Abstract
Background:
To evaluate the efficacy and safety of programmed intermittent epidural bolus (PIEB) in parturients
Methods:
The PubMed, Embase, and the Cochrane Library (from inception to July 2021) were searched for identification of randomized placebo-controlled trials in which PIEB was applied in parturients. The outcomes were the effect of analgesia, satisfaction score, mode of delivery, duration of labor, neonatal condition, and adverse events. The pooled odds ratios (OR), weighted mean difference (WMD), and 95% confidence intervals (CIs) were calculated using random- and fixed-effects models.
Results:
PIEB was found to be associated with decreased total consumption of ropivacaine (WMD = −15.83, 95% CI: −19.06 to −12.60, P < .00001; I2 = 61%; P for heterogeneity = .04), total consumption of sufentanil (WMD = −4.93, 95% CI: −6.87 to 2.98, P < .00001; I2 = 68%; P for heterogeneity = .05), numbers of patients who require patient-controlled epidural analgesia bolus (OR = 0.27, 95% CI: 0.14–0.51, P < .0001; I2 = 65%; P for heterogeneity = .01), the number of attempts (WMD = −4.12, 95% CI: −7.21 to −1.04, P = .009; I2 = 100%; P for heterogeneity < .00001), rate of breakthrough pain (OR = 0.47, 95% CI: 0.28–0.80, P = .005; I2 = 47%; P for heterogeneity = .09). Eight studies focus on the duration of analgesia. After by meta-analysis, we found that the pain visual analogue scale (VAS) score at 30 minutes, 2 hours, 4 hours, and 5 hours in PIEB group was significantly lower when compared with control group, (WMD = −0.15, 95% CI: −0.26 to −0.04, P = .006; I2 = 0%; P for heterogeneity = .64), (WMD = −0.79, 95% CI: −1.32 to 0.25, P = .004; I2 = 97%; P for heterogeneity < .00001), (WMD = −1.00, 95% CI: −1.08 to −0.91, P < .00001; I2 = 0%; P for heterogeneity = .67), (WMD = −1.81, 95% CI: −3.23 to −0.39, P = .01; I2 = 98%; P for heterogeneity < .00001), respectively. Nineteen studies discussed the mode of delivery between 2 groups. The results suggest that the rate of normal delivery is significantly higher in PIEB group compared with control group (OR = 1.37, 95% CI: 1.08–1.75, P = .01). The time of first and second stage of labor are significantly shorter in PIEB group compared with control group, the result is (WMD = −10.52, 95% CI: −14.74 to 4.76, P < .00001; I2 = 0%; P for heterogeneity = .86), (WMD = −1.48, 95% CI: −2.26 to −0.69, P = .0002; I2 = 35%; P for heterogeneity = .10), respectively. Thirteen studies concerned the satisfaction score of patients. The satisfaction score of patients in the PIEB group was significantly higher when compared with control group (WMD = 0.91, 95% CI: 0.42–1.39, P = .0003; I2 = 98%; P for heterogeneity < .00001). The Apgar score at 1, 5 minutes in PIEB group are significantly higher (WMD = 0.07, 95% CI: 0.02–0.13 P = .007; I2 = 55%; P for heterogeneity = .04), (WMD = −0.08, 95% CI: −0.12 to −0.05, P < .00001; I2 = 21%; P for heterogeneity = .27), respectively.
Conclusions:
PIEB is a good alternative for labor analgesia with better analgesic effect, maternal and infant outcome.
Keywords: analgesic consumption, labor analgesia, meta-analysis, programmed intermittent epidural bolus
1. Introduction
The patient-controlled epidural analgesia (PCEA) technique has been recently set up as a preferred mode of epidural drug delivery and used widely. Programmed intermittent epidural bolus (PIEB) is a new way of injecting anesthetics into the epidural space through an epidural catheter at fixed time intervals.[1,2] It can be used as a background administration with the PCEA technique.[3] Although the use of a continuous epidural infusion (CEI) is gaining popularity,[4] it is unknown if the PIEB could improve the analgesic function. Earlier studies have indicated that PIEB, as compared CEI, can increase maternal satisfaction score and decrease the consumption of epidural drugs.[5–8] This may be ascribed to a more extensive spread of epidural solution when delivered as a bolus rather than continuous infusion.[9]
In this meta-analysis, we sought to compare the efficacy of PIEB with that of CEI with respect to the maintenance of epidural analgesia during labor, duration of labor, mode of delivery, side effects, and satisfaction score.
2. Methods
Our systematic review was carried out according to the guidelines of the preferred reporting items for systematic reviews and meta-analyses.[10] We prospectively registered our system review at PROSPERO (Registration number: CRD42016038820). The proposed study utilizes published data, as such, there is no need for ethical approval. We followed the methods of several articles.[11–14]
3. Data sources and search strategy
The PubMed, Cochrane Library databases, and Embase were searched from inception to July 2021 for relevant studies comparing PIEB with CEI for labor analgesia. The following search terms were used: “programmed intermittent epidural bolus”, “regular intermittent bolus”, “automated intermittent epidural bolus”, “automated intermittent”, “intermittent epidural”, “continuous epidural infusion”, “continuous infusion”, “epidural bolus”, and “labor analgesia”. A manual search of reference sections of included trials, published meta-analyses, and pertinent review articles was conducted to identify additional articles. If duplicated data were found, only the most recent, the largest or the most complete studies were included.
4. Data selection
Original studies included were based on patient, intervention, comparison, outcome, and study design as follows: P: American Society of Anesthesiologists grade I/ II pregnant women undergoing delivery; I and C: PIEB and CEI; O: the rate of side effect (pruritus, hypotension, shivering, nausea, vomiting), the mode of delivery, pain score, total dose of ropivacaine, satisfaction score, duration of labor, duration of second stage of labor, duration of analgesia, number of oxytocin used, fetal birthweight, Apgar score; and S: only randomized controlled trials (RCTs) were included. Only English language studies were selected.
5. Data extraction
Patient characteristics (number of patients, age, gender, type of anaesthesia) and trial design (intervention, control, follow-up time, and reported outcomes) were accounted for. If the data mentioned above were unavailable in the article, the corresponding authors were contacted for missing information. All of the data were independently extracted using a standard data collection form by 2 reviewers (XX Wang and ZQ Xin), and then the collected data were checked and entered into Review Manager analysis software (RevMan) Version 5.3. All discrepancies were checked, and a consensus was achieved by discussion with a third author (XL Zhang). A record of reasons for excluding studies was kept.
6. Assessment of study quality and risk of bias
A critical evaluation of the quality of the included studies was conducted by 2 reviewers (XX Wang and ZQ Xin) using a 5-point Jadad scale.[15] The main categories of the Jadad scale consist of the following 5 areas of evaluation: “was the study described as randomized?”, “was the method used to generate the sequence of randomization described and appropriate (random numbers, computer-generated, etc.)?”, “was the study described as double-blind?”, “was the method of double-blinding described and appropriate (identical placebo, active placebo, dummy, etc.)?”, and “was there a description of withdrawals and drop-outs?”. Studies with a score of 4 to 5 were included in this analysis.
Two reviewers (XX Wang and XL Zhang) independently evaluated the risk of bias according to the recommendations from the Cochrane Collaboration.[16] The principal categories consisted of random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, and selective reporting and other bias. Each domain was measured as “high risk”, “low risk”, or “unclear risk”. A designation of “low risk” was for items with sufficient and correct information and a designation of “high risk” was for incorrectly reported items. If the information of an item was insufficient or unsanctioned, it was designated as “unclear risk”.
