Abstract
A 17-year-old boy was admitted for management of ventricular fibrillation (VF) with intermittent Brugada pattern on ECG. On evaluation, cardiac MRI revealed myocardial scar and mediastinal lymphadenopathy. 18-Fluorodeoxyglucose positron emission tomography scan showed inflammation in the heart, lungs, and lymph nodes. He was diagnosed as a case of cardiac sarcoidosis (CS) and treated with steroids. However, there was a reactivation of cardiac inflammation and the development of a second VF storm. Following catheter ablation, the patient's arrhythmia improved. This report highlights the inflammation due to CS mimicking channelopathic features.
Keywords: Cardiac sarcoidosis, Ventricular fibrillation storm, Inflammatory cardiomyopathy, Brugada phenocopy, Brugada syndrome, Catheter ablation, Epicardial ablation
1. Introduction
Cardiac inflammation and inflammatory mediators can cause ion channel dysfunction [1]. Inflammation and fibrosis in the right ventricular outflow tract (RVOT) can have Brugada phenotypical presentation due to sodium channel dysfunction. We describe a case of ventricular fibrillation (VF) storm diagnosed in a patient with cardiac sarcoidosis (CS) presenting with Brugada phenotype.
2. Case report
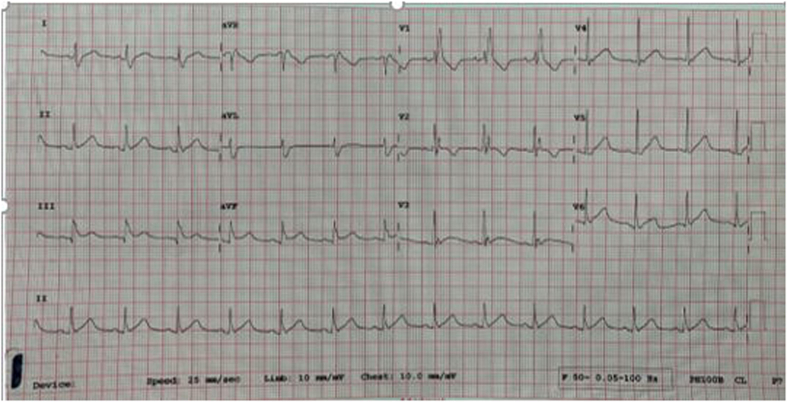
A 17-year-old boy was referred after being resuscitated from sudden cardiac death (SCD). He was diagnosed to have recurrent seizures and treated with intravenous phenytoin. He worsened, with increasing seizure frequency after the phenytoin infusion. VF was found to be the cause for seizing activity and referred to our institute for arrhythmia management. He presented to the emergency room (ER) with an electrical storm. His resting ECGs showed a dynamic type I Brugada pattern (Fig. 1a and b). He had frequent runs of VF and polymorphic ventricular tachycardia (PMVT) (Fig. 1d and e). Short-coupled premature ventricular complexes (PVCs) of different morphologies (suggestive of pleomorphism), were observed to trigger VF (Fig. 1e). He also developed occasional monomorphic ventricular tachycardia (MMVT) and intermittent atrial fibrillation (AF) (Fig. 1e, c).
Fig. 1.
a- Baseline ECG; b- 12 lead ECG showing type I Brugada pattern, inset showing 2 mm coved type ST elevation in lead V2; c- Follow-up ECG during VT-Storm, showing the progression of disease in the form of RBBB with underlying atrial fibrillation; d- Single strip ECG showing polymorphic VT induced by short-coupled PVC; e− Single strip ECG showing PVCs of more than one morphology (black arrows) and VF; f- Single strip ECG showing monomorphic VT. ECG-electrocardiograph, VT-ventricular tachycardia; RBBB- right bundle branch block; PVC- premature ventricular contraction; VF-ventricular fibrillation.
He had no significant past medical ailments, and there was no family history of SCD. Transthoracic echocardiogram revealed a structurally normal heart. He was febrile and physical examination was otherwise unremarkable. In view of the VF storm, the patient was intubated and maintained on mechanical ventilatory support. Intravenous phenytoin was stopped and antipyretics, isoprenaline and cold saline infusion were initiated in the ER to control the VF storm. The VF episodes subsided over the next 30 minutes, and there was no further episode throughout the hospital stay. Twenty-four hours later, the patient was weaned off the ventilator, and isoprenaline infusion was discontinued. Due to the unavailability of quinidine, the patient was started on oral cilostazol and theophylline as prophylaxis to prevent VF.
