Abstract
Schistosoma haematobium is the leading cause of urogenital schistosomiasis and it is recognised as a class 1 carcinogen due to the robust association of infection with bladder cancer. In schistosomes, tetraspanins (TSPs) are abundantly present in different parasite proteomes and could be potential diagnostic candidates due to their accessibility to the host immune system. The large extracellular loops of six TSPs from the secretome (including the soluble excretory/secretory products, tegument and extracellular vesicles) of S. haematobium (Sh-TSP-2, Sh-TSP-4, Sh-TSP-5, Sh-TSP-6, Sh-TSP-18 and Sh-TSP-23) were expressed in a bacterial expression system and polyclonal antibodies were raised to the recombinant proteins to confirm the anatomical sites of expression within the parasite. Sh-TSP-2, and Sh-TSP-18 were identified on the tegument, whereas Sh-TSP-4, Sh-TSP-5, Sh-TSP-6 and Sh-TSP-23 were identified both on the tegument and internal tissues of adult parasites. The mRNAs encoding these TSPs were differentially expressed throughout all schistosome developmental stages tested. The potential diagnostic value of three of these Sh-TSPs was assessed using the urine of individuals (stratified by infection intensity) from an endemic area of Zimbabwe. The three Sh-TSPs were the targets of urine IgG responses in all cohorts, including individuals with very low levels of infection (those positive for circulating anodic antigen but negative for eggs by microscopy). This study provides new antigen candidates to immunologically diagnose S. haematobium infection, and the work presented here provides compelling evidence for the use of a biomarker signature to enhance the diagnostic capability of these tetraspanins.
Author summary
Schistosoma haematobium, the leading cause of urogenital schistosomiasis, affects millions of people worldwide. Infection with this parasite is associated with different clinical complications such as squamous cell carcinoma and genital malignancy in women. Despite its importance, there is a lack of sensitive and specific diagnostics that support control and elimination initiatives against this devastating disease. Herein, we have characterised six molecules belonging to the tetraspanin family of membrane proteins, providing details about their relative expression during parasite’s development and their localization in adult forms of S. haematobium. Furthermore, we have characterised the antibody responses against three of these molecules in urine from infected human subjects from an endemic area, providing compelling evidence for the use of these molecules to diagnose urogenital schistosomiasis.
Introduction
Schistosomiasis is a parasitic disease caused by blood dwelling trematodes from the genus Schistosoma and it is the second most important parasitic disease next to malaria in terms of social, economic and public health impact [1]. Over 250 million people are affected and 700 million people live in areas at risk [2]. Most of the schistosomiasis burden is found in Sub-Saharan Africa (SSA) [3], where around 280,000 people die annually [2]. S. haematobium, the causative agent of urogenital schistosomiasis, is endemic in the Middle East and some African countries [4, 5], and has been sporadically detected in India [6] and France [7]. The disease affects more than 90 million people, with nearly 150,000 deaths per year [8].
Pathogenesis associated with urogenital schistosomiasis is mainly caused by eggs trapped in tissues, particularly in the bladder [9]. The adult female worms live in the perivesicular veins, where a single female adult worm, when paired, can release hundreds of eggs per day after 4–7 weeks of infection [10]. Some of the eggs penetrate the bladder wall and get excreted through urine; however, half of the produced eggs are carried away with the bloodstream and trapped in the tissues of different urogenital organs [10]. The eggs trapped in the capillary beds of the bladder stimulate an immune response [9], affecting the bladder wall, leading to general urinary dysfunction and progressing to obstructive renal pathology [11, 12]. Infected individuals can also develop squamous cell carcinoma (an especially aggressive type of bladder cancer) [13, 14]; indeed, the International Agency for Research on Cancer has classified this parasite as a class 1 carcinogen [15]. In female patients, urogenital schistosomiasis may increase the risk of acquiring HIV infection [16].
Microscopic examination of urine to detect schistosome eggs is the gold standard test for diagnosis of S. haematobium infection [17]. Even though this technique is simple to perform and inexpensive [18], it cannot detect the acute stage of the disease (early infection, when no eggs are released) and it is influenced by day-to-day variability in egg excretion [19]. The circulating cathodic antigen (CCA) rapid diagnostic test is more sensitive than egg microscopy and detects the presence of a schistosome glycan in patient’s urine [20]; however, the sensitivity of this test is low in areas where S. mansoni and S. haematobium are co-endemic [21]. A different schistosome glycan, the circulating anodic antigen (CAA) has also be used to diagnose S. haematobium infections in the urine samples in low endemic settings [22, 23]. The CAA test applies a luminescent highly sensitive up-converting reporter particle technology (UCP) in a lateral flow (LF) based assay format. The UCP-LF CAA test is also applicable to blood-based samples and detects all Schistosoma species (potentially down to the level of a single worm [24]). However, it requires a basic laboratory and can be costly when requiring highest sensitivity; the test (yet) has not been commercialised. Molecular techniques like PCR are highly specific; however, DNA-based diagnosis of helminths requires expensive equipment and reagents [25]. Detecting antibodies produced against the different developmental stages of S. haematobium can help in the diagnosis of patients with a light egg load (or acute infection when the microscopic examination is still negative) in low-level endemic areas [26]. Despite the fact that antibody detection cannot differentiate between past and active infections [5, 10, 27] it could help in determining re-emergence of schistosomiasis in certain areas and the diagnosis of travellers [28] as well as supporting schistosomiasis control and elimination initiatives, with particular focus on post-elimination surveillance [29].
Recently, we documented the proteomic composition of the soluble excretory/secretory (ES) products, extracellular vesicles (EVs) and tegument from S. haematobium adult worms, revealing many different proteins, including tetraspanins (TSPs) [30, 31]. TSPs are a family of proteins that consist of four transmembrane domains, a small extracellular loop (SEL) and a large extracellular loop (LEL). TSPs are involved in many cellular activities such as differentiation, adhesion and division [32]. In platyhelminths, TSPs play an important role in tegument formation, maturation and stability [33–35]. In schistosomes, TSP LELs have been tested as vaccine candidates [36]; indeed, Sm-TSP-2 from S. mansoni has completed phase I clinical trials [37]. Furthermore, TSPs from other platyhelminths such as Taenia solium and Schistosoma japonicum have been suggested as potential diagnostic candidates [38, 39] and a secretome-wide immunomics/proteomics analysis of S. haematobium has highlighted the diagnostic efficacy of several of these molecules [29].
Herein, we characterise six S. haematobium TSPs (Sh-TSPs) that might be playing key roles in host-parasite interactions since they were found in the tegument, ES products and EVs of adult S. haematobium worms. We also assess the diagnostic efficacy of some of these TSPs in recombinant form using the urine of individuals with different S. haematobium infection intensities. This study not only provides important molecular information about this family of proteins and implicates them in host-parasite interactions, but also contributes to the first steps towards the generation of new diagnostic tools for this devastating disease.
Materials and methods
Ethics statement
The collection of urine from individuals from Zimbabwe was approved by the Medical Research Council of Zimbabwe; Approval MRCZ/A/1710. Written informed consent was required and obtained.
All experimental procedures performed on animals in this study were approved by the James Cook University (JCU) animal ethics committee through project A2391. All experiments were performed in accordance with the 2007 Australian Code of Practice for the Care and Use of Animals for Scientific Purposes and the 2001 Queensland Animal Care and Protection Act.
Animals
Male BALB/c mice (6–8 week) were purchased from the Animal Resource Centre, Perth, Australia and maintained at the AITHM animal facilities on the JCU Cairns campus. Mice were kept in cages under controlled temperature and light with free access to pelleted food and water as described previously [40].
Human urine samples
A total of 73 urine samples from S. haematobium infected individuals from an endemic area and 9 urine samples from Australian volunteer donors that had never travelled to schistosomiasis endemic areas (non-endemic negative) were collected as described previously [31]. In line with WHO criteria, the infection level of urine samples collected from the endemic area were classified as either high (>50 eggs/10 mL of urine) (n = 20), medium (11–49 eggs/10 mL of urine) (n = 20), low (0.3–10 eggs/10 mL of urine) (n = 30) and egg negative (0 eggs/10 mL of urine) (n = 3). To confirm the presence or absence of infection, egg negative urines obtained from patients from the endemic area were also tested for the presence of CAA using the UCAA2000 format of the UCP-LF CAA test as described previously [41]. Of these samples, all were positive for CAA. The collected urine samples were aliquoted and placed at -80°C until further use.
Schistosoma haematobium material
S. haematobium-infected (Egyptian strain) Bulinus truncatus snails were provided by the Biomedical Research Institute, MD, USA. Snails were removed from the tank with a pair of forceps and washed several times with water to remove debris and rotifers, then transferred to a Petri dish and incubated without water at 27°C in the dark for 2 h. Water was added and the snails were placed under light for 1.5 h at 28–30°C. Cercariae were concentrated using a 20 μm pore size sieve and then used to infect BALB/c mice (1,000/mouse) by tail penetration [42]. S. haematobium adult worms were obtained by perfusion of mice at 16 weeks post-infection [42]. Freshly perfused S. haematobium adult worms were fixed in paraformaldehyde, embedded in paraffin and cryostatically sectioned into 7.0 μm sections [43].