7. Statistical analysis
The weighted mean difference (WMD) or odds ratio (OR) with 95% confidence intervals (CIs) was used as a common measure of the effect between the 2 groups. The meta-analysis was conducted using Review Manager, Version 5.3 (The Cochrane Collaboration, Software Update, Oxford, UK). Statistical heterogeneity across studies was usually investigated using the I2 statistic. When I2 values of less than 50% were determined, heterogeneity could be accepted, and the fixed-effects model was adopted. Otherwise, the randomized-effects model was adopted. Subgroup analysis was also carried out to investigate potential sources of between-study heterogeneity. Publication bias was evaluated in funnel plot. A P value of <.05 was considered statistically significant.
8. Results
8.1. Identification of eligible studies
In total, 696 potentially relevant abstracts were identified. After duplicates were removed, 694 unique abstracts remained. After examining the abstracts, 43 publications seemed to meet the inclusion criteria. Of these, 24 were excluded for the following reasons: unpublished studies,[17–20] retrospective study,[21–23] cohort study,[24,25] review,[26] letter,[27] no available data on the outcome of interest in,[28–37] and nonEnglish language.[38] Finally, the remaining 25 studies[1,5–7,39–59] with existing data met our selection criteria and were enrolled in the systematic review. A flow diagram of the search strategy and study selection is illustrated in Figure 1.
Figure 1.
Flow diagram of the search strategy and study selection.
9. Study characteristics
The characteristics of all the included studies are presented in Table 1. All subjects were pregnant patients undergoing delivery. The quality of the included studies was evaluated by a Jadad score. The highest Jadad score of the included studies was 4, and the mean score was 3.3 (range 2–4). Fifteen studies have high score (≥4). These studies were published between 2004 and 2020. The sample size of the included studies ranged from 40 to 2865. All were RCTs and a quality assessment of the 25 RCTs is presented. The baseline characteristics of patients were reported in all trials, and all trials mentioned the method of random selection (Fig. 2).
Table 1.
Characteristic of included studies.
| Author | No. of patients (PIEB/CEI) | Country | PIEB group | CEI group | Outcomes | Jadad score |
| A. Ojo 2020 | 61/59 | USA | 6-mL programmed intermittent epidural boluses every 45 min | Continuous epidural infusion at 8 mL/h | Patient-controlled epidural analgesia consumption per hour, a need for physician interventions, patterns of patient-controlled epidural analgesia use, motor blockade, number of patients who developed hypotension, pain scores, duration of second stage of labor, mode of delivery, and maternal satisfaction | 4 |
| Capogna 2011 | 75/70 | Italy | Levobupivacaine 0.0625% with sufentanil 0.5 g/mL, after an initial epidural loading dose of 20 mL, 10 mL every hour beginning 60 min after the initial dose | Levobupivacaine 0.0625% with sufentanil 0.5 g/mL, 10 mL/h, beginning immediately after the initial dose | Total dose of levobupivacaine/total dose of sufentanil/patients requiring PCEA boluses/PCEA boluses for each patient/motor block occurred at least once/motor block occurred/instrumental delivery/cesarean delivery | 4 |
| Chua 2004 | 21/21 | Singapore | 0.1% ropivacaine and fentanyl 2 μg/mL, 5 mL boluses were given hourly, with the first bolus 30 min postinduction | 0.1% ropivacaine and fentanyl 2 μg/mL, at the rate of 5 mL/h was initiated in the minute after CSE | Duration of analgesia/need for supplemental analgesia/hourly consumption of epidural bupivacaine + fentanyl solution/sensory block/motor block | 4 |
| Fan 2019 | 1454/1411 | China | Hourly PIEB dose of 10 mL was given starting 75 min post the loading dose | Maintained at a constant speed of 10 mL/h for CEI group | Baseline demographic characteristics of epidural labor analgesia patients; incidence of maternal fever; visual analog scale pain scores over time; epidural sensory levels over time; epidural, obstetric, and neonatal outcomes | 4 |
| Fang 2016 | 100/100 | China | In the PIEB group, the background infusion dose was 8mL/h, the administration rate was 6mL/min, and the dose was once an hour | The CEI group was continuously administered at a rate of 8 mL/h | Baseline characteristics; pain VAS Score at various time points; the duration and interval of uterine contractions, fetal heart rate, labor analgesia time, delivery method, and ratio of oxytocin use in the 2 groups | 2 |
| Feng 2014 | 66/66 | China | A bolus dose (10 mL of 0.08% ropivacaine + 0.4 μg/mL sufentanil) was manually administrated once an hour | A bolus dose (10 mL of 0.08% ropivacaine + 0.4 μg/mL sufentanil) was manually administrated at a constant rate of 10 mL/h | Cervical dilatation/sensory block levels/apgar score/number of epidural boluses/consumption of ropivacaine, sufentanil/neonatal weight/instrumental delivery/duration of analgesia, labor/artificial rupture of membranes/rupture of the membranes to delivery/number of vaginal examinations | 4 |
| Fettes 2006 | 20/20 | UK | An infusion of ropivacaine 2 mg/mL with fentanyl 2 mg/mL at hourly boluses of 10 mL | An infusion of ropivacaine 2 mg/mL with fentanyl 2 mg/mL at 10 mL/h | Caesarean section/first stage of labour second stage of labour/duration of epidural/ VAS pre-epidural/ropivacaine dose/epidural bolus given/2 epidural boluses or more/cervical dilatation | 3 |
| Fidkowski 2019 | 41/34 | USA | Epidural analgesia regimen of bupivacaine 0.125% with fentanyl 2 μg/mL at PIEB 10 mL every 60 min | Epidural analgesia regimen of bupivacaine 0.125% with fentanyl 2 μg/mL at 10 mL/h continuous infusion | Average pain scores; demographic data; vaginal delivery; cesarean delivery; duration epidural analgesia; maximum bromage score; lowest dermatomal sensory level; physician administered epidural bolus; patient satisfaction | 4 |
| Haidl 2020 | 75/75 | Norway | PIEB + PCEA (5 mL bolus every hour, 5 mL PCEA bolus lockout 20 min) using a solution of bupivacaine 1mg/mL, fentanyl 2 mcg/mL, and adrenaline 2 mcg/mL | CEI + PCEA (5 mL/h, 5 mL PCEA bolus, lockout 20 min) using a solution of bupivacaine 1mg/mL, fentanyl 2 mcg/mL, and adrenaline 2 mcg/mL | Baseline characteristics; total epidural solution consumption; No. of completed PCEA boluses;/rejected PCEA boluses/participants needing any further physician intervention/physician administered manual boluses of the study epidural solution/rescue bupivacaine boluses/supplemental spinal injections/unilateral epidural effect/new epidural catheter; time from epidural placement to birth (min); modified bromage score at 60 min; modified bromage score at delivery; mode of delivery; hypotension; nausea; pruritus; satisfaction with treatment | 4 |
| Ji 2016 | 25/25 | China | Epidural boluses of LA 8 mL each 60 min | Maintained at a constant speed of 8 mL/h | Baseline characteristics, gestational age, uterine orifice size before analgesia, and artificial membrane rupture rate between the 2 groups; comparison of the time for maternal block level to reach T10, PCEA usage and breakthrough pain; pain VAS score at various time points; maternal delivery and newborn Apgar score | 2 |
