Abstract
On the basis of biochemical, phenotypic, and 16S rRNA analyses, Helicobacter cinaedi was isolated from the colon, liver, and mesenteric lymph nodes of a 2-year-old rhesus monkey with chronic diarrhea. Histologically, the liver had mild to moderate biliary hyperplasia and hypertrophy with periportal inflammation and fibrosis. Colonic and cecal lesions consisted of diffuse chronic inflammation and glandular hyperplasia extending the length of the crypts. This is the first observation of H. cinaedi associated with active hepatitis and colitis in a nonhuman primate.
A campylobacter-like organism classified as Campylobacter cinaedi (CLO-1A) was first isolated from the lower bowels of homosexuals with proctitis and colitis (25). Along with Helicobacter fennelliae, it was reclassified as a helicobacter due to its sheathed flagella and 16S rRNA analysis and DNA-DNA hybridization results (12). It has also been isolated from the blood of homosexual patients with human immunodeficiency virus as well as of children and adult females (3, 14, 15, 16, 19, 20, 23, 24, 26). A Helicobacter cinaedi-associated syndrome, consisting of bacteremia and fever accompanied by leukocytosis and thrombocytopenia, is also present. Recurrent cellulitis and/or arthritis has been noted in a high percentage of infected immunocompromised patients (1, 13). H. cinaedi is primarily recovered from immunocompromised individuals but also has been isolated from chronic alcoholics, immunocompetent males and females, and children (16, 26).
Since H. cinaedi has been isolated from the normal intestinal flora of hamsters, it has been suggested that pet hamsters serve as a reservoir for transmission to humans (11, 16). Dogs have also been purported to be reservoir hosts in cases where H. cinaedi (based on fatty acid analysis and DNA-DNA hybridization) was isolated from feces (18). Although H. cinaedi has been shown experimentally to produce diarrhea and bacteremia in infant pigtail macaques, H. cinaedi has not been isolated from either diarrheic or normal Old World primates. The purpose of this study was to characterize by biochemistry and 16S rRNA analysis H. cinaedi from inflamed colon, mesenteric lymph nodes, and diseased liver of a juvenile macaque with chronic diarrhea.
A 2-year-old, domestically raised female rhesus monkey of Indian origin with chronic diarrhea and weight loss was euthanatized and submitted for necropsy. The entire length of the colon was noticeably distended with liquid contents. The mucosa was reddened, with punctate multifocal erosions being noted. The mesenteric lymph nodes draining the lower bowel were enlarged to two to three times the normal size. Full-thickness colonic biopsy samples were collected at necropsy from anterior, transverse, and descending portions of the large intestine; the cecum; the liver; and the mesenteric lymph nodes. Representative samples were fixed in 10% buffered formalin and processed for routine histopathologic analysis. The remainder of the samples were placed in 20% glycerol with brucella broth and frozen at −70°C before microaerobic culture. After transport to the Massachusetts Institute of Technology in dry ice, the samples were homogenized with phosphate-buffered saline. A portion of the tissue homogenate was passed through a 0.45-μm-pore-size filter and plated onto CVA media (Remel Labs, Lenexa, Kans.) containing cefoperazone, vancomycin, and amphotericin B and Helicobacter-selective media containing nalidixic acid, polymyxin B, amphotericin B, bacitracin, and vancomycin. The remaining unfiltered homogenate was streaked on CVA or Helicobacter-selective media. The cultures were incubated for 14 days in vented jars containing N2, H2, and CO2 (85:10:5). To analyze the bacteria biochemically and obtain genomic DNA for 16S rRNA sequencing, cultures of bacteria compatible with Helicobacter spp. were subsequently passaged on sheep blood agar plates. Biochemical analyses following a previously described protocol were performed on isolated bacteria (5).
