be associated with an increased risk of in-hospital mortality in COVID patients.
In a previous analysis of the full PEPCOV cohort, COVID status was not associated with a higher risk of PE diagnosis in the ED[16]. The present study suggests that PE severity is different between COVID and non-COVID patients, with COVID patients presenting PE with a more distal topography, and less risk of RVD and a lower sPESI score. It is reported that the higher incidence of thromboembolism in COVID patient may be caused by hypercoagulability subsequent to an hyperinflammatory state[20]. These prothrombic abnormalities are associated with a higher risk of PE or disseminated intravascular coagulation, which is associated with a higher risk of mortality [21]. Autopsy studies reported that both macro- and microvascular thrombosis occurs frequently in COVID patients[22]. This may be in contrast with the physiopathology of PE in non-COVID patients, which are predominantly subsequent to DVT and may explain the difference in PE size in these two groups. Another plausible reason for the smaller size of PEs in COVID patients lies in the fact that other symptoms (many of which are not directly related to PE but to COVID itself) may lead patients to visit the ED and thereby be diagnosed with PE at an earlier stage. This would also explain at least partially that COVID patients present in the ED with PEs of lesser severity, with a lower percentage of RVD and a lower sPESI score.
Despite this milder severity, patients with COVID exhibited a higher in-hospital mortality for trivial reasons: COVID patients included in this study presented some symptoms of severity that led to ED visit and subsequent CTPA. The median age in this sample was 67 years, with a reported mortality in similar populations ranging from 10 to 20% in previous studies[23,24]. Nonetheless, there are scarce reports assessing risk factors for in-hospital mortality among COVID patients with PE. This study confirms what was described in non-COVID patients: higher sPESI scores and the presence of RVD are associated with a higher risk of mortality. Accordingly, the present findings suggest that the usual tools for PE risk stratification may be valid even in COVID patients[19,25,26]. Nonetheless, the poorer prognosis associated with RVD could be more related to acute changes in the pulmonary vascular system pressure resulting from the extensive lung parenchymal lesions caused by COVID itself rather than to circulatory obstructions by clots, which involve smaller arteries than in non-COVID patients. Interestingly, the size of the PE does not seem to have prognostic value in COVID patients: even with subsegmental PEs, 11 out of 56 (20%) of COVID patients died in the hospital. Since subsegmental PEs are usually associated with a very low risk of mortality, this suggests that COVID itself rather than PE largely drives the prognosis of patients with PE[27].
Finally, it is remarkable that the number of severity markers present in a particular patient, either COVID or non-COVID, was directly correlated with in-hospital mortality. However, while non-COVID patients with a low risk PE (non-lobar or larger PE, no RVD and sPESI=0) had a very low risk of mortality (0% in our series) as previously reported, this was not the case for COVID patients who had a substantial mortality risk (8.7% in our series) and warranted hospital admission and closer monitoring.
4.1. Limitations
This study has some limitations. Firstly, the collection of data was retrospective, and it is likely that some data were not clearly reported in the medical notes, and therefore, might not have been properly collected in our database. This limitation, inherent to retrospective chart reviews, may be of limited extent because we mostly analyzed data that were reliably reported in medical charts, such as CTPA reports with characteristics of emboli. However, some items comprised in the sPESI score may not have been routinely collected and reported in the medical notes, particularly regarding past medical history. Another major limitation is the assessment of RVD that was only adjudicated in CTPA reports. The diagnostic performance of CTPA alone to diagnose RVD is unknown, and it is likely that the analysis of cardiac biomarkers and systematic echocardiography would have been more precise to detect the presence of RVD. Of note, the recent European guidelines recommend that PE severity be assessed depending on the numbers of markers of evidence of RVD, assessed with these three options[28]. The fact that only one was used to assess RVD in this study may explain the absence of association with in-hospital mortality in non-COVID patients with PE. An additional limitation is that there might have been a selection bias as described previously. Which patients underwent a CTPA for suspected PE was not uniform across the different EDs, and whether this represents the usual ED patient management is unknown, as patients were included during a very peculiar time (COVID outbreak). The 5% mortality rate of non-COVID patients with PE is consistent with what has previously been reported, suggesting that these results are likely valid in unselected ED patients with thromboembolism. Moreover, the present results only apply for PE diagnosed in the ED, and this is a very particular scenario, especially during the first pandemic wave[29,30]. Since then, some additional pandemic waves have passed and prevalence and/or severity of PE could have changed, making our findings during the first wave not directly applicable to these successive waves. Therefore, readers should to be into account that COVID patients developing PE during hospitalization or in further pandemic waves could exhibit a different severity pattern with a different relationship with mortality. Finally, there was no size calculation and, accordingly, we could have committed a type-II error in some of our estimations.