| Leo 2010 | 31/31 | Singapore | 0.1% ropivacaine + fentanyl 2 μg/mL, automated mandatory boluses of 5 mL/h | 0.1% ropivacaine + fentanyl 2 μg/mL, basal continuous infusion of 5 mL/h | Hourly consumption of ropivacaine/sensory block/pain scores/duration of labor, 2nd stage/mode of delivery/apgar scores/satisfaction/shivering/pruritus/nausea/vomiting/breakthrough pain/cervical dilation/VAS/oxytocin infusion/sensory level/time to 1st breakthrough pain | 4 |
| Lim 2005 | 30/30 | Singapore | Levobupivacaine 0.1% with fentanyl 2 μg/mL, 5 mL epidural boluses every half hour | Levobupivacaine 0.1% with fentanyl 2 μg/mL at a rate of 10 mL/h | Breakthrough pain/ pruritus/nausea/vomiting/shivering/ hypotension/satisfaction score/pain scores/mode of delivery/Apgar scores at 5 min/cervical dilatation/use of oxytocin/motor block | 4 |
| Lim 2010 | 25/26 | Singapore | 2.5 mL automated intermittent epidural boluses of ropivacaine 0.1% plus fentanyl 2 μg/mL delivered over a 2-min period every 15 min | Continuous epidural infusion of ropivacaine 0.1% plus fentanyl 2 μg/mL at 10 mL/h | Breakthrough pain/pruritus/nausea/vomiting/shivering/hypotension/foetal bradycardia, birth weight/urinary catheter inserted/mode of delivery/duration of labour, second stage of labour/total ropivacaine dose/Apgar score/satisfaction score/mean time to first breakthrough pain/pain score/sensory level/oxytocin being administered/crvical dilation | 3 |
| Lin 2016 | 102/98 | China | 0.1% ropivacaine mixed with sufentanil 0.3 μg/mL; an hourly IEB of 5 mL and mixed with a PCEA bolus of 5 mL | 0.1% ropivacaine mixed with sufentanil 0.3 μg/mL; CEI at a rate of 5 mL/h and mixed with a PCEA bolus of 5 mL | Demographic properties/cervical dilation/and VAS scores/delivery mode/duration of first stage of labor/duration of second stage of labor/Apgar score at 1 min, Apgar score at 5 min, dosage consumption of ropivacaine, rescue medication dose by PCEA, Time to reach maximum block height (T10) | 4 |
| Morau 2019 | 124/125 | France | Received an hourly bolus of 8 mL (injection rate of 250 mL/h) beginning 60 min after the loading dose. | A continuous infusion rate of 8 mL/h was immediately commenced in the PCEA group | Maternal characteristics; reasons for instrumental vaginal delivery; primary outcome and detailed analysis of events included in the composite endpoint; presence of a motor block; data recorded during labour | 3 |
| Nunes 2016 | 33/60 | Portugal | Deliver 10 mL of ropivacaine 0.15% plus sufentanil 0.2g/mL solution every hour beginning 60 min after the administration of the initial epidural loading dose | Deliver the ropivacaine 0.15% plus sufentanil 0.2g/mL solution at a rate of 5 mL/h, with PCEA boluses of 5 mL with a lockout interval of 20 min, and a per hour maximum volume of 15 mL | Subject and labor characteristics, Apgar scores at 1st and 5th minutes | 3 |
| Riazanova 2019 | 42/38 | Russia | Programmed intermittent epidural boluses of LA 8.0 mL each 30 min with patient controlled epidural analgesia, LA bolus 8.0 mL, lockout interval 30 min | Patient-controlled epidural analgesia was conducted (8.0 mL LA lockoutinterval 30 min) with continuous background infusion of ropivacaine hydrochloride 0.08% with an infusion rate of 8.0 mL/h | General characteristics of examined patients; indices of blood pressure (BP) and heart rate (HR) during labour pain relief; assessment of pain level using VAS and assessment of motor block using the Bromage scale in different study stages; Apgar score; duration of delivery with epidural analgesia and local anaesthetic consumption | 3 |
| Rodríguez–Campoó 2018 | 100/95 | Spain | A 2 mL/h continuous infusion plus a 7 mL/30 min PIEB bolus | A continuous infusion of 5 mL/h plus 6 mL/20 min PCEA | Total levobupivacaine dose; pain control; subject satisfaction; type of delivery; vaginal tears; episiotomy | 4 |
| Sia 2007 | 21/21 | Singapore | 0.1% ropivacaine + fentanyl 2 μg/mL, lockout 10 min, automated mandatory boluses of 5 mL/h | 0.1% ropivacaine + fentanyl 2 μg/mL, lockout 10 min, basal continuous infusion of 5 mL/h | Epidural ropivacaine consumed per hour/number of self-bolus/time to the first self-bolus/number of breakthrough pain requiring an anesthesiologist's intervention/nausea/vomiting/ pruritus/duration of labor/duration of second stage/mode of delivery/fetal birthweight/Apgar score/satisfaction score/use of oxytocin at time/cervical dilatation/ pain score/lowest systolic blood/maximum dermatomal block to cold/lower limb motor block | 4 |
| Sia 2013 | 51/51 | Singapore | 0.1% ropivacaine + fentanyl 2 μg/mL, automated boluses of 5 mL in addition to the patient-controlled boluses | 0.1% ropivacaine + fentanyl 2 μg/mL, PCEA with basal infusion 5 mL/h | Baseline characteristics/breakthrough pain/side-effects/obstetric and neonatal outcomes | 4 |
| Song 2020 | 38/40 | China | The pump was programmed to administer the first bolus of 8 mL 1 h after initiation and every hour afterward | The epidural pump was programmed to deliver at a constant rate of 8 mL/h | Demographic and baseline characteristics; analgesia characteristics and labor outcomes; pain VAS score at various time points | 4 |
| Wang 2016 | 100/100 | China | Programmed intermittent epidural boluses of LA 10 mL each 30 min with patient controlled epidural analgesia, lockout interval 30 min | Maintained at a constant speed of 10 mL/h for CEI group. | Baseline characteristics; pain VAS score at various time points; thoracic sensory block level; PCA frequency and total medication; maternal delivery, blood loss, satisfaction scores and newborn Apgar scores; hypotension, nausea, vomiting, itching and others. | 2 |
| Wang 2017 | 62/62 | China | 10 mL/60 min, pulse dosing starts 60 min after the first dose is injected | Maintained at a constant speed of 10 mL/h for CEI group. | Baseline characteristics; pain VAS score at various time points; time to add medication for the first time, the number of PCEA compressions and the total amount of epidural analgesia used by the parturient; satisfaction and newborn situation; the duration and interval of uterine contractions, labor analgesia time, delivery method, and ratio of oxytocin use in the 2 groups | 2 |
| Wong 2006 | 63/63 | USA | 6 mL bolus every 30 min beginning 45 min after the intrathecal injection | 12 mL/h infusion beginning 15 min the after the intrathecal injection | Labor pain/epidural bupivacaine dose/epidural fentanyl dose/time to first PCEA request/PCEA bupivacaine dose/manual bolus (number of subjects, number per subject)/manual bupivacaine dose (mg/h)/total bupivacaine dose | 4 |
| Zhao 2013 | 29/28 | China | Delevered a 3 mL bolus at a rate of 60mL/h per 30 min | maintained at a constant speed of 6 mL/h for CEI group | Baseline characteristics; maternal labor, mode of delivery; oxytocin use, postpartum hemorrhage, and newborn Apgar score | 3 |
CEI = continuous epidural infusion, PCEA = patient-controlled epidural analgesia, PIEB = programmed intermittent epidural bolus, VAS = visual analogue scale.
