Abstract
Coronavirus disease 2019 (COVID-19) is characterized by heterogeneity of possible cardiovascular manifestations. Spontaneous coronary artery dissection is a rare cause of acute coronary syndrome, the development of which in patients with COVID-19 has been described and studied insufficiently. A 35-year-old male patient presented to our hospital with an acute coronary syndrome a few weeks after mild COVID-19. According to coronary angiography, a dissection of ramus intermedius was detected. Successful stenting was performed. Subsequently, the patient had relapses of chest pain, which led to two repeated coronary angiographies. The patient had been diagnosed with consecutive dissections of right coronary artery and distal branch of ramus intermedius. Repeated stenting of dissected segments of right coronary artery and ramus intermedius was not performed. Afterward, the patient’s condition remained stable and he was successfully discharged. One of the main pathophysiological mechanisms of cardiovascular complications in COVID-19 is probably the virus-triggered hyperinflammation and massive release of cytokines. A systemic inflammatory response may initiate inflammation of the vascular wall and other target tissues. The results of histological studies confirm the direct infection of endothelial cells 2019-nCoV with the development of diffuse endothelial inflammation (endotheliitis). It is possible that in patients with a genetic predisposition to artery dissection, COVID-19 may be a trigger of spontaneous coronary artery dissection.
Keywords: Acute coronary syndrome, coronavirus disease 2019, spontaneous coronary artery dissection, case report
Introduction
According to currently limited data presented, coronavirus disease 2019 (COVID-19) is characterized by heterogeneity of possible cardiovascular manifestations: acute coronary syndrome (ACS), myocarditis, cardiomyopathy, and so on. 1 According to the data obtained in the most affected regions of Italy and China, the number of hospitalizations for ACS and percutaneous coronary interventions (PCI) during the COVID-19 outbreak is significantly reduced (by 30%–50%), which has a number of likely explanations. 2 Nevertheless, taking into account the results of previously published meta-analyses that demonstrated the negative impact of seasonal epidemics of influenza virus and other viral respiratory diseases on cardiovascular morbidity and mortality, the delayed negative effect of COVID-19 can be predicted.3–5
In the absence of systematic data on the possible impact of COVID-19 on the risk of ACS, as well as the clinical and pathophysiological features of the course of ACS in patients with an established diagnosis of an acute respiratory syndrome caused by the new coronavirus (SARS-CoV-2), a description of individual clinical cases is one of the important and accessible sources of information.
Case presentation
In the morning of 8 May, a 35-year-old man was admitted to the emergency department complaining of episodes of pressure pain behind the sternum, lasting from 2–5 min to 1 h; the pain radiated to the left upper limb, arose after gardening works and persisted both at rest and during moderate physical exertion.
It is known from the anamnesis that, since April 2019, the patient had complaints of blurred vision; the patient was observed by an ophthalmologist with a diagnosis Serpiginous Choroiditis of both eyes. He was initially received pulse therapy with glucocorticoids (methylprednisolone in a cumulative dose of 1000 mg); then, he administrated methylprednisolone at a dose of 4 mg/day per os. On 20 April, the patient noted weakness, fever up to 38°C, nasal congestion, anosmia, dry cough, and chest congestion. The repeated polymerase chain reaction (PCR) test for 2019-nCoV was positive. The patient was hospitalized at the clinic for patients with COVID-19, where he was staying from 21 April to 3 May 2020, with a diagnosis of Coronavirus disease 2019 (mild case), nasopharyngitis. During hospitalization, computed tomography (CT) scanning of the chest was performed, according to which no pathological changes were detected. In the course of hospitalization, there was a moderate increase of C-reactive protein (up to 12.5 mg/L, reference values—up to 8.0 mg/L). During hospitalization for COVID-2019, the patient received hydroxychloroquine and clarithromycin.
It is also known from the anamnesis that the patient has no established cardiovascular diseases. Among cardiovascular risk factors were active smoking for 26 years (20 cigarettes/day), overweight (height = 171 cm, weight = 85 kg, body mass index (BMI) = 29.1 kg/m2). The patient denied the use of psychoactive substances (cocaine, etc.).
