Abstract
Memory T cells are crucial players in vertebrate adaptive immunity but their development is incompletely understood. Here, we describe a method to produce human memory-like T cells from naive human T cells in culture. Using commercially available human T-cell differentiation kits, both purified naive CD8+ T cells and purified naive CD4+ T cells were activated via T-cell receptor signaling and appropriate cytokines for several days in culture. All the T-cell activators were then removed from the medium and the cultures were continued in hypoxic condition (1% O2 atmosphere) for several more days; during this period, most of the cells died, but some survived in a quiescent state for a month. The survivors had small round cell bodies, expressed differentiation markers characteristic of memory T cells and restarted proliferation when the T-cell activators were added back. We could also induce memory-like T cells from naive human T cells without hypoxia, if we froze the activated T cells or prepared the naive T cells from chilled filter buffy coats.
Keywords: memory T cell, ex vivo differentiation, naive T cell, CD4, CD8
Pre-activated human naïve CD4+ or CD8+ T cells were cultured in activator-free medium to induce cell death for 5 to 10 days. Although no cell survived in normal oxygen condition (20% O2), 2 to 30% of the activated T cells could survive in hypoxia (1% O2) and became memory-like T cells. We named this simple procedure as ‘the Death Assay’.
Graphical Abstract
Graphical Abstract.
Introduction
Human T lymphocytes (both CD8+ T cells and CD4+ T cells) can give rise to have four types of memory T cells; stem cell memory T cells (TSCM cells), central memory T cells (TCM cells), effector memory T cells (TEM cells), and tissue-resident memory T cells (TRM cells) [1]. Although these memory T cells are the crucial for vertebrate adaptive immunity, the cell and molecular mechanisms by which they are produced are not well understood. Because memory T cells can persist in a quiescent state for several decades, other group gave an opinion that they resemble a form of adult somatic stem cells [2].
In a previous report, we studied the differentiation of adult rodent oligodendrocyte precursor cells (adult OPCs) in culture [3], which also share features with quiescent adult somatic stem cells [4]. Oligodendrocytes are myelinating cells in the central nervous system (CNS). And adult OPCs are conserved in CNS for life-long time in a quiescent state. When myelin sheath is destroyed, adult OPCs start proliferation and differentiation into oligodendrocytes to repair the damaged region. Actively proliferating perinatal OPCs purified from postnatal day 7 rodent optic nerves, when cultured in the serum-free medium containing platelet-derived growth factor and thyroid hormone in 20% O2, all stop proliferating and differentiate into oligodendrocytes after a limited number of cell divisions [5]. In the same conditions, however, but in 1–1.5% O2, only about half the cells became oligodendrocytes, while the rest became quiescent adult-like OPCs [3].
Antibody-based adaptive immune system and myelin sheath of axon are the unique components of jawed vertebrates (Gnathostomata) [6, 7]. They are thought to have evolved at the same stage. Although both memory T cells and adult OPCs share many features with quiescent adult somatic stem cells, because of their plenty of number (in human peripheral blood, 1–7% of the leukocytes are memory CD8+ T cells and 3–11% of them are memory CD4+ T cells [1], and 3–8% of nucleated cells in human CNS are adult OPCs [4]), they have not been classified as somatic stem cells by the classical definition [8]. These common properties between memory T cells and adult OPCs raised the possibility that a hypoxic environment might also promote the development of memory T cells. To test this possibility, we tried to induce human naive T cells to differentiate into memory T cells in hypoxic culture conditions.
Methods
Ethical review and informed consent
Preparation of peripheral blood mononuclear cells (PBMCs) from healthy donors was included in the study approved by the institutional review board of the Saitama Medical University (permission number 906; UMIN000038311). Written informed consent was obtained in accordance with the Declaration of Helsinki.
Purification of human naive T lymphocytes from peripheral blood
Human PBMCs were prepared from whole blood of healthy donors by Ficoll density gradient centrifugation using Ficoll-Paque PLUS (#17-1440-02, GE Healthcare) and SepMate-50 tubes (#86450, STEMCELL Technologies) [9, 10]. For the purification of human naive CD8+ T cells, an EasySep human naive CD8+ T-cell isolation kit II (#17928, STEMCELL Technologies) was used. The purity of naive CD8+ T cell was up to 97%. And for the purification of human naive CD4+ T cells, an EasySep human naive CD4+ T cell isolation kit II (#17555, STEMCELL Technologies) was used. The purification procedures were carried out according to the supplier’s instructions. The purity of naive CD4+ T cell was up to 98%.
