Abstract
Context:
Immunohistochemistry (IHC) to differentiate germ cell tumors.
Aims:
The aim of the study is to differentiate seminomatous and nonseminomatous germ cell tumors (GCTs) with morphological overlap using a minimal and affordable panel of IHC markers.
Settings and Design:
This is a retrospective observational study.
Subjects and Methods:
All testicular GCTs (TGCT) which were diagnosed on biopsies and/or resection specimens (prechemotherapy) between January 2014 and June 2019. The demographic, clinical, and imaging findings were noted from the medical records. Hematoxylin and eosin (H and E)-stained sections were reviewed for morphology. The IHC markers constituted Octamer-binding transcription factor (OCT) 3/4, glypican 3 (GPC3), CD117, CD30, placental-like alkaline phosphatase, Sal-like protein 4, and β-human chorionic gonadotropin (HCG). IHC markers were performed in various combinations depending on the morphology, and a panel constituting OCT 3/4, CD117, GPC3, and CD30 was performed on cases with diagnostic dilemma and morphological overlaps.
Statistical Analysis Used:
Sensitivity, specificity, positive (PPV), and negative predictive value (NPV) were calculated for suggested panel of IHC OCT 3/4, CD117, GPC3, and CD30.
Results:
The study included 36 patients with TGCT with a mean age of 27 (15–58) years. Nonseminomatous tumors were the most common (86%). The concise panel was performed in 20/36 (56%) tumors to resolve the diagnosis. The sensitivity, specificity, PPV, and NPV for OCT3/4 were 80%, 55%, 31%, and 92% in seminomas and 65%, 100%, 100%, and 46% in embryonal carcinomas (EC), for CD117 was 89%, 82%, 73%, and 93% in seminomas and 60%, 77%, 60%, and 77% in yolk sac tumors (YST), for GPC3 was 95%, 90%, 95%, and 90% in YST, CD30 96%, 100%, 100%, and 91% in ECs, respectively.
Conclusions:
Designing a novel concise and affordable IHC panel constituting OCT 3/4, CD117, GPC3, and CD30 has good sensitivity and specificity in differentiating seminomas, YST, and EC, respectively. Additional markers, namely β-HCG, can be used in identifying the choriocarcinoma component.
Keywords: Germ cell tumors, immunohistochemistry, panel, Testis
INTRODUCTION
Testicular cancer represents 1% of malignancies in men. The incidence varies across the globe with reportedly higher occurrence in the developed and Western countries. India has the lowest age-standardized incidence of 0.5/100,000 men.[1] They exhibit biological and morphological diversity with some of them having overlapping features making them difficult to classify based only on histopathology.[2] Appropriate classification of germ cell tumor components is essential as biological behavior and therapeutic decisions are dependent on it. The predominance of embryonal carcinoma component in a Stage I nonseminomatous tumor requires additional therapy after orchidectomy, as it is commonly associated with high risk of failure on surveillance alone. Similarly, tumors showing early carcinomatous differentiation in seminoma may be managed more aggressively than pure seminomas at some institutions.[3]
Testicular germ cell tumors (TGCT) may be seminomatous or nonseminomatous germ cell tumors (NSGCT). Embryonal carcinoma, yolk sac tumor (YST), choriocarcinoma, teratoma, and mixed germ cell tumors (GCT) are included in NSGCT. Seminomas usually arise later in life, with a mean age of 35 years at the first presentation compared to 25 years for NSGCT.[4] Approximately 15% of TGCTs are mixed tumors, which contain both seminomas and nonseminoma elements. NSGCTs are often treated with surgery and chemotherapy, and their cure rates depend on the stage of the disease. The cure rate reaches up to 99% in the early stages of NSGCTs and decreases from 90% in those who had good prognostic category to 50% in those who had poor prognostic features.[5]
Since the outcome of the TGCTs depends on its pathological type and tumor stage, it is important to categorize the subtype. Immunohistochemistry (IHC) is useful to differentiate various germ cell tumors (GCT), especially those which present with morphologic overlap and provide a definitive diagnosis.