Figure 2.
A. Graph of review authors’ assessments of risk of bias for each Cochrane item. B. Summary of review authors’ assessments of risk of bias for each Cochrane item and each included study.
9.1. Meta-analyses of outcomes
9.1.1. Basic situation comparison
We conducted a systematic review of the basic conditions of the 2 groups of patients before the implementation of labor analgesia. These basic conditions include the patient's age, height, weight, BMI, basal blood pressure and heart rate before analgesia, gestational age, usage of oxytocin, and cervical dilatation before analgesia. By meta-analysis, we found that the basic conditions of the 2 groups of patients before labor analgesia were not statistically significant (Table 2).
Table 2.
Basic conditions of the 2 groups.
| Variable | Number of studies | RR (95% CI) | I2 | Effects models | P value |
| Age | 18 | −0.16 (−0.38–0.07) | 37 | Fixed effects models | .18 |
| Height | 21 | −0.80 (−0.28–0.26) | 37 | Fixed effects models | .94 |
| Weight | 21 | −0.41 (−1.51–0.69) | 51 | Random effects models | .47 |
| BMI | 7 | 0.01 (−0.16–0.18) | 1 | Fixed effects models | .92 |
| Maternal systolic BP | 8 | 0.02 (−2.00–2.05) | 0 | Fixed effects models | .98 |
| Maternal diastolic BP | 4 | −0.94 (−2.84–0.96) | 0 | Fixed effects models | .33 |
| Maternal heart rate | 4 | −1.39 (−3.97–1.18) | 29 | Fixed effects models | .29 |
| Cervical dilation at initiation of analgesia | 20 | −0.03 (−0.10–0.04) | 55 | Random effects models | .43 |
| Oxytocin infusion | 5 | 1.34 (0.83–2.15) | 30 | Fixed effects models | .23 |
| Gestational age | 18 | 0.02 (−0.11–0.15) | 60 | Random effects models | .75 |
BMI = body mass index, BP = blood pressure, CI = confidence interval, RR = risk ratios.
9.1.2. The effect of analgesia between 2 groups
PIEB was associated with decreases in total consumption of ropivacaine (5 RCTs; WMD = −15.83, 95% CI: −19.06 to −12.60, P < .00001; I2 = 61%; P for heterogeneity = .04) (Fig. 3), total consumption of sufentanil (3 RCTs; WMD = −4.93, 95% CI: −6.87 to 2.98, P < .00001; I2 = 68%; P for heterogeneity = .05) (Fig. 4), numbers of patients who require PCEA bolus (6 RCTs; OR = 0.27, 95% CI: 0.14–0.51, P < .0001; I2 = 65%; P for heterogeneity = .01) (Fig. 5), the number of attempts (6 RCTs; WMD = −4.12, 95% CI: −7.21 to −1.04, P = .009; I2 = 100%; P for heterogeneity < .00001) (Fig. 6), rate of breakthrough pain (6 RCTs; OR = 0.47, 95% CI: 0.28–0.80, P = .005; I2 = 47%; P for heterogeneity = .09) (Fig. 7). Eight studies focus on the duration of analgesia. The aggregated results of these 8 studies suggest that PIEB was not associated with a significant increase in the duration of analgesia (WMD = −0.11, 95% CI: −23.48 to 23.25, P = .99; I2 = 91%; P for heterogeneity < .00001) (Fig. 8). Fifteen studies concerned the pain visual analogue scale (VAS) score at various time points. After by meta-analysis, we found that the pain VAS score at 30 minutes, 2 hours, 4 hours, and 5 hours in PIEB group was significantly lower when compered with control group, (WMD = −0.15, 95% CI: −0.26 to −0.04, P = .006; I2 = 0%; P for heterogeneity = .64), (WMD = −0.79, 95% CI: −1.32 to 0.25, P = .004; I2 = 97%; P for heterogeneity < .00001), (WMD = −1.00, 95% CI: −1.08 to −0.91, P < .00001; I2 = 0%; P for heterogeneity = .67), (WMD = −1.81, 95% CI: −3.23 to −0.39, P = .01; I2 = 98%; P for heterogeneity < .00001), respectively (Fig. 9).
Figure 3.
Meta-analysis of the net change in total consumption of ropivacaine. CI = confidence interval, PIEB = programmed intermittent epidural bolus, SD = standard deviation.
Figure 4.
Meta-analysis of the net change in total consumption of sufentanil. CI = confidence interval, PIEB = programmed intermittent epidural bolus, SD = standard deviation.
Figure 5.
Meta-analysis of the net change in rate of patients who need additional PCEA bolus. CI = confidence interval, PIEB = programmed intermittent epidural bolus.
Figure 6.
Meta-analysis of the net change on the number of attempts. CI = confidence interval, PIEB = programmed intermittent epidural bolus, SD = standard deviation.
Figure 7.
Meta-analysis of the net change on the rate of breakthrough pain. CI = confidence interval, PIEB = programmed intermittent epidural bolus.
Figure 8.
Meta-analysis of the net change on the duration of analgesia. CI = confidence interval, PIEB = programmed intermittent epidural bolus, SD = standard deviation.
Figure 9.
Meta-analysis of the net change on the pain VAS score at various time points. CI = confidence interval, PIEB = programmed intermittent epidural bolus, SD = standard deviation.
9.1.3. Labor in delivery
Nineteen studies discussed the mode of delivery between 2 groups. Fourteen studies with a total of 1574 patients reported the rate of normal delivery. There are no heterogeneity noted among the studies (I2 = 0%; P = .86), and a fixed-effects model was selected. The results suggest that the rate of normal delivery is significantly higher in PIEB group compared with control group (OR = 1.37, 95% CI: 1.08–1.75, P = .01). Seventeen studies compared the rate of instrumental vaginal delivery. The heterogeneity between 2 groups are accepted (I2 = 6%; P = .39), and a fixed-effects model was selected. The rate of instrumental vaginal delivery has no significant difference between 2 groups (OR = 0.83, 95% CI: 0.68–1.02, P = .07). Sixteen studies reported the rate of cesarean delivery. There are no heterogeneity noted among the studies (I2 = 0%; P = .97), and a fixed-effects model was selected. The rate of cesarean delivery has no significant difference between 2 groups (OR = 0.89, 95% CI: 0.65–1.20, P = .44) (Fig. 10). Seventeen studies discussed the time of labor, the heterogeneity between 2 groups are accepted and a fixed-effects model was selected. After comparison, the time of first and second stage of labor are significantly shorter in PIEB group compared with control group, the result is (WMD = −10.52, 95% CI: −14.74 to 4.76, P < .00001; I2 = 0%; P for heterogeneity = .86), (WMD = −1.48, 95% CI: −2.26 to −0.69, P = .0002; I2 = 35%; P for heterogeneity = .10), respectively (Fig. 11). Thirteen studies concerned the satisfaction score of patients. After by meta-analysis, we found that the satisfaction score of patients in the PIEB group was significantly higher when compared with control group (WMD = 0.91, 95% CI: 0.42–1.39, P = .0003; I2 = 98%; P for heterogeneity < .00001) (Fig. 12).
Figure 10.
Meta-analysis of the net change in rate of mode of delivery. CI = confidence interval, PIEB = programmed intermittent epidural bolus.
Figure 11.
Meta-analysis of the net change in time of labor. CI = confidence interval, PIEB = programmed intermittent epidural bolus, SD = standard deviation.
Figure 12.
Meta-analysis of the net change on the satisfaction score. CI = confidence interval, PIEB = programmed intermittent epidural bolus, SD = standard deviation.
