Abstract
Introduction
Inflammatory bowel disease (IBD) is associated with greater risk of thromboembolic events (TEs) due to the link between systemic inflammation and hypercoagulability. This study assessed the rates of TEs among patients with IBD versus patients without immune-mediated disease (IMD) and the cost of TEs among patients with IBD in the United States.
Methods
This study used the IBM MarketScan® Commercial and Medicare Supplemental Databases (2014–2018). To assess the incremental rates of TEs (deep vein thrombosis [DVT], pulmonary embolism [PE], ischemic stroke [IS], myocardial infarction [MI]), patients with IBD were matched to patients without IMD. Unadjusted and adjusted incidence rate ratios (IRRs) of TEs were used to compare cohorts. To assess the cost of TEs, patients with IBD with TEs were matched to patients with IBD without TEs. Costs were assessed 30 days and 1 year post index date.
Results
There were 34,687 matched pairs included in the rates of TE analyses. Compared to patients without IMD, patients with IBD had greater rates of DVT (adjusted IRR [95% confidence interval] 2.44 [2.00, 2.99]; p < 0.01) and PE (1.90 [1.42, 2.54]; p < 0.01). Increased rates were not observed for IS and MI. There were 1885 matched pairs included in the cost of TE analyses. Patients with IBD with TEs incurred greater healthcare costs over 30 days and 1 year versus patients without TEs (adjusted total cost difference: 30 days $20,784; 1 year $44,630; p < 0.01 for both).
Conclusions
Patients with IBD experienced greater rates of DVT and PE compared to patients without IMD; this elevated risk was associated with a substantial economic burden.
Supplementary Information
The online version contains supplementary material available at 10.1007/s12325-021-01973-7.
Keywords: Economic burden, Healthcare costs, Inflammatory bowel disease, Thromboembolic events
Key Summary Points
| Why carry out this study? |
| Inflammatory bowel disease (IBD) has been linked to an increased risk of thromboembolic events (TEs) |
| Given the serious nature of TEs, there is a growing interest in understanding the incremental rates of TEs associated with IBD as well as the economic burden of TEs among patients with IBD; however, there is limited evidence available on these topics |
| What was learned from the study? |
| Compared to patients without immune-mediated diseases, patients with IBD had significantly increased rates of deep vein thrombosis (adjusted incidence rate ratios [IRR]: 2.44; p < 0.01) and pulmonary embolism (adjusted IRR: 1.90; p < 0.01), although increased rates were not observed for ischemic stroke and myocardial infarction |
| Patients with IBD with TEs had significantly higher healthcare costs 30 days and 1 year following their TE event compared to patients with IBD without TEs (adjusted total cost difference: 30 days $20,784; 1 year $44,630; both p < 0.01) |
Introduction
Inflammatory bowel disease (IBD) is a risk factor for vascular complications due to the complex interplay of systemic inflammation, hypercoagulability and predisposing factors (e.g., comorbidities, medications) [1, 2]. Patients with IBD face a 2–3-fold greater risk of thromboembolic events (TEs) relative to patients without IBD and the general population [1, 3, 4]. TEs, which encompass venous events (i.e., deep vein thrombosis [DVT] and pulmonary embolism [PE]) and arterial events (i.e., ischemic stroke [IS] and myocardial infarction [MI]), have been associated with increased morbidity and mortality among patients with IBD [1, 5]. For example, patients with IBD with TEs have been shown to experience more than one episode of a TE in atypical sites (e.g., intracardiac, cerebral, innominate veins) [6], and mortality has been reported to range from 8 to 25% of patients with IBD who experienced TE [6, 7]. The increased morbidity and mortality associated with TE among patients with IBD have been reported to negatively affect patients’ quality of life and result in extensive healthcare resource use [8].
IBD and TEs are each associated with a considerable economic burden [9–12]. An economic report cited direct and indirect costs associated with IBD between $14.6 and $31.6 billion in 2014 [9]. Regarding TEs, conservative cost estimates of the annual incident venous TEs (VTEs) have been reported to range from $7 to $10 billion annually and have been linked to > 500,000 annual hospitalizations in the United States (US) [11, 13]. Despite awareness regarding the high economic burden associated with IBD and TEs individually, the magnitude of the economic burden among patients with IBD who experience TEs is largely unknown. While there have been studies on healthcare resource use and costs of TE among patients with cancer and those undergoing surgery (e.g., hip replacement), less is known about the economic burden of TE among patients with immune-mediated diseases (IMDs), such as IBD [9–12, 14–16]. As IBD is a key risk factor for TEs, there is an impetus to characterize the burden in this patient population.
The clinical management of IBD has dramatically changed following the approval of several advanced therapies for IBD [17, 18]. As a result, there is a need to systematically assess the association between IBD and TE in the current therapeutic landscape. Such an assessment can serve as a benchmark for newer agents for IBD [18]. To obtain a comprehensive understanding of the burden of TEs in patients with IBD, this study characterized the incremental rates of TEs among patients with IBD versus patients without IMDs. The cost of TEs among patients with IBD in the US was also assessed.
Methods
Data Source and Compliance with Ethics Guidelines
To assess the incremental rates and cost of TE, this study used the IBM MarketScan® Commercial and Medicare Supplemental Databases (2014–2018). Informed consent and consent to publish were not applicable to this study. Institutional review board approval was not required for this study. The pre-existing, retrospective data from the IBM MarketScan® Commercial and Medicare Supplemental Databases are represented by IBM MarketScan to be fully de-identified in accordance with the Health Insurance Portability and Accountability Act. The data were provided under license agreement with IBM.
Study Design and Sample Selection
This retrospective cohort study consisted of two parts. The first part assessed the rates of TEs among patients with IBD compared to patients without IMDs and was based on a subgroup analysis of a previously published claims-based study that assessed the rates of TEs among patients with IMDs compared to patients without IMDs [19]. In that study, IMDs encompassed IBD in addition to the most common IMDs including ankylosing spondylitis, atopic dermatitis, multiple sclerosis, plaque psoriasis, psoriatic arthritis, rheumatoid arthritis and systemic lupus erythematosus. Patients with ≥ 2 diagnoses of IMDs on distinct dates were matched on age, sex and index year (i.e., the day after a randomly selected date of an IMD diagnosis for the IMD cohort and a randomly selected medical claim for the non-IMD cohort) to patients with no evidence of IMD. All patients were required to have 1 year of continuous enrollment prior to the index date (baseline period), 30 days of continuous enrollment following the index date and be ≥ 18 years. Within the sample of patients with IMD, those with IBD during the baseline period and their matched non-IMD control patients were included for the present analysis. The study period was defined as the time from the index date until the earliest of patient death, end of continuous eligibility or end of data availability.
