Abstract
Background
Cases of coronavirus disease 2019 (COVID-19) have emerged in discrete waves. We explored temporal trends in the reporting of COVID-19 in inflammatory bowel disease (IBD) patients.
Methods
The Surveillance Epidemiology of Coronavirus Under Research Exclusion for Inflammatory Bowel Disease (SECURE-IBD) is an international registry of IBD patients diagnosed with COVID-19. The average percent changes (APCs) were calculated in weekly reported cases of COVID-19 during the periods of March 22 to September 12, September 13 to December 12, 2020, and December 13 to July 31, 2021.
Results
Across 73 countries, 6404 cases of COVID-19 were reported in IBD patients. COVID-19 reporting decreased globally by 4.2% per week (95% CI, −5.3% to −3.0%) from March 22 to September 12, 2020, then climbed by 10.2% per week (95% CI, 8.1%-12.3%) from September 13 to December 12, 2020, and then declined by 6.3% per week (95% CI, −7.8% to −4.7%). In the fall of 2020, weekly reporting climbed in North America (APC, 11.3%; 95% CI, 8.8-13.8) and Europe (APC, 17.7%; 95% CI, 12.1%-23.5%), whereas reporting was stable in Asia (APC, −8.1%; 95% CI, −15.6-0.1). From December 13, 2020, to July 31, 2021, reporting of COVID-19 in those with IBD declined in North America (APC, −8.5%; 95% CI, −10.2 to −6.7) and Europe (APC, −5.4%; 95% CI, −7.2 to −3.6) and was stable in Latin America (APC, −1.5%; 95% CI, −3.5% to 0.6%).
Conclusions
Temporal trends in reporting of COVID-19 in those with IBD are consistent with the epidemiological patterns COVID-19 globally.
Keywords: inflammatory bowel disease, COVID-19, global, temporal
Introduction
Since the World Health Organization declared coronavirus disease 2019 (COVID-19) a global pandemic on March 11, 2020,1 the number of cases has dramatically risen across the world: from 200 000 in mid-March 2020 to 200 million by July 2021.2 Spread of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus has varied by geography, striking Asia and Europe initially, followed by North America and then Latin America. Public health measures such as hand hygiene, physical distancing, masks, home isolation, societal restrictions or lockdowns, contact tracing, and testing have reduced rates of viral transmission.3 However, implementation of these measures has varied by region and despite these efforts, multiple waves of infection have ensued.4
The highest prevalence of inflammatory bowel disease (IBD) is in countries of the Western world, though the incidence of IBD is rapidly rising in newly industrialized countries in Asia, Africa, and Latin America.5-10 Patients with IBD may have an increased risk of infection and complications of COVID-19 due to disease activity and drug-induced immunocompromised status.11-13 Consequently, several gastroenterology organizations have published guidelines to support clinical management during the pandemic and to promote practices to minimize the spread of infection in patients with IBD.14,15 Guidelines, in conjunction with public health measures, may have reduced the infection of COVID-19 in those with IBD over the course of the pandemic.16,17
We explored temporal trends in the submission of cases of COVID-19 in patients with IBD to an international, voluntary reporting registry, stratified across geographic regions throughout the world compared with global trends of reporting COVID-19 in the general population.
Materials and Methods
The Surveillance Epidemiology of Coronavirus Under Research Exclusion for Inflammatory Bowel Disease (SECURE-IBD) is an international registry of IBD patients with COVID-19 (www.covidibd.org).18-20 Physicians enter anonymized COVID-19 cases in people with Crohn’s disease or ulcerative colitis into a secure online REDCap (Research Electronic Data Capture) database. Reporting into the SECURE-IBD registry occurs for both ambulatory and hospitalized individuals with IBD who test positive for COVID-19. To allow for assessment of outcomes of COVID-19, cases are reported after the resolution of acute disease or death. The database undergoes quality control regularly with record matching (eg, age, sex, country) used to remove potential duplicate cases.19 Additionally, data of COVID-19 cases in the general population were obtained from Johns Hopkins University’s COVID-19 database (https://github.com/CSSEGISandData/COVID-19).21
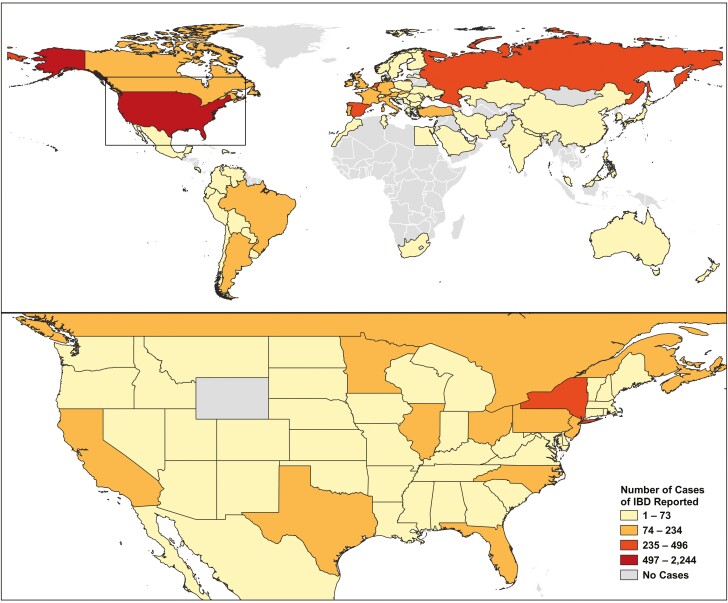
A choropleth map to illustrate geographic differences in reported cases was created using QGIS 3.44 (Open Source Geospatial Foundation, Chicago, Illinois, USA, https://qgis.org/) and stratified using Jenks Natural Breaks.22,23 A total of 46 reports that did not contain a country of origin were excluded from all analyses. For the United States, an additional choropleth map was designed denoting the distribution of reported cases by state. The maps were based on available data as of July 31, 2021. Reported cases of COVID-19 in patients with IBD were stratified into regions based on the United Nations Geoscheme:24 North America (the US and Canada), Latin America and the Caribbean (including Mexico), Asia, Europe, Africa, and Oceania. The proportion of COVID-19 among those with IBD was calculated per 100 000 persons diagnosed with COVID-19 in the continent based on data from the John Hopkins University COVID-19 database.21
