Abstract
In endocrine and reproductive endocrine literature, adult female acne is considered as a possible clinical expression of hyperandrogenism, with most polycystic ovary syndrome (PCOS) guidelines considering acne as a condition of androgen excess. Adult female acne, however, in the dermatological literature is considered as an inflammatory skin disease and new guidelines on adult female acne have been produced by dermatological societies, with little perspective from any endocrine or reproductive endocrine points of view. An expert task force was appointed by the AE-PCOS society to determine the current state of knowledge and provide evidence-based recommendations that could be valid for all specialists taking care of female adult acne. The following are the recommendations (level of evidence A or B): (1) diagnosis of female adult acne is mainly clinical, but a grading tool is needed for optimizing the treatment; (2) measurement of serum androgen values (total testosterone, free testosterone, and dehydroepiandrosterone sulfate) by high-quality assays is recommended in all women with adult acne; (3) in women with adult acne and proven hyperandrogenism, oral combined estroprogestins should be added to the topical or systemic treatment of acne, independently of severity of acne; (4) all second- and third-generation estroprogestins may be used, independently of the estrogen dose and progestin component; (5) spironolactone may be added to estroprogestins in women with moderate or severe hyperandrogenic adult acne, not responding to usual treatments; (6) estroprogestins may be used in nonhyperandrogenic patients with adult acne as second-line therapy.
Keywords: acne, female adult acne, hyperandrogenism, polycystic ovary syndrome, estroprogestins, antiandrogens
In endocrine and reproductive endocrine literature, adult female acne is considered as a possible clinical expression of hyperandrogenism (androgen excess), with most polycystic ovary syndrome (PCOS) guidelines considering acne as a condition of androgen excess [1-4]. Adult female acne, however, in the dermatological literature is considered as an inflammatory skin disease determined by several factors that may or may not include hyperandrogenism. Recently, new guidelines on diagnosing and treating adult female acne have been produced by dermatological societies [5, 6] with little perspective from any endocrine or reproductive endocrine points-of-view.
Therefore, the Androgen Excess and PCOS (AE-PCOS) Society appointed an expert task force not only to evaluate the current state of knowledge regarding the relationship of hyperandrogenism with adult female acne, but also to propose consistent strategies to evaluate and treat these patients. This publication provides evidence-based and expert opinion–based recommendations for the evaluation, diagnosis, and treatment of adult female acne, and its association with hyperandrogenism, for use by healthcare providers of different specialties who care for these women.
Methods
AE-PCOS Society Task force
The AE-PCOS Society Board appointed an expert task force from pertinent medical disciplines (dermatology, endocrinology, reproductive endocrinology) to determine the current state of knowledge concerning adult female acne and its relationship to androgen excess, and to provide evidence-based recommendations that could be valid for all specialists taking care of this disorder. Members of the Task force were selected from those researchers who had authored many original articles in the field. All members of the task force declared no conflicts of interest or financial interests that might interfere with their objectivity and collaborative effort.
Process
Each section of this review was prepared by at least 2 investigators and then reviewed by all members of the task force. Analyzed papers included individual studies, systematic reviews, and abstracts published in the English scientific literature. Peer-reviewed studies evaluating adult acne vulgaris (acne) published through 2020 were reviewed. Criteria for inclusion/exclusion of the published papers relating to each section were agreed upon by at least 2 reviewers in each area and arbitrated by a third when necessary. Levels of evidence of the were assessed and graded from A to D [7, 8]. Levels of evidence A or B were needed for recommendations regarding diagnosis and therapy of female adult acne.
The final manuscript was reviewed and approved by the AE-PCOS Society Board of Directors. Institutional Review Board approval was not obtained because the study reviewed publicly available medical literature.
Results and Discussion
Pathophysiology of Adult Acne
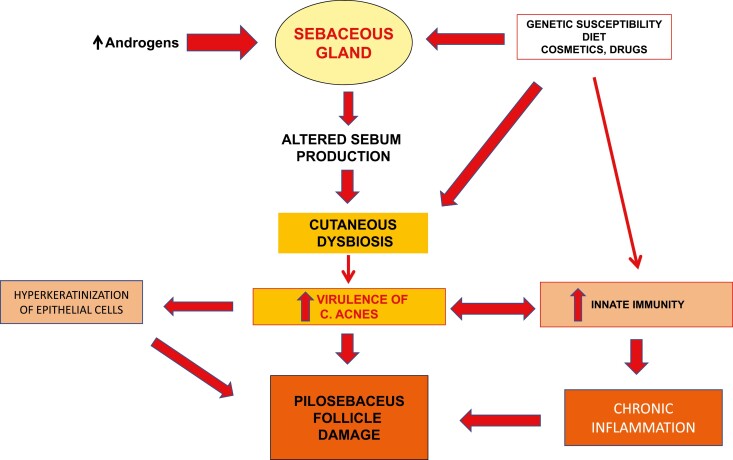
Acne is a complex and multifactorial inflammatory disease that involves several factors: (1) excessive and altered sebum production; (2) cutaneous dysbiosis with selection of virulent subtypes of Cutibacterium acnes (C. acnes) and/or of other bacterial agents; (3) abnormalities of differentiation and proliferation of keratinocytes of the pilosebaceous follicle; (4) inflammation and activation of the innate immune response. These key processes are strictly interrelated and may be influenced by several hormonal and genetic factors. In Fig. 1, the possible pathogenetic mechanisms determining female adult acne are shown.
Figure 1.
Pathophysiology of female adult acne.
1. Excessive and altered sebum production
Excessive and altered production of sebum is generally the first step in the pathophysiology of acne [9-11]. Sebocytes, the major cell type in sebaceous glands, are differentiated epithelial cells that synthesize and accumulate lipids in their cytoplasm and then undergo disintegration releasing their content (the sebum) into the follicle. Human sebum is mainly composed of triglycerides and fatty acids (57.5%) but also of cholesterol esters and squalene. Via the hair canal, sebum reaches the skin surface, where it has several functions, including antimicrobial and anti-inflammatory activity [9, 12, 13].
Increased sebum production correlates with the severity of clinical manifestations of acne [14]. However, it is insufficient to produce the inflammatory processes characteristic of acne. Rather, an altered sebum lipid profile also is required, which not only reduces the protective barrier function of the skin but actively participates in the inflammation that is essential for the development of acne [9-11, 15, 16]. Several studies examining the type of altered sebum lipid production accompanying acne have identified an increased ratio of saturated/mono-unsaturated fatty acids, a reduction of linoleic acid and an increased formation of squalene and lipid peroxides [16].
2. Cutaneous dysbiosis with selection of virulent subtypes of Cutibacterium acnes (C. acnes) and/or of other bacterial agents
Cutibacterium acnes (previously known as Propionibacterium acnes) has been identified as the most common bacterial agent activating the inflammatory and immune responses that are essential in acne pathogenesis [17, 18].
However, C. acnes is part of the normal skin microbiota [19] and acne onset and progression require cutaneous dysbiosis favoring the selection of more virulent C. acnes strains [20, 21]. Therefore, acne is associated with both a loss of diversity of skin microbiota due to selection of a predominant phylotype 1A1 of C. acnes, and activation of virulent genes within these bacterial subtypes [20]. With altered sebum influencing the skin microbiome composition, sebum triglycerides are broken down into free fatty acids by virulent C. acnes strains to perpetuate further C. acnes colonization and promote inflammation [11].
Other bacteria may be important in this process, since proliferation or selection of virulent strains of other bacteria such as Staphylococcus epidermidis, Staphylococcus aureus, Klebsiella pneumoniae, Streptococcus, and Enterobacter may be found in acne patients [19-21]. It has been recently reported that adult acne on the back is often linked to cutaneous dysbiosis with predominance of virulent strains of Enterobacter [21].
3. Abnormalities of differentiation and proliferation of keratinocytes
Acne is not only characterized by inflammatory skin lesions but also by the formation of comedones. C. acnes plays an important role in this process favoring hyperkeratinization of epithelial cells and resultant clogging of the pilosebaceous follicle, ultimately leading to the formation of comedones [15] as well as activation of several immune and inflammatory responses.
4. Inflammation and activation of innate immune response
Cutaneous dysbiosis with selection of virulent strains of C. acnes and of other bacteria induces activation of innate immunity, several factors of which directly lead to inflammation and pathological damage [10, 15, 20, 22, 23].
