Abstract
Previous studies have described reverse-transcription loop-mediated isothermal amplification (RT-LAMP) for the rapid detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in nasopharyngeal/oropharyngeal swab and saliva samples. This multisite clinical evaluation describes the validation of an improved sample preparation method for extraction-free RT-LAMP and reports clinical performance of four RT-LAMP assay formats for SARS-CoV-2 detection. Direct RT-LAMP was performed on 559 swabs and 86,760 saliva samples and RNA RT-LAMP on extracted RNA from 12,619 swabs and 12,521 saliva samples from asymptomatic and symptomatic individuals across health care and community settings. For direct RT-LAMP, overall diagnostic sensitivity (DSe) was 70.35% (95% CI, 63.48%–76.60%) on swabs and 84.62% (95% CI, 79.50%–88.88%) on saliva, with diagnostic specificity of 100% (95% CI, 98.98%–100.00%) on swabs and 100% (95% CI, 99.72%–100.00%) on saliva, compared with quantitative RT-PCR (RT-qPCR); analyzing samples with RT-qPCR ORF1ab CT values of ≤25 and ≤33, DSe values were 100% (95% CI, 96.34%–100%) and 77.78% (95% CI, 70.99%–83.62%) for swabs, and 99.01% (95% CI, 94.61%–99.97%) and 87.61% (95% CI, 82.69%–91.54%) for saliva, respectively. For RNA RT-LAMP, overall DSe and diagnostic specificity were 96.06% (95% CI, 92.88%–98.12%) and 99.99% (95% CI, 99.95%–100%) for swabs, and 80.65% (95% CI, 73.54%–86.54%) and 99.99% (95% CI, 99.95%–100%) for saliva, respectively. These findings demonstrate that RT-LAMP is applicable to a variety of use cases, including frequent, interval-based direct RT-LAMP of saliva from asymptomatic individuals who may otherwise be missed using symptomatic testing alone.
Rapid diagnostic testing to identify and isolate symptomatic and asymptomatic individuals potentially transmitting infectious viral pathogens is an essential requirement of any pandemic response. The novel betacoronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), initially identified after an outbreak of viral pneumonia in Wuhan, China, in December 2019,1 has rapidly spread throughout the world, causing >223 million confirmed cases and >4.6 million deaths (https://coronavirus.jhu.edu, last accessed September 10, 2021).
Conventional diagnostics for SARS-CoV-2 consist of RNA enrichment followed by reverse-transcription quantitative real-time PCR (RT-qPCR) against one or more viral gene targets.2 However, this method requires sample inactivation, RNA extraction, and RT-qPCR thermal cycling, meaning that the time from sample to result can often be several hours, and requires centralized equipment and personnel trained in Good Laboratory Practice to perform testing.
The utility of reverse-transcription loop-mediated isothermal amplification (RT-LAMP) for the detection of SARS-CoV-2 both from extracted RNA (RNA RT-LAMP) and directly from nasopharyngeal (NP)/oropharyngeal (OP) swabs (direct RT-LAMP)3 has previously been shown. RT-LAMP utilizes a rapid and stable DNA polymerase that amplifies target nucleic acids at a constant temperature. This removes the requirement for conventional thermal cycling, allowing RT-LAMP reactions to be performed in shorter reaction times using less sophisticated platforms.
In a study of 196 clinical samples,3 testing of RNA extracted from NP/OP swabs collected into viral transport media (VTM) using RNA RT-LAMP demonstrated a diagnostic sensitivity (DSe) of 97% and a diagnostic specificity (DSp) of 99% in comparison to RT-qPCR of the ORF1ab region of SARS-CoV-2. For direct RT-LAMP on crude swab samples, the DSe and DSp were 67% and 97%, respectively. When a CT cutoff for RT-qPCR of <25 was considered, reflecting the increased likelihood of detecting viral RNA from active viral replication, the DSe of direct RT-LAMP increased to 100%.3
However, the collection of a swab sample is invasive, and during the time of the pandemic, there have been considerable shortages in swab supplies. Exploring the use of alternative sample types that are both easy to collect and more comfortable from a sampling perspective4, 5, 6 is desirable, particularly when repeated sampling is performed.7, 8, 9 Saliva presents an ideal biofluid that fulfills both these objectives, and previous studies have shown that SARS-CoV-2 is readily detectable in such a sample type.10, 11, 12, 13, 14, 15, 16, 17, 18 To improve the diagnostic sensitivity of previously described saliva direct RT-LAMP,3 optimization of saliva preparation for the detection of SARS-CoV-2 was undertaken utilizing a cohort of 3100 saliva samples from an asymptomatic population19 of health care workers; saliva was diluted 1:1 in Mucolyse (Pro-Lab, Toronto, ON, Canada), followed by a 1 in 10 dilution in 10% (w/v) Chelex 100 Resin (Bio-Rad, Hercules, CA), ending with a 98°C heat step before RT-LAMP, which resulted in optimal sensitivity and specificity.
Despite the benefits of this optimization, the protocol added additional steps and reagents that increased chance for user error and made the automation of the process more challenging. This study therefore aimed to investigate a simpler process using a novel reagent, RapiLyze (OptiGene Ltd, Camberley, UK), which is a sample dilution buffer, followed by a 2-minute heat step. This novel sample preparation method was evaluated in combination with direct RT-LAMP using samples collected from symptomatic National Health Service (NHS) patients and symptomatic and asymptomatic health care staff.
Materials and Methods
Ethical Statement
All relevant ethical guidelines have been followed, and any necessary institutional review board and/or ethics committee approvals have been obtained. The study followed National Research Ethics Service Committee West Midlands, South Birmingham 2002/201 Amendment Number 4. All necessary written participant consent has been obtained, and the appropriate institutional forms have been archived.
Testing Sites
The OptiGene Ltd SARS-CoV-2 RT-LAMP assay was evaluated in nine sites, comprising Basingstoke and North Hampshire Hospital and Royal Hampshire County Hospital, Hampshire Hospitals NHS Foundation Trust; University Hospital Southampton; Animal and Plant Health Agency/Medical Research Council Lifecourse Epidemiology Unit (University of Southampton); Public Health Lab Manchester/Central Manchester University Hospitals NHS Foundation Trust; Leeds Teaching Hospitals NHS Trust; University Hospitals Birmingham NHS Foundation Trust/Institute of Cancer and Genomic Science University of Birmingham; High Containment Microbiology, National Infection Service, Public Health England, Porton Down; and Public Health University Laboratory, Gibraltar Health Authority, Gibraltar, UK.
Clinical Samples
NP and OP swabs were collected from asymptomatic and symptomatic individuals and placed in VTM.
Drooled saliva samples were collected at the start of the day, before eating, drinking, teeth brushing, or using a mouthwash. Saliva was transferred into the specimen pot directly or via a clean teaspoon, according to a standardized protocol. Samples from University Hospitals Birmingham deposited saliva straight into the collection pot.
Log Reduction of SARS-CoV-2 for the Heat and Lysis Steps Used Independently and Sequentially
The viral inactivation kinetics of the best sample preparation condition(s) were determined, evaluating the effect of the heat and lysis steps on the viral inactivation of SARS-CoV-2, as determined by infectivity assays. All inactivation experiments were conducted under containment level 3 containment and as such were undertaken at Animal and Plant Health Agency. Heat inactivation experiments were conducted utilizing high-titer live SARS-CoV-2 virus spiked into pools of saliva collected from Animal and Plant Health Agency staff or in tissue culture supernatant. Early experiments demonstrated that saliva had a high toxicity for tissue culture cells, even after heat inactivation, demonstrating that toxicity was likely not enzymatic. As such, further inactivation was undertaken on live virus tissue culture supernatant. Comparison was also undertaken of β-propiolactone inactivated virus and live virus.
RNA Extraction
RNA was extracted using a range of different methods available at each participating site.
Maxwell RSC Viral Total Nucleic Acid Purification Kit
In a class 1 microbiological safety cabinet within a containment level 3 laboratory, 200 μL of sample was added to 223 μL of prepared lysis solution (including 5 μL per reaction of Genesig Easy RNA Internal extraction control; Primerdesign Ltd, Chandler's Ford, UK). Samples were then inactivated for 10 minutes at room temperature within the microbiological safety cabinet and 10 minutes at 56°C on a heat block before automated RNA extraction using a Maxwell RSC48 Instrument (Promega UK Ltd, Southampton, UK). RNA was eluted in 50 μL of nuclease-free water.
MagMAXCORE Nucleic Acid 140 Purification Kit
A total of 10 μL of sample [diluted in 190 μL diethylpyrocarbonate (DEPC) treated water] was added to 700 μL of prepared lysis solution. Samples were then inactivated for 10 minutes at room temperature within the safety cabinet before automated RNA extraction using a Kingfisher Flex (Thermo Fisher, Basingstoke, UK). RNA was eluted in 90 μL of nuclease-free water.
Roche FLOW System
RNA was extracted on a MagNA Pure 96 extraction robot using the MagNA Pure 96 DNA and Viral Nucleic Acid Small Volume kit (Roche, Basel, Switzerland) and the Pathogen 200 universal protocol version 4.0.
Qiagen QIAsymphony
RNA was extracted using the QIASymphony Virus/Bacteria Mini Kit (Qiagen, Hilden, Germany) by the CellFree200 Default IC protocol with a 60 μL extract elution volume.
SARS-CoV-2 Real-Time RT-qPCR
RNA was analyzed using a range of different methods available at each site.
CerTest VIASURE SARS-CoV-2 Real-Time qPCR Assay
Single-step RT-qPCR against the ORF1ab region and N1 gene target of SARS-CoV-2 was performed using the CerTest VIASURE SARS-CoV-2 real-time PCR kit (CerTest Biotech SL, Zaragoza, Spain), according to the manufacturer's instructions for use on either the Thermo Fisher QuantStudio 5 or BioMolecular Systems (Upper Coomera, QLD, Australia) Magnetic Induction Cycler (MIC) instruments, using 5 μL of extracted RNA per reaction. RNA extracted using the Maxwell RSC Viral Total Nucleic Acid Purification Kit was analyzed using this assay.
COVID-19 Genesig Real-Time qPCR Assay
Single-step RT-qPCR against the ORF1ab region of SARS-CoV-2 was performed using the COVID-19 Genesig Real-Time PCR assay (Primerdesign Ltd), according to the manufacturer's instructions for use on BioMolecular Systems MIC instruments, using 5 μL of extracted RNA per reaction. RNA extracted using the Maxwell RSC Viral Total Nucleic Acid Purification Kit was analyzed using this assay.