His initial laboratory tests revealed normal blood counts and electrolytes levels except for elevated C-reactive protein (CRP) (80 mg/l). Late gadolinium-enhanced cardiac magnetic resonance imaging (CMRI) revealed mid-septal and right ventricular (RV) free wall scarring with multiple mediastinal and cervical lymph node enlargement (Fig. 2a). Following that, the patient underwent a refurbished single-chamber implantable cardioverter-defibrillator (ICD) implantation. Due to the presence of lymphadenopathy with myocardial scar, 18-fluorodeoxyglucose positron emission tomography, computed tomography (FDG-PET CT) was performed. FDG-PET-CT identified abnormal uptake in the heart, right lung, and lymph nodes. In the heart, focal areas of increased uptake were found in the posterior interventricular septum and anterolateral papillary muscle of the left ventricle (LV) (Fig. 2b). In the lungs, there was diffuse ground glass uptake in the right upper lobe. Metabolically active right supraclavicular, paratracheal, and hilar nodes were seen on PET-CT scan (Fig. 2b). Excision biopsy and histological study of the right supraclavicular node showed reactive hyperplasia without any evidence of granuloma. Tests for tuberculosis (acid-fast stain, culture for mycobacterium, and Gene-Xpert test) in the biopsy specimen and interferon-gamma release assay were negative. The serum angiotensin enzyme level was normal. Genetic testing did not reveal pathogenic mutations consistent with BrS. ECG screening among family members with modified lead positions did not reveal Brugada pattern.
Fig. 2.
a- Late gadolinium-enhanced cardiac MRI showing scar (white arrows) on the left side of the interventricular septum and right ventricular free wall; b- Cardiac 18-PET-CT scan with 18-FDG showing multiple mediastinal and right supraclavicular lymph node uptake along with myocardial uptake at baseline; c- PET scan after three months of therapy showing resolution of lymph node. See text for details. MRI- Magnetic Resonance Imaging; PET CT- Positron Emission Tomography and Computed Tomography; 18-FDG- 18-fluorodeoxyglucose.
A diagnosis of inflammatory cardiomyopathy with mediastinal lymphadenopathy due to clinical CS presenting as Brugada phenocopy was considered and oral steroid therapy was initiated. During index hospitalization, the patient developed a new-onset right bundle branch block (RBBB) with fractionated QRS (fQRS) in the precordial leads, and this pattern persisted during follow-up. Four months later, the patient presented with a single episode of appropriate ICD shock. The repeat FDG-PET showed resolution of lung and lymph nodes lesions, albeit an increase in myocardial uptake in the interventricular septum and LV papillary muscle was observed (Fig. 2c). Two days later, the patient presented with a VF storm. Monomorphic short-coupled PVCs triggered VF episodes. The ECG showed early repolarization pattern in inferior leads in between VF episodes (supplementary image 1). He also developed intermittent atrial fibrillation. He was afebrile. He was initiated on isoprenaline infusion, and hypothermia was induced. Due to failure of medical therapy, the patient underwent emergency catheter ablation. The right ventricular endo-epicardial electroanatomical mapping was performed using the CARTO 3 system (Biosense Webster Inc., Diamond Bar, CA, USA), as per institutional protocol. In the lab, monomorphic PVC of the RVOT morphology triggering VFs was spontaneously induced (Fig. 3b). The earliest activation was localized to the RVOT free wall, where the local bipolar electrogram (EGM) preceded QRS onset by 42msec. Pace-mapping at this site showed a good morphology match (Fig. 3c).
Fig. 3.
a- ECG showing RBBB with ST-elevation V1– V3 (type 1 Brugada pattern); b- ECG showing PVC (second beat) from RVOT; c- ECG showing good pace map for RVOT PVC; d- ECG post-ablation showing persistent RBBB but ST-elevation settled; e, f- 3-D CARTO guided voltage map (normal >1.5 mV, border zone 0.5–1.5 mV, scar <0.5 mV of right ventricle endocardial (e) and epicardial (f) anteroposterior view. The white arrow in figure e showing the earliest site for clinical PVC (orange dot). Blue dots-isolated late potentials (red arrow); Light orange dots-fractionated signals, Red dots-ablation sites. See text for details. RBBB-Right bundle branch block; PVC- premature ventricular contraction; RVOT-right ventricular outflow tract.
During the initial ablation at this site, there was an acceleration of PVCs and VF. Following the termination of VF, the lesion was consolidated. Both endocardial and epicardial substrate mapping of RV showed scar (Fig. 3e and f) with fractionated and late potentials, in the RVOT and peritricuspid region. These abnormal EGMs were ablated. Following ablation, PES was performed, and there was no inducible VT or VF. Warm saline was irrigated in the pericardial space, and PES was performed. There was no inducible tachyarrhythmia or appearance of the Brugada pattern. Post substrate modification, type 1 Brugada pattern resolved (Fig. 3a and d). He was continued on oral steroids and cilostazol for 12 months. The PET scan repeated after 12 months did not show any cardiac or extracardiac inflammation.
3. Discussion
Inflammatory cardiomyopathy with mediastinal lymphadenopathy and lung involvement is likely to be a granulomatous disease such as CS and tuberculosis. In the absence of tissue diagnosis, the patient was diagnosed to have clinical CS according to updated Japanese circulation society guidelines [2]. In CS, primary pathology is granulomatous inflammation affecting the myocardium and conduction system. Due to the heterogeneous cardiac involvement, CS manifestations can mimic many other primary arrhythmic disorders like ARVD, CPVT, etc. [3,4] The presence of cardiac inflammation, mediastinal lymphadenopathy, multifocal myocardial scar with abnormal substrate in the RVOT and peritricuspid region, conduction system abnormality makes us consider cardiac sarcoidosis as the primary etiology for VF storm. The inflammation and scar due to sarcoidosis is the likely cause for the Brugada phenocopy in this case.