In this paper, we reviewed a series of 36 patients with TGCTs treated in our institute between January 2014 and June 2019 with IHC to find the most suitable panel for routine diagnostic application.
SUBJECTS AND METHODS
A retrospective observational study was carried out on all TGCTs which were diagnosed on biopsies and/or resection specimens (prechemotherapy) between January 2014 and June 2019. The demographic, clinical, and imageology findings were noted from the medical records. Surgical specimens were fixed in 10% buffered formalin and processed for paraffin-embedded sections. Four-μm sections stained with hematoxylin and eosin (H and E) were reviewed for morphology.
For IHCs, additional 4 μm sections were mounted on poly-l-lysine coded slides and were run on the Leica Bond 3 Autostainer with (heat-induced epitope retrieval). The markers performed were Octamer-binding transcription factor (OCT) 3/4, CD117, GPC3, CD30, sal-like protein 4 (SALL4), β-human chorionic gonadotropin human chorionic gonadotropin (HCG), and placental-like alkaline phosphatase (PLAP) wherever necessary and were categorized according to the criteria of WHO (2016) classification of TGCT.[6] Source and clone of IHC markers used for the study were noted [Table 1]. Slides were reviewed along with a positive control.
Table 1.
Source and clone of immunohistochemistry markers used in the study
| ANTIBODY | CLONE | COMPANY |
|---|---|---|
| OCT 3/4 | SEMGC | BIOCARE |
| CD117 | YR145 | CELLMARQUE |
| GPC3 | 1G12 | CELLMARQUE |
| CD30 | BCL H2 | DAKO |
| SALL4 | 6E3 (MMA) | BIOCARE |
| βHCG | MMA | PATH INSITU |
| PLAP | 8A9 | DAKO |
OCT 3/4: Octamer binding transcription factor 3/4; CD117: c-kit; GPC3: Glypican 3; CD30: SALL4: Sal-Like Protein 4; βHCG: β- Human chorionic gondotropin; PLAP: Placental alkaline phosphatase
All tumors were classified after running a panel of IHCs that constituted OCT3/4, CD117, GPC3, CD30, PLAP, SALL4, and β-HCG in various combinations depending on the cases in study. SALL4 or/and OCT3/4 were added as “entry” marker in cases of metastasis of unknown origin to determine the nature of tumor. On their positivity, the cases were evaluated along the lines of GCT. β-HCG was performed wherever there was a suspicion of choriocarcinoma component. Other IHC markers were as per the case demand. With the experience over a period of 5 years, a panel of four IHC markers was designed with an intention to cover the most common of the GCTs and their combinations. These included OCT3/4, CD117, GPC3, and CD30.
The sensitivity, specificity, positive, and negative predictive values for the proposed panel of IHC markers were calculated.
RESULTS
The study included 36 patients with TGCT with age ranging from 15 to 58 (mean 27) years. Most (28/36) of them were high inguinal orchidectomy (HIO) specimens and the others included cervical lymph nodes (4), undescended testis (2), retroperitoneal lymph node (1), and mediastinal mass (1). The cases other than orchidectomy specimens were proven to have TGCTs at a later date.
Histopathological examination showed germ cell tumor with different morphology [Figure 1]. IHC was done as per proposed panel along with additional markers wherever required [Figures 2 and 3].
Figure 1.
