Abstract
Stem cell-based therapy is a promising approach for treating a variety of disorders, including acute brain insults and neurodegenerative diseases. Stem cells such as mesenchymal stem cells (MSCs) secrete extracellular vesicles (EVs), circular membrane fragments (30 nm−1 μm) that are shed from the cell surface, carrying several therapeutic molecules such as proteins and microRNAs. Because EV-based therapy is superior to cell therapy in terms of scalable production, biodistribution, and safety profiles, it can be used to treat brain diseases as an alternative to stem cell therapy. This review presents evidences evaluating the role of stem cell-derived EVs in stroke, traumatic brain injury, and degenerative brain diseases, such as Alzheimer’s disease and Parkinson’ disease. In addition, stem cell-derived EVs have better profiles in biocompatibility, immunogenicity, and safety than those of small chemical and macromolecules. The advantages and disadvantages of EVs compared with other strategies are discussed. Even though EVs obtained from native stem cells have potential in the treatment of brain diseases, the successful clinical application is limited by the short half-life, limited targeting, rapid clearance after application, and insufficient payload. We discuss the strategies to enhance the efficacy of EV therapeutics. Finally, EV therapies have yet to be approved by the regulatory authorities. Major issues are discussed together with relevant advances in the clinical application of EV therapeutics.
Keywords: Exosomes, Extracellular vesicles, Mesenchymal stem cells, Neurodegeneration, Stroke, Traumatic brain injury
INTRODUCTION
Stem cell-based therapy is a promising approach for treating acute brain insults such as stroke and traumatic brain injury (TBI), and neurodegenerative diseases including Alzheimer’s disease (AD), Parkinson’s disease (PD), and Huntington’s disease (HD). Stem cells such as mesenchymal stem cells (MSCs) secrete extracellular vesicles (EVs), which carry several molecules such as proteins and microRNAs (miRNAs). Recent preclinical studies suggest that stem cell-derived EVs can be used to treat brain illness as an alternative to stem cell application.
This review presents evidence regarding the role of stem cell-derived EVs in acute and chronic neurological diseases in addition to discussing the advantages and disadvantages of EVs therapy versus other strategies. Major issues in the clinical application of EV therapeutics are discussed together with relevant advances in EV production/enrichment, isolation/purification, and quantification/characterization.
ADVANTAGES OF STEM CELL-DERIVED EXTRACELLULAR VESICLES OVER OTHER THERAPEUTIC STRATEGIES
Several therapeutic strategies have been introduced for prevent-ing or slowing the progression of brain damages and each has its own advantages and disadvantages (Table 1).
Table 1.
Strategies for acute and degenerative brain diseases
| Small chemicals or macromolecules | Drug delivery system | Stem cells | Stem cell-derived EV therapy | |
|---|---|---|---|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
EV, extracellular vesicle; MoA, mode of action; BBB, blood-brain barrier; MSCs, mesenchymal stem cells; FDA, Food and Drug Administration; RCT, randomized controlled trial.
Small chemicals or macromolecules showed a limited efficacy due to single mechanism of action and complex pathophysiology of brain diseases. For example, over 1,000 neuroprotective agents for acute stroke have been investigated in preclinical studies with promising results, but failed when tested in human (1). Similarly, trophic factors tested in various neurological diseases such as PD failed to show beneficial effects (2). For neuroprotection, a single target of neuroprotection will not provide the expected therapeutic effects, and signals that mediate cell death during the acute stage of ischemic insult might promote repair during the recovery phase (3). As a result, pleiotropic multi-target agents that act via multiple mechanisms of action to interrupt multiple steps may be more fruitful (4). In addition, almost no macromolecules and 98% of all small molecules do not cross the blood-brain barrier (BBB). Therefore, non-active vesicles such as adeno-associated virus capsids and polymer- or lipid-base nanoparticles were used to overcome the limitation. However, the use of drug delivery system increases the risk of toxicity, immunogenicity, and infection (5).
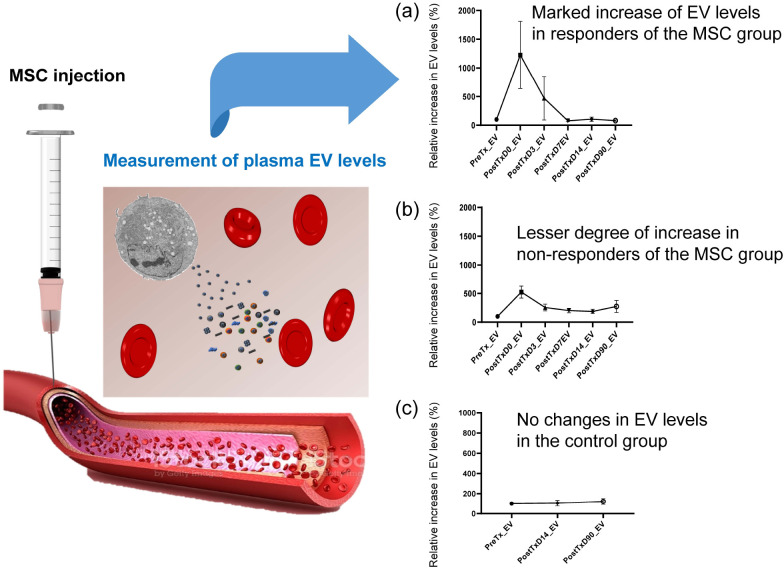
Cell-based therapy is a promising therapeutic approach against a range of neurological diseases. Unlike to small chemicals or macromolecules, MSCs harbor specific functions, such as regenerative, cytoprotective, and immunomodulatory properties. Application of MSC transplantation was safe in patients with neurological diseases. However, the beneficial effects were diverse among patients. For example, four randomized clinical trials (RCTs) of stem cells have been conducted in stroke patients, with mixed results (6-9). There are several possible reasons for this inconsistent result, including heterogeneity of patients, timing of therapy, and donor-to-donor or batch-to-batch variations. Our pre-specified biomarker sub-study showed that circulating EVs were markedly increased immediately after intravenous injection of MSCs (10). In this study, the number of the circulating EVs varied among patients after the application of the same dose of MSCs, and was associated with motor function improvement, as assessed by clinical assessment and multimodal magnetic resonance imaging (MRI) as shown in Fig. 1 (10). These data raised the possibility of the use of MSC-EVs, instead of MSCs per se, given that the number of EVs determines the effects of MSC-based therapy. By contrast, plasma levels of trophic factors remained unchanged after the intravenous injection of autologous MSCs although the level of trophic factors in brain-derived EVs were increased. These findings suggest that the paracrine effects of MSCs are modulated by trophic factors in the brain indirectly via MSC-EVs.
Fig. 1.
Association between elevated levels of circular extracellular vesicles (EVs) and stroke outcome after mesenchymal stem cell (MSC) injection. Modified from Bang et al. (10) The levels of circulating EVs increased im-mediately after intravenous injection of autologous MSCs (A and B), but not after placebo treatment (C, right lanes). Although patients of the MSC group re-ceived the same number of MSCs (1 × 106 cells/kg), the levels of circulating EVs were varied among patients; (A) marked increase of EV levels in patients who showed clinically significant improvement, and (B) lesser degree of increase in those who showed no clinically significant improvement. The circulating EVs levels were correlated with improvement in MRI indices of neuroplasticity as well as in motor function.
