Technique Video
Presentation of different steps of procedure for arthroscopic rotator cuff repair with harvest of coracoacromial ligament autograft for footprint stimulation and augmentation, including preoperative assessment, patient positioning, portal placement, and surgical technique steps.
Abstract
Failure of rotator cuff repair can be a disastrous clinical outcome. Although failure is a multifactorial issue, recent interest has piqued in understanding the biology of the insertional components of the supraspinatus and infraspinatus at the footprint. When the torn tendon is of poor quality, especially if it is diminutive or thin, rotator cuff repair augmentation should be considered to aid in long-term healing. Various allograft options have been described in the past, and more recently, xenografts and synthetics have become more commonly used. The use of autografts in the treatment of insertional footprint deficiency has great potential; however, few grafts have been described. This study describes the surgical technique for footprint augmentation in arthroscopic supraspinatus repair using harvested autologous coracoacromial ligament tissue.
Rotator cuff repair (RCR) is one of the most common surgical procedures performed by orthopaedic surgeons. The advances in arthroscopic techniques over the past 2 decades have transformed surgical management in RCR. Although most patients who undergo RCR achieve successful postoperative outcomes, repair failure remains a concern. Many factors must be considered in the case of failed RCR, including fixation method construct and rigidity, tear characteristics (i.e., size, number of tendons involved, and chronicity), patient health characteristics, and biological factors. Incomplete or incompetent healing at the supraspinatus footprint related to tissue biology has been implicated as a crucial reason for failure. Failure of tendon healing to bone at the supraspinatus footprint is a multifactorial issue. For healing to occur, there must be adequate blood supply to the footprint, a pliable and reducible cuff tendon, sufficient cuff tissue, and appropriate reduction and tension of the healing tissue as remodeling and healing occur.
Various forms of augmentation have been described in RCR surgery. In the scenario in which the tendon of desired repair is diminutive or thin, few options exist to augment the tissue at the footprint. This study describes a technique for footprint augmentation in arthroscopic supraspinatus repair using harvested autologous coracoacromial (CA) ligament tissue.
Surgical Technique
Fig 1, Fig 2, Fig 3, Fig 4, Fig 5, Fig 6, Fig 7, Fig 8, Fig 9 and Video 1 show the surgical technique.
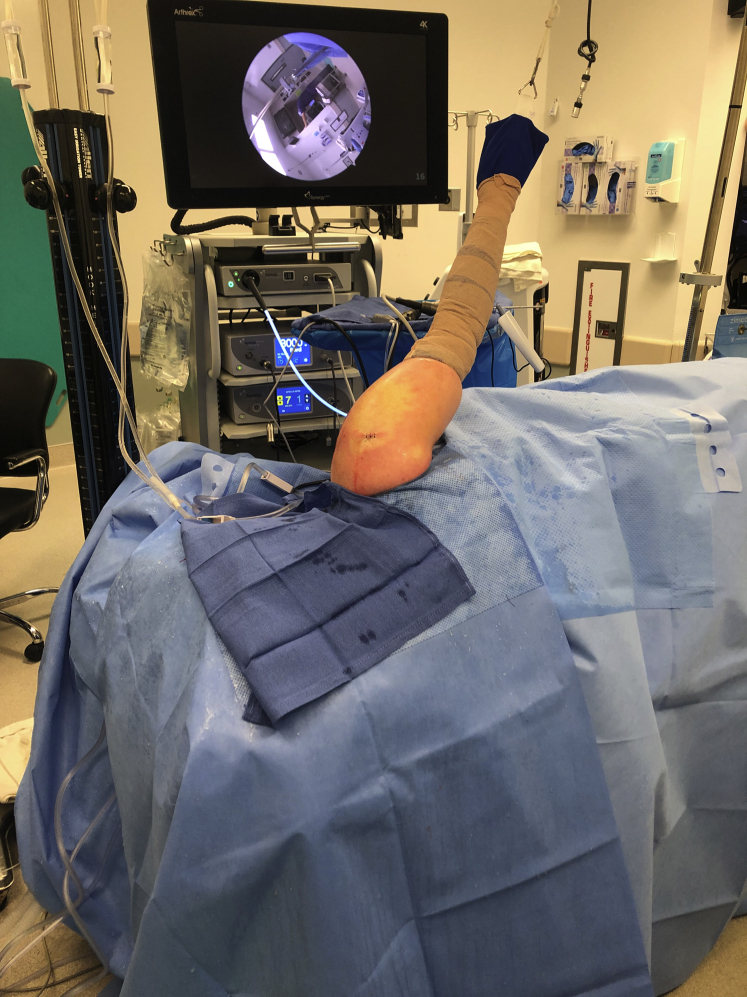
Fig 1.