The consequence is a profound alteration of the innate immune response that maintains the cutaneous dysbiosis, the inflammatory process, and the acne lesions [20, 23].
Role of androgens in pathogenesis of female adult acne
Within the pilosebaceous unit, androgen receptors are present in the dermal papilla, the outer root sheath, and the sebaceous glands [24, 25]. 5α-Reductase isoforms, type 1 and type 2, also are present and can convert testosterone to dihydrotestosterone [24], thereby amplifying local androgen action. Specifically, both 5α-reductase isoforms exist in the outer root sheath, and to a lesser extent in the dermal papilla, with greater expression of 5α-reductase type 1 than type 2 [24, 25]. In addition, the skin expresses all necessary enzymes to convert dehydroepiandrosterone sulfate (DHEAS) to dihydrotestosterone [25]. Aromatase also exists in sebaceous glands and the outer root sheath of hair follicles and can diminish local androgen action through aromatization [25, 26].
Androgen metabolism within the pilosebaceous unit is further modified by several other steroid enzymes (sulfotransferase, 3β-hydroxysteroid dehydrogenase, and 17β-hydroxysteroid dehydrogenase) [25-27]. Sebaceous glands also possess receptors for several growth factors, including epidermal growth factor, insulin-like growth factor I, and keratinocyte growth factor, which further modify sebum production [25].
Hyperandrogenism favors the development of acne by increasing sebum production but mainly by altering sebum lipid profile [28]. The correlation between androgen excess and development of adult female acne is well documented [28-32]. Because in most instances androgen excess starts during puberty, persistent adult female acne (a form of adult acne beginning during adolescence and continuing until adulthood) is associated more with hyperandrogenism than adult female acne beginning in later life [33].
During puberty, acne is more common than during adulthood but is less correlated with androgen excess. It has been suggested that adolescent acne depends on rapid changes in androgen production with consequent increase in sebum production. However, other hormones like insulin like growth factor-1 may play a major role in adolescent (and also be important in adult) acne [34, 35].
In adult female acne, circulating levels of an androgen metabolite, androsterone glucuronide, largely derived from androstenedione, have been found preferentially increased [36-38]. Interestingly, androsterone glucuronide levels are increased in up to 60% of the patients with normal androgen levels [38], and this has been interpreted as indicating increased androgen sensitivity. Consistently with this hypothesis, estroprogestins may improve acne in patients with normal androgen levels [5, 39].
It is also possible that alternative pathways of androgen production are increased in women with adult acne [28]. New pathways of androgen production are currently being studied, and 11-oxo androgens of adrenal origin have been found to be quantitatively greater in the circulation than levels of the usual androgens assessed [40]. No data on oxy-androgen levels in adult female acne are available and more studies are needed to understand the mechanisms that may link androgens to acne also in women with normal circulating levels of the commonly measured androgens.
Conclusions. 1. Increased androgen production plays a major role in inducing the sebum alterations that participate to the pathogenesis of adult acne (level of evidence B).
Clinical Presentation of Female Adult Acne
General aspects
In adult women acne is a common problem that may represent a continuation of adolescent acne (persistent acne) often recurring several years after its disappearance in adolescence (recidivant acne) while in other patients it may first occur during adulthood (late-onset acne). Some researchers consider adult acne only the form that presents in women after age 25 years, but all patients having acne during their adult life (18 years or more) should be considered affected by adult acne [41-45]. There are 2 forms of adult acne: The first is found in 80% of patients and is characterized by the presence of superficial inflammatory lesions (papules, pustules) all over the face with few nodules. The second form, which is less common (20% of adult women with acne), is localized mainly in the mandibular region, chin, and is characterized by many closed comedones and cysts with a few inflammatory lesions [46].
In most studies, adult acne becomes less common after the 30s [47, 48], but in some patients, it may continue to a later age, and a form of menopausal acne has been reported.
Acne grading tools
In clinical practice and in studies assessing the effects of possible therapies, utilization of reliable methods for assessing the severity of acne is essential.
Many acne grading tools have been suggested [49-52], but most are of limited usefulness and there is no current consensus on which scale to use. While most clinicians utilize a simplified subjective scale dividing acne into mild, moderate, and severe forms, it would be better to rely on a semi-objective grading tool. Different methods using varying criteria have been suggested and include counting acne lesions, grading severity of acne by comparing the patient presentation with photographs or figures, multimodal imaging by use of specialized photographic equipment and computer algorithms.
Most experts believe that the use of a counting method (perhaps by a multimodal imaging system) would provide the best results for epidemiological and clinical research, while a grading method would be best suited for the clinical setting.
In a recent analysis using quality criteria [52], the best acne grading tools were the Global Evaluation Acne grading scale [53], which assesses the severity of acne on the face on a scale from 0 (no acne) to 5 (severe acne with many inflammatory lesions across the face and many nodules), and the Burke and Cunliffe counting method [54, 55], mainly if it includes a multimodal imaging assessment [56].
It has been also suggested that, in patients having mild or moderate acne according to the Global Evaluation Acne method (scores 0-3), a careful evaluation of the mandibular region may permit better scoring of adult acne [57]. This method is called AFAST (adult female acne scoring tool).
Conclusions. 1. Diagnosis of acne is mainly clinical, but a grading tool is needed for optimizing the treatment (level of evidence B).
Psychological consequences of female adult acne. the role of androgen excess and/or PCOS
Adult acne may induce significant disfigurement resulting in anxiety [58], depression [58, 59], and impaired quality of life [59].
It is unclear whether women with adult acne and hyperandrogenism experience more severe emotional distress than women with acne and no hyperandrogenism. Only limited data are available in women with PCOS and acne vs women with PCOS and no acne [58-60]. Women with PCOS have lower QOL scores for the acne domain compared to controls [61], and this relationship is mediated by depression. However, severity of acne does not always correlate with body image distress scores or self-esteem [62].
Conclusions. 1. Women with adult acne have a high prevalence of abnormal scores for anxiety, depression, and impaired quality of life (level of evidence B).
Epidemiology of Female Adult Acne
General aspects
The Global Burden of Disease Project estimated that the global prevalence of acne in women is 9.8% [63]. In a meta-analysis of 25 Chinese studies (83 008 subjects), the overall prevalence rates across all age groups were 10.2% [64]. However, prevalence of acne is strictly related to age and is remarkably high in primary and secondary students (50-55%), still high in undergraduate students (40%), and then further decreases during adult age, becoming quite uncommon in menopausal age [47, 48, 65, 66]. The prevalence of acne in adult female population is unclear but probably around 15% to 20% [47, 48, 66].
Prevalence of hyperandrogenism in women with adult acne
Female adult acne may be associated with hirsutism and available data show a prevalence of hirsutism in women with adult acne of about 20% to 30% [67-71].
Several studies have assessed androgen excess in women with adult acne, but the results are quite variable (between 18% and 88%) because of different settings and used assays [31, 72, 73]. In the largest study, 55% of 835 women with adult acne had increased androgen levels [73] and it may be estimated that prevalence of hyperandrogenism in adult acne is about 50% (level of evidence C). Finally, in a small study, 9 out 15 patients with adult acne and normal androgen levels had increased androgen metabolites suggesting some form of androgen excess or of increased androgen sensitivity [38].
Prevalence of adult acne in women with hyperandrogenism or polycystic ovary syndrome
Data on the prevalence of acne among various forms of androgen excess are also extremely variable [74, 75] depending on setting or used assays.
Because PCOS is by far the most common cause of hyperandrogenism [76], more information may be obtained by the studies that assessed prevalence of PCOS in women with adult acne. Studies in different ethnic groups, using the Rotterdam criteria, found a prevalence of 17% to 27% of PCOS in women with acne [77, 78]. Higher prevalence of PCOS (51%) has been reported in patients with severe forms of acne [79].
A recent meta-analysis has assessed the prevalence of acne in adult women with PCOS and in the general adult female population [32]. Based on 31 studies [80-110] which included data of 23 426 women with PCOS and 1 896 979 healthy controls without PCOS, the prevalence of acne among adult women with and without PCOS was 42% and 17%, respectively. Because in the same meta-analysis the overall pooled prevalence of acne among women with PCOS was 1.6-fold higher than non-PCOS counterparts [32], we estimated that the real prevalence of adult acne in women with PCOS is about 30% to 40%, These data (17% of patients with acne in adult female population and 30% to 40% in women with PCOS) are similar to those found in previous studies [47, 48, 77, 78].