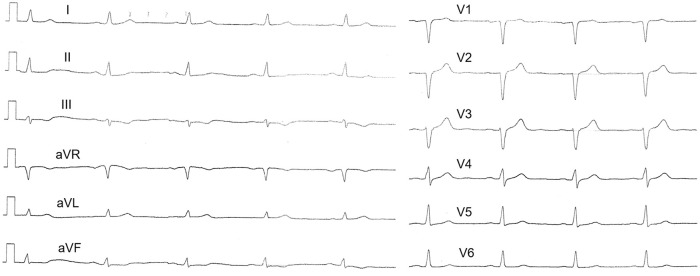
On admission to the emergency department on 8 May, according to objective examination, the parameters were as follows: blood pressure = 126/78 mm Hg, heart rate = 84 beats per minute, respiratory rate = 18 per minute, additional respiratory sounds in the lungs were not auscultated, saturation (on room air) = 99%. On admission, a high-sensitive cardiac troponin was within the reference values. The patient was given loading doses of acetylsalicylic acid, clopidogrel, and heparin. The electrocardiogram (ECG) on admission is shown in Figure 1.
Figure 1.
ECG on admission. Slight ST segment elevation in the right precordial leads with reciprocal changes in the lateral wall of left ventricle.
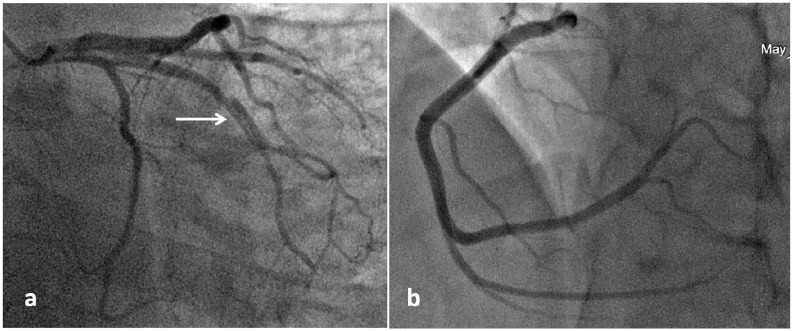
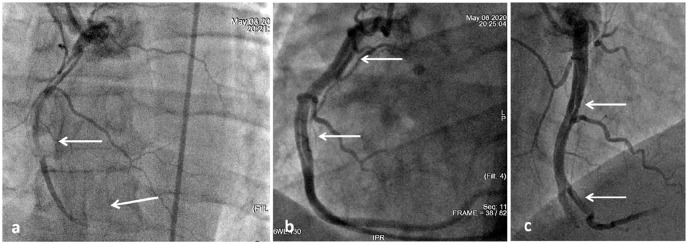
Taking into account the clinical picture of ACS with ST segment elevation, the patient underwent coronary angiography. A linear dissection of ramus intermedius (RI) was established over 20 mm with stenosis of the lumen up to 80% in diameter against the background of thrombolysis in myocardial infarction (TIMI) 3 blood flow in all coronary arteries (Figure 2).
Figure 2.
Coronary angiography on admission: (a) RI dissection is indicated by a white arrow and (b) intact right coronary artery.
It was decided to stent RI dissection area with a drug-coated sirolimus stent. TIMI 3 blood flow on control angiograms is shown in Figure 3. The results of laboratory tests are presented in Table 1.
Figure 3.
Stenting of RI: (a) the arrow indicates the inflated balloon at the dissection segment and (b) control angiogram after stenting of RI (stent is indicated by an arrow).
Table 1.
Laboratory test results.
| Parameter, units | Values | Reference values |
|---|---|---|
| Red blood cells, n × 109/L | 5.57 | 4.0–5.6 |
| Hemoglobin, g/L | 158 | 130–170 |
| White blood cells, n × 109/L | 10.8 | 4.0–9.0 |
| Cardiac troponin I at admission, ng/mL | 0.00 | <0.05 |
| Cardiac troponin I at 6 h, ng/mL | 2.98 | <0.05 |
| Cardiac troponin I at 12 h, ng/mL | 3.35 | <0.05 |
| D-dimer, ng/mL | 234 | <250 |
| Creatinine, µmol/L | 98 | <120 |
| eGFR, mL/min/1.73 m2 | 86 | >90 |
| Glucose, mmol/L | 5.6 | <6.1 |
| Uric acid, µmol/L | 372 | <420 |
| Total cholesterol, mmol/L | 4.16 | – |
| HDL-C, mmol/L | 0.74 | – |
| Triglycerides, mmol/L | 1.52 | – |
| LDL-C, mmol/L | 1.98 | – |
| C-reactive protein, mg/L | 5.00 | <8.00 |
eGFR: estimated glomerular filtration rate (CKD-EPI); HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol.
