Abstract
The coronavirus disease 2019 (COVID-19) is an ongoing global pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Patients with severe COVID-19 exhibit hyper-inflammatory responses characterized by excessive activation of myeloid cells, including monocytes, macrophages, and neutrophils, and a plethora of pro-inflammatory cytokines and chemokines. Accumulating evidence also indicates that hyper-inflammation is a driving factor for severe progression of the disease, which has prompted the development of anti-inflammatory therapies for the treatment of patients with COVID-19. Corticosteroids, IL-6R inhibitors, and JAK inhibitors have demonstrated promising results in treating patients with severe disease. In addition, diverse forms of exosomes that exert anti-inflammatory functions have been tested experimentally for the treatment of COVID-19. Here, we briefly describe the immunological mechanisms of the hyper-inflammatory responses in patients with severe COVID-19. We also summarize current anti-inflammatory therapies for the treatment of severe COVID-19 and novel exosome-based therapeutics that are in experimental stages.
Keywords: Anti-inflammation, COVID-19, Exosome, Hyper-inflammation, Therapy
INTRODUCTION
The coronavirus disease 2019 (COVID-19) was first found in patients with unidentified pneumonia in Wuhan, China, in December 2019 and rapidly spread worldwide (1). The World Health Organization (WHO) announced the COVID-19 outbreak to be a pandemic on 11 March, 2020. COVID-19 is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a positive-sense single-stranded RNA virus with high sequence homology to bat coronaviruses (CoVs). Other CoVs have exhibited severe infections in humans. These include SARS-CoV-1 and Middle East respiratory syndrome (MERS)-CoV, which emerged in 2003 and 2012, respectively (2). SARS-CoV-2 uses its spike protein to bind the angiotensin-converting enzyme 2 (ACE2) receptor expressed on the cell membrane for entry into host cells (3, 4). Although SARS-CoV-2 is not as lethal as MERS-CoV and SARS-CoV-1 (5), its substantial spread has resulted in severe casualties and caused overwhelming pressure for the medical system worldwide.
Nearly 20% of patients with COVID-19 experience severe disease (6, 7). Accumulating evidence suggests that hyper-inflammatory responses of the host to SARS-CoV-2 infection lead to severe forms of COVID-19 (8, 9). Immunopathological features, such as excess infiltration of patients’ lungs by macrophages and neutrophils, as well as increased serum cytokine and chemokine levels, are characteristics of severe COVID-19 (6, 10, 11). Anti-inflammatory therapies, including corticosteroids, are currently considered the standard of care in treating patients with severe COVID-19 (12). The clinical efficacy of other anti-inflammatory therapies is being investigated in various clinical trials.
Here, we review the mechanisms of hyper-inflammatory responses in COVID-19 and describe recent advances in anti-inflammatory therapies for COVID-19. We also highlight the potential roles of exosomes as novel anti-inflammatory therapeutics for the treatment of COVID-19.
THE DYSREGULATED IMMUNE RESPONSE IN PATIENTS WITH COVID-19
Severe COVID-19 pathology results from massive initial viral replication that arises because the SARS-CoV-2 can evade and inhibit the host innate immune recognition system and interferon (IFN) responses (13). Type I IFNs (IFN-αs and IFN-β) and type III IFNs (IFN-λs) are the major first-line defenses against viruses (14), but SARS-CoV-2 has developed various strategies to evade and suppress the production and functions of type I and III IFNs and IFN-stimulated genes (ISGs) (15-17). These mechanisms allow SARS-CoV-2 to replicate robustly, leading to excessive activation of monocytes, macrophages, and neutrophils. Excessively activated myeloid cells then produce excessive pro-inflammatory cytokines and chemokines, resulting in hyper-inflammatory responses (13, 18-20).
Excessive neutrophil activation and infiltration
Patients with severe COVID-19 exhibit increased neutrophil counts with a high neutrophil-to-lymphocyte ratio as an independent risk factor for severity (21-23). A transcriptome analysis of bronchoalveolar lavage fluid (BALF) from patients with COVID-19 showed that SARS-CoV-2 infection induces excessive neutrophil infiltration compared to other forms of pneumonia (24). In addition, patients with severe COVID-19 exhibit increased tissue infiltration of neutrophils in the upper airways of the lungs (25) and the bronchoalveolar space (26, 27). A recent study found that NSP10 of SARS-CoV-2 interacts with the NF-κB repressor NKRF to induce IL-8 production, which augments IL-8-mediated chemotaxis of neutrophils and the over-exuberant host inflammatory responses in COVID-19 (28). Furthermore, increased formation of neutrophil extracellular traps (NETs), which are net-like structures composed of DNA, antimicrobials and oxidant enzymes released by neutrophils, exacerbates lung injury and inflammation in patients with severe COVID-19 (29, 30). These findings imply that the dysregulated activation of neutrophils contributes to hyper-inflammatory responses in severe cases of COVID-19.
Dysregulated activation of macrophages and monocytes
Macrophage activation, especially in the lungs, plays a key role in the progression of dysregulated immune responses in patients with severe COVID-19. Single-cell RNA sequencing (scRNA-seq) analysis of BALF from patients with COVID-19 has shown elevated numbers of pro-inflammatory macrophages in the lungs of such patients (24, 26). The lung macrophages of such patients have shown increased expression of pro-inflammatory cytokine genes, such as IL1B, IL6, and TNF, as well as chemokine genes, such as MCP1/CCL2, MIP1A/CCL3, MIP1B/CCL4, and MCP3/CCL7 (24, 26). Dysregulated activation of monocytes also contributes to severe progression of COVID-19. Elevated numbers of inflammatory monocytes have been identified in the blood of patients with severe COVID-19 (31, 32). A recent large-scale single-cell transcriptome atlas study claimed that monocytes in the peripheral blood are key contributors to the cytokine storm in such patients (32). This is supported by a study showing that inflammatory monocytes in the blood of patients with COVID-19 exhibit elevated gene expression related to classical M1 macrophages (33). Strikingly, scRNA-seq analyses showed that pro-inflammatory cytokines trigger the activation and expansion of circulating monocytes, suggesting positive feedback between the activation of monocytes and production of pro-inflammatory cytokines (33, 34). These observations collectively suggest that SARS-CoV-2 infection triggers dysregulated activation of macrophages and monocytes, resulting in the secretion of a plethora of pro-inflammatory cytokines and chemokines.