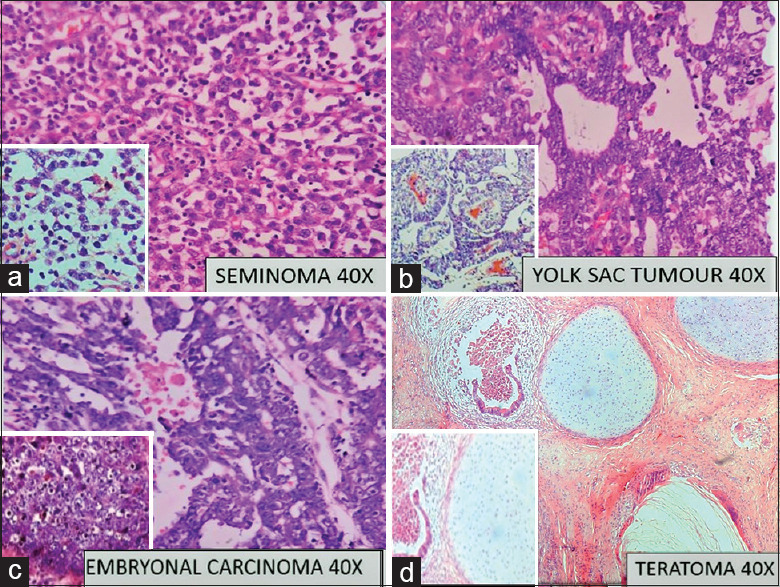
Photomicrograph of (a) Seminoma composed of sheets polygonal cells; Inset: tumor cells against a lymphocytic background; Hematoxylin and eosin (H and E, ×10). (b) Yolk sac tumor with microcystic pattern; Inset: Schiller-Duval bodies, H and E, ×40. (c) Embryonal carcinoma with sheets of polygonal cells with prominent nucleoli, H and E, ×40; Inset: Solid area in embryonal carcinoma. (d) Teratoma with mature epithelial and cartilaginous components; Inset: Immature neural elements; H and E, ×40
Figure 2.
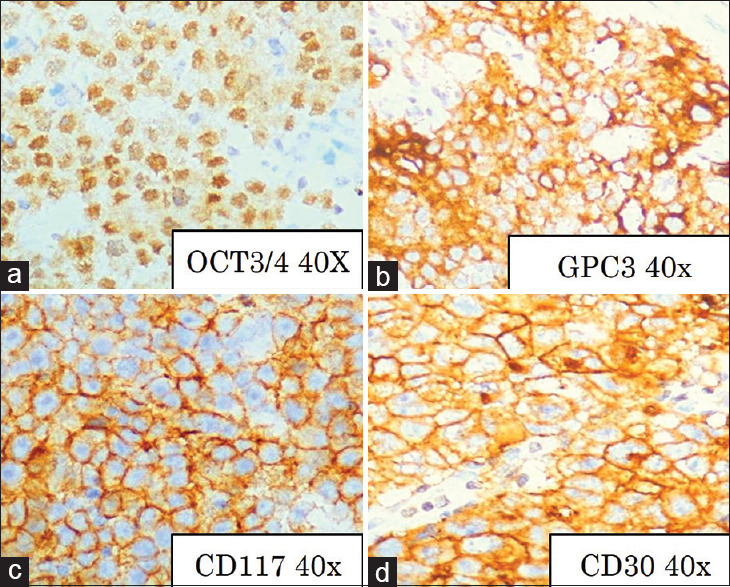
Immunohistochemistry: (a) Octamer binding transcription factor 3/4 positivity in embryonal carcinoma, Octamer binding transcription factor 3/4 × 40. (b) Glypican 3 positivity in yolk sac tumor, Glypican 3 × 40. (c) CD 117 positivity in seminoma, CD 117, ×40; (d) CD30 positivity in embryonal carcinoma, CD 30, ×40
Figure 3.
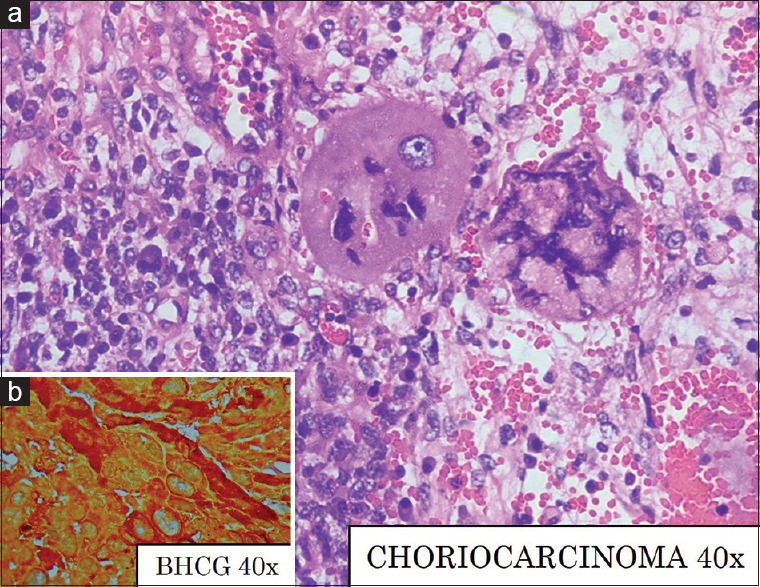
(a) Choriocarcinoma with syncytiotrophoblasts and cytotrophoblasts with areas of hemorrhage, H and E, ×40 (b) β-human chorionic gonadotropin positivity, β-human chorionic gonadotropin, ×40
Further analysis showed mixed GCTs to be most common (21) with the most common combination being YST with embryonal carcinoma (10/21) [Table 2]. SALL4 was positive in 7/8, OCT3/4 in 13/26, CD117 in 11/26, CD30 in 25/36, GPC3 in 21/31, PLAP in 2/3, and β-HCG in 2/4 cases.