There are limited data to statistically compare the prevalence of acne in nonhirsute PCOS women with nonhirsute non-PCOS controls; however, a subanalysis of the few available studies showed a similar prevalence of acne in both groups [32], suggesting that in PCOS acne is generally associated with hirsutism. It remains unclear whether acne alone may be sufficient to make diagnosis of biological hyperandrogenism as stated in PCOS guidelines. More studies are needed to clarify this important issue.
Conclusions. 1. In women with PCOS, prevalence of adult acne is higher (1.6-fold) than in the general female population (level of evidence A).
Diagnosis of Hyperandrogenism in Women with Adult Acne
In women with adult acne, because of the high prevalence of hyperandrogenism, a detailed medical history and laboratory work up is necessary for identifying patients who present androgen excess and for determining the underlying disorder.
A comprehensive reproductive history including menstrual cycle, and hormonal and ovarian sonographic evaluation is needed for determining whether the patient is affected by PCOS [1-4]. The presence of other possible cutaneous signs of androgen excess (ie, hirsutism, virilization, female pattern hair loss) may suggest that acne is linked to hyperandrogenism and/or PCOS [28, 30, 76]. If a patient is diagnosed with PCOS, guidelines for additional screening have been established [3, 4].
The AE-PCOS task force agrees that measurement of androgens in blood should be performed in all women with adult acne and should evaluate serum total and free testosterone and DHEAS levels. When measuring total testosterone, extraction and chromatography performed before immunoassay or liquid chromatography mass spectrometry are important to avoid interference by steroids of similar configuration [111, 112]. In fact, most commercial assays provide inaccurate results and should be avoided [112-114].
Circulating free testosterone concentrations are more sensitive than the measurement of total testosterone levels for establishing the presence of androgen excess [112]. Again, most commercial laboratories use direct radioimmunoassay or chemiluminescent assays that rely on the competitive binding of a nontestosterone analog for measuring free testosterone, which are notoriously inaccurate [113]. The most accurate assays compute measurement of percent free testosterone concentration as the product of the percent free testosterone and the total testosterone concentration. Measurement of the percent free testosterone requires accurate techniques (equilibrium dialysis and ultracentrifugation) [112, 114]. In any of these free testosterone assays, a high-quality total testosterone measurement is needed.
Because equilibrium dialysis assays are cumbersome and not available to many laboratories, a possible alternative is to perform concomitant measurement of sex hormone–binding globulin along with the total testosterone measurement; this permits the calculation of free androgen index [112] and/or the free testosterone concentration [114]. This estimate of free testosterone concentration has good concordance and correlation with free testosterone measured by equilibrium dialysis but requires an accurate method for measuring the total testosterone values [114].
In women with adult acne, measurement of serum DHEAS may be also useful because some studies reported increased DHEAS in many women with acne [31]. Commercial assays are generally useful for this purpose although they yield higher results than the recently available, more specific liquid chromatography mass spectrometry assays. Testing for other androgens, such as androstenedione and DHEA, does not appear to add much to the detection of hyperandrogenism in women with adult acne [30]. Similarly, testing for androgen metabolites cannot be routinely suggested.
Adult acne may depend on other hyperandrogenic disorders including nonclassic congenital adrenal hyperplasia, Cushing’s syndrome, and androgen-producing tumors [30, 112]. Extremely high androgen levels suggest the presence of an androgen secreting tumor while 17-hydroxyprogesterone measurement during the follicular phase at 8 am is needed for diagnosis of nonclassic 21-hydroxylase deficiency [112]. If signs of Cushing’s syndrome coexist, several 24-hour measurements for free urinary cortisol and/or serial salivary cortisol levels eventually followed by an overnight dexamethasone suppression test should also be performed [112].
Conclusions. 1. Measurement of serum androgen values (total testosterone, free testosterone, and DHEAS) by high-quality assays should be performed in women with adult acne (level of evidence A)
Treatment of Female Adult Acne
1. General issues
Most dermatological experts suggest a stepwise approach to female adult acne treatment that utilizes different drugs depending on the severity and the type of the individual’s acne lesions [5, 15, 115, 116]. Therefore, a careful scoring of acne before treating these patients is essential.
The general principles of this stepwise approach are simple: (1) topical agents for mild forms of acne; (2) topical agents plus an oral antibiotic or hormonal therapy for moderate forms of acne; (3) topical agents, oral antibiotics and hormonal therapy, or an oral retinoid (isotretinoin) in severe forms of acne.
The choice of topical agents depends on the type of acne, comedonal or inflammatory, as well as its severity. Benzoyl peroxide and topical antibiotics target inflammatory lesions, whereas adapalene, tretinoin, and tazarotene are retinoids that target comedones. Azelaic acid targets both.
For mild adult acne, benzoyl peroxide is often the first choice if it is well tolerated in terms of dryness. Topical antibiotics can be effective but after a short period of time bacterial resistance may occur. Because of resistance, topical antibiotics should not be used as monotherapy for acne. Most European experts do not use topical antibiotics in acne while the American Academy of Dermatology suggests using topical antibiotics only in combination with benzoyl peroxide to avoid bacterial resistance [5]. For comedones, topical retinoids should be tried in a stepwise manner starting with the least potent (and irritating) adapalene and gradually moving to the stronger tretinoin and finally, the most potent, tazarotene. Several other topical retinoids, with different potencies, are available.
In moderate forms of adult acne, oral antibiotics are often indicated. However, because of the common occurrence of antibiotic resistance, use of antibiotics that are needed for other conditions is discouraged. Doxycycline, lymecycline, or minocycline are the antibiotics of choice, but courses should be limited to 3 to 6 months at most [117, 118]. Addition of a topical retinoid will address the comedonal component of the acne if present. If there is evidence of androgen excess or other hormonal alterations, treatment with combined (estrogen and progesterone) oral contraceptives and/or antiandrogens such as spironolactone may be used with or instead of antibiotics. It must be reminded that antiandrogens have teratogenic effects and cannot be used alone but always need contraception.
In severe female adult acne, oral retinoids (primarily isotretinoin) are often the treatment of choice, but teratogenicity and other possible side effects should be carefully considered.
Some authors have suggested that antiandrogens associated with topical therapy may be a valuable alternative in both moderate and severe acne and that their use should be recommended independently on androgen values [119]. In particular, the use of spironolactone alone (without estroprogestins and oral antibiotics) is gaining popularity in some countries [120, 121]. However, no sufficient data comparing spironolactone use in normoandrogenic vs hyperandrogenic women with adult acne are available and the teratogen effect of the antiandrogen products (see below) always require contraception (generally by oral combined estroprogestins).
In selected patients, radiofrequency, laser, and light treatments are available [122] but cost and evidence for their long-term efficacy are scant.
2. Treatment of adult acne in hyperandrogenic women
In adult women, acne may be an expression of hyperandrogenism and, as previously reported, increased androgen levels are present in about 50% of the patients [73]. In addition, about 50% to 60% of women with adult acne and normal androgen levels present increased androgen metabolites suggesting some form of androgen excess [38]. Therefore, treatment of hyperandrogenism is part of all protocols of acne therapy but different guidelines exist.
The American Academy of Dermatology includes oral estroprogestin or antiandrogen therapy as second-line treatment in women with moderate or severe adult acne who do not respond to topical and antibiotic therapy [5], but independently of androgen levels (there is no recommendation to measure androgens in women with adult acne).
European dermatologists (and many American dermatologists) use hormonal treatment in patients with mild acne when there is evidence of hyperandrogenism, and in moderate acne, also without evidence of androgen excess [6, 123]. In all patients, treatment with estroprogestins or antiandrogens is used along with other acne treatment (topical or systemic depending on the severity of the acne manifestations).
In the AE-PCOS task force view, it is important to treat hyperandrogenism independently of the severity of acne, when clinical or biological evidence of it exists. Of course, these patients will also need topical and eventually systemic treatment for their acne.
The mainstay of treatment of adult acne in hyperandrogenic women is the use of combined oral estroprogestins. On a theoretical basis, less androgenic progestins should be of greater benefit, but there is no strong clinical evidence to support this. A systemic Cochrane review of randomized trials showed the superiority of estroprogestins over placebo [124]. Because estrogens have a potent effect on inhibiting sebum production, doses of ethynyl-estradiol (EE2) higher than 20 μg have been considered to be more beneficial. However, no good, randomized data support this, and the efficacy of doses lower than 20 μg of EE2 estroprogestin has been documented [125]. Probably the potency of EE2 also renders estroprogestin with low estrogen concentrations able to sufficiently suppress androgen secretion and action.