For temporal analyses, cases of IBD were amalgamated on a weekly basis, starting the week of March 22-28, 2020, and ending the week of July 25-31, 2021. The first week of data capture in SECURE-IBD (March 13-21) was excluded from analysis to account for low awareness at the launch of the registry. Temporal analyses were stratified by North America (the US and Canada), Latin America and the Caribbean (including Mexico), Asia, and Europe. Individual-level temporal trend analyses of Africa and Oceania were excluded due to reporting only 42 cases of COVID-19 in IBD over the course of the pandemic, likely reflecting the low number of COVID-19 infections in Oceania and low prevalence of IBD in Africa. A separate analysis of temporal trends was carried out for the 4 Census Regions of the United States: Midwest, Northeast, South, and West.25
Weekly reporting of cases was used in Stata V15.1 to determine the average percent change (APC) in weekly cases of COVID-19 in those with IBD. Poisson regression was used on counts or negative binomial regression if appropriate, using week as the predictor variable. Temporal analyses were stratified by the first (March 22-28 to September 6-12, 2020), second (September 13-19 to December 6-12, 2020), and third period (December 13-19, 2020, to July 25–31, 2021) of the pandemic. The transition date in September was selected to correspond to simulation models predicting the onset of the second wave of COVID-19 in the general population,26 whereas the transition date in December corresponds to the onset of the approval of the first COVID-19 vaccines.27
Ethical Considerations
The UNC-Chapel Hill Office for Human Research Ethics concluded that the storage and analysis of de-identified data for SECURE-IBD registry does not constitute human subjects research and does not require IRB approval. The University of Calgary’s Conjoint Health Research Ethics Board approved the protocol for data visualization as containing adequate safeguards for the protection of privacy (ethics certificate: REB20-0535) due to the data being de-identified.
Results
During the period of March 13, 2020, to July 31, 2021, a total of 6404 cases from 73 countries and dependencies and 50 states and territories within the United States were reported to the SECURE-IBD registry. Table 1 shows the proportion of cases reported to SECURE-IBD by total COVID-19 cases within world regions and US regions. Figure 1 shows the distribution of reported cases globally and a breakdown of states within the United States. An online dashboard with an interactive map displaying the current global distribution of patients with IBD and COVID-19 is available and is updated monthly (https://kaplan-secure-ibd-ucalgary.hub.arcgis.com/).
Table 1.
Proportion of COVID-19 in patients with IBD by population size and number of COVID-19 cases in the population stratified by continents and quadrants within the United States: March 13, 2020 to July 31, 2021.
| COVID-19 Cases Reported in the SECURE-IBD Registry | COVID-19 Cases Reported in the Countries or States Reporting in the SECURE-IBD Registry | Proportion of COVID-19 Cases reported to the SECURE-IBD (per 100 000)∗ | |
|---|---|---|---|
| World∗∗ | 6,404 | 178,155,029 | 3.59 (3.51, 3.68) |
| Africa | 29 | 3,944,809 | 0.74 (0.49, 1.06) |
| Asia | 412 | 50,300,051 | 0.82 (0.73, 0.90) |
| Europe | 3,110 | 48,398,556 | 6.43 (6.20, 6.66) |
| Latin America | 342 | 39,057,623 | 0.47 (0.76, 0.97) |
| North America | 2,416 | 36,416,733 | 6.63 (6.37, 6.90) |
| Oceania | 13 | 37,257 | 34.89 (18.58, 59.66) |
| United States∗∗∗ | 2,231 | 34,817,970 | 6.41 (6.14, 6.68) |
| Midwest | 681 | 7,495,739 | 9.09 (8.42, 9.79) |
| Northeast | 596 | 5,843,208 | 10.20 (9.40, 11.05) |
| South | 629 | 13,823,528 | 4.55 (4.20, 4.92) |
| West | 254 | 7,655,495 | 3.32 (2.92, 3.75) |
∗Proportion of COVID-19 cases reported to the SECURE-IBD registry relative to all COVID-19 cases in the general population among countries or states reporting data into the SECURE-IBD registry (per 100,000).
∗∗826 reported cases of COVID-19 in patients with IBD did not report the country of origin.
∗∗∗Five reported cases of COVID-19 in patients with IBD were from United States territories outside of the 4 regions listed here.
Figure 1.
Map of COVID-19 in IBD patients reported to SECURE-IBD: March 13 to July 25-31, 2021. Online interactive dashboard that is updated weekly: https://kaplan-secure-ibd-ucalgary.hub.arcgis.com/.