Corman et al2 Real-Time qPCR Assay
Single-step RT-qPCR against the E gene target of SARS-CoV-2 was performed with the Corman et al2 primers using the AgPath-ID PCR kit (Thermo Fisher), according to manufacturer's instructions for use on an Aria qPCR Cycler (Agilent, Cheadle, UK); and results were analyzed using the Agilent AriaMX 1.5 software, using 5 μL of extracted RNA per reaction. RNA extracted using the MagMAXCORE Nucleic acid purification kit were analyzed using this assay.
RT-qPCR was performed on an Applied Biosystems Fast 7500 PCR thermocycler in standard run mode using the SARS-CoV-2 E gene Sarbeco assay using MS2 as an internal extraction control and aliquots of SARS-CoV-2/England/2/2020 as a positive control. The master mix comprised E-gene F and R primers and TM-P (400, 400, and 200 nmol/L final concentration, respectively), MS2 primers and TM probe (20, 20, and 40 nmol/L final concentration, respectively), and 4× TaqMan Fast Virus 1-Step Master Mix made up with molecular-grade nuclease free water (Ambion, Austin, TX) to a final volume of 15 μL. A total of 5 μL of AVE buffer extract was used as a template and added to the 15 μL mastermix. Cycling conditions were 55°C for 10 minutes, followed by 94°C for 3 minutes and 45 cycles of 95°C for 15 seconds and 58°C for 30 seconds.
SARS-CoV-2 (2019-nCoV) CDC qPCR Probe Assay
Single-step RT-qPCR against the N1 and N2 gene targets of SARS-CoV-2 was performed using Integrated Design Technologies kit (catalog number 10006606; Coralville, IA), according to the manufacturer's instructions for use on either an LC480 II or ABI 7500 FAST instrument. RNA extracted on Qiagen QIAsymphony and the Roche FLOW system was analyzed using this RT-qPCR assay.
RT-LAMP
RT-LAMP assays were performed using OptiGene Ltd COVID-19_RT-LAMP kits, as described previously,3 with the following modifications. The COVID-19_RNA RT-LAMP KIT-500 kit was used for RNA RT-LAMP, and the COVID-19_Direct PLUS RT-LAMP KIT-500 was used for direct RT-LAMP directly on oropharyngeal/nasopharyngeal swabs or saliva samples. The COVID-19_Direct PLUS RT-LAMP KIT-500 kit also includes a sample preparation buffer, RapiLyze. For RNA RT-LAMP, 5 μL of extracted RNA was added to the reaction. For the Direct PLUS RT-LAMP, 50 μL sample (swab VTM or neat saliva) was added to 50 μL RapiLyze, vortexed, and placed in a dry heat block preheated to 98°C for 2 minutes. A total of 5 μL of the treated sample was added to each reaction.
The anneal temperature that confirmed a positive result for direct RT-LAMP was modified to 81.5°C and 85.99°C because of the effect of RapiLyze buffer on the reaction.
SARS-CoV-2 Viral Culture of Clinical Samples across a CT Range
For culture, 100 and 100 μL of a 1 in 10 dilution of samples 1 to 6 (predicted lower CT values) and 100 μL samples 7 to 26 (with higher predicted CT values) were added to 25-cm2 flasks containing 80% confluent Vero E6 cells and allowed to adsorb for 1 hour. A total of 5 mL of minimum essential medium (Gibco) + HEPES (Gibco, Thermo Fisher, Basingstoke, UK) + 4% fetal calf serum (Sigma, St. Louis, MO) + 1 × antibiotic-antimycotic (Gibco) was added to each flask and incubated for 1 week at 37°C. Two negative control flasks, to which 100 μL minimum essential medium + 4% fetal calf serum was added in place of sample, were set up in parallel. Cultures were checked visually for cytopathic effect (CPE). Where CPE was not observed after 1 week, 500 μL of supernatant was passed into a fresh flask containing Vero E6 cells for a further two passages. At the beginning and end of each passage, 140 μL of supernatant was collected for SARS-CoV-2 RT-qPCR, as described before.
To determine the sensitivity of the isolation method for SARS-CoV-2 from clinical samples, a virus stock titered by plaque assay (HCM/V/53), a passage 3 working bank grown from SARS-CoV-2 Strain England 2, from Public Health England, was diluted in minimum essential medium to give virus dilutions containing 1000, 100, 10, 1, 0.1, and 0.01 plaque-forming units. The virus was added to duplicate flasks containing Vero E6 cells and AVL. After 72 hours of incubation, flasks were checked for CPE, and for those where CPE was observed, the supernatant was collected for RT-qPCR. Any flasks not showing CPE after 7 days were passed on to fresh cells and resampled, as described above.
Statistical Analysis
Overall diagnostic sensitivity and specificity (including 95% Clopper-Pearson CIs) were calculated by the aggregation of individual site data for each method (RNA and direct RT-LAMP) for each sample type (swabs and saliva). To demonstrate the effectiveness of detecting samples with higher viral load, confusion matrices are quoted where the threshold for positive sample inclusion varies (ie, for CT ≤25, only positive samples with CT ≤25 are included).
To account for site heterogeneity, a bivariate meta-analysis model is additionally applied at the site level to produce a summary of sensitivity and specificity for each method and sample type.20 Within-study variability for sensitivity and specificity are assumed to follow independent binomial distributions:
| (1) |
where represent the number testing positive for site i respectively, and represent the number testing positive and negative by RT-qPCR for site i respectively. The between-study heterogeneity is represented by a bivariate normal distribution for the logit-transformed sensitivity and specificity
| (2) |
where represent the expected logit sensitivity and specificity, , represent the between-study variance in the logit sensitivity and specificity, and represents the covariance between the logit sensitivity and specificity. For direct RT-LAMP, we fit a univariate normal distribution for the logit-transformed sensitivity only, because of the absence of false positives across all sites.
In addition, the sensitivity as a function of viral load was assessed for RNA RT-LAMP and direct RT-LAMP on both swab and saliva samples. This was performed through the conversion of each sample CT value to viral load in gene copies/mL for all sample sets. As the relationship between CT value and viral load varied according to the RT-qPCR method used, a dilution series was utilized for each method to standardize these values for two of the four aforementioned RT-qPCR methods (CerTest VIASURE SARS-CoV-2 real-time PCR kit and Corman et al2 RT-qPCR assay E gene), which was used for testing 100% of the swab samples, 90% of the saliva samples used for direct RT-LAMP, and 83% of the saliva samples used for RNA RT-LAMP. The logarithm of the viral load was then fitted to the CT values for both methods using linear regression followed by converting the CT values to viral load based on which method had been used to evaluate the samples. For the remaining samples (n = 56) that utilized one of the other two RT-qPCR methods for which viral load was not standardized against a CT value, the conversion derived from the CerTest VIASURE SARS-CoV-2 real-time PCR kit dilution series was applied, with the assumption that the N gene CT values are the most similar.21, 22, 23
For the CerTest VIASURE SARS-CoV-2 real-time PCR kit, the following relationship between log viral load and CT value was applied:
| (3) |
and similarly, for the Corman et al2 RT-qPCR assay:
| (4) |
where V represents the viral load in copies/mL.
Viral load was grouped according to the following categories (in copies/mL): ≤103, >103 to ≤104, >104 to ≤105, >105 to ≤106, >106 to ≤107, and >107; then, the diagnostic sensitivity was calculated according to viral load group with associated Clopper-Pearson 95% CIs.
The site meta-analysis was produced using R 3.5.3 (The R Project for Statistical Computing; https://cran.r-project.org/bin/windows/base/old/3.5.3). Confusion matrices, sensitivity, specificity, sensitivity as a function of viral load calculations, and the production of scatter graphs showing the relationship between RT-LAMP results and CT were performed using Python 3.8.6 (https://www.python.org).
Results
Optimization of Sample Preparation Conditions
Heat inactivation experiments demonstrated that SARS-CoV-2 was completely inactivated by heating at 60°C (20 minutes plus) or ≥70°C (after 2, 5, or 10 minutes) (Supplemental Table S1). More important, optimized RapiLyze Sample Lysis Buffer did not inactivate live virus on its own without a heat step. Furthermore, inactivation at 56°C was not 100% effective at shorter incubation times, and additionally showed a loss in sensitivity following a 4 × twofold dilution (P07102) (Supplemental Table S2) at 10 and 30 minutes. Following optimization of heat inactivation of live virus, pretreatment of samples was assessed to determine any impact of pretreatment on assay sensitivity. Interestingly, a pretreatment of 70°C for 5 minutes performed on spiked samples before the proposed direct RT-LAMP assay had no effect on subsequent LAMP or PCR results. It recommended that even if a pretreatment is effective in inactivating the virus that downstream processes are performed in UV hoods or with effective air-flow management to prevent cross-contamination of the direct RT-LAMP assay. Comparison of β-propiolactone inactivated virus and live virus demonstrated that β-propiolactone inactivation has resulted in lower sensitivity of detection using direct RT-LAMP. β-Propiolactone inactivated virus is therefore not an ideal substitute for live virus in spiking experiments. Any conclusions on assay sensitivity or performance have consequently been drawn from experiments on spiking of live virus in tissue culture supernatant or saliva performed in containment. Spiking of live virus into pooled saliva has demonstrated that direct detection by RT-LAMP is possible in samples that give a CT of <25/26 with extraction and PCR.
RNA RT-LAMP
VTM from 12,619 NP/OP swabs were assayed. A total of 265 swab samples were from known symptomatic individuals, and 2073 swab samples were from known asymptomatic individuals. The clinical status of the remaining samples (n = 10,281) was unknown.
A total of 12,521 neat saliva samples were assayed, none of which was from known symptomatic individuals. A total of 12,365 of these samples were from known asymptomatic individuals. The clinical status of the remaining saliva samples (n = 156) was unknown.
Direct RT-LAMP
VTM from 559 NP/OP swabs were assayed. A total of 170 swab samples were from known symptomatic individuals, and 310 samples were from known asymptomatic individuals; and the clinical status of the remaining swab samples (n = 79) was unknown.
A total of 86,760 neat saliva samples were assayed. A total of 93 samples were from known symptomatic individuals, and 86,593 samples were from known asymptomatic individuals. The clinical status of the remaining samples (n = 74) was unknown. In addition, 10 separate longitudinal daily saliva samples were provided from one individual as a time course from development of symptoms to 3 days after resolution of symptoms.