EVs are circular membrane fragments measuring 30 nm to 1 μm in diameter that are shed from the cell surface. EVs represent a heterogeneous group of vesicles released from multiple cell types in the brain (neuron, astrocyte, oligodendrocytes, microglia, and endothelial cells/pericyte). EVs mediate cell-cell interaction and the complex and versatile EV signaling was shown to regulate neurogenesis, angiogenesis, and inflammation (11). EVs are important in sustaining the cellular function in the CNS, but they also participate in the pathophysiology of underlying neurodegenerative diseases. EVs are implicated in disease spreading by misfolded and pathological proteins engaged in transferring pathogenic molecules to neighboring cells, such as Aβ42 in AD, Huntingtin protein in HD, α-synclein, leucine-rich receptor kinase 2, vacuolar-sorting protein 35 in PD, and prion proteins (PrPc and PrPsc) in Prion disease (12). For example, EVs are involved in complex mechanisms of secretion, diffusion and degradation of Aβ or tau proteins. A study analyzing the physical properties of individual EVs using electrostatic force microscopy showed that EVs carried higher levels of Aβ42 when treated with to neuroblastoma cells with higher concentrations of Aβ42 oligomers, implying that it acts as a transport vesicle (13).
Stem cell-derived EVs are considered as naturally therapeutic agents and innate drug delivery systems for therapy of brain diseases. Unlike a sole protein or small molecule, EVs contain molecules with heterogenous function. EVs contain cellular proteins, DNAs, and RNAs. Among them, most studies have focused on the regulatory roles of non-coding RNAs components, such as miRNAs, in the CNS. EVs can also capture and transfer whole mitochondria or mitochondrial fragments, and mitochondrial transfer of stem cells to injured cells may be bene-ficial in ischemic diseases and mitochondria-related diseases, such as age-related neurodegenerative diseases (14-16). EVs exhibit multiple benefits related to biocompatibility, immunogenicity, stability, pharmacokinetics, biodistribution, and cellular uptake mechanism (17). EVs can transfer intravesicular cargo and vesicular membrane-bound receptors to recipient cells. EVs can cross the BBB and actively target specific cell types (18). Unlike to cell-based therapy, EVs can avoid the first pass effect and cell-mediated adverse effects, such as as tumor formation, coagulopathy, and vascular occlusion, In addition, because lipids are essential components of EV membrane, nucleic acid components enriched in EVs are protected against RNase in the blood. A biodistribution study showed that systemically injected MSC-derived EVs can target the site of injury in pro-portion to the degree of tissue damage suggesting the possible role of signals from damaged tissue in homing EVs but not liposomes (19, 20). Further, EVs secreted by stem cells carry more complex cargo than those secreted by other cells (21), and EVs bearing MSC-specific membrane proteins on their surfaces potentially show disease-targeting ability as infused MSCs (22, 23).
Various cells have been used as a source of EVs. HEK293 cells are most commonly used as a source of EVs due to their high EV production capacity and easy transfection. However, HEK293 cells carry minimal intrinsic biological cargo, and the results of HEK293 cells cannot be translated to MSCs that are hard to transfect (24). MSCs represent a better source of EVs because the paracrine activity is responsible for at least 80% of their positive effects (25). As a result, most groups demonstrated therapeutic effects of MSC-EVs in preclinical models. However, EVs from different MSC sources exhibit different properties and carry cargo with distinct effects in the same diseases. The profile of miRNA within EVs varied greatly among the three common sources of MSCs, i.e., bone marrow, adipose, and umbilical cord (only 11 miRNAs were common), and the number of miRNAs was the highest in umbilical cord MSC-derived EVs (26). Compared with EVs derived from other MSC sources, EVs from umbilical cord showed superior therapeutic immunomodulation and protective effect (27). Further, fetal stem cells, such as umbilical cord/amniotic fluid stem exhibit a cellular phenotype intermediate between embryonic stem cells (ESCs)/induced pluripotent stem cells (iPSC) and adult MSCs (28).
It remains unknown whether MSCs are the best EV source for treatment of neurological disease. MSCs may not be an ideal cell source for EV manufacture on a clinical scale due to their limited lifespan, heterogeneity and batch-to-batch or donor-to-donor variations (29). ESCs or iPSCs, or ESC/iPSC-derived MSCs show better profiles in terms of cell numbers, sene-scence, and donor variation, while ethical concerns of ESCs are the main disadvantage in addition to the risk of immune response and teratoma involving iPSCs. Upadhya et al. showed that EVs isolated from iPSC-derived NSCs were enriched with miRNAs and proteins involved in neuroprotective, anti-apoptotic, anti-inflammatory, BBB repairing, neurogenic, and Aβ reducing activities, and are used in treating neurodegenerative disorders (30). Webb and colleagues found differences between EVs derived from different cell sources. Compared with MSC-EVs, NSC-derived EVs were superior in terms of modulation of post-stroke systemic immune response, neuroprotection, and functional recovery (31).
APPLICATIONS OF EXTRACELLULAR VESICLES IN BRAIN DISEASES
Cerebrovascular disease
Many preclinical studies have recently shown that stem cell-derived EVs can be used in stroke therapy (32). In 2013, Xin et al. reported that intravenous injection of MSC-EVs in a rat model of stroke improved the neurological outcomes and increased angiogenesis and neurogenesis (33). Other investigators have also demonstrated the beneficial effects of stem cell-derived EVs in various animal models of stroke. Several advances in EV-based strategy have been reported: (a) the use of EVs derived from stem cells other than MSCs, such as ESCs, neural stem cells (NSCs), and iPSC-derived MSCs/NSCs (31, 34, 35), (b) application of EVs intranasally (34), (c) production of EVs via 3D dynamic culture to increase the synthesis and regulate the payload of EVs (36) and stimulation with ischemic brain extracts (37, 38), and (d) evaluation of therapeutic effects of EVs on stroke in large animal models such as pigs and monkeys (35, 39). Stem cell-derived EVs contain many molecules that may have therapeutic effects in stroke, and miRNA-mediated effects was most widely studied. EV cargo miRNAs and other components related to the action of stem cell-derived EV in animal models of stroke were summarized elsewhere (32).
Traumatic brain injury
TBI represents acute damage of brain tissue caused by trauma. Together with stroke, TBI is one of the most common causes of disability and death in adults. Gao et al. showed that EVs derived from human umbilical cord blood-derived endothelial colony-forming cells have beneficial effects on BBB integrity in mice with TBI (40). Astrocyte-derived EVs carrying gap junction alpha 1 (GJA1) transmitted to neurons reduced apoptosis, increased mitochondrial function, and alleviated neuronal damage (41). Zhang et al. showed that MSC-derived EVs significantly improved functional recovery in rats after TBI, by promoting angiogenesis and neurogenesis and reducing neuroinflammation (42). In a porcine model of TBI, administration of EVs excreted from human MSCs reduced brain edema and lesion size, and improved BBB integrity (43). The therapeutic effects of EVs in TBI are attributed to their miRNA content. Yin et al. showed that miR-21-5p contained within EVs secreted from neurons promotes microglial M2 polarization to alleviate neuroinflammation after TBI (44). In contrast, Long and colleagues showed that miR-873a-5p carried by astrocyte-derived EVs suppresses neuroinflammation by inhibiting the NF-κB signaling pathway in neurons after TBI (45).
