Abstract
Purpose
The aim of this study was to prospectively compare the therapy response and safety of microwave (MWA) and radiofrequency ablation (RFA) for the treatment of liver metastases using a dual ablation system.
Methods
Fifty patients with liver metastases (23 men, mean age: 62.8 ± 11.8 years) were randomly assigned to MWA or RFA for thermal ablation using a one generator dual ablation system. Magnetic resonance imaging (MRI) was acquired before treatment and 24 h post ablation. The morphologic responses to treatment regarding size, volume, necrotic areas, and diffusion characteristics were evaluated by MRI. Imaging follow-up was obtained for one year in three months intervals, whereas clinical follow-up was obtained for two years in all patients.
Results
Twenty-six patients received MWA and 24 patients received RFA (mean diameter: 1.6 cm, MWA: 1.7 cm, RFA: 1.5 cm). The mean volume 24 h after ablation was 37.0 cm3 (MWA: 50.5 cm3, RFA: 22.9 cm3, P < 0.01). The local recurrence rate was 0% (0/26) in the MWA-group and 8.3% (2/24) in the RFA-group (P = 0.09). The rate of newly developed malignant formations was 38.0% (19/50) for both groups (MWA: 38.4%, RFA: 37.5%, P = 0.07). The overall survival rate was 70.0% (35/50) after two years (MWA: 76.9%, RFA: 62.5%, P = 0.60). No major complications were reported.
Conclusion
In conclusion, MWA and RFA are both safe and effective methods for the treatment of liver metastases with MWA generating greater volumes of ablation. No significant differences were found for overall survival, rate of neoplasm, or major complications between both groups.
Abbreviations: ADC, apparent diffusion coefficient; DR, distant recurrence; DWI, diffusion-weighted imaging; LTP, local tumor progression; MWA, microwave ablation; RFA, radiofrequency ablation; TACE, transarterial chemoembolization
Keywords: Ablation Techniques, Liver, Metastasis, Magnetic resonance imaging, Medical Oncology
Highlights
-
•
A dual ablation system allows for MWA and RFA treatment using the same hardware.
-
•
Both methods are safe and effective for the treatment of liver metastases.
-
•
MWA generates greater volumes of ablation and larger ablative margins compared to RFA.
1. Introduction
Liver metastases are common in different types of cancer diseases. Due to the dual blood supply, many tumors such as colorectal, breast, and pancreatic carcinomas metastasize to the liver. Surgical resection is commonly seen as the standard of care [1], [2] and perioperative chemotherapy can reduce the progression of operable colorectal liver metastases [1], [2], [3]. Although resection for non-colorectal liver metastasis remains controversial, studies indicate a survival benefit in selected patients [4], [5]. For unresectable metastases, systemic therapy represents the standard treatment [1], [2], [3], [6]. However, thermal ablation plus systemic therapy showed promising results regarding the progression free survival [6], [7].
Thermal ablation, including cryoablation, radiofrequency ablation (RFA), and microwave ablation (MWA), was originally used for minimally invasive therapy of inoperable hepatocellular carcinomas. Due to the low complication and recurrence rates, thermal ablation became a treatment option for secondary liver malignancies. However, MWA has shown some benefits to RFA, such as larger ablation volumes, increased tissue temperature, homogeneous cell death, shortened treatment duration, reduced susceptibility to heat-sink effect, and higher rate of local tumor control [8], [9], [10], [11], [12], [13], [14], [15]. Moreover, MWA might require more energy to achieve a similar ablation volume in different tumor types [16]. The treatment response is commonly described by the extent of necrotic areas, decrease in tumor volume, and changes of magnetic resonance (MR)-based diffusion parameters. To our knowledge, the value of these quantitative index parameters for the response to ablation has not been evaluated.
Thus, the aim of the current study was to determine and compare the therapy response and safety of microwave (MWA) and radiofrequency ablation (RFA) for the treatment of liver metastases in a prospectively randomized trial using a dual ablation system.
2. Methods
2.1. Study Design
The current study was approved by the institutional ethical committee of our University Hospital Frankfurt (IRB number 322/13) and informed consent existed from every patient. All methods were performed in accordance with HIPAA guidelines. This study was partially supported by Medicor (MMS Medicor Medical Supplies GmbH, Kerpen, Germany) for statistical analysis. The study design and data analysis were performed by the authors who are not affiliated to this company.
All patients met general thermal ablation inclusion criteria as follows: (a) Thermal ablation as option for therapy, (b) written and verbal consent of ablation, (c) age over 18 years, (d) diagnosis of liver metastases histologically and/or radiologically confirmed, (e) MRI examination with 1.5 Tesla (T) or 3 T, (f) good general condition, (g) lesions with axial diameter < 5 cm, (h) clinically planned therapy, (i) maximum of 5 liver metastases (j) treatment decision in a multidisciplinary tumor board. Exclusion criteria were as follows: (a) presence of extrahepatic metastasis (b) general contraindication for MRI such as pacemaker, (c) pregnant or breast-feeding women, (d) second carcinoma, (e) allergy to gadolinium-based contrast materials, (f) contraindication for thermal ablation. Before patients were enrolled in the study, most of them had undergone a combination therapy of surgery, transarterial chemoembolization (TACE), systemic chemotherapy and/or radiation. None of the patients received additional TACE, radiation, or surgery after beginning of the study.
All patients were randomly assigned (1:1) to either the MWA-group or RFA-group between January 2014 and April 2016. Randomized treatment allocation was performed using a permuted block design to ensure similar group sizes.
2.2. Patient Characteristics
A total of 50 patients (23 men and 27 women, mean age: 62.8 ± 11.8 years, range: 40–91 years), who met the clinical inclusion and exclusion criteria, underwent a CT-guided percutaneous thermal ablation of liver metastasis. One single patient was wrongly assigned to the MWA group instead of the RFA group. Therefore, the MWA group consisted of 26 patients (mean age: 62.7 years, range: 43–91years), the RFA group of 24 patients (mean age: 63.3 years, range: 41–85 years). There were no significant differences of age between the two groups (P = 0.77).
