Abstract
Skeletal muscle‐related symptoms are common in both acute coronavirus disease (Covid)‐19 and post‐acute sequelae of Covid‐19 (PASC). In this narrative review, we discuss cellular and molecular pathways that are affected and consider these in regard to skeletal muscle involvement in other conditions, such as acute respiratory distress syndrome, critical illness myopathy, and post‐viral fatigue syndrome. Patients with severe Covid‐19 and PASC suffer from skeletal muscle weakness and exercise intolerance. Histological sections present muscle fibre atrophy, metabolic alterations, and immune cell infiltration. Contributing factors to weakness and fatigue in patients with severe Covid‐19 include systemic inflammation, disuse, hypoxaemia, and malnutrition. These factors also contribute to post‐intensive care unit (ICU) syndrome and ICU‐acquired weakness and likely explain a substantial part of Covid‐19‐acquired weakness. The skeletal muscle weakness and exercise intolerance associated with PASC are more obscure. Direct severe acute respiratory syndrome coronavirus (SARS‐CoV)‐2 viral infiltration into skeletal muscle or an aberrant immune system likely contribute. Similarities between skeletal muscle alterations in PASC and chronic fatigue syndrome deserve further study. Both SARS‐CoV‐2‐specific factors and generic consequences of acute disease likely underlie the observed skeletal muscle alterations in both acute Covid‐19 and PASC.
Keywords: Covid‐19, Skeletal muscle, Inflammation, Muscle wasting, Metabolism
Introduction
There is a clear heterogeneity in the clinical presentation of patients with coronavirus disease (Covid)‐19. 1 , 2 The majority of patients with Covid‐19 infections are asymptomatic or only experience mild symptoms, do not require hospitalization, and recover relatively quickly. 3 Also, the majority of those with more severe Covid‐19 survive hospitalization. 4 A substantial proportion of post‐Covid‐19 patients, however, suffers from persistent symptoms such as fatigue and exercise intolerance. 5 Recently, this condition has been termed ‘post‐acute sequelae of Covid‐19’ (PASC). At 4–7 months following initial onset of Covid‐19 in hospitalized patients, the most common symptoms reported in patients with PASC were dyspnoea, fatigue, and muscle weakness, each seen in approximately 53–63% of patients. 6 , 7 , 8 These symptoms were more common in women and patients with higher disease severity of the acute Covid‐19 episode, whereas dyspnoea was not explained by long‐term pulmonary abnormalities. 6
Skeletal muscle‐related symptoms are common in both acute Covid‐19 and PASC. These include muscle pain (myalgia), muscle weakness (mild to severe), fatigue, and exercise intolerance. 9 , 10 Myalgia and arthralgia are also common persistent symptoms in patients with PASC and are more notable in patients who were prone positioned during intensive care unit (ICU) admission. 11
The high prevalence of skeletal muscle‐related symptoms hints towards structural and functional alterations in skeletal muscle in patients with Covid‐19. 10 Due to the heterogeneity of patients and their clinical presentations, these muscle‐related symptoms may vary substantially and are likely to be multifactorial in nature. Patients with Covid‐19 may develop an acute myopathy during their acute viral illness, although the myopathy itself is rarely the presenting symptom, 12 , 13 and muscle weakness can range from mild to severe. Furthermore, during hospitalization, patients with severe Covid‐19 are susceptible to the development of an acute myopathy, known as critical illness myopathy. 14 , 15 Risk factors to developing critical illness myopathy include respiratory failure, severity of the underlying illness, the need for mechanical ventilation, the administration of intravenous corticosteroids or neuromuscular junction blocking agents, and sepsis, most of which are commonly encountered in hospitalized patients with Covid‐19. 16 , 17 It remains unclear whether Covid‐19‐related myopathy and critical illness myopathy have a common underlying mechanism. Other risk factors such as systemic inflammation, hypoxaemia, extended periods of (forced) inactivity or bed rest, and various medications may promote or exacerbate muscle weakness, fatigue, and exercise intolerance in Covid‐19 patients. 10 , 18 , 19 , 20
In this narrative review, we focus on the skeletal muscle alterations in acute Covid‐19 and PASC. We highlight key clinical findings, describe the cellular and molecular pathways that are involved, and discuss these in the light of the severe acute respiratory syndrome coronavirus (SARS‐CoV) outbreak of 2003, acute respiratory distress syndrome (ARDS), pneumonitis, post‐intensive care syndrome, critical illness myopathy, and post‐viral fatigue syndrome. Subsequently, we discuss risk factors and confounding factors in acute Covid‐19 and PASC.
Structure and function of the neuromuscular system in Covid‐19
This section highlights the specific muscular alterations found in patients with Covid‐19, with particular focus on hospitalized patients with acute Covid‐19, as the majority of current studies included patients that required (intensive) hospital care or were deceased.
Skeletal muscle weakness
Patients admitted to the ICU with Covid‐19 show significant reductions in skeletal muscle mass and strength over the course of their hospital stay. 20 Bedridden, intensive‐care patients with severe Covid‐19 showed a 30% decrease in cross‐sectional area of the rectus femoris, with a reduction in thickness of the anterior compartment of the quadriceps muscle of almost 20% after 10 days. 20 Knee‐extensor and arm‐flexor weakness was observed in 75–85% of a cohort of 41 previously hospitalized patients (40–88 years old) recovering from Covid‐19. 21 These values for the Covid‐19 cohort were comparable with those seen in patients with ARDS, who failed to recover completely even 5 years after initial hospitalization. 22 In hospitalized patients with Covid‐19, the impairment in quadriceps strength was significantly associated with a longer hospital stay. In other cohorts, muscle strength negatively correlated with the length of stay in the ICU. 21 , 23 In general, hospitalized patients, particularly those that require intensive care, require a long rehabilitation process after discharge, 24 and low muscle mass is an independent risk factor for mortality. 25 It is therefore not surprising that patients with Covid‐19 with pre‐existing sarcopenia took twice as long to discharge and had an eight times higher mortality rate than those without sarcopenia. 26 While it remains unclear whether the SARS‐CoV‐2 virus directly causes muscle atrophy and weakness, muscle weakness is an evident clinical symptom in patients with Covid‐19.
The Covid‐19‐induced muscle weakness closely resembles that seen in ICU‐acquired weakness, which extends hospital stay and mortality in the short term and results in sustained physical disability in the long term. 27 Although comparative studies are needed, it is reasonable to expect that the skeletal muscle alterations in a subgroup of hospitalized patients with Covid‐19 fall under this broader category of generic ICU‐acquired weakness. Some of the major risk factors for intensive care‐acquired muscle weakness are time spent at ICU, age, female sex, systemic inflammation, and sepsis. 28 Muscle weakness can originate from muscle atrophy, reduced specific tension (force relative to size), or both. Qualitative differences in muscle tissue (i.e. fibrosis, fat infiltration, and a selective loss of myosin) and impairments in the neuromuscular activation can contribute to a lower specific tension. The increased echogenicity in rectus femoris muscle in severe Covid‐19 patients suggests that indeed qualitative tissue impairments, potentially due to fat infiltration and fibrosis, contribute to muscle weakness. 20
The molecular mechanisms of ICU‐acquired atrophy include an activation of the muscle ubiquitin‐proteasome pathway in the diaphragm and limb muscles of patients on mechanical ventilation, 29 as atrogin and muscle ring finger‐1 (MuRF1) are elevated in septic ICU patients. 30 Hyperactivation of autophagy contributes to muscle loss in critical illness myopathy, as it does in cancer cachexia, sepsis, chemotherapy, disuse, and chronic obstructive pulmonary disease (COPD). 31
Nerve function impairments in acute Covid‐19
A wide spectrum of neurological complications have been described in patients with Covid‐19 32 , 33 , 34 , 35 as well as in Middle East respiratory syndrome (MERS)‐related coronavirus. 36 The reported sensory loss and paresthesia, along with the muscle weakness, are in line with an acute inflammatory sensory and motor polyradiculoneuropathy or Guillain‐Barré syndrome. This syndrome is thought to be due to viral antigen mimicry resulting in an immune response in peripheral nerves ultimately leading to muscle denervation. 37
Neuromuscular problems are common ICU‐acquired complications, affecting about 40% of all patients, 38 and include a decreased muscle membrane excitability. 39 This causes a prolonged duration of the compound muscle action potential (CMAP) and slowing of motor conduction velocities. 39 Prolonged CMAP duration is one of the most characteristic indicators of critical illness myopathy. 39 Decreased muscle membrane excitability, slowing of nerve conduction velocities, and axonal degeneration directly impact skeletal muscle activation, and thus function. 40 This reduction in the ability of maximal voluntary activation causes muscle weakness independent from a loss of muscle mass. Molecular pathways of axonal degeneration in critical illness remain to be clarified, but pro‐inflammatory and neurotoxic mediators produced through nuclear factor‐κB (NF‐κB)‐regulated inflammation signalling have been implicated. 40 As such, it is likely that peripheral nerve damage contributes to a lower voluntary muscle activation in patients with Covid‐19.