Subsequently, the patient received therapy consistent with the clinical recommendations, including triple antithrombotic therapy using acetylsalicylic acid, clopidogrel, and infusion of unfractionated heparin intravenously.
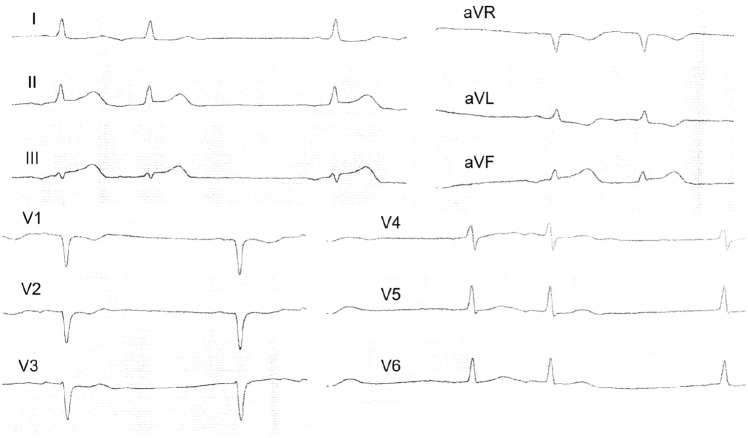
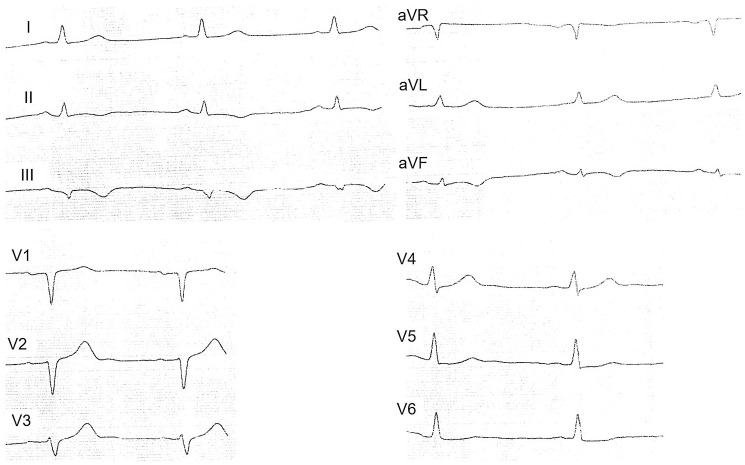
At 7:20 p.m. of the same day, in the intensive care unit, the patient again had anginal pain syndrome without significant effect from nitroglycerin; the pain was relieved with morphine. ECG was performed (see Figure 4).
Figure 4.
ECG during the recurrence of chest pain. The ST segment elevation in the leads II, III, and aVF with reciprocal changes.
Given the appearance of ST segment elevation in the leads from the inferior wall of the left ventricle, the patient underwent repeated angiography (see Figure 5).
Figure 5.
Repeated coronary angiography: (a) defect of contrasting of the RCA with stenosis by 90% (upper white arrow), occlusion of the distal segment (lower white arrow); (b) and (c) linear dissections of the RCA and the initial segment of the posterior lateral branch of the RCA (indicated by arrows).
It was established that the stented segment and other sections of the left coronary artery (LCA) had no stenosis. Blood flow along the right coronary artery (RCA) TIMI 0–1 with occlusion in the distal section. Also, stenosis of the initial and middle sections of the RCA was detected (up to 60%–75%) over the entire length according to the type of possible spasm in comparison with the initial coronary angiograms. After two injections of contrast into the RCA and 2.0 mL of a 0.01% solution of nitroglycerin, recanalization was observed with the restoration of the initial caliber of the artery with visualization of linear dissections throughout the initial and middle sections. According to the control angiography, after 15 min (see Figure 6), a decrease in the length of the dissected segment in the initial and middle sections of the RCA and posterior lateral branch of the RCA was established. Taking into account the TIMI 3 blood flow, extended lesions of the RCA, and the posterolateral branch of the RCA, spontaneous positive dynamics in relation to this dissected segment, conservative management were applied.
Figure 6.