RNA RT-LAMP on NP/OP Swabs
A total of 12,619 swab samples were assayed by RNA RT-LAMP, of which 254 were RT-qPCR positive and 12,365 were RT-qPCR negative. RNA RT-LAMP detected 244 of the 254 positives (Figure 1 and Table 1). Only one of the 12,365 samples negative by RT-qPCR was positive by RNA RT-LAMP. A total of 588 samples were tested in duplicate, and 12,031 were tested as single replicates. Of those samples tested in duplicate, seven were detected by RNA RT-LAMP in only a single replicate (CT values of 27.00, 32.66, 33.14, 33.16, 34.07, 35.05, and 37.20); all of these had received at least one freeze thaw before analysis. Overall DSe was 96.06% (95% CI, 92.88%–98.12%) and overall DSp was 99.99% (95% CI, 99.95%–100.00%), which is corrected to DSe of 95.98% (95% CI, 92.70%–97.83%) and DSp of 99.99% (95% CI, 99.94%–100.00%) after site meta-analysis. Diagnostic sensitivity of samples with a CT ≤25 (n = 123) was 100.00% (95% CI, 96.76%–100.00) and specificity was 99.99% (95% CI, 99.95%–100.00), and of samples with a CT ≤33 (n = 180), sensitivity was 98.65% (95% CI, 96.10%–99.72%) and specificity was 99.99% (95% CI, 99.95%–100.00%).
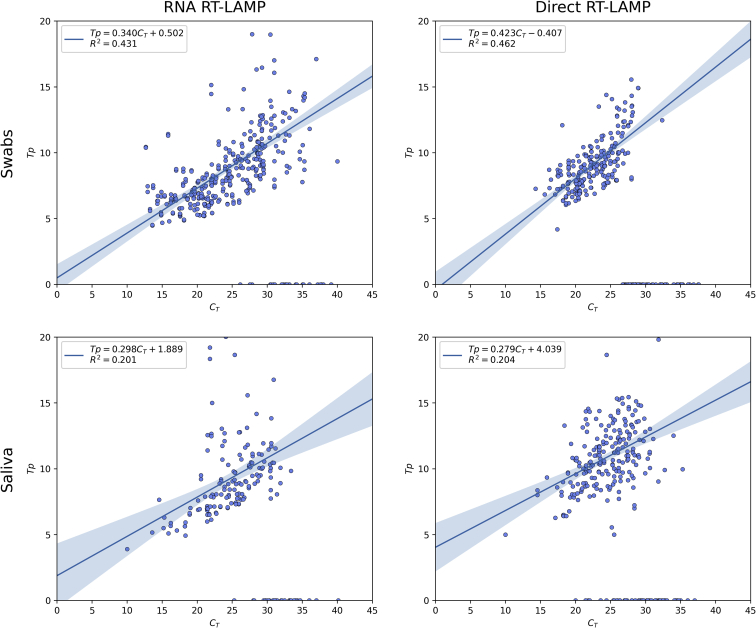
Figure 1.
Time to positivity (Tp) in minutes plotted against RT-qPCR CT for each combination of method and sample type. Samples that were negative by RT-qPCR are not shown. Samples that were negative by reverse-transcription loop-mediated isothermal amplification (RT-LAMP) are shown with 0 time to positivity. Results of linear ordinary least squared regression are shown for samples that were RT-LAMP positive with the regression line and corresponding 95% CI represented by the blue lines and light blue shaded regions, respectively.
Table 1.
DSe and DSp of RNA RT-LAMP on Swabs Compared with RT-qPCR
| CT value | Variable | RT-qPCR Pos | RT-qPCR Neg | Total | DSe or DSp | % | 95% CI |
|---|---|---|---|---|---|---|---|
| CT <45 swab | RNA RT-LAMP Pos | 244∗ | 1 | 245 | DSe | 96.06 | 92.88–98.12 |
| RNA RT-LAMP Neg | 10 | 12,364 | 12,374 | DSp | 99.99 | 99.95–100 | |
| Total | 254 | 12,365 | |||||
| CT <33 swab | RNA RT-LAMP Pos | 219 | 1 | 220 | DSe | 98.95 | 96.10–99.72 |
| RNA RT-LAMP Neg | 3 | 12,364 | 12,367 | DSp | 99.9 | 99.95–100 | |
| Total | 222 | 12,365 | |||||
| CT <25 swab | RNA RT-LAMP Pos | 112 | 1 | 113 | DSe | 100 | 96.76–100 |
| RNA RT-LAMP Neg | 0 | 12,364 | 12,364 | DSp | 99.9 | 99.95–100 | |
| Total | 112 | 12,365 |
DSe, diagnostic sensitivity; DSp, diagnostic specificity; Neg, negative; Pos, positive; RT-LAMP, reverse-transcription loop-mediated isothermal amplification.
Five samples included in this number were positive by RT-qPCR but did not have an associated CT value because of being assayed on a platform that did not produce a CT value.
Direct RT-LAMP on NP/OP Swabs
A total of 559 swab samples were assayed by direct RT-LAMP, of which 199 were RT-qPCR positive and 360 were RT-qPCR negative. Direct RT-LAMP detected 140 of the 199 samples positive by RT-qPCR (Figure 1 and Table 2). A total of 195 samples were tested in duplicate, and 364 were tested as single replicates. Seven of 195 samples tested in duplicate were positive by direct RT-LAMP in only one replicate (CT values of 27.51, 27.95, 28.15, 28.15, 28.87, 28.92, and 28.95); all these samples had received at least one freeze thaw before analysis. Overall diagnostic sensitivity was 70.35% (95% CI, 63.48%–76.60%), and specificity was 100% (95% CI, 98.98%–100%). After correction by site meta-analysis, the DSe is corrected to 67.59% (95% CI, 53.71%–78.94%). Diagnostic sensitivity of samples with a CT ≤25 (n = 113) was 100% (95% CI, 96.34%–100%) and specificity was 100% (95% CI, 98.98%–100%), and of samples with a CT ≤33 (n = 182), sensitivity was 77.78% (95% CI, 70.99%–83.62%) and specificity was 100% (95% CI, 98.98%–100%).
Table 2.
DSe and DSp of Direct RT-LAMP on Swabs Compared with RT-qPCR
| CT value | Variable | RT-qPCR Pos | RT-qPCR Neg | Total | DSe or DSp | % | 95% CI |
|---|---|---|---|---|---|---|---|
| CT <45 swab | Direct RT-LAMP Pos | 140 | 0 | 140 | DSe | 70.35 | 63.48–76.60 |
| Direct RT-LAMP Neg | 59 | 360 | 419 | DSp | 100 | 98.98–100 | |
| Total | 199 | 360 | |||||
| CT <33 swab | Direct RT-LAMP Pos | 140 | 0 | 140 | DSe | 77.78 | 70.99–83.62 |
| Direct RT-LAMP Neg | 40 | 360 | 400 | DSp | 100 | 98.98–100 | |
| Total | 180 | 360 | |||||
| CT <25 swab | Direct RT-LAMP Pos | 99 | 0 | 99 | DSe | 100 | 96.34–100 |
| Direct RT-LAMP Neg | 0 | 360 | 360 | DSp | 100 | 98.98–100 | |
| Total | 99 | 360 |
DSe, diagnostic sensitivity; DSp, diagnostic specificity; Neg, negative; Pos, positive; RT-LAMP, reverse-transcription loop-mediated isothermal amplification.
RNA RT-LAMP on Saliva
Saliva samples numbering 12,521 were assayed by RNA RT-LAMP, of which 155 were RT-qPCR positive and 12,366 were RT-qPCR negative. RNA RT-LAMP detected 133 of the 155 samples that were positive by RT-qPCR (Figure 1 and Table 3). Only one of the 12,366 samples negative by RT-qPCR was positive by RNA RT-LAMP. A total of 44 samples were tested in duplicate, and 12,477 were tested as single replicates. All samples tested in duplicate were positive in both replicates. Overall diagnostic sensitivity was 80.65% (95% CI, 73.54%–86.54%) and specificity was 99.99% (95% CI, 99.95%–100%), which is corrected to DSe of 79.05% (95% CI, 68.87%–86.55%) and DSp of 99.99% (95% CI, 99.74%–100%) after site meta-analysis. Diagnostic sensitivity of samples with a CT ≤25 (n = 74) was 100% (95% CI, 93.73%–100%) and specificity was 99.99% (95% CI, 99.95%–100%), and of samples with a CT ≤33 (n = 150), sensitivity was 87.32% (95% CI, 80.71%–92.31%) and specificity was 99.95% (95% CI, 99.95%–100.00%).
Table 3.
DSe and DSp of RNA RT-LAMP on Saliva Compared with RT-qPCR
| CT value | Variable | RT-qPCR Pos | RT-qPCR Neg | Total | DSe or DSp | % | 95% CI |
|---|---|---|---|---|---|---|---|
| CT <45 saliva | RNA RT-LAMP Pos | 125 | 1 | 126 | DSe | 80.65 | 73.54–86.54 |
| RNA RT-LAMP Neg | 30 | 12,365 | 12,395 | DSp | 99.99 | 99.95–100 | |
| Total | 155 | 12,366 | |||||
| CT <33 saliva | RNA RT-LAMP Pos | 124 | 1 | 125 | DSe | 87.32 | 80.71–92.31 |
| RNA RT-LAMP Neg | 18 | 12,365 | 12,383 | DSp | 99.99 | 99.95–100 | |
| Total | 142 | 12,366 | |||||
| CT <25 saliva | RNA RT-LAMP Pos | 57 | 1 | 58 | DSe | 100 | 93.73–100 |
| RNA RT-LAMP Neg | 0 | 12,365 | 12,365 | DSp | 99.99 | 99.95–100 | |
| Total | 57 | 12,366 |
DSe, diagnostic sensitivity; DSp, diagnostic specificity; Neg, negative; Pos, positive; RT-LAMP, reverse-transcription loop-mediated isothermal amplification.
Direct RT-LAMP on Saliva
A total of 86,760 saliva samples were tested by direct RT-LAMP, of which 247 were RT-qPCR positive and 7195 were RT-qPCR negative (79,318 were negative on RT-LAMP but were not tested by RT-qPCR) (Figure 1 and Table 4). Direct RT-LAMP detected 209 of the 247 samples positive by RT-qPCR. A total of 83 samples were tested in duplicate, and 86,677 were tested as single replicates. Nine of the 83 samples tested in duplicate were negative in one of the duplicates, and all these samples had received at least one freeze thaw before analysis (CT values of 20.27, 21.28, 22.01, 24.42, 25.85, 27.35, 28.52, and 30.37). Overall diagnostic sensitivity was 84.62% (95% CI, 79.50%–88.88%), and specificity was 100% (95% CI, 99.72%–100%). After correction by site meta-analysis, DSe is corrected to 84.24% (95% CI, 55.03%–95.89%). Diagnostic specificity was calculated using only the confirmed RT-qPCR negative samples. Diagnostic sensitivity of samples with a CT ≤25 (n = 126) was 99.01% (95% CI, 94.61%–99.97%) and specificity was 100.00% (95% CI, 99.72%–100%), and of samples with a CT ≤33 (n = 237), sensitivity was 87.61% (95% CI, 82.69%–91.54%) and specificity was 100% (95% CI, 99.72%–100%).
Table 4.