Muscle histopathologic alterations
Data on histopathologic alterations in patients with Covid‐19 or PASC are scarce. An unpublished case from a middle‐aged female patient with Covid‐19‐related critical illness myopathy, but no other comorbidities, is presented in Figure 1. The histopathological equivalent of cell death is the presence of necrotic fibres (Figure 1A). Necrosis results from toxic, inflammatory or mechanical injury, and infiltrating immune cells (mainly macrophages) clean up the remaining debris. Muscle fibre regeneration, and atrophy (Figure 1A + 1B), disruption in sarcomeric ultrastructure, and myosin loss (myosinolysis; Figure 1C and 1D) are seen in this patient with Covid‐19. A selective loss of myosin directly impairs crossbridge cycling, force production, and specific tension. These observations resemble those seen in critical illness myopathy, 41 , 42 , 43 , 44 and are cellular explanations of muscle weakness in patients with Covid‐19. Skeletal muscle from deceased patients following the 2003 outbreak of SARS‐CoV‐related pneumonitis also showed a heterogeneous presentation of myofibre necrosis, muscle atrophy, myofibrillar disarray, and loss of Z disk organization. 45 Circulating creatine kinase levels are classically elevated in Covid‐19 up to levels comparable with those seen in rhabdomyolysis, 46 indicative of rapid skeletal muscle breakdown. Activation of the ubiquitin‐proteasome pathway and calpain activation have been suggested as possible contributors to the observed proteolysis and atrophy in critically ill patients, 47 but it is unclear if Covid‐19 can also activate these pathways independently. 48
Figure 1.
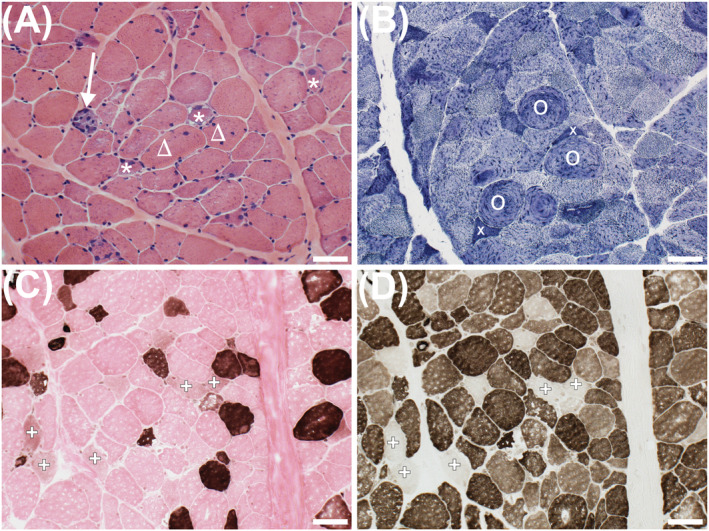
Frozen sections of the vastus lateralis from a young female patient hospitalized with Covid‐19. Haematoxylin and eosin stain (A) shows a necrotic fibre replaced by macrophages (arrow), regenerating fibres (stars), one of which has subsarcolemmal vacuoles, and fibres with internalized nuclei (triangles). NADH‐reacted section (B) shows atrophic angulated fibres (x) and a disrupted mitochondrial network (O). ATPase‐reacted sections show type I fibres (dark at pH 4.3; C). At pH 9.4 (D), both type I and type II fibres are stained. Failed staining indicates the absence of functional myosin ATPase, indicative of myosinolysis (+; C, D). Scale bar = 50 μm.
A comprehensive histopathological, virological, immunological, and ultrastructural study of skeletal muscle samples from people who died of severe Covid‐19 showed substantial infiltration of leukocytes, T‐cells, and natural killer cells compared with critically‐ill patients without Covid‐19. 46 This suggests a clear contribution of immune cell involvement in severe Covid‐19.
We provide evidence of the occurrence of internalized nuclei in a patient with Covid‐19 (Figure 1A). Indeed, signs of degenerating muscle fibres were more frequent in autopsy material from deceased Covid‐19 patients. 46 Internalized nuclei are a histological indication that muscle regeneration has occurred and are seen in critical illness myopathy 49 and COPD. 50 It is likely that regeneration and repair of injured cells are required to fully recover from Covid‐19‐related muscle alterations.
An ultrastructural analysis of capillaries showed thickening and multiple layers of basement membranes in patients who died of Covid‐19. 46 Endothelial cells frequently had swollen mitochondria and a granular appearance of cytoplasm with numerous ribosomes, indicative of ongoing regenerative processes, 46 which has been previously observed in severe acute respiratory syndrome (SARS). 51 These observations confirm a regenerative process after a primary injury in acute Covid‐19. Despite these observations, it remains unknown whether the skeletal muscle regenerative capacity is impaired in Covid‐19, as seen in other diseases such as COPD. 50
Fibrosis
Pulmonary fibrosis is an important clinical observation in patients with severe Covid‐19 and is strongly associated with dysregulation of the transforming growth factor β (TGF‐β) pathway following SARS‐CoV‐2 infection. 52 Members of the TGF‐β pathway have also been implicated in skeletal muscle fibrosis. 53 Autopsies from 26 patients with Covid‐19 showed an increased expression of angiotensin I‐converting enzyme 2 (ACE2) and genes involved in fibrosis, as well as a two‐fold higher degree of fibrosis in the diaphragm muscle, compared with ICU patients without Covid‐19. 29 This fibrosis contributes to a lower specific force generating capacity of the diaphragm and possibly to dyspnoea. 29 Whether similar results are observed in limb skeletal muscles in patients with Covid‐19 is currently unknown. Patients recovering from critical illness myopathy have a distinct gene expression signature where genes involved in skeletal muscle regeneration and extracellular matrix deposition are altered, contributing to the development of fibrosis. 27 This may be an important factor to understand the persistent muscle weakness in patients with PASC.
Exercise intolerance and metabolic alterations
Daily activities require not only an adequate skeletal muscle mass to produce force and power but also an optimal metabolic capacity to sustain function. As fatigue and exercise intolerance are common clinical symptoms in patients with Covid‐19 and PASC, it is possible that metabolic function of the skeletal muscle is impaired. 54 Post‐Covid‐19 patients who had been mechanically ventilated for a median of 13 days showed poor cardiorespiratory fitness during clinical rehabilitation, with peak oxygen uptake corresponding to ~50% of predicted values. 55 After 3 months post‐hospital discharge, recovering patients with Covid‐19 were primarily limited by reduced muscle mass, low oxidative capacity or both, rather than by a cardiac or ventilatory exercise limitation. 55
The ACE2 is generally regarded as the host cell entry point of SARS‐CoV‐2. 56 Because ACE2 is expressed in endothelial cells, endothelial dysfunction has been implicated in the pathophysiology of Covid‐19. 57 SARS‐CoV‐2‐infected endothelial cells lead to vascular inflammation and increased permeability, ultimately culminating in a pro‐coagulant, pro‐inflammatory, and pro‐oxidant condition that contributes to endothelial injury and reduces systemic perfusion and oxygen delivery. 58 Indeed, skeletal muscle flow‐mediated dilation during exercise is reduced in young adults with Covid‐19 indicating a lower vascular function and a higher arterial stiffness compared with healthy controls, 59 even in a case study of a young pre‐symptomatic Covid‐19 patient. 60 Autopsy material from patients with SARS showed oedema and infiltration of monocytes and lymphocytes in vessel walls and the circumference of small veins and arteries in striated muscles in the lower limbs. 51
Mitochondrial dysfunction is not well studied in patients with Covid‐19 or PASC. A lower ADP‐stimulated mitochondrial respiration and an attenuated sensitivity to ADP have been observed in a young Covid‐19 patient before symptoms developed, suggestive of early alterations in skeletal mitochondrial function and substrate utilization. 60 Our example from Figure 1 also shows a disrupted mitochondrial network (Figure 1B), but it is unclear if mitochondrial density and respiration are altered.
