Abstract
Objectives:
To determine the long-term effect on the stability of dentin-resin interfaces after the addition of polylactide (PLA) capsules containing proanthocyanidin (PAC) to adhesive resin.
Methods:
Sub-micron (SM) and micron (M) size capsules containing PACs were produced using a combination of emulsification and solvent evaporation techniques and characterized. Human dentin surfaces (n = 8) were etched (35% glycolic acid) and primed (15% enriched Vitis vinifera extract solution), followed by the application of an experimental adhesive containing 0 (control), 1.5 wt% of SM or M PAC-filled PLA capsules light cured for 40 s. A crown was built using commercial composite. After 24 h-immersion (37°C) in simulated body fluid, specimens were serially sectioned into resin-dentin beams. Microtensile bond strength (TBS), micro-permeability and fracture pattern were assessed immediately and after 1 and 2 years. Data were statistically analyzed using two-way ANOVA and post-hoc test (α = 0.05).
Results:
Polydisperse capsules were manufactured with average diameter of 0.36 μm and 1.08 μm for SM and M, respectively. The addition of capsules did not affect TBS (p = 0.889). After 2 years, TBS significantly decreased in SM (p = 0.006), whereas M showed similar initial values (p = 0.291). Overall, less micro-permeability was found in M than the control and SM group (p < 0.001). After 2 years, fractured surfaces from capsule-containing groups failed within the adhesive layer while control fractured at the bottom of the hybrid layer.
Significance:
The addition of PAC-filled PLA microcapsules in a dental adhesive did not affect the bond strength while increased and sustained the protection against micro-permeability in the interface, likely due to release of PACs.
Keywords: proanthocyanidins, bond strength, polylactide capsules, bioactive release, sustained release
1. Introduction
Secondary caries and marginal fracture are the primary causes of failure of resin composite restorations [1,2]. The strategies developed to increase the lifespan of these restorations integrate protective mechanisms within the composite [3], the adhesive material [4] and/or the dentin [5]; and are directed to increase the endurance of the hybrid layer and prevent future caries disease events. Still, the resin-dentin interface remains susceptible to long-term breakdown due to several mechanisms linked to plasticization and hydrolysis of the adhesive polymer network [6,7] and the activation of endogenous proteolytic enzymes in the dentin extracellular matrix [8,9].
Proanthocyanidins (PACs) are biopolymers shown to reduce the long-term biodegradation of the adhesive interface by decreasing collagen solubilization, increasing the dentin matrix stiffness, and reducing the surface hydrophilicity and resin-dentin interfacial permeability [5,10]. Application of PACs are explored mostly as a pre-adhesive step in the bonding protocol or as a combination with an etchant (acidic primer) for immediate interaction with dentin [11]. The availability of PACs for on-demand and sustained delivery is an adjunctive approach for boosting the actions of PACs at the dentin-resin interface. This strategy could be achieved by the addition of polylactide (PLA) capsules containing PACs as core material into the adhesive resin. A previous study optimized the encapsulation parameters to produce capsules compatible with dentin adhesion, with a targeted long degradation time while preserving PAC bioactivity [12]. Microencapsulation techniques have been used to deliver drugs with an on-demand release due to targeted degradation of the shell material. Polylactide, a hydrophobic biobased polymer, is commonly used as shell wall material due to its biocompatibility and biodegradability, decomposing into naturally occurring metabolites through hydrolysis or enzymatic processes [13,14]. The core material can be released from the capsules by diffusion through the shell wall, hydrolytic degradation of the walls, or a combination of both mechanisms [15]. Encapsulation represents a versatile technology that allows tunning of the release rate by modifying various factors such as the molecular weight of shell polymer, porosity, and capsule size, among others [16]. In restorative dentistry, few studies reported using encapsulation to deliver self-healing agents resulting in the prevention of microcracks propagation within the resin composite and adhesive [17]. Others focused on providing anti-inflammatory medication to therapeutically address inflammation in deep cavities [18] or releasing fluoride to reduce the attachment of bacterial plaque [19].