Purification of human naive T lymphocytes from Filter Buffy Coats (FBC)
Used leukocyte depletion filters were back-flushed by 50 ml of PBS (#14190-144, Gibco)/2% FBS (#173012, Sigma-Aldrich)/2 mM EDTA (#15575-020, Invitrogen) twice [11]. Total 100 ml of eluted fluid was centrifuged 300 g for 8 min at 20°C. Upper cell-free phase was discarded by aspiration. Blood cell suspensions were segregated by Ficoll density gradient centrifugation to prepare PBMCs. An EasySep human naive CD8+ T-cell isolation kit II and an EasySep human naive CD4+ T cell isolation kit II were used for the purification of naive CD8+ T cells and naive CD4+ T cells.
Culture of human T lymphocytes
RPMI 1640/10% FBS/2-Me medium (for 50 ml; Advanced RPMI 1640 medium (#12633-012, Gibco) 44.5 ml, heat-inactivated FBS (#173012, Sigma-Aldrich) 5 ml, x100 PSG (#10378-016, Gibco) 0.5 ml, 50 mM 2-Me (#21438–82, Nacalai Tesque) 0.05 ml) was used as the basal medium.
CD8+ T-cell activation
Purified human naive CD8+ cells were suspended with human CD8+ T-cell activation medium (50 μl of Dynabeads human T-activator CD3/CD28 (#11131D, Gibco) and 50 μl of IL-2 (10 μg/ml, #202-IL, R&D Systems) were added to 50 ml of the basal medium) at density of 1.0 × 105 cells/ml. Cells were inoculated in non-coated 24-well microplate (1 ml/well, #1820-024, IWAKI). Cells were cultured in 5% CO2 at 37°C for 7–15 days.
CD4+ Th1 differentiation
CD4+ Th1 cell differentiation was induced by using CellXVivo human Th1 cell differentiation kit (#CDK001, R&D Systems). Purified human naive CD4+ cells were suspended with human Th1 differentiation medium (the basal medium containing Human Th1 reagent 1 and 2) at density of 1.0 × 105 cells/ml. Cells were inoculated in mouse anti-human CD3 antibody (included in the kit) coated 24-well microplate (1 ml/well). Cells were cultured in 5% CO2 at 37°C for 7–15 days.
CD4+ Th2 differentiation
CD4+ Th2 cell differentiation was induced by using CellXVivo human Th2 cell differentiation kit (#CDK002, R&D Systems). Purified human naive CD4+ cells were suspended with human Th2 differentiation medium (the basal medium containing Human Th2 reagent 1~4) at density of 1.0 × 105 cells/ml. Cells were inoculated in mouse anti-human CD3 antibody (included in the kit) coated 24-well microplate (1 ml/well). Cells were cultured in 5% CO2 at 37°C for 8–17 days.
The Death Assay for activated CD8+ T cells
Activated CD8+ T cells were harvested and suspended with 2.5 ml of PBS/2% FBS/1 mM EDTA. To strip away Dynabeads human T-activator CD3/CD28, after 20 times of pipetting, cells were transferred to a 5 ml round-bottom tube (#720028, Corning). The tube was set on the EasySep magnet (#18000, STEMCELL Technologies) for 3 min. After then cells were transferred to a new tube and harvested again. These cells were re-suspended with the basal medium at density of 2.0 × 105 cells/ml. Cultures were carried out in non-coated 50 ml tissue culture flasks (5 ml/flask, #353108, Corning). Cell cultures were continued in 1% O2 or 20% O2, with 5% CO2 at 37°C for in a multigas incubator MCO-5M-PJ (PHC Holdings Corp.).
The Death Assay for activated CD4+ T cells
CD4+ T cells in either Th1 differentiation medium or Th2 differentiation medium were harvested and washed with PBS/2% FBS, then they were re-suspended with the basal medium at density of 2.0 × 105 cells/ml. Cultures were carried out in non-coated 50 ml tissue culture flasks (5 ml/flask). Cell cultures were continued in 1% O2 or 20% O2, with 5% CO2 at 37°C in a multigas incubator MCO-5M-PJ.