The second part of this study assessed the cost of TEs among patients with IBD. Patients whose first diagnosis of an IMD was IBD were separated into two cohorts. Patients in the TE cohort were required to have a TE (see Supplementary Material for diagnosis codes to identify TEs) after the first diagnosis of IBD. Patients in the no TE cohort were required to have no evidence of a TE. Patients with TEs were matched on age, sex and year of first IBD diagnosis to patients without TEs. All patients were required to have 6 months of continuous enrollment prior to the index date, 1 year of continuous enrollment following the index date and be ≥ 18 years. The index date for the TE cohort was defined as the date of first TE after IBD diagnosis. For the no TE cohort, the index date was assigned so that the duration between the first IBD diagnosis to the index date matched the corresponding duration of the TE patient. The study period was defined as the 30-day and 1-year periods following the index date (inclusive). Data processing and statistical analyses were performed using SAS Enterprise Guide version 7.15 (SAS Institute Inc., Cary, NC).
Study Measures and Outcomes
Patient Characteristics
For both parts of this study, patient characteristics assessed during the baseline period included demographics, age at index date, IMD and non-IMD drugs, and select comorbidities (Supplementary Material). The comorbidities selected for this analysis included risk factors for TE and potential confounders for the association between IMD and TE based on the literature and clinical guidance. In the analysis of the rates of TEs, patients with IBD were compared to patients without IMD. In the analysis of the cost of TEs, patients with IBD with TEs were compared to patients with IBD without TEs. Continuous variables were described using means and standard deviations; categorical variables were described using counts and percentages. Statistical comparisons for matched samples were performed using Wilcoxon signed-rank tests for continuous variables and McNemar tests for categorical variables.
Unadjusted and Adjusted Incremental Risks of TEs
Using the sample of IBD and matched non-IMD patients, patients with venous and arterial TEs were identified as those with ≥ 1 diagnosis code from the International Classification of Diseases, Ninth/Tenth Revision, Clinical Modification for DVT, PE, IS or MI (Supplementary Material) during the study period. Incidence rates (IRs) of TEs, overall and separately for DVT, PE, IS and MI, were calculated as the total number of TEs divided by the total patient-years during the study period. Both unadjusted and adjusted incidence rate ratios (IRRs) were used to compare the IR of TEs among patients in the IBD cohort versus patients in the non-IMD cohort. The 95% confidence intervals (CI) and p values were estimated using generalized estimating equations. The adjusted IRRs were adjusted for age at index date, female sex, baseline comorbidities (i.e., cancer, cardiovascular diseases, chronic kidney disease, chronic obstructive pulmonary disease, type 1 and type 2 diabetes, fractures, peripheral vascular disease and pregnancy), baseline non-IMD drugs (i.e., hormone replacement therapies, testosterone replacement therapies, oral contraceptives) and baseline TEs of interest.
Healthcare Costs of TEs
Unadjusted and adjusted costs and costs differences between patients with IBD in the TE and no TE cohorts were assessed during the 30-day and 1-year periods following the index date. Statistical comparisons for unadjusted costs were performed using the Wilcoxon signed-rank test for continuous variables. Adjusted costs were estimated using generalized estimating equations. The adjusted models were adjusted for the following variables: age at index date, female sex, index year, healthcare plan type (capitation), baseline comorbidities (i.e., cancer, cardiovascular diseases, chronic kidney disease, chronic obstructive pulmonary disease, type 1 and type 2 diabetes, fractures, peripheral vascular disease and pregnancy), baseline non-IMD drugs (i.e., hormone replacement therapies, testosterone replacement therapies, oral contraceptives) and baseline IMD drugs (i.e., tumor necrosis factor [TNF] inhibitors, interleukin inhibitors, other biologics, methotrexate, other non-biologics, 5-aminosalicyclic-acid derivative agents, glucocorticoids and nonsteroidal anti-inflammatory drugs [NSAIDs]).
TE-related healthcare costs (i.e., cost of medical visits with a TE diagnosis) were also reported during the 30-day and 1-year periods following the index date.
Results
Risks of TEs in IBD
Patient Characteristics
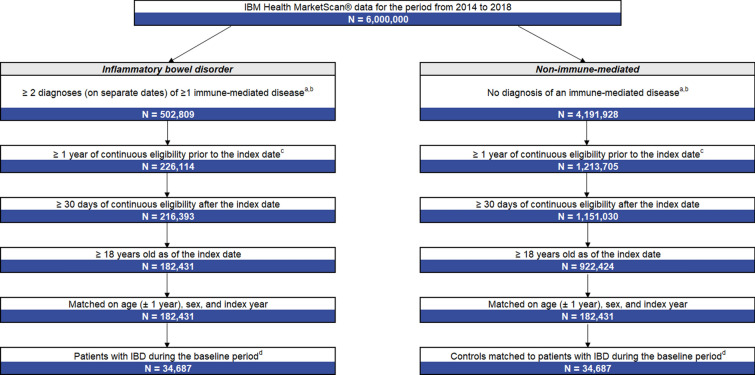
After applying the sample selection criteria and matching 1:1 on age, sex and index year, the IBD and non-IMD cohorts each comprised 34,687 patients (Fig. 1). In both cohorts, the mean age was 49.0 years and 54.4% of patients were female (Table 1). In the IBD cohort, a significantly greater proportion of patients had ≥ 1 baseline TE compared to patients in the non-IMD cohort (4.7% vs. 2.6%; p < 0.01). The mean Charlson Comorbidity Index was significantly higher among patients in the IBD cohort compared with patients in the non-IMD cohort (0.7 vs. 0.4, p < 0.01). The most prevalent comorbidities were cardiovascular diseases and hypertension (cardiovascular diseases [44.6% vs. 42.2%]; hypertension [32.5% vs. 30.5%]; both p < 0.01).