Mechanistic model of hyper-inflammation in severe COVID-19
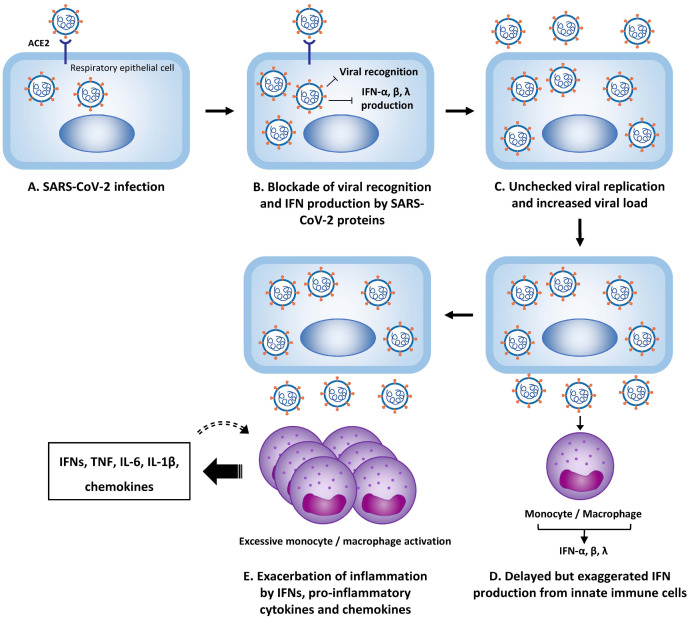
A mechanistic model that explains the contribution of delayed IFN responses to the exacerbated inflammatory response in patients with severe COVID-19 has been proposed (Fig. 1) (13, 20). After SARS-CoV-2 infection of respiratory epithelial cells, the virus efficiently evades host innate immune recognition and IFN responses by blocking the type I and III IFN responses. The viral load rapidly increases, and myeloid cells, such as monocytes and macrophages, are stimulated by viral components via Toll-like receptors (TLRs). Then the monocytes and macrophages produce type I and III IFNs. Positive feedback occurs between the production of IFNs and chemokines and the accumulation and activation of monocytes and macrophages, thus producing large amounts of pro-inflammatory cytokines, such as TNF, IL-6, and IL-1β. This model explains how delayed but exaggerated IFN responses contribute to hyper-inflammation and severe progression of COVID-19.
Fig. 1.
Mechanistic model of hyper-inflammation in COVID-19. After respiratory epithelial cells are infected (A), SARS-CoV-2 proteins block viral-recognition signaling and type I and III interferon (IFN) responses (B). The viral load increases (C) and myeloid cells, such as monocytes and macrophages, are stimulated by viral components via Toll-like receptors, producing type I and III IFNs (D). IFNs further stimulate the production of chemokines and induce the accumulation and activation of monocytes and macrophages, thus producing excessive amounts of pro-inflammatory cytokines (E). This process can be amplified by a positive feedback mechanism.
CURRENT ANTI-INFLAMMATORY THERAPIES FOR COVID-19
Accumulating evidence of hyper-inflammation in patients with severe COVID-19 has provoked the development of anti-inflammatory therapies. Currently, more than 6000 clinical trials testing various therapeutics for treating COVID-19 are registered at clinicaltrials.gov, many of them with anti-inflammatory therapeutics. Various anti-inflammatory agents, such as corticosteroids, IL-6R inhibitors, and JAK inhibitors, have already been shown to be effective in ameliorating hyper-inflammation in COVID-19 (Table 1).
Table 1.
Mechanisms of action and therapeutic efficacy of current anti-inflammatory therapeutics for severe COVID-19
| Therapeutics | Mechanisms of action and therapeutic efficacy |
|---|---|
| Corticosteroids |
|
| IL-6R inhibitors |
|
| JAK inhibitors |
|
Corticosteroids
Glucocorticoids strongly inhibit the immune system. Glucocorticoids function as glucocorticoid receptor (GR) agonists. Binding of the glucocorticoids to the GR activates the receptor to exert anti-inflammatory effects, such as suppressing the production of pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, etc.) (35, 36). A controlled, open-label, randomized RECOVERY trial evaluated the efficacy of the glucocorticoid dexametha-sone in hospitalized patients with COVID-19 (12). There were 2104 COVID-19 patients who received 6 mg of dexamethasone once daily for up to 10 days; these patients were compared with 4321 patients who received usual care (12). The trial demonstrated a lower 28-day death rate in the dexamethasone group versus the usual care group (22.9% versus 25.7%; P < 0.001) (12). In particular, the use of dexamethasone significantly lowered the 28-day death rate for those who received either invasive mechanical ventilation or oxygen alone at randomization, but not for those receiving no respiratory support (12), which implies that dexamethasone is effective only in severe patients with ongoing hyper-inflammation. Early meta-analyses also suggested therapeutic use of corticosteroids in severe COVID-19 patients who require respiratory support or mechanical ventilation (37, 38).
