Abstract
Atherosclerosis is a chronic inflammatory disease of the arterial wall, characterized by the formation of plaques containing lipid, connective tissue and immune cells in the intima of large and medium-sized arteries. Over the past three decades, a substantial reduction in cardiovascular mortality has been achieved largely through LDL-cholesterol-lowering regimes and therapies targeting other traditional risk factors for cardiovascular disease, such as hypertension, smoking, diabetes mellitus and obesity. However, the overall benefits of targeting these risk factors have stagnated, and a huge global burden of cardiovascular disease remains. The indispensable role of immunological components in the establishment and chronicity of atherosclerosis has come to the forefront as a clinical target, with proof-of-principle studies demonstrating the benefit and challenges of targeting inflammation and the immune system in cardiovascular disease. In this Review, we provide an overview of the role of the immune system in atherosclerosis by discussing findings from preclinical research and clinical trials. We also identify important challenges that need to be addressed to advance the field and for successful clinical translation, including patient selection, identification of responders and non-responders to immunotherapies, implementation of patient immunophenotyping and potential surrogate end points for vascular inflammation. Finally, we provide strategic guidance for the translation of novel targets of immunotherapy into improvements in patient outcomes.
Subject terms: Atherosclerosis, Innate immune cells, Inflammation, Immunotherapy, Adaptive immunity
In this Review, the authors provide an overview of the immune cells involved in atherosclerosis, discuss preclinical research and published and ongoing clinical trials assessing the therapeutic potential of targeting the immune system in atherosclerosis, highlight emerging therapeutic targets from preclinical studies and identify challenges for successful clinical translation.
Key points
Inflammation is an important component of the pathophysiology of cardiovascular disease; an imbalance between pro-inflammatory and anti-inflammatory processes drives chronic inflammation and the formation of atherosclerotic plaques in the vessel wall.
Clinical trials assessing canakinumab and colchicine therapies in atherosclerotic cardiovascular disease have provided proof-of-principle of the benefits associated with therapeutic targeting of the immune system in atherosclerosis.
The immunosuppressive adverse effects associated with the systemic use of anti-inflammatory drugs can be minimized through targeted delivery of anti-inflammatory drugs to the atherosclerotic plaque, defining the window of opportunity for treatment and identifying more specific targets for cardiovascular inflammation.
Implementing immunophenotyping in clinical trials in patients with atherosclerotic cardiovascular disease will allow the identification of immune signatures and the selection of patients with the highest probability of deriving benefit from a specific therapy.
Clinical stratification via novel risk factors and discovery of new surrogate markers of vascular inflammation are crucial for identifying new immunotherapeutic targets and their successful translation into the clinic.
Introduction
Atherosclerosis, the major cause of cardiovascular disease (CVD), is a chronic inflammatory disease triggered by the accumulation of cholesterol-containing LDL particles in the arterial wall1. The gold standard of treatment for atherosclerosis is the prevention of cardiovascular events by targeting modifiable risk factors and the re-establishment of arterial flow by percutaneous or surgical procedures2,3. However, the therapeutic benefit of these strategies on cardiovascular outcomes has stagnated and a huge global burden of CVD remains4.
Evidence for the role of inflammation in atherosclerosis has accumulated over the past 35 years (Fig. 1). Attilio Maseri (1935–2021) was one of the first investigators to foresee the importance of inflammation as a component of the pathogenesis of acute coronary syndromes5,6. The arterial wall is populated by various immune cells, both in healthy individuals and in patients with disease7,8. The innate immune system is the first line of defence against invading pathogens and the innate immune response is usually initiated by pattern recognition receptors, including Toll-like receptors (TLRs)9,10. The innate immune response induces the activation of antigen-presenting cells such as macrophages and dendritic cells that mediate antigen presentation, co-stimulation and cytokine production in the immune synapse to trigger the adaptive immune response. The adaptive immune response involves B cells and T cells and is slower but more specific and long-lived than the innate immune response. Athero-inflammation involves the activation of both innate and adaptive immune responses, with both inherently linked8,11 (Fig. 2). Immune cells in the arteries are activated owing to persistent inflammatory stimuli or a failure in the resolution of inflammation, leading to chronic inflammation, a hallmark of CVD12. To understand atherogenesis, we must consider the interplay between cellular immunity and lipid retention13 and the complex crosstalk between and within immune and non-immune cells, as well as the advantages and disadvantages of the experimental models used in this research field (Box 1).
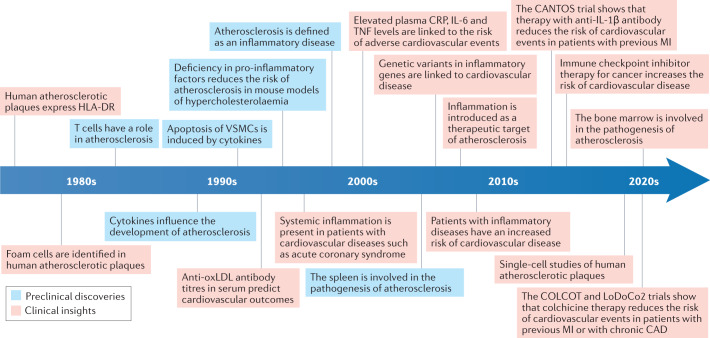
Fig. 1. History of research into the role of inflammation in atherosclerosis.
The timeline shows the main milestones in the past four decades of research into the role of inflammation in atherosclerosis. In the 1980s, the introduction of immunohistochemical techniques to study atherosclerotic plaques provided evidence of HLA-DR expression in human atherosclerotic plaques, followed by identification of monocytes, macrophages and T cells in the plaque29,44,263–269. In the 1990s, studies showed the presence of pro-inflammatory cytokines, such as tumour necrosis factor (TNF), in atherosclerotic plaques270–274, and the association between high plasma C-reactive protein (CRP) levels and coronary artery disease (CAD)5,275. During this decade, the first mouse models of hypercholesterolaemia with an inflammatory gene knockout were developed274,276,277 and titres of antibodies against oxidized LDL (oxLDL) in the serum were shown to predict cardiovascular disease outcomes278. In the 2000s, studies demonstrated the association between increased levels of inflammatory markers and increased risk of cardiovascular events279,280. An increased risk of cardiovascular disease was shown in patients with inflammatory diseases281–283, and several studies demonstrated the association between elevated levels of CRP, IL-6 and TNF in the plasma and worse clinical outcomes in patients with cardiovascular disease115,127,284,285. This finding led to the introduction of inflammation as a therapeutic target in cardiovascular disease286. In the late 2010s, studies showed that immune checkpoint inhibitor treatment increased the risk of cardiovascular disease in patients with cancer287,288. In the past decade, clinical trials investigated whether targeting inflammation in cardiovascular disease is beneficial32,34,135. Numerous studies also demonstrated the involvement of the bone marrow in atherosclerosis172,177,178 and performed single-cell analysis of plaque immune cells19,25. Preclinical discoveries are shown in blue boxes and clinical discoveries in red boxes. MI, myocardial infarction; VSMC, vascular smooth muscle cell.
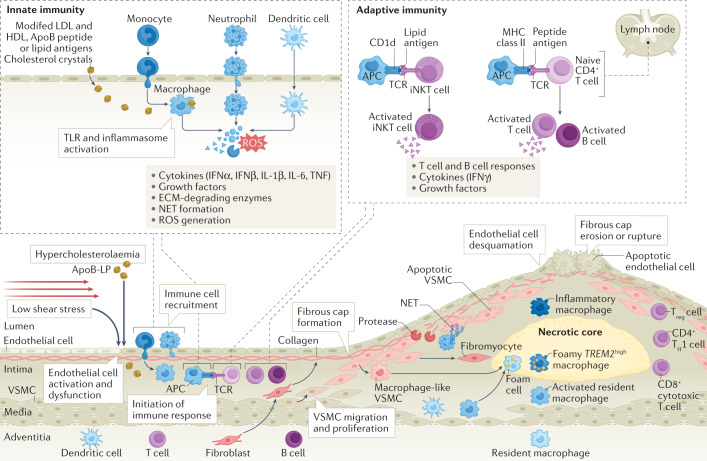
Fig. 2. Inflammation in atherosclerosis.
In medium and large arteries, haemodynamic forces create areas of low shear stress that are often predictors of atherosclerotic plaque location. As the atherosclerotic plaque begins to form, circulating apolipoprotein B (ApoB)-containing lipoproteins (ApoB-LP) and ApoB peptides enter the subendothelial space, where they can be modified and recognized by innate immune cells as danger signals. These danger signals activate Toll-like receptor (TLR) signalling and the inflammasome in innate immune cells, eliciting responses that drive inflammation, including production and secretion of cytokines, release of neutrophil extracellular traps (NETs), upregulation of co-stimulatory molecules and promotion of monocyte recruitment to the plaque289. Macrophages derived from monocyte differentiation, local proliferation or from transdifferentiation of vascular smooth muscle cells (VSMCs) take up lipoproteins present in the plaque and become lipid-laden foam cells that lay the foundation for the formation of the plaque necrotic core. At the immune synapse, antigen-presenting cells (APCs), including macrophages, dendritic cells and B cells, present lipid antigens to invariant natural killer T (iNKT) cells and peptide antigens to T cells, the latter engaging adaptive T cell and B cell responses. Antigen presentation occurs in the plaque and in secondary lymph organs, such as the lymph node8. Together, all these processes contribute to endothelial dysfunction, leading to further aggravation of inflammation through continued monocyte recruitment, increased uptake of lipoproteins adding to the plaque lipid burden, VSMC activation and proliferation, and fibroblast migration contributing to fibrous cap formation. ECM, extracellular matrix; IFN, interferon; MHC, major histocompatibility complex; ROS, reactive oxygen species; TCR, T cell receptor; TH1, T helper 1; TNF, tumour necrosis factor; Treg cell, regulatory T cell.
A unique aspect that sets aside atherogenesis from other chronic inflammatory diseases is the crucial role of lipid particles in the induction of atherogenesis. Modified lipoproteins, such as oxidized LDL (oxLDL), trigger the immune response through a unique property, whereby these particles can act as both antigens activating the adaptive immune response8,14 and adjuvant molecular patterns activating the innate immune response15,16. In advanced atherosclerosis, complex chronic inflammatory processes result in the generation of a plaque with a thin fibrous cap and a large necrotic core, or in plaque erosion or other plaque morphologies associated with clinical vulnerability to rupture, which lead to ischaemic events17. The complexity of inflammation in atherosclerosis has been emphasized by single-cell studies in humans and mice showing the high heterogeneity of vascular leukocytes in atherosclerotic lesions18–27. This heterogeneity underscores the importance of targeting specific cell subsets to inhibit atherosclerosis progression while maintaining tissue homeostasis. Superimposing the single-cell transcriptional landscape of leukocytes from mouse and human atherosclerotic plaques will help identify the different pathways, genes or cells that can be used in animal models to study human disease. Moreover, emerging evidence now shows that atherogenesis is a multiorgan process with contributions from organs such as the bone marrow and spleen28,29. In particular, the presence of clonal haematopoiesis of indeterminate potential (CHIP), an age-related process in which certain somatic mutations in bone marrow progenitor cells confer a competitive advantage leading to the expansion of specific cell clones, has been proposed as a risk factor for CVD30,31.
The first proof of the benefits of targeting inflammation in CVD in humans came from the 2017 CANTOS trial32, which showed improved clinical outcomes in patients with a history of myocardial infarction (MI) who received treatment with antibodies against IL-1β (canakinumab) compared with those who received placebo (Table 1). This finding was quickly followed by evidence from two clinical trials published in 2019 and 2020 showing that the anti-inflammatory effects of colchicine therapy reduced the risk of cardiovascular events in patients with recent MI33 or coronary artery disease (CAD)34. Evidence for the role of inflammation in CVD has also been described in other disease settings. Patients with chronic inflammatory diseases such as lupus or rheumatoid arthritis (RA) have an increased risk of CVD (tenfold and twofold, respectively) compared with healthy controls, and this risk significantly correlates with the magnitude of systemic inflammation35. Moreover, checkpoint inhibitor therapies used for several cancer types to improve tumour surveillance by the immune system are associated with an increased risk of CVD, adding to the challenges in the cardio-oncology field36,37. Together, these studies highlight immunotherapeutics as the next step in CVD therapy that will provide an opportunity to surpass the ceiling reached with the current management of classic risk factors for CVD to address the residual cardiovascular risk38. At present, the challenge lies in identifying crucial effectors of atherosclerosis-specific inflammation among the plethora of inflammatory mediators while sparing the host defence.
Table 1.
Immunotherapies proven to be effective in phase III clinical trials in cardiovascular disease
| Trial (year) | Agent | Drug target | Trial design | Patient cohort | Primary end point | Main outcomes | Ref. |
|---|---|---|---|---|---|---|---|
| CANTOS (2017) | Canakinumab | Inhibition of the IL-1β pathway | Randomized, double-blind, placebo-controlled | 10,061 patients with previous MI and elevated plasma CRP levels | Non-fatal MI, non-fatal stroke or death from cardiovascular causes | The 150-mg dose of canakinumab reduced cardiovascular events compared with placebo, independent of lipid level reductions | 32 |
| COLCOT (2019) | Colchicine | Broad cellular effects, including inhibition of tubulin polymerization, alteration of leukocyte responsiveness, and inhibition of inflammasome assembly and IL-1 release | Randomized, double-blind, placebo-controlled | 4,745 patients with MI within 30 days before enrolment | Death from cardiovascular causes, resuscitated cardiac arrest, MI, stroke, or hospitalization for angina leading to coronary revascularization | Colchicine decreased the risk of the composite end point compared with placebo | 33 |
| LoDoCo2 (2020) | Colchicine | Broad cellular effects, including inhibition of tubulin polymerization, alteration of leukocyte responsiveness, and inhibition of inflammasome assembly and IL-1 release | Randomized, double-blind, placebo-controlled | 5,522 patients with chronic coronary artery disease | Death from cardiovascular causes, spontaneous MI, ischaemic stroke or ischaemia-driven coronary revascularization | Colchicine decreased the risk of the composite end point compared with placebo | 34 |
CRP, C-reactive protein; MI, myocardial infarction.
In this Review, we discuss the therapeutic potential of targeting the immune system in atherosclerosis. First, we provide an overview of immune cells involved in CVD. Next, we summarize the published and ongoing clinical trials targeting the immune system in atherosclerosis and identify important challenges that need to be addressed to advance the translation of novel immunotherapeutics into the clinic. Finally, we highlight the new therapeutic targets emerging from preclinical studies with the biggest potential for translational pay-off in the medium term.
Box 1 Can we learn from mouse models?
Laboratory mice have provided invaluable insights into the mammalian immune system, diseases and drug development. These models are economical, easy to breed and straightforward to manipulate genetically and for these reasons, they are here to stay. The generation of Apoe−/−290 and Ldlr−/−291 mouse models has led to scientific advances in the field of lipoprotein metabolism as well as in research on inflammation in atherosclerosis, and validated the discovery of PCSK9 as a novel therapy292,293. However, the translation of beneficial responses to therapeutics from mice to humans has not always been successful294–297.
The cardiovascular system of mice and humans differs in the levels of shear stress in the vasculature298, the degree of fibrosis299 and the content of T cells (higher in humans25). Furthermore, atherosclerotic lesions in mice form predominantly at the aortic root, a pattern observed in patients with familial hypercholesterolaemia but not relevant to the general population299. Plaque rupture frequency in mice is very low and when it happens spontaneously (usually in the brachiocephalic artery), the rupture is not at the same site as in humans (carotid artery)300. Moreover, only a few genes linked to atherosclerosis in mice have shown a genetic association with human atherosclerosis, raising questions as to the use of mouse models of atherosclerosis301. As a result, to study the spectrum of human cardiovascular disease thoroughly we have to use several experimental models.
The immune system of mice and humans is also dissimilar; mice have higher lymphocyte levels (70–90%) and fewer neutrophils (10–25%) in the blood. Toll-like receptor (TLR) expression, antibody subsets, levels of defensins and nitric oxide production are also different in mice and humans, as reviewed previously294. In addition, many cytokines and chemokines in humans have no known orthologues in mice and vice versa295. These differences are partly attributable to variations in protein expression and signalling. Genomic comparisons revealed substantial transcriptional overlap between mice and humans but raised noteworthy differences302. Moreover, the immune response varies between mouse strains as a result of genetic variations and polymorphisms arising from genetic drift and/or intentional breeding303.
Pig models have similar cardiovascular anatomical features to and higher genetic homology with humans. For instance, as in humans, pigs have ten TLRs (TLR1–10) and duplication of the IL1B gene, and pig TLRs have significant homology with their human counterparts304. However, distinguishing dendritic cells from macrophages and B cells in pigs is difficult owing to common markers in these cell types, and variations in the morphology and function of neutrophils have been reported between pig breeds305. Lymph node histology is also different between pigs and humans; in pigs, the medullary tissue is located in the periphery and the cortical part in the central area305. Finally, there is a severe lack of reagents for pig models compared with their availability for humans and mouse models. Altogether, the study of the immune system and its role in cardiovascular diseases in pig models presents challenges.
Organoid systems and lab-on-a-chip technology are being devised to fill the gap in translation between mouse models and humans306. In the meantime, we strive to improve mouse models. To reduce variability, laboratory mice are kept in specific pathogen-free conditions, leading to a low density of mature T cells, scarceness of neutrophils and low lipopolysaccharide responsiveness compared with mice in the wild, which more closely resembles the human immune system294,307,308. Therefore, part of the problem is not inherent in the use of mouse models per se but how we use them. Perhaps better models will emerge by dialling back our efforts towards pathogen-free environments. Moreover, humanized mice are a powerful tool to improve research into human cardiac disease309.
In summary, mouse models are still the foundation of basic research and offer too many advantages to be discarded. Albeit useful, no organoid or lab-on-a-chip system can fully reproduce the advantage of a structured immune system. Therefore, several questions must be considered when choosing a model: What aspect of human cardiovascular disease is addressed with the model? Does the model recapitulate the human immune response in the disease condition or a particular stage of disease? Is the species or strain appropriate to model the question? Are there reagents available to study the immune system? How could the genetic background influence the study outcome? Considering these questions, we need to keep learning from a variety of biological systems, using each one to address the appropriate question to which it can provide the answer.
Immune cells involved in atherosclerosis
In this section, we summarize the functional diversity of innate and adaptive immune cells in atherosclerosis and refer to previous reviews for further in-depth discussion. The role of platelets and other non-immune cells in inflammation have been previously reviewed39–41.
Monocytes
Monocytes are present in the blood, bone marrow and spleen during homeostasis. Monocytes can be classified into two main populations: classical monocytes (Ly6Chigh in mice and CD14+CD16− in humans) and non-classical monocytes (Ly6Clow in mice and CD14lowCD16+ in humans). In atherosclerosis, classical monocytes are recruited to atherosclerotic plaques after engagement of the chemokine receptors CCR2, CCR5 and CX3CR1 (refs11,42). In the plaque, monocytes differentiate into dendritic cells and macrophages that show high functional and phenotypic heterogeneity43. In both mice42,44 and humans45, an increase in the blood monocyte pool is associated with increased severity of atherosclerosis. Preclinical studies in mice have demonstrated that splenic Ly6Chigh monocytes contribute to both the growing atheroma and plaque instability29,46. However, monocyte recruitment also has an important role in atherosclerosis regression47, and ‘patrolling’ Ly6Clow monocytes, which are derived from Ly6Chigh monocytes, are important for endothelial cell maintenance48. Hypercholesterolaemia, stress, inflammation and other risk factors for atherosclerosis can induce emergency haematopoiesis, including extramedullary haematopoiesis in the spleen29, and contribute to disease progression by skewing haematopoietic stem cells in the bone marrow towards monopoiesis29,44,49.
Macrophages
Two distinct resident macrophage populations are found in mouse arteries, one in the intima and the other in the adventitia50. Both macrophage populations originate from embryonic precursors and their survival depends on the presence of colony-stimulating factor 1. Resident adventitial macrophages are replenished by bone-marrow-derived monocytes in the period immediately after birth and are maintained by local proliferation in adulthood51. In atherogenesis, monocytes reconstitute the population of resident macrophages in the arterial intima during early stages of atherosclerosis50, whereas local proliferation of lesional macrophages contributes to macrophage accumulation in advanced lesions52. In both health and disease, adventitial macrophages expressing lymphatic vessel endothelial hyaluronic acid receptor 1 (LYVE1) prevent unfavourable arterial remodelling, largely through the regulation of collagen production in medial vascular smooth muscle cells (VSMCs)53. Arterial intima-resident macrophages have a pro-atherogenic function, and ablation of these macrophages prevents lesion formation50. A subset of LYVE1+ vascular macrophages expressing the innate immune receptor C-type lectin CLEC4A2 has anti-atherogenic functions and the ablation of this macrophage population increases lesion formation54.