Carboxyfluorescein succinimidyl ester (CFSE) labeling
CFSE labeling was carried out by using a CFSE Cell Division Assay Kit (#10009853, Cayman Chemical Company). In short, naive CD8+ T cells were activated and induced to differentiate for 8 days. After that, to synchronize their cell cycles, cells were cultured in activator free medium overnight. Cells were washed twice with PBS/2% FBS. 2.0 × 107 cells were suspended with 0.5 ml of pre-warmed PBS. After then 0.5 ml of 2× CFSE staining solution was added. Cells were incubated in a water bath at 37°C for 15 min. To stop the reaction, 5 ml of basal medium added to the cells. Cells were washed three times with 10 ml of basal medium, then they were assessed by the Death Assay.
Fluorescent-activated cell sorting (FACS) analysis
Cells were washed twice with PBS/2% FBS. After then cells were suspended with PBS/2% FBS (1.0 × 107 cells/ml). For 0.1 ml of cell suspensions, 5 μl of FITC conjugated mouse anti-human CD45RA monoclonal antibody (200 μg/ml, HI100, #11-0458-42, Invitrogen), 5 μl of PE-conjugated mouse anti-human CD62L monoclonal antibody (200 μg/ml, DREG-56, #12-0629-42, Invitrogen), 5 μl of PE-Cyanine7-conjugated mouse anti-human CD127 monoclonal antibody (200 μg/ml, eBioRDR5, #25-1278-42, Invitrogen), and 7 μl of 7-AAD (50 μg/ml, #559925, BD Biosciences) were added. Cells were stored on ice, in dark, for 20 min. Then, cells were washed once with 5 ml of PBS/2% FBS and re-suspended with 0.5 ml of PBS/2% FBS. FACS analysis was carried out using FACSCanto II (Becton Dickinson). Data analysis was done using FACSDiva ver. 6.1 (Becton Dickinson). The gate including cells that are not stained by the 7-AAD was set to incorporate only viable cells for analysis. For fluorescence analysis of multiplex-stained cells, fluorescence signals were compensated based on signal intensity of single-stained cells for each fluorescent dye.
FACS-sorting
CD8+ T cells cultured in basal medium for 27 days in 1% O2 condition were harvested and rinsed with PBS/2% FBS. Cells were suspended in 50 μl of PBS/2% FBS. After the addition of 7 μl of 7-AAD (50 μg/ml), the cell suspension was stored on ice for 20 min in dark. Cells were rinsed with PBS/2% FBS and suspended with 0.5 ml of PBS/2% FBS. FACS-sorting was carried out using FACSAria II (Becton Dickinson). The group of viable cells, which were not stained by 7-AAD were sorted. The total number of the cells of which we have sorted was 1.8 × 104 cells. Each 9.0 × 103 cells were suspended with 0.2 ml of medium (for one group human CD8+ T-cell activation medium was used, for the other group basal medium was used) and seeded in a well of the 96-well plate (#167008, Thermo Fisher Scientific).
Results
We first tried to induce memory CD8+ T cells. In the study where we cultured of perinatal OPCs in hypoxic conditions (1–1.5% O2) terminally differentiated oligodendrocytes and adult-like OPCs appeared at the same time [3]. If a similar phenomenon occurred with CD8+ T cells, both terminally differentiated effector CD8+ T cells and memory CD8+ T cells might develop together when under hypoxic conditions. However, when human naive CD8+ T cells (Supplementary Fig. 1A) prepared from healthy donors (20–21 years old; 20–21 years) were cultured in human T-activator CD3/CD28 and IL-2 containing medium for 8 days in either 20% O2 or 1% O2, there was not so many differences in the proportion of activated T cells that developed when assessed by FACS analysis (Fig. 1). Of note, the down regulation of CD127 was faster in 1% O2 (27.8%) than in 20% O2 (48.7%). It might reflect the accelerated differentiation of CD8+ T cells in hypoxic culture [12].
Fig. 1.
Activated CD8+ T cells in either normal or hypoxic culture. Naive CD8+ T cells derived from a healthy donor were cultured in human T-activator CD3/CD28 and IL-2 containing medium for 8 days. (A) in 20% O2 condition and (B) in 1% O2 condition. The 7-AAD-negative cells in the area gated as P1 in the FSC/SSC panel were considered as living cells. The expression pattern of CD45RA, CD62L, and CD127 of living cells were analyzed by FACS. The numbers on the FACS-plot panel mean the frequencies (%) of population of cells. We repeated this experiment four times.