4.2. Conclusion
In this retrospective analysis of patients with PE diagnosed in the ED from two cohorts in Spain and France, patients with COVID exhibited PEs of less severity. However, COVID was associated with an increased risk of in-hospital mortality. This study suggests that the usual tools for risk stratification, specifically the sPESI and RVD, are still valid in COVID patients, while the size of a PE is not valid in this particular population infected by SARS-CoV-2.
Declaration of Competing Interest
None.
Appendix
The SIESTA network is formed by the following researchers and centers (all from Spain):
Steering Committee: Òscar Miró, Sònia Jiménez (Hospital Clínic, Barcelona), Juan González del Castillo, Francisco Javier Martín-Sánchez, Eric Jorge García-Lamberechts (Hospital Clínico San Carlos, Madrid), Pere Llorens (Hospital General de Alicante), Guillermo Burillo-Putze (Hospital Universitario de Canarias, Tenerife), Alfonso Martín (Hospital Universitario Severo Ochoa de Leganés, Madrid), Pascual Piñera Salmerón (Hospital General Universitario Reina Sofía, Murcia), Aitor Alquézar-Arqué (Hospital de la Santa Creu i Sant Pau), Javier Jacob (Hospital Universitari de Bellvitge, Barcelona).
Participating centres:
-
1
Hospital Universitario Doctor Peset Aleixandre de Valencia: María Luisa López Grima.
-
2
Hospital Universitario y Politécnico La Fe de Valencia: Javier Millán.
-
3
Hospital Universitario General de Alicante: Bárbara Peña, Begoña Espinosa.
-
4
Hospital Clínico Universitario de Valencia: José Noceda.
-
5
Hospital Arnau de Vilanova de Valencia: María José Cano.
-
6
Hospital Francesc de Borja de Gandía, Valencia: María José Fortuny Bayarri.
-
7
Hospital General Universitario de Elche, Alicante: Blas Jiménez.
-
8
Hospital Marina Baixa de Villajoyosa de Alicante: Juan Miguel Porrino.
-
9
Hospital Virgen de los Lirios, Alcoy Alicante: Napoleón Meléndez.
-
10
Hospital Universitario Vinalopó de Elche (Alicante): Matilde González Tejera.
-
11
Hospital Universitario de Torrevieja de Alicante: Rigoberto del Rio.
-
12
Hospital Lluis Alcanys de Xativa: Carles Pérez García.
-
13
Hospital Universitario de La Ribera de Valencia: José Vicente Brasó Aznar.
-
14
Hospital de la Vega Baja Orihuela de Alicante: María Carmen Ponce.
-
15
Hospital Universitario Sant Joan Alicante: Elena Díaz Fernández.
-
16
Hospital General de Requena de Valencia: Laura Ejarque Martinez.
-
17
Hospital de Lliria de Valencia: Ana Peiró Gómez.
-
18
Hospital de la Santa Creu i Sant Pau (Barcelona): Aitor Alquezar, Josep Guardiola.
-
19
Hospital Clinic (Barcelona): Carlos Cardozo.
-
20
Hospital Universitari de Bellvitge de Hospitalet de Llobregat (Barcelona): Ferran Llopis-Roca.
-
21
Hospital Universitari Germans Trias i Pujol de Badalona (Barcelona): Josep Maria Mòdol Deltell.
-
22
Hospital de Terrassa (Barcelona): Josep Tost.
-
23
Hospital del Mar (Barcelona): Alfons Aguirre Tejedo.
-
24
Hospital Universitari Joan XXIII (Tarragona): Anna Palau.
-
25
Hospital Universitari de Girona Dr. Josep Trueta (Girona): Maria Adroher Muñoz.
-
26
Hospital Universitari de Vic (Barcelona): Lluís LLauger García.
-
27
Hospital de Sant Pau i Santa Tecla (Tarragona): Enrique Martín Mojarro.