Fig. 1.
Sample selection of patients with IBD and without IMD. IBD inflammatory bowel disease, IMD immune-mediated disease. aIMD included ankylosing spondylitis, atopic dermatitis, inflammatory bowel disease, multiple sclerosis, plaque psoriasis, psoriatic arthritis, rheumatoid arthritis and systemic lupus erythematosus. bQualifying diagnoses were identified on the inpatient and/or outpatient services claims datasets. cFor patients with ≥ 1 IMD, the index date was the day after a randomly selected date of a claim for an IMD. For patients without IMD, the index date was the day after a randomly selected date of a claim. dThe baseline period was the 12-month period prior to the index date
Table 1.
Patient demographics and clinical characteristics measured during the baseline perioda—patients with IBD and matched patients with non-immune-mediated disease
| Inflammatory bowel disease | Non-immune-mediated | p value | |
|---|---|---|---|
| (n = 34,687) | (n = 34,687) | ||
| Age at index date (years) | |||
| Mean ± SD | 49.0 ± 16.1 | 49.0 ± 16.1 | 0.14 |
| Sex (female), n (%) | 18,880 (54.4) | 18,880 (54.4) | – |
| Index year, n (%) | |||
| 2015 | 9422 (27.2) | 9422 (27.2) | – |
| 2016 | 9282 (26.8) | 9282 (26.8) | – |
| 2017 | 7945 (22.9) | 7945 (22.9) | – |
| 2018 | 8038 (23.2) | 8038 (23.2) | – |
| Immune-mediated disease, n (%) | |||
| Inflammatory bowel disease | 34,687 (100) | 0 (0) | – |
| Rheumatoid arthritis | 642 (1.9) | 0 (0) | – |
| Psoriasis | 539 (1.6) | 0 (0) | – |
| Ankylosing spondylitis | 238 (0.7) | 0 (0) | – |
| Atopic dermatitis | 180 (0.5) | 0 (0) | – |
| Systemic lupus erythematosus | 162 (0.5) | 0 (0) | – |
| Psoriatic arthritis | 118 (0.3) | 0 (0) | – |
| Multiple sclerosis | 116 (0.3) | 0 (0) | – |
| History of thromboembolic events, n (%) | 1613 (4.7) | 887 (2.6) | < 0.01 |
| Deep vein thrombosis | 810 (2.3) | 326 (0.9) | < 0.01 |
| Ischemic stroke | 443 (1.3) | 327 (0.9) | < 0.01 |
| Pulmonary embolism | 355 (1.0) | 144 (0.4) | < 0.01 |
| Myocardial infarction | 306 (0.9) | 212 (0.6) | < 0.01 |
| Charlson Comorbidity Index | |||
| Mean ± SD | 0.7 ± 1.4 | 0.4 ± 1.1 | < 0.01 |
| Comorbidities, n (%) | |||
| Cancer | 3412 (9.8) | 2318 (6.7) | < 0.01 |
| Cardiovascular diseases | 15,474 (44.6) | 14,622 (42.2) | < 0.01 |
| Atherosclerosis | 2415 (7.0) | 2005 (5.8) | < 0.01 |
| Atrial fibrillation | 1253 (3.6) | 895 (2.6) | < 0.01 |
| Heart failure | 963 (2.8) | 687 (2.0) | < 0.01 |
| Hyperlipidemia | 10,336 (29.8) | 10,350 (29.8) | 0.90 |
| Hypertension | 11,266 (32.5) | 10,582 (30.5) | < 0.01 |
| Chronic kidney disease | 1313 (3.8) | 843 (2.4) | < 0.01 |
| Chronic obstructive pulmonary disease | 1596 (4.6) | 1043 (3.0) | < 0.01 |
| Diabetes | |||
| Type 1 | 457 (1.3) | 405 (1.2) | 0.08 |
| Type 2 | 3782 (10.9) | 3937 (11.4) | 0.05 |
| Fracture (hip or leg) | 224 (0.6) | 221 (0.6) | 0.89 |
| Peripheral vascular disease | 1797 (5.2) | 1096 (3.2) | < 0.01 |
| Pregnancyb | 855 (4.5) | 1077 (5.7) | < 0.01 |
| Common classes of drugs, n (%) | |||
| Non-immune-mediating drugs | |||
| Anticoagulants | 1377 (4.0) | 903 (2.6) | < 0.01 |
| Hormone replacement therapiesb | 1424 (7.5) | 1199 (6.4) | < 0.01 |
| Testosterone replacement therapiesb | 448 (2.8) | 327 (2.1) | < 0.01 |
| Oral contraceptivesb | 2,570 (13.6) | 2133 (11.3) | < 0.01 |
| Immune-mediating drugsc | |||
| Biologics | 4089 (11.8) | 2 (0) | – |
| TNF inhibitors | 3835 (11.1) | 2 (0) | – |
| Interferon beta-1a | 13 (0) | 0 (0) | – |
| Interleukin inhibitors | 194 (0.6) | 0 (0) | – |
| Other biologics | 152 (0.4) | 0 (0) | – |
| JAK inhibitors | 19 (0.1) | 1 (0) | – |
| Non-biologic immunomodulators | 6695 (19.3) | 180 (0.5) | – |
| Methotrexate | 797 (2.3) | 15 (0) | – |
| S1P receptor modulators | 5 (0) | 0 (0) | – |
| Other non-biologic immunomodulators | 6078 (17.5) | 170 (0.5) | – |
| 5-Aminosalicyclic-acid derivative agents | 15,811 (45.6) | 23 (0.1) | – |
| Glucocorticoids | 13,058 (37.6) | 5730 (16.5) | < 0.01 |
| NSAIDs | 5864 (16.9) | 6556 (18.9) | < 0.01 |
JAK Janus kinase, NSAIDs nonsteroidal anti-inflammatory drugs, S1P sphingosine 1-phosphate, SD standard deviation, TNF tumor necrosis factor
aThe baseline period was defined as 1-year period prior to the index date
bThe proportions of patients with pregnancy, hormone replacement therapies and oral contraceptives were reported out of the total number of females in each group. The proportion of patients with testosterone replacement therapies was reported out of the total number of males in each group
cSome of the biologics and other immune-mediating drugs had minimal utilization in the non-immune-mediated cohort. These drugs are occasionally used off-label for conditions that were not included the present study. In addition, there is always a risk of misclassification of patients in a retrospective claims study given the use of real-world data, but such misclassification would likely be inconsequential
Incidence of TEs in Patients with IBD
During the study period, the total person-years for the IBD and non-IMD cohorts were 47,233 and 44,315, respectively (Table 2). The unadjusted rates of DVT were 0.089 and 0.024 per person-year for the IBD and non-IMD cohorts, respectively, while the unadjusted rates of PE were 0.048 and 0.015 per person-year. Patients with IBD had a 3.75 (95% CI 3.02, 4.64) and 3.15 (95% CI 2.34, 4.24) times greater rate of having DVT and PE, respectively (p < 0.01 for both), compared to patients without IMD. The increased risk of VTE in patients with IBD remained significant after adjustment (adjusted IRR [95% CI] DVT 2.44 [2.00, 2.99], PE 1.90 [1.42, 2.54], both p < 0.01). No significant increase in rates was observed for arterial events (IS: 1.32 [95% CI 1.03, 1.69]; MI: 0.81 [95% CI 0.9, 1.11]). After adjustments, such increases were not observed (adjusted IRRs: IS 1.15 [0.89, 1.50], p = 0.278; MI 0.62 [0.44, 0.88], p = 0.008).