S. haematobium schistosomula were obtained by transforming cercariae in vitro by mechanical transformation as described previously [42]. Schistosomula were resuspended to a density of 1,000/ml in modified Basch media supplemented with 4x antibiotic/antimycotic (10,000 units/mL of penicillin, 10,000 μg/mL of streptomycin, and 25 μg/mL of amphotericin B) (Thermo Fisher Scientific, USA) and incubated at 37°C in 5% CO₂. Media was changed daily and schistosomula (1,000) were collected at 24 hours, 3 and 5 days and immediately stored in 500 μl of TRI reagent (Sigma-Aldrich, USA) at -80°C until further use.
RNA extraction, cDNA synthesis and RT-qPCR
Total RNA from S. haematobium from 24 h, 3 d and 5 d schistosomula was extracted using TRI reagent as per the manufacturer’s instruction. The RNA pellets were finally resuspended in 12 μl RNAse-free water and incubated for 5 min at 55°C. First strand cDNA was synthesized using the total volume of the RNA suspension and Superscript III reverse transcriptase (ThermoFisher Scientific, USA) according to the manufacturer’s instructions. The cDNA of S. haematobium adult, miracidia, cercariae and egg stages were provided by the Biomedical Research Institute, MD, USA.
Real time quantitative PCR (RT-qPCR) was performed to check the expression levels of Sh-tsp genes in different life stages (adult, egg, miracidia, cercaria and schistosomula (24 h, 3 days and 5 days)). Each qRT-PCR reaction consisted of 5 μl of SYBR green premix EX Taq (2x) (Qiagen, Netherlands), 1 μl (10 mM) of each forward and reverse primer (S1 Table), 1μl (50 ng) of the first–strand cDNA and sterile water to a final volume of 10 μl. The reactions were performed on a Rotor-Gene Q (Qiagen, Netherlands) using the following conditions: initial denaturation at 95°C for 10 min (1 cycle) followed by 40 cycles of denaturation (95°C for 10 sec), annealing (50°C for 15 sec) and extension (72°C for 20 sec). Sh-tsp expression was normalised to a housekeeping gene (α-tubulin, accession number XM_012938434.1) as described before [44] and relative expression levels were calculated using the 2−ΔΔCt method using egg as a reference group [45].
Phylogenetic analysis
A phylogenetic analysis was performed on the 6 Sh-TSPs of interest and 32 TSP sequences from different organisms (Bos taurus, Clonorchis sinensis, Danio rerio, Homo sapiens, Mus musculus, S. japonicum, S. mansoni and Opisthorchis viverrini) as well as from other TSPs present in the genome of S. haematobium obtained from the NCBI database (the list and accession numbers of all sequences are detailed in S2 Table). A multiple sequence alignment was carried out using the alignment program MUSCLE [46]. PhyML (v 20160207) [47] was used for maximum-likelihood (ML) phylogenetic analyses of the amino acid sequences using default parameters (substitution model: ‘LG’; number of relative substitution categories: ‘4’; alpha value for the discrete Gamma model: ‘e’, which gets the maximum likelihood estimate; bootstrap values obtained using ‘approximate Bayes branch support’). The tree was visualized with The Interactive Tree of Life (iTOF) online phylogeny tool (https://itol.embl.de/) [48] using default parameters.
Cloning of Sh-tsp cDNAs
The cDNAs encoding for the open reading frames of 5 Sh-TSPs (MS3_09198, MS3_01370, MS3_01153, MS3_05226 and MS3_05289) were obtained from the S. haematobium genome (www.parasite.wormbase.org), while the cDNA encoding for the open reading frame of Sh-TSP-2 was obtained from GenBank (accession number MK238557). For ease of reading and to standardise the nomenclature with S. mansoni tetraspanins, we have renamed the proteins (see S3 Table for matching codes and names). The LEL regions from the 6 Sh-TSPs were identified using TMpred (https://embnet.vital-it.ch/software/TMPRED_form.html) and amplified by PCR using oligonucleotide primers flanking these regions and S. haematobium adult cDNA as a template (S4 Table). The PCR products of Sh-TSP-4, Sh- TSP-5, Sh-TSP-6, Sh-TSP-18 and Sh-TSP-23 were NcoI/XhoI cloned into pET-32aΔHis such that they were in frame with the N-terminal thioredoxin (TRX) and C-terminal 6xHis tags. The vector pET-32aΔHis is an in-house modified version of pET-32a (Novagen, USA) which has the N-terminal 6xHis-tag absent to facilitate efficient purification after cleavage of the TRX tag if such removal is deemed necessary. The Sh-TSP-2 PCR product was NdeI/XhoI cloned into pET41a (Novagen, USA) to facilitate native N-terminal expression without the GST fusion tag but retaining the C-terminal 6xHis tag. Recombinant vectors were transformed into E. coli TOP10 strains (ThermoFisher Scientific, USA) and recombination confirmed by sequencing.
Protein expression
Recombinant plasmids were transformed into E. coli BL21(DE3) (ThermoFisher Scientific, USA) and resultant colonies were inoculated into 10 mL of Luria broth containing 100 μg/ml ampicillin (LBamp) and incubated overnight at 37°C with shaking at 200 rpm. Overnight culture was seeded (1/100) into 500 mL of fresh LBamp and incubated at 37°C with shaking at 200 rpm until OD600 = 0.5–1.0 (approximately 3 h), whereupon expression was induced by the addition of 1 mM isopropyl β-D-1-thiogalactopyranoside (IPTG) (Bioline, UK). Cultures continued incubating for 4 h (Sh-TSP-4, Sh-TSP-5, Sh-TSP-6 and Sh-TSP-23) or 16 h (Sh-TSP-18) and were then harvested by centrifugation at 8,000 g for 20 min at 4°C. Sh-TSP-2 was expressed as for Sh-TSP-23 except that LB supplemented with 50 μg/ml kanamycin was used instead of LBamp.
Each harvested pellet was resuspended in 50 mL of lysis buffer (50 mM sodium phosphate pH 8, 40 mM imidazole and 300 mM NaCl), freezed/thawed 3 times and then sonicated 10 times (5 sec bursts) at 4°C. Then, for the soluble proteins (Sh-TSP-2, Sh-TSP-6 and Sh-TSP-23 –determined by a small-scale pilot expression) the bacterial lysate was centrifuged at 20,000 g for 20 min at 4°C and the supernatant decanted and stored at -80°C. In the case of insoluble proteins (Sh-TSP-4, Sh-TSP-5 and Sh-TSP-18 –determined by small-scale pilot expression), Triton X-100 was added to a final concentration of 3% after sonication, the mixture incubated for 1 h at 4°C with gentle shaking and then pelleted at 20,000 g for 20 min at 4°C. The supernatant was removed, the pellet washed twice with 30 mL of lysis buffer (with centrifugation at 20,000 g for 20 min at 4°C after each wash) and the final pellet resuspended in 20 mL of solubilisation buffer (50 mM sodium phosphate, 40 mM imidazole, 300 mM NaCl and 6 M urea). The resuspension was incubated at 4°C overnight with gentle shaking, centrifuged at 20,000 g for 20 min at 4°C and the supernatant decanted and stored at -80°C.
An empty pET-32aΔHis vector was transformed into E. coli BL21(DE3) and expressed and purified as described above for Sh-TSP-23 to obtain the TRX tag, which was used as a control in ELISAs.
Protein purification
Recombinant proteins were purified by Ni2+ IMAC using an AKTA Prime UPC FPLC (GE Healthcare, USA). Each recombinant protein solution was diluted 1:4 in lysis buffer (soluble proteins) or solubilization buffer (insoluble proteins) and filtered through a 0.45 μm filter. The solutions were then applied to a 1 mL His-Trap IMAC column (GE Healthcare, USA), equilibrated with lysis buffer (soluble proteins) or solubilization buffer (insoluble proteins), at a flow rate of 1 mL/min. Bound proteins were washed with 10 column volumes (CV) of lysis buffer (soluble proteins) or solubilization buffer (insoluble proteins) and then eluted using lysis buffer (soluble proteins) or solubilization buffer (insoluble proteins) with an increasing linear gradient of imidazole (100–500 mM). Fractions containing purified recombinant proteins were combined and buffer exchanged into PBS (soluble proteins) or 1x PBS, 300 mM NaCl and 6 M urea (insoluble proteins) using a 3 kDa MWCO Amicon Ultra-15 centrifugal filter. The identity of expressed proteins was confirmed by SDS-PAGE and Western blot using anti-His monoclonal antibodies.
Polyclonal antibody production
Three male BALB/c mice (6 weeks old) were immunized subcutaneously with 50 μg of each recombinant protein emulsified with Alum adjuvant (Thermo Fisher Scientific, USA) and boosted twice at two weekly intervals using the same amount of protein as described previously [43]. Blood was collected from each mouse before immunization and two weeks after the final immunization. Sera was collected by allowing the blood to clot followed by centrifugation at 10,000 g for 10 min and then stored at -20°C.