The sequence of the 16S rRNA gene of the bacterial isolate (MIT 00-6197) cultured from the liver was determined. The 16S rRNA cistrons were amplified with eight sequencing primers used for Helicobacter sp. 16S rRNA and for intervening sequences present at Escherichia coli position 210 using previously described methods (10). PCR was performed in thin-walled tubes with a Perkin-Elmer 9700 thermocycler. One microliter of the DNA template was added to a reaction mixture (50-μl final volume) containing 20 pmol of each primer, 40 nmol of deoxynucleoside triphosphates, and 1 U of Taq 2000 polymerase (Stratagene, La Jolla, Calif.) in buffer containing Taqstart antibody (Sigma Chemical Co.). In a hot-start protocol, samples were preheated at 95°C for 8 min followed by amplification using these conditions: denaturation at 95°C for 45 s, annealing at 60°C for 45 s, and elongation for 1.5 min with an additional 5 s for each cycle. A total of 30 cycles were performed and then followed by a final elongation step at 72°C for 10 min. The results of PCR amplification were examined by electrophoresis in a 1% agarose gel. DNA was stained with ethidium bromide and visualized under short-wavelength UV light. Purified DNA from PCR was sequenced using an ABI Prism cycle-sequencing kit (BigDye Terminator Cycle Sequencing kit with AmpliTaq DNA polymerase FS; Perkin-Elmer). The primers used for sequencing have been previously described. Quarter dye chemistry was used with 80 μM primers and 1.5 μl of PCR product utilized in a final volume of 20 μl. Cycle sequencing was performed using an ABI 9700 (Perkin-Elmer) with 25 cycles of denaturation at 96°C for 10 s and annealing and extension at 60°C for 4 min. Sequencing reactions were run on an ABI 377 DNA sequencer. Sequence data were entered into RNA, a program set for data entry, editing, sequence alignment, secondary-structure comparison, similarity matrix generation, and dendrogram construction for 16S rRNA in Microsoft QuickBasic for use with personal computers, and sequences were aligned as previously described (17). Our database contains over 1,000 sequences obtained in our laboratory and over 500 obtained from GenBank. Dendrograms were constructed by the neighbor-joining method (21).