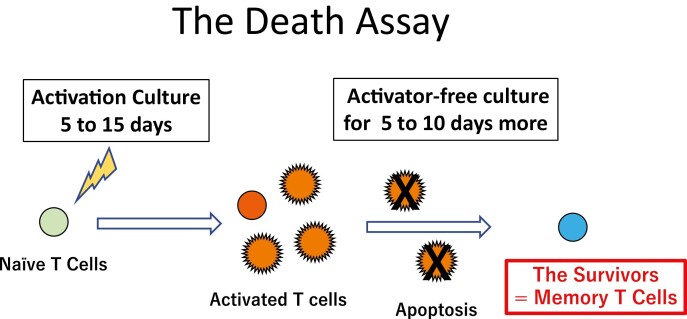
To determine whether there were any memory CD8+ T cells in our cultures, we withdrew all T-cell activators from the medium and the cultures were continued for 8 more days in either 20% O2 or 1% O2. This depletion of both antigenic stimulation and activating cytokines should induce the death of the effector T cells, so we considered the surviving cells to be memory CD8+ T cells [13]. We therefore called this screening procedure the Death Assay (Fig. 2). Although all activated CD8+ T cells died in the Death Assay in 20% O2 (Supplementary Fig. 2), several activated CD8+ T cells (4.2% of the cells preactivated in 20% O2 and 14.7% of the cells preactivated in 1% O2) could survive in the Death Assay in 1% O2 (Fig. 3). The pattern of forward scatter (FSC) and side scatter (SSC) showed that the surviving cells shrank in size and had a decreased number of cytoplasmic granules. During the Death Assay in 1% O2, the survivors acquired the human memory T cell marker CD127 (92.4% of the survivors of the cells preactivated in 20% O2 and 84.6% of the survivors of the cells preactivated in 1% O2) [14, 15]. The majority of the CD127+ cells were CD45RA+/CD62L+ TSCM cells (73.3% of the survivors of the cells preactivated in 20% O2 and 74.0% of the survivors of the cells preactivated in 1% O2). CD45RA-/ CD62L+ TCM cells (20.7% of the survivors of the cells preactivated in 20% O2 and 20.5% of the survivors of the cells preactivated in 1% O2) and CD45RA-/CD62L- TEM cells (2.5% of the survivors of the cells preactivated in 20% O2 and 2.4% of the survivors of the cells preactivated in 1% O2) also existed.
Fig. 2.
The scheme of ‘the Death Assay’. Naive T cells activated for 5–15 days were transferred into activator-free medium and cultured for 5–10 days more in ether 20% or 1% O2 culture condition. Starvation of activating signals induces the cell death of activated T cell. The survivors after this procedure might be the memory T cells.
Fig. 3.
Activated CD8+ T cells survived in 8 days of the Death Assay in hypoxia. Naive CD8+ T cells activated for 8 days were transferred into activator-free medium and cultured for 8 days more in 1% O2 condition. (A) cells preactivated in 20% O2 condition and (B) cells preactivated in 1% O2 condition. The 7-AAD-negative cells in the area gated as P1 in the FSC/SSC panel were considered as living cells. TSCM; Stem cell memory T cell, TCM; Central memory T cell, TEM; Effector memory T cell. Arrows showed the expected positions of each memory T cell. The numbers on the FACS-plot panel mean the frequencies (%) of population of cells. We repeated this experiment three times.
If these survivors had differentiated into CD8+ memory T cells, they had to have stopped dividing. To confirm their quiescence, a CFSE cell division assay was carried out [16]. After the 8 days of activation, CD8+ T cells were stained with CSFE and divided into two groups. One group was cultured in activator-free medium for the Death Assay in 1% O2; the other was cultured in activation medium as a control. As shown in Fig. 4, whereas the control cells divided seven to eight times during 6 days of culture, the survivors of the Death Assay in 1% O2 divided only once during the same period.
Fig. 4.
CD8+ T cells in the Death Assay in hypoxia were quiescent. Naive CD8+ T cells were cultured in activating medium in 20% O2 condition for 8 days. Cells were stained with CFSE and divided into two groups. One group was transferred into activator-free medium and the Death Assay was carried out in 1% O2 condition. The other group continued activation culture in 1% O2 condition as a control. FACS analysis was carried out on day 1 and day 6, and the number of cell division was estimated by the counts of CFSE stain. The scale on the vertical axis is an integer and the scale on the horizontal axis is logarithmic. The experiment was carried out one time.