-
28
Clinica Sagrada Familia (Barcelona): Arturo Huerta.
-
29
Hospital Clínico San Carlos (Madrid): Francisco Javier Martin Sánchez, Marcos Fragiel.
-
30
Hospital Universitario La Paz (Madrid): Alejandro Martín Quirós.
-
31
Hospital Universitario de la Princesa (Madrid): Carmen del Arco Galán.
-
32
Hospital Universitario Severo Ochoa de Leganés (Madrid): Alfonso Martín, Esther Álvarez.
-
33
Hospital Universitario Rey Juan Carlos (Madrid): Belen Rodríguez Miranda.
-
34
Hospital Universitario del Henares (Madrid): Martín Ruiz Grinspan.
-
35
Hospital Universitario de Fuenlabrada (Madrid): María Jesús Domínguez.
-
36
Hospital Universitario Infanta Cristina de Parla (Madrid): Francisco Javier Teigell.
-
37
Hospital Comarcal El Escorial (Madrid): Sara Gayoso Martín.
-
38
Clínica Universidad Navarra de Madrid: Nieves López-Laguna.
-
39
Hospital Universitario de Salamanca: Angel García.
-
40
Complejo Asistencial Universitario de León: Marta Iglesias Vela.
-
41
Hospital Universitario de Burgos: María Pilar López Díaz.
-
42
Hospital Universitario Rio Hortega (Valladolid): Virginia Carbajosa.
-
43
Complejo Asistencial de Soria: Fahd Beddar Chaib.
-
44
Hospital Universitario Regional de Málaga: Manuel Salido.
-
45
Hospital Universitario Juan Ramón Jiménez: María José Marchena González.
-
46
Hospital Costa del Sol de Marbella: Carmen Agüera Urbano.
-
47
Hospital Valle de los Pedroches de Pozoblanco (Córdoba): Jorge Pedraza García.
-
48
Hospital Virgen del Rocío de Sevilla: Amparo Fernández de Simón Almela.
-
49
Complejo Hospitalario Universitario de A Coruña: Ricardo Calvo López.
-
50
Hospital Universitario Lucus Augusti Lugo: Juan José López Díaz.
-
51
Complejo Hospitalario Universitario de Vigo. Hospital Álvaro Cunqueiro: María Teresa Maza Vera.
-
52
Hospital Universitario General de Albacete: Francisco Javier Lucas-Imbernón.
-
53
Hospital Virgen de la Luz (Cuenca): Félix González Martínez.
-
54
Hospital Nuestra Señora del Prado de Talavera de la Reina (Toledo): Ricardo Juárez.
-
55
Hospital Universitario de Canarias (Tenerife): Lissete Traveria.
-
56
Hospital Universitario de Gran Canaria Dr. Negrín: José Pavón Monzo.
-
57
Hospital Universitario Central Asturias: Pablo Herrero Puente.
-
58
Hospital Universitario de Cabueñes (Gijón): Ana Patricia Niembro Valdés.
-
59
Hospital Clínico Universitario Virgen de la Arrixaca: Eva Quero Motto.
-
60
Hospital General Universitario Reina Sofía de Murcia: Pascual Piñera, Jose Andres Sanchez Nicolas.
-
61
Hospital San Pedro de Logroño: Noemí Ruiz de Lobera.
-
62
Hospital Clínico Universitario Lozano Blesa: Jose María Ferreras Amez.
The PEPCOV network is formed by the following researchers and centers (all from France):
-
1
CHU Nice: Celine Occelli.
-
2
CHU Nancy: Tahar Chouihed, Adrien Bassand.
-
3
CHU Hôpital Européen Georges-Pompidou, Paris: Richard Chocron
-
4
CHU Louis Mourier, Paris: Nicolas Javaud.
-
5
CHU Tours: Saïd Laribi, Laurent Brunereau.
-
6
CHU Nantes: Emannuel Montassier.
-
7
CHU Cochin: Jennifer Truchot.
-
8
CHU Nîmes: Pierre-Géraud Claret.
-
9
CHU Tenon: Hélène Goulet.
-
10
CHU Lariboisière, Paris: Xavier Eyer.
-
11
CHU Saint Louis, Paris: Olivier Peyrony.
-
12
CHU Pitié-Salpêtrière, Paris: Marie Drogrey, Samia Boussouar, Victoria Donciu.