DSe and DSp of Direct RT-LAMP on Saliva Compared with RT-qPCR
| CT value | Variable | RT-qPCR Pos | RT-qPCR Neg | Total | DSe or DSp | % | 95% CI |
|---|---|---|---|---|---|---|---|
| CT <45 saliva | Direct RT-LAMP Pos | 209 | 0 | 209 | DSe | 84.62 | 79.50–88.88 |
| Direct RT-LAMP Neg | 38 | 7195∗ | 7233 | DSp | 100.00 | 99.95–100.00 | |
| Total | 247 | 7195 | |||||
| CT <33 saliva | Direct RT-LAMP Pos | 205 | 0 | 205 | DSe | 87.61 | 82.69–91.54 |
| Direct RT-LAMP Neg | 29 | 7195∗ | 7224 | DSp | 100.0 | 99.95–100.00 | |
| Total | 234 | 7195 | |||||
| CT <25 saliva | Direct RT-LAMP Pos | 100 | 0 | 100 | DSe | 99.01 | 94.61–99.97 |
| Direct RT-LAMP Neg | 1 | 7195∗ | 7196 | DSp | 100.0 | 99.95–100.00 | |
| Total | 101 | 7195 |
DSe, diagnostic sensitivity; DSp, diagnostic specificity; Neg, negative; Pos, positive; RT-LAMP, reverse-transcription loop-mediated isothermal amplification.
A total of 85,177 samples were negative on RT-LAMP, but only 7196 were confirmed negative by RT-qPCR. Only those that were confirmed negative by RT-qPCR were included in the calculations.
Relationship between CT Value and Tp
The relationship between CT value and time to positivity (Tp) was explored with the results shown in Figure 1. Although there is a weak linear relationship between CT value and Tp across all methods and sample types, a stronger linear relationship was observed in swab samples, with R2 = 0.431 for RNA RT-LAMP and R2 = 0.462 for direct RT-LAMP. There was a notably weaker linear relationship in saliva samples: R2 = 0.201 for RNA RT-LAMP and R2 = 0.204 for direct RT-LAMP. For RNA RT-LAMP, there was a notable increase in Tp variance () after CT = 20 across both sample types. On saliva samples, for CT ≤20, and for CT >20; on swabs samples, for CT ≤20, and for CT >20. Given the relationship between CT value and viral load, this indicates that Tp is not a reliable indicator for viral load beyond the CT = 20 threshold.
SARS-CoV-2 Viral Culture of Clinical Samples across a CT Range
Although not a large sample size, a negative result via direct RT-LAMP indicates that the presence of culturable virus is less probable; and for samples with a CT >25 (RDRP/ORF1ab target), recoverable virus is less likely (Table 5). The sensitivity of 1 plaque-forming unit/mL of the viral culture assay is presented in Supplemental Table S3. No CPE was observed in the flasks inoculated with 0.1 or 0.01 plaque-forming units after the two passes. AVL samples were taken from the flasks at the beginning and end of each passage, and the CT values of the extracted nucleic acids are shown in Supplemental Table S3.
Table 5.
Viral Culture of Positive VTM from Oropharyngeal Swabs and Assay Results
| Sample | Direct RT-LAMP | RNA RT-LAMP | CT values for each RT-qPCR assay |
CPE | ||
|---|---|---|---|---|---|---|
| Genesig RDRP gene | VIASURE |
SARS-CoV-2 E gene Sarbeco assay | ||||
| ORF1ab | ||||||
| 1 | POS | POS | 19.9 | 18.7 | 17.8 | CPE+ |
| 2 | POS | POS | 21.3 | 19.9 | 19.0 | CPE+ |
| 3 | POS | POS | 21.6 | 19.1 | 18.5 | CPE+ |
| 4 | POS | POS | 22.6 | 20.8 | 19.8 | CPE+ |
| 5 | POS | POS | 22.9 | 21.6 | 21.0 | CPE+ |
| 6 | POS | POS | 23.7 | 20.6 | 20.6 | CPE+ |
| 7 | NEG | POS | — | ND | ND | No CPE |
| 8 | NEG | POS | 39.2 | ND | ND | No CPE |
| 9 | NEG | POS | 35.2 | ND | ND | No CPE |
| 10 | NEG | NEG | 34.6 | ND | ND | No CPE |
| 11 | NEG | POS | 35.4 | ND | ND | No CPE |
| 12 | NEG | POS | 36.2 | ND | ND | No CPE |
| 13 | POS | POS | 35.8 | ND | ND | No CPE |
| 14 | POS | POS | 34.5 | ND | ND | No CPE |
| 15 | NEG | POS | 35.1 | ND | ND | No CPE |
| 16 | POS | POS | 30.0 | ND | ND | No CPE |
| 17 | POS | POS | 32.3 | ND | ND | No CPE |
| 18 | NEG | POS | 34.6 | ND | ND | No CPE |
| 19 | POS | POS | 31.3 | ND | ND | No CPE |
| 20 | NEG | POS | 30.3 | ND | ND | No CPE |
| 21 | NEG | POS | 30.0 | ND | ND | No CPE |
| 22 | NEG | POS | 31.5 | ND | ND | No CPE |
| 23 | NEG | POS | 30.7 | ND | ND | CPE+ |
| 24 | POS | POS | 29.9 | ND | ND | No CPE |
| 25 | POS | POS | 29.4 | ND | ND | No CPE |
| 26 | NEG | POS | 28.2 | ND | ND | CPE+ |
Samples were taken through three passages.
CPE, cytopathic effect; ND, not done; NEG, negative; POS, positive; RT-LAMP, reverse-transcription loop-mediated isothermal amplification; VTM, viral transport media.
Individual Time Course
In the time course experiment, SARS-CoV-2 RNA was detected from day 5 (at the onset of symptoms) up to day 12 after suspected initial exposure using direct RT-LAMP and up to day 13 by RNA RT-LAMP, encompassing the full 6 days where symptoms were recorded (Supplemental Table S4).
Performance of RT-LAMP across Viral Load Groups
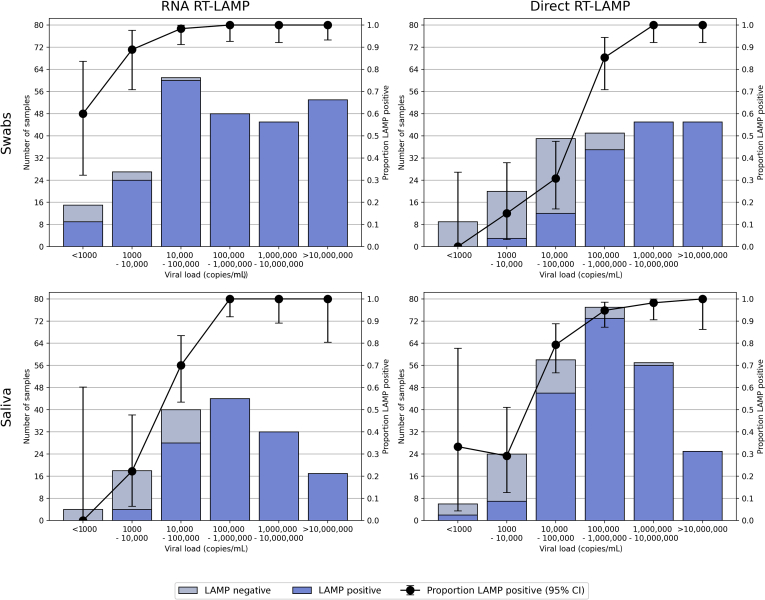
The sensitivity of the RNA and direct RT-LAMP assays across viral load groups is shown in Figure 2. For RNA RT-LAMP, samples that were positive by RT-qPCR containing >105 copies/mL were consistently identified as positive, with no samples returning a negative result. Below this copy number, sensitivity is reduced for both saliva and NP/OP swab samples, reaching approximately 60% in NP/OP swab samples exclusively with viral loads <103 copies/mL, and an approximately linear drop in sensitivity from 100% to 0% between viral loads of 105 and 103 copies/mL, respectively, in saliva samples. For direct RT-LAMP, all but one saliva sample were detected above 106 copies/mL. On swab samples, sensitivity is reduced on samples containing <105 copies/mL, dropping from 85% at viral loads of 105 to 106 copies/mL, to 30% in the 104 to 105 range. On saliva samples, sensitivity is reduced in the 104 to 105 range to a sensitivity of 80% but then reduces further within the 103 to 104 range, to 30%.
Figure 2.
Performance of the RNA reverse-transcription loop-mediated isothermal amplification (RT-LAMP) and direct RT-LAMP assays on both saliva and swab samples, according to viral load groupings.
