Abstract
With reports from Australia, Canada, USA, Hawaii and Colorado documenting a link between cannabis and congenital anomalies (CAs), this relationship was investigated in Europe. Data on 90 CAs were accessed from Eurocat. Tobacco and alcohol consumption and median household income data were from the World Bank. Amphetamine, cocaine and last month and daily use of cannabis from the European Monitoring Centre for Drugs and Drug Addiction. Cannabis herb and resin Δ9-tetrahydrocannabinol concentrations were from published reports. Data were processed in R. Twelve thousand three hundred sixty CA rates were sourced across 16 nations of Europe. Nations with a higher or increasing rate of daily cannabis use had a 71.77% higher median CA rates than others [median ± interquartile range 2.13 (0.59, 6.30) v. 1.24 (0.15, 5.14)/10 000 live births (P = 4.74 × 10−17; minimum E-value (mEV) = 1.52]. Eighty-nine out of 90 CAs in bivariate association and 74/90 CAs in additive panel inverse probability weighted space-time regression were cannabis related. In inverse probability weighted interactive panel models lagged to zero, two, four and six years, 76, 31, 50 and 29 CAs had elevated mEVs (< 2.46 × 1039) for cannabis metrics. Cardiovascular, central nervous, gastrointestinal, genital, uronephrology, limb, face and chromosomalgenetic systems along with the multisystem VACTERL syndrome were particularly vulnerable targets. Data reveal that cannabis is related to many CAs and fulfil epidemiological criteria of causality. The triple convergence of rising cannabis use prevalence, intensity of daily use and Δ9-tetrahydrocannabinol concentration in herb and resin is powerfully implicated as a primary driver of European teratogenicity, confirming results from elsewhere.
Keywords: tobacco, alcohol, cannabis, cannabinoid, cancer, cancerogenesis, mutagenesis, oncogenesis, genotoxicity, epigenotoxicity, chromosomal toxicity
Introduction
Whilst it is often said that prenatal cannabis exposure has relatively benign implications in postnatal life [1–3], recent independent reports from Hawaii [4], Colorado [5], Canada [6, 7], Australia [8] and USA [9–11] indicate that dozens of congenital anomalies (CAs; birth defects) are likely epidemiologically causally associated with rising rates of community cannabis consumption. Systems that are particularly affected include the cardiovascular, gastrointestinal, chromosomal, genitourinary, limb and body wall systems. Concerns regarding prenatal exposure were provided with heightened salience by reports from many places indicating increased use of cannabis and cannabinoid products by pregnant women in recent times [12], by rates of cannabis use in pregnancy amongst teenagers as high as 25% [13], by increased use of cannabis in pregnancy since the COVID-19 pandemic [12] and by reports that 69% of cannabis dispensaries positively recommend cannabis use to women whilst pregnant [14]. Moreover, recent reports note a quadruple convergence of rising rates of cannabis use, Δ9-tetrahydrocannabinol (THC) potency, intensity of daily use and cannabis use disorder in Europe, suggesting that the modern era is actually experiencing a confluence of concerning teratogenic trends [15, 16].
The implications of cannabinoid genotoxicity are further highlighted with the recent data suggesting that multiple cancers (of breast, pancreas, thyroid, liver and acute lymphoid and myeloid leukaemias) are also epidemiologically causally related to cannabis use [17, 18] and, with the formal experimental demonstration in mice, that epigenomic programming actually controls the organism-wide ageing epigenomic cassettes [19]. The recent demonstration that cannabis is a major driver of the rise in USA paediatric cancer rates underscores the transgenerational nature of this mutagenesis [17, 20]. These data together indicate that cannabinoid-related epigenomic disturbances likely have broad public health implications for diverse communities extending to cancerogenesis on the one hand and pan-systemic ageing on the other and including transgenerational effects.
Key to any consideration of the possible causal relationships of cannabis with mutagenesis and teratogenesis is the elucidation of the biological pathways, which may underlie any apparently causal relationship. Multiple cannabinoids have long been known to be toxic to chromosomes, genes, DNA strands, DNA nucleosides, the epigenome, sperm, oocytes, mitosis and meiosis and the male and female reproductive tracts in multiple respects [21–38]. Several studies have also shown cannabis to have a major effect perturbing DNA methylation [31–37], with these changes shown to be inheritable to subsequent generations [31–37], to perturb DNA methylation in the nucleus accumbens of offspring and affect behaviour [33, 34], to be seen in human sperm [31, 32] and to improve in both rats and humans after cessation of cannabis exposure [32]. Cannabis has an adverse effect on protein synthesis including histone formation, a change which necessarily opens up chromatin for aberrant gene expression [40, 41]. Thus cannabinoids derange the ‘histone code’. They also adversely affect tubulin synthesis and the post-translational modifications (particularly glycosylation) of tubulin, which have been collectively referred to as the ‘tubulin code’ [42], which adversely affects both the microtubules of the mitotic spindle and the anaphase separation of chromosomes, and also the motility of sperm flagella and their ability to maintain linear forward progression in fertilization assays [42]. Numerous cannabinoids have adverse effects on mitochondrial metabolism in neurons, sperm, lymphocytes and pulmonocytes [22, 23, 43–51], which necessarily impairs the supply of methyl, acetyl, ubiquinyl, propyl, adenosine-ribose and many other groups for the epigenomic machinery, impairs ATP energy supply for the numerous energy-dependent genomic and epigenomic reactions required for normal genome maintenance and also deranges the delicate mitonuclear balance [52, 53].
Ready acces to various European metrics of cannabis exposure indicating the quadruple convergence of rising cannabinoid exposure [15, 16] along with access to comprehensive congenital anomaly rates from multiple national European registries together with newer statistical techniques allowing the examination of multiple models in a single analytical run for all anomalies considered together in a space-time context have provided an ideal opportunity to investigate these relationships in the contemporary European context. The hypothesis to be tested in this investigation was that the well-described genotoxicity and epigenotoxicity of cannabinoids seen in vitro may be manifested clinically in vivo at the level of child population health with various of the described metrics for cannabis use. Furthermore, we sought to employ statistical techniques of formal causal inference to allow epidemiologically causal relationships to be investigated beyond merely those of simple association. It was also relevant to compare links described in other jurisdictions with the European findings and to compare the relative effects of the known teratogens tobacco and alcohol.
In terms of anomaly classes of special interest, we were particularly interested to study those that had been previously identified in the literature as being cannabis related, such as chromosomal and genetic, cardiovascular, central nervous, gastrointestinal, urogenital and nephrological and limb anomalies [4–9, 15–18, 20, 54, 55]. Interestingly, the presence in the European data of a rare multisystem anomaly known as vertebral, anorectal, cardiac, tracheo-esophageal fistula ± oesophageal atresia, renal anomalies and limb abnormalities (VATER/VACTERL), which was described from Great Ormond St Hospital as a group of co-occurring anomalies [56] whose aetiology was recently ascribed to inhibition of sonic hedgehog signalling in utero, which is a known target of many cannabinoids [57], was of particular interest. All hypotheses were formulated prior to study commencement.
Methods
Data
Data on total congenital anomaly rates per 10 000 live births was downloaded from the Eurocat website for each nation and for each year separately for all available CAs [58]. Data on national birth rates and populations were taken from the World Bank [59]. Data on tobacco (percentage smoking) and alcohol (per capita annual litres of alcohol consumption) use were taken from the World Health Organization (WHO) Global Health Observatory [60]. Data on drug use were taken from the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) [61]. EMCDDA data on cannabis use and potency were supplemented by data provided in the recent report from the Manthey group relating to monthly and daily cannabis use, which was itself derived from EMCDDA [16]. Median household income data were derived from the World Bank [62]. Nations were chosen on the basis of base population, the availability of comprehensive data for 2010–2019, and for their place in the Supplementary Fig. 4 of Manthey and colleagues relating to rising/high or falling/low daily cannabis exposure [16]. Nations were divided dichotomously into rising or falling groups based on the findings presented in this figure.
Derived Data
As noted in the Introduction, Europe has been subject to a convergence of rising indices of cannabinoid exposure in the past decade. It was therefore of interest to see if a combination of these variables may have more explanatory power than the simple covariates themselves. Hence, last month cannabis use was multiplied by the THC potency of cannabis herb and cannabis resin to form compound covariates. Similarly these metrics were also multiplied by daily use rates to gain compound indices of use-intensity-potency for each nation. Quintiles of substance exposure were calculated by dividing the total range across the whole period into five equal parts with the ggplot2 function cut_number.
Data Interpolation
Data interpolation was undertaken for drug use for years for which data were missing. For nations with no data relating to drug exposure in any year, their data field was allowed to remain entirely missing. Both raw data and data after interpolation are provided in the online files in the Mendeley data repository.
Statistics
Data were processed in R studio 1.4.1717 based on R 4.1.1 from the comprehensive R Archive Network. Data were log transformed based on the Shapiro–Wilk test. Negative or zero rates were arcsinh transformed where required. Normally distributed data are quoted as mean ± SEM. Non-normally distributed data are quoted as median and interquartile range. Data were manipulated in dplyr, and graphs were drawn in ggplot2, both from the tidyverse [63]. All artwork is original and is not under pre-existing copyright. Linear regression was conducted in Base R. Linear models were reduced by the classical method of manual deletion of the least significant term [64]. Overall or marginal effects of additive or interactive models were calculated from the margins package [65]. Point estimates for the E-value and its 95% lower bound were calculated using the R package E-value [66–69]. Relative risk (RR), attributable fraction in the exposed (AFE) and population attributable risk (PAR) were calculated in the package epiR version 2.0.38 [70]. The R package ranger was used to conduct random forest regressions [71], and the package vip was used to construct variable importance plots [72]. Heatmaps were drawn using the R Package gplots [73]. Multiple models were analysed simultaneously as described below using purrr-map pipelines (from tidyverse [63]) incorporating functions from the R packages broom [74, 75], dplyr, margins and E-value. P-values were corrected for multiple testing by the algorithms of Bonferroni [76], Holm [77] and false discovery rate [78].
Multivariable regression was conducted using panel regression using the R package plm [79]. Panel regression was chosen for several reasons, including the fact that space and time could be considered simultaneously without consuming degrees of freedom, that models could be inverse probability weighted (IPW), that temporal lagging could be conducted, that models can be incorporated into purrr-map analytical pipelines and that model objects contained a standard deviation that allowed E-values to be calculated. All panel models contained all drug and income covariates. All panel models were IPW. Panel models were conducted using the two-ways method, which allows space and time to be studied simultaneously.
Causal Inference
The use of inverse probability weighting makes all groups in observational studies comparable, effectively pseudo-randomizing the study groups and transforms findings from mere associations into a formal causal paradigm. For this, the R package ipw was employed [80]. One of the classic issues faced by observational studies is that low-level findings may be due to an unidentified confounder variable, which is not controlled in the analysis. This issue is addressed by the use of the E-value (or expected value) [66], which is the degree of association required of some unobserved extraneous covariate with both the exposure of concern and the outcome of interest to explain away the observed effect. Both a point estimate and the 95% lower confidence interval are calculated. In the published literature, minimum E-values (mEV) >1.25 are said to indicate potentially causal effects [81].
Data Availability
Input and output data have been provided online through the Mendeley data repository doi: 10.17632/vd6mt5r5jm.1. Four files with R source code running to 18 830 lines of code are also supplied.
Ethics
Ethical approval for this study was provided from the Human Research Ethics Committee of the University of Western Australia number RA/4/20/4724 on 24 September 2021.
Results
Introduction
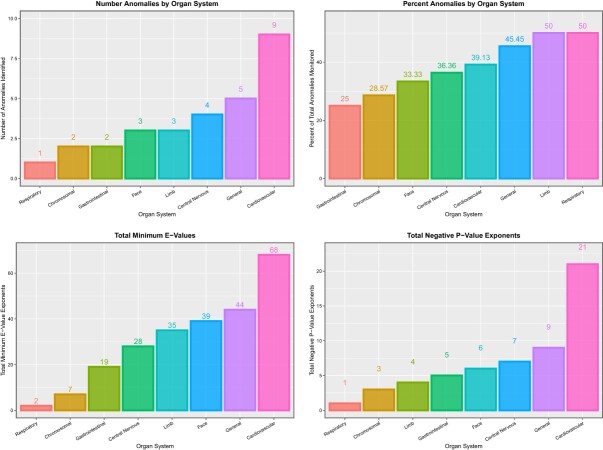
As shown in eTable 1, data on 90 CAs were downloaded from the Eurocat website. The Eurocat dataset has a major advantage in that it provides a total rate of anomalies so that foetuses that are not live born, either due to the severity of their condition or due to early termination for anomaly, are included in the total rates. These anomalies are listed in abbreviated form in this table. The key to their full name is shown in eTable 2. Data were derived from the 16 nations indicated. Measures for the compound derived indices of cannabinoid exposure are also shown in eTable 1. eTable 2 also provides the system assignment used. In most cases, this is self-evident. However, the eye is derived from both face structures in its anterior segments, and the retina and optic nerve are derived from outgrowths of the forebrain [82]. Whilst lens and glaucoma abnormalities have been assigned alongside face structures, eye anomalies overall have been assigned to the central nervous system (CNS). eTable 2B lists the anomalies by organ system. A summary of the numbers of anomalies in each system is provided in eTable 3. eTable 4 provides a breakdown of the numbers of anomalies in each system by system both as numbers and as percentages of the totals.
Overall Picture
eTable 5 indicates the assignment of nations into those in which daily cannabis use was either high/increasing or low/decreasing as documented in eFig. 4 of Manthey [16]. When the log (as arcsinh) of the anomaly rate is graphed against time, the results depicted in Fig. 1 are shown. The median (interquartile range) in the decreasing group is 1.041 (0.149, 2.338) and in the increasing group is 1.50 (0.56, 2.54). These results correspond to raw (sinh) anomaly rates of 1.24 (0.149, 5.136) and 2.13 (0.589, 6.300)/10 000 live births, respectively, indicating a 71.77% elevation in the increasing cannabis use intensity countries (t = 8.204, df = 4660, P = 2.99 × 10−16). At linear regression against time, this finding is also significant (β-estimate = 0.2506 (0.1092, 0.3089), P = 4.74 × 10−17), which correspond to point estimates for the E-value of 1.63 and 1.52 for its lower bound (mEV). E-values exceeding 1.25 are said in the literature to indicate likely causal relationships [81].
Figure 1:
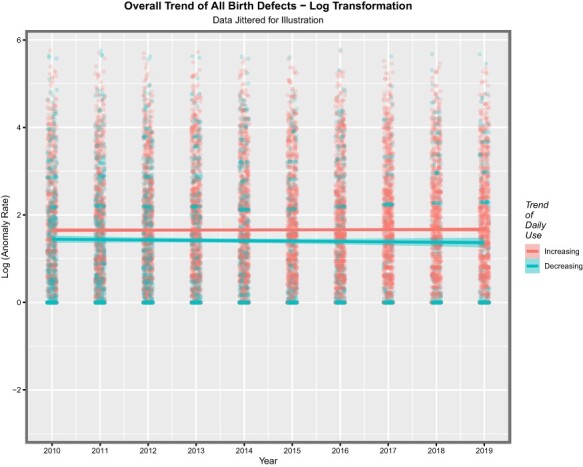
Overall trend of all CAs after log transformation
Substance and Time Trends—Continuous Analysis
eFigure 1A and B shows the rates of the 90 CAs across time. The figure has been split into two as there are so many anomalies to aid with presentation and readability. Genetic/chromosomal, cardiovascular and CNS anomalies are noted to feature amongst those which are rising. This list includes holoprosencephaly (a severe facial deformity) and VACTERL (a complex multisystem series of anomalies). eFigures 2A and 2B show the CA rate as a function of tobacco exposure. Only a few CAs are noted to rise. Again when alcohol is considered in eFig. 3, only a few anomalies are noted to rise. eFigures 4 and 5 perform similar roles for amphetamine and cocaine exposures, and it is noted that many more CAs are associated with positive gradients from these agents.
A similar exercise may be done for the THC concentration of cannabis herb. This is shown in eFig. 6. Here, all the CAs in eFig. 6A and half those in eFig. 6B are noted to demonstrate a positive relationship with rising herb THC concentration. This pattern is continued when last month cannabis exposure is considered in Fig. 2A and B, where all the CAs in Fig. 2A are noted to be rising with cannabis exposure. When daily cannabis use interpolated is considered, this pattern is again repeated as shown in Fig. 3A. The pattern is again repeated in the compound indices of last month herb and resin THC concentrations × interpolated daily use shown in eFigs 7 and 8.
Figure 2:
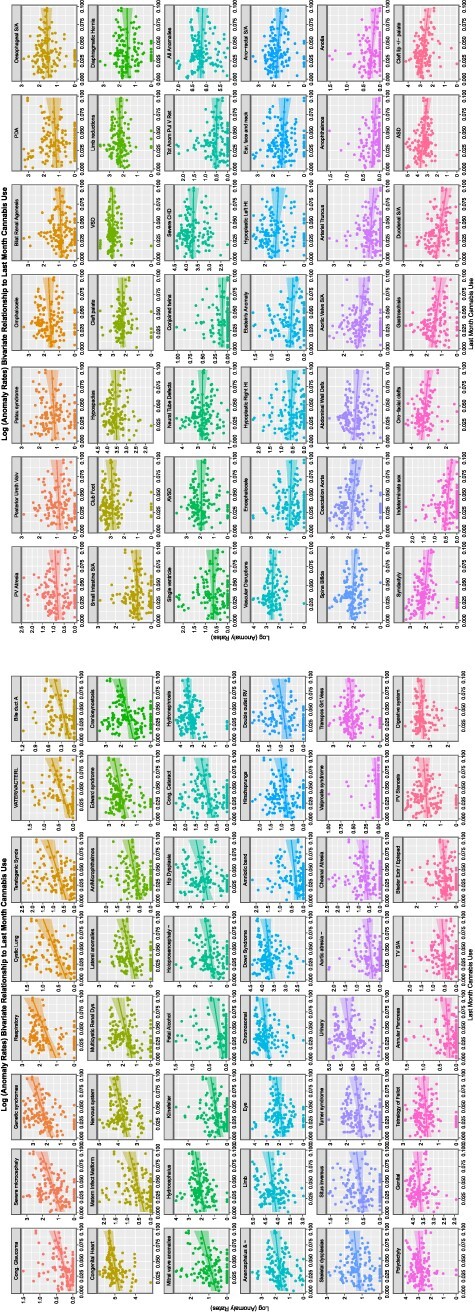
Time trends of CAs
Figure 3:
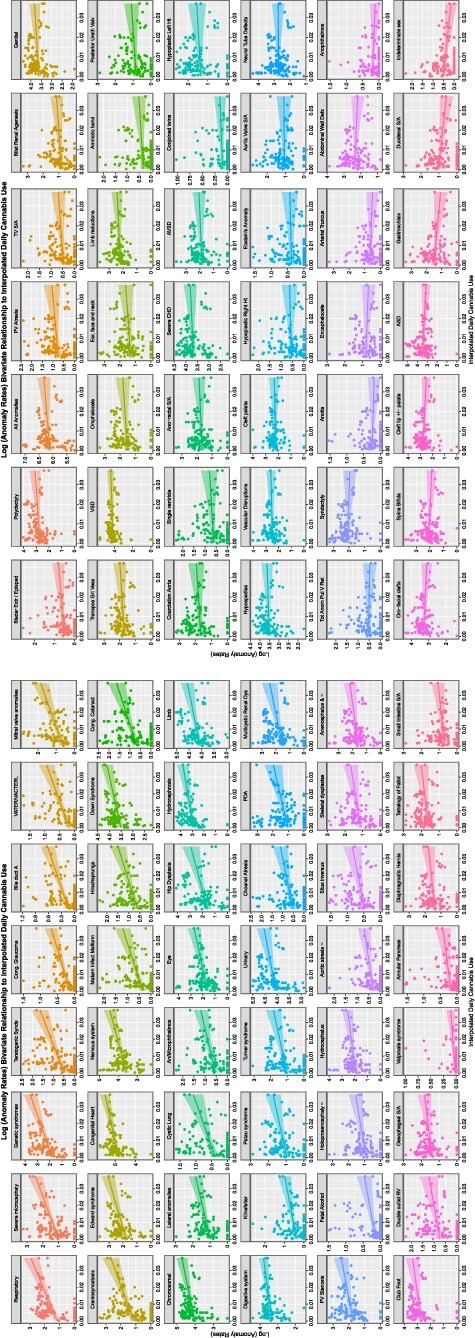
Trends of CAs with daily cannabis use (interpolated)
These relationships are formally analysed by linear regression in eTables 6–15 for each substance, respectively. These eTables list the usual metrics for linear models along with the applicable E-value point estimates and lower bounds. For the series of substances tobacco, alcohol, amphetamines and cocaine, 3, 12, 23 and 68 anomalies had elevated mEV, respectively. For the series of substances last month cannabis use, herbal THC concentration, resin THC concentration, daily use interpolated, last month cannabis use × herbal THC content × daily use interpolated (LMC_Herb_Daily) and last month cannabis use × resin THC content × daily use interpolated (LMC_Resin_Daily), the applicable numbers were 23, 45, 34, 41 and 42, respectively.
These data are summarized in Table 1. As the table is rather dense with information, these results are illustrated graphically for comparison in eFig. 9. Here, the number of anomalies for each substance is shown in panel A, and the cumulative exponents of the mEV in Panel B and the cumulative negative exponents of the P-values in Panel C. In each case, indices for cannabis exposure outperform teratogenic indices for tobacco, alcohol and amphetamines. eFigure 10 presents a study of the marginal or overall effects. Panel A shows the cumulative percentage average marginal effect, panel B the log of the mean percentage change, panel C the log of the standard error of the percentage change and panel D presents the SEM/average marginal effect ratio as a measure of the variability of the indices. In the first three cases, cannabis indices are noted to be higher than those of the other substances. The variability of the cannabinoid indices is lower than that of tobacco, alcohol and amphetamines (Panel D).
Table 1:
Summary table of bivariate continuous relationships by substance
| Substance | Number of terms with elevated E-values | Sum of P-value exponents | Sum of mEV exponents | Total % change | Mean % change | S.E. % change | Median % change | First quartile % change | Third quartile % change |
|---|---|---|---|---|---|---|---|---|---|
| Tobacco | 3 | 3 | 0 | 6.45 | 2.15 | 1.56 | 1.18 | 0.63 | 3.19 |
| Alcohol | 12 | 24 | 0 | 99.40 | 8.28 | 2.79 | 6.88 | 2.21 | 8.43 |
| Amphetamine | 23 | 45 | 0 | 185.25 | 8.05 | 2.51 | 3.21 | 0.72 | 10.20 |
| Cocaine | 68 | 268 | 0 | 991.55 | 14.58 | 1.65 | 10.71 | 3.19 | 20.85 |
| LM_Cannabis | 23 | 48 | 35 | 15 019.46 | 653.02 | 111.64 | 468.06 | 250.90 | 959.87 |
| Herb_THC | 45 | 107 | 55 | 9780.06 | 217.33 | 40.28 | 112.64 | 26.28 | 295.60 |
| Resin_THC | 34 | 75 | 8 | 2261.72 | 66.52 | 12.01 | 47.18 | 12.18 | 86.21 |
| LM_Cann_×_Herb | 42 | 107 | 68 | 20 370.06 | 485.00 | 73.45 | 325.67 | 146.58 | 653.78 |
| LM_Cann_×_Resin | 38 | 72 | 3 | 3500.09 | 92.11 | 15.32 | 61.13 | 15.19 | 132.36 |
| Daily_Interpol | 41 | 116 | 241 | 61 794.77 | 1507.19 | 226.63 | 1145.78 | 467.88 | 2180.68 |
| Herb_×_Day_Int | 41 | 111 | 18 | 6736.93 | 164.32 | 24.53 | 131.28 | 59.70 | 230.31 |
| Resin_×_Day_Int | 42 | 90 | NA | 2860.11 | 68.10 | 11.06 | 50.12 | 11.35 | 92.23 |
Since some marginal effect data are not distributed normally, the median and first and third quartile data are shown in eFig. 11. In all cases, the cannabis indices are substantially higher than those of the other substances. eTable 16 presents a categorization of the organ systems affected by their substance exposures by numbers of anomalies. These data are also presented as a heatmap in Fig. 4. Cardiovascular, chromosomal, gastrointestinal and uronephrological anomalies are noted to be prominent. eFigure 12 presents the number of systems impacted by each substance.
Figure 4:
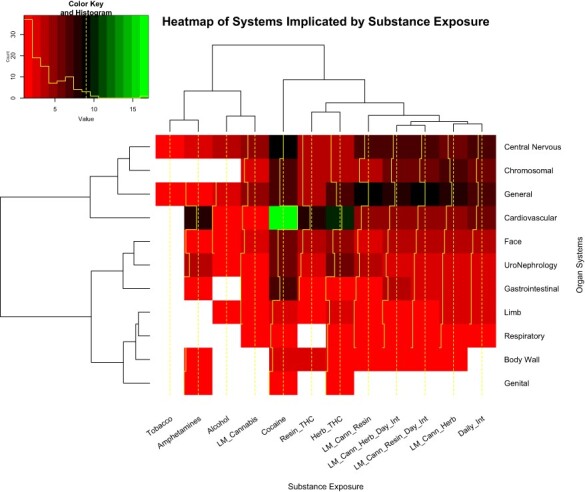
Heatmap of systems by substance exposure
In the genomic and epigenomic literature, volcano plots are commonly used to represent the significance levels against the fold change in gene expression. The equivalent in this work might be to chart the significance level against the mEV as a measure of fold change implied in the data. eFigures 13, 14 and 15 do this for tobacco, alcohol and amphetamines. The eFigures are noted to be rather lean and to have relatively low levels of significance and mEV. eFigure 16 performs a similar role for cocaine, and this eFigure is noted to be heavily populated and quantitatively much greater. eFigure 17, Figs 5 and 6 and eFigs 18 and 19 perform this role for herbal THC content, last month cannabis use, daily cannabis use interpolated, LMC_Herb_Daily and LMC_Resin_Daily, respectively. These figures are noted to be more densely populated and quantitatively much higher than those for the other substances.
Figure 5:
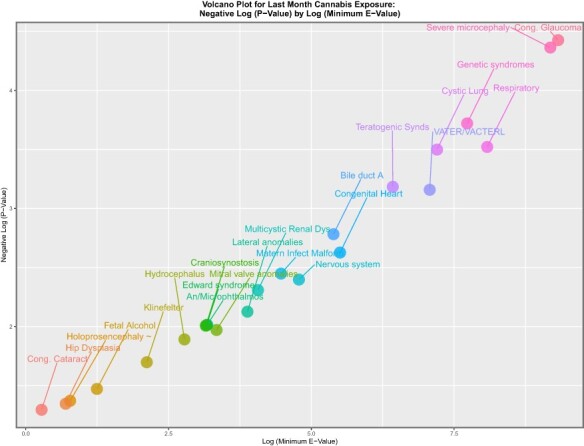
Volcano plot of significance (negative log (P-value)) against log (mEV) for past month cannabis use
Figure 6:
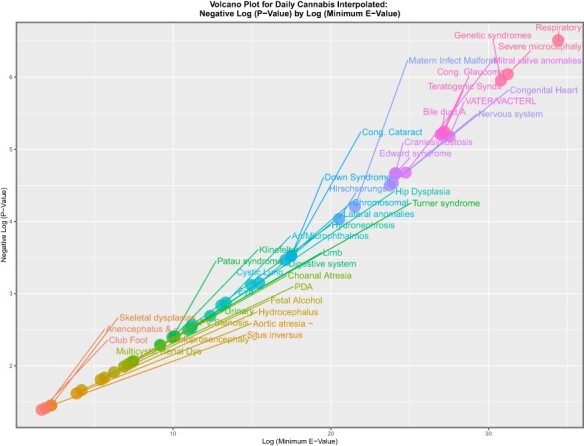
Volcano plot of significance (negative log (P-value)) against log (mEV) for daily cannabis use interpolated
Categorical Analysis
The data also lend itself to categorization for the purposes of calculating key epidemiological indices, including RR, AFE and PAR. For this purpose, substance exposure was divided into quintiles and boxplots as shown in eFigs 20–24 for tobacco, alcohol, amphetamines, cocaine and LMC_Resin_Daily, which contrast the highest and lowest quintiles of substance exposures. Where notches do not overlap, this indicates a statistically significant difference. This method also allows the calculation of highest:lowest quintile ratios for each anomaly by substance.