-
13
CHU Strasbourg: Pascal Bilbaut.
-
14
CHU Avicenne, Bobigny: Frederic Adned.
-
15
SHU Sant Antoine, Paris: Pierre-Alexis Raynal.
-
16
CHU Rouen: Mehdi Taalba.
References
- 1.Guan W-J, Ni Z-Y, Hu Y, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area | Critical Care Medicine | JAMA | JAMA Network. Accessed 12 January 2021. https://jamanetwork.com/journals/jama/fullarticle/2765184?resultClick=1. [DOI] [PMC free article] [PubMed]
- 3.Poissy J, Goutay J, Caplan M, et al. Pulmonary embolism in patients With COVID-19. Circulation. 2020;142:184–186. doi: 10.1161/CIRCULATIONAHA.120.047430. [DOI] [PubMed] [Google Scholar]
- 4.García-Ortega A, Oscullo G, Calvillo P, López-Reyes R, Méndez R, Gómez-Olivas JD, et al. Incidence, risk factors, and thrombotic load of pulmonary embolism in patients hospitalized for COVID-19 infection. J Infect. 2021 doi: 10.1016/j.jinf.2021.01.003. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fauvel C, Weizman O, Trimaille A, Mika D, Pommier T, Pace N, et al. Pulmonary embolism in COVID-19 patients: a French multicentre cohort study. Eur Heart J. 2020;41:3058–3068. doi: 10.1093/eurheartj/ehaa500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jiménez Hernández S, Lozano Polo L, Suñen Cuquerella G, Peña Pardo B, Espinosa B, Cardozo C, et al. Clinical findings, risk factors, and final outcome in patients diagnosed with pulmonary thromboembolism and COVID-19 in hospital emergency departments. Emergencias. 2020;32:253–257. [PubMed] [Google Scholar]
- 7.Miró Ò, Llorens P, Aguirre A, Lozano L, Beaune S, Roussel M, et al. Association between Covid-19 and pulmonary embolism (AC-19-PE study) Thromb Res. 2020;196:322–324. doi: 10.1016/j.thromres.2020.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tan BK, Mainbourg S, Friggeri A, Bertoletti L, Douplat M, Dargaud Y, et al. Arterial and venous thromboembolism in COVID-19: a study-level meta-analysis. Thorax. 2021;76:970–979. doi: 10.1136/thoraxjnl-2020-215383. [DOI] [PubMed] [Google Scholar]
- 9.Espallargas I, Rodríguez Sevilla JJ, Rodríguez Chiaradía DA, Salar A, Casamayor G, Villar-Garcia J, et al. CT imaging of pulmonary embolism in patients with COVID-19 pneumonia: a retrospective analysis. Eur Radiol. 2020 doi: 10.1007/s00330-020-07300-y. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Davide D, Giandola T, Maino C, Pecorelli A, Capodaglio C, Ragusi M, et al. Acute pulmonary embolism in hospitalized patients with SARS-CoV-2-related pneumonia: multicentric experience from Italian endemic area. Radiol Med. 2021 doi: 10.1007/s11547-020-01328-2. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wiener RS, Schwartz LM, Woloshin S. When a test is too good: how CT pulmonary angiograms find pulmonary emboli that do not need to be found. BMJ. 2013;347(2) doi: 10.1136/bmj.f3368. jul02f3368-f3368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Miró Ò, Llorens P, Jiménez S, et al. Frequency, risk factors, clinical characteristics, and outcomes of spontaneous pneumothorax in patients with coronavirus disease 2019: A Case-Control, Emergency Medicine-Based Multicenter Study. Chest. 2020 doi: 10.1016/j.chest.2020.11.013. Published online November 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Miró Ò, González Del Castillo J. Collaboration among Spanish emergency departments to promote research: on the creation of the SIESTA (Spanish Investigators in Emergency Situations TeAm) network and the coordination of the UMC-19 (Unusual Manifestations of COVID-19) macroproject. Emergencias. 2020;32:269–277. [PubMed] [Google Scholar]