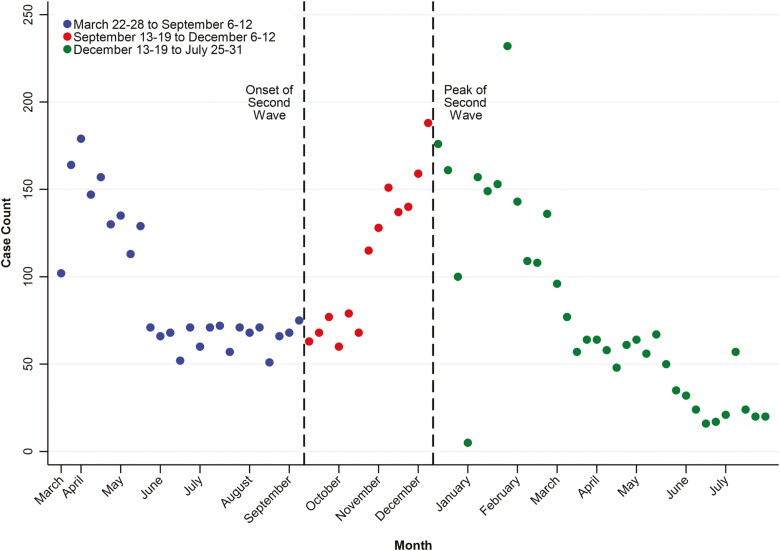
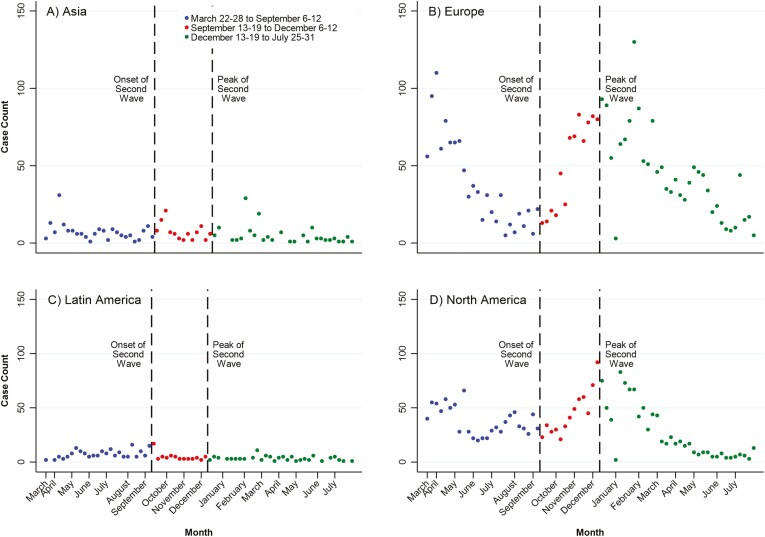
Table 2 shows the weekly temporal trend analyses, and Figures 2-4 display the weekly number of cases by world regions and US regions. Overall, reporting of COVID-19 in patients with IBD across the world declined by 4.2% per week (95% CI, −5.3% to −3.0%) from March 22-28 to September 6-12, 2020, then rose by 10.2% per week (95% CI, 8.1% to 12.3%) from September 13-19 to December 6-12. It then declined by 6.2% per week (95% CI, −7.5% to −4.9%) from December 13-19, 2020, to July 25-31, 2021 (Table 1, Figure 2). Temporal patterns varied by geography (Table 1, Figure 3). Europe and North America shared temporal patterns following a first and second wave of reporting COVID-19 in those with IBD (Table 1, Figure 3). In contrast, reporting in Asia declined by 3.7% per week (95% CI, −6.9% to −0.4%) from March 22-28 to September 6-12 and then stabilized thereafter. In Latin America, reporting increased by 3.5% per week (95% CI, 0.9% to 6.2%) from March 22-28 to September 6-12 and then significantly decreased thereafter (−10.2%; 95% CI, −16.2% to −3.8%). Supplemental Figure 1 displays the temporal patterns of COVID-19 diagnoses in the general population for Asia, Europe, Latin America, and North America.
Table 2.
Average weekly percent change of reports of COVID-19 in IBD patients stratified by continents and quadrants within the United States: March 22-28 to September 6-12; September 13-19 to December 6-12, 2020; and December 13-19, 2020, to July 25-31, 2021
| Region | March 22-28 to September 6-12 | September 13-19 to December 6-12 | December 13-19, 2020, to July 25-31, 2021 |
|---|---|---|---|
| World | −4.2 (−5.3,−3.0) | 10.2 (8.1, 12.3) | −6.3 (−7.8,−4.7) |
| Asia | −3.7 (−6.9,−0.4) | −8.1 (−15.6, 0.1) | −5.3 (−8.7,−1.8) |
| Europe | −8.9 (−10.8,−6.9) | 17.7 (12.1, 23.5) | −5.4 (−7.2,−3.6) |
| Latin America | 3.4 (1.2, 5.6) | −10.2 (−16.2,−3.8) | −1.5 (−3.5, 0.6) |
| North America | −1.9 (−3.5,−0.4) | 11.1 (8.1, 14.1) | −8.5 (−10.2,−6.7) |
| United States | −1.7 (−3.3,−0.1) | 10.9 (7.7, 14.2) | −9.2 (−11.2,−7.1) |
| Midwest | −0.4 (−3.0. 2.3) | 17.0 (10.8, 23.6) | −9.8 (−12.5,−7.0) |
| Northeast | −10.0 (−12.3,−7.6) | 19.2 (11.5, 27.5) | −8.2 (−11.5,−4.9) |
| South | 5.5 (2.6, 8.5) | −0.8 (−6.8, 5.5) | −5.9 (−7.7,−4.0) |
| West | −0.4 (−3.5, 2.8) | 9.5 (−0.9, 20.9) | −6.8 (−10.2,−3.2) |
Figure 2.