IL-6R inhibitors
IL-6 is an important cytokine involved in the hyper-inflammatory response in patients with severe COVID-19, which promp-ted the use of selective IL-6 inhibitors for treatment of these patients. Tocilizumab, a recombinant humanized monoclonal antibody for IL-6 receptor (IL-6R), exerts therapeutic effects by blocking the binding of IL-6 to IL-6R (39). Tocilizumab was previously found be effective against the cytokine release syndrome resulting from chimeric antigen-receptor T-cell therapy (40). Several studies have shown the therapeutic effect of tocilizumab in treating severe COVID-19 by rapidly decreasing inflammatory markers, improving oxygenation, and reducing the death rate in COVID-19 patients who are on mechanical ventilation (41-43). Nevertheless, there is a debate regarding the therapeutic effect of tocilizumab in treating COVID-19. On the one hand, Gupta et al. conducted a multicenter cohort study of 4485 COVID-19 patients with intensive care unit (ICU) admission and reported that the risk of in-hospital death was lower in patients treated with tocilizumab in the first two days of ICU admission (44). On the other hand, Stone et al. did a randomized, double-blind, placebo-controlled trial with 243 COVID-19 patients with hyper-inflammation and concluded that tocilizumab was not effective at preventing intubation or death in moderately ill patients hospitalized with COVID-19 (45). However, Leaf et al. argued that the result was severely underpowered, by pointing out that the percentage of patients with primary outcomes such as intubation or death was 12.5% in the placebo group, which is far lower than the expected 30% (46). They claimed that the percentage of primary outcomes and number of patients enrolled (n = 243) would have made it nearly impossible for the trial to have demonstrated a therapeutic effect (46). Salama et al. also reported that tocili-zumab did not improve survival in a randomized trial of hos-pitalized COVID-19 patients with pneumonia, but treatment reduced the likelihood of progression to mechanical ventilation or death (47). Similarly, in a randomized trial of 452 hospitalized patients with severe COVID-19 accompanied by pneumonia, Rosas et al. reported that the use of tocilizumab did not lead to improved clinical status or lower death rate than did a placebo at 28 days (48). Another monoclonal anti-body for IL-6R, sarilumab, has also had controversial results in COVID-19 patients (49, 50). Thus, the therapeutic efficacy of the IL-6R inhibitor for COVID-19 patients should be carefully examined in further clinical studies.
JAK inhibitors
Inhibitors of the Janus kinases (JAKs) are powerful anti-inflammatory agents that effectively ameliorate various inflammatory diseases, such as rheumatoid arthritis (51). JAK inhibitors suppress the kinase activity of JAKs by competitively binding to the ATP-binding site of JAKs, thereby inhibiting signal transduction of a wide variety of cytokines (51). In a preclinical study, baricitinib, a clinically approved JAK1/JAK2 inhibitor, suppressed the production of pro-inflammatory cytokines and chemokines from lung macrophages and the recruitment of neutrophils in SARS-CoV-2-infected rhesus macaques (52). In addition, a double-blind, randomized, placebo-controlled trial evaluating the effect of baricitinib plus remdesivir in 1033 hospitalized adults with COVID-19 reported that this combination was more effective than remdesivir alone in reducing the recovery time and im-proving clinical status among patients with severe COVID-19 who were receiving high-flow oxygen or non-invasive ventilation (53). In a retrospective, uncontrolled patient cohort with moderate-to-severe COVID-19, treatment with baricitinib plus hydroxychloroquine demonstrated clinical improvement in 11 of 15 patients (54). In July 2021, baricitinib was approved by the US Food and Drug Administration as a single treatment for hospitalized patients with COVID-19.
Another JAK inhibitor, ruxolitinib, demonstrated clinical improvement in 18 critically ill COVID-19 patients with acute respiratory distress syndrome (ARDS) (55). In a prospective, multicenter, single-blind, randomized controlled phase II trial involving 43 patients, no significant difference was observed in ruxolitinib-treated patients compared to controls, though ruxolitinib recipients had faster clinical improvement (56).
EXOSOMES AS POTENTIAL THERAPEUTICS IN COVID-19
Characteristics of exosomes
Extracellular vesicles (EVs) are natural nanoparticles secreted by the cell. They are classified into three subgroups: exosomes, microvesicles, and apoptotic bodies, which have different biological properties in their biogenesis, content, and size (exosomes, 30-150 nm; microvesicles, 0.1-1 μm; and apoptotic bodies, 1-5 μm) (57, 58). Exosomes are enclosed by a single lipid bilayer which are generated by inward budding of vesicles into endosomes that mature into multivesicular bodies or by direct budding of lipid vesicles from the plasma membrane (59). Exosomes are known to be secreted by all cell types and are present in various body fluids (60-65). Exosomes participate in intercellular delivery of diverse biological molecules, such as nucleic acids (DNA, RNA), proteins, lipids, and carbohydrates. Many efforts have been made to apply exosomes for various therapeutic application via the engineering of exosomes or exosome-producing cells for incorporating active pharmaceutical ingredients (APIs) into exosomes and inducing targetability to specific cells or organs (66-68).
Exosome-based therapeutics for treating COVID-19
Mesenchymal stem cells (MSCs), which are multipotent adult stem cells, are an easily accessible type of stem cell that are present in various human tissues. Considerable interest in MSCs has been raised for their therapeutic efficacy in tissue repair and in suppressing inflammation. Interestingly, enough research has shown that MSCs exert therapeutic effects by secreting EVs, not by a differentiation mechanism (69, 70). In line with this finding, MSC-derived exosomes have demonstrated regenerative potential, immune-modulatory functions, and anti-inflammatory effects (71).
Recent studies have highlighted the therapeutic potential of MSC-derived exosomes for treating COVID-19. In a prospective, non-randomized, open-label cohort study, the efficacy of exosomes derived from allogenic bone-marrow (BM) MSCs was evaluated in 24 COVID-19 patients with moderate-to-severe ARDS (72). BM MSC-derived exosomes demonstrated a survival rate of 83%, with 17 of 24 (71%) patients recovered showing no adverse effects observed within 72 hours of exosome administration (72). In addition, improved respiratory function (PaO2/FiO2) and reduced neutrophil count and acute phase reactants (i.e., C-reactive protein, ferritin, and D-dimer) were observed (72). However, the clinical outcome of this study must be carefully interpreted because little information was provided about the characteristics, biological properties, or proposed biological or therapeutic actions of the BM-MSC-derived exosomes used in this study (73). Four clinical trials, mostly phase 1 or 2, evaluating the therapeutic effect of MSC-derived exosomes in COVID-19-associated pneumonia are currently in progress (NCT04276987, NCT04798716, NCT04602442, NCT04491240, Table 2).