Immunohistochemistry
Immunohistochemistry was performed to determine the anatomic sites of Sh-TSP expression in sections of adult worms. Adult worm sections from S. haematobium were de-paraffinized using 2 x 3 min washes each of 100% and 50% xylene and rehydrated in an ethanol series. Antigen retrieval was performed by boiling the slides in citrate buffer (10 mM sodium citrate, pH 6) for 40 minutes followed by Tris buffer (10mM Tris, 1 mM EDTA, 0.05% Tween, pH 9.0), for 20 minutes. Subsequently, sections were blocked with 10% goat serum for 1 hour at RT. After washing 3 times with TBS/0.05% Tween-20 (TBST), sections were incubated with anti-Sh-TSP antisera (diluted 1:50 in 1% BSA/TBST) overnight at 4°C and then washed with TBST (3 x 5 min). Sections were finally probed with goat-anti-mouse IgG-Alexa Fluor 647 (Sigma-Aldrich, USA) (diluted 1:200 in 1% BSA/TBST) for 1 h in the dark at RT. After a final washing step with TBST, slides were mounted with Entellan mounting medium (Millipore, Germany) and covered with coverslips. The images were acquired by Nuance software with an AxioImager M1 fluorescence microscope (ZEISS, Germany).
Indirect enzyme-linked immunosorbent assay
An enzyme-linked immunosorbent assay (ELISA) was performed to assess the diagnostic efficacy of the recombinantly expressed Sh-TSPs using the serum of infected mice (a pool of four independent S. haematobium infections and another pool of four independent S. mansoni infections) and the urine from naturally infected Zimbabwean people. Sera from naïve mice or urine from non-endemic individuals were used as negative controls. Microtiter plate wells (Greiner Bio-One, Austria) were coated with 50 μl (2 μg/ml) of protein (either Sh-TSP2, Sh-TSP-4, Sh-TSP-5, Sh-TSP-6, Sh-TSP-18 or Sh-TSP-23 for the mouse ELISAs and either Sh-TSP-4, Sh-TSP-5, Sh-TSP-18 or a combination of the three antigens for the human ELISAs) in 0.1 M carbonate-bicarbonate buffer (pH 9.6) and incubated overnight at 4°C. The plates were washed 3 times with PBST and blocked with 5% milk at 37°C for 1 h. Then, plates were washed 3 times with PBST and 50 μl of human urine (diluted 1:50 in PBST) or mouse serum (diluted 1:5,000 in PBST) was added and incubated at 37°C for 1 h. After washing with PBST 3 times, 50 μl of HRP-conjugated anti-human IgG (Sigma-Aldrich, USA) or anti-mouse IgG (diluted 1:5,000 in PBST) was added, incubated at 37°C for 1 h and washed 3 times with PBST. Finally, 50 μl of 3,3’,5,5’-tetramethylbenzidine (TMB) (Thermo Fisher Scientific, USA) was added and incubated for 12 minutes at RT in the dark. The reaction was stopped with 3 M HCl and the absorbance was measured at 450 nm using a POLARstar Omega (BMG Labtech, Australia).
Statistics
All statistics were performed using GraphPad Prism 9.0. For both serum and urine ELISAs, significance between groups was determined using the Student’s t test and reactivity cut-off values were determined as the average reactivity +3SD of the non-endemic negative group. Receiver Operating Characteristic (ROC) curves were used to calculate sensitivity, specificity and the area under the curve (AUC).
Results
General characteristics of Sh-TSPs
Six Sh-TSPs (Sh-TSP-2, Sh-TSP-4, Sh-TSP-5, Sh-TSP-6, Sh-TSP-18 and Sh-TSP-23) were selected based on previous proteomic analyses from the adult worm tegument, ES and EVs (Table 1) [31]. The sequences of all Sh-TSPs were interrogated and each ORF contained four transmembrane domains, one SEL, one LEL and three intracellular regions. Each LEL contained the four-cysteine residues (forming two disulfide bonds) as well as the CCG motif characteristic of TSPs [49]. Full-length cDNA sequence, predicted amino acid sequence and LEL sizes are shown in Table 1. The amino acid sequence similarities between the Sh-TSPs described here and their S. mansoni homologs ranged from 71–93% (when entire ORFs were compared) (S1 Fig) and 70.2–84% (when just the LEL regions were compared) (S2 Fig).
Table 1. Characteristics of Schistosoma haematobium tetraspanins.
Aas: aminoacids; bp: base pairs; LEL: large extracellular loop; SEL: small extracellular loop.
| TSPs | Length (bp) | Size (# aas) | Weight (kDa) | SEL position | Inner loop position | LEL position | Cytoplasmic tail | Originally identified from [30,31] |
|---|---|---|---|---|---|---|---|---|
| Sh-TSP-2 | 660 | 219 | 24.4 | 31–79 | 74–80 | 102–183 | 213–219 | Tegument 120k EVs 15k EVs |
| Sh-TSP-4 | 837 | 278 | 30.9 | 36–76 | 100–108 | 130–250 | 272–278 | Tegument 120k EVs 15k EVs |
| Sh-TSP-5 | 822 | 273 | 30.4 | 31–79 | 97–109 | 173–239 | 265–273 | Secretome 120k EVs 15k EVs |
| Sh-TSP-6 | 675 | 224 | 24.9 | 36–54 | 87–95 | 108–190 | 212–225 | Tegument 15k EVs |
| Sh-TSP-18 | 888 | 295 | 32.9 | 32–66 | 87–95 | 118–266 | 283–225 | 120k EVs 15k EVs |
| Sh-TSP-23 | 654 | 218 | 24.2 | 36–56 | 72–81 | 103–184 | 205–218 | Tegument 120k EVs 15k EVs |
Phylogeny of Sh-TSPs
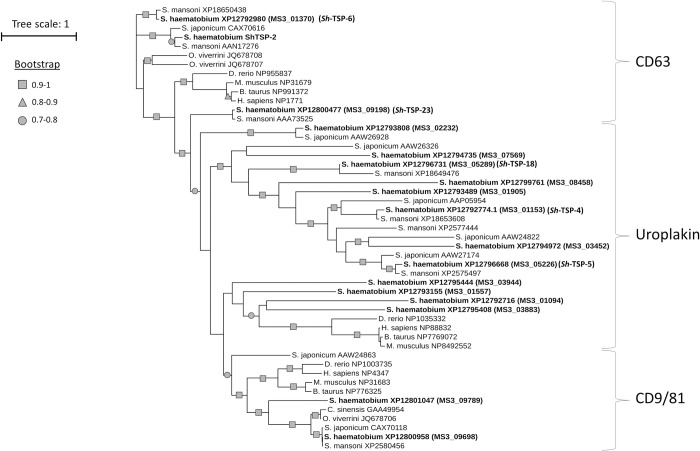
A phylogenetic analysis between the studied Sh-TSPs and other well-characterised TSPs from related trematodes was performed. Sh-TSP-2, Sh-TSP-6 and Sh-TSP-23 grouped together in the CD63 clade of TSPs and clustered together with other well characterised CD63-like TSPs from S. mansoni (AAN17276, AAA73525 and XP_018650438), S. japonicum (CAX70616) and O. viverrini (JQ678707.1 and JQ678708.1) and bootstrapping values > 0.7 (Fig 1). Sh-TSP-4, Sh-TSP-5 and Sh-TSP-18 clustered under the uroplakin family of TSPs together with other TSPs from S. mansoni (XP_018649476, XP_18653608, XP_002577444 and XP_002575497), S. japonicum (AAW26298, AAW26326, AAW24822, AAP05954 and AAW27174) and S. haematobium (MS3_02232, MS3_07569, MS3_08458, MS3_01905, MS3_03452, MS3_03944, MS3_01557, MS3_01094, MS3_03883) and bootstrap values mostly > 0.9 (Fig 1).
Fig 1. Phylogenetic analysis of Schistosoma haematobium tetraspanins and homologs from related organisms.
A multiple sequence alignment was carried out using MUSCLE. PhyML was used for a maximum-likelihood phylogenetic analysis with bootstrapping, and results were visualized with The Interactive Tree of Life (iTOF) online phylogeny tool (https://itol.embl.de/) using default parameters.
Protein expression and purification
The LEL region from each of the Sh-tsp cDNA was cloned into a bacterial expression system and sequences were validated by Sanger sequencing. The Sh-TSP-6, Sh-TSP-23 and Sh-TSP-2 LELs were expressed as soluble proteins, while Sh-TSP-4, Sh-TSP-5 and Sh-TSP-18 were expressed as inclusion bodies and 6 M urea was used for solubilisation. The expected sizes of the expressed Sh-TSPs were confirmed by SDS-PAGE (S3A Fig) and a western-blot using a monoclonal anti-His antibody (S3B Fig).