Global temporal trends in reporting of COVID-19 in patients with IBD from the SECURE-IBD registry: March 22-28 to September 6-12; September 13-19 to December 6-12, 2020; and December 13-19, 2020 to July 25-31, 2021.
Figure 3.
Global regional temporal trends in reporting of COVID-19 in patients with IBD from the SECURE-IBD registry: (A) Asia, (B) Europe, (C) Latin America, and (D) North America: March 22-28 to September 6-12; September 13-19 to December 6-12, 2020; and December 13-19, 2020 to July 25-31, 2021.
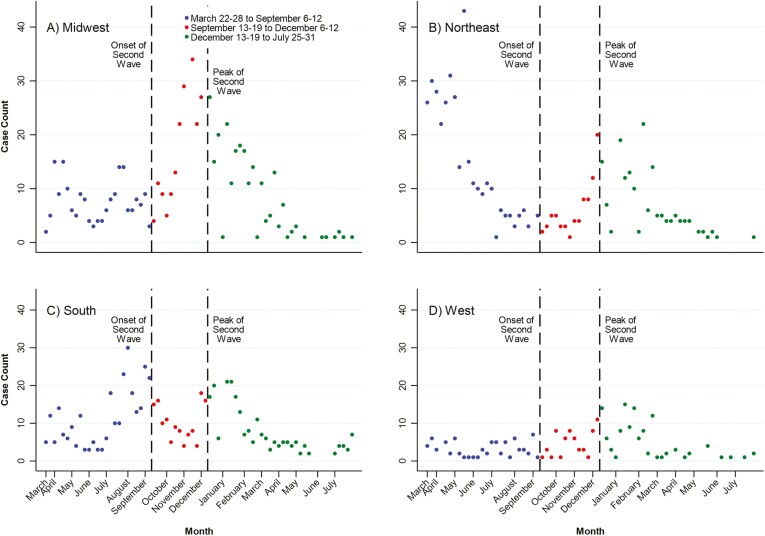
Stratified analyses within the United States showed geographic heterogeneity during the first 2 periods of the pandemic (Table 1, Figure 4). The Midwest was stable (APC, −0.4%; 95% CI, −2.3-1.6) before September 13-19, but it increased leading up to December 6-12, (APC, 17.0%; 95% CI, 10.8-23.6). In contrast, the Northeast decreased (APC, −10.0%; 95% CI, −12.3 to −7.6) and then increased (APC, 19.2%; 95% CI, 11.5-27.5). The South increased (APC, 5.5%; 95% CI, 2.6-8.5) and then stabilized (APC, −0.8; 95% CI, −6.8-5.5). The weekly reporting of COVID-19 in patients with IBD was stable in the West during both the first (APC, −0.4%; 95% CI, −3.5-2.8) and second (APC, 9.5%; 95% CI, −0.9-20.9) periods. However, after December 13-19, reporting of COVID-19 in those with IBD in the United States steadily decreased by 9.2% per week (95% CI, −11.2 to −7.12), and this decline was observed in all 4 regions of the US (Table 1, Figure 4).
Figure 4.
United States regional temporal trends in reporting of COVID-19 in patients with IBD from the SECURE-IBD registry: (A) Midwest, (B) Northeast, (C) South, and (D) West: March 22-28 to September 6-12; September 13-19 to December 6-12, 2020; and December 13-19, 2020 to July 25-31, 2021.
Discussion
Globally, the trends of COVID-19 in those with IBD declined by 4.2% per week through the spring and summer of 2020, followed by a steadily rising rate of 10.2% per week in the fall of 2020. After mid-December, rates declined by 6.3% per week up to July 2021. The temporal trends in reporting COVID-19 in patients with IBD parallels the epidemiological patterns of the different waves of COVID-19 in the general population. The waves of reporting COVID-19 in people with IBD were largely driven by cases in Europe and North America due to escalating cases of COVID-19 in the general population in these continents and the high prevalence of IBD in the Western world.6,28
Although our study was not designed to explain observed temporal trends, we can speculate several factors that may explain the temporal patterns of reporting of COVID-19 in those with IBD throughout 2021 into the SECURE-IBD registry. First, reporting of COVID-19 in those with IBD was similar to the frequency of COVID-19 infections in the general population and the corresponding prevalence of IBD in the general population.6,28 North America and Europe had the highest incidence of COVID-19 in the general population and, likewise, reporting COVID-19 in those with IBD was highest in these regions. North America and Europe have higher prevalence of IBD in the general population compared with Asia and Latin America; this higher prevalence contributed to greater case ascertainments of COVID-19 in the IBD population in North American and European countries. In Oceania, which has a prevalence of IBD similar to North America and Europe,6 public health measures resulted in low penetration of COVID-19 in the general population;4 therefore, the absolute number of cases reported from Oceania was low, but the proportion of IBD cases with COVID-19 relative to the number of cases of COVID-19 in the general population was high.