Bacteria were isolated in microaerobic cultures from the ascending colon, liver, and mesenteric lymph nodes. The isolate grew at 37°C in the presence of 1% glycine but not at 42°C; it was oxidase and catalase positive, reduced nitrate to nitrite, but was negative for urease, alkaline phosphatase hydrolysis, and γ-glutamyl transpeptidase. It was sensitive to nalidixic acid (30-μg disk) but resistant to cephalothin (30 μg). The 16S rRNA sequence of the monkey isolate (MIT 00-6197) was identical to that of the type strain of H. cinaedi, ATCC 35638 (M88150).
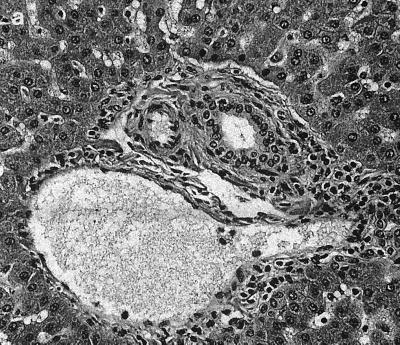
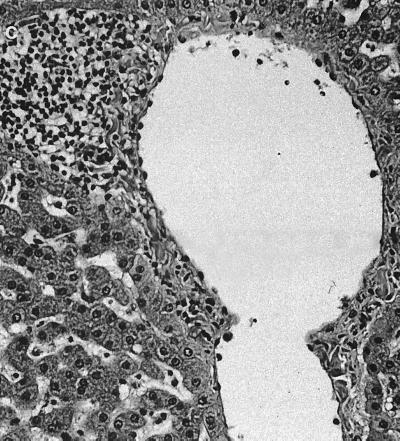
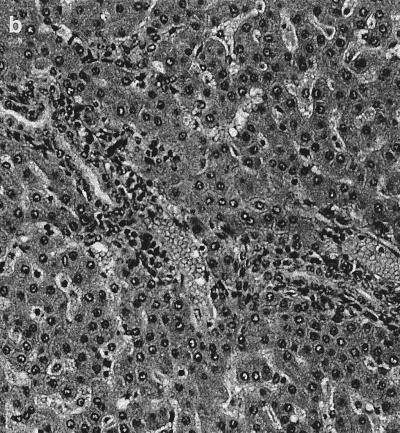
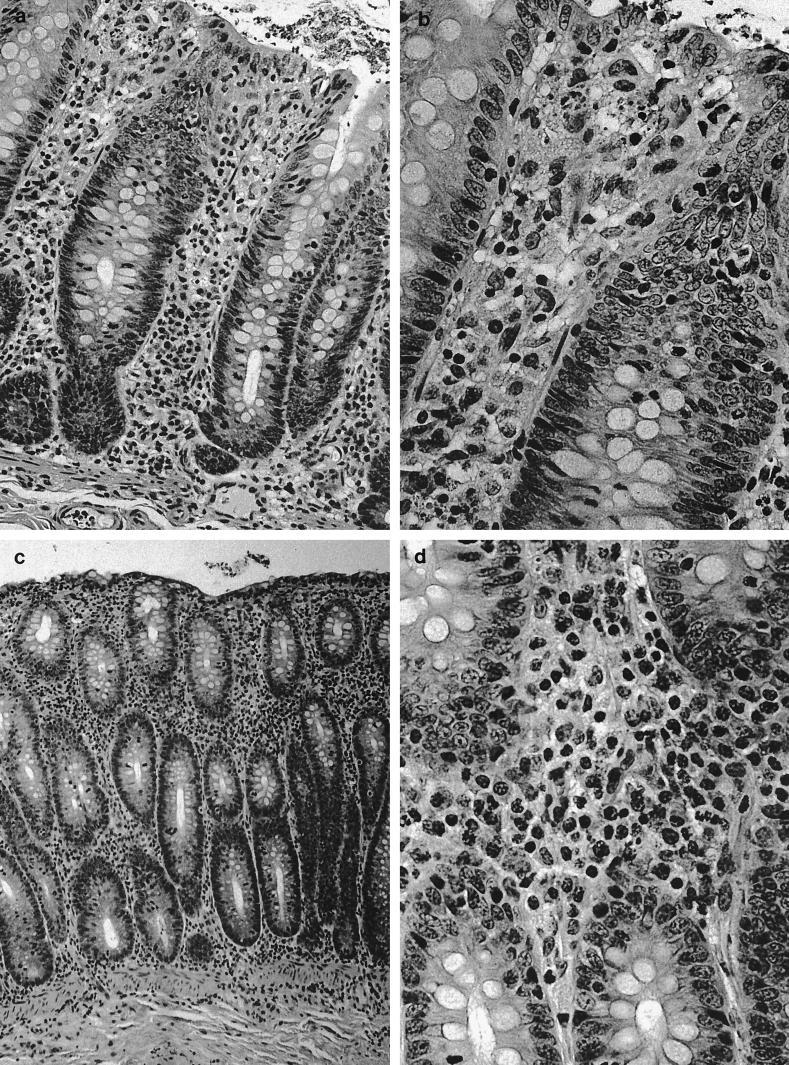
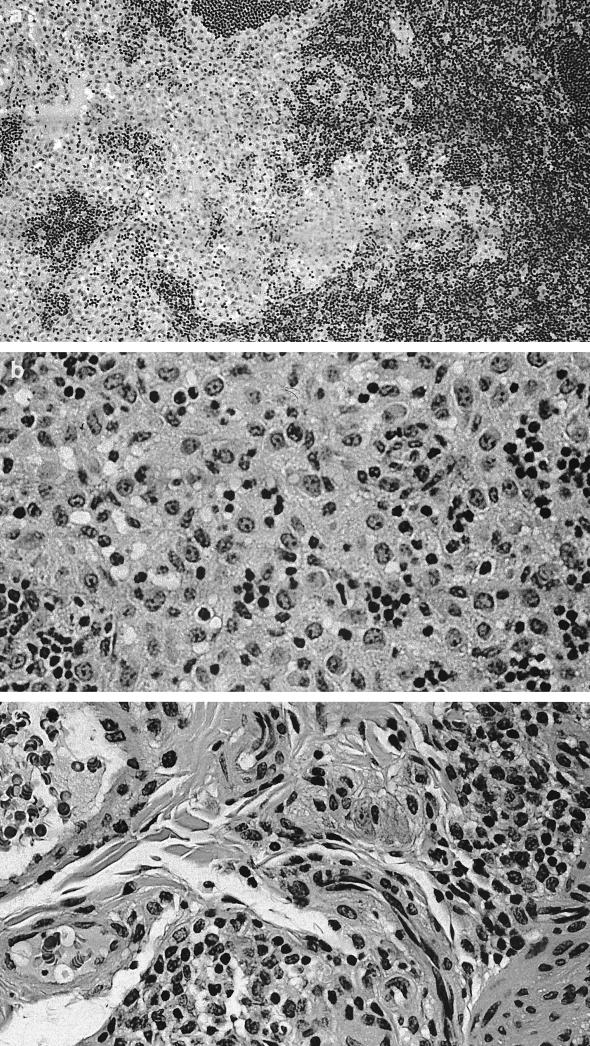
Significant histological changes were restricted to the lower bowel, draining lymph nodes, and liver. Portal regions in the liver were frequently characterized by low to rarely moderate numbers of lymphocytes and plasma cells, with fewer granulocytes and multifocal, mild peribiliary fibroblast proliferation with collagen deposition (Fig. 1a). There was mild to moderate biliary epithelial hypertrophy, which formed prominent elongated bile duct profiles extending from the portal triad regions (Fig. 1b). There also was occasional mild to moderate lymphoplasmacytic infiltration adjacent to centrolobular veins and randomly within the hepatic lobules (Fig. 1c). The lamina propria of the cecum contained large numbers of mononuclear cells that varied regionally but were composed predominantly of plasma cells and macrophages, with fewer lymphocytes and polymorphonuclear cells. Glands were often lined by a highly cellular and very basophilic population of epithelial cells that appeared to be hyperplastic (Fig. 2a). In some