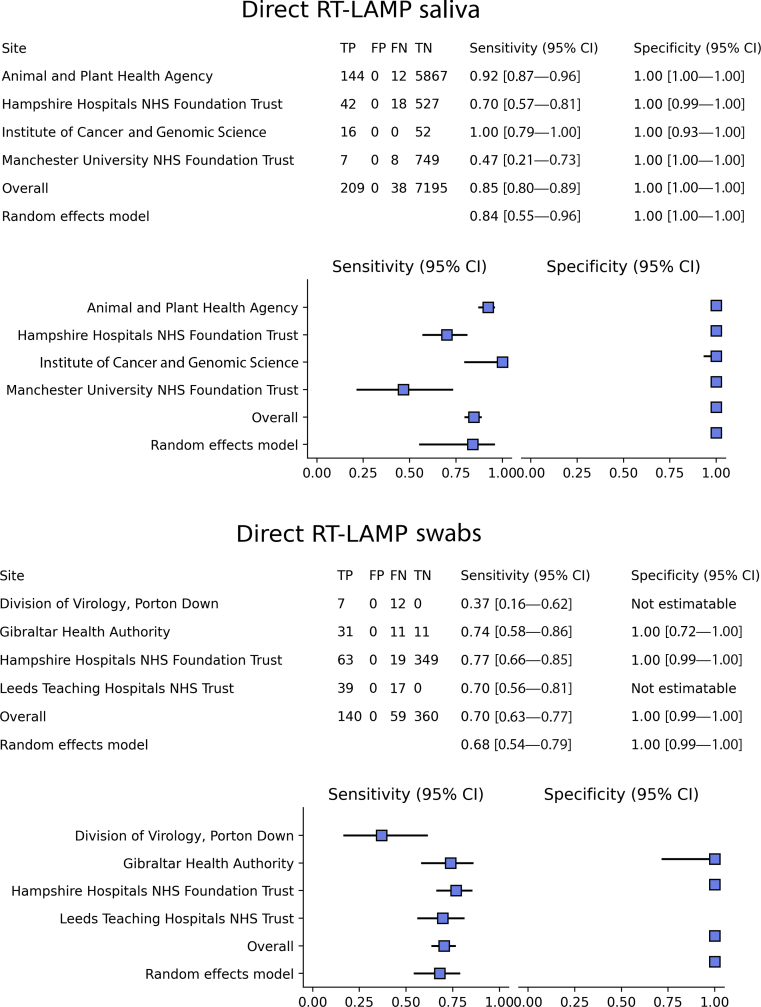
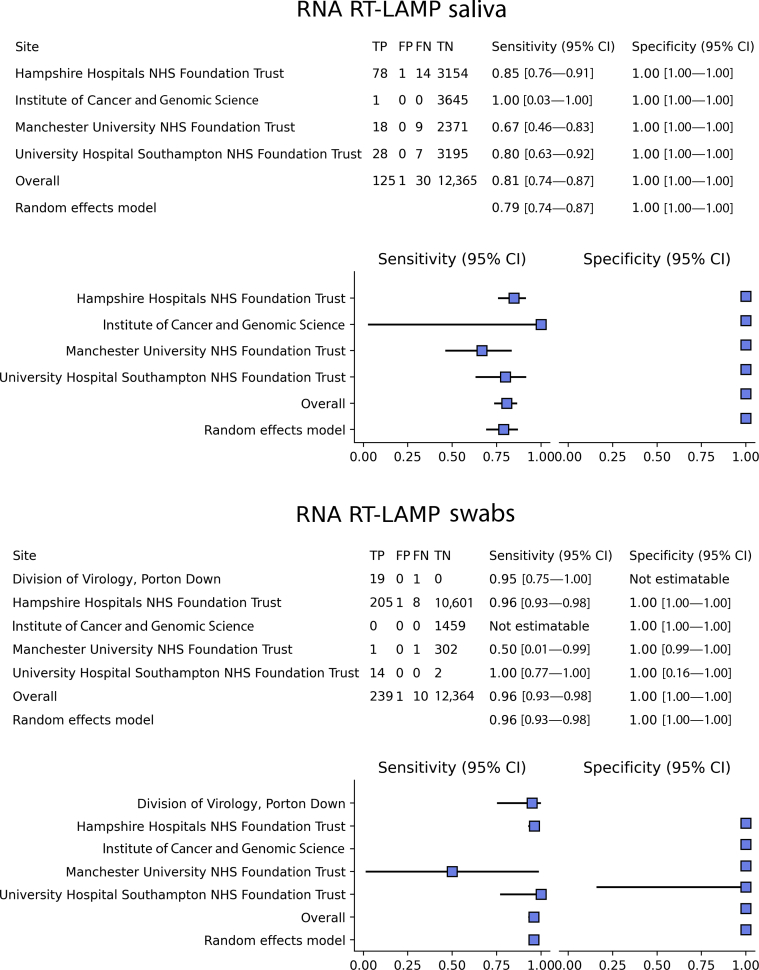
Site Meta-Analysis
Site-level confusion matrices, sensitivity, and specificity per method and sample type are shown in Figures 3 and 4. For specificity, heterogeneity between sites was minimal for all combinations of method and sample type, with the random effects model matching the overall aggregated sample calculation. For sensitivity, heterogeneity was minimal between sites for RNA RT-LAMP. However, for direct RT-LAMP, sensitivity showed significant overall heterogeneity (bivariate model variance: on saliva samples; on swab samples). Between-site variations in the viral load of the samples tested contributed a minority of the heterogeneity, but sensitivity was consistently high in samples with higher viral loads (ie, >106 copies/mL) (Figure 2), while being more heterogeneous between sites in samples with lower viral loads. Sensitivity at lower viral loads was highest in the sites with the most established testing programs.
Figure 3.
Forest plots for direct reverse-transcription loop-mediated isothermal amplification (RT-LAMP) per sample type, showing site heterogeneity in sensitivity and specificity, with overall estimates and the resulting expected sensitivity and specificity retrieved from each respective bivariate random effects model. FN, false negative; FP, false positive; NHS, National Health Service; TN, true negative; TP, true positive.
Figure 4.
Forest plots for RNA reverse-transcription loop-mediated isothermal amplification (RT-LAMP) per sample type, showing site heterogeneity in sensitivity and specificity, with overall estimates and the resulting expected sensitivity and specificity retrieved from each respective bivariate random effects model. FN, false negative; FP, false positive; NHS, National Health Service; TN, true negative; TP, true positive.
Discussion
Testing of human populations for SARS-CoV-2 nucleic acid has been hampered by both logistical (eg, swab availability) and physical (eg, discomfort from repeated swab testing) constraints. The aim of this study was to evaluate an optimized sample preparation method, building on previously published methods for the extraction-free detection of SARS-CoV-2 by RT-LAMP primarily from saliva.3,19 The salivary glands are reported to be early targets of SARS-CoV-2 infection,24 and studies have demonstrated the detection of high viral loads of SARS-CoV-2 from saliva, similar to those observed from nasopharyngeal/oropharyngeal swabs.15,25, 26, 27 Collection of saliva is noninvasive and does not require a trained individual or specialist consumables for collection of a quality sample. Utilizing a noninvasive sampling method should open testing to more individuals who dislike or are unable to tolerate having a nasopharyngeal/oropharyngeal swab taken.28 In addition, several studies have demonstrated that SARS-CoV-2 viral RNA could be detected from saliva for a similar duration after onset of clinical signs when compared with combined NP/OP swabs,29, 30, 31 highlighting saliva as a valuable tool for SARS-CoV-2 detection.
Direct detection negates the requirement for RNA extraction,32,33 for which there has previously been competition for reagents and often requires expensive extraction equipment, including liquid handling automation. This extraction-free method decreases turnaround time from sample collection to result. The direct RT-LAMP method is straightforward and rapid, allowing the test to be performed in a wide range of settings, including near patient hospital laboratories and pop-up or mobile laboratories. However, previously evaluated extraction-free sample preparation methods using RT-LAMP from saliva samples have demonstrated reduced sensitivity,3,19 likely because of the inhibitory factors found within saliva that may affect molecular tests, such as RT-LAMP and RT-qPCR.34,35 The simple sample preparation method evaluated in the study aimed to improve on these methods by utilizing the addition of a novel proprietary reagent, RapiLyze, designed to neutralize common sample inhibitors. A subsequent heat step of 98°C for 2 minutes before addition to the RT-LAMP master mix renders SARS-CoV-2 inactive, as confirmed by infectivity analysis using live virus inactivation studies (Supplemental Tables S1 and S2). Downstream steps are then able to proceed outside of traditional containment-level laboratory settings, broadening its clinical utility.
This study utilized high numbers of combined nasopharyngeal/oropharyngeal swabs (n = 559) and saliva samples (n = 86,760) for the evaluation of this novel sample preparation method in combination with the direct RT-LAMP assay. RNA RT-LAMP was also performed on >25,000 samples for comparison, providing updated values for the performance of the assay reported previously.3,19,36 Correlation between CT value and sample viral copy number has been demonstrated within this and other studies, with lower CT values (CT <25 and <33) indicating a higher probability that the sample contains recoverable active virus, and consequently the likelihood that the individual may be infectious to others.4,25,37, 38, 39, 40 As a result, the RNA and direct RT-LAMP assays were compared with RT-qPCR results in groups with three different CT cutoff values: <45, <33, and <25. This was completed so that the performance of the assays in different clinical scenarios (use case) could be determined: i) CT <45: does the RT-LAMP assay (either RNA or direct) compare with RT-qPCR for all reportable CT values?; ii) CT <33: can the RT-LAMP assay detect those individuals who have medium-high levels of viral RNA in their specimens, with an ORF1ab target being analogous with viral copy number because it is exclusively a genomic target?22; and iii) CT <25: can the RT-LAMP assay detect those individuals who have a high level of viral RNA in their specimens?
Diagnostic sensitivity for RNA RT-LAMP on swab and saliva samples was improved when compared with a previous report utilizing this method,3 with values of >96% and >80%, respectively, when considering all CT values, and 100% for both sample types when considering CT <25, with these samples having a high probability of containing replicating virus for >24,000 samples tested. Direct RT-LAMP sensitivity on swab samples was also improved from the previous method, with 100% sensitivity for CT <25, 77.78% for CT <33, and 70.35% for CT <45 across 559 samples used for this evaluation. In contrast, sensitivity for direct RT-LAMP on saliva was in general higher than that determined for swabs (CT <33 = 87.61%, and CT <45 = 84.62%), apart from the group with CT values of <25, which had a reported sensitivity of 99.01%. These results support previous reports that demonstrate comparable performance when comparing paired swabs and saliva samples,41,42 and that one sample type is not superior to the other. Interestingly, the diagnostic sensitivity for RNA and direct RT-LAMP for saliva samples was almost equivalent (80.65% and 84.62%, respectively), suggesting that RNA extraction may not even be required when performing testing on saliva samples. Direct RT-LAMP also demonstrates a higher sensitivity than a wide variety of lateral flow tests in the CT < 25, CT ≥ 25, and overall categories, with the overall sensitivity of direct RT-LAMP on saliva samples achieving a higher overall sensitivity than 94 of 96 lateral flow tests previously evaluated.43 The correlation between PCR CT value and the direct RT-LAMP Tp was weaker for saliva than for swabs, which may reflect the PCR CT value being from a nasopharyngeal swab and recognized time course differences between initial viral infection of the salivary glands and later infection of the respiratory tract.26,30
Previous studies have described the importance of identifying asymptomatic individuals, particularly those with high viral loads.28,44, 45, 46, 47, 48 The ability of the direct RT-LAMP assay to reliably detect individuals with medium-high viral loads in a simple to collect, noninvasive sampling process highlights the suitability of this assay for both symptomatic and asymptomatic population screening. This is particularly important in health care and care home staff, where the use of asymptomatic COVID-19 screening would reduce the risk of onward transmission of SARS-CoV-2, consequently maintaining NHS capacity and Social Care capacity, and more important, reducing the risk to vulnerable individuals present within those environments.36
When designing surveillance strategies for asymptomatic infection testing as an intervention to reduce transmission, frequency of testing and result turnaround time may be considered more significant than diagnostic sensitivity.49 Gold standard tests with high sensitivity, such as RT-qPCR, generally need to be performed in centralized testing facilities, often resulting in increased reporting times, leading to a less effective control of viral transmission.49 In contrast, point-of-care tests, such as lateral flow tests,43,50 or those requiring only a basic/mobile laboratory set-up, such as direct RT-LAMP, which have the ability to produce rapid results, can be performed frequently (eg, daily or multiple times per week). Consequently, the likelihood of sampling an individual when his/her viral load is highest, as seen in the early, often presymptomatic stages of infection, increases, maximizing the probability of rapidly detecting infectious cases, allowing prompt isolation. In this use case, sampling and testing frequency using a rapid assay with suitable accuracy in detection of medium-high viral loads, but not necessarily optimal sensitivity over the whole range, including low to very low viral loads, is desirable or necessary.49,51 Frequent on-site testing of asymptomatic NHS health care workers using direct RT-LAMP has been successfully implemented in the pilot study described herein; and continues to be utilized. Direct RT-LAMP has also been used in a mass community based pilot in school and higher education settings,36 to identify those individuals who may have been missed when surveillance relies only on symptomatic individuals coming forward for testing. With the use of mobile or pop-up laboratories, direct RT-LAMP could also be used for risk-based mass testing (eg, targeting specific geographic areas or vulnerable groups). The potential also exists for lyophilization of the direct RT-LAMP reagents reported in other studies,52,53 which would minimize the necessity for trained personnel by reducing pipetting steps and the requirement for a cold chain, allowing greater capacity of the assay in multi-use case scenarios, including point of care, and in low- and middle-income countries.