Table 2.
(A) Histomorphological distribution of all TGCT (n=36), (B) distribution of mixed germ cell tumors (n=21)
| A) Histomorphology | Number | B) Mixed GCT components | Number |
|---|---|---|---|
| Seminoma | 5 | YST + EC | 10 |
| Yolk sac tumour | 2 | YST + EC + Teratoma | 8 |
| Embryonal carcinoma | 6 | YST + EC + Choriocarcinoma | 1 |
| Choriocarcinoma | 1 | YST + EC + Seminoma | 1 |
| Teratoma | 1 | EC + Seminoma | 1 |
| Mixed GCT | 21 | Total | 21 |
| Total | 36 |
YST: Yolk sac tumor, EC: Embryonal carcinoma
SALL4 was performed in 8 that included undescended testis (2), cervical lymph node (2), retroperitoneal lymph node (1), mediastinum (1), and HIO (2) and it was positive in 7. One specimen (undescended testis) was negative for SALL - 4 and was diffusely positive for CD30 only, and hence, a diagnosis of embryonal carcinoma was given in correlation with the morphology.
The panel of OCT3/4, CD117, GPC3, and CD30 was done in twenty tumors, which were resolved as two seminomas, two YSTs, four ECs, one choriocarcinoma, and eleven mixed GCTs.
The sensitivity, specificity, positive, and negative predictive values for each marker of the designated IHC panel were calculated [Table 3].
Table 3.
(A) Sensitivity and Specificity; (B) Positive predictive value (PPV) and Negative predictive value (NPV) of the panel of IHC markers used
| OCT3/4 | CD117 | GPC3 | CD30 | Specificity | OCT3/4 | CD117 | GPC3 | CD30 | |
|---|---|---|---|---|---|---|---|---|---|
| A) Sensitivity | |||||||||
| Seminoma | 80% | 89% | 0% | 0% | Seminoma | 55% | 82% | 0% | 0% |
| YST | 0% | 75% | 95% | 0% | YST | 0% | 80% | 90% | 0% |
| EC | 65% | 0% | 0% | 96% | EC | 100% | 0% | 0% | 100% |
|
| |||||||||
| OCT3/4 | CD117 | GPC3 | CD30 | NPV | OCT3/4 | CD117 | GPC3 | CD30 | |
|
| |||||||||
| B) PPV | |||||||||
| Seminoma | 31% | 73% | 0% | 0% | Seminoma | 92% | 93% | 0% | 0% |
| YST | 0% | 60% | 95% | 0% | YST | 0% | 89% | 90% | 0% |
| EC | 100% | 0% | 0% | 100% | EC | 46% | 0% | 0% | 91% |
DISCUSSION
TGCT usually presents between 20 and 45 years and rarely in patients younger than 15 years or older than 60 years.[7] The median age in the present study was 27 years. Fosså et al. reported that age is an adverse prognostic factor for predicting relapse.[8]
TGCTs are theorized to have a common cell of origin but differ with respect to degree of differentiation; seminomas are viewed as transformed primitive germ cells and are pluripotent. Embryonal carcinoma is the least differentiated, whereas YST, choriocarcinoma, and teratoma are more differentiated.[9]
Identification of the subtypes of GCTs is important in determining prognosis, designing appropriate therapies, and diagnosing metastases or relapses. This might be challenging when there is overlap of morphologic features. Examples of the morphological overlap may include (1) areas with architecture considered typical of YST (e.g. glandular, early secretary endometrium-like) but with markedly atypical cytological features; (2) atypical areas within or adjacent to seminoma that suggest an early transformation to embryonal carcinoma; and (3) the interface of embryonal carcinoma and solid YST with cytological features unlike the typical YST. Such cases warrant an assistance of IHC for identification and better characterization into the various subtypes.[2]
The usual IHC markers used in GCTs are PLAP, SALL4, OCT 3/4, CD117, Glypican 3 (GPC3), CD30, and β-HCG, applied in combination as and when the morphology deems it necessary. In our study, all the patients were <60 years of age and none showed morphological features of lymphoma on H and E sections, and hence, life cycle assessment was not included in the suggested panel of IHC markers.