As shown in eTable 17, many substances demonstrate higher anomaly rates in the highest quintiles. However, the indices of cannabis exposure, which include daily exposure, have the greatest number of elevated ratios. These data are illustrated graphically in eFig. 25. eTable 18 shows the number of organ systems affected for LMC_Resin_Daily as a function of the total number of anomalies in each organ system. The table is ordered from those with the greatest percentage of anomalies per system. In fact, all 11 measured body systems are represented at levels above 50% in these data with general, chromosomal, uronephrolgical, limb, body wall, respiratory, cardiovascular, face, gastrointestinal and CNS anomalies more than 80% represented. These data are presented graphically in eFig. 26, which highlights these findings. eTables 19–29 and Tables 2 and 3 show the formal quantitative analysis of this data concatenated as a series of two-by-two epidemiological tables in long format. Many interesting features emerge from these tables, including that foetal alcohol syndrome is at the top of the tobacco, alcohol and herb THC, last month herb THC, last month Resin THC and LMC_Resin_Daily tables with mEVs of 8.67, 34.24, 13.44, 9.69, 7.37 and 43.19, respectively. Interestingly, VACTERL syndrome is near the top of the alcohol, amphetamine, cocaine, last month cannabis, last month cannabis × daily cannabis interpolated, last month cannabis herb, LMC_Herb_Daily and LMC_Resin_Daily lists, with mEVs of 5.93, 4.64, 24.90, 11.35, 26.43, 4.92, 34.61 and 43.19, respectively. t-test results are listed along with their accompanying P-values in eTable 29.
Table 2:
Summary table of bivariate categorical relationships comparing the highest and lowest quintiles of last month cannabis × herb THC concentration × daily cannabis use interpolated
| Anomaly | Total numbers of anomalies in quintile 5 | Total numbers of normals in quintile 5 | Total numbers of anomalies in quintile 1 | Total numbers of normals in quintile 1 | RR | RR (lower bound) | RR (upper bound) | AFE | AFE (lower bound) | AFE (upper bound) | PAR | PAR (lower bound) | PAR (upper bound) | Chi-square | P-value | E-value esimate | E-value (95% lower bound) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VATER/VACTERL | 726 | 12 566 202 | 3 | 2 833 401 | 54.5626 | 17.5566 | 169.5705 | 0.9817 | 0.9430 | 0.9941 | 0.9776 | 0.9308 | 0.9928 | 157.1010 | 2.43E-36 | 108.62 | 34.61 |
| Hip dysplasia | 11 592 | 12 555 336 | 359 | 2 833 045 | 7.2802 | 6.5544 | 8.0864 | 0.8626 | 0.8474 | 0.8763 | 0.8367 | 0.8192 | 0.8525 | 1887.9451 | 0.00E + 00 | 14.04 | 12.59 |
| Respiratory | 6122 | 12 560 806 | 230 | 2 833 174 | 6.0013 | 5.2611 | 6.8456 | 0.8334 | 0.8099 | 0.8539 | 0.8032 | 0.7766 | 0.8266 | 924.2930 | 2.57E-203 | 11.48 | 10.00 |
| Matern infect malform | 1279 | 12 565 649 | 54 | 3 405 791 | 6.4191 | 4.8890 | 8.4280 | 0.8442 | 0.7955 | 0.8813 | 0.8100 | 0.7533 | 0.8537 | 237.0546 | 8.63E-54 | 12.32 | 9.25 |
| Teratogenic synds | 2284 | 12 564 644 | 111 | 3 405 734 | 5.5766 | 4.6093 | 6.7468 | 0.8207 | 0.7830 | 0.8518 | 0.7826 | 0.7393 | 0.8188 | 397.6438 | 8.97E-89 | 10.63 | 8.69 |
| Edward syndrome | 9689 | 12 557 239 | 676 | 3 405 169 | 3.8844 | 3.5931 | 4.1994 | 0.7426 | 0.7217 | 0.7619 | 0.6941 | 0.6710 | 0.7156 | 1354.3539 | 8.72E-297 | 7.23 | 6.65 |
| Cystic lung | 1413 | 12 565 516 | 86 | 3 405 759 | 4.4529 | 3.5818 | 5.5358 | 0.7754 | 0.7208 | 0.8194 | 0.7309 | 0.6697 | 0.7809 | 217.0697 | 1.97E-49 | 8.37 | 6.62 |
| Foetal alcohol | 784 | 12 566 144 | 45 | 3 405 800 | 4.7217 | 3.4964 | 6.3764 | 0.7882 | 0.7140 | 0.8432 | 0.7454 | 0.6618 | 0.8084 | 124.8482 | 2.75E-29 | 8.91 | 6.45 |
| Bile duct A | 600 | 12 566 328 | 34 | 3 405 811 | 4.7826 | 3.3854 | 6.7566 | 0.7909 | 0.7046 | 0.8520 | 0.7485 | 0.6512 | 0.8186 | 96.2678 | 5.02E-23 | 9.04 | 6.23 |
| Mitral valve anomalies | 2120 | 12 564 808 | 119 | 2 833 285 | 4.0167 | 3.3395 | 4.8312 | 0.7510 | 0.7006 | 0.7930 | 0.7111 | 0.6559 | 0.7575 | 255.3200 | 8.99E-58 | 7.50 | 6.13 |
| Severe microcephaly | 5083 | 12 561 845 | 381 | 3 405 464 | 3.6157 | 3.2582 | 4.0124 | 0.7234 | 0.6931 | 0.7508 | 0.6730 | 0.6397 | 0.7032 | 670.9093 | 3.17E-148 | 6.69 | 5.97 |
| Double outlet RV | 1768 | 12 565 160 | 105 | 2 833 298 | 3.7964 | 3.1180 | 4.6224 | 0.7366 | 0.6793 | 0.7837 | 0.6953 | 0.6331 | 0.7470 | 204.1802 | 1.28E-46 | 7.05 | 5.69 |
| Turner syndrome | 3364 | 12 563 564 | 260 | 3 405 585 | 3.5065 | 3.0909 | 3.9780 | 0.7148 | 0.6765 | 0.7486 | 0.6635 | 0.6217 | 0.7007 | 432.5232 | 2.29E-96 | 6.47 | 5.63 |
| Cong. glaucoma | 773 | 12 566 155 | 53 | 3 405 792 | 3.9528 | 2.9925 | 5.2211 | 0.7470 | 0.6658 | 0.8085 | 0.6991 | 0.6096 | 0.7681 | 109.4087 | 6.60E-26 | 7.37 | 5.43 |
| Lateral anomalies | 3182 | 12 563 747 | 213 | 2 833 191 | 3.3682 | 2.9320 | 3.8694 | 0.7031 | 0.6589 | 0.7416 | 0.6590 | 0.6117 | 0.7006 | 332.4899 | 1.38E-74 | 6.19 | 5.31 |
| Genetic syndromes | 9209 | 12 557 719 | 815 | 3 405 030 | 3.0623 | 2.8507 | 3.2897 | 0.6735 | 0.6492 | 0.6960 | 0.6187 | 0.5928 | 0.6430 | 1040.5508 | 1.38E-228 | 5.58 | 5.15 |
| Hirschsprungs | 1877 | 12 565 051 | 152 | 3 405 693 | 3.3467 | 2.8368 | 3.9482 | 0.7012 | 0.6475 | 0.7467 | 0.6487 | 0.5906 | 0.6985 | 231.4082 | 1.47E-52 | 6.15 | 5.12 |
| Nervous system | 43 028 | 12 523 900 | 3327 | 2 830 077 | 2.9159 | 2.8150 | 3.0205 | 0.6571 | 0.6448 | 0.6689 | 0.6099 | 0.5969 | 0.6225 | 3899.4204 | 0.00E + 00 | 5.28 | 5.08 |
| Patau syndrome | 3497 | 12 563 431 | 303 | 3 405 542 | 3.1279 | 2.7815 | 3.5174 | 0.6803 | 0.6405 | 0.7157 | 0.6260 | 0.5834 | 0.6643 | 403.7380 | 4.23E-90 | 5.71 | 5.01 |
| Klinefelter | 989 | 12 565 939 | 81 | 3 405 764 | 3.3091 | 2.6384 | 4.1503 | 0.6978 | 0.6210 | 0.7591 | 0.6450 | 0.5623 | 0.7120 | 120.6415 | 2.29E-28 | 6.07 | 4.72 |
| Chromosomal | 61 670 | 12 505 258 | 6213 | 3 399 632 | 2.6901 | 2.6209 | 2.7611 | 0.6283 | 0.6185 | 0.6378 | 0.5708 | 0.5605 | 0.5808 | 6018.9417 | 0.00E + 00 | 4.82 | 4.68 |
| Anencephalus and ∼ | 5369 | 12 561 559 | 517 | 3 405 328 | 2.8145 | 2.5716 | 3.0803 | 0.6447 | 0.6111 | 0.6754 | 0.5881 | 0.5527 | 0.6206 | 551.8625 | 2.48E-122 | 5.07 | 4.58 |
| Situs inversus | 1252 | 12 565 676 | 109 | 3 405 736 | 3.1130 | 2.5596 | 3.7860 | 0.6788 | 0.6093 | 0.7359 | 0.6244 | 0.5503 | 0.6863 | 143.8198 | 1.95E-33 | 5.68 | 4.56 |
| Craniosynostosis | 5180 | 12 561 748 | 503 | 3 405 342 | 2.7910 | 2.5469 | 3.0585 | 0.6417 | 0.6074 | 0.6730 | 0.5849 | 0.5488 | 0.6181 | 527.1098 | 6.01E-117 | 5.03 | 4.53 |
| Holoprosencephaly ∼ | 2849 | 12 564 079 | 274 | 3 405 571 | 2.8180 | 2.4894 | 3.1899 | 0.6451 | 0.5983 | 0.6865 | 0.5885 | 0.5393 | 0.6325 | 293.2214 | 4.94E-66 | 5.08 | 4.42 |
| Small intestine S/A | 1223 | 12 565 705 | 113 | 3 405 732 | 2.9332 | 2.4191 | 3.5566 | 0.6591 | 0.5866 | 0.7188 | 0.6033 | 0.5268 | 0.6675 | 131.8108 | 8.23E-31 | 5.31 | 4.27 |
| An/microphthalmos | 1558 | 12 565 370 | 159 | 3 405 686 | 2.6556 | 2.2558 | 3.1263 | 0.6234 | 0.5567 | 0.6801 | 0.5657 | 0.4964 | 0.6255 | 148.9350 | 1.48E-34 | 4.75 | 3.94 |
| Down syndrome | 34 645 | 12 532 283 | 4052 | 3 401 793 | 2.3172 | 2.2431 | 2.3938 | 0.5684 | 0.5542 | 0.5823 | 0.5089 | 0.4944 | 0.5230 | 2722.9178 | 0.00E + 00 | 4.06 | 3.91 |
| Limb | 61 519 | 12 505 409 | 6073 | 2 827 331 | 2.2839 | 2.2246 | 2.3449 | 0.5622 | 0.5505 | 0.5735 | 0.5117 | 0.4998 | 0.5232 | 4007.1357 | 0.00E + 00 | 4.00 | 3.88 |
| Choanal atresia | 1427 | 12 565 502 | 150 | 3 405 695 | 2.5783 | 2.1791 | 3.0506 | 0.6121 | 0.5411 | 0.6722 | 0.5539 | 0.4806 | 0.6169 | 131.1477 | 1.15E-30 | 4.60 | 3.78 |
| Multicystic renal dys | 6976 | 12 559 952 | 814 | 3 405 031 | 2.3226 | 2.1600 | 2.4975 | 0.5695 | 0.5370 | 0.5996 | 0.5099 | 0.4770 | 0.5408 | 549.2833 | 9.01E-122 | 4.08 | 3.74 |
| Omphalocele | 4382 | 12 562 547 | 503 | 3 405 342 | 2.3610 | 2.1529 | 2.5892 | 0.5765 | 0.5355 | 0.6138 | 0.5171 | 0.4754 | 0.5555 | 354.1107 | 2.70E-79 | 4.15 | 3.73 |
| Skeletal dysplasias | 2792 | 12 564 137 | 323 | 3 405 522 | 2.3427 | 2.0878 | 2.6286 | 0.5731 | 0.5210 | 0.6196 | 0.5137 | 0.4608 | 0.5614 | 222.8260 | 1.09E-50 | 4.12 | 3.59 |
| Congenital heart | 118 263 | 12 448 665 | 12 766 | 2 820 638 | 2.0887 | 2.0510 | 2.1271 | 0.5212 | 0.5124 | 0.5299 | 0.4704 | 0.4617 | 0.4791 | 6594.4726 | 0.00E + 00 | 3.60 | 3.52 |
| Annular pancreas | 274 | 12 566 654 | 24 | 3 405 821 | 3.0941 | 2.0386 | 4.6961 | 0.6768 | 0.5095 | 0.7871 | 0.6223 | 0.4457 | 0.7426 | 31.2763 | 1.12E-08 | 5.64 | 3.49 |
| Cong. cataract | 2204 | 12 564 724 | 259 | 3 405 586 | 2.3063 | 2.0277 | 2.6231 | 0.5664 | 0.5068 | 0.6188 | 0.5068 | 0.4466 | 0.5605 | 171.4996 | 1.74E-39 | 4.04 | 3.47 |
| Hydrocephalus | 7481 | 12 559 447 | 961 | 3 404 884 | 2.1098 | 1.9727 | 2.2563 | 0.5260 | 0.4931 | 0.5568 | 0.4661 | 0.4334 | 0.4970 | 497.3813 | 1.76E-110 | 3.64 | 3.36 |
| Conjoined twins | 202 | 12 566 726 | 17 | 3 405 828 | 3.2203 | 1.9631 | 5.2827 | 0.6895 | 0.4906 | 0.8107 | 0.6360 | 0.4253 | 0.7694 | 24.0045 | 4.81E-07 | 5.89 | 3.34 |
| Amniotic band | 960 | 12 565 968 | 112 | 3 405 733 | 2.3230 | 1.9101 | 2.8251 | 0.5695 | 0.4765 | 0.6460 | 0.5100 | 0.4162 | 0.5888 | 75.5777 | 1.76E-18 | 4.08 | 3.23 |
| Digestive system | 24 256 | 12 542 673 | 2761 | 2 830 643 | 1.9808 | 1.9043 | 2.0602 | 0.4951 | 0.4749 | 0.5146 | 0.4445 | 0.4246 | 0.4638 | 1205.8838 | 1.61E-264 | 3.37 | 3.22 |
| Vascular disruptions | 8534 | 12 558 394 | 1018 | 2 832 385 | 1.8901 | 1.7712 | 2.0170 | 0.4709 | 0.4354 | 0.5042 | 0.4207 | 0.3861 | 0.4534 | 381.4750 | 2.97E-85 | 3.19 | 2.94 |
| Club foot | 16 165 | 12 550 763 | 2412 | 3 403 433 | 1.8163 | 1.7403 | 1.8957 | 0.4494 | 0.4254 | 0.4725 | 0.3911 | 0.3680 | 0.4133 | 770.9346 | 5.63E-170 | 3.03 | 2.88 |
| Neural tube defects | 12 692 | 12 554 236 | 1891 | 3 403 954 | 1.8190 | 1.7332 | 1.9090 | 0.4502 | 0.4230 | 0.4762 | 0.3919 | 0.3658 | 0.4169 | 607.4520 | 2.00E-134 | 3.04 | 2.86 |
| AVSD | 5451 | 12 561 478 | 796 | 3 405 049 | 1.8559 | 1.7229 | 1.9992 | 0.4612 | 0.4196 | 0.4998 | 0.4024 | 0.3624 | 0.4400 | 274.2795 | 6.63E-62 | 3.12 | 2.84 |
| Genital | 29 065 | 12 537 863 | 3695 | 2 829 709 | 1.7735 | 1.7139 | 1.8352 | 0.4361 | 0.4165 | 0.4551 | 0.3870 | 0.3681 | 0.4053 | 1108.3316 | 2.55E-243 | 2.94 | 2.82 |
| Hypoplastic left ht | 3339 | 12 563 589 | 486 | 3 405 359 | 1.8620 | 1.6930 | 2.0479 | 0.4629 | 0.4093 | 0.5117 | 0.4041 | 0.3525 | 0.4516 | 169.3353 | 5.17E-39 | 3.13 | 2.78 |
| Urinary | 54 851 | 12 512 077 | 8623 | 3 397 222 | 1.7239 | 1.6853 | 1.7635 | 0.4199 | 0.4066 | 0.4329 | 0.3629 | 0.3503 | 0.3752 | 2274.3562 | 0.00E + 00 | 2.84 | 2.76 |
| PV atresia | 1449 | 12 565 479 | 203 | 3 405 642 | 1.9345 | 1.6702 | 2.2406 | 0.4831 | 0.4013 | 0.5537 | 0.4237 | 0.3445 | 0.4934 | 80.3873 | 1.54E-19 | 3.28 | 2.73 |
| Abdominal wall defx | 6953 | 12 559 976 | 1072 | 3 404 773 | 1.7578 | 1.6483 | 1.8746 | 0.4311 | 0.3933 | 0.4665 | 0.3735 | 0.3376 | 0.4075 | 303.5942 | 2.71E-68 | 2.91 | 2.68 |
| Hydronephrosis | 17 099 | 12 549 829 | 2725 | 3 403 120 | 1.7006 | 1.6332 | 1.7707 | 0.4120 | 0.3877 | 0.4353 | 0.3553 | 0.3325 | 0.3774 | 679.2268 | 4.92E-150 | 2.79 | 2.65 |
| Hypospadias | 24 229 | 12 542 699 | 3233 | 2 830 171 | 1.6897 | 1.6289 | 1.7528 | 0.4082 | 0.3861 | 0.4295 | 0.3601 | 0.3391 | 0.3805 | 804.4393 | 2.92E-177 | 2.77 | 2.64 |
| Polydactyly | 17 729 | 12 549 199 | 2904 | 3 402 941 | 1.6546 | 1.5909 | 1.7207 | 0.3956 | 0.3714 | 0.4189 | 0.3399 | 0.3173 | 0.3618 | 646.9932 | 5.03E-143 | 2.70 | 2.56 |
| Diaphragmatic hernia | 3749 | 12 563 179 | 590 | 3 405 255 | 1.7221 | 1.5789 | 1.8783 | 0.4193 | 0.3667 | 0.4676 | 0.3623 | 0.3126 | 0.4084 | 154.3957 | 9.49E-36 | 2.84 | 2.54 |
| Severe CHD | 28 086 | 12 538 842 | 4742 | 3 401 103 | 1.6052 | 1.5566 | 1.6553 | 0.3770 | 0.3576 | 0.3959 | 0.3226 | 0.3045 | 0.3401 | 927.5716 | 4.98E-204 | 2.59 | 2.49 |
| TV S/A | 851 | 12 566 077 | 123 | 3 405 722 | 1.8751 | 1.5521 | 2.2653 | 0.4667 | 0.3557 | 0.5586 | 0.4078 | 0.3014 | 0.4979 | 43.8914 | 1.74E-11 | 3.16 | 2.48 |
| Bilat renal agenesis | 1477 | 12 565 451 | 226 | 3 405 619 | 1.7712 | 1.5398 | 2.0373 | 0.4354 | 0.3506 | 0.5092 | 0.3776 | 0.2973 | 0.4488 | 65.8245 | 2.46E-16 | 2.94 | 2.45 |
| Valproate syndrome | 78 | 12 566 850 | 6 | 3 405 839 | 3.5232 | 1.5357 | 8.0827 | 0.7162 | 0.3489 | 0.8763 | 0.6650 | 0.2758 | 0.8451 | 10.0679 | 0.0008 | 6.50 | 2.44 |
| Aortic atresia ∼ | 775 | 12 566 153 | 92 | 2 833 312 | 1.8993 | 1.5301 | 2.3575 | 0.4735 | 0.3465 | 0.5758 | 0.4232 | 0.3003 | 0.5246 | 35.0195 | 1.63E-09 | 3.21 | 2.43 |
| Oesophageal S/A | 3351 | 12 563 577 | 553 | 3 405 292 | 1.6423 | 1.5010 | 1.7968 | 0.3911 | 0.3338 | 0.4435 | 0.3357 | 0.2824 | 0.3851 | 119.2583 | 4.60E-28 | 2.67 | 2.37 |
| Tetralogy of Fallot | 4324 | 12 562 604 | 723 | 3 405 122 | 1.6209 | 1.4981 | 1.7536 | 0.3830 | 0.3325 | 0.4298 | 0.3282 | 0.2813 | 0.3720 | 147.3533 | 3.28E-34 | 2.62 | 2.36 |
| All anomalies | 358 462 | 12 208 466 | 64 516 | 3 341 329 | 1.5058 | 1.4934 | 1.5184 | 0.3359 | 0.3304 | 0.3414 | 0.2847 | 0.2796 | 0.2897 | 9542.4380 | 0.00E + 00 | 2.38 | 2.35 |
| Blader extr/epispad | 812 | 12 566 116 | 125 | 3 405 720 | 1.7605 | 1.4583 | 2.1253 | 0.4320 | 0.3143 | 0.5295 | 0.3744 | 0.2635 | 0.4686 | 35.5906 | 1.22E-09 | 2.92 | 2.28 |
| Transpos grt vess | 4214 | 12 562 714 | 733 | 3 405 112 | 1.5581 | 1.4405 | 1.6852 | 0.3582 | 0.3058 | 0.4066 | 0.3051 | 0.2571 | 0.3500 | 124.8471 | 2.75E-29 | 2.49 | 2.24 |
| Hypoplastic right ht | 891 | 12 566 037 | 142 | 3 405 703 | 1.7005 | 1.4245 | 2.0300 | 0.4119 | 0.2980 | 0.5074 | 0.3553 | 0.2489 | 0.4466 | 35.3481 | 1.38E-09 | 2.79 | 2.20 |
| Limb reductions | 7801 | 12 559 127 | 1431 | 3 404 414 | 1.4774 | 1.3965 | 1.5631 | 0.3231 | 0.2839 | 0.3602 | 0.2731 | 0.2376 | 0.3069 | 186.6616 | 8.51E-43 | 2.32 | 2.14 |
| Spina bifida | 5766 | 12 561 163 | 1075 | 3 404 770 | 1.4537 | 1.3620 | 1.5514 | 0.3121 | 0.2658 | 0.3554 | 0.2630 | 0.2215 | 0.3024 | 128.3342 | 4.74E-30 | 2.27 | 2.06 |
| PDA | 3491 | 12 563 437 | 639 | 3 405 206 | 1.4806 | 1.3609 | 1.6109 | 0.3246 | 0.2652 | 0.3792 | 0.2744 | 0.2208 | 0.3243 | 84.2905 | 2.14E-20 | 2.32 | 2.06 |
| Anorectal S/A | 3764 | 12 563 164 | 698 | 3 405 147 | 1.4615 | 1.3481 | 1.5844 | 0.3158 | 0.2582 | 0.3688 | 0.2664 | 0.2146 | 0.3147 | 85.8211 | 9.85E-21 | 2.28 | 2.03 |
| PV stenosis | 5594 | 12 561 334 | 1067 | 3 404 778 | 1.4209 | 1.3308 | 1.5170 | 0.2962 | 0.2486 | 0.3408 | 0.2488 | 0.2063 | 0.2889 | 111.7550 | 2.02E-26 | 2.19 | 1.99 |
| Tot anom pul V ret | 662 | 12 566 266 | 114 | 3 405 731 | 1.5738 | 1.2901 | 1.9198 | 0.3646 | 0.2249 | 0.4791 | 0.3110 | 0.1837 | 0.4185 | 20.3466 | 3.23E-06 | 2.52 | 1.90 |
| Eye | 6732 | 12 560 196 | 1334 | 3 404 511 | 1.3677 | 1.2897 | 1.4504 | 0.2688 | 0.2246 | 0.3105 | 0.2244 | 0.1854 | 0.2615 | 110.1067 | 4.64E-26 | 2.08 | 1.90 |
| Single ventricle | 1051 | 12 565 877 | 191 | 3 405 654 | 1.4913 | 1.2782 | 1.7399 | 0.3294 | 0.2177 | 0.4252 | 0.2788 | 0.1783 | 0.3670 | 26.1624 | 1.57E-07 | 2.35 | 1.87 |
| Arterial truncus | 841 | 12 566 088 | 150 | 3 405 695 | 1.5195 | 1.2772 | 1.8078 | 0.3419 | 0.2170 | 0.4468 | 0.2901 | 0.1774 | 0.3874 | 22.6105 | 9.92E-07 | 2.41 | 1.87 |
| Encephalocele | 1558 | 12 565 370 | 297 | 3 405 548 | 1.4217 | 1.2558 | 1.6095 | 0.2966 | 0.2037 | 0.3787 | 0.2491 | 0.1666 | 0.3234 | 31.2049 | 1.16E-08 | 2.20 | 1.82 |
| VSD | 55 036 | 12 511 892 | 11 869 | 3 393 976 | 1.2567 | 1.2321 | 1.2818 | 0.2043 | 0.1883 | 0.2199 | 0.1680 | 0.1544 | 0.1815 | 514.0649 | 4.14E-114 | 1.82 | 1.77 |
| Ebstein’s anomaly | 514 | 12 566 414 | 95 | 3 405 750 | 1.4663 | 1.1781 | 1.8251 | 0.3180 | 0.1512 | 0.4521 | 0.2684 | 0.1200 | 0.3918 | 11.8921 | 0.0003 | 2.29 | 1.64 |
| Orofacial clefts | 15 026 | 12 551 902 | 2772 | 2 830 632 | 1.2222 | 1.1737 | 1.2727 | 0.1818 | 0.1480 | 0.2143 | 0.1535 | 0.1240 | 0.1819 | 94.6207 | 1.15E-22 | 1.74 | 1.63 |
| Posterior ureth valv | 1806 | 12 565 122 | 389 | 3 405 456 | 1.2582 | 1.1277 | 1.4039 | 0.2052 | 0.1132 | 0.2877 | 0.1689 | 0.0905 | 0.2405 | 16.9660 | 1.90E-05 | 1.83 | 1.51 |
| Ear, face and neck | 3203 | 12 563 725 | 711 | 3 405 134 | 1.2209 | 1.1256 | 1.3242 | 0.1809 | 0.1116 | 0.2449 | 0.1481 | 0.0895 | 0.2029 | 23.2624 | 7.07E-07 | 1.74 | 1.50 |
| Coarctation aorta | 3788 | 12 563 141 | 874 | 3 404 971 | 1.1746 | 1.0913 | 1.2642 | 0.1487 | 0.0837 | 0.2090 | 0.1208 | 0.0666 | 0.1718 | 18.4386 | 8.77E-06 | 1.63 | 1.41 |
| Cleft lip ± palate | 8444 | 12 558 484 | 2027 | 3 403 818 | 1.1290 | 1.0756 | 1.1851 | 0.1143 | 0.0703 | 0.1562 | 0.0921 | 0.0560 | 0.1269 | 24.1059 | 4.56E-07 | 1.51 | 1.36 |
| Cleft palate | 6486 | 12 560 443 | 1286 | 2 832 118 | 1.1371 | 1.0711 | 1.2072 | 0.1206 | 0.0664 | 0.1717 | 0.1006 | 0.0546 | 0.1444 | 17.7599 | 1.25E-05 | 1.53 | 1.35 |
| Gastroschisis | 1953 | 12 564 975 | 453 | 3 405 392 | 1.1684 | 1.0549 | 1.2942 | 0.1441 | 0.0520 | 0.2273 | 0.1170 | 0.0406 | 0.1873 | 8.9283 | 0.0014 | 1.61 | 1.30 |
| Anophthalmos | 211 | 12 566 717 | 51 | 3 405 794 | 1.1213 | 0.8258 | 1.5224 | 0.1081 | −0.2109 | 0.3431 | 0.0871 | −0.1679 | 0.2864 | 0.5387 | 0.2315 | 1.49 | 1.00 |
| Anotia | 368 | 12 566 560 | 87 | 3 405 758 | 1.1464 | 0.9075 | 1.4481 | 0.1277 | −0.1019 | 0.3094 | 0.1033 | −0.0833 | 0.2577 | 1.3150 | 0.1257 | 1.56 | 1.00 |
| Aortic valve S/A | 1432 | 12 565 496 | 452 | 3 405 393 | 0.8586 | 0.7725 | 0.9544 | −0.1647 | −0.2946 | −0.0478 | −0.1252 | −0.2193 | −0.0383 | 7.9990 | 0.0023 | 1.60 | — |
| ASD | 17 175 | 12 549 753 | 5504 | 3 400 341 | 0.8457 | 0.8204 | 0.8717 | −0.1825 | −0.2189 | −0.1471 | −0.1382 | −0.1646 | −0.1123 | 117.5198 | 1.10E-27 | 1.65 | — |
| Duodenal S/A | 1390 | 12 565 539 | 451 | 3 405 394 | 0.8353 | 0.7511 | 0.9289 | −0.1972 | −0.3313 | −0.0766 | −0.1489 | −0.2448 | −0.0604 | 11.0618 | 0.0004 | 1.68 | — |