areas, small amounts of cellular debris were present within the superficial lamina propria (Fig. 2b). There were moderate to large numbers of mononuclear cells dispersed throughout the lamina propria of the colon. These cells, in many foci, were predominantly plasma cells, with fewer macrophages, lymphocytes, and polymorphonuclear cells. In some areas, larger cells with abundant pale cytoplasm consistent with macrophages predominated. The white blood cells occasionally extended into the submucosa, where they were often perivascular. The epithelium lining the mid to deep glands was highly cellular, with prominent basophilic nuclei and abundant basophilic to pale eosinophilic cytoplasm, which had variable amounts of mucoid differentiation. Small amounts of cellular debris were present within the superficial lamina propria, frequently at sites with erosions of the overlying epithelium. In some areas, the epithelium did not have any mucoid differentiation and the glands were elongated and had apparent hyperplasia of the epithelium. The mesenteric lymph nodes were markedly hypercellular (Fig. 3a). The cortex contained large numbers of small lymphocytes and occasional cortical follicles. The medulla was filled by large macrophages (Fig. 3b). Large numbers of mixed inflammatory cells were also found within the mesentery adjacent to the cecum (Fig. 3c).
FIG. 1.
Liver. (a) Portal inflammation with mild peribiliary collagen deposition. (b) Elongated bile duct profiles. (c) Hematoxylin and eosin stain showing centrolobular inflammation. Magnification, ×156 for all panels.
FIG. 2.
(a and b) Cecal inflammation. Magnifications, ×162 and ×324, respectively. (c and d) Hematoxylin and eosin stain showing colonic inflammation. Magnifications, ×81 and ×324, respectively.
FIG. 3.
Mesenteric lymph node. (a) Marked hypercellularity of cortex and medulla. Magnification, ×80. (b) Medullary histiocytic infiltrate. Magnification, ×160. (c) Hematoxylin and eosin stain showing inflamed cecal mesentery. Magnification, ×320.