Neurodegenerative diseases
Similar to acute brain insults, such as stroke and TBI, EVs play an important role in neurodegenerative diseases, as both disease biomarkers and therapeutic targets. Dysregulation of circul-ating levels of specific EV-miRNAs has been reported in patients with neurodegenerative diseases. Recent evidences implicate EVs in the etiology and spread of neurogenerative disease. In preclinical models of AD, EVs reduced oxidative stress and neuroinflammation, inhibited the progression of neurodegeneration, and induced clearance of amyloid and neurofibrillary tangles (46, 47). Intranasal administration of MSC-derived EVs and iPSC-derived NSCs inhibited microglial activation, increased synaptogenesis, and rescued memory loss in animal models of AD (30, 48, 49). The beneficial effects of EVs obtained from MSCs preconditioned by cytokines or hypoxia and 3D culture methods were also reported recently in animal models of AD (48, 50, 51). Narbute and colleagues showed that intranasal administration of stem cell-derived EVs from teeth improved motor symptoms and normalized tyrosine hydroxylase expression in a rat model of PD (52). Several EV-miRNAs have been reported to show potential effects in AD models, such as miR-124a, miR-146a, miR-21, and miR-29b (53). Katsuda et al. showed that adipose tissue-derived MSCs secrete EVs carrying enzymatically active neprilysin, the most important Aβ-degrading enzyme in the brain (54).
Relatively few studies carried out to data have revealed the effects of stem cell-derived EV therapy in PD. Engineered EVs have been used to regulate specific proteins related to PD pathogenesis, such as antioxidant catalase, α-synuclein, and dopamine. The α-synuclein protein accumulates in brains of individuals with PD. Investigators have designed shRNA minicircles delivered by RVG EVs to treat dopaminergic neurons and reduce α-synuclein aggregation in PD (55, 56). Chen et al. showed that EVs from umbilical cord MSCs inhibited apoptosis by inducing autophagy in an in vitro model of PD (57). Upregulation of autophagy may clear accumulated α-synuclein, and miRNAs play major regulators of autophagy pathway (58). In HD, EVs transport mutant huntingtin between cells and trigger HD-related behavior and pathology (59). Didiot et al. reported the efficacy of small-interfering RNAs-loaded EVs delivered to the brain of a HD model in silencing HD mRNA suggesting the role of EVs as a gene-modifying strategy (60).
LIMITATIONS OF CURRENT MSC-EV THERAPEUTICS
Although the use of stem cell-derived EV therapy has several benefits in preclinical studies, there exist some limitations of the use of EVs obtained from naïve stem cells for patients with brain diseases.
Donor heterogeneity
One of the main obstacles hindering the clinical application of MSCs and EV therapeutics is the large variability in cell quality, due to the usage of different donors and their tissues, known as donor heterogeneity. Interestingly, Wang et al. showed that individual MSC-EV preparations from healthy human donors may differ in their therapeutic potency, suggesting donor-to-donor variation (61). The therapeutic potential of independent MSC-EV preparation may differ due to donor age, comorbidity (obesity and disease condition), artificial niche of MSCs (preconditioning or external stimuli), and culture methods used (62, 63). Along with the development of a production method that minimizes donor-to-donor and batch-to-batch variations, a robust quality control for each EV production lot is required.
Inherited undesirable features of MSC-EVs
Nalamolu et al. tested the efficacy of EVs secreted by MSCs under standard culture conditions against post-stroke brain damage and neurological outcomes in a rat model of stroke. The treatment attenuated ischemic brain damage without improving the post-stroke neurological outcome, suggesting the need for modification of MSC culture conditions (64).
Even though native EVs have potential in the treatment of brain disease, the successful clinical application is limited by the short half-life, limited targeting, rapid clearance after application, and insufficient payload (65). Although native EVs cross the BBB under stroke-like, inflamed conditions, whether they can cross the intact BBB has yet to be firmly established (66, 67). In both preclinical and human studies, blood levels of EVs decreased rapidly after systemic application of EVs, and EVs accumulated in the lung, liver and spleen until day 10 after administration (68, 69). In addition, the circulation time of EVs is shortened by macrophage/microglial clearance.
ENHANCING THE EFFICACY OF THERAPEUTIC EXTRACELLULAR VESICLES
Two strategies to enhance the efficacy of EV therapeutics.
Production or selection of optimal EVs
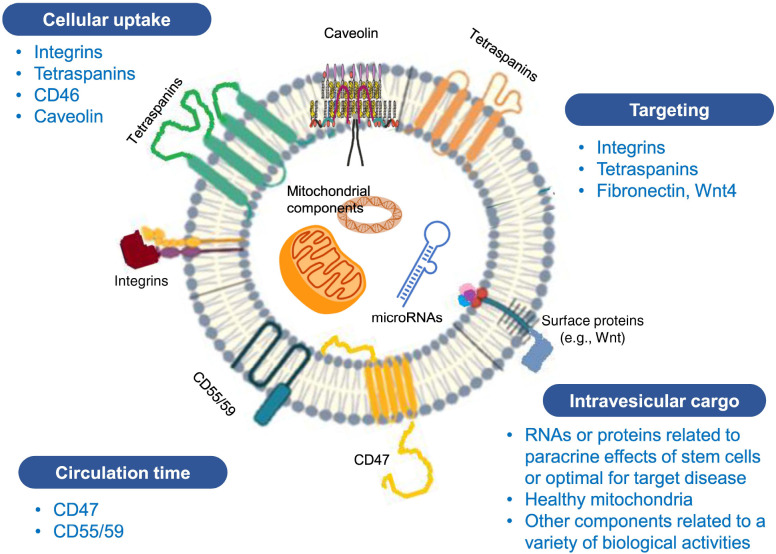
Brain pharmacokinetics of EVs may differ among EVs of differ-ent origin (68) and may depend on the characteristics of EV membrane proteins or receptors. For example, CD46, integrins, and intercellular adhesion molecule-1 on the surface of EVs were associated with the rate and mechanisms of BBB crossing of EVs (70, 71). In addition to surface molecules, intravesicular miRNAs and VEGF-A may also influence the BBB integrity (72, 73). Caveolin-mediated endocytosis and integrins and phosphatidylserine ligand-receptor interaction are involved in EV uptake by recipient cells in the brain (65). Membranous lipid-draft protein caveolin-1 and phosphatidylserine were highly expressed in microvesicles than in exosomes (74). CD47, a transmembrane protein that enables cancer cells to evade clearance by macrophages (‘don’t eat me signal’), prolongs the circulation time of EV after systemic administration (75). EV surface features, such as tetraspanin (CD63) and integrin profiles, may influence the targeting capacity (76). Selection of EVs with optimal characteristics in terms of surface and cargo molecules represents a safer alternative than bioengineered EVs. Ideal EVs for brain therapeutics exhibit the aforementioned characteristics of surface molecules and intravesicular cargo, as shown in Fig. 2.
Fig. 2.
Surface molecules and cargo related to efficacy and biodistribution of stem cell-derived extracellular vesicles (EVs).
Modification of EV culture conditions or exposure to external stimuli may enhance the therapeutic potential of MSC-EVs (77, 78). Preconditioning (hypoxia, ischemia or inflammation), supplementation of culture medium with bioactive factors, and modification of cell-cell interaction (spheroid culture) or cell-substrate interaction (collagen microgel) were used to enhance their therapeutic properties and possibly minimize donor variation. Interestingly, our and other studies showed that the cellular yield of EVs was increased compared with standard 2D culture and EV cargo undetected by standard 2D conditioning of cells were enriched in EVs obtained from 3D culture (36, 79).
Bioengineering of source cells or EVs
Engineered EVs may be used to enhance the efficacy of cell-derived EVs for treating neurological diseases. In this review, artificial engineering techniques for surface modification and cargo loading to enhance therapeutic efficacy of EVs are not discussed in detail because MSCs are hard to transfect and this topic was covered by other reviews (24, 80, 81). EV surface engineering can be achieved indirectly by genetic modification of the EV-secreting cells or via direct modification of EV surface to improve stability, targeting ability and EV tracking (82). However, such EV surface engineering techniques may be associated with toxicity and alteration of the characteristics of stem cell-derived EVs.
