Abstract
Background
Vitamin D status of pregnant women is associated with body composition of the offspring. The objective of this study was to assess whether the association between maternal vitamin D status and neonatal adiposity is modified by maternal adiposity preconception.
Methods
Healthy mothers and their term appropriate weight for gestational age (AGA) infants (n = 142; 59% male, Greater Montreal, March 2016-2019) were studied at birth and 1 month postpartum (2-6 weeks). Newborn (24-36 h) serum was collected to measure total 25-hydroxyvitamin D [25(OH)D] (immunoassay); maternal pre-pregnancy BMI was obtained from the medical record. Anthropometry, body composition (dual-energy X-ray absorptiometry) and serum 25(OH)D were measured at 2-6 weeks postpartum in mothers and infants. Mothers were grouped into 4 categories based on their vitamin D status (sufficient 25(OH)D ≥ 50 nmol/L vs. at risk of being insufficient < 50 nmol/L) and pre-pregnancy BMI (< 25 vs. ≥25 kg/m2): insufficient-recommended weight (I-RW, n = 24); insufficient-overweight/obese (I-OW/O, n = 21); sufficient-recommended weight (S-RW, n = 69); and sufficient-overweight/obese (S-OW/O, n = 28). Partial correlation and linear fixed effects model were used while adjusting for covariates.
Results
At birth, infant serum 25(OH)D mean concentrations were below 50 nmol/L, the cut-point for sufficiency, for both maternal pre-pregnancy BMI categories; 47.8 [95%CI: 43.8, 51.9] nmol/L if BMI < 25 kg/m2 and 38.1 [95%CI: 33.5, 42.7] nmol/L if BMI ≥25 kg/m2. Infant serum 25(OH)D concentrations at birth (r = 0.77; P < 0.0001) and 1 month (r = 0.59, P < 0.0001) were positively correlated with maternal postpartum serum 25(OH)D concentrations. Maternal serum 25(OH)D concentration was weakly correlated with maternal percent whole body fat mass (r = − 0.26, P = 0.002). Infants of mothers in I-OW/O had higher fat mass versus those of mothers in S-OW/O (914.0 [95%CI: 766.4, 1061.6] vs. 780.7 [95%CI: 659.3, 902.0] g; effect size [Hedges’ g: 0.42]; P = 0.04 adjusting for covariates) with magnitude of difference of 220.4 g or ~ 28% difference.
Conclusions
Maternal and neonatal vitamin D status are positively correlated. In this study, maternal adiposity and serum 25(OH)D < 50 nmol/L are dual exposures for neonatal adiposity. These findings reinforce the importance of vitamin D supplementation early in infancy irrespective of vitamin D stores acquired in utero and maternal weight status.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12884-022-04403-w.
Keywords: Mother-infant dyads, Body composition, Vitamin D status
Introduction
Adverse nutritional exposures in utero and in infancy impair growth [1] and increase the risk of chronic conditions later in life [2, 3]. A burgeoning body of evidence suggests that maternal-fetal transfer of vitamin D is associated with a lean body mass phenotype in childhood [4]. This is a very complex physiological phenomenon with multiple factors to consider, some of which are modifiable. Maternal excess adiposity preconception is a modifiable correlate of low vitamin D status in the neonate [5, 6]. This is potentially due to vitamin D sequestration in maternal adipose tissue, volumetric dilution [7] and consequently hindered placental transfer of vitamin D [5]. Lower cord 25-hydroxyvitamin D [25(OH)D] concentrations are observed in newborns of mothers with BMI over 30 kg/m2 compared to those born to mothers with BMI in the recommended range (18.5 to 24.9 kg/m2), even though the mothers, on average, had sufficient vitamin D status (25(OH)D ≥ 50 nmol/L) in the third trimester [5]. In addition, neonates born to mothers with pre-gravid BMI over 25 kg/m2 [8] and those with gestational weight gain above the Institute of Medicine (IOM) guidelines [9] have elevated neonatal whole body fat mass and percentage body fat [10].
In Canada, 10-15% of women do not have sufficient vitamin D status [11–13]. Based on pregnancy cohort studies, inadequate maternal 25(OH)D during gestation associates with higher abdominal adiposity in neonates [14], as well as higher body fat (%) in children at 5 to 9.5 years of age [15, 16]. Similarly, mothers in the highest quartile of vitamin D status in the third trimester had children with greater lean mass (%) at 4 years of age compared to those of mothers in the lowest quartile [17]. These patterns prevailed even after adjusting for sociodemographic factors as well as maternal BMI before [14, 17] or during [15, 16] pregnancy.
Recent evidence suggests that fetal exposure to both low maternal vitamin D status and excess adiposity is associated with body composition of the offspring. The majority of these studies used BMI as a proxy measure of adiposity. Studies reporting upon lean and fat mass partitioning in both mother and neonate are scarce. The aim of the current study was to explore the correlates of maternal and neonatal vitamin D status and to assess whether the association between maternal vitamin D status and neonatal adiposity is modified by maternal adiposity preconception.
Materials and methods
Study design and population
Participants included mother-infant pairs (n = 142) who were recruited at the Lakeshore General Hospital, located in greater Montréal, Québec, Canada as part of a trial of vitamin D supplementation in breastfed infants, from March 2016 through to March 2019. The present study includes data collected prior to hospital discharge as previously published [18], and data from the baseline visit (2-6 weeks postpartum) before entering a trial (NCT02563015). The inclusion criteria for this analysis were healthy, singleton, term born infants of appropriate weight for gestational age (AGA) [19]. Exclusion criteria were: infants born to mothers with gestational diabetes or hypertension in the present pregnancy, comorbidities (liver, renal, celiac and Crohn’s diseases), medications that are known to impact vitamin D metabolism or limit growth, as well as smoking or illicit drugs [20–22].
Obstetric history, demographic and lifestyle surveys
Prior to hospital discharge, the obstetric history including pre-pregnancy weight, weight at delivery, parity, and mode of delivery were obtained from the medical record. The mother’s pre-pregnancy BMI was then calculated using mothers’ weight prior to pregnancy and measured height at the postpartum visit. Demographic information surveyed included: maternal age and self-reported population group (white/all other groups combined including if unknown) which was defined according to the proposed guideline from Canadian Institute for Health Information [23], and in doing so any mixed ancestry were categorized as other groups. The highest level of education completed (elementary/high school, college/vocational school, or university), and household income were surveyed according to the median annual income for Canadian families with children and collapsed into ≥70,000, < 70,000 Canadian dollars (CAD), or not reported [24]. Lifestyle factors were surveyed as reported previously [18] including maternal multivitamin supplement use (yes/no) and the frequency (every day, almost every day, 2-3/week or less); and exercise habits (yes/no), typical frequency (none, 1-2 h/wk., ≥ 3 h/wk), and intensity (low, moderate or high), during the 3 months prior to pregnancy and then separately for across pregnancy. Moreover, whether the infants had received vitamin D supplements containing 400 IU/d prescribed by their physician prior to hospital discharge was surveyed at the follow-up visit.
Biochemistry measurements
Capillary blood was sampled from the neonates between 24 and 36 h of life [18]. Mothers and their infants participated in a postnatal visit at 1 month (± 0.5 month) at the Mary Emily Clinical Nutrition Research Unit, McGill University. In the non-fasted state, capillary blood samples (0.4-0.5 ml) were collected from infants by heel lance; and a maternal venous sample (5 ml) was taken to assess vitamin D status which does not vary significantly between delivery and 1 mo postpartum [25, 26]. Total serum 25(OH)D using an automated chemiluminescence immunoassay (Liaison, DiaSorin Inc.). The laboratory maintained a certificate of proficiency from the Vitamin D External Quality Assessment Scheme. Vitamin D control samples from the National Institute of Standards and Technology (NIST) quality assurance program were implemented in routine quality control measures. The inter-assay coefficient of variation for NIST972a (levels 1 to 4) was on average < 10% and the accuracy 97.4% of certified values. The inter-assay coefficient of variation for an internal laboratory control (62.8 nmol/L) human serum sample was 8.2% across all assays. Deming regression was used to standardize the original measured 25(OH)D values for mothers and infants to NIST reference measurements: standardized concentration = 0.9634 (Liaison concentration + 3.122 nmol/L). In a subgroup of mothers and infants (n = 83), total 25(OH)D was in agreement (mean difference = − 0.8) with liquid chromatography tandem mass spectroscopy (Queen’s University, Kingston, Ontario, Canada) using an assay certified by the Vitamin D Standardization-Certification Program. In the current study, the cut-point for sufficiency of vitamin D status in mother-infant dyads was set at ≥50 nmol/L of serum 25(OH)D in accordance with the IOM [27] and since vitamin D status above this cut-point also positively relates to lean mass at 3-4 y [17, 28]. For ease of readability, individuals with serum 25(OH)D concentration < 50 nmol/L, were termed as insufficient to reflect the increasing risk of being insufficient as serum 25(OH)D falls below 50 nmol/L. The population cut-point of 40 nmol/L of serum 25(OH)D was not used in the present analysis since the assessment was at the level of individual mother-infant dyads.
Skin pigmentation and UVB exposure
Skin tone of the infant was measured at the research facility by taking the average of three measurements at the inner upper arm for constitutive pigmentation (basal color) using a spectrophotometer (CM-700d/600d, Konica Minolta, USA). Individual typological angle (ITAo) was calculated with the L* and b* values using published equations [29]. Infants were classified into two skin tone groups (F I-III; F IV-VI) based on Fitzpatrick scales [30, 31]. Based on the strength of solar UVB, vitamin D synthesizing/vitamin D non-synthesizing periods (April 1st-October 31st/November 1st-March 31st) [32] and season (winter, spring, summer, fall) [18] at birth were used as proxies for potential vitamin D synthesis.