OCT 3/4 is an octamer binding transcription factor that has been detected in tumor germ cell which has pluripotent potential.[10,11] It is mostly detected in seminomas/dysgerminomas and embryonal carcinomas (EC) but negative in YST. It is located in the cell nuclei.[12] Several researchers have reported that OCT 3/4 was positive in 100% seminoma and negative in normal testicular tissues.[13,14] Hattab et al. also demonstrated that OCT 3/4 is more specific and sensitive than PLAP in the diagnosis of TGCTs.[15] OCT3/4 was positive in 13 cases in the present study.
GPC3 is one of the highly over-expressed genes found in YST but not other nonseminomatous tumors of the testis. Zynger et al. confirmed the GPC3 differential expression pattern in YST compared with embryonal carcinoma and further demonstrated the lack of GPC3 expression in adjacent nonneoplastic testicular parenchyma, intratubular germ cell neoplasia, seminomas, teratomas with mature elements, and the majority of EC and teratomas with immature elements. On the basis of their findings, they concluded that GPC3 may have diagnostic value in identifying nonseminomatous components and distinguishing YSTs from other germ cell tumor subtypes.[16] Similar observations were made in the present study and GPC3 was positive and more specific in the yolk sac component. However, GPC3 was positive in 33% of immature teratomas in our study which was also observed and explained by Zynger et al. that expression of GPC3 in extraembryonic testicular germ cell tumors may occur as the cells acquire a fetal or placental phenotype and could explain why some immature teratomatous elements were positive whereas all teratomas with mature components were negative.[16]
Alpha Feto-Protein (AFP) is generally not a very sensitive marker of yolk sac differentiation, showing positive staining in only 55%–75% of YSTs.[17] Ye and Ulbright found that AFP was much less sensitive than GPC3 in solid Yolk sac tumor (YST) with both markers being 100% specific in differentiating from seminomas. They also highlighted that despite improved sensitivity of GPC3 over AFP for solid Yolk sac tumor (YST) intensity of staining was less in comparison to other patterns.[18] AFP was not done in the present study. Zynger et al. in their study found that choriocarcinoma was another component accentuated by GPC3, with all cases demonstrating staining.[19]
CD117 (c-kit) is expressed in seminomas of the testis and dysgerminomas of the ovary, morphologically, and genetically identical tumors. Immunohistologically, the product is detectable as distinct membranous positive staining in up to 100% of cases, whereas NSGCTs are either negative or show cytoplasmic staining in occasional cells. Based on these results, it has been suggested that downregulation of c-kit might be a critical step during differentiation from seminomas to NSGCT.[20] CD117 was positive in 11 cases in the present study, in the seminomas and seminomatous component of NSGCT. CD117 was also positive in 21% yolk sac tumor (3/14 cases) and was positive in 36% of Mixed GCTs consisting of seminoma and Yolk sac tumor components (5/14 cases). Hence, it does not help in differentiation of yolk sac tumor and seminoma in mixed germ cell tumors when used in isolation.