Acne can occur, paradoxically, with the use of some oral contraceptives, and this is due to a particular sensitivity to the type of progestin. In this instance, use of a less androgenic progestin, or altering the EE2 dose, is usually effective. Accordingly, while all combined oral estroprogestins have efficacy for acne on a theoretical basis, less androgenic progestins should be preferred; additionally, the estrogen dose of 20 μg of EE2 is also preferred. The choice of the estroprogestin product should be individualized according to the age, body mass index, and cardiovascular risk [126].
Use of progestins alone, such as those used for contraception (oral, injected, and implanted forms or intra-uterine devices), are not indicated for the treatment of acne, and may indeed exacerbate the problem.
Antiandrogens are not approved by the FDA for use in acne, nor are they considered as first-line therapy in all women with acne vulgaris [127] but have been extensively used by both dermatologists and endocrinologists. However, their use cannot be recommended without contraception.
The most used antiandrogen, spironolactone, has been shown to decrease sebum production [128]. Small placebo-controlled prospective studies have shown improvement in acne and sebum production in doses up to 200 mg daily for 3 months [129]. Retrospective review of 85 women receiving spironolactone (50-100 mg daily) as a single drug or adjunctive therapy for up to 24 months has shown that two-thirds of individuals experienced greater than 50% improvement in acne [130]. Another review of 64 Asian women receiving spironolactone (200 mg daily for 8 weeks with dose reduction for 12 weeks; total 20 weeks) reported good to excellent clinical improvement in all individuals [131]. A systematic review demonstrated the effectiveness of spironolactone at a dose of 100 mg per day in adult women with acne [132]. Spironolactone has few side effects (mainly polymenorrhea), but like all antiandrogens it may cause fetal harm, birth defects, incomplete masculinization of the male fetus, and decreased offspring survival, and it should be used only with contraception.
Because oral contraceptives are generally sufficient to improve sebum production and acne in hyperandrogenic adult women, the AE-PCOS task force recommends adding spironolactone only when moderate or severe adult acne is present and does not respond to the usual therapy including oral antibiotics and estroprogestins. Use of spironolactone may be also needed when other cutaneous symptoms of hyperandrogenism, such as female pattern hair loss or hirsutism, are present. When spironolactone is associated with estroprogestins, menstrual abnormalities are minimized.
Other antiandrogens, while potentially useful [30, 127], are not generally used. Flutamide has been associated with altered liver enzymes, even at a dose of 125 mg/day [28], which has been reported being the minimally effective dose [133]. In adult acne, in many countries but not in the United States, oral contraceptives containing cyproterone acetate have been largely used, but recent reports of meningiomas occurring in patients using these products have created alarm [134]. While this association has been proved only in patients using daily doses of cyproterone acetate of 10 mg or more, it is the opinion of the Committee that prolonged use of estroprogestins containing low doses of cyproterone should be avoided in patients with acne. Finasteride (5 mg daily), a 5α-reductase inhibitor of the type II isoenzyme, is not highly effective for acne [127], and data on dutasteride, a dual 5α-reductase inhibitor of type 1 and type 2 isoenzymes, in women with acne [28] are few.
There has also been recent interest in the use of topical antiandrogens. A recent phase 3 trial of topical clascoterone cream, 1%, an androgen receptor inhibitor, demonstrated efficacy and safety for adult facial acne [135] and has been approved in the United States for use in acne. However, data are few and, at the moment, such treatment cannot be recommended.
In hyperandrogenic adult acne, oral retinoids should be used only in patients with a severe form who do not respond to the associated treatment with estroprogestin and spironolactone (level of evidence C).
In patients with moderate or severe adult acne, when there is no evidence of androgen excess (no hirsutism, no increase of androgens in blood, no PCOS), oral combined estroprogestins (level of evidence B) and spironolactone (level of evidence C) may be added to the treatment when other treatments fail.
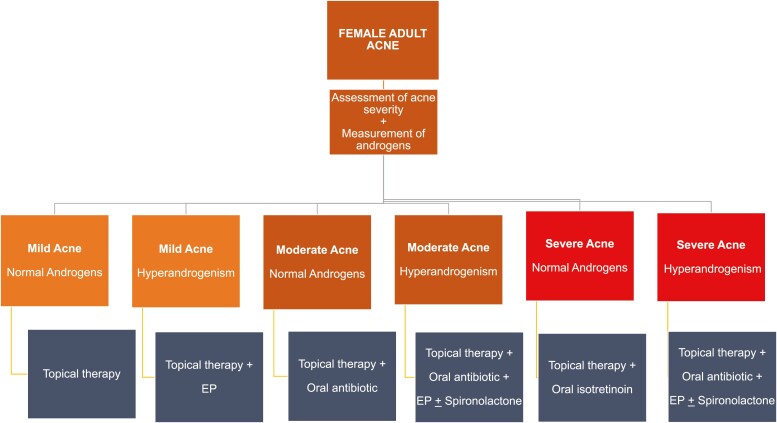
In Fig. 2, an algorithm for first-line treatment of female adult acne according to androgen levels is presented.
Figure 2.
Algorithm for first-line treatment of female adult acne according to blood androgen levels. Estroprogestins may be used in nonhyperandrogenic patients with moderate or severe adult acne as second-line therapy. EP = oral combined estroprogestins.
Conclusions. 1. In women with adult acne and proven hyperandrogenism, oral combined estroprogestins should be added to the topical or systemic treatment of acne, independently of severity of acne (level of evidence A). 2. All second and third-generation estroprogestins may be used, independently of the estrogen dose and progestin component (level of evidence B) 3. Spironolactone may be added to estroprogestins in patients with moderate or severe hyperandrogenic adult acne, not responding to usual treatment (level of evidence B) 4. Estroprogestins may be used in nonhyperandrogenic patients with adult acne as second-line therapy (level of evidence B)
Summary Recommendations for Diagnosis and Treatment of Female Adult Acne
The AE-PCOS task force, after a careful evaluation of all available data, issued some recommendations for diagnosis and treatment of female adult acne.
Diagnosis of female adult acne is mainly clinical, but a grading tool is needed for optimizing the treatment (level of evidence B).
Measurement of serum androgen values (total testosterone, free testosterone, and DHEAS) by high-quality assays is recommended in all women with adult acne (level of evidence A).
In women with adult acne and proven hyperandrogenism, oral combined estroprogestins should be added to the topical or systemic treatment of acne, independently of severity of acne (level of evidence A).
All second- and third-generation estroprogestins may be used, independently of the estrogen dose and progestin component (level of evidence B).
Spironolactone may be added to estroprogestins in women with moderate or severe hyperandrogenic adult acne not responding to usual treatments (level of evidence B).
Estroprogestins may be used in nonhyperandrogenic patients with adult acne as second-line therapy (level of evidence B).
Glossary
Abbreviations
- DHEAS
dehydroepiandrosterone sulfate
- EE2
ethynyl-estradiol
- PCOS
polycystic ovary syndrome
Financial Support
No specific funding was sought for the study.
Author contributions
E.C. organized the study and participated in the collection of literature data, manuscript writing and critical discussion. He read and approved the final manuscript. B.D. participated in the collection of literature data, manuscript writing and critical discussion. He read and approved the final manuscript. W.A.L. participated in the collection of literature data, manuscript writing and critical discussion. She read and approved the final manuscript. W.G.A. participated in the collection of literature data, manuscript writing and critical discussion. He read and approved the final manuscript. A.D. participated in the collection of literature data, manuscript writing and critical discussion. She read and approved the final manuscript. J.J.K. participated in the collection of literature data, manuscript writing and critical discussion. She read and approved the final manuscript. A.R.L. participated in the collection of literature data, manuscript writing and critical discussion. He read and approved the final manuscript. F.R.T. participated in the collection of literature data, manuscript writing and critical discussion. He read and approved the final manuscript. D.D. participated in the collection of literature data, manuscript writing and critical discussion. He read and approved the final manuscript.
Disclosure Summary
The authors have no competing interests to declare.