Anthropometric measurements
At the research facility, infant weight, length and head circumference were measured using standard methodology [33]. Weight, length, and BMI for age z-scores were calculated using WHO growth standards and software (WHO AnthroPlus, Switzerland). Measured maternal weight and height were used to calculate BMI (kg/m2) [33]. Total weight gain in pregnancy was estimated by subtracting pre-pregnancy weight from the weight obtained at delivery [34] and classified as inadequate, adequate, or excess weight gain according to pre-pregnancy BMI [9].
Body composition measurements
Body composition of infants (infant whole body software) and mothers (whole body software) was assessed in a three-compartment model using a fan-beam dual-energy X-ray absorptiometer (DXA; APEX version 13.3:3, Hologic 4500A Discovery Series, Bedford, MA) as reported in detail [33]. For quality control and quality assurance purposes, a spine phantom (Hologic phantom; No. 14774) was used at each study visit and the coefficient of variation for bone mineral content, bone mineral density, and bone area were < 1%; the radiographic uniformity tests were within established limits across the study. Whole body scans provided lean mass (g) excluding bone mineral content, fat mass (g and %); from these values, lean mass index (LMI, lean body mass (kg)/stature (m)2), and fat mass index (FMI, fat mass (kg)/stature (m)2) were then calculated using standing height for mothers and crown heel length for infants.
Power analysis and sample size estimation
This was a convenience sample of 142 mother-infant dyads in a cross-sectional analysis at birth and 1 month postpartum as part of a randomized trial [35], and thus a retrospective power was calculated based on changes in the primary outcome (fat mass) between mothers with pre-pregnancy ≥25 kg/m2 and 25(OH)D < 50 (n = 21) and those with pre-pregnancy ≥25 kg/m2 and 25(OH)D ≥ 50 (n = 28). Power was estimated to be 75% using the procedure described by Kononoff [36] for specific data sets accounting for the fixed effects of gestational weight gain, neonatal sex, gestational age, UVB period at birth, actual age of infant at the postnatal visit, and infant length in linear fixed effects design [37].
Statistical analysis
Data analyses were conducted using Statistical Analysis System (SAS; version 9.4, SAS Institute Inc., Cary, NC). Descriptive characteristics for mothers and infants were expressed as mean (95% confidence interval) or n (%). Mothers were classified into 1 of 4 groups according to serum 25(OH)D concentrations (insufficient: 25(OH)D < 50, sufficient: 25(OH)D ≥ 50 nmol/L) and pre-pregnancy BMI (recommended BMI: < 25, overweight/obese: ≥25 kg/m2). The 2 × 2 design of maternal vitamin D status and pre-pregnancy BMI formed 4 groups of interest, I-RW: insufficient-recommended weight (25(OH)D < 50 and BMI < 25 kg/m2), I-OW/O: insufficient-overweight/obese (25(OH)D < 50 and BMI ≥25 kg/m2), S-RW: sufficient-recommended weight (25(OH)D ≥ 50, BMI: < 25 kg/m2), and S-OW/O: sufficient-overweight/obese (25(OH)D ≥ 50 nmol/L, BMI ≥25 kg/m2). For maternal and neonatal characteristics at delivery and 1 mo postpartum, data were compared among these groups using a linear fixed effects model, fit using SAS PROC MIXED [37] for continuous variables, followed by post hoc Tukey’s tests with Tukey-Kramer adjustment for multiple comparisons. Chi-square or Fisher’s exact tests (frequency analysis) were used to test for differences in proportions. According to the product of the interaction effect of maternal vitamin D status and pre-pregnancy BMI, the 4 groups of interests (I-RW, I-OW/O, S-RW, and S-OW/O), frequency analysis creates 3-way crosstabulation tables. Each of the categorical variables was used in the model to stratify the crosstabulation tables followed by the the interaction term (maternal 25(OH)D*pre-pregnancy BMI) creating two 2-way tables of 25(OH)D and pre-pregnancy BMI, for each level of the categorical variable.
The interrelationship among maternal pre-pregnancy BMI, maternal 25(OH)D, and the interaction effect of these two variables with neonatal body composition (lean and fat mass, related percentages, and indices) was tested using a linear fixed effects model, fit using SAS PROC MIXED [37], and post hoc Tukey’s tests adjusted for multiple comparisons using Tukey-Kramer adjustment. Disjunctive cause criterion [38] was used for selection of covariates in the model and in accordance with factors known to be associated with body composition or vitamin D status. Fixed effects included in the model were gestational weight gain [10], neonatal sex [35], gestational age (GA) [39], UVB period at birth [32], actual age of the infants at the postnatal visit [39], and infant length [1]. Other variables that were considered in these analyses were: maternal age, family income, self-reported population group, education, multivitamin supplement use, physical activity during pregnancy as well as changes in neonatal serum 25(OH)D concentration during the postpartum period (delta 25(OH)D). These variables did not improve the model as judged by Bayesian information criterion (BIC), thus were removed from the final model. We further performed sex-stratified analysis to determine whether the associations between maternal pre-pregnancy BMI, maternal vitamin D status and neonatal body composition differ by sex. In this analysis, the three-way interaction of maternal pre-pregnancy BMI, maternal vitamin D status, and sex which was otherwise tested using the same model, appeared non-significant and was removed. The two way interaction term (maternal pre-pregnancy BMI*maternal vitamin D status) and sex were retained in the final model.
In order to determine correlates of serum 25(OH)D concentrations in mother-infant dyads, maternal and neonatal serum 25(OH)D were separately modelled (using a linear fixed effects model fit using SAS PROC MIXED) against maternal pre-pregnancy BMI (healthy BMI: < 25, overweight/obese: ≥25 kg/m2), postpartum BMI (healthy:18.5-24.9, overweight 25.0-29.9, obese ≥30 kg/m2), postpartum FMI [40] (low to normal: 4-9, excess fat: > 9-13, obese: > 13 kg/m2), gestational weight gain [9] (inadequate, adequate, and excess), and other important lifestyle or demographic factors including exercise before and during pregnancy (yes/no) as two separate variables, multivitamin supplement use prior to/during pregnancy (yes/no), UVB period at birth/at delivery (vitamin D synthesizing, non-synthesizing period), parity (primiparous, multiparous), maternal education (elementary/high school, college/vocational school, university), and income (≥70,000, < 70,000 CAD, or not reported). Postpartum FMI [38] was substituted in place of BMI, and differences among categories otherwise tested using the same model.
In these regression models for maternal 25(OH)D, the frequency of multivitamin supplements, the frequency and intensity of the exercise prior to/during pregnancy as well as season at birth did not improve the models and were thus removed. For the regression analysis of neonatal 25(OH)D, neonatal sex (male, female) and skin tone (F I-III, F IV-VI) were additional fixed factors included in the model. Normality of continuous data were tested using Kolmogorov-Smirnov and Shapiro-Wilk tests and the residuals were normally distributed. Levene and Bartlett tests of homogeneity of variances were used to confirm the assumptions of the post-hoc testing. Correlation tests were used to identify linear relationships between continuous variables including maternal and neonatal 25(OH)D, and maternal 25(OH)D and maternal body composition (SAS PROC CORR and PROC GLM using MANOVA option). For all tests, a P-value of < 0.05 was used to guide interpretation of the results.
Results
Maternal characteristics according to vitamin D status and pre-pregnancy BMI categories
Groups categorized based on maternal 25(OH)D and pre-pregnancy BMI were not different in terms of maternal age, country of birth, self-reported population group and education (Table 1). Although the majority of mothers (92.3% overall) took a multivitamin supplement during pregnancy, the proportion was lower in I-RW compared to the other groups. No differences were observed in the dose of vitamin D taken. A higher proportion of mothers in I-OW/O had household annual income < 70,000 CAD compared to the other groups. At the postpartum visit 98.6% of mothers were breastfeeding, 2 mothers discontinued breastfeeding between recruitment at birth and the baseline visit due to milk insufficiency.
Table 1.
