Dear editor,
We read with interest the letter published recently by Costa et al. in the journal of Infection. They analyzed the difference between the viral loads of the SARS-CoV-2 Alpha and Delta variants using the parameters of clinical presentation, time to testing from symptoms onset, age and vaccination status.1 A new variant of concern (VOC), the Omicron variant (B.1.1.529), emerged in South Africa in November 2021, and rapidly spread throughout the world.2 Recent data suggest that this variant is more transmissible,3 less sensitive to vaccination,4 and causes less severe outcomes than the Delta variant.5 In vitro studies have demonstrated changes in cell entry and cellular tropism with the Omicron variant that might explain its greater transmissibility and reduced severity.6 , 7 However, clinical data comparing Delta and Omicron infections remain scarce, especially for ambulatory patients. We therefore examined the virological features of these two variants found in patients attending testing center.
All positive specimens detected at the Toulouse University Hospital drive-through testing center between December 15 and 31, 2021 were screened for SARS-CoV-2 variant. We used the Thermo Fisher® TaqPath™ COVID-19 CE-IVD RT-PCR kit (TaqPath) for SARS-CoV-2 detection and variant screening. A deletion at position 69-70 in the spike (S) gene of Omicron variant leads to a loss of detection of this target in the TaqPath assay and allows the discrimination with the Delta variant (Omicron: S-/ Delta: S+) .8 The TaqPath profiles and whole genome sequences (PacBio technology) of a subset of 560 positive specimens were 100% concordant. Viral loads (log10 copies/ml) were determined using a calibration curve obtained with the TaqPath N gene Ct values and digital droplet RT-PCR (RT-ddPCR) (BioRad, Hercules, CA). Data on patient symptomatology and vaccination status were collected at sampling.
Among the 12 949 tests performed during this period, 975 Delta variant infections (median age= 31[20–42]; 52.7% men) and 1578 Omicron infections (median age= 28[22–38]; 49.2% men) have been diagnosed. The Omicron variant was detected in 10% of SARS-CoV-2 infections between December 15 and December 19, 55% of infections during December 20–26, and in 82% of infections during December 27–31. The patients’ characteristics are shown in the Supplementary Table.
In bivariate analysis, the nasopharyngeal (NP) viral loads of patients infected with the Omicron variant were lower than those of Delta-infected patients (p = 0.04), although the Omicron-infected patients had more mild symptoms (63.2% [60.7%−79.8%]) than those infected with the Delta variant (51.8% [48.6%−55.0%]; p < 0.01). The proportion of infections in vaccinated patients (2 or 3 doses) was higher with the Omicron variant (68.7% [66.3%−82.5%]) than with the Delta variant (52.6% [49.4%−55.7%]; p < 0.01).
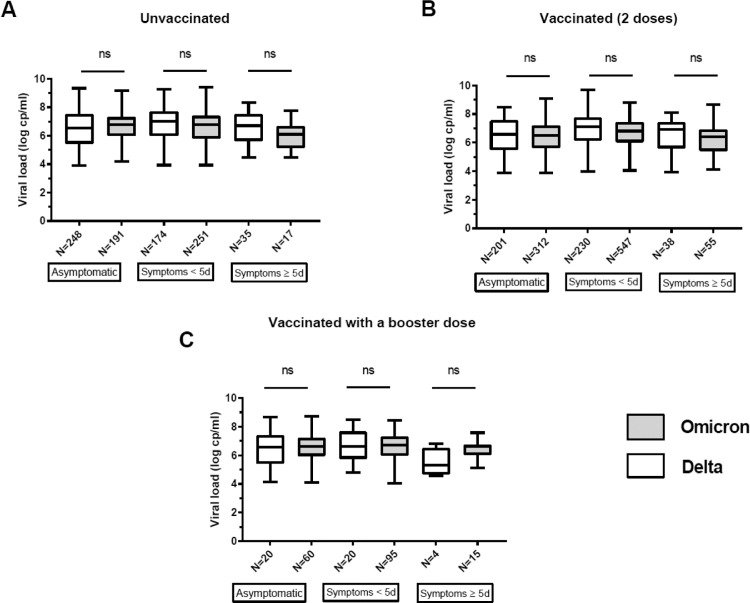
Multivariate analysis identified several characteristics that were independently associated with Omicron infections (Table 1 ). Omicron infections resulted in more symptomatic cases (OR=1.24; p < 0.01), were more frequent in vaccinated patients (OR=1.48; p < 0.01), and in young patients (OR=0.99; p < 0.01) (Table 1). The nasopharyngeal viral loads of Delta and Omicron infections were not significantly different, after adjustment for age, sex, symptoms and vaccination status (Table 1, Fig. 1 ). After stratification on age (interactions with vaccination and symptoms, p < 0.01), our final model showed that Omicron infections were more frequent than Delta infections in vaccinated patients. This was true for all age categories (<22 years (OR= 1.85; p < 0.0.01); 22–39 years (OR=1.32; p < 0.01); ≥40 years (OR=1.39; p < 0.01)) (Table 1). Omicron infections were associated with more symptomatic forms only in 22–39 year-old patients (OR=1.29; p < 0.01).