Data Availability
All data analyzed during this study are included in the data repositories listed in References.
References
- 1. Rotterdam ESHRE/ASRM Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril. 2004;81:19-25. [DOI] [PubMed] [Google Scholar]
- 2. Azziz R, Carmina E, Dewailly D, et al. . Position statement: criteria for defining polycystic ovary syndrome as a predominantly hyperandrogenic syndrome: an androgen excess society guideline. J Clin Endocrinol Metab. 2006;91:4237-4455. [DOI] [PubMed] [Google Scholar]
- 3. Legro RS, Arslanian SA, Ehrmann DA, et al. . Diagnosis, and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2013;98:4565-4592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Teede HJ, Misso ML, Costello MF, et al. ; International PCOS Network. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertil Steril. 2018; 110: 364-379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Zaenglein AL, Pathy AL, Schlosser BJ, et al. . Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-73.e33. [DOI] [PubMed] [Google Scholar]
- 6. Poli F, Auffret N, Claudel JP, Leccia MT, Dreno B. AFAST: an adult female acne treatment algorithm for daily clinical practice. Eur J Dermatol. 2018;28:101-1033. [DOI] [PubMed] [Google Scholar]
- 7. Burls A. AGREE II-improving the quality of clinical care. Lancet. 2010;376:1128-1129. [DOI] [PubMed] [Google Scholar]
- 8. National Health and Medical Research Council (NHMRC). NHMRC additional levels of evidence and grades for recommendations for developers of guidelines – Stage 2 consultation – early 2008 – end June 2009. https://www.mja.com.au/sites/default/files/NHMRC.levels.of.evidence.2008-09.pdf
- 9. Zouboulis C. Acne and sebaceous gland function. Clin Dermatol. 2004;22:360-366. [DOI] [PubMed] [Google Scholar]
- 10. Kurokawa I, Danby FW, Ju Q, et al. . New developments in our understanding of acne pathogenesis and treatment. Exp Dermatol. 2009;18:821-832. [DOI] [PubMed] [Google Scholar]
- 11. Clayton RW, Göbel K, Niessen CM, Paus R, Van Steensel MMA, Lim X. Homeostasis of the sebaceous gland and mechanisms of acne pathogenesis. Br J Dermatol. 2019;181:677-690. [DOI] [PubMed] [Google Scholar]
- 12. Downie MM, Kealey T. Human sebaceous glands engage in aerobic glycolysis and glutaminolysis. Br J Dermatol. 2004;151:320-327. [DOI] [PubMed] [Google Scholar]
- 13. Tóth BI, Oláh A, Szöllosi AG, Czifra G, Bíró T. Sebocytes makeup: novel mechanisms and concepts in the physiology of the human sebaceous glands. Pflugers Arch. 2011;461:593-6066. [DOI] [PubMed] [Google Scholar]
- 14. Harris HH, Downing DT, Stewart ME, Strauss JS. Sustainable rates of sebum secretion in acne patients and matched normal control subjects. J Am Acad Dermatol. 1983;8:200-203. [DOI] [PubMed] [Google Scholar]
- 15. Das S, Reynolds RV. Recent advances in acne pathogenesis: implications for therapy. Am J Clin Dermatol. 2014;15:479-888. [DOI] [PubMed] [Google Scholar]
- 16. Zouboulis CC, Jourdan E, Picardo M. Acne is an inflammatory disease and alterations of sebum composition initiate acne lesions. J Eur Acad Dermatol Venereol. 2014;28:527-322. [DOI] [PubMed] [Google Scholar]
- 17. Leeming JP, Holland KT, Cuncliffe WJ. The microbial colonization of inflamed acne vulgaris lesions. Br J Dermatol. 1988;118:203-208. [DOI] [PubMed] [Google Scholar]
- 18. Leyden JJ, McGinley KJ, Kligman AM. Tetracycline and minocycline treatment. Arch Dermatol. 1982;118:19-22. [PubMed] [Google Scholar]
- 19. Weyrich LS, Dixit S, Farrer AG, Cooper AJ. The skin microbiome: associations between altered microbial communities and disease. Australasian J Dermatol. 2015;56:268-274. [DOI] [PubMed] [Google Scholar]
- 20. Dreno B, Pecastaings S, Corvec S, Veraldi S, Khammari A, Roques C. Cutibacterium acnes (Propionibacterium acnes) and acne vulgaris: a brief look at the latest updates. J Eur Acad Dermatol Venereol. 2018;32(Suppl 2):5-14. [DOI] [PubMed] [Google Scholar]
- 21. Dagnelle MA, Montassier E, Khammari A, Mounier C, Corvec S, Dreno B. Inflammatory skin is associated with changes in the skin microbiota composition on the back of severe acne patients. Exp Dermatol. 2019;28:961-967. [DOI] [PubMed] [Google Scholar]
- 22. Contassot E, French LE. New insights into acne pathogenesis: Propionibacterium acnes activates the inflammasome. J Invest Dermatol. 2014;134:310-313. [DOI] [PubMed] [Google Scholar]
- 23. O Neill AM, Gallo RL. Host-microbiome interactions and recent progress into understanding the biology of acne vulgaris. Microbiome. 2018;6:177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Sawaya ME, Price VH. Different levels of 5alpha-reductase type I and II, aromatase, and androgen receptor in hair follicles of women and men with androgenetic alopecia. J Invest Dermatol. 1997;109:296-300. [DOI] [PubMed] [Google Scholar]
- 25. Del Rosso JO, Kircik LH, Stein Gold L, Thiboutot D. Androgens, androgen receptors, and the skin: from the laboratory to the clinic with emphasis on clinical and therapeutic implications. J Drugs Dermatol. 2020;19:30-35. [PubMed] [Google Scholar]
- 26. Sawaya ME, Pennys NS. Immunohistochemical distribution of aromatase and 3B-hydroxysteroid dehydrogenase in human hair follicle and sebaceous gland. J Cutan Pathol. 1992;19:309-314. [DOI] [PubMed] [Google Scholar]
- 27. Thiboutot D, Gilliland K, Light J, Lookingbill D. Androgen metabolism in sebaceous glands from subjects with and without acne. Arch Dermatol. 1999;135:1041-1045. [DOI] [PubMed] [Google Scholar]
- 28. Carmina E. Cutaneous manifestations of polycystic ovary syndrome. Curr Opin Endocrinol Metab Res. 2020;12:49-52. [Google Scholar]
- 29. Reingold SB, Rosenfield RL. The relationship of mild hirsutism or acne in women to androgens. Arch Dermatol. 1987;123:209-212. [PubMed] [Google Scholar]
- 30. Carmina E, Lobo RA. Hirsutism, alopecia, and acne. In: Becker KL, ed. Principles and Practice of Endocrinology and Metabolism. 3rd ed. Lippincott, Williams and Wilkins; 2001:991-1008. [Google Scholar]
- 31. Slayden SM, Moran C, Sams WM Jr, Boots LR, Azziz R. Hyperandrogenemia in patients presenting with acne. Fertil Steril. 2001;275:889-892. [DOI] [PubMed] [Google Scholar]
- 32. Ramezani Tehrani F, Behboudi-GandevanI S, Bidhendi Yarandi R, Ghare Naz MS, Carmina E. Prevalence of acne vulgaris among women with polycystic ovary syndrome: a systemic review and meta-analysis. Gynecol Endocrinol. 2021;37:392-405. [DOI] [PubMed] [Google Scholar]
- 33. Sardana K, Bansal P, Sharma LK, Garga UC, Vats G. A study comparing the clinical and hormonal profile of late onset and persistent acne in adult females. Int J Dermatol. 2020;59:428-433. [DOI] [PubMed] [Google Scholar]
- 34. Clayton RW, Göbel K, Niessen CM, Paus R, van Steensel MAM, Lim X. Homeostasis of the sebaceous gland and mechanisms of acne pathogenesis. Br J Dermatol. 2019;181:677-690. [DOI] [PubMed] [Google Scholar]
- 35. Kurokawa I, Layton AM, Ogawa R. Updated treatment for acne: targeted therapy based on pathogenesis. Dermatol Ther (Heidelberg). 2021;11:1129-1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Carmina E, Stanczyk FZ, Matteri RK, Lobo RA. Serum androsterone conjugates differentiate between acne and hirsutism in hyperandrogenic women. Fertil Steril. 1991;55:872-876. [PubMed] [Google Scholar]