Maternal characteristics according to maternal pre-pregnancy BMI and postpartum vitamin D status
| Characteristic1 | All | 25(OH)D < 50 nmol/L | 25(OH)D ≥ 50 nmol/L | P-value2 | ||||
|---|---|---|---|---|---|---|---|---|
| BMI < 25 kg/m2 (n = 24) | BMI ≥ 25 kg/m2 (n = 21) | BMI < 25 kg/m2 (n = 69) | BMI ≥ 25 kg/m2 (n = 28) | 25(OH)D | BMI | 25(OH)D*BMI | ||
| Age at delivery, y | 32.2 (31.4, 32.9) | 31.0 (28.9, 33.0) | 32.7 (30.1, 35.2) | 31.9 (31.0, 32.8) | 33.5 (31.7, 35.2) | 0.29 | 0.05 | 0.93 |
| Parity, n (%) | ||||||||
| Primiparous | 44 (31.0) | 12 (50.0) | 6 (28.6) | 21 (30.4) | 5 (17.9) | 0.23 | 0.0009 | 0.28 |
| Multiparous | 98 (69.0) | 12 (50.0) | 15 (71.4) | 48 (69.6) | 23 (82.1) | < 0.0001 | 0.03 | 0.04 |
| Pre-pregnancy BMI, kg/m2 | 24.6 (23.8, 25.3) | 22.4 (21.6, 23.2) | 30.0 (28.1, 31.8) | 21.8 (21.4, 22.2) | 29.2 (27.3, 31.0) | 0.22 | < 0.0001 | 0.85 |
| Gestational weight gain, kg | 13.6 (12.6, 14.6) | 13.3 (10.2, 16.5) | 12.0 (9.3, 14.6) | 14.8 (13.5, 16.1) | 12.0 (9.7, 14.4) | 0.49 | 0.07 | 0.53 |
| Serum 25(OH)D, nmol/L | 67.4 (63.1, 71.7) | 40.0 (36.2, 43.9) | 40.4 (36.8, 43.9) | 83.3 (78.1, 88.6) | 71.8 (64.8, 78.8) | < 0.0001 | 0.10 | 0.08 |
| Maternal birthplace, n (%) | ||||||||
| Canada | 90 (63.4) | 13 (54.2) | 8 (38.1) | 50 (72.5) | 19 (67.9) | < 0.0001 | 0.0001 | 0.35 |
| Elsewhere | 52 (36.6) | 11 (45.8) | 13 (61.9) | 19 (27.5) | 9 (32.1) | 0.58 | 0.27 | 0.11 |
| Self-reported population group, n (%) | ||||||||
| White | 79 (55.6) | 8 (37.5) | 7 (33.3) | 47 (71.0) | 17 (60.7) | < 0.0001 | 0.0002 | 0.16 |
| All other groups3 | 63 (44.4) | 16 (62.5) | 14 (66.7) | 22 (29.0) | 11 (39.3) | 0.80 | 0.20 | 0.32 |
| Supplement use4, n (%) | ||||||||
| Yes | 131 (92.3) | 20 (83.3) | 20 (95.2) | 65 (94.2) | 26 (92.9) | < 0.0001 | 0.0007 | 0.02 |
| No | 11 (7.7) | 4 (16.7) | 1 (4.8) | 4 (5.8) | 2 (7.1) | 0.76 | 0.13 | 0.62 |
| Vitamin D dosage, IU/d | 501.5 (478.6, 524.3) | 470 (402.8, 537.2) | 513.2 (458.8, 567.5) | 504.6 (472, 537.1) | 510.9 (453.7, 568.1) | 0.53 | 0.34 | 0.48 |
| Education, n (%) | ||||||||
| Elementary/high school | 13 (9.2) | 4 (17.8) | 4 (19.0) | 4 (5.8) | 1 (3.6) | 0.41 | 0.41 | 0.28 |
| College/vocational school | 30 (21.1) | 5 (13.3) | 1 (4.8) | 15 (21.7) | 9 (32.1) | 0.001 | 0.07 | 0.33 |
| University | 99 (69.7) | 15 (68.9) | 16 (76.2) | 50 (72.5) | 18 (64.3) | 0.0002 | 0.002 | 0.01 |
| Family annual income5, n (%) | ||||||||
| ≥ 70,000 CAD | 80 (56.3) | 15 (62.5) | 5 (23.8) | 42 (60.9) | 18 (64.3) | < 0.0001 | 0.0001 | 0.67 |
| < 70,000 CAD | 41 (28.9) | 4 (16.7) | 13 (61.9) | 18 (26.1) | 6 (21.4) | 0.27 | 0.64 | 0.001 |
| Not reported | 21 (14.8) | 5 (20.8) | 3 (14.3) | 9 (13.0) | 4 (14.3) | 0.28 | 0.13 | 1.00 |
Abbreviations: 25(OH)D 25-hydroxyvitamin D, BMI Body mass index, CAD Canadian dollar
1Data are mean (lower and upper 95% confidence limits) or n (%)
2Data were compared using a linear fixed effects model for continuous variables followed by post hoc Tukey’s tests with Tukey-Kramer adjustment for multiple comparisons and Chi-square or Fisher exact tests for categorical variables (using frequency procedure to create 3-way crosstabulation tables; categorical variables were used in the model to stratify the crosstabulation tables followed by the last two variables: maternal 25(OH)D*pre-pregnancy BMI, creating two 2-way tables of 25(OH)D and pre-pregnancy BMI, for each level of the categorical variables)
3Other groups included: South Asian, Chinese, Black, Filipino, Latin American, Arab, Southeast Asian, West Asian, Korean, Japanese, or other
4Use of prenatal supplement containing vitamin D during pregnancy
5The median income (in Canadian dollars) for Canadian families with children
Neonatal characteristics at birth and postpartum according to maternal vitamin D status and pre-pregnancy BMI
Newborns (59% male) were not different among groups in terms of GA, age at the postpartum visit, UVB period at birth, and anthropometric measurements (Table 2). At birth, on average, infant serum 25(OH)D concentrations were below the cut-point for sufficiency of 50 nmol/L for both maternal pre-pregnancy BMI categories of within or above the recommended range (47.8 [95%CI: 43.8, 51.9] vs. 38.1 [95%CI: 33.5, 42.7]). At birth and 1 month of age, infants of mothers in I-RW and I-OW/O had significantly lower serum 25(OH)D concentrations compared to infants born to mothers in S-RW and S-OW/O. In addition, more infants in I-OW/O were male compared to the other groups whereas a higher proportion of infants in S-RW had skin tone F I-III compared to the rest of the groups.
Table 2.
Neonatal characteristics according to maternal pre-pregnancy BMI and postpartum vitamin D status
| Characteristic1 | All | 25(OH)D < 50 nmol/L | 25(OH)D ≥ 50 nmol/L | P-value2 | ||||
|---|---|---|---|---|---|---|---|---|
| BMI < 25 kg/m2 (n = 24) | BMI ≥ 25 kg/m2 (n = 21) | BMI < 25 kg/m2 (n = 69) | BMI ≥ 25 kg/m2 (n = 28) | 25(OH)D | BMI | 25(OH)D*BMI | ||
| Birth | ||||||||
| Gestational age, wk | 39.64 (39.5, 39.8) | 39.9 (39.4, 40.4) | 39.7 (39.2, 40.1) | 39.7 (39.4, 39.9) | 39.3 (38.9, 39.8) | 0.13 | 0.13 | 0.90 |
| Sex, n (%) | ||||||||
| Male | 83 (58.5) | 13 (54.2) | 14 (66.7) | 40 (58.0) | 16 (57.1) | 0.002 | 0.01 | 0.04 |
| Female | 59 (41.5) | 11 (45.8) | 7 (33.3) | 29 (42.0) | 12 (42.9) | 0.003 | 0.01 | 0.47 |
| UVB period3, n (%) | ||||||||
| Synthesizing | 83 (58.5) | 14 (58.3) | 9 (42.9) | 43 (62.3) | 17 (60.7) | < 0.0001 | 0.001 | 0.34 |
| Non-synthesizing | 59 (41.5) | 10 (41.7) | 12 (57.1) | 26 (37.7) | 11 (39.3) | 0.05 | 0.09 | 0.06 |
| Weight, kg | 3.4 (3.3, 3.5) | 3.4 (3.3, 3.6) | 3.5 (3.3, 3.7) | 3.3 (3.3, 3.4) | 3.4 (3.2, 3.6) | 0.12 | 0.44 | 0.69 |
| Weight z score | 0.2 (0.0, 0.3) | 0.3 (−0.1, 0.7) | 0.3 (− 0.1, 0.7) | 0.1 (− 0.1, 0.2) | 0.2 (− 0.1, 0.5) | 0.20 | 0.68 | 0.54 |
| Serum 25(OH)D, nmol/L | 44.5 (41.3, 47.6) | 29.3 (24.8, 33.7) | 26.6 (23.0, 30.2) | 54.3 (50.0, 58.6) | 46.7 (40.8, 52.5) | < 0.0001 | 0.07 | 0.38 |
| Postnatal visit | ||||||||
| Age, mo | 0.7 (0.6, 0.8) | 0.7 (0.6, 0.8) | 0.6 (0.5, 0.8) | 0.7 (0.7, 0.8) | 0.7 (0.6, 0.8) | 0.37 | 0.25 | 0.51 |
| Weight, kg | 4.0 (3.9, 4.1) | 4.1 (3.8, 4.3) | 3.9 (3.7, 4.2) | 3.9 (3.8, 4.1) | 4.0 (3.8, 4.2) | 0.61 | 0.68 | 0.29 |
| Weight z score | −0.1 (− 0.2, 0.0) | 0.1 (−2.0, 0.5) | − 0.01 (− 0.4, 0.4) | −0.2 (− 0.4, − 0.03) | −0.02 (− 0.4, 0.3) | 0.23 | 0.86 | 0.28 |
| Length, cm | 53.1 (52.7, 53.4) | 53.8 (52.9, 54.8) | 53.0 (52.2, 53.8) | 52.9 (52.4, 53.3) | 53.0 (52.1, 53.9) | 0.22 | 0.34 | 0.19 |
| Length z score | −0.01 (− 0.2, 0.1) | 0.4 (− 0.1, 0.8) | 0.1 (− 0.3, 0.4) | −0.2 (− 0.4, 0.1) | −0.03 (− 0.4, 0.3) | 0.06 | 0.57 | 0.20 |
| Head circumference, cm | 36.4 (36.2, 36.6) | 36.5 (35.9, 37.0) | 36.5 (35.9, 37.0) | 36.3 (36.1, 36.6) | 36.7 (36.2, 37.2) | 0.95 | 0.40 | 0.43 |
| Head circumference z score | 0.1 (−0.02, 0.3) | 0.2 (− 0.3, 0.6) | 0.3 (− 0.1, 0.7) | −0.03 (− 0.2, 0.2) | 0.3 (0.01, 0.7) | 0.71 | 0.13 | 0.43 |
| Serum 25(OH)D, nmol/L | 54.7 (51.9, 57.5) | 45.1 (40.3, 49.9) | 42.3 (34.5, 50.1) | 61.9 (57.9, 65.9) | 54.5 (49.3, 59.7) | < 0.0001 | 0.08 | 0.42 |
| Skin tone4, n (%) | ||||||||
| F I-III | 110 (77.5) | 17 (70.8) | 14 (66.7) | 59 (85.5) | 20 (71.4) | < 0.0001 | < 0.0001 | 0.04 |