Table 2.
Ongoing clinical trials evaluating the efficacy of exosome therapeutics in COVID-19
| Therapeutic exosomes | Delivery method | Dosage | Phase | NCT number |
|---|---|---|---|---|
| Exosomes overexpressing CD24 | Inhalation | 1010 particles in 4 ml normal saline | 2 | NCT04969172 |
| CovenD24 (exosomes overexpressing CD24) | Inhalation | 109, 1010particles | 2 | NCT04902183 |
| Ardoxso (MSC-derived exosomes) | Intravenous infusion | 2 × 109, 4 × 109, 8 × 109 particles | 1, 2 | NCT04798716 |
| EXO 1, EXO 2 (MSC-derived exosomes) | Inhalation | 0.5-2 × 1010 particles in 3 ml special solution | 2 | NCT04602442 |
| EXO-CD24 (exosomes overexpressing CD24) | Inhalation | 1 × 108-1 × 1010 particles per 2 ml saline | 1 | NCT04747574 |
| EXO 1, EXO 2 (MSC-derived exosomes) | Inhalation | 0.5-2 × 1010 particles in 3 ml special solution | 1 | NCT04491240 |
| CSTC-Exo (COVID-19-specific T cell-derived exosomes) | Inhalation | 2 × 108 particles in 3 ml | 1 | NCT04389385 |
| MSC-derived exosomes | Inhalation | 2 × 108 particles in 3 ml | 1 | NCT04276987 |
Engineered exosomes demonstrating potent anti-inflammatory effects have potential to act as effective immunomodulators to ameliorate the excessive inflammation observed in patients with severe COVID-19. Recently, a clinical trial (NCT0474574, Table 2) conducted in Israel completed a phase 1 trial, in which 30 patients with moderate or worse COVID-19 were treated with CD24-expressing exosomes (EXO-CD24). More results are awaited regarding the therapeutic effect of anti-inflammatory exosomes in relieving immunopathogenesis in severe COVID-19.
Therapeutic exosome platform technologies for efficient intracellular cargo delivery
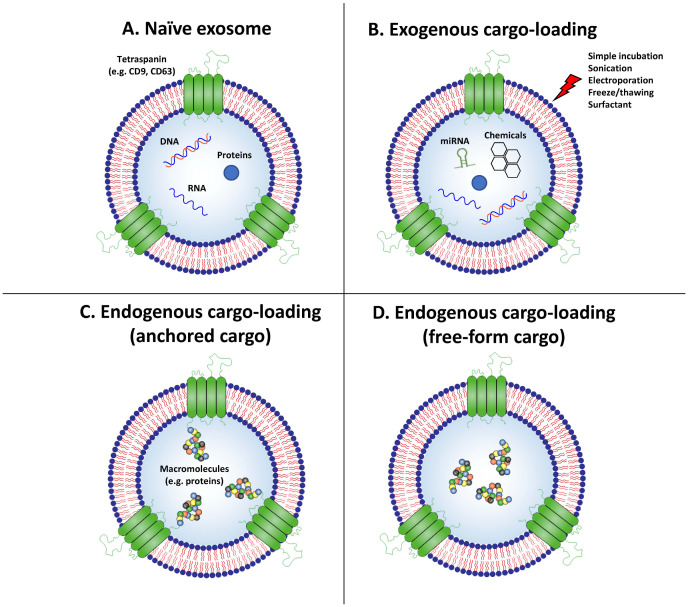
Various exosome engineering platform technologies that can generate exosomes armed with therapeutic cargo have been developed. APIs could be ‘post-incorporated’ into isolated exosomes through exogenous methods, or ‘pre-incorporated’ into exosomes through endogenous methods by modifying the exosome-producing cells (Fig. 2). Exogenous cargo loading methods involves loading APIs into exosomes through methods such as sonication, electroporation, freeze-thaw cycles, and extrusion (Fig. 2B) (74-79). However, a major caveat of these methods is damage to the exosomal membranes during the loading process (80). Endogenous cargo loading uses exosome-producing cells to load APIs into exosomes during natural exosome biogenesis. For example, biological agents can be endogenously incorporated into exosomes by genetically modifying the exosome-producing cells to overexpress the desired proteins or nucleic acids, which are then naturally loaded into exosomes. Macrophage-derived EVs loaded with IL-10 by transfecting the IL10 gene to EV-producing cells exerted therapeutic efficacy in ischemia/reperfusion induced acute kidney injury (AKI) by ameliorating the renal tubular injury and inflammation and driving M2 macrophage polarization via targeted delivery of EVs to macrophages (81). Endogenous cargo loading can be improved by inducing additional modification to the cargo, such as by anchoring the cargo onto the inner/outer membrane of exosomes via conjugation with membrane proteins of exosomes, such as PTGFRN (Fig. 2C) (82). However, the drawback of this approach is that APIs remain attached to the membrane of exosomes after delivery to the target cell, which may dramatically restrict its biological function. Alternatively, a novel technology called EXosomes for Protein Loading via Optically Reversible protein-protein interaction (EXPLOR) has been developed which could load non-anchored free-form proteins into exosomes using light-induced hetero-dimerizing modules, cryptochrome 2 (CRY2), and the N-terminal of CRY-interacting basic-helix-loop-helix 1 (CIBN) isolated from Arabidopsis thaliana (83). CRY2 and CIBN undergo hetero-dimerization in a blue light-specific manner, but reversibly and rapidly dissociate with each other in the absence of blue light (84, 85). By fusing CRY2 with the cargo protein and CIBN with the exosomal membrane protein CD9, cargo proteins can be loaded into exosomes with high yield under blue light via the natural exosome biogenesis pathway (Fig. 2D).
Fig. 2.