These quiescent survivors could be maintained in the same condition of the Death Assay in 1% O2 for a month (Supplementary Fig. 3). On day 27 in the Death Assay in 1% O2, 0.8% of the cells were still alive. To determine whether these long-lived cells could be reactivated, we FACS-sorted the survivors and divided them into two groups. One group continued the Death Assay in 1% O2 for 6 more days, and the other was cultured in human T-activator CD3/CD28 and IL-2 containing medium for 6 days in 1% O2. On day 33 of the Death Assay in 1% O2, the quiescent cells were small and round (Fig. 5A). On day 6 of the reactivation, the cells had enlarged and become irregular in shape—resembling blast cells; they also had increased their number (Fig. 5B). These observations suggested that the surviving CD8+ T cells, after the long days of the Death Assay in 1% O2 fulfilled the minimum criteria of memory CD8+ T cells in culture.
Fig. 5.
Long-term living CD8+ T cells in the Death Assay in hypoxia could be reactivated. Naive CD8+ T cells were cultured in activating medium in 20% O2 condition for 7 days. Then cells were transferred into activator-free medium and the Death Assay was carried out in 1% O2 condition. On day 27 in the Death Assay in hypoxia, living cells were FACS-sorted. Cells were divided into two groups. Each 9.0 × 103 cells were suspended with 0.2 ml of medium and seeded in a 96-well plate. One group continued the Death Assay in hypoxia for 6 days more. And the others were cultured in the activation medium for 6 days. Reactivation cultures were carried out in 1% O2 condition, too. The phase-contrast microscopic images were shown in (A) the Death Assay in hypoxia on day 33, in (B) the reactivation culture on day 6. Black bars mean 100 μm. The experiment was carried out one time.
We then used a similar approach to induce memory CD4+ T cells [17]. We cultured purified naive CD4+ T cells (Supplementary Fig. 1B) from healthy donors (20–21 years) in either Th1 differentiation medium (containing IL-12, INF-γ) or Th2 differentiation medium (containing IL-2, IL-4, IL-7, and TSLP) in anti-human CD3 antibody-coated wells. Activation to induce differentiation was carried out in either 20% O2 or 1% O2 (8 days for CD4+ Th1 culture or 10 days for CD4+ Th2 culture). The cells were then transferred to non-coated culture flasks and cultured in the activator-free medium for the Death Assay. As anticipated, after 7 days, there were no survivors in 20% O2 (CD4+ Th1; Supplementary Fig. 4, CD4+ Th2; Supplementary Fig. 5). By contrast, both CD4+ Th1 cells (10.9% of the cells preactivated in 20% O2 and 45.1% of the cells preactivated in 1% O2) and CD4+ Th2 cells (27.1% of the cells preactivated in 20% O2 and 32.6% of the cells preactivated in 1% O2) survived after 7 days of the Death Assay in 1% O2 (Figs 6 and 7). Similar to CD8+ T cells, during the Death Assay in 1% O2 the CD4+ T cells decreased in size, had a reduced number of cytoplasmic granules, and increased their expression of CD127. The majority of the surviving CD4+ T cells had CD45RA-/ CD62L+/CD127+ phenotype, similar to TCM cells, and they also could persist in a quiescent state for a month during the Death Assay in 1% O2 (Supplementary Fig. 6).
Fig. 6.
CD4+ Th1 cells survived in the Death Assay in hypoxia. Naive CD4+ T cells prepared from healthy donors were activated in CD4+ Th1 differentiation medium. (A) Cells activated in CD4+ Th1 differentiation medium for 8 days in 1% O2 condition. (B) After the 8 days of activation culture, CD4+ Th1 cells were transferred into the activator-free medium and the Death Assay was carried out for 7 days in 1% O2 condition. FACS analysis was carried out according to the method for CD8+ T cells. Arrows showed the expected positions of each memory T cell. The 7-AAD-negative cells in the area gated as P1 in the FSC/SSC panel were considered as living cells. 93.9% of the survived CD4+ Th1 was CD127 positive. The survivors were, TCM; 73.5%, TEM; 10.5%, TSCM; 15.4%. The numbers on the FACS-plot panel mean the frequencies (%) of population of cells.
Fig. 7.