Table 2.
Unadjusted and adjusted incidence rate ratios during the study period
| Unadjusted | Adjusteda | |||||
|---|---|---|---|---|---|---|
| Inflammatory bowel disease | Non-immune-mediated | Rate ratio of IBD vs. non-immune-mediated (95% CI)b | p value | Rate ratio of IBD vs. non-immune-mediated (95% CI)b | p value | |
| (n = 34,687) | (n = 34,687) | |||||
| Total person-years | 47,233 | 44,315 | ||||
| Total number of TEs | 9087 | 4186 | ||||
| Event rate per year | 0.192 | 0.094 | 2.04 (1.78, 2.34) | < 0.01 | 1.49 (1.30, 1.71) | < 0.01 |
| Total number of DVT events | 4188 | 1049 | ||||
| Event rate per year | 0.089 | 0.024 | 3.75 (3.02, 4.64) | < 0.01 | 2.44 (2.00, 2.99) | < 0.01 |
| Total number of PE events | 2273 | 677 | ||||
| Event rate per year | 0.048 | 0.015 | 3.15 (2.34, 4.24) | < 0.01 | 1.90 (1.42, 2.54) | < 0.01 |
| Total number of IS events | 2163 | 1535 | ||||
| Event rate per year | 0.046 | 0.035 | 1.32 (1.03, 1.69) | 0.026 | 1.15 (0.89, 1.50) | 0.278 |
| Total number of MI events | 901 | 1046 | ||||
| Event rate per year | 0.019 | 0.024 | 0.81 (0.59, 1.11) | 0.187 | 0.62 (0.44, 0.88) | 0.008 |
CI confidence interval, DVT deep vein thromboembolism, IBD inflammatory bowel disease, IS ischemic stroke, MI myocardial infarction, PE pulmonary embolism, TE thromboembolic event. The study period was defined as the time from the index date until the earliest of patient death, end of continuous eligibility or end of data availability
aThe adjusted incidence rate ratios controlled for the following: cohort assignment, age at index date, sex (female), baseline comorbidities, baseline non-immune-mediating drugs and baseline TE of interest (yes/no)
bIncidence rate ratios, 95% confidence intervals and p-values were estimated using generalized linear models with a Poisson distribution and a sandwich (robust) variance estimator. An offset was used to account for varying lengths of follow-up time
Healthcare Costs of TEs in IBD
Patient Characteristics
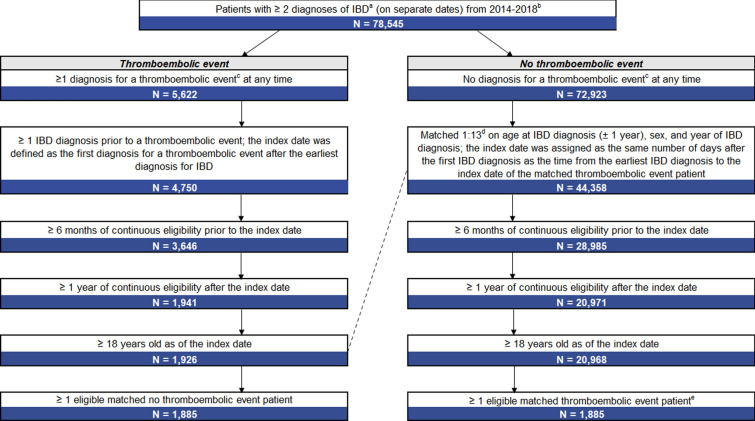
In the analysis on healthcare costs, a total of 1885 matched pairs of patients with IBD were included in the TE cohort and no TE cohort (Fig. 2). In both cohorts, the mean age was 58.9 years and 52.6% of patients were female (Table 3). In the TE cohort, the most common index event was DVT (47.1%), followed by IS (25.8%), MI (17.2%) and PE (15.8%). The mean Charlson Comorbidity Index was significantly higher among patients with TE versus no TE (1.2 vs. 0.5, p < 0.01). The most common comorbidities included hypertension (TE cohort: 49.7%; no TE cohort: 35.4%; p < 0.01) and hyperlipidemia (TE cohort: 35.2%; no TE cohort: 30.9%; p = 0.004).
Fig. 2.