Arterial macrophages have distinct functional and ontogenetic signatures and this plasticity reflects the heterogeneous environment of atherosclerotic plaques, which is increasingly being appreciated. Genetic lineage tracing and monocyte fate mapping studies have started exploring the contributions of monocytes to specific macrophage subpopulations in atherosclerosis20,47 and have helped to understand how local progenitor cells and proliferation of resident macrophages contribute to plaque progression50,52,55. Three main macrophage populations with different inflammatory properties have been identified in single-cell studies of human19 and mouse18,27 atherosclerotic plaques, suggesting that macrophage heterogeneity in the plaques cannot be explained simply by the M1–M2 macrophage polarization paradigm56. Strikingly, a pro-inflammatory macrophage population found in mice and humans expresses high levels of IL-1β18,19, a well-recognized immune target in atherosclerosis, further highlighting the relevance of this cytokine for atherosclerosis progression. Another population of the identified macrophage subsets has a more resident-like phenotype and is enriched in transcripts of proteins involved in antigen presentation and endocytosis18,25.
Foam cells are a hallmark of atherosclerosis. These cells are derived from macrophages, dendritic cells and VSMCs57. Foam cells drive necrotic core formation through uptake of intraplaque lipids, which leads to increased endoplasmic reticulum stress and cell death57. A single-cell study of mouse atherosclerotic lesions showed that plaque Trem2high macrophages, a subset that has also been identified in adipose tissue, express genes associated with lipid handling and have a profile consistent with a foamy macrophage phenotype27. TREM2high macrophages in human and mouse atherosclerotic lesions do not express genes encoding inflammatory factors, suggesting that these subsets have a homeostatic lipid-handling role in the plaques18,22,25,58. The profile of this macrophage subset is consistent with evidence showing that intracellular accumulation of desmosterol, a precursor in cholesterol biosynthesis, maintains macrophage homeostasis through the activation of transcription of liver X receptor target genes and the suppression of inflammation18,22,25,58. This discovery draws important parallels between the pathophysiology of CVD and obesity, highlighting a common blueprint between the two most prevalent metabolic diseases at present59,60. At the same time, these findings call into question the concept of lipid-driven inflammation. Further studies are warranted to reconcile inflammatory and lipid drivers of the disease. Another aspect of plaque macrophage biology to consider is the role of these cells in plaque rupture and thrombosis through the production of matrix metalloproteinases and tissue factor61, and the coordination of intraplaque efferocytosis, a crucial mechanism for resolving inflammation in atherosclerosis62 (Box 2).
Box 2 Rebalancing the immune system in cardiovascular disease.
The balance between pro-inflammatory and anti-inflammatory immune processes is important for tissue homeostasis and to control inflammation. A failure to resolve acute inflammation results in the development of chronic inflammation, as seen in atherosclerosis. Crucial mechanisms in the resolution of inflammation in atherosclerosis involve efferocytosis and a rebalance of the levels of pro-inflammatory lipid mediators towards specialized pro-resolving mediators (SPMs). Non-specific targeting of inflammation in cardiovascular disease might affect immune subsets with homeostatic functions and induce the inhibition of endogenous plaque-resolving immune processes, such as efferocytosis.
Targeting efferocytosis
Defective efferocytosis and lack of immunomodulation promote an inflammatory environment in the atherosclerotic plaque, the formation of the necrotic core and plaque destabilization owing to secondary necrosis of apoptotic cells62. Efferocytosis is mediated through phagocytic receptors, such as tyrosine–protein kinase MER (MERTK) or LDL-receptor-related protein 1 (LRP1), and apoptotic cell ligands310–312. In atherosclerosis, impaired efferocytosis can be attributed to the downregulation or cleavage of efferocytosis receptors310,312 and dysregulated expression of ‘eat me’ signals313,314. Atherosclerotic mice with increased MERTK expression have higher levels of efferocytosis and less necrotic core formation than control Ldlr−/− mice310,311. Loss of LRP1 in macrophages or haematopoietic cells in atheroprone mice leads to increased lesion area and necrotic core size315, highlighting the potential of therapies aimed at increasing efferocytosis. One avenue for increasing efferocytosis is masking the ‘don’t eat me’ signal CD47 on apoptotic cells. Blocking CD47 with a neutralizing antibody improved efferocytosis and ameliorated atherosclerosis in Apoe−/− mice314. Drugs targeting CD47 (Hu5F9-G4 and TTI-621) are currently being tested in clinical studies as cancer therapies316,317. However, the use of anti-CD47 in a clinical setting might have various adverse effects because of the role of CD47 in the regulation of other cellular processes318, such as anaemia owing to high CD47 expression on haematopoietic stem cells and erythrocytes319. In addition, total loss of CD47 or its ligand thrombospondin, which is associated with the regulation of inflammatory responses rather than efferocytosis, increased the size of the necrotic core in mice320. Therefore, the pharmacological properties of the antibody and target accessibility should be considered before advancing this therapy into the clinical arena. Of note, concomitant inhibition of CD47 and tumour necrosis factor (TNF) using anti-CD47 antibody therapy and commercially available anti-TNF antibodies, such as infliximab or etanercept, offers a synergistic benefit in the clearance of apoptotic cells in mice314. The observation that anti-TNF therapy reduces the risk of future cardiovascular events in patients with rheumatoid arthritis321 provides a strong rationale for combining anti-inflammatory and pro-efferocytic therapies for the treatment of advanced atherosclerosis.
Specialized pro-resolving mediators
Mediators involved in the resolution of inflammation in atherosclerosis include IL-10, annexin A1 and SPMs, such as resolving D1, 15-epi-lipoxin A4 and resolvin E1 (ref.322). Chronic inflammation in mouse and human atherosclerotic plaques is characterized by an imbalance between SPMs and pro-inflammatory mediators, such as leukotrienes323. In addition, a low resolvin D1 to leukotriene ratio in saliva has been proposed as a biomarker of the presence of non-resolving inflammation324.
In the atherosclerotic plaque, lipid and peptide SPMs signal through N-formyl peptide receptor 2 (FPR2) and chemokine-like receptor 1 (CMKLR1), which are both G protein-coupled receptors. Systemic administration of SPMs, including resolvin D1, 15-epi-lipoxin A4, Ac2-26 (a synthetic analogue of annexin A1) and resolvin E1, reduced atherosclerosis in mouse and rabbit models of advanced atherosclerosis249,323,325–328. These studies highlight that restoring the balance of pro-inflammatory and pro-resolving mediators to induce the resolution of inflammation is an exciting therapeutic avenue, especially given that atherosclerosis in a clinical setting is usually treated once plaques and non-resolving inflammation have been established. Resolvin E1 analogues have been tested in a phase II trial for the treatment of ocular inflammation but did not improve outcomes compared with placebo329. However, before moving into a clinical setting in cardiovascular disease, the effect of activation of immunosuppressive mechanisms should be evaluated. Most of the above-mentioned mediators have systemic roles in maintaining tissue homeostasis. However, specific delivery of Ac2-26 using monocyte–macrophage-targeting nanoparticles increased plaque stability in mice249. Therefore, selective targeting of specific pro-resolving or pro-inflammatory cell types in atherosclerosis will most probably mediate the most beneficial outcomes in the clinic.
Dendritic cells
Dendritic cells are another crucial cell type driving atherosclerotic plaque inflammation that bridges the innate and adaptive immune responses. Dendritic cells can be classified into three main subsets: plasmacytoid dendritic cells, type 1 conventional dendritic cells (cDC1s) and type 2 conventional dendritic cells (cDC2s). Plasmacytoid dendritic cells are generally located in blood and lymphoid tissues. After encountering pathogens, these cells produce large amounts of type I interferon (IFN). By contrast, conventional dendritic cells are found in lymphoid and non-lymphoid sites. cDC1s are involved in cross-presentation of antigens and drive cytotoxic immune responses, whereas cDC2s are involved in T cell priming63.
In humans, plaque dendritic cell numbers positively correlate with plaque vulnerability64. Dendritic cells have been found to have both pro-atherogenic and anti-atherogenic functions in mouse models, as reviewed previously8. Dendritic cells elicit an adaptive immune response that encompasses both T cells and B cells8. During atherosclerosis regression in mice, dendritic cells can leave the lesions and migrate to the lymphatic tissue in a process mediated by the chemokine ligands CCL19 and CCL21 and their receptor CCR7 on the surface of dendritic cells65. Dendritic cells expressing CCL17 have a pro-atherogenic role in mice66. CD103+ cDC1s can promote atheroprotective regulatory T (Treg) cell responses67. Loss of myeloid differentiation factor 88 (MyD88) signalling in CD11c+ dendritic cells leads to loss of Treg cells and increased atherogenesis in mice68. By contrast, plasmacytoid dendritic cells have been reported to have both pro-atherogenic and anti-atherogenic roles in mice, possibly owing to subtle cellular heterogeneity in this subset69,70.
Neutrophils
Neutrophils are involved in all stages of atherosclerosis71. In mice, neutrophil depletion reduces atherosclerosis, whereas increased levels of circulating neutrophils exacerbate plaque formation, suggesting a role of this cell type in lesion development72. Neutrophils promote vascular inflammation through the secretion of reactive oxygen species, which leads to increased permeability of the endothelial cell barrier73. Neutrophils attract monocytes via secretion of chemotactic molecules and can activate macrophages via extrusion of their nuclear material as neutrophil extracellular traps (NETs)74. NETs contain histone H4, which binds to VSMCs and induces cell lysis, resulting in plaque destabilization75. In addition, NETs induce plaque erosion and platelet aggregation, leading to thrombosis76. Overall, neutrophils have a pro-atherogenic role. However, during thrombotic events, neutrophils have reparative functions through the promotion of endothelial repair and angiogenesis77.
T cells
T cells are important for atherosclerosis initiation and progression, as reviewed previously78,79. A mass cytometry study revealed that T cells outnumber macrophages in human carotid artery plaques25, in contrast to plaques in mice, in which the overall proportion of T cells is lower24. T cells in human atherosclerotic plaques show more activation-related and exhaustion-related gene expression than peripheral blood T cells. High expression of the inhibitory molecule PD1 as a consequence of chronic antigen stimulation can result in inefficient T cell effector function and dysregulation of the immune response within the plaque19,25. Once activated, T cells directly mediate effector functions in the arterial wall or help B cells produce antibodies. CD4+ T cells are the most abundant T cells in mouse atherosclerotic plaques, and are polarized predominantly towards a pro-inflammatory phenotype (T helper 1 (TH1) cells)79. CD4+ T cells have been shown to both protect against and promote atherogenesis depending on the subset involved. TH1 cells have been consistently shown to have pro-atherogenic roles, whereas Treg cells are thought to have atheroprotective roles via IL-10 and TGFβ secretion78. The role of TH2 cells and TH17 cells in atherosclerosis is controversial78. Phenotyping of CD4+ T cells in a mouse model of atherosclerosis with the use of single-cell RNA sequencing revealed a CD4+ T cell population80 that shared transcriptional similarities with apolipoprotein B (ApoB)-reactive CD4+ T cells81. During atherosclerosis progression, ApoB-reactive CD4+ T cells undergo a transition from a Treg cell to a pro-inflammatory phenotype, which might contribute to further disease progression81.
CD8+ T cells in atherosclerotic lesions have also been found to have dual functions, with pro-atherogenic effects mediated by IFNγ production and macrophage activation, and atheroprotective effects via B cell modulation78. CD8+ T cells in mice have been identified as drivers of plaque inflammation and apoptosis, promoting unstable plaque phenotypes and plaque erosion82,83. CD8+ T cells outnumber CD4+ T cells in advanced human atherosclerotic plaques25,82, and an increase in CD8+ T cell numbers in blood is associated with the presence of CAD84,85.
Invariant natural killer T (iNKT) cells are a distinct subset of T cells that express unique invariant T cell receptors and natural killer cell surface molecules, such as CD161 (also known as NK1.1 in mice) and killer cell immunoglobulin-like receptors (analogous to the Ly49 family in mice)86. Given the central role of lipids in atherosclerosis, iNKT cells are a relevant cell type because they respond to lipid antigens presented by CD1d on antigen-presenting cells. In mice, iNKT cells are considered to be pro-atherogenic owing to their production of pro-inflammatory cytokines such as IFNγ86. In humans, rupture-prone plaques have higher numbers of iNKT cells than stable plaques87 but the exact mechanism underlying this observation is unknown.
B cells
B cell subpopulations make different contributions to atherogenesis88. B cells are central to humoral immunity and mediate the production of antibodies against oxidation-specific epitopes to help dampen inflammation. B cells are classified into two lineages: B1 cells, which are mainly produced in the fetal liver, and B2 cells, which originate in the bone marrow. B1 cells are further subdivided into B1a and B1b subsets. B2 cells can differentiate into transitional (T1 and T2 marginal zone progenitor) B cells, marginal zone B cells, follicular B cells and antibody-secreting plasma cells88. In atherosclerosis, B cells are not always found in the plaque and are more commonly localized in the adventitia or in node-like structures, referred to as tertiary lymphoid organs, that form in the adventitia as a result of chronic inflammation79. B1 cells have been described as atheroprotective in mice owing to the production of IgM antibodies that block the uptake of oxLDL by macrophages in lesions16,89. By contrast, B2 cells have been shown overall to be pro-atherogenic, through antibody responses formed via germinal centre B cell reactions that further drive adaptive immunity88. In mice fed a high-cholesterol diet, subsets of B2 cells with atheroprotective functions arise in secondary lymphoid organs, such as the lymph node (T2 marginal zone progenitor B cells)90 and the spleen (marginal zone B cells)91. These subsets act either through PDL1-mediated suppression of T follicular helper cells91 or via IL-10, although the role of IL-10 varies in different mouse models (IL-10 was shown to have a role in Apoe−/− mice90 but not in Ldlr−/− chimeric mice92) and is dependent on the microbiome93 and the radioresistance of B cell subsets94.
Clinical trials of immunotherapies in CVD
Over the past 5 years, promising results from clinical trials targeting inflammation in CVD have been reported. In this section, we summarize the positive phase III trials, promising phase II studies, ongoing trials and trials with neutral results, and the lessons learnt from these studies (Fig. 3).
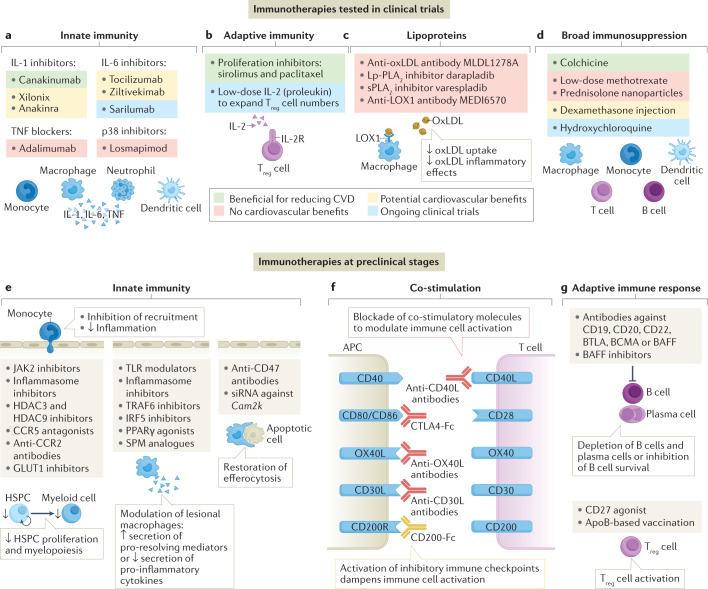
Fig. 3. Targeting the immune system in atherosclerosis.
a–d | Immunotherapies for the treatment of atherosclerosis that showed benefit (green), no benefit (red) or potential benefit (yellow) in reducing inflammation or cardiovascular events in clinical trials or currently being tested in ongoing clinical trials (blue) are shown. Therapeutics targeting innate immunity include IL-1 inhibitors, IL-6 inhibitors, tumour necrosis factor (TNF) blockers and p38 inhibitors (panel a). Therapeutics targeting adaptive immunity include local proliferation inhibitors in drug-eluting stents and low-dose IL-2 targeting regulatory T (Treg) cells (panel b). Therapeutics targeting lipoproteins to reduce inflammation include antibodies against oxidized LDL (oxLDL), lipoprotein-associated phospholipase A2 (Lp-PLA2), secretory phospholipase A2 (sPLA2) and lectin-like oxidized LDL receptor 1 (LOX1) (panel c). Therapeutics with broad immunosuppressive effects include colchicine, low-dose methotrexate, glucocorticoids and hydroxychloroquine (panel d). See Tables 1,2 and 3 and Supplementary Table 1 for further details. e–g | Overview of therapeutics in preclinical development targeting innate immunity (panel e), co-stimulation pathways (panel f) and B cell and T cell regulation (panel g). APC, antigen-presenting cell; ApoB, apolipoprotein B; BAFF, B cell activating factor; BCMA, B cell maturation antigen; BTLA, B and T lymphocyte attenuator; CCR, C-C chemokine receptor; CD30L, CD30 ligand; CD40L, CD40 ligand; CTLA4, cytotoxic T lymphocyte antigen 4; CVD, cardiovascular disease; GLUT1, glucose transporter 1; HDAC, histone deacetylase; HSPC, haematopoietic stem and progenitor cell; IRF5, interferon regulatory factor 5; OX40L, OX40 ligand; PPARγ, peroxisome proliferator-activated receptor-γ; siRNA, small interfering RNA; SPM, specialized pro-resolving mediators; TLR, Toll-like receptor; TRAF6, tumour necrosis factor receptor-associated factor 6.
Phase III clinical trials showing cardiovascular benefits
Two immunotherapeutics have been successful in improving the cardiovascular outcomes of patients with CVD: canakinumab32 and colchicine33,34,95 (Table 1).
Canakinumab
The CANTOS trial32 was a double-blind, randomized, controlled trial investigating the effects of canakinumab, a monoclonal antibody against the pro-inflammatory cytokine IL-1β, in patients with recent MI. In total, 10,061 patients with a history of MI who were receiving optimal management for cardiovascular risk factors and had high-sensitivity C-reactive protein (hsCRP) levels of >2 mg/l were randomly assigned to receive canakinumab or placebo. Canakinumab was administered subcutaneously at doses of 50 mg, 150 mg or 300 mg every 3 months. Patients were followed up for a median of 3.7 years. The 150-mg canakinumab dose led to a significantly lower rate of recurrent cardiovascular events than placebo, independently of lipid-level lowering (HR 0.85, 95% CI 0.74–0.98; P = 0.021)32. No effect was observed on total mortality, owing to a small but significant increased risk of infection with canakinumab. Notably, among patients receiving canakinumab, those with a reduction in on-treatment hsCRP levels to <2 mg/l benefited the most from the treatment, and the effect of canakinumab at reducing hsCRP levels was dose-dependent96. A subanalysis extended the scope of the effects of canakinumab beyond IL-1β by showing that the modulation of plasma IL-6 levels is associated with the beneficial effects of canakinumab in reducing the risk of cardiovascular events97. Moreover, canakinumab reduced cancer mortality98. The CANTOS trial demonstrated for the first time the proof-of-principle that therapeutic targeting of the immune system can be beneficial for cardiovascular outcomes in patients.
Colchicine
Colchicine, which is widely used for the treatment of gout and pericarditis, decreases inflammation by inhibiting cytoskeletal microtubule formation99,100. Colchicine has broad cellular effects, including reduction of monocyte and neutrophil motility and inhibition of inflammasome assembly in vitro101. The LoDoCo2 trial34 included 5,522 patients with stable chronic CAD. After 1 month of open-label use of colchicine (0.5 mg once daily), patients were randomly assigned to receive colchicine or placebo and followed up for a median of 28.6 months. Patients receiving colchicine had a 31% reduction in the incidence of the primary composite end point of cardiovascular death, MI, ischaemic stroke and ischaemia-driven coronary revascularization compared with patients receiving placebo (HR 0.69, 95% CI 0.57–0.83; P < 0.001). Unfortunately, data on the effects of colchicine on inflammatory markers are not available. The results of this trial are consistent with those of two phase II trials investigating colchicine, LoDoCo95 (in patients with stable chronic CAD) and COLCOT33 (in patients with MI), and provide further support for the potential benefits of anti-inflammatory therapy in patients with acute coronary disease. Taken together, these trials demonstrated that anti-inflammatory therapies are efficacious in reducing cardiovascular events in patients with stable CVD. Although CANTOS and LoDoCo2 have not yet changed the treatment strategy in cardiovascular risk management in clinical practice, these trials are a crucial milestone for the clinical translation of immunomodulatory therapeutics in CVD. Both treatments target innate immunity, offering proof in humans of the importance of the innate response of the immune system in triggering inflammation in atherosclerosis.