The rationale for the use of PAC-filled capsules is to provide the adhesive system with a response capacity. Thus, when hydrolysis occurs, fluids reach and degrade the wall shell of the capsules releasing PACs. Released PACs will then interact with the dentin matrix, reinforcing the adhesive interface by protecting the dentin extracellular matrix from further damage. Hence, the objective of this study was to determine and compare the long-term stability of the adhesive interface of experimental dental adhesives containing micron- and sub-micron-sized PLA PAC-filled capsules to a PAC biomodified dentin. The null hypothesis was that the incorporation of capsules in the adhesive resin will not affect the short- and long-term bond strength and interfacial micro-permeability of a dentin-PAC-resin adhesive interface.
2. Materials and methods
2.1. Preparation and characterization of capsules
2.1.1. Preparation of VVe-filled capsules
Enriched grape seed extract from Vitis vinifera (VVe), a rich source of oligomeric proanthocyanidins biopolymers, was encapsulated in sub-micron and micron PLA capsules using an oil-in-water (O/W) emulsion technique (Fig. 1) [12]. PLA (Ingeo 4043D, NatureWorks; Mw ~150,000 Da) is a water-immiscible polymer whereas VVe is a water-soluble compound; therefore, to create a single emulsion, a solvent mixture was used to co-dissolve both polymer and core materials. The oil phase (O) was a mixture of 1 mL of 150 mg/mL VVe in methanol (MeOH) and 3 mL of 5 wt.% PLA in dichloromethane (DCM) solutions. Miscibility of MeOH and DCM with each other allows for co-dissolving both VVe and PLA in a mixture of these two organic solvents at critical mixing ratios. In addition, when the volume of DCM in the mixture is greater than that of MeOH, the overall solution shows immiscibility with water, enabling the emulsification of the oil solution in the continuous aqueous phase (W). For all experiments, the continuous phase was a solution of 2.5 wt% polyvinyl alcohol (PVA, Mw 13,000–23,000, 87–89% hydrolyzed) in distilled water. For fabrication of micron capsules, emulsions were created by adding the oil phase dropwise to 300 mL of continuous phase while homogenizing the emulsions using a homogenizer (OMNI GLH- 01) for 10 minutes. Emulsions for fabrication of sub-micron capsules were created by emulsifying 1 mL of the oil phase in a test tube containing 4 mL of continuous phase using a horn sonicator (750 W, Cole Parmer) under pulsed mode (20% amplitude, 0.2 s pulse, 0.2 s pause) for one minute. Emulsions were then transferred to a beaker containing 200 mL of continuous aqueous phase under continuous agitation at 800 rpm. The process was repeated until all oil solution was emulsified in the aqueous phase. Following emulsification using either approach, emulsions were mechanically agitated at 800 rpm for five hours to allow diffusion and evaporation of solvents and formation of solid polymer capsules. Capsules were then centrifuged (4000 rpm for 10 min) and washed with distilled water four times to remove any residual surfactants. Collected capsules were then lyophilized overnight to remove water from the capsules.
Figure 1.