| Indeterminate sex | 421 | 12 566 507 | 161 | 3 405 684 | 0.7087 | 0.5910 | 0.8498 | −0.4111 | −0.6921 | −0.1767 | −0.2974 | −0.4795 | −0.1376 | 13.9470 | 9.40E-05 | 2.17 | — |
| Syndactyly | 5199 | 12 561 729 | 1713 | 3 404 132 | 0.8225 | 0.7788 | 0.8687 | −0.2157 | −0.2840 | −0.1512 | −0.1623 | −0.2110 | −0.1155 | 49.3507 | 1.07E-12 | 1.73 | — |
Table 3:
Summary table of bivariate categorical relationships comparing the highest and lowest quintiles oflast month cannabis × resin THC concentration × daily cannabis use interpolated
| Anomaly | Total numbers of anomalies in quintile 5 | Total numbers of normals in quintile 5 | Total numbers of anomalies in Quintile 1 | Total numbers of normals in quintile 1 | RR | RR (Lower bound) | RR (upper bound) | AFE | AFE (lower bound) | AFE (upper bound) | PAR | PAR (lower bound) | PAR (upper bound) | Chi-squared | P-value | E-value esimate | E-value (95% lower bound) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Foetal alcohol | 686 | 10 335 677 | 5 | 3 967 367 | 52.6610 | 21.8492 | 126.9237 | 0.9810 | 0.9542 | 0.9921 | 0.9739 | 0.9375 | 0.9891 | 251.5786 | 5.88E-57 | 104.82 | 43.19 |
| Amniotic band | 862 | 10 335 501 | 59 | 3 967 313 | 5.6078 | 4.3077 | 7.3002 | 0.8217 | 0.7679 | 0.8630 | 0.7690 | 0.7044 | 0.8196 | 209.0833 | 1.09E-47 | 10.69 | 8.08 |
| Multicystic renal dys | 6362 | 10 330 000 | 684 | 3 966 688 | 3.5700 | 3.2993 | 3.8630 | 0.7199 | 0.6969 | 0.7411 | 0.6500 | 0.6242 | 0.6741 | 1143.2133 | 6.70E-251 | 6.60 | 6.05 |
| VATER/VACTERL | 726 | 10 335 637 | 61 | 3 690 483 | 4.2494 | 3.2723 | 5.5182 | 0.7647 | 0.6944 | 0.8188 | 0.7054 | 0.6251 | 0.7685 | 139.8292 | 1.45E-32 | 7.97 | 6.00 |
| Hypoplastic right ht | 961 | 10 335 402 | 97 | 3 967 276 | 3.8027 | 3.0861 | 4.6856 | 0.7370 | 0.6760 | 0.7866 | 0.6695 | 0.6004 | 0.7266 | 182.0095 | 8.82E-42 | 7.07 | 5.62 |
| Teratogenic synds | 2000 | 10 334 363 | 219 | 3 967 154 | 3.5053 | 3.0489 | 4.0300 | 0.7147 | 0.6720 | 0.7519 | 0.6442 | 0.5965 | 0.6862 | 353.4852 | 3.69E-79 | 6.47 | 5.55 |
| Hip dysplasia | 11 179 | 10 325 184 | 1325 | 3 689 220 | 3.0124 | 2.8457 | 3.1889 | 0.6680 | 0.6486 | 0.6864 | 0.5972 | 0.5762 | 0.6172 | 1593.9238 | 0.00E + 00 | 5.47 | 5.14 |
| Omphalocele | 4163 | 10 332 199 | 562 | 3 966 810 | 2.8432 | 2.6035 | 3.1050 | 0.6483 | 0.6159 | 0.6779 | 0.5712 | 0.5366 | 0.6032 | 591.8580 | 4.94E-131 | 5.13 | 4.65 |
| Valproate syndrome | 72 | 10 336 290 | 4 | 3 967 368 | 6.9089 | 2.5243 | 18.9091 | 0.8553 | 0.6039 | 0.9471 | 0.8102 | 0.5075 | 0.9269 | 19.1507 | 6.04E-06 | 13.30 | 4.49 |
| Edward syndrome | 8773 | 10 327 590 | 1265 | 3 966 107 | 2.6619 | 2.5096 | 2.8235 | 0.6243 | 0.6015 | 0.6458 | 0.5457 | 0.5216 | 0.5685 | 1147.9334 | 6.31E-252 | 4.77 | 4.46 |
| Matern infect malform | 1134 | 10 335 229 | 147 | 3 967 225 | 2.9609 | 2.4935 | 3.5160 | 0.6623 | 0.5990 | 0.7156 | 0.5863 | 0.5183 | 0.6446 | 169.0136 | 6.08E-39 | 5.37 | 4.42 |
| Patau syndrome | 3186 | 10 333 177 | 457 | 3 966 915 | 2.6759 | 2.4260 | 2.9515 | 0.6263 | 0.5878 | 0.6612 | 0.5477 | 0.5072 | 0.5849 | 419.5932 | 1.50E-93 | 4.79 | 4.29 |
| Ebstein’s anomaly | 449 | 10 335 913 | 56 | 3 967 316 | 3.0775 | 2.3311 | 4.0628 | 0.6751 | 0.5710 | 0.7539 | 0.6002 | 0.4882 | 0.6877 | 69.8286 | 3.23E-17 | 5.61 | 4.09 |
| Lateral anomalies | 2900 | 10 333 462 | 405 | 3 690 140 | 2.5566 | 2.3042 | 2.8367 | 0.6089 | 0.5660 | 0.6475 | 0.5342 | 0.4898 | 0.5749 | 336.8852 | 1.52E-75 | 4.55 | 4.04 |
| Genetic syndromes | 9047 | 10 327 316 | 1435 | 3 965 937 | 2.4198 | 2.2888 | 2.5584 | 0.5868 | 0.5631 | 0.6091 | 0.5064 | 0.4821 | 0.5296 | 1032.5849 | 7.42E-227 | 4.27 | 4.01 |
| Cystic lung | 1210 | 10 335 153 | 176 | 3 967 196 | 2.6388 | 2.2529 | 3.0908 | 0.6210 | 0.5561 | 0.6765 | 0.5422 | 0.4744 | 0.6012 | 156.3959 | 3.47E-36 | 4.72 | 3.93 |
| Situs inversus | 1098 | 10 335 264 | 159 | 3 967 214 | 2.6506 | 2.2445 | 3.1302 | 0.6227 | 0.5545 | 0.6805 | 0.5440 | 0.4727 | 0.6056 | 142.7689 | 3.30E-33 | 4.74 | 3.92 |
| Hydronephrosis | 15 960 | 10 320 403 | 2758 | 3 964 614 | 2.2211 | 2.1332 | 2.3127 | 0.5498 | 0.5312 | 0.5676 | 0.4688 | 0.4502 | 0.4868 | 1580.8321 | 0.00E + 00 | 3.87 | 3.69 |
| Posterior urethral valve | 1700 | 10 334 662 | 277 | 3 967 096 | 2.3556 | 2.0747 | 2.6746 | 0.5755 | 0.5180 | 0.6261 | 0.4949 | 0.4366 | 0.5471 | 185.8450 | 1.28E-42 | 4.14 | 3.57 |
| Severe microcephaly | 4513 | 10 331 849 | 774 | 3 966 599 | 2.2380 | 2.0737 | 2.4153 | 0.5532 | 0.5178 | 0.5860 | 0.4722 | 0.4367 | 0.5054 | 452.6242 | 9.68E-101 | 3.90 | 3.57 |
| Limb | 55 575 | 10 280 787 | 9536 | 3 681 008 | 2.0808 | 2.0362 | 2.1265 | 0.5194 | 0.5089 | 0.5297 | 0.4433 | 0.4329 | 0.4536 | 4590.7979 | 0.00E + 00 | 3.58 | 3.49 |
| Anencephalus and ∼ | 4844 | 10 331 519 | 860 | 3 966 512 | 2.1619 | 2.0107 | 2.3245 | 0.5374 | 0.5027 | 0.5698 | 0.4564 | 0.4219 | 0.4889 | 456.2601 | 1.57E-101 | 3.75 | 3.44 |
| AVSD | 4990 | 10 331 373 | 900 | 3 966 472 | 2.1281 | 1.9823 | 2.2846 | 0.5301 | 0.4955 | 0.5623 | 0.4491 | 0.4150 | 0.4812 | 456.1556 | 1.65E-101 | 3.68 | 3.38 |
| Polydactyly | 15 794 | 10 320 569 | 2964 | 3 964 408 | 2.0453 | 1.9666 | 2.1271 | 0.5111 | 0.4915 | 0.5299 | 0.4303 | 0.4112 | 0.4488 | 1334.9209 | 1.46E-292 | 3.51 | 3.35 |
| Abdominal wall defx | 6525 | 10 329 837 | 1234 | 3 966 138 | 2.0296 | 1.9098 | 2.1568 | 0.5073 | 0.4764 | 0.5364 | 0.4266 | 0.3965 | 0.4552 | 542.2794 | 3.01E-120 | 3.48 | 3.23 |
| Urinary | 48 201 | 10 288 162 | 9559 | 3 957 814 | 1.9354 | 1.8935 | 1.9783 | 0.4833 | 0.4719 | 0.4945 | 0.4033 | 0.3923 | 0.4141 | 3621.1633 | 0.00E + 00 | 3.28 | 3.19 |
| Turner syndrome | 2965 | 10 333 397 | 551 | 3 966 822 | 2.0654 | 1.8859 | 2.2620 | 0.5158 | 0.4698 | 0.5579 | 0.4350 | 0.3900 | 0.4767 | 255.4275 | 8.52E-58 | 3.55 | 3.18 |
| Chromosomal | 53 757 | 10 282 606 | 10 726 | 3 956 646 | 1.9237 | 1.8843 | 1.9639 | 0.4802 | 0.4693 | 0.4908 | 0.4003 | 0.3899 | 0.4106 | 3983.8113 | 0.00E + 00 | 3.26 | 3.18 |
| Blader extr/epispad | 809 | 10 335 554 | 144 | 3 967 229 | 2.1564 | 1.8061 | 2.5746 | 0.5363 | 0.4463 | 0.6116 | 0.4552 | 0.3668 | 0.5313 | 75.8075 | 1.56E-18 | 3.74 | 3.01 |
| Club foot | 14 653 | 10 321 710 | 2999 | 3 964 373 | 1.8754 | 1.8032 | 1.9505 | 0.4668 | 0.4454 | 0.4873 | 0.3875 | 0.3672 | 0.4071 | 1018.4416 | 8.81E-224 | 3.16 | 3.01 |
| Hypoplastic left ht | 2946 | 10 333 417 | 580 | 3 966 793 | 1.9496 | 1.7835 | 2.1311 | 0.4871 | 0.4393 | 0.5308 | 0.4069 | 0.3612 | 0.4495 | 224.1839 | 5.53E-51 | 3.31 | 2.97 |
| Aortic atresia ∼ | 695 | 10 335 668 | 115 | 3 690 429 | 2.1578 | 1.7714 | 2.6284 | 0.5366 | 0.4355 | 0.6195 | 0.4604 | 0.3608 | 0.5444 | 61.3019 | 2.45E-15 | 3.74 | 2.94 |
| Conjoined twins | 190 | 10 336 173 | 28 | 3 967 345 | 2.6045 | 1.7516 | 3.8729 | 0.6161 | 0.4291 | 0.7418 | 0.5369 | 0.3456 | 0.6723 | 24.1230 | 4.52E-07 | 4.65 | 2.90 |
| Holoprosencephaly ∼ | 2601 | 10 333 762 | 519 | 3 966 853 | 1.9236 | 1.7506 | 2.1136 | 0.4801 | 0.4288 | 0.5269 | 0.4003 | 0.3513 | 0.4456 | 191.9022 | 6.11E-44 | 3.26 | 2.90 |
| Respiratory | 5185 | 10 331 178 | 1028 | 3 689 517 | 1.8009 | 1.6843 | 1.9255 | 0.4447 | 0.4063 | 0.4806 | 0.3711 | 0.3350 | 0.4053 | 305.6756 | 9.56E-69 | 3.00 | 2.76 |
| VSD | 46 424 | 10 289 939 | 10 401 | 3 956 971 | 1.7132 | 1.6772 | 1.7499 | 0.4163 | 0.4038 | 0.4286 | 0.3401 | 0.3285 | 0.3514 | 2532.8001 | 0.00E + 00 | 2.82 | 2.74 |
| Neural tube defects | 11 338 | 10 325 024 | 2487 | 3 964 885 | 1.7498 | 1.6755 | 1.8274 | 0.4285 | 0.4032 | 0.4528 | 0.3514 | 0.3279 | 0.3741 | 655.9895 | 5.57E-145 | 2.90 | 2.74 |
| Bilat renal agenesis | 1487 | 10 334 875 | 306 | 3 967 066 | 1.8652 | 1.6493 | 2.1094 | 0.4639 | 0.3937 | 0.5259 | 0.3847 | 0.3186 | 0.4444 | 101.8620 | 2.98E-24 | 3.14 | 2.68 |
| PV stenosis | 5118 | 10 331 245 | 1128 | 3 966 244 | 1.7415 | 1.6328 | 1.8575 | 0.4258 | 0.3876 | 0.4616 | 0.3489 | 0.3136 | 0.3824 | 291.9487 | 9.35E-66 | 2.88 | 2.65 |
| Choanal atresia | 1240 | 10 335 123 | 256 | 3 967 116 | 1.8592 | 1.6251 | 2.1269 | 0.4621 | 0.3847 | 0.5298 | 0.3830 | 0.3103 | 0.4481 | 84.2572 | 2.17E-20 | 3.12 | 2.63 |
| Down syndrome | 29 415 | 10 306 948 | 6805 | 3 960 567 | 1.6591 | 1.6160 | 1.7034 | 0.3973 | 0.3812 | 0.4129 | 0.3226 | 0.3080 | 0.3370 | 1450.7519 | 9.84E-318 | 2.70 | 2.61 |
| Nervous system | 36 794 | 10 299 568 | 8033 | 3 682 511 | 1.6354 | 1.5964 | 1.6753 | 0.3885 | 0.3736 | 0.4031 | 0.3189 | 0.3053 | 0.3322 | 1632.9280 | 0.00E + 00 | 2.65 | 2.57 |
| Double outlet RV | 1617 | 10 334 745 | 325 | 3 690 219 | 1.7764 | 1.5769 | 2.0012 | 0.4371 | 0.3658 | 0.5003 | 0.3639 | 0.2976 | 0.4240 | 91.8471 | 4.68E-22 | 2.95 | 2.53 |
| Encephalocele | 1438 | 10 334 925 | 310 | 3 967 062 | 1.7805 | 1.5748 | 2.0129 | 0.4383 | 0.3650 | 0.5032 | 0.3606 | 0.2927 | 0.4220 | 87.2573 | 4.76E-21 | 2.96 | 2.53 |
| Skeletal dysplasias | 2380 | 10 333 983 | 531 | 3 966 841 | 1.7204 | 1.5659 | 1.8900 | 0.4187 | 0.3614 | 0.4709 | 0.3423 | 0.2898 | 0.3910 | 130.9758 | 1.25E-30 | 2.83 | 2.51 |
| Bile duct A | 509 | 10 335 854 | 101 | 3 967 272 | 1.9343 | 1.5625 | 2.3947 | 0.4830 | 0.3600 | 0.5824 | 0.4030 | 0.2866 | 0.5005 | 38.0366 | 3.47E-10 | 3.28 | 2.50 |
| TV S/A | 767 | 10 335 596 | 159 | 3 967 214 | 1.8515 | 1.5609 | 2.1964 | 0.4599 | 0.3593 | 0.5447 | 0.3809 | 0.2869 | 0.4626 | 51.5808 | 3.44E-13 | 3.11 | 2.50 |
| All anomalies | 309 546 | 10 026 817 | 75 745 | 3 891 627 | 1.5686 | 1.5563 | 1.5810 | 0.3625 | 0.3574 | 0.3675 | 0.2912 | 0.2867 | 0.2957 | 12 889.1510 | 0.00E + 00 | 2.51 | 2.49 |
| Severe CHD | 25 055 | 10 311 308 | 6065 | 3 961 307 | 1.5856 | 1.5418 | 1.6307 | 0.3693 | 0.3514 | 0.3868 | 0.2974 | 0.2813 | 0.3130 | 1058.4314 | 1.79E-232 | 2.55 | 2.46 |
| Anorectal S/A | 3430 | 10 332 933 | 792 | 3 966 581 | 1.6623 | 1.5387 | 1.7958 | 0.3984 | 0.3501 | 0.4431 | 0.3237 | 0.2799 | 0.3648 | 169.8277 | 4.03E-39 | 2.71 | 2.45 |
| Craniosynostosis | 4319 | 10 332 043 | 1018 | 3 966 355 | 1.6284 | 1.5210 | 1.7435 | 0.3859 | 0.3425 | 0.4264 | 0.3123 | 0.2732 | 0.3493 | 199.8699 | 1.11E-45 | 2.64 | 2.41 |
| Cong. cataract | 1905 | 10 334 457 | 435 | 3 966 938 | 1.6809 | 1.5146 | 1.8654 | 0.4051 | 0.3398 | 0.4639 | 0.3298 | 0.2705 | 0.3843 | 97.6924 | 2.44E-23 | 2.75 | 2.40 |
| Eye | 5774 | 10 330 589 | 1380 | 3 965 992 | 1.6060 | 1.5144 | 1.7031 | 0.3773 | 0.3397 | 0.4128 | 0.3045 | 0.2708 | 0.3367 | 254.7824 | 1.18E-57 | 2.59 | 2.40 |
| Vascular disruptions | 7593 | 10 328 770 | 1732 | 3 688 812 | 1.5653 | 1.4857 | 1.6491 | 0.3611 | 0.3269 | 0.3936 | 0.2941 | 0.2634 | 0.3234 | 288.0838 | 6.50E-65 | 2.51 | 2.34 |
| Cong. glaucoma | 650 | 10 335 713 | 140 | 3 967 232 | 1.7821 | 1.4846 | 2.1391 | 0.4388 | 0.3264 | 0.5325 | 0.3611 | 0.2575 | 0.4502 | 39.5357 | 1.61E-10 | 2.96 | 2.33 |
| PV atresia | 1435 | 10 334 928 | 331 | 3 967 042 | 1.6640 | 1.4766 | 1.8753 | 0.3990 | 0.3228 | 0.4667 | 0.3243 | 0.2553 | 0.3868 | 71.2770 | 1.55E-17 | 2.72 | 2.32 |
| Anotia | 298 | 10 336 065 | 59 | 3 967 313 | 1.9387 | 1.4663 | 2.5632 | 0.4842 | 0.3180 | 0.6099 | 0.4042 | 0.2477 | 0.5281 | 22.3830 | 1.12E-06 | 3.29 | 2.29 |
| Hirschsprungs | 1562 | 10 334 800 | 366 | 3 967 006 | 1.6381 | 1.4619 | 1.8355 | 0.3895 | 0.3159 | 0.4552 | 0.3156 | 0.2495 | 0.3759 | 73.7104 | 4.52E-18 | 2.66 | 2.28 |
| Tetralogy of Fallot | 3815 | 10 332 548 | 943 | 3 966 430 | 1.5528 | 1.4460 | 1.6675 | 0.3560 | 0.3084 | 0.4003 | 0.2854 | 0.2434 | 0.3251 | 148.8534 | 1.54E-34 | 2.48 | 2.25 |
| Limb reductions | 6606 | 10 329 757 | 1663 | 3 965 710 | 1.5247 | 1.4449 | 1.6089 | 0.3441 | 0.3079 | 0.3785 | 0.2749 | 0.2431 | 0.3054 | 240.0232 | 1.94E-54 | 2.42 | 2.25 |
| Genital | 24 721 | 10 311 642 | 6108 | 3 684 437 | 1.4451 | 1.4052 | 1.4861 | 0.3080 | 0.2884 | 0.3271 | 0.2470 | 0.2299 | 0.2637 | 672.8740 | 1.18E-148 | 2.25 | 2.16 |
| Hypospadias | 20 687 | 10 315 676 | 5119 | 3 685 426 | 1.4429 | 1.3995 | 1.4877 | 0.3069 | 0.2854 | 0.3278 | 0.2461 | 0.2274 | 0.2643 | 558.8957 | 7.31E-124 | 2.24 | 2.15 |
| Spina bifida | 5056 | 10 331 306 | 1317 | 3 966 056 | 1.4735 | 1.3868 | 1.5656 | 0.3214 | 0.2789 | 0.3613 | 0.2549 | 0.2182 | 0.2899 | 159.0619 | 9.07E-37 | 2.31 | 2.12 |
| Diaphragmatic hernia | 3337 | 10 333 026 | 866 | 3 966 507 | 1.4790 | 1.3725 | 1.5938 | 0.3239 | 0.2714 | 0.3726 | 0.2571 | 0.2117 | 0.2999 | 106.7022 | 2.59E-25 | 2.32 | 2.09 |
| Congenital heart | 96 438 | 10 239 925 | 24 784 | 3 665 761 | 1.3893 | 1.3701 | 1.4088 | 0.2802 | 0.2701 | 0.2902 | 0.2229 | 0.2143 | 0.2315 | 2169.7116 | 0.00E + 00 | 2.12 | 2.08 |
| Annular pancreas | 286 | 10 336 077 | 63 | 3 967 309 | 1.7425 | 1.3265 | 2.2889 | 0.4261 | 0.2461 | 0.5631 | 0.3492 | 0.1862 | 0.4795 | 16.3330 | 2.66E-05 | 2.88 | 1.98 |
| Cleft lip ± palate | 7984 | 10 328 379 | 2236 | 3 965 136 | 1.3705 | 1.3077 | 1.4363 | 0.2703 | 0.2353 | 0.3038 | 0.2112 | 0.1818 | 0.2396 | 175.0977 | 2.85E-40 | 2.08 | 1.94 |
| Transpos grt vess | 3798 | 10 332 564 | 1042 | 3 966 330 | 1.3990 | 1.3063 | 1.4983 | 0.2852 | 0.2345 | 0.3326 | 0.2238 | 0.1809 | 0.2644 | 93.0853 | 2.50E-22 | 2.15 | 1.94 |
| Oesophageal S/A | 2927 | 10 333 436 | 814 | 3 966 559 | 1.3802 | 1.2771 | 1.4916 | 0.2755 | 0.2169 | 0.3296 | 0.2155 | 0.1664 | 0.2618 | 66.7118 | 1.57E-16 | 2.10 | 1.87 |
| Hydrocephalus | 6444 | 10 329 919 | 1841 | 3 965 531 | 1.3435 | 1.2757 | 1.4149 | 0.2557 | 0.2161 | 0.2932 | 0.1989 | 0.1659 | 0.2305 | 125.8285 | 1.68E-29 | 2.02 | 1.87 |
| Orofacial clefts | 13 773 | 10 322 590 | 3735 | 3 686 809 | 1.3166 | 1.2699 | 1.3651 | 0.2405 | 0.2125 | 0.2674 | 0.1892 | 0.1658 | 0.2119 | 223.9970 | 6.07E-51 | 1.96 | 1.86 |
| Gastroschisis | 1815 | 10 334 548 | 497 | 3 966 875 | 1.4017 | 1.2693 | 1.5479 | 0.2866 | 0.2122 | 0.3540 | 0.2250 | 0.1622 | 0.2831 | 44.9227 | 1.02E-11 | 2.15 | 1.85 |
| Arterial truncus | 750 | 10 335 613 | 195 | 3 967 177 | 1.4763 | 1.2611 | 1.7282 | 0.3226 | 0.2070 | 0.4213 | 0.2560 | 0.1570 | 0.3435 | 23.7801 | 5.40E-07 | 2.31 | 1.83 |
| Digestive system | 20 789 | 10 315 573 | 5754 | 3 684 791 | 1.2900 | 1.2529 | 1.3282 | 0.2248 | 0.2019 | 0.2471 | 0.1761 | 0.1570 | 0.1947 | 294.3454 | 2.81E-66 | 1.90 | 1.82 |
| Single ventricle | 913 | 10 335 450 | 245 | 3 967 127 | 1.4303 | 1.2422 | 1.6470 | 0.3009 | 0.1950 | 0.3928 | 0.2372 | 0.1475 | 0.3175 | 25.0121 | 2.85E-07 | 2.21 | 1.79 |
| Syndactyly | 4503 | 10 331 859 | 1346 | 3 966 027 | 1.2841 | 1.2082 | 1.3647 | 0.2212 | 0.1724 | 0.2672 | 0.1703 | 0.1305 | 0.2083 | 65.1528 | 3.47E-16 | 1.89 | 1.71 |
| Cleft palate | 5688 | 10 330 674 | 1609 | 3 688 936 | 1.2622 | 1.1943 | 1.3340 | 0.2077 | 0.1627 | 0.2504 | 0.1619 | 0.1250 | 0.1973 | 68.3469 | 6.86E-17 | 1.84 | 1.68 |
| An/microphthalmos | 1347 | 10 335 016 | 401 | 3 966 972 | 1.2893 | 1.1533 | 1.4414 | 0.2244 | 0.1329 | 0.3062 | 0.1729 | 0.0987 | 0.2410 | 20.0633 | 3.75E-06 | 1.90 | 1.57 |
| Aortic valve S/A | 1234 | 10 335 128 | 388 | 3 966 985 | 1.2207 | 1.0891 | 1.3682 | 0.1808 | 0.0818 | 0.2691 | 0.1376 | 0.0594 | 0.2093 | 11.7826 | 2.99E-04 | 1.74 | 1.40 |
| Mitral valve anomalies | 1674 | 10 334 689 | 520 | 3 690 024 | 1.1494 | 1.0417 | 1.2683 | 0.1300 | 0.0400 | 0.2115 | 0.0992 | 0.0290 | 0.1643 | 7.7067 | 0.0028 | 1.56 | 1.25 |
| PDA | 2871 | 10 333 491 | 1012 | 3 966 360 | 1.0889 | 1.0136 | 1.1698 | 0.0816 | 0.0134 | 0.1451 | 0.0604 | 0.0092 | 0.1088 | 5.4322 | 0.0099 | 1.40 | 1.13 |
| Coarctation aorta | 3241 | 10 333 122 | 1161 | 3 966 212 | 1.0715 | 1.0020 | 1.1458 | 0.0667 | 0.0020 | 0.1272 | 0.0491 | 0.0010 | 0.0949 | 4.0768 | 0.0217 | 1.35 | 1.05 |
| Duodenal S/A | 1275 | 10 335 088 | 473 | 3 966 900 | 1.0346 | 0.9310 | 1.1498 | 0.0335 | −0.0741 | 0.1303 | 0.0244 | −0.0536 | 0.0967 | 0.3999 | 0.2636 | 1.22 | 1.00 |
| Klinefelter | 798 | 10 335 564 | 279 | 3 967 094 | 1.0978 | 0.9579 | 1.2582 | 0.0891 | −0.0439 | 0.2052 | 0.0660 | −0.0332 | 0.1558 | 1.8022 | 0.0897 | 1.43 | 1.00 |
| Small intestine S/A | 1053 | 10 335 310 | 394 | 3 966 978 | 1.0258 | 0.9137 | 1.1517 | 0.0252 | −0.0945 | 0.1317 | 0.0183 | −0.0680 | 0.0976 | 0.1862 | 0.3330 | 1.19 | 1.00 |
| Anophthalmos | 191 | 10 336 172 | 78 | 3 967 295 | 0.9399 | 0.7223 | 1.2231 | −0.0640 | −0.3845 | 0.1824 | −0.0454 | −0.2604 | 0.1329 | 0.2130 | 0.3222 | 1.32 | — |
| ASD | 13 356 | 10 323 007 | 6183 | 3 961 190 | 0.8291 | 0.8045 | 0.8545 | −0.2061 | −0.2430 | −0.1703 | −0.1409 | −0.1646 | −0.1176 | 149.0682 | 1.39E-34 | 1.70 | — |
| Ear, face and neck | 2475 | 10 333 888 | 1232 | 3 966 140 | 0.7711 | 0.7202 | 0.8256 | −0.2969 | −0.3886 | −0.2112 | −0.1982 | −0.2541 | −0.1448 | 55.9168 | 3.78E-14 | 1.92 | — |
| Indeterminate sex | 303 | 10 336 060 | 241 | 3 967 132 | 0.4826 | 0.4075 | 0.5715 | −1.0722 | −1.4542 | −0.7497 | −0.5972 | −0.7550 | −0.4536 | 74.4764 | 3.07E-18 | 3.56 | — |
| Tot anom pul V ret | 526 | 10 335 837 | 216 | 3 967 156 | 0.9347 | 0.7978 | 1.0951 | −0.0699 | −0.2535 | 0.0868 | −0.0495 | −0.1742 | 0.0619 | 0.6988 | 0.2016 | 1.34 | — |
The numbers of anomalies with elevated mEVs from this analysis by substance are shown in tabular and graphical formats in eTable 30 and eFig. 27.
If one studies the lists for the cannabis metrics closely, it is noted that 89 of the 90 (98.9%) CAs demonstrate elevated mEVs by one or more indices of cannabinoid exposure. The sole exception is indeterminate sex. This anomaly, however, is mentioned positively in the tables shown below. Complete coverage of this set of 89 CAs can be achieved by considering together the three covariates cannabis herb THC concentration, daily cannabis use and LMC_Resin_Daily.
Forest Regression
Since the analysis clearly needs to move from bivariate into multivariable regression, a salient issue is which variable/s should be used as the key metric of cannabis exposure moving forwards? This issue is not immediately apparent in the rich European dataset.