Indirect evidence supports the presence of mitochondrial stress in patients with Covid‐19. Growth differentiation factor 15 (GDF15) is released from skeletal muscle upon activation of the mitochondrial unfolded protein response, which is activated upon mitochondrial proteotoxic stress. 61 Circulating GDF15 concentrations were elevated in ~80% of patients hospitalized with Covid‐19, compared with age‐stratified healthy individuals, and higher concentrations were associated with detectable SARS‐CoV‐2 viremia, hypoxaemia, soluble ACE2 levels, and a higher risk for mortality and cardiovascular disease. 62 The source of GDF15 in Covid‐19 is currently unknown. Release of the myokine GDF15 has been associated with skeletal muscle bioenergetic dysfunction in older people with sarcopenia 63 and sepsis. 64 Plasma GDF15 levels and gene expression in the skeletal muscle are also elevated in patients with intensive care‐acquired weakness. 65 GDF15 acts to increase mitochondrial function, fatty acid oxidation, and insulin sensitivity and can also reduce food intake. 61 How GDF15 contributes to skeletal muscle dysfunction and other symptoms in patients with Covid‐19 warrants further study.
Mitochondrial dysfunction and metabolic alterations are typically observed in patients with critical illness myopathy. 66 Suppression of fatty acid oxidation and NADH‐linked (mitochondrial complex I‐supported) respiration occur within days of admission to the ICU. 67 Sepsis triggers profound long‐lasting ultrastructural defects in skeletal muscle mitochondria. 68 , 69 Critical illness is also associated with the loss of mitochondrial complex subunits and lower expression of peroxisome‐proliferator activated receptor gamma co‐activator (PGC)‐1α, 70 suggesting impaired mitochondrial biogenesis. Mitochondrial dysfunction in critical illness myopathy 69 alters the cellular ATP/AMP balance and activates AMPK, which subsequently enhances the transcriptional activity of FoxO3 to induce muscle atrophy. 71 Similarly, alterations in the molecular machinery of mitochondrial dynamics (increasing fission and decreasing fusion) contribute to muscle atrophy. 72
Recovery from critical illness is strongly associated with restoration of oxidative phosphorylation 67 and the activation of mitochondrial biogenesis, 70 with eventual survivors showing higher intramuscular ATP content than non‐survivors. 69 Low muscle mitochondrial content in ICU survivors contributes to long‐term weakness. 73 Whether more subtle alterations in oxidative phosphorylation, metabolic flexibility, or mitochondrial redox homeostasis persist in Covid‐19 remains to be determined.
Factors contributing to muscle weakness and exercise intolerance in patients with Covid‐19
The cause of the exercise intolerance and muscle atrophy seen in patients with Covid‐19 and PASC is likely multifactorial. While it is clear that multiple comorbidities exacerbate Covid‐19 disease severity and symptoms, 74 , 75 Covid‐19 might act as a second hit, exacerbating muscle weakness and exercise intolerance. Contributing factors are probably similar to those seen in other chronic diseases, critical illness myopathy, and ageing. We describe the most widely acknowledged factors here, namely, systemic inflammation, enforced physical inactivity or disuse, viral infiltration, hypoxaemia, malnutrition, and certain medications (Figure 2). Increasing evidence shows that not all patients with PASC were hospitalized during the acute infection. 76 This suggests that other factors than those associated with critical illness may contribute to muscle weakness and exercise intolerance.
Figure 2.
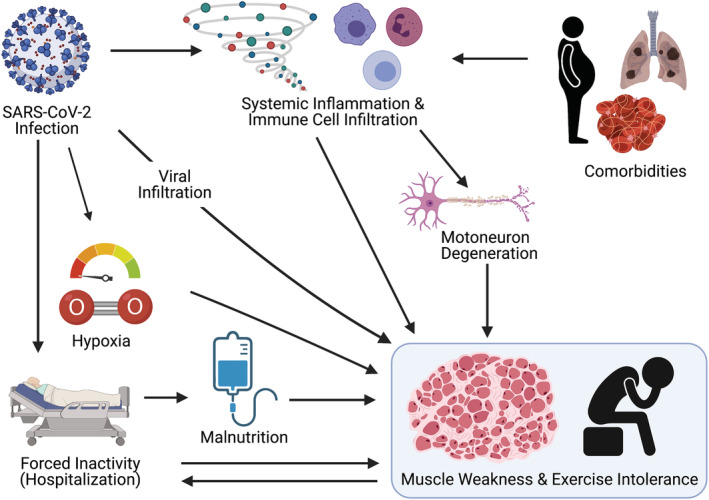
A graphical overview of the factors that contribute to muscle weakness and fatigue in patients with Covid‐19. Severe SARS‐CoV‐2 infection is characterized by a dysregulated host response that can directly affect neuronal function and various inflammatory cytokines and immune cells contributing to muscle atrophy. The concomitant physical inactivity and, in some cases, hypoxaemia and malnutrition are additional factors that likely contribute to alterations in skeletal muscle structure and function. Impaired muscle weakness and exercise intolerance lead to further inactivity at vicious cycle.
Systemic inflammation
Systemic inflammation plays a critical role in Covid‐19 morbidity and disease progression. 77 , 78 Blood plasma levels of C‐reactive protein (CRP), interleukin (IL)‐1β, IL‐6, IL‐8, IL‐10, IL‐2 receptor, soluble tumour necrosis factor receptor‐1 (sTNFR1), and tumour necrosis factor‐α (TNF‐α) were higher in severe cases of Covid‐19 compared with those showing mild symptoms. 79 , 80 These increased levels of pro‐inflammatory mediators cause a so‐called ‘cytokine storm’, contributing to excessive and uncontrolled inflammation. 78 The inflammatory profile of patients with severe Covid‐19 is different to other inflammatory muscle wasting conditions seen in, for instance, diabetes, cancer, or COPD, 75 , 81 , 82 , 83 likely because of the viral nature of the disease. Although the link between systemic inflammation and muscle wasting has been widely studied in other diseases and conditions, 81 , 82 , 84 the link between circulating cytokines, muscle wasting and altered metabolism has not yet been made in patients with Covid‐19. It is, however, likely that the cytokine storm of pro‐inflammatory mediators plays an important role in the development of skeletal muscle atrophy and altered metabolic function in acute patients with Covid‐19.
Interleukin‐6 has been shown to be the best early biomarker for disease severity, followed by CRP, IL‐10, IL‐8, and IL‐1β. 80 , 85 Results from patients with severe SARS and MERS have suggested that elevated levels of IL‐6 are associated with long‐term dependency on mechanical ventilation—likely through disease severity—and may underpin a mechanistic link to the poor clinical outcome of patients with severe Covid‐19 after a prolonged stay at the ICU. 86 IL‐6, which is markedly elevated in patients with Covid‐19, has pleiotropic pro‐inflammatory effects. 78 Elevated levels of IL‐6 in skeletal muscle promote both uptake and oxidation of glucose and fat oxidation and alter insulin signalling. 87 Further, it has been suggested that IL‐6 may acutely alter brain serotonergic function and motor neuron activation, reducing exercise capacity in healthy athletes, independent of other contributing factors. 88
Systemic TNF‐α infusion in healthy volunteers increased protein breakdown and inhibited muscle protein synthesis. 89 Activation of TNF‐α receptors induce the NF‐κB pathway, regulating expression of the muscle‐specific ubiquitin ligase MuRF‐1, 30 , 82 , 90 which is involved in protein breakdown. NF‐κB also lowers the skeletal muscle regenerative potential by inhibition of satellite cell activation. 30 , 90 TNF‐α induces an NF‐κB‐dependent reduction in muscle oxidative capacity and transcription of regulators of mitochondrial biogenesis. 91 Lastly, NF‐κB contributes to a loss of motoneuron excitation by inducing axonal degradation. 92
C‐reactive protein (CRP) can induce cellular apoptosis, phagocytosis, and lower muscle protein synthesis, 84 and CRP levels correlate with Covid‐19‐related cachexia. 48 Because CRP does not consistently go back to pre‐infection levels 3 months after infection in patients with Covid‐19, 5 this may have implications for understanding PASC.