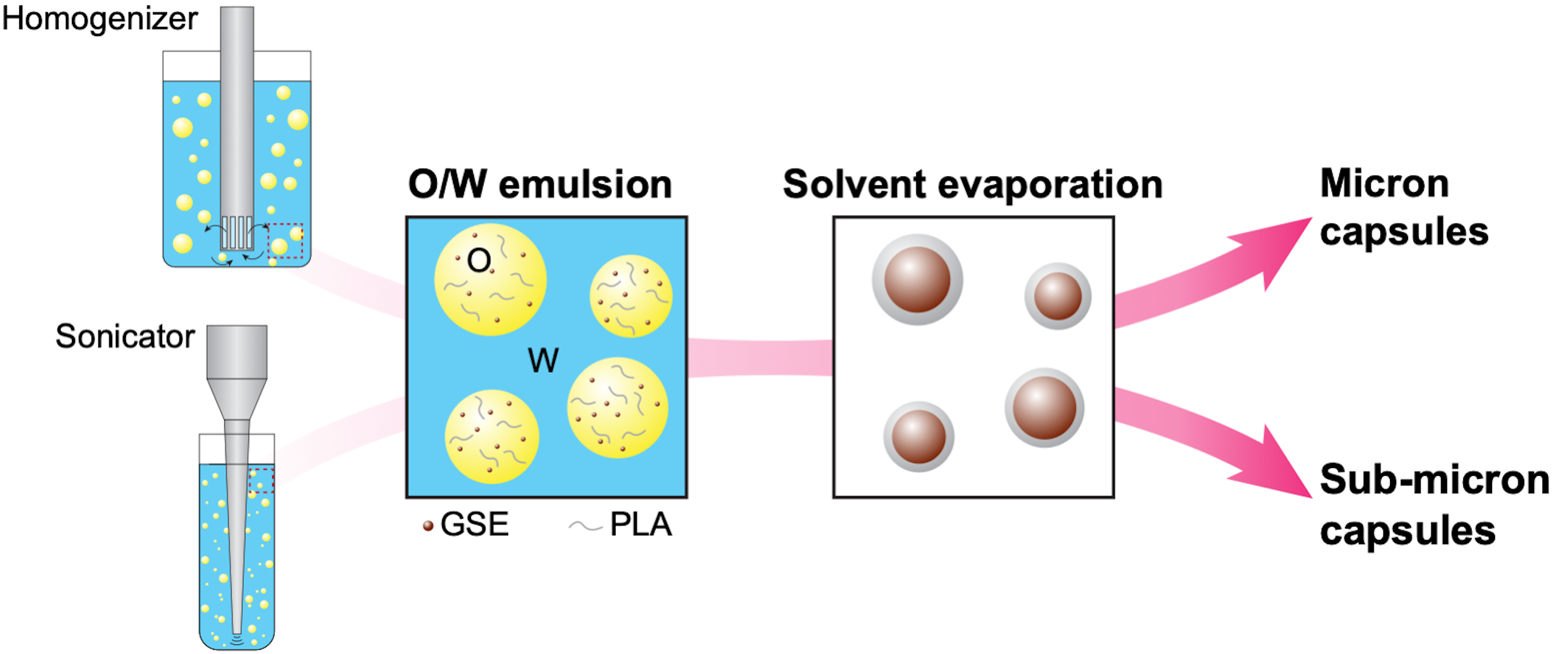
Schematic representation of the encapsulation technique
2.1.2. Characterization of capsules
The morphology and size distribution of capsules were characterized using scanning electron microscopy (SEM, Quanta 450 FEG ESEM) and dynamic light scattering (DLS, Litesizer 500, Anton Paar), respectively. The core loading of capsules was determined using UV-visible spectroscopy (UV-2401 PC, Shimadzu, Japan) according to a method reported in our previous work [12]. Briefly, capsules were first dissolved in a 40:60 vol/vol mixture of tetrahydrofuran and methanol at 1 g/L concentration. The solution was diluted with distilled water at a 1:9 volume ratio to precipitate and remove the shell polymer. The absorbance of VVe at 280 nm was then measured and compared with a linear calibration curve created using known concentrations of VVe in the same solvent mixture to calculate the concentration of VVe (cvv). Capsule core loading was calculated as,
| (1) |
where Vs is the volume of diluted solution used in UV-vis measurements, d is the dilution ratio (10 in this study), and Mcap is the mass of capsules.
2.2. Adhesion studies
2.2.1. Preparation of experimental adhesives
Experimental adhesives containing either VVe-filled sub-micron or micron PLA capsules were prepared at a 1.5 wt% capsule concentration, based on an initial study assessing the effect of capsule concentration on the adhesive viscosity. Capsules were added to the resin mixture (Table 1) and mixed with magnetic stirring under a light-protected environment to prevent polymerization. Control group used the same resin mixture without the addition of capsules.