CD30 and cytokeratin expressions are used typically in the distinction of YST vs. EC. CD30 may show very weak expression in the otherwise classical embryonal carcinoma or lost in postchemotherapy or treated embryonal carcinoma at metastatic sites.[21] OCT3/4 is reported to be a more sensitive marker of embryonal carcinoma than CD30.[22]
A number of studies recommended OCT3/4 for seminoma, GPC3, AFP and/or cytokeratin for YST, CD30 and/or cytokeratin for EC.[2,3,23,24,25] In addition, Idrees et al. also recommended D2-40 to differentiate seminoma from other GCTs.[23] Recent markers such as SOX2 for EC and SOX17 for seminoma were introduced but are not easily available.[2,25,26]
The present study used a panel of IHC using OCT3/4, CD117, GPC3, and CD30 to subtype TGCTs with frequency positivity similar to other studies [Table 4].
Table 4.
Comparison of present study with others using similar IHC panel
| Studies for comparison | IHC Markers | Seminoma | Embryonal Carcinoma | Yolk sac tumor | Mature teratoma | Immature teratoma | Choriocarcinoma |
|---|---|---|---|---|---|---|---|
| Zhang et al.[4] n=145 | OCT 3/4 | 100% | 100% | 0% | 27% | 0% | - |
| Gopalan et al.[2] n=50 | OCT 3/4 | 100% | 100% | 0% | 0% | 0% | 0% |
| CD117 | 100% | 6% | 35% | 0% | 0% | 0% | |
| CD30 | 0% | 98% | 0% | 0% | 0% | 0% | |
| Kao et al.[24] n=52 | GPC3 | 0% | - | 97% | - | - | - |
| CD117 | 98% | - | 59% | - | - | - | |
| Zynger[19] n=71 | GPC3 | 0% | 8% | 100% | 0% | 38% | 100% |
| Teng et al.[27] n=63 | OCT 3/4 | 100% | 88% | 17% | 0% | 0% | 0% |
| CD117 | 91% | 0% | 17% | 10% | 0% | 0% | |
| CD30 | 0% | 75% | 17% | 0% | 0% | 0% | |
| Present study n=36 | OCT 3/4 | 93% | 65% | 0% | 0% | 0% | 0% |
| CD117 | 92% | 0% | 21% | 0% | 0% | 0% | |
| CD30 | 0% | 96% | 0% | 0% | 0% | 0% | |
| GPC3 | 0% | 0% | 96% | 0% | 33% | 30% |
The prognosis of GCT and initial management decisions are dictated by the clinical stage of the disease, which is based on histopathological findings and pathological stage of the primary tumor, post orchidectomy serum tumor markers, and the presence and extent of metastatic disease as determined by physical examination and staging imaging studies. Radical orchidectomy establishes the histological diagnosis and primary T stage, provides important prognostic information from the tumor histology, and is curative in 80% to 85% and 70% to 80% of clinical stage 1 seminoma and clinical stage 1 NSGCT, respectively.[28] Hence, diagnosis, differentiation, and percentage of tumor components of mixed germ cell tumors have a crucial role in patient care.