Lateral shoulder positioning and setup for arthroscopy, shown from behind the posterior side of the patient (right shoulder).
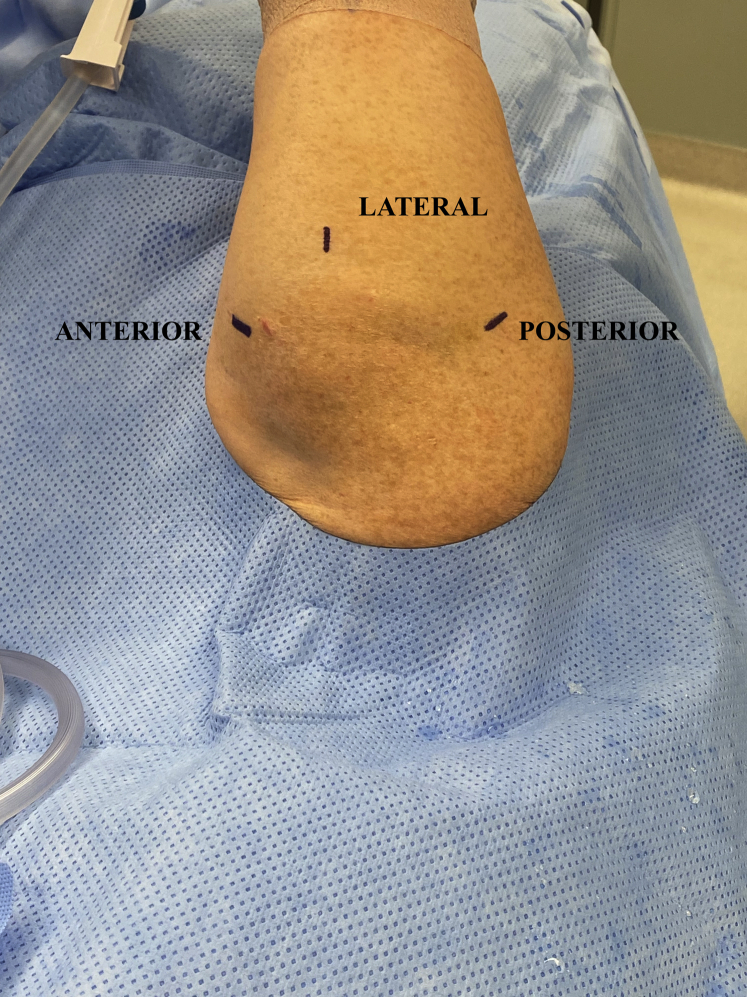
Fig 2.
Anterior, lateral, and posterior arthroscopic portals used in shoulder arthroscopy from the lateral decubitus position (right shoulder) for the described procedure, as seen from the head of the operating room table.
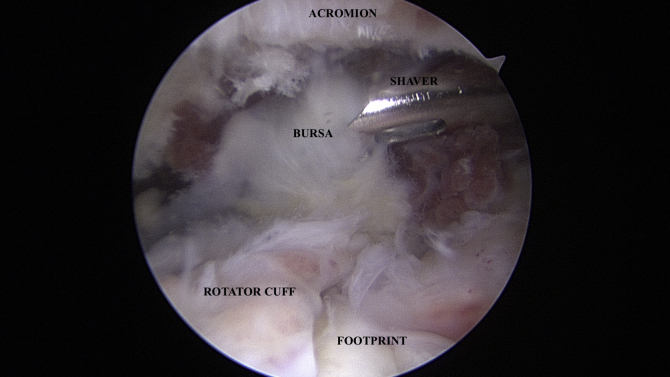
Fig 3.
Viewing from the posterior portal in the subacromial space, meticulous exposure is performed to visualize the coracoacromial ligament (right shoulder). Superiorly, the acromion is visualized, with the shaver immediately inferior actively removing bursal tissue. The supraspinatus rotator cuff tissue and footprint can be visualized inferiorly.