Several experiments typical of a diagnostic performance evaluation were not performed as part of this study, as they had been performed and reported previously. This included both analytical specificity, which, when tested against a panel of respiratory pathogens causing indistinguishable clinical signs to COVID-19, demonstrated a high level of analytical specificity (100% in this case)3; and analytical sensitivity of the direct RT-LAMP, which is reported to detect 1000 copies/mL.3,36,41 In addition, the RNA and direct RT-LAMP assays evaluated as part of this study have been shown to reliably detect the emerging variants of concern, including the B.1.1.7 alpha variant, the 501Y.V2 beta variant, the P1 gamma variant, and the new rapidly spreading B.1.617.2 delta variant54,55 (https://www.gov.uk/government/collections/new-sars-cov-2-variant, last accessed June 2021). The emergence of further variants of concern could lead to a criticism of the RT-LAMP assay because of its reliance on a single target, ORF1ab, where mutations in the target region in a sample could lead to false negatives. For RT-qPCR, this has been observed during the current pandemic,56, 57, 58 where at least a dual target assay is recommended.59 However, this is less likely to occur for the RT-LAMP assay used in this pilot evaluation. First, this is because of the multiple sets of primer pairs utilized, three pairs, with two pairs within the target region. This builds in redundancy to mutation not unlike a duplex RT-qPCR. Second, the ORF1ab region is highly conserved and crucial for viral replication and fitness in SARS-CoV-2. As a result, these regions are well maintained using a proofreading system via the nsp14 protein,60 resulting in a more stable genome compared with many other RNA viruses.
The authors highlight the importance of incorporating an inhibition control into the next iteration of the RT-LAMP assays. The paired RT-LAMP and RT-qPCR data from this study show a good correlation, and any false-negative results were likely due to the analytical sensitivity of the RT-LAMP assay, not sample-driven inhibition. To this end, a control primer set by OptiGene Ltd was evaluated (PS-0010), targeting the human ribosomal protein LO gene. Preliminary analysis of the inhibition control primers showed consistent detection across 279 saliva and 381 combined nasopharyngeal/oropharyngeal swab samples using both RNA and direct RT-LAMP (B.A., S.P.K., A.G., unpublished data). Incorporation of this inhibition control into the RT-LAMP assays would alleviate a potential limitation of the current assays and further support quality assurance for use within a clinical diagnostic setting. One further limitation to LAMP assays is the potential for contamination from assay product, which can be significant. LAMP assays produce vast amounts that can persist in the environment, not only causing potential false-positive results in subsequent testing but also anomalous results in laboratory workers who are part of a SARS-CoV-2 testing program.61 Therefore, it is crucial that appropriate waste streams are in place to mitigate this risk.
This study demonstrated high sensitivity and specificity for a novel sample preparation method used for SARS-CoV-2 direct RT-LAMP, particularly in samples from which the individual would likely be considered infectious, highlighting the usefulness of saliva as a simple to collect, noninvasive sample type. The highly sensitive RNA RT-LAMP assay provides a rapid alternative with a reliance on differing reagents and equipment to RT-qPCR testing, thus providing additional diagnostic capacity and redundancy through diversity. Direct RT-LAMP may complement existing surveillance tools for SARS-CoV-2 testing, including other point-of-care and laboratory-based diagnostics, and is applicable to a variety of clinical scenarios, such as frequent, interval-based testing of asymptomatic individuals who may be missed when reliance is on symptomatic testing alone. However, care should be taken when considering frequency of testing, messaging around the role and interpretation of asymptomatic rapid tests, integration of data storage and access, and the challenges faced when scaling up surveillance to large populations.
The role out of a new testing strategy can often throw up interesting and unexpected experiences. These collective experiences and lessons learnt from setting up an NHS asymptomatic staff testing program using direct RT-LAMP will be shared in a future publication.
Conclusions
Rapid diagnostic testing at scale to identify and isolate symptomatic and asymptomatic individuals potentially transmitting infectious SARS-CoV-2 is an essential part of the response to the COVID-19 pandemic. RT-LAMP on both extracted RNA and directly on crude samples potentially provides faster turnaround times than reverse transcription quantitative real-time PCR testing, with a higher sensitivity and specificity than antigen lateral flow devices. Increasing evidence points to potential benefits of SARS-CoV-2 testing using saliva rather than nasopharyngeal/oropharyngeal swabs; therefore, a multisite evaluation of an improved simple sample preparation method for direct SARS-CoV-2 RT-LAMP was undertaken. This study demonstrated that the RNA RT-LAMP assay has high sensitivity and specificity, providing a rapid alternative to RT-qPCR testing with a reliance on differing reagents and equipment. The simple SARS-CoV-2 direct RT-LAMP preparation method also demonstrated high sensitivity and specificity for detecting SARS-CoV-2 in saliva and nasopharyngeal/oropharyngeal swabs from asymptomatic and symptomatic individuals, notably in saliva samples from which the individual would likely be considered infectious. The findings highlight the usefulness of saliva as a simple to collect, noninvasive sample type, potentially applicable for interval-based testing of asymptomatic individuals.
Footnotes
OptiGene Ltd. supplied reagents free of charge for this study. Supported in part by a UK Department of Health and Social Care award to the University of Southampton grant reference number 2020/032 (feasibility study for city-wide testing using saliva 315 based LAMP testing). The views expressed are those of the authors and not necessarily those of the Department of Health and Social Care. K.M.G. is supported by the UK Medical Research CouncilMC_UU_12011/4, the National Institute for Health Research (NIHR) Senior Investigator NF-SI-0515-318 10042 and NIHR Southampton Biomedical Research Center IS-BRC-1215-20004, and the British Heart FoundationRG/15/17/3174. For part of this project, E.L.A.H. was on secondment at GeneSys Biotech Ltd, which was part supported by The Pirbright Institute Flexible Talent Mobility Account under Biotechnology and Biological Sciences Research Council grant BB/S507945/1. A.D.B. is currently supported by a Cancer Research UK Advanced Clinician Scientist award C31641/A23923, and his laboratory is supported by CRUK Center Birmingham C17422/A25154 and the Birmingham Experimental Cancer Medicine CenterC11497/A25127. V.L.F., S.P.K., B.A., and Z.D. were on secondment to the Department of Health and Social Care for a period during this study. Animal and Plant Health Agency laboratory activities and expertise were supported by both the Safe and Certain projectAPHACSKL0085 and Defra projectAPHANSOM0416.
S.P.K. and D.B. contributed equally to this work as co-authors.
V.L.F. contributed as senior author.
Disclosures: M.A. is an employee of OptiSense Limited, and N.M. is an employee of GeneSys Biotech Limited; neither had any role in study design or data analysis. K.M.G. is part of an academic consortium that has received research funding from Abbott Nutrition, Nestec, BenevolentAI Bio Ltd, and Danone.
Supplemental material for this article can be found at http://doi.org/10.1016/j.jmoldx.2021.12.007.
Supplemental Data
References
- 1.Zhang J., Dong X., Cao Y., Yuan Y., Yang Y., Yan Y., Akdis C., Gao Y. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy. 2020;75:1730–1741. doi: 10.1111/all.14238. [DOI] [PubMed] [Google Scholar]
- 2.Corman V.M., Landt O., Kaiser M., Molenkamp R., Meijer A., Chu D.K.W., Bleicker T., Brünink S., Schneider J., Schmidt M.L., Mulders D.G.J.C., Haagmans B.L., Van Der Veer B., Van Den Brink S., Wijsman L., Goderski G., Romette J.L., Ellis J., Zambon M., Peiris M., Goossens H., Reusken C., Koopmans M.P.G., Drosten C. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance. 2020;25:23–30. doi: 10.2807/1560-7917.ES.2020.25.3.2000045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fowler V.L., Armson B., Gonzales J.L., Wise E.L., Howson E.L.A., Vincent-Mistiaen Z., Fouch S., Maltby C.J., Grippon S., Munro S., Jones L., Holmes T., Tillyer C., Elwell J., Sowood A., de Peyer O., Dixon S., Hatcher T., Patrick H., Laxman S., Walsh C., Andreou M., Morant N., Clark D., Moore N., Houghton R., Cortes N.J., Kidd S.P. A highly effective reverse-transcription loop-mediated isothermal amplification (RT-LAMP) assay for the rapid detection of SARS-CoV-2 infection. J Infect. 2020;82:117–125. doi: 10.1016/j.jinf.2020.10.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Moreira V.M., Mascarenhas P., Machado V., Botelho J., Mendes J.J., Taveira N., Almeida M.G. Diagnosis of SARS-Cov-2 infection by RT-PCR using specimens other than naso- and oropharyngeal swabs: a systematic review and meta-analysis. Diagnostics (Basel) 2021;11:363. doi: 10.3390/diagnostics11020363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.de Paula Eduardo F., Bezinelli L.M., de Araujo C.A.R., Moraes J.V.V., Birbrair A., Pinho J.R.R., Hamerschlak N., Al-Hashimi I., Heller D. Self-collected unstimulated saliva, oral swab, and nasopharyngeal swab specimens in the detection of SARS-CoV-2. Clin Oral Investig. 