Additive and interactive linear model of all covariates against (log) CA rates were constructed, and the final forms of these linear models after model reduction are shown in eTable 31. A three-way interaction between tobacco, alcohol and LMC_Herb_Daily was used in the interactive model. Forest regression was conducted on these models using the Gini (‘impurity’) index as the measure of variable importance. This procedure derived the results shown in tabular form in eTable 32 and in graphical form as variable importance plots in Fig. 7. It is clear from these analyses that for both models compound indices of daily cannabis exposure and cannabis herb THC concentration are the most powerful covariates for entry into multivariable regression.
Figure 7:
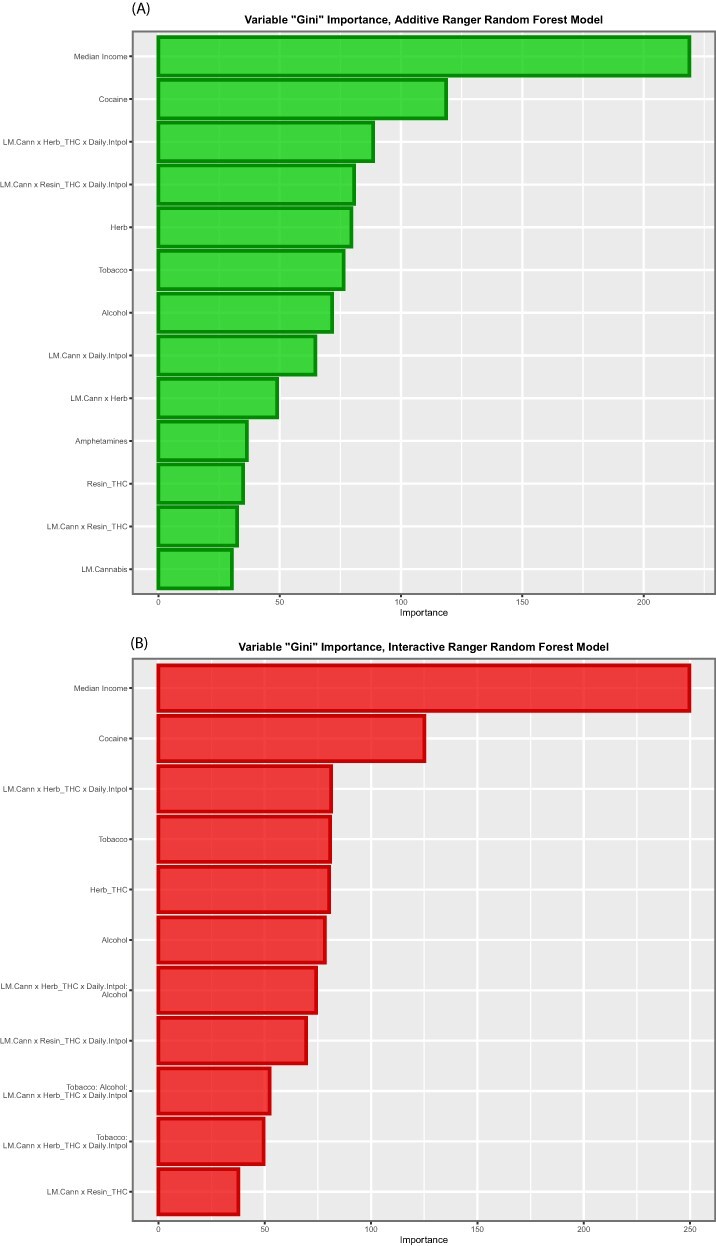
Variable importance plots for (A) additive and (B) interactive random forest models (at zero lags)
Multivariable Regression
Additive Models
For these reasons, the three covariates cannabis herb THC concentration, LMC_Herb_Daily and LMC_Resin_Daily were selected for use in multivariable regression. They were first applied together with the other substances and median household income in an additive panel regression model. All panel regression models were IPW. eTable 33 shows the result of these regressions for 196 statistically significant positive terms From this eTable, 86 terms were extracted, which included a term for the metrics of cannabis (eTable 34). The most significant terms for each separate CA were then selected out, leaving 76 anomalies, which appear in Table 4. Interestingly, this table is headed by cardiovascular disorders, genital disorders, microphthalmos, polydactyly, nervous system disorders, patent ductus arteriosus (PDA), atrial septal defect (ASD) and genetic and facial anomalies, which have all been previously reported to be cannabis associated [4–8, 18, 55]. It is also noted that the P-values in this table ascend from 1.5 × 10−23 (for teratogenic syndromes) and the mEV descend from 8.2 × 109.
Table 4:
Multivariate additive IPW panel regression results
| Anomaly | Term | Mean rate | β-estimate | Standard error | Sigma | T-statistic | Adj. R2 | P-value | E-value estimate | E-value (95% lower bound) | P-value exponent | E-value exponent | % Increment | P—Bonferroni | P—false discovery rate | P—Holm |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Congenital heart | Herb | 82.6649 | 8.8588 | 0.9896 | 0.2846 | 8.9516 | 0.5218 | 5.16E-14 | 3.99E + 12 | 8.19E + 09 | 14 | 9 | 3.07 | 3.82E-12 | 9.55E-13 | 3.67E-12 |
| Genital | Herb | 22.6289 | 7.7667 | 1.3378 | 0.3848 | 5.8055 | 0.5099 | 9.98E-08 | 1.90E + 08 | 3.89E + 05 | 8 | 5 | 11.20 | 7.39E-06 | 8.21E-07 | 6.59E-06 |
| An/microphthalmos | LpmHerbDailyInt | 0.9004 | 12.2684 | 1.3776 | 0.7287 | 8.9056 | −0.0865 | 4.98E-14 | 9.00E + 06 | 3.11E + 05 | 14 | 5 | 281.40 | 3.68E-12 | 9.55E-13 | 3.58E-12 |
| Aortic valve S/A | LpmHerbDailyInt | 1.3094 | 14.1512 | 1.6110 | 0.8522 | 8.7841 | −0.0520 | 8.93E-14 | 7.31E + 06 | 2.53E + 05 | 14 | 5 | 193.51 | 6.61E-12 | 1.32E-12 | 6.25E-12 |
| Polydactyly | Herb | 10.3496 | 9.1270 | 1.6788 | 0.4957 | 5.4367 | 0.3313 | 4.52E-07 | 3.79E + 07 | 9.12E + 04 | 7 | 4 | 24.48 | 3.34E-05 | 2.57E-06 | 2.80E-05 |
| Nervous system | Herb | 26.1182 | 7.7253 | 1.4583 | 0.4194 | 5.2975 | 0.3384 | 8.58E-07 | 3.80E + 07 | 7.80E + 04 | 7 | 4 | 9.70 | 6.35E-05 | 4.53E-06 | 5.23E-05 |
| PDA | LpmHerbDailyInt | 2.9086 | 11.1711 | 1.5486 | 0.8192 | 7.2137 | 0.3160 | 1.57E-10 | 4.90E + 05 | 1.69E + 04 | 10 | 4 | 87.11 | 1.16E-08 | 1.94E-09 | 1.08E-08 |
| ASD | Herb | 21.4739 | 9.2217 | 1.9147 | 0.5653 | 4.8163 | 0.1194 | 5.80E-06 | 5.59E + 06 | 1.35E + 04 | 5 | 4 | 11.80 | 4.29E-04 | 2.42E-05 | 3.37E-04 |
| Patau syndrome | LpmHerbDailyInt | 1.7507 | 9.5652 | 1.3862 | 0.7333 | 6.9003 | 0.0513 | 6.74E-10 | 2.86E + 05 | 9.88E + 03 | 10 | 3 | 144.73 | 4.99E-08 | 7.13E-09 | 4.59E-08 |
| Ear, face and neck | Herb | 2.4050 | 18.1635 | 4.1690 | 1.2309 | 4.3568 | 0.0233 | 3.46E-05 | 1.36E + 06 | 3.27E + 03 | 4 | 3 | 105.35 | 0.00255941 | 1.07E-04 | 0.00179197 |
| Teratogenic synds | LpmResinDailyInt | 1.2227 | 5.9944 | 0.4423 | 0.6403 | 13.5526 | 0.4714 | 1.50E-23 | 1.00E + 04 | 2.93E + 03 | 23 | 3 | 178.34 | 1.11E-21 | 1.11E-21 | 1.11E-21 |
| Arterial truncus | Herb | 1.0030 | 10.0572 | 2.3589 | 0.6965 | 4.2636 | 0.0782 | 4.91E-05 | 1.02E + 06 | 2.45E + 03 | 4 | 3 | 252.62 | 0.00363094 | 1.40E-04 | 0.00240427 |
| Hypospadias | Herb | 19.9122 | 7.3602 | 1.7734 | 0.5101 | 4.1502 | 0.4986 | 7.65E-05 | 1.01E + 06 | 2.07E + 03 | 4 | 3 | 12.72 | 0.00566086 | 2.02E-04 | 0.00359541 |
| Orofacial clefts | Herb | 14.1998 | 8.6815 | 2.1057 | 0.6057 | 4.1229 | 0.1253 | 8.45E-05 | 9.25E + 05 | 1.90E + 03 | 4 | 3 | 17.84 | 0.00625453 | 2.09E-04 | 0.00388795 |
| Posterior urethral valve | Herb | 1.2952 | 8.8449 | 2.2296 | 0.6583 | 3.9670 | 0.2628 | 1.45E-04 | 4.08E + 05 | 982.80 | 3 | 2 | 195.63 | 0.01072607 | 3.15E-04 | 0.00594282 |
| Matern infect malform | LpmResinDailyInt | 0.8059 | 5.4136 | 0.4771 | 0.6906 | 11.3481 | 0.3104 | 4.07E-19 | 2.51E + 03 | 732.22 | 19 | 2 | 270.57 | 3.02E-17 | 1.51E-17 | 2.97E-17 |
| Urinary | Herb | 36.0821 | 6.0183 | 1.5679 | 0.4629 | 3.8386 | 0.1964 | 0.0002 | 2.75E + 05 | 661.40 | 3 | 2 | 7.02 | 0.016893 | 4.57E-04 | 0.00867478 |
| Limb | Herb | 37.7465 | 5.5628 | 1.5229 | 0.4380 | 3.6527 | 0.5350 | 0.0004 | 2.09E + 05 | 428.32 | 3 | 2 | 6.71 | 0.0325785 | 7.97E-04 | 0.01540875 |
| Tetralogy of Fallot | LpmHerbDailyInt | 3.1167 | 8.3069 | 1.7397 | 0.9203 | 4.7748 | −0.0571 | 6.85E-06 | 7.38E + 03 | 254.72 | 5 | 2 | 81.30 | 5.07E-04 | 2.55E-05 | 3.83E-04 |
| Hydrocephalus | LpmHerbDailyInt | 5.3737 | 7.0000 | 1.4664 | 0.7757 | 4.7735 | −0.0047 | 6.88E-06 | 7.37E + 03 | 254.19 | 5 | 2 | 47.15 | 5.09E-04 | 2.55E-05 | 3.83E-04 |
| Multicystic renal dys | Herb | 3.5369 | 9.7517 | 2.7887 | 0.8234 | 3.4969 | 0.3369 | 0.0007 | 9.59E + 04 | 230.40 | 3 | 2 | 71.64 | 0.05402309 | 0.00125635 | 0.02336134 |
| Syndactyly | Herb | 4.2302 | 7.3389 | 2.1361 | 0.6307 | 3.4357 | 0.0478 | 0.0009 | 7.94E + 04 | 190.72 | 3 | 2 | 59.90 | 0.0660369 | 0.00150084 | 0.02766411 |
| Encephalocele | LpmHerbDailyInt | 1.1446 | 6.2213 | 1.4122 | 0.7470 | 4.4055 | −0.0748 | 2.88E-05 | 3.91E + 03 | 134.71 | 4 | 2 | 221.37 | 0.00212862 | 1.01E-04 | 0.00155332 |
| Annular pancreas | LpmHerbDailyInt | 0.2372 | 3.0116 | 0.6855 | 0.3626 | 4.3930 | −0.0313 | 3.02E-05 | 3.83E + 03 | 131.85 | 4 | 2 | 1068.20 | 0.00223154 | 1.01E-04 | 0.00159826 |
| Severe microcephaly | LpmHerbDailyInt | 2.6832 | 7.5642 | 1.7358 | 0.9182 | 4.3578 | 0.1977 | 3.45E-05 | 3.60E + 03 | 124.06 | 4 | 2 | 94.43 | 0.00255012 | 1.07E-04 | 0.00179197 |
| Spina bifida | Herb | 4.3705 | 5.6604 | 1.7243 | 0.5091 | 3.2828 | 0.1039 | 0.0015 | 4.96E + 04 | 118.87 | 2 | 2 | 57.97 | 0.10797166 | 0.00234721 | 0.04231322 |
| Down syndrome | LpmHerbDailyInt | 20.7996 | 3.3573 | 0.7784 | 0.4117 | 4.3132 | 0.2441 | 4.08E-05 | 3.34E + 03 | 114.86 | 4 | 2 | 12.18 | 0.00301611 | 1.21E-04 | 0.00203791 |
| VATER/VACTERL | Herb | 0.4798 | 6.1281 | 1.9199 | 0.5522 | 3.1919 | 0.3359 | 0.0020 | 4.86E + 04 | 99.31 | 2 | 1 | 528.09 | 0.14505544 | 0.00302199 | 0.05315528 |
| Situs inversus | LpmHerbDailyInt | 0.6187 | 3.5795 | 0.8573 | 0.4535 | 4.1752 | 0.2135 | 6.81E-05 | 2.63E + 03 | 90.49 | 4 | 1 | 409.53 | 0.00503876 | 1.87E-04 | 0.00326839 |
| Diaphragmatic hernia | LpmHerbDailyInt | 2.5089 | 7.1983 | 1.7493 | 0.9253 | 4.1151 | −0.0659 | 8.49E-05 | 2.37E + 03 | 81.54 | 4 | 1 | 100.99 | 0.00628208 | 2.09E-04 | 0.00388795 |
| Cleft lip ± palate | LpmHerbDailyInt | 8.6449 | 5.8082 | 1.4199 | 0.7511 | 4.0904 | 0.0181 | 9.29E-05 | 2.27E + 03 | 78.14 | 4 | 1 | 29.31 | 0.00687259 | 2.22E-04 | 0.00408641 |
| Anotia | LpmHerbDailyInt | 0.2330 | 1.8699 | 0.4817 | 0.2548 | 3.8816 | 0.0073 | 0.0002 | 1.59E + 03 | 54.40 | 3 | 1 | 1087.45 | 0.01452356 | 4.03E-04 | 0.00765431 |
| Severe CHD | LpmHerbDailyInt | 21.9866 | 4.5125 | 1.1816 | 0.6251 | 3.8189 | −0.0172 | 0.0002 | 1.43E + 03 | 48.79 | 3 | 1 | 11.52 | 0.01809651 | 4.66E-04 | 0.00904826 |
| Hydronephrosis | LpmHerbDailyInt | 13.5286 | 5.9134 | 1.5490 | 0.8194 | 3.8176 | 0.2531 | 0.0002 | 1.42E + 03 | 48.68 | 3 | 1 | 18.73 | 0.01817657 | 4.66E-04 | 0.00904826 |
| Vascular disruptions | LpmResinDailyInt | 6.9668 | 2.9516 | 0.4384 | 0.6346 | 6.7330 | 0.1494 | 1.64E-09 | 137.29 | 39.78 | 9 | 1 | 31.30 | 1.21E-07 | 1.51E-08 | 1.10E-07 |
| Genetic syndromes | LpmHerbDailyInt | 6.5208 | 6.4410 | 1.7660 | 0.9342 | 3.6473 | −0.0163 | 0.0004 | 1.06E + 03 | 36.18 | 3 | 1 | 38.86 | 0.03265865 | 7.97E-04 | 0.01540875 |
| Hypoplastic left ht | LpmHerbDailyInt | 2.3257 | 5.8086 | 1.6505 | 0.8731 | 3.5193 | 0.0102 | 0.0007 | 851.29 | 28.93 | 3 | 1 | 108.95 | 0.05015837 | 0.00119425 | 0.02236792 |
| VSD | Herb | 37.2628 | 5.3939 | 1.9550 | 0.5772 | 2.7591 | 0.1610 | 0.0070 | 9.87E + 03 | 23.25 | 2 | 1 | 6.80 | 0.5185063 | 0.00925904 | 0.13313 |
| Gastroschisis | LpmResinDailyInt | 2.3468 | 3.5118 | 0.6263 | 0.9067 | 5.6071 | 0.1511 | 2.19E-07 | 67.37 | 19.34 | 7 | 1 | 92.92 | 1.62E-05 | 1.52E-06 | 1.42E-05 |
| Blader extr/epispad | LpmResinDailyInt | 0.7056 | 1.9668 | 0.3513 | 0.5085 | 5.5991 | −0.0206 | 2.26E-07 | 67.03 | 19.24 | 7 | 1 | 309.03 | 1.67E-05 | 1.52E-06 | 1.45E-05 |
| Indeterminate sex | Herb | 0.5005 | 5.2208 | 1.9503 | 0.5758 | 2.6769 | −0.0617 | 0.0088 | 7.66E + 03 | 17.93 | 2 | 1 | 506.25 | 0.65226722 | 0.01105538 | 0.14179341 |
| Cong. glaucoma | LpmResinDailyInt | 0.2853 | 2.2735 | 0.4156 | 0.6016 | 5.4708 | 0.0968 | 3.91E-07 | 61.80 | 17.70 | 7 | 1 | 764.30 | 2.89E-05 | 2.41E-06 | 2.46E-05 |
| Ebstein’s anomaly | Herb | 0.4111 | 3.4408 | 1.3011 | 0.3842 | 2.6445 | 0.0493 | 0.0096 | 6.93E + 03 | 16.18 | 2 | 1 | 616.34 | 0.71306363 | 0.01188439 | 0.14453992 |
| All anomalies | LpmHerbDailyInt | 249.8954 | 2.1805 | 0.6920 | 0.3661 | 3.1510 | 0.2718 | 0.0022 | 451.56 | 15.11 | 2 | 1 | 1.01 | 0.16299042 | 0.00332634 | 0.0572669 |
| Chromosomal | LpmHerbDailyInt | 35.9025 | 2.9271 | 0.9356 | 0.4949 | 3.1286 | 0.1256 | 0.0024 | 434.41 | 14.51 | 2 | 1 | 7.06 | 0.17465658 | 0.00349313 | 0.0590056 |
| Cong. cataract | LpmResinDailyInt | 1.1682 | 3.0794 | 0.5990 | 0.8672 | 5.1406 | 0.0874 | 1.56E-06 | 50.12 | 14.29 | 5 | 1 | 186.66 | 1.15E-04 | 7.68E-06 | 9.35E-05 |
| Abdominal wall defx | LpmResinDailyInt | 5.7704 | 2.2561 | 0.4415 | 0.6392 | 5.1100 | 0.3983 | 1.77E-06 | 49.16 | 14.00 | 5 | 1 | 37.79 | 1.31E-04 | 8.17E-06 | 1.04E-04 |
| Craniosynostosis | LpmHerbDailyInt | 2.9964 | 4.9570 | 1.6039 | 0.8485 | 3.0905 | 0.2027 | 0.0027 | 406.88 | 13.56 | 2 | 1 | 84.56 | 0.19616883 | 0.00384645 | 0.06362232 |
| AVSD | Herb | 4.0854 | 6.2821 | 2.4410 | 0.7207 | 2.5735 | −0.0096 | 0.0117 | 5.57E + 03 | 12.89 | 1 | 1 | 62.02 | 0.86452845 | 0.0141726 | 0.16355944 |
| Lateral anomalies | Herb | 1.8149 | 6.2780 | 2.4679 | 0.7098 | 2.5438 | 0.1470 | 0.0127 | 6.26E + 03 | 12.32 | 1 | 1 | 139.61 | 0.94057227 | 0.01517052 | 0.16523567 |
| Limb reductions | LpmResinDailyInt | 5.3892 | 2.1126 | 0.4390 | 0.6355 | 4.8126 | 0.1210 | 5.89E-06 | 40.69 | 11.52 | 5 | 1 | 40.46 | 4.36E-04 | 2.42E-05 | 3.37E-04 |
| Mitral valve anomalies | LpmHerbDailyInt | 1.6067 | 5.6814 | 1.9368 | 1.0205 | 2.9334 | 0.3100 | 0.0043 | 316.61 | 10.29 | 2 | 1 | 157.70 | 0.31625901 | 0.00596715 | 0.09614151 |
| Conjoined twins | Herb | 0.1128 | 1.9128 | 0.7687 | 0.2270 | 2.4882 | −0.0424 | 0.0147 | 4.28E + 03 | 9.78 | 1 | 0 | 2246.24 | 1 | 0.01721843 | 0.17590719 |
| Skeletal dysplasias | LpmHerbDailyInt | 1.8518 | 4.7853 | 1.6649 | 0.8807 | 2.8743 | −0.0040 | 0.0050 | 280.35 | 9.18 | 2 | 0 | 136.83 | 0.37296464 | 0.00678118 | 0.10080125 |
| Transpos grt vess | Herb | 3.4440 | 5.7348 | 2.3746 | 0.7011 | 2.4151 | 0.1216 | 0.0177 | 3.42E + 03 | 7.70 | 1 | 0 | 73.57 | 1 | 0.0201855 | 0.17730503 |
| Anorectal S/A | LpmResinDailyInt | 3.1853 | 1.9684 | 0.4825 | 0.6985 | 4.0795 | 0.1261 | 9.67E-05 | 25.47 | 7.06 | 4 | 0 | 68.46 | 0.00715222 | 2.24E-04 | 0.00415602 |
| Choanal atresia | LpmResinDailyInt | 0.9191 | 1.9449 | 0.4883 | 0.7070 | 3.9826 | 0.0914 | 1.37E-04 | 23.94 | 6.61 | 3 | 0 | 237.25 | 0.01014212 | 3.07E-04 | 0.00575634 |
| Foetal alcohol | LpmResinDailyInt | 0.2577 | 0.8498 | 0.2158 | 0.3124 | 3.9386 | 0.3406 | 0.0002 | 23.27 | 6.41 | 3 | 0 | 846.15 | 0.0118676 | 3.39E-04 | 0.00641492 |
| Single ventricle | LpmResinDailyInt | 0.7525 | 1.2747 | 0.3735 | 0.5407 | 3.4128 | 0.0451 | 0.0010 | 16.57 | 4.43 | 3 | 0 | 289.77 | 0.07114277 | 0.00158095 | 0.02884166 |
| Hip dysplasia | LpmResinDailyInt | 6.3491 | 2.3960 | 0.7482 | 1.0831 | 3.2023 | 0.2890 | 0.0019 | 14.45 | 3.80 | 2 | 0 | 34.34 | 0.1404818 | 0.00298897 | 0.05315528 |
| Cleft palate | Herb | 5.4928 | 6.5824 | 2.9987 | 0.8625 | 2.1951 | −0.0213 | 0.0308 | 2.07E + 03 | 3.68 | 1 | 0 | 46.13 | 1 | 0.03302199 | 0.21425568 |
| Eye | LpmResinDailyInt | 3.6805 | 1.3682 | 0.4656 | 0.6741 | 2.9384 | 0.2060 | 0.0042 | 12.16 | 3.11 | 2 | 0 | 59.25 | 0.30932487 | 0.00594856 | 0.09614151 |
| Respiratory | LpmResinDailyInt | 3.2843 | 1.7470 | 0.5971 | 0.8644 | 2.9258 | 0.5774 | 0.0044 | 12.06 | 3.08 | 2 | 0 | 66.39 | 0.32335886 | 0.00598813 | 0.09614151 |
| Bilat renal agenesis | LpmHerbDailyInt | 1.5241 | 3.3242 | 1.4461 | 0.7649 | 2.2988 | −0.0651 | 0.0238 | 103.84 | 3.01 | 1 | 0 | 166.25 | 1 | 0.02669179 | 0.21425568 |
| PV atresia | LpmHerbDailyInt | 1.1589 | 3.3990 | 1.4839 | 0.7850 | 2.2905 | −0.0789 | 0.0243 | 102.37 | 2.95 | 1 | 0 | 218.63 | 1 | 0.02684011 | 0.21425568 |
| Bile duct A | LpmResinDailyInt | 0.2854 | 0.9697 | 0.3556 | 0.5147 | 2.7273 | 0.1397 | 0.0077 | 10.58 | 2.63 | 2 | 0 | 764.03 | 0.56702174 | 0.00994775 | 0.13792421 |
| Edward syndrome | LpmResinDailyInt | 4.9591 | 1.4804 | 0.5490 | 0.7947 | 2.6968 | 0.2853 | 0.0083 | 10.37 | 2.57 | 2 | 0 | 43.97 | 0.61721838 | 0.0106417 | 0.14179341 |
| Neural tube defects | LpmHerbDailyInt | 9.0634 | 2.2506 | 1.0304 | 0.5451 | 2.1841 | 0.1583 | 0.0315 | 85.16 | 2.32 | 1 | 0 | 27.96 | 1 | 0.03303907 | 0.21425568 |
| Holoprosencephaly ∼ | LpmResinDailyInt | 1.6371 | 1.0551 | 0.4262 | 0.6170 | 2.4756 | 0.1276 | 0.0152 | 8.95 | 2.12 | 1 | 0 | 133.20 | 1 | 0.01751788 | 0.17590719 |
| Hypoplastic right ht | LpmHerbDailyInt | 0.6594 | 2.3160 | 1.0852 | 0.5741 | 2.1342 | −0.0720 | 0.0355 | 78.11 | 2.06 | 1 | 0 | 384.25 | 1 | 0.03650427 | 0.21425568 |
| Aortic atresia ∼ | LpmResinDailyInt | 0.5426 | 0.8590 | 0.3779 | 0.5470 | 2.2732 | −0.0431 | 0.0254 | 7.81 | 1.74 | 1 | 0 | 401.89 | 1 | 0.02769426 | 0.21425568 |
| Digestive system | Herb | 17.0099 | 3.1511 | 1.5699 | 0.4515 | 2.0072 | 0.1302 | 0.0478 | 1.15E + 03 | 1.63 | 1 | 0 | 14.90 | 1 | 0.04779159 | 0.21425568 |
| Club foot | LpmResinDailyInt | 10.6471 | 1.0225 | 0.4687 | 0.6785 | 2.1818 | 0.3969 | 0.0317 | 7.35 | 1.57 | 1 | 0 | 20.48 | 1 | 0.03303907 | 0.21425568 |
| Small intestine S/A | LpmResinDailyInt | 1.0797 | 1.1091 | 0.5432 | 0.7863 | 2.0420 | −0.0879 | 0.0440 | 6.68 | 1.30 | 1 | 0 | 201.96 | 1 | 0.04465153 | 0.21425568 |
These data are summarized in Table 5, which summarizes the key metrics for these results. mEV exponents are illustrated graphically in Fig. 8 and negative P-value exponents are shown graphically in Fig. 9. Figure 10 shows the number of significant associated anomalies (Panel A), the cumulative E-value exponents (Panel B) and the cumulative P-value negative exponents (Panel C). Figure 11 portrays the overall marginal effects as (A) sum, (B) mean and (C) median values.