| F IV-VI | 32 (22.5) | 7 (29.2) | 7 (33.3) | 10 (14.5) | 8 (28.6) | 0.48 | 0.72 | 0.75 |
Abbreviations: 25(OH)D 25-hydroxyvitamin D, F Fitzpatrick, UVB Ultraviolet B
1Data are mean (lower and upper 95% confidence limits) or n (%)
2Data were compared using a linear fixed effect model for continuous variables followed by post hoc Tukey’s tests with Tukey-Kramer adjustment for multiple comparisons; and Chi-square or Fisher exact tests for categorical variables (using frequency procedure to create 3-way cross tabulation tables; categorical variables were used in the model to stratify the crosstabulation tables followed by the last two variables: maternal 25(OH)D*pre-pregnancy BMI, creating two 2-way tables of 25(OH)D and pre-pregnancy BMI, for each level of the categorical variables)
3Vitamin D synthesizing: April 1st-October 31st or vitamin D non-synthesizing: November 1st-March 31st
4Classified based on Fitzpatrick descriptions: F I-III (light) and F IV-VI (dark) [30, 31]
Overall, the majority (95.0%) of infants received daily vitamin D supplements (containing 400 IU vitamin D) between discharge from hospital and the follow-up visit. Infant mean serum 25(OH)D concentrations significantly increased during the postnatal period (birth: 44.5 [95%CI: 41.3, 47.6] vs. 1 month: 54.7 [95%CI: 51.9, 57.5] nmol/L; P < 0.0001). Infants born with serum 25(OH)D ≥ 50 nmol/L had significantly lower (P < 0.0001) mean change in serum 25(OH)D concentration (1.4, 95%CI: − 3.0, 5.8 nmol/L) compared to neonates born with 25(OH)D 30-49.9 nmol/L (12.9, 95%CI: 10.2, 15.7 nmol/L), and those deficient < 30 nmol/L (17.7, 95%CI: 12.5, 22.8 nmol/L). Infant serum 25(OH)D concentrations at birth (r = 0.77; P < 0.0001) and at 1 month (r = 0.59, P < 0.0001) were positively correlated with maternal serum 25(OH)D concentrations. These correlations remained evident after adjusting for parity, maternal multivitamin supplement use, gestational age at birth, sex, UVB period at birth, and infant skin tone.
Maternal vitamin D status, indicators of adiposity and whole-body lean mass
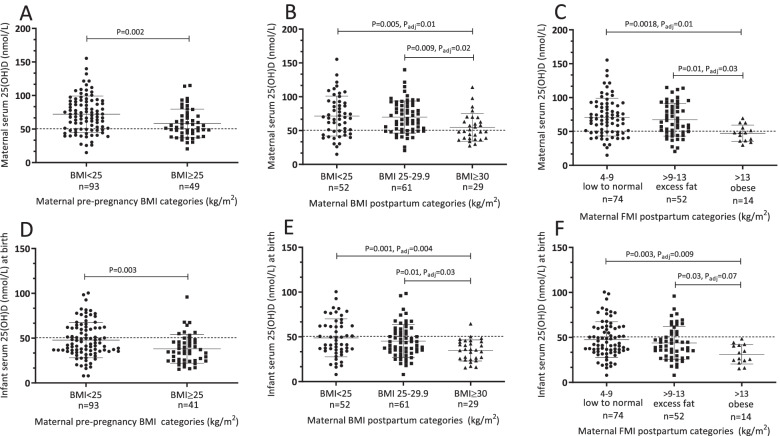
In mothers, serum 25(OH)D concentrations were weakly, positively correlated with whole body lean mass (r = 0.23, P = 0.006) and weakly, inversely correlated with percent whole body fat mass (r = − 0.26, P = 0.002). Maternal serum 25(OH)D was on average higher in mothers with pre-pregnancy BMI < 25 kg/m2 (Fig. 1A) compared to ≥25 kg/m2. Similarly, serum 25(OH)D was lower with postpartum BMI < 25 kg/m2 or 25-29.9 kg/m2 (Fig. 1B) compared to mothers with BMI ≥30 kg/m2. However, on average, maternal serum 25(OH)D concentrations were ≥ 50 nmol/L in all BMI categories (68.3 and 57.1% of mothers with BMI < 25 and ≥ 25 kg/m2 had 25(OH)D concentrations ≥50 nmol/L, respectively). Likewise, serum 25(OH)D of mothers with low to normal or excess FMI categories was higher compared to mothers with FMI in the obese range (Fig. 1C). Appropriateness of gestational weight gain was not related to maternal serum 25(OH)D (Supplementary Table 1). Among important correlates of vitamin D status, mothers who self-reported being physically active (indoor and outdoor combined) 3 months prior to conception or during pregnancy had higher 25(OH)D concentrations postpartum versus mothers who were not active. Additionally, self-reported population group (white/all other groups) was a prominent correlate of maternal serum 25(OH)D. Other tested covariates such as parity, multivitamin supplement use, education level and family income were not related.
Fig. 1.
Maternal and neonatal serum 25(OH)D according to maternal BMI and FMI categories. Serum 25(OH)D concentrations of A mothers according to pre-pregnancy BMI categories (healthy: BMI < 25 or overweight/obese: BMI ≥25 kg/m2), B serum 25(OH)D concentrations of mothers according to postpartum BMI categories (healthy: BMI < 25, overweight: BMI 25-29.9, obese: BMI ≥30 kg/m2, C mothers according to their postpartum fat mass index (FMI) categories (low to normal: 4-9, excess fat: > 9-13, and obese: > 13 kg/m2). Serum 25(OH)D concentrations of infants at birth according to mothers, D pre-pregnancy BMI categories, E postnatal BMI categories and, F FMI categories. Data were compared using a linear fixed effects model, maternal pre-pregnancy, postpartum BMI and FMI as categorical fixed effects followed by post hoc Tukey’s tests with Tukey-Kramer adjustment for multiple comparisons. Data are mean ± SD
Neonatal vitamin D status and maternal indicators of adiposity
The mean serum 25(OH)D was on average higher in infants of mothers with pre-pregnancy BMI (Fig. 1D) < 25 kg/m2 compared to ≥25 kg/m2. Infant 25(OH)D was also higher in infants of mothers with postpartum BMI < 25 kg/m2 or 25-29.9 kg/m2 (Fig. 1E) compared to mothers with BMI ≥30 kg/m2. Similarly, serum 25(OH)D concentrations of infants born to mothers in the low to normal category of FMI were higher compared to infants born to mothers in the obese category (Fig. 1F). However, on average, infant serum 25(OH)D concentrations were below the cut-point for sufficiency of 50 nmol/L for all maternal BMI (38.7 and 20.4% of infants of mothers with < 25 and ≥ 25 kg/m2 had vitamin D sufficiency) or FMI categories (39.2, 30.8, and 0% of infants of mothers with FMI of low to normal: 4-9, excess fat: > 9-13, and obese: > 13 kg/m2, respectively). The relationship among neonatal vitamin D status and maternal indicators of adiposity and other maternal factors is shown in detail in (Supplementary Table 2).
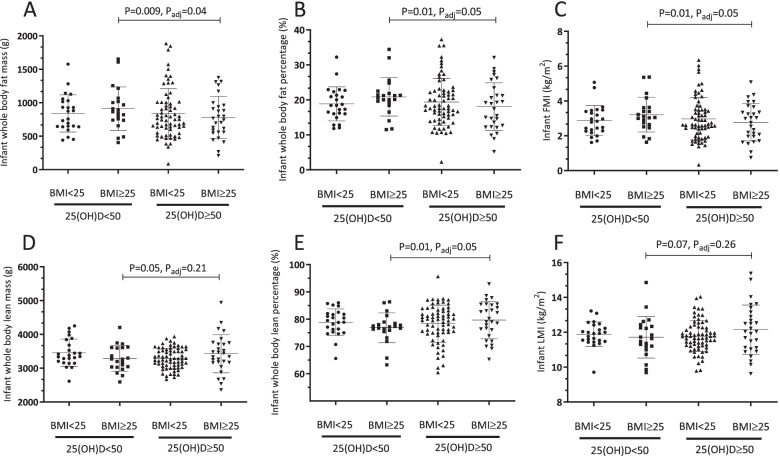
Maternal pre-pregnancy BMI (< 25 kg/m2 or ≥ 25 kg/m2) and maternal serum 25(OH)D (< 50 or ≥ 50 nmol/L) as independent categorical variables were not associated with any of neonatal indicators of adiposity (Table 3 and Supplementary Table 3). However, a significant interaction effect of maternal pre-pregnancy BMI and 25(OH)D concentration was observed for neonatal fat mass (Table 3), fat percentage and FMI adjusting for multiple covariates (Supplementary Table 3). After pairwise comparison tests, infants of mothers in the I-OW/O group with elevated pre-pregnancy BMI (≥25 kg/m2) and vitamin D insufficiency (25(OH)D < 50 nmol/L) had significantly higher fat mass (Fig. 2A), fat percentage (Fig. 2B), and FMI (Fig. 2C) compared to infants born to mothers in the S-OW/O group with BMI > 25 kg/m2 but vitamin D sufficiency (25(OH)D ≥ 50 nmol/L). The magnitude of difference in whole body fat mass observed between I-OW/O and S-OW/O groups was 220.4 g (95% CI: 56.4, 384.3) representing ~ 28% difference with effect size of 0.42. Whole body fat mass, fat percentage, and FMI were also higher in female infants versus males (P < 0.05) (Supplemental Fig. 1A, B, C) and in infants born in the vitamin D synthesizing period (UVB period) versus those born vitamin D non-synthesizing period (P < 0.05) (Supplemental Table 3). Other covariates linked to greater infant adiposity indicators were infant age and length (Table 3 and Supplemental Table 3). In these adjusted mixed models, gestational weight gain was not associated with neonatal fat mass or any other adiposity indicators.