Promising phase II clinical trials
Several cytokine blockers have shown promising results in phase II trials (Table 2). Cytokine blockers are the first line of biologics for the treatment of chronic inflammatory diseases, including RA, inflammatory bowel disease and psoriasis102–104. Therefore, an arsenal of potential therapeutics for CVD is available, some of which will soon be available as generic drugs (such as tumour necrosis factor (TNF) blockers).
Table 2.
Potentially effective immunotherapies in phase II clinical trials in cardiovascular disease
| Study (year) | Agent | Drug target | Study design | Patient cohort | Primary end point | Main outcomes | Ref. |
|---|---|---|---|---|---|---|---|
| El Sayed et al. (2016) | Xilonix | Monoclonal antibody specifically targeting IL-1α | Randomized, placebo-controlled | 43 patients undergoing percutaneous SFA revascularization | Clinically significant target vessel restenosis, time to restenosis and incidence of major adverse cardiovascular events | At 12 months of follow-up, no difference between Xilonix and placebo; at 3 months, trend towards decreased restenosis (0% versus 10%) and cardiovascular events (9% versus 24%) in the Xilonix versus placebo groups | 112 |
| MRC-ILA heart study (2015) | Anakinra | IL-1 receptor antagonist | Randomized, double-blind, placebo-controlled | 182 patients with NSTE–ACS presenting <48 h from onset of chest pain | hsCRP AUC over the first 7 days after treatment initiation | Decrease in hsCRP levels after 14 days of treatment with anakinra; similar risk of MACE at 30 days and 3 months but significant increase in MACE at 1 year in the anakinra group compared with the placebo group | 111 |
| VCU-ART3 (2020) | Anakinra | IL-1 receptor antagonist | Randomized, double-blind, placebo-controlled | 99 patients with STEMI | hsCRP AUC at baseline and at 72 h and 14 days after treatment initiation | Decrease in hsCRP AUC after 14 days of treatment with anakinra; reduced incidence of new-onset heart failure, death and hospitalization for heart failure in the anakinra group compared with the placebo group | 110 |
| DANCE (2018) | Dexamethasone delivered to the adventitial tissue surrounding target lesions | Broad anti-inflammatory effect | Prospective, single-group, open- label; data compared with findings from contemporary trials | 262 patients with symptomatic PAD receiving PTA (n = 124) or atherectomy (n = 159) | 12-month primary patency (composite of freedom from binary restenosis and clinically driven target-lesion revascularization) | Reduced restenosis after 12 months of follow-up | 243 |
| Kleveland et al. (2016) | Tocilizumab | Monoclonal antibody against IL-6 receptor | Randomized, double-blind, placebo-controlled | 117 patients with NSTEMI, included in the randomization at a median of 2 days after symptom onset | hsCRP AUC at 1–3 days of treatment initiation | Tocilizumab reduced hsCRP levels compared with placebo | 119 |
| ASSAIL-MI (2021) | Tocilizumab | Monoclonal antibody against IL-6 receptor | Randomized, double-blind, placebo-controlled | 199 patients within 6 h of STEMI and undergoing PCI | Myocardial salvage index measured by MRI 3–7 days after treatment initiation | Tocilizumab increased the myocardial salvage index and reduced CRP levels compared with placebo | 118 |
| RESCUE (2021) | Ziltivekimab | Monoclonal antibody against IL-6 | Randomized, double-blind, placebo-controlled | 264 patients with chronic kidney disease and hsCRP >2 mg/l | hsCRP measured 12 weeks after treatment initiation | Ziltivekimab reduced hsCRP levels at all doses compared with placebo | 120 |
AUC, area under the curve; CRP, C-reactive protein; hsCRP, high-sensitivity C-reactive protein; MACE, mayor adverse cardiovascular events; MI, myocardial infarction; NSTE–ACS, non-ST-segment elevation acute coronary syndrome; NSTEMI, non-ST-segment elevation myocardial infarction; PAD, peripheral artery disease; PCI, percutaneous coronary intervention; PTA, percutaneous transluminal angioplasty; SFA, superficial femoral artery; STEMI, ST-segment elevation myocardial infarction.
IL-1 blockade
IL-1 is a pro-inflammatory cytokine that drives inflammation in atherosclerosis105. Both isoforms of IL-1, IL-1α and IL-1β, are involved in atherosclerosis. Studies in mice have shown that IL-1α has a role in the remodelling of arteries during early atherogenesis, whereas IL-1β mainly drives vascular inflammation in later stages of atherosclerosis106. However, IL-1β had a protective role in advanced atherosclerosis in mice through the promotion and maintenance of a fibrous cap rich in VSMCs and collagen107. Additionally, IL-1α forms a link between the immune system and coagulation through the activation of IL-1α by thrombin, underscoring the importance of this isoform in the pathogenesis of adverse cardiovascular events108. In humans, the levels of IL-1β in the coronary arteries are higher in patients with CAD than in patients with non-ischaemic cardiomyopathy109, and this cytokine is considered to be therapeutically tractable. Several options are available for IL-1 blockade, including canakinumab (selective IL-1β targeting), anakinra (an IL-1 receptor antagonist, which thereby targets IL-1α and IL-1β) and xilonix (a monoclonal antibody specifically targeting IL-1α). In two separate studies, therapy with anakinra significantly reduced hsCRP levels in the acute setting in patients with ACS compared with placebo110,111. Therapy with xilonix plus standard of care showed a non-significant trend towards a reduction in restenosis and the incidence of major adverse cardiovascular events compared with standard of care only in patients undergoing percutaneous femoral artery revascularization112. Whereas the CANTOS trial highlighted the relevance of targeting IL-1β in stable CAD, these studies illustrate the importance of IL-1 as a target in the acute setting of thrombotic events. Additional studies in larger patient groups should be performed to further assess the effect of these therapeutics on cardiovascular outcomes.
IL-6 blockade
IL-6 is a pro-inflammatory cytokine involved in the innate immune response and a downstream mediator of a cytokine cascade featuring TNF and IL-1. IL-6 is a central stimulus for the acute phase response. In particular, IL-6 stimulates the production of CRP, among other acute phase reactants, in hepatocytes113. IL-6 signalling contributes to atherosclerosis and plaque destabilization in mice114. Data from humans show that elevated IL-6 levels in the plasma are associated with an increased risk of MI, and genetic studies have provided evidence of a causal role for IL-6 receptor signalling in CVD115–117. Therapy with tocilizumab, a monoclonal antibody targeting the IL-6 receptor, reduced hsCRP levels in patients with ST-segment elevation MI (STEMI)118 or non-STEMI119 compared with placebo. Tocilizumab therapy also significantly increased the myocardial salvage index in patients with STEMI118; however, the absolute difference between the tocilizumab and placebo groups was only 5.6%, meaning that this increase might be of limited clinical relevance. In a phase II trial published in 2021, IL-6 blocking with the antibody ziltivekimab reduced hsCRP levels in patients with chronic kidney disease, who are at high risk of atherosclerosis120. These studies demonstrate the efficacy of IL-6 blockade for inflammation reduction. Follow-up studies, including the ZEUS trial121, will provide a more complete picture of the clinical relevance of IL-6-targeted therapies in CVD.
Blockade of other cytokines
Alternatives to IL-1 and IL-6 blockade include TNF or IL-23 blockers, given that preclinical and clinical research has demonstrated a pro-atherogenic role for these cytokines122–124. TNF is a pro-inflammatory cytokine and is produced by several cells involved in atherosclerosis, including macrophages and VSMCs125. In mice, TNF deficiency reduced atherogenesis126. In humans, TNF is present in atherosclerotic plaques and the levels of TNF in peripheral blood predict future coronary events in patients with MI125,127. In observational studies in patients with arthritis, inflammation was a strong risk factor for cardiovascular events and TNF blockade resulted in reduced atherogenesis and lower incidence of cardiovascular events compared with patients with arthritis who did not receive TNF-blocking therapy35. However, in clinical trials in patients with heart failure, TNF blockade had no efficacy or even worsened the clinical outcome128,129. Therefore, TNF blockers might not be suitable for patients with substantial deterioration of left ventricular systolic function.
IL-23 is present in human atherosclerotic plaques, and high plasma levels of IL-23 are associated with increased mortality in patients with carotid artery stenosis123. Studies in mice have shown that IL-23 drives TH17 cell function, contributing to the aggravation of atherosclerosis130–132. Despite the pro-atherogenic role of IL-23 in mice, several meta-analyses of studies in patients with psoriasis showed either no effect or possible worsening of cardiovascular outcomes after treatment with IL-23 blockers (ustekinumab and briakinumab) compared with placebo133,134. These studies were primarily designed to assess the effect of the IL-23 blockers on psoriasis and, therefore, conclusions cannot be drawn about their effect on inflammation in atherosclerosis. Other alternative therapeutic targets currently being tested in trials, including hydroxychloroquine and low-dose IL-2, are discussed in Box 3 and Table 3.
Table 3.
Ongoing randomized controlled trials targeting the immune system in atherosclerosis
| Trial name (number) | Agent | Drug target | Trial design | Patient cohort | Primary end point | Ref. |
|---|---|---|---|---|---|---|
| OXI (NCT02648464) | Hydroxychloroquine | Broad immunosuppression | Phase IV | 125 patients with MI | Rate of cardiovascular adverse events (MI, death, hospitalization for unstable angina and heart failure) | 252 |
| CHANGAN (NCT02874287) | Hydroxychloroquine | Broad immunosuppression | Phase IV | 35 patients with CAD and hsCRP >1 mg/l | Change in fasting hsCRP level | 253 |
| LILACS (NCT03113773) | Low-dose IL-2 | Induces expansion of regulatory T cell numbers | Phase I–II | 41 patients with a history of CAD or acute coronary syndrome | Safety, tolerability and circulating regulatory T cell levels | 254 |
| IVORY (NCT04241601) | Low-dose IL-2 | Induces expansion of regulatory T cell numbers | Phase II | 60 patients with ACS and hsCRP >2 mg/l | Change in vascular inflammation, as measured by FDG PET–CT | 255 |
| NCT04762472 | Montelukast | Leukotriene receptor | Phase IV | 200 adults asymptomatic for atherosclerotic disease and exposed to air pollution | Subclinical atherosclerosis (as measured by brachial flow-mediated dilatation, carotid intima–media thickness and blood inflammatory markers) | 256 |
| NCT04616872 | Methotrexate delivered in LDL-like nanoparticles | Broad immunosuppression | Phase II–III | 40 patients with multivessel CAD and hsCRP >2 mg/l | Reduction in plaque volume, measured by CTA | 257 |
| SARIPET (NCT04350216) | Sarilumab | Monoclonal antibody against IL-6 receptor | Phase IV | 20 patients with active rheumatoid arthritis and CRP levels >1 mg/dl | Changes in carotid atheroma plaque assessed by ultrasonography | 258 |
| PAC-MAN (NCT04148833) | Paclitaxel | Proliferation | Phase II–III | 40 patients with CAD | Low-attenuation plaque volume measured by CTA | 259 |
| GOLDILOX (NCT04610892) | MEDI6570 | Antibody against LOX1 receptor (blocks uptake of oxidized LDL) | Phase IIb | 792 patients with a history of MI | Non-calcified plaque volume measured by CTA | 260 |
| CLEAR-Synergy (NCT03048825) | Colchicine | Broad immunosuppression | Phase III | 7,000 patients with MI | MACE | 261 |
| CONVINCE (NCT02898610) | Colchicine | Broad immunosuppression | Phase III | 2,623 patients with ischaemic stroke or at high risk of transient ischaemic attack | Recurrence of non-fatal ischaemic stroke or non-fatal MACE, or vascular-related death | 262 |
| ZEUS (NCT05021835) | Ziltivekimab | Monoclonal antibody against IL-6 | Phase III | 6,200 patients with chronic kidney disease and CRP ≥2 mg/l | Time to first MACE | 121 |
ACS, acute coronary syndrome; CAD, coronary artery disease; CRP, C-reactive protein; CTA, computed tomography angiography; FDG, fluorodeoxyglucose; hsCRP, high-sensitivity C-reactive protein; LOX1, lectin-like oxidized LDL receptor 1; MACE, major adverse cardiovascular events; MI, myocardial infarction.
Box 3 Ongoing clinical trials targeting atherosclerosis.
Hydroxychloroquine is an antimalarial and a disease-modifying antirheumatic drug used for the treatment of inflammatory rheumatic diseases, especially systemic lupus erythematosus (SLE) and rheumatoid arthritis (RA)330. In lysosomes, hydroxychloroquine inhibits the degradation of cargo by increasing the pH and preventing the activity of lysosomal enzymes. This drug can inhibit nucleic acid sensors, such as cyclic GMP–AMP synthase, and prevents ligand binding to Toll-like receptor 7 (TLR7) and TLR9, thereby reducing the production of pro-inflammatory cytokines, including type I interferons330. In observational studies, hydroxychloroquine therapy was associated with a 72% decrease in the risk of cardiovascular events in patients with RA and a 68% reduction in thromboembolic events in patients with SLE331,332. Hydroxychloroquine is currently being tested in two clinical trials in patients with coronary artery disease252,253 (Table 3).
The cytokine IL-2 is essential for the growth and survival of regulatory T (Treg) cells, which have a role in the control of inflammation. Low-dose IL-2 therapy has been trialled in patients with SLE, RA and psoriasis333. The principle of using low doses of IL-2 for the treatment of inflammatory diseases is based on the differential sensitivity of distinct immune cell subsets to IL-2. Among all T cell and natural killer cell subsets, Treg cells typically respond to the lowest concentrations of IL-2 owing to elevated surface expression of the IL-2 receptor subunit-α (also known as CD25) and the high-affinity IL-2 receptor complex in this cell subset. Low-dose IL-2 therapy increases the number of Treg cells and the expression of functional markers, such as CD25, in patients with other inflammatory diseases334,335. High-dose IL-2 therapy administered to patients with cancer is associated with adverse effects, but this severe toxicity is not justifiable in the setting of autoimmune diseases333. The ongoing phase II LILACS trial254,336 is testing low-dose IL-2 therapy in patients with stable ischaemic heart disease and patients with acute coronary syndrome, with preliminary results showing effective expansion of Treg cells with the therapy. We look forward to the results of these and other exciting trials listed in Table 3.
Challenges
Several strategies for targeting inflammation in CVD have been tested in clinical trials but have not resulted in the reduction of inflammation markers and/or cardiovascular events (Supplementary Table 1). Notable examples are methotrexate and a p38 inhibitor, which did not reduce cardiovascular events or mortality in patients with CVD135,136. The majority of the trials that did not show efficacy of the drug being tested included unselected patient cohorts; therefore, a potential explanation for the lack of efficacy might be the heterogeneity of the patient group. The CANTOS trial32 was the first trial to take a step towards the use of precision medicine by specifically selecting patients with an increased residual inflammatory risk (measured as hsCRP >2 mg/l). However, the trials investigating colchicine also included unselected patient groups and did show beneficial effects on cardiovascular outcomes33,34. This finding illustrates that failure to demonstrate efficacy might also be mechanism-based and that inhibiting inflammation in CVD is effective provided the correct inflammatory target or drug is chosen.
The variability of disease settings in clinical trials of CVD might explain the lack of beneficial effects of p38 inhibitors. p38 is an intracellular kinase that is activated in CVD by several stressors, such as oxLDL and hypertension, and is involved in the stabilization of mRNA encoding several inflammatory mediators that are crucial in CVD137,138. The first study of the p38 inhibitor losmapimod in CVD included patients with stable atherosclerosis139. Vascular inflammation was assessed with fluorodeoxyglucose (FDG) PET–CT imaging. Losmapimod therapy did not significantly reduce the overall uptake of FDG in the index vessel compared with placebo but reduced inflammation in the most inflamed regions139. However, losmapimod had no effect on clinical outcomes in subsequent trials that included larger cohorts of patients with acute MI136,140, suggesting that p38 might have a selective role in chronic stable CVD, which is consistent with the role of p38 in prolonging inflammatory responses via modulation of mRNA stability138.
Other studies have also used FDG PET–CT imaging to assess vascular inflammation, such as the GLACIER trial141. The trial included 147 patients with stable atherosclerotic disease who were randomly assigned to receive a single dose of the anti-oxLDL antibody MLDL1278A, multiple doses of MLDL1278A or placebo. None of the MLDL1278A regimens had a significant effect on carotid plaque inflammation, possibly owing to the concomitant use of lipid-lowering medication, which might have masked the effect of passive vaccination with MLDL1278A141. This study also raises questions about the use of imaging as a surrogate end point for cardiovascular events. New PET–CT imaging tracers that can detect meaningful cardiovascular inflammation more accurately than FDG are needed142. An imaging technique developed in the past 4 years that is based on CT angiography showed that changes in the CT attenuation index of perivascular adipose tissue might be a marker of coronary perivascular inflammation associated with cardiovascular outcomes143,144. Further improvements in the imaging of atherosclerosis will facilitate the development of valid surrogate end points of cardiovascular outcomes. Although cardiovascular surrogate end points are at present not sufficiently specific and, therefore, have not reached the benchmark of a clinical trial, developments in the field of machine learning could be used to combine multiple surrogate end points for a more accurate prediction of clinical outcomes145,146.
Considering the above-mentioned successes in therapeutic targeting of the immune system in atherosclerosis, the number of ongoing trials in this setting is surprisingly low. One reason could be the high costs of clinical trials in CVD, which make this area less attractive for industry investments. Trials in CVD are event-driven rather than symptom-driven and, therefore, require high patient numbers and long follow-up. Therefore, identifying reliable surrogate markers of vascular inflammation is crucial to facilitate the design of small proof-of-principle trials, allowing rapid innovation and reduced risks. One crucial need is the early identification of patients who are likely to respond to a specific treatment and patients who would not benefit from the interruption of a specific inflammatory pathway. This concept is well exemplified by the CANTOS trial32, which demonstrated that patients with the larger reductions in hsCRP levels with canakinumab therapy derived the largest clinical benefit from the treatment. Patients with a decrease in hsCRP levels greater than the median percentage reduction had a 27% reduction in cardiovascular events compared with a reduction of only 5% in those patients with a decrease in hsCRP levels that was lower than the median96. Moreover, the fall in hsCRP levels has so far gone hand in hand with outcome benefits in the majority of clinical trials of anti-inflammatory therapies in CVD. In the future, new surrogate end points that are based on immunophenotyping and/or imaging could be used in clinical trials, provided that an association with cardiovascular outcomes is demonstrable.
Looking to the future, the secondary effects of anti-inflammatory therapies should be carefully considered. Canakinumab administration was associated with a major reduction in the incidence of lung cancer compared with placebo in the CANTOS trial98. By contrast, in the CIRT trial135,147, methotrexate was linked to a small increase in the incidence of skin cancer compared with placebo, emphasizing the complexity of the effects of immunotherapy on CVD and cancer. Immunosuppression and chronic inflammation can both increase the risk of cancer147. Furthermore, preclinical studies have spotlighted the existence of an immune-mediated link between MI and breast cancer that can accelerate cancer progression148. An increasing number of studies have also shown that immune checkpoint inhibitor therapies might increase the risk of CVD in patients with cancer36,37, whereas inhibition of adaptive immunity increases the risk of cancer through disruption of antitumour immunity149. Now that anti-inflammatory therapies in CVD are close to implementation in clinical practice, unravelling the complex immunological relationship between cancer and CVD is crucial.
Finally, the pathogenesis of CVD is multifactorial, and several types of coronary culprit lesions lead to the same clinical presentation and syndromes17. Different disease settings have distinct immune signatures, as illustrated by the different signatures in plaque erosion and rupture150, which calls for the identification of the disease setting in which a therapy will be most successful. Implementing deep immunophenotyping strategies can improve the selection of patients with the highest likelihood of benefiting from a specific therapy and facilitate rapid identification of responders and non-responders to therapy151. Immunophenotyping of patients with CVD is still in its infancy; however, a few of the currently available markers could guide patient selection, such as hsCRP and IL-6 levels in the plasma96,97,152. The discovery of CHIP as a novel risk factor of atherosclerosis will potentially enable further risk stratification of patients153. For example, a re-analysis of CANTOS data suggested that anti-inflammatory treatment might be more effective in patients carrying CHIP-associated gene variants154. Taken together, extensive immunophenotyping and immune-based risk stratification might facilitate patient selection and stratification and identification of treatment responders, allowing efficient design of clinical trials and realizing the potential of targeted immunomodulatory therapies for CVD.
In summary, the challenges in addressing the low-grade inflammation associated with CVD are manifold and encompass the need for careful risk–benefit assessment, the existence of several coronary syndromes with potentially different endotypes and pathogenesis, our current inability to identify responders to treatment early, and our reliance on ‘hard’ clinical end points in trial design owing to the limitations of our current imaging techniques. Further understanding of the immune signature of CVD together with the evolution of cardiovascular imaging technologies will accelerate the translation of therapies targeting inflammation from the preclinical to the clinical arena.