9.1.4. Neonatal condition
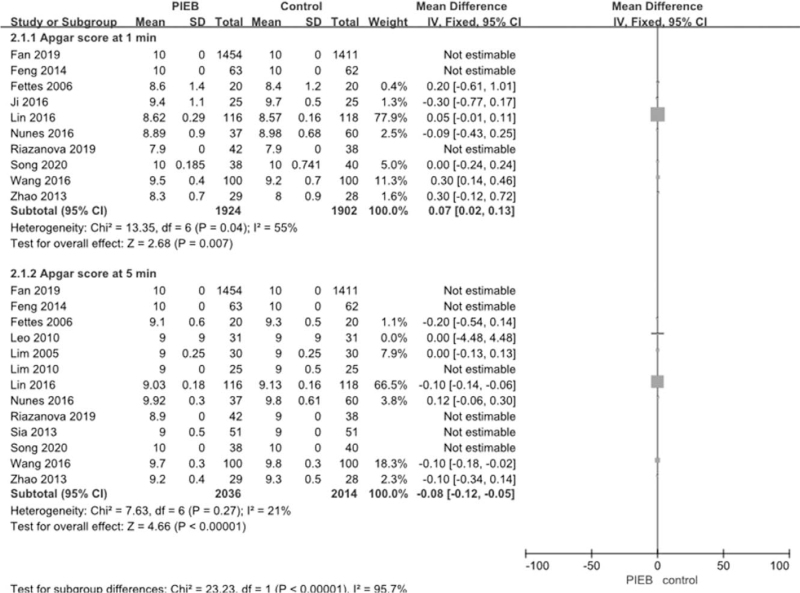
PIEB was not associated with increase the rate of bradycardia (4 RCTs; OR = 1, 95% CI: 0.30–3.39, P = 1; I2 = 0%; P for heterogeneity = .71) (Fig. 13), and the fetal heart rate between 2 groups was no significantly difference (4 RCTs; WMD = −0.61, 95% CI: −2.53 to 1.31, P = .53; I2 = 0%; P for heterogeneity = .69) (Fig. 14). Fifteen studies reported the Apgar score between 2 groups. When compared with the control group, Apgar score at 1, 5 minutes in PIEB group are significantly higher (WMD = 0.07, 95% CI: 0.02 to 0.12 P = .009; I2 = 49%; P for heterogeneity = .06), (WMD = −0.08, 95% CI: −0.12 to −0.05, P < .00001; I2 = 21%; P for heterogeneity = .27), respectively (Fig. 15)
Figure 13.
Meta-analysis of the net change on the rate of bradycardia. CI = confidence interval, PIEB = programmed intermittent epidural bolus.
Figure 14.
Meta-analysis of the net change on the fetal heart rate. CI = confidence interval, PIEB = programmed intermittent epidural bolus, SD = standard deviation.
Figure 15.
Meta-analysis of the net change on the Apgar score at 1, 5 minutes. CI = confidence interval, PIEB = programmed intermittent epidural bolus, SD = standard deviation.
10. Adverse events
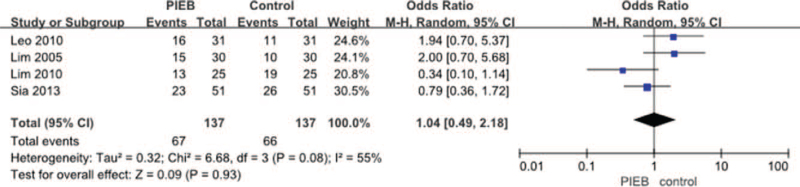
PIEB was not associated with decreases the rate of pruritus (10 RCTs; OR = 1.02, 95% CI: 0.71–1.45, P = .92; I2 = 0%; P for heterogeneity = .95) (Fig. 16), hypotension (9 RCTs; OR = 0.77, 95% CI: 0.33–1.77, P = .53; I2 = 0%; P for heterogeneity = .61) (Fig. 17), shivering (4 RCTs; OR = 1.04, 95% CI: 0.49–2.18, P = .93; I2 = 55%; P for heterogeneity = .08) (Fig. 18), nausea (10 RCTs; OR = 2.31, 95% CI: 0.95–5.59, P = .06; I2 = 0%; P for heterogeneity = .79) (Fig. 19), and vomiting (9 RCTs; OR = 2.08, 95% CI: 0.61–7.09, P = .24; I2 = 0%; P for heterogeneity = .38) (Fig. 20).
Figure 16.
Meta-analysis of the net change in rate of pruritus. CI = confidence interval, PIEB = programmed intermittent epidural bolus.
Figure 17.
Meta-analysis of the net change in rate of hypotension. CI = confidence interval, PIEB = programmed intermittent epidural bolus.
Figure 18.
Meta-analysis of the net change in rate of shivering. CI = confidence interval, PIEB = programmed intermittent epidural bolus.
Figure 19.
Meta-analysis of the net change in rate of nausea. CI = confidence interval, PIEB = programmed intermittent epidural bolus.
Figure 20.
Meta-analysis of the net change in rate of vomiting. CI = confidence interval, PIEB = programmed intermittent epidural bolus.
11. Discussion
The finding of our systematic review and meta-analysis study show that PIEB was associated with decreases in total consumption of ropivacaine, sufentanil, numbers of patients who require PCEA bolus, the number of attempts, and rate of breakthrough pain. At the same time, the time of the first and second stage of labor is significantly shorter and the satisfaction score of patients was significantly higher in the PIEB group compared with the control group. There have been similar systematic reviews before, but the included literature is less.[8] On this basis, this study included more studies for analysis (24 RCTs), thereby improving the credibility of the synthetic analysis data. Also, this study added VAS scores at different time points and neonatal Apgar scores for meta-analysis based on previous studies. In this way, the influence of PIEB on maternal and infant outcomes is more systematic and comprehensive. Our study found that the pain VAS score at 30 minutes, 2 hours, 4 hours, and 5 hours in the PIEB group was significantly lower when compared with the control group and the rate of normal delivery, Apgar score at 1, 5 minutes were significantly higher in PIEB group when compared with the control group. This study is the first to conduct a meta-analysis of the basic conditions of the included patients to further demonstrate the balance of the general conditions of the 2 groups of patients so that the outcome indicators are comparable. In this meta-analysis, eleven trials reported adverse effects and the incidence of pruritus, hypotension, shivering, nausea, and vomiting were similar in both PIEB and control groups.
Programmed intermittent epidural analgesia and continuous epidural analgesia are the 2 main technical methods of labor analgesia, and their different administration methods have different effects on the outcome of the mother and the baby. The speed at which the infusion bolus is delivered and the pressure generated in the epidural space also affects dispersion. Theoretically, intermittent boluses injected at higher pressure should add more widespread and uniform epidural solution dispersion.[59,60,61] Experimentally, the use of intermittent boluses had been found to result in a greater spread of infusate when compared with a continuous infusion, despite a similar rate of infusion.[8] In vitro studies,[62] it confirmed that when a constant rate of 10.5 mL/h is used for continuous administration, most of the drug solution flows out through the proximal hole of the spinal epidural catheter; and when a single injection is used, the epidural both the proximal and distal holes of the lumen catheter have liquid outflow, suggesting that when the same dose is taken, a single injection will have a wider range of drug block. It may also be that programmed epidural analgesia is better than continuous epidural analgesia.[63,64] In this study, we also found that, compared with continuous epidural analgesia, programmed epidural analgesia showed good analgesic effects. PIEB was associated with decreases in total consumption of ropivacaine, total consumption of sufentanil, numbers of patients who require PCEA bolus, the number of attempts, and rate of breakthrough pain. We also found that the pain VAS score at 30 minutes, 2 hours, 4 hours, and 5 hours in the PIEB group was significantly lower when compared with the control group.