Table 5:
Summary table of multivariate additive IPW panel regression results by substance
| Covariate | Number of positive terms | Sum mEV exponents | Sum P-value exponents | Mean % increment | Median % increment | Sum % increment |
|---|---|---|---|---|---|---|
| Alcohol | 16 | 0 | 35 | −11.22 | −6.37 | −179.46 |
| Herb_THC | 28 | 57 | 92 | 184.08 | 41.25 | 5154.28 |
| Amphetamines | 13 | 0 | 22 | 8.19 | 2.75 | 106.52 |
| Cocaine | 28 | 0 | 117 | −31.23 | −14.19 | −874.33 |
| LM.Cann_Resin_THC_Daily.Int | 28 | 41 | 116 | 187.10 | 97.71 | 5238.80 |
| Median_Income | 30 | 16 | 156 | 211.34 | 113.06 | 6340.35 |
| LM.Cann_Daily.Int | 37 | 0 | 77 | −16.78 | −8.99 | −620.91 |
| Tobacco | 22 | 0 | 40 | 2.09 | 0.68 | 45.91 |
Figure 8:
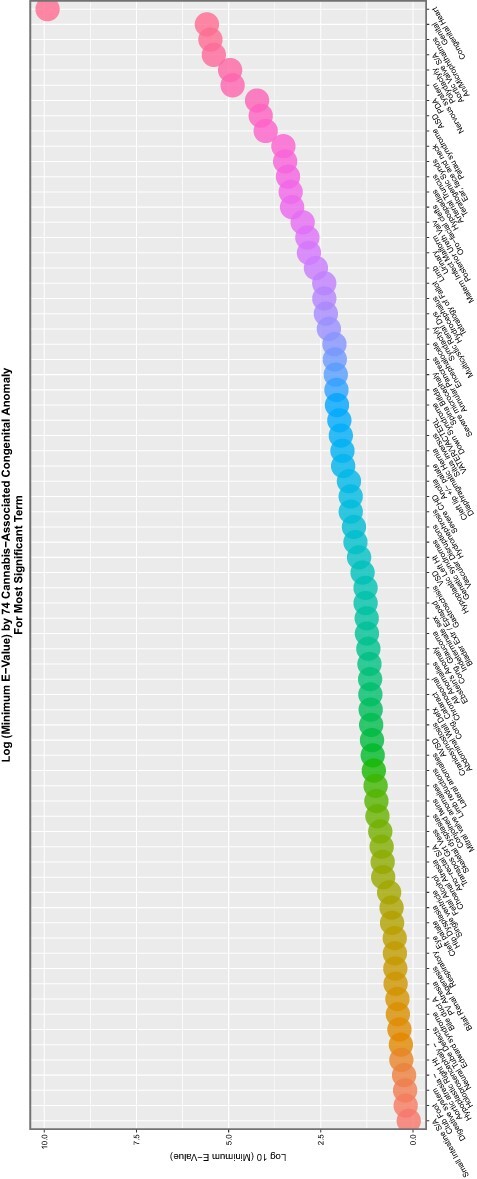
Scatterplot of log (base 10) mEV by congenital anomaly type from additive multivariable IPW panel model
Figure 9:
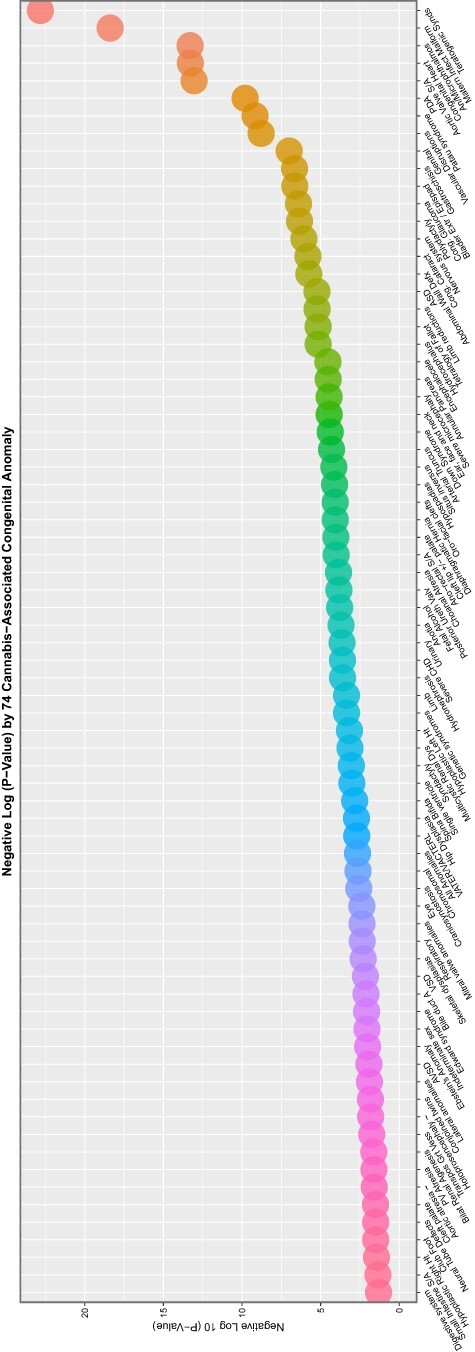
Scatterplot of negative log (base 10) P-value by congenital anomaly type from additive multivariable IPW panel model
Figure 10:
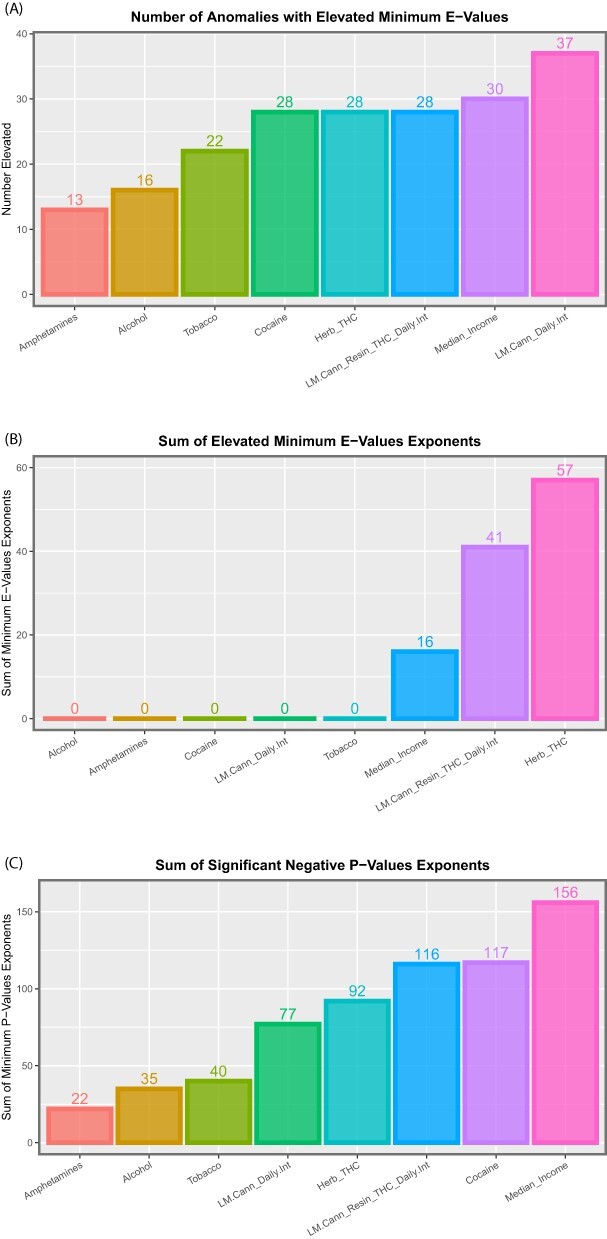
Summaries of E- and P-values by substance type for (A) number of anomalies with elevated mEV, (B) the sum of the mEV exponents and (C) the sum of the negative exponents of the significant P-values for the additive IPW multivariable panel model
Figure 11:
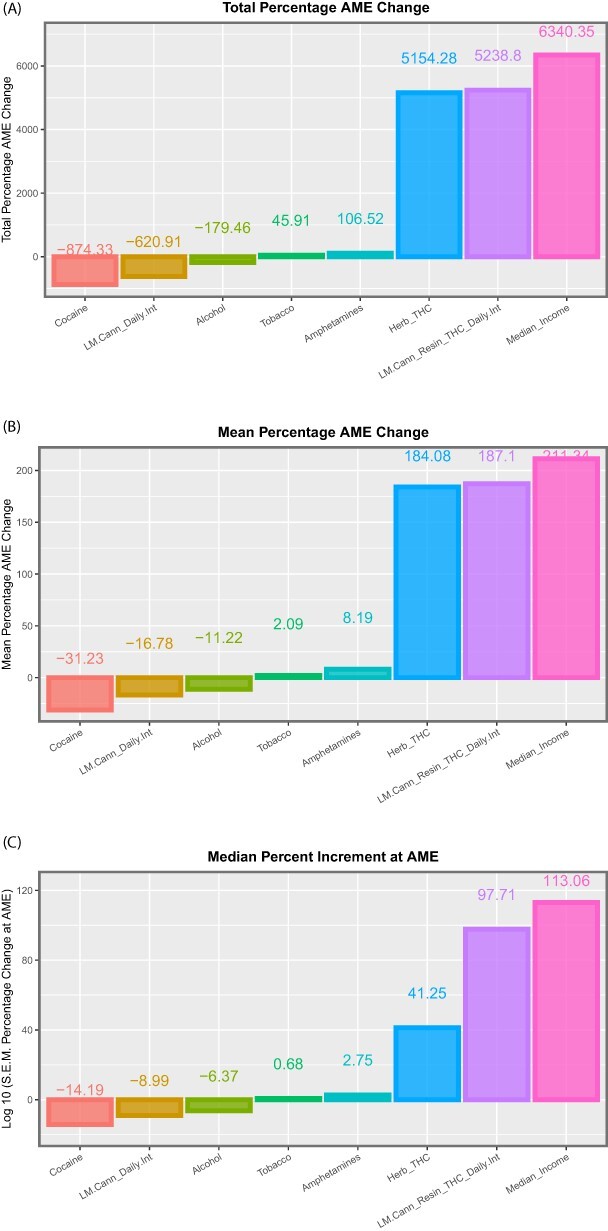
Summaries of marginal (overall) effects by substance type for (A) total percentage change at average marginal effect (AME), (B) the mean percentage change at AME and (C) the median percentage change at AME for the additive IPW multivariable panel model
Table 6 shows these data by organ system. It is noted that the table is headed by CAs affecting the face, genitalia, limbs and uronephrological systems, each of which show 100% of anomalies affected. Summary metrics relating to E- and P-values are shown in Fig. 12, and studies relating to marginal effects are illustrated in Fig. 13.
Table 6:
Summary table of multivariate additive IPW panel regression results by organ system
| System | Number positive terms | Total system count | % Anomalies in system | Sum mEV exponents | Sum P-value exponents | Sum % increment | Mean % increment | Median % increment |
|---|---|---|---|---|---|---|---|---|
| Face | 9 | 9 | 100.00 | 10 | 32 | 2607.48 | 289.72 | 133.20 |
| Genital | 2 | 2 | 100.00 | 6 | 10 | 517.44 | 258.72 | 258.72 |
| Limb | 6 | 6 | 100.00 | 9 | 21 | 186.38 | 31.06 | 29.41 |
| Uronephrology | 7 | 7 | 100.00 | 11 | 24 | 781.02 | 111.57 | 71.64 |
| Cardiovascular | 19 | 23 | 82.61 | 36 | 85 | 3004.32 | 158.12 | 87.11 |
| Central nervous | 9 | 11 | 81.82 | 18 | 41 | 883.79 | 98.20 | 59.25 |
| General | 9 | 11 | 81.82 | 9 | 57 | 4756.37 | 528.49 | 270.57 |
| Body wall | 3 | 4 | 75.00 | 3 | 16 | 231.69 | 77.23 | 92.92 |
| Gastrointestinal | 5 | 8 | 62.50 | 2 | 12 | 2117.53 | 423.51 | 201.96 |
| Chromosomal | 4 | 7 | 57.14 | 5 | 17 | 234.61 | 58.65 | 41.41 |
| Respiratory | 1 | 2 | 50.00 | 0 | 2 | 66.39 | 66.39 | 66.39 |
Figure 12:
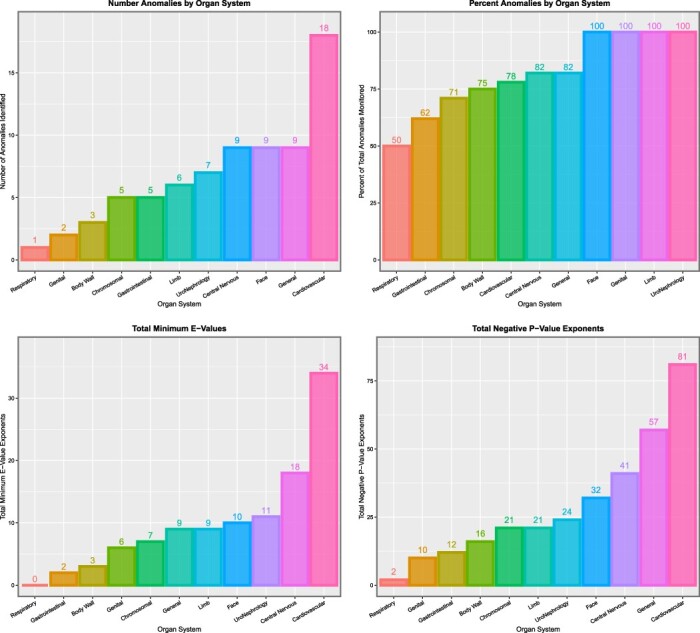
Summaries of E- and P-values by organ system. (A) Number of anomalies with elevated mEV. (B) Percentage of anomalies with elevated mEV. (C) The sum of the mEV exponents. (D) The sum of the negative exponents of the significant P-values for the additive IPW multivariable panel model
Figure 13:
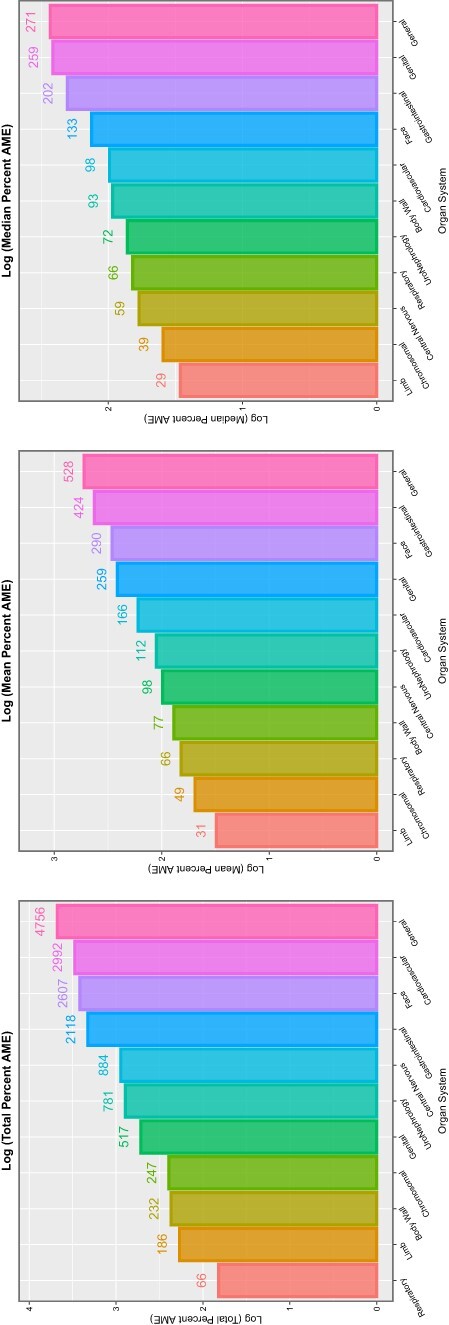
Summaries of marginal (overall) effects by organ system for (A) total percentage change at average marginal effect (AME), (B) the mean percentage change at AME and (C) the median percentage change at AME for the additive IPW multivariable panel model
This exercise can be repeated for the three cannabis metrics providing complete coverage of the 89 anomalies shown in bivariate analysis to be cannabis related. As shown in eTables 35–41 and eFigs 28–33, this procedure selects out 69 CAs and finds that last month cannabis use × daily cannabis use interpolated is by far the most powerful predictive covariate in this set.
Interactive Panel Models
Zero Temporal Lags
An IPW interactive model, including a three-way interaction between tobacco, herb THC concentration and LMC_Resin_Daily, was examined. Two hundred sixty-seven significant positive terms were returned from this procedure (eTable 44), of which 155 included cannabis-related terms (eTable 45, ordered by anomaly). These are ordered by P-value (eTable 45), by mEV (eTable 46) and by marginal effect size (eTable 47). Finally, the most significant terms for the 76 CAs implicated are listed in the descending order of mEV from 2.87 × 1016 in eTable 48. Data are summarized by substance exposure term in eTable 49. P-value negative exponents (eFig. 34) and mEV exponents (eFig. 35) are aggregated and illustrated in eFig. 36, and summary data for marginal effects are shown in eFig. 37.
Tabular data summarizing these data by organ system are shown in eTable 50, and they are illustrated graphically in eFigs 38 and 39.
Two Temporal Lags
When all the independent variables are lagged to two years, the results shown in eTable 51 are obtained for significant positive terms in an IPW model featuring an interaction between tobacco and LMC_Resin_Daily. The results are summarized in eTable 52, which show that LMC_Resin_Daily is the most salient term by number of anomalies implicated and the sum of negative P-value exponents and mEV exponents and that herb THC concentration is the most salient factor by marginal effect size estimates. eTable 53 extracts cannabis-related terms noting duplicate entries for congenital heart disease under various cannabis-related terms and PDA. eTable 54 lists these anomalies in order, eTable 55 lists them by ascending P-values and eTable 56 lists them by descending mEV. In eTable 56, duplicate anomaly entries have been removed, leaving only the most signifcant terms. eTable 57 lists the applicable marginal effects. eFigure 40 lists these P-value negative exponents graphically, and eFig. 41 similarly lists the exponents of mEV. Summary data for numbers of anomalies and P- and E-values are shown in eFig. 42, and cannabis-related terms are noted to clearly outperform tobacco, alcohol and other substances. Similar findings are clearly shown upon consideration of marginal effects in eFig. 43.
Data are summarized by organ system in eTable 58 and presented graphically in eFigs 44 and 45. When considering E- and P-values, the cardiovascular system and CNS are the most affected (eFig. 44). When marginal effects are considered, the gastrointestinal tract is also particularly affected (eFig. 45).
Four Temporal Lags
In an IPW panel regression model featuring an interaction between LMC_Resin_Daily and LMC_Herb_Daily, the 169 positive and significant terms noted in eTable 59 are returned. Data are summarized in eTable 60. Fifty-five cannabis-related terms are extracted in eTable 61. The most significant terms for 50 unique anomalies are extracted and appear in eTable 62. P- and E-values are illustrated by anomaly in eFigs 47 and 48 and summary E- and P-values are illustrated in eFig. 48. This chart is again dominated by terms including daily cannabis exposure. Marginal effects are shown in eFig. 49. The mean and median marginal effect charts are again dominated by LMC_Herb_Daily.
These data are summarized in eTable 63 and are depicted graphically in the following eFigures. From eFig. 50, it is clear that cardiovascular and CNS anomalies dominate the picture. In considering marginal effects, it is clear that genital, face and limb anomalies dominate the total, mean and median marginal effects graphs.
Six Temporal Lags
When six temporal lags are considered in an IPW panel model again, featuring an interaction between LMC_Herb_Daily and LMC_Resin_Daily, 119 significant positive terms were returned (eTable 64). These are summarized in Table 7. Thirty cannabis-related terms may be extracted from these results (eTable 65). When the most significant of these are retained and listed in order of descending mEV, the results shown in Table 8 are revealed, which relate to 29 distinct CAs. The results in this table are remarkable for the very high mEVs noted, which decline from 3.61 × 1033 to 18.45. Genetic, cardiovascular, face and limb anomalies are notable. The table may be ordered in ascending order by P-values (eTable 66) or by marginal effects (eTable 67). Figure 14 illustrates the applicable P-value negative exponents, and Fig. 15 the mEV exponents. Summaries of the number of anomalies affected by substance, and E- and P-values are shown graphically in Fig. 16. Marginal effects are shown in Fig. 17. Here, the most efficacious terms are all related to compound daily indices of cannabis exposure, with the most salient term in each case being the interaction between LMC_Herb_Daily and LMC_Resin_Daily.
Table 7:
Summary table of multivariate interactive IPW panel regression results by substance at 6 years lag
| Covariate | Number of positive terms | Sum mEV exponents | Sum P-value exponents | Mean % increment | Median % increment | Sum % increment |
|---|---|---|---|---|---|---|
| Alcohol | 11 | 0 | 18 | −6.43 | −5.05 | −70.76 |
| Herb_THC | 12 | 59 | 21 | 197.78 | 107.17 | 2373.35 |
| Amphetamines | 7 | 0 | 8 | −15.53 | −6.75 | −108.74 |
| Cocaine | 12 | 0 | 15 | −6.05 | −4.91 | −72.63 |
| LM.Cann_Herb_THC_Daily.Int | 10 | 69 | 22 | 469.61 | 200.11 | 4696.14 |
| LM.Cann_Resin_THC_Daily.Int | 10 | 21 | 16 | 10.31 | 4.58 | 103.05 |
| LM.Cann_Herb_THC_Daily.Int x LM.Cann_Resin_THC_Daily.Int | 8 | 115 | 14 | 1103.35 | 539.30 | 8826.77 |
| Median_Income | 27 | 0 | 50 | 0.00 | 0.00 | 0.04 |
| Tobacco | 22 | 0 | 67 | 4.3718 | 2.2772 | 96.1797 |
Table 8:
Multivariate interactive IPW panel regression results by substance at 6 years lag for most significant anomaly term
| Anomaly | Term | Mean Anomaly Rate | ß-Estimate | Standard error | Sigma | T-statistic | Adj. R2 | P-value | E-value estimate | E-value (lower bound) | P-value exponent | Lower E-value exponent | % Increment | P—Bonferroni | P—false discovery rate | P—Holm |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ear, face and neck | lag(LpmResinDailyInt, 6):lag(LpmHerbDailyInt, 6) | 2.4050 | 124.8613 | 31.4481 | 0.7528 | 3.9704 | 0.2145 | 0.0005 | 7.09E + 65 | 3.61E + 33 | 3 | 33 | 198.54 | 0.0139 | 0.0028 | 0.0120 |
| Hip dysplasia | lag(LpmResinDailyInt, 6):lag(LpmHerbDailyInt, 6) | 6.3491 | 107.5064 | 34.7070 | 0.8308 | 3.0975 | −0.3248 | 0.0045 | 2.76E + 51 | 1.40E + 19 | 2 | 19 | 75.21 | 0.1310 | 0.0131 | 0.0908 |
| Hirschsprungs | lag(LpmHerbDailyInt, 6) | 0.9900 | 21.0139 | 4.0517 | 0.2960 | 5.1864 | 0.5242 | 0.0000 | 2.30E + 28 | 6.01E + 17 | 4 | 17 | 457.40 | 0.0005 | 0.0005 | 0.0005 |
| VATER/VACTERL | lag(LpmResinDailyInt, 6):lag(LpmHerbDailyInt, 6) | 0.4798 | 66.1544 | 22.7293 | 0.5441 | 2.9105 | −0.3275 | 0.0071 | 2.25E + 48 | 1.15E + 16 | 2 | 16 | 995.22 | 0.2072 | 0.0139 | 0.1260 |
| PDA | lag(LpmHerbDailyInt, 6) | 2.9086 | 59.4896 | 12.9845 | 0.9484 | 4.5816 | 0.0630 | 0.0001 | 1.23E + 25 | 3.21E + 14 | 4 | 14 | 155.69 | 0.0027 | 0.0009 | 0.0025 |
| Anophthalmos | lag(LpmResinDailyInt, 6):lag(LpmHerbDailyInt, 6) | 0.2059 | 22.2169 | 7.9565 | 0.1905 | 2.7923 | −0.1420 | 0.0095 | 2.52E + 46 | 1.28E + 14 | 2 | 14 | 2319.08 | 0.2754 | 0.0155 | 0.1274 |
| Conjoined twins | lag(LpmResinDailyInt, 6):lag(LpmHerbDailyInt, 6) | 0.1128 | 20.6507 | 7.4064 | 0.1773 | 2.7882 | −0.2137 | 0.0096 | 2.16E + 46 | 1.10E + 14 | 2 | 14 | 4233.14 | 0.2781 | 0.0155 | 0.1274 |
| Congenital heart | lag(Herb, 6) | 82.6649 | 10.2501 | 2.2265 | 0.1814 | 4.6037 | 0.4037 | 0.0001 | 4.35E + 22 | 1.40E + 13 | 4 | 13 | 5.48 | 0.0026 | 0.0009 | 0.0025 |
| Single ventricle | lag(Herb, 6) | 0.7525 | 24.7634 | 5.8151 | 0.4736 | 4.2585 | 0.1922 | 0.0002 | 9.19E + 20 | 2.97E + 11 | 3 | 11 | 601.77 | 0.0065 | 0.0016 | 0.0058 |
| Skeletal dysplasias | lag(LpmHerbDailyInt, 6) | 1.8518 | 30.4938 | 8.0752 | 0.5898 | 3.7762 | −0.0234 | 0.0008 | 5.40E + 20 | 1.41E + 10 | 3 | 10 | 244.54 | 0.0231 | 0.0039 | 0.0191 |
| Syndactyly | lag(LpmResinDailyInt, 6):lag(LpmHerbDailyInt, 6) | 4.2302 | 49.3778 | 19.3916 | 0.4642 | 2.5464 | −0.3173 | 0.0169 | 2.19E + 42 | 1.11E + 10 | 1 | 10 | 112.88 | 0.4902 | 0.0213 | 0.1403 |
| Tot anom pul V ret | lag(Herb, 6) | 0.6242 | 21.1817 | 5.7989 | 0.4723 | 3.6527 | 0.2061 | 0.0011 | 1.06E + 18 | 3.41E + 08 | 2 | 8 | 725.46 | 0.0319 | 0.0046 | 0.0253 |
| Coarctation aorta | lag(Herb, 6) | 3.3779 | 23.2510 | 7.0103 | 0.5710 | 3.3167 | 0.3213 | 0.0026 | 2.48E + 16 | 8.00E + 06 | 2 | 6 | 134.06 | 0.0756 | 0.0095 | 0.0574 |
| Limb | lag(LpmResinDailyInt, 6):lag(LpmHerbDailyInt, 6) | 37.7465 | 42.1716 | 18.0460 | 0.4320 | 2.3369 | −0.3162 | 0.0271 | 7.63E + 38 | 3.88E + 06 | 1 | 6 | 12.65 | 0.7862 | 0.0302 | 0.1403 |
| Anencephalus and ∼ | lag(LpmHerbDailyInt, 6) | 3.5488 | 14.5166 | 4.6598 | 0.3404 | 3.1153 | 0.6029 | 0.0043 | 1.43E + 17 | 3.74E + 06 | 2 | 6 | 127.60 | 0.1254 | 0.0131 | 0.0908 |
| Hydrocephalus | lag(LpmHerbDailyInt, 6) | 5.3737 | 15.9980 | 5.4132 | 0.3954 | 2.9554 | 0.3363 | 0.0064 | 1.95E + 16 | 5.10E + 05 | 2 | 5 | 84.27 | 0.1858 | 0.0139 | 0.1218 |
| Cong. glaucoma | lag(LpmHerbDailyInt, 6) | 0.2853 | 21.4190 | 7.3379 | 0.5360 | 2.9189 | −0.0901 | 0.0070 | 1.24E + 16 | 3.24E + 05 | 2 | 5 | 1587.21 | 0.2031 | 0.0139 | 0.1260 |
| Down syndrome | lag(LpmHerbDailyInt, 6) | 20.7996 | 15.6869 | 5.3918 | 0.3938 | 2.9094 | 0.0850 | 0.0072 | 1.10E + 16 | 2.88E + 05 | 2 | 5 | 21.77 | 0.2078 | 0.0139 | 0.1260 |
| VSD | lag(Herb, 6) | 37.2628 | 8.3593 | 2.8717 | 0.2339 | 2.9109 | 0.2060 | 0.0071 | 2.66E + 14 | 8.60E + 04 | 2 | 4 | 12.15 | 0.2070 | 0.0139 | 0.1260 |
| Edward syndrome | lag(Herb, 6) | 4.9591 | 22.3653 | 7.9587 | 0.6483 | 2.8102 | 0.1626 | 0.0091 | 8.63E + 13 | 2.79E + 04 | 2 | 4 | 91.31 | 0.2639 | 0.0155 | 0.1274 |
| ASD | lag(Herb, 6) | 21.4739 | 20.3234 | 7.4426 | 0.6062 | 2.7307 | −0.1801 | 0.0110 | 3.55E + 13 | 1.15E + 04 | 1 | 4 | 21.09 | 0.3189 | 0.0168 | 0.1274 |
| Patau syndrome | lag(LpmHerbDailyInt, 6) | 1.7507 | 20.3273 | 7.7899 | 0.5690 | 2.6094 | 0.2459 | 0.0146 | 2.63E + 14 | 6.86E + 03 | 1 | 3 | 258.66 | 0.4237 | 0.0202 | 0.1403 |
| Eye | lag(Herb, 6) | 3.6805 | 20.1616 | 7.6754 | 0.6252 | 2.6268 | 0.1091 | 0.0140 | 1.11E + 13 | 3.59E + 03 | 1 | 3 | 123.03 | 0.4069 | 0.0202 | 0.1403 |
| Foetal alcohol | lag(LpmHerbDailyInt, 6) | 0.2577 | 7.1157 | 2.7893 | 0.2037 | 2.5511 | 0.1258 | 0.0167 | 1.27E + 14 | 3.31E + 03 | 1 | 3 | 1757.20 | 0.4849 | 0.0213 | 0.1403 |
| Aortic atresia ∼ | lag(LpmResinDailyInt, 6):lag(LpmHerbDailyInt, 6) | 0.5426 | 32.6821 | 15.3377 | 0.3672 | 2.1308 | −0.1736 | 0.0424 | 3.02E + 35 | 1.54E + 03 | 1 | 3 | 880.06 | 1.0000 | 0.0424 | 0.1403 |
| Respiratory | lag(Herb, 6) | 3.2843 | 18.7230 | 7.6259 | 0.6211 | 2.4552 | 0.4014 | 0.0208 | 1.64E + 12 | 527.8115 | 1 | 2 | 137.88 | 0.6034 | 0.0251 | 0.1403 |
| Digestive system | lag(Herb, 6) | 17.0099 | 6.9501 | 2.9249 | 0.2382 | 2.3762 | 0.2960 | 0.0248 | 6.76E + 11 | 217.9977 | 1 | 2 | 26.62 | 0.7206 | 0.0288 | 0.1403 |
| All anomalies | lag(Herb, 6) | 249.8954 | 4.5283 | 2.0545 | 0.1673 | 2.2041 | 0.5142 | 0.0362 | 9.90E + 10 | 31.4602 | 1 | 1 | 1.81 | 1.0000 | 0.0389 | 0.1403 |
| Choanal atresia | lag(Herb, 6) | 0.9191 | 18.4092 | 8.5330 | 0.6950 | 2.1574 | −0.0434 | 0.0400 | 5.87E + 10 | 18.4553 | 1 | 1 | 492.69 | 1.0000 | 0.0415 | 0.1403 |
Figure 14:
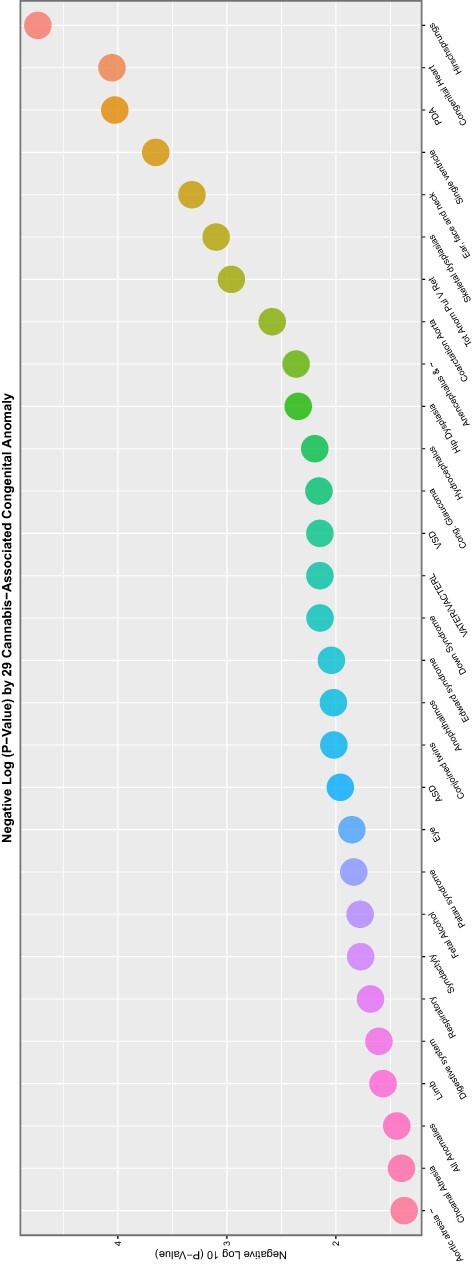
Scatterplot of negative log (base 10) P-value by congenital anomaly type from interactive multivariable IPW panel model temporally lagged by six years
Figure 15:
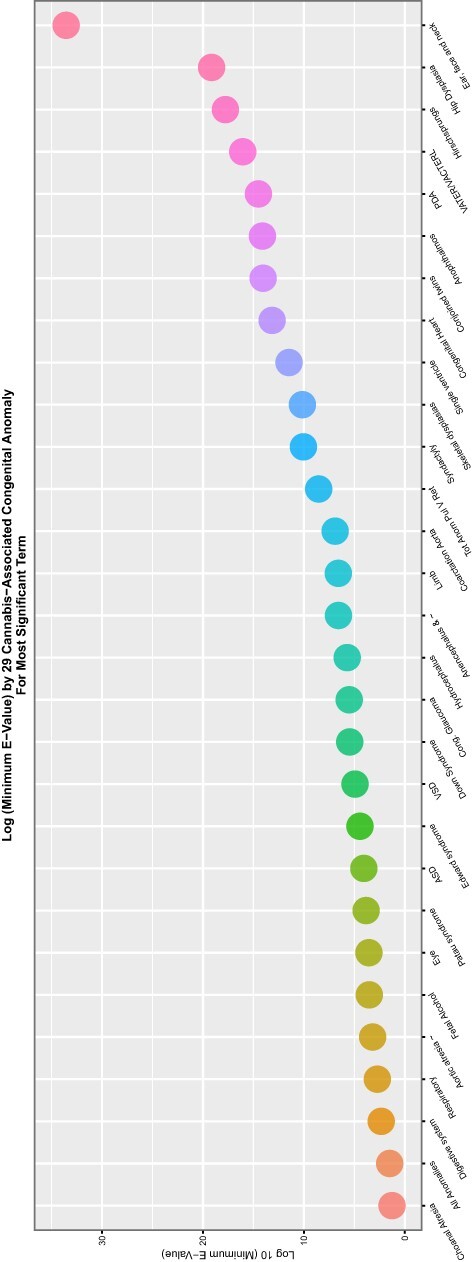
Scatterplot of log (base 10) mEV by congenital anomaly type from interactive multivariable IPW panel model temporally lagged by six years
Figure 16:
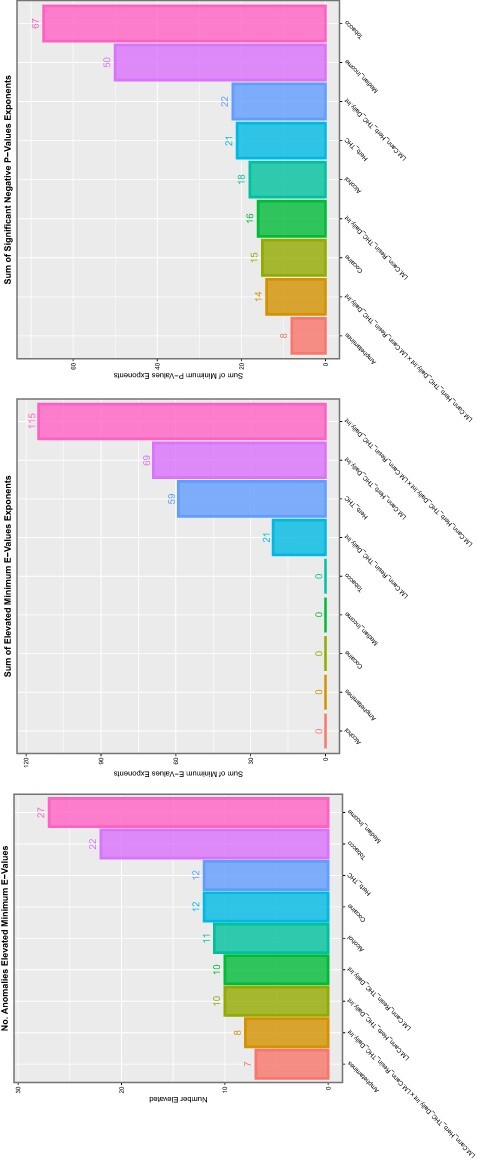
Summaries of E- and P-values by substance type for (A) number of anomalies with elevated mEV, (B) the sum of the mEV exponents and (C) the sum of the negative exponents of the significant P-values for the interactive multivariable IPW panel model temporally lagged by six years
Figure 17:
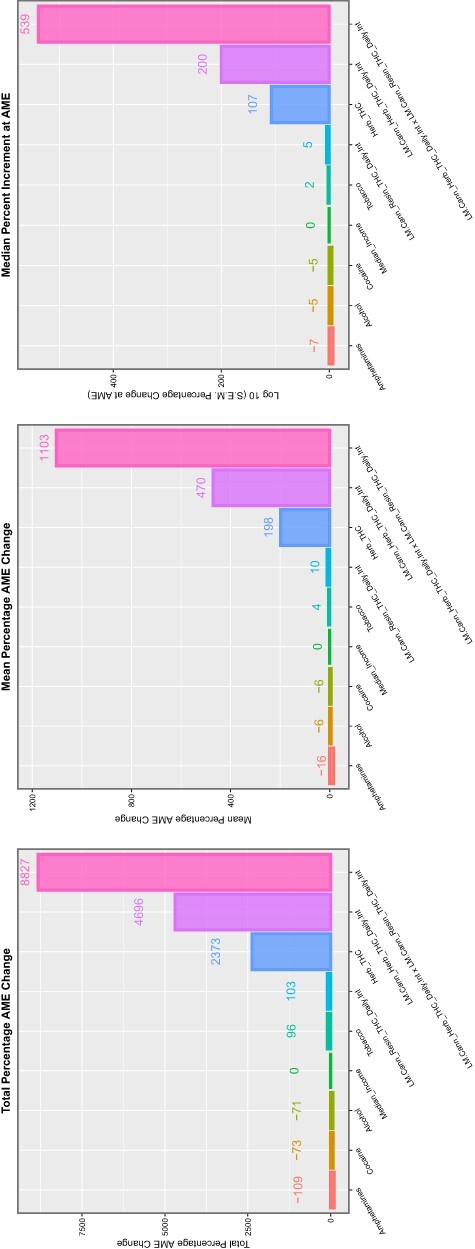
Summaries of marginal (overall) effects by substance type for (A) total percentage change at average marginal effect (AME), (B) the mean percentage change at AME and (C) the median percentage change at AME for the interactive multivariable IPW panel model temporally lagged by six years
Table 9 summarizes the organ systems implicated. Results are illustrated graphically in Fig. 18. Cardiovascular, general, limb and respiratory systems are all prominently represented. General, face and CNS effects are all prominently represented on the marginal effects summary shown in Fig. 19.
Table 9:
Summary table of multivariate interactive IPW panel regression results by organ system at 6 years lag