Other, less well‐studied cytokines that are elevated in Covid‐19 also promote muscle atrophy, metabolic alterations, and neural dysfunction, such as IL‐2 receptor. 40 Further research is clearly needed to confirm which inflammatory factors contribute to muscle wasting and dysfunction in patients with Covid‐19 and PASC and may provide further mechanistic insight in the cellular and molecular pathways leading to skeletal muscle alterations in Covid‐19.
Viral infiltration
Deceased patients with Covid‐19 show more myositis in skeletal muscle than patients dying without Covid‐19. 29 , 46 However, while a recent case report described the detection of viral particles in the skeletal muscle 5 weeks following the infection, 93 in a study of autopsy material from deceased patients with Covid‐19, the detection of viral load was low or negative in most skeletal and cardiac muscles, probably due to circulating viral RNA rather than genuine infection of myocytes. 46 Nevertheless, a direct cytotoxic effect of viral particles on the skeletal muscle cannot be excluded. Viral particles were also found in diaphragm muscle of deceased patients with severe Covid‐19, 29 and post mortem examinations of patients who succumbed to SARS in 2005 showed the presence of viral particles in atrophic skeletal muscle fibres. 45
While it has been unclear whether viral infiltration is a contributing factor for skeletal muscle adaptations, viral infection in general is known to modulate mitochondrial function and alter gene expression levels of the host cell. 94 Different viruses affect mitochondrial dynamics through mitochondrial fusion or fission proteins and can induce mitophagy to clear damaged mitochondria to enhance cell survival and viral persistence, which is associated with physiological alterations such as oxidative stress, hypoxia, and dysregulated calcium homeostasis. 94 , 95 Clearly, more fundamental and clinical work is required to better understand viral infiltration of skeletal muscle and the possible functional consequences.
Muscle disuse
Disuse presents a major risk factor for patients with severe Covid‐19 who require intensive care and mechanical ventilation. 20 Physical inactivity or (forced) disuse alters muscle contractile properties and metabolic health, leading to further physical inactivity and a vicious cycle. Low physical activity also significantly increases the risk of hospitalization, ICU admission, and mortality. 96
de Andrade‐Junior et al. 20 observed a ~30% reduction in rectus femoris cross‐sectional area in patients with severe Covid‐19 after ~10 days, which is larger than seen after disuse only. During disuse, muscle atrophy appears rapidly and in a non‐linear manner, with a decline in muscle mass of 5% after short‐term (10 days) and up to ~20% after long‐term disuse (6 weeks). 97 This disuse‐related loss of muscle mass and size accounted for ~80% of a decrease in strength of the knee extensor muscles, with the remaining 20% being due to qualitative alterations, such as a lower specific tension. 97 A lower force generating capacity and power is largely attributable to a loss of contractile material due to a decrease in the cross‐sectional area of single muscle fibres. 19 , 97 It is particularly the fast‐twitch, type II muscle fibres that show more atrophy. 30 Because type II fibres produce up to five times more power than type I fibres, 98 this specific type II fibre atrophy results in larger declines in power, thus impacting activities of daily living more than a loss of strength per se. Type II atrophy or a lower specific tension are not yet reported in patients with Covid‐19 but are described in ICU‐acquired weakness. 99
Not only does disuse cause a loss of muscle mass and strength, disuse also induces alterations in metabolism that ultimately result in increased fatigability, exercise intolerance, and lower VO2max. 100 , 101 Short‐term bed rest resulted in impairments in peripheral oxygen delivery and altered metabolic pathways upstream of mitochondrial respiration, 102 but the location and cause of these are unknown. A lower mitochondrial density and oxidative phosphorylation capacity have been observed following prolonged physical inactivity. 101 Disuse‐induced changes in skeletal muscle mitochondrial morphology, such as mitochondrial fission, alterations in transcription, translation and import of mitochondrial proteins, as well as changes in membrane lipid profiles, reactive oxygen species emission, and impaired Ca2+ handling can underlie the lower oxidative phosphorylation capacity. 101
Hypoxaemia
Pulmonary abnormalities are present in virtually all patients with severe Covid‐19, as hypoxia is a criterion for marking Covid‐19 as severe. Hypoxia poses a challenge for tissue oxygen delivery, and common features of the human response to hypoxia include weight loss, muscle wasting, and tissue metabolic alterations. 103 The molecular response to hypoxia is primarily initiated by activation of the hypoxia‐inducible factor (HIF) family of transcription factors and associated regulatory proteins, which orchestrate angiogenesis and erythropoiesis as means to combat tissue hypoxia. 104 Stabilization of HIF1α activates the glycolysis 104 and suppresses oxidative metabolism. 105 It has been postulated that either tissue hypoxia or oxidative stress, and the consequent downstream activation of HIF signalling, might explain the metabolic responses reported in muscle samples from ICU patients and that this pathway might therefore be a novel target for therapeutic intervention. 106 There has been intense speculation on the possible roles played by HIF pathway activation in the pathogenesis of Covid‐19, 107 , 108 and these hypotheses warrant further investigation.
Malnutrition
Proteins and amino acids are important substrates for muscle build‐up and maintenance. As such, appropriate nutrition of patients with acute Covid‐19 is required. A multicentre study in the Netherlands revealed a high prevalence of malnutrition in patients with Covid‐19. 109 While 67% of patients with Covid‐19 were overweight, 36% were found to be malnourished at hospital admission. One in five patients (and more than one‐third of ICU patients) showed serious acute weight loss of more than 5 kg during hospitalization. In total, 73% of patients were at high risk of developing sarcopenia. Contributing factors to this high incidence of malnutrition were the high prevalence of nutritional complaints, including loss of appetite (58%), feeling of being full (49%), loss and change of taste (both 33%), and loss of smell (27%). Notably, just 7% of patients did not experience any nutritional complaint. Because nutritional abnormalities may persist long after discharge, dietary counselling should be considered in hospitalized patients with Covid‐19. Although it is less clear how malnutrition affects Covid‐19 outcome and recovery, it is clear that nutritional complaints in patients with Covid‐19 contribute to the weight loss observed during the acute phase of the infection. 110
Adverse effects of medication
Several medications used in the management of patients with acute Covid‐19 are known to have side effects on skeletal muscle structure and function. Chloroquine and its derivative hydroxychloroquine have been used in the first year of the Covid‐19 pandemic. Its use can lead to myopathy, characterized by the accumulation of autophagic vacuoles with abnormally increased acid phosphatase (lysosomal enzyme) reactivity, and detection of myeloid or curvilinear bodies. 111 In patients with Covid‐19 admitted to the ICU, correlations between hydrocortisone dose with handgrip strength and walking distance were indicative of possible links between medication and skeletal muscle function. 20 The absence of a correlation between hydrocortisone dose with rectus femoris cross‐sectional area, thickness of the anterior compartment of the quadriceps muscle, and echogenicity, however, suggests that confounding factors such as disease severity, duration at the ICU, and other factors may play a more important role.
