Abstract
Using a rabbit model of systemic infection, we show that it is possible to differentiate infections caused by Candida dubliniensis and other Candida species by detecting the antibody response mounted by the infected animals. These results confirm our previous observation in a patient with C. dubliniensis candidemia and suggest that detection of C. dubliniensis-specific antibodies is useful in the diagnosis of invasive candidiasis caused by this yeast.
Candida dubliniensis has been predominantly recovered from the oral cavities of human immunodeficiency virus-infected individuals (6, 10, 11). However, in recent years, C. dubliniensis has been increasingly recovered from individuals not infected with human immunodeficiency virus (6, 7, 12), including individuals with invasive candidiasis (4, 4a, 5, 9). At present, the identification of systemic infections caused by C. dubliniensis relies on the isolation of the yeast from clinical specimens followed by identification by conventional methods used in the clinical microbiology laboratory. We have previously shown the existence of antigenic differences between C. dubliniensis and its close relative Candida albicans (3), and we have shown that it was possible to differentiate candidemia caused by the two organisms due to the specific antibody response detected in a C. dubliniensis-infected patient (9). In the present study, a rabbit model of systemic infection was developed to confirm the existence of differences in the antibody response which can be used in the serodiagnosis of infections caused by C. dubliniensis.
Apart from Candida dubliniensis CD33 (11), which was from the University of Dublin strain collection, all of the yeast strains used in this study were obtained from the National Collection of Pathogenic Fungi (Bristol, United Kingdom). Yeast strains were routinely grown in medium 199 (Sigma Chemical Co., St. Louis, Mo.) as previously described (2), and their cell walls were extracted in the presence of dithiothreitol (Sigma) as reported previously (8). New Zealand White rabbits were intravenously inoculated with 2 × 106 blastospores as described previously (2). In some experiments, a group of rabbits were immunized subcutaneously with two immunoreactive antigens from C. dubliniensis with molecular masses of 34 kDa or between 160 and 170 kDa. For the initial immunization, antigens were dissolved in 0.5 ml of complete Freund's adjuvant (Sigma) and injected subcutaneously into two rows of five sites equidistantly spaced along the rabbit's back. Subsequent immunizations were performed every week with the antigens in incomplete Freund's adjuvant. Sera were adsorbed with C. albicans NCPF 3153 blastospores using previously described methods (8). Indirect immunofluorescence and Western blotting assays were performed as previously described (2). To obtain the immunoreactive bands of 34 kDa or 160 to 170 kDa from the cell wall of C. dubliniensis, a dithiothreitol extract (300 μg of protein) was separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and electroblotted onto a nitrocellulose membrane as described previously (2). Bands of 34 kDa and 160 to 170 kDa were excised, dissolved in 0.5 ml of dimethyl sulfoxide (Sigma), and diluted with 0.5 ml of complete or incomplete Freund's adjuvant (Sigma).
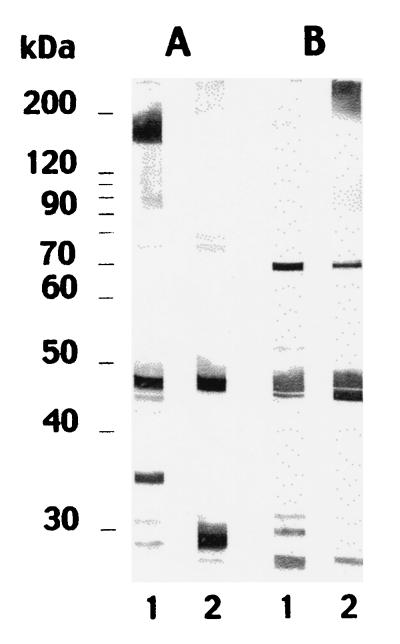
It was not possible to discriminate between rabbit systemic infections caused by C. dubliniensis NCPF 3949 and C. albicans NCPF 3153 on the basis of the detection of antibodies directed to the cell wall surface by indirect immunofluorescence. However, the antibody response against antigens extracted from the cell wall was more informative. Adsorbed sera from rabbits infected with C. dubliniensis NCPF 3949 reacted predominantly with four antigens in the C. dubliniensis NCPF 3949 extract with molecular masses of 160 to 170, 45, 34, and 29 kDa and with two antigens in the C. albicans NCPF 3153 extract with molecular masses of 45 and 29 kDa (Fig. 1). Similar results were observed with cell wall extracts from C. dubliniensis CD33. Adsorbed sera from rabbits infected with C. albicans NCPF 3153 reacted predominantly with five antigens in the C. dubliniensis extract with molecular masses of 70, 45, 32, 30, and 28 kDa and with five antigens in the C. albicans extract with molecular masses of >200, 70, 45, 44, and 28 kDa (Fig. 1).