Although H. cinaedi has been isolated from multiple organs and body fluids from both immunocompromised and immunocompetent patients, it has not been previously isolated from inflamed tissues of nonhuman primates (8). Interestingly, however, both H. cinaedi and H. fennelliae can cause diarrhea and bacteremia when inoculated into pigtail macaques (4). Recently, a novel Helicobacter sp. has been isolated from cotton-top tamarins with colitis (22). An organism with this morphology has also been noted by electron microscopy in inflamed colonic tissue of cotton top tamarins (2). Though the causative role of a novel Helicobacter sp. isolated from cotton top tamarins with ulcerative colitis has not been established, the systemic immune response to this helicobacter in infected monkeys is robust and appears to be specific (22, 29).
The liver lesions in the macaque described in this report were reminiscent of early liver lesions caused by Helicobacter hepaticus in A/JCr mice (7, 9, 27). In infected mice, the organism causes a multifocal hepatic lesion, with cholangitis and vasculitis, which progresses in severity to include bile duct hyperplasia, hepatomegaly, oval cell hyperplasia, hepatocellular proliferation, and, in aged A/JCr mice, hepatoma or hepatocellular carcinoma (7, 9, 28). The H. cinaedi-associated liver lesions noted in the young primate consisted of multifocal hepatitis with attendant oval cell hyperplasia.
The mechanism whereby certain species of helicobacters, whose normal ecological niche is the lower intestine, colonize the liver is unknown. The bacteria may gain access to the liver by initial M-cell uptake, with colonization of the liver via the portal circulation and final sequestration of the bacteria in the biliary tract. Alternatively, there may be direct translocation through enterocytes or migration of the helicobacters from the lumen of the gut into the bile duct. The isolation of H. cinaedi from the mesenteric lymph nodes is supportive of either translocation or M-cell uptake for colonization of the liver.
Although H. cinaedi has been isolated from diarrheic and bacteremic children and adults with gastroenteritis, it is unknown whether it can cause hepatitis in humans. We predict that with the use of appropriate diagnostic media and microaerobic culture conditions. H. cinaedi along with other Helicobacter spp. will be increasingly isolated from cases of hepatobiliary disease in humans and various species of animals (6). In addition, it will be interesting to study whether novel Helicobacter spp. recently isolated from inflamed colons of rhesus monkeys maintained in the same colony can also induce hepatitis (8). Given the zoonotic implication of H. cinaedi transmission from hamsters to humans, animal technicians should wear protective clothing and appropriate face masks to minimize exposure to fecal-oral spread of H. cinaedi and other potentially zoonotic enteric helicobacters (8, 22).
Acknowledgments
This work was supported by NIH grants R01CA67529 and R01DK52413 and a grant from Merck Laboratories.
REFERENCES
- 1.Burman W J, Cohn D L, Reves R R, Wilson M L. Multifocal cellulitis and monoarticular arthritis as manifestations of H. cinaedi bacteremia. Clin Infect Dis. 1995;20:564–570. doi: 10.1093/clinids/20.3.564. [DOI] [PubMed] [Google Scholar]
- 2.Chalifoux L V, Brieland J K, King N W. Evolution and natural history of colonic disease in cotton-top tamarins (Saguinus oedipus) Dig Dis Sci. 1985;30:54S–58S. doi: 10.1007/BF01296976. [DOI] [PubMed] [Google Scholar]