Sh-TSPs are expressed throughout all life stages
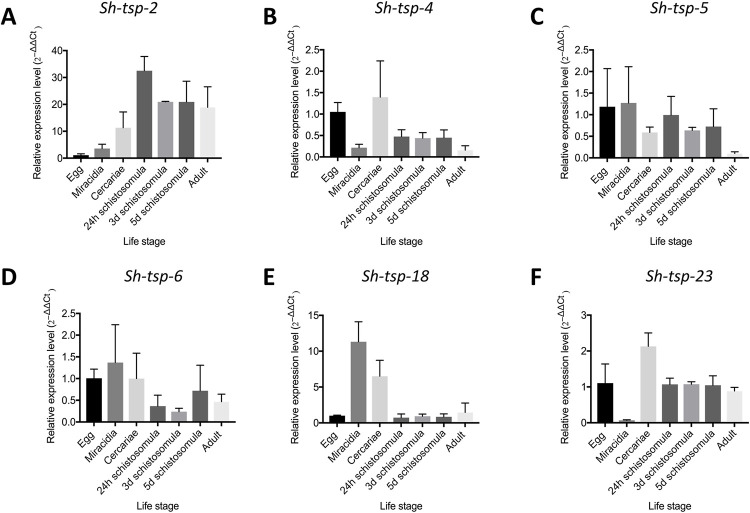
The transcriptional patterns of all Sh-tsp mRNAs were analysed in different life stages of S. haematobium: adult, egg, miracidia, cercaria and schistosomula (24 h, 3 d and 5 d) by RT-qPCR. All Sh-tsp mRNAs were expressed throughout all life stages tested (Fig 2). Overall, the expression levels of Sh-tsp-2 and Sh-TSP-18 were the highest (Fig 2A and 2D). Sh-tsp-2 expression peaked at 24 h schistosomula and decreased in subsequent developmental stages (Fig 2A). Similarly, the highest expression level of Sh-tsp-5, Sh-tsp-6 and Sh-tsp-18 was identified in the miracidia while the lowest level of expression was observed in adult, 3-day schistosomula and 24 h schistosomula, respectively (Fig 2B, 2F and 2D). In the case of Sh-tsp-23, the highest expression level was observed in cercariae and the lowest expression level was observed in miracidia (Fig 2C). The expression level of Sh-tsp-4 was highest in cercariae and lowest in adult life stages (Fig 2E).
Fig 2. Expression levels of Schistosoma haematobium tetraspanins at different life stages.
Relative mRNA expression levels of Sh-tsp-2 (A), Sh-tsp-4 (B), Sh-tsp-5 (C), Sh-tsp-6 (D), Sh-tsp-18 (E) and Sh-tsp-23 (F) were analyzed by qPCR. Relative mRNA expression levels were normalised to a housekeeping gene (α-tubulin), calculated using the 2−ΔΔCt method using the egg stage as a reference group.
Sh-TSPs are expressed in the tegument and internal organs of S. haematobium adult worms
To determine the location of Sh-TSPs in the adult worms, sections from S. haematobium adult worms were probed with mouse polyclonal antibodies produced against each Sh-TSP (Fig 3). Sh-TSP-2 and Sh-TSP-18 were identified on the tegument of the worms, whereas Sh-TSP-4, Sh-TSP-5, Sh-TSP-6 and Sh-TSP-23 were identified both on the tegument and internal organs of adult worms (Fig 3). S. haematobium adult worm sections were not recognised by the negative control anti-TRX antibody (Fig 3).
Fig 3. Localisation of Schistosoma haematobium tetraspanins in adult worms.
Immunolocalisation of the S. haematobium tetraspanins (TSPs) Sh-TSP-2, Sh-TSP-4, Sh-TSP-5, Sh-TSP-6, Sh-TSP-18, Sh-TSP-23 and thioredoxin (TRX) in adult worm sections. Sections were probed with anti-S. haematobium TSP sera followed by goat-anti-mouse IgG-Alexa Fluor 647.
Sh-TSPs are recognized by the sera of infected mice
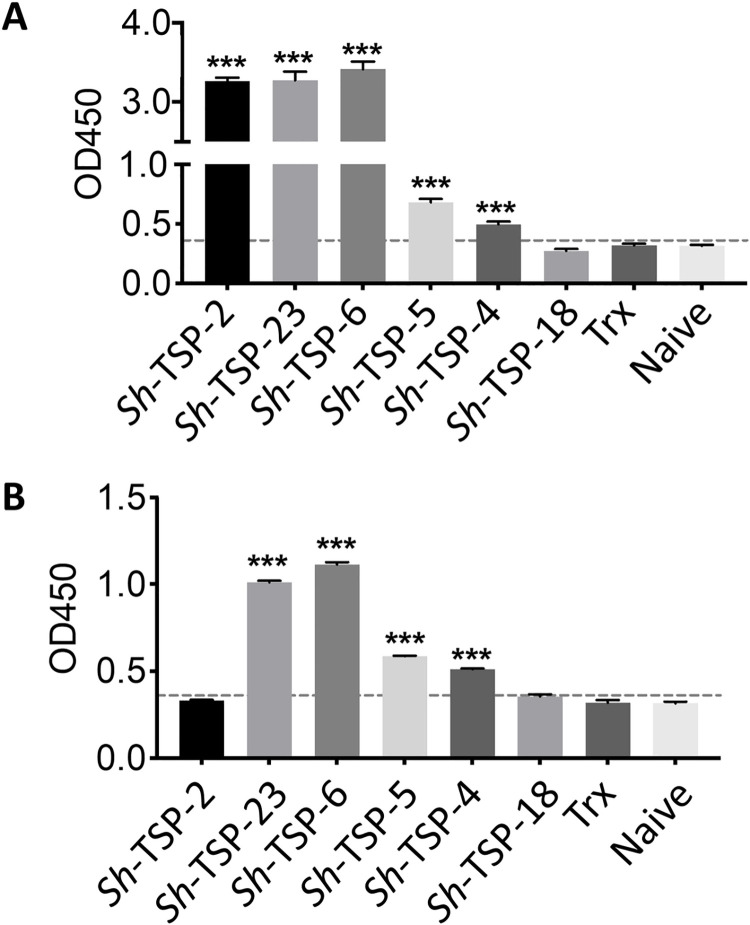
As a first step towards the assessment of TSPs as diagnostic candidates, we performed an indirect ELISA to analyse the immunogenicity of these TSPs in mice. All Sh-TSPs except Sh-TSP-18 were the target of significantly greater antibody responses (P<0.001) in the sera of mice experimentally infected with S. haematobium compared to uninfected mouse sera (Fig 4A). To assess cross-reactivity with S. mansoni, an indirect ELISA was performed using the sera of mice experimentally infected with S. mansoni, and Sh-TSP-4, Sh-TSP-5, Sh-TSP-6 and Sh-TSP-23 were the target of significantly greater (P<0.001) antibody levels in S. mansoni infected mice compared to uninfected mice (Fig 4B).
Fig 4. Schistosoma haematobium tetraspanins are recognized by antibodies in the sera of infected mice.
Bar graph showing the detection of S. haematobium tetraspanins (TSPs) using S. haematobium infected mouse serum (A) and S. mansoni infected mouse serum (B). The reactivity cut-off points were determined by comparing the average OD450 of infected mouse serum with the average plus 3 standard deviations of naïve mouse serum. A statistical analysis between infected vs. naïve mice was performed using a Student’s t test. ***P<0.001.
Sh-TSPs are recognized by antibodies in the urine of naturally infected human subjects from an endemic area
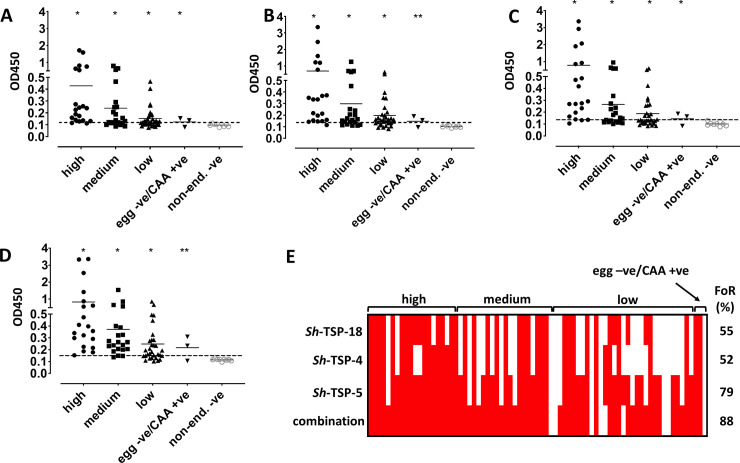
Since the diagnostic efficacy of Sh-TSP-2, Sh-TSP-6 and Sh-TSP-23 has already been evaluated in a previous publication [29], herein we tested the diagnostic efficacy of the uroplakin members Sh-TSP-4, Sh-TSP-5 and Sh-TSP-18 via their recognition by antibodies in urine from infected individuals from an endemic area in Zimbabwe. All Sh-TSPs tested were the targets of significantly higher antibody levels in infected individuals compared to negative controls (urine samples from a non-endemic area), either using individual TSPs (Fig 5A, 5B and 5C) or a combination of all TSPs (Fig 5D). Since all tested proteins were expressed as thioredoxin-fused molecules, the thioredoxin tag obtained from the empty vector was used as a control. Only four sera from individuals with a high intensity infection cross-reacted with this tag, whereas thioredoxin was not recognized by other infected individuals (S4 Fig). Frequency of recognition (FoR) was above 50% for every single antigen (52% for Sh-TSP-4, 79% for Sh-TSP-5 and 55% for Sh-TSP-18,), and 88% when the three antigens were combined (Fig 5E). No correlation was observed between egg counts and OD for any of the proteins tested.
Fig 5. Recognition of six Schistosoma haematobium tetraspanins by urine antibodies from Zimbabwean individuals with different infection intensities.
The antibody level was measured by indirect ELISA and indicated by OD values: Sh-TSP-4 (A), Sh-TSP-5 (B), Sh-TSP-18 (C) and the combination of all three antigens (D). All the data was entered in GraphPad Prism 9 and analysed by Student’s t test (infected vs. non endemic negative). * P < 0.05, ** P < 0.01. Urine of non-infected individuals from a non-endemic area was used as negative control. The reactivity cut-off values were determined by adding the average and 3x standard deviations of non-endemic negative individuals (indicated by broken lines). (E) Frequency of recognition (FoR) patterns based on ELISA IgG responses to E. coli-expressed recombinant proteins. FoR percentages among the infected populations (sensitivity) are displayed on the right-hand side of the image.