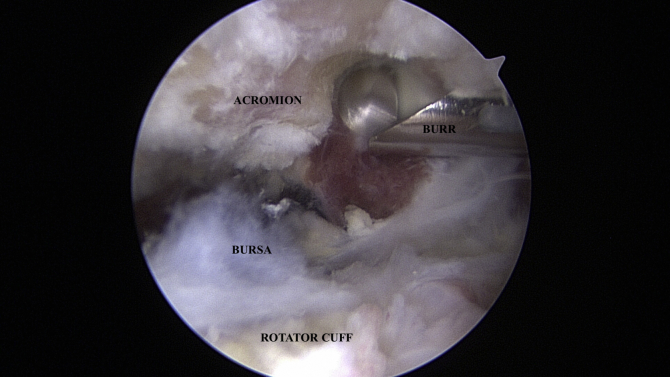
Fig 4.
Viewing from the posterior portal in the subacromial space, partial acromial undersurface contouring is performed with a burr (right shoulder). Superiorly, the acromion is visualized, with the motorized burr removing bone from lateral (right) to medial (left). The adjacent bursal tissue and rotator cuff is visualized inferior to the acromion.
Fig 5.
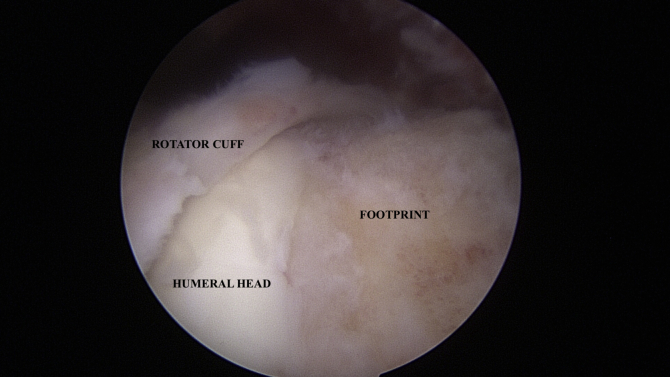
Viewing from the posterior portal in the subacromial space, a massive tear is noted in the supraspinatus and infraspinatus (labeled “rotator cuff”) with retraction to the level of the humeral head. Lateral (right) to the exposed humeral head is the bony footprint of the supraspinatus.
Fig 6.
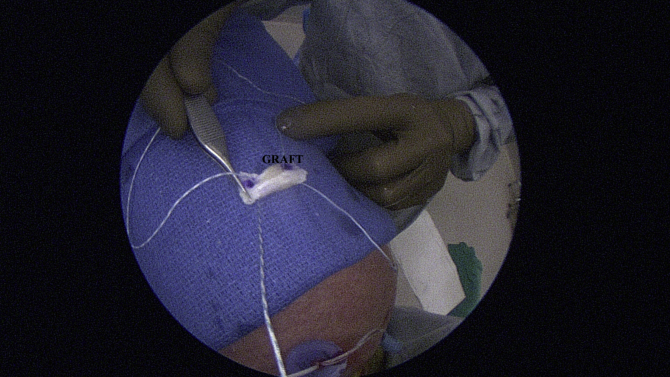
Photograph from operating room Mayo stand showing the coracoacromial ligament graft measuring approximately 15 mm wide by 8 mm long by 3 mm thick. The sutures from 2 medial-row footprint anchors are placed 8 mm apart in the graft (marked with purple dots).
Fig 7.