Skeletal muscle adaptations in patients with post‐acute sequelae of SARS‐CoV‐2
Persisting skeletal muscle‐related symptoms may exist throughout Covid‐19 severity grades. Classically, critical illness myopathy is only seen in ICU‐admitted patients, but also non‐admitted patients with mild disease show low exercise capacity and low fat‐free mass index. 5 Symptoms in patients with PASC are remarkably similar to those seen with (post‐viral) chronic fatigue syndrome (CFS), including profound fatigue, sleep disturbances, neurocognitive changes, and post‐exertional malaise. 112 CFS, or myalgic encephalomyelitis (ME), is a debilitating disease of unknown aetiology or therapy. Viral infections are sometimes mentioned as trigger. A more pronounced immune response to exercise and altered muscle metabolic function are evident in CFS/ME, with a larger reliance on glycolysis for energy production. 113 Because exercise intolerance and muscle weakness are complex phenomena, with interaction between physical and psychological factors, 6 , 11 it is currently unknown what the possible mechanisms are that can explain these symptoms in patients with PASC. Likely, other mechanisms than those traditionally described above are involved, but no systematic study has been published yet. It is speculated that hypoxia, malnutrition, and medication may play a smaller role, and other factors such as low‐grade systemic inflammation, physical inactivity, persisting viral load, and possibly specific genotypes (such as higher skeletal muscle ACE protein content in women 114 ) might play important roles.
Improving skeletal muscle function after Covid‐19
There is not yet a consensus on optimal management strategies to improve fatigue and exercise tolerance in patients with acute Covid‐19 and PASC. Detailed clinical recommendations for hospital‐based physical therapists managing patients with Covid‐19 have been published, 115 , 116 but recommendations for exercise therapy are still suboptimal. Rehabilitation, in the form of regular physical exercise, has proven to be effective in avoiding and improving the debilitating effects of critical illness myopathy. Immediate ambulation in ICU‐admitted patients has proven to be feasible and results in positive effects. 117 , 118 Recent work has highlighted the potential effectiveness of neuromuscular electrical stimulation on the recovery of ICU‐admitted patients with Covid‐19. 119
Physical therapy after acute Covid‐19 infection may be beneficial in the respiratory and physical rehabilitation of patients with Covid‐19. Physical exercise reduces local and systemic inflammation and can induce whole‐body metabolic alterations, mainly induced by various myokines. 82 , 120 Symptoms of under‐strain and over‐strain should, however, be closely monitored in both ICU and non‐ICU Covid‐19 patients. Particularly in patients with PASC, post‐exertional malaise is a complicating factor for optimizing exercise therapy. Clearly, a better clinical and fundamental understanding of post‐exertional malaise will provide important new avenues for a more optimal, personalized rehabilitation process.
Conclusions
Muscle weakness and exercise intolerance are key symptoms of patients with acute Covid‐19 and PASC. Our current evidence base suggests that reduced force generating capacity, decreased neural activation, fibre atrophy, necrosis, fibrosis, and alterations in blood flow and metabolic function may underlie these symptoms. Contributing factors will vary between patients, but likely include systemic inflammation, viral infection, and (forced) inactivity. Comorbidities, malnutrition, hypoxaemia, and certain medications can worsen skeletal muscle atrophy and metabolic alterations in some (hospitalized) patients. The observation that a significant subgroup of patients reports clinical symptoms of exercise intolerance and muscle weakness for up to a year post‐infection suggests this to be a multifactorial problem. Clearly, future studies across larger cohorts and from different genetic backgrounds are required to completely disentangle the molecular signature of Covid‐19‐related skeletal muscle alterations and its pathogenicity. Tailored interventions based on a more in‐depth, fundamental understanding of skeletal muscle alterations and the underlying determinants are needed in order to attenuate clinical symptoms and improve quality of life in patients with acute Covid‐19 and PASC.
Conflict of interest
Madu N. Soares, Moritz Eggelbusch, Elie Naddaf, Karin H. L. Gerrits, Marike van der Schaaf, Bram van den Borst, W. Joost Wiersinga, Michele van Vugt, Peter J. M. Weijs, Andrew J. Murray, and Rob C. I. Wüst declare that they have no conflict of interest.
Acknowledgements
The authors of this manuscript certify that they comply with the ethical guidelines for authorship and publishing in the Journal of Cachexia, Sarcopenia and Muscle. 121
Soares M. N., Eggelbusch M., Naddaf E., Gerrits K. H. L., van der Schaaf M., van den Borst B., Wiersinga W. J., van Vugt M., Weijs P. J. M., Murray A. J., and Wüst R. C. I. (2022) Skeletal muscle alterations in patients with acute Covid‐19 and post‐acute sequelae of Covid‐19, Journal of Cachexia, Sarcopenia and Muscle, 13, 11–22, 10.1002/jcsm.12896
Madu N. Soares and Moritz Eggelbusch have equal contribution.
References
- 1. Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID‐19): a review. JAMA 2020;324:782–793. [DOI] [PubMed] [Google Scholar]
- 2. Sun P, Qie S, Liu Z, Ren J, Li K, Xi J. Clinical characteristics of hospitalized patients with SARS‐CoV‐2 infection: a single arm meta‐analysis. J Med Virol 2020;92:612–617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Yanes‐Lane M, Winters N, Fregonese F, Bastos M, Perlman‐Arrow S, Campbell JR, et al. Proportion of asymptomatic infection among COVID‐19 positive persons and their transmission potential: a systematic review and meta‐analysis. PLoS ONE 2020;15:e0241536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Karagiannidis C, Mostert C, Hentschker C, Voshaar T, Malzahn J, Schillinger G, et al. Case characteristics, resource use, and outcomes of 10 021 patients with COVID‐19 admitted to 920 German hospitals: an observational study. Lancet Respir Med 2020;8:853–862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. van den Borst B, Peters JB, Brink M, Schoon Y, Bleeker‐Rovers CP, Schers H, et al. Comprehensive health assessment 3 months after recovery from acute coronavirus disease 2019 (COVID‐19). Clin Infect Dis 2021;73:e1089–e1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Huang C, Huang L, Wang Y, Li X, Ren L, Gu X, et al. 6‐month consequences of COVID‐19 in patients discharged from hospital: a cohort study. Lancet 2021;397:220–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Sudre CH, Murray B, Varsavsky T, Graham MS, Penfold RS, Bowyer RC, et al. Attributes and predictors of long COVID. Nat Med 2021;27:626–631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Nalbandian A, Sehgal K, Gupta A, Madhavan MV, McGroder C, Stevens JS, et al. Post‐acute COVID‐19 syndrome. Nat Med 2021;27:601–615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Carfi A, Bernabei R, Landi F. Persistent symptoms in patients after acute COVID‐19. JAMA 2020;324:603–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Pleguezuelos E, Del Carmen A, Llorensi G, Carcole J, Casarramona P, Moreno E, et al. Severe loss of mechanical efficiency in COVID‐19 patients. J Cachexia Sarcopenia Muscle 2021;12:1056–1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Gautam N, Madathil S, Tahani N, Bolton S, Parekh D, Stockley J, et al. Medium‐term outcome of severe to critically ill patients with SARS‐CoV‐2 infection. Clin Infect Dis 2021. 10.1093/cid/ciab341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Suwanwongse K, Shabarek N. Rhabdomyolysis as a presentation of 2019 novel coronavirus disease. Cureus 2020;12:e7561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Valente‐Acosta B, Moreno‐Sanchez F, Fueyo‐Rodriguez O, Palomar‐Lever A. Rhabdomyolysis as an initial presentation in a patient diagnosed with COVID‐19. BMJ Case Rep 2020;13:e236719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Frithiof R, Rostami E, Kumlien E, Virhammar J, Fallmar D, Hultstrom M, et al. Critical illness polyneuropathy, myopathy and neuronal biomarkers in COVID‐19 patients: a prospective study. Clin Neurophysiol 2021;132:1733–1740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Pitscheider L, Karolyi M, Burkert FR, Helbok R, Wanschitz JV, Horlings C, et al. Muscle involvement in SARS‐CoV‐2 infection. Eur J Neurol 2021;28:3411–3417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Douglass JA, Tuxen DV, Horne M, Scheinkestel CD, Weinmann M, Czarny D, et al. Myopathy in severe asthma. Am Rev Respir Dis 1992;146:517–519. [DOI] [PubMed] [Google Scholar]