FIG. 1.
Western blots of 10% (wt/vol) acrylamide slab gels loaded with extracts from C. dubliniensis NCPF 3949 blastospores grown at 37°C (lanes 1) and C. albicans NCPF 3153 germ tubes (lanes 2), stained with the anti-C. dubliniensis-adsorbed serum (A lanes) or with the anti-C. albicans-adsorbed serum (B lanes). Molecular masses (in kilodaltons) of standard proteins are shown on the left.
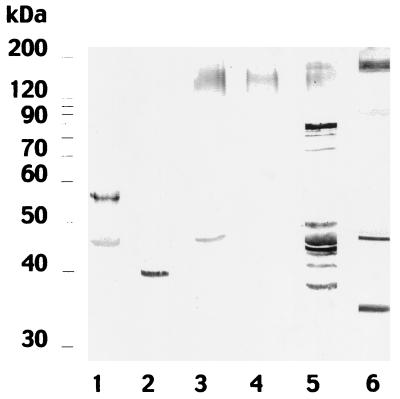
Since the antibody response against C. dubliniensis cell wall antigens permitted discrimination between infections caused by C. dubliniensis from those caused by C. albicans, it was of interest to investigate whether infections caused by other Candida species could also be differentiated. Antisera from rabbits infected with Candida krusei NCPF 3100 recognized two antigens with molecular masses of 57 and 45 kDa, while sera from rabbits infected with Candida parapsilosis NCPF 3104 reacted with an antigen with a molecular mass of 39 kDa (Fig. 2). The antisera from the rabbits infected with Candida guilliermondii NCPF 3099 or Candida glabrata NCPF 3203 reacted with polydispersed components of molecular masses between 130 and 150 kDa (Fig. 2). The sera from rabbits infected with Candida tropicalis NCPF 3111 stained polydispersed components of molecular masses between 130 and 160 kDa and several antigens with molecular masses between 38 and 88 kDa (Fig. 2).
FIG. 2.
Western blots of 10% (wt/vol) acrylamide slab gels loaded with cell wall extracts from C. dubliniensis NCPF 3949, stained with C. albicans-adsorbed sera from rabbits infected by C. krusei NCPF 3100 (lane 1), C. parapsilosis NCPF 3104 (lane 2), C. guilliermondii NCPF 3099 (lane 3), C. glabrata NCPF 3203 (lane 4), C. tropicalis NCPF 3111 (lane 5), and C. dubliniensis NCPF 3949. Molecular masses (in kilodaltons) of standard proteins are shown on the left.