2022;26:1561–1567. doi: 10.1007/s00784-021-04129-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Teo A.K.J., Choudhury Y., Tan I.B., Cher C.Y., Chew S.H., Wan Z.Y., Cheng L.T.E., Oon L.L.E., Tan M.H., Chan K.S., Hsu L.Y. Saliva is more sensitive than nasopharyngeal or nasal swabs for diagnosis of asymptomatic and mild COVID-19 infection. Sci Rep. 2021;11:3134. doi: 10.1038/s41598-021-82787-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ott I.M., Strine M.S., Watkins A.E., Boot M., Kalinich C.C., Harden C.A., Vogels C.B.F., Casanovas-Massana A., Moore A.J., Muenker M.C., Nakahata M., Tokuyama M., Nelson A., Fournier J., Bermejo S., Campbell M., Datta R., Cruz C.S.D., Farhadian S.F., Ko A.I., Iwasaki A., Grubaugh N.D., Wilen C.B., Wyllie A.L. Stability of SARS-CoV-2 RNA in nonsupplemented saliva. Emerg Infect Dis. 2021;27:1146–1150. doi: 10.3201/eid2704.204199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Matic N., Stefanovic A., Leung V., Lawson T., Ritchie G., Li L., Champagne S., Romney M.G., Lowe C.F. Practical challenges to the clinical implementation of saliva for SARS-CoV-2 detection. Eur J Clin Microbiol Infect Dis. 2021;40:447–450. doi: 10.1007/s10096-020-04090-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fajnzylber J., Regan J., Coxen K., Corry H., Wong C., Rosenthal A., Worrall D., Giguel F., Piechocka-trocha A., Atyeo C., Fischinger S., Chan A., Flaherty K.T., Hall K., Dougan M., Ryan E.T., Gillespie E., Chishti R., Li Y., Jilg N., Hanidziar D., Baron R.M., Baden L., Tsibris A.M., Armstrong K.A., Kuritzkes D.R., Alter G., Walker B.D., Yu X., Li J.Z., Consortium M. SARS-CoV-2 viral load is associated with increased disease severity and mortality. Nat Commun. 2020;11:5493. doi: 10.1038/s41467-020-19057-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Borghi E., Massa V., Zuccotti G., Wyllie A.L. Testing saliva to reveal the submerged cases of the COVID-19 iceberg. Front Microbiol. 2021;12:10–12. doi: 10.3389/fmicb.2021.721635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee R.A., Herigon J.C., Benedetti A., Pollock N.R., Denkinger C.M. Performance of saliva, oropharyngeal swabs, and nasal swabs for SARS-CoV-2 molecular detection: a systematic review and meta-analysis. J Clin Microbiol. 2021;59 doi: 10.1128/JCM.02881-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wong S.C.Y., Tse H., Siu H.K., Kwong T.S., Chu M.Y., Yat F., Yau S., Yu I., Cheung Y., Wing C., Tse S., Poon K.C., Cheung K.C., Wu T.C., Wai J., Chan M., Cheuk W., Lung D.C. Posterior oropharyngeal saliva for the detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Clin Infect Dis. 2020;2:2939–2946. doi: 10.1093/cid/ciaa797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Iwasaki S., Fujisawa S., Nakakubo S., Kamada K., Yamashita Y., Fukumoto T., Sato K., Oguri S., Taki K., Senjo H., Sugita J., Hayasaka K., Konno S., Nishida M., Teshima T. Comparison of SARS-CoV-2 detection in nasopharyngeal swab and saliva. J Infect. 2020;81:e145–e147. doi: 10.1016/j.jinf.2020.05.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Aydin S., Benk I.G., Geckil A.A. May viral load detected in saliva in the early stages of infection be a prognostic indicator in COVID-19 patients? J Virol Methods. 2021;294:114198. doi: 10.1016/j.jviromet.2021.114198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhu J., Guo J., Xu Y., Chen X. Viral dynamics of SARS-CoV-2 in saliva from infected patients. J Infect. 2020;81:e48–e50. doi: 10.1016/j.jinf.2020.06.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Azzi L., Baj A., Alberio T., Lualdi M., Veronesi G., Carcano G., Ageno W., Gambarini C., Maffioli L., Saverio S.D., Gasperina D.D., Genoni A.P., Premi E., Donati S., Azzolini C., Grandi A.M., Dentali F., Tangianu F., Sessa F., Maurino V., Tettamanti L., Siracusa C., Vigezzi A., Monti E., Iori V., Iovino D., Ietto G., Grossi P.A., Tagliabue A., Fasano M. Rapid salivary test suitable for a mass screening program to detect SARS-CoV-2: a diagnostic accuracy study. J Infect. 2020;81:e75–e78. doi: 10.1016/j.jinf.2020.06.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yokota I., Shane P.Y., Okada K., Unoki Y., Yang Y., Inao T., Sakamaki K., Iwasaki S., Hayasaka K., Sugita J., Nishida M., Fujisawa S., Teshima T. Mass screening of asymptomatic persons for severe acute respiratory syndrome coronavirus 2 using saliva. Clin Infect Dis. 2021;73:e559–e565. doi: 10.1093/cid/ciaa1388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Masse S., Bonnet C., Vilcu A.M., Benamar H., Swital M., van der Werf S., Carrat F., Hanslik T., Blanchon T., Falchi A. Are posterior oropharyngeal saliva specimens an acceptable alternative to nasopharyngeal sampling for the monitoring of sars-cov-2 in primary-care settings? Viruses. 2021;13:1–11. doi: 10.3390/v13050761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Howson E.L.A., Kidd S.P., Armson B., Goring A., Sawyer J., Cassar C., Cross D., Lewis T., Hockey J., Rivers S., Cawthraw S., Banyard A., Anderson P., Rahou S., Andreou M., Morant N., Clark D., Walsh C., Laxman S., Houghton R., Slater-Jefferies J., Costello P., Brown I., Cortes N., Godfrey K.M., Fowler V.L. Preliminary optimisation of a simplified sample preparation method to permit direct detection of SARS-CoV-2 within saliva samples using reverse-transcription loop-mediated isothermal amplification (RT-LAMP) J Virol Methods. 2021;289:114048. doi: 10.1016/j.jviromet.2020.114048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Reitsma J.B., Glas A.S., Rutjes A.W.S., Scholten R.J.P.M., Bossuyt P.M., Zwinderman A.H. Bivariate analysis of sensitivity and specificity produces informative summary measures in diagnostic reviews. J Clin Epidemiol. 2005;58:982–990. doi: 10.1016/j.jclinepi.2005.02.022. [DOI] [PubMed] [Google Scholar]
- 21.Bhargava A. Dynamic aspects of ORF1ab and N RNA cycle threshold values among COVID-19 patients in China. Infect Genet Evol. 2021;87:104657. doi: 10.1016/j.meegid.2020.104657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Alexandersen S., Chamings A., Bhatta T.R. SARS-CoV-2 genomic and subgenomic RNAs in diagnostic samples are not an indicator of active replication. Nat Commun. 2020;11:6059. doi: 10.1038/s41467-020-19883-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xu T., Chen C., Zhu Z., Cui M., Chen C., Dai H., Xue Y. Clinical features and dynamics of viral load in imported and non-imported patients with COVID-19. Int J Infect Dis. 2020;94:68–71. doi: 10.1016/j.ijid.2020.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Huang N., Pérez P., Kato T., Mikami Y., Okuda K., Gilmore R.C., et al. SARS-CoV-2 infection of the oral cavity and saliva. Nat Med. 2021;27:892–903. doi: 10.1038/s41591-021-01296-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.To K.K.W., Tsang O.T.Y., Leung W.S., Tam A.R., Wu T.C., Lung D.C., Yip C.C.Y., Cai J.P., Chan J.M.C., Chik T.S.H., Lau D.P.L., Choi C.Y.C., Chen L.L., Chan W.M., Chan K.H., Ip J.D., Ng A.C.K., Poon R.W.S., Luo C.T., Cheng V.C.C., Chan J.F.W., Hung I.F.N., Chen Z., Chen H., Yuen K.Y. Temporal profiles of viral load in posterior oropharyngeal saliva samples and serum antibody responses during infection by SARS-CoV-2: an observational cohort study. Lancet Infect Dis. 2020;20:565–574. doi: 10.1016/S1473-3099(20)30196-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tan S.H., Allicock O., Armstrong-Hough M., Wyllie A.L. Saliva as a gold-standard sample for SARS-CoV-2 detection. Lancet Respir Med. 2021;2600:19–21. doi: 10.1016/S2213-2600(21)00178-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Savela E.S., Winnett A., Romano A.E., Porter M.K., Shelby N., Akana R., Ji J. Quantitative SARS-CoV-2 viral-load curves in paired saliva and nasal swabs inform appropriate respiratory sampling site and analytical test sensitivity required for earliest viral detection. medRxiv. 2021 doi: 10.1101/2021.04.02.21254771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yang Q., Saldi T.K., Gonzales P.K., Lasda E., Decker C.J., Tat K.L., Fink M.R., Hager C.R., Davis J.C., Ozeroff C.D., Muhlrad D., Clark S.K., Fattor W.T., Meyerson N.R., Paige C.L., Gilchrist A.R., Barbachano-Guerrero A., Worden-Sapper E.R., Wu S.S., Brisson G.R., McQueen M.B., Dowell R.D., Leinwand L., Parker R., Sawyer S.L. Just 2% of SARS-CoV-2−positive individuals carry 90% of the virus circulating in communities. Proc Natl Acad Sci U S A. 2021;118:1–6. doi: 10.1073/pnas.2104547118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Uwamino Y., Nagata M., Aoki W., Fujimori Y., Nakagawa T., Yokota H., Sakai-Tagawa Y., Iwatsuki-Horimoto K., Shiraki T., Uchida S., Uno S., Kabata H., Ikemura S., Kamata H., Ishii M., Fukunaga K., Kawaoka Y., Hasegawa N., Murata M. Accuracy and stability of saliva as a sample for reverse transcription PCR detection of SARS-CoV-2. J Clin Pathol. 2021;74:67–68. doi: 10.1136/jclinpath-2020-206972. [DOI] [PubMed] [Google Scholar]
- 30.Wyllie A.L., Fournier J., Casanovas-Massana A., Campbell M., Tokuyama M., Vijayakumar P., et al. Saliva or nasopharyngeal swab specimens for detection of SARS-CoV-2. N Engl J Med. 2020;383:1283–1286. doi: 10.1056/NEJMc2016359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vogels C.B.F., Watkins A.E., Harden C.A., Brackney D.E., Shafer J., Wang J., Caraballo C., Kalinich C.C., Ott I.M., Fauver JR S., Kudo E., Lu P., Venkataraman A., Tokuyama M., Moore A.J., Muenker M.C. Casanovas-Massana A, Fournier J, Bermejo S, Campbell M, Datta R, Nelson A, Yale IMPACT Research Team, Dela Cruz CS, Ko AI, Iwasaki A, Krumholz HM, Matheus JD, Hui P, Liu C, Farhadian SF, Sikka R, Wyllie AL, Grubaugh ND. SalivaDirect: a simplified and flexible platform to enhance SARS-CoV-2 testing capacity. Med. 2021;2:263–280.e6. doi: 10.1016/j.medj.2020.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Buck M.D., Poirier E.Z., Cardoso A., Frederico B., Canton J., Barrell S., et al. SARS-CoV-2 detection by a clinical diagnostic RT-LAMP assay. Wellcome Open Res. 2021;6:9. doi: 10.12688/wellcomeopenres.16517.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kellner M.J., Matl M., Ross J.J., Schnabl J., Handler D., Heinen R., Schaeffer J., Hufnagl P., Indra A., Dekens M.P., Fritsche-Polanz R., Födinger M., Zuber J., Allerberger F., Pauli A., Brennecke J., BioCenter Program V. Head-to-head comparison of direct-input RT-PCR and RT-LAMP against RT-qPCR on extracted RNA for rapid SARS-CoV-2 diagnostics. medRxiv. 2021 doi: 10.1101/2021.01.19.21250079. [DOI] [Google Scholar]