Second, those with IBD who were actively flaring or taking corticosteroids may have been at higher risk of acquiring COVID-19 and experiencing more severe outcomes.18,20,29 Additionally, those with IBD may have followed public health guidelines more strictly than the general population due to fears associated with being immunocompromised. Leading IBD organizations like the International Organization for the study of IBD (IOIBD),14 British Society of Gastroenterology,15 Crohn’s and Colitis Foundation,30 and Crohn’s and Colitis Canada31 held symposia and struck taskforces to develop recommendations to guide patients with IBD during the pandemic. Communication strategies directed at the IBD community and their health care providers focused on avoiding flare of disease activity by maintaining medical management and implementing protective measures to reduce transmission: home isolation, physical distancing, and hand hygiene, followed later by advocating for the use of masks. Early analyses in Asia and Europe suggest that these public health measures may have reduced transmission of SARS-CoV-2 in the IBD population.17,32
Third, the availability of COVID-19 vaccines may have influenced temporal trend patterns. Throughout 2021, a steady decline in reporting of COVID-19 in those with IBD was reported in Europe and North America. The increased penetrance of vaccinations in those with IBD may explain the drop in cases of COVID-19 throughout Europe and North America.33 Access to vaccines was initially highest in countries with higher prevalence of IBD (ie, North America, Europe). Unfortunately, vaccine utilization is not recorded in the SECURE-IBD database, and thus, future studies are necessary to explore the impact of COVID-19 vaccines on temporal patterns in those with IBD.
Geographic heterogeneity in temporal trends of reporting COVID-19 in those with IBD occurred across the world but are best exemplified in the United States. The Northeast, in particular New York state, was the earliest to be inundated by COVID-19 but ultimately instituted public health measures that successfully controlled transmission. Reporting of IBD into the SECURE-IBD registry was initially high and then steadily decreased along with overall COVID-19 cases in the region. Whereas the South reported fewer overall COVID-19 cases in the beginning of the pandemic. By June 2020, the proportion of cases reported in the South was the highest in the nation and then subsequently declined.25 Moreover, COVID-19 has continuously climbed in the Midwest. The West reported low numbers of cases throughout the year. The heterogeneity in reporting across different regions in the United States may be explained by variation in public health measures across US states, penetrance of vaccination, and the transmission of variants (eg, Delta). Further, reporting practices of gastroenterologists may vary regionally, with possibly greater reporting in cities compared with rural regions.
The SECURE-IBD international registry was developed to track COVID-19-related outcomes in patients with IBD. Surveillance of reporting across time and geography offers the most complete picture of the impact of COVID-19 in the IBD population. The registry is strong because it is a large international collaborative effort that, to date, has identified more than 6000 SARS-CoV-2 infections in patients with IBD. However, important limitations of the registry should be noted, as they may explain some of temporal patterns observed in our study. First, cases are voluntarily reported by gastroenterologists; therefore, underreporting of cases is likely to occur, leading to underestimation of the true incidence. Furthermore, those with IBD on antitumor necrosis factor therapy may have milder manifestation of COVID-19, leading to under diagnosis of COVID-19.34 Also, cases are typically reported after the COVID-19 course has resolved, which means that weekly reports do not correspond to the exact date of diagnosis. Second, reporting by physicians may have waned over time, leading to declining temporal trends due to reporting bias. Also, the majority of patients are PCR-positive from nasopharyngeal swab: as such, regions with restricted or limited testing may underdiagnose the true prevalence of COVID-19 in patients with IBD, particularly missing mildly or asymptomatic individuals.
Although the SECURE-IBD registry records severe complications of COVID-19 (ie, hospitalization and death), these outcomes were too few to allow for temporal trend analyses stratified by COVID-19 severity. Furthermore, we lacked information on the penetrance of different medications for treatment of IBD in the general population stratified by different regions, which prevented us from conducting temporal analyses on the role of therapeutics on temporal patterns of COVID-19 reporting in the IBD population. Future temporal analyses of the pandemic will provide larger sample sizes and longer duration, leading to more robust insight into the temporal trends of COVID-19 in the IBD population.
Conclusions
After one year of reporting COVID-19 in those with IBD into the SECURE-IBD registry, our temporal trend findings are consistent with the epidemiological patterns COVID-19 globally. The highest incidence of COVID-19 in IBD occurs in the Western world where the prevalence of IBD predominates. Temporal trends in reporting show that most regions in the world have followed the patterns of their general populations. Europe and North America have observed a parabolic curve with downward trends throughout 2021 that may be secondary to public health measures, vaccination programs, or methodological considerations such as reporting biases. Africa, Asia, Latin America, and Oceania have few reported cases of COVID-19 in their IBD populations throughout the pandemic. Future studies should focus on the effectiveness of vaccination programs in conjunction with public health measures to reduce the transmission of SARS-CoV-2 in patients with IBD.
Supplementary Material
Acknowledgments
The authors would like to acknowledge all health care providers who reported cases to the SECURE-IBD registry. A list acknowledging all reporters can be found at https://covidibd.org/reporter-acknowledgment/.
Contributor Information
Gilaad G Kaplan, Departments of Medicine and Community Health Sciences, University of Calgary, Calgary, Alberta, Canada.
Fox E Underwood, Departments of Medicine and Community Health Sciences, University of Calgary, Calgary, Alberta, Canada.
Stephanie Coward, Departments of Medicine and Community Health Sciences, University of Calgary, Calgary, Alberta, Canada.
Manasi Agrawal, The Henry D. Janowitz Division of Gastroenterology, Icahn School of Medicine at Mount Sinai, New York, New York, USA.