Predictive accuracy of Sh-TSPs for the diagnosis of Schistosoma haematobium infection
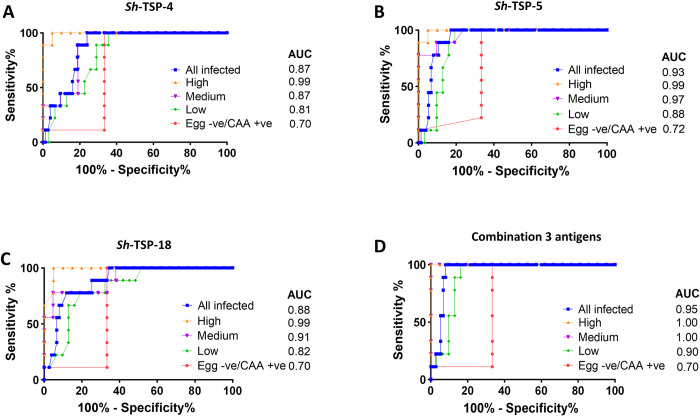
The diagnostic accuracy of Sh-TSPs was measured by calculating the area under the curve (AUC) of the receiver operating characteristic (ROC) curve generated for each antigen (Fig 6). All three antigens showed a diagnostic accuracy of 0.99 for individuals with high egg burdens (Fig 6). In the case of medium egg burden individuals, the highest accuracy of diagnosis was obtained with Sh-TSP-5 (0.97), followed by Sh-TSP-18 (0.91) and Sh-TSP-4 (0.87) (Fig 6). For individuals with low egg burdens, the highest accuracy of detection was also obtained with Sh-TSP-5 (0.88), followed by Sh-TSP-18 (0.82) and Sh-TSP-4 (0.81) (Fig 6). In the case of egg negative but CAA positive individuals, the highest accuracy of detection was obtained with Sh-TSP-5 (0.72) followed by Sh-TSP-4 and Sh-TSP-18 (both with 0.70) (Fig 6).
Fig 6. Receiver operating characteristic (ROC) curves analysis of Schistosoma haematobium tetraspanins.
The diagnostic accuracy of S. haematobium TSPs to detect antibodies in the urine of infected individuals with differing infection status was measured by the area under the ROC curve (AUC). Sh-TSP-4 (A), Sh-TSP-5 (B), Sh-TSP-18 (C) and the combination of all three antigens (D). Urine of non-infected individuals from non-endemic area was used as negative control.
The combination of Sh-TSP-4, Sh-TSP-5 and Sh-TSP-18 resulted in higher AUCs for all cohorts. An AUC of 1.0 was obtained in the case of high and medium egg burden individuals, while in the case of low and egg negative/CAA positive burden individuals, accuracy of detection was 0.90 and 0.70, respectively.
Additionally, the AUC for all infected was 0.95 when using the combination of Sh-TSP-4, Sh-TSP-5 and Sh-TSP-18 antigens, while it was 0.87, 0.93, 0.88 and for Sh-TSP-4, Sh-TSP-5 and Sh-TSP-18, respectively.
Discussion
TSPs are a family of proteins that consist of four transmembrane domains, a SEL and a LEL. The LEL portion of TSPs contains between four and eight cysteine residues that form two to four disulfide bonds, facilitating specific protein-protein interactions with laterally associated proteins and other ligands [50]. TSPs are involved in many activities of cells such as differentiation, adhesion, and division [32]. They can also play an important role in host-parasite interactions, and some TSPs have been tested as potential vaccines against a range of trematode infections [36, 51–53]. The first TSP identified in S. mansoni was Sm23 [54] and, since then, other TSPs have been found in the proteome of schistosomes [55–58]. Recently, the S. haematobium ortholog of Sm23 (Sh-TSP-23), and 5 other additional TSPs were identified in a proteomic analysis of the ES products, EVs and tegument from S. haematobium adult worms [31] and, in the present study we have characterized these Sh-TSPs, and assessed the potential of three of them for diagnosing infection using the urine of naturally infected individuals with different infection intensities.
Sh-TSP-2, Sh-TSP-6 and Sh-TSP-23 are grouped together in the CD63 family of TSPs, together with known S. mansoni and S. japonicum vaccine candidates [36, 52], whereas Sh-TSP-4, Sh-TSP-5 and Sh-TSP-18 are clustered under the uroplakin family of TSPs. All Sh-TSPs formed a single clade distinct from vertebrate TSPs. Interestingly, CD63 antigens such as Sh-TSP-2 and Sm-TSP-2 have been studied as diagnostic markers and vaccine candidates in the past [29, 36]. CD63 and uroplakin members have been suggested to have similar functional properties in S. japonicum [59], although the exact roles for uroplakins, either in the infection process or in pathogenesis, is still unkown and further research is needed.
All Sh-tsp genes were expressed throughout all the assessed life stages of the parasite, albeit with differing expression patterns, as has been reported for S. japonicum [38] and S. mansoni [60]. Taken together with their detected presence in tegumental, EV and ES proteomic studies, this data suggests that all Sh-TSPs are, upon initial infection, continuously exposed to the immune system. The highest levels of expression for Sh-TSP-5, Sh-TSP-6, Sh-TSP-18, were observed in miracidia, which implies that these Sh-TSPs may have specific roles in the intermediate snail host. For Sh-TSP-4, Sh-TSP-23 and Sh-TSP-2, the highest level of expression was observed in cercariae and 24 h schistosomula, respectively. Similarly, the highest expression level of Sm-TSP-2 was detected in egg [34]. Furthermore, the highest expression level for the CD63-like TSPs from the human carcinogenic liver fluke, O. viverrini (Ov-TSP-2 and Ov-TSP-3) was observed in the egg stage [35]. These S. mansoni and O. viverrini TSPs have been shown to be involved in tegument formation, maturation and stability, and thus, homologous Sh-TSPs may also be involved in tegument formation and EV secretion [61–64].
Sh-TSP-4, Sh-TSP-5, Sh-TSP-6 and Sh-TSP-23 are located on the tegument surface as well as on various internal organs of adult flukes. Similarly, Sj-TSP-5 and Sj-TSP-6 are also located on the tegument and internal organs of adult worms whereas Sj-TSP-3 Sj-TSP-1 are located only in the internal organs of adult worms [59]. In contrast, Sh-TSP-2 and Sh-TSP-18 are located exclusively on the tegument of adult S. haematobium worms, and other TSPs from S. mansoni, O. viverrini and S. japonicum [35, 36, 59, 65] also display this localisation pattern. These tegumental proteins might play an important role in tegument biogenesis and turnover [35, 36, 65]. Since the tegument is the most susceptible structure to host-mediated immune attack [66], the LEL regions of TSPs in trematodes have been tested as vaccine candidates. Immunization of mice with the LEL of two S. mansoni TSPs (Sm-TSP-1 and Sm-TSP-2) significantly decreased adult worm and liver egg burdens [36] and Sm-TSP-2 has completed phase I clinical trials [67]. Sm23 is one of the independently tested WHO vaccine candidates [68] and its S. japonicum ortholog (Sj23) was also found to be an efficacious vaccine in animal models of schistosomiasis [69, 70]. Indeed, Sm23 has been shown to be efficacious when delivered as a DNA vaccine [71], and immunization of mice with a fusion of the Sj23 TSP with other vaccine candidates significantly reduced the worm burden and liver eggs in a subsequent S. japonicum challenge [69, 70]. Furthermore, antibodies produced against a TSP present in O. viverrini EVs blocked the internalization of EVs by cholangiocytes and decreased the production of cytokines that stimulate tumorigenesis [61], while TSPs present in S. haematobium EVs elicited protection in a heterologous model of schistosomiasis [30]. These observations demonstrated the usefulness of TSPs in vaccine strategies.