Table 3.
Correlates of neonatal body composition
| Fixed effects modela | Regression coefficients | 95% Confidence intervals | P-value | Adjusted P-value |
|---|---|---|---|---|
| Neonatal whole-body fat mass g (R2 0.38, R2adj 0.37)a | ||||
| Sexb of infant (Ref: female) | − 119.85 | − 219.24, − 20.45 | 0.02 | |
| Gestational age at birth, wk | −15.38 | −67.13, 36.38 | 0.56 | |
| Infant age, mo | 435.35 | 187.62, 683.09 | 0.001 | |
| Infant length, cm | 58.80 | 29.99, 87.57 | < 0.0001 | |
| UVB period at birthc (Ref: non-synthesizing period) | 105.28 | 8.94, 201.63 | 0.03 | |
| Gestational weight gain, kg | 6.57 | −1.61, 14.76 | 0.11 | |
| Maternal pre-pregnancy BMId (Ref: < 25 kg/m2) | − 45.26 | − 173.54, 83.02 | 0.12 | |
| Maternal 25(OH)De (Ref: ≥50 nmol/L) | − 40.57 | − 176.04, 94.91 | 0.09 | |
| BMI*25(OH)D interaction (pairwise comparisons) | 0.02 | |||
| BMI ≥ 25, 25(OH)D < 50 vs BMI ≥ 25, 25(OH)D ≥ 50 | 220.40 | 56.19, 384.60 | 0.009 | 0.04 |
| BMI ≥ 25, 25(OH)D < 50 vs BMI < 25, 25(OH)D < 50 | 215.70 | 44.73, 386.68 | 0.01 | 0.07 |
| BMI ≥ 25, 25(OH)D < 50 vs BMI < 25, 25(OH)D ≥ 50 | 175.14 | 28.38, 321.89 | 0.02 | 0.09 |
| BMI ≥ 25, 25(OH)D ≥ 50 vs BMI < 25, 25(OH)D < 50 | −4.70 | − 161.54, 152.15 | 0.95 | 0.99 |
| BMI ≥ 25, 25(OH)D ≥ 50 vs BMI < 25, 25(OH)D ≥ 50 | −45.26 | −173.54, 83.02 | 0.49 | 0.90 |
| BMI < 25, 25(OH)D < 50 vs BMI < 25, 25(OH)D ≥ 50 | −40.57 | −176.04, 94.91 | 0.55 | 0.93 |
| Neonatal whole-body lean mass g (R2 0.53, R2adj 0.52) | ||||
| Sex of infant (Ref: female) | 174.52 | 73.41, 275.64 | 0.0009 | |
| Gestational age at birth, wk | 32.29 | −20.36, 84.94 | 0.23 | |
| Infant age, mo | 294.98 | 42.95, 547.00 | 0.02 | |
| Infant length, cm | 102.26 | 72.97, 131.55 | < 0.0001 | |
| UVB period (Ref: non-synthesizing period) | −68.86 | − 166.88, 29.16 | 0.17 | |
| Gestational weight gain, kg | −4.56 | −12.89, 3.76 | 0.28 | |
| Maternal pre-pregnancy BMI (Ref: < 25 kg/m2) | 124.67 | −5.83, 255.17 | 0.06 | |
| Maternal 25(OH)D (Ref: ≥50 nmol/L) | 55.67 | −82.15, 193.49 | 0.43 | |
| Maternal pre-pregnancy BMI*25(OH)D | 0.04 | |||
| BMI ≥ 25, 25(OH)D < 50 vs BMI ≥ 25, 25(OH)D ≥ 50 | − 164.58 | −384.34, 55.19 | 0.05 | 0.21 |
| BMI ≥ 25, 25(OH)D < 50 vs BMI < 25, 25(OH)D < 50 | −95.57 | − 324.39, 133.24 | 0.28 | 0.70 |
| BMI ≥ 25, 25(OH)D < 50 vs BMI < 25, 25(OH)D ≥ 50 | −39.91 | − 236.31, 156.50 | 0.60 | 0.95 |
| BMI ≥ 25, 25(OH)D ≥ 50 vs BMI < 25, 25(OH)D < 50 | 69.00 | − 140.90, 278.91 | 0.39 | 0.83 |
| BMI ≥ 25, 25(OH)D ≥ 50 vs BMI < 25, 25(OH)D ≥ 50 | 124.67 | −47.01, 296.35 | 0.06 | 0.24 |
| BMI < 25, 25(OH)D < 50 vs BMI < 25, 25(OH)D ≥ 50 | 55.67 | − 125.64, 236.97 | 0.43 | 0.85 |
Abbreviations: 25(OH)D 25-hydroxyvitamin D, BMI Body mass index, UVB Ultraviolet B
aData were compared using a linear fixed effect model for continuous variables followed by post hoc Tukey’s tests with Tukey-Kramer adjustment for multiple comparisons
bSex of infant (male vs. female)
cUVB period (April 1st-October 31st or November 1st-March 31st)
dMaternal pre-pregnancy BMI (BMI < 25 kg/m2 or BMI ≥25 kg/m2)
eMaternal serum 25(OH)D (≥ or < 50 nmol/L)
Fig. 2.
The interaction effect of maternal pre-pregnancy BMI and maternal 25(OH)D status with neonatal body composition. A Whole-body fat mass, B whole-body fat percentage, C fat mass index (FMI), D whole-body lean mass, E whole-body lean percentage, and F lean mass index (LMI). Data were compared using a linear fixed effects model, maternal pre-pregnancy BMI and 25(OH)D interaction as a categorical fixed effect followed by post hoc Tukey’s tests with Tukey-Kramer adjustment for multiple comparisons. Data are mean ± SD
Similar to neonatal indicators of adiposity, maternal pre-pregnancy BMI (< 25 kg/m2 or ≥ 25 kg/m2) and maternal serum 25(OH)D (< 50 or ≥ 50 nmol/L) were not independently linked to lean mass, lean percentage and LMI in the infants (Table 3 and Supplementary Table 4). A significant interaction effect of maternal pre-pregnancy BMI and 25(OH)D concentration was observed for neonatal lean mass (Table 3 and Fig. 2D) and lean percentage (Fig. 2E) but not LMI (Fig. 2F). However, after pairwise comparison tests, only infants of mothers in I-OW/O with BMI ≥25 kg/m2 before pregnancy and vitamin D insufficiency (25(OH)D < 50 nmol/L) had significantly lower lean percentage compared to infants born to mothers in S-OW/O with BMI ≥25 kg/m2 but with vitamin D sufficiency 25(OH)D ≥ 50 nmol/L (P = 0.05) (Supplementary Table 4). Whole body lean mass, percentage lean mass, and LMI were also observed to be higher in male infants versus females (P < 0.05) (Supplemental Fig. 1D, E, F). In addition, only percentage lean mass appeared to be lower in infants born in the vitamin D synthesizing period (UVB period) versus those born in vitamin D non-synthesizing period (P = 0.0482) (Supplemental Table 4). Infant age and length were important correlates of lean mass and percentage lean mass, (Table 3 and Supplemental Table 3).
Discussion
According to our data, healthy neonates dually exposed to insufficient maternal vitamin D status and elevated pre-pregnancy BMI had higher whole body fat mass (∆ 220.4 g, ~ 28% difference) compared to those of mothers with elevated pre-pregnancy BMI yet vitamin D sufficient. Our results are unique in examining maternal adiposity and vitamin D status as dual exposures in programming of neonatal body composition and complement other reports that separately link maternal vitamin D status to adiposity in the neonatal period [14] or in childhood [15, 16], and maternal overweight/obese pre-gravid BMI with neonatal adiposity [8, 41].
Most national guidelines for a healthy pregnancy [42, 43] do not have a specific recommendation on vitamin D supplementation for pregnant women with an elevated BMI; nor their newborn. Although the majority of mothers took multivitamin supplements containing vitamin D during pregnancy, 64.3% of infants of mothers with elevated pre-pregnancy BMI had serum 25(OH)D < 50 nmol/L. If maternal serum 25(OH)D was < 50 nmol/L all infants had 25(OH)D < 50 nmol/L and the majority (71.4%) were vitamin D deficient. These observations reinforce the importance of encouraging overweight/obese women to seek nutrition counselling prior to conception [44] or if pregnant to initiate the consultation and multivitamin supplementation as soon as possible to help establish vitamin D stores in the fetus and ultimately in the newborn [5]. This should be followed by neonatal vitamin D supplementation [27].