New targets for clinical translation
Advances in our understanding of the pathogenesis of atherosclerosis have highlighted several potential cellular and molecular therapeutic targets. In this section, we focus on a selection of the most promising areas supported by the convergence of several lines of evidence from CVD and other diseases, and which are, therefore, closer to translation to patient therapies in the medium term (Fig. 3).
Immunometabolism and trained immunity
Targeting immunometabolic processes is a promising strategy for modulating inflammation and immunity. Atherosclerosis-associated changes in blood and bone marrow are regulated by immunometabolic events155. In mice, a Western diet and hyperglycaemia have been shown to induce epigenetic reprogramming of myeloid progenitors, which resulted in sustained monocyte and macrophage pro-inflammatory priming, thereby driving tissue inflammation and CVD156–158. These effects persisted even after restoring lipid and glucose levels to normal levels owing to the phenomenon of ‘trained immunity’, whereby transcriptomic, epigenetic and metabolic rewiring of innate immune cells leads to an altered response towards a second challenge159.
Epigenetic regulation is of particular interest because of the potential for pharmacological inhibition. Histone deacetylases (HDACs) repress gene expression by removing open-chromatin acetylation marks. Broad HDAC inhibition in atherosclerotic mice showed mixed results160–162, whereas inhibition or genetic deletion of HDAC3 or HDAC9 reduced atherosclerosis in mice163–165. Variants in HDAC9 have been associated with abdominal aortic calcification and ischaemic stroke in genome-wide association studies in humans166,167, highlighting the clinical potential of specific HDAC targeting in CVD.
Targeting metabolic rewiring is an alternative strategy because increased glucose metabolism in human and mouse haematopoietic stem and progenitor cells (HSPCs) dictates myeloid lineage commitment168. Glucose transporter 1 (GLUT1), a ubiquitously expressed glucose transporter, is a well-recognized target in other inflammatory conditions169. GLUT1 deficiency in bone marrow cells resulted in reduced HSPC proliferation, myelopoiesis and atherogenesis in mice170. However, further investigation of the effects of GLUT1 inhibition in humans is necessary, because patients with GLUT1 deficiency syndrome have neurological symptoms, such as epilepsy171.
Targeting CHIP
The discovery of CHIP has led to the identification of new potential targets. The most commonly occurring variants associated with CHIP are loss-of-function variants in DNTM3A, ASXL1 and TET2 and gain-of-function variants in JAK2 (JAK2V617F), that all result in growth and survival advantages in the cells carrying the gene variant172. Mice with TET2 deficiency or carrying the Jak2V617F variant showed accelerated atherogenesis30,153,173,174. Both macrophages from Tet2-knockout mice and peripheral blood monocytes from patients with aortic valve stenosis carrying a DNTMA3 or TET2 variant produce high levels of IL-1β and show NLRP3 inflammasome priming30,153,175. NLRP3 inflammasome inhibition by administration of MCC950 prevented TET2-dependent atherosclerosis progression in mice in vivo30,153. Similarly, clonal haematopoiesis driven by TET2 deficiency aggravated heart failure, cardiac dysfunction and obesity in mice, whereas NLRP3 inhibition with MCC950 protected against the development of heart failure and insulin sensitivity176–178. Activation of the absent in melanoma 2 (AIM2) inflammasome has been associated with Jak2V617F-driven atherosclerosis in mice. In a mouse model of Jak2V617F-driven atherosclerosis, deletion of the genes encoding for essential components that act downstream of the AIM2 inflammasome, such as caspase 1, caspase 11 and gasdermin D, induced a more stable plaque phenotype179 (Box 4). Taken together, the findings of these studies highlight the potential of targeting CHIP-driven inflammation with the use of NLRP3 or AIM2 inflammasome inhibitors.
JAK2 inhibitors could represent an alternative strategy for targeting inflammation in atherogenesis. Ruxolitinib and fedratinib are FDA-approved drugs for the treatment of myeloproliferative neoplasms and are currently being tested for use in other inflammatory conditions, such as RA180. Both drugs were effective in reducing inflammation and atherosclerosis in mouse and rabbit models of atherosclerosis174,181. Although treatment with the JAK1–JAK2 inhibitor ruxolitinib reduced atherosclerotic plaque size in mice with Jak2V617F-dependent atherosclerosis174,179, the treatment also increased necrotic core size and reduced cap thickness, resulting in an unstable plaque phenotype179. Therefore, a more specific JAK2 inhibitor, such as fedratinib, might be of interest in CVD.
Box 4 Eliciting an innate response: TLRs and inflammasomes.
TLRs
Toll-like receptors (TLRs) are a family of ten proteins in humans that recognize pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs), triggering an innate immune response10. Whereas the intracellular receptors TLR3 (ref.337) and TLR7 (ref.338) are involved in anti-atherogenic processes, the extracellular sensors TLR2 and TLR4 are thought to initiate the immune response in the arterial wall by recognizing modified lipoproteins in concert with scavenger receptors15,339, leading to cell activation and induction of pro-inflammatory cytokines, such as IL-1β, while also priming the inflammasome, which regulates IL-1β production340. TLR2 and TLR4 are significantly upregulated in human atherosclerotic tissue341 and on circulating monocytes from patients with acute coronary syndrome342, linking TLR levels to the risk of cardiovascular events338,343. Although TLR4 induces the most powerful responses of any TLR in pathogen-related situations, mutations and/or deletions of Tlr4 in experimental models of atherosclerosis produced mixed results, with a varying degree of reduction or no effect on atherosclerotic lesion size344. By contrast, deletion of Tlr2 consistently reduced lipid deposition and macrophage content in atherosclerotic lesions in mice345. Moreover, inflammation in human atherosclerosis has been shown to be driven by TLR2 via the signalling adaptor MyD88 (ref.217). Overall, this evidence strongly suggests that TLR2 is one of the most pro-atherogenic TLRs.
Inflammasomes
Inflammasomes are multimeric protein complexes that form in response to endogenous and exogenous danger signals, and promote pro-inflammatory cytokine production (including IL-1β and IL-18) and pyroptotic cell death346. The NLRP3 inflammasome is an important driver of lipid-driven vascular inflammation and atherosclerosis. Triggers for the activation of the NLRP3 inflammasome include potassium efflux, mitochondrial reactive oxygen species and cathepsin B346. All these triggers are likely to be present in the atheroma; however, a crucial driver of NLRP3 inflammasome activation in the arterial intima is cathepsin B activation downstream of cholesterol crystal, oxidized LDL and calcium phosphate crystal accumulation in phagocytic cells347,348.
A crucial role for the absent in melanoma 2 (AIM2) inflammasome in atherosclerosis is emerging. In atherosclerotic mice, AIM2 inflammasome activation resulted in the production of IL-1β and IL-18 accompanied by an unstable plaque phenotype349. By contrast, Aim2 deletion or pharmacological inhibition with an AIM2-antagonizing synthetic oligonucleotide increased plaque stability. Interestingly, AIM2 activation was also shown to be involved in atherosclerosis driven by clonal haematopoiesis179,350. AIM2-dependent inflammasome formation depends on the detection of cytosolic double-stranded DNA179, possibly downstream of atherosclerosis-associated necrosis and apoptosis. AIM2 activation then leads to the release of cellular contents into the extracellular space, thereby driving inflammation.
Under normal conditions, the levels of NLRP3 or AIM2 inflammasome complexes in cells are minimal351, and the inflammasomes remain in an inactive state through ubiquitination352. Both, NLRP3 and AIM2 inflammasomes require an initial priming signal, which promotes the expression of proteins involved in inflammasome signalling in a nuclear factor-κB-dependent fashion and stimulates inflammasome deubiquitination351,352. Subsequently, activation signals promote the assembly and activation of the inflammasome, enabling the proteolytic function of caspase 1 (ref.346). In addition to the processing of IL-1 cytokines, inflammasome activation facilitates the cleavage of gasdermin D, thereby inducing pore formation in the plasma membrane and pyroptosis, which is critical for the release of IL-1β and IL-18 to further promote inflammation in atherosclerosis353.
Targeting monocyte recruitment
Monocyte recruitment in atherosclerosis depends on the CCR2, CCR5 and CX3CR1 chemokine receptors182. Genetic deletion of Ccr2 or its ligand Ccl2 reduced bone marrow-derived monocytosis and atherosclerotic lesion size in mice42,183–185. Similarly, mice with MI treated with a small interfering RNA (siRNA) targeting Ccr2 had decreased monocyte recruitment to the infarct area and reduced disease severity186. In humans, genetic predisposition to elevated plasma CCL2 levels is associated with an increased risk of stroke, MI and CAD, and increased CCL2 levels in the blood and atherosclerotic plaques correlate with a higher risk of stroke and with markers of plaque destabilization187. MLN1202, a CCR2-blocking antibody, reduced hsCRP levels in patients at risk of atherosclerotic CVD188. Pharmacological inhibition of CCR5 with the FDA-approved CCR5 antagonist maraviroc reduced atherosclerosis in Ldlr−/− mice189,190. Interestingly, treatment with maraviroc also led to reduced atheroprogression in patients with HIV infection and high risk of CVD compared with baseline, as assessed by intima–media thickness191,192. However, given that circulating monocytes traffic into tissues during homeostasis, inflammation and inflammation resolution47,193, the effect of targeting monocyte recruitment on these processes will need monitoring.
Reprogramming inflammatory macrophages
Macrophage polarization is orchestrated by key master regulators, including nuclear factor-κB, the STAT family, peroxisome proliferator-activated receptor-γ (PPARγ) and the interferon regulatory factor (IRF) family194. Reprogramming pro-inflammatory macrophage populations that drive vascular inflammation towards homeostatic pro-resolving phenotypes could reduce disease burden. Pioglitazone is an FDA-approved PPARγ agonist that induces a pro-resolving macrophage phenotype by reducing pro-inflammatory cytokine production and promoting monocyte differentiation into alternatively activated macrophages195–197. In mice with atherosclerosis, administration of pioglitazone reduced macrophage content and increased plaque stability198,199. Clinical studies investigating the role of pioglitazone in patients with CVD and/or type 2 diabetes mellitus showed atheroprotective effects and a reduction of cardiovascular events with pioglitazone therapy200–203, highlighting the therapeutic potential of this drug in CVD.
In mouse models of CVD, global or myeloid-specific IRF5 deficiency reduced atherosclerosis and improved plaque stability204,205, and IRF5 inhibition with nanoparticles decreased myocardial infarct size206. The transcription factor IRF5 induces a pro-inflammatory phenotype in mouse and human macrophages207. Therefore, IRF5 is a promising therapeutic target in CVD. Inhibitors of IRF5 have proven to be therapeutically effective in mouse models of systemic lupus erythematosus208,209.
Targeting the inflammasomes
Selective inhibition of the NLRP3 inflammasome with MCC950 reduced atherosclerosis in hypercholesterolaemic or diabetic mice210,211. MCC950 has been tested in phase II trials in patients with RA, but the trials had to be discontinued owing to liver toxicity212. The interest in using NLRP3 inflammasome inhibitors for the treatment of chronic inflammatory and neuroinflammatory diseases is increasing and these agents are being tested in clinical trials213. The NLRP3 inflammasome inhibitor OLT1177 has been assessed in phase I–II clinical trials in patients with osteoarthritis214, acute gout213 or heart failure215 and has shown high tolerability. OLT1177 is also currently being tested in a study in patients with COVID-19 (ref.216).
Alternative approaches to targeting the inflammasome in atherosclerosis include the prevention of inflammasome priming with the use of TLR inhibitors217, targeting the AIM2 inflammasome179 and inhibition of caspase 1 (Box 4). The catalytic activity of caspase 1 is required to convert pro-IL-1β into its active form downstream of NLRP3 and AIM2. The caspase 1 inhibitor VX-765 reduces atherosclerosis in mice218. However, phase II trials of the caspase 1 inhibitors VX-740 and VX-765 in patients with psoriasis or epilepsy revealed drug-induced hepatotoxicity and further development was stopped219, highlighting the challenges presented by inhibition of inflammasomes.
Targeting the adaptive immune system
Immune recognition of LDL and oxLDL moieties leads to the generation of autoantibodies and oxLDL-reactive T cells14,220. Immunization with ApoB-derived antigens induces atheroprotective effects in mice and rabbits via diverse mechanisms including the induction of a humoral antibody response, Treg cell activation, suppression of CD4+ T cells and reduction of dendritic cell numbers in the plaque221–223. However, passive immunization with MLDL1278A, an anti-oxLDL antibody, added to lipid-lowering therapies did not reduce cardiovascular events in patients with stable atherosclerotic disease, as discussed above141. To improve the translation of ApoB-based immunization therapies from the preclinical to the clinical setting, Wolf and colleagues used in silico prediction methods to identify ApoB peptides that would bind to various major histocompatibility complex class II variants81. Using the in silico methods, the investigators identified 30 ApoB peptides that successfully induced a response in human T cells in vitro81.
Another approach to targeting adaptive immune cells is the direct targeting of atherogenic B cell subsets224. B cell depletion therapies are already in clinical use for the treatment of RA and multiple sclerosis, and studies in mice have shown that preferential B2 cell depletion with the use of an anti-CD20 antibody reduces atherosclerosis225,226. A single dose of rituximab, a B cell-depleting anti-CD20 antibody, was safe and efficiently depleted B cells in patients with acute STEMI227. Antibodies for B cell depletion targeting CD19 (blinatumomab and inebilizumab), CD22 (inotuzumab ozogamicin) or B cell maturation antigen (belantamab mafodotin and AMG420) have been approved or are currently in clinical development for the treatment of multiple sclerosis and cancer. Other promising strategies targeting B cells include: impairment of B cell survival and proliferation (with atacicept, belimumab, blisibimod and ianalumab), modulation of B cell receptor signalling (with acalabrutinib, epratuzumab and ibrutinib), antibody neutralization (with omalizumab), and the modulation of B cell co-stimulation (with abatacept)224,228.
Targeting co-stimulation pathways
Immune checkpoints are immune regulatory co-stimulatory molecules that provide stimulatory or inhibitory signals to adaptive and innate immune cells229. Immune checkpoints modulate the immune response in CVD229. In vivo studies in mice identified crucial co-stimulatory axes in atherosclerosis with the use of genetic deletion and agonistic and antagonist antibodies: activation of CD27–CD70, B and T lymphocyte attenuator (BTLA), CD200 receptor (CD200R)–CD200 and CD80/CD86–CTLA4 (cytotoxic T lymphocyte antigen 4) pathways or inhibition of CD40–CD40 ligand, OX40–OX40 ligand and CD30–CD30 ligand pathways might be beneficial therapeutic strategies in atherosclerosis230–237. Multiple immune checkpoint inhibitors and agonists targeting the above pathways are in clinical development for the treatment of cancer and RA238,239. In preclinical models, specific inhibition of tumour necrosis factor receptor-associated factor 6 (TRAF6), downstream of the pro-inflammatory CD40 signalling pathway, with small-molecule inhibitors resulted in plaque stabilization without inducing adverse effects and sparing host defence240. Similarly, CD200R expression is restricted to the myeloid compartment, making the CD200–CD200R pathway amenable for selective targeting of the monocyte–macrophage axis locally and in the bone marrow in CVD234.
Targeting the atherosclerotic plaque
Long-term immunosuppression might disrupt cardiovascular homeostasis and host defence241. Local delivery of drugs has been used in the clinic in the vascular field for many years with the use of drug-eluting stents containing sirolimus or paclitaxel, both of which have anti-inflammatory properties242. Furthermore, microneedle injections of dexamethasone in the adventitia prevents restenosis in patients who have undergone percutaneous transluminal angioplasty243.
An alternative strategy for minimizing the systemic adverse effects of off-target cell activation with systemic immunosuppressive approaches and improving accessibility to the cell type of interest is the use of cell-targeted delivery approaches. Nanoparticles have a high engagement with myeloid cells and can be modified to target specific subsets with ligand-decorated nanomaterials244. Nanoparticles have been used to target macrophages in several trials in patients with CVD245,246. Flores and colleagues used PEGylated, single-wall carbon nanotubes to deliver a downstream inhibitor of the anti-phagocytic CD47 pathway to lesional macrophages in mice, which resulted in a reduced plaque burden without toxic effects247. Administration of macrophage-targeted nanoparticles carrying siRNA against Camk2g increased plaque stability in mice owing to improved efferocytosis, leading to rebalancing of the immune system in atherosclerosis248 (Box 2). Nanoparticles decorated with collagen type IV accumulate in the atherosclerotic lesion shoulder and the use of these nanoparticles for the targeted delivery of IL-10 or the anti-inflammatory annexin A1 biomimetic Ac2-26 peptide stabilized atherosclerotic lesions in mice249,250. TRAF6 inhibitors or pioglitazone delivered with nanoparticles was also effective in increasing plaque stability in atherosclerotic mice198,240. These studies highlight the potential of modulating the immune system in CVD by specifically targeting atherosclerotic plaques to avoid toxic effects associated with systemic immunosuppression approaches.
Conclusions
Cardiovascular research lags behind oncology and rheumatology in recognizing the effects of chronic inflammation on CVD and translating inflammatory targets to human cardiovascular therapy. Although our understanding of the role of inflammation in atherosclerosis has improved substantially over the past two decades, the nuanced balance between pro-inflammatory and anti-inflammatory cells required for homeostasis remains elusive. To identify new therapeutic targets in atherosclerosis, we need to improve our interpretation of the determinants of this equilibrium. Single-cell biology approaches can accelerate clinical translation by facilitating the examination of immune signatures in patients with CVD. Identification of culprit cell types with the use of multiomics approaches could help identify the most suitable patient population for clinical trials and support target selection and informed decision-making in a clinical setting, moving towards personalized medicine. In addition, it is imperative to determine the window of opportunity for anti-inflammatory therapy in atherosclerosis, in which the benefits of immune system inhibition outweigh the systemic immunosuppressive effects. More targeted approaches using biologics or vaccination might allow specific targeting of atherosclerotic inflammation and thus minimize off-target effects. The development of mRNA vaccines has revolutionized the field of RNA-based therapeutics, extending the toolkit for vaccines against atherosclerosis and for previously ‘undruggable’ targets251. The association of CHIP with CVD risk exemplifies the importance of patient stratification beyond the use of traditional risk factors to define the patient population that will benefit from treatment. It is time to take inflammation seriously as a pathogenic driver of CVD and direct resources towards mechanistic and translational studies to find the cause of and a remedy for inflammation in this context. There has never been a more exciting time for research in cardiovascular inflammation.
Supplementary information
Acknowledgements
This article is dedicated to Prof. Attilio Maseri (1935–2021), who indicated the way for many of us to follow. The authors received funding from the British Heart Foundation (PG/18/1/33430 and PG/19/41/344), the European Commission under the Seventh Framework Programme (FP7/2007-2013, grant agreement number HEALTH-F2-2013-602114 (Athero-B-Cell), HEALTH-F2-2013-602222 (Athero-Flux), HEALTH.2012-1.2-1, contract number 305739 RiskyCAD, and (TAXINOMISIS) grant agreement H2020-SC1-2016-2017, 797788 STRIKING STREAKS), The Kennedy Trustees and the Novo Nordisk Foundation (NNF15CC0018346 and NNF0064142).
Author contributions
All the authors contributed substantially to all aspects of the article.
Competing interests
The authors declare no competing interests.
Footnotes
Peer review information
Nature Reviews Cardiology thanks C.J. Binder, who co-reviewed with F. Porsch; A. Abbate; K. Ley; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Suzanne E. Engelen, Alice J. B. Robinson, Yasemin-Xiomara Zurke.
Supplementary information
The online version contains supplementary material available at 10.1038/s41569-021-00668-4.