Nucleic acids, proteins, and small molecules can be loaded into EVs. To improve the loading efficacy, EV can be physically or chemically manipulated. However, most studies focused on specific EV-miRNAs or EV proteins to evaluate the mechanisms of EVs, and used engineered EVs from non-stem cells (e.g., HEK293) that contain selected EV cargos. It is likely that multiple EV-associated cargos rather than a single candidate molecule elicit therapeutic effects of MSC-EVs, in a synergistic manner. Further, active bioengineering techniques recently introduced may influence the characteristics of EVs and induce toxicity or be clinically undesirable. All the techniques can increase loading efficiency; however, they are often associated with negative effects such as loss of membrane integrity, aggregate formation and cargo impurity (83). Although bioengineering of EV-producing cells is attractive, most investigators prefer post-EV modification for rapid results and yield as well as viability for clinical feasi-bility (65).
EVs harbor bioactive molecules. Among them, miRNAs regulate gene expression and protein synthesis, while proteins have biochemical effects. The majority of RNAs found in EVs are less than 200 nucleotides in length. Unlike the abundance of ribosomal RNAs in the parent cells, EVs are mainly enriched in small RNAs, such as miRNAs, long non-coding RNAs, and circular RNAs (84). The miRNA is of prime importance in mediating therapeutic effects (32, 85). Differential miRNA expression in angiogenesis, neuroprotection, and immunomodulation may be associated with stroke and neurodegenerative diseases (32). For example, loading MSCs with miR-124, a neuroprotectant and inflammatory modulator induced cortical neurogenesis via EVs (86). Application of EVs from MSCs overexpressing miRNA-133b showed neurite remodeling and neuroprotection (87). In addition, the miRNA-17-92 cluster is associated with neuroplasticity, and treatment of EVs from MSCs loaded with these miRNAs increased neural plasticity and functional recovery in a rat model of stroke compared with EVs derived from naïve MSCs (88).
Brain-derived neurotrophic factor (BDNF) is a clinically relevant candidate for drug delivery, potentially for neuroprotective effects both at and across the BBB (89). BDNF plays important roles in a variety of brain diseases. BDNF rescues neurons from apoptotic cell death, promotes neuronal development and regeneration of synaptic connections, and improves the overall neuroplasticity of recovery from brain injury and cognitive processes (90). Further, BDNF is expressed in many different brain regions and is decreased during the aging process. The levels of BDNF and netrin-1 (a laminin-related hormones) are strongly reduced in Parkinson’s disease brains and gut tissues (91). D’Souza and colleagues susscessfuly transferred the plasmid DNA expressing BDNF and mitochondria/mitochondrial DNA to brain endothelial cells (89). Yang et al. evaluated the potential therapeutic effects of EV-mediated targeted delivery of NGF in ischemic cortex (92). In this study, HEK293 cells were transfected with RVG-Lamp2b and NGF vectors. Systemic administration of EVs resulted in a burst of encapsulated NGF protein released in the brain. Similarly, Zha et al. encapsulated the VEGF gene into chondrogenic ATDC5-derived EVs, which induced vascularized bone regeneration (93).
Therapeutic agents such as curcumin and catalase can be loaded in EVs to enhance the therapeutic potential of naïve EVs. For example, catalase, a potent antioxidant, was loaded into EVs ex vivo using different methods, and treatment via EVs provided neuroprotective effects in both in vitro and in vivo models of PD (94). Conversely, curcumin which exhibits anti-inflammatory and anti-oxidant properties, was loaded by passive incubation into MSCs and HEK293 cells resulting in enhanced protective effects in models of osteoarthritis and myocardial infarction, respectively (95, 96).
CLINICAL SCALE PRODUCTION OF EXTRACELLULAR VESICLES
The clinical use of EVs requires mass production of EVs. Strategies to increase the yield of EV production include large-scale methods for EV generation, such as artificial EV generation (e.g., extrusion via porous membrane) and large-scale natural EV generation (e.g., bioreactor use) as well as aforementioned methods for modification of culture conditions or external stimuli (97, 98). Various methods of EV production, including different bioreactors and isolation methods, are being used for clinical trials of EV therapeutics (98). In addition to culture of EV source cells, isolation methods also affect the EV cargos. Therefore, selection and validation of isolation methods of choice are needed to avoid confounding results regarding EV-specific content and function.
Haraszti et al. showed that microcarrier-based 3D culture and tangential flow filtration (TFF) facilitate scalable production of biologically active EVs from umbilical cord-derived MSCs. The yield of EVs using this combination system was robust compared with 2D-cultures (99). We have recently introduced a novel method for clinical scale MSC-EV production using a micro-patterned well-based 3D-spheroid system. Using this culture method, we were able to upregulate miRNAs related to neurogenesis/axonal outgrowth and reduce the donor variation.
CONCLUSIONS
Stem cell-derived EV therapy represents a promising approach for patients with acute and degenerative brain disease, as MSC therapies have already been tested in clinical trials. EV-mediated therapy is superior to cell therapy in terms of scalable production, biodistribution, and safety profiles. However, continuous efforts are needed to control the heterogeneity of cargo, opti-mization of EV surface molecules, and increasing EV production yield.
Currently, MSC-EV therapy is still in the process of development. The results of clinical trials of the application of MSC-EVs have been reported in graft versus host disease, chronic kidney disease, and COVID-19, which showed no adverse effects related to the administration of MSC-EVs (100-102). In addition, clinical safety and possible beneficial effects of MSC-EVs or secretome were reported in patients with alveolar bone regeneration, alopecia, Meniere’s disease undergoing intracochlear application, and refractory macular degeneration (103-106). As of June 2021, only two clinical trials are evaluating the role of EV therapeutics in brain diseases including a small clinical trial of safety involving intravenous application of MSC-EVs engineered to express miR-124 in stroke patients (clinicalTrial.gov identifier NCT3384433) and a phase I/II clinical trial of safety and efficacy of EVs derived from allogeneic adipose MSCs administered for nasal drip in patients with AD (NCT0438982).
EV therapies have yet to be approved by the regulatory authorities. Therefore, further studies evaluating the efficacy of MSC-EVs in RCTs are required. In addition, the conventional 2D culture method has been used in the aforementioned clinical studies. EV characterization and isolation methods show substantial heterogeneity. Therefore, further clinical studies are needed to address the limitations of clinical progression of EV therapeutics, such as scalability and GMP of source cells and EV preparation, in addition to extensive quality control. Finally, the biodistribution of EVs and the route and dose of application should be defined as they differ depending on the characterization of target diseases, in terms of acute insults vs. chronic neurodegeneration and intact vs. inflamed BBB.
ACKNOWLEDGEMENTS
This research was in part supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI14C 3484).
Footnotes
CONFLICTS OF INTEREST
The authors have no conflicting interests.