Table 1.
Adhesive formulation and experimental group nomenclature according to capsule size.
| Resin mixture | Capsule size | Group |
|---|---|---|
| 41.25 wt% Bisphenol A glycidyl methacrylate (BisGMA) 12 wt% Triethylene glycol dimethacrylate (TEGDMA) |
1.5 wt% sub-micron | SM |
| 6 wt% Hydroxyethyl methacrylate (HEMA) 0.15 wt% Camphorquinone (CQ) |
1.5 wt% micron | M |
| 0.6 wt% Ethyl-4-dimethylamino benzoate (EDMAB) 40 % Ethanol |
0% | Control |
2.2.2. Preparation of Resin-dentin Interfaces
Sound human third molars (IRB no. 2018–0346) were selected and ground flat with silicon carbide (SiC) abrasive paper (180-, 320-grit, Buehler, Lake Bluff, IL, USA) to remove the occlusal enamel and expose mid-coronal dentin. Dentin surfaces were polished (600-grit for 20 seconds) to produce a smear layer. Specimens were randomly divided into 3 groups (n = 8) according to the experimental adhesive. Dentin surfaces were etched with 35 wt% glycolic acid (pH = 1.30, Sigma Aldrich, St. Louis, MO, USA) for 15 s and rinsed with ultrapure water (15 s). A bioactive primer consisting of 15 wt% VVe diluted in HEPES solution (pH = 7.2) was actively applied for 1 min and rinsed for 15 s. Then, two layers of the experimental adhesive containing 0% (control), 1.5 wt% of sub-micron (SM) or micron (M) VVe-filled PLA spheres were applied, air-dried for solvent evaporation and light cured for 40s (600 mW/cm2, Optilux 501, Kerr Corp., Brea, CA, USA). A commercial light-curable resin composite (Filtek Supreme Ultra, Lot: N820171, 3M ESPE, St. Paul, MO, USA) was used to incrementally build a crown-like block. All specimens were immersed in simulated body fluid (SBF: 50 mM HEPES, 5 mM CaCl2·2 H2O, 0.001 mM ZnCl2, 150 mM NaCl, and 3 mM NaN3, pH = 7.4) [20], at 37°C. SBF was replaced every 15 days.
2.2.3. Interfacial bond strength
After 24 h-immersion in SBF, specimens were serially sectioned into resin-dentin beams with a cross-sectional area of 0.8 ± 0.05 mm2. Five resin-dentin beams per tooth were selected and fixed by the edges to a Ciucchi jig and tested in tension (Bisco, Schaumburg, IL, USA) at a crosshead speed of 1 mm/min. Microtensile bond strength (TBS) was calculated by dividing the fracture load by the cross-sectional area of the interface and results were expressed in MPa. TBS was assessed after 24 h, 1, and 2 years of SBF immersion at 37°C. Normality of distribution was confirmed with Shapiro-Wilk test (p > 0.1). Levene’s test assessed intragroup variability and found to be not homogenously distributed (p = 0.014). Data were then statistically analyzed using two-way ANOVA (variables: time and adhesives), followed by Games-Howell post hoc tests (α = 0.05).
2.2.4. Interfacial micro-permeability of the adhesive interface
The micro-permeability of the adhesive interface was determined by the infiltration of fluorescent dye into interfacial micro-porosities. As per an established protocol [21], resin-dentin beams (n = 9) from each group were randomly selected, embedded in epoxy resin and polished with SiC papers (320-, 600-, 800-, and 1200-grit). Specimens were kept immersed in freshly prepared 0.1 wt% rhodamine-B solution (RITC/Rhodamine-B; Sigma Aldrich) dissolved in 0.01 M PBS (pH = 7.2) for 1 h at room temperature without light exposure, followed by a water rinse for 1 min. The fluorescence emission intensity (FEI) was analyzed under a Fluorescence microscope (EVOS FL Auto, Life Technologies, Thermo Fisher Scientific, Waltham, MA, USA) using three images taken from each specimen at ×20 magnification. FEI values of interfacial micro-permeability were obtained using a parallel line profile traced below the adhesive interface by an image analysis software (ImageJ 1.48p, National Institutes of Health, USA). The FEI data variability was assessed using Levene’s test (p < 0.001). Data were statistically analyzed using two-way ANOVA (variables: time and adhesives), followed by Games-Howell post hoc test (α = 0.05).