- 37. Carmina E, Godwin AJ, Stanczyk FZ, Lippman JS, Lobo RA. The association of serum androsterone glucuronide with inflammatory lesions in women with adult acne. J Endocrinol Invest. 2002;25:765-768. [DOI] [PubMed] [Google Scholar]
- 38. Carmina E, Lobo RA. Evidence for increased androsterone metabolism in some normoandrogenic women with acne. J Clin Endocrinol Metab. 1993;76:1111-1114. [DOI] [PubMed] [Google Scholar]
- 39. Lucky AW, Henderson TA, Olson WH, Robisch DM, Lebwohl M, Swinyer LJ. Effectiveness of norgestimate and ethinyl estradiol in treating moderate acne vulgaris. J Am Acad Dermatol. 1997;37:746-754. [DOI] [PubMed] [Google Scholar]
- 40. Pretorius E, Arlt W, Storbeck KH. A new dawn for androgens: novel lessons from 11-oxygenated C19 androgens. Mol Cell Endocrinol. 2017;441:76-85. [DOI] [PubMed] [Google Scholar]
- 41. Zeichner JA, Baldwin HE, Cook-Bolden FE, Eichenfield LF, Fallon-Friedlander S, Rodriguez DA. Emerging issues in adult female acne. J Clin Aesthet Dermatol. 2017;10:37-46. [PMC free article] [PubMed] [Google Scholar]
- 42. Schmitt JV, Masuda PY, Miot HA. Acne in women: clinical patterns in different age-groups. An Bras Dermatol 2009;84:349-354. [DOI] [PubMed] [Google Scholar]
- 43. Dréno B, Layton A, Zouboulis CC, et al. . Adult female acne: a new paradigm. J Eur Acad Dermatol Venereol. 2013;27:1063-1070. [DOI] [PubMed] [Google Scholar]
- 44. Dréno B, Thiboutot D, Layton AM, Berson D, Perez M, Kang S; Global Alliance to Improve Outcomes in Acne . Large-scale international study enhances understanding of an emerging acne population: adult females. J Eur Acad Dermatol Venereol. 2015;29:1096-1106. [DOI] [PubMed] [Google Scholar]
- 45. Bagatin E, Proença de Freitas TH, Rivitti-Machado MC, Medeiros Ribeiro B, Nunes S, Dias da Rocha MA. Adult female acne: a guide to clinical practice. An Bras Dermatol. 2019;94:62-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Preneau S, Dreno B. Female acne - a different subtype of teenager acne? J Eur Acad Dermatol Venereol. 2012;26:277-282. [DOI] [PubMed] [Google Scholar]
- 47. Bhate K, Williams HC. Epidemiology of acne vulgaris. Br J Dermatol. 2013;168:474-485. [DOI] [PubMed] [Google Scholar]
- 48. Heng AHS, Chen FT. Systematic review of the epidemiology of acne vulgaris. Sci Rep. 2020;10:5754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Lucky AW, Barber BL, Girman CJ, Williams J, Ratterman J, Waldstreicher J. A multirater validation study to assess the reliability of acne lesion counting. J Am Acad Dermatol. 1996;35:559-565. [DOI] [PubMed] [Google Scholar]
- 50. Witkowski JA, Parish LC. The assessment of acne: an evaluation of grading and lesion counting in the measurement of acne. Clin Dermatol. 2004;22:394-397. [DOI] [PubMed] [Google Scholar]
- 51. Tan JKL, Jones E, Allen E, Pripotnev S, Raza A, Wolfe B. Evaluation of essential clinical components and features of current acne global grading scales. J Am Acad Dermatol. 2013;69:754-761. [DOI] [PubMed] [Google Scholar]
- 52. Agnew T, Furber G, Leach M, Segal L. A comprehensive critique and review of published measures of acne severity. J Clin Aesthet Dermatol. 2016;9:40-52. [PMC free article] [PubMed] [Google Scholar]
- 53. Dreno B, Poli F, Pawin H, et al. . Development and evaluation of a Global Acne Severity Scale (GEA Scale) suitable for France and Europe. J Eur Acad Dermatol Venereol. 2011;25:43-48. [DOI] [PubMed] [Google Scholar]
- 54. Burke BM, Cunliffe WJ. The assessment of acne vulgaris—the Leeds technique. Br J Dermatol. 1984;111:83-92. [DOI] [PubMed] [Google Scholar]
- 55. O’Brien SC, Lewis JB, Cunliffe WJ. The Leeds revised acne grading system. J Dermatol Treat. 1998;9:215-220. [Google Scholar]
- 56. Bergman H, Tsai KY, Seo SJ, Kvedar JC, Watson AJ. Remote assessment of acne: the use of acne grading tools to evaluate digital skin images. Telemed J E Health. 2009;15:426-430. [DOI] [PubMed] [Google Scholar]
- 57. Auffret N, Claudel JP, Leccia MT, Poli F, Farhi D, Dréno B. AFAST - adult female acne scoring tool: an easy-to-use tool for scoring acne in adult females. J Eur Acad Dermatol Venereol. 2016;30:824-828. [DOI] [PubMed] [Google Scholar]
- 58. Samuels DV, Rosenthal R, Lin R, Chaudhari S, Natsuaki MN. Acne vulgaris and risk of depression and anxiety: a meta-analytic review. J Am Acad Dermatol. 2020;83:532-541. [DOI] [PubMed] [Google Scholar]
- 59. Yazici K, Baz K, Yazici AE, et al. . Disease-specific quality of life is associated with anxiety and depression in patients with acne. J Eur Acad Dermatol Venereol. 2004;18:435-439. [DOI] [PubMed] [Google Scholar]
- 60. Picardi A, Lega I, Tarolla E. Suicide risk in skin disorders. Clin Dermatol. 2013;31:47-56. [DOI] [PubMed] [Google Scholar]
- 61. Barnard L, Ferriday D, Guenther N, Strauss B, Balen AH, Dye I. Quality of life and psychological wellbeing in polycystic ovary syndrome. Hum Reprod. 2007;22:2279-2286. [DOI] [PubMed] [Google Scholar]
- 62. Bazarganipour F, Ziaei S, Montazeri A, Foroozanfard F, Kazemnejad A, Faghihzadeh S. Body image satisfaction and self-esteem status among the patients with polycystic ovary syndrome. Iran J Reprod Med. 2013;11:829-836. [PMC free article] [PubMed] [Google Scholar]
- 63. Vos T, Flaxman AD, Naghavi M, et al. . Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990– 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2163-2196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Li D, Chen Q, Liu Y, Liu T, Tang W, Li S. The prevalence of acne in Mainland China: a systematic review and meta-analysis. BMJ Open. 2017;7:e015354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Zore T, Lizneva D, Brakta S, Walker W, Suturina L, Azziz R. Minimal differences in phenotype between adolescents and young adults with polycystic ovary syndrome. Fertil Steril. 2019;111:389-396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Perkins AC, Cheng CE, Hillebrand GG, Miyamoto K, Kimball AB. Comparison of the epidemiology of acne vulgaris among Caucasian, Asian, Continental Indian, and African American women. J Eur Acad Dermatol Venereol. 2011;25:1054-1060. [DOI] [PubMed] [Google Scholar]
- 67. Goulden V, Clark SM, Cunliffe WJ. Post-adolescent acne: a review of clinical features. Br J Dermatol. 1997;136:66-70. [PubMed] [Google Scholar]
- 68. Cibula D, Hill M, Vohradnikova O, Kuzel D, Fanta M, Zivny J. The role of androgens in determining acne severity in adult women. Br J Dermatol. 2000;143:399-404. [DOI] [PubMed] [Google Scholar]
- 69. Borgia F, Cannavò S, Guarneri F, Cannavò SP, Vaccaro M, Guarneri B. Correlation between endocrinological parameters and acne severity in adult women. Acta Derm Venereol. 2004;84:201-204. [DOI] [PubMed] [Google Scholar]
- 70. Suh DH, Kim BY, Min SU, et al. . A multicenter epidemiological study of acne vulgaris in Korea. Int J Dermatol. 2011;50:673-681. [DOI] [PubMed] [Google Scholar]
- 71. Wang YY, Li SW, Luo S, et al. . How to evaluate acne in reproductive-age women: an epidemiological study in Chinese communities. Biomed Res Int. 2019:6126808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Lucky AW, McGuire J, Rosenfield RL, Lucky PA, Rich BH. Plasma androgens in women with acne vulgaris. J Invest Dermatol. 1983;81:70-74. [DOI] [PubMed] [Google Scholar]