References
- 1.Libby P, Ridker PM, Maseri A. Inflammation and atherosclerosis. Circulation. 2002;105:1135–1143. doi: 10.1161/hc0902.104353. [DOI] [PubMed] [Google Scholar]
- 2.Arnett DK, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;140:e596–e646. doi: 10.1161/CIR.0000000000000678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Neumann FJ, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 2020;41:407–477. doi: 10.1093/eurheartj/ehz425. [DOI] [PubMed] [Google Scholar]
- 4.Timmis A, et al. European Society of Cardiology: cardiovascular disease statistics 2019. Eur. Heart J. 2020;41:12–85. doi: 10.1093/eurheartj/ehz859. [DOI] [PubMed] [Google Scholar]
- 5.Liuzzo G, et al. The prognostic value of C-reactive protein and serum amyloid a protein in severe unstable angina. N. Engl. J. Med. 1994;331:417–424. doi: 10.1056/NEJM199408183310701. [DOI] [PubMed] [Google Scholar]
- 6.Biasucci LM, et al. Elevated levels of interleukin-6 in unstable angina. Circulation. 1996;94:874–877. doi: 10.1161/01.cir.94.5.874. [DOI] [PubMed] [Google Scholar]
- 7.Galkina E, et al. Lymphocyte recruitment into the aortic wall before and during development of atherosclerosis is partially L-selectin dependent. J. Exp. Med. 2006;203:1273–1282. doi: 10.1084/jem.20052205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Roy P, Orecchioni M, Ley K. How the immune system shapes atherosclerosis: roles of innate and adaptive immunity. Nat. Rev. Immunol. 2021 doi: 10.1038/s41577-021-00584-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006;124:783–801. doi: 10.1016/j.cell.2006.02.015. [DOI] [PubMed] [Google Scholar]
- 10.O’Neill LAJ, Golenbock D, Bowie AG. The history of Toll-like receptors–redefining innate immunity. Nat. Rev. Immunol. 2013;13:453–460. doi: 10.1038/nri3446. [DOI] [PubMed] [Google Scholar]
- 11.Tabas I, Lichtman AH. Monocyte-macrophages and T cells in atherosclerosis. Immunity. 2017;47:621–634. doi: 10.1016/j.immuni.2017.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Moore KJ, Tabas I. Macrophages in the pathogenesis of atherosclerosis. Cell. 2011;145:341–355. doi: 10.1016/j.cell.2011.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tsiantoulas D, et al. APRIL limits atherosclerosis by binding to heparan sulfate proteoglycans. Nature. 2021;597:92–96. doi: 10.1038/s41586-021-03818-3. [DOI] [PubMed] [Google Scholar]
- 14.Stemme S, et al. T lymphocytes from human atherosclerotic plaques recognize oxidized low density lipoprotein. Proc. Natl Acad. Sci. USA. 1995;92:3893–3897. doi: 10.1073/pnas.92.9.3893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Stewart CR, et al. CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nat. Immunol. 2010;11:155–161. doi: 10.1038/ni.1836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Binder CJ, Papac-Milicevic N, Witztum JL. Innate sensing of oxidation-specific epitopes in health and disease. Nat. Rev. Immunol. 2016;16:485–497. doi: 10.1038/nri.2016.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Naghavi M, et al. From vulnerable plaque to vulnerable patient: a call for new definitions and risk assessment strategies: Part I. Circulation. 2003;108:1664–1672. doi: 10.1161/01.CIR.0000087480.94275.97. [DOI] [PubMed] [Google Scholar]
- 18.Zernecke A, et al. Meta-analysis of leukocyte diversity in atherosclerotic mouse aortas. Circ. Res. 2020;127:402–426. doi: 10.1161/CIRCRESAHA.120.316903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Depuydt MAC, et al. Microanatomy of the human atherosclerotic plaque by single-cell transcriptomics. Circ. Res. 2020;127:1437–1455. doi: 10.1161/CIRCRESAHA.120.316770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lin JD, et al. Single-cell analysis of fate-mapped macrophages reveals heterogeneity, including stem-like properties, during atherosclerosis progression and regression. JCI Insight. 2019;4:e124574. doi: 10.1172/jci.insight.124574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Winkels H, et al. Atlas of the immune cell repertoire in mouse atherosclerosis defined by single-cell RNA-sequencing and mass cytometry. Circ. Res. 2018;122:1675–1688. doi: 10.1161/CIRCRESAHA.117.312513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim K, et al. Transcriptome analysis reveals nonfoamy rather than foamy plaque macrophages are proinflammatory in atherosclerotic murine models. Circ. Res. 2018;123:1127–1142. doi: 10.1161/CIRCRESAHA.118.312804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.McArdle S, et al. Migratory and dancing macrophage subsets in atherosclerotic lesions. Circ. Res. 2019;125:1038–1051. doi: 10.1161/CIRCRESAHA.119.315175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cole JE, et al. Immune cell census in murine atherosclerosis: cytometry by time of flight illuminates vascular myeloid cell diversity. Cardiovasc. Res. 2018;114:1360–1371. doi: 10.1093/cvr/cvy109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fernandez DM, et al. Single-cell immune landscape of human atherosclerotic plaques. Nat. Med. 2019;25:1576–1588. doi: 10.1038/s41591-019-0590-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gu W, et al. Adventitial cell atlas of wt (wild type) and ApoE (apolipoprotein E)-deficient mice defined by single-cell RNA sequencing. Arterioscler. Thromb. Vasc. Biol. 2019;39:1055–1071. doi: 10.1161/ATVBAHA.119.312399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cochain C, et al. Single-cell RNA-seq reveals the transcriptional landscape and heterogeneity of aortic macrophages in murine atherosclerosis. Circ. Res. 2018;122:1661–1674. doi: 10.1161/CIRCRESAHA.117.312509. [DOI] [PubMed] [Google Scholar]
- 28.McAlpine CS, et al. Sleep modulates haematopoiesis and protects against atherosclerosis. Nature. 2019;566:383–387. doi: 10.1038/s41586-019-0948-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Robbins CS, et al. Extramedullary hematopoiesis generates Ly-6C high monocytes that infiltrate atherosclerotic lesions. Circulation. 2012;125:364–374. doi: 10.1161/CIRCULATIONAHA.111.061986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fuster JJ, et al. Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science. 2017;355:842–847. doi: 10.1126/science.aag1381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Soehnlein O, Libby P. Targeting inflammation in atherosclerosis–from experimental insights to the clinic. Nat. Rev. Drug Discov. 2021;20:589–610. doi: 10.1038/s41573-021-00198-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ridker PM, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 2017;377:1119–1131. doi: 10.1056/NEJMoa1707914. [DOI] [PubMed] [Google Scholar]
- 33.Tardif J-C, et al. Efficacy and safety of low-dose colchicine after myocardial infarction. N. Engl. J. Med. 2019;381:2497–2505. doi: 10.1056/NEJMoa1912388. [DOI] [PubMed] [Google Scholar]
- 34.Nidorf SM, et al. Colchicine in patients with chronic coronary disease. N. Engl. J. Med. 2020;383:1838–1847. doi: 10.1056/NEJMoa2021372. [DOI] [PubMed] [Google Scholar]
- 35.Full LE, Monaco C. Targeting inflammation as a therapeutic strategy in accelerated atherosclerosis in rheumatoid arthritis. Cardiovasc. Ther. 2011;29:231–242. doi: 10.1111/j.1755-5922.2010.00159.x. [DOI] [PubMed] [Google Scholar]
- 36.Drobni ZD, et al. Association between immune checkpoint inhibitors with cardiovascular events and atherosclerotic plaque. Circulation. 2020;142:2299–2311. doi: 10.1161/CIRCULATIONAHA.120.049981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Poels K, et al. Immune checkpoint inhibitor therapy aggravates T cell-driven plaque inflammation in atherosclerosis. JACC Cardiovasc. Oncol. 2020;2:599–610. doi: 10.1016/j.jaccao.2020.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ridker PM. How common is residual inflammatory risk? Circ. Res. 2017;120:617–619. doi: 10.1161/CIRCRESAHA.116.310527. [DOI] [PubMed] [Google Scholar]
- 39.Allahverdian S, Chaabane C, Boukais K, Francis GA, Bochaton-Piallat M-L. Smooth muscle cell fate and plasticity in atherosclerosis. Cardiovasc. Res. 2018;114:540–550. doi: 10.1093/cvr/cvy022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Doran AC, Meller N, McNamara CA. Role of smooth muscle cells in the initiation and early progression of atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2008;28:812–819. doi: 10.1161/ATVBAHA.107.159327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lievens D, von Hundelshausen P. Platelets in atherosclerosis. Thromb. Haemost. 2011;106:827–838. doi: 10.1160/TH11-08-0592. [DOI] [PubMed] [Google Scholar]
- 42.Combadière C, et al. Combined inhibition of CCL2, CX3CR1, and CCR5 abrogates Ly6Chi and Ly6Clo monocytosis and almost abolishes atherosclerosis in hypercholesterolemic mice. Circulation. 2008;117:1649–1657. doi: 10.1161/CIRCULATIONAHA.107.745091. [DOI] [PubMed] [Google Scholar]
- 43.Tacke F, et al. Monocyte subsets differentially employ CCR2, CCR5, and CX3CR1 to accumulate within atherosclerotic plaques. J. Clin. Invest. 2007;117:185–194. doi: 10.1172/JCI28549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Swirski FK, et al. Ly-6Chi monocytes dominate hypercholesterolemia-associated monocytosis and give rise to macrophages in atheromata. J. Clin. Invest. 2007;117:195–205. doi: 10.1172/JCI29950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shimizu Y, et al. Radiation exposure and circulatory disease risk: Hiroshima and Nagasaki atomic bomb survivor data, 1950-2003. BMJ. 2010;340:b5349. doi: 10.1136/bmj.b5349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Swirski FK, et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science. 2009;325:612–616. doi: 10.1126/science.1175202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rahman K, et al. Inflammatory Ly6Chi monocytes and their conversion to M2 macrophages drive atherosclerosis regression. J. Clin. Invest. 2017;127:2904–2915. doi: 10.1172/JCI75005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Woollard KJ, Geissmann F. Monocytes in atherosclerosis: subsets and functions. Nat. Rev. Cardiol. 2010;7:77–86. doi: 10.1038/nrcardio.2009.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Schloss MJ, Swirski FK, Nahrendorf M. Modifiable cardiovascular risk, hematopoiesis, and innate immunity. Circ. Res. 2020;126:1242–1259. doi: 10.1161/CIRCRESAHA.120.315936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Williams JW, et al. Limited proliferation capacity of aortic intima resident macrophages requires monocyte recruitment for atherosclerotic plaque progression. Nat. Immunol. 2020;21:1194–1204. doi: 10.1038/s41590-020-0768-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ensan S, et al. Self-renewing resident arterial macrophages arise from embryonic CX3CR1+ precursors and circulating monocytes immediately after birth. Nat. Immunol. 2016;17:159–168. doi: 10.1038/ni.3343. [DOI] [PubMed] [Google Scholar]
- 52.Robbins CS, et al. Local proliferation dominates lesional macrophage accumulation in atherosclerosis. Nat. Med. 2013;19:1166–1172. doi: 10.1038/nm.3258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lim HY, et al. Hyaluronan receptor LYVE-1-expressing macrophages maintain arterial tone through hyaluronan-mediated regulation of smooth muscle cell collagen. Immunity. 2018;49:326–341.e7. doi: 10.1016/j.immuni.2018.06.008. [DOI] [PubMed] [Google Scholar]
- 54.Park I, et al. C-type lectin receptor CLEC4A2 promotes tissue adaptation of macrophages and protects against atherosclerosis. Nat. Commun. 2022;13:215. doi: 10.1038/s41467-021-27862-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Weinberger T, et al. Ontogeny of arterial macrophages defines their functions in homeostasis and inflammation. Nat. Commun. 2020;11:4549. doi: 10.1038/s41467-020-18287-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Murray PJ. Macrophage polarization. Annu. Rev. Physiol. 2017;79:541–566. doi: 10.1146/annurev-physiol-022516-034339. [DOI] [PubMed] [Google Scholar]
- 57.Owsiany KM, Alencar GF, Owens GK. Revealing the origins of foam cells in atherosclerotic lesions. Arterioscler. Thromb. Vasc. Biol. 2019;39:836–838. doi: 10.1161/ATVBAHA.119.312557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Spann NJ, et al. Regulated accumulation of desmosterol integrates macrophage lipid metabolism and inflammatory responses. Cell. 2012;151:138–152. doi: 10.1016/j.cell.2012.06.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Voisin M, et al. Inhibiting LXRα phosphorylation in hematopoietic cells reduces inflammation and attenuates atherosclerosis and obesity in mice. Commun. Biol. 2021;4:420. doi: 10.1038/s42003-021-01925-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Jaitin DA, et al. Lipid-associated macrophages control metabolic homeostasis in a Trem2-dependent manner. Cell. 2019;178:686–698.e14. doi: 10.1016/j.cell.2019.05.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Deguchi JO, et al. Inflammation in atherosclerosis: visualizing matrix metalloproteinase action in macrophages in vivo. Circulation. 2006;114:55–62. doi: 10.1161/CIRCULATIONAHA.106.619056. [DOI] [PubMed] [Google Scholar]
- 62.Kojima Y, Weissman IL, Leeper NJ. The role of efferocytosis in atherosclerosis. Circulation. 2017;135:476–489. doi: 10.1161/CIRCULATIONAHA.116.025684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Merad M, Sathe P, Helft J, Miller J, Mortha A. The dendritic cell lineage: ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Annu. Rev. Immunol. 2013;31:563–604. doi: 10.1146/annurev-immunol-020711-074950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Yilmaz A, et al. Emergence of dendritic cells in rupture-prone regions of vulnerable carotid plaques. Atherosclerosis. 2004;176:101–110. doi: 10.1016/j.atherosclerosis.2004.04.027. [DOI] [PubMed] [Google Scholar]
- 65.Trogan E, et al. Gene expression changes in foam cells and the role of chemokine receptor CCR7 during atherosclerosis regression in ApoE-deficient mice. Proc. Natl Acad. Sci. USA. 2006;103:3781–3786. doi: 10.1073/pnas.0511043103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Weber C, et al. CCL17-expressing dendritic cells drive atherosclerosis by restraining regulatory T cell homeostasis in mice. J. Clin. Invest. 2011;121:2898–2910. doi: 10.1172/JCI44925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Choi JH, et al. Flt3 signaling-dependent dendritic cells protect against atherosclerosis. Immunity. 2011;35:819–831. doi: 10.1016/j.immuni.2011.09.014. [DOI] [PubMed] [Google Scholar]
- 68.Subramanian M, Thorp E, Hansson GK, Tabas I. Treg-mediated suppression of atherosclerosis requires MYD88 signaling in DCs. J. Clin. Invest. 2013;123:179–188. doi: 10.1172/JCI64617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Niessner A, et al. Pathogen-sensing plasmacytoid dendritic cells stimulate cytotoxic T-cell function in the atherosclerotic plaque through interferon-α. Circulation. 2006;114:2482–2489. doi: 10.1161/CIRCULATIONAHA.106.642801. [DOI] [PubMed] [Google Scholar]
- 70.MacRitchie N, et al. Plasmacytoid dendritic cells play a key role in promoting atherosclerosis in apolipoprotein e-deficient mice. Arterioscler. Thromb. Vasc. Biol. 2012;32:2569–2579. doi: 10.1161/ATVBAHA.112.251314. [DOI] [PubMed] [Google Scholar]
- 71.Silvestre-Roig C, Braster Q, Ortega-Gomez A, Soehnlein O. Neutrophils as regulators of cardiovascular inflammation. Nat. Rev. Cardiol. 2020;17:327–340. doi: 10.1038/s41569-019-0326-7. [DOI] [PubMed] [Google Scholar]
- 72.Zernecke A, et al. Protective role of CXC receptor 4/CXC ligand 12 unveils the importance of neutrophils in atherosclerosis. Circ. Res. 2008;102:209–217. doi: 10.1161/CIRCRESAHA.107.160697. [DOI] [PubMed] [Google Scholar]
- 73.Wang L, et al. ROS-producing immature neutrophils in giant cell arteritis are linked to vascular pathologies. JCI Insight. 2020;5:e139163. doi: 10.1172/jci.insight.139163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Warnatsch A, Ioannou M, Wang Q, Papayannopoulos V. Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science. 2015;349:316–320. doi: 10.1126/science.aaa8064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Silvestre-Roig C, et al. Externalized histone H4 orchestrates chronic inflammation by inducing lytic cell death. Nature. 2019;569:236–240. doi: 10.1038/s41586-019-1167-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Fuchs TA, et al. Extracellular DNA traps promote thrombosis. Proc. Natl Acad. Sci. USA. 2010;107:15880–15885. doi: 10.1073/pnas.1005743107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Soehnlein O, et al. Atherosclerosis: neutrophil-derived cathelicidin protects from neointimal hyperplasia. Sci. Transl. Med. 2011;3:103ra98. doi: 10.1126/scitranslmed.3002531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Saigusa R, Winkels H, Ley K. T cell subsets and functions in atherosclerosis. Nat. Rev. Cardiol. 2020;17:387–401. doi: 10.1038/s41569-020-0352-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ketelhuth DFJ, Hansson GK. Adaptive response of T and B cells in atherosclerosis. Circ. Res. 2016;118:668–678. doi: 10.1161/CIRCRESAHA.115.306427. [DOI] [PubMed] [Google Scholar]
- 80.Winkels H, Wolf D. Heterogeneity of T cells in atherosclerosis defined by single-cell RNA-sequencing and cytometry by time of flight. Arterioscler. Thromb. Vasc. Biol. 2021;41:549–563. doi: 10.1161/ATVBAHA.120.312137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wolf D, et al. athogenic autoimmunity in atherosclerosis evolves from initially protective apolipoprotein B100-reactive CD4+ T-regulatory cells. Circulation. 2020;142:1279–1293. doi: 10.1161/CIRCULATIONAHA.119.042863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Kyaw T, et al. Cytotoxic and proinflammatory CD8+ T lymphocytes promote development of vulnerable atherosclerotic plaques in ApoE-deficient mice. Circulation. 2013;127:1028–1039. doi: 10.1161/CIRCULATIONAHA.112.001347. [DOI] [PubMed] [Google Scholar]
- 83.van Duijn J, Kuiper J, Slütter B. The many faces of CD8+ T cells in atherosclerosis. Curr. Opin. Lipidol. 2018;29:411–416. doi: 10.1097/MOL.0000000000000541. [DOI] [PubMed] [Google Scholar]
- 84.Hwang Y, et al. Expansion of CD8+ T cells lacking the IL-6 receptor α chain in patients with coronary artery diseases (CAD) Atherosclerosis. 2016;249:44–51. doi: 10.1016/j.atherosclerosis.2016.03.038. [DOI] [PubMed] [Google Scholar]
- 85.Bergström I, Backteman K, Lundberg A, Ernerudh J, Jonasson L. Persistent accumulation of interferon-γ-producing CD8+CD56+ T cells in blood from patients with coronary artery disease. Atherosclerosis. 2012;224:515–520. doi: 10.1016/j.atherosclerosis.2012.07.033. [DOI] [PubMed] [Google Scholar]
- 86.Getz GS, Reardon CA. Natural killer T cells in atherosclerosis. Nat. Rev. Cardiol. 2017;14:304–314. doi: 10.1038/nrcardio.2017.2. [DOI] [PubMed] [Google Scholar]
- 87.Bobryshev YV, Lord RSA. Co-accumulation of dendritic cells and natural killer T cells within rupture-prone regions in human atherosclerotic plaques. J. Histochem. Cytochem. 2005;53:781–785. doi: 10.1369/jhc.4B6570.2005. [DOI] [PubMed] [Google Scholar]
- 88.Sage AP, Tsiantoulas D, Binder CJ, Mallat Z. The role of B cells in atherosclerosis. Nat. Rev. Cardiol. 2019;16:180–196. doi: 10.1038/s41569-018-0106-9. [DOI] [PubMed] [Google Scholar]
- 89.Kyaw T, et al. B1a B lymphocytes are atheroprotective by secreting natural IgM that increases IgM deposits and reduces necrotic cores in atherosclerotic lesions. Circ. Res. 2011;109:830–840. doi: 10.1161/CIRCRESAHA.111.248542. [DOI] [PubMed] [Google Scholar]
- 90.Strom AC, et al. B regulatory cells are increased in hypercholesterolaemic mice and protect from lesion development via IL-10. Thromb. Haemost. 2015;114:835–847. doi: 10.1160/TH14-12-1084. [DOI] [PubMed] [Google Scholar]
- 91.Nus M, et al. Marginal zone B cells control the response of follicular helper T cells to a high-cholesterol diet. Nat. Med. 2017;23:601–610. doi: 10.1038/nm.4315. [DOI] [PubMed] [Google Scholar]
- 92.Sage AP, et al. Regulatory B cell-specific interleukin-10 is dispensable for atherosclerosis development in mice. Arterioscler. Thromb. Vasc. Biol. 2015;35:1770–1773. doi: 10.1161/ATVBAHA.115.305568. [DOI] [PubMed] [Google Scholar]
- 93.Rosser EC, et al. Regulatory B cells are induced by gut microbiota-driven interleukin-1β and interleukin-6 production. Nat. Med. 2014;20:1334–1339. doi: 10.1038/nm.3680. [DOI] [PubMed] [Google Scholar]
- 94.Riggs JE, Lussier AM, Lee SK, Appel MC, Woodland RT. Differential radiosensitivity among B cell subpopulations. J. Immunol. 1988;141:1799–1807. [PubMed] [Google Scholar]
- 95.Nidorf SM, Eikelboom JW, Budgeon CA, Thompson PL. Low-dose colchicine for secondary prevention of cardiovascular disease. J. Am. Coll. Cardiol. 2013;61:404–410. doi: 10.1016/j.jacc.2012.10.027. [DOI] [PubMed] [Google Scholar]
- 96.Ridker PM, et al. Relationship of C-reactive protein reduction to cardiovascular event reduction following treatment with canakinumab: a secondary analysis from the CANTOS randomised controlled trial. Lancet. 2018;391:319–328. doi: 10.1016/S0140-6736(17)32814-3. [DOI] [PubMed] [Google Scholar]
- 97.Ridker PM, et al. Modulation of the interleukin-6 signalling pathway and incidence rates of atherosclerotic events and all-cause mortality: analyses from the Canakinumab Anti-Inflammatory Thrombosis Outcomes Study (CANTOS) Eur. Heart J. 2018;39:3499–3507. doi: 10.1093/eurheartj/ehy310. [DOI] [PubMed] [Google Scholar]
- 98.Ridker PM, et al. Effect of interleukin-1β inhibition with canakinumab on incident lung cancer in patients with atherosclerosis: exploratory results from a randomised, double-blind, placebo-controlled trial. Lancet. 2017;390:1833–1842. doi: 10.1016/S0140-6736(17)32247-X. [DOI] [PubMed] [Google Scholar]
- 99.Daskalov, I. & Valova-Ilieva, T. Management of acute pericarditis: treatment and follow-up. ESChttps://www.escardio.org/Journals/E-Journal-of-Cardiology-Practice/Volume-15/Management-of-acute-pericarditis-treatment-and-follow-up (2017).