The outcome of programmed intermittent epidural labor analgesia on the length of labor and delivery methods are not the same. Sia et al[53] performed a randomized controlled study comparing CEI administered as 5 mL/h with PIEB es administered as 5 mL every 60 minutes, with all groups using 0.1% ropivacaine with 2 μg/mL fentanyl. It is noted that the time to delivery in the PIEB group was significantly longer than the control group. At the same time, some studies have reached the opposite conclusion.[43,48,65] We conducted a meta-analysis on the time of labor and the mode of delivery. A systematic review and meta-analysis of the existing data found that the procedural epidural analgesia group had a significantly shorter time in the first and second stages of labor when compared with the continuous epidural analgesia group. It may be that the PIEB was more prone to an extensive nerve block, which makes the soft birth canal cervix looser and soft, and the fetal head descends more smoothly. The results of this study also confirmed the above view. The study found that the normal delivery rate of the PIEB group was higher than the control group. This result also shows that the PIEB mode has less effect on contractions than continuous epidural analgesia, and can effectively reduce pelvic floor muscle tension. The production process went smoothly and maternal satisfaction was also higher.
Observing the effects of different modes of labor analgesia on newborns is the first time to be reported in the form of systematic reviews. By observing the effects of different modes of labor analgesia on the Apgar score of newborns at 1, 5 minutes after birth, we can make a more comprehensive and objective analysis to explore the effects of different modes of labor analgesia on maternal and infant outcomes. According to the results of this study, compared with the continuous epidural analgesia group, the newborns in the programmed intermittent epidural analgesia group had significantly higher Apgar scores at 1 and 5 minutes after delivery, and the Apgar score level was closer to the normal level, which also reflected it less impact on newborns.
Perioperative adverse clinical side effects relevant to anesthesia in parturients were pruritus, hypotension, shivering, nausea, and vomiting, which affect the patients’ clinical prognosis. In our study, we observed that the adverse side effects in the PIEB group have no statistically different from the control group.
In our meta-analysis, the patients enrolled were less homogeneous. Fifteen studies had Jadad scores equal to 4 and were of high quality. The participants in all studies were well matched (eg, sex, age, American Society of Anesthesiologists grade, administration time, etc). However, results in this current meta-analysis should be interpreted with careful consideration given the limitations inherent in the design of the study. First, some of the major outcomes had small sample sizes, which might result in a small-study effect. Second, the kind and dosage applied in trials was different, which might have influenced outcomes. Third, this meta-analysis was based on studies published in English, which might have generated bias. Finally, we selected published studies, and many studies were not registered in clinical trial databases.
An interesting clue on the topic may be helpful for future research. In our meta-analysis, we found that the duration of analgesia in the 2 groups has no difference and the studies concerned the consumption of stupefacient was little, so attention should be paid to it. Further studies may also focus on the safety of different types of anesthesia. Moreover, in our study, anesthesia types were only compared when they were used in patients who labor, so other medical situations in which these types are used should also be studied.
12. Conclusions
In conclusion, PIEB is a good alternative for labor analgesia with better analgesic effect, maternal and infant outcome. However, some of the results in our meta-analysis should be interpreted carefully because of the clinical heterogeneity and insufficient data.
Author contributions
XXW and ZQX conceived the study, participated in the study design, collected the data, and drafted the manuscript. XXW, ZQX, and XLZ participated in the study design, collected the data, performed the statistical analysis and contributed to drafting the manuscript. HJG, ZXZ, JX, YLZ, SZY, and HYL helped to perform the statistical analysis and revised the manuscript critically to ensure all important intellectual content was present. All authors read and approved the final manuscript.
Conceptualization: Jing Xiao.
Data curation: Xian-xue Wang, Xiao-lan Zhang, Hua-jing Guo, Yun-lin Zhang.
Formal analysis: Shu-zhen Yuan.
Investigation: Zi-qin Xin.
Methodology: Zi-qin Xin.
Project administration: Zhao-xia Zhang.
Resources: Zi-qin Xin.
Software: Xian-xue Wang, Hua-jing Guo.
Supervision: Hai-yan Liu.
Validation: Jing Xiao.
Visualization: Hai-yan Liu.
Writing – original draft: Xian-xue Wang.
Writing – review & editing: Xian-xue Wang.
Footnotes
Abbreviations: CEI = continuous epidural infusion, CI = confidence interval, OR = odds ratios, PCEA = patient-controlled epidural analgesia, PIEB = programmed intermittent epidural bolus, RCTs = randomized controlled trials, VAS = visual analogue scale, WMD = weighted mean difference.
How to cite this article: Wang Xx, Zhang Xl, Zhang Zx, Xin Zq, Guo Hj, Liu Hy, Xiao J, Zhang Yl, Yuan Sz. Programmed intermittent epidural bolus in parturients: a meta-analysis of randomized controlled trials. Medicine. 2022;101:5(e28742).
XxW, XlZ, and ZxZ contributed equally.
Funding was received from Technology Research and Development Fund Project of Changde Science and Technology Bureau, Project Number: 2019S183.
The authors have no conflicts of interest to disclose.
The datasets generated during and/or analyzed during the current study are publicly available.