This study adds that maternal 25(OH)D concentration is an influential factor in neonatal vitamin D status at birth and within the neonatal period. We observed a positive correlation between maternal and neonatal vitamin D status at birth, which decreases 23.4% through the neonatal period, in line with other reports [45, 46]. This is attributed to the fact that the fetus is fully reliant on maternal-fetal transfer of vitamin D [47], however, shortly after birth the majority of infants commenced routine supplements containing 400 IU/d vitamin D and were breastfed. Overall, the majority of infants at birth (67.6%) had serum 25(OH)D < 50 nmol/L, whereas at the postpartum visit, this declined to 40%. Even though 4 ~ 6 week is not long enough to see a plateau in the response to vitamin D supplementation [48–50], the increments in vitamin D status of infants with 25(OH)D < 50 nmol/L agree with other reports [51, 52]. The response of infants to vitamin D supplementation inversely related to basal status resulting in a greater increment in serum 25(OH)D in neonates with vitamin D deficiency. Thus, 400 IU/d of vitamin D is suitable for healthy term-born infants, even if born with vitamin D deficiency.
Vitamin D status of mothers could be a proxy for other healthy behaviors, reflect quality of diet, or time spent outside. In our study exercise (combined indoor and outdoor) in pregnancy was associated with 14.8 nmol/L higher 25(OH)D concentration in mothers of all BMI ranges. This is of relevance since exercise tends to take place outdoors [53] with a positive association between exercise and time spent outside [54, 55]. Outdoor activity and being more exposed to sunlight promotes vitamin D synthesis and combined with vitamin D mobilization from adipose tissue [56] supports achievement and maintenance of higher vitamin D status. Physical activity is encouraged as part of a healthy lifestyle in pregnancies without contraindications [57].
The genes that regulate fat distribution, adiposity [58], and skeletal muscle phenotypes [59] are responsive to environmental and lifestyle exposures. Achieving appropriate body weight and being physically active preconception and during pregnancy may determine body composition in the offspring [60]. Regular physical activity shifts the increased energy demands to maternal muscle mass and away from the adipocytes of the fetus leading to proportionately increased lean mass and decreased adipose tissue [60]. This pattern is consistent with the observations in our study and is likely due to mobilization of vitamin D from fat tissue into circulation [56]. Therefore, it can be inferred that genetic, behavioral and environmental interactions contribute to variations in fat-muscle partitioning in early development.
In this study, we did not observe an association between maternal vitamin D status and infant lean body mass, potentially due to all infants being born term and AGA. In comparison to infants born AGA, body composition partitioning differs among infants at the extremes of birth weight, small or large for gestational age [61]. Infants born large for gestational age tend to have vitamin D insufficiency [62, 63], possibly due to entrapment of vitamin D in fat tissue compared to AGA. In addition, by including only non-smoking mothers we eliminated smoking as a confounder as neonates born to smoking mothers have lower vitamin D status [64] as well as lower lean body mass [65, 66]. Lack of association between vitamin D and infant lean body mass might also reflect the rapid growth spurt in the first month of life. Furthermore, sex differences in infant body composition emerged early postnatally. This effect was independent of the UVB period at birth, GA, maternal supplement use, and maternal and neonatal vitamin D status. Male infants have more muscle mass due to the anabolic effect of testosterone which temporarily surges postnatally within 1 ~ 3 months postpartum [67, 68]. In contrast, in our study and others, females have greater stores of fat mass irrespective of vitamin D status [69].
Strengths, limitations, and further research
Strengths of this study are inclusion of infants of a diverse ancestry which aids the generalizability of the findings to multi-ethnic populations and assessment of body composition early in postnatal period in mother infant dyads. There are however some limitations including that pre-pregnancy weight was obtained from medical records, some of which could be self-reported. Pre-pregnancy BMI as a proxy for overweight and obesity may underestimate or misclassified adiposity [70], however, the three-compartmental model of DXA confirmed excess adipose tissue. We used an immunoassay to measure total serum 25(OH)D which is not a gold standard technique, nonetheless, the manufacturer (DiaSorin) is certified by the Vitamin D Standardization-Certification Program [71]. Furthermore, we used rigorous quality assurance measures and standardized 25(OH)D concentrations to NIST standard reference materials. Given the design of the study, maternal 25(OH)D concentrations were only measured at the postpartum visit and not at delivery. This may not be a major limitation as maternal 25(OH)D concentrations are not significantly different between 36 weeks of GA and 1 month postpartum [25, 26]. Additionally, our analysis might have been statistically underpowered to detect relationships such as the association between gestational weight gain and maternal or neonatal vitamin D status, future larger studies are needed. Lastly, whether the body composition pattern extends later into childhood requires a longitudinal study.
Conclusion
In otherwise healthy mother-infant dyads, maternal overweight/obesity and serum 25(OH)D < 50 nmol/L are dual exposures that associate with neonatal serum 25(OH)D < 50 nmol/L as well as higher adiposity. More concerning, 71.4% of neonates in this cohort were vitamin D deficient. These results reinforce the importance of postnatal vitamin D supplementation in infants born to mothers with BMI ≥25 kg/m2. In the event of low maternal-fetal transfer of vitamin D, postnatal supplementation with 400 IU/d of vitamin D readily builds vitamin D stores and in doing so may limit the impact of fetal exposures.
Supplementary Information
Additional file 1: Supplemental Table 1. Maternal serum 25(OH)D postpartum based on maternal characteristics. Supplemental Table 2. Neonatal serum 25(OH)D at birth based on maternal characteristics. Supplemental Table 3. Correlates of neonatal body fat mass. Supplemental Table 4. Correlates of neonatal body lean mass. Supplemental Figure 1. Sex dimorphism in neonatal body composition.
Acknowledgments
The authors would like to thank the following people for their invaluable contributions towards the success of this important work: Dr. Sarah Kimmins from McGill University for providing guidance and feedback throughout this project. Dr. Roger Cue from McGill University for his valuable guidance and insightful advice throughout statistical analyses; Sherry Agellon, Paula Lavery, and Maggie Yuan from McGill University for their long-standing assistance in laboratory measurements across the study; Veronique Menard, Erika de Risi, Kristina Mullahoo, Laura Glenn, Sharina Patel, and Zahra Farahnak for their involvement in recruitment at the Lakeshore General Hospital. The authors would also like to particularly thank all the families that participated in this study. At the time of the research, Dr. Hope A. Weiler was funded by a salary award from the Canada Research Chairs Program (2015-2018: 950-230633); Maryam Razaghi and Nathalie Gharibeh received Graduate Excellence Fellowships from McGill University. The funding agencies were not involved in the study design, collection, management, analysis, and interpretation of the data or in the preparation, review, or approval of the manuscript.
Abbreviations
- 25(OH)D
25-hydroxyvitamin D
- AGA
Appropriate for gestational age
- BMI
Body mass index
- UVB
Ultraviolet B
Authors’ contributions
HAW, SW, DM, FR, and GJ designed the study. HAW and CAV supervised the study. HAW, CAV, MR, NG, OFS conducted the study; HAW, CAV, MR, NG, OFS collected the data; MR, NG and OFS performed the laboratory analyses. MR performed statistical analysis; MR wrote the final manuscript with the intellectual aid and comments of HAW. All authors (MR, NG, CAV, OFS, SW, DM, FR, GJ, and HAW) have read and approved the manuscript.
Funding
This research was supported by funding from the Canadian Institutes of Health Research: MOP-142391; Canada Foundation for Innovation: 202820.
Availability of data and materials
The dataset used and/or analysed during the current study will not be made publicly available because permission to share data was not requested at the time of obtaining participant consent but are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
Prior to collection of data and blood samples, parents provided written informed consent. The study was approved by the St. Mary’s Hospital Research Ethics Committee (Montréal, Québec, Canada) that oversees research at the Lakeshore General Hospital. Both ethics approval (REB# 15-34) and trial registration with clinicaltrials.gov (NCT02563015) were completed prior to beginning recruitment. This study was also approved by the Health Canada Research Ethics Board (REB 2019-033H) and Privacy Management Division (HC-PR-2019-000024). All methods were carried out in accordance with relevant guidelines and regulations.
Consent for publication
Not applicable. There are no details, images, or videos related to an individual participant.