- 17. Latronico N, Tomelleri G, Filosto M. Critical illness myopathy. Curr Opin Rheumatol 2012;24:616–622. [DOI] [PubMed] [Google Scholar]
- 18. Baldwin KM, Haddad F. Skeletal muscle plasticity: cellular and molecular responses to altered physical activity paradigms. Am J Phys Med Rehabil 2002;81:S40–S51. [DOI] [PubMed] [Google Scholar]
- 19. Booth FW. Effect of limb immobilization on skeletal muscle. J Appl Physiol Respir Environ Exerc Physiol 1982;52:1113–1118. [DOI] [PubMed] [Google Scholar]
- 20. de Andrade‐Junior MC, de Salles ICD, de Brito CMM, Pastore‐Junior L, Righetti RF, Yamaguti WP. Skeletal muscle wasting and function impairment in intensive care patients with severe COVID‐19. Front Physiol 2021;12:640973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Paneroni M, Simonelli C, Saleri M, Bertacchini L, Venturelli M, Troosters T, et al. Muscle strength and physical performance in patients without previous disabilities recovering from COVID‐19 pneumonia. Am J Phys Med Rehabil 2021;100:105–109. [DOI] [PubMed] [Google Scholar]
- 22. Herridge MS, Tansey CM, Matte A, Tomlinson G, Diaz‐Granados N, Cooper A, et al. Functional disability 5 years after acute respiratory distress syndrome. N Engl J Med 2011;364:1293–1304. [DOI] [PubMed] [Google Scholar]
- 23. Van Aerde N, Van den Berghe G, Wilmer A, Gosselink R, Hermans G, COVID‐19 Consortium . Intensive care unit acquired muscle weakness in COVID‐19 patients. Intensive Care Med 2020;46:2083–2085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Looijaard WG, Dekker IM, Stapel SN, Girbes AR, Twisk JW, Oudemans‐van Straaten HM, et al. Skeletal muscle quality as assessed by CT‐derived skeletal muscle density is associated with 6‐month mortality in mechanically ventilated critically ill patients. Crit Care 2016;20:386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Weijs PJ, Looijaard WG, Dekker IM, Stapel SN, Girbes AR, Oudemans‐van Straaten HM, et al. Low skeletal muscle area is a risk factor for mortality in mechanically ventilated critically ill patients. Crit Care 2014;18:R12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kim JW, Yoon JS, Kim EJ, Hong HL, Kwon HH, Jung CY, et al. Prognostic implication of baseline sarcopenia for length of hospital stay and survival in patients with coronavirus disease 2019. J Gerontol A Biol Sci Med Sci 2021;76:e110–e116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Walsh CJ, Batt J, Herridge MS, Mathur S, Bader GD, Hu P, et al. Transcriptomic analysis reveals abnormal muscle repair and remodeling in survivors of critical illness with sustained weakness. Sci Rep 2016;6:29334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Yang T, Li Z, Jiang L, Wang Y, Xi X. Risk factors for intensive care unit‐acquired weakness: a systematic review and meta‐analysis. Acta Neurol Scand 2018;138:104–114. [DOI] [PubMed] [Google Scholar]
- 29. Shi Z, de Vries HJ, Vlaar APJ, van der Hoeven J, Boon RA, Heunks LMA, et al. Diaphragm pathology in critically ill patients with COVID‐19 and postmortem findings from 3 medical centers. JAMA Intern Med 2021;181:122–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Bloch S, Polkey MI, Griffiths M, Kemp P. Molecular mechanisms of intensive care unit‐acquired weakness. Eur Respir J 2012;39:1000–1011. [DOI] [PubMed] [Google Scholar]
- 31. Sartori R, Romanello V, Sandri M. Mechanisms of muscle atrophy and hypertrophy: implications in health and disease. Nat Commun 2021;12:330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Naddaf E, Laughlin RS, Klein CJ, Toledano M, Theel ES, Binnicker MJ, et al. Guillain‐Barre syndrome in a patient with evidence of recent SARS‐CoV‐2 infection. Mayo Clin Proc 2020;95:1799–1801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Scheidl E, Canseco DD, Hadji‐Naumov A, Bereznai B. Guillain‐Barre syndrome during SARS‐CoV‐2 pandemic: a case report and review of recent literature. J Peripher Nerv Syst 2020;25:204–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Andalib S, Biller J, Di Napoli M, Moghimi N, McCullough LD, Rubinos CA, et al. Peripheral nervous system manifestations associated with COVID‐19. Curr Neurol Neurosci Rep 2021;21:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Caress JB, Castoro RJ, Simmons Z, Scelsa SN, Lewis RA, Ahlawat A, et al. COVID‐19‐associated Guillain‐Barre syndrome: the early pandemic experience. Muscle Nerve 2020;62:485–491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Kim JE, Heo JH, Kim HO, Song SH, Park SS, Park TH, et al. Neurological complications during treatment of Middle East respiratory syndrome. J Clin Neurol 2017;13:227–233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Bahouth S, Chuang K, Olson L, Rosenthal D. COVID‐19 related muscle denervation atrophy. Skeletal Radiol 2021;50:1717–1721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Maramattom BV, Bhattacharjee S. Neurological complications with COVID‐19: a contemporaneous review. Ann Indian Acad Neurol 2020;23:468–476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Allen DC, Arunachalam R, Mills KR. Critical illness myopathy: further evidence from muscle‐fiber excitability studies of an acquired channelopathy. Muscle Nerve 2008;37:14–22. [DOI] [PubMed] [Google Scholar]
- 40. Mohammadi B, Schedel I, Graf K, Teiwes A, Hecker H, Haameijer B, et al. Role of endotoxin in the pathogenesis of critical illness polyneuropathy. J Neurol 2008;255:265–272. [DOI] [PubMed] [Google Scholar]
- 41. Lacomis D, Giuliani MJ, Van Cott A, Kramer DJ. Acute myopathy of intensive care: clinical, electromyographic, and pathological aspects. Ann Neurol 1996;40:645–654. [DOI] [PubMed] [Google Scholar]
- 42. al‐Lozi MT, Pestronk A, Yee WC, Flaris N, Cooper J. Rapidly evolving myopathy with myosin‐deficient muscle fibers. Ann Neurol 1994;35:273–279. [DOI] [PubMed] [Google Scholar]
- 43. Bierbrauer J, Koch S, Olbricht C, Hamati J, Lodka D, Schneider J, et al. Early type II fiber atrophy in intensive care unit patients with nonexcitable muscle membrane. Crit Care Med 2012;40:647–650. [DOI] [PubMed] [Google Scholar]
- 44. Wollersheim T, Woehlecke J, Krebs M, Hamati J, Lodka D, Luther‐Schroeder A, et al. Dynamics of myosin degradation in intensive care unit‐acquired weakness during severe critical illness. Intensive Care Med 2014;40:528–538. [DOI] [PubMed] [Google Scholar]
- 45. Leung TW, Wong KS, Hui AC, To KF, Lai ST, Ng WF, et al. Myopathic changes associated with severe acute respiratory syndrome: a postmortem case series. Arch Neurol 2005;62:1113–1117. [DOI] [PubMed] [Google Scholar]
- 46. Aschman T, Schneider J, Greuel S, Meinhardt J, Streit S, Goebel HH, et al. Association between SARS‐CoV‐2 infection and immune‐mediated myopathy in patients who have died. JAMA Neurol 2021;78:948–960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Friedrich O, Reid MB, Van den Berghe G, Vanhorebeek I, Hermans G, Rich MM, et al. The sick and the weak: neuropathies/myopathies in the critically ill. Physiol Rev 2015;95:1025–1109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Morley JE, Kalantar‐Zadeh K, Anker SD. COVID‐19: a major cause of cachexia and sarcopenia? J Cachexia Sarcopenia Muscle 2020;11:863–865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Hund E. Myopathy in critically ill patients. Crit Care Med 1999;27:2544–2547. [DOI] [PubMed] [Google Scholar]