References
- [1].Song YJ, Du WJ, Zhou SQ, et al. Effect of dural puncture epidural technique combined with programmed intermittent epidural bolus on labor analgesia onset and maintenance: a randomized controlled trial. Anesth Analg 2021;132:971–8. [DOI] [PubMed] [Google Scholar]
- [2].Kim YJ, Lee DK, Kwon HJ, et al. Programmed intermittent epidural bolus versus continuous epidural infusion in major upper abdominal surgery: a retrospective comparative study. J Clin Med 2021;10:5382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Wang LY, Wu ZH, Hu LJ, et al. Programmed intermittent epidural bolus for post-cesarean delivery analgesia: a randomized controlled double-blind trial. J Anesth 2021;doi: 10.1007/s00540-021-03002-x. [DOI] [PubMed] [Google Scholar]
- [4].Gaiser RR, Lewin SB, Cheek TG, et al. Effects of immediately initiating an epidural infusion in the combined spinal and epidural technique in nulliparous parturients. Reg Anesth Pain Med 2000;25:223–7. [DOI] [PubMed] [Google Scholar]
- [5].Chua SM, Sia AT. Automated intermittent epidural boluses improve analgesia induced by intrathecal fentanyl during labour. Can J Anaesth 2004;51:581–5. [DOI] [PubMed] [Google Scholar]
- [6].Lim Y, Sia AT, Ocampo C. Automated regular boluses for epidural analgesia: a comparison with continuous infusion. Int J Obstet Anesth 2005;14:305–9. [DOI] [PubMed] [Google Scholar]
- [7].Fettes PD, Moore CS, Whitesides JB, et al. Intermittent vs continuous administration of epidural ropivacaine with fentanyl for analgesia during labour. Br J Anaesth 2006;97:359–64. [DOI] [PubMed] [Google Scholar]
- [8].Xu JQ, Zhou J, Xiao HR, et al. A systematic review and meta analysis comparing programmed intermittent bolus and continuous infusion as the background infusion for parturient-controlled epidural analgesia. Sci Rep 2019;9:2583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Kaynar AM, Shankar KB. Epidural infusions: continuous or bolus? [letter]. Anesth Analg 1999;89:534. [DOI] [PubMed] [Google Scholar]
- [10].Moher D, Liberati A, Tetzlaff J, Altman DG. PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 2010;8:336–41. [DOI] [PubMed] [Google Scholar]
- [11].Zhou AG, Wang XX, Pan DB, et al. Preoperative antihypertensive medication in relation to postoperative atrial fibrillation in patients undergoing cardiac surgery: a meta-analysis. Biomed Res Int 2017;2017:1203538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Chiu YH, Chang KV, Chen IJ, et al. Utility of sonoelastography for the evaluation of rotator cuff tendon and pertinent disorders: a systematic review and meta-analysis. Eur Radiol 2020;30:6663–72. [DOI] [PubMed] [Google Scholar]
- [13].Chiu YH, Chang KV, Wu WT, et al. Comparative effectiveness of injection therapies for hemiplegic shoulder pain in stroke: a systematic review and network meta-analysis. Pharmaceuticals (Basel) 2021;14:788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Chen KC, Lee TM, Wu WT, et al. Assessment of tongue strength in sarcopenia and sarcopenic dysphagia: a systematic review and meta-analysis. Front Nutr 2021;8:684840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 1996;17:01–12. [DOI] [PubMed] [Google Scholar]
- [16].Higgins JP, Altman DG, Gøtzsche PC, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ 2011;343:d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Ferreira E, Veiga M, Freitas J, Nunes S, et al. Labor analgesia: analyzing outcomes: a randomized controlled trial. 33rd Annual European Society of Regional Anaesthesia and Pain Therapy. Seville, Spain; 2014: pp. e196. [Google Scholar]
- [18].Nunes J, Nunes S, Veiga M, et al. Programmed intermittent boluses: are we improving epidural labour analgesia? European Anaesthesiology Congress. Stockholm, Sweden; 2014: 182–3. [Google Scholar]
- [19].Mavridou I, Husain T, Fernando R, et al. An evaluation of a programmed intermittent epidural bolus technique for labour analgesia. Annual Meeting of the Obstetric Anaesthetists’ Association. Bournemouth, United Kingdom; 2013: pp. S12. [Google Scholar]
- [20].Stirparo S, Camorcia M, Farcomeni A, et al. Maternal motor block during the second stage of labor and labor outcome: a comparison between programmed intermittent epidural bolus (PIEB) and continuous epidural infusion (CEI) analgesia. European Anaesthesiology Congress. Amsterdam, Netherlands; 2011: pp. 3. [Google Scholar]
- [21].Mckenzie CP, Cobb B, Riley ET, et al. Programmed intermittent epidural boluses for maintenance of labor analgesia: an impact study. Int J Obstet Anesth 2016;26:32–8. [DOI] [PubMed] [Google Scholar]
- [22].Rodriguez Gonzalez IP, Espinosa Dominguez E, Quesada Garcia C, et al. Comparison between different epidural analgesia modalities for labor. Rev Esp Anestesiol Reanim 2019;66:417–24. [DOI] [PubMed] [Google Scholar]
- [23].Tien M, Allen TK, Mauritz A, et al. A retrospective comparison of programmed intermittent epidural bolus with continuous epidural infusion for maintenance of labor analgesia. Curr Med Res Opin 2016;32:1435–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Delgado C, Ciliberto C, Bollag L, et al. Continuous epidural infusion versus programmed intermittent epidural bolus for labor analgesia: optimal configuration of parameters to reduce physician-administered top-ups. Curr Med Res Opin 2018;34:649–56. [DOI] [PubMed] [Google Scholar]
- [25].Bullingham A, Liang S, Edmonds E, et al. Continuous epidural infusion vs programmed intermittent epidural bolus for labour analgesia: a prospective, controlled, before-and-after cohort study of labour outcomes. Br J Anaesth 2018;121:432–7. [DOI] [PubMed] [Google Scholar]
- [26].Halpern SH, Carvallo B. Patient-controlled epidural analgesia for labor. Anesth Analg 2009;108:921–8. [DOI] [PubMed] [Google Scholar]
- [27].Kang S, Jeon S, Choe JH, et al. Comparison of analgesic effects of programmed intermittent epidural bolus and continuous epidural infusion after total knee arthroplasty. Korean J Anesthesiol 2013;65: (Suppl 6): S130–1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Leone Roberti Maggiore U, Silanos R, Carlevaro S, et al. Programmed intermittent epidural bolus versus continuous epidural infusion for pain relief during termination of pregnancy: a prospective, double-blind, randomized trial. Int J Obstet Anesth 2016;25:37–44. [DOI] [PubMed] [Google Scholar]
- [29].Vallejo MC, Ramesh V, Phelps AL, et al. Epidural labor analgesia: continuous infusion versus patient-controlled epidural analgesia with background infusion versus without a background infusion. J Pain 2007;8:970–5. [DOI] [PubMed] [Google Scholar]
- [30].Wong CA, McCarthy RJ, Hewlett B. The effect of manipulation of the programmed intermittent bolus time interval and injection volume on total drug use for labor epidural analgesia: a randomized controlled trial. Anesth Analg 2011;112:904–11. [DOI] [PubMed] [Google Scholar]
- [31].Wiesmann T, Hoff L, Prien L, et al. Programmed intermittent epidural bolus versus continuous epidural infusion for postoperative analgesia after major abdominal and gynecological cancer surgery: a randomized, triple-blinded clinical trial. BMC Anesthesiol 2018;18:154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Diez-Picazo LD, Guasch E, Brogly N, et al. Is breakthrough pain better managed by adding programmed intermittent epidural bolus to a background infusion during labor epidural analgesia? A randomized controlled trial. Minerva Anestesiol 2019;85:1097–104. [DOI] [PubMed] [Google Scholar]
- [33].Satomi S, Kakuta N, Murakami C, et al. The efficacy of programmed intermittent epidural bolus for postoperative analgesia after open gynecological surgery: a randomized double-blinded study. Biomed Res Int 2018;6297247.doi: 10.1155/2018/6297247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Kanczuk ME, Barrett NM, Arzola C, et al. Programmed intermittent epidural bolus for labor analgesia during first stage of labor: a biased-coin up-and-down sequential allocation trial to determine the optimum interval time between boluses of a fixed volume of 10 mL of bupivacaine 0.0625% with fentanyl 2 μg/mL. Anesth analg 2017;124:537–41. [DOI] [PubMed] [Google Scholar]