Sample selection of patients with IBD with and without TE. IBD inflammatory bowel disease, TE thromboembolic event. aPatients were not allowed to have a diagnosis of any of the following IMDs prior to their first IBD diagnosis: ankylosing spondylitis, atopic dermatitis, multiple sclerosis, plaque psoriasis, psoriatic arthritis, rheumatoid arthritis and systemic lupus erythematosus. bQualifying diagnoses for autoimmune disorders were identified on the inpatient and/or outpatient services claims datasets. All patients were required to have eligibility data. cTEs included deep vein thrombosis, pulmonary embolism, ischemic stroke and myocardial infarction. dMatching between the TE cohort and no TE cohort was done with a 1:13 ratio to keep as many eligible patients with TEs as possible. eFor TE patients with > 1 eligible matched no TE patient, 1 no TE patient was randomly selected
Table 3.
Patient demographics and clinical characteristics measured during the baseline period—patients with IBD with and without TE
| Thromboembolic event | No thromboembolic event | p value | |
|---|---|---|---|
| (n = 1885) | (n = 1885) | ||
| Age at index date (years) | |||
| Mean ± SD | 58.9 ± 15.5 | 58.9 ± 15.5 | < 0.01 |
| Sex (female), n (%) | 991 (52.6) | 991 (52.6) | – |
| Index year, n (%) | |||
| 2015 | 331 (17.6) | 338 (17.9) | 0.39 |
| 2016 | 649 (34.4) | 657 (34.9) | 0.54 |
| 2017 | 504 (26.7) | 485 (25.7) | 0.16 |
| 2018 | 401 (21.3) | 405 (21.5) | 0.65 |
| Immune-mediated diseasea, n (%) | |||
| Inflammatory bowel disease | 1885 (100) | 1885 (100) | – |
| Psoriatic arthritis | 37 (2.0) | 16 (0.8) | < 0.01 |
| Systemic lupus erythematosus | 19 (1.0) | 17 (0.9) | 0.74 |
| Psoriasis | 13 (0.7) | 3 (0.2) | 0.01 |
| Rheumatoid arthritis | 6 (0.3) | 3 (0.2) | 0.32 |
| Multiple sclerosis | 5 (0.3) | 3 (0.2) | 0.48 |
| Ankylosing spondylitis | 4 (0.2) | 3 (0.2) | 0.71 |
| Atopic dermatitis | 1 (0.1) | 3 (0.2) | 0.32 |
| Charlson Comorbidity Index | |||
| Mean ± SD | 1.2 ± 1.8 | 0.5 ± 1.0 | < 0.01 |
| Comorbidities, n (%) | |||
| Cancer | 301 (16.0) | 196 (10.4) | < 0.01 |
| Cardiovascular diseases | 1,155 (61.3) | 897 (47.6) | < 0.01 |
| Atherosclerosis | 328 (17.4) | 139 (7.4) | < 0.01 |
| Atrial fibrillation | 179 (9.5) | 73 (3.9) | < 0.01 |
| Heart failure | 152 (8.1) | 38 (2.0) | < 0.01 |
| Hyperlipidemia | 663 (35.2) | 582 (30.9) | < 0.01 |
| Hypertension | 937 (49.7) | 667 (35.4) | < 0.01 |
| Chronic kidney disease | 180 (9.5) | 68 (3.6) | < 0.01 |
| Chronic obstructive pulmonary disease | 201 (10.7) | 82 (4.4) | < 0.01 |
| Diabetes | |||
| Type 1 | 40 (2.1) | 16 (0.8) | < 0.01 |
| Type 2 | 374 (19.8) | 250 (13.3) | < 0.01 |
| Fracture (hip or leg) | 24 (1.3) | 9 (0.5) | < 0.01 |
| Peripheral vascular disease | 217 (11.5) | 92 (4.9) | < 0.01 |
| Pregnancyb | 16 (1.6) | 14 (1.4) | 0.68 |
| Common classes of drugs, n (%) | |||
| Non-immune-mediating drugs | |||
| Anticoagulants | 321 (17.0) | 57 (3.0) | < 0.01 |
| Hormone replacement therapiesb | 60 (6.1) | 64 (6.5) | 0.71 |
| Testosterone replacement therapiesb | 31 (3.5) | 24 (2.7) | 0.35 |
| Oral contraceptivesb | 61 (6.2) | 70 (7.1) | 0.37 |
| Immune-mediating drugs | |||
| Biologics | 168 (8.9) | 159 (8.4) | 0.59 |
| TNF inhibitors | 157 (8.3) | 156 (8.3) | 0.95 |
| Interferon beta-1a | 0 (0) | 0 (0) | – |
| Interleukin inhibitors | 5 (0.3) | 2 (0.1) | 0.26 |
| Other biologics | 7 (0.4) | 3 (0.2) | 0.22 |
| JAK inhibitors | 0 (0) | 0 (0) | – |
| Non-biologic immunomodulators | 354 (18.8) | 308 (16.3) | 0.04 |
| Methotrexate | 41 (2.2) | 26 (1.4) | 0.07 |
| S1P receptor modulators | 1 (0.1) | 0 (0) | – |
| Other non-biologic immunomodulators | 317 (16.8) | 284 (15.1) | 0.14 |
| 5-Aminosalicyclic-acid derivative agents | 781 (41.4) | 816 (43.3) | 0.25 |
| Glucocorticoids | 693 (36.8%) | 404 (21.4%) | < 0.01 |
| NSAIDs | 235 (12.5%) | 204 (10.8%) | 0.11 |
The baseline period was defined as 1-year period prior to the index date
IBD inflammatory bowel disease, JAK Janus kinase, NSAIDs nonsteroidal anti-inflammatory drugs, S1P sphingosine 1-phosphate, SD standard deviation, TNF tumor necrosis factor
aIMDs were reported at any time prior to the index date
bThe proportions of patients with pregnancy, hormone replacement therapies, and oral contraceptives were reported out of the total number of females in each group. The proportion of patients with testosterone replacement therapies was reported out of the total number of males in each group
Healthcare Costs
Patients with IBD in the TE cohort incurred greater costs compared with patients with IBD in the no TE cohort. The total 30-day all-cause healthcare costs were $24,061 and $1715 for the TE and no TE cohorts, respectively (unadjusted cost difference $22,346, p < 0.01; data not shown). After adjustments, the 30-day cost difference between the cohorts was $20,784 (p < 0.01) (Table 4).
Table 4.