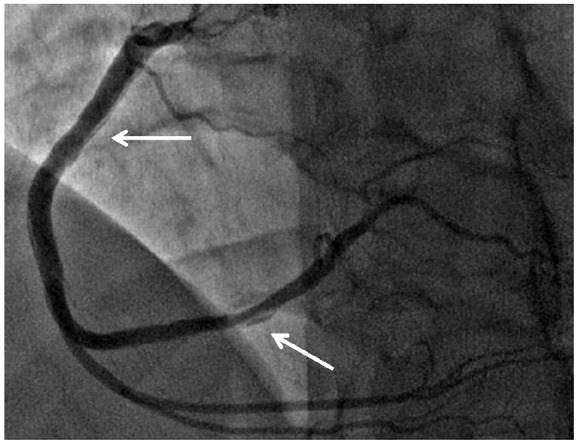
Dissection of the RCA after 15 min. Dissection is indicated by white arrows.
Over the next 72 h, the patient’s condition remained stable, and chest pain did not recur. In the morning of 14 May, the patient again felt a pain behind the sternum, lasting up to 10–15 min and stopping on its own. ECG was performed, and found that there were no significant dynamic changes from previous ECGs (see Figure 7).
Figure 7.
ECG from 14 May 2020.
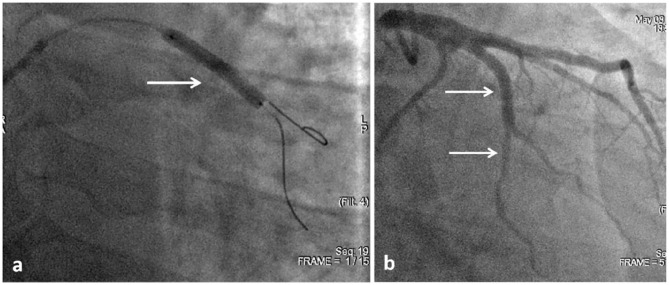
It was decided to conduct control angiography. It was detected that there was no change either in the RCA or the posterolateral branch of the RCA in comparison with the previous examination. The stented segment of the RI LCA was passable; however, in one of the RI branches, immediately after its bifurcation, stenosis of up to 85% in diameter with a length of 20–25 mm was detected, which was absent in the previous two angiograms (see Figure 8). With intracoronary administration of 2 mL of 0.01% nitroglycerin, there were no changes in the degree of stenosis in the affected artery.
Figure 8.
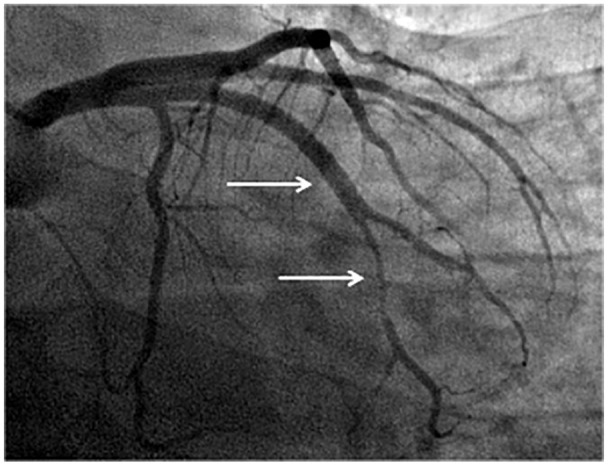
Angiography from 14 May 2020. The stented segment of the RI is passed without contrast defects (upper white arrow); in one of the branches of RI stenosis with a length of 20–25 mm up to 85% in diameter (lower white arrow).
Subsequently, anginal pain did not recur. The patient was discharged from the clinic on 20 May 2020. Written informed consent has been obtained from the patient for publication of the case report and accompanying images.
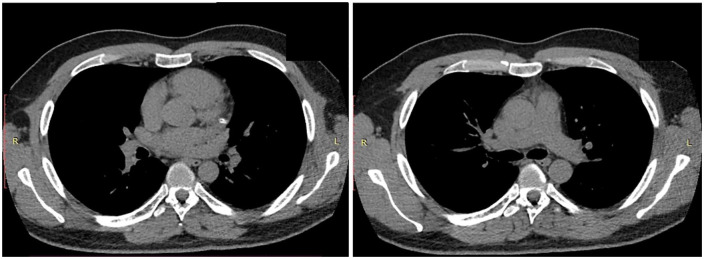
It should be noted that, during the hospitalization, the patient was consulted by a rheumatologist. Additional examinations were performed (antinuclear antibodies, HLA-B27 test, rheumatoid factor, antibodies to β2-macroglobulin—all tests were negative), which allowed the rheumatologist to exclude the presence of chronic systemic inflammatory disease. CT of the chest in dynamics from April 2020 was also performed, which also showed no pathological changes (see Figure 9).