Colorectal carcinoma was the primary tumor in 54% (MWA: 13, RFA: 14) breast cancer in 22% (MWA: 6, RFA: 5), pancreatic carcinoma in 6.0% (MWA: 3), ovarian carcinoma in 4% (MWA: 2), and other carcinomas in 14% of all cases (Table 1). In the RFA-group 91.6% (22/24) of patients were treated with surgical resection before ablative therapy, 70.8% (17/24) underwent TACE, and 66.6% (16/24) received a systemic chemotherapy before study inclusion. In the MWA-group we had a similar rate of combined treatment, 88.5% (23/26) performed previous surgery and 53.8% (14/26) were treated with TACE before the start of the study.
Table 1.
Patient characteristics.
| MWA (n = 26) | RFA (n = 24) | |
|---|---|---|
| Mean age | 62.7 ± 11.8 years | 63.3 ± 11.8 years |
| Gender female male |
14 12 |
13 11 |
| Tumor Colon cancer Breast cancer Pancreatic carcinoma Ovarian carcinoma Cholangiocarcinoma Neuroendocrine tumor Esophageal cancer Uveal melanoma Oropharyngeal cancer |
13 6 3 2 1 1 0 0 0 |
14 5 0 0 2 0 1 1 1 |
2.3. Pre-Ablation Assessment and Ablation Procedure
For each patient, recent imaging studies were used to assess size, lesion volume, anatomical position, and diffusion characteristics on the day before the examination. Imaging was performed with routine contrast-enhanced magnetic resonance imaging (MRI) using a 1.5-T or 3-T systems (Magnetom Avanto and Magnetom Prismafit, Siemens Healthcare, Erlangen, Germany) in transverse section with 5-mm thickness.
Patients were clinically monitored during the procedure including blood pressure, electrocardiography, and pulse oximetry measurements. After positioning the patient in a supine position with arms elevated above the head, a combination of sedative and analgesic medication with diazepam (Diazepam, ratiopharm GmbH, Ulm, Germany; 0.1–0.2 mg/kg body weight) and piritramide (Piritramid, Hameln Pharma Plus GmbH, Hameln, Germany; 0.2 mg/kg body weight) were administered through an intravenous catheter. All ablations were performed under CT planning and guidance (Somatom Sensation 128, Siemens) with the following parameters: 5-mm collimation, 30 mAs, 120 kV, 5-mm section-thickness and activated real-time tube modulation (CARE Dose 4D, Siemens) [17].
After disinfection of the treatment area, the RFA electrode applicator or MWA antenna were inserted percutaneously. The interventional radiologists controlled the ablation procedures using repeated CT single-image acquisitions. The end of the ablation treatment was determined by CT imaging showing complete tumor coverage. Patients were observed and monitored for the following 12 h [17].
Following the standard grading system, major complications were defined as events that lead to substantial morbidity and disability and increased level of care or extension of the hospital stay; all other occurring complications (e.g., moderate local pain) were determined as minor [18], [19].
2.4. Technical Features
The dual ablation system, Amica (MMS Medicor Medical Supplies GmbH, Kerpen, Germany), was used to generate both, RFA and MWA. The use of a unique apparatus in this study minimized system-related biases in the comparison between RFA and MWA, like hardware or software differences or manufacturer approach. RFA used output frequencies of 450 kHz and an output power of 200 W. The duration of the RFA procedure was between 10 and 20 min. The RFA technique is based on the biophysical interaction between high frequency alternating current and biological tissue. An electric field is established between the electrodes, which induces ionic oscillatory motion and results in heat generation [20].
MWA used output frequencies of 2450 MHz and a maximum output power of 140 W. The MWA procedure was performed in three steps with rising output powers (45–60 W for 1 min, 65–80 W for 1 min, and 85–100 W for 5–10 min). Since the water molecule has an electric dipole moment, the electric field of the microwaves excites harmonic oscillations in the water, resulting in heating. Other molecules are heated by convection, since macromolecules are not directly affected by microwaves [20]. The MWA applicator had a straight water-cooled shaft with a mini-choked coaxial antenna operated with continuous power delivery. Each procedure was performed using a single antenna. The MWA provided by the chosen system was previously shown to be safe and effective in the treatment of hepatic malignancies [21]. A single electrode device was used for RFA and MWA ablation. Each device performed one ablation zone without overlapping areas of smaller tumor lesions. Ablation of bigger lesions were performed with position change of the applicator system.
2.5. Imaging and Follow-up Protocol
First postprocedural imaging was obtained one day after ablation as unenhanced and contrast-enhanced (Gadovist 1 mmol/ml, Bayer Healthcare, Berlin, Germany) T1- and T2-weighted MRI scans were acquired in transverse and sagittal orientation with 5- or 6-mm slice thickness using a 1.5-T system (Magnetom Avanto, Siemens) or 3-T system (Prisma; Siemens). The applied sequence protocol consisted of the following parameters: diffusion-transverse, T2w-coronal and transverse, T1w-FLASH-2D transverse, EP-2D-Diff (b50, b400, b800) HASTE, TSE, in- and opposed phase, and contrast-enhanced FLASH 2D dynamic phase. Further imaging follow-up was obtained after three, six and twelve months using the same MRI protocol. Clinical follow-up with survival rates were obtained for two years.
2.6. Definitions and Method of Measurement
Technical success was defined as correct intervention accomplishment according to protocol and complete coverage of the index tumor through the ablation zone in CT at the end of ablation. Technical efficacy was determined as complete coverage of the index tumor through the ablation zone in first contrast-enhanced MRI 24 h post-ablation showing a thin symmetric rim of peripheral enhancement [19].
Recurrence of tumor disease was differentiated in local tumor progression (LTP) and distant recurrence (DR). LTP was defined as a tumor focus connected to the edge of an ablation zone that was previously considered technically efficient in 24-hour post-ablation MRI. DR was determined as a new intrahepatic lesion in untreated parenchyma or extrahepatic progress [19]. Recurrences were evaluated in contrast-enhanced follow-up MRI after three, six, and twelve months.
Evaluation of the treatment response included the following parameters: Ablative margin, ablation zone, ablation volume, and necrosis zone post ablation. The ablation margin was determined by exactly measuring the liver lesion and positioning it at the same spot on the MRI after ablation. The distance between the lesion and the ablation zone was defined as the ablation margin. In addition, diffusion-weighted imaging (DWI) with apparent diffusion coefficient (ADC) was used to assess therapy response. These parameters allow for further characterization of tumor lesions and were used to evaluate the response to chemotherapy in liver metastasis in previous studies [22], [23], [24].
2.7. Statistical Analysis
The total number of single interventions was categorized in two groups according to the used system (RFA, MWA). Parameters collected were age, gender, primary tumor, technical success and efficacy, complications, one-year mortality, LTP and DR at twelve-months follow-up, tumor diameter, one-year and two years survival rates, ADC and ablation volume on MRI and ablative margin size.
Minimum and maximum values with standard deviations, as well as medians with ranges were assessed. The Kolmogorov-Smirnov test was used to determine whether the measurements were normally distributed. Since variables were not normally distributed, Friedman test was used to determine whether differences between pre- and post-ablation measurements were significant. Kaplan-Meier analysis was performed to calculate survival rates. Statistical software (BiAS for Windows, version 11.06; epsilon-Verlag, Darmstadt, Germany) was used and a P-value < 0.05 indicated statistical significance.
3. Results
Technical success was achieved in all interventions with RFA and MWA. Technical efficacy was reached in 26/26 (100%) MWA and 24/24 (100%) RFA interventions. We recorded no intraprocedural death. The one-year mortality rate after ablation in this trial was 14.0% (7/50) with 12.5% (3/24) in the RFA and 15.4% (4/26) in the MWA-group (P = 0.99). There were no major complications observed in either group. The minor complications included low-grade subcapsular bleeding and a small liver abscess in the RFA group that did not require treatment. All patients received previous treatment like surgery, TACE, radiotherapy and/or chemotherapy. TACE as single treatment was performed in 31 cases, including 14 in the MWA-group and 17 in the RFA-group. The combination of surgery and TACE was performed in 28 patients, including 13 in MWA-group and 15 in RFA-group.
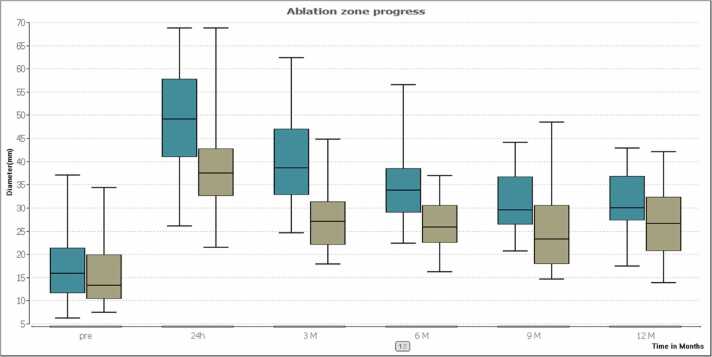
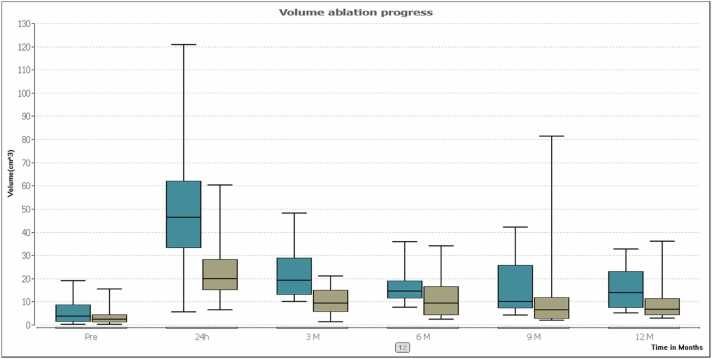
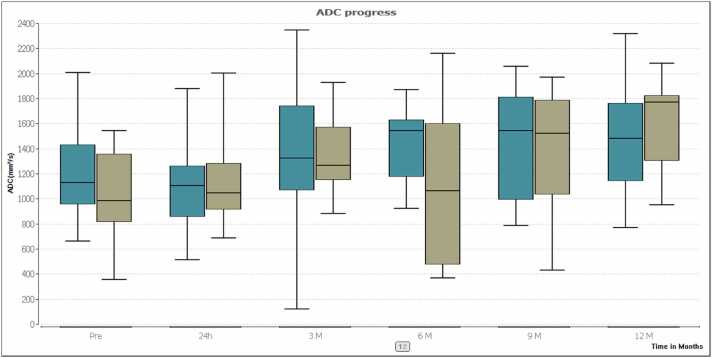
The mean transverse initial tumor diameter was 17.2 ± 8.1 mm (range: 6.3–37.1 mm) in the group of patients with MWA treatment and 15.3 ± 6.1 mm (range: 7.6–34.4 mm) in the RFA-group. Differences between index tumors were not significant (P = 0.32). Ablation zone progress is presented in Table 2 and Fig. 1. The mean initial tumor volume was 3.7 cm³ in the MWA-group and 3.4 cm³ in the RFA-group. Differences between index tumors were not significant (P > 0.05). Volume changes post ablation are shown in Table 3 and Fig. 2. The mean transverse initial tumor ADC value was 1216.7 mm²/s in the MWA-group and 1038.7 mm²/s in the RFA-group (P > 0.05). ADC values post ablation are shown in Table 4 and Fig. 3.
Table 2.
Comparison of ablation zone diameter after treatment.
| Diameter (mm) | Pre-Ablation | 24 h | 3 Months | 6 Months | 9 Months | 12 Months |
|---|---|---|---|---|---|---|
|
MWA Mean SD Median Range |
17.2 8.1 15.90 6.3–37.1 |
49.2 11.1 49.2 26.2–68.8 |
40.1 10.1 38.65 24.7–62.4 |
34.3 7.9 33.8 22.5–56.6 |
31.2 7.2 29.5 20.8–44.1 |
30.5 7.1 29.9 17.5–42.9 |
|
RFA Mean SD Median Range |
15.3 6.1 13.3 7.6–34.4 |
39.7 11.4 37.45 21.6–68.8 |
27.2 6.4 27.1 18.0–44.8 |
26.3 5.7 25.8 16.3–36.9 |
25.7 10.2 23.25 14.8–48.5 |
26.7 7.9 26.6 14.0–42.1 |
Fig. 1.
The box plots compare the medians of the ablation zone progress after treatment between MWA (turquoise) and RFA (olive). Results indicate that larger ablation zones were achieved with MWA compared to RFA after 24 h and 3 months. But no significant differences were found after the 12 months follow-up (P = 0.189).
Table 3.
Comparison of volume development post ablation.
| Volume (cm³) | Pre-Ablation | 24 h | 3 Months | 6 Months | 9 Months | 12 Months |
|---|---|---|---|---|---|---|
|
MWA Mean SD Median Range |
3.7 4.9 3.8 0.5–19.09 |
50.5 27.5 46.4 5.7–120.7 |
22.1 11.5 19.1 10.2–48.2 |
17.9 9.1 14.5 7.8–35.9 |
15.5 11.9 10.0 4.5–42.1 |
15.6 9.2 13.8 5.2–32.8 |
|
RFA Mean SD Median Range |
3.4 3.4 2.3 0.4–15.5 |
22.9 11.8 20.0 6.7–60.4 |
10.1 5.4 9.4 1.5–21.1 |
11.7 9.5 9.3 2.6–34.0 |
14.3 23.9 6.5 2.3–81.3 |
9.6 9.2 6.7 3.1–35.9 |
Fig. 2.
The box plots show a comparison of the median volume ablation progress after treatment between MWA (turquoise) and RFA (olive). MWA results revealed larger volumes for MWA compared to RFA after 3 months (P < 0.001) but no significant difference after 12 months follow-up (P = 0.037).
Table 4.
Comparison of ADC values post ablation.
| ADC (mm²/s) | Pre-Ablation | 24 h | 3 Months | 6 Months | 9 Months | 12 Months |
|---|---|---|---|---|---|---|
|
MWA Mean SD Median Range |
1216.7 369.9 1131 665–2007 |
1096.7 300.1 1106 518–1879 |
1383.7 502.4 1322.5 126–2348 |
1429.6 295.5 1544 927–1869 |
1448.8 458.9 1543 791–2057 |
1497.6 447.7 1481 773–2316 |
|
RFA Mean SD Median Range |
1038.7 342.1 986 359–1543 |
1111.7 313.74 1046 692–2002 |
1324.1 281.8 1266.5 886–1930 |
1089.2 654.3 1065 371–2158 |
1387.9 484.6 1522.5 434–1970 |
1575 364.8 1769 955–2080 |
Fig. 3.
The box plots show a comparison of ADC progress after treatment between MWA (turquoise) and RFA (olive). ADC values showed no significant differences between RFA and MWA after 12 months (P = 0.566).
Regarding the ablative margin, the study showed significantly larger sizes for MWA with 17.9 mm compared to RFA with 14.3 mm (P = 0.034) after three months. The ablative margins are shown in Table 5. Local tumor progression (LTP) at twelve months follow-up was documented in 0% (0/26) of MWA interventions and in 8.3% (2/24) of RFA interventions. Distant intrahepatic recurrence (DR) within twelve months occurred in 38.4% (10/26) of patients in the MWA-group and in 37.5% (9/24) of patients in the RFA-group. Differences between the groups were not significant (all P ≥ 0.07). Case examples are shown in Figs. 4a-d and 5a-d.
Table 5.
Comparison of diameter of ablative margin.
| Diameter (mm) | 24 h | 3 Months | 6 Months | 9 Months | 12 Months | LTP |
|---|---|---|---|---|---|---|
|
MWA Mean SD Median Range |
24.2 11.6 22.7 5.6–49.3 |
17.9 6.6 17.7 8.4–30.9 |
17.5 8.3 19.7 6.7–35.4 |
16.9 6.95 18.6 6.4–27 |
15.9 9.1 12.8 3.7–31.3 |
0/24 |
|
RFA Mean SD Median Range |
22.1 11.7 20.1 6.2–50.3 |
14.3 7.5 13.5 0.2–29.0 |
10.7 3.6 11.2 5.4–16.9 |
8.1 6.9 9.4 7.2–27 |
8.3 6.1 9.5 4.9–22.8 |
2/26 |
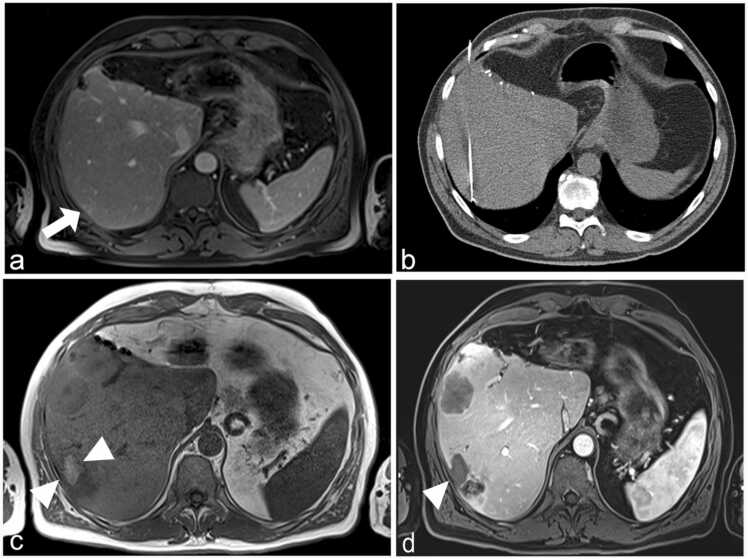
Fig. 4.
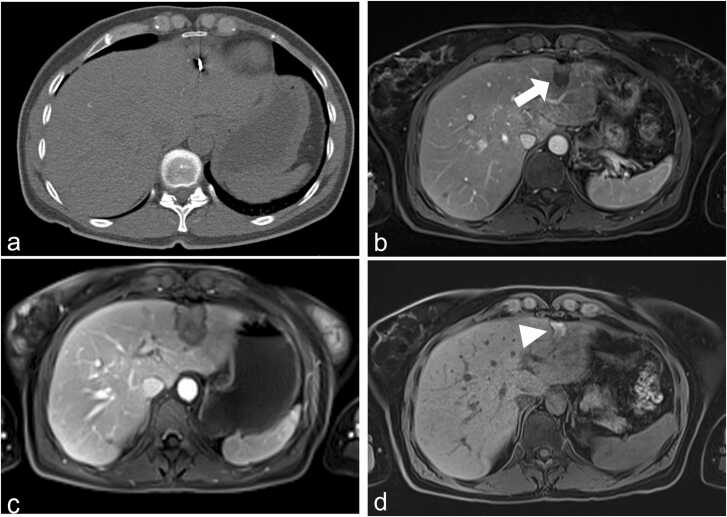
61-year-old male patient with recurrent colorectal liver metastasis (arrow) (a). The RFA electrode applicator was placed under CT guidance and thermal ablation was performed with maximal RF energy (220 W) (b). The ablation zone (arrowheads) is visible in unenhanced (c) and contrast-enhanced MR (d) after 12 months.
Fig. 5.
47-year-old female patient with colorectal liver metastasis who underwent MWA treatment. The MWA antenna was placed under CT guidance and thermal ablation was performed with 140 W (a). Contrast-enhanced MRI after 24 h shows the ablation zone (b). Contrast-enhanced (c) and unenhanced (d) MRI after 12 months show a complete remission with A0 ablation (arrows). The scar demonstrates this with a moderate high signal intensity (arrowhead).
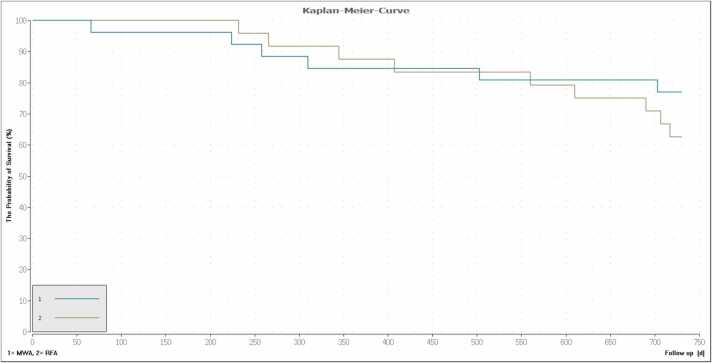
The one-year survival rates were 87.5% (21/24) in the RFA and 84.6% (22/26) in the MWA-group. The two-year survival rates from the day of treatment were 62.5% (15/24) for RFA and 76.9% (20/26) for MWA. The one- and two-year survival rates showed no significant differences between the two groups (P ≥ 0.60). All causes of death were tumor-associated. The corresponding Kaplan-Meier curves are shown in Fig. 6.
Fig. 6.
Survival curve showing the difference between MWA (turquoise) and RFA (olive) for the follow-up period of two years. The one-year survival rate from the day of therapy was 86.0% (43/50). (87.5% (21/24) for RFA and 84.6% (22/26) for MWA). The two-year survival rate was 70.0% (35/50) (62.5% (15/24) for RFA and 76.9% (20/26) for MWA). The differences were not significant (P ≥ 0.11).
4. Discussion
The main aim of this study was to prospectively evaluate and compare therapy response of RFA and MWA in patients with hepatic metastases using a one-generator system. We found out that MWA and RFA are both safe and effective methods for the treatment of liver metastasis. No significant differences were found for overall survival, rate of neoplasm, or major complications between both groups. In addition, MWA generated greater ablation volumes compared to RFA.
During the follow-up period of twelve months, the number of LTP was lower in the MWA-group compared to the RFA-group. The results showed that RFA could not reach the desired size of an ablation margin greater than 10 mm in all patients after 12 months post ablation. Shady et al. reported no LTP for margins over 10 mm regardless of the thermal modality used [25], [26], [27]. A smaller ablation margin is generally associated with a higher LTP [28]. However, an ablation margin of 5–10 mm also showed good results in several previous studies [17], [28], [29], [30], [31]. An ablation zone with an ablation margin greater than 5 mm postablation was in former studies associated with the best local tumor control [32], [33].
In our study we reported two cases of LTP in the RFA-group, both with ablative margin greater than 5 mm after 3 months postablation. Especially mutant RAS metastases which are associated with an earlier and higher rate of LTP should be treated with ablative margins greater than 10 mm [34], [35], [36]. Future studies with a more patients should be carried out to obtain more accurate results on the clinical outcomes and to identify differences in long-term LTP between the systems.
Our results showed that larger ablation zones and volumes can be achieved with MWA in comparison to RFA. One reason may be the fact that efficiency of MWA is unlike RFA not affected by perivascular tumors [25]. This fact also may correlate with greater ablative margins achieved in MWA-group and can be supported by lower LTP rates of MWA in comparison with RFA. However, no significant results for ablation volumes between MWA and RFA were found after the follow-up period of twelve months. This may be explained by the small patient cohorts in this study. As index tumors were similar, it would be interesting to evaluate the possibility of sparing normal liver parenchyma while creating sufficient margins due to a more precise ablation zone formation.
To our knowledge, no previous studies have compared the ADC change after thermal ablation. However, few studies evaluated the ADC change after chemotherapy. In our study, we demonstrated that metastatic lesions have higher ADC values than normal hepatic parenchyma before treatment which supports the findings of a previous study by Koh et al. [24]. In this study, the authors showed that patients with high ADC values before treatment were not responsive to chemotherapy. Moreover, Koh and Zheng revealed that ADC has increased over time in the effective group with chemotherapy [24], [30]. In our study, pre-treatment ADC values were higher in the MWA than in RFA-group. In both groups there were significant higher ADC values 24 h and 3 months after ablation (P = 0.005 vs. P = 0.024). Nevertheless, there were no significant differences for ADC parameters between the two groups after twelve months follow-up (P = 0.566).
In our study we found no significant differences for overall survival after one year between RFA and MWA. After the clinical follow-up of two years, we found that more patients with MWA therapy have survived in comparison to RFA; however, differences regarding the overall survival were not significant. Few studies have evaluated the survival rates after MWA and have shown similar outcomes compared to our study [21], [37]. Furthermore, the results of this study demonstrated that the evaluated one-generator MWA and RFA system is safe and efficient. A direct comparison of the ablated areas showed that MWA can induce a significantly larger necrotic area [17], [21]. The higher mortality in the MWA group after the follow-up period of one year may be explained by a higher number of pancreatic carcinomas and consequently poor prognosis of this disease. Further clinical evaluation of the effect of the primary origin would be interesting.
In addition, our analysis revealed that there was no strong correlation between ADC values and the mean tumor diameter before or after treatment. There were certain limitations in our study. First, the assessed patient sample size was relatively small. Second, we included patients with different tumor entities. A dedicated analysis of this system in a specific tumor subtype would be helpful, especially that the influence of primary tumor origin analysis and genetic mutations as predictors for oncological outcomes were not assessed in this study [35], [37]. Third, the Amica system was used to compare MWA and RFA, however, there are more than ten other systems used for the thermal ablation. Fourth, ablation of liver metastasis in patients with pancreatic carcinoma is generally not considered as standard of care. However, in selected patients with pancreatic carcinoma the resection or ablation of hepatic metastases might be a therapeutic option [14], [21], [38]. Finally, this study did not consider individual differences between the RFA and MWA-group.
In conclusion, MWA and RFA are both safe and effective methods for the treatment of liver metastases with MWA generating greater volumes of ablation. No significant differences were found for overall survival, rate of neoplasm, or major complications between both groups.
Compliance with Ethical Standards
Funding
This study was partially funded by Medicor (MMS Medicor Medical Supplies GmbH, Kerpen, Germany) for the statistical analysis of this work. The study design and data analysis were performed by the authors who are not affiliated to this company.
Compliance with Ethical Standards
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The current study was approved by the institutional ethical committee of our University Hospital Frankfurt (IRB number 322/13).
Informed consent
Informed consent was obtained from all individual participants included in the study.
CRediT authorship contribution statement
Thomas J. Vogl: Conceptualization, Methodology, Software, Validation, Formal analysis, Investigation, Resources, Data curation, Writing – original draft, Visualization, Supervision, Project administration, Funding acquisition. Yousef Jaraysa: Methodology, Software, Validation, Formal analysis, Investigation, Data curation, Writing – original draft, Visualization. Simon S. Martin: Software, Writing – original draft, Writing – review & editing. Tatjana Gruber-Rouh: Conceptualization, Resources, Writing – review & editing, Supervision. Rock H. Savage: Writing – original draft, Writing – review & editing. Nour-Eldin A. Nour-Eldin: Resources, Supervision. Amela Mehmedovic: Methodology, Software, Validation, Formal analysis, Investigation, Data curation, Writing – original draft, Visualization.
Consent for publication
Consent for publication was obtained for every individual person’s data included in the study.
Declaration of Competing Interest
Dr. Martin has received speakers’ fees from Siemens Healthineers. The other authors declare that they have no conflict of interest.
Acknowledgements
The current study was approved by the institutional ethical committee of our University Hospital Frankfurt (IRB number 322/13) and informed consent existed from every patient.
References
- 1.Ohlsson B., Stenram U., Tranberg K.G. Resection of colorectal liver metastases: 25-year experience. World J. Surg. 1998;22(3):268–276. doi: 10.1007/s002689900381. discussion 276-7. [DOI] [PubMed] [Google Scholar]
- 2.Scheele J., Stang R., Altendorf-Hofmann A., Paul M. Resection of colorectal liver metastases. World J. Surg. 1995;19(1):59–71. doi: 10.1007/BF00316981. [DOI] [PubMed] [Google Scholar]