REFERENCES
- 1.Bang OY. Neuroprotective strategies for acute ischemic stroke: recent progress and future perspectives. Precis Future Med. 2017;1:115–121. doi: 10.23838/pfm.2017.00149. [DOI] [Google Scholar]
- 2.Kordower JH, Bjorklund A. Trophic factor gene therapy for Parkinson's disease. Mov Disord. 2013;28:96–109. doi: 10.1002/mds.25344. [DOI] [PubMed] [Google Scholar]
- 3.Lo EH. A new penumbra: transitioning from injury into repair after stroke. Nat Med. 2008;14:497–500. doi: 10.1038/nm1735. [DOI] [PubMed] [Google Scholar]
- 4.Lyden PD. Cerebroprotection for acute ischemic stroke: looking ahead. Stroke. 2021;52:3033–3044. doi: 10.1161/STROKEAHA.121.032241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rufino-Ramos D, Albuquerque PR, Carmona V, Perfeito R, Nobre RJ, Pereira de Almeida L. Extracellular vesicles: novel promising delivery systems for therapy of brain diseases. J Control Release. 2017;262:247–258. doi: 10.1016/j.jconrel.2017.07.001. [DOI] [PubMed] [Google Scholar]
- 6.Lee JS, Hong JM, Moon GJ, et al. A long-term follow-up study of intravenous autologous mesenchymal stem cell transplantation in patients with ischemic stroke. Stem Cells. 2010;28:1099–1106. doi: 10.1002/stem.430. [DOI] [PubMed] [Google Scholar]
- 7.Prasad K, Sharma A, Garg A, et al. Intravenous autologous bone marrow mononuclear stem cell therapy for ischemic stroke: a multicentric, randomized trial. Stroke. 2014;45:3618–3624. doi: 10.1161/STROKEAHA.114.007028. [DOI] [PubMed] [Google Scholar]
- 8.Hess DC, Wechsler LR, Clark WM, et al. Safety and efficacy of multipotent adult progenitor cells in acute ischaemic stroke (MASTERS): a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Neurol. 2017;16:360–368. doi: 10.1016/S1474-4422(17)30046-7. [DOI] [PubMed] [Google Scholar]
- 9.Chung JW, Chang WH, Bang OY, et al. Efficacy and safety of intravenous mesenchymal stem cells for ischemic stroke. Neurology. 2021;96:e1012–e1023. doi: 10.1212/WNL.0000000000011440. [DOI] [PubMed] [Google Scholar]
- 10.Bang OY, Kim EH, Cho YH. Circulating extracellular vesicles in stroke patients treated with mesenchymal stem cells: a biomarker analysis of a randomized trial. Stroke [Epub Ahead of Print] 2022 doi: 10.1161/STROKEAHA.121.036545. https://doi.org/10.1161/STROKEAHA.121.036545. [DOI] [PubMed] [Google Scholar]
- 11.Zagrean AM, Hermann DM, Opris I, Zagrean L, Popa-Wagner A. Multicellular crosstalk between exosomes and the neurovascular unit after cerebral ischemia. Therapeutic implications. Front Neurosci. 2018;12:811. doi: 10.3389/fnins.2018.00811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brites D. Regulatory function of microRNAs in microglia. Glia. 2020;68:1631–1642. doi: 10.1002/glia.23846. [DOI] [PubMed] [Google Scholar]
- 13.Choi Y, Kim SM, Heo Y, Lee G, Kang JY, Yoon DS. Nanoelectrical characterization of individual exosomes secreted by Abeta42-ingested cells using electrostatic force microscopy. Nanotechnology. 2021;32:025705. doi: 10.1088/1361-6528/abba58. [DOI] [PubMed] [Google Scholar]
- 14.Crewe C, Funcke JB, Li S, et al. Extracellular vesicle-based interorgan transport of mitochondria from energetically stressed adipocytes. Cell Metab. 2021;33:1853–1868. doi: 10.1016/j.cmet.2021.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.O'Brien CG, Ozen MO, Ikeda G, et al. Mitochondria-rich extracellular vesicles rescue patient-specific cardiomyocytes from doxorubicin injury: insights into the SENECA trial. JACC CardioOncol. 2021;3:428–440. doi: 10.1016/j.jaccao.2021.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.D'Souza A, Burch A, Dave KM, et al. Microvesicles transfer mitochondria and increase mitochondrial function in brain endothelial cells. J Control Release. 2021;338:505–526. doi: 10.1016/j.jconrel.2021.08.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nam GH, Choi Y, Kim GB, Kim S, Kim SA, Kim IS. Emerging prospects of exosomes for cancer treatment: from conventional therapy to immunotherapy. Adv Mater. 2020;32:e2002440. doi: 10.1002/adma.202002440. [DOI] [PubMed] [Google Scholar]
- 18.Otero-Ortega L, Laso-Garcia F, Frutos MCG, et al. Low dose of extracellular vesicles identified that promote recovery after ischemic stroke. Stem Cell Res Ther. 2020;11:70. doi: 10.1186/s13287-020-01601-1.3fe897b11bcf4a0fac4bf1ae968c9f3f [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wen S, Dooner M, Papa E, et al. Biodistribution of mesenchymal stem cell-derived extracellular vesicles in a radiation injury bone marrow murine model. Int J Mol Sci. 2019;20:5468. doi: 10.3390/ijms20215468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mirzaaghasi A, Han Y, Ahn SH, Choi C, Park JH. Biodistribution and pharmacokinectics of liposomes and exosomes in a mouse model of sepsis. Pharmaceutics. 2021;13:427. doi: 10.3390/pharmaceutics13030427.cfac682a986d4bfaaa66a2fa978302fe [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lai RC, Tan SS, Teh BJ, et al. Proteolytic potential of the MSc exosome proteome: implications for an exosome-mediated delivery of therapeutic proteasome. Int J Proteomics. 2012;2012:971907. doi: 10.1155/2012/971907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Biancone L, Bruno S, Deregibus MC, Tetta C, Camussi G. Therapeutic potential of mesenchymal stem cell-derived microvesicles. Nephrol Dial Transplant. 2012;27:3037–3042. doi: 10.1093/ndt/gfs168. [DOI] [PubMed] [Google Scholar]
- 23.Karp JM, Leng Teo GS. Mesenchymal stem cell homing: the devil is in the details. Cell Stem Cell. 2009;4:206–216. doi: 10.1016/j.stem.2009.02.001. [DOI] [PubMed] [Google Scholar]
- 24.Le Saux S, Aubert-Pouessel A, Mohamed KE, et al. Interest of extracellular vesicles in regards to lipid nanoparticle based systems for intracellular protein delivery. Adv Drug Deliv Rev. 2021;176:113837. doi: 10.1016/j.addr.2021.113837. [DOI] [PubMed] [Google Scholar]
- 25.Muhammad SA, Nordin N, Mehat MZ, Fakurazi S. Comparative efficacy of stem cells and secretome in articular cartilage regeneration: a systematic review and meta-analysis. Cell Tissue Res. 2019;375:329–344. doi: 10.1007/s00441-018-2884-0. [DOI] [PubMed] [Google Scholar]
- 26.Zheng X, Hermann DM, Bahr M, Doeppner TR. The role of small extracellular vesicles in cerebral and myocardial ischemia-Molecular signals, treatment targets, and future clinical translation. Stem Cells. 2021;39:403–413. doi: 10.1002/stem.3329. [DOI] [PubMed] [Google Scholar]
- 27.Cai J, Wu J, Wang J, et al. Extracellular vesicles derived from different sources of mesenchymal stem cells: therapeutic effects and translational potential. Cell Biosci. 2020;10:69. doi: 10.1186/s13578-020-00427-x.9e02821a62804b5f991722cc01a1967f [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Loukogeorgakis SP, De Coppi P. Concise review: amniotic fluid stem cells: the known, the unknown, and potential regenerative medicine applications. Stem Cells. 2017;35:1663–1673. doi: 10.1002/stem.2553. [DOI] [PubMed] [Google Scholar]