Adjusted cost differences among patients with IBD during the 30-day and 1-year study period
| 30-Day study period post TE | 1-Year study period post TE | |||||||
|---|---|---|---|---|---|---|---|---|
| IBD patients with TEs | IBD patients with no TEs | Cost difference | p value | IBD patients with TEs | IBD patients with no TEs | Cost difference | p value | |
| (n = 1885) | (n = 1885) | (n = 1885) | (n = 1885) | |||||
| All-cause healthcare costs ($)a,b | ||||||||
| Total costs | 22,669 | 1886 | 20,784 | < 0.01 | 67,054 | 22,424 | 44,630 | < 0.01 |
| Medical costs | 21,433 | 1078 | 20,355 | < 0.01 | 53,801 | 12,769 | 41,032 | < 0.01 |
| Inpatient | 18,601 | 215 | 18,386 | < 0.01 | 34,290 | 3537 | 30,753 | < 0.01 |
| Outpatient | 1979 | 749 | 1229 | < 0.01 | 17,108 | 8557 | 8551 | < 0.01 |
| Emergency department | 987 | 40 | 948 | < 0.01 | 2806 | 631 | 2175 | < 0.01 |
| Pharmacy costs | 1199 | 751 | 448 | < 0.01 | 13,663 | 9367 | 4296 | < 0.01 |
IBD inflammatory bowel disease
aAdjusted results include predicted costs and p-values, estimated using generalized estimating equations with a Tweedie distribution. The adjusted models controlled for the following: cohort assignment, age at index date, sex (female), index year, plan type (capitation), baseline comorbidities, baseline non-immune-mediating medications and select baseline immune-mediating treatments
bAll costs were inflated to 2019 US dollars
The total 1-year all-cause healthcare costs were $71,004 and $20,418 for the TE and no TE cohorts, respectively (unadjusted cost difference $50,586, p < 0.01; data not shown). After adjustments, the 1-year cost difference between the cohorts was $44,630 (p < 0.01; Table 4). TE-related costs were $20,036 and $25,311 in the 30-day and 1-year period for the TE cohort.
Discussion
The present study characterized the rates of TEs among patients with IBD versus patients without IMD as well as the cost of TEs in patients with IBD with and without TEs. Overall, patients with IBD experienced significantly greater rates of TEs compared to patients without IMD. More specifically, patients with IBD had increased rates of DVT and PE, before and after adjustments for potential confounding factors. These trends were not observed for IS and MI. Patients with IBD with TEs incurred significantly greater costs relative to patients with IBD without TEs over the 30-day and 1-year study periods.
In this study, the increased risk of TEs associated with IBD was mainly due to increased risk of VTE (i.e., DVT and PE), which aligns with prior research. The analysis of the risks of TEs was a subgroup analysis of previously published results among a sample of patients with IMDs [19]. In that study, the incremental risks of TEs were 1.78 and 1.66 for DVT and PE, respectively, among patients with IMD versus patients without IMD, while the present study found incremental risks of 2.44 and 1.90 among patients with IBD compared to the non-IMD cohort. The observed incremental risk of venous events in the IBD cohort vs. non-IMD cohort in this study was greater than the incremental risk observed among the IMD cohort versus non-IMD cohort in the previously published study [19]. The magnitude of the rates of DVT and PE in this study (2.44 and 1.90, respectively) also align with a meta-analysis that found that patients with IBD experienced an approximate two-fold increased risk for VTE compared to patients without IBD [20]. Similarly, the present study’s finding that there was no increased risk of arterial TEs (i.e., IS and MI) among patients with IBD also aligns with prior evidence [21]. A systematic review across 33 studies found no increased risk of arterial TEs (relative risk: 1.15, 95% CI 0.91, 1.45) [21]. However, to date, results regarding the risk of arterial TEs in patients with IBD have been inconsistent due to factors such as type of arterial TE, severity of IBD and patient characteristics [4, 21, 22].
The association between coagulation and inflammation is suggested to increase the risk of VTE in patients with IBD [22–25]. As such, the greater risk of VTEs among patients with IBD may be due to the close relationship between systemic inflammation and hypercoagulability in patients with IBD [1]. While the specific mechanisms that contribute to TE have yet to be elucidated, factors that promote thrombosis within the microvasculature of the intestinal tract are believed to have a critical role in pathogenesis [1]. Due to the interplay of IBD as a risk factor of TEs, there is a growing impetus to develop therapies to improve outcomes in this patient population [26]. It has been reported that certain treatments (i.e., corticosteroids, JAK inhibitors) [27, 28] may exacerbate the risk of TEs; the potential association between IBD therapies and increasing risk of DVT and PE requires further investigation [1]. The elevated risk of TE should be carefully considered when selecting treatment options to minimize risks.
Patients with IBD are a heterogeneous population due to differences in prognosis and disease severity and activity. As such, the risk for TEs could vary within the population. A limitation of this study is that the claims data did not have the clinical details necessary to stratify the analysis by disease activity and severity as well as behavioral factors such as smoking. Future studies with these clinical data elements available should evaluate how the incremental risk of TEs vary by IBD disease severity/activity and behavioral factors. Additionally, this study focused on quantifying the overall incremental risk of TEs associated with IBD, including the IBD disease itself and treatments for IBD. It would be helpful for future studies to determine how much of the incremental risk of TEs associated with IBD can be attributed to the IBD disease itself, such as the inflammatory and hypercoagulable state, and how much can be attributed to the treatments for IBD, such as corticosteroid use.
This study showed that, among patients with IBD, those with TEs incurred higher costs than those without TEs in both the 30-day and 1-year periods following a TE. To date, few studies have examined healthcare costs of TEs in patients with IBD; however, previous results show that patients across other disease areas with TEs experience a significant economic burden compared to healthy patients [29, 30]. One study estimated that annualized median costs were $17,512 and $18,901 for patients with DVT and PE, respectively, versus $680 in the control group [29]. However, the costs associated with TEs in the IBD cohort may differ from costs of TEs in the general population as well as in patients with non-IMDs due to differences in both disease pathogenesis and progression [1]. Moreover, a history of TEs represents a significant risk factor for recurrent events [31, 32]; the risk of recurrence in patients with history of VTE is 5–7% per year, which is 50 times higher than the risk in patients without a history of VTE [33]. Therefore, it is important to note that the economic burden of TEs extends beyond the diagnosis and treatment of the initial event as costs of recurring TEs and associated long-term effects can cumulatively add to the overall cost burden. Inpatient costs were the main driver behind the difference in healthcare costs between patients with IBD with TEs and patients with IBD without TEs. The difference in inpatient costs accounted for 88% and 70% of the adjusted total cost difference in the 30-day and 1-year periods, respectively. This may be due to the acute TE on index date and the resulting inpatient stay from the TE. The insight gained from the present analysis has the potential to inform healthcare stakeholders and serve as a benchmark for future efforts to improve the clinical management in this patient population.