| System | Number positive terms | System count | % Anomalies by system | Sum mEV exponents | Sum P-value exponents | Sum % increments | Mean % increments | Median % increments |
|---|---|---|---|---|---|---|---|---|
| Limb | 3 | 6 | 50 | 35 | 4 | 200.74 | 66.91 | 75.21 |
| Respiratory | 1 | 2 | 50 | 2 | 1 | 137.88 | 137.88 | 137.88 |
| General | 5 | 11 | 45.45 | 44 | 9 | 7231.90 | 1446.38 | 995.22 |
| Cardiovascular | 9 | 23 | 39.13 | 68 | 21 | 2557.51 | 284.17 | 134.06 |
| Central nervous | 4 | 11 | 36.36 | 28 | 7 | 2653.98 | 663.50 | 125.32 |
| Face | 3 | 9 | 33.33 | 39 | 6 | 2278.44 | 759.48 | 492.69 |
| Chromosomal | 2 | 7 | 28.57 | 7 | 3 | 349.97 | 174.98 | 174.98 |
| Gastrointestinal | 2 | 8 | 25 | 19 | 5 | 484.03 | 242.01 | 242.01 |
Figure 18:
Summaries of E- and P-values by organ system. (A) Number of anomalies with elevated mEV. (B) Percentage of anomalies with elevated mEV. (C) The sum of the mEV exponents. (D) The sum of the negative exponents of the significant P-values for the interactive multivariable IPW panel model temporally lagged by six years
Figure 19:
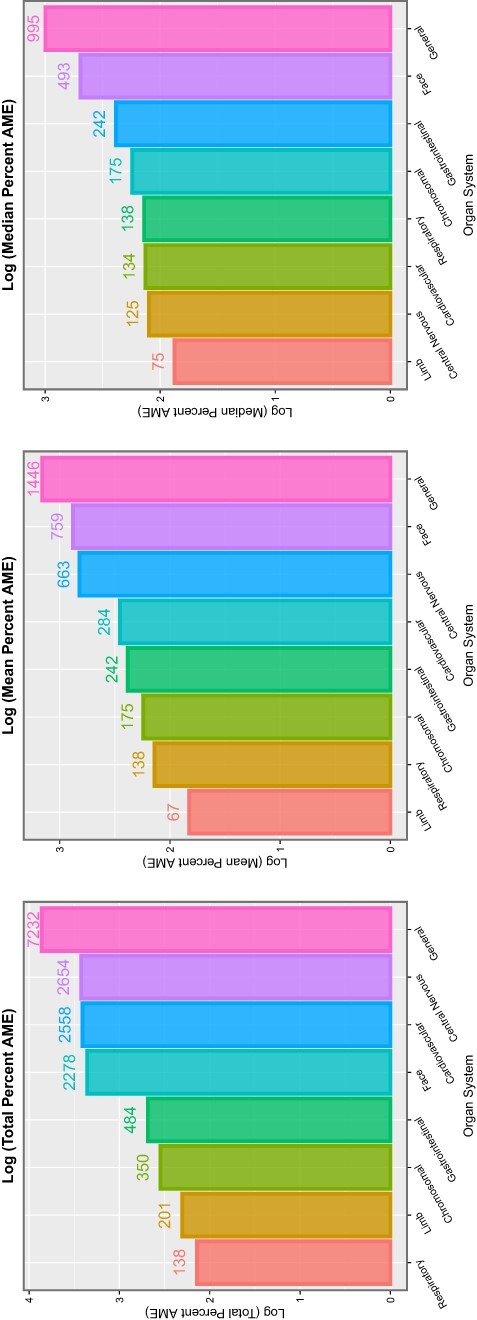
Summaries of marginal (overall) effects by organ system for (A) total percentage change at average marginal effect (AME), (B) the mean percentage change at AME and (C) the median percentage change at AME for the interactive multivariable IPW panel model temporally lagged by six years
Discussion
Main Results
By using available European national datasets on congenital birth anomalies against various metrics of cannabis or other drug exposure, sophisticated analysis was able to associate all 90 tracked congenital birth anomalies with various metrics of cannabis exposure or combinations thereof. This is true both for birth defects considered in aggregate across the spectrum (Fig. 1 and accompanying analysis) and for CAs considered individually and by organ system. It was also shown convincingly both by random forest regression and by multivariable panel regression that compound indices of daily cannabis use were generally the most powerfully predictive covariates, confirming earlier concerns that it is the convergence of metrics of high cannabinoid exposure that most places infants and populations at genotoxic risk [15, 16].
Since all multivariable regression models were IPW, a pseudo-randomized analytical paradigm was created from which causal inferences could properly be drawn. Formal processes of causal inference were reinforced by the widespread use of E-values from both categorical two-by-two tables and linear and panel models.
Considered in aggregate whole, the CA rate was 71.8% higher in countries with high or increasing daily cannabis use, a result that was shown to be significant at the P = 4.74 × 10−17 level and was associated with an mEV of 1.52, which exceeds the epidemiological threshold for causality [81].
At bivariate analysis, 89 of 90 CAs were shown to be linked with the metrics of cannabinoid exposure, the sole exception being indeterminate sex. However, this anomaly was found to be cannabis related at multivariable panel regression in additive models and in interactive models lagged to zero and four years (Table 4 and eTables 34–35, 43–48, 59–62). For the series of substances tobacco, alcohol, amphetamines and cocaine, 3, 12, 23 and 68 anomalies had elevated mEV, respectively. For the metrics last month cannabis use, cannabis herb THC, cannabis resin THC, daily cannabis use interpolated, LMC_Herb_Daily and LMC_Resin_Daily, 23, 45, 34, 41, 41 and 42 mEVs were elevated.
Categorical Metrics
At categorical analysis, 84/90 CAs demonstrated elevated highest:lowest quintile ratios for LMC_Resin_Daily and 100% of CAs in the uronephrology, respiratory, limb, general, chromosomal and body wall classes were implicated. Eighty-two and 83 CAs had elevated mEVs for parameters including LMC_Resin_Daily and LMC_Herb_Daily, respectively (eTable 30 and eFig. 27).
The cannabinoid parameter with the highest variable importance on random forest and panel regression was LMC_Herb_Daily. Results for categorization on this covariate are shown in Table 2. It is of interest that the table is headed by VACTERL syndrome with highest to lowest quintile RR of 54.56 (17.55, 169.57), AFE 98.17% (94.30%, 99.41%) and PAR 97.76% (93.08%, 99.28%), P = 2.43 × 10−36 and mEV = 34.61.
All anomalies are shown in this table as having RR of 1.51 (1.49, 1.52), AFE 33.59% (33.04%, 34.14%) and PAR 28.47% (27.96%, 28.97%), P < 2.2 × 10−307 and mEV = 2.35.
The CNS anomalies severe microcephaly, nervous system, anencephalus and similar hydrocephalus, neural tube defects, spina bifida and encephalocele were associated with AFEs of 72.34% (69.31%, 75.08%), 65.71% (64.48%, 66.89%), 64.47% (641.11%, 67.54%), 52.60% (49.31%, 55.68%), 52.60% (49.31%, 55.68%), 45.02% (42.30%, 47.62%), 31.21% (26.58%, 35.54%) and 29.66% (20.37%, 37.87%) and mEVs of 5.97, 5.08, 4.58, 3.36, 2.86, 2.06 and 1.82, respectively.
Facial anomalies have been shown to be developmentally related to abnormalities of CNS development since the head and the brain form due to the facial organizer and the forebrain organizer, which have interrelated and interconnected form and functions developmentally [57, 82]. Congenital glaucoma, holoprosencephaly, anophthalmos/microphthalmos, choanal atresia, congenital cataract, eye anomalies, orofacial clefts, ear, face and neck anomalies, cleft lip ± cleft palate and cleft palate were associated with AFEs of 74.70% (66.58%), 64.51% (59.83%, 68.65%), 62.34% (55.67%, 68.01%), 61.21% (54.11%, 67.22%), 56.64% (50.68%, 61.88%), 26.88% (22.46%,m 31.05%), 18.18% (14.80%, 21.43%), 18.09% (11.16%, 24.49%), 11.43% (7.03%,15.62%) and 12.06% (6.64%, 17.76%) and mEVs of 5.43, 4.42, 3.94, 3.78, 3.47, 1.90, 1.63, 1.50, 1.36 and 1.35, respectively.
The cardiovascular anomalies mitral valve anomalies, double outlet right ventricle, congenital heart disorders, pulmonary valve atresia, aortic atresia and similar, vascular disruptions, tricuspid valve stenosis or atresia, hypoplastic left heart, atrioventricular septal defect (AVSD), hypoplastic right heart, tetralogy of Fallot, severe congenital heart disease, total anomalous pulmonary venous return, transposition of the great vessels, arterial truncus, single ventricle, PDA, Ebstein’s anomaly, pulmonary valve stenosis, ventricular septal defect (VSD) and coarctation of the aorta were associated with AFEs of 75.10% (70.6%, 79.30%), 73.66% (67.93%, 78.37%), 52.12% (51.24%, 52.99%), 48.31% (40.13%, 55.37%), 47.35% (34.65%, 57.58%), 47.09% (43.54%, 50.42%), 46.67% (35.57%, 55.86%), 46.29% (40.93%, 51.17%), 46.12% (41.96%, 49.98%), 41.19% (29.80%, 503.74%), 38.30% (33.25%, 42.98%), 37.70% (35.76%, 39.59%), 36.46% (22.49%, 47.91%), 35.82% (30.58%, 40.66%), 34.19% (21.70%, 44.68%), 32.94% (21.77%, 42.52%), 32.46% (26.52%, 37.92%), 31.80% (15.12%, 45.12%), 29.62% (24.86%, 34.08%), 20.43% (18.83%, 21.99%) and 14.87% (8.37%, 20.90%) and mEVs of 6.13, 5.69, 3.52, 2.73, 2.43, 2.94, 2.48, 2.78, 2.84, 2.20, 2.36, 2.49, 1.90, 2.24, 1.87, 1.87, 2.06, 1.64, 1.99, 1.77, and 1.41, respectively.
The limb anomalies hip dysplasia, limb anomalies, skeletal dysplasia, polydactyly and limb reductions were associated with AFEs of 86.26% (84.74%, 87.63%), 56.22% (55.05%, 57.35%), 57.31% (52.10%, 61.96%), 39.56% (37.14%, 41.89%) and 32.31% (28.39%, 36.02%) and mEVs of 12.59, 3.83, 3.59, 2.56 and 2.14, respectively.
The genetic syndromes Trisomy 18 (Edwards syndrome), Turner syndrome (monosomy X), genetic syndromes, Trisomy 13 (Patau syndrome), Klinefelter (male disomy X), chromosomal anomalies and Trisomy 21 (Downs syndrome) were noted to have AFEs of 74.26% (72.17%, 76.19%), 71.48% (67.65%, 74.86%), 67.35% (64.92%, 69.60%), 68.03% (64.05%, 71.57%), 69.78% (62.10%, 75.91%), 6.83% (61.85%, 63.78%) and 56.84% (55.42%, 58.23%) and mEVs of 6.65, 5.63, 5.15, 5.01, 4.72, 4.68 and 3.91, respectively.
Considering LMC_Resin_Daily and comparing the highest and lowest quintiles for all anomalies, genetic disorders, central nervous, cardiovascular, facial, limb and VACTERL disorders, the following notable observations were reported (Table 3). For the sequence of disorders all anomalies, teratogenic syndromes, Trisomy 18, Trisomy 13, genetic syndromes, Turners syndrome, Trisomy 21 and chromosomal disorders, the applicable P-values were <2.2 × 10−307, 3.69 × 10−79, 6.31 × 10−252, 1.50 × 10−93, 7.42 × 10−227, 8.52 × 10−58, 9.84 × 10−318 and <2.23 × 10−307, with associated mEVs of 2.49, 5.55, 4.46, 4.29, 4.01, 3.18, 2.61 and 3.18, respectively. For the CNS disorders severe microcephaly and anencephalus and similar, the P-values were 9.68 × 10−101 and 1.57 × 10−101 and the mEVs were 3.57 and 3.44, respectively. For the cardiovascular disorders, congenital heart disease, severe congenital heart disease, VSD, AVSD, vascular disruptions, aortic atresia and similar, and tetralogy of Fallot and Ebstein’s anomaly, the applicable P-values were <2.2 × 10−307, 1.79 × 10−232, <2.2 × 10−307, 1.65 × 10−101, 6.50 × 10−65, 2.45 × 10−45, 1.54 × 10−34 and 3.23 × 10−17, respectively, and the relevant mEVs were 2.08, 2.46, 2.74, 3.38, 2.34, 2.94, 2.25 and 4.09, respectively. For the facial anomalies holoprosencephaly and orofacial clefts, the applicable P-values were 6.11 × 10−44 and 6.07 × 10−51, respectively, and the applicable mEVs were 2.90 and 1.86, respectively. For the limb anomalies limb anomalies and limb reduction anomalies, the P-values were <2.3 × 10−307 and 1.94 × 10−54, respectively, and the relevant mEVs were 3.49 and 2.25, respectively. For VACTERL, the relevant P-value was 1.45 × 10−32 and mEV was 6.00.
Panel Regression
At additive IPW panel regression, 74 CAs were shown to be cannabis related. The introduction of interactive terms in the regression had the effect of increasing this number somewhat to 76 CAs. The number of implicated CAs dropped to 31, 50 and 29 CAs in interactive panel models lagged by two, four and six years.
In additive panel models, the most affected organ systems were uronephrology, face, central nervous body wall and cardiovascular systems, but 50% or more of CAs in all organ systems studied had elevated mEVs. Chromosomal, face, central nervous and cardiovascular systems had highest cumulative mEVs.
This pattern persisted with the introduction of interactive terms and temporal lagging. At six years of temporal lag in an interactive IPW panel, model body wall, genital, CNS and cardiovascular system had the highest percentage of CAs implicated. Cardiovascular, uronephrology, central nervous and general classes had the highest cumulative mEVs.
Interpretation
Several features stand out as major implications from the substantial body of evidence presented, namely the strength of reported effects (documented in the tables); the breadth of reported affects comprehensively across the spectrum of CAs and anomaly classes; the consistency with in vitro mechanistic studies (discussed below); the consistency with animal studies [83–85]; the consistency with results reported from elsewhere [4–9, 18, 55, 86–88]; concordance with recent cancer analyses [17, 18, 20, 89] and the implications of presumptive cannabinoid genotoxicity for cellular ageing community-wide [19].
A critical concern is that increasing European cannabis exposure is associated with a broad spectrum of CAs, in fact extending to the totality of anomalies reported by European data. This finding is compounded when one notes that cannabis exposure has also been linked with breast, thyroid, liver and pancreatic cancer and acute lymphoid leukaemia [17] and acute myeloid leukaemia of childhood [18], which latter constitute heritable mutagenic and teratogenic malignant syndromes.
However, cancer and CAs are relatively rare disorders compared to the widespread availability in the community of a known genotoxin, including allowing its entry into the food chain. Moreover, as our results clearly indicate that it is the convergence of increased cannabis prevalence, intensity and concentration of exposure driving the expected total dose–response relationships, which is the leading risk factor for heritable genotoxic outcomes, it is likely that these changes will leverage multiplicatively off each other in populations where policies favouring cannabis liberalization are in play.
Since cannabis is a known major epigenomic toxin [31–37, 90] and since DNA methylation has been shown to be a direct cause of mammalian ageing [19], it becomes difficult to avoid the conclusion that increasing cannabis use will actually increase the epigenomic age of widely exposed populations.
Furthermore, since many epigenomic effects are known to be heritable to several subsequent generations, this implies the multigenerational transmission of these changes in addition to described diagnosable genotoxic [18] and/or neurotoxic [91–94] outcomes. It also includes the disconcerting possibilities that babies will be born ‘old’ with advanced epigenomic ages and may continue to age in an accelerated manner as has been demonstrated in clinical populations [95] or develop abnormally due to large-scale epigenomic dysregulation.
Commentary on Specific Defects by Systems
Total Anomalies
All anomalies were significantly elevated on bivariate continuous (P = 0.0006, mEV = 8.41, eTable 12) and categorical [RR = 1.57 (1.56, 1.58), P < 2.2 × 10−307 and mEV = 2.49; Table 3] analyses. All anomalies were significantly associated with cannabis exposure on additive IPW panel regression (P = 0.0022, mEV = 451.56.41; Table 4) and in interactive IPW panel models lagged to zero (P = 0.0067, mEV = 7.60 × 1012; eTable 46), four (P = 4.20 × 10−6, mEV = 1.11 × 104; eTable 62) and six (P = 0.0362, mEV = 31.46; Table 8) years.
This important finding has been replicated in Colorado and Canada [5, 7]. No comparable metric exists for this datapoint in USA datasets generally.
Cardiovascular System
Cardiovascular disorders are widely acknowledged to be the commonest CAs. Cardiovascular disorders associated with metrics of cannabis use on bivariate analysis were: aortic atresia and similar, aortic valve stenosis or atresia, arterial truncus, ASD, AVSD, coarctation of aorta, congenital heart disease, double outlet right ventricle, Ebstein’s anomaly, hypoplastic left heart syndrome, hypoplastic right heart syndrome, mitral valve anomalies, PDA, pulmonary valve atresia, pulmonary valve stenosis, severe congenital heart disease, single ventricle, tetralogy of Fallot, total anomalous pulmonary venous return, transposition of the great vessels, tricuspid valve stenosis or atresia, vascular disruptions and VSD.
It is thus of interest that total cardiovascular disorders were noted to also be increased in association with cannabis use in the northern Canadian Provinces and in the US state of Colorado [5, 7]. Similarly, it is important to note that ASD was noted to occur with elevated rates in the US states of Hawaii and Colorado, across the USA and in Australia, (and presumably also in Canada as it is the commonest cardiovascular anomaly) [4–7, 55]; VSD was noted to be increased in Australia, USA Colorado, Hawaii and in other series [4–7, 55, 86]; PDA was noted to be increased in Colorado, Australia and USA [5, 8, 55]; tetralogy of Fallot was increased in Hawaii, Australia and USA [4, 8, 55, 96]; AVSD was noted to be elevated also in USA (manuscript submitted) and pulmonary valve atresia and stenosis was associated in USA [55].
Chromosomal Disorders
The chromosomal disorders identified in this study as being linked to indices of cannabis exposure were: Chromosomal, Downs syndrome (Trisomy 21), Edward syndrome (Trisomy 18), Genetic syndromes, Klinefelter (Male XXY), Patau syndrome (Trisomy 13) and Turner syndrome (Female XO). It is of considerable importance that elevated rates of Downs syndrome have previously been identified in association with cannabis exposure in Colorado [5], Australia [8], Canada [7], Hawaii [4] and USA [11, 55]; of Edwards syndrome in USA (manuscript submitted); of Patau syndrome in USA [18, 55]; of Turner syndrome in USA [18, 55]; of Turner syndrome in Australia [8] and of chromosomal anomalies in USA, Canada and Australia [7, 8, 18, 55].
In this context, it becomes important to observe that cannabis use has also been linked with testicular cancer in several studies [97–102] and with acute lymphoid leukaemia [17, 18, 20]. These disorders invariably or generally involve major rearrangements, translocations or deletions of chromosomes 12 and 19, respectively [100, 102–111].
If one aggregates all of these chromosomal disorders together, noting that chromosomes 12, 13, 18, 9, 21 and X are of 133, 114, 80, 57, 48 and 153 MB in length, it becomes clear that this provides clinical evidence of direct cannabinoid genotoxicity to 585 MB of the 3000 MB of the human DNA or 19.5%, which is clearly a not inconsiderable fraction of the human genome.