Figure 9.
Chest computed tomography (17 May).
Discussion
Early clinical experience suggests that cardiovascular complications are one of the leading causes of death associated with COVID-19. 6 In such a case, adverse cardiovascular events, including ACS, developing after the resolution of COVID-19 (as in the case of influenza), may also have a significant effect on the long-term survival of patients. 7 Spontaneous coronary artery dissection (SCAD) is a rare cause of ACS, which, according to various sources, accounts for 1%–4% of cases, mainly among women. 8 The causes and pathogenesis of SCAD have not yet been specifically established; however, among other things, it is known about the frequent association of SCAD and chronic systemic inflammatory diseases. 9 In addition, in 2019, the first SCAD genetic risk factor was identified, associated with the PHACTR1/EDN1 gene variant. 10 This may indicate that the most important mechanisms for the development of SCAD are impaired angiogenesis, connective tissue metabolism in the vascular wall, and regulation of the vascular tone.
Until now, the results of several clinical cases of SCAD in patients with COVID-19 have been published. In one of the described clinical cases, spontaneous dissection of RCA (intramural hematoma with rupture of intima) was detected in a patient with peripheral artery disease and SARS-CoV-2. 11 In the case described by K. Kumar et al., 12 left anterior descending artery dissection was observed in a 48-year-old woman without established cardiovascular disease with COVID-19, which was asymptomatic. L. Fernandez Gasso et al. 13 described a case of spontaneous dissection of the first obtuse marginal branch artery and distal left anterior descending artery in a 39-year-old patient on the eighth day of severe SARS-CoV-2 case. The case we have presented is unique for several reasons. A multivascular SCAD was demonstrated in a young patient without established cardiovascular diseases, which developed after the resolution of the COVID-19 mild case. The dynamic performance of angiography due to the course of the disease also provides important information on the sequential development of multivascular SCAD amid invasive intervention.
Now, it is generally known that COVID-19 is characterized by a variety of possible cardiovascular manifestations. 14 One of the main pathophysiological mechanisms of development thereof is probably the virus-triggered hyperinflammation and massive release of cytokines. 15 A systemic inflammatory response may initiate inflammation of the vascular wall and other target tissues. It is known that 2019-nCoV uses the ACE2 receptor for the invasion of the cell, which is also expressed on endothelial cells and vascular smooth muscle cells. 16 The results of histological studies confirm the direct infection of endothelial cells 2019-nCoV with the development of diffuse endothelial inflammation (endotheliitis). 17 Endothelial dysfunction and massive death of endotheliocytes cause the development of disorders in the hemostatic system, regulation of the vascular tone, and so on. These changes can be observed in the coronary arteries and their vasa vasorum. 18 Depending on the initial status of the patient and the “vulnerabilities” they have, these factors may cause various cardiovascular events. So, it is possible that in patients with severe atherosclerosis, atherosclerotic plaque destabilization may start with the development of various ischemic catastrophes; in patients with a genetic predisposition to dissection, spontaneous artery dissection may develop.10,19
Another important aspect of the described case is the indication of taking glucocorticoids in pulse therapy 1 month before COVID-19 disease and more than 1.5 months before SCAD. A case of SCAD in a young patient that developed after intravenous administration of hydrocortisone in a total dose of 150 mg for optic neuritis was previously described. 20 Taking into account a number of cardiovascular side effects of glucocorticoids and data on their possible effect on the development of aneurysm and aortic dissection, a number of authors suggest that taking high doses of glucocorticoids can be considered as a potential SCAD trigger.21–23
Thus, in this clinical case, there are at least several potential SCAD triggers. However, long intervals between exposure thereof and the cardiovascular event make it difficult to establish possible causal relationships and assess the contribution of each of these factors.
Conclusion
Thus, in a young patient after COVID-19, we observed left ventricular myocardial infarction caused by spontaneous dissection of RI and RCA. Currently, there are no recognized recommendations on how to guide such a group of patients. The accumulation of clinical experience will allow us to develop a unified approach to treating this group of patients.