- 29.Johnson J, Shojaee M, Mitchell Crow J, Khanabdali R. From mesenchymal stromal cells to engineered extracellular vesicles: a new therapeutic paradigm. Front Cell Dev Biol. 2021;9:705676. doi: 10.3389/fcell.2021.705676.3397dc7845c744088a445a1eeff9085c [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Upadhya R, Madhu LN, Attaluri S, et al. Extracellular vesicles from human iPSC-derived neural stem cells: miRNA and protein signatures, and anti-inflammatory and neurogenic properties. J Extracell Vesicles. 2020;9:1809064. doi: 10.1080/20013078.2020.1809064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Webb RL, Kaiser EE, Scoville SL, et al. Human neural stem cell extracellular vesicles improve tissue and functional recovery in the murine thromboembolic stroke model. Transl Stroke Res. 2018;9:530–539. doi: 10.1007/s12975-017-0599-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bang OY, Kim EH. Mesenchymal stem cell-derived extracellular vesicle therapy for stroke: challenges and progress. Front Neurol. 2019;10:211. doi: 10.3389/fneur.2019.00211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Xin H, Li Y, Cui Y, Yang JJ, Zhang ZG, Chopp M. Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J Cereb Blood Flow Metab. 2013;33:1711–1715. doi: 10.1038/jcbfm.2013.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kalani A, Chaturvedi P, Kamat PK, et al. Curcumin-loaded embryonic stem cell exosomes restored neurovascular unit following ischemia-reperfusion injury. Int J Biochem Cell Biol. 2016;79:360–369. doi: 10.1016/j.biocel.2016.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Webb RL, Kaiser EE, Jurgielewicz BJ, et al. Human neural stem cell extracellular vesicles improve recovery in a porcine model of ischemic stroke. Stroke. 2018;49:1248–1256. doi: 10.1161/STROKEAHA.117.020353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cha JM, Shin EK, Sung JH, et al. Efficient scalable production of therapeutic microvesicles derived from human mesenchymal stem cells. Sci Rep. 2018;8:1171. doi: 10.1038/s41598-018-19211-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Moon GJ, Sung JH, Kim DH, et al. Application of mesenchymal stem cell-derived extracellular vesicles for stroke: biodistribution and microRNA Study. Transl Stroke Res. 2019;10:509–521. doi: 10.1007/s12975-018-0668-1. [DOI] [PubMed] [Google Scholar]
- 38.Lee JY, Kim E, Choi SM, et al. Microvesicles from brain-extract-treated mesenchymal stem cells improve neurological functions in a rat model of ischemic stroke. Sci Rep. 2016;6:33038. doi: 10.1038/srep33038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Medalla M, Chang W, Calderazzo SM, et al. Treatment with mesenchymal-derived extracellular vesicles reduces injury-related pathology in pyramidal neurons of monkey perilesional ventral premotor cortex. J Neurosci. 2020;40:3385–3407. doi: 10.1523/JNEUROSCI.2226-19.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gao W, Li F, Liu L, et al. Endothelial colony-forming cell-derived exosomes restore blood-brain barrier continuity in mice subjected to traumatic brain injury. Exp Neurol. 2018;307:99–108. doi: 10.1016/j.expneurol.2018.06.001. [DOI] [PubMed] [Google Scholar]
- 41.Chen W, Zheng P, Hong T, et al. Astrocytes-derived exosomes induce neuronal recovery after traumatic brain injury via delivering gap junction alpha 1-20 k. J Tissue Eng Regen Med. 2020;14:412–423. doi: 10.1002/term.3002. [DOI] [PubMed] [Google Scholar]
- 42.Zhang Y, Chopp M, Zhang ZG, et al. Systemic administration of cell-free exosomes generated by human bone marrow derived mesenchymal stem cells cultured under 2D and 3D conditions improves functional recovery in rats after traumatic brain injury. Neurochem Int. 2017;111:69–81. doi: 10.1016/j.neuint.2016.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Williams AM, Bhatti UF, Brown JF, et al. Early single-dose treatment with exosomes provides neuroprotection and improves blood-brain barrier integrity in swine model of traumatic brain injury and hemorrhagic shock. J Trauma Acute Care Surg. 2020;88:207–218. doi: 10.1097/TA.0000000000002563. [DOI] [PubMed] [Google Scholar]
- 44.Yin Z, Han Z, Hu T, et al. Neuron-derived exosomes with high miR-21-5p expression promoted polarization of M1 microglia in culture. Brain Behav Immun. 2020;83:270–282. doi: 10.1016/j.bbi.2019.11.004. [DOI] [PubMed] [Google Scholar]
- 45.Long X, Yao X, Jiang Q, et al. Astrocyte-derived exosomes enriched with miR-873a-5p inhibit neuroinflammation via microglia phenotype modulation after traumatic brain injury. J Neuroinflammation. 2020;17:89. doi: 10.1186/s12974-020-01761-0.a3069d49259d46648e8f7b05c9024674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Soares Martins T, Trindade D, Vaz M, et al. Diagnostic and therapeutic potential of exosomes in Alzheimer's disease. J Neurochem. 2021;156:162–181. doi: 10.1111/jnc.15112. [DOI] [PubMed] [Google Scholar]
- 47.Elia CA, Tamborini M, Rasile M, et al. Intra-cerebral injection of extracellular vesicles from mesenchymal stem cells exerts reduced abeta plaque burden in early stages of a preclinical model of Alzheimer's disease. Cells. 2019;8:1059. doi: 10.3390/cells8091059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Losurdo M, Pedrazzoli M, D'Agostino C, et al. Intranasal delivery of mesenchymal stem cell-derived extracellular vesicles exerts immunomodulatory and neuroprotective effects in a 3xTg model of Alzheimer's disease. Stem Cells Transl Med. 2020;9:1068–1084. doi: 10.1002/sctm.19-0327.2ac1911ed4e44248968ed57781e81261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ma X, Huang M, Zheng M, et al. ADSCs-derived extracellular vesicles alleviate neuronal damage, promote neurogenesis and rescue memory loss in mice with Alzheimer's disease. J Control Release. 2020;327:688–702. doi: 10.1016/j.jconrel.2020.09.019. [DOI] [PubMed] [Google Scholar]
- 50.Cone AS, Yuan X, Sun L, et al. Mesenchymal stem cell-derived extracellular vesicles ameliorate Alzheimer's disease-like phenotypes in a preclinical mouse model. Theranostics. 2021;11:8129–8142. doi: 10.7150/thno.62069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cui GH, Wu J, Mou FF, et al. Exosomes derived from hypoxia-preconditioned mesenchymal stromal cells ameliorate cognitive decline by rescuing synaptic dysfunction and regulating inflammatory responses in APP/PS1 mice. FASEB J. 2018;32:654–668. doi: 10.1096/fj.201700600R. [DOI] [PubMed] [Google Scholar]
- 52.Narbute K, Pilipenko V, Pupure J, et al. Intranasal administration of extracellular vesicles derived from human teeth stem cells improves motor symptoms and normalizes tyrosine hydroxylase expression in the substantia nigra and striatum of the 6-Hydroxydopa-mine-treated rats. Stem Cells Transl Med. 2019;8:490–499. doi: 10.1002/sctm.18-0162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cui GH, Zhu J, Wang YC, Wu J, Liu JR, Guo HD. Effects of exosomal miRNAs in the diagnosis and treatment of Alzheimer's disease. Mech Ageing Dev. 2021;200:111593. doi: 10.1016/j.mad.2021.111593. [DOI] [PubMed] [Google Scholar]