CD4+ Th2 cells survived in the Death Assay in hypoxia. Naive CD4+ T cells prepared from healthy donors were activated in CD4+ Th2 differentiation medium. (A) Cells activated in CD4+ Th2 differentiation medium for 10 days in 1% O2 condition. (B) After the 10 days of activation culture, CD4+ Th2 cells were transferred into the activator-free medium and the Death Assay was carried out for 7 days in 1% O2 condition. FACS analysis was carried out according to the method for CD8+ T cells. The 7-AAD-negative cells in the area gated as P1 in the FSC/SSC panel were considered as living cells. 96.6% of the survived CD4+ Th2 was CD127 positive. The survivors were, TCM; 93.9%, TEM; 1.4%, TSCM; 4.6%. The numbers on the FACS-plot panel mean the frequencies (%) of population of cells. We repeated this experiment twice.
Taken together, these results showed that the Death Assay in hypoxia could be used as a general method to induce memory-like T cells from naive T cells, but was hypoxia an essential requirement or was it just a nonspecific stressor? On the contrary to the starting hypothesis, a hypoxic environment was not an essential requirement to induce memory-like T cells, because there were a few exceptions. The first exception was freezing the activated T cells before the Death Assay. Naive CD8+ T cells and naive CD4+ T cells were prepared from healthy donors and activated to induce differentiation as before, we then froze the cells at −80°C and stored them at this temperature overnight. The frozen cells were defrosted, washed and re-inoculated in the activator-free medium in either 20% O2 or 1% O2 for 7 days. As shown in Fig. 8, the results were surprising: 20.2% of CD8+ T cells and 2.3% of CD4+ Th2 cells in 20% O2; in 1% O2, 36.0% of CD8+ T cells and 27.1% of CD4+ Th2 cells survived. Moreover, the cell sizes, shapes, and CD marker expressions of the survivors in 20% O2 were same as those of the survivors in the Death Assay in hypoxia. We tested three kinds of cell freezing buffers—90% FBS/10% DMSO, Bambanker (serum-free) and Cell Reservoir One (serum-free, DMSO-free)—and all of them worked similarly. Thus, it seemed that freezing, like hypoxia, was sufficient to induce naive T cells to develop into memory-like T cells.
Fig. 8.
Frozen activated-T cells could survive in the Death Assay in 20% O2. (A) Naive CD8+ T cells prepared from healthy donors were activated for 8 days in 20% O2 condition. The activated cells were stored at −80°C. Cells were defrosted and assessed in the Death Assay in 20% O2 condition for 7 days. (B) Naive CD4+ T cells prepared from healthy donors were activated for 10 days in Th2 differentiation medium in 20% O2 condition. The activated cells were stored at −80°C Cells were defrosted and assessed in the Death Assay in 20% O2 condition for 7 days. The 7-AAD-negative cells in the area gated as P1 in the FSC/SSC panel were considered as living cells. The numbers on the FACS-plot panel mean the frequencies (%) of population of cells. We repeated the both experiments three times.
The second exception was preparing naive T cells from chilled Filter Buffy Coats (FBCs) [11]. We had purified both naive CD8+ T cells and naive CD4+ T cells from PBMCs trapped in used leukocyte depletion filters that had been used to concentrate red blood cells: they were stored at 4°C for 3 days before we began our preparation. The recovery rate of naive T cells from FBCs was less than 1% of that from fresh blood. Naive T cells recovered from FBCs were activated in culture for 5 days and transferred into activator-free medium in either 20% O2 or 1% O2 for 8 days. As shown in Fig. 9, 1.7% of CD8+ T cells and 12.2% of CD4+ Th2 cells survived in 20% O2: in 1% O2, 5.9 % of CD8+ T cells and 17.5% of CD4+ Th2 cells survived. The phenotypes of the survivors in 20% O2 were the same as those of the survivors in the Death Assay in hypoxia. Thus, it seemed that chilled storing the blood leukocytes on the buffy coat filters was sufficient to bypass hypoxia step in the Death Assay for activated T cells to develop into memory-like T cells.
Fig. 9.
Naive T cells prepared from chilled FBCs endured the Death Assay in 20% O2. Naive CD8+ T cells and naive CD4+ T cells were prepared from the chilled FBCs. (A) Naive CD8+ T cells were activated for 5 days in 20% O2 condition. The activated cells assessed in the Death Assay in 20% O2 condition for 8 days. (B) Naive CD4+ T cells were activated for 5 days in Th2 differentiation medium in 20% O2 condition. The activated cells were assessed in the Death Assay in 20% O2 condition for 8 days. The 7-AAD-negative cells in the area gated as P1 in the FSC/SSC panel were considered as living cells. The numbers on the FACS-plot panel mean the frequencies (%) of population of cells. We repeated this experiment twice.