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval: Our institution does not require ethical approval for reporting individual cases or case series.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent: Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
ORCID iD: Vadim Genkel  https://orcid.org/0000-0001-5902-3803
https://orcid.org/0000-0001-5902-3803
References
- 1. Mahmud E, Dauerman HL, Welt FG, et al. Management of acute myocardial infarction during the COVID-19 pandemic. J Am Coll Cardiol 2020; 76: 1375–1384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Piccolo R, Bruzzese D, Mauro C, et al. Population trends in rates of percutaneous coronary revascularization for acute coronary syndromes associated with the COVID-19 outbreak. Circulation 2020; 141: 2035–2037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Nguyen JL, Yang W, Ito K, et al. Seasonal influenza infections and cardiovascular disease mortality. JAMA Cardiol 2016; 1: 274–281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Zheng YY, Ma YT, Zhang JY, et al. COVID-19 and the cardiovascular system. Nat Rev Cardiol 2020; 17(5): 259–260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Barnes M, Heywood AE, Mahimbo A, et al. Acute myocardial infarction and influenza: a meta-analysis of case-control studies. Heart 2015; 101(21): 1738–1747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Paramasivam A, Priyadharsini JV, Raghunandhakumar S, et al. A novel COVID-19 and its effects on cardiovascular disease. Hypertens Res 2020; 43(7): 729–730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Zhu H, Rhee JW, Cheng P, et al. Cardiovascular complications in patients with COVID-19: consequences of viral toxicities and host immune response. Curr Cardiol Rep 2020; 22(5): 32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Gilhofer TS, Saw J. Spontaneous coronary artery dissection: update 2019. Curr Opin Cardiol 2019; 34(6): 594–602. [DOI] [PubMed] [Google Scholar]
- 9. Garcia-Guimarães M, Bastante T, Antuña P, et al. Spontaneous coronary artery dissection: mechanisms, diagnosis and management. Eur Cardiol 2020; 15: e03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Adlam D, Olson TM, Combaret N, et al. Association of the PHACTR1/EDN1 genetic locus with spontaneous coronary artery dissection. J Am Coll Cardiol 2019; 73: 58–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Courand PY, Harbaoui B, Bonnet M, et al. Spontaneous coronary artery dissection in a patient with COVID-19. J Am Coll Cardiol Cardiovasc Interv 2020; 13: e107–e108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kumar K, Vogt JC, Divanji PH, et al. Spontaneous coronary artery dissection of the left anterior descending artery in a patient with COVID-19 infection. Catheter Cardiovasc Interv. Epub ahead of print 7 May 2020. DOI: 10.1002/ccd.28960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Fernandez Gasso L, Maneiro Melon NM, Sarnago Cebada F, et al. Multivessel spontaneous coronary artery dissection presenting in a patient with severe acute SARS-CoV-2 respiratory infection. Eur Heart J 2020; 41: 3100–3101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Fried JA, Ramasubbu K, Bhatt R, et al. The variety of cardiovascular presentations of COVID-19. Circulation 2020; 141: 1930–1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kang Y, Chen T, Mui D, et al. Cardiovascular manifestations and treatment considerations in COVID-19. Heart 2020; 106: 1132–1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020; 181(2): 271–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Varga Z, Flammer AJ, Steiger P, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020; 395:1417–1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Mulligan-Kehoe MJ, Simons M. Vasa vasorum in normal and diseased arteries. Circulation 2014; 129(24): 2557–2566. [DOI] [PubMed] [Google Scholar]
- 19. Turley TN, O’Byrne MM, Kosel ML, et al. Identification of susceptibility loci for spontaneous coronary artery dissection. JAMA Cardiol 2020; 5: 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Keir ML, Dehghani P. Corticosteroids and spontaneous coronary artery dissection: a new predisposing factor? Can J Cardiol 2016; 32(3): 395.e7–395.e8. [DOI] [PubMed] [Google Scholar]
- 21. Macaya F, Salinas P, Gonzalo N, et al. Spontaneous coronary artery dissection: contemporary aspects of diagnosis and patient management. Open Heart 2018; 5(2): e000884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Saçar M, Baltalarlı M, Adalı F, et al. Acute aortic dissection due to long-term steroid therapy for Addison’s disease. Turk Gogus Kalp Dama 2011; 19: 086–088. [Google Scholar]
- 23. Verlaeckt E, Van de Bruaene L, Coeman M, et al. Spontaneous coronary artery dissection in a patient with hereditary polycystic kidney disease and a recent liver transplant: a case report. Eur Heart J Case Rep 2019; 3(4): 1–5. [DOI] [PMC free article] [PubMed] [Google Scholar]