This study should be considered within the context of specific limitations. First, as with all observational claims-based studies, the administrative claims databases used in this study may be subject to coding errors or data omission. Second, claims-based data only comprise diagnostic and procedure codes that are recorded for reimbursement rather than research purposes. As a result, the impact of variables that can affect TE outcomes, such as the prescribing behavior of healthcare providers and clinical measures (e.g., body mass index, smoking status and mobility status) could not be accounted for as this information is unavailable in claims data. Although other unmeasured and unobserved confounders could not be adjusted for, this study was able to adjust for a comprehensive list of factors known to affect TE outcomes. Third, this study was conducted in a commercially insured US population. Therefore, the results might not be generalizable to Medicare and Medicaid populations, which have different clinical and severity profiles than the commercially insured population. Medicare and Medicaid patient populations may be older and have more severe patient profiles than commercially insured patients, so the cost estimate of TE burden in those populations could potentially be higher. Thus, the cost analysis in this dataset may be a conservative estimate of the TE burden. Lastly, due to the time interval analyzed in the present study (i.e., 2014–2018), the generalizability of the study’s findings is limited. For example, recently approved treatments for IBD may be underrepresented; furthermore, patients may not have used the recorded medication as prescribed after filling the prescription.
Conclusions
Patients with IBD experienced greater rates of DVT and PE compared to patients without IMD; this elevated risk was associated with a substantial economic burden. This study suggests that the risk of VTEs should be carefully considered in the management and treatment of patients with IBD.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
Funding
Sponsorship for this study and Rapid Service and Open Access Fee were funded by Arena Pharmaceuticals, Inc. The study sponsor was involved in all stages of the study’s design, data interpretation and the decision to submit for publication.
Medical writing, editorial and other assistance
Editorial assistance in the preparation of this article was provided by Dr. Gloria DeWalt of Analysis Group, Inc. Support for this assistance was funded by Arena Pharmaceuticals, Inc.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole and have given their approval for this version to be published.
Author contributions
Study design: All authors. Formal analysis: Fan Mu, Miriam L. Zichlin, Hongbo Yang, Emma Billmyer, and Nathaniel Downes. Data interpretation, manuscript writing and editing: All authors.
Compliance with ethics guidelines
Informed consent and consent to publish were not applicable to this study. Institutional review board approval was not required for this study. The pre-existing, retrospective data from the IBM MarketScan® Commercial and Medicare Supplemental Databases are represented by IBM MarketScan to be fully de-identified in accordance with the Health Insurance Portability and Accountability Act. The data were provided under license agreement with IBM.
Data availability
The data used in this study were accessed via licensing agreement with IBM and are not publicly available.
References
- 1.Zezos P, Kouklakis G, Saibil F. Inflammatory bowel disease and thromboembolism. World J Gastroenterol. 2014;20(38):13863–13878. doi: 10.3748/wjg.v20.i38.13863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yarur AJ, Deshpande AR, Pechman DM, et al. Inflammatory bowel disease is associated with an increased incidence of cardiovascular events. Am J Gastroenterol. 2011;106(4):741–747. doi: 10.1038/ajg.2011.63. [DOI] [PubMed] [Google Scholar]
- 3.Bafford AC, Cross RK. Risk of venous thromboembolism in patients with inflammatory bowel disease extends beyond hospitalization. Inflamm Bowel Dis. 2020;26(11):1769–1770. doi: 10.1093/ibd/izaa003. [DOI] [PubMed] [Google Scholar]
- 4.Manea M, Marcu D, Motofei I, et al. Cardiovascular risk in patients with inflammatory bowel diseases: a review. Rom Biotechnol Lett. 2019;24(2):366–373. doi: 10.25083/rbl/24.2/366.373. [DOI] [Google Scholar]
- 5.Quera R, Shanahan F. Thromboembolism—an important manifestation of inflammatory bowel disease. Am J Gastroenterol. 2004;99(10):1971–1973. doi: 10.1111/j.1572-0241.2004.40923.x. [DOI] [PubMed] [Google Scholar]
- 6.Jackson LM, O'Gorman PJ, O'Connell J, et al. Thrombosis in inflammatory bowel disease: clinical setting, procoagulant profile and factor V Leiden. QJM. 1997;90(3):183–188. doi: 10.1093/qjmed/90.3.183. [DOI] [PubMed] [Google Scholar]
- 7.Talbot RW, Heppell J, Dozois RR, et al. Vascular complications of inflammatory bowel disease. Mayo Clin Proc. 1986;61(2):140–145. doi: 10.1016/S0025-6196(12)65200-8. [DOI] [PubMed] [Google Scholar]
- 8.Bryant RV, Jairath V, Curry N, et al. Thrombosis in inflammatory bowel disease: are we tailoring prophylaxis to those most at risk? J Crohns Colitis. 2014;8(2):166–171. doi: 10.1016/j.crohns.2013.09.007. [DOI] [PubMed] [Google Scholar]
- 9.Mehta F. Report: economic implications of inflammatory bowel disease and its management. Am J Manag Care. 2016;22(3 Suppl):s51–60. [PubMed] [Google Scholar]