Engineering methods for loading therapeutic agents into exosomes. The techniques for loading cargo into exosomes can be divided into four approaches. (A) Using naïve exosomes (e.g., MSC-derived exosomes) requires relatively simple techniques to generate therapeutic exosomes, but the drawback is the difficulty in controlling the bioactive molecules contained in the exosomes. (B) Exogenous cargo loading is based on the use of sonication, repeated freeze/thaw cycles, or electroporation to destabilize the integrity of exosomal membranes and thus allow drugs to be introduced into the exosomes. (C, D) Endogenous cargo loading spontaneously loads molecules of interest by hijacking the natural exosome biogenesis pathway. These techniques are divided into two approaches based on whether the cargo is anchored onto the exosomal membrane (C) or resides as a free form inside the lumen of the exosome (D).
EXPLOR technology has been applied to generate anti-inflammatory exosomes loaded with anti-inflammatory proteins that inhibit the NF-κB signaling pathway. Exosomes loaded with super-repressor IκB (srIκB), a dominant active form of IκBα, generated by EXPLOR technology has demonstrated a promising therapeutic effect in inflammatory diseases, such as sepsis (86-88). SrIκB is a degradation-resistant form of NF-κB-inhibiting protein IκBα, which blocks the nuclear translocation of NF-κB even when pro-inflammatory stimulus is present. Administration of exosomes loaded with srIκB (Exo-srIκB) to septic mouse models ameliorated the death rate and systemic inflammation by reducing the levels of circulating pro-inflammatory cytokines and alleviating acute organ injury (86). Intravital imaging revealed that the administered exosomes are taken up mainly by neutrophils and monocytes, which are attractive target cells for treating hyper-inflammation in COVID-19 (86). Exosomes are cleared by phagocytic cells, such as macrophages and neutrophils, after systemic injection, which makes these cells a primary target for exosomal therapeutics (89, 90). In an AKI model of C57BL/6 mice, Exo-srIκB treatment has demonstrated an anti-inflammatory effect by decreasing gene expression of pro-inflammatory cytokines and adhesion molecules in post-ischemic kidneys (88). In addition, Exo-srIκB administration affects the post-ischemic kidney immune-cell population, reducing the neutrophil, monocyte, and macrophage populations (88). As suppression of the NF-κB pathway has been implied as a potential therapeutic approach for treating patients with severe COVID-19 (91, 92), results demonstrating the therapeutic efficacy of Exo-srIκB in various inflammation-related disorders suggest a possibility of applying Exo-srIκB in the treatment of hyper-inflammation in patients with severe COVID-19.
SUMMARY AND PERSPECTIVE
Excessive activation of myeloid cells, such as monocytes, macrophages, and neutrophils, and exaggerated production of pro-inflammatory cytokines result in hyper-inflammation, leading to severe progression of COVID-19. Anti-inflammatory agents, such as dexamethasone and baricitinib, have shown promising results in ameliorating hyper-inflammation in patients with severe COVID-19, which implies that targeting hyper-inflammation is an appropriate strategy for ameliorating the severity of COVID-19. In addition, exosomes have arisen as a novel therapeutic modality for treating dysregulated inflammatory responses in COVID-19, either as a naïve form or a bioactive cargo delivery vehicle. Promising results of anti-inflammatory exosome therapeutics in ameliorating various inflammatory diseases have indicated the possibility of applying exosomes in the treatment of hyper-inflammation in COVID-19. Recent studies and clinical trials have reported a therapeutic potential of anti-inflammatory exosomes in treating patients with COVID-19. Nonetheless, more research is needed, as well as randomized clinical trials, with enough enrolled patients to verify the efficacy of exosomes in treating patients with severe COVID-19.
Footnotes
CONFLICTS OF INTEREST
H.C. is a minor shareholder of ILIAS Biologics Inc. The authors have no additional financial interests.
REFERENCES
- 1.Zhu N, Zhang DY, Wang WL, et al. A novel Coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fung TS, Liu DX. Human Coronavirus: host-pathogen interaction. Annu Rev Microbiol. 2019;73:529–557. doi: 10.1146/annurev-micro-020518-115759. [DOI] [PubMed] [Google Scholar]
- 3.Lu RJ, Zhao X, Li J, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565–574. doi: 10.1016/S0140-6736(20)30251-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wan YS, Shang J, Graham R, Baric RS, Li F. Receptor recognition by the novel Coronavirus from Wuhan: an analysis based on decade-long structural studies of SARS Coronavirus. J Virol. 2020;94:e00127–20. doi: 10.1128/JVI.00127-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fauci AS, Lane HC, Redfield RR. Covid-19-navigating the uncharted. N Engl J Med. 2020;382:1268–1269. doi: 10.1056/NEJMe2002387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323:2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mehta P, McAuley DF, Brown M, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gustine JN, Jones D. Immunopathology of Hyperinflammation in COVID-19. Am J Pathol. 2021;191:4–17. doi: 10.1016/j.ajpath.2020.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Del Valle DM, Kim-Schulze S, Huang HH, et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat Med. 2020;26:1636–1643. doi: 10.1038/s41591-020-1051-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Group RC, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384:693–704. doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kim YM, Shin EC. Type I and III interferon responses in SARS-CoV-2 infection. Exp Mol Med. 2021;53:750–760. doi: 10.1038/s12276-021-00592-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mesev EV, LeDesma RA, Ploss A. Decoding type I and III interferon signalling during viral infection. Nat Microbiol. 