Ryan C Ungaro, The Henry D. Janowitz Division of Gastroenterology, Icahn School of Medicine at Mount Sinai, New York, New York, USA.
Erica J Brenner, Division of Pediatric Gastroenterology, Department of Pediatrics, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA.
Richard B, Gearry, Department of Medicine, University of Otago, Christchurch, New Zealand.
Michele Kissous-Hunt, The Henry D. Janowitz Division of Gastroenterology, Icahn School of Medicine at Mount Sinai, New York, New York, USA; New York Gastroenterology Associates, New York, New York, USA.
James D Lewis, Department of Epidemiology and Biostatistics, The University of Pennsylvania, Philadelphia, Pennsylvania, USA.
Siew C Ng, Department of Medicine and Therapeutics, Institute of Digestive Disease, State Key Laboratory of Digestive Disease, Li Ka Shing Institute of Health Science, The Chinese University of Hong Kong, Hong Kong SAR, The People’s Republic of China.
Jean-Francois Rahier, Department of Gastroenterology, Université Catholique de Louvain, Yvoir, Belgium.
Walter Reinisch, Department Internal Medicine III, Division Gastroenterology & Hepatology, Medical University of Vienna, Vienna, Austria.
Flavio Steinwurz, Department of Gastroenterology, Hospital Israelita Albert Einstein, São Paulo, Brazil.
Xian Zhang, Department of Gastroenterology, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA.
Michael D Kappelman, Division of Pediatric Gastroenterology, Department of Pediatrics, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA.
Jean-Frederic Colombel, The Henry D. Janowitz Division of Gastroenterology, Icahn School of Medicine at Mount Sinai, New York, New York, USA.
Author Contributions
G.G.K.: conceptualization, formal analysis, investigation, methodology, writing of the original draft.
F.E.U. and S.C.: formal analysis, investigation, methodology, software, visualization, writing, review, and editing of article.
R.C.U. and E.J.B.: conceptualization, data curation, formal analysis, writing, review, and editing of article.
R.B.G., M.K.H, J.D.L., S.C.N., J.F.R, W.R., F.S., and M.A.: conceptualization, formal analysis, writing, review, and editing of article.
X.Z.: data curation, formal analysis, writing, review, and editing of article.
M.D.K.: conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, writing, review, and editing of article.
J.F.C.: conceptualization, data curation, formal analysis, investigation, writing, review, and editing of article.
Funding
This work was funded by the Helmsley Charitable Trust (2003-04445), National Center for Advancing Translational Sciences (UL1TR002489), a T32DK007634, and a K23KD111995-01A1. Additional funding provided by Pfizer, Takeda, Janssen, AbbVie, Lilly, Genentech, Boehringer Ingelheim, Bristol Myers Squibb, Celtrion, and Arenapharm. Canadian Institutes of Health Research Operating Grant: COVID-19 Rapid Research Funding Opportunity funding reference number VR5-172684.
Conflicts of Interest
G.G.K.: Honoraria for speaking or consultancy from AbbVie, Janssen, Pfizer, and Takeda. He has received research support from Ferring, Janssen, AbbVie, GlaxoSmith Kline, Merck, and Shire. Ownership of a patent: TREATMENT OF INFLAMMATORY DISORDERS, AUTOIMMUNE DISEASE, AND PBC. UTI Limited Partnership, assignee. Patent WO2019046959A1. PCT/CA2018/051098. September 7, 2018.
R.C.U.: Has served as an advisory board member or consultant for Eli Lilly, Janssen, Pfizer, and Takeda; research support from AbbVie, Boehringer Ingelheim, and Pfizer.
R.B.G.: Speaker fees and Scientific Advisory Boards for AbbVie and Janssen.
M.K.H.: Speaker/consultant for AbbVie, Janssen, Takeda.
J.D.L.: Personal fees from Johnson & Johnson Consumer Inc, grants, personal fees and other from Takeda Pharmaceuticals, personal fees and nonfinancial support from AbbVie, grants and personal fees from Janssen Pharmaceuticals, personal fees from Eli Lilly and Company, personal fees from Samsung Bioepis, personal fees from UCB, personal fees from Bristol-Myers Squibb, grants and personal fees from Nestle Health Science, personal fees from Bridge Biotherapeutics, personal fees from Celgene, personal fees from Merck, personal fees and other from Pfizer, personal fees from Gilead, personal fees from Arena Parmaceuticals, personal fees from Protagonist Therapeutics, personal fees from Entasis Therapeutics.
S.C.N.: Honoraria for speaking or consultancy from AbbVie, Janssen, Ferring, Tillotts and Takeda; research support from Ferring and AbbVie.
J.F.R.: Lecture fees from AbbVie, MSD, Takeda, Pfizer, Ferring, and Falk; consulting fees from AbbVie, Takeda, Hospira, Mundipharma, MSD, Pfizer, GlaxoSK, and Amgen; and research support from Takeda and AbbVie.