Discussion
In previous studies by others, depletion of activation signals from cultures of activated T cells induced all the cells die [18]. In this study, we showed that hypoxia during the period of deprivation allowed some cells to survive in a quiescent state. The survivors expressed differentiation markers characteristic of memory T cells, and they restarted proliferation when the T cell activators were added back. We could also induce memory-like T cells from naive T cells without hypoxia, if we froze the activated T cells or prepared the naive T cells from chilled FBCs. In some ways, this phenomenon resembles stress-inducible cellular hormesis described by others [19]. Stress signaling might support the survival of deprived activated T cells in various ways. For example, hypoxia-inducible factors might contribute to the T-cell survival in hypoxia [20]. In the case of both freezing the activated T cells and preparing the naive T cells from chilled FBCs, cold shock proteins might act to promote cell survival [21].
Normally, T cells in humans never experience freezing or chilling, but they can experience hypoxic stress. Lymphocytes in lymphatic vessels, for example, especially in lymphatic capillaries are in one of the most hypoxic regions of the human body, with a mean pO2 of ~8 mm Hg [22], which is similar to the T cells maintained in our 1% O2 culture (pO2 ~10 mm Hg) [3].
The memory-like phenotypic changes induced in activated human T cells during the starvation of activating signals, including cell shrinkage, up-regulation of CD127 expression, and cell-cycle arrest, resemble the differentiation-induced by mitogen withdrawal that has been studied for decades in many types of cultured developing mammalian cells, including developing neurons, astrocytes, and oligodendrocytes [23, 24].
It will be interesting to compare the gene and/or protein expression in the memory-like T cells induced in our culture by hypoxia to the expression in the memory-like T cells induced by freezing, as it might provide some novel insights into the molecular mechanisms of T cell memory developmental. Most importantly, it remains to be determined if the memory-like T cells induced in our cultures can act as memory T cells in vivo. Nonetheless, our finding showed how a small twist in culture conditions can produce a major change in the fate of human T cells.
Supplementary Material
Acknowledgements
The authors thank the volunteers providing their blood. We also present our special thanks to Masami Bessho for his organization of the research team. We greatly appreciate Naofumi Tomura for his superior coordination of this project. And we appreciate Martin C. Raff for his helpful discussion and very kind supports including English editing. Used leukocyte depletion filters were gifts from the Japanese Red Cross Society (R020020).
Glossary
Abbreviations
- CFSE
carboxyfluorescein succinimidyl ester
- CNS
central nervous system
- FACS
fluorescent-activated cell sorting
- FSC
forward scatter
- OPCs
oligodendrocyte precursor cells
- PBMCs
peripheral blood mononuclear cells
- SSC
side scatter
- TSLP
thymic stromal lymphopoietin
- TSCM cells
stem cell memory T cells
- TCM cells
central memory T cells
- TEM cells
effector memory T cells
- TRM cells
tissue-resident memory T cells
Funding
This study was supported by the Super Highway grant (SHW2019-03) from Japan Science and Technology Agency (JST).
Conflict of interest
All authors declare no competing financial interests.
Author contributions
Y.T. was the principal investigator and made the research plan and performed experiments and prepared the manuscript. Y.A. and S.O. contributed experimental planning, performed experiments and edited the manuscript. Y.N. and Y.M. contributed to FACS analysis. T.M. provided research support and study coordination, and edited the manuscript.
Data availability
All data generated during this study are included in this published article and its online supplementary information file.