All Sh-TSPs except Sh-TSP-18 were significantly recognized by serum antibodies from mice experimentally infected with S. haematobium. Similarly, TSPs from other platyhelminths such as T. solium, O. viverrini and S. japonicum can be recognized by the antibodies of infected humans and experimental animals [35, 38, 39, 65]. This, together with the localisation studies and gene expression patterns, suggests that these TSPs are accessible to antibodies and are immunogenic during natural infection. Indeed, we have shown Sh-TSP2, Sh-TSP-6 and Sh-TSP-23 to be the targets of IgG responses in the serum and urine of individuals from S. haematobium-endemic areas [29]. Interestingly, Sh-TSP2 and Sh-TSP-18 were not recognized by antibodies from S. mansoni infected mice, an observation consistent with the preferential recognition of Sh-TSP-2 by S. haematobium-infected as opposed to S. mansoni-infected individuals [29], implying the potential usefulness of Sh-TSP-2 and Sh-TSP-18 in the diagnosis of S. haematobium infections where both species are co-endemic. Motivated by these results, and despite the low recognition of Sh-TSP-18 by the serum of infected mice, we decided to test the utility of Sh-TSP-4, Sh-TSP-5 and Sh-TSP-18 (the diagnostic efficacy of Sh-TSP2, Sh-TSP-6 and Sh-TSP-23 has been reported elsewhere [29]) as biomarkers of S. haematobium infection by using them to detect antibodies in the urine of individuals infected with the parasite. Urine, not serum, was selected as the diagnostic fluid due to the relative ease of sample collection, especially in field conditions [72]. In certain infectious disease states, the use of urine over serum for diagnosis may be at the expense of sensitivity, but we posit that, for S. haematobium infections, any potential decrease in detection limits may be mitigated by an increased level of IgG present in the urine, relative to that produced by kidney excretion, due to the serum antibody leakage into the bladder caused by egg-induced damage. Each Sh-TSP was the target of significantly elevated antibody levels in all cohorts, including egg negative/CAA positive urine samples, compared to uninfected subjects, highlighting the increased diagnostic sensitivity of this approach compared to egg microscopy. Further, the use of Sh-TSP combinations in diagnosis increased the predictive accuracy of infection by more comprehensively capturing the breadth of the anti-schistosome antibody response than any single antigen. This is likely explained by the different FoR patterns for each antigen (due to the variation in life-stage expression of each molecule, different antigens may be present in individuals with acute or patent infections) combining to form a “consensus” FoR for the infected population. With regards to diagnostic use, preparations of defined, recombinant antigens can offer an advantage over crude protein mixtures (such as SEA or SWAP) in that they represent a more standardised and sustainable resource for diagnosis. Indeed, the defined antigen preparations described herein have AUC and FoR values which exceed that of SEA [31], making them a potentially effective, as well as rigorous, tool for the non-invasive diagnosis of S. haematobium infection.
In the present study, we have fully characterised different Sh-TSPs in order to advance the knowledge on this important family of proteins. In addition, this is the first study assessing the diagnostic efficacy of uroplakin-like Sh-TSPs, and only the second study describing the use of defined recombinant antigens in the diagnosis of S. haematobium infection, and provides the first steps towards the generation of new diagnostic tools against this devastating disease.
Supporting information
A multiple sequence alignment was carried out using MUSCLE with defaults and visualised using JalView with default ClustalX colouring.
(TIF)
A multiple sequence alignment was carried out using MUSCLE with defaults and visualised using JalView.
(TIF)
1μg of each TSP was electrophoresed on SDS-PAGE and Comassie-stained: Protein ladder (A1), Sh-TSP-2 (A2), Sh-TSP-4 (A3), Sh-TSP-18 (A4), Sh-TSP-5 (A5), Sh-TSP-23 (A6) and Sh-TSP-6 (A7). Western blot of TSPs using an anti-His monoclonal antibody: Protein ladder (B1), Sh-TSP-2 (B2), Sh-TSP-4 (B3), Sh-TSP-18 (B4), Sh-TSP-5 (B5), Sh-TSP-23 (B6) and Sh-TSP-6 (B7). The expected size of recombinant Sh-TSP-2 was 12.4 kDa, since it was expressed without a tag. For Sh-TSP-4, Sh-TSP-5, Sh-TSP-6, Sh-TSP-18 and Sh-TSP-23, since they contained the TRX-tag from the cloning vector, their expected size ranged from ~28 kDa to ~38 kDa.
(TIF)
The antibody level was measured by indirect ELISA and indicated by OD values. Urine of non-infected individuals from non-endemic area was used as negative control. The reactivity cut-off values were determined by adding the average and 3x standard deviation of non-endemic negative individuals (indicated by broken lines).
(TIF)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Acknowledgments
The authors thank the study participants, as well as the parents/legal guardians and their teachers in Zimbabwe for their support of this study. They are very grateful for the cooperation of the Ministry of Health and Child Welfare in Zimbabwe. For their technical support, they would like to thank the members of the Department of Biochemistry at the University of Zimbabwe.
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
This work was supported by a program grant (APP# 1037304) from the National Health and Medical Research Council (NHMRC) and a Senior Principal Research fellowship from NHMRC to AL (APP# 1117504). GGM received funding from the Australian Institute of Tropical Health and Medicine PhD scholarship. J.S. is a Miguel Servet Fellow funded by Instituto de Salud Carlos III (CP17III/00002). The funders had no role in study design, data collection, analysis and publication.
References
- 1.CDC. Schistosomiasis [cited 2016 10/09/2016]. https://www.cdc.gov/parasites/schistosomiasis/biology.html].
- 2.Hotez P, Alvarado M, Basáñez M-G, Bolliger I, Bourne R, Boussinesq M, et al. The Global Burden of Disease Study 2010: Interpretation and Implications for the Neglected Tropical Diseases. PLoS Neglected Tropical Diseases 2014;8(7):e2865. doi: 10.1371/journal.pntd.0002865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Steinmann P, Keiser J, Bos R, Tanner M, Utzinger J. Schistosomiasis and water resources development: systematic review, meta-analysis, and estimates of people at risk. The Lancet Infectious Diseases. 2006;6(7):411–25. doi: 10.1016/S1473-3099(06)70521-7 [DOI] [PubMed] [Google Scholar]
- 4.Colley DG, Bustinduy AL, Secor E, King CH. Human schistosomiasis. Lancet. 2014;383(9936):2253–64. doi: 10.1016/S0140-6736(13)61949-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gryseels B, Polman K, Clerinx J, Kestens L. Human schistosomiasis. Lancet. 2006;368(9541):1106–18. doi: 10.1016/S0140-6736(06)69440-3 . [DOI] [PubMed] [Google Scholar]
- 6.Chitsulo L, Engels D, Montresor A, Savioli L. The global status of schistosomiasis and its control. Acta Tropica. 2000;77(1):41–51. doi: 10.1016/s0001-706x(00)00122-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Boissier J, Grech-Angelini S, Webster BL, Allienne J-F, Huyse T, Mas-Coma S, et al. Outbreak of urogenital schistosomiasis in Corsica (France): an epidemiological case study. The Lancet Infectious Diseases. 2016;16(8):971–9. doi: 10.1016/S1473-3099(16)00175-4 [DOI] [PubMed] [Google Scholar]
- 8.Berger S. Schistosoma haematobium 2017 edition. Los Angeles, CA: GIDEON Informatics Inc; 2017. [Google Scholar]
- 9.Odegaard JI, Hsieh MH. Immune responses to Schistosoma haematobium infection. Parasite Immunology. 2014;36(9):428–38. doi: 10.1111/pim.12084 [DOI] [PubMed] [Google Scholar]
- 10.Schistosomiasis Gryseels B. Infectious Disease Clinics of North America. 2012;26(2):383–97. doi: 10.1016/j.idc.2012.03.004 [DOI] [PubMed] [Google Scholar]