WR: Served as a speaker for Abbott Laboratories, AbbVie, Aesca, Aptalis, Astellas, Centocor, Celltrion, Danone Austria, Elan, Falk Pharma GmbH, Ferring, Immundiagnostik, Mitsubishi Tanabe Pharma Corporation, MSD, Otsuka, PDL, Pharmacosmos, PLS Education, Schering-Plough, Shire, Takeda, Therakos, Vifor, and Yakult. He has been a consultant for Abbott Laboratories, AbbVie, Aesca, Algernon, Amgen, AM Pharma, AMT, AOP Orphan, Arena Pharmaceuticals, Astellas, Astra Zeneca, Avaxia, Roland Berger GmBH, Bioclinica, Biogen IDEC, Boehringer-Ingelheim, Bristol-Myers Squibb, Cellerix, Chemocentryx, Celgene, Centocor, Celltrion, Covance, Danone Austria, DSM, Elan, Eli Lilly, Ernest & Young, Falk Pharma GmbH, Ferring, Galapagos, Genentech, Gilead, Grünenthal, ICON, Index Pharma, Inova, Intrinsic Imaging, Janssen, Johnson & Johnson, Kyowa Hakko Kirin Pharma, Lipid Therapeutics, LivaNova, Mallinckrodt, Medahead, MedImmune, Millenium, Mitsubishi Tanabe Pharma Corporation, MSD, Nash Pharmaceuticals, Nestle, Nippon Kayaku, Novartis, Ocera, OMass, Otsuka, Parexel, PDL, Periconsulting, Pharmacosmos, Philip Morris Institute, Pfizer, Procter & Gamble, Prometheus, Protagonist, Provention, Robarts Clinical Trial, Sandoz, Schering-Plough, Second Genome, Seres Therapeutics, Setpointmedical, Sigmoid, Sublimity, Takeda, Therakos, Theravance, Tigenix, UCB, Vifor, Zealand, Zyngenia, and 4SC. He has been an advisory board member for Abbott Laboratories, AbbVie, Aesca, Amgen, AM Pharma, Astellas, Astra Zeneca, Avaxia, Biogen IDEC, Boehringer-Ingelheim, Bristol-Myers Squibb, Cellerix, Chemocentryx, Celgene, Centocor, Celltrion, Danone Austria, DSM, Elan, Ferring, Galapagos, Genentech, Grünenthal, Inova, Janssen, Johnson & Johnson, Kyowa Hakko Kirin Pharma, Lipid Therapeutics, MedImmune, Millenium, Mitsubishi Tanabe Pharma Corporation, MSD, Nestle, Novartis, Ocera, Otsuka, PDL, Pharmacosmos, Pfizer, Procter & Gamble, Prometheus, Sandoz, Schering-Plough, Second Genome, Setpointmedical, Takeda, Therakos, Tigenix, UCB, Zealand, Zyngenia, and 4SC. He has received research funding from Abbott Laboratories, AbbVie, Aesca, Centocor, Falk Pharma GmbH, Immundiagnsotik, and MSD.
F.S.: Speaker for Janssen, Pfizer, and Takeda; and advisory board member for Amgen, Janssen, Pfizer, Sandoz, and Takeda.
M.D.K.: Consulted for AbbVie, Janssen, Pfizer, and Takeda, is a shareholder in Johnson & Johnson, and has received research support from Pfizer, Takeda, Janssen, AbbVie, Lilly, Genentech, Boehringer Ingelheim, Bristol Myers Squibb, Celtrion, and Arenapharm.
J.F.C.: Research grants from AbbVie, Janssen Pharmaceuticals and Takeda; receiving payment for lectures from AbbVie, Amgen, Allergan, Inc. Ferring Pharmaceuticals, Shire, and Takeda; receiving consulting fees from AbbVie, Amgen, Arena Pharmaceuticals, Boehringer Ingelheim, Celgene Corporation, Celltrion, Eli Lilly, Enterome, Ferring Pharmaceuticals, Genentech, Janssen Pharmaceuticals, Landos, Ipsen, Medimmune, Merck, Novartis, Pfizer, Shire, Takeda, Tigenix, Viela bio; and holds stock options in Intestinal Biotech Development and Genfit.
All other others have nothing to declare.
References
- 1. WHO Director-General. WHO Director-General’s opening remarks at the media briefing on COVID-19 - 11 March 2020. Accessed November 1, 2020. https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020
- 2. Wiersinga WJ, Rhodes A, Cheng AC, et al. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. Jama. 2020;324:782–793. [DOI] [PubMed] [Google Scholar]
- 3. Pan A, Liu L, Wang C, et al. Association of public health interventions with the epidemiology of the COVID-19 outbreak in Wuhan, China. Jama. 2020;323:1915–1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Han E, Tan MMJ, Turk E, et al. Lessons learnt from easing COVID-19 restrictions: an analysis of countries and regions in Asia Pacific and Europe. Lancet. 2020;396:1525–1534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Molodecky NA, Soon IS, Rabi DM, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012;142:46–54.e42; quiz e30. [DOI] [PubMed] [Google Scholar]
- 6. Ng SC, Shi HY, Hamidi N, et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2017;390:2769–2778. [DOI] [PubMed] [Google Scholar]
- 7. Kaplan GG. The global burden of IBD: from 2015 to 2025. Nat Rev Gastroenterol Hepatol. 2015;12:720–727. [DOI] [PubMed] [Google Scholar]
- 8. Kaplan GG, Windsor JW.. The four epidemiological stages in the global evolution of inflammatory bowel disease. Nat Rev Gastroenterol Hepatol. 2021;18:56–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kotze PG, Underwood FE, Damião AOMC, et al. Progression of inflammatory bowel diseases throughout Latin America and the caribbean: a systematic review. Clin Gastroenterol Hepatol. 2020;18:304–312. [DOI] [PubMed] [Google Scholar]
- 10. Kaplan GG, Ng SC.. Globalisation of inflammatory bowel disease: perspectives from the evolution of inflammatory bowel disease in the UK and China. Lancet Gastroenterol Hepatol. 2016;1:307–316. [DOI] [PubMed] [Google Scholar]