2019;4:914–924. doi: 10.1038/s41564-019-0421-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Park A, Iwasaki A. Type I and type III interferons - induction, signaling, evasion, and application to combat COVID-19. Cell Host Microbe. 2020;27:870–878. doi: 10.1016/j.chom.2020.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ribero MS, Jouvenet N, Dreux M, Nisole S. Interplay between SARS-CoV-2 and the type I interferon response. PLoS Pathog. 2020;16:e1008737. doi: 10.1371/journal.ppat.1008737.fbb3406527044db2bf04bc34a750f74e [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Choi H, Shin EC. Roles of type I and III interferons in COVID-19. Yonsei Med J. 2021;62:381–390. doi: 10.3349/ymj.2021.62.5.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Perico L, Benigni A, Casiraghi F, Ng LFP, Renia L, Remuzzi G. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat Rev Nephrol. 2021;17:46–64. doi: 10.1038/s41581-020-00357-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee JS, Park S, Jeong HW, et al. Immunophenotyping of COVID-19 and influenza highlights the role of type I interferons in development of severe COVID-19. Sci Immunol. 2020;5:eabd1554. doi: 10.1126/sciimmunol.abd1554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Galani IE, Rovina N, Lampropoulou V, et al. Untuned antiviral immunity in COVID-19 revealed by temporal type I/III interferon patterns and flu comparison. Nat Immunol. 2021;22:32–40. doi: 10.1038/s41590-020-00840-x. [DOI] [PubMed] [Google Scholar]
- 21.Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel Coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liu J, Liu Y, Xiang P, et al. Neutrophil-to-lymphocyte ratio predicts critical illness patients with 2019 coronavirus disease in the early stage. J Transl Med. 2020;18:206. doi: 10.1186/s12967-020-02374-0.0ff25af72de3466cbc47d442fad1e6c3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Carissimo G, Xu W, Kwok I, et al. Whole blood immunophenotyping uncovers immature neutrophil-to-VD2 T-cell ratio as an early marker for severe COVID-19. Nat Commun. 2020;11:5243. doi: 10.1038/s41467-020-19080-6.d967340f6639442885027de8b53a2e43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhou Z, Ren L, Zhang L, et al. Heightened innate immune responses in the respiratory tract of COVID-19 patients. Cell Host Microbe. 2020;27:883–890. doi: 10.1016/j.chom.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chua RL, Lukassen S, Trump S, et al. COVID-19 severity correlates with airway epithelium-immune cell interactions identified by single-cell analysis. Nat Biotechnol. 2020;38:970–979. doi: 10.1038/s41587-020-0602-4. [DOI] [PubMed] [Google Scholar]
- 26.Liao M, Liu Y, Yuan J, et al. Single-cell landscape of bronchoalveolar immune cells in patients with COVID-19. Nat Med. 2020;26:842–844. doi: 10.1038/s41591-020-0901-9. [DOI] [PubMed] [Google Scholar]
- 27.Wauters E, Van Mol P, Garg AD, et al. Discriminating mild from critical COVID-19 by innate and adaptive immune single-cell profiling of bronchoalveolar lavages. Cell Res. 2021;31:272–290. doi: 10.1038/s41422-020-00455-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li J, Guo M, Tian X, et al. Virus-host interactome and proteomic survey reveal potential virulence factors influencing SARS-CoV-2 pathogenesis. Med (N Y) 2021;2:99–112. doi: 10.1016/j.medj.2020.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Middleton EA, He XY, Denorme F, et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood. 2020;136:1169–1179. doi: 10.1182/blood.2020007008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zuo Y, Yalavarthi S, Shi H, et al. Neutrophil extracellular traps in COVID-19. JCI Insight. 2020;5:e138999. doi: 10.1172/jci.insight.138999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bost P, Giladi A, Liu Y, et al. Host-viral infection maps reveal signatures of severe COVID-19 patients. Cell. 2020;181:1475–1488. doi: 10.1016/j.cell.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ren X, Wen W, Fan X, et al. COVID-19 immune features revealed by a large-scale single-cell transcriptome atlas. Cell. 2021;184:1895–1913. doi: 10.1016/j.cell.2021.01.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wen W, Su W, Tang H, et al. Immune cell profiling of COVID-19 patients in the recovery stage by single-cell sequencing. Cell Discov. 2020;6:31. doi: 10.1038/s41421-020-0168-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Guo C, Li B, Ma H, et al. Single-cell analysis of two severe COVID-19 patients reveals a monocyte-associated and tocilizumab-responding cytokine storm. Nat Commun. 2020;11:3924. doi: 10.1038/s41467-020-17834-w.725c63210c6543e5bbfeb15fc0ba1839 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Williams DM. Clinical pharmacology of corticosteroids. Respir Care. 2018;63:655–670. doi: 10.4187/respcare.06314. [DOI] [PubMed] [Google Scholar]
- 36.Brattsand R, Linden M. Cytokine modulation by glucocorticoids: mechanisms and actions in cellular studies. Aliment Pharmacol Ther. 1996;10 Suppl 2:81–90. discussion 91–82. doi: 10.1046/j.1365-2036.1996.22164025.x. [DOI] [PubMed] [Google Scholar]
- 37.Pulakurthi YS, Pederson JM, Saravu K, et al. Corticosteroid therapy for COVID-19: A systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore) 2021;100:e25719. doi: 10.1097/MD.0000000000025719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Johns M, George S, Taburyanskaya M, Poon YK. A review of the evidence for corticosteroids in COVID-19. J Pharm Pract. 2021:897190021998502. doi: 10.1177/0897190021998502. [DOI] [PubMed] [Google Scholar]
- 39.Sebba A. Tocilizumab: the first interleukin-6-receptor inhibitor. Am J Health Syst Pharm. 2008;65:1413–1418. doi: 10.2146/ajhp070449. [DOI] [PubMed] [Google Scholar]