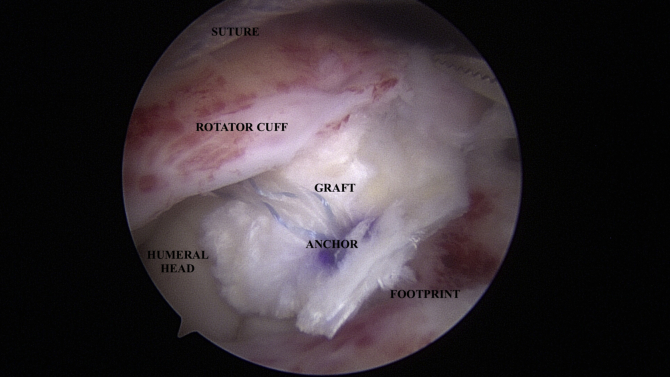
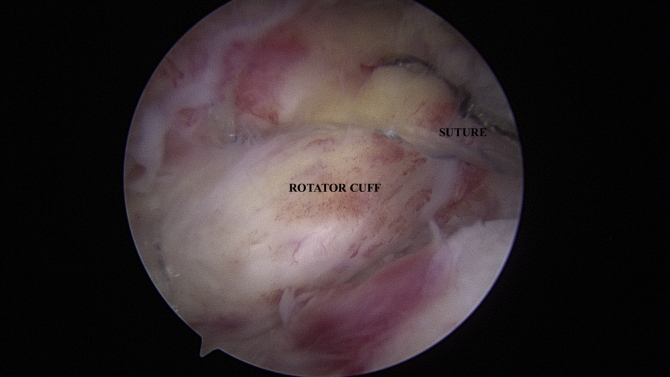
Viewing from the posterior portal in the subacromial space, the coracoacromial ligament is observed to be approximated to the footprint with the sutures from the medial-row anchors traveling through the coracoacromial ligament graft (deep) and then the native rotator cuff tissue (superficial).
Fig 8.
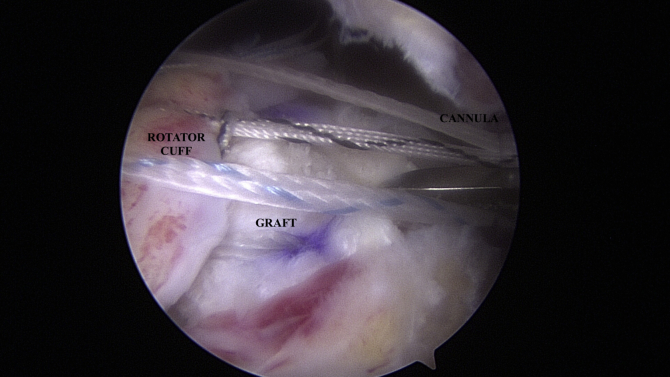
Viewing from the posterior portal in the subacromial space, the medial row and luggage-tag sutures are tensioned over the native cuff (superficial) and coracoacromial ligament autograft (deep) as the sutures are pulled from medial (left) to lateral (right) while being loaded into a lateral-row anchor.
Fig 9.
Viewing from the posterior portal in the subacromial space, the final double-row construct shows appropriate tension and compression of the cuff and graft at the supraspinatus insertion.
Patient Setup
The patient is positioned in the lateral decubitus position (Fig 1) and is stabilized with a beanbag and chest support posts. The operative extremity is prepared and draped in the usual sterile fashion. The arm is held in traction with 15 lb.
Arthroscopic Procedure
Standard posterior, anterior, and lateral portals are used (Fig 2). The standard posterior portal is established first to enter the glenohumeral joint. The joint is insufflated with arthroscopic fluid. The anterior portal is established in the rotator interval. A standard diagnostic arthroscopy is performed. The trocar and camera are then removed from the glenohumeral joint, and the subacromial space is entered. The lateral portal is established under needle visualization. Bursectomy is performed with a motorized shaver. The undersurface of the acromion is visualized with an electrocautery device and a shaver. The CA ligament is visualized from its insertion on the acromion down to the origin on the coracoid. Meticulous debridement of synovial and bursal tissue is performed around the CA ligament to isolate the structure (Fig 3). The CA ligament is then harvested by first transecting the ligament off of the acromial attachment using a Samurai blade (Stryker, Kalamazoo, MI). While the free end is being controlled with a tissue grasper, the distal end of the CA ligament is transected in the rotator interval near the coracoid using an electrocautery device. The harvested graft is then removed from the subacromial space. The graft should measure approximately 15 mm in length, 8 mm in width, and 3 mm in thickness. The total time required for graft harvest and preparation is approximately 10 to 15 minutes.
Limited acromioplasty is performed (Fig 4). As the supraspinatus is identified, special care is taken to note the presence of a tear and the characteristics of the tissue. Specifically, the augmentation technique described should be considered in the setting of a supraspinatus tendon with a full-thickness tear with moderate retraction and tissue that is diminutive or thin (Fig 5).