- 100.Hui M, et al. The British Society for Rheumatology guideline for the management of gout. Rheumatology. 2017;56:1056–1059. doi: 10.1093/rheumatology/kex150. [DOI] [PubMed] [Google Scholar]
- 101.Paschke S, et al. Technical advance: inhibition of neutrophil chemotaxis by colchicine is modulated through viscoelastic properties of subcellular compartments. J. Leukoc. Biol. 2013;94:1091–1096. doi: 10.1189/jlb.1012510. [DOI] [PubMed] [Google Scholar]
- 102.Aaltonen KJ, et al. Systematic review and meta-analysis of the efficacy and safety of existing TNF blocking agents in treatment of rheumatoid arthritis. PLoS ONE. 2012;7:e30275. doi: 10.1371/journal.pone.0030275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Yamamoto-Furusho JK. Inflammatory bowel disease therapy: blockade of cytokines and cytokine signaling pathways. Curr. Opin. Gastroenterol. 2018;34:187–193. doi: 10.1097/MOG.0000000000000444. [DOI] [PubMed] [Google Scholar]
- 104.Reich K, et al. Tildrakizumab versus placebo or etanercept for chronic plaque psoriasis (reSURFACE 1 and reSURFACE 2): results from two randomised controlled, phase 3 trials. Lancet. 2017;390:276–288. doi: 10.1016/S0140-6736(17)31279-5. [DOI] [PubMed] [Google Scholar]
- 105.Grebe A, Hoss F, Latz E. NLRP3 inflammasome and the IL-1 pathway in atherosclerosis. Circ. Res. 2018;122:1722–1740. doi: 10.1161/CIRCRESAHA.118.311362. [DOI] [PubMed] [Google Scholar]
- 106.Vromman A, et al. Stage-dependent differential effects of interleukin-1 isoforms on experimental atherosclerosis. Eur. Heart J. 2019;40:2482–2491. doi: 10.1093/eurheartj/ehz008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Gomez D, et al. Interleukin-1β has atheroprotective effects in advanced atherosclerotic lesions of mice. Nat. Med. 2018;24:1418–1429. doi: 10.1038/s41591-018-0124-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Burzynski LC, et al. The coagulation and immune systems are directly linked through the activation of interleukin-1α by thrombin. Immunity. 2019;50:1033–1042.e6. doi: 10.1016/j.immuni.2019.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Galea J, et al. Interleukin-1β in coronary arteries of patients with ischemic heart disease. Arterioscler. Thromb. Vasc. Biol. 1996;16:1000–1006. doi: 10.1161/01.atv.16.8.1000. [DOI] [PubMed] [Google Scholar]
- 110.Abbate A, et al. Interleukin-1 blockade inhibits the acute inflammatory response in patients with ST-segment-elevation myocardial infarction. J. Am. Heart Assoc. 2020;9:e014941. doi: 10.1161/JAHA.119.014941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Morton AC, et al. The effect of interleukin-1 receptor antagonist therapy on markers of inflammation in non-ST elevation acute coronary syndromes: The MRC-ILA heart study. Eur. Heart J. 2015;36:377–384. doi: 10.1093/eurheartj/ehu272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.El Sayed H, Kerensky R, Stecher M, Mohanty P, Davies M. A randomized phase II study of Xilonix, a targeted therapy against interleukin 1α, for the prevention of superficial femoral artery restenosis after percutaneous revascularization. J. Vasc. Surg. 2016;63:133–141.e1. doi: 10.1016/j.jvs.2015.08.069. [DOI] [PubMed] [Google Scholar]
- 113.Ridker PM. Anticytokine agents: targeting interleukin signaling pathways for the treatment of atherothrombosis. Circ. Res. 2019;124:437–450. doi: 10.1161/CIRCRESAHA.118.313129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Zhang K, et al. Interleukin 6 destabilizes atherosclerotic plaques by downregulating prolyl-4-hydroxylase α1 via a mitogen-activated protein kinase and c-Jun pathway. Arch. Biochem. Biophys. 2012;528:127–133. doi: 10.1016/j.abb.2012.09.007. [DOI] [PubMed] [Google Scholar]
- 115.Ridker PM, Rifai N, Stampfer MJ, Hennekens CH. Plasma concentration of interleukin-6 and the risk of future myocardial infarction among apparently healthy men. Circulation. 2000;101:1767–1772. doi: 10.1161/01.cir.101.15.1767. [DOI] [PubMed] [Google Scholar]
- 116.Sarwar N, et al. Interleukin-6 receptor pathways in coronary heart disease: a collaborative meta-analysis of 82 studies. Lancet. 2012;379:1205–1213. doi: 10.1016/S0140-6736(11)61931-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Swerdlow DI, et al. The interleukin-6 receptor as a target for prevention of coronary heart disease: a mendelian randomisation analysis. Lancet. 2012;379:1214–1224. doi: 10.1016/S0140-6736(12)60110-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Broch K, et al. Randomized trial of interleukin-6 receptor inhibition in patients with acute ST-segment elevation myocardial infarction. J. Am. Coll. Cardiol. 2021;77:1845–1855. doi: 10.1016/j.jacc.2021.02.049. [DOI] [PubMed] [Google Scholar]
- 119.Kleveland O, et al. Effect of a single dose of the interleukin-6 receptor antagonist tocilizumab on inflammation and troponin T release in patients with non-ST-elevation myocardial infarction: a double-blind, randomized, placebo-controlled phase 2 trial. Eur. Heart J. 2016;37:2406–2413. doi: 10.1093/eurheartj/ehw171. [DOI] [PubMed] [Google Scholar]
- 120.Ridker PM, et al. IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (RESCUE): a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet. 2021;397:2060–2069. doi: 10.1016/S0140-6736(21)00520-1. [DOI] [PubMed] [Google Scholar]
- 121.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT05021835 (2021).
- 122.Tousoulis DI, Oikonomou E, Economou EK, Crea F, Kaski JC. Inflammatory cytokines in atherosclerosis: current therapeutic approaches. Eur. Heart J. 2016;37:1723–1735. doi: 10.1093/eurheartj/ehv759. [DOI] [PubMed] [Google Scholar]
- 123.Abbas A, et al. Sinterleukin 23 levels are increased in carotid atherosclerosis possible role for the interleukin 23/interleukin 17 axis. Stroke. 2015;46:793–799. doi: 10.1161/STROKEAHA.114.006516. [DOI] [PubMed] [Google Scholar]
- 124.Ohta H, et al. Disruption of tumor necrosis factor-α gene diminishes the development of atherosclerosis in ApoE-deficient mice. Atherosclerosis. 2005;180:11–17. doi: 10.1016/j.atherosclerosis.2004.11.016. [DOI] [PubMed] [Google Scholar]
- 125.Barath P, et al. Detection and localization of tumor necrosis factor in human atheroma. Am. J. Cardiol. 1990;65:297–302. doi: 10.1016/0002-9149(90)90291-8. [DOI] [PubMed] [Google Scholar]
- 126.Brånén L, et al. Inhibition of tumor necrosis factor-α reduces atherosclerosis in apolipoprotein E knockout mice. Arterioscler. Thromb. Vasc. Biol. 2004;24:2137–2142. doi: 10.1161/01.ATV.0000143933.20616.1b. [DOI] [PubMed] [Google Scholar]
- 127.Ridker PM, et al. Elevation of tumor necrosis factor-α and increased risk of recurrent coronary events after myocardial infarction. Circulation. 2000;101:2149–2153. doi: 10.1161/01.cir.101.18.2149. [DOI] [PubMed] [Google Scholar]
- 128.Chung ES, Packer M, Lo KH, Fasanmade AA, Willerson JT. Randomized, double-blind, placebo-controlled, pilot trial of infliximab, a chimeric monoclonal antibody to tumor necrosis factor-α, in patients with moderate-to-severe heart failure: results of the Anti-TNF Therapy Against Congestive Heart Failure (ATTACH) trial. Circulation. 2003;107:3133–3140. doi: 10.1161/01.CIR.0000077913.60364.D2. [DOI] [PubMed] [Google Scholar]
- 129.Mann DL, et al. Targeted anticytokine therapy in patients with chronic heart failure: results of the randomized etanercept worldwide evaluation (RENEWAL) Circulation. 2004;109:1594–1602. doi: 10.1161/01.CIR.0000124490.27666.B2. [DOI] [PubMed] [Google Scholar]
- 130.Gao Q, et al. A critical function of Th17 proinflammatory cells in the development of atherosclerotic plaque in mice. J. Immunol. 2010;185:5820–5827. doi: 10.4049/jimmunol.1000116. [DOI] [PubMed] [Google Scholar]
- 131.Langrish CL, et al. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 2005;201:233–240. doi: 10.1084/jem.20041257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Ma S, et al. The immunomodulatory effect of bone marrow stromal cells (BMSCs) on interleukin (IL)-23/IL-17-mediated ischemic stroke in mice. J. Neuroimmunol. 2013;257:28–35. doi: 10.1016/j.jneuroim.2013.01.007. [DOI] [PubMed] [Google Scholar]
- 133.Tzellos T, Kyrgidis A, Zouboulis CC. Re-evaluation of the risk for major adverse cardiovascular events in patients treated with anti-IL-12/23 biological agents for chronic plaque psoriasis: a meta-analysis of randomized controlled trials. J. Eur. Acad. Dermatol. Venereol. 2013;27:622–627. doi: 10.1111/j.1468-3083.2012.04500.x. [DOI] [PubMed] [Google Scholar]
- 134.Ryan C, et al. Association between biologic therapies for chronic plaque psoriasis and cardiovascular events: a meta-analysis of randomized controlled trials. JAMA. 2011;306:864–871. doi: 10.1001/jama.2011.1211. [DOI] [PubMed] [Google Scholar]
- 135.Ridker PM, et al. Low-dose methotrexate for the prevention of atherosclerotic events. N. Engl. J. Med. 2019;380:752–762. doi: 10.1056/NEJMoa1809798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.O’Donoghue ML, et al. Effect of losmapimod on cardiovascular outcomes in patients hospitalized with acute myocardial infarction: a randomized clinical trial. JAMA. 2016;315:1591–1599. doi: 10.1001/jama.2016.3609. [DOI] [PubMed] [Google Scholar]
- 137.Martin ED, Felice De Nicola G, Marber MS. New therapeutic targets in cardiology: p38 alpha mitogen-activated protein kinase for ischemic heart disease. Circulation. 2012;126:357–368. doi: 10.1161/CIRCULATIONAHA.111.071886. [DOI] [PubMed] [Google Scholar]
- 138.Dean JLE, Brook M, Clark AR, Saklatvala J. p38 Mitogen-activated protein kinase regulates cyclooxygenase-2 mRNA stability and transcription in lipopolysaccharide-treated human monocytes. J. Biol. Chem. 1999;274:264–269. doi: 10.1074/jbc.274.1.264. [DOI] [PubMed] [Google Scholar]
- 139.Elkhawad M, et al. Effects of p38 mitogen-activated protein kinase inhibition on vascular and systemic inflammation in patients with atherosclerosis. Imaging. 2012;5:911–922. doi: 10.1016/j.jcmg.2012.02.016. [DOI] [PubMed] [Google Scholar]
- 140.Newby LK, et al. Losmapimod, a novel p38 mitogen-activated protein kinase inhibitor, in non-ST-segment elevation myocardial infarction: a randomised phase 2 trial. Lancet. 2014;384:1187–1195. doi: 10.1016/S0140-6736(14)60417-7. [DOI] [PubMed] [Google Scholar]
- 141.Lehrer Graiwer J, et al. FDG-PET imaging for oxidized LDL in stable atherosclerotic disease: a phase II study of safety, tolerability, and anti-inflammatory activity. JACC Cardiovasc. Imaging. 2015;8:493–494. doi: 10.1016/j.jcmg.2014.06.021. [DOI] [PubMed] [Google Scholar]
- 142.Ćorović A, Wall C, Mason JC, Rudd JHF, Tarkin JM. Novel positron emission tomography tracers for imaging vascular inflammation. Curr. Cardiol. Rep. 2020;22:119. doi: 10.1007/s11886-020-01372-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Oikonomou EK, et al. Non-invasive detection of coronary inflammation using computed tomography and prediction of residual cardiovascular risk (the CRISP CT study): a post-hoc analysis of prospective outcome data. Lancet. 2018;392:929–939. doi: 10.1016/S0140-6736(18)31114-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Antonopoulos AS, et al. Detecting human coronary inflammation by imaging perivascular fat. Sci. Transl. Med. 2017;9:eaal2658. doi: 10.1126/scitranslmed.aal2658. [DOI] [PubMed] [Google Scholar]
- 145.Krittanawong C, et al. Machine learning prediction in cardiovascular diseases: a meta-analysis. Sci. Rep. 2020;10:16057. doi: 10.1038/s41598-020-72685-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Padmanabhan S, Tran TQB, Dominiczak AF. Artificial intelligence in hypertension: seeing through a glass darkly. Circ. Res. 2021;128:1100–1118. doi: 10.1161/CIRCRESAHA.121.318106. [DOI] [PubMed] [Google Scholar]
- 147.Greten FR, Grivennikov SI. Inflammation and cancer: triggers, mechanisms, and consequences. Immunity. 2019;51:27–41. doi: 10.1016/j.immuni.2019.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Koelwyn GJ, et al. Myocardial infarction accelerates breast cancer via innate immune reprogramming. Nat. Med. 2020;26:1452–1458. doi: 10.1038/s41591-020-0964-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Egen JG, Ouyang W, Wu LC. Human anti-tumor immunity: insights from immunotherapy clinical trials. Immunity. 2020;52:36–54. doi: 10.1016/j.immuni.2019.12.010. [DOI] [PubMed] [Google Scholar]
- 150.Leistner DM, et al. Differential immunological signature at the culprit site distinguishes acute coronary syndrome with intact from acute coronary syndrome with ruptured fibrous cap: results from the prospective translational OPTICO-ACS study. Eur. Heart J. 2020;41:3549–3560. doi: 10.1093/eurheartj/ehaa703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Hamers AAJ, et al. Human monocyte heterogeneity as revealed by high-dimensional mass cytometry. Arterioscler. Thromb. Vasc. Biol. 2019;39:25–36. doi: 10.1161/ATVBAHA.118.311022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Kott KA, et al. Single-cell immune profiling in coronary artery disease: the role of state-of-the-art immunophenotyping with mass cytometry in the diagnosis of atherosclerosis. J. Am. Heart Assoc. 2020;9:e017759. doi: 10.1161/JAHA.120.017759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Jaiswal S, et al. Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. N. Engl. J. Med. 2017;377:111–121. doi: 10.1056/NEJMoa1701719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Svensson E, et al. TET2-driven clonal hematopoiesis predicts enhanced response to canakinumab in the CANTOS trial: an exploratory analysis [abstract] Circulation. 2019;138(Suppl. 1):15111. [Google Scholar]
- 155.Pålsson-McDermott EM, O’Neill LAJ. Targeting immunometabolism as an anti-inflammatory strategy. Cell Res. 2020;30:300–314. doi: 10.1038/s41422-020-0291-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Edgar L, et al. Hyperglycemia induces trained immunity in macrophages and their precursors and promotes atherosclerosis. Circulation. 2021;144:961–982. doi: 10.1161/CIRCULATIONAHA.120.046464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Seijkens T, et al. Hypercholesterolemia-induced priming of hematopoietic stem and progenitor cells aggravates atherosclerosis. FASEB J. 2014;28:2202–2213. doi: 10.1096/fj.13-243105. [DOI] [PubMed] [Google Scholar]
- 158.Christ A, et al. Western diet triggers NLRP3-dependent innate immune reprogramming. Cell. 2018;172:162–175.e14. doi: 10.1016/j.cell.2017.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Netea MG, et al. Trained immunity: a program of innate immune memory in health and disease. Science. 2016;352:427. doi: 10.1126/science.aaf1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Bowes AJ, Khan MI, Shi Y, Robertson L, Werstuck GH. Valproate attenuates accelerated atherosclerosis in hyperglycemic ApoE-deficient mice: evidence in support of a role for endoplasmic reticulum stress and glycogen synthase kinase-3 in lesion development and hepatic steatosis. Am. J. Pathol. 2009;174:330–342. doi: 10.2353/ajpath.2009.080385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Manea SA, et al. Pharmacological inhibition of histone deacetylase reduces NADPH oxidase expression, oxidative stress and the progression of atherosclerotic lesions in hypercholesterolemic apolipoprotein E-deficient mice; potential implications for human atherosclerosis. Redox Biol. 2020;28:101338. doi: 10.1016/j.redox.2019.101338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Choi JH, et al. Trichostatin A exacerbates atherosclerosis in low density lipoprotein receptor-deficient mice. Arterioscler. Thromb. Vasc. Biol. 2005;25:2404–2409. doi: 10.1161/01.ATV.0000184758.07257.88. [DOI] [PubMed] [Google Scholar]
- 163.Hoeksema MA, et al. Targeting macrophage histone deacetylase 3 stabilizes atherosclerotic lesions. EMBO Mol. Med. 2014;6:1124–1132. doi: 10.15252/emmm.201404170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Cao Q, et al. Histone deacetylase 9 represses cholesterol efflux and alternatively activated macrophages in atherosclerosis development. Arterioscler. Thromb. Vasc. Biol. 2014;34:1871–1879. doi: 10.1161/ATVBAHA.114.303393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Asare Y, et al. Histone deacetylase 9 activates IKK to regulate atherosclerotic plaque vulnerability. Circ. Res. 2020;127:811–823. doi: 10.1161/CIRCRESAHA.120.316743. [DOI] [PubMed] [Google Scholar]
- 166.Malhotra R, et al. HDAC9 is implicated in atherosclerotic aortic calcification and affects vascular smooth muscle cell phenotype. Nat. Genet. 2019;51:1580–1587. doi: 10.1038/s41588-019-0514-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Bellenguez C, et al. Genome-wide association study identifies a variant in HDAC9 associated with large vessel ischemic stroke. Nat. Genet. 2012;44:328–333. doi: 10.1038/ng.1081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Oburoglu L, et al. Glucose and glutamine metabolism regulate human hematopoietic stem cell lineage specification. Cell Stem Cell. 2014;15:169–184. doi: 10.1016/j.stem.2014.06.002. [DOI] [PubMed] [Google Scholar]
- 169.Chen Z, Dudek J, Maack C, Hofmann U. Pharmacological inhibition of GLUT1 as a new immunotherapeutic approach after myocardial infarction. Biochem. Pharmacol. 2021;190:114597. doi: 10.1016/j.bcp.2021.114597. [DOI] [PubMed] [Google Scholar]