Central Nervous System
The disorders of the CNS that were noted to be elevated on bivariate analyses were: anophthalmos/microphthalmos, anencephalus and similar, anophthalmos, craniosynostosis, encephalocele, eye, hydrocephalus, nervous system, neural tube defects, severe microcephaly and spina bifida. This is consistent with published data. The neural tube defects anencephaly, spina bifida and encephalocele were previously noted to be elevated in Canada after cannabis exposure. Hydrocephaly and microcephaly were previously identified in the Hawaiian series [4]. Hydrocephalus and microcephalus were positively identified with prenatal cannabis exposure in USA [55]. Microcephalus was also associated with cannabis use in Colorado [5].
Face
The facial anomalies that were positively associated on bivariate testing with metrics of cannabis exposure were: anotia, choanal atresia, cleft lip ± palate, cleft palate, congenital cataract, congenital glaucoma, ear, face and neck, holoprosencephaly and similar and orofacial clefts.
Cleft lip with and without cleft palate and cleft palate were also associated with cannabis exposure in USA [55]. Holoprosencephaly was also likely cannabis related in USA [55]. Choanal atresia was positively associated with cannabis use in Hawaii and USA [4, 55].
Gastrointestinal Disorders
The gastrointestinal disorders that were identified in bivariate analyses as being cannabis related were: annular pancreas, anorectal stenosis or atresia, bile duct atresia, digestive system disorders, duodenal stenosis or atresia, Hirschsprungs disease, oesophageal stenosis or atresia and small intestine stenosis or atresia. Of this list, gastrointestinal disorders, biliary atresia and small bowel stenosis or atresia were strongly identified in the USA datasets [55]. Pyloric stenosis and atresia and large bowel and anorectal stenosis and atresia were positively related to antenatal cannabis exposure in Hawaii [4] but were not tracked in Europe. Large bowel disorders and Hirschsprung’s disease were strongly cannabis related in USA [55]. Gastrointestinal disorders, including small and large bowel stenoses and atresias, were also noted to be positively associated with cannabis exposure in Australia [8].
Limb Disorders
The limb disorders that were positively associated with cannabis exposure on bivariate analysis were: club foot, hip dysplasia, limb, limb reductions, polydactyly and syndactyly. Interestingly, syndactyly, polydactyly and reduction deformity of the upper limbs were first identified in the Hawaiian series [4]. Reduction of the lower limbs was a positive finding in the USA series [55]. Musculoskeletal disorders were also noted to be elevated in Colorado in association with increased cannabis exposure.
Interestingly, recent reports from France and Germany [112–115], where cannabis use is rising, relate to ‘outbreaks’ of babies borne without limbs but no such reports have been issued from nearby Switzerland where cannabis is not allowed to enter the food chain.
An increased incidence of limb defects is reminiscent of the notorious teratogenic agent thalidomide to which we directly owe the modern system of pharmacological regulation and safeguards [116]. Concerningly 21 of the 31 CAs described following thalidomide exposure are documented in the present series in association with cannabis exposure [116–122]. Similarly, cannabis shares most (12/13) of the same mechanisms of molecular action as thalidomide .
Body Wall
One of the CAs for which the strongest evidence exists for cannabis association is gastroschisis [4, 123–128]. Moreover, gastroschisis was shown to be 3-fold elevated after multivariable adjustment for all likely confounders in a careful Canadian study [125]. It was therefore of considerable interest to investigate the relationships in the European dataset.
The body wall anomalies with which cannabis was found to be significantly associated were: abdominal wall defect, diaphragmatic hernia, gastroschisis and omphalocele. Gastroschisis has been previously linked with cannabis use also in Hawaii [4], Colorado [5], Australia [8] and Canada [7, 125, 129–131]. Omphalocele was also associated with cannabis use both in Australia [8] and in preclinical series in animals [85]. Diaphragmatic hernia and gastroschisis have previously been linked with cannabis use in USA [87].
Uronephrological Disorders
Uronephrolgical disorders that were found to be elevated on bivariate analyses included: bilateral renal agenesis, bladder extrophy/epispadias, hydronephrosis, hypospadias, multicystic renal disorders, posterior urethral valve and urinary disorders. Obstructive genitourinary disorders was positively associated with prenatal cannabis use in Hawaii [4] and USA [55] and congenital posterior urethral valve was also positively associated in USA [55]. Hence, the European series significantly extends work on this body system.
Genital Disorders
The genital disorder that was found to be elevated after increased population cannabis exposure in bivariate analyses was genital disorder. Indeterminate sex was also noted to be elevated in several multivariable IPW panel analyses. Genitourinary anomalies were also noted to be elevated in Colorado [5], and epispadias was found to be elevated in USA data [55] and hypospadias in Australia [8].
Respiratory
The respiratory defects that were identified on bivariate analysis to be linked with cannabis exposure were cystic lung and respiratory anomalies. Respiratory anomalies were previously identified as a cannabis-associated defect both in Colorado [5] and in USA [55]. Cystic anomalies of the lung have been described as occurring in association with cannabis exposure in later life [132–134].
General
The CAs considered under the ‘general’ category and found to be elevated at bivariate analyses were: all anomalies, amniotic band, conjoined twins, foetal alcohol syndrome, lateral anomalies, maternal infections resulting in malformations, situs inversus, skeletal dysplasias, teratogenic syndromes, valproate syndrome and VATER/VACTERL.
The presence of foetal alcohol syndrome on this list is noteworthy for several reasons. There is evidence of co-abuse of substances of addiction. Many studies document the gateway effect of both cannabis and alcohol leading on to further drug use. Interestingly the foetal alcohol syndrome has been shown to be mediated epigenetically partly via the cannabinoid receptor type 1 (CB1R) [135–138].
Of particular interest is the rare syndrome VATER/VACTERL. Whilst for a long period the reason that these multiple syndromes were observed together was unclear, this has recently been attributed to the inhibition of the key human morphogen sonic hedgehog [57]. Sonic hedgehog plays a pivotal and key and irreplaceable role in human morphogenesis at many points in most organ systems. The positive association of cannabis exposure with this syndrome is important in that it demonstrates at once both that the general view that cannabis is not associated with defined CAs is incorrect and also that cannabis can affect the development of many body systems
Cannabinoids have also been shown to inhibit other key body morphogens, including retinoic acid signalling [139–141], wnt signalling [142–147], the hippo pathway [31], notch signalling [148–152], fibroblast growth factor [153, 154], including transactivation of the FGF1R by CB1R [155], and bone morphogenetic proteins [156–158].
Epidemiological Causal Assignment
Inverse probability weighting effectively removes the non-comparability of groups of interest and creates a situation where groups can be properly compared. It is now well established that inverse probability weighting has the effect of ‘pseudo-randomizing’ an observational dataset, which transforms its findings from merely observational-level associations to replicate a randomized trial in a real-world setting. The reanalysis of several observational trials using this procedure has been done and was shown to reliably replicate the results of subsequent randomized clinical trials [159]. Therefore, the use of this procedure strengthens the present findings.
The other major weakness of observational studies is that uncontrolled extraneous factor(s) might account for the apparently causal nature of an association. The E-value quantifies the degree of association required of some unmeasured confounding covariate with both the exposure of concern and the outcome of interest to obviate the described effect. The E-value usually associated with causality is 1.25 [81], and an E-value of 9, such as is represented by the tobacco–lung cancer relationship, represents a large effect [160]. Many of the mEV reported herein are much larger than this, and range up to 2.46 × 1039. The E-values in Table 8 range at six years lag from 18.45 to 3.61 × 1033 so that they are clearly in a range far beyond what could reasonably be ascribed to extraneous confounding.
Hence, it can be properly stated that the reported results in this study fulfil the epidemiological criteria of causality. Naturally, this comment is not intended to imply that further experimental, laboratory, genomic and epigenomic studies are not required for, indeed, our results underscore the importance of such further and critically important mechanistic investigations.
Mechanisms and Pathways
The mechanisms by which cannabis exposure might induce genotoxicity or epigenotoxicity are numerous and diverse. Plethoric effects on many reproductive organs, sperm stem cells, spermatids, oocytes, cell division, chromosomes, DNA bases, genes, histones and the epigenome have all been described [21–38]. Partly because there are eight different receptors for cannabinoids described [161], the subject is complex and has been reviewed in considerable detail elsewhere [18, 31, 35, 36, 53, 90, 162–167]. It is intended here to make only a few remarks by way of introduction and overview.
Epigenomic Mechanisms
Much elegant work has been undertaken in recent years documenting that quite widespread changes of DNA methylation are induced by cannabis exposure [37] and that these can be passed to subsequent generations of mice [33–37] and can affect the epigenetic regulation of key medium spiny neurons in the nucleus accumbens of the brain, which is a key appetitive driver centre in subsequent generations, which, in turn, affects the proclivity to develop major disorders, such as (opioid and cocaine) drug dependency syndromes [33–37]. It has also been shown that the epigenomic changes in rat and human sperm are equivalent at the pathway level and that such changes in rats were transmissible to subsequent generations [31]. Indeed, it was recently shown that the cessation of cannabis exposure for 17 weeks allows many of the epigenomic changes seen in rats and humans to reverse [32].
Relative Histone Deficiency
It has been shown both in classical focussed investigations of histone synthesis [28–30, 41, 168, 169] and of proteomic screens [40] that histone synthesis in cannabis-exposed tissues is greatly reduced, sometimes by as much as 50%. This has far-reaching implications for epigenomic dysregulation but remains relatively unexplored by modern methods. This area of induced epimutagensis is clearly a very fertile area for further research in much the same way that classical mutagenesis studies have proved to be invaluable as a resource for investigation of gene and protein structure and function in classical genomics.
Epigenomic Implications of Mitochondriopathy
The mitochondriopathic effects of cannabinoids have been richly investigated in considerable detail but remain largely overlooked in the current broader investigative environment.
In their inner and outer membrane complexes and intermembrane spaces, mitochondria carry the full complement of cannabinoid signal reception and transduction machinery as is found in the plasmalemma. They are therefore competent to receive and transduced signals from lipophilic cannabinoids, which will clearly move freely across lipid bilayer membranes.
Whilst many of the proteins found within mitochondria are encoded by the 16 KB of mitochondrial DNA, some are not. Hence, there must be a coordination in the expression of the mitochondrial and nuclear genes to allow normal mitochondrial function. Similar remarks apply to the supply of nuclear energy and epigenomic substrates. This mitonuclear communication occurs through various small-molecule shuttles, including nicotinamide mononucleotide and glutamate/aspartate [52]. This indirect system is known as mitonuclear balance [52].
Mitochondrial inhibition from cannabinoids has been shown to occur through many mechanisms, including uncoupling oxidative phosphorylation by way of uncoupling protein 2 and increasing transmembrane proton leak, and a reduction of many of the key cytochromes of the electron transport chain, including the F1 ATPase itself, which finally harnesses the proton motive force to drive ATP synthesis [40].
Since mitochondria generate the bulk of the small molecules that are used as epigenomic substrates and co-factors (such as methyl, acetyl, phosphate, phosphoribosyl and many other groups) and the bulk of cellular ATP for the largely energy-dependent genomic and epigenomic reactions, it follows that inhibition of intermediary mitochondrial metabolism necessarily has a profound impact on epigenomic expression by limiting the supply of both substrates and energy. Moreover, mitochondrial inhibition will also greatly perturb the indirect pathways of mitonuclear balance.
Indeed, membranous structural continuity has recently been demonstrated between several subcellular organelles and the nucleus [170].
These areas would appear to provide fertile areas for further detailed mechanistic studies.
Cannabinoid DNA Methylome—Ageing DNA Methylome
Derangement of the epigenome is one of the major potential concerns to emerge from this study. Given cannabis exposure has previously been linked with accelerated organismal ageing clinically [95] and since both ageing and cannabinoid pathophysiology are mediated importantly through the epigenome, one fertile field of further investigation would be the intersection and interaction of these two DNA methylomes, including DNA methylation ages [171–173]. Such studies would powerfully inform the multisystem derangements described herein.
Cannabis-induced changes to histone phosphorylation and acetylation states have also previously been documented [41]. The serious changes induced in sperm motility by altered tubulin glycosylation have also recently been documented and were alluded to above as part of the important ‘tubulin code’ [42]. Hence, important work on comparative cannabinoid perturbations of proteomes, phosphoproteomes and post-translational proteomic changes, including altered glycan physiology (as a major post-translational modification) [174, 175] and their altered functional metabolomic and epigenomic implications, are similarly large gaps in the literature, which remain to be addressed.
Non-Coding RNA Expression
The areas of cannabinoid impacts on signalling RNAs of various classes, such as short interfering RNAs, long non-coding RNAs and promoter, enhancer and superenhancer expression and activity are relatively unexplored to our knowledge and also constitute very important areas for further research, especially in view of the organ-specific CAs and tumourigenesis described and alluded to above. Cannabinoid-induced changes to transfer RNAs and gonadal piwi interacting RNAs have been described [176].
Further Research
The present findings strongly indicate the need for further research in both epidemiological and mechanistic fields. Geospatial statistical studies could be extended by the use of smaller geographical units and the use of formal spatiotemporal geostatistical methods. Mechanistic studies are also indicated into the myriad of cannabinoid effects on reproductive toxicity, its effects on mitosis and meiosis, chromosomal and genomic toxicity and the many complex and interacting epigenetic mechanisms, which are likely to be interactively in play.
Generalizability
We feel that these data are widely generalizable for several reasons, including the internal consistency of results within this study, their external consistency with other studies of large datasets published internationally [4–9, 17, 18, 55, 86, 87], the demonstration that the criteria of epidemiological causality are fulfilled (by inverse probability weighting and E-value studies), their robust mechanistic support from the cellular and molecular literature in laboratory investigations [24–26, 28–30, 168, 169, 177–180] and strong support from the preclinical prenatal cannabinoid exposure literature involving several model animal species [83, 85].
The European and USA datasets together comprise the majority of the global datasets on these issues. The fact that similar results have been found in both datasets provides strong external validation a posteriori to both sets of studies of the veracity of their findings.
Strengths and Limitations
This study has a number of strengths and limitations. Strengths include the use of a full panel of substance-related and economic covariates, the use of the formal quantitative techniques of causal inference, the use of multipanelled graphs to visualize whole datasets at one glance and the use of space-time panel regression. We also used formal random forest regression techniques to evaluate variable importance formally. The study has been done on a background of a current understanding of both the cannabis-related epidemiological and mechanistic literature. Its shortcomings relate mainly to the fact that in common with many large epidemiological studies individual participant level data on cannabinoid and substance exposure is not available. Linear interpolation was used to complete some drug data fields.
Conclusion
Using various metrics of cannabis exposure, this study provides compelling evidence of cannabis exposure in the aetiology of a broad range of CAs observed in Europe. Particularly notable on this list were all anomalies and anomalies of the cardiovascular, central nervous, gastrointestinal, chromosomal and genetic, genital, uronephrological, limb and body wall systems and the multisystem disorder VACTERL. These results are consistent with and substantially extend recently documented results from several other jurisdictions [4–9, 17, 18, 55, 86, 87]. They are also consistent with in vitro and preclinical studies from the 1960s to the present time [24–26, 28–30, 168, 169, 177–180]. Data specifically implicate high-intensity daily use and amply confirm concerns raised in relation to the triple epidemiological convergence of rising cannabis use prevalence, rising cannabis use intensity and rising THC concentrations in cannabis herb and resins as a particularly potent driver of cannabis-related disorders [15, 16]. Results are also in accord with recent reports of the epidemiological implication of cannabis with cancers of many types [17, 18, 20, 89] and, since epigenotoxicity has been definitively linked with cellular ageing [19], carry far-reaching implications for programmes and policies, which would lead to widespread genotoxic/epigenotoxic damage across the community under the erroneous assumption that the known epigenotoxic/genotoxic effects of cannabinoids are of minimal clinical significance. The present results amply document the fallacy of this assumption and its severe and ominous implications for population health policy. Further laboratory research, particularly relating to the genotoxic, epigenotoxic, metabolomic-mitochondriopathic-epigenomic and chromosomal toxicity of diverse cannabinoids together with high-resolution spatiotemporal epidemiological studies are strongly indicated.
Supplementary Material
Acknowledgements
All authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Prof. Mark Stevenson at the University of Melbourne is very gratefully thanked for specially preparing a new version of epiR to handle the very large integers involved in this analysis. Prof. Maya Mathur of Stanford Medical School is thanked for her advice, assistance and guidance with E-values.
Contributor Information
Albert Stuart Reece, Division of Psychiatry, University of Western Australia, Crawley, Western Australia 6009, Australia; School of Medical and Health Sciences, Edith Cowan University, Joondalup, Western Australia 6027, Australia.
Gary Kenneth Hulse, Division of Psychiatry, University of Western Australia, Crawley, Western Australia 6009, Australia; School of Medical and Health Sciences, Edith Cowan University, Joondalup, Western Australia 6027, Australia.
Data Availability
All data generated or analysed during this study are included in this published article and its supplementary information files. Data along with the relevant R code have been made publicly available on the Mendeley Database Repository and can be accessed from this URL doi: 10.17632/vd6mt5r5jm.1.
Supplementary data
Supplementary data is available at EnvEpig online.
Funding
No funding was provided for this study. No funding organization played any role in the design and conduct of the study; collection, management, analysis and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Conflict of interest statement
The authors declare that they have no competing interests.
Declarations
Ethics Approval and Consent to Participate. The Human Research Ethics Committee of the University of Western Australia provided ethical approval for the study to be undertaken 24 September 2021 (No. RA/4/20/4724).
Consent for Publication
Not applicable.
Authors’ Contributions
A.S.R. assembled the data, designed and conducted the analyses and wrote the first manuscript draft. G.K.H. provided technical and logistic support, co-wrote the paper, assisted with gaining ethical approval and provided advice on manuscript preparation and general guidance to study conduct. A.S.R. had the idea for the article, performed the literature search, wrote the first draft and is the guarantor for the article.
Abbreviations.
| Acronym | Expanded meaning |
|---|---|
| AME | Average marginal effect |
| AFE | Attributable fraction in the exposed |
| ASD | Atrial septal defect |
| AVSD | Atrioventricular septal defect |
| CA | Congenital anomaly |
| CAR | Congenital anomaly rate |
| CB1R | Cannabinoid receptor type 1 |
| EMCDDA | European Monitoring Centre for Drugs and Drug Addiction |
| E-Value | Expected value |
| IPW | Inverse probability weighted |
| LMC_Herb_Daily | Last month cannabis use × herbal THC content × daily use interpolated |
| LMC_Resin_Daily | Last month cannabis use × cannabis resin THC content × daily use interpolated |
| mEV | Minimum E-value |
| PAR | Population attributable risk |
| PDA | Patent ductus arteriosus |
| RR | Relative rate |
| THC | Δ9-tetrahydrocannabinol |
| VATER/VACTERL | Vertebral, anorectal, cardiac, tracheo-esophageal fistula ± oesophageal atresia, renal anomalies and limb abnormalities syndrome |
| VSD | Ventricular septal defect |
| WHO | World Health Organization |
References
- 1. Volkow ND, Compton WM, Wargo EM. The risks of marijuana use during pregnancy. JAMA 2017;317:129–30. [DOI] [PubMed] [Google Scholar]
- 2. Volkow ND, Han B, Compton WM. et al. Marijuana use during stages of pregnancy in the United States. Ann Intern Med 2017;166:763–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Volkow ND, Han B, Einstein EB. et al. Prevalence of substance use disorders by time since first substance use among young people in the US. JAMA Pediatr 2021;175:640–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Forrester MB, Merz RD. Risk of selected birth defects with prenatal illicit drug use, Hawaii, 1986-2002. J Toxicol Environ Health 2007;70:7–18. [DOI] [PubMed] [Google Scholar]
- 5. Reece AS, Hulse GK. Cannabis teratology explains current patterns of coloradan congenital defects: the contribution of increased cannabinoid exposure to rising teratological trends. Clin Pediatr (Phila) 2019;58:1085–123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Reece AS, Hulse GK. Cannabis consumption patterns explain the east-west gradient in Canadian neural tube defect incidence: an ecological study. Glob Pediatr Health 2019;6:2333794X19894798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Reece AS, Hulse GK. Canadian cannabis consumption and patterns of congenital anomalies: an ecological geospatial analysis. J Addict Med 2020;14:e195–e210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Reece AS, Hulse GK. Broad Spectrum epidemiological contribution of cannabis and other substances to the teratological profile of northern New South Wales: geospatial and causal inference analysis. BMC Pharmacol Toxicol 2020;21:75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Reece AS, Hulse GK. Cannabis and pregnancy don’t mix. Mo Med 2020;117:530–1. [PMC free article] [PubMed] [Google Scholar]
- 10. Reece AS, Hulse GK. Cannabis in pregnancy – rejoinder, exposition and cautionary tales. Psychiatric Times, 2020. https://www.bing.com/search?q=Cannabis+in+Pregnancy+%E2%80%93+Rejoinder%2C+Exposition+and+Cautionary+Tales&cvid=22538e0124c04711b92017489c63214a&aqs=edge.69i57.439j0j1&pglt=43&FORM=ANSPA1&PC=U531 (20 November 2021, date last accessed).
- 11. Reece AS, Hulse GK. Epidemiological overview of multidimensional chromosomal and genome toxicity of cannabis exposure in congenital anomalies and cancer development. Sci Rep 2021;11:13892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Young-Wolff KC, Ray GT, Alexeeff SE. et al. Rates of prenatal cannabis use among pregnant women before and during the COVID-19 pandemic. JAMA 2021;326:1745–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Young-Wolff KC, Tucker LY, Alexeeff S. et al. Trends in self-reported and biochemically tested marijuana use among pregnant females in California from 2009-2016. JAMA 2017;318:2490–1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Dickson B, Mansfield C, Guiahi M. et al. Recommendations from cannabis dispensaries about first-trimester cannabis use. Obstet Gynecol 2018;131:1031–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Reece AS, Hulse GK. Quadruple convergence – rising cannabis prevalence, intensity, concentration and use disorder treatment. Lancet Reg Health - Europe 2021;100245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Manthey J, Freeman TP, Kilian C. et al. Public health monitoring of cannabis use in Europe: prevalence of use, cannabis potency, and treatment rates. Lancet Reg Health - Europe 2021;10:100227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Reece AS, Hulse GK. Cannabinoid exposure as a major driver of pediatric acute lymphoid Leukaemia rates across the USA: combined geospatial, multiple imputation and causal inference study. BMC Cancer 2021;21:984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Reece AS, Hulse GK. Epidemiological overview of multidimensional chromosomal and genome toxicity of cannabis exposure in congenital anomalies and cancer development. Sci Rep 2021;11:13892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Lu Y, Brommer B, Tian X. et al. Reprogramming to recover youthful epigenetic information and restore vision. Nature 2020;588:124–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Reece AS, Hulse GK. A geospatiotemporal and causal inference epidemiological exploration of substance and cannabinoid exposure as drivers of rising US pediatric cancer rates. BMC Cancer 2021;21:197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Chioccarelli T, Cacciola G, Altucci L. et al. Cannabinoid receptor 1 influences chromatin remodeling in mouse spermatids by affecting content of transition protein 2 mRNA and histone displacement. Endocrinology 2010;151:5017–29. [DOI] [PubMed] [Google Scholar]
- 22. Rossato M, Ion Popa F, Ferigo M. et al. Human sperm express cannabinoid receptor Cb1, the activation of which inhibits motility, acrosome reaction, and mitochondrial function. J Clin Endocrinol Metab 2005;90:984–91. [DOI] [PubMed] [Google Scholar]
- 23. Rossato M, Pagano C, Vettor R. The cannabinoid system and male reproductive functions. J Neuroendocrinol 2008;20:90–3. [DOI] [PubMed] [Google Scholar]
- 24. Leuchtenberger C, Leuchtenberger R. Morphological and cytochemical effects of marijuana cigarette smoke on epithelioid cells of lung explants from mice. Nature 1971;234:227–9. [DOI] [PubMed] [Google Scholar]
- 25. Leuchtenberger C, Leuchtenberger R, Schneider A. Effects of marijuana and tobacco smoke on human lung physiology. Nature 1973;241:137–9. [DOI] [PubMed] [Google Scholar]
- 26. Stenchever MA, Kunysz TJ, Allen MA. Chromosome breakage in users of marihuana. Am J Obstet Gynecol 1974;118:106–13. [DOI] [PubMed] [Google Scholar]
- 27. Russo C, Ferk F, Mišík M. et al. Low doses of widely consumed cannabinoids (cannabidiol and cannabidivarin) cause DNA damage and chromosomal aberrations in human-derived cells. Arch Toxicol 2019;93:179–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Zimmerman AM, Zimmerman S. Cytogenetic studies of cannabinoid effects. In: Braude MC, Zimmerman AM (eds), Genetic and Perinatal Effects of Abused Substances. 1. New York: Academic Press Inc., 1987, 95–112. Harcourt, Brace Jovanovich. [Google Scholar]
- 29. Zimmerman AM, Zimmerman S, Raj AY. Effects of cannabinoids on spermatogensis in mice. In: Nahas GG, Sutin KM, Harvey DJ. et al. (eds), Marijuana and Medicine. 1 1st edn. Totowa, NY: Humana Press, 1999, 347–58. [Google Scholar]
- 30. Zimmerman AM, Raj AY. Influence of cannabinoids on somatic cells in vivo. Pharmacology 1980;21:277–87. [DOI] [PubMed] [Google Scholar]
- 31. Murphy SK, Itchon-Ramos N, Visco Z. et al. Cannabinoid exposure and altered DNA methylation in rat and human sperm. Epigenetics 2018;13:1208–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Schrott R, Murphy SK, Modliszewski JL. et al. Refraining from use diminishes cannabis-associated epigenetic changes in human sperm. Environ Epigenet 2021;7:dvab009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. DiNieri JA, Wang X, Szutorisz H. et al. Maternal cannabis use alters ventral striatal dopamine D2 gene regulation in the offspring. Biol Psychiatry 2011;70:763–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Szutorisz H, DiNieri JA, Sweet E. et al. Parental THC exposure leads to compulsive heroin-seeking and altered striatal synaptic plasticity in the subsequent generation. Neuropsychopharmacology 2014;39:1315–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Szutorisz H, Hurd YL. Epigenetic effects of cannabis exposure. Biol Psychiatry 2016;79:586–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Szutorisz H, Hurd YL. High times for cannabis: Epigenetic imprint and its legacy on brain and behavior. Neurosci Biobehav Rev 2018;85:93–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Watson CT, Szutorisz H, Garg P. et al. Genome-wide DNA methylation profiling reveals epigenetic changes in the rat nucleus accumbens associated with cross-generational Effects of adolescent THC exposure. Neuropsychopharmacology 2015;40:2993–3005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Morishima A. Effects of cannabis and natural cannabinoids on chromosomes and ova. NIDA Res Monogr 1984;44:25–45. [PubMed] [Google Scholar]
- 39. DiNieri JA, Wang X, Szutorisz H. et al. Maternal cannabis use alters ventral striatal dopamine D2 gene regulation in the offspring. Biol Psychiatry 2011;70:763–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Wang J, Yuan W, Li MD. Genes and pathways co-associated with the exposure to multiple drugs of abuse, including alcohol, amphetamine/ methamphetamine,cocaine, marijuana, morphine, and/or nicotine: a review of proteomics analyses. Mol Neurobiol 2011;44:269–86. [DOI] [PubMed] [Google Scholar]
- 41. Mon MJ, Haas AE, Stein JL. et al. Influence of psychoactive and nonpsychoactive cannabinoids on chromatin structure and function in human cells. Biochem Pharmacol 1981;30:45–58. [DOI] [PubMed] [Google Scholar]
- 42. Gadadhar S, Alvarez Viar G, Hansen JN. et al. Tubulin glycylation controls axonemal dynein activity, flagellar beat, and male fertility. Science 2021;371:6525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Chan JZ, Duncan RE. Regulatory effects of cannabidiol on mitochondrial functions: a review. Cells 2021;10:1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Ryan D, Drysdale AJ, Lafourcade C. et al. Cannabidiol targets mitochondria to regulate intracellular Ca2+ levels. J Neurosci 2009;29:2053–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Winklmayr M, Gaisberger M, Kittl M. et al. Dose-dependent cannabidiol-induced elevation of intracellular calcium and apoptosis in human articular chondrocytes. J Orthop Res 2019;37:2540–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Hebert-Chatelain E, Desprez T, Serrat R. et al. A cannabinoid link between mitochondria and memory. Nature 2016;539:555–9. [DOI] [PubMed] [Google Scholar]
- 47. Chiu P, Karler R, Craven C. et al. The influence of delta9-tetrahydrocannabinol, cannabinol and cannabidiol on tissue oxygen consumption. Res Commun Chem Pathol Pharmacol 1975;12:267–86. [PubMed] [Google Scholar]
- 48. Koch M, Varela L, Kim JG. et al. Hypothalamic POMC neurons promote cannabinoid-induced feeding. Nature 2015;519:45–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Harkany T, Horvath TL. (S)Pot on mitochondria: cannabinoids disrupt cellular respiration to limit neuronal activity. Cell Metab 2017;25:8–10. [DOI] [PubMed] [Google Scholar]
- 50. Sarafian TA, Habib N, Oldham M. et al. Inhaled marijuana smoke disrupts mitochondrial energetics in pulmonary epithelial cells in vivo. Am J Physiol 2006;290:L1202–9. [DOI] [PubMed] [Google Scholar]
- 51. Sarafian TA, Kouyoumjian S, Khoshaghideh F. et al. Delta 9-tetrahydrocannabinol disrupts mitochondrial function and cell energetics. Am J Physiol 2003;284:L298–306. [DOI] [PubMed] [Google Scholar]
- 52. Canto C, Menzies KJ, Auwerx J. NAD(+) metabolism and the control of energy homeostasis: a balancing act between mitochondria and the nucleus. Cell Metab 2015;22:31–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Reece AS, Hulse GK. Chromothripsis and epigenomics complete causality criteria for cannabis- and addiction-connected carcinogenicity, congenital toxicity and heritable genotoxicity. Mutat Res 2016;789:15–25. [DOI] [PubMed] [Google Scholar]
- 54. Reece AS, Hulse GK. Cannabinoid exposure as a major driver of pediatric acute lymphoid Leukaemia rates across the USA: combined geospatial, multiple imputation and causal inference study. BMC Cancer 2021;21:684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Reece AS, Hulse GK. Cannabis in pregnancy – rejoinder, exposition and cautionary tales. Psychiatric Times, 10 October 2020. https://www.bing.com/search?q=Cannabis+in+Pregnancy+%E2%80%93+Rejoinder%2C+Exposition+and+Cautionary+Tales&cvid=22538e0124c04711b92017489c63214a&aqs=edge.69i57.439j0j1&pglt=43&FORM=ANSPA1&PC=U531. (20 November 2021, date last accessed).