Competing interests
The authors state no conflicts of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Bi WG, Nuyt AM, Weiler H, Leduc L, Santamaria C, Wei SQ. Association between vitamin D supplementation during pregnancy and offspring growth, morbidity, and mortality: a systematic review and meta-analysis. JAMA Pediatr. 2018;172(7):635–645. doi: 10.1001/jamapediatrics.2018.0302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Barouki R, Gluckman PD, Grandjean P, Hanson M, Heindel JJ. Developmental origins of non-communicable disease: implications for research and public health. Environ Health. 2012;11(1):42. doi: 10.1186/1476-069X-11-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gluckman PD, Hanson MA, Cooper C, Thornburg KL. Effect of in utero and early-life conditions on adult health and disease. N Engl J Med. 2008;359(1):61–73. doi: 10.1056/NEJMra0708473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ma K, Wei SQ, Bi WG, Weiler HA, Wen SW. Effect of vitamin D supplementation in early life on children’s growth and body composition: a systematic review and meta-analysis of randomized controlled trials. Nutrients. 2021;13(2):524. doi: 10.3390/nu13020524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Josefson JL, Feinglass J, Rademaker AW, Metzger BE, Zeiss DM, Price HE, et al. Maternal obesity and vitamin D sufficiency are associated with cord blood vitamin D insufficiency. J Clin Endocrinol Metab. 2013;98(1):114–119. doi: 10.1210/jc.2012-2882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bodnar LM, Catov JM, Roberts JM, Simhan HN. Prepregnancy obesity predicts poor vitamin D status in mothers and their neonates. J Nutr. 2007;137(11):2437–2442. doi: 10.1093/jn/137.11.2437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pourshahidi LK. Vitamin D and obesity: current perspectives and future directions. Proc Nutr Soc. 2015;74(2):115–124. doi: 10.1017/S0029665114001578. [DOI] [PubMed] [Google Scholar]
- 8.Hull HR, Dinger MK, Knehans AW, Thompson DM, Fields DA. Impact of maternal body mass index on neonate birthweight and body composition. Am J Obstet Gynecol. 2008;198(4):416.e1–416.e6. doi: 10.1016/j.ajog.2007.10.796. [DOI] [PubMed] [Google Scholar]
- 9.Institute of Medicine. Weight gain during pregnancy: reexamining the guidelines. Washington, DC: National Academies Press; 2010. [PubMed]
- 10.Hull HR, Thornton JC, Ji Y, Paley C, Rosenn B, Mathews P, et al. Higher infant body fat with excessive gestational weight gain in overweight women. Am J Obstet Gynecol. 2011;205(3):211.e1–211.e7. doi: 10.1016/j.ajog.2011.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ward LM. Vitamin D deficiency in the 21st century: a persistent problem among Canadian infants and mothers. Can Med Assoc J. 2005;172(6):769–770. doi: 10.1503/cmaj.050177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Weiler HA, Leslie WD, Krahn J, Steiman PW, Metge CJ. Canadian Aboriginal women have a higher prevalence of vitamin D deficiency than non-Aboriginal women despite similar dietary vitamin D intakes. J Nutr. 2007;137(2):461–465. doi: 10.1093/jn/137.2.461. [DOI] [PubMed] [Google Scholar]
- 13.Godel JC, Society CP. First Nations I. Committee MH Vitamin D supplementation: recommendations for Canadian mothers and infants. Paediatr Child Health. 2007;12(7):583–589. [PMC free article] [PubMed] [Google Scholar]
- 14.Tint MT, Chong MF, Aris IM, Godfrey KM, Quah PL, Kapur J, et al. Association between maternal mid-gestation vitamin D status and neonatal abdominal adiposity. Int J Obes. 2018;42(7):1296–1305. doi: 10.1038/s41366-018-0032-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Boyle V, Thorstensen E, Thompson J, McCowan L, Mitchell E, Godfrey K, et al. The relationship between maternal 25-hydroxyvitamin D status in pregnancy and childhood adiposity and allergy: an observational study. Int J Obes. 2017;41(12):1755–1760. doi: 10.1038/ijo.2017.182. [DOI] [PubMed] [Google Scholar]
- 16.Krishnaveni GV, Veena SR, Winder NR, Hill JC, Noonan K, Boucher BJ, et al. Maternal vitamin D status during pregnancy and body composition and cardiovascular risk markers in Indian children: the Mysore Parthenon study. Am J Clin Nutr. 2011;93(3):628–635. doi: 10.3945/ajcn.110.003921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Harvey NC, Moon RJ, Sayer AA, Ntani G, Davies JH, Javaid MK, et al. Maternal antenatal vitamin D status and offspring muscle development: findings from the Southampton women’s survey. J Clin Endocrinol Metab. 2014;99(1):330–337. doi: 10.1210/jc.2013-3241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Weiler HA, Vanstone CA, Razaghi M, Gharibeh N, Patel S, Wei SQ, et al. Disparities in vitamin D status of newborn infants from a diverse sociodemographic population in Montreal, Canada. J Nutr. 2021:nxab344. 10.1093/jn/nxab344 Online ahead of print. PMID: 34612495. [DOI] [PMC free article] [PubMed]
- 19.Arbuckle TE, Wilkins R, Sherman GJ. Birth weight percentiles by gestational age in Canada. Obstet Gynecol. 1993;81(1):39–48. [PubMed] [Google Scholar]
- 20.Nørgård B, Fonager K, Sørensen HT, Olsen J. Birth outcomes of women with celiac disease: a nationwide historical cohort study. Am J Gastroenterol. 1999;94(9):2435–2440. doi: 10.1111/j.1572-0241.1999.01370.x. [DOI] [PubMed] [Google Scholar]
- 21.Stephansson O, Larsson H, Pedersen L, Kieler H, Granath F, Ludvigsson JF, et al. Crohn's disease is a risk factor for preterm birth. Clin Gastroenterol Hepatol. 2010;8(6):509–515. doi: 10.1016/j.cgh.2010.02.014. [DOI] [PubMed] [Google Scholar]
- 22.Vielwerth S, Jensen R, Larsen T, Greisen G. The impact of maternal smoking on fetal and infant growth. Early Hum Dev. 2007;83(8):491–495. doi: 10.1016/j.earlhumdev.2006.09.010. [DOI] [PubMed] [Google Scholar]
- 23.Canadian Institute for Health Information. Proposed standards for race-based and indigenous identity data collection and health reporting in Canada. Ottawa: CIHI; 2020.
- 24.Statistics Canada. Table 11-10-0190-01 Market income, government transfers, total income, income tax and after-tax income by economic family type. 2015-2019. 10.25318/1110019001-eng. Accessed 15 Jul 2021.
- 25.March KM, Chen NN, Karakochuk CD, Shand AW, Innis SM, Von Dadelszen P, et al. Maternal vitamin D3 supplementation at 50 μg/d protects against low serum 25-hydroxyvitamin D in infants at 8 wk of age: a randomized controlled trial of 3 doses of vitamin D beginning in gestation and continued in lactation. Am J Clin Nutr. 2015;102(2):402–410. doi: 10.3945/ajcn.114.106385. [DOI] [PubMed] [Google Scholar]
- 26.Thiele DK, Ralph J, El-Masri M, Anderson CM. Vitamin D3 supplementation during pregnancy and lactation improves vitamin D status of the mother–infant dyad. J Obstet Gynecol Neonatal Nurs. 2017;46(1):135–147. doi: 10.1016/j.jogn.2016.02.016. [DOI] [PubMed] [Google Scholar]
- 27.Institute of Medicine. Dietary reference intakes for calcium and vitamin D. Washington, DC: The National Academies Press; 2011. [PubMed]
- 28.Hazell T, Gallo S, Vanstone C, Agellon S, Rodd C, Weiler H. Vitamin D supplementation trial in infancy: body composition effects at 3 years of age in a prospective follow-up study from Montreal. Pediatr Obes. 2017;12(1):38–47. doi: 10.1111/ijpo.12105. [DOI] [PubMed] [Google Scholar]
- 29.Chardon A, Cretois I, Hourseau C. Skin colour typology and suntanning pathways. Int J Cosmet Sci. 1991;13(4):191–208. doi: 10.1111/j.1467-2494.1991.tb00561.x. [DOI] [PubMed] [Google Scholar]
- 30.Del Bino S, Sok J, Bessac E, Bernerd F. Relationship between skin response to ultraviolet exposure and skin color type. Pigment Cell Res. 2006;19(6):606–614. doi: 10.1111/j.1600-0749.2006.00338.x. [DOI] [PubMed] [Google Scholar]
- 31.Reeder AI, Hammond VA, Gray AR. Questionnaire items to assess skin color and erythemal sensitivity: reliability, validity, and “the dark shift”. Cancer Epidemiol Biomark Prev. 2010;19(5):1167–1173. doi: 10.1158/1055-9965.EPI-09-1300. [DOI] [PubMed] [Google Scholar]
- 32.Webb AR, Kline L, Holick MF. Influence of season and latitude on the cutaneous synthesis of vitamin D3: exposure to winter sunlight in Boston and Edmonton will not promote vitamin D3 synthesis in human skin. J Clin Endocrinol Metab. 1988;67(2):373–378. doi: 10.1210/jcem-67-2-373. [DOI] [PubMed] [Google Scholar]
- 33.Gharibeh N, Razaghi M, Vanstone CA, Wei S, McNally D, Rauch F, et al. Maternal vitamin D status and gestational weight gain as correlates of neonatal bone mass in healthy term breastfed young infants from Montreal, Canada. Nutrients. 2021;13(12):4189. doi: 10.3390/nu13124189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Widen EM, Factor-Litvak PR, Gallagher D, Paxton A, Pierson RN, Heymsfield SB, et al. The pattern of gestational weight gain is associated with changes in maternal body composition and neonatal size. Matern Child Health J. 2015;19(10):2286–2294. doi: 10.1007/s10995-015-1747-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Razaghi M, Gharibeh N, Vanstone CA, Sotunde OF, Khamessan A, Wei SQ, et al. Correction of neonatal vitamin D status using 1000 IU/d of vitamin D increased lean body mass by 12 months of age compared to 400 IU/d: a randomized controlled trial. Am J Clin Nutr. 2022. In press. [DOI] [PMC free article] [PubMed]
- 36.Kononoff PJ, Hanford KJ. Estimating statistical power of mixed models used in dairy nutrition experiments. J Dairy Sci. 2006;89(10):3968–3971. doi: 10.3168/jds.S0022-0302(06)72439-0. [DOI] [PubMed] [Google Scholar]
- 37.Roy J. SAS for mixed models. 2nd ed. Cary, NC: Taylor & Francis, SAS Institute Inc; 2007.