CONCLUSION
Identifying the components of GCTs, especially in a mixed GCT, becomes important as it influences treatment protocols, prognosis as well as in prediction of recurrence. The present study tested a concise and affordable panel of IHC markers OCT 3/4, CD117, GPC3, and CD30 which showed good sensitivity and specificity in differentiating seminomas, YST, and EC components. Additional markers, namely β-HCG, can be used in identifying the choriocarcinoma component.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Anjanappa M, Kumar A, Mathews S, Joseph J, Jagathnathkrishna KM, James FV. Testicular seminoma: Are clinical features and treatment outcomes any different in India? Indian J Cancer. 2017;54:385–7. doi: 10.4103/ijc.IJC_100_17. [DOI] [PubMed] [Google Scholar]
- 2.Gopalan A, Dhall D, Olgac S, Fine SW, Korkola JE, Houldsworth J, et al. Testicular mixed germ cell tumors: A morphological and immunohistochemical study using stem cell markers, OCT3/4, SOX2 and GDF3, with emphasis on morphologically difficult-to-classify areas. Mod Pathol. 2009;22:1066–74. doi: 10.1038/modpathol.2009.66. [DOI] [PubMed] [Google Scholar]
- 3.Tiwana K, Nibhoria S, Yadav A, Monga T, Bajaj A. Testicular germ cell tumours: A review of genetics, morphology and immunohistochemistry. Nat J Lab Med. 2015;4:30–5. [Google Scholar]
- 4.Zhang T, Ji L, Liu B, Guan W, Liu Q, Gao Y. Testicular germ cell tumours: A clinicopathological and immunohistochemical analysis of 145 cases. Int J Clin Exp Pathol. 2018;11:4622–9. [PMC free article] [PubMed] [Google Scholar]
- 5.Chieffi P, Chieffi S. An update on newly discovered immunohistochemical biomarkers for the diagnosis of human testicular germ cell tumours. Histol Histopathol. 2014;29:999–1006. doi: 10.14670/HH-29.999. [DOI] [PubMed] [Google Scholar]
- 6.Williamson SR, Delahunt B, Magi-Galluzzi C, Algaba F, Egevad L, Ulbright TM, et al. The world health organization 2016 classification of testicular germ cell tumours: A review and update from the international society of urological pathology testis consultation panel. Histopathology. 2017;70:335–46. doi: 10.1111/his.13102. [DOI] [PubMed] [Google Scholar]
- 7.Vasdev N, Moon A, Thorpe AC. Classification, epidemiology and therapies for testicular germ cell tumours. Int J Dev Biol. 2013;57:133–9. doi: 10.1387/ijdb.130031nv. [DOI] [PubMed] [Google Scholar]
- 8.Fosså SD, Cvancarova M, Chen L, Allan AL, Oldenburg J, Peterson DR, et al. Adverse prognostic factors for testicular cancer-specific survival: A population-based study of 27,948 patients. J Clin Oncol. 2011;29:963–70. doi: 10.1200/JCO.2010.32.3204. [DOI] [PubMed] [Google Scholar]
- 9.Chaganti RS, Houldsworth J. Genetics and biology of adult human male germ cell tumors. Cancer Res. 2000;60:1475–82. [PubMed] [Google Scholar]
- 10.Hansis C, Grifo JA, Krey LC. Oct-4 expression in inner cell mass and trophectoderm of human blastocysts. Mol Hum Reprod. 2000;6:999–1004. doi: 10.1093/molehr/6.11.999. [DOI] [PubMed] [Google Scholar]
- 11.Okamoto K, Okazawa H, Okuda A, Sakai M, Muramatsu M, Hamada H. A novel octamer binding transcription factor is differentially expressed in mouse embryonic cells. Cell. 1990;60:461–72. doi: 10.1016/0092-8674(90)90597-8. [DOI] [PubMed] [Google Scholar]
- 12.Looijenga LH, Stoop H, de Leeuw HP, de Gouveia Brazao CA, Gillis AJ, van Roozendaal KE, et al. POU5F1 (OCT3/4) identifies cells with pluripotent potential in human germ cell tumors. Cancer Res. 2003;63:2244–50. [PubMed] [Google Scholar]
- 13.Cao D, Li J, Guo CC, Allan RW, Humphrey PA. SALL4 is a novel diagnostic marker for testicular germ cell tumors. Am J Surg Pathol. 2009;33:1065–77. doi: 10.1097/PAS.0b013e3181a13eef. [DOI] [PubMed] [Google Scholar]