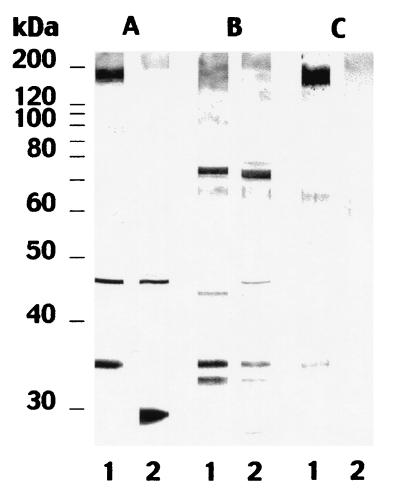
The results of the studies described above led to the identification of two antigens with molecular masses of 160 to 170 and 34 kDa which are potentially specific for C. dubliniensis. To further characterize these antigens, rabbits were immunized separately with antigen extracts and the resulting antisera were used to stain cell wall extracts from several Candida species. The serum directed against the 160- to 170-kDa antigen reacted with this antigen and with two bands of 65 and 34 kDa in the C. dubliniensis extract (Fig. 3, lane C1). This antiserum also stained faint bands of >200, 65, and 60 kDa in the C. albicans extract (Fig. 3, lane C2). The serum directed against the 34-kDa antigen reacted with several bands (73, 65, 45, 44, 34, and 32 kDa) present in extracts from both C. dubliniensis and C. albicans (Fig. 3, B lanes). However, this antiserum also showed a faint reactivity with the 160- to 170-kDa region in the C. dubliniensis extract and with a band of >200 kDa in the C. albicans extract. The antisera directed against both the 160- to 170-kDa antigen and the 34-kDa antigen showed a positive reaction by indirect immunofluorescence with C. dubliniensis blastospores but not with C. albicans blastospores (data not shown).
FIG. 3.
Western blots of 10% (wt/vol) acrylamide slab gels loaded with extracts from C. dubliniensis NCPF 3949 grown at 37°C (lanes 1) and C. albicans NCPF 3153 grown at 37°C (lanes 2), stained with serum from a rabbit infected by C. dubliniensis (A lanes) or sera from rabbits immunized with the C. dubliniensis 34-kDa antigen (B lanes) or the C. dubliniensis 160- to 170-kDa antigen (C lanes). Molecular masses (in kilodaltons) of standard proteins are shown on the left.
As C. dubliniensis can be recovered from cases of invasive infection, it is clear that routine diagnostic laboratories should be in a position to rapidly and accurately identify this species. Due to its close similarity with C. albicans, identification of C. dubliniensis infections may be problematic in most clinical mycology laboratories, and currently, identification is made following isolation of the fungus in culture. However, recovery from blood may be difficult, since the sensitivity of blood culture methods such as lysis centrifugation varies from 28 to 78%, depending on the number of deep-tissue sites infected by Candida (1). In this regard, antibody detection may help to identify some cases of C. dubliniensis invasive infections, providing a specific response can be demonstrated (9).
In the present study, we have shown that it is possible to differentiate between experimentally induced infections caused by C. dubliniensis and C. albicans and between infections caused by C. dubliniensis and other clinically significant Candida species on the basis of the antibody response mounted by the infected animals. Rabbits infected with C. dubliniensis induced a specific antibody response directed against two cell wall components of 160 to 170 and 34 kDa not found in animals infected with C. albicans or with any of the other clinically relevant Candida species studied. These components seem to be antigenically related and located on the cell wall surface. The results presented in this study confirm and extend our previous observation of a patient with C. dubliniensis candidemia, who exhibited an antibody response against an antigen of 160 to 170 kDa which was not observed in patients with C. albicans candidemia (9). If the antibody responses detected in rabbits infected by C. tropicalis, C. glabrata, C. krusei, C. parapsilosis, or C. guillermondii could be extrapolated to humans, it is unlikely that patients with infections caused by these Candida species would produce an antibody response similar to that observed in the patient infected by C. dubliniensis.
In conclusion, we present evidence for the existence of a specific antibody response in rabbits infected with C. dubliniensis which is similar to that observed in a patient with C. dubliniensis candidemia. We believe that detection of antibodies specific for C. dubliniensis antigens may have a clinical application in the diagnosis of invasive candidiasis caused by this yeast, especially in patients with negative blood cultures.
Acknowledgments
This investigation was supported in part by grants UPV 093.327-G01/98 from the Universidad del País Vasco and PM99-0033 from the Spanish Ministerio de Ciencia y Tecnología.