The footprint is then prepared gently to expose a bleeding surface. Two FiberTak DR anchors (Arthrex, Naples, FL) loaded with tape are placed at the footprint, 8 mm apart. The suture tape from the anchors is passed through the CA ligament graft at both ends of the graft, 8 mm apart (Fig 6). The CA ligament graft is transferred back into the subacromial space and is advanced down the sutures to lie directly on the anchors at the supraspinatus footprint (Fig 7). The tape from the anchors is then used take spaced bites in the supraspinatus tendon using a Scorpion device (Arthrex). Additionally, 2 luggage-tag sutures are placed in the supraspinatus at the anterior and posterior margins of the tear using FiberLink (Arthrex) and passing with the Scorpion device. The sutures are docked into 2 lateral-row SwiveLock anchors (Arthrex) in a crossing fashion (Fig 8). The final double-row construct achieves appropriate reduction, tension, and compression of the cuff at the footprint (Fig 9).
The arthroscopic portals are closed with interrupted nylon sutures. Postoperative dressings are placed. The patient is placed in a sling with an abduction pillow. Postoperative recovery is similar to a standard RCR protocol. A sling is used for 6 weeks. Table 1 provides a detailed breakdown of the postoperative rehabilitation protocol.
Table 1.
Postoperative Rehabilitation Protocol After RCR With CA Ligament Harvest and Augmentation
| General recommendations |
|
|
|
| Weeks 0-4: primary goals—eliminate swelling and regain PROM |
|
|
|
|
|
|
| Weeks 4-8: primary goal—regain full PROM |
|
|
|
| Weeks 8-12: primary goal—regain full AROM |
|
|
|
| Weeks 12-16: primary goal—begin resistive rotator cuff strengthening |
|
|
|
|
|
|
| 4-6 mo: primary goal—increase shoulder and rotator cuff strengthening |
|
|
|
|
|
|
ABER, abduction with external rotation; ABIR, abduction with internal rotation; AROM, active range of motion; CA, coracoacromial; ER, external rotation; PROM, passive range of motion; PT, physical therapy; RCR, rotator cuff repair; ROM, range of motion.
Discussion
The clinical scenario of a failed RCR is difficult to manage. A growing understanding of the biological and mechanical parameters of the torn rotator cuff has allowed us to refine which patients might be at risk. Mirzayan et al.1 suggested patient age older than 65 years, tear size greater than 2 cm, retraction greater than 2 cm, involvement of 2 or more tendons, and fatty muscle infiltration as reasons to consider augmentation. In a recent systematic review, Bailey et al.2 reported a lower retear rate and improved American Shoulder and Elbow Surgeons scores in patients treated with RCR when augmented or interposed with graft. Much of the recent interest in augmentation has revolved around the use of xenografts3 and synthetics,4 which—along with human allograft—have significant disadvantages including cost and risk of disease transmission. Use of an autograft would be ideal for the setting of cuff deficiency or footprint augmentation to avoid the aforementioned disadvantages while providing the optimal biological and vascular environment for tissue healing.
Although the use of CA ligament autograft has not previously been described in the setting of RCR, it has a successful track record when used for stabilizing procedures for acromioclavicular separation, such as the modified Weaver-Dunn procedure.5 Previous anatomic dissections of the CA ligament have approximated the length at nearly 46 mm, with a width of 30 mm at the coracoid and 15 mm at the acromion.6 Whereas the use of the CA ligament has been assessed in biomechanical studies of the Weaver-Dunn procedure, no previous biomechanical studies have been performed on the use of the CA ligament in RCR.7
As with any surgical technique, advantages, disadvantages, and technical pearls must be considered prior to implementation of CA ligament harvest and augmentation in clinical practice. One key technical pearl for this procedure is the use of 2 to 3 luggage-tag sutures in the supraspinatus separate from the medial-row anchor sutures; this technique aids in balanced mobilization of the cuff over the graft onto the footprint. A previous biomechanical investigation has described this method as having significantly improved bone-tendon contact at the footprint as compared with the traditional double-row construct.8 The main potential downside of using a CA ligament graft is the extra time required to perform graft harvest, although this could be minimized after streamlined techniques and a brief learning curve. The manner in which this study performed the harvest also resulted in an avascular free autograft, which decreases some of the biological advantage compared with a vascularized autograft. Finally, it must be noted that the CA ligament does serve as a secondary static stabilizer of the glenohumeral joint, especially in the setting of rotator cuff tear.9 Although CA ligament release in isolation has not been shown to lead to anterosuperior escape, there may be a theoretical risk associated with harvest in the setting of RCR. The key benefits of using the CA ligament for augmentation include autogenous graft, robust tissue quality, and facile harvest with no added morbidity or accessory incisions. For these reasons, use of the CA ligament autograft for augmentation to aid in footprint healing with RCR is a promising technique.