- 54.Katsuda T, Tsuchiya R, Kosaka N, et al. Human adipose tissue-derived mesenchymal stem cells secrete functional neprilysin-bound exosomes. Sci Rep. 2013;3:1197. doi: 10.1038/srep01197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Li X, Zhang J, Zhang X, Dong M. Puerarin suppresses MPP(+)/MPTP-induced oxidative stress through an Nrf2-dependent mechanism. Food Chem Toxicol. 2020;144:111644. doi: 10.1016/j.fct.2020.111644. [DOI] [PubMed] [Google Scholar]
- 56.Izco M, Blesa J, Schleef M, et al. Systemic exoso-mal delivery of shRNA minicircles prevents parkinsonian pathology. Mol Ther. 2019;27:2111–2122. doi: 10.1016/j.ymthe.2019.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chen HX, Liang FC, Gu P, et al. Exosomes derived from mesenchymal stem cells repair a Parkinson's disease model by inducing autophagy. Cell Death Dis. 2020;11:288. doi: 10.1038/s41419-020-2473-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Akkoc Y, Gozuacik D. MicroRNAs as major regulators of the autophagy pathway. Biochim Biophys Acta Mol Cell Res. 2020;1867:118662. doi: 10.1016/j.bbamcr.2020.118662. [DOI] [PubMed] [Google Scholar]
- 59.Jeon I, Cicchetti F, Cisbani G, et al. Human-to-mouse prion-like propagation of mutant huntingtin protein. Acta Neuropathol. 2016;132:577–592. doi: 10.1007/s00401-016-1582-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Didiot MC, Hall LM, Coles AH, et al. Exosome-mediated delivery of hydrophobically modified siRNA for Huntingtin mRNA silencing. Mol Ther. 2016;24:1836–1847. doi: 10.1038/mt.2016.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wang C, Borger V, Sardari M, et al. Mesenchymal stromal cell-derived small extracellular vesicles induce ischemic neuroprotection by modulating leukocytes and specifically neutrophils. Stroke. 2020;51:1825–1834. doi: 10.1161/STROKEAHA.119.028012. [DOI] [PubMed] [Google Scholar]
- 62.Costa LA, Eiro N, Fraile M, et al. Functional heterogeneity of mesenchymal stem cells from natural niches to culture conditions: implications for further clinical uses. Cell Mol Life Sci. 2021;78:447–467. doi: 10.1007/s00018-020-03600-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.de Almeida Fuzeta M, Bernardes N, Oliveira FD, et al. Scalable production of human mesenchymal stromal cell-derived extracellular vesicles under serum-/xeno-free conditions in a microcarrier-based bioreactor culture system. Front Cell Dev Biol. 2020;8:553444. doi: 10.3389/fcell.2020.553444.b500df7c63514a22843cbaa239e4e991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nalamolu KR, Venkatesh I, Mohandass A, et al. Exosomes treatment mitigates ischemic brain damage but does not improve post-stroke neurological outcome. Cell Physiol Biochem. 2019;52:1280–1291. doi: 10.33594/000000090.5c9033bb723d440d8d5b69c822e60a2a [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Khan H, Pan JJ, Li Y, Zhang Z, Yang GY. Native and bioengineered exosomes for ischemic stroke therapy. Front Cell Dev Biol. 2021;9:619565. doi: 10.3389/fcell.2021.619565.bc5a49d581024dae96b73af82975cf39 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chen CC, Liu L, Ma F, et al. Elucidation of exosome migration across the blood-brain barrier model in vitro. Cell Mol Bioeng. 2016;9:509–529. doi: 10.1007/s12195-016-0458-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Yuan D, Zhao Y, Banks WA, et al. Macrophage exosomes as natural nanocarriers for protein delivery to inflamed brain. Biomaterials. 2017;142:1–12. doi: 10.1016/j.biomaterials.2017.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wiklander OP, Nordin JZ, O'Loughlin A, et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J Extracell Vesicles. 2015;4:26316. doi: 10.3402/jev.v4.26316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Gholamrezanezhad A, Mirpour S, Bagheri M, et al. In vivo tracking of 111In-oxine labeled mesenchymal stem cells following infusion in patients with advanced cirrhosis. Nucl Med Biol. 2011;38:961–967. doi: 10.1016/j.nucmedbio.2011.03.008. [DOI] [PubMed] [Google Scholar]
- 70.Banks WA, Sharma P, Bullock KM, Hansen KM, Ludwig N, Whiteside TL. Transport of extracellular vesicles across the blood-brain barrier: brain pharmacokinetics and effects of inflammation. Int J Mol Sci. 2020;21:4407. doi: 10.3390/ijms21124407.9296d89a0dfe4a7daf2af5cddf484712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Saint-Pol J, Gosselet F, Duban-Deweer S, Pottiez G, Karamanos Y. Targeting and crossing the blood-brain barrier with extracellular vesicles. Cells. 2020;9:851. doi: 10.3390/cells9040851.0a4624f7e87c4c2ca8ba2f9122e46635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zhao Z, Zlokovic BV. Remote control of BBB: A tale of exosomes and microRNA. Cell Res. 2017;27:849–850. doi: 10.1038/cr.2017.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zhao C, Wang H, Xiong C, Liu Y. Hypoxic glioblastoma release exosomal VEGF-A induce the permeability of blood-brain barrier. Biochem Biophys Res Commun. 2018;502:324–331. doi: 10.1016/j.bbrc.2018.05.140. [DOI] [PubMed] [Google Scholar]
- 74.Durcin M, Fleury A, Taillebois E, et al. Characterisation of adipocyte-derived extracellular vesicle sub-types identifies distinct protein and lipid signatures for large and small extracellular vesicles. J Extracell Vesicles. 2017;6:1305677. doi: 10.1080/20013078.2017.1305677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wei Z, Chen Z, Zhao Y, et al. Mononuclear phagocyte system blockade using extracellular vesicles modified with CD47 on membrane surface for myocardial infarction reperfusion injury treatment. Biomaterials. 2021;275:121000. doi: 10.1016/j.biomaterials.2021.121000. [DOI] [PubMed] [Google Scholar]
- 76.Murphy DE, de Jong OG, Brouwer M, et al. Extracellular vesicle-based therapeutics: natural versus engineered targeting and trafficking. Exp Mol Med. 2019;51:1–12. doi: 10.1038/s12276-019-0223-5.316df075eed04434ba364fbd33b3895c [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Cunningham CJ, Redondo-Castro E, Allan SM. The therapeutic potential of the mesenchymal stem cell secretome in ischaemic stroke. J Cereb Blood Flow Metab. 2018;38:1276–1292. doi: 10.1177/0271678X18776802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Park KS, Bandeira E, Shelke GV, Lasser C, Lotvall J. Enhancement of therapeutic potential of mesenchymal stem cell-derived extracellular vesicles. Stem Cell Res Ther. 2019;10:288. doi: 10.1186/s13287-019-1398-3.0efc678e600d441d89f07b17ce05981c [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Millan C, Prause L, Vallmajo-Martin Q, Hensky N, Eberli D. Extracellular vesicles from 3D engineered microtissues harbor disease-related cargo absent in EVs from 2D cultures. Adv Healthc Mater. 2021:e2002067. doi: 10.1002/adhm.202002067. [DOI] [PubMed] [Google Scholar]