REFERENCES
- 1.Berenguer J, Buck M, Witebsky F, Stock F, Pizo P A, Walsh T J. Lysis-centrifugation blood cultures in the detection of tissue proven invasive candidiasis: disseminated versus single organ infection. Diagn Microbiol Infect Dis. 1993;17:103–109. doi: 10.1016/0732-8893(93)90020-8. [DOI] [PubMed] [Google Scholar]
- 2.Bikandi J, San Millán R, Regúlez P, Moragues M D, Quindós G, Pontón J. Detection of antibodies to Candida albicans germ tubes during experimental infections by different Candida species. Clin Diagn Lab Immunol. 1998;5:369–374. doi: 10.1128/cdli.5.3.369-374.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bikandi J, San Millán R, Moragues M D, Cebas G, Clarke M, Coleman D C, Sullivan D J, Quindós G, Pontón J. Rapid identification of Candida dubliniensis by indirect immunofluorescence based on differential localization of antigens on C. dubliniensis blastospores and Candida albicans germ tubes. J Clin Microbiol. 1998;36:2428–2433. doi: 10.1128/jcm.36.9.2428-2433.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brandt M E, Harrison L H, Pass M, Sofair A N, Huie S, Li R K, Morrison C J, Warnock D W, Hajjeh R A. Candida dubliniensis fungemia: the first four cases in North America. Emerg Infect Dis. 2000;6:46–49. doi: 10.3201/eid0601.000108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4a.Marriott D, Laxton M, Harkness J. Candida dubliniensis candidemia in Australia. Emerg Infect Dis. 2001;7:479. doi: 10.3201/eid0703.010326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Meis J F, Ruhnke M, De Pauw B E, Odds F C, Siegert W, Verweij P E. Candida dubliniensis candidemia in patients with chemotherapy induced neutropenia and bone marrow transplantation. Emerg Infect Dis. 1999;5:150–153. doi: 10.3201/eid0501.990119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Moran G P, Sullivan D J, Henman M C, McCreary C E, Harrington B J, Shanley D B, Coleman D C. Antifungal drug susceptibilities of oral Candida dubliniensis isolates from human immunodeficiency virus (HIV)-infected and non-HIV-infected subjects and generation of stable fluconazole-resistant derivatives in vitro. Antimicrob Agents Chemother. 1997;41:617–623. doi: 10.1128/aac.41.3.617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Polacheck I, Strahilevitz J, Sullivan D, Donnelly S, Salkin I F, Coleman D C. Recovery of Candida dubliniensis from non-human immunodeficiency virus-infected patients in Israel. J Clin Microbiol. 2000;38:170–174. doi: 10.1128/jcm.38.1.170-174.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ponton J, Jones J M. Analysis of cell wall extracts of Candida albicans by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and Western blot techniques. Infect Immun. 1986;53:565–572. doi: 10.1128/iai.53.3.565-572.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Salesa R, Moragues M D, Sota R, Pemán J, Quindós G, Pontón J. Specific antibody response in a patient with Candida dubliniensis fungemia. Rev Iberoam Micol. 2001;18:42–44. [PubMed] [Google Scholar]
- 10.Sullivan D J, Moran G, Donnelly S, Gee S, Pinjon E, McCartan B, Shanley D B, Coleman D C. Candida dubliniensis: an update. Rev Iberoam Micol. 1999;16:72–76. [PubMed] [Google Scholar]
- 11.Sullivan D J, Westerneng T J, Haynes K A, Bennett D E, Coleman D C. Candida dubliniensis sp. nov.: phenotypic and molecular characterization of a novel species associated with oral candidosis in HIV-infected individuals. Microbiology. 1995;141:1507–1521. doi: 10.1099/13500872-141-7-1507. [DOI] [PubMed] [Google Scholar]
- 12.Willis A M, Coulter W A, Sullivan D J, Coleman D C, Hayes J R, Bell P M, Lamey P J. Isolation of C. dubliniensis from insulin-using diabetes mellitus patients. J Oral Pathol Med. 2000;29:86–90. doi: 10.1034/j.1600-0714.2000.290206.x. [DOI] [PubMed] [Google Scholar]