Footnotes
The authors report the following potential conflicts of interest or sources of funding: O.L. is a consultant for Arthrex and Linvatec. Full ICMJE author disclosure forms are available for this article online, as supplementary material.
Supplementary Data
Presentation of different steps of procedure for arthroscopic rotator cuff repair with harvest of coracoacromial ligament autograft for footprint stimulation and augmentation, including preoperative assessment, patient positioning, portal placement, and surgical technique steps.
References
- 1.Mirzayan R., Weber A.E., Petrigliano F.A., Chahla J. Rationale for biologic augmentation of rotator cuff repairs. J Am Acad Orthop Surg. 2019;27:468–478. doi: 10.5435/JAAOS-D-18-00281. [DOI] [PubMed] [Google Scholar]
- 2.Bailey J.R., Kim C., Alentorn-Geli E., et al. Rotator cuff matrix augmentation and interposition: A systematic review and meta-analysis. Am J Sports Med. 2019;47:1496–1506. doi: 10.1177/0363546518774762. [DOI] [PubMed] [Google Scholar]
- 3.Bushnell B.D., Bishai S.K., Krupp R.J., et al. Treatment of partial-thickness rotator cuff tears with a resorbable bioinductive bovine collagen implant: 1-Year results from a prospective multicenter registry. Orthop J Sport Med. 2021;9 doi: 10.1177/23259671211027850. 23259671211027850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baldwin M., Nagra N.S., Greenall G., et al. Use of implantable meshes for augmented rotator cuff repair: A systematic review and meta-analysis. BMJ Open. 2020;10 doi: 10.1136/bmjopen-2020-039552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Le Hanneur M., Delgrande D., Lafosse T., Werthel J.D., Hardy P., Elhassan B. Triple-bundle anatomical reconstruction using the coracoacromial ligament and the short head of biceps tendon to stabilize chronic acromioclavicular joint dislocations: A cadaver feasibility study. Orthop Traumatol Surg Res. 2018;104:27–32. doi: 10.1016/j.otsr.2017.11.003. [DOI] [PubMed] [Google Scholar]
- 6.Moinfar A.R., Murthi A.M. Anatomy of the pectoralis minor tendon and its use in acromioclavicular joint reconstruction. J Shoulder Elbow Surg. 2007;16:339–346. doi: 10.1016/j.jse.2006.09.007. [DOI] [PubMed] [Google Scholar]
- 7.Le Hanneur M., Thoreson A., Delgrande D., et al. Biomechanical comparison of anatomic and extra-anatomic reconstruction techniques using local grafts for chronic instability of the acromioclavicular joint. Am J Sports Med. 2018;46:1927–1935. doi: 10.1177/0363546518770603. [DOI] [PubMed] [Google Scholar]
- 8.Urch E., Lin C.C., Itami Y., et al. Improved rotator cuff footprint contact characteristics with an augmented repair construct using lateral edge fixation. Am J Sports Med. 2020;48:444–449. doi: 10.1177/0363546519888182. [DOI] [PubMed] [Google Scholar]
- 9.Arntz C., Jackins S., Matsen F. Prosthetic replacement of the shoulder for the treatment of defects in the rotator cuff and the surface of the glenohumeral joint. J Bone Joint Surg Am. 1993;75:485–491. doi: 10.2106/00004623-199304000-00002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Presentation of different steps of procedure for arthroscopic rotator cuff repair with harvest of coracoacromial ligament autograft for footprint stimulation and augmentation, including preoperative assessment, patient positioning, portal placement, and surgical technique steps.
Presentation of different steps of procedure for arthroscopic rotator cuff repair with harvest of coracoacromial ligament autograft for footprint stimulation and augmentation, including preoperative assessment, patient positioning, portal placement, and surgical technique steps.