- 170.Sarrazy V, et al. Disruption of Glut1 in hematopoietic stem cells prevents myelopoiesis and enhanced glucose flux in atheromatous plaques of ApoE−/− mice. Circ. Res. 2016;118:1062–1077. doi: 10.1161/CIRCRESAHA.115.307599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Klepper J, et al. Glut1 deficiency syndrome (Glut1DS): state of the art in 2020 and recommendations of the international Glut1DS study group. Epilepsia Open. 2020;5:354–365. doi: 10.1002/epi4.12414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Jaiswal S, Libby P. Clonal haematopoiesis: connecting ageing and inflammation in cardiovascular disease. Nat. Rev. Cardiol. 2020;17:137–144. doi: 10.1038/s41569-019-0247-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Wang W, et al. Macrophage inflammation, erythrophagocytosis, and accelerated atherosclerosis in JAK2V617F mice. Circ. Res. 2018;123:E35–E47. doi: 10.1161/CIRCRESAHA.118.313283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Tang Y, et al. Inhibition of JAK2 suppresses myelopoiesis and atherosclerosis in ApoE−/− mice. Cardiovasc. Drugs Ther. 2020;34:145–152. doi: 10.1007/s10557-020-06943-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Abplanalp WT, et al. Association of clonal hematopoiesis of indeterminate potential with inflammatory gene expression in patients with severe degenerative aortic valve stenosis or chronic postischemic heart failure. JAMA Cardiol. 2020;5:1170–1175. doi: 10.1001/jamacardio.2020.2468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Sano S, et al. Tet2-mediated clonal hematopoiesis accelerates heart failure through a mechanism involving the IL-1β/NLRP3 INFLAMMASOME. J. Am. Coll. Cardiol. 2018;71:875–886. doi: 10.1016/j.jacc.2017.12.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Wang Y, et al. Tet2-mediated clonal hematopoiesis in nonconditioned mice accelerates age-associated cardiac dysfunction. JCI Insight. 2020;5:e135204. doi: 10.1172/jci.insight.135204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Fuster JJ, et al. TET2-loss-of-function-driven clonal hematopoiesis exacerbates experimental insulin resistance in aging and obesity. Cell Rep. 2020;33:108326. doi: 10.1016/j.celrep.2020.108326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Fidler TP, et al. The AIM2 inflammasome exacerbates atherosclerosis in clonal haematopoiesis. Nature. 2021;592:296–301. doi: 10.1038/s41586-021-03341-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Schwartz DM, et al. JAK inhibition as a therapeutic strategy for immune and inflammatory diseases. Nat. Rev. Drug Discov. 2017;16:843–862. doi: 10.1038/nrd.2017.201. [DOI] [PubMed] [Google Scholar]
- 181.Yang X, et al. Inhibition of JAK2/STAT3/SOCS3 signaling attenuates atherosclerosis in rabbit. BMC Cardiovasc. Disord. 2020;20:133. doi: 10.1186/s12872-020-01391-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Hilgendorf I, Swirski FK, Robbins CS. Monocyte fate in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2015;35:272–279. doi: 10.1161/ATVBAHA.114.303565. [DOI] [PubMed] [Google Scholar]
- 183.Soehnlein O, et al. Distinct functions of chemokine receptor axes in the atherogenic mobilization and recruitment of classical monocytes. EMBO Mol. Med. 2013;5:471–481. doi: 10.1002/emmm.201201717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Boring L, Gosling J, Cleary M, Charo IF. Decreased lesion formation in CCR2−/− mice reveals a role for chemokines in the initiation of atherosclerosis. Nature. 1998;394:894–897. doi: 10.1038/29788. [DOI] [PubMed] [Google Scholar]
- 185.Gu L, et al. Absence of monocyte chemoattractant protein-1 reduces atherosclerosis in low density lipoprotein receptor-deficient mice. Mol. Cell. 1998;2:275–281. doi: 10.1016/s1097-2765(00)80139-2. [DOI] [PubMed] [Google Scholar]
- 186.Majmudar MD, et al. Monocyte-directed RNAi targeting CCR2 improves infarct healing in atherosclerosis-prone mice. Circulation. 2013;127:2038–2046. doi: 10.1161/CIRCULATIONAHA.112.000116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Georgakis MK, et al. Monocyte-chemoattractant protein-1 levels in human atherosclerotic lesions associate with plaque vulnerability. Arterioscler. Thromb. Vasc. Biol. 2021;41:2038–2048. doi: 10.1161/ATVBAHA.121.316091. [DOI] [PubMed] [Google Scholar]
- 188.Gilbert J, et al. Effect of CC chemokine receptor 2 CCR2 blockade on serum C-reactive protein in individuals at atherosclerotic risk and with a single nucleotide polymorphism of the monocyte chemoattractant protein-1 promoter region. Am. J. Cardiol. 2011;107:906–911. doi: 10.1016/j.amjcard.2010.11.005. [DOI] [PubMed] [Google Scholar]
- 189.Cipriani S, et al. Efficacy of the CCR5 antagonist maraviroc in reducing early, ritonavir-induced atherogenesis and advanced plaque progression in mice. Circulation. 2013;127:2114–2124. doi: 10.1161/CIRCULATIONAHA.113.001278. [DOI] [PubMed] [Google Scholar]
- 190.Veillard NR, et al. Antagonism of RANTES receptors reduces atherosclerotic plaque formation in mice. Circ. Res. 2004;94:253–261. doi: 10.1161/01.RES.0000109793.17591.4E. [DOI] [PubMed] [Google Scholar]
- 191.Maggi P, et al. Effects of therapy with maraviroc on the carotid intima media thickness in HIV-1/HCV co-infected patients. In Vivo. 2017;31:125–132. doi: 10.21873/invivo.11035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Francisci D, et al. Maraviroc intensification modulates atherosclerotic progression in HIV-suppressed patients at high cardiovascular risk. A randomized, crossover pilot study. Open Forum Infect. Dis. 2019;6:ofz112. doi: 10.1093/ofid/ofz112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Shi C, Pamer EG. Monocyte recruitment during infection and inflammation. Nat. Rev. Immunol. 2011;11:762–774. doi: 10.1038/nri3070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Lawrence T, Natoli G. Transcriptional regulation of macrophage polarization: enabling diversity with identity. Nat. Rev. Immunol. 2011;11:750–761. doi: 10.1038/nri3088. [DOI] [PubMed] [Google Scholar]
- 195.Smith, U. Pioglitazone: mechanism of action. Int. J. Clin. Pract. Suppl. (121), 13–18 (2001). [PubMed]
- 196.Rigamonti E, Chinetti-Gbaguidi G, Staels B. Regulation of macrophage functions by PPAR-α, PPAR-γ, and LXRs in mice and men. Arterioscler. Thromb. Vasc. Biol. 2008;28:1050–1059. doi: 10.1161/ATVBAHA.107.158998. [DOI] [PubMed] [Google Scholar]
- 197.Bouhlel MA, et al. PPARγ activation primes human monocytes into alternative M2 macrophages with anti-inflammatory properties. Cell Metab. 2007;6:137–143. doi: 10.1016/j.cmet.2007.06.010. [DOI] [PubMed] [Google Scholar]
- 198.Nakashiro S, et al. Pioglitazone-incorporated nanoparticles prevent plaque destabilization and rupture by regulating monocyte/macrophage differentiation in ApoE−/− mice. Arterioscler. Thromb. Vasc. Biol. 2016;36:491–500. doi: 10.1161/ATVBAHA.115.307057. [DOI] [PubMed] [Google Scholar]
- 199.Chang K, et al. Pioglitazone suppresses inflammation in vivo in murine carotid atherosclerosis: novel detection by dual-target fluorescence molecular imaging. Arterioscler. Thromb. Vasc. Biol. 2010;30:1933–1939. doi: 10.1161/ATVBAHA.110.206342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Pfützner A, et al. Improvement of cardiovascular risk markers by pioglitazone is independent from glycemic control: results from the pioneer study. J. Am. Coll. Cardiol. 2005;45:1925–1931. doi: 10.1016/j.jacc.2005.03.041. [DOI] [PubMed] [Google Scholar]
- 201.Erdmann E, et al. The effect of pioglitazone on recurrent myocardial infarction in 2,445 patients with type 2 diabetes and previous myocardial infarction. results from the PROactive (PROactive 05) study. J. Am. Coll. Cardiol. 2007;49:1772–1780. doi: 10.1016/j.jacc.2006.12.048. [DOI] [PubMed] [Google Scholar]
- 202.Langenfeld MR, et al. Pioglitazone decreases carotid intima-media thickness independently of glycemic control in patients with type 2 diabetes mellitus: results from a controlled randomized study. Circulation. 2005;111:2525–2531. doi: 10.1161/01.CIR.0000165072.01672.21. [DOI] [PubMed] [Google Scholar]
- 203.de Jong M, van der Worp HB, van der Graaf Y, Visseren FLJ, Westerink J. Pioglitazone and the secondary prevention of cardiovascular disease. A meta-analysis of randomized-controlled trials. Cardiovasc. Diabetol. 2017;16:134. doi: 10.1186/s12933-017-0617-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Seneviratne AN, et al. Interferon regulatory factor 5 controls necrotic core formation in atherosclerotic lesions by impairing efferocytosis. Circulation. 2017;136:1140–1154. doi: 10.1161/CIRCULATIONAHA.117.027844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Leipner J, et al. Myeloid cell-specific Irf5 deficiency stabilizes atherosclerotic plaques in Apoe−/− mice. Mol. Metab. 2021;53:101250. doi: 10.1016/j.molmet.2021.101250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Courties G, et al. In vivo silencing of the transcription factor IRF5 reprograms the macrophage phenotype and improves infarct healing. J. Am. Coll. Cardiol. 2014;63:1556–1566. doi: 10.1016/j.jacc.2013.11.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Krausgruber T, et al. IRF5 promotes inflammatory macrophage polarization and TH1-TH17 responses. Nat. Immunol. 2011;12:231–238. doi: 10.1038/ni.1990. [DOI] [PubMed] [Google Scholar]
- 208.Song S, et al. Inhibition of IRF5 hyperactivation protects from lupus onset and severity. J. Clin. Invest. 2020;130:6700–6717. doi: 10.1172/JCI120288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Ban T, et al. Genetic and chemical inhibition of IRF5 suppresses pre-existing mouse lupus-like disease. Nat. Commun. 2021;12:4379. doi: 10.1038/s41467-021-24609-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Sharma A, et al. Specific NLRP3 inhibition protects against diabetes-associated atherosclerosis. Diabetes. 2021;70:772–787. doi: 10.2337/db20-0357. [DOI] [PubMed] [Google Scholar]
- 211.Van Der Heijden T, et al. NLRP3 inflammasome inhibition by MCC950 reduces atherosclerotic lesion development in apolipoprotein e-deficient mice–brief report. Arterioscler. Thromb. Vasc. Biol. 2017;37:1457–1461. doi: 10.1161/ATVBAHA.117.309575. [DOI] [PubMed] [Google Scholar]
- 212.Mangan MSJ, et al. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat. Rev. Drug Discov. 2018;17:588–606. doi: 10.1038/nrd.2018.97. [DOI] [PubMed] [Google Scholar]
- 213.Klück V, et al. Dapansutrile, an oral selective NLRP3 inflammasome inhibitor, for treatment of gout flares: an open-label, dose-adaptive, proof-of-concept, phase 2a trial. Lancet Rheumatol. 2020;2:e270–e280. doi: 10.1016/s2665-9913(20)30065-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01768975 (2014).
- 215.Wohlford GF, et al. Phase 1B, randomized, double-blinded, dose escalation, single-center, repeat dose safety and pharmacodynamics study of the oral NLRP3 indibitor dapansutrile in subjects with NYHA II-III systolic heart failure. J. Cardiovasc. Pharmacol. 2020;77:49–60. doi: 10.1097/FJC.0000000000000931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT04540120 (2021).
- 217.Monaco C, et al. Toll-like receptor-2 mediates inflammation and matrix degradation in human atherosclerosis. Circulation. 2009;120:2462–2469. doi: 10.1161/CIRCULATIONAHA.109.851881. [DOI] [PubMed] [Google Scholar]
- 218.Li Y, et al. VX-765 attenuates atherosclerosis in ApoE deficient mice by modulating VSMCs pyroptosis. Exp. Cell Res. 2020;389:111847. doi: 10.1016/j.yexcr.2020.111847. [DOI] [PubMed] [Google Scholar]
- 219.MacKenzie SH, Schipper JL, Clark AC. The potential for caspases in drug discovery. Curr. Opin. Drug Discov. Dev. 2010;13:568–576. [PMC free article] [PubMed] [Google Scholar]
- 220.Nilsson J, Hansson GK. Vaccination strategies and immune modulation of atherosclerosis. Circ. Res. 2020;126:1281–1296. doi: 10.1161/CIRCRESAHA.120.315942. [DOI] [PubMed] [Google Scholar]
- 221.Chyu KY, et al. CD8+ T cells mediate the athero-protective effect of immunization with an ApoB-100 peptide. PLoS ONE. 2012;7:e30780. doi: 10.1371/journal.pone.0030780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Dunér P, et al. Antibodies against apoB100 peptide 210 inhibit atherosclerosis in apoE−/− mice. Sci. Rep. 2021;11:9022. doi: 10.1038/s41598-021-88430-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Herbin O, et al. Regulatory T-cell response to apolipoprotein B100-derived peptides reduces the development and progression of atherosclerosis in mice. Arterioscler. Thromb. Vasc. Biol. 2012;32:605–612. doi: 10.1161/ATVBAHA.111.242800. [DOI] [PubMed] [Google Scholar]
- 224.Pattarabanjird T, Li C, McNamara C. B cells in atherosclerosis: mechanisms and potential clinical applications. JACC Basic Transl. Sci. 2021;6:546–563. doi: 10.1016/j.jacbts.2021.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Ait-Oufella H, et al. B cell depletion reduces the development of atherosclerosis in mice. J. Exp. Med. 2010;207:1579–1587. doi: 10.1084/jem.20100155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Kyaw T, et al. Conventional B2 B cell depletion ameliorates whereas its adoptive transfer aggravates atherosclerosis. J. Immunol. 2010;185:4410–4419. doi: 10.4049/jimmunol.1000033. [DOI] [PubMed] [Google Scholar]
- 227.Zhao TX, et al. Rituximab in patients with acute ST-elevation myocardial infarction: an experimental medicine safety study. Cardiovasc. Res. 2021 doi: 10.1093/cvr/cvab113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Porsch F, Binder CJ. Impact of B-cell-targeted therapies on cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 2019;39:1705–1714. doi: 10.1161/ATVBAHA.119.311996. [DOI] [PubMed] [Google Scholar]
- 229.Kusters PJH, Lutgens E, Seijkens TTP. Exploring immune checkpoints as potential therapeutic targets in atherosclerosis. Cardiovasc. Res. 2018;114:368–377. doi: 10.1093/cvr/cvx248. [DOI] [PubMed] [Google Scholar]
- 230.Foks AC, et al. Interruption of the OX40–OX40 ligand pathway in LDL receptor-deficient mice causes regression of atherosclerosis. J. Immunol. 2013;191:4573–4580. doi: 10.4049/jimmunol.1200708. [DOI] [PubMed] [Google Scholar]
- 231.Lutgens E, et al. Requirement for CD154 in the progression of atherosclerosis. Nat. Med. 1999;5:1313–1316. doi: 10.1038/15271. [DOI] [PubMed] [Google Scholar]
- 232.Foks AC, et al. Interference of the CD30–CD30L pathway reduces atherosclerosis development. Arterioscler. Thromb. Vasc. Biol. 2012;32:2862–2868. doi: 10.1161/ATVBAHA.112.300509. [DOI] [PubMed] [Google Scholar]
- 233.Winkels H, et al. CD27 co-stimulation increases the abundance of regulatory T cells and reduces atherosclerosis in hyperlipidaemic mice. Eur. Heart J. 2017;38:3590–3599. doi: 10.1093/eurheartj/ehx517. [DOI] [PubMed] [Google Scholar]
- 234.Kassiteridi C, et al. CD200 limits monopoiesis and monocyte recruitment in atherosclerosis. Circ. Res. 2021;129:280–295. doi: 10.1161/CIRCRESAHA.119.316062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Poels K, et al. Antibody-mediated inhibition of CTLA4 aggravates atherosclerotic plaque inflammation and progression in hyperlipidemic mice. Cells. 2020;9:1987. doi: 10.3390/cells9091987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Schönbeck U, Sukhova GK, Shimizu K, Mach F, Libby P. Inhibition of CD40 signaling limits evolution of established atherosclerosis in mice. Proc. Natl Acad. Sci. USA. 2000;97:7458–7463. doi: 10.1073/pnas.97.13.7458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Douna H, et al. B- and T-lymphocyte attenuator stimulation protects against atherosclerosis by regulating follicular B cells. Cardiovasc. Res. 2020;116:295–305. doi: 10.1093/cvr/cvz129. [DOI] [PubMed] [Google Scholar]
- 238.Waldman AD, Fritz JM, Lenardo MJ. A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat. Rev. Immunol. 2020;20:651–668. doi: 10.1038/s41577-020-0306-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Genovese MC, et al. Abatacept for rheumatoid arthritis refractory to tumor necrosis factor α inhibition. N. Engl. J. Med. 2005;353:1114–1123. doi: 10.1056/NEJMoa050524. [DOI] [PubMed] [Google Scholar]
- 240.Seijkens TTP, et al. Targeting CD40-induced TRAF6 signaling in macrophages reduces atherosclerosis. J. Am. Coll. Cardiol. 2018;71:527–542. doi: 10.1016/j.jacc.2017.11.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Giugliano GR, Giugliano RP, Gibson CM, Kuntz RE. Meta-analysis of corticosteroid treatment in acute myocardial infarction. Am. J. Cardiol. 2003;91:1055–1059. doi: 10.1016/s0002-9149(03)00148-6. [DOI] [PubMed] [Google Scholar]
- 242.Torii S, et al. Drug-eluting coronary stents: insights from preclinical and pathology studies. Nat. Rev. Cardiol. 2020;17:37–51. doi: 10.1038/s41569-019-0234-x. [DOI] [PubMed] [Google Scholar]
- 243.Razavi MK, Donohoe D, D’Agostino RB, Jaff MR, Adams G. Adventitial drug delivery of dexamethasone to improve primary patency in the treatment of superficial femoral and popliteal artery disease: 12-month results from the DANCE clinical trial. JACC Cardiovasc. Interv. 2018;11:921–931. doi: 10.1016/j.jcin.2017.12.015. [DOI] [PubMed] [Google Scholar]
- 244.Teunissen AJP, et al. Embracing nanomaterials’ interactions with the innate immune system. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2021;13:e1719. doi: 10.1002/wnan.1719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.van der Valk FM, et al. Prednisolone-containing liposomes accumulate in human atherosclerotic macrophages upon intravenous administration. Nanomed. Nanotechnol. Biol. Med. 2015;11:1039–1046. doi: 10.1016/j.nano.2015.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Fitzgerald K, et al. A highly durable RNAi therapeutic inhibitor of PCSK9. N. Engl. J. Med. 2017;376:41–51. doi: 10.1056/NEJMoa1609243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Flores AM, et al. Pro-efferocytic nanoparticles are specifically taken up by lesional macrophages and prevent atherosclerosis. Nat. Nanotechnol. 2020;15:154–161. doi: 10.1038/s41565-019-0619-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Tao W, et al. SiRNA nanoparticles targeting CaMKIIγ in lesional macrophages improve atherosclerotic plaque stability in mice. Sci. Transl. Med. 2020;12:eaay1063. doi: 10.1126/scitranslmed.aay1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Fredman G, et al. Targeted nanoparticles containing the proresolving peptide Ac2-26 protect against advanced atherosclerosis in hypercholesterolemic mice. Sci. Transl. Med. 2015;7:275ra20. doi: 10.1126/scitranslmed.aaa1065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Kamaly N, et al. Targeted interleukin-10 nanotherapeutics developed with a microfluidic chip enhance resolution of inflammation in advanced atherosclerosis. ACS Nano. 2016;10:5280–5292. doi: 10.1021/acsnano.6b01114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Tsimikas S. RNA-targeted therapeutics for lipid disorders. Curr. Opin. Lipidol. 2018;29:459–466. doi: 10.1097/MOL.0000000000000549. [DOI] [PubMed] [Google Scholar]
- 252.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02648464 (2020).
- 253.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02874287 (2021).
- 254.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT03113773 (2021).
- 255.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT04241601 (2020).
- 256.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT04762472 (2021).
- 257.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT04616872 (2020).
- 258.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT04350216 (2020).
- 259.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT04148833 (2020).
- 260.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT04610892 (2021).
- 261.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT03048825 (2020).
- 262.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02898610 (2017).