- 10.Park KT, Ehrlich OG, Allen JI, et al. The cost of inflammatory bowel disease: an initiative from the Crohn’s and colitis foundation. Inflamm Bowel Dis. 2020;26(1):1–10. doi: 10.1093/ibd/izz104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Grosse SD, Nelson RE, Nyarko KA, et al. The economic burden of incident venous thromboembolism in the United States: a review of estimated attributable healthcare costs. Thromb Res. 2016;137:3–10. doi: 10.1016/j.thromres.2015.11.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vekeman F, LaMori JC, Laliberte F, et al. Risks and cost burden of venous thromboembolism and bleeding for patients undergoing total hip or knee replacement in a managed-care population. J Med Econ. 2011;14(3):324–334. doi: 10.3111/13696998.2011.578698. [DOI] [PubMed] [Google Scholar]
- 13.Centers for Disease Control and Prevention (CDC) Venous thromboembolism in adult hospitalizations—United States, 2007–2009. MMWR Morb Mortal Wkly Rep. 2012;61(22):401–404. [PubMed] [Google Scholar]
- 14.Kourlaba G, Relakis J, Mylonas C, et al. The humanistic and economic burden of venous thromboembolism in cancer patients: a systematic review. Blood Coagul Fibrinolysis. 2015;26(1):13–31. doi: 10.1097/MBC.0000000000000193. [DOI] [PubMed] [Google Scholar]
- 15.Sakon M, Maehara Y, Kobayashi T, et al. Economic burden of venous thromboembolism in patients undergoing major abdominal surgery. Value Health Reg Issues. 2015;6:73–79. doi: 10.1016/j.vhri.2015.03.017. [DOI] [PubMed] [Google Scholar]
- 16.Khorana AA, McCrae K, Milentijevic D, et al. The risk of recurrent VTE and major bleeding in a commercially-insured population of cancer patients treated with anticoagulation. Am J Hematol. 2019;94(2):E58–E61. doi: 10.1002/ajh.25361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee HS, Park SK, Park DI. Novel treatments for inflammatory bowel disease. Korean J Intern Med. 2018;33(1):20–27. doi: 10.3904/kjim.2017.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Olivera PA, Lasa JS, Bonovas S, et al. Safety of Janus Kinase inhibitors in patients with inflammatory bowel diseases or other immune-mediated diseases: a systematic review and meta-analysis. Gastroenterology. 2020;158(6):1554–1573 e1512. doi: 10.1053/j.gastro.2020.01.001. [DOI] [PubMed] [Google Scholar]
- 19.Setyawan J, Mu F, Yarur A, et al. Risk of thromboembolic events and associated risk factors, including treatments, in patients with immune-mediated diseases. Clin Ther. 2021;43(8):1392–1407.e1. doi: 10.1016/j.clinthera.2021.06.008. [DOI] [PubMed] [Google Scholar]
- 20.Yuhara H, Steinmaus C, Corley D, et al. Meta-analysis: the risk of venous thromboembolism in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2013;37(10):953–962. doi: 10.1111/apt.12294. [DOI] [PubMed] [Google Scholar]
- 21.Fumery M, Xiaocang C, Dauchet L, et al. Thromboembolic events and cardiovascular mortality in inflammatory bowel diseases: a meta-analysis of observational studies. J Crohns Colitis. 2014;8(6):469–479. doi: 10.1016/j.crohns.2013.09.021. [DOI] [PubMed] [Google Scholar]
- 22.Biondi RB, Salmazo PS, Bazan SGZ, et al. Cardiovascular risk in individuals with inflammatory bowel disease. Clin Exp Gastroenterol. 2020;13:107–113. doi: 10.2147/CEG.S243478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tezel A, Demir M. Inflammatory bowel disease and thrombosis. Turk J Haematol. 2012;29(2):111–119. doi: 10.5505/tjh.2012.04557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Owczarek D, Cibor D, Glowacki MK, et al. Inflammatory bowel disease: epidemiology, pathology and risk factors for hypercoagulability. World J Gastroenterol. 2014;20(1):53–63. doi: 10.3748/wjg.v20.i1.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cheng K, Faye AS. Venous thromboembolism in inflammatory bowel disease. World J Gastroenterol. 2020;26(12):1231–1241. doi: 10.3748/wjg.v26.i12.1231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Papa A, Gerardi V, Marzo M, et al. Venous thromboembolism in patients with inflammatory bowel disease: focus on prevention and treatment. World J Gastroenterol. 2014;20(12):3173–3179. doi: 10.3748/wjg.v20.i12.3173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sarlos P, Szemes K, Hegyi P, et al. Steroid but not biological therapy elevates the risk of venous thromboembolic events in inflammatory bowel disease: a meta-analysis. J Crohns Colitis. 2018;12(4):489–498. doi: 10.1093/ecco-jcc/jjx162. [DOI] [PubMed] [Google Scholar]
- 28.Verden A, Dimbil M, Kyle R, et al. Analysis of spontaneous postmarket case reports submitted to the FDA regarding thromboembolic adverse events and JAK inhibitors. Drug Saf. 2018;41(4):357–361. doi: 10.1007/s40264-017-0622-2. [DOI] [PubMed] [Google Scholar]
- 29.MacDougall DA, Feliu AL, Boccuzzi SJ, et al. Economic burden of deep-vein thrombosis, pulmonary embolism, and post-thrombotic syndrome. Am J Health Syst Pharm. 2006;63(20 Suppl 6):S5–15. doi: 10.2146/ajhp060388. [DOI] [PubMed] [Google Scholar]
- 30.Spyropoulos AC, Lin J. Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations. J Manag Care Pharm. 2007;13(6):475–486. doi: 10.18553/jmcp.2007.13.6.475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Samama MM. An epidemiologic study of risk factors for deep vein thrombosis in medical outpatients: the Sirius study. Arch Intern Med. 2000;160(22):3415–3420. doi: 10.1001/archinte.160.22.3415. [DOI] [PubMed] [Google Scholar]
- 32.Alikhan R, Cohen AT, Combe S, et al. Risk factors for venous thromboembolism in hospitalized patients with acute medical illness: analysis of the MEDENOX Study. Arch Intern Med. 2004;164(9):963–968. doi: 10.1001/archinte.164.9.963. [DOI] [PubMed] [Google Scholar]
- 33.Fahrni J, Husmann M, Gretener SB, et al. Assessing the risk of recurrent venous thromboembolism–a practical approach. Vasc Health Risk Manag. 2015;11:451–459. doi: 10.2147/VHRM.S83718. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data used in this study were accessed via licensing agreement with IBM and are not publicly available.