- 40.Le RQ, Li L, Yuan W, et al. FDA approval summary: Tocilizumab for treatment of chimeric antigen receptor T cell-induced severe or life-threatening cytokine release syndrome. Oncologist. 2018;23:943–947. doi: 10.1634/theoncologist.2018-0028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xu X, Han M, Li T, et al. Effective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci U S A. 2020;117:10970–10975. doi: 10.1073/pnas.2005615117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sciascia S, Apra F, Baffa A, et al. Pilot prospective open, single-arm multicentre study on off-label use of tocilizumab in patients with severe COVID-19. Clin Exp Rheumatol. 2020;38:529–532. [PubMed] [Google Scholar]
- 43.Guaraldi G, Meschiari M, Cozzi-Lepri A, et al. Tocilizumab in patients with severe COVID-19: a retrospective cohort study. Lancet Rheumatol. 2020;2:e474–e484. doi: 10.1016/S2665-9913(20)30173-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gupta S, Wang W, Hayek SS, et al. Association between early treatment with Tocilizumab and mortality among critically ill patients with COVID-19. JAMA Intern Med. 2021;181:41–51. doi: 10.1001/jamainternmed.2020.6252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Stone JH, Frigault MJ, Serling-Boyd NJ, et al. Efficacy of Tocilizumab in patients hospitalized with Covid-19. N Engl J Med. 2020;383:2333–2344. doi: 10.1056/NEJMoa2028836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Leaf DE, Gupta S, Wang W. Tocilizumab in Covid-19. N Engl J Med. 2021;384:86–87. doi: 10.1056/NEJMc2032911. [DOI] [PubMed] [Google Scholar]
- 47.Salama C, Han J, Yau L, et al. Tocilizumab in patients hospitalized with Covid-19 pneumonia. N Engl J Med. 2021;384:20–30. doi: 10.1056/NEJMoa2030340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rosas IO, Brau N, Waters M, et al. Tocilizumab in hospitalized patients with severe Covid-19 pneumonia. N Engl J Med. 2021;384:1503–1516. doi: 10.1056/NEJMoa2028700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Della-Torre E, Campochiaro C, Cavalli G, et al. Interleukin-6 blockade with sarilumab in severe COVID-19 pneumonia with systemic hyperinflammation: an open-label cohort study. Ann Rheum Dis. 2020;79:1277–1285. doi: 10.1136/annrheumdis-2020-218122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lescure FX, Honda H, Fowler RA, et al. Sarilumab in patients admitted to hospital with severe or critical COVID-19: a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Respir Med. 2021;9:522–532. doi: 10.1016/S2213-2600(21)00099-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Morinobu A. JAK inhibitors for the treatment of rheumatoid arthritis. Immunol Med. 2020;43:148–155. doi: 10.1080/25785826.2020.1770948. [DOI] [PubMed] [Google Scholar]
- 52.Hoang TN, Pino M, Boddapati AK, et al. Baricitinib treatment resolves lower-airway macrophage inflammation and neutrophil recruitment in SARS-CoV-2-infected rhesus macaques. Cell. 2021;184:460–475. doi: 10.1016/j.cell.2020.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kalil AC, Patterson TF, Mehta AK, et al. Baricitinib plus Remdesivir for hospitalized adults with Covid-19. N Engl J Med. 2021;384:795–807. doi: 10.1056/NEJMoa2031994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Titanji BK, Farley MM, Mehta A, et al. Use of Baricitinib in patients with moderate to severe Coronavirus disease 2019. Clin Infect Dis. 2021;72:1247–1250. doi: 10.1093/cid/ciaa879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Capochiani E, Frediani B, Iervasi G, et al. Ruxo-litinib rapidly reduces acute respiratory distress syndrome in COVID-19 disease. Analysis of data collection From RESPIRE protocol. Front Med (Lausanne) 2020;7:466. doi: 10.3389/fmed.2020.00466.9e8bebcd1b2d49f2a1dfaa4c777d894e [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cao Y, Wei J, Zou L, et al. Ruxolitinib in treatment of severe coronavirus disease 2019 (COVID-19): a multicenter, single-blind, randomized controlled trial. J Allergy Clin Immunol. 2020;146:137–146. doi: 10.1016/j.jaci.2020.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200:373–383. doi: 10.1083/jcb.201211138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Thery C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol. 2002;2:569–579. doi: 10.1038/nri855. [DOI] [PubMed] [Google Scholar]
- 59.Pegtel DM, Gould SJ. Exosomes. Annu Rev Biochem. 2019;88:487–514. doi: 10.1146/annurev-biochem-013118-111902. [DOI] [PubMed] [Google Scholar]
- 60.Peng H, Ji W, Zhao R, et al. Exosome: a significant nano-scale drug delivery carrier. J Mater Chem B. 2020;8:7591–7608. doi: 10.1039/D0TB01499K. [DOI] [PubMed] [Google Scholar]
- 61.Caby MP, Lankar D, Vincendeau-Scherrer C, Raposo G, Bonnerot C. Exosomal-like vesicles are present in human blood plasma. Int Immunol. 2005;17:879–887. doi: 10.1093/intimm/dxh267. [DOI] [PubMed] [Google Scholar]
- 62.Pisitkun T, Shen RF, Knepper MA. Identification and proteomic profiling of exosomes in human urine. Proc Natl Acad Sci U S A. 2004;101:13368–13373. doi: 10.1073/pnas.0403453101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Michael A, Bajracharya SD, Yuen PS, et al. Exosomes from human saliva as a source of microRNA biomarkers. Oral Dis. 2010;16:34–38. doi: 10.1111/j.1601-0825.2009.01604.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Admyre C, Johansson SM, Qazi KR, et al. Exosomes with immune modulatory features are present in human breast milk. J Immunol. 2007;179:1969–1978. doi: 10.4049/jimmunol.179.3.1969. [DOI] [PubMed] [Google Scholar]
- 65.Vojtech L, Woo S, Hughes S, et al. Exosomes in human semen carry a distinctive repertoire of small non-coding RNAs with potential regulatory functions. Nucleic Acids Res. 2014;42:7290–7304. doi: 10.1093/nar/gku347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Jafari D, Shajari S, Jafari R, et al. Designer exosomes: a new platform for biotechnology therapeutics. BioDrugs. 2020;34:567–586. doi: 10.1007/s40259-020-00434-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zipkin M. Exosome redux. Nat Biotechnol. 2019;37:1395–1400. doi: 10.1038/s41587-019-0326-5. [DOI] [PubMed] [Google Scholar]