2.2.5. Morphology of the adhesive interface and fracture pattern
Resin-dentin beams incubated for 24 months in SBF were selected to cross-sectionally assess the adhesive interface and determine the penetration of the capsules into the hybrid layer. The adhesive interface was treated with 10% phosphoric acid for 3 s followed by 5% sodium hypochlorite for 10 min. In addition, three debonded specimens of each group and time point were selected to analyze the morphology and fracture pattern of the interface. All specimens were fixed in a stub, sputter-coated, and examined by scanning electron microscopy (JEOL JSM-6510LV, JEOL USA Inc., Peabody, MA, USA).
3. Results
3.1. Characterization of capsules
Sub-micron- and micron-sized PLA capsules filled with VVe were manufactured using a combination of emulsification and solvent evaporation techniques. Both sub-micron and micron capsules are polydisperse in size with a smooth surface morphology (Fig. 2). Size distribution analysis showed an average diameter (Davg) of 0.36 μm and 1.08 μm for sub-micron and micron capsules, respectively. Histogram plots of size distribution of capsules are depicted in Figure 2. The percentage of VVe core loading found for sub-micron capsules was ~15%, and for microcapsules was ~21%.
Figure 2.

Size distribution and representative scanning electron microscopy images of sub-micron (A) and micron (B) capsules. The average diameter (Davg) for sub-micron and micron capsules were 0.36 μm and 1.08 μm, respectively. Images exposed polydisperse size capsules with smooth surfaces. Scale bar: 2 μm.
3.2. Dentin-Resin interface bond strength
Microtensile bond strength values and standard deviations are shown in Figure 3A. There was a significant interaction among the studied factors (adhesives vs time, p = 0.039). Overall, the addition of capsules did not negatively affect the dentin-PAC-resin interface bond strength (p = 0.889) when compared to control at any time point (p > 0.06). However, time significantly influenced the sub-micron group, decreasing the bond strength by 25% after 2 years (p = 0.006). The micron-sized capsule group exhibited a significant reduction after 1 year (p = 0.004) yet recovered by the second year with values similar to that of 24 h (p = 0.291). Storage time in SBF did not influence the adhesive bond strength of control group (p = 0.608).
Figure 3.

(A) Microtensile bond strength (in MPa, mean and standard deviation) and (B) Interfacial Micro-permeability (FEI- fluorescence emission intensity, mean values and standard error) of experimental groups as a function of time (24 h, 1 and 2 years). Different uppercase letters indicate statistically significant differences (p < 0.05) among time points within each experimental adhesive group. Same symbol indicates lack of statistical difference among adhesives within each time point (p > 0.05).
3.3. Interfacial micro-permeability of the adhesive interface
No significant interaction among studied factors (adhesive and time) was found (p = 0.295, Figure 3B). However, significant differences were found among adhesives (p < 0.001), with lower micro-permeability (low fluorescence intensity) in the micron-sized capsule group when compared to control and sub-micron group (p < 0.001).
3.4. Morphology of adhesive interface and fracture pattern
Representative cross-sectional images of the adhesive interfaces obtained after 24 h incubation are shown in Figure 4. Sub-micron and micron groups showed resin-dentin interfaces with polydisperse capsules spread within the adhesive layer. Overall, larger agglomerations of capsules were observed in the sub-micron group whereas micron-sized capsules were evenly distributed in the adhesive. A thicker adhