Abstract
The susceptibility of Aspergillus fumigatus to mulundocandin, an echinocandin-like compound, and other antifungal agents was assessed by the National Committee for Clinical Laboratory Standards (NCCLS) M38-P method, a 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenyl-amino)carbonyl]-2H-tetrazolium hydroxide (XTT)-based colorimetric assay, and determination of morphologic alterations by microscopy. In contrast to the NCCLS M38-P method, which does not predict the activity in vivo, the XTT-based assay showed that A. fumigatus is susceptible to mulundocandin. Thus, the XTT-based assay might be useful for determination of the susceptibilities of molds to echinocandins. Further evaluation is warranted.
The National Committee for Clinical Laboratory Standards (NCCLS) is developing a reference method for broth dilution antifungal susceptibility testing of filamentous fungi (12). This method does not, however, incorporate standardized testing of new classes of antifungal agents such as the macrocyclic lipopeptide antifungal agents (9). Examples of antifungal agents from the macrocyclic lipopeptide class are caspofungins, FK463, and mulundocandin (5). These compounds are echinocandin-like lipopeptide antifungal agents, which are assumed to exert their antifungal activity through the inhibition of β-(1,3)-d-glucan synthesis (9). Mulundocandin is an echinocandin-like lipopeptide obtained from a variant of Aspergillus sydowii (13). It has antifungal activity against Candida strains, including fluconazole-resistant isolates, and is being developed by Aventis (Romainville, France). However, this compound is poorly active against other non-Candida albicans isolates and is inactive against Cryptococcus neoformans (5). The biological activity of mulundocandin and structural elucidation studies with the compound have been described previously (5, 11).
In addition to the NCCLS proposed methodology, several studies have demonstrated the use of alternative assays that may be useful for the susceptibility testing of fungi, including Aspergillus species. These include colorimetric susceptibility assays based upon the use of the tetrazolium salt 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) or Alamar Blue (7, 15) and agar-based assays such as the E-test (3). MTT has also been shown to be useful in this context for the susceptibility testing of yeasts (2), as has a related tetrazolium salt, 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenyl-amino)carbonyl]-2H-tetrazolium hydroxide (XTT) (4, 6, 14). In the study described here, we have used the XTT-based assay in order to evaluate the effects of mulundocandin on Aspergillus fumigatus in vitro and compared this technique with the microdilution and morphology assays performed by the NCCLS M38-P method. We focused on A. fumigatus because it is the primary pathogen (responsible for approximately 90% of human aspergillosis) causing life-threatening invasive aspergillosis, pulmonary aspergilloma, allergic bronchopulmonary aspergillosis, and extrapulmonary infections (for a review, see reference 10).
The organisms used included the following clinical isolates: A. fumigatus WGHU 8896, WGHU 8901, and BP 9220 (kind gifts from M. Richardson, Mycology Reference Laboratory, Glasgow, United Kingdom). A. fumigatus ATCC 9643, a reference strain, was also used in our study. The isolates were grown and maintained on potato dextrose agar slants. The susceptibilities of the isolates to mulundocandin (prepared at Aventis, Frankfurt, Germany), amphotericin B and flucytosine (Sigma Chemical Co., St. Louis, Mo.), and fluconazole (synthesized at Aventis) were assessed by the broth microdilution technique, which is similar to the M38-P method for the susceptibility testing of filamentous fungi (12). Conidia were counted with a hemocytometer, and 104 conidia/ml (100-μl aliquots) were inoculated into wells of 96-well microtiter plates containing 100 μl of RPMI 1640 medium (12). The antifungal agents tested were amphotericin B and itraconazole (0.015 to 16 μg/ml), fluconazole and flucytosine (0.13 to 64 μg/ml), and mulundocandin (0.1 to 256 μg/ml). The plates were incubated for 72 h at 35°C. The MICs of amphotericin B and mulundocandin were defined as the lowest drug concentrations that caused 100% inhibition of fungal growth compared to the growth in the control well, while for fluconazole and flucytosine the MICs were defined as the lowest concentrations of the antifungal agents that cause 80% inhibition compared to the growth in the control wells. The MICs were determined visually. The second susceptibility assay was performed as described above, with the following additions. Three hours prior to reading of the standard visual MIC, the plates were agitated to resuspend the cell pellets in the wells of flat-bottom plates. Unlike the microtiter plates with U-shaped wells used in the M38-P method, we used flat-bottom plates in our study because such plates were used previously in colorimetric assays (7). Thereafter, 50-μl aliquots of a solution containing 200 μg of XTT (Sigma) per ml and 20 μg of phenazene methylsulfate (Sigma) per ml were added in order to measure the metabolic activities of untreated (control) and antifungal agent-treated cells. The plates were incubated for 3 h at 35°C to allow color development. The absorbance at 492 nm was read with a Titertek microtiter plate reader. The absorbance values for antifungal agent-treated cells were compared with the absorbance values for untreated control cells, and the inhibitory concentration that resulted in an 80% reduction in metabolism (IC80) was calculated from dose-response curves.
The effects of mulundocandin on the morphology of A. fumigatus cells were assessed by using the minimum effective concentrations (MECs), as described previously (8). Briefly, the plates were prepared in exactly the same way as they were for testing by the M38-P method, and the effects of mulundocandin on the morphologies of A. fumigatus cells were assessed by viewing the cells under a light microscope after 18 h of incubation at 35°C. The MEC was defined as the minimum concentration of mulundocandin that produced morphologic alterations of A. fumigatus cells (abnormal hyphal growth with highly branched tips, swollen germ tubes, and distended balloon-like cells). To critically evaluate the effects of mulundocandin on the morphologies of A. fumigatus cells, the cells were examined by scanning electron microscopy (SEM). The cells were taken from microtiter plates prepared by the M38-P method and included untreated control cells taken from growth control wells. Cells were also taken from wells which contained the corresponding IC80 (see description of the XTT-based assay above) of mulundocandin. The cells were harvested, centrifuged, and subjected to three washes of 10 min each with 0.15 M phosphate-buffered saline (pH 7.2). Thereafter, the cells were fixed in 3% glutaraldehyde (cacodylate buffer) for 1 h. The samples were then washed three times (10 min each time) with buffer and postfixed in 1% OsO4 (prepared in cacodylate buffer). The samples were dehydrated with increasing concentrations of ethanol, immersed in hexamethyldisilazane (Sigma) for 5 min, dried, and then sputter coated. Samples were viewed with a JEOL 480 scanning electron microscope.
Using the M38-P method, we observed that the A. fumigatus isolates showed different susceptibilities to the antifungal agents. For example, we noted that they were poorly susceptible to mulundocandin (MIC range, 64 to 128 μg/ml), as has also been shown in a previous study (5). In contrast, amphotericin B (MIC range, 0.03 to 0.12) and itraconazole (MIC range, 0.5 to 1.0 μg/ml) were inhibitory, while fluconazole (MIC range, 16 to 64 μg/ml) and flucytosine (MIC range, 0.5 to 16 μg/ml) exhibited poor or moderate levels of inhibitory activity, respectively.
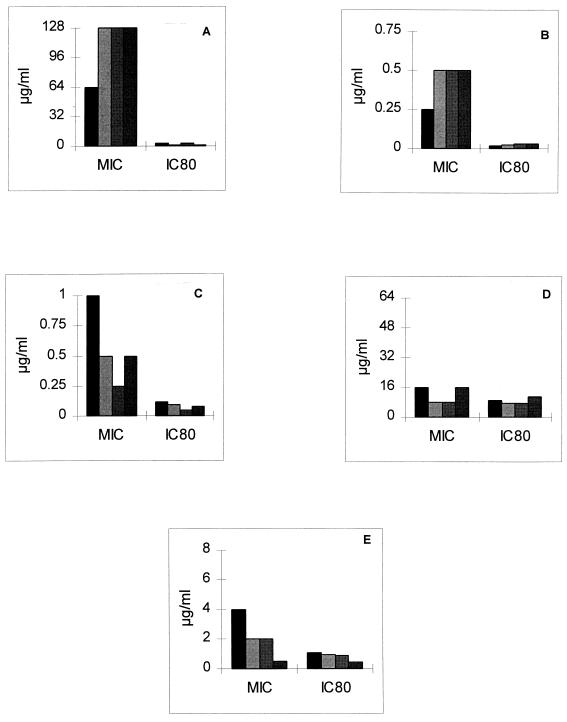
Previous studies have shown that tetrazolium salts are useful in assessing more quantitatively the effects of antifungal agents in MIC experiments (6, 14). Colorimetric methods with tetrazolium salts such as XTT and MTT are based on the fact that these salts are taken up by living cells and are reduced by mitochondrial dehydrogenase of the fungi to colored tetrazolium formazan products that are determined spectrophotometrically. While both XTT and MTT are useful, XTT provides two main advantages over MTT: it yields a water-soluble formazan product, eliminating the need for a cell lysis step, and it does not require dimethyl sulfoxide as a solubilizing agent for the reduced product. Tetrazolium salts are useful for determination of antifungal susceptibility, as they generate clear-cut endpoints based on a visually detectable color change that is quantifiable (6, 7). Hence, using the XTT-based assay, we examined the susceptibilities of all isolates to each of the antifungal agents. Figure 1 shows that amphotericin B was uniformly active against all of the isolates tested and that the XTT IC80s for inhibition were low, in agreement with the MICs (Fig. 1). The IC80s and MICs of fluconazole and flucytosine were very similar, with the MICs being elevated. By contrast, by the XTT-based assay mulundocandin showed a marked reduction in metabolism, with IC80s being between 1/20 and 1/40 of the MIC (depending on the isolate tested) determined by the NCCLS M38-P method. These data suggest that mulundocandin possesses activity against Aspergillus isolates, despite the high MICs obtained by the M38-P method. Moreover, this in vitro susceptibility is in agreement with the in vivo data demonstrating the efficacy of this agent in murine models of aspergillosis (unpublished data).
FIG. 1.
Activities of antifungal agents against four A. fumigatus isolates, as measured by the M38-P method (MIC) and the XTT-based assay (IC80). The values are in micrograms per milliliter; and the graphs represent the activities of mulundocandin (A), amphotericin B (B), itraconazole (C), fluconazole (D), and flucytosine (E).
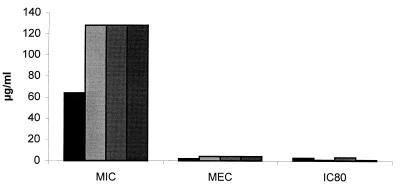
In order to confirm our findings that mulundocandin was active in vitro against Aspergillus isolates, we prepared similar microtiter plates and determined the MEC. We observed that mulundocandin does affect the morphology of Aspergillus cells when it is tested at low concentrations (sub-MICs). For example, incubation (for 18 h at 35°C) of A. fumigatus cells in the presence of sub-MICs of mulundocandin had a significant effect on the morphology of the fungus. These cells were much shorter and malformed compared with untreated control cells (Fig. 2). We also noted that there was a good correlation between the MECs and the IC80s by the XTT-based assay (Fig. 3). By contrast, the correlation of the MIC generated by the M38-P method with either the MEC or the IC80 was very poor (Fig. 3). Our results are consistent with those of Chiou et al. (C. C. Chiou, N. Mavrogiorgos, and T. J. Walsh, Abstr. 40th Intersci Conf. Antimicrob. Agents Chemother., abstr. 940, 2000), who used the MTT-based assay to investigate the antifungal activity of echinocandin in combination with other cell wall-active antifungal agents. Their data showed a correlation between the values obtained by the MTT-based assay and hyphal damage.
FIG. 2.
Effect of mulundocandin on the morphology of A. fumigatus WGHU 8896 hyphae. Hyphae were observed by SEM following 18 h of incubation at 35°C in the absence (A) or presence (B) of a sub-MIC of mulundocandin (1.0 μg/ml). The concentration tested corresponds to the MEC for this isolate.
FIG. 3.
Comparison of the activities of mulundocandin against four A. fumigatus isolates as determined by the classical methods of determination of the MIC, MEC, and IC80. All values are in micrograms per milliliter.
Overall the XTT-based antifungal assay allows one to determine the anti-Aspergillus activities of antifungal agents of the macrocyclic lipopeptide antifungal class and other classes of antifungal molecules. Some molecules of the lipopeptide class do show moderate or poor activity against Aspergillus spp. by the classical MIC testing method, as was previously seen with cilofungin, whereby the compound was inactive when it was tested by classical MIC methods but was observed to be efficacious in a murine model of aspergillosis (1). In the present study, mulundocandin showed very poor activity by the M38-P method, although it was active by both the MEC assay and the XTT-based assay. The XTT-based assay in combination with the MEC assay might allow one to fully appreciate the anti-Aspergillus activities of antifungal agents such as mulundocandin. However, it is important to emphasize that the XTT-based assay alone can determine the susceptibility of A. fumigatus to echinocandins. Examination by SEM is not an integral part of the test and was used in our study to critically evaluate the morphologic changes induced by the lipopeptide. Further studies with a large panel of filamentous fungi should be undertaken to evaluate the clinical usefulness of the XTT-based method for determination of the susceptibilities of these pathogens to echinocandin-like compounds.
Acknowledgments
We thank Mohammad A. Hossain for critical review of the manuscript.
REFERENCES
- 1.Beaulieu D, Tang J, Zeckner D J, Parr T R., Jr Correlation of cilofungin in vivo efficacy with its activity against Aspergillus fumigatus (1,3)-β-d-glucan synthase. FEMS Microbiol Lett. 1993;108:133–138. doi: 10.1111/j.1574-6968.1993.tb06088.x. [DOI] [PubMed] [Google Scholar]
- 2.Clancy C J, Nguyen M H. Comparison of a photometric method with standardized methods of antifungal susceptibility testing of yeasts. J Clin Microbiol. 1997;35:2878–2882. doi: 10.1128/jcm.35.11.2878-2882.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Colombo A L, Barchiesi F, McGough D A, Fothergill A W, Rinaldi M G. Evaluation of the E test versus a microtiter broth method for antifungal susceptibility testing of yeasts against fluconazole and itraconazole. J Antimicrob Chemother. 1995;36:93–100. doi: 10.1093/jac/36.1.93. [DOI] [PubMed] [Google Scholar]
- 4.Hawser S. Comparisons of the susceptibilities of planktonic and adherent Candida albicans to antifungal agents: a modified XTT tetrazolium assay using synchronized C. albicans cells. J Med Vet Mycol. 1996;34:149–152. [PubMed] [Google Scholar]
- 5.Hawser S, Borgonovi M, Markus A, Isert D. Mulundocandin, an echinocandin-like lipopeptide antifungal agent: biological activities in vitro. J Antibiot. 1999;52:305–310. doi: 10.7164/antibiotics.52.305. [DOI] [PubMed] [Google Scholar]
- 6.Hawser S P, Norris H, Jessup C J, Ghannoum M A. Comparison of a 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenyl-amino)carbonyl]-2H-tetrazolium hydroxide (XTT) colorimetric method with the standardized National Committee for Clinical Laboratory Standards method of testing clinical yeast isolates for susceptibility to antifungal agents. J Clin Microbiol. 1998;36:1450–1452. doi: 10.1128/jcm.36.5.1450-1452.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jahn B, Stuben A, Bhakdi S. Colorimetric susceptibility testing for Aspergillus fumigatus: comparison of menadione-augmented 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide and Alamar Blue tests. J Clin Microbiol. 1996;34:2039–2041. doi: 10.1128/jcm.34.8.2039-2041.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kurtz M B, Heath I B, Marrinan J. Morphological effects of lipopeptides against Aspergillus fumigatus correlate with activities against (1,3)-β-d-glucan synthase. Antimicrob Agents Chemother. 1994;38:1480–1489. doi: 10.1128/aac.38.7.1480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kurtz M B, Douglas C M. Lipopeptide inhibitors of fungal glucan synthase. J Med Vet Mycol. 1997;35:79–86. doi: 10.1080/02681219780000961. [DOI] [PubMed] [Google Scholar]
- 10.Latge J P. Aspergillus fumigatus and aspergillosis. Clin Microbiol Rev. 1999;12:310–350. doi: 10.1128/cmr.12.2.310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mukhopadhyay T, Ganguli B N, Fehlhaber H W, Kogler H, Vertesy L. Mulundocandin, a new lipopeptide antibiotic. II. Structure elucidation. J Antibiot (Tokyo) 1987;40:281–289. doi: 10.7164/antibiotics.40.281. [DOI] [PubMed] [Google Scholar]
- 12.National Committee for Clinical Laboratory Standards. Reference method for broth dilution antifungal susceptibility testing of conidium-forming filamentous fungi. Proposed standard M38-P. Wayne, Pa: National Committee for Clinical Laboratory Standards; 1998. [Google Scholar]
- 13.Roy K, Mukhopadhyay T, Reddy C G S, Desikan K R, Ganguli B N. Mulundocandin, a new lipopeptide antibiotic. I. Taxonomy, fermentation, isolation and characterization. J Antibiot (Tokyo) 1987;40:275–280. doi: 10.7164/antibiotics.40.275. [DOI] [PubMed] [Google Scholar]
- 14.Tellier R, Krajden M, Grigoriew G A, Campbell I. Innovative endpoint determination system for antifungal susceptibility testing of yeasts. Antimicrob Agents Chemother. 1992;36:1619–1625. doi: 10.1128/aac.36.8.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tiballi R N, He X, Zarins L T, Revankar S G, Kauffman C A. Use of a colorimetric system for yeast susceptibility testing. J Clin Microbiol. 1995;33:915–917. doi: 10.1128/jcm.33.4.915-917.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]