- 14.Miró Ò, Llorens P, Jiménez S, Piñera P, Burillo-Putze G, Martín A, Martín-Sánchez FJ, González Del Castillo J. Spanish Investigators on Emergency Situations TeAm (SIESTA) network. Frequency of five cardiovascular/hemostatic entities as primary manifestations of SARS-CoV-2 infection: results of the UMC-19-S2. Int J Cardiol. 2021;330:268–272. doi: 10.1016/j.ijcard.2021.01.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Freund Y, Drogrey M, Cachanado M, Bloom B. Prevalence of pulmonary embolism in ED patients with Suspected COVID-19: the truth remains unknown. Acad Emerg Med. 2020;27:1218. doi: 10.1111/acem.14138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Freund Y, Drogrey M, Miró Ò, et al. Association between pulmonary embolism and COVID-19 in emergency department patients undergoing computed tomography pulmonary angiogram: the PEPCOV International Retrospective Study. Acad Emerg Med. n/a(n/a). doi:10.1111/acem.14096. [DOI] [PubMed]
- 17.Alquézar-Arbé A, Piñera P, Jacob J, et al. Impact of the COVID-19 pandemic on hospital emergency departments: results of a survey of departments in 2020 - the Spanish ENCOVUR study. Emergencias. 2020;32(5):320–331. [PubMed] [Google Scholar]
- 18.López-Barbeito B, García-Martínez A, Coll-Vinent B, Placer A, Font C, Vargas CR, et al. Factors associated with revisits by patients with SARS-CoV-2 infection discharged from a hospital emergency department. Emergencias. 2020;32:386–394. [PubMed] [Google Scholar]
- 19.Jiménez D. Simplification of the Pulmonary Embolism Severity Index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med. 2010;170:1383. doi: 10.1001/archinternmed.2010.199. [DOI] [PubMed] [Google Scholar]
- 20.Piazza G, Morrow DA. Diagnosis, management, and pathophysiology of arterial and venous thrombosis in COVID-19. JAMA. 2020;324(24):2548–2549. doi: 10.1001/jama.2020.23422. [DOI] [PubMed] [Google Scholar]
- 21.Goswami J, MacArthur TA, Sridharan M, et al. A review of pathophysiology, clinical features, and management options of COVID-19 associated coagulopathy. Shock. 2020 doi: 10.1097/SHK.0000000000001680. Publish Ahead of Print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Edler C, Schröder AS, Aepfelbacher M, et al. Correction to: Dying with SARS-CoV-2 infection-an autopsy study of the first consecutive 80 cases in Hamburg, Germany. Int J Legal Med. 2020;134(5):1977. doi: 10.1007/s00414-020-02336-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rosenthal N, Cao Z, Gundrum J, Sianis J, Safo S. Risk factors associated with in-hospital mortality in a us national sample of patients with COVID-19. JAMA Netw Open. 2020;3(12) doi: 10.1001/jamanetworkopen.2020.29058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rieg S, von Cube M, Kalbhenn J, et al. COVID-19 in-hospital mortality and mode of death in a dynamic and non-restricted tertiary care model in Germany. PLOS ONE. 2020;15(11) doi: 10.1371/journal.pone.0242127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Goldhaber SZ. Assessing the prognosis of acute pulmonary embolism. Chest. 2008;133(2):334–336. doi: 10.1378/chest.07-2464. [DOI] [PubMed] [Google Scholar]
- 26.Sanchez O, Trinquart L, Planquette B, et al. Echocardiography and pulmonary embolism severity index have independent prognostic roles in pulmonary embolism. Eur Respir J. 2013;42(3):681–688. doi: 10.1183/09031936.00097512. [DOI] [PubMed] [Google Scholar]
- 27.Carrier M, Klok FA. Symptomatic subsegmental pulmonary embolism: to treat or not to treat? Hematol Am Soc Hematol Educ Program. 2017;2017(1):237–241. doi: 10.1182/asheducation-2017.1.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Konstantinides SV, Meyer G, Becattini C, Bueno H, Geersing GJ, Harjola VP, et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS): The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC) Eur Respir J. 2019;54 doi: 10.1183/13993003.01647-2019. Oct 9. [DOI] [PubMed] [Google Scholar]
- 29.Freund Y. The challenge of emergency medicine facing the COVID-19 outbreak. Eur J Emerg Med. 2020;27:155. doi: 10.1097/MEJ.0000000000000699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Miró Ò. COVID-19: one threat, one world, one response (magical thinking) Eur J Emerg Med. 2020;27:165–166. doi: 10.1097/MEJ.0000000000000707. [DOI] [PMC free article] [PubMed] [Google Scholar]