- 38.VanderWeele TJ. Principles of confounder selection. Eur J Epidemiol. 2019;34(3):211–219. doi: 10.1007/s10654-019-00494-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Harvey NC, Poole J, Javaid MK, Dennison EM, Robinson S, Inskip HM, et al. Parental determinants of neonatal body composition. J Clin Endocrinol Metab. 2007;92(2):523–526. doi: 10.1210/jc.2006-0456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kelly TL. Technical considerations in advanced clinical body composition evaluation. Age. 2009;40(41):42. [Google Scholar]
- 41.Sewell MF, Huston-Presley L, Super DM, Catalano P. Increased neonatal fat mass, not lean body mass, is associated with maternal obesity. Am J Obstet Gynecol. 2006;195(4):1100–1103. doi: 10.1016/j.ajog.2006.06.014. [DOI] [PubMed] [Google Scholar]
- 42.Vitner D, Harris K, Maxwell C, Farine D. Obesity in pregnancy: a comparison of four national guidelines. J Matern Fetal Neonatal Med. 2019;32(15):2580–2590. doi: 10.1080/14767058.2018.1440546. [DOI] [PubMed] [Google Scholar]
- 43.Hynes C, Jesurasa A, Evans P, Mitchell C. Vitamin D supplementation for women before and during pregnancy: an update of the guidelines, evidence, and role of GPs and practice nurses. Br J Gen Pract. 2017;67(662):423–424. doi: 10.3399/bjgp17X692489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Green-Raleigh K, Lawrence JM, Chen H, Devine O, Prue C. Pregnancy planning status and health behaviors among nonpregnant women in a California managed health care organization. Perspect Sex Reprod Health. 2005;37(4):179–183. doi: 10.1363/psrh.37.179.05. [DOI] [PubMed] [Google Scholar]
- 45.Maghbooli Z, Hossein-Nezhad A, Shafaei AR, Karimi F, Madani FS, Larijani B. Vitamin D status in mothers and their newborns in Iran. BMC Pregnancy Childbirth. 2007;7(1):1. doi: 10.1186/1471-2393-7-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Saraf R, Morton SM, Camargo CA, Jr, Grant CC. Global summary of maternal and newborn vitamin D status - a systematic review. Matern Child Nutr. 2016;12(4):647–668. doi: 10.1111/mcn.12210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Dawodu A, Tsang RC. Maternal vitamin D status: effect on milk vitamin D content and vitamin D status of breastfeeding infants. Adv Nutr. 2012;3(3):353–361. doi: 10.3945/an.111.000950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gallo S, Comeau K, Vanstone C, Agellon S, Sharma A, Jones G, et al. Effect of different dosages of oral vitamin D supplementation on vitamin D status in healthy, breastfed infants: a randomized trial. J Am Med Assoc. 2013;309(17):1785–1792. doi: 10.1001/jama.2013.3404. [DOI] [PubMed] [Google Scholar]
- 49.Harris Susan SS. Plasma vitamin D and 25OHD responses of young and old men to supplementation with vitamin D3. J Am Coll Nutr. 2002;21(4):357–362. doi: 10.1080/07315724.2002.10719235. [DOI] [PubMed] [Google Scholar]
- 50.Holick MF, Biancuzzo RM, Chen TC, Klein EK, Young A, Bibuld D, et al. Vitamin D2 is as effective as vitamin D3 in maintaining circulating concentrations of 25-hydroxyvitamin D. J Clin Endocrinol Metab. 2008;93(3):677–681. doi: 10.1210/jc.2007-2308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gallo S, Phan A, Vanstone CA, Rodd C, Weiler HA. The change in plasma 25-hydroxyvitamin D did not differ between breast-fed infants that received a daily supplement of ergocalciferol or cholecalciferol for 3 months. J Nutr. 2013;143(2):148–153. doi: 10.3945/jn.112.167858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zittermann A, Pilz S, Berthold HK. Serum 25-hydroxyvitamin D response to vitamin D supplementation in infants: a systematic review and meta-analysis of clinical intervention trials. Eur J Nutr. 2020;59(1):359–369. doi: 10.1007/s00394-019-01912-x. [DOI] [PubMed] [Google Scholar]
- 53.Shanahan DF, Franco L, Lin BB, Gaston KJ, Fuller RA. The benefits of natural environments for physical activity. Sports Med. 2016;46(7):989–995. doi: 10.1007/s40279-016-0502-4. [DOI] [PubMed] [Google Scholar]
- 54.Bélanger M, Gallant F, Doré I, O'Loughlin JL, Sylvestre M-P, Abi Nader P, et al. Physical activity mediates the relationship between outdoor time and mental health. Prev Med Rep. 2019;16:101006. doi: 10.1016/j.pmedr.2019.101006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gray C, Gibbons R, Larouche R, Sandseter EBH, Bienenstock A, Brussoni M, et al. What is the relationship between outdoor time and physical activity, sedentary behaviour, and physical fitness in children? A systematic review. Int J Environ Res Public Health. 2015;12(6):6455–6474. doi: 10.3390/ijerph120606455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hengist A, Perkin O, Gonzalez J, Betts J, Hewison M, Manolopoulos K, et al. Mobilising vitamin D from adipose tissue: the potential impact of exercise. Nutr Bull. 2019;44(1):25–35. doi: 10.1111/nbu.12369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Davies GA, Wolfe LA, Mottola MF, MacKinnon C, Arsenault MY, Bartellas E, et al. Exercise in pregnancy and the postpartum period. J Obstet Gynaecol Can. 2003;25(6):516–529. doi: 10.1016/s1701-2163(16)30313-9. [DOI] [PubMed] [Google Scholar]
- 58.Fernandez J, Klimentidis YC, Dulin-Keita A, Casazza K. Genetic influences in childhood obesity: recent progress and recommendations for experimental designs. Int J Obes. 2012;36(4):479–484. doi: 10.1038/ijo.2011.236. [DOI] [PubMed] [Google Scholar]
- 59.Roth SM. Genetic aspects of skeletal muscle strength and mass with relevance to sarcopenia. BoneKEy Rep. 2012;1:58. [DOI] [PMC free article] [PubMed]
- 60.Archer E. The childhood obesity epidemic as a result of nongenetic evolution: the maternal resources hypothesis. Mayo Clin Proc. 2015;90(1):77–92. [DOI] [PMC free article] [PubMed]
- 61.Larsson A, Ottosson P, Törnqvist C, Olhager E. Body composition and growth in full-term small for gestational age and large for gestational age Swedish infants assessed with air displacement plethysmography at birth and at 3-4 months of age. PLoS One. 2019;14(5):e0207978. doi: 10.1371/journal.pone.0207978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yilmaz S, Aktulay A, Demirtas C, Engin-Ustun Y. Low cord blood serum levels of vitamin D: cause or effect of fetal macrosomia? Clin Exp Obstet Gynecol. 2015;42(4):501–504. [PubMed] [Google Scholar]
- 63.Wen J, Kang C, Wang J, Cui X, Hong Q, Wang X, et al. Association of maternal serum 25-hydroxyvitamin D concentrations in second and third trimester with risk of macrosomia. Sci Rep. 2018;8(1):6169. doi: 10.1038/s41598-018-24534-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kiely M, O’Donovan SM, Kenny LC, Hourihane JOB, Irvine AD, Murray DM. Vitamin D metabolite concentrations in umbilical cord blood serum and associations with clinical characteristics in a large prospective mother-infant cohort in Ireland. J Steroid Biochem Mol Biol. 2017;167:162–168. doi: 10.1016/j.jsbmb.2016.12.006. [DOI] [PubMed] [Google Scholar]
- 65.Bernstein IM, Plociennik K, Stahle S, Badger GJ, Secker-Walker R. Impact of maternal cigarette smoking on fetal growth and body composition. Am J Obstet Gynecol. 2000;183(4):883–886. doi: 10.1067/mob.2000.109103. [DOI] [PubMed] [Google Scholar]
- 66.Samper M, Jiménez-Muro A, Nerín I, Marqueta A, Ventura P, Rodríguez G. Maternal active smoking and newborn body composition. Early Hum Dev. 2012;88(3):141–145. doi: 10.1016/j.earlhumdev.2011.07.015. [DOI] [PubMed] [Google Scholar]
- 67.Rey RA. Mini-puberty and true puberty: differences in testicular function. Ann Endocrinol. 2014;75(2):58–63. doi: 10.1016/j.ando.2014.03.001. [DOI] [PubMed] [Google Scholar]
- 68.Kuiri-Hänninen TT. Activation of the hypothalamic-pituitary-gonadal axis in infancy: minipuberty. Horm Res Paediatr. 2014;82(2):73–80. doi: 10.1159/000362414. [DOI] [PubMed] [Google Scholar]
- 69.Fields DA, Krishnan S, Wisniewski AB. Sex differences in body composition early in life. Gend Med. 2009;6(2):369–375. doi: 10.1016/j.genm.2009.07.003. [DOI] [PubMed] [Google Scholar]
- 70.Shah NR, Braverman ER. Measuring adiposity in patients: the utility of body mass index (BMI), percent body fat, and leptin. PLoS One. 2012;7(4):e33308. doi: 10.1371/journal.pone.0033308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.DiaSorin Inc . LIAISON® 25 OH Vitamin D TOTAL Assay. 2012. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Supplemental Table 1. Maternal serum 25(OH)D postpartum based on maternal characteristics. Supplemental Table 2. Neonatal serum 25(OH)D at birth based on maternal characteristics. Supplemental Table 3. Correlates of neonatal body fat mass. Supplemental Table 4. Correlates of neonatal body lean mass. Supplemental Figure 1. Sex dimorphism in neonatal body composition.
Data Availability Statement
The dataset used and/or analysed during the current study will not be made publicly available because permission to share data was not requested at the time of obtaining participant consent but are available from the corresponding author on reasonable request.