- 3.Cimolai N, Gill M J, Jones A, Flores B, Stamm W E, Laurie W, Madden B, Shahrabadi M S. “Campylobacter cinaedi” bacteremia: case report and laboratory findings. J Clin Microbiol. 1987;25:942–943. doi: 10.1128/jcm.25.5.942-943.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Flores B M, Fennell C L, Kuller L, Bronsdon M A, Morton W R, Stamm W E. Experimental infection of pig-tailed macaques (Macaca nemestrina) with Campylobacter cinaedi and Campylobacter fennelliae. Infect Immun. 1990;58:3947–3953. doi: 10.1128/iai.58.12.3947-3953.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fox J G, Chien C C, Dewhirst F E, Paster B J, Shen Z, Melito P L, Woodward D L, Rodgers F G. Helicobacter canadensis sp. nov. isolated from humans with diarrhea as an example of an emerging pathogen. J Clin Microbiol. 2000;38:2546–2549. doi: 10.1128/jcm.38.7.2546-2549.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fox J G, Dewhirst F E, Shen Z, Fen Y, Taylor N S, Paster B, Erickson R L, Lau C N, Correa P, Araya J C, Roa I. Hepatic Helicobacter species identified in bile and gallbladder tissue from Chileans with chronic cholecystitis. Gastroenterology. 1998;114:755–763. doi: 10.1016/s0016-5085(98)70589-x. [DOI] [PubMed] [Google Scholar]
- 7.Fox J G, Dewhirst F E, Tully J G, Paster B J, Yan L, Taylor N S, Collins M J, Jr, Gorelick P L, Ward J M. Helicobacter hepaticus sp. nov., a microaerophilic bacterium isolated from livers and intestinal mucosal scrapings from mice. J Clin Microbiol. 1994;32:1238–1245. doi: 10.1128/jcm.32.5.1238-1245.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fox, J. G., L. Handt, S. Xu, Z. Shen, F. E. Dewhirst, C. Dangler, K. Lodge, S. Motzel, and H. Klein. Novel Helicobacter spp isolated from colonic tissue of rhesus monkeys with chronic idiopathic colitis. J. Med. Microbiol., in press. [DOI] [PubMed]
- 9.Fox J G, Li X, Yan L, Cahill R J, Hurley R, Lewis R, Murphy J C. Chronic proliferative hepatitis in A/JCr mice associated with persistent Helicobacter hepaticus infection: a model of helicobacter-induced carcinogenesis. Infect Immun. 1996;64:1548–1558. doi: 10.1128/iai.64.5.1548-1558.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fox J G, Yan L L, Dewhirst F E, Paster B J, Shames B, Murphy J C, Hayward A, Belcher J C, Mendes E N. Helicobacter bilis sp. nov., a novel Helicobacter species isolated from bile, livers, and intestines of aged, inbred mice. J Clin Microbiol. 1995;33:445–454. doi: 10.1128/jcm.33.2.445-454.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gebhart C J, Fennell C L, Murtaugh M P, Stamm W E. Campylobacter cinaedi is normal intestinal flora in hamsters. J Clin Microbiol. 1989;27:1692–1694. doi: 10.1128/jcm.27.7.1692-1694.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kiehlbauch J A, Brenner D J, Cameron D N, Steigerwalt A G, Makowski J M, Baker C N, Patton C M, Wachsmuth I K. Genotypic and phenotypic characterization of Helicobacter cinaedi and Helicobacter fennelliae strains isolated from humans and animals. J Clin Microbiol. 1995;33:2940–2947. doi: 10.1128/jcm.33.11.2940-2947.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kiehlbauch J A, Tauxe R V, Baker C N, Wachsmuth I K. Helicobacter cinaedi-associated bacteremia and cellulitis in immunocompromised patients. Ann Intern Med. 1994;121:90–93. doi: 10.7326/0003-4819-121-2-199407150-00002. [DOI] [PubMed] [Google Scholar]
- 14.Mammen M P, Jr, Aronson N E, Edenfield W J, Endy T P. Recurrent Helicobacter cinaedi bacteremia in a patient infected with human immunodeficiency virus: case report. Clin Infect Dis. 1995;21:1055. doi: 10.1093/clinids/21.4.1055. [DOI] [PubMed] [Google Scholar]