- 3.Pathak S., Jones R., Tang J.M., Parmar C., Fenwick S., Malik H., Poston G. Ablative therapies for colorectal liver metastases: a systematic review. Colorectal Dis. 2011;13(9):e252–e265. doi: 10.1111/j.1463-1318.2011.02695.x. [DOI] [PubMed] [Google Scholar]
- 4.Schwarz C., Kaczirek K., Bodingbauer M. Liver resection for non-colorectal metastases. Eur. Surg. 2018;50(3):113–116. doi: 10.1007/s10353-018-0528-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gandy R.C., Bergamin P.A., Haghighi K.S. Hepatic resection of non-colorectal non-endocrine liver metastases. ANZ J. Surg. 2017;87(10):810–814. doi: 10.1111/ans.13470. [DOI] [PubMed] [Google Scholar]
- 6.Ruers T., Punt C., Van Coevorden F., Pierie J.P., Borel-Rinkes I., Ledermann J.A., Poston G., Bechstein W., Lentz M.A., Mauer M., Van Cutsem E., Lutz M.P., Nordlinger B. A.L.u.-t.i.d.C.A.O. Eortc Gastro-Intestinal Tract Cancer Group, G. the National Cancer Research Institute Colorectal Clinical Study, Radiofrequency ablation combined with systemic treatment versus systemic treatment alone in patients with non-resectable colorectal liver metastases: a randomized EORTC Intergroup phase II study (EORTC 40004) Ann. Oncol. Off. J. The Eur. Soc. Med. Oncol. 2012;23(10):2619–2626. doi: 10.1093/annonc/mds053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Poulou L.S., Thanos L., Ziakas P.D., Merikas E., Achimastos A., Gennatas C., Syrigos K.N. Thermal ablation may improve outcomes in patients with colorectal liver metastasis: a case-control study. J. BUON. 2017;22(3):673–678. [PubMed] [Google Scholar]
- 8.Andreano A., Brace C.L. A comparison of direct heating during radiofrequency and microwave ablation in ex vivo liver. Cardiovasc. Intervent. Radiol. 2013;36(2):505–511. doi: 10.1007/s00270-012-0405-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hoffmann R., Rempp H., Clasen S. Mikrowellenablation. Der Radiol. 2012;52(1):22–28. doi: 10.1007/s00117-011-2208-9. [DOI] [PubMed] [Google Scholar]
- 10.Tsai S., Pawlik T.M. Outcomes of ablation versus resection for colorectal liver metastases: are we comparing apples with oranges? Ann. Surg. Oncol. 2009;16(9):2422–2428. doi: 10.1245/s10434-009-0491-8. [DOI] [PubMed] [Google Scholar]
- 11.Fan W.J., Li X., Zhang L., Jiang H., Zhang J.L. Comparison of Microwave Ablation and Multipolar Radiofrequency Ablation In Vivo Using Two Internally Cooled Probes. Am. J. Roentgenol. 2012;198(1):W46–W50. doi: 10.2214/AJR.11.6707. [DOI] [PubMed] [Google Scholar]
- 12.Erce C., Parks R.W. Interstitial ablative techniques for hepatic tumours. Brit. J. Surg. 2003;90(3):272–289. doi: 10.1002/bjs.4091. [DOI] [PubMed] [Google Scholar]
- 13.Mcgahan J.P., Browning P.D., Brock J.M., Tesluk H. Hepatic Ablation Using Radiofrequency Electrocautery. Investig. Radiol. 1990;25(3):267–270. doi: 10.1097/00004424-199003000-00011. [DOI] [PubMed] [Google Scholar]
- 14.Yun B.L., Lee J.M., Baek J.H., Kim S.H., Lee J.Y., Han J.K., Choi B.I. Radiofrequency ablation for treating liver metastases from a non-colorectal origin. Korean J. Radiol. 2011;12(5):579–587. doi: 10.3348/kjr.2011.12.5.579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Takahashi H., Kahramangil B., Kose E., Berber E. HPB (Oxford),; 2018. A comparison of microwave thermosphere versus radiofrequency thermal ablation in the treatment of colorectal liver metastases; pp. 1157–1162. [DOI] [PubMed] [Google Scholar]
- 16.Heerink W.J., Solouki A.M., Vliegenthart R., Ruiter S.J.S., Sieders E., Oudkerk M., de Jong K.P. The relationship between applied energy and ablation zone volume in patients with hepatocellular carcinoma and colorectal liver metastasis. Eur. Radiol. 2018;28(8):3228–3236. doi: 10.1007/s00330-017-5266-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vogl T.J., Basten L.M., Nour-Eldin N.A., Kaltenbach B., Bodelle B., Wichmann J.L., Ackermann H., Naguib N.N.N. Evaluation of microwave ablation of liver malignancy with enabled constant spatial energy control to achieve a predictable spherical ablation zone. Int. J. Hyperth. 2018;34(4):492–500. doi: 10.1080/02656736.2017.1358408. [DOI] [PubMed] [Google Scholar]
- 18.Smolock A.R., Lubner M.G., Ziemlewicz T.J., Hinshaw J.L., Kitchin D.R., Brace C.L., Lee F.T. Microwave ablation of hepatic tumors abutting the diaphragm is safe and effective. Am. J. Roentgenol. 2015;204(1):197–203. doi: 10.2214/AJR.14.12879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ahmed M., Solbiati L., Brace C.L., Breen D.J., Callstrom M.R., Charboneau J.W., Chen M.-H., Choi B.I., De Baère T., Dodd III G.D. Image-guided tumor ablation: standardization of terminology and reporting criteria—a 10-year update. Radiology. 2014;273(1):241–260. doi: 10.1148/radiol.14132958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Vogl T.J., Helmberger T.K., Mack M.G., Reiser M.F. Springer; 2008. Percutaneous Tumor Ablation in Medical Radiology. [Google Scholar]
- 21.Vogl T.J., Hagar A., Nour-Eldin N.A., Gruber-Rouh T., Eichler K., Ackermann H., Bechstein W.O., Naguib N.N. High-frequency versus low-frequency microwave ablation in malignant liver tumours: evaluation of local tumour control and survival. Int. J. Hyperth. 2016;32(8):868–875. doi: 10.1080/02656736.2016.1212107. [DOI] [PubMed] [Google Scholar]
- 22.Le Bihan D. Molecular diffusion, tissue microdynamics and microstructure. NMR Biomed. 1995;8(7–8):375–386. doi: 10.1002/nbm.1940080711. [DOI] [PubMed] [Google Scholar]
- 23.Le Bihan D. Intravoxel incoherent motion imaging using steady‐state free precession. Magn. Reson. Med. 1988;7(3):346–351. doi: 10.1002/mrm.1910070312. [DOI] [PubMed] [Google Scholar]