- 34.Ochert A.S., Boulter A.W., Birnbaum W., Johnson N.W., Teo C.G. Inhibitory effect of salivary fluids on PCR: potency and removal. PCR Methods Appl. 1994;3:365–368. doi: 10.1101/gr.3.6.365. [DOI] [PubMed] [Google Scholar]
- 35.Wilson I.G. Inhibition and facilitation of nucleic acid amplification. Appl Environ Microbiol. 1997;63:3741–3751. doi: 10.1128/aem.63.10.3741-3751.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Department of Health and Social Care . Department of Health and Social Care; London, UK: 2020. Rapid Evaluation of OptiGene RT-LAMP Assay (Direct and RNA Formats) [Google Scholar]
- 37.La Scola B., Le Bideau M., Andreani J., Hoang V.T., Grimaldier C., Colson P., Gautret P., Raoult D. Viral RNA load as determined by cell culture as a management tool for discharge of SARS-CoV-2 patients from infectious disease wards. Eur J Clin Microbiol Infect Dis. 2020;39:1059–1061. doi: 10.1007/s10096-020-03913-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bullard J., Dust K., Funk D., Strong J.E., Alexander D., Garnett L., Boodman C., Bello A., Hedley A., Schiffman Z., Doan K., Bastien N., Li Y., van Caeseele P.G., Poliquin G. Predicting infectious severe acute respiratory syndrome coronavirus 2 from diagnostic samples. Clin Infect Dis. 2020;71:2663–2666. doi: 10.1093/cid/ciaa638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wölfel R., Corman V.M., Guggemos W., Seilmaier M., Zange S., Müller M.A., Niemeyer D., Jones T.C., Vollmar P., Rothe C., Hoelscher M., Bleicker T., Brünink S., Schneider J., Ehmann R., Zwirglmaier K., Drosten C., Wendtner C. Virological assessment of hospitalized patients with COVID-2019. Nature. 2020;581:465–469. doi: 10.1038/s41586-020-2196-x. [DOI] [PubMed] [Google Scholar]
- 40.Marc A., Kerioui M., Blanquart F., Bertrand J., Mitjà O., Corbacho-Monné M., Marks M., Guedj J. Quantifying the relationship between SARS-CoV-2 viral load and infectiousness. Elife. 2021;10 doi: 10.7554/eLife.69302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fowler D.V., Douglas D.A., Godfrey P.K., Williams P.A., Beggs P.A., Kidd S., Cortes D.N., Wilcox P.M., Davies D.K., Smith M., Hill P.D.S. Critical evaluation of the methodology used by Wilson-Davies et al., (2020) entitled “Concerning the Optigene Direct LAMP assay, and it`s use in at-risk groups and hospital staff.”. J Infect. 2021;82:282–327. doi: 10.1016/j.jinf.2021.01.012. [DOI] [PubMed] [Google Scholar]
- 42.Hanson K.E., Barker A.P., Hillyard D.R., Gilmore N., Barrett J.W., Orlandi R.R., Shakir S.M. Self-collected anterior nasal and saliva specimens versus healthcare worker-collected nasopharyngeal swabs for the molecular detection of SARS-CoV-2. J Clin Microbiol. 2020;58:e01824-20. doi: 10.1128/JCM.01824-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Scheiblauer H., Filomena A., Nitsche A., Puyskens A., Corman V.M., Drosten C., Zwirglmaier K., Lange C., Emmerich P., Müller M., Knauer O., Nübling C.M. Comparative sensitivity evaluation for 122 CE-marked rapid diagnostic tests for SARS-CoV-2 antigen, Germany, September 2020 to April 2021. Eurosurveillance. 2021;26:1–13. doi: 10.2807/1560-7917.ES.2021.26.44.2100441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nikolai L.A., Meyer C.G., Kremsner P.G., Velavan T.P. Asymptomatic SARS coronavirus 2 infection: invisible yet invincible. Int J Infect Dis. 2020;100:112–116. doi: 10.1016/j.ijid.2020.08.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Marks M., Millat P., Ouchi D., Roberts C., Alemany A., Corbacho-Monné M., Ubals M., Vall-Mayans M., G-Beiras C, Prat N., Ara J., Clotet B., Mitjà O. Transmission of COVID-19 in 282 clusters in Catalonia, Spain: a cohort study. Lancet Infect Dis. 2021;21:6290. doi: 10.1016/S1473-3099(20)30985-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Byambasuren O., Cardona M., Bell K., Clark J., McLaws M.L., Glasziou P. Estimating the extent of asymptomatic COVID-19 and its potential for community transmission: systematic review and meta-analysis. J Assoc Med Microbiol Infect Dis Canada. 2020;5:223–234. doi: 10.3138/jammi-2020-0030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Buitrago-Garcia D., Egli-Gany D., Counotte M.J., Hossmann S., Imeri H., Ipekci A.M., Salanti G., Low N. Occurrence and transmission potential of asymptomatic and presymptomatic SARS-CoV-2 infections: a living systematic review and meta-analysis. PLoS Med. 2020;17 doi: 10.1371/journal.pmed.1003346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Johansson M.A., Quandelacy T.M., Kada S., Prasad P.V., Steele M., Brooks J.T., Slayton R.B., Biggerstaff M., Butler J.C. SARS-CoV-2 transmission from people without COVID-19 symptoms. JAMA Netw Open. 2021;4:1–8. doi: 10.1001/jamanetworkopen.2020.35057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Larremore D.B., Wilder B., Lester E., Shehata S., Burke J.M., Hay J.A., Tambe M., Mina M.J., Parker R. Test sensitivity is secondary to frequency and turnaround time for COVID-19 screening. Sci Adv. 2021;7:1–11. doi: 10.1126/sciadv.abd5393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lee L.Y.W., Rozmanowski S., Pang M., Charlett A., Anderson C., Hughes G.J., Barnard M., Peto L., Vipond R., Sienkiewicz A., Hopkins S., Bell J., Crook D.W., Gent N., Walker A.S., Peto T.E.A., Eyre D.W. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infectivity by viral load, S gene variants and demographic factors, and the utility of lateral flow devices to prevent transmission. Clin Infect Dis. 2022;74:407–415. doi: 10.1093/cid/ciab421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kissler S.M., Fauver J.R., Mack C., Olesen S.W., Tai C., Shiue K.Y., Kalinich C.C., Jednak S., Ott I.M., Vogels C.B.F., Wohlgemuth J., Weisberger J., DiFiori J., Anderson D.J., Mancell J., Ho D.D., Grubaugh N.D., Grad Y.H. Viral dynamics of acute SARS-CoV-2 infection and applications to diagnostic and public health strategies. PLoS Biol. 2021;19:1–17. doi: 10.1371/journal.pbio.3001333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Howson E.L.A., Armson B., Madi M., Kasanga C.J., Kandusi S., Sallu R., Chepkwony E., Siddle A., Martin P., Wood J., Mioulet V., King D.P., Lembo T., Cleaveland S., Fowler V.L. Evaluation of two lyophilized molecular assays to rapidly detect foot-and-mouth disease virus directly from clinical samples in field settings. Transbound Emerg Dis. 2017;64:861–871. doi: 10.1111/tbed.12451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Armson B., Walsh C., Morant N., Fowler V.L., Knowles N.J., Clark D. The development of two field-ready reverse transcription loop-mediated isothermal amplification assays for the rapid detection of Seneca Valley virus 1. Transbound Emerg Dis. 2019;66:497–504. doi: 10.1111/tbed.13051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Horby P., Huntley C., Davies N., Edmunds J., Ferguson N., Medley G., Hayward A., Cevik M., Semple C. Sage; London, UK: 2021. NERVTAG Note on B.1.1.7 Severity; pp. 5–8. [Google Scholar]
- 55.Tegally H., Wilkinson E., Giovanetti M., Iranzadeh A., Fonseca V., Giandhari J., et al. Emergence and rapid spread of a new severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2) lineage with multiple spike mutations in South Africa. medRxiv. 2020 doi: 10.1101/2020.12.21.20248640. [Preprint] doi: [DOI] [Google Scholar]
- 56.Vanaerschot M., Mann S.A., Webber J.T., Kamm J., Bell S.M., Bell J., Hong S.N., Nguyen M.P., Chan L.Y., Bhatt K.D., Tan M., Detweiler A.M., Espinosa A., Wu W., Batson J., Dynerman D., Wadford D.A., Puschnik A.S., Neff N., Ahyong V., Miller S., Ayscue P., Tato C.M., Paul S., Kistler A.L., DeRisi J.L., Crawford E.D. Identification of a polymorphism in the N gene of SARS-CoV-2 that adversely impacts detection by reverse transcription-PCR. J Clin Microbiol. 2020;59:e02369-20. doi: 10.1128/JCM.02369-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Hasan M.R., Sundararaju S., Manickam C., Mirza F., Al-Hail H., Lorenz S., Tang P. A novel point mutation in the N gene of SARS-CoV-2 may affect the detection of the virus by reverse transcription-quantitative PCR. J Clin Microbiol. 2021;59:e03278-20. doi: 10.1128/JCM.03278-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Artesi M., Bontems S., Göbbels P., Franckh M., Maes P., Boreux R., Meex C., Melin P., Hayette M.-P., Bours V., Durkin K. A recurrent mutation at position 26340 of SARS-CoV-2 is associated with failure of the E gene quantitative reverse transcription-PCR utilized in a commercial dual-target diagnostic assay. J Clin Microbiol. 2020;58:e01598-20. doi: 10.1128/JCM.01598-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wang R., Hozumi Y., Yin C., Wei G.-W. Mutations on COVID-19 diagnostic targets. Genomics. 2020;112:5204–5213. doi: 10.1016/j.ygeno.2020.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rausch J.W., Capoferri A.A., Katusiime M.G., Patro S.C., Kearney M.F. Low genetic diversity may be an Achilles heel of SARS-CoV-2. Proc Natl Acad Sci U S A. 2020;117:24614–24616. doi: 10.1073/pnas.2017726117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Robinson-McCarthy L.R., Mijalis A.J., Filsinger G.T., de Puig H., Donghia N.M., Schaus T.E., Rasmussen R.A., Ferreira R., Lunshof J.E., Chao G., Ter-Ovanesyan D., Dodd O., Kuru E., Sesay A.M., Rainbow J., Pawlowski A.C., Wannier T.M., Angenent-Mari N.M., Najjar D., Yin P., Ingber D.E., Tam J.M., Church G.M. Laboratory-generated DNA can cause anomalous pathogen diagnostic test results. Microbiol Spectr. 2021;9:1–9. doi: 10.1128/Spectrum.00313-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.