- 15.Ng V L, Hadley W K, Fennell C L, Flores B M, Stamm W E. Successive bacteremias with “Campylobacter cinaedi” and “Campylobacter fennelliae” in a bisexual male. J Clin Microbiol. 1987;25:2008–2009. doi: 10.1128/jcm.25.10.2008-2009.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Orlicek S L, Welch D F, Kuhls T L. Septicemia and meningitis caused by Helicobacter cinaedi in a neonate. J Clin Microbiol. 1993;31:569–571. doi: 10.1128/jcm.31.3.569-571.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Paster B J, Dewhirst F E. Phylogeny of campylobacter, wolinellas, Bacteroides gracilis, and Bacteroides ureolyticus by 16S ribosomal ribonucleic acid sequencing. Int J Syst Bacteriol. 1988;38:56–62. [Google Scholar]
- 18.Patton C M, Shaffer N, Edmonds P, Barrett T J, Lambert M A, Baker C, Perlman D M, Brenner D J. Human disease associated with “Campylobacter upsatensis” (catalase-negative or weakly positive Campylobacter species) in the United States. J Clin Microbiol. 1989;27:66–73. doi: 10.1128/jcm.27.1.66-73.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Quinn T C, Goodell S E, Fennell C L, Wang S P, Schuffler M D, Holmes K K, Stamm W E. Infections with Campylobacter jejuni and Campylobacter-like organisms in homosexual men. Ann Intern Med. 1984;101:187–192. doi: 10.7326/0003-4819-101-2-187. [DOI] [PubMed] [Google Scholar]
- 20.Quinn T C, Stamm W E, Goodell S E, Mkrtichian E, Benedetti J, Corey L, Schuffler M D, Holmes K K. The polymicrobial origin of intestinal infections in homosexual men. N Engl J Med. 1983;309:576–582. doi: 10.1056/NEJM198309083091002. [DOI] [PubMed] [Google Scholar]
- 21.Saitou N, Nei M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol. 1987;4:406–425. doi: 10.1093/oxfordjournals.molbev.a040454. [DOI] [PubMed] [Google Scholar]
- 22.Saunders K E, Shen Z, Dewhirst F E, Paster B J, Dangler C A, Fox J G. Novel intestinal Helicobacter species isolated from cotton-top tamarins (Saguinus oedipus) with chronic colitis. J Clin Microbiol. 1999;37:146–151. doi: 10.1128/jcm.37.1.146-151.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sullivan A K, Nelson M R, Walsh J, Gazzard B G. Recurrent Helicobacter cinaedi cellulitis and bacteremia in a patient with HIV infection. Int J STD AIDS. 1997;8:59–60. doi: 10.1258/0956462971918634. [DOI] [PubMed] [Google Scholar]
- 24.Tee W, Street A C, Spelman D, Munckhof W, Mijch A. Helicobacter cinaedi bacteremia: varied clinical manifestations in three homosexual males. Scand J Gastroenterol. 1996;28:199–203. doi: 10.3109/00365549609049078. [DOI] [PubMed] [Google Scholar]
- 25.Totten P A, Fennel C L, Tenover F C. Campylobacter cinaedi (sp. nov.) and Campylobacter fennelliae (sp. nov.): two new Campylobacter species associated with enteric disease in homosexual men. J Infect Dis. 1985;151:131–139. doi: 10.1093/infdis/151.1.131. [DOI] [PubMed] [Google Scholar]
- 26.Vandamme P, Falsen E, Pot B, Kersters K, De Ley J. Identification of Campylobacter cinaedi isolated from blood and feces of children and adult females. J Clin Microbiol. 1990;28:1016–1020. doi: 10.1128/jcm.28.5.1016-1020.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ward J M, Anver M R, Haines D C, Benveniste R E. Chronic active hepatitis in mice caused by Helicobacter hepaticus. Am J Pathol. 1994;145:959–968. [PMC free article] [PubMed] [Google Scholar]
- 28.Ward J M, Fox J G, Anver M R, Haines D C, George C V, Collins M J, Gorelick P L, Nagashima K, Gonda M A, Gilden R V, Tully J G, Russell R J, Benveniste R E, Paster B J, Dewhirst F E, Donovan J C, Anderson L M, Rice J M. Chronic active hepatitis and associated liver tumors in mice caused by a persistent bacterial infection with a novel Helicobacter species. J Natl Cancer Inst. 1994;86:1222–1227. doi: 10.1093/jnci/86.16.1222. [DOI] [PubMed] [Google Scholar]
- 29.Whary M T, Saunders K E, Esteves M I, Wood J, Peck O C, Fox J G. A novel Helicobacter sp. isolated from inflamed colonic tissue in cotton top tamarins is associated with a specific humoral immune response. Gastroenterology. 1999;116:A845. [Google Scholar]