- 24.Koh D.M., Scurr E., Collins D., Kanber B., Norman A., Leach M.O., Husband J.E. Predicting response of colorectal hepatic metastasis: value of pretreatment apparent diffusion coefficients. AJR Am. J. Roentgenol. 2007;188(4):1001–1008. doi: 10.2214/AJR.06.0601. [DOI] [PubMed] [Google Scholar]
- 25.Shady W., Petre E.N., Do K.G., Gonen M., Yarmohammadi H., Brown K.T., Kemeny N.E., D’Angelica M., Kingham P.T., Solomon S.B., Sofocleous C.T. Percutaneous Microwave versus Radiofrequency Ablation of Colorectal Liver Metastases: Ablation with Clear Margins (A0) Provides the Best Local Tumor Control. J. Vasc. Interv. Radiol. JVIR. 2018;29(2):268–275. doi: 10.1016/j.jvir.2017.08.021. e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shady W., Petre E.N., Gonen M., Erinjeri J.P., Brown K.T., Covey A.M., Alago W., Durack J.C., Maybody M., Brody L.A., Siegelbaum R.H., D’Angelica M.I., Jarnagin W.R., Solomon S.B., Kemeny N.E., Sofocleous C.T. Percutaneous radiofrequency ablation of colorectal cancer liver metastases: factors affecting outcomes-a 10-year experience at a single center. Radiology. 2016;278(2):601–611. doi: 10.1148/radiol.2015142489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Benson, 3rd A.B., Venook A.P., Cederquist L., Chan E., Chen Y.J., Cooper H.S., Deming D., Engstrom P.F., Enzinger P.C., Fichera A., Grem J.L., Grothey A., Hochster H.S., Hoffe S., Hunt S., Kamel A., Kirilcuk N., Krishnamurthi S., Messersmith W.A., Mulcahy M.F., Murphy J.D., Nurkin S., Saltz L., Sharma S., Shibata D., Skibber J.M., Sofocleous C.T., Stoffel E.M., Stotsky-Himelfarb E., Willett C.G., Wu C.S., Gregory K.M., Freedman-Cass D. Colon cancer, version 1.2017, NCCN clinical practice guidelines in oncology. J. Natl. Compr Canc. Netw. 2017;15(3):370–398. doi: 10.6004/jnccn.2017.0036. [DOI] [PubMed] [Google Scholar]
- 28.Bhardwaj N., Strickland A.D., Ahmad F., Atanesyan L., West K., Lloyd D.M. A comparative histological evaluation of the ablations produced by microwave, cryotherapy and radiofrequency in the liver. Pathology. 2009;41(2):168–172. doi: 10.1080/00313020802579292. [DOI] [PubMed] [Google Scholar]
- 29.Koh D.M., Scurr E., Collins D.J., Pirgon A., Kanber B., Karanjia N., Brown G., Leach M.O., Husband J.E. Colorectal hepatic metastases: quantitative measurements using single-shot echo-planar diffusion-weighted MR imaging. Eur. Radiol. 2006;16(9):1898–1905. doi: 10.1007/s00330-006-0201-x. [DOI] [PubMed] [Google Scholar]
- 30.Zheng D.X., Meng S.C., Liu Q.J., Li C.T., Shang X.D., Zhu Y.S., Bai T.J., Xu S.M. Predicting liver metastasis of gastrointestinal tract cancer by diffusion-weighted imaging of apparent diffusion coefficient values. World J. Gastroenterol. 2016;22(10):3031–3037. doi: 10.3748/wjg.v22.i10.3031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Curley S.A. Radiofrequency ablation of malignant liver tumors. Ann. Surg. Oncol. 2003;10(4):338–347. doi: 10.1245/aso.2003.07.017. [DOI] [PubMed] [Google Scholar]
- 32.Sotirchos V.S., Petrovic L.M., Gonen M., Klimstra D.S., Do R.K.G., Petre E.N., Garcia A.R., Barlas A., Erinjeri J.P., Brown K.T., Covey A.M., Alago W., Brody L.A., DeMatteo R.P., Kemeny N.E., Solomon S.B., Manova-Todorova K.O., Sofocleous C.T. Colorectal cancer liver metastases: biopsy of the ablation zone and margins can be used to predict oncologic outcome. Radiology. 2016;280(3):949–959. doi: 10.1148/radiol.2016151005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang X.D., Sofocleous C.T., Erinjeri J.P., Petre E.N., Gonen M., Do K.G., Brown K.T., Covey A.M., Brody L.A., Alago W., Thornton R.H., Kemeny N.E., Solomon S.B. Margin size is an independent predictor of local tumor progression after ablation of colon cancer liver metastases. Cardiovasc. Interv. Radiol. 2013;36(1):166–175. doi: 10.1007/s00270-012-0377-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Odisio B.C., Yamashita S., Huang S.Y., Harmoush S., Kopetz S.E., Ahrar K., Shin Chun Y., Conrad C., Aloia T.A., Gupta S., Hicks M.E., Vauthey J.N. Local tumour progression after percutaneous ablation of colorectal liver metastases according to RAS mutation status. Brit. J. Surg. 2017;104(6):760–768. doi: 10.1002/bjs.10490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shady W., Petre E.N., Vakiani E., Ziv E., Gonen M., Brown K.T., Kemeny N.E., Solomon S.B., Solit D.B., Sofocleous C.T. Kras mutation is a marker of worse oncologic outcomes after percutaneous radiofrequency ablation of colorectal liver metastases. Oncotarget. 2017;8(39):66117–66127. doi: 10.18632/oncotarget.19806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Calandri M., Yamashita S., Gazzera C., Fonio P., Veltri A., Bustreo S., Sheth R.A., Yevich S.M., Vauthey J.N., Odisio B.C. Ablation of colorectal liver metastasis: interaction of ablation margins and RAS mutation profiling on local tumour progression-free survival. Eur. Radiol. 2018;28(7):2727–2734. doi: 10.1007/s00330-017-5273-2. [DOI] [PubMed] [Google Scholar]
- 37.Veltri A., Guarnieri T., Gazzera C., Busso M., Solitro F., Fora G., Racca P. Vol. 117. 2012. Long-term outcome of radiofrequency thermal ablation (RFA) of liver metastases from colorectal cancer (CRC): size as the leading prognostic factor for survival; pp. 1139–1151. (Radiologia Medica). [DOI] [PubMed] [Google Scholar]
- 38.Shi S., Yu X.J. Time to think: Selecting patients who may benefit from synchronous resection of primary pancreatic cancer and liver metastases. World J. Gastroenterol. 2018;24(33):3677–3680. doi: 10.3748/wjg.v24.i33.3677. [DOI] [PMC free article] [PubMed] [Google Scholar]