- 50. Theriault ME, Pare ME, Lemire BB, Maltais F, Debigare R. Regenerative defect in vastus lateralis muscle of patients with chronic obstructive pulmonary disease. Respir Res 2014;15:35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Ding Y, Wang H, Shen H, Li Z, Geng J, Han H, et al. The clinical pathology of severe acute respiratory syndrome (SARS): a report from China. J Pathol 2003;200:282–289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Stukalov A, Girault V, Grass V, Karayel O, Bergant V, Urban C, et al. Multilevel proteomics reveals host perturbations by SARS‐CoV‐2 and SARS‐CoV. Nature 2021;594:246–252. [DOI] [PubMed] [Google Scholar]
- 53. Shi A, Hillege MMG, Wüst RCI, Wu G, Jaspers RT. Synergistic short‐term and long‐term effects of TGF‐β1 and 3 on collagen production in differentiating myoblasts. Biochem Biophys Res Commun 2021;547:176–182. [DOI] [PubMed] [Google Scholar]
- 54. Ayres JS. A metabolic handbook for the COVID‐19 pandemic. Nat Metab 2020;2:572–585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Blokland IJ, Ilbrink S, Houdijk H, Dijkstra JW, van Bennekom CAM, Fickert R, et al. Exercise capacity after mechanical ventilation because of COVID‐19: cardiopulmonary exercise tests in clinical rehabilitation. Ned Tijdschr Geneeskd 2020;164:D5253. [PubMed] [Google Scholar]
- 56. Tortorici MA, Veesler D. Structural insights into coronavirus entry. Adv Virus Res 2019;105:93–116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Evans PC, Rainger GE, Mason JC, Guzik TJ, Osto E, Stamataki Z, et al. Endothelial dysfunction in COVID‐19: a position paper of the ESC Working Group for Atherosclerosis and Vascular Biology, and the ESC Council of Basic Cardiovascular Science. Cardiovasc Res 2020;116:2177–2184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Jin Y, Ji W, Yang H, Chen S, Zhang W, Duan G. Endothelial activation and dysfunction in COVID‐19: from basic mechanisms to potential therapeutic approaches. Signal Transduct Target Ther 2020;5:293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Ratchford SM, Stickford JL, Province VM, Stute N, Augenreich MA, Koontz LK, et al. Vascular alterations among young adults with SARS‐CoV‐2. Am J Physiol Heart Circ Physiol 2021;320:H404–H410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Trinity JD, Craig JC, Fermoyle CC, McKenzie AI, Lewis MT, Park SH, et al. Impact of presymptomatic COVID‐19 on vascular and skeletal muscle function: a case study. J Appl Physiol 1985;2021:1961–1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Chung HK, Ryu D, Kim KS, Chang JY, Kim YK, Yi HS, et al. Growth differentiation factor 15 is a myomitokine governing systemic energy homeostasis. J Cell Biol 2017;216:149–165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Myhre PL, Prebensen C, Strand H, Roysland R, Jonassen CM, Rangberg A, et al. Growth differentiation factor 15 provides prognostic information superior to established cardiovascular and inflammatory biomarkers in unselected patients hospitalized with COVID‐19. Circulation 2020;142:2128–2137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Romanello V. The interplay between mitochondrial morphology and myomitokines in aging sarcopenia. Int J Mol Sci 2020;22:91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Fujita Y, Ito M, Ohsawa I. Mitochondrial stress and GDF15 in the pathophysiology of sepsis. Arch Biochem Biophys 2020;696:108668. [DOI] [PubMed] [Google Scholar]
- 65. Bloch SA, Lee JY, Syburra T, Rosendahl U, Griffiths MJ, Kemp PR, et al. Increased expression of GDF‐15 may mediate ICU‐acquired weakness by down‐regulating muscle microRNAs. Thorax 2015;70:219–228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. McKenna HT, Murray AJ. Reconsidering critical illness as an uncharacterised acquired mitochondrial disorder. J Intensive Care Soc 2020;21:102–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. McKenna HT, O'Brien KA, Fernandez BO, Minnion M, Tod A, McNally BD, et al. Divergent trajectories of cellular bioenergetics, intermediary metabolism and systemic redox status in survivors and non‐survivors of critical illness. Redox Biol 2021;41:101907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Brealey D, Karyampudi S, Jacques TS, Novelli M, Stidwill R, Taylor V, et al. Mitochondrial dysfunction in a long‐term rodent model of sepsis and organ failure. Am J Physiol Regul Integr Comp Physiol 2004;286:R491–R497. [DOI] [PubMed] [Google Scholar]
- 69. Brealey D, Brand M, Hargreaves I, Heales S, Land J, Smolenski R, et al. Association between mitochondrial dysfunction and severity and outcome of septic shock. Lancet 2002;360:219–223. [DOI] [PubMed] [Google Scholar]
- 70. Carre JE, Orban JC, Re L, Felsmann K, Iffert W, Bauer M, et al. Survival in critical illness is associated with early activation of mitochondrial biogenesis. Am J Respir Crit Care Med 2010;182:745–751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Batt J, dos Santos CC, Cameron JI, Herridge MS. Intensive care unit‐acquired weakness: clinical phenotypes and molecular mechanisms. Am J Respir Crit Care Med 2013;187:238–246. [DOI] [PubMed] [Google Scholar]
- 72. Romanello V, Sandri M. The connection between the dynamic remodeling of the mitochondrial network and the regulation of muscle mass. Cell Mol Life Sci 2021;78:1305–1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Dos Santos C, Hussain SN, Mathur S, Picard M, Herridge M, Correa J, et al. Mechanisms of chronic muscle wasting and dysfunction after an intensive care unit stay. A pilot study. Am J Respir Crit Care Med 2016;194:821–830. [DOI] [PubMed] [Google Scholar]
- 74. Smith RL, Soeters MR, Wüst RCI, Houtkooper RH. Metabolic flexibility as an adaptation to energy resources and requirements in health and disease. Endocr Rev 2018;39:489–517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Wüst RCI, Degens H. Factors contributing to muscle wasting and dysfunction in COPD patients. Int J Chron Obstruct Pulmon Dis 2007;2:289–300. [PMC free article] [PubMed] [Google Scholar]
- 76. Logue JK, Franko NM, McCulloch DJ, McDonald D, Magedson A, Wolf CR, et al. Sequelae in adults at 6 months after COVID‐19 infection. JAMA Netw Open 2021;4:e210830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Yang J, Zheng Y, Gou X, Pu K, Chen Z, Guo Q, et al. Prevalence of comorbidities and its effects in patients infected with SARS‐CoV‐2: a systematic review and meta‐analysis. Int J Infect Dis 2020;94:91–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Tang Y, Liu J, Zhang D, Xu Z, Ji J, Wen C. Cytokine storm in COVID‐19: the current evidence and treatment strategies. Front Immunol 2020;11:1708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Mulchandani R, Lyngdoh T, Kakkar AK. Deciphering the COVID‐19 cytokine storm: systematic review and meta‐analysis. Eur J Clin Invest 2021;51:e13429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. McElvaney OJ, McEvoy NL, McElvaney OF, Carroll TP, Murphy MP, Dunlea DM, et al. Characterization of the inflammatory response to severe COVID‐19 illness. Am J Respir Crit Care Med 2020;202:812–821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Londhe P, Guttridge DC. Inflammation induced loss of skeletal muscle. Bone 2015;80:131–142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Zhou J, Liu B, Liang C, Li Y, Song YH. Cytokine signaling in skeletal muscle wasting. Trends Endocrinol Metab 2016;27:335–347. [DOI] [PubMed] [Google Scholar]
- 83. Kumar NB. Does COVID‐19‐related cachexia mimic cancer‐related cachexia? Examining mechanisms, clinical biomarkers, and potential targets for clinical management. J Cachexia Sarcopenia Muscle 2021;12:519–522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Wahlin‐Larsson B, Wilkinson DJ, Strandberg E, Hosford‐Donovan A, Atherton PJ, Kadi F. Mechanistic links underlying the impact of C‐reactive protein on muscle mass in elderly. Cell Physiol Biochem 2017;44:267–278. [DOI] [PubMed] [Google Scholar]