- [35].Bittencourt R, Arzola C, Zakus P, et al. A biased coin up-and-down sequential allocation trial to determine the optimum programmed intermittent epidural bolus time interval between 5 mL boluses of bupivacaine 0.125% with fentanyl 2 μg·mL−1. Can J Anaesth 2019;66:1075–81. [DOI] [PubMed] [Google Scholar]
- [36].Su PP, Peniche A, Clelland E, et al. Comparison of programmed intermittent epidural bolus and continuous epidural infusion for post-operative analgesia after major abdominal surgery: a randomized controlled trial. J Clin Anesth 2020;64:109850. [DOI] [PubMed] [Google Scholar]
- [37].Roofthooft E, Barbe A, Schildermans J, et al. Programmed intermittent epidural bolus vs. patient-controlled epidural analgesia for maintenance of labour analgesia: a two-centre, double-blind, randomised study. Anaesthesia 2020;75:1635–42. [DOI] [PubMed] [Google Scholar]
- [38].De Eccher L, Martino C, Bacchilega I, et al. Epidural analgesia during labor: intermittent bolus or patients controlled administrations? Minerva Anestesiol 2002;68:83–7. [PubMed] [Google Scholar]
- [39].Ojo OA, E.Mehdiratta J, Gamez BH, et al. Comparison of programmed intermittent epidural boluses with continuous epidural infusion for the maintenance of labor analgesia: a randomized, controlled, double-blind study. Anesth Analg 2020;130:426–35. [DOI] [PubMed] [Google Scholar]
- [40].Capogna G, Camorcia M, Stirparo S, et al. Programmed intermittent epidural bolus versus continuous epidural infusion for labor analgesia: the efects on maternal motor function and labor outcome. A randomized double-blind study in nulliparous women. Anesth Analg 2011;113:826–31. [DOI] [PubMed] [Google Scholar]
- [41].Fan Y, Hou W, Feng S, et al. Programmed intermittent epidural bolus decreases the incidence of intra-partum fever for labor analgesia in primiparous women: a randomized controlled study. Arch Gynecol Obstet 2019;300:1551–7. [DOI] [PubMed] [Google Scholar]
- [42].Fang XD, Xie L, Chen XX. Clinical efficacy of programmed intermittent epidural bolus and continuous epidural infusion for labor analgesia. J Clin Anesthesiol 2016;32:757–60. [Google Scholar]
- [43].Feng SW, Xu SQ, Ma L, et al. Regular intermittent bolus provides similar incidence of maternal fever compared with continuous infusion during epidural labor analgesia. Saudi Med J 2014;35:1237–42. [PMC free article] [PubMed] [Google Scholar]
- [44].Fidkowski CW, Shah S, Alsaden MR. Programmed intermittent epidural bolus as compared to continuous epidural infusion for the maintenance of labor analgesia: a prospective randomized single-blinded controlled trial. Korean J Anesthesiol 2019;72:472–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Haidl F, Rosseland LA, Rørvik AM, et al. Programmed intermittent boluses vs continuous epidural infusion in labor using an adrenaline containing solution: a randomized trial. Acta Anaesthesiol Scand 2020;64:1505–12. [DOI] [PubMed] [Google Scholar]
- [46].Ji J, Xu ZD, Jin C, et al. A randomized comparison between intermittent bolus and continuous infusion in combination with patient-controlled epidural analgesia in labor. Shanghai Med J 2016;39:461–5. [Google Scholar]
- [47].Leo S, Ocampo CE, Lim Y, et al. A randomized comparison of automated intermittent mandatory boluses with a basal infusion in combination with patient-controlled epidural analgesia for labor and delivery. Int J Obstet Anesth 2010;19:357–64. [DOI] [PubMed] [Google Scholar]
- [48].Lim Y, Chakravarty S, Ocampo CE, et al. Comparison of automated intermittent low volume bolus with continuous infusion for labour epidural analgesia. Anaesth Intensive Care 2010;38:894–9. [DOI] [PubMed] [Google Scholar]
- [49].Lin Y, Li Q, Liu JL, et al. Comparison of continuous epidural infusion and programmed intermittent epidural bolus in labor analgesia. Ther Clin Risk Manag 2016;12:1107–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Morau E, Jaillet M, Storme B, et al. Does programmed intermittent epidural bolus improve childbirth conditions of nulliparous women compared with patient-controlled epidural analgesia?: A multicentre, randomised, controlled, triple-blind study. Eur J Anaesthesiol 2019;36:755–62. [DOI] [PubMed] [Google Scholar]
- [51].Nunes J, Nunes S, Veiga M, et al. A prospective, randomized, blinded-endpoint, controlled study – continuous epidural infusion versus programmed intermittent epidural bolus in labor analgesia. Rev Bras Anestesiol 2016;66:439–44. [DOI] [PubMed] [Google Scholar]
- [52].Riazanova OV, Alexandrovich YS, Guseva YV, et al. A randomized comparison of low dose ropivacaine programmed intermittent epidural bolus with continuous epidural infusion for labour analgesia. Rom J Anaesth Intensive Care 2019;26:25–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Rodríguez-Campoó MB, Curto A, González M, et al. Patient intermittent epidural boluses (PIEB) plus very low continuous epidural infusion (CEI) versus patient-controlled epidural analgesia (PCEA) plus continuous epidural infusion (CEI) in primiparous labour: a randomized trial. J Clin Monit Comput 2019;33:879–85. [DOI] [PubMed] [Google Scholar]
- [54].Sia AT, Lim Y, Ocampo C. A comparison of a basal infusion with automated mandatory boluses in parturient-controlled epidural analgesia during labor. Anesth Analg 2007;104:673–8. [DOI] [PubMed] [Google Scholar]
- [55].Sia AT, Leo S, Ocampo CE. A randomised comparison of variable-frequency automated mandatory boluses with a basal infusion for patient-controlled epidural analgesia during labour and delivery. Anaesthesia 2013;68:267–75. [DOI] [PubMed] [Google Scholar]
- [56].Wang ZH, Xu SQ, Feng SW, et al. Efficacy of programmed intermittent epidural bolus for labor analgesia in parturients and the effect on neonates. Chin J Anesthesiol 2016;36:1134–7. [Google Scholar]
- [57].Wang ZH, Feng SW, Xu SQ, et al. Comparison of programmed intermittent epidural bolus with continuous epidural infusion at different time intervals for epidural labor analgesia. J Clin Anesthesiol 2017;33:755–9. [Google Scholar]
- [58].Wong CA, Ratliff JT, Sullivan JT, et al. A randomized comparison of programmed intermittent epidural bolus with continuous epidural infusion for labor analgesia. Anesth Analg 2006;102:904–9. [DOI] [PubMed] [Google Scholar]
- [59].Zhao JR, Wu J, Li SH, et al. Comparison of automated mandatory bolus and continuous epidural infusion in parturient controlled epidural analgesia during labor. Shanghai Med J 2013;36:504–6. [Google Scholar]
- [60].Carvalho B, George RB, Cobb B, et al. Implementation of programmed intermittent epidural bolus for the maintenance of labor analgesia. Anesth Analg 2016;123:965–71. [DOI] [PubMed] [Google Scholar]
- [61].Klumpner TT, Lange EM, Ahmed HS, et al. An in vitro evaluation of the pressure generated during programmed intermittent epidural bolus injection at varying infusion delivery speeds. J Clin Anesth 2016;34:632–7. [DOI] [PubMed] [Google Scholar]
- [62].Kaynar AM, Shankar KB. Epidural infusion: continuous or bolus? Anesth Analg 1999;89:534. [DOI] [PubMed] [Google Scholar]
- [63].Chen L, Wu Y, Cai Y, et al. Comparison of programmed intermittent bolus infusion and continuous infusion for postoperative patient-controlled analgesia with thoracic paravertebral block catheter: a randomized, double-blind, controlled trial. Reg Anesth Pain Med 2019;44:240–5. [DOI] [PubMed] [Google Scholar]
- [64].Zhou SQ, Wang J, Du WJ, et al. Optimum interval time of programmed intermittent epidural bolus of ropivacaine 0.08% with sufentanyl 0.3 μg/mL for labor analgesia: a biased-coin up-and-down sequential allocation trial. Chin Med J 2020;133:517–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [65].Holgado CM, Girones A, Tapia N, et al. Labour outcomes with epidural analgesia: an observational before-and-after cohort study comparing continuous infusion versus programmed intermittent bolus plus patient-controlled analgesia. Minerva Anestesiol 2020;86:1277–86. [DOI] [PubMed] [Google Scholar]