- 11. Suárez-Fariñas M, Tokuyama M, Wei G, et al. Intestinal inflammation modulates the expression of ACE2 and TMPRSS2 and potentially overlaps with the pathogenesis of SARS-CoV-2-related disease. Gastroenterology. 2021;160:287–301.e20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kaplan GG, Ng SC.. Understanding and preventing the global increase of inflammatory bowel disease. Gastroenterology. 2017;152:313–321.e2. [DOI] [PubMed] [Google Scholar]
- 13. Singh S, Al-Darmaki A, Frolkis AD, et al. Postoperative mortality among patients with inflammatory bowel diseases: a systematic review and meta-analysis of population-based studies. Gastroenterology. 2015;149:928–937. [DOI] [PubMed] [Google Scholar]
- 14. Rubin DT, Abreu MT, Rai V, Siegel CA, International Organization for the Study of Inflammatory Bowel D. . Management of patients with crohn’s disease and ulcerative colitis during the coronavirus disease-2019 pandemic: results of an international meeting. Gastroenterology. 2020;159(1):6–13 e16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. British Society of Gastroenterology. BSG multi-society guidance on further recovery of endoscopy services during the post-pandemic phase of COVID-19. Accessed November 1, 2020. https://www.bsg.org.uk/covid-19-advice/bsg-multi-society-guidance-on-further-recovery-of-endoscopy-services-during-the-post-pandemic-phase-of-covid-19/
- 16. An P, Ji M, Ren H, et al. Prevention of COVID-19 in patients with inflammatory bowel disease in Wuhan, China. Lancet Gastroenterol Hepatol. 2020;5:525–527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Fiorino G, Gilardi D, Radice S, et al. Absence of COVID-19 infection in patients accessing IBD unit at humanitas, milan: implications for postlockdown measures. Am J Gastroenterol. 2020;115:1719–1721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Brenner EJ, Ungaro RC, Gearry RB, et al. Corticosteroids, but not TNF antagonists, are associated with adverse COVID-19 outcomes in patients with inflammatory bowel diseases: results from an international registry. Gastroenterology. 2020;159:481–491.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Brenner EJ, Ungaro RC, Colombel JF, Kappelman MD.. IBD in the COVID-19 era: the value of international collaboration. Lancet Gastroenterol Hepatol. 2020;5:887–888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Ungaro RC, Brenner EJ, Gearry RB, et al. Effect of IBD medications on COVID-19 outcomes: results from an international registry. Gut. 2021;70:725–732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Dong E, Du H, Gardner L.. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect Dis. 2020;20:533–534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Jenks GF. The data model concept in statistical mapping. Int Yearb Carto. 1967;7:186–190. [Google Scholar]
- 23. Windsor JW, Underwood FE, Brenner E, et al. Data visualization in the era of COVID-19: an interactive map of the SECURE-IBD registry. Am J Gastroenterol. 2020;115:1923–1924. [DOI] [PubMed] [Google Scholar]
- 24. United Nations Statistics Division. Standard country or area codes for statistical use (M49). Accessed November 1, 2020. https://unstats.un.org/unsd/methodology/m49/
- 25. Oster AM, Kang GJ, Cha AE, et al. Trends in number and distribution of COVID-19 hotspot Counties - United States, March 8-July 15, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1127–1132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Cacciapaglia G, Cot C, Sannino F.. Second wave COVID-19 pandemics in Europe: a temporal playbook. Sci Rep. 2020;10:15514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Jackson LA, Anderson EJ, Rouphael NG, et al. ; mRNA-1273 Study Group. . An mRNA vaccine against SARS-CoV-2 - preliminary report. N Engl J Med. 2020;383:1920–1931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Coward S, Clement F, Benchimol EI, et al. Past and future burden of inflammatory bowel diseases based on modeling of population-based data. Gastroenterology. 2019;156:1345–1353.e4. [DOI] [PubMed] [Google Scholar]
- 29. Ricciuto A, Lamb CA, Benchimol EI, et al. Inflammatory bowel disease clinical activity is associated with COVID-19 severity especially in younger patients. J Crohns Colitis. 2021:jjab172. doi: 10.1093/ecco-jcc/jjab172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Crohn’s and Colitis Foundation. COVID-19 (Coronavirus): What IBD Patients Should Know. Accessed November 1, 2020. https://www.crohnscolitisfoundation.org/coronavirus/what-ibd-patients-should-know
- 31. Crohn’s and Colitis Canada. COVID-19 and IBD Recommendations. Accessed November 1, 2020. https://crohnsandcolitis.ca/Living-with-Crohn-s-Colitis/COVID-19-and-IBD
- 32. An P, Ji M, Ren H, et al. Prevention of COVID-19 in patients with inflammatory bowel disease in Wuhan, China. Lancet Gastroenterol Hepatol. 2020;5:525–527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wong S-Y, Dixon R, Pazos VM, Gnjatic S, Colombel J-F, Cadwell K.. Serological response to COVID-19 vaccination in IBD patients receiving biologics. medRxiv. 2021:2021.03.17.21253848. doi: 10.1101/2021.03.17.21253848. [DOI] [Google Scholar]
- 34. Papa A, Gasbarrini A, Tursi A, Epidemiology and the impact of therapies on the outcome of COVID-19 in patients with inflammatory bowel disease. Am J Gastroenterol. 2020;115:1722–1724. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.