- 85. Nagant C, Ponthieux F, Smet J, Dauby N, Doyen V, Besse‐Hammer T, et al. A score combining early detection of cytokines accurately predicts COVID‐19 severity and intensive care unit transfer. Int J Infect Dis 2020;101:342–345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. McClafferty B, Umer I, Fye G, Kepko D, Kalayanamitra R, Shahid Z, et al. Approach to critical illness myopathy and polyneuropathy in the older SARS‐CoV‐2 patients. J Clin Neurosci 2020;79:241–245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Glund S, Deshmukh A, Long YC, Moller T, Koistinen HA, Caidahl K, et al. Interleukin‐6 directly increases glucose metabolism in resting human skeletal muscle. Diabetes 2007;56:1630–1637. [DOI] [PubMed] [Google Scholar]
- 88. Robson‐Ansley PJ, de Milander L, Collins M, Noakes TD. Acute interleukin‐6 administration impairs athletic performance in healthy, trained male runners. Can J Appl Physiol 2004;29:411–418. [DOI] [PubMed] [Google Scholar]
- 89. Bach E, Nielsen RR, Vendelbo MH, Moller AB, Jessen N, Buhl M, et al. Direct effects of TNF‐α on local fuel metabolism and cytokine levels in the placebo‐controlled, bilaterally infused human leg: increased insulin sensitivity, increased net protein breakdown, and increased IL‐6 release. Diabetes 2013;62:4023–4029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Friedrich O. Critical illness myopathy: sepsis‐mediated failure of the peripheral nervous system. Eur J Anaesthesiol Suppl 2008;42:73–82. [DOI] [PubMed] [Google Scholar]
- 91. Remels AH, Gosker HR, Schrauwen P, Hommelberg PP, Sliwinski P, Polkey M, et al. TNF‐α impairs regulation of muscle oxidative phenotype: implications for cachexia? FASEB J 2010;24:5052–5062. [DOI] [PubMed] [Google Scholar]
- 92. Yang X, Zeng Q, Baris M, Tezel G. Transgenic inhibition of astroglial NF‐κB restrains the neuroinflammatory and neurodegenerative outcomes of experimental mouse glaucoma. J Neuroinflammation 2020;17:252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Dodig DD, Lu DJ‐Q, Gordon K. Late breaking news e‐poster presentation. Neuromuscul Disord 2020;30:S167. [Google Scholar]
- 94. Li X, Wu K, Zeng S, Zhao F, Fan J, Li Z, et al. Viral infection modulates mitochondrial function. Int J Mol Sci 2021;22:4260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Elesela S, Lukacs NW. Role of mitochondria in viral infections. Life (Basel) 2021;11:232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Sallis R, Young DR, Tartof SY, Sallis JF, Sall J, Li Q, et al. Physical inactivity is associated with a higher risk for severe COVID‐19 outcomes: a study in 48 440 adult patients. Br J Sports Med 2021;55:1099–1105. [DOI] [PubMed] [Google Scholar]
- 97. Marusic U, Narici M, Simunic B, Pisot R, Ritzmann R. Nonuniform loss of muscle strength and atrophy during bed rest: a systematic review. J Appl Physiol 1985;2021:194–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Meijer JP, Jaspers RT, Rittweger J, Seynnes OR, Kamandulis S, Brazaitis M, et al. Single muscle fibre contractile properties differ between body‐builders, power athletes and control subjects. Exp Physiol 2015;100:1331–1341. [DOI] [PubMed] [Google Scholar]
- 99. Lad H, Saumur TM, Herridge MS, Dos Santos CC, Mathur S, Batt J, et al. Intensive care unit‐acquired weakness: not just another muscle atrophying condition. Int J Mol Sci 2020;21:7840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Booth FW, Roberts CK, Laye MJ. Lack of exercise is a major cause of chronic diseases. Compr Physiol 2012;2:1143–1211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Hyatt H, Deminice R, Yoshihara T, Powers SK. Mitochondrial dysfunction induces muscle atrophy during prolonged inactivity: a review of the causes and effects. Arch Biochem Biophys 2019;662:49–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Zuccarelli L, Baldassarre G, Magnesa B, Degano C, Comelli M, Gasparini M, et al. Peripheral impairments of oxidative metabolism after a 10‐day bed rest are upstream of mitochondrial respiration. J Physiol 2021;599:4813–4829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Raguso CA, Guinot SL, Janssens JP, Kayser B, Pichard C. Chronic hypoxia: common traits between chronic obstructive pulmonary disease and altitude. Curr Opin Clin Nutr Metab Care 2004;7:411–417. [DOI] [PubMed] [Google Scholar]
- 104. Semenza GL. Hypoxia‐inducible factors in physiology and medicine. Cell 2012;148:399–408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Kim JW, Tchernyshyov I, Semenza GL, Dang CV. HIF‐1‐mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab 2006;3:177–185. [DOI] [PubMed] [Google Scholar]
- 106. McKenna HT, Murray AJ, Martin DS. Human adaptation to hypoxia in critical illness. J Appl Physiol 1985;2020:656–663. [DOI] [PubMed] [Google Scholar]
- 107. Serebrovska ZO, Chong EY, Serebrovska TV, Tumanovska LV, Xi L. Hypoxia, HIF‐1α, and COVID‐19: from pathogenic factors to potential therapeutic targets. Acta Pharmacol Sin 2020;41:1539–1546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Afsar B, Kanbay M, Afsar RE. Hypoxia inducible factor‐1 protects against COVID‐19: a hypothesis. Med Hypotheses 2020;143:109857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Wierdsma NJ, Kruizenga HM, Konings LA, Krebbers D, Jorissen JR, Joosten MI, et al. Poor nutritional status, risk of sarcopenia and nutrition related complaints are prevalent in COVID‐19 patients during and after hospital admission. Clin Nutr ESPEN 2021;43:369–376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Anker MS, Landmesser U, von Haehling S, Butler J, Coats AJS, Anker SD. Weight loss, malnutrition, and cachexia in COVID‐19: facts and numbers. J Cachexia Sarcopenia Muscle 2021;12:9–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Naddaf E, Paul P, AbouEzzeddine OF. Chloroquine and hydroxychloroquine myopathy: clinical spectrum and treatment outcomes. Front Neurol 2020;11:616075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Poenaru S, Abdallah SJ, Corrales‐Medina V, Cowan J. COVID‐19 and post‐infectious myalgic encephalomyelitis/chronic fatigue syndrome: a narrative review. Ther Adv Infect Dis 2021;8:20499361211009385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Rutherford G, Manning P, Newton JL. Understanding muscle dysfunction in chronic fatigue syndrome. J Aging Res 2016;2016:2497348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Perez‐Valera M, Martinez‐Canton M, Gallego‐Selles A, Galvan‐Alvarez V, Gelabert‐Rebato M, Morales‐Alamo D, et al. Angiotensin‐converting enzyme 2 (SARS‐CoV‐2 receptor) expression in human skeletal muscle. Scand J Med Sci Sports 2021;31:2249–2258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Felten‐Barentsz KM, van Oorsouw R, Klooster E, Koenders N, Driehuis F, Hulzebos EHJ, et al. Recommendations for hospital‐based physical therapists managing patients with COVID‐19. Phys Ther 2020;100:1444–1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Bij de Vaate E, Gerrits KHL, Goossens PH. Personalized recovery of severe COVID19: rehabilitation from the perspective of patient needs. Eur J Clin Invest 2020;50:e13325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Sommers J, Klooster E, Zoethout SB, van den Oever HLA, Nollet F, Tepaske R, et al. Feasibility of exercise testing in patients who are critically ill: a prospective, observational multicenter study. Arch Phys Med Rehabil 2019;100:239–246. [DOI] [PubMed] [Google Scholar]
- 118. Sommers J, Engelbert RH, Dettling‐Ihnenfeldt D, Gosselink R, Spronk PE, Nollet F, et al. Physiotherapy in the intensive care unit: an evidence‐based, expert driven, practical statement and rehabilitation recommendations. Clin Rehabil 2015;29:1051–1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Burgess LC, Venugopalan L, Badger J, Street T, Alon G, Jarvis JC, et al. Effect of neuromuscular electrical stimulation on the recovery of people with COVID‐19 admitted to the intensive care unit: a narrative review. J Rehabil Med 2021;53:jrm00164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. do Brito Valente AF, Jaspers RT, Wüst RCI. Regular physical exercise mediates the immune response in atherosclerosis. Exerc Immunol Rev 2021;27:42–53. [PubMed] [Google Scholar]
- 121. von Haehling S, Morley JE, Coats AJS, Anker SD. Ethical guidelines for publishing in the Journal of Cachexia, Sarcopenia and Muscle: update 2019. J Cachexia Sarcopenia Muscle 2019;10:1148–1145. [DOI] [PMC free article] [PubMed] [Google Scholar]