In the present patient, there are other possibilities to consider apart from CS as the primary diagnosis, namely: inflammation playing a causal role in the pathogenesis of Brugada syndrome (BrS); CS inflammation acting as a second hit for the expression of BrS or fortuitous occurrence of both conditions. Histological studies in BrS have demonstrated the presence of inflammation, cardiomyopathy changes, and viral genome in the RV [5]. Pieroni et al. [6] showed that arrhythmia inducibility was strongly associated with RVOT inflammation in BrS patients. Bonny et al. found a higher level of inflammatory marker-CRP in symptomatic BrS compared to asymptomatic patients [7]. Also, Li et al. showed the presence of isolated cardiac inflammation by PET in the BrS VF storm [8]. Further studies are needed to address the role of inflammation in the etiopathogenesis of BrS.
Apart from a genetic abnormality, channelopathies have been shown to occur due to inflammatory cytokine-mediated ion channel and gap junction protein dysfunction, causing ventricular arrhythmias [1,9]. Besides, the myocardial inflammatory process often results in fibrosis, which can give rise to ventricular arrhythmia. Superimposed inflammation or exacerbation of smouldering inflammation in the back ground of fibrosis due to CS, is probably responsible for the VF storm in the patient.
4. Conclusion
Inflammatory cardiomyopathy like cardiac sarcoidosis can mimic channelopathy like Brugada syndrome. Active inflammation due to sarcoidosis could serve as a potential therapeutic target in prevention of VF storm. Cardiac PET and CMRI should be used to identify the inflammation and risk-stratify Brugada phenocopy.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of competing interest
None of the authors have any conflicts of interest.
Footnotes
Peer review under responsibility of Indian Heart Rhythm Society.
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ipej.2021.10.004.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
figs1.
References
- 1.Lazzerini P.E., Capecchi P.L., Laghi-Pasini F., Boutjdir M. Autoimmune channelopathies as a novel mechanism in cardiac arrhythmias. Nat Rev Cardiol. 2017;14:521–535. doi: 10.1038/nrcardio.2017.61. [DOI] [PubMed] [Google Scholar]
- 2.Terasaki F., Yoshinaga K. New guidelines for diagnosis of cardiac sarcoidosis in Japan. Ann Nucl Cardiol. 2017;3:42–45. [Google Scholar]
- 3.Vasaiwala S.C., Finn C., Delpriore J., Leya F., Gagermeier J., Akar J.G., Santucci P., Dajani K., Bova D., Picken M.M., Basso C., Marcus F., Wilber D.J. Prospective study of cardiac sarcoid mimicking arrhythmogenic right ventricular dysplasia. J Cardiovasc Electrophysiol. 2009;20:473–476. doi: 10.1111/j.1540-8167.2008.01351.x. [DOI] [PubMed] [Google Scholar]
- 4.Benjamin M.M., Hayes K., Field M.E., Scheinman M.M., Hoffmayer K.S. Bidirectional ventricular tachycardia in cardiac sarcoidosis. J Arrhythm. 2017;33(1):69–72. doi: 10.1016/j.joa.2016.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Frustaci A., Priori S.G., Pieroni M., et al. Cardiac histological substrate in patients with clinical phenotype of Brugada syndrome. Circulation. 2005;112:3680–3687. doi: 10.1161/CIRCULATIONAHA.105.520999. [DOI] [PubMed] [Google Scholar]
- 6.Pieroni M., Notarstefano P., Oliva A., et al. Electroanatomic and pathologic right ventricular outflow tract abnormalities in patients with Brugada syndrome. J Am Coll Cardiol. 2018;72:2747–2757. doi: 10.1016/j.jacc.2018.09.037. [DOI] [PubMed] [Google Scholar]
- 7.Bonny A., Tonet J., Márquez M.F., De Sisti A., Temfemo A., Himbert C., Gueffaf F., Larrazet F., Ditah I., Frank R., Hidden-Lucet F., Fontaine G. C-reactive protein levels in the Brugada syndrome. Cardiol Res Pract. 2011:1–6. doi: 10.4061/2011/341521. 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li A., Tung R., Shivkumar K., Bradfield J.S. Brugada syndrome-Malignant phenotype associated with acute cardiac inflammation? HeartRhythm Case Rep. 2017 Jun 20;3(8):384–388. doi: 10.1016/j.hrcr.2017.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lazzerini P.E., Capecchi P.L., Laghi-Pasini F. Systemic inflammation and arrhythmic risk: lessons from rheumatoid arthritis. Eur Heart J. 2017;38:1717–1727. doi: 10.1093/eurheartj/ehw208. [DOI] [PubMed] [Google Scholar]