- 73. da Cunha MG, Fonseca FL, Machado CD. Androgenic hormone profile of adult women with acne. Dermatology. 2013;226: 167-171. [DOI] [PubMed] [Google Scholar]
- 74. Azziz R, Sanchez LA, Knochenhauer ES, et al. . Androgen excess in women: experience with over 1000 consecutive patients. J Clin Endocrinol Metab. 2004;89:453-462. [DOI] [PubMed] [Google Scholar]
- 75. Carmina E, Rosato F, Jannì A, Rizzo M, Longo RA. Relative prevalence of different androgen excess disorders in 950 women referred because of clinical hyperandrogenism. J Clin Endocrinol Metab. 2006;91:2-6. [DOI] [PubMed] [Google Scholar]
- 76. Azziz R, Carmina E, Chen Z, et al. . Polycystic ovary syndrome. Nat Rev Dis Primers. 2016;2:16057. [DOI] [PubMed] [Google Scholar]
- 77. Bunker GB, Newton JA, Kilborn J, et al. . Most women with acne have polycystic ovaries. Br J Dermatol. 1989;121:675-680. [DOI] [PubMed] [Google Scholar]
- 78. Kelekci KH, Kelekci S, Incki K, Ozdemir O, Yilmaz B. Ovarian morphology, and prevalence of polycystic ovary syndrome in reproductive aged women with or without mild acne. Int J Dermatol. 2010;49:775-779. [DOI] [PubMed] [Google Scholar]
- 79. Maluqi AH. The frequency of polycystic ovary syndrome in females with acne vulgaris. J Cosmetic Dermatol. 2010;9:142-148. [DOI] [PubMed] [Google Scholar]
- 80. Uysal G, Sahin Y, Unluhizarci K, et al. . Is acne a sign of androgen excess disorder or not? Eur J Obstet Gynecol Reprod Biol. 2017;211:21-25. [DOI] [PubMed] [Google Scholar]
- 81. Kazemi M, Pierson RA, Lujan ME, et al. . Comprehensive evaluation of type 2 diabetes and cardiovascular disease risk profiles in reproductive-age women with polycystic ovary syndrome: a large Canadian cohort. J Obstet Gynaecol Canada. 2019;41: 1453-1460. [DOI] [PubMed] [Google Scholar]
- 82. Villarroel C, Trejo L, Muñoz A, Kohen P, Fuentes A, Devoto L. Assessment of diagnostic competence of plasmatic androgens on polycystic ovary syndrome based on receiver operator characteristic curves. Gynecol Endocrinol. 2010;26:600-606. [DOI] [PubMed] [Google Scholar]
- 83. Feng JG, Guo Y, Ma L, Xing J, Sun R, Zhu W. . Prevalence of dermatologic manifestations and metabolic biomarkers in women with polycystic ovary syndrome in North China. J Cosmetic Dermatol. 2018;17:511-517. [DOI] [PubMed] [Google Scholar]
- 84. Tan J, Wang Q-Y, Feng G-M, Li X-Y, Huang W. Increased risk of psychiatric disorders in women with polycystic ovary syndrome in Southwest China. Chin Med J. 2017;130:262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Zhang HY, Guo CX, Zhu FF, Qu PP, Lin WJ, Xiong J. Clinical characteristics, metabolic features, and phenotype of Chinese women with polycystic ovary syndrome: a large-scale case–control study. Arch Gynecol Obstet. 2013;287:525-531. [DOI] [PubMed] [Google Scholar]
- 86. Lauritsen MP, Bentzen J, Pinborg A, et al. . The prevalence of polycystic ovary syndrome in a normal population according to the Rotterdam criteria versus revised criteria including anti-Müllerian hormone. Hum Reprod. 2014;29:791-801. [DOI] [PubMed] [Google Scholar]
- 87. Taponen S, Ahonkallio S, Martikainen H, et al. . Prevalence of polycystic ovaries in women with self‐reported symptoms of oligomenorrhoea and/or hirsutism: Northern Finland Birth Cohort 1966 Study. Hum Reprod. 2004;19:1083-1088. [DOI] [PubMed] [Google Scholar]
- 88. Dalamaga M, Papadavid E, Basios G, et al. . Ovarian SAHA syndrome is associated with a more insulin-resistant profile and represents an independent risk factor for glucose abnormalities in women with polycystic ovary syndrome: a prospective controlled study. J Am Acad Dermatol. 2013;69:922-930. [DOI] [PubMed] [Google Scholar]
- 89. Jacob R, Jude K, Chandrasekhar R, Sasikala K. Prevalence of acne among women with poly cystic ovarian syndrome – a clinical study. Scrutiny Int Res J Health Med Sci. 2014;1(1):7-13. [Google Scholar]
- 90. Mangalath AAM, Alias A, Sajith M, Nimbargi V, Kumdale S. Sociodemographic characteristics, and clinical presentation of infertile women with polycystic ovary syndrome in a tertiary care hospital. Int J Infertil Fet Med. 2018;9:14-18. [Google Scholar]
- 91. Rashidi BH, Shams S, Shariat M, Jaliseh HK, Mohebi M, Haghollahi F. Evaluation of serum hepcidin and iron levels in patients with PCOS: a case-control study. J Endocrinol Invest. 2017;40:779-784. [DOI] [PubMed] [Google Scholar]
- 92. Shishehgar F, Mirmiran P, Rahmati M, Tohidi M, Tehrani FR. Does a restricted energy low glycemic index diet have a different effect on overweight women with or without polycystic ovary syndrome? BMC Endocr Dis. 2019;19:93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Tehrani FR, Rashidi H, Khomami MB, Tohidi M, Azizi F. The prevalence of metabolic disorders in various phenotypes of polycystic ovary syndrome: a community-based study in Southwest of Iran. Reprod Biol Endocrinol. 2014;12:89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Hosseini MS, Dizavi A, Rostami H, Parastouei K, Esfandiari S. Healthy eating index in women with polycystic ovary syndrome: a case-control study. Int J Reprod BioMed 2017;15:575. [PMC free article] [PubMed] [Google Scholar]
- 95. Belosi C, Selvaggi L, Apa R, et al. . Is the PCOS diagnosis solved by ESHRE/ASRM 2003 consensus or could it include ultrasound examination of the ovarian stroma? Hum Reprod. 2006;21:3108-3115. [DOI] [PubMed] [Google Scholar]
- 96. Akram M, Roohi N. Endocrine correlates of polycystic ovary syndrome in Pakistani women. J Coll Physicians Surg Pak. 2015;25:22-26. [PubMed] [Google Scholar]
- 97. Musmar S, Afaneh A, Mo’alla H. Epidemiology of polycystic ovary syndrome: a cross sectional study of university students at An-Najah national university-Palestine. Reprod Biol Endocrinol. 2013;11:47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Sharif E, Rahman S, Zia Y, Rizk NM. The frequency of polycystic ovary syndrome in young reproductive females in Qatar. Int J Women Health. 2017;9:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Chen CH, Wang PH, Hsieh MT, et al. . Sexual orientations of women with polycystic ovary syndrome: clinical observation in Taiwan. Taiwan J Obstet Gynecol. 2014;53:542-546. [DOI] [PubMed] [Google Scholar]
- 100. Ercan C, Coksuer H, Aydogan U, et al. . Sexual dysfunction assessment and hormonal correlations in patients with polycystic ovary syndrome. Int J impotence Res. 2013;25:127-132. [DOI] [PubMed] [Google Scholar]
- 101. Eser A, Erpolat S, Kaygusuz I, Balci H, Kosus A. Investigation of Demodex folliculorum frequency in patients with polycystic ovary syndrome. An Brasil Dermatol. 2017;92:807-810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Vural B, Caliskan E, Turkoz E, Kilic T, Demirci A. Evaluation of metabolic syndrome frequency and premature carotid atherosclerosis in young women with polycystic ovary syndrome. Hum Reprod. 2005;20:2409-2413. [DOI] [PubMed] [Google Scholar]
- 103. Hart K, Barr S, Reeves S, Sharp K, Jeanes Y. Suboptimal dietary intake is associated with cardiometabolic risk factors in women with polycystic ovary syndrome. Nut Diet. 2016;73:1771-183 . [Google Scholar]