- 68.Kim J, Song Y, Park CH, Choi C. Platform technologies and human cell lines for the production of therapeutic exosomes. Extracell Vesicles Circ Nucl Acids. 2021;2:3–17. doi: 10.20517/evcna.2020.01. [DOI] [Google Scholar]
- 69.Caplan AI, Dennis JE. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006;98:1076–1084. doi: 10.1002/jcb.20886. [DOI] [PubMed] [Google Scholar]
- 70.Baek G, Choi H, Kim Y, Lee HC, Choi C. Mesenchymal stem cell-derived extracellular vesicles as therapeutics and as a drug delivery platform. Stem Cells Transl Med. 2019;8:880–886. doi: 10.1002/sctm.18-0226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Suh JH, Joo HS, Hong EB, Lee HJ, Lee JM. Therapeutic application of exosomes in inflammatory diseases. Int J Mol Sci. 2021;22:1144. doi: 10.3390/ijms22031144.aa70761881b343689fa667cffd5933d7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Sengupta V, Sengupta S, Lazo A, Woods P, Nolan A, Bremer N. Exosomes derived from bone marrow mesenchymal stem cells as treatment for severe COVID-19. Stem Cells Dev. 2020;29:747–754. doi: 10.1089/scd.2020.0080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lim SK, Giebel B, Weiss DJ, Witwer KW, Rohde E. Re: "Exosomes Derived from Bone Marrow Mesenchymal Stem Cells as Treatment for Severe COVID-19" by Sengupta et al. Stem Cells Dev. 2020;29:877–878. doi: 10.1089/scd.2020.0089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sun D, Zhuang X, Xiang X, et al. A novel nanoparticle drug delivery system: the anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Mol Ther. 2010;18:1606–1614. doi: 10.1038/mt.2010.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kim MS, Haney MJ, Zhao Y, et al. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine. 2016;12:655–664. doi: 10.1016/j.nano.2015.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Alvarez-Erviti L, Seow Y, Yin H, Betts C, Lakhal S, Wood MJ. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol. 2011;29:341–345. doi: 10.1038/nbt.1807. [DOI] [PubMed] [Google Scholar]
- 77.Lamichhane TN, Jeyaram A, Patel DB, et al. Oncogene knockdown via active loading of small rnas into extracellular vesicles by sonication. Cell Mol Bioeng. 2016;9:315–324. doi: 10.1007/s12195-016-0457-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hood JL. Post isolation modification of exosomes for nanomedicine applications. Nanomedicine (Lond) 2016;11:1745–1756. doi: 10.2217/nnm-2016-0102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kamerkar S, LeBleu VS, Sugimoto H, et al. Exosomes facilitate therapeutic targeting of oncogenic KRAS in pancreatic cancer. Nature. 2017;546:498–503. doi: 10.1038/nature22341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hood JL, Scott MJ, Wickline SA. Maximizing exosome colloidal stability following electroporation. Anal Biochem. 2014;448:41–49. doi: 10.1016/j.ab.2013.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Tang TT, Wang B, Wu M, et al. Extracellular vesicle-encapsulated IL-10 as novel nanotherapeutics against ischemic AKI. Sci Adv. 2020;6:eaaz0748. doi: 10.1126/sciadv.aaz0748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Dooley K, McConnell RE, Xu K, et al. A versatile platform for generating engineered extracellular vesicles with defined therapeutic properties. Mol Ther. 2021;29:1729–1743. doi: 10.1016/j.ymthe.2021.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yim N, Ryu SW, Choi K, et al. Exosome engineering for efficient intracellular delivery of soluble proteins using optically reversible protein-protein interaction module. Nat Commun. 2016;7:12277. doi: 10.1038/ncomms12277.5ecca384bd0343d7bad8c5a0f51a75de [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Shalitin D, Yang H, Mockler TC, et al. Regulation of Arabidopsis cryptochrome 2 by blue-light-dependent phosphorylation. Nature. 2002;417:763–767. doi: 10.1038/nature00815. [DOI] [PubMed] [Google Scholar]
- 85.Kennedy MJ, Hughes RM, Peteya LA, Schwartz JW, Ehlers MD, Tucker CL. Rapid blue-light-mediated induction of protein interactions in living cells. Nat Methods. 2010;7:973–975. doi: 10.1038/nmeth.1524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Choi H, Kim Y, Mirzaaghasi A, et al. Exosome-based delivery of super-repressor IkappaBalpha relieves sepsis-associated organ damage and mortality. Sci Adv. 2020;6:eaaz6980. doi: 10.1126/sciadv.aaz6980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Sheller-Miller S, Radnaa E, Yoo JK, et al. Exosomal delivery of NF-kappaB inhibitor delays LPS-induced preterm birth and modulates fetal immune cell profile in mouse models. Sci Adv. 2021;7:eabd3865. doi: 10.1126/sciadv.abd3865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kim S, Lee SA, Yoon H, et al. Exosome-based delivery of super-repressor IkappaBalpha ameliorates kidney ischemia-reperfusion injury. Kidney Int. 2021;100:570–584. doi: 10.1016/j.kint.2021.04.039. [DOI] [PubMed] [Google Scholar]
- 89.Choi H, Choi Y, Yim HY, Mirzaaghasi A, Yoo JK, Choi C. Biodistribution of exosomes and engineering strategies for targeted delivery of therapeutic exosomes. Tissue Eng Regen Med. 2021;18:499–511. doi: 10.1007/s13770-021-00361-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Imai T, Takahashi Y, Nishikawa M, et al. Macro-phage-dependent clearance of systemically administered B16BL6-derived exosomes from the blood circulation in mice. J Extracell Vesicles. 2015;4:26238. doi: 10.3402/jev.v4.26238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Kircheis R, Haasbach E, Lueftenegger D, Heyken WT, Ocker M, Planz O. NF-kappaB pathway as a potential target for treatment of critical stage COVID-19 patients. Front Immunol. 2020;11:598444. doi: 10.3389/fimmu.2020.598444.679bf59e1bb3412f81768c659425f182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Hariharan A, Hakeem AR, Radhakrishnan S, Reddy MS, Rela M. The role and therapeutic potential of nf-kappa-b Pathway in Severe COVID-19 patients. Inflammopharmacology. 2021;29:91–100. doi: 10.1007/s10787-020-00773-9. [DOI] [PMC free article] [PubMed] [Google Scholar]