- 263.Jonasson L, Holm J, Skalli O, Gabbiani G, Hansson GK. Expression of class II transplantation antigen on vascular smooth muscle cells in human atherosclerosis. J. Clin. Invest. 1985;76:125–131. doi: 10.1172/JCI111934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Hansson GK, Jonasson L, Holm J, Claesson-Welsh L. Class II MHC antigen expression in the atherosclerotic plaque: smooth muscle cells express HLA-DR, HLA-DQ and the invariant gamma chain. Clin. Exp. Immunol. 1986;64:261–268. [PMC free article] [PubMed] [Google Scholar]
- 265.Vedeler CA, Nyland H, Matre R. In situ characterization of the foam cells in early human atherosclerotic lesions. Acta Pathol. Microbiol. Immunol. Scand. C. 1984;92:133–137. doi: 10.1111/j.1699-0463.1984.tb00064.x. [DOI] [PubMed] [Google Scholar]
- 266.Aqel NM, Ball RY, Waldmann H, Mitchinson MJ. Identification of macrophages and smooth muscle cells in human atherosclerosis using monoclonal antibodies. J. Pathol. 1985;146:197–204. doi: 10.1002/path.1711460306. [DOI] [PubMed] [Google Scholar]
- 267.Hansson GK, Holm J, Jonasson L. Detection of activated T lymphocytes in the human atherosclerotic plaque. Am. J. Pathol. 1989;135:169–175. [PMC free article] [PubMed] [Google Scholar]
- 268.Jonasson L, Holm J, Skalli O, Bondjers G, Hansson GK. Regional accumulations of T cells, macrophages, and smooth muscle cells in the human atherosclerotic plaque. Arteriosclerosis. 1986;6:131–138. doi: 10.1161/01.atv.6.2.131. [DOI] [PubMed] [Google Scholar]
- 269.Emeson EE, Robertson AL. T lymphocytes in aortic and coronary intimas: their potential role in atherogenesis. Am. J. Pathol. 1988;130:369–376. [PMC free article] [PubMed] [Google Scholar]
- 270.Amento EP, Ehsani N, Palmer H, Libby P. Cytokines and growth factors positively and negatively regulate interstitial collagen gene expression in human vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 1991;11:1223–1230. doi: 10.1161/01.atv.11.5.1223. [DOI] [PubMed] [Google Scholar]
- 271.Warner SJC, Libby P. Human vascular smooth muscle cells. Target for and source of tumor necrosis factor. J. Immunol. 1989;142:100–109. [PubMed] [Google Scholar]
- 272.Warner SJC, Auger KR, Libby P. Human interleukin 1 induces interleukin I gene expression in human vascular smooth muscle cells. J. Exp. Med. 1987;165:1316–1331. doi: 10.1084/jem.165.5.1316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Geng YJ, Wu Q, Muszynski M, Hansson GK, Libby P. Apoptosis of vascular smooth muscle cells induced by in vitro stimulation with interferon-γ, tumor necrosis factor-α, and interleukin-1β. Arterioscler. Thromb. Vasc. Biol. 1996;16:19–27. doi: 10.1161/01.atv.16.1.19. [DOI] [PubMed] [Google Scholar]
- 274.De Villiers WJS, et al. Macrophage phenotype in mice deficient in both macrophage-colony- stimulating factor (Op) and apolipoprotein E. Arterioscler. Thromb. Vasc. Biol. 1998;18:631–640. doi: 10.1161/01.atv.18.4.631. [DOI] [PubMed] [Google Scholar]
- 275.Berk BC, Weintraub WS, Alexander RW. Elevation of C-reactive protein in ‘active’ coronary artery disease. Am. J. Cardiol. 1990;65:168–172. doi: 10.1016/0002-9149(90)90079-g. [DOI] [PubMed] [Google Scholar]
- 276.Rajavashisth T, et al. Heterozygous osteopetrotic (op) mutation reduces atherosclerosis in LDL receptor-deficient mice. J. Clin. Invest. 1998;101:2702–2710. doi: 10.1172/JCI119891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Smith JD, et al. Decreased atherosclerosis in mice deficient in both macrophage colony-stimulating factor (op) and apolipoprotein E. Proc. Natl Acad. Sci. USA. 1995;92:8264–8268. doi: 10.1073/pnas.92.18.8264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Salonen JT, et al. Autoantibody against oxidised LDL and progression of carotid atherosclerosis. Lancet. 1992;339:883–887. doi: 10.1016/0140-6736(92)90926-t. [DOI] [PubMed] [Google Scholar]
- 279.The Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature. 2007;447:661–678. doi: 10.1038/nature05911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Samani NJ, et al. Genomewide association analysis of coronary artery disease. N. Engl. J. Med. 2007;357:443–453. doi: 10.1056/NEJMoa072366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Roman MJ, et al. Prevalence and correlates of accelerated atherosclerosis in systemic lupus erythematosus. N. Engl. J. Med. 2003;349:2399–2406. doi: 10.1056/NEJMoa035471. [DOI] [PubMed] [Google Scholar]
- 282.Aviña-Zubieta JA, et al. Risk of cardiovascular mortality in patients with rheumatoid arthritis: a meta-analysis of observational studies. Arthritis Care Res. 2008;59:1690–1697. doi: 10.1002/art.24092. [DOI] [PubMed] [Google Scholar]
- 283.Ha C, Magowan S, Accortt NA, Chen J, Stone CD. Risk of arterial thrombotic events in inflammatory bowel disease. Am. J. Gastroenterol. 2009;104:1445–1451. doi: 10.1038/ajg.2009.81. [DOI] [PubMed] [Google Scholar]
- 284.Ridker PM, Buring JE, Shih J, Matias M, Hennekens CH. Prospective study of C-reactive protein and the risk of future cardiovascular events among apparently healthy women. Circulation. 1998;98:731–733. doi: 10.1161/01.cir.98.8.731. [DOI] [PubMed] [Google Scholar]
- 285.Ross R. Inflammation or atherogenesis. N. Engl. J. Med. 1999;340:115–126. doi: 10.1056/NEJM199901143400207. [DOI] [PubMed] [Google Scholar]
- 286.Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 2005;352:1685–1695. doi: 10.1056/NEJMra043430. [DOI] [PubMed] [Google Scholar]
- 287.Tomita Y, et al. Acute coronary syndrome as a possible immune-related adverse event in a lung cancer patient achieving a complete response to anti-PD-1 immune checkpoint antibody. Ann. Oncol. 2017;28:2893–2895. doi: 10.1093/annonc/mdx326. [DOI] [PubMed] [Google Scholar]
- 288.Bar J, et al. Acute vascular events as a possibly related adverse event of immunotherapy: a single-institute retrospective study. Eur. J. Cancer. 2019;120:122–131. doi: 10.1016/j.ejca.2019.06.021. [DOI] [PubMed] [Google Scholar]
- 289.Hansson GK, Hermansson A. The immune system in atherosclerosis. Nat. Immunol. 2011;12:204–212. doi: 10.1038/ni.2001. [DOI] [PubMed] [Google Scholar]
- 290.Maeda N. Development of apolipoprotein E-deficient mice. Arterioscler. Thromb. Vasc. Biol. 2011;31:1957–1962. doi: 10.1161/ATVBAHA.110.220574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Ishibashi S, et al. Hypercholesterolemia in low density lipoprotein receptor knockout mice and its reversal by adenovirus-mediated gene delivery. J. Clin. Invest. 1993;92:883–893. doi: 10.1172/JCI116663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Shapiro MD, Tavori H, Fazio S. PCSK9 from basic science discoveries to clinical trials. Circ. Res. 2018;122:1420–1438. doi: 10.1161/CIRCRESAHA.118.311227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Maxwell KN, Breslow JL. Adenoviral-mediated expression of Pcsk9 in mice results in a low-density lipoprotein receptor knockout phenotype. Proc. Natl Acad. Sci. USA. 2004;101:7100–7105. doi: 10.1073/pnas.0402133101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Mestas J, Hughes CCW. Of mice and not men: differences between mouse and human immunology. J. Immunol. 2004;172:2731–2738. doi: 10.4049/jimmunol.172.5.2731. [DOI] [PubMed] [Google Scholar]
- 295.Shay T, et al. Conservation and divergence in the transcriptional programs of the human and mouse immune systems. Proc. Natl Acad. Sci. USA. 2013;110:2946–2951. doi: 10.1073/pnas.1222738110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Von Herrath MG, Nepom GT. Lost in translation: barriers to implementing clinical immunotherapeutics for autoimmunity. J. Exp. Med. 2005;202:1159–1162. doi: 10.1084/jem.20051224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Graham AL. Naturalizing mouse models for immunology. Nat. Immunol. 2021;22:111–117. doi: 10.1038/s41590-020-00857-2. [DOI] [PubMed] [Google Scholar]
- 298.Greve JM, et al. Allometric scaling of wall shear stress from mice to humans: quantification using cine phase-contrast MRI and computational fluid dynamics. Am. J. Physiol. Hear. Circ. Physiol. 2006;291:1700–1708. doi: 10.1152/ajpheart.00274.2006. [DOI] [PubMed] [Google Scholar]
- 299.Golforoush P, Yellon DM, Davidson SM. Mouse models of atherosclerosis and their suitability for the study of myocardial infarction. Basic Res. Cardiol. 2020;115:73. doi: 10.1007/s00395-020-00829-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Schwartz SM, Galis ZS, Rosenfeld ME, Falk E. Plaque rupture in humans and mice. Arterioscler. Thromb. Vasc. Biol. 2007;27:705–713. doi: 10.1161/01.ATV.0000261709.34878.20. [DOI] [PubMed] [Google Scholar]
- 301.Pasterkamp G, et al. Human validation of genes associated with a murine atherosclerotic phenotype. Arterioscler. Thromb. Vasc. Biol. 2016;36:1240–1246. doi: 10.1161/ATVBAHA.115.306958. [DOI] [PubMed] [Google Scholar]
- 302.Breschi A, Gingeras TR, Guigó R. Comparative transcriptomics in human and mouse. Nat. Rev. Genet. 2017;18:425–440. doi: 10.1038/nrg.2017.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Sellers RS. Translating mouse models: immune variation and efficacy testing. Toxicol. Pathol. 2017;45:134–145. doi: 10.1177/0192623316675767. [DOI] [PubMed] [Google Scholar]
- 304.Mair KH, et al. The porcine innate immune system: an update. Dev. Comp. Immunol. 2014;45:321–343. doi: 10.1016/j.dci.2014.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Pabst R. The pig as a model for immunology research. Cell Tissue Res. 2020;380:287–304. doi: 10.1007/s00441-020-03206-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Low LA, Mummery C, Berridge BR, Austin CP, Tagle DA. Organs-on-chips: into the next decade. Nat. Rev. Drug Discov. 2021;20:345–361. doi: 10.1038/s41573-020-0079-3. [DOI] [PubMed] [Google Scholar]
- 307.Masopust D, Sivula CP, Jameson SC. Of mice, dirty mice, and men: using mice to understand human immunology. J. Immunol. 2017;199:383–388. doi: 10.4049/jimmunol.1700453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Abolins S, et al. The comparative immunology of wild and laboratory mice, Mus musculus domesticus. Nat. Commun. 2017;8:14811. doi: 10.1038/ncomms14811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Proto JD, et al. Hypercholesterolemia induces T cell expansion in humanized immune mice. J. Clin. Invest. 2018;128:2370–2375. doi: 10.1172/JCI97785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Cai B, et al. MerTK receptor cleavage promotes plaque necrosis and defective resolution in atherosclerosis. J. Clin. Invest. 2017;127:564–568. doi: 10.1172/JCI90520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Doran AC, et al. CAMKIIγ suppresses an efferocytosis pathway in macrophages and promotes atherosclerotic plaque necrosis. J. Clin. Invest. 2017;127:4075–4089. doi: 10.1172/JCI94735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Thorp E, et al. Shedding of the Mer tyrosine kinase receptor is mediated by ADAM17 protein through a pathway involving reactive oxygen species, protein kinase Cδ, and p38 mitogen-activated protein kinase (MAPK) J. Biol. Chem. 2011;286:33335–33344. doi: 10.1074/jbc.M111.263020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Kojima Y, et al. Cyclin-dependent kinase inhibitor 2B regulates efferocytosis and atherosclerosis. J. Clin. Invest. 2014;124:1083–1097. doi: 10.1172/JCI70391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Kojima Y, et al. CD47-blocking antibodies restore phagocytosis and prevent atherosclerosis. Nature. 2016;536:86–90. doi: 10.1038/nature18935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Overton CD, Yancey PG, Major AS, Linton MF, Fazio S. Deletion of macrophage LDL receptor-related protein increases atherogenesis in the mouse. Circ. Res. 2007;100:670–677. doi: 10.1161/01.RES.0000260204.40510.aa. [DOI] [PubMed] [Google Scholar]
- 316.Advani R, et al. CD47 blockade by Hu5F9-G4 and rituximab in non-Hodgkin’s lymphoma. N. Engl. J. Med. 2018;379:1711–1721. doi: 10.1056/NEJMoa1807315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Ansell SM, et al. Phase I study of the CD47 blocker TTI-621 in patients with relapsed or refractory hematologic malignancies. Clin. Cancer Res. 2021;27:2190–2199. doi: 10.1158/1078-0432.CCR-20-3706. [DOI] [PubMed] [Google Scholar]
- 318.Brown EJ, Frazier WA. Integrin-associated protein (CD47) and its ligands. Trends Cell Biol. 2001;11:130–135. doi: 10.1016/s0962-8924(00)01906-1. [DOI] [PubMed] [Google Scholar]
- 319.Buatois V, et al. Preclinical development of a bispecific antibody that safely and effectively targets CD19 and CD47 for the treatment of B-cell lymphoma and leukemia. Mol. Cancer Ther. 2018;17:1739–1751. doi: 10.1158/1535-7163.MCT-17-1095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.Moura R, et al. Thrombospondin-1 deficiency accelerates atherosclerotic plaque maturation in ApoE−/− mice. Circ. Res. 2008;103:1181–1189. doi: 10.1161/CIRCRESAHA.108.185645. [DOI] [PubMed] [Google Scholar]
- 321.Westlake SL, et al. Tumour necrosis factor antagonists and the risk of cardiovascular disease in patients with rheumatoid arthritis: a systematic literature review. Rheumatology. 2011;50:518–531. doi: 10.1093/rheumatology/keq316. [DOI] [PubMed] [Google Scholar]
- 322.Bäck M, Yurdagul A, Tabas I, Öörni K, Kovanen PT. Inflammation and its resolution in atherosclerosis: mediators and therapeutic opportunities. Nat. Rev. Cardiol. 2019;16:389–406. doi: 10.1038/s41569-019-0169-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Fredman G, et al. An imbalance between specialized pro-resolving lipid mediators and pro-inflammatory leukotrienes promotes instability of atherosclerotic plaques. Nat. Commun. 2016;7:12859. doi: 10.1038/ncomms12859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 324.Thul S, Labat C, Temmar M, Benetos A, Bäck M. Low salivary resolvin D1 to leukotriene B4 ratio predicts carotid intima media thickness: a novel biomarker of non-resolving vascular inflammation. Eur. J. Prev. Cardiol. 2017;24:903–906. doi: 10.1177/2047487317694464. [DOI] [PubMed] [Google Scholar]
- 325.Laguna-Fernandez A, et al. ERV1/ChemR23 signaling protects against atherosclerosis by modifying oxidized low-density lipoprotein uptake and phagocytosis in macrophages. Circulation. 2018;138:1693–1705. doi: 10.1161/CIRCULATIONAHA.117.032801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Hasturk H, et al. Resolvin E1 (RvE1) attenuates atherosclerotic plaque formation in diet and inflammation-induced atherogenesis. Arterioscler. Thromb. Vasc. Biol. 2015;35:1123–1133. doi: 10.1161/ATVBAHA.115.305324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 327.Salic K, et al. Resolvin E1 attenuates atherosclerosis in absence of cholesterol-lowering effects and on top of atorvastatin. Atherosclerosis. 2016;250:158–165. doi: 10.1016/j.atherosclerosis.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 328.Petri MH, et al. Aspirin-triggered lipoxin A4 inhibits atherosclerosis progression in apolipoprotein E−/− mice. Br. J. Pharmacol. 2017;174:4043–4054. doi: 10.1111/bph.13707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02329743 (2019).
- 330.Schrezenmeier E, Dörner T. Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nat. Rev. Rheumatol. 2020;16:155–166. doi: 10.1038/s41584-020-0372-x. [DOI] [PubMed] [Google Scholar]
- 331.Sharma TS, et al. Hydroxychloroquine use is associated with decreased incident cardiovascular events in rheumatoid arthritis patients. J. Am. Heart Assoc. 2016;5:e002867. doi: 10.1161/JAHA.115.002867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Jung H, et al. The protective effect of antimalarial drugs on thrombovascular events in systemic lupus erythematosus. Arthritis Rheum. 2010;62:863–868. doi: 10.1002/art.27289. [DOI] [PubMed] [Google Scholar]
- 333.Graßhoff H, et al. Low-dose IL-2 therapy in autoimmune and rheumatic diseases. Front. Immunol. 2021;12:902. doi: 10.3389/fimmu.2021.648408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 334.Von Spee-Mayer C, et al. Low-dose interleukin-2 selectively corrects regulatory T cell defects in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 2016;75:1407–1415. doi: 10.1136/annrheumdis-2015-207776. [DOI] [PubMed] [Google Scholar]
- 335.Zhao TX, Newland SA, Mallat Z. 2019 ATVB plenary lecture: Interleukin-2 therapy in cardiovascular disease: the potential to regulate innate and adaptive immunity. Arterioscler. Thromb. Vasc. Biol. 2020;40:853–864. doi: 10.1161/ATVBAHA.119.312287. [DOI] [PubMed] [Google Scholar]
- 336.Zhao TX, et al. Low dose interleukin-2 in patients with stable ischaemic heart disease and acute coronary syndrome (LILACS) Eur. Heart J. 2020;41:e022452. doi: 10.1136/bmjopen-2018-022452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Cole JE, et al. Unexpected protective role for Toll-like receptor 3 in the arterial wall. Proc. Natl Acad. Sci. USA. 2011;108:2372–2377. doi: 10.1073/pnas.1018515108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 338.Salagianni M, et al. Toll-like receptor 7 protects from atherosclerosis by constraining inflammatory macrophage activation. Circulation. 2012;126:952–962. doi: 10.1161/CIRCULATIONAHA.111.067678. [DOI] [PubMed] [Google Scholar]
- 339.Cole JE, Kassiteridi C, Monaco C. Toll-like receptors in atherosclerosis: a ‘Pandora’s box’ of advances and controversies. Trends Pharmacol. Sci. 2013;34:629–636. doi: 10.1016/j.tips.2013.09.008. [DOI] [PubMed] [Google Scholar]
- 340.Dinarello CA. Interleukin-1β and the autoinflammatory diseases. N. Engl. J. Med. 2009;360:2467–2470. doi: 10.1056/NEJMe0811014. [DOI] [PubMed] [Google Scholar]
- 341.Edfeldt K, Swedenborg J, Hansson GK, Yan ZQ. Expression of toll-like receptors in human atherosclerotic lesions: a possible pathway for plaque activation. Circulation. 2002;105:1158–1161. [PubMed] [Google Scholar]
- 342.Methe H, et al. Expansion of circulating Toll-like receptor 4-positive monocytes in patients with acute coronary syndrome. Circulation. 2005;111:2654–2661. doi: 10.1161/CIRCULATIONAHA.104.498865. [DOI] [PubMed] [Google Scholar]
- 343.Mullick AE, et al. Increased endothelial expression of Toll-like receptor 2 at sites of disturbed blood flow exacerbates early atherogenic events. J. Exp. Med. 2008;205:373–383. doi: 10.1084/jem.20071096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Michelsen KS, et al. Lack of Toll-like receptor 4 or myeloid differentiation factor 88 reduces atherosclerosis and alters plaque phenotype in mice deficient in apolipoprotein E. Proc. Natl Acad. Sci. USA. 2004;101:10679–10684. doi: 10.1073/pnas.0403249101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Liu X, et al. Toll-like receptor 2 plays a critical role in the progression of atherosclerosis that is independent of dietary lipids. Atherosclerosis. 2008;196:146–154. doi: 10.1016/j.atherosclerosis.2007.03.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Schroder K, Tschopp J. The inflammasomes. Cell. 2010;140:821–832. doi: 10.1016/j.cell.2010.01.040. [DOI] [PubMed] [Google Scholar]
- 347.Duewell P, et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature. 2010;464:1357–1361. doi: 10.1038/nature08938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Hornung V, et al. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nat. Immunol. 2008;9:847–856. doi: 10.1038/ni.1631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 349.Lüsebrink E, et al. AIM2 stimulation impairs reendothelialization and promotes the development of atherosclerosis in mice. Front. Cardiovasc. Med. 2020;7:223. doi: 10.3389/fcvm.2020.582482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 350.Paulin N, et al. Double-strand DNA sensing Aim2 inflammasome regulates atherosclerotic plaque vulnerability. Circulation. 2018;138:321–323. doi: 10.1161/CIRCULATIONAHA.117.033098. [DOI] [PubMed] [Google Scholar]
- 351.Bauernfeind FG, et al. Cutting edge: NF-κB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. J. Immunol. 2009;183:787–791. doi: 10.4049/jimmunol.0901363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Py BF, Kim MS, Vakifahmetoglu-Norberg H, Yuan J. Deubiquitination of NLRP3 by BRCC3 critically regulates inflammasome activity. Mol. Cell. 2013;49:331–338. doi: 10.1016/j.molcel.2012.11.009. [DOI] [PubMed] [Google Scholar]
- 353.Kayagaki N, et al. Caspase-11 cleaves gasdermin D for non-canonical inflammasome signalling. Nature. 2015;526:666–671. doi: 10.1038/nature15541. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.