- 56. Hosptial GOS . VACTERL association: information for families Great Ormond St., London, United Kingdom: Great Ormond St., London, United Kingdom. 2019. (Updated 2021). https://media.gosh.nhs.uk/documents/VACTERL_association_F1612_FINAL_Sep19.tif (29 October 2021, date last accessed).
- 57. Fish EW, Murdaugh LB, Zhang C. et al. Cannabinoids Exacerbate Alcohol Teratogenesis by a CB1-Hedgehog Interaction. Sci Rep 2019;9:16057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Commission E . Eurocat Data: Prevalence Charts and Tables Brussels, Belgium: European Commission; 2021. https://eu-rd-platform.jrc.ec.europa.eu/eurocat/eurocat-data/prevalence_en (29 October 2021, date last accessed). [Google Scholar]
- 59. Bank TW . The World Bank: crude data: birth rate 2021. https://data.worldbank.org/indicator/SP.DYN.CBRT.IN (29 October 2021, date last accessed).
- 60. Organization WH . Global Health Observatory. Geneva, Switzerland: WHO, 2021. https://www.who.int/data/gho/data/indicators/indicator-details/GHO/total-recorded-unrecorded-alcohol-per-capita-15--consumption (1 October 2021, date last accessed). [Google Scholar]
- 61. European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) . European Monitoring Centre for Drugs and Drug Addiction (EMCDDA): Statistical Bulletin 2021 — prevalence of drug use Lisbon, Portugal: European Monitoring Centre for Drugs and Drug Addiction (EMCDDA). 2021https://www.emcdda.europa.eu/data/stats2021/gps_en. (20 November 2021, date last accessed).
- 62. Bank TW . The World Bank: Crude Data: Adjusted net national income per capita (current US$): The World Bank. 2021. https://data.worldbank.org/indicator/NY.ADJ.NNTY.PC.CD (29 October 2021, date last accessed).
- 63. Wickham H, Averick M, Bryan J. et al. Welcome to the Tidyverse. J Open Source Softw 2019;4:1686–91. [Google Scholar]
- 64. Crawley MJ. The R Book. Hoboken, NJ: Wiley, 2007, P. 941. [Google Scholar]
- 65. Leeper TJ. Margins: marginal effects for model objects. R package version 0.3.26; 2021. 2021.
- 66. Mathur M. Package ‘EValue’: CRAN. 2020. https://cran.r-project.org/web/packages/EValue/EValue.tif (14 August 2020, date last accessed).
- 67. Mathur MB, Ding P, Riddell CA. et al. Web site and r package for computing E-values. Epidemiology 2018;29:e45–e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. VanderWeele TJ, Ding P. Sensitivity analysis in observational research: introducing the E-value. Ann Intern Med 2017;167:268–74. [DOI] [PubMed] [Google Scholar]
- 69. VanderWeele TJ, Mathur MB. Commentary: developing best-practice guidelines for the reporting of E-values. Int J Epidemiol 2020;49:1495–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Stevenson M, Sergeant E, Nunes T. et al. epiR: Tools for the analysis of epidemiological data. 2020. https://fvas.unimelb.edu.au/research/groups/veterinary-epidemiology-melbourne; https://www.ausvet.com.au/ (1 November 2020, date last accessed).
- 71. Wright MN, Ziegler A. Ranger: a fast implementation of random forests for high dimensional data in C++ and R. J Stat Softw 2017;77:1–17. [Google Scholar]
- 72. Greenwell BM, Boehmke BC. Variable importance plots—an introduction to the vip package. R J 2021;12:343–66. [Google Scholar]
- 73. Warnes GR, Bolker B, Bonebakker L. et al. gplots: Various R programming tools for plotting data. R package version 311 [Internet]. 2020. https://CRAN.R-project.org/package=gplots (updated October 29, 2021; 29 October 2021, date last accessed).
- 74. Robinson D, Hayes A, Couch S. Broom: convert statistical objects into tidy tibbles: R package version 0.7.0. 2020. https://CRAN.R-project.org/package=broom (12 December 2020, date last accessed).
- 75. Bolker B, Robinson D. Broom.mixed: tidying methods for mixed models. R package version 0.2.7.: R – CRAN. 2020. http://github.com/bbolker/broom.mixed (12 December 2020, date last accessed).
- 76. Bonferroni CE. Teoria statistica delle classi e calcolo delle probabilità Editor. Neil J Salkind: Encycl Res Des 1936. [Google Scholar]
- 77. Holm S, Simple Sequentially A. Rejective Multiple Test Procedure. Scand J Stat 1979;6:65–70. [Google Scholar]
- 78. Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc: Ser B (Methodological) 1995;57:289–300. [Google Scholar]
- 79. Croissant Y, Millo G, Tappe K. et al. Package ‘plm’. https://cran.r-project.org/web/packages/plm/index.html: CRAN: Central "R"Archive Network. 2014. https://cran.r-project.org/web/packages/plm/plm.tif (7 May 2020, date last accessed).
- 80. Van der Wal WM, Geskus RB. IPW: an R package for inverse probabilty weighting. J Stat Softw 2011;43:1–23. [Google Scholar]
- 81. VanderWeele TJ, Ding P, Mathur M. Technical considerations in the use of the E-value. J Causal Inference 2019;7:1–11. [Google Scholar]
- 82. Carlson BM. Human Embryology and Developmental Biology 6th edn. Philadelphia: Elsevier, 2019, 506. [Google Scholar]
- 83. Geber WF, Schramm LC. Teratogenicity of marihuana extract as influenced by plant origin and seasonal variation. Arch Int Pharmacodyn Ther 1969;177:224–30. [PubMed] [Google Scholar]
- 84. Geber WF, Schramm LC. Effect of marihuana extract on fetal hamsters and rabbits. Toxicol Appl Pharmacol 1969;14:276–82. [DOI] [PubMed] [Google Scholar]
- 85. Graham JDP. Cannabis and health. In: Graham JDP (ed.), Cannabis and Health. 1 1st edn. London, New York, San Francisco: Academic Press, 1976, 271–320. [Google Scholar]
- 86. Jenkins KJ, Correa A, Feinstein JA. et al. Noninherited risk factors and congenital cardiovascular defects: current knowledge: a scientific statement from the American Heart Association Council on Cardiovascular Disease in the Young: endorsed by the American Academy of Pediatrics. Circulation 2007;115:2995–3014. [DOI] [PubMed] [Google Scholar]
- 87. Van Gelder MMHJ, Donders ART, Devine O. et al. Using Bayesian models to assess the effects of under-reporting of cannabis use on the association with birth defects, national birth defects prevention study, 1997-2005. Paediatr Perinat Epidemiol 2014;28:424–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Van Gelder MMHJ, Reefhuis J, Caton AR. et al. Maternal periconceptional illicit drug use and the risk of congenital malformations. Epidemiology 2009;20:60–6. [DOI] [PubMed] [Google Scholar]
- 89. Reece AS, Hulse GK. Causal inference multiple imputation investigation of the impact of cannabinoids and other substances on ethnic differentials in US testicular cancer incidence. BMC Pharmacol Toxicol 2021;22:40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Reece AS, Hulse GK. Impacts of cannabinoid epigenetics on human development: reflections on Murphy et. al. ‘cannabinoid exposure and altered DNA methylation in rat and human sperm’ epigenetics 2018; 13: 1208-1221. Epigenetics 2019;14:1041–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Reece AS, Hulse GK. Epidemiological associations of various substances and multiple cannabinoids with autism in USA. Clin Pediatr Open Access 2019;4:1–20. [Google Scholar]
- 92. Reece AS, Hulse GK. Effect of cannabis legalization on US autism incidence and medium term projections. Clin Pediatr Open Access 2019;4:1–17. [Google Scholar]
- 93. Reece AS, Hulse GK. Gastroschisis and autism-dual canaries in the Californian coalmine. JAMA Surg 2019;154:366–7. [DOI] [PubMed] [Google Scholar]
- 94. Reece AS, Hulse GK. Co-occurrence across time and space of drug- and cannabinoid- exposure and adverse mental health outcomes in the National Survey of Drug Use and Health: combined geotemporospatial and causal inference analysis. BMC Public Health 2020;20:1655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Reece AS, Norman A, Hulse GK. Cannabis exposure as an interactive cardiovascular risk factor and accelerant of organismal ageing: a longitudinal study. BMJ Open 2016;6:e011891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Reece AS, Hulse GK. Contemporary epidemiology of rising atrial septal defect trends across USA 1991-2016: a combined ecological geospatiotemporal and causal inferential study. BMC Pediatr 2020;20:539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Callaghan RC, Allebeck P, Akre O. et al. Cannabis use and incidence of testicular cancer: a 42-year follow-up of Swedish men between 1970 and 2011. Cancer Epidemiol Biomarkers Prev 2017;26:1644–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Daling JR, Doody DR, Sun X. et al. Association of marijuana use and the incidence of testicular germ cell tumors. Cancer 2009;115:1215–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Gurney J, Shaw C, Stanley J. et al. Cannabis exposure and risk of testicular cancer: a systematic review and meta-analysis. BMC Cancer 2015;15:897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Lacson JC, Carroll JD, Tuazon E. et al. Population-based case-control study of recreational drug use and testis cancer risk confirms an association between marijuana use and nonseminoma risk. Cancer 2012;118:5374–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Song A, Myung NK, Bogumil D. et al. Incident testicular cancer in relation to using marijuana and smoking tobacco: A systematic review and meta-analysis of epidemiologic studies. Urol Oncol 2020;38:642.e1-.e9. [DOI] [PubMed] [Google Scholar]
- 102. Trabert B, Sigurdson AJ, Sweeney AM. et al. Marijuana use and testicular germ cell tumors. Cancer 2011;117:848–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Birger Y, Shiloh R, Izraeli S. Mechanisms of leukemia evolution: lessons from a congenital syndrome. Cancer Cell 2019;36:115–7. [DOI] [PubMed] [Google Scholar]
- 104. Bonaventure A, Harewood R, Stiller CA. et al. Worldwide comparison of survival from childhood leukaemia for 1995-2009, by subtype, age, and sex (CONCORD-2): a population-based study of individual data for 89 828 children from 198 registries in 53 countries. Lancet Haematol 2017;4:e202–e17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Bröske AM, Vockentanz L, Kharazi S. et al. DNA methylation protects hematopoietic stem cell multipotency from myeloerythroid restriction. Nat Genet 2009;41:1207–15. [DOI] [PubMed] [Google Scholar]
- 106. Geurts van Kessel A, Suijkerbuijk RF, Sinke RJ. et al. Molecular cytogenetics of human germ cell tumours: i(12p) and related chromosomal anomalies. Eur Urol 1993;23:23–8. discussion 9. [DOI] [PubMed] [Google Scholar]
- 107. Gurney JK, Florio AA, Znaor A. et al. International trends in the incidence of testicular cancer: lessons from 35 years and 41 countries. Eur Urol 2019;76:615–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Hanna NH, Einhorn LH. Testicular cancer—discoveries and updates. N Engl J Med 2014;371:2005–16. [DOI] [PubMed] [Google Scholar]
- 109. Rapley EA, Turnbull C, Al Olama AA. et al. A genome-wide association study of testicular germ cell tumor. Nat Genet 2009;41:807–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Gilbert D, Rapley E, Shipley J. Testicular germ cell tumours: predisposition genes and the male germ cell niche. Nat Rev Cancer 2011;11:278–88. [DOI] [PubMed] [Google Scholar]
- 111. Oosterhuis JW, Looijenga LHJ. Germ cell tumors from a developmental perspective: cells of origin, pathogenesis, and molecular biology (emerging patterns). In: Nogales FF, Jimenez RE (eds), Pathology and Biology of Human Germ Cell Tumors. Berlin, Heidelberg: Springer Berlin Heidelberg, 2017, 23–129. [Google Scholar]
- 112. Robinson M. Babies born with deformed hands spark investigation in Germany. CNN News, 2019. https://edition.cnn.com/2019/09/16/health/hand-deformities-babies-gelsenkirchen-germany-intl-scli-grm/index.html (2019, date last accessed).
- 113. Agence France-Presse in Paris . France to investigate cause of upper limb defects in babies. The Guardian [Internet], 3 November 2018. https://www.theguardian.com/world/2018/oct/21/france-to-investigate-cause-of-upper-limb-defects-in-babies (3 November 2018, date last accessed).
- 114. Gant J. Scientists are baffled by spatter of babies born without hands or arms in France, as investigation fails to discover a cause. Daily Mail [Internet], 14 July 2019. https://www.dailymail.co.uk/news/article-7242059/Scientists-baffled-babies-born-without-hands-arms-France-probe-fails-discover-cause.html (14 July 2019, date last accessed).
- 115. Willsher K. Baby arm defects prompt nationwide investigation in France. Guardian [Internet], 3 November 2018. https://www.theguardian.com/world/2018/oct/31/baby-arm-defects-prompt-nationwide-investigation-france (November 2018, date last accessed).
- 116. Rehman W, Arfons LM, Lazarus HM. The rise, fall and subsequent triumph of thalidomide: lessons learned in drug development. Ther Adv Hematol 2011;2:291–308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Ing GM, Olman CL, Oyd JR. Drug-induced (Thalidomide) malformations. Can Med Assoc J 1962;87:1259–62. [PMC free article] [PubMed] [Google Scholar]
- 118. McBride WG. Thalidomide and congenital malformations. Lancet 1962;2:1358–9.13988811 [Google Scholar]
- 119. Melchert M, List A. The thalidomide saga. Int J Biochem Cell Biol 2007;39:1489–99. [DOI] [PubMed] [Google Scholar]
- 120. Therapontos C, Erskine L, Gardner ER. et al. Thalidomide induces limb defects by preventing angiogenic outgrowth during early limb formation. Proc Natl Acad Sci U S A 2009;106:8573–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Vargesson N. Thalidomide-induced limb defects: resolving a 50-year-old puzzle. Bioessays 2009;31:1327–36. [DOI] [PubMed] [Google Scholar]
- 122. Vargesson N. Thalidomide-induced teratogenesis: history and mechanisms. Birth Defects Res C Embryo Today 2015;105:140–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. David AL, Holloway A, Thomasson L. et al. A case-control study of maternal periconceptual and pregnancy recreational drug use and fetal malformation using hair analysis. PLoS One 2014;9:e111038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Draper ES, Rankin J, Tonks AM. et al. Recreational drug use: a major risk factor for gastroschisis?. Am J Epidemiol 2008;167:485–91. [DOI] [PubMed] [Google Scholar]
- 125. Skarsgard ED, Meaney C, Bassil K. et al. Maternal risk factors for gastroschisis in Canada. Birth Defects Res A Clin Mol Teratol 2015;103:111–8. [DOI] [PubMed] [Google Scholar]
- 126. Torfs CP, Velie EM, Oechsli FW. et al. A population-based study of gastroschisis: demographic, pregnancy, and lifestyle risk factors. Teratology 1994;50:44–53. [DOI] [PubMed] [Google Scholar]
- 127. van Gelder MM, Reefhuis J, Caton AR. et al. Maternal periconceptional illicit drug use and the risk of congenital malformations. Epidemiology 2009;20:60–6. [DOI] [PubMed] [Google Scholar]
- 128. Werler MM, Sheehan JE, Mitchell AA. Association of vasoconstrictive exposures with risks of gastroschisis and small intestinal atresia. Epidemiology 2003;14:349–54. [PubMed] [Google Scholar]
- 129. Alshehri A, Emil S, Laberge JM. et al. Canadian Pediatric Surgery N. Outcomes of early versus late intestinal operations in patients with gastroschisis and intestinal atresia: results from a prospective national database. J Pediatr Surg 2013;48:2022–6. [DOI] [PubMed] [Google Scholar]
- 130. Bassil KL, Yang J, Arbour L. et al. Spatial variability of gastroschisis in Canada, 2006-2011: An exploratory analysis. Can J Public Health 2016;107:e62–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Moore A, Roulean J, Skarsgard E. Congenital anomalies in Canada, 2013. A perinatal health surveillance report. Chapter 7. In: Public Health Agency of Canada HC (eds), Gastroschisis. Ottawa: Health Canada, 2013, 57–63. [Google Scholar]
- 132. Reece AS. Cannabis as a cause of giant cystic lung disease. QJM 2008;101:503. [DOI] [PubMed] [Google Scholar]
- 133. Reece AS. Giant cystic lung disease with mediastinal compression in a short-term heavy cannabis smoker. BMJ Case Rep 2011;2011:bcr0420102934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Reece AS. Response to aldington: cannabis and lung cancer. Eur Resp J 2008;32:238–9. [DOI] [PubMed] [Google Scholar]
- 135. Seleverstov O, Tobiasz A, Jackson JS. et al. Maternal alcohol exposure during mid-pregnancy dilates fetal cerebral arteries via endocannabinoid receptors. Alcohol (Fayetteville), NY 2017;61:51–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Subbanna S, Nagre NN, Shivakumar M. et al. CB1R-mediated activation of caspase-3 causes epigenetic and neurobehavioral abnormalities in postnatal ethanol-exposed mice. Front Mol Neurosci 2018;11:45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Subbanna S, Nagre NN, Umapathy NS. et al. Ethanol exposure induces neonatal neurodegeneration by enhancing CB1R Exon1 histone H4K8 acetylation and up-regulating CB1R function causing neurobehavioral abnormalities in adult mice. Int J Neuropsychopharmacol 2014;18:5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Subbanna S, Psychoyos D, Xie S. et al. Postnatal ethanol exposure alters levels of 2-arachidonylglycerol-metabolizing enzymes and pharmacological inhibition of monoacylglycerol lipase does not cause neurodegeneration in neonatal mice. J Neurochem 2015;134:276–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Fraher D, Ellis MK, Morrison S. et al. Lipid abundance in zebrafish embryos is regulated by complementary actions of the endocannabinoid system and retinoic acid pathway. Endocrinology 2015;156:3596–609. [DOI] [PubMed] [Google Scholar]
- 140. Kučukalić S, Ferić Bojić E, Babić R. et al. Genetic susceptibility to posttraumatic stress disorder: analyses of the oxytocin receptor, retinoic acid receptor-related orphan receptor A and cannabinoid receptor 1 genes. Psychiatr Danub 2019;31:219–26. [DOI] [PubMed] [Google Scholar]
- 141. Lee YS, Jeong WI. Retinoic acids and hepatic stellate cells in liver disease. J Gastroenterol Hepatol 2012;27:75–9. [DOI] [PubMed] [Google Scholar]
- 142. Vallee A, Lecarpentier Y, Guillevin R. et al. Effects of cannabidiol interactions with Wnt/beta-catenin pathway and PPARgamma on oxidative stress and neuroinflammation in Alzheimer’s disease. Acta Biochim Biophys Sin (Shanghai) 2017;49:853–66. [DOI] [PubMed] [Google Scholar]
- 143. Nallathambi R, Mazuz M, Namdar D. et al. Identification of synergistic interaction between cannabis-derived compounds for cytotoxic activity in colorectal cancer cell lines and colon polyps that induces apoptosis-related cell death and distinct gene expression. Cannabis Cannabinoid Res 2018;3:120–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Petko J, Tranchina T, Patel G. et al. Identifying novel members of the Wntless interactome through genetic and candidate gene approaches. Brain Res Bull 2018;138:96–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Xian X, Tang L, Wu C. et al. miR-23b-3p and miR-130a-5p affect cell growth, migration and invasion by targeting CB1R via the Wnt/beta-catenin signaling pathway in gastric carcinoma. Onco Targets Ther 2018;11:7503–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. McKenzie MG, Cobbs LV, Dummer PD. et al. Non-canonical Wnt signaling through Ryk regulates the generation of somatostatin- and parvalbumin-expressing cortical interneurons. Neuron 2019;103:853–64 e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Nalli Y, Dar MS, Bano N. et al. Analyzing the role of cannabinoids as modulators of Wnt/beta-catenin signaling pathway for their use in the management of neuropathic pain. Bioorg Med Chem Lett 2019;29:1043–6. [DOI] [PubMed] [Google Scholar]
- 148. Frampton G, Coufal M, Li H. et al. Opposing actions of endocannabinoids on cholangiocarcinoma growth is via the differential activation of Notch signaling. Exp Cell Res 2010;316:1465–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149. Tanveer R, Gowran A, Noonan J. et al. The endocannabinoid, anandamide, augments Notch-1 signaling in cultured cortical neurons exposed to amyloid-beta and in the cortex of aged rats. J Biol Chem 2012;287:34709–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Xapelli S, Agasse F, Sarda-Arroyo L. et al. Activation of type 1 cannabinoid receptor (CB1R) promotes neurogenesis in murine subventricular zone cell cultures. PLoS One 2013;8:e63529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Kim D, Lim S, Park M. et al. Ubiquitination-dependent CARM1 degradation facilitates Notch1-mediated podocyte apoptosis in diabetic nephropathy. Cell Signal 2014;26:1774–82. [DOI] [PubMed] [Google Scholar]
- 152. Niu F, Zhao S, Xu CY. et al. Potentiation of the antitumor activity of adriamycin against osteosarcoma by cannabinoid WIN-55,212-2. Oncol Lett 2015;10:2415–21. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 153. Aguado T, Romero E, Monory K. et al. The CB1 cannabinoid receptor mediates excitotoxicity-induced neural progenitor proliferation and neurogenesis. J Biol Chem 2007;282:23892–8. [DOI] [PubMed] [Google Scholar]
- 154. Williams EJ, Walsh FS, Doherty P. The FGF receptor uses the endocannabinoid signaling system to couple to an axonal growth response. J Cell Biol 2003;160:481–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Asimaki O, Leondaritis G, Lois G. et al. Cannabinoid 1 receptor-dependent transactivation of fibroblast growth factor receptor 1 emanates from lipid rafts and amplifies extracellular signal-regulated kinase 1/2 activation in embryonic cortical neurons. J Neurochem 2011;116:866–73. [DOI] [PubMed] [Google Scholar]
- 156. Birerdinc A, Jarrar M, Stotish T. et al. Manipulating molecular switches in brown adipocytes and their precursors: a therapeutic potential. Prog Lipid Res 2013;52:51–61. [DOI] [PubMed] [Google Scholar]
- 157. Richard D, Picard F. Brown fat biology and thermogenesis. Front Biosci (Landmark Ed) 2011;16:1233–60. [DOI] [PubMed] [Google Scholar]
- 158. Xu TR, Yang Y, Ward R. et al. Orexin receptors: multi-functional therapeutic targets for sleeping disorders, eating disorders, drug addiction, cancers and other physiological disorders. Cell Signal 2013;25:2413–23. [DOI] [PubMed] [Google Scholar]
- 159. Hernán MA. Methods of public health research — strengthening causal inference from observational data. N Engl J Med 2021;385:1345–8. [DOI] [PubMed] [Google Scholar]
- 160. Pearl J, Mackaenzie D. The Book of Why. The New Science of Cause and Effect. New York: Basic Books, 2019, P. 418. [Google Scholar]
- 161. Cutando L, Maldonado R, Ozaita A. Microglial activation and cannabis exposure. In: Preedy V (ed.), Handbook of Cannabis and Related Pathologies: Biology, Pharmacology, Diagnosis and Treatment. 1. New York: Academic Press, 2017, 401–12. [Google Scholar]
- 162. Reece AS. Chronic toxicology of cannabis. Clin Toxicol (Phila) 2009;47:517–24. [DOI] [PubMed] [Google Scholar]
- 163. Zumbrun EE, Sido JM, Nagarkatti PS. et al. Epigenetic regulation of immunological alterations following prenatal exposure to marijuana cannabinoids and its long term consequences in offspring. J Neuroimmune Pharmacol 2015;10:245–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164. Miller ML, Chadwick B, Dickstein DL. et al. Adolescent exposure to Delta(9)-tetrahydrocannabinol alters the transcriptional trajectory and dendritic architecture of prefrontal pyramidal neurons. Mol Psychiatry 2018;24:588–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165. Richardson KA, Hester AK, McLemore GL. Prenatal cannabis exposure - The "first hit" to the endocannabinoid system. Neurotoxicol Teratol 2016;58:5–14. [DOI] [PubMed] [Google Scholar]
- 166. Cecil CA, Walton E, Smith RG. et al. DNA methylation and substance-use risk: a prospective, genome-wide study spanning gestation to adolescence. Transl Psychiatry 2016;6:e976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167. Fernandez-Ruiz J, Gomez M, Hernandez M. et al. Cannabinoids and gene expression during brain development. Neurotox Res 2004;6:389–401. [DOI] [PubMed] [Google Scholar]
- 168. Zimmerman AM, Stich H, San R. Nonmutagenic action of cannabinoids in vitro. Pharmacology 1978;16:333–43. [DOI] [PubMed] [Google Scholar]
- 169. Zimmerman S, Zimmerman AM. Genetic effects of marijuana. Int J Addict 1990;25:19–33. [DOI] [PubMed] [Google Scholar]
- 170. Ali M, Boosi Narayana Rao K, Majumder P. et al. Alterations in inter-organelle crosstalk and Ca(2+) signaling through mitochondria during proteotoxic stresses. Mitochondrion 2021;57:37–46. [DOI] [PubMed] [Google Scholar]
- 171. Bocklandt S, Lin W, Sehl ME. et al. Epigenetic predictor of age. PLoS One 2011;6:e14821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172. Horvath S. DNA methylation age of human tissues and cell types. Genome Biol 2013;14:R115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173. Teschendorff AE, West J, Beck S. Age-associated epigenetic drift: implications, and a case of epigenetic thrift?. Hum Mol Genet 2013;22:R7–R15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174. Hou H, Yang H, Liu P. et al. Profile of immunoglobulin G N-glycome in COVID-19 patients: a case-control study. Front Immunol 2021;12:748566–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175. Reece AS, Wang W, Hulse GK. Pathways from epigenomics and glycobiology towards novel biomarkers of addiction and its radical cure. Med Hypotheses 2018;116:10–21. [DOI] [PubMed] [Google Scholar]
- 176. Meccariello R, Santoro A, D’Angelo S. et al. The epigenetics of the endocannabinoid system. Int J Mol Sci 2020;21:1113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177. Nahas GG. Cannabis physiopathology epidemiology detection. CRC Press Revivals 1990;1990. [Google Scholar]
- 178. Nahas GG. Keep off the Grass. Middlebury: Paul S. Eriksson, 1990, P. 300. [Google Scholar]
- 179. Nahas GG, Morishima A, Desoize B. Effects of cannabinoids on macromolecular synthesis and replication of cultured lymphocytes. Fed Proc 1977;36:1748–52. [PubMed] [Google Scholar]
- 180. Nahas GG, Suciu-Foca N, Armand JP. et al. Inhibition of cellular mediated immunity in marihuana smokers. Science 1974;183:419–20. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Input and output data have been provided online through the Mendeley data repository doi: 10.17632/vd6mt5r5jm.1. Four files with R source code running to 18 830 lines of code are also supplied.
All data generated or analysed during this study are included in this published article and its supplementary information files. Data along with the relevant R code have been made publicly available on the Mendeley Database Repository and can be accessed from this URL doi: 10.17632/vd6mt5r5jm.1.