- 11.van der Werf MJ, de Vlas SJ, Brooker S, Looman CW, Nagelkerke NJ, Habbema JD, et al. Quantification of clinical morbidity associated with schistosome infection in sub-Saharan Africa. Acta Tropica. 2003;86(2–3):125–39. doi: 10.1016/s0001-706x(03)00029-9 . [DOI] [PubMed] [Google Scholar]
- 12.Kjetland EF, Ndhlovu PD, Mduluza T, Gomo E, Gwanzura L, Mason PR, et al. Simple clinical manifestation of genital Schistosoma haematobium infection in rural Zimbabweab women American Journal of Tropical Medicine and Hygiene. 2005;72(3):311–9. [PubMed] [Google Scholar]
- 13.Shiff C, Veltri R, Naples J, Quartey J, Otchere J, Anyan W, et al. Ultrasound verification of bladder damage is associated with known biomarkers of bladder cancer in adults chronically infected with Schistosoma haematobium in Ghana. Trans R Soc Trop Med Hyg. 2006;100(9):847–54. Epub 2006/01/31. doi: 10.1016/j.trstmh.2005.10.010 . [DOI] [PubMed] [Google Scholar]
- 14.McManus DP, Dunne DW, Sacko M, Utzinger J, Vennervald BJ, Zhou XN. Schistosomiasis. Nat Rev Dis Primers. 2018;4(1):13. Epub 2018/08/11. doi: 10.1038/s41572-018-0013-8 . [DOI] [PubMed] [Google Scholar]
- 15.IARC. Biological agents. A review of human carcinogens. IARC monographs on the evaluation of carcinogenic risks to humans / World Health Organization, International Agency for Research on Cancer. 2012;100(Pt B):1. [Google Scholar]
- 16.Ndhlovu PD, Mduluza T, Kjetland EF, Midzi N, Nyanga L, Gundersen SG, et al. Prevalence of urinary schistosomiasis and HIV in females living in a rural community of Zimbabwe: does age matter? Transactions of the Royal Society of Tropical Medicine and Hygiene. 2007;101(5):433–8. doi: 10.1016/j.trstmh.2006.08.008 [DOI] [PubMed] [Google Scholar]
- 17.Ross AG, Bartley Pb Fau—Sleigh AC, Sleigh Ac Fau—Olds GR, Olds Gr Fau—Li Y, Li Y Fau—Williams GM, Williams Gm Fau—McManus DP, et al. Schistosomiasis. New England Journal of Medicine. 346(16):1212–20. [DOI] [PubMed] [Google Scholar]
- 18.Stete K, Krauth SJ, Coulibaly JT, Knopp S, Hattendorf J, Müller I, et al. Dynamics of Schistosoma haematobium egg output and associated infection parameters following treatment with praziquantel in school-aged children. Parasites & vectors. 2012;5(1):298–. doi: 10.1186/1756-3305-5-298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Braun-Munzinger RA, Southgate BA. Repeatability and reproducibility of egg counts of Schistosoma haematobium in urine. Trop Med Parasitol. 1992;43(3):149–54. Epub 1992/09/01. . [PubMed] [Google Scholar]
- 20.Kremsner PG, De Jonge N, Simarro PP, Mühlschlegel F, Mir M, Sima FO, et al. Quantitative determination of circulating anodic and cathodic antigens in serum and urine of individuals infected with Schistosoma intercalatum. Transactions of the Royal Society of Tropical Medicine and Hygiene. 1993;87(2):167–9. doi: 10.1016/0035-9203(93)90474-5 [DOI] [PubMed] [Google Scholar]
- 21.Ashton RA, Stewart BT, Petty N, Lado M, Finn T, Brooker S, et al. Accuracy of circulating cathodic antigen tests for rapid mapping of Schistosoma mansoni and S. haematobium infections in Southern Sudan. Tropical Medicine & International Health. 2011;16(9):1099–103. doi: 10.1111/j.1365-3156.2011.02815.x [DOI] [PubMed] [Google Scholar]
- 22.Knopp S, Corstjens PL, Koukounari A, Cercamondi CI, Ame SM, Ali SM, et al. Sensitivity and Specificity of a Urine Circulating Anodic Antigen Test for the Diagnosis of Schistosoma haematobium in Low Endemic Settings. PLoS neglected tropical diseases. 2015;9(5):e0003752. Epub 2015/05/15. doi: 10.1371/journal.pntd.0003752 ; PubMed Central PMCID: PMC4431728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Corstjens P, de Dood CJ, Knopp S, Clements MN, Ortu G, Umulisa I, et al. Circulating Anodic Antigen (CAA): A Highly Sensitive Diagnostic Biomarker to Detect Active Schistosoma Infections-Improvement and Use during SCORE. Am J Trop Med Hyg. 2020;103(1_Suppl):50–7. Epub 2020/05/14. doi: 10.4269/ajtmh.19-0819 ; PubMed Central PMCID: PMC7351307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Langenberg MCC, Hoogerwerf MA, Janse JJ, van Lieshout L, Corstjens P, Roestenberg M, et al. Katayama Syndrome Without Schistosoma mansoni Eggs. Ann Intern Med. 2019;170(10):732–3. Epub 2019/01/08. doi: 10.7326/L18-0438 . [DOI] [PubMed] [Google Scholar]
- 25.Gordon CA, Gray DJ, Gobert GN, McManus DP. DNA amplification approaches for the diagnosis of key parasitic helminth infections of humans. Molecular and Cellular Probes. 2011;25(4):143–52. doi: 10.1016/j.mcp.2011.05.002 [DOI] [PubMed] [Google Scholar]
- 26.Clerinx J, Van Gompel A. Schistosomiasis in travellers and migrants. Travel Medicine and Infectious Disease. 2011;9(1):6–24. doi: 10.1016/j.tmaid.2010.11.002 [DOI] [PubMed] [Google Scholar]
- 27.Bergquist R, Johansen MV, Utzinger J. Diagnostic dilemmas in helminthology: what tools to use and when? Trends in Parasitology. 2009;25(4):151–6. doi: 10.1016/j.pt.2009.01.004 [DOI] [PubMed] [Google Scholar]
- 28.Allen GPR, Sleigh AC, Li Y, Davis GM, Williams GM, Jiang Z, et al. Schistosomiasis in the People’s Republic of China: prospects and challenges for the 21st century. Clinical Microbiology Reviews. 2001;14(2):270–95. doi: 10.1128/CMR.14.2.270-295.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pearson MS, Tedla BA, Mekonnen GG, Proietti C, Becker L, Nakajima R, et al. Immunomics-guided discovery of serum and urine antibodies for diagnosing urogenital schistosomiasis: a biomarker identification study. The Lancet Microbe. 2021;2(11):e617–e26. doi: 10.1016/S2666-5247(21)00150-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mekonnen GG, Tedla BA, Pickering D, Becker L, Wang L, Zhan B, et al. Schistosoma haematobium Extracellular Vesicle Proteins Confer Protection in a Heterologous Model of Schistosomiasis. Vaccines (Basel). 2020;8(3). Epub 2020/07/30. doi: 10.3390/vaccines8030416 ; PubMed Central PMCID: PMC7563238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sotillo J, Pearson MS, Becker L, Mekonnen GG, Amoah AS, van Dam G, et al. In-depth proteomic characterization of Schistosoma haematobium: Towards the development of new tools for elimination. PLoS neglected tropical diseases. 2019;13(5):e0007362. Epub 2019/05/16. doi: 10.1371/journal.pntd.0007362 ; PubMed Central PMCID: PMC6538189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hemler ME. Tetraspanin proteins mediate cellular penetration, invasion, and fusion events and define anovel type of membrane microdomain Annual Review of Cell and Developmental Biology. 2003;19(1):397–422. doi: 10.1146/annurev.cellbio.19.111301.153609 [DOI] [PubMed] [Google Scholar]
- 33.Piratae S, Tesana S, Jones MK, Brindley PJ, Loukas A, Lovas E, et al. Molecular characterization of a tetraspanin from the human liver fluke, Opisthorchis viverrini. PLoS neglected tropical diseases. 2012;6(12):e1939. doi: 10.1371/journal.pntd.0001939 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tran MH, Freitas TC, Cooper L, Gaze S, Gatton ML, Jones MK, et al. Suppression of mRNAs encoding tegument tetraspanins from Schistosoma mansoni results in impaired tegument turnover. PLoS Pathogens. 2010;6 (4):e1000840. doi: 10.1371/journal.ppat.1000840 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chaiyadet S, Krueajampa W, Hipkaeo W, Plosan Y, Piratae S, Sotillo J, et al. Suppression of mRNAs encoding CD63 family tetraspanins from the carcinogenic liver fluke Opisthorchis viverrini results in distinct tegument phenotypes. Scientific reports. 2017;7(1):14342. Epub 2017/11/01. doi: 10.1038/s41598-017-13527-5 ; PubMed Central PMCID: PMC5662742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tran MH, Pearson MS, Bethony JM, Smyth DJ, Jones MK, Duke M, et al. Tetraspanins on the surface of Schistosoma mansoni are protective antigens against schistosomiasis. Nature Medicine. 2006;12(7):835–40. doi: 10.1038/nm1430 [DOI] [PubMed] [Google Scholar]
- 37.ClinicalTrials.gov. A phase I study of the safety, reactogenicity, and immunogenicity of Sm-TSP-2/alhydrogel with or without GLA-AF for intestinal schistosomiasis in healthy adults.