References
- 1. Farber DL, Yudanin NA, Restifo NP.. Human memory T cells: generation, compartmentalization and homeostasis. Nat Rev Immunol 2014, 14, 24–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Fearon DT, Manders P, Wagner SD.. Arrested differentiation, the self-renewing memory lymphocyte, and vaccination. Science 2001, 293, 248–50. [DOI] [PubMed] [Google Scholar]
- 3. Tokumoto Y, Tamaki S, Kabe Y, Takubo K, Suematsu M.. Quiescence of adult oligodendrocyte precursor cells requires thyroid hormone and hypoxia to activate Runx1. Sci Rep 2017, 7, 1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Richardson WD, Young KM, Tripathi RB, McKenzie I.. NG2-glia as multipotent neural stem cells: fact or fantasy? Neuron 2011, 70, 661–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Raff M. Intracellular developmental timers. Cold Spring Harb Symp Quant Biol 2007; 72: 431–443. [DOI] [PubMed] [Google Scholar]
- 6. Flajnik MF. A cold-blooded view of adaptive immunity. Nat Rev Immunol 2018, 18, 438–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Verkhratsky A, Ho MS, Parpura V.. Evolution of Neuroglia. Adv Exp Med Biol 2019, 1175, 15–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Goodell MA, Nguyen H, Shroyer N.. Somatic stem cell heterogeneity: diversity in the blood, skin and intestinal stem cell compartments. Nat Rev Mol Cell Biol 2015, 16, 299–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Araki Y, Fann M, Wersto R, Weng NP.. Histone acetylation facilitates rapid and robust memory CD8 cell response through differential expression of effector molecules (eomesodermin and its target: perforin and granzyme B). J Immunol 2008;180(12): 8102–8108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Araki Y, Wang Z, Zang C, et al.. Genome-wide analysis of histone methylation reveals chromatin state-based regulation of gene transcription and function of memory CD8+ T cells. Immunity 2009, 30, 912–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Meyer TP, Zehnter I, Hofmann B, et al.. Filter Buffy Coats (FBC): a source of peripheral blood leukocytes recovered from leukocyte depletion filters. J Immunol Methods 2005, 307, 150–66. [DOI] [PubMed] [Google Scholar]
- 12. Gropper Y, Feferman T, Shalit T, Salame TM, Porat Z, Shakhar G.. Culturing CTLs under hypoxic conditions enhances their cytolysis and improves their anti-tumor function. Cell Rep 2017, 20, 2547–55. [DOI] [PubMed] [Google Scholar]
- 13. Klenerman P, Hill A.. T cells and viral persistence: lesson from diverse infections. Nat. Immunol. 2005; 6(9): 873–879. [DOI] [PubMed] [Google Scholar]
- 14. Boettler T, Panther E, Bengsch B, et al.. Expression of the interleukin-7 receptor alpha chain (CD127) on virus-specific CD8+ T cells identifies functionally and phenotypically defined memory T cells during acute resolving hepatitis B virus infection. J Virol 2006, 80, 3532–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kaech SM, Tan JT, Wherry EJ, Konieczny BT, Surh CD, Ahmed R.. Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nat Immunol 2003, 4, 1191–8. [DOI] [PubMed] [Google Scholar]
- 16. Lyons AB. Analysing cell division in vivo and in vitro using flow cytometric measurement of CFSE dye dilution. J Immunol Methods 2000, 243, 147–54. [DOI] [PubMed] [Google Scholar]
- 17. Zhu J, Yamane H, Paul WE.. Differentiation of effector CD4 T cell populations. Annu. Rev. Immunol. 2010; 28: 445–489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Zhan Y, Carrington EM, Zhang Y, Heinzel S, Lew AM.. Life and death of activated T cells: How are they different from naïve T cells? Front Immunol 2017, 8, 1809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Zhou DR, Eid R, Miller KA, Boucher E, Mandato CA, Greenwood MT.. Intracellular second messengers mediate stress inducible hormesis and Programmed Cell Death: A review. Biochim Biophys Acta Mol Cell Res 2019, 1866, 773–92. [DOI] [PubMed] [Google Scholar]
- 20. Keith B, Johnson RS, Simon MC.. HIF1a and HIF2a: sibling rivalry in hypoxic tumor growth and progression. Nat. Rev. Cancer 2012; 12(1): 9–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Lindquist JA, Mertens PR.. Cold shock proteins: from cellular mechanisms to pathophysiology and disease. Cell Commun Signal 2018, 16, 63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Ohhashi T, Mizuno R, Ikomi F, Kawai Y.. Current topics of physiology and pharmacology in the lymphatic system. Pharmacol Ther 2005, 105, 165–88. [DOI] [PubMed] [Google Scholar]
- 23. Reynolds BA, Tetzlaff W, Weiss S.. A multipotent EGF-responsive striatal embryonic progenitor cell produces neurons and astrocytes. J Neurosci 1992, 12, 4565–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Raff MC, Miller RH, Noble M.. A glial progenitor cell that develops in vitro into an astrocyte or an oligodendrocyte depending on culture medium. Nature 1983, 303, 390–6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated during this study are included in this published article and its online supplementary information file.