- 80.Pauwels MJ, Vandendriessche C, Vandenbroucke RE. Special delEVery: extracellular vesicles as promising delivery platform to the brain. Biomedicines. 2021;9:1734. doi: 10.3390/biomedicines9111734.e7a441730e224e57b41c1cc1ac4d03c7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wiklander OPB, Brennan MA, Lotvall J, Breakefield XO, El Andaloussi S. Advances in therapeutic applications of extracellular vesicles. Sci Transl Med. 2019;11:eaav8521. doi: 10.1126/scitranslmed.aav8521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lino MM, Simoes S, Tomatis F, et al. Engineered extracellular vesicles as brain therapeutics. J Control Release. 2021;338:472–485. doi: 10.1016/j.jconrel.2021.08.037. [DOI] [PubMed] [Google Scholar]
- 83.Pedrioli G, Piovesana E, Vacchi E, Balbi C. Extracellular vesicles as promising carriers in drug delivery: considerations from a cell biologist's perspective. Biology 10. 2021;(Basel):376. doi: 10.3390/biology10050376.f9bc366be8bb4dff8bff7af1fac4404a [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Crescitelli R, Lasser C, Szabo TG, et al. Distinct RNA profiles in subpopulations of extracellular vesicles: apoptotic bodies, microvesicles and exosomes. J Extracell Vesicles. 2013;2:1–10. doi: 10.3402/jev.v2i0.20677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Zhang ZG, Buller B, Chopp M. Exosomes - beyond stem cells for restorative therapy in stroke and neurological injury. Nat Rev Neurol. 2019;15:193–203. doi: 10.1038/s41582-018-0126-4. [DOI] [PubMed] [Google Scholar]
- 86.Yang J, Zhang X, Chen X, Wang L, Yang G. Exosome mediated delivery of miR-124 promotes neurogenesis after ischemia. Mol Ther Nucleic Acids. 2017;7:278–287. doi: 10.1016/j.omtn.2017.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Xin H, Li Y, Liu Z, et al. MiR-133b promotes neural plasticity and functional recovery after treatment of stroke with multipotent mesenchymal stromal cells in rats via transfer of exosome-enriched extracellular particles. Stem Cells. 2013;31:2737–2746. doi: 10.1002/stem.1409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Xin H, Katakowski M, Wang F, et al. MicroRNA cluster miR-17-92 cluster in exosomes enhance neuroplasticity and functional recovery after stroke in rats. Stroke. 2017;48:747–753. doi: 10.1161/STROKEAHA.116.015204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.D'Souza A, Dave KM, Stetler RA, D SM. Targeting the blood-brain barrier for the delivery of stroke therapies. Adv Drug Deliv Rev. 2021;171:332–351. doi: 10.1016/j.addr.2021.01.015. [DOI] [PubMed] [Google Scholar]
- 90.Poo MM. Neurotrophins as synaptic modulators. Nat Rev Neurosci. 2001;2:24–32. doi: 10.1038/35049004. [DOI] [PubMed] [Google Scholar]
- 91.Ahn EH, Kang SS, Ye K. Netrin-1/receptors regulate the pathogenesis in Parkinson's diseases. Precis Future Med. 2021;5:50–61. doi: 10.23838/pfm.2020.00177. [DOI] [Google Scholar]
- 92.Yang J, Wu S, Hou L, et al. Therapeutic effects of simultaneous delivery of nerve growth factor mRNA and protein via exosomes on cerebral ischemia. Mol Ther Nucleic Acids. 2020;21:512–522. doi: 10.1016/j.omtn.2020.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Zha Y, Li Y, Lin T, Chen J, Zhang S, Wang J. Progenitor cell-derived exosomes endowed with VEGF plasmids enhance osteogenic induction and vascular remodeling in large segmental bone defects. Theranostics. 2021;11:397–409. doi: 10.7150/thno.50741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Haney MJ, Klyachko NL, Zhao Y, et al. Exosomes as drug delivery vehicles for Parkinson's disease therapy. J Control Release. 2015;207:18–30. doi: 10.1016/j.jconrel.2015.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kang JY, Kim H, Mun D, Yun N, Joung B. Co-delivery of curcumin and miRNA-144-3p using heart-targeted extracellular vesicles enhances the therapeutic efficacy for myocardial infarction. J Control Release. 2021;331:62–73. doi: 10.1016/j.jconrel.2021.01.018. [DOI] [PubMed] [Google Scholar]
- 96.Li S, Stockl S, Lukas C, et al. Curcumin-primed human BMSC-derived extracellular vesicles reverse IL-1beta-induced catabolic responses of OA chondrocytes by upregulating miR-126-3p. Stem Cell Res Ther. 2021;12:252. doi: 10.1186/s13287-021-02317-6.d245f02e52e44186be57f0aa14740f2e [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Hahm J, Kim J, Park J. Strategies to enhance extracellular vesicle production. Tissue Eng Regen Med. 2021;18:513–524. doi: 10.1007/s13770-021-00364-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Grangier A, Branchu J, Volatron J, et al. Technological advances towards extracellular vesicles mass production. Adv Drug Deliv Rev. 2021;176:113843. doi: 10.1016/j.addr.2021.113843. [DOI] [PubMed] [Google Scholar]
- 99.Haraszti RA, Miller R, Stoppato M, et al. Exosomes produced from 3D cultures of MSCs by tangential flow filtration show higher yield and improved activity. Mol Ther. 2018;26:2838–2847. doi: 10.1016/j.ymthe.2018.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Kordelas L, Rebmann V, Ludwig AK, et al. MSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease. Leukemia. 2014;28:970–973. doi: 10.1038/leu.2014.41. [DOI] [PubMed] [Google Scholar]
- 101.Nassar W, El-Ansary M, Sabry D, et al. Umbilical cord mesenchymal stem cells derived extracellular vesicles can safely ameliorate the progression of chronic kidney diseases. Biomater Res. 2016;20:21. doi: 10.1186/s40824-016-0068-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Sengupta V, Sengupta S, Lazo A, Woods P, Nolan A, Bremer N. Exosomes derived from bone marrow mesenchymal stem cells as treatment for severe COVID-19. Stem Cells Dev. 2020;29:747–754. doi: 10.1089/scd.2020.0080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Katagiri W, Osugi M, Kawai T, Hibi H. First-in-human study and clinical case reports of the alveolar bone regeneration with the secretome from human mesenchymal stem cells. Head Face Med. 2016;12:5. doi: 10.1186/s13005-016-0101-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Fukuoka H, Suga H. Hair regeneration treatment using adipose-derived stem cell conditioned medium: follow-up with trichograms. Eplasty. 2015;15:e10. [PMC free article] [PubMed] [Google Scholar]
- 105.Warnecke A, Prenzler N, Harre J, et al. First-in-human intracochlear application of human stromal cell-derived extracellular vesicles. J Extracell Vesicles. 2021;10:e12094. doi: 10.1002/jev2.12094.ad77c6d5597f4f8db4edd230ec54d8d1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Zhang X, Liu J, Yu B, Ma F, Ren X, Li X. Effects of mesenchymal stem cells and their exosomes on the healing of large and refractory macular holes. Graefes Arch Clin Exp Ophthalmol. 2018;256:2041–2052. doi: 10.1007/s00417-018-4097-3. [DOI] [PubMed] [Google Scholar]
- 107.Szebeni J, Muggia F, Gabizon A, Barenholz Y. Activation of complement by therapeutic liposomes and other lipid excipient-based therapeutic products: prediction and prevention. Adv Drug Deliv Rev. 2011;63:1020–1030. doi: 10.1016/j.addr.2011.06.017. [DOI] [PubMed] [Google Scholar]