- 38.Wang L, Giri BR, Chen Y, Xia T, Liu J, Li H, et al. Molecular characterization, expression profile, and preliminary evaluation of diagnostic potential of CD63 in Schistosoma japonicum. Parasitol Res. 2018;117(11):3625–31. Epub 2018/09/05. doi: 10.1007/s00436-018-6063-8 . [DOI] [PubMed] [Google Scholar]
- 39.Hancock K, Pattabhi S, Whitfield FW, Yushak ML, Lane WS, Garcia HH, et al. Characterization and cloning of T24, a Taenia solium antigen diagnostic for cysticercosis. Molecular and biochemical parasitology. 2006;147(1):109–17. Epub 2006/03/17. doi: 10.1016/j.molbiopara.2006.02.004 . [DOI] [PubMed] [Google Scholar]
- 40.Eichenberger RM, Ryan S, Jones L, Buitrago G, Polster R, Montes de Oca M, et al. Hookworm Secreted Extracellular Vesicles Interact With Host Cells and Prevent Inducible Colitis in Mice. Frontiers in immunology. 2018;9:850. Epub 2018/05/16. doi: 10.3389/fimmu.2018.00850 ; PubMed Central PMCID: PMC5936971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Corstjens PL, De Dood CJ, Kornelis D, Fat EM, Wilson RA, Kariuki TM, et al. Tools for diagnosis, monitoring and screening of Schistosoma infections utilizing lateral-flow based assays and upconverting phosphor labels. Parasitology. 2014;141(14):1841–55. Epub 2014/06/17. doi: 10.1017/S0031182014000626 ; PubMed Central PMCID: PMC4265670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tucker MS, Karunaratne LB, Lewis FA, Freitas TC, Liang Y-s. Schistosomiasis. Current protocols in immunology / edited by John E Coligan [et al]. 2013;103(103):Unit 19.1. doi: 10.1002/0471142735.im1901s103 [DOI] [PubMed] [Google Scholar]
- 43.Pearson MS, Pickering DA, McSorley HJ, Bethony JM, Tribolet L, Dougall AM, et al. Enhanced protective efficacy of a chimeric form of the schistosomiasis vaccine antigen Sm-TSP-2. PLoS neglected tropical diseases. 2012;6(3):e1564. Epub 2012/03/20. doi: 10.1371/journal.pntd.0001564 ; PubMed Central PMCID: PMC3302818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lancelot J, Caby S, Dubois-Abdesselem F, Vanderstraete M, Trolet J, Oliveira G, et al. Schistosoma mansoni Sirtuins: characterization and potential as chemotherapeutic targets. PLoS neglected tropical diseases. 2013;7(9):e2428. Epub 2013/09/27. doi: 10.1371/journal.pntd.0002428 ; PubMed Central PMCID: PMC3772001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25(4):402–8. Epub 2002/02/16. doi: 10.1006/meth.2001.1262 . [DOI] [PubMed] [Google Scholar]
- 46.Edgar RC. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004;32(5):1792–7. Epub 2004/03/23. doi: 10.1093/nar/gkh340 ; PubMed Central PMCID: PMC390337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Guindon S, Lethiec F, Duroux P, Gascuel O. PHYML Online—a web server for fast maximum likelihood-based phylogenetic inference. Nucleic Acids Res. 2005;33(Web Server issue):W557–9. Epub 2005/06/28. doi: 10.1093/nar/gki352 ; PubMed Central PMCID: PMC1160113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Letunic I, Bork P. Interactive Tree Of Life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021;49(W1):W293–W6. Epub 2021/04/23. doi: 10.1093/nar/gkab301 ; PubMed Central PMCID: PMC8265157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Boucheix C, Rubinstein E. Tetraspanins. Cellular and molecular life sciences: CMLS. 2001;58(9):1189–205. Epub 2001/10/02. doi: 10.1007/PL00000933 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Levy S, Shoham T. The tetraspanin web modulates immune-signalling complexes. Nature Reviews Immunology. 2005;5(2):136–48. doi: 10.1038/nri1548 [DOI] [PubMed] [Google Scholar]
- 51.Chaiyadet S, Sotillo J, Krueajampa W, Thongsen S, Brindley PJ, Sripa B, et al. Vaccination of hamsters with Opisthorchis viverrini extracellular vesicles and vesicle-derived recombinant tetraspanins induces antibodies that block vesicle uptake by cholangiocytes and reduce parasite burden after challenge infection. PLoS neglected tropical diseases. 2019;13(5):e0007450. Epub 2019/05/29. doi: 10.1371/journal.pntd.0007450 ; PubMed Central PMCID: PMC6555531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhang W, Li J, Duke M, Jones MK, Kuang L, Zhang J, et al. Inconsistent protective efficacy and marked polymorphism limits the value of Schistosoma japonicum tetraspanin-2 as a vaccine target. PLoS neglected tropical diseases. 2011;5(5):e1166. Epub 2011/06/10. doi: 10.1371/journal.pntd.0001166 ; PubMed Central PMCID: PMC3104969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Dang Z, Yagi K, Oku Y, Kouguchi H, Kajino K, Watanabe J, et al. Evaluation of Echinococcus multilocularis tetraspanins as vaccine candidates against primary alveolar echinococcosis. Vaccine. 2009;27(52):7339–45. Epub 2009/09/29. doi: 10.1016/j.vaccine.2009.09.045 . [DOI] [PubMed] [Google Scholar]
- 54.Gaugitsch HW, Hofer E, Huber NE, Schnabl E, Baumruker T. A new superfamily of lymphoid and melanoma cell proteins with extensive homology to Schistosoma mansoni antigen Sm23. European journal of immunology. 1991;21(2):377–83. Epub 1991/02/01. doi: 10.1002/eji.1830210219 [DOI] [PubMed] [Google Scholar]
- 55.Braschi S, Wilson RA. Proteins exposed at the adult schistosome surface revealed by biotinylation. Molecular & Cellular Proteomics. 2006;5(2):347–56. doi: 10.1074/mcp.M500287-MCP200 [DOI] [PubMed] [Google Scholar]
- 56.Braschi S, Curwen RS, Ashton PD, Verjovski-Almeida S, Wilson A. The tegument surface membranes of the human blood parasite Schistosoma mansoni: a proteomic analysis after differential extraction. Proteomics. 2006;6(5):1471. doi: 10.1002/pmic.200500368 [DOI] [PubMed] [Google Scholar]
- 57.van Balkom BWM, van Gestel RA, Brouwers JFHM, Krijgsveld J, Tielens AGM, Heck AJR, et al. Mass spectrometric analysis of the Schistosoma mansoni tegumental sub-proteome. Journal of Proteome Research. 2005;4(3):958. doi: 10.1021/pr050036w [DOI] [PubMed] [Google Scholar]
- 58.Sotillo J, Pearson M, Becker L, Mulvenna J, Loukas A. A quantitative proteomic analysis of the tegumental proteins from Schistosoma mansoni schistosomula reveals novel potential therapeutic targets. International Journal For Parasitology. 2015;45(8):505–16. doi: 10.1016/j.ijpara.2015.03.004 [DOI] [PubMed] [Google Scholar]
- 59.Jiang Y, Xu X, Qing X, Pan W. Identification and characterization of six novel tetraspanins from Schistosoma japonicum. Parasit Vectors. 2011;4:190. Epub 2011/10/01. doi: 10.1186/1756-3305-4-190 ; PubMed Central PMCID: PMC3203850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fitzpatrick JM, Peak E, Perally S, Chalmers IW, Barrett J, Yoshino TP, et al. Anti-schistosomal intervention targets identified by lifecycle transcriptomic analyses. PLoS neglected tropical diseases. 2009;3(11):e543. Epub 2009/11/04. doi: 10.1371/journal.pntd.0000543 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chaiyadet S, Sotillo J, Smout M, Cantacessi C, Jones MK, Johnson MS, et al. Carcinogenic liver fluke secretes extracellular vesicles that promote cholangiocytes to adopt a tumorigenic phenotype. Journal of Infectious Disease. 2015;212(10):1636–45. doi: 10.1093/infdis/jiv291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Mulvenna J, Moertel L, Jones MK, Nawaratna S, Lovas EM, Gobert GN, et al. Exposed proteins of the Schistosoma japonicum tegument. International Journal for Parasitology. 2010;40 (5):543–54. doi: 10.1016/j.ijpara.2009.10.002 [DOI] [PubMed] [Google Scholar]
- 63.Mulvenna J, Sripa B, Brindley PJ, Gorman J, Jones MK, Colgrave ML, et al. The secreted and surface proteomes of the adult stage of the carcinogenic human liver fluke Opisthorchis viverrini. Proteomics. 2010;10(5):1063–78. doi: 10.1002/pmic.200900393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sotillo J, Pearson M, Potriquet J, Becker L, Pickering D, Mulvenna J, et al. Extracellular vesicles secreted by Schistosoma mansoni contain protein vaccine candidates. International Journal For Parasitology. 2016;46(1):1–5. doi: 10.1016/j.ijpara.2015.09.002 [DOI] [PubMed] [Google Scholar]
- 65.Piratae S, Tesana S, Jones MK, Brindley PJ, Loukas A, Lovas E, et al. Molecular characterization of a tetraspanin from the human liver fluke, Opisthorchis viverrini. PLoS neglected tropical diseases. 2012;6(12):e1939. Epub 2012/12/14. doi: 10.1371/journal.pntd.0001939 ; PubMed Central PMCID: PMC3516575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Loukas A, Tran M, Pearson MS. Schistosome membrane proteins as vaccines. Int J Parasitol. 2007;37(3–4):257–63. Epub 2007/01/16. doi: 10.1016/j.ijpara.2006.12.001 . [DOI] [PubMed] [Google Scholar]
- 67.Keitel WA, Potter GE, Diemert D, Bethony J, El Sahly HM, Kennedy JK, et al. A phase 1 study of the safety, reactogenicity, and immunogenicity of a Schistosoma mansoni vaccine with or without glucopyranosyl lipid A aqueous formulation (GLA-AF) in healthy adults from a non-endemic area. Vaccine. 2019;37(43):6500–9. Epub 2019/09/14. doi: 10.1016/j.vaccine.2019.08.075 ; PubMed Central PMCID: PMC6771426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bergquist NR, Colley DG. Schistosomiasis Vaccine:Research to Development. Parasitology Today. 1998;14(3):99–104. doi: 10.1016/s0169-4758(97)01207-6 [DOI] [PubMed] [Google Scholar]
- 69.Dai Y, Zhu Y, Harn DA, Wang X, Tang J, Zhao S, et al. DNA vaccination by electroporation and boosting with recombinant proteins enhances the efficacy of DNA vaccines for Schistosomiasis japonica. Clinical and Vaccine Immunology. 2009;16(12):1796–803. doi: 10.1128/CVI.00231-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhu L, Liu H-F, Lu M-B, Long Q-K, Shi Y-E, Yu L-J. Construction, purification, and evaluation of multivalent DNA vaccine against Schistosoma japonicum. Parasitology Research. 2011;108(1):115–21. doi: 10.1007/s00436-010-2040-6 [DOI] [PubMed] [Google Scholar]
- 71.Da’Dara AA, Skelly PJ, Walker CM, Harn DA. A DNA-prime/protein-boost vaccination regimen enhances Th2 immune responses but not protection following Schistosoma mansoni infection. Parasite Immunol. 2003;25(8–9):429–37. Epub 2003/12/04. doi: 10.1111/j.1365-3024.2003.00651.x . [DOI] [PubMed] [Google Scholar]
- 72.Noya O, Alarcon de Noya B, Losada S, Colmenares C, Guzman C, Lorenzo MA, et al. Laboratory diagnosis of Schistosomiasis in areas of low transmission: a review of a line of research. Mem Inst Oswaldo Cruz. 2002;97 Suppl 1:167–9. Epub 2002/11/12. doi: 10.1590/s0074-02762002000900032 . [DOI] [PubMed] [Google Scholar]