Table 1.
Multivariate analysis of the factors associated with Omicron variant infections.
| Delta (ref) /Omicron | Initial and final analyses |
Age-dependent analyses (final) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| < 22 years | 22–39 years | ≥ 40 years | ||||||||||
| OR | 95% CI | P value | OR | 95% CI | P value | OR | 95% CI | P value | OR | 95% CI | P value | |
| Age | 0.99 | [0.98;0.99] | <0.01 | Not Applicable | Not Applicable | Not Applicable | ||||||
| Gender (Female) | 1.11 | [0.95;−1.31] | ns | |||||||||
| Vaccination* | 1.48 | [1.37;1.61] | <0.01 | 1.85 | [1.55;2.21] | <0.01 | 1.32 | [1.17;1.49] | <0.01 | 1.39 | [1.19;1.61] | <0.01 |
| Symptoms⁎⁎ | 1.24 | [1.08;1.42] | <0.01 | 1.29 | [1.05;1.58] | 0.01 | ||||||
| Nasopharyngeal RNA viral load | 0.98 | [0.91;1.05] | ns | |||||||||
compared to unvaccinated patients.
compared to asymptomatic patients.
Fig. 1.
SARS-CoV-2 RNA loads in nasopharyngeal specimens from infected individuals. Data are shown as medians (midlines) plus interquartile ranges (IQR) (top and bottom box edges). Whiskers represent the upper and lower values. The SARS-CoV-2 Delta (white) and Omicron (gray) RNA loads were compared between patients in the same category (i.e. asymptomatic/ symptomatic < 5 days or > 5 days) and according to their vaccination status: A. Unvaccinated B. Vaccinated 2 doses C. Vaccinated 3 doses. The number of patients in each group and the p values (Mann-Whitney U-test) are shown. ns: not significant.
The SARS-CoV-2 pandemic has evolved since its beginning in late 2019, with the continuous emergence of new variants. Those with the highest transmissibility became prevalent and responsible for epidemic waves. The Omicron variant is no exception and outcompeted the Delta variant in many countries within a few weeks.2 The emergence of these variants challenges the effectiveness of our current vaccines and monoclonal antibodies. In vitro studies indicate that monoclonal antibodies and antibody-enriched plasma are much less effective against the Omicron variant .4 Our data provide clinical evidence that the Omicron variant is responsible for a greater proportion of vaccine breakthrough infections than is the Delta variant.
The transmissibility of the Alpha and Delta variants has been linked to higher NP viral loads.1 , 9 However, our data demonstrate that the increased transmissibility of the Omicron variant is not explained by higher nasopharyngeal viral load. Danish data also found similar nasopharyngeal viral load between Delta and Omicron infections.3 Whether higher viral loads can be found in more superficial samples such as saliva and nasal specimens as suggested by some should be further investigated.10 Ex-vivo studies on cultures of human bronchus and lung explants found that Omicron replicated faster in bronchial tissue than did Delta but that it replicated less efficiently in lungs .7 Perhaps the reduced severity of Omicron infections is due to a change in host cell tropism and faster replication in the upper respiratory tract.