- 14.Santagata S, Ligon KL, Hornick JL. Embryonic stem cell transcription factor signatures in the diagnosis of primary and metastatic germ cell tumors. Am J Surg Pathol. 2007;31:836–45. doi: 10.1097/PAS.0b013e31802e708a. [DOI] [PubMed] [Google Scholar]
- 15.Hattab EM, Tu PH, Wilson JD, Cheng L. OCT4 immunohistochemistry is superior to placental alkaline phosphatase (PLAP) in the diagnosis of central nervous system germinoma. Am J Surg Pathol. 2005;29:368–71. doi: 10.1097/01.pas.0000149709.19958.a7. [DOI] [PubMed] [Google Scholar]
- 16.Zynger DL, Dimov ND, Luan C, Teh BT, Yang XJ. Glypican 3: A novel marker in testicular germ cell tumors. Am J Surg Pathol. 2006;30:1570–5. doi: 10.1097/01.pas.0000213322.89670.48. [DOI] [PubMed] [Google Scholar]
- 17.Emerson RE, Ulbright TM. The use of immunohistochemistry in the differential diagnosis of tumours of the testis and paratestis. Semin Diagn Pathol. 2005;22:33–50. doi: 10.1053/j.semdp.2005.11.003. [DOI] [PubMed] [Google Scholar]
- 18.Ye H, Ulbright TM. Difficult differential diagnosis in testicular pathology. Arch Pathol Lab Med. 2002;136:435–66. doi: 10.5858/arpa.2011-0475-RA. [DOI] [PubMed] [Google Scholar]
- 19.Zynger DL, McCallum JC, Luan C, Chou PM, Yang XJ. Glypican 3 has a higher sensitivity than alpha-fetoprotein for testicular and ovarian yolk sac tumour: Immunohistochemical investigation with analysis of histological growth patterns. Histopathology. 2010;56:750–7. doi: 10.1111/j.1365-2559.2010.03553.x. [DOI] [PubMed] [Google Scholar]
- 20.Tickoo SK, Hutchinson B, Bacik J, Mazumdar M, Motzer RJ, Bajorin DF, et al. Testicular seminoma: A clinicopathologic and immunohistochemical study of 105 cases with special reference to seminomas with atypical features. Int J Surg Pathol. 2002;10:23–32. doi: 10.1177/106689690201000105. [DOI] [PubMed] [Google Scholar]
- 21.Berney DM, Shamash J, Pieroni K, Oliver RT. Loss of CD30 expression in metastatic embryonal carcinoma: The effects of chemotherapy? Histopathology. 2001;39:382–5. doi: 10.1046/j.1365-2559.2001.01226.x. [DOI] [PubMed] [Google Scholar]
- 22.Liu DL, Lu YP, Shi HY, Zhu SX, Lü JW, Li WF, et al. Expression of CD117 in human testicular germ cell tumors and its diagnostic value for seminoma and nonseminoma. Zhonghua Nan Ke Xue. 2008;14:38–41. [PubMed] [Google Scholar]
- 23.Idrees M, Saxena R, Cheng L, Ulbright TM, Badve S. Podoplanin, a novel marker for seminoma: A comparison study evaluating immunohistochemical expression of podoplanin and OCT3/4. Ann Diagn Pathol. 2010;14:331–6. doi: 10.1016/j.anndiagpath.2010.05.008. [DOI] [PubMed] [Google Scholar]
- 24.Kao CS, Idrees MT, Young RH, Ulbright TM. Solid pattern yolk sac tumor: A morphologic and immunohistochemical study of 52 cases. Am J Surg Pathol. 2012;36:360–7. doi: 10.1097/PAS.0b013e31823c510b. [DOI] [PubMed] [Google Scholar]
- 25.de Jong J, Stoop H, Gillis AJ, van Gurp RJ, van de Geijn GJ, Boer MD, et al. Differential expression of SOX17 and SOX2 in germ cells and stem cells has biological and clinical implications. J Pathol. 2008;215:21–30. doi: 10.1002/path.2332. [DOI] [PubMed] [Google Scholar]
- 26.Nonaka D. Differential expression of SOX2 and SOX17 in testicular germ cell tumours. Am J Clin Pathol. 2004;28:1163–8. doi: 10.1309/AJCP7MNCNBCRN8NO. [DOI] [PubMed] [Google Scholar]
- 27.Teng LH, Lu DH, Xu QZ, Fu YJ, Yang H, He ZL. Expression and diagnostic significance of OCT4, CD117 and CD30 in germ cell tumors. Zhonghua bing li xue za zhi= Chinese journal of pathology. 2005 Nov 1;34(11):711–5. [PubMed] [Google Scholar]
- 28.Partin A, Peters C, Kavoussi L, Dmochowski R, Wein A. 12th ed. Philadelphia: Campbell 12edition Elsevier; 2020. Campbell Walsh Wein Urology; pp. 1686–7. [Google Scholar]