- 104. Kumarendran B, O’Reilly MW, Manolopoulos KN, et al. . Polycystic ovary syndrome, androgen excess, and the risk of nonalcoholic fatty liver disease in women: a longitudinal study based on a United Kingdom primary care database. PLoS Med. 2018; 15(3):e1002542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Balachandran K, Sumilo D, O’Reilly MW, et al. . Increased risk of obstructive sleep apnoea in women with polycystic ovary syndrome: a population-based cohort study. Eur J Endocrinol. 2019;180:265-272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Welt C, Arason G, Gudmundsson J, et al. . Defining constant versus variable phenotypic features of women with polycystic ovary syndrome using different ethnic groups and populations. J Clin Endocrinol Metab. 2006;91:4361-4368. [DOI] [PubMed] [Google Scholar]
- 107. Welt C, Gudmundsson J, Arason G, et al. . Characterizing discrete subsets of polycystic ovary syndrome as defined by the Rotterdam criteria: the impact of weight on phenotype and metabolic features. J Clin Endocrinol Metab. 2006;91:4842-4848. [DOI] [PubMed] [Google Scholar]
- 108. Bird ST, Hartzema AG, Brophy JM, Etminan M, Delaney JA. Risk of venous thromboembolism in women with polycystic ovary syndrome: a population-based matched cohort analysis. CMAJ. 2013;185:E115-E120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Bird ST, Hartzema AG, Etminan M, Brophy JM, Delaney JA. Polycystic ovary syndrome and combined oral contraceptive use: a comparison of clinical practice in the United States to treatment guidelines. Gynecol Endocrinol. 2013;29:365-369. [DOI] [PubMed] [Google Scholar]
- 110. Cao NT, Le MT, Nguyen VQH, et al. . Defining polycystic ovary syndrome phenotype in Vietnamese women. J Obst Gynaecol Res. 2019;45:2209-2219. [DOI] [PubMed] [Google Scholar]
- 111. Vesper HW, Bhasin S, Wang C, et al. . Interlaboratory comparison study of serum total testosterone [corrected] measurements performed by mass spectrometry methods. Steroids. 2009;74:498-503. [DOI] [PubMed] [Google Scholar]
- 112. Carmina E, Stanczyk F, Lobo RA. Evaluation of hormonal status. Strauss JF III, Barbieri RL, eds. In: Yen and Jaffe’s Reproductive Endocrinology: Physiology, Pathophysiology and Clinical Management. 8th ed. Elsevier Saunders; 2018:887-915. [Google Scholar]
- 113. Rosner W. An extraordinary inaccurate assay for free testosterone is still with us. J Clin Endocrinol Metab. 2001;86:2903. [DOI] [PubMed] [Google Scholar]
- 114. Rosner W, Auchus RJ, Azziz R, Sluss PM, Raft H. Position statement: utility, limitations and pitfalls in measuring testosterone: an Endocrine Society position statement. J Clin Endocrinol Metab. 2007;92:405-413. [DOI] [PubMed] [Google Scholar]
- 115. Hauk L. Acne vulgaris: treatment guidelines from the AAD. Am Fam Physician. 2017;95:740-741. [PubMed] [Google Scholar]
- 116. Oge LK, Broussard A, Marshall MD. Acne vulgaris: diagnosis and treatment. Am Fam Physician. 2019;100:475-484. [PubMed] [Google Scholar]
- 117. Bienenfeld A, Nagler AR, Orlow SJ. Oral antibacterial therapy for acne vulgaris: an evidence-based review. Am J Clin Dermatol. 2017;18:469-490. [DOI] [PubMed] [Google Scholar]
- 118. Moore A, Ling M, Bucko A, Manna V, Rueda MJ. Efficacy and safety of subantimicrobial dose, modified-release doxycycline 40 mg versus doxycycline 100 mg versus placebo for the treatment of inflammatory lesions in moderate and severe acne: a randomized, double-blinded, controlled study. J Drugs Dermatol. 2015;14:581-586. [PubMed] [Google Scholar]
- 119. Bienenfeld A, Azarchi S, Lo Sicco K, Marchbein S, Shapiro J, Nagler AR. Androgens in women: androgen-mediated skin disease and patient evaluation. J Am Acad Dermatol. 2019;80:1497-1506. [DOI] [PubMed] [Google Scholar]
- 120. Barbieri JS, Choi JK, Mitra N, Margolis DJ. Frequency of treatment switching for spironolactone compared to oral tetracycline-class antibiotics for women with acne: a retrospective cohort study 2010-2016. J Drugs Dermatol. 2018;17:632-638. [PubMed] [Google Scholar]
- 121. Han JJ, Faletsky A, Barbieri JS, Mostaghimi A. New acne therapies and updates on use of spironolactone and isotretinoin: a narrative review. Dermatol Ther (Heidelb). 2021;11:79-91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Tong LX, Brauer JA. Lasers, light, and the treatment of acne: a comprehensive review of the literature. J Drugs Dermatol. 2017;16:1095-1102. [PubMed] [Google Scholar]
- 123. Arowojolu AO, Gallo MF, Lopez LM, et al. . Combined oral contraceptive pills for treatment of acne. Cochrane Database Syst Rev. 2012;(7):CD004425. [DOI] [PubMed] [Google Scholar]
- 124. Thorneycroft IH, Stanczyk FZ, Bradshaw KD, Ballagh SA, Nichols M, Weber ME. Effect of low dose oral contraceptives on androgenic markers and acne. Contraception 1999;60:255-267. [DOI] [PubMed] [Google Scholar]
- 125. Carmina E. Oral contraceptives and cardiovascular risk in Polycystic Ovary Syndrome. J Endocrinol Invest. 2013;36:358-363. [DOI] [PubMed] [Google Scholar]
- 126. Carmina E, Lobo RA. A comparison of the relative efficacy of antiandrogens for the treatment of acne in hyperandrogenic women. Clin Endocrinol. (Oxf) 2002;57:231-234. [DOI] [PubMed] [Google Scholar]
- 127. Brown J, Farquhar C, Lee O, Toomath R, Jepson RG. Spironolactone versus placebo or in combination with steroids for hirsutism and/or acne (review). Cochrane Database Syst Rev. 2009:CD000194.19370553 [Google Scholar]
- 128. Thiboutot D, Chen W. Update, and future of hormonal therapy in acne. Dermatology. 2003;206:57-67. [DOI] [PubMed] [Google Scholar]
- 129. Muhlemann MF, Carter GD, Cream JJ, Wise P. Oral spironolactone: an effective treatment for acne vulgaris in women. Br J Dermatol. 1986;115:227-232. [DOI] [PubMed] [Google Scholar]
- 130. Goodfellow A, Alaghband-Zadeh J, Carter G, et al. . Oral spironolactone improves acne vulgaris and reduces sebum excretion. Br J Dermatol. 1984;111:209-214. [DOI] [PubMed] [Google Scholar]
- 131. Shaw JC. Low-dose adjunctive spironolactone in the treatment of acne in women: a retrospective analysis of 85 consecutively treated patients. J Am Acad Dermatol. 2000;43:498-502. [DOI] [PubMed] [Google Scholar]
- 132. Layton AM, Eady EA, Whitehouse H, Del Rosso JQ, Fedorowicz Z, van Zuuren EJ. Oral spironolactone for acne vulgaris in adult females: a hybrid systemic review. Am J Clin Dermatol. 2017;18:169-191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Calaf J, Lopez E, Millet A, et al. ; Spanish Working Group for Hirsutism . Long-term efficacy and tolerability of flutamide combined with oral contraceptive in moderate to severe hirsutism: a 12-month, double-blind, parallel clinical trial. J Clin Endocrinol Metab. 2007;92:3446-3452. [DOI] [PubMed] [Google Scholar]
- 134.Press Release from European Medicines Agency. Restrictions in use of cyproterone due to meningioma risk. Accessed February 14, 2020. https://www.ema.europa.eu/en/news/restrictions-use-cyproterone-due-meningioma-risk
- 135. Hebert A, Thiboutot D, Gold LS, et al. . Efficacy and safety of topical clascoterone cream, 1%, for treatment in patients with facial acne. Two phase 3 randomized clinical trials. JAMA Dermatol. 2020;156:621-630. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data analyzed during this study are included in the data repositories listed in References.