Our study suggests that the Omicron variant is more contagious mainly because of vaccine escape resulting from the spike mutation that alters virus neutralization rather than because of greater virus shedding in the nasopharynx. Although we did no follow-up and collected no detailed clinical data, a strength of our study is that the specimens were collected from unselected individuals in a homogeneous population, all within a short time frame. We believe our findings will help identify the factors underlying the spread of the Omicron variant and the measures needed to control the pandemic.
Funding
No specific funding.
Declaration of Competing Interest
The authors declare no conflict of interest.
Acknowledgments
The English text was edited by Dr Owen Parkes.
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.jinf.2022.01.036.
Appendix. Supplementary materials
References
- 1.Rosa C., Beatriz O., Alma B.M., Eliseo A., de Michelena Paula M.C.C., et al. RNA viral loads of SARS-CoV-2 Alpha and Delta variants in nasopharyngeal specimens at diagnosis stratified by age, clinical presentation and vaccination status. J Infect. 2021 doi: 10.1016/j.jinf.2021.12.018. S0163-4453(21)00641-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.European Centre for Disease Prevention and Control; 2022. Weekly Epidemiological Update: Omicron Variant of Concern (VOC) – Week 1 (Data as of 7 January 2022) EU/EEA.https://www.ecdc.europa.eu/en/news-events/weekly-epidemiological-update-omicron-variant-concern-voc-week-1-data-7-january-2022 Available at. Accessed January 13, 2022. [Google Scholar]
- 3.L.F. Plesner, M.L. Hvas, J. Denwood Matthew, C.L. Engbo, M.C. Holten, S.R. Leo, et al. SARS-CoV-2 Omicron VOC transmission in Danish households. medRxiv 2021:2021.12.27.21268278. doi: 10.1101/2021.12.27.21268278. [DOI]
- 4.Delphine P., Nell S., Piet M., Florence G.B., Cyril P., Julian B., et al. Considerable escape of SARS-CoV-2 Omicron to antibody neutralization. Nature. 2021:1–7. doi: 10.1038/s41586-021-04389-z. [DOI] [PubMed] [Google Scholar]
- 5.Caroline M., Richard F., Mande T., Anchen L., Teshlin A., Boniswa K. Characteristics and outcomes of hospitalized patients in South Africa during the COVID-19 Omicron wave compared with previous waves. JAMA. 2021 doi: 10.1001/jama.2021.24868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hanjun Z., Lu L., Zheng P., Lin-Lei C., Xinjin M., Chuyuan Z., et al. SARS-CoV-2 Omicron variant shows less efficient replication and fusion activity when compared with delta variant in TMPRSS2-expressed cells. Emerg Microbes Infect. 2021:1–18. doi: 10.1080/22221751.2021.2023329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.HKUMed finds Omicron SARS-CoV-2 can infect faster and better than Delta in human bronchus but with less severe infection in lung. Available at https://www.med.hku.hk/en/news/press/20211215-omicron-sars-cov-2-infection. Accessed January 13, 2022, n.d.
- 8.Anthony Li, Allison M., Megan C., Hugh Guan T. Omicron and S-gene target failure cases in the highest COVID-19 case rate region in Canada - December 2021. J Med Virol. 2021 doi: 10.1002/jmv.27562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rosa C., Felipe B., Estela G., Alma B., Eliseo A., Diego C., et al. Initial viral load and decay kinetics of SARS-CoV-2 lineage B.1.1.7 in the upper respiratory tract of adults and children. J Infect. 2021;83(4):496–522. doi: 10.1016/j.jinf.2021.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.M. Gert, H. Nei-yuan, I. Arash, D. Deelan, E. Annabel, C. Chun-yat, et al. Saliva swabs are the preferred sample for Omicron detection. MedRxiv 2021:2021.12.22.21268246. doi: 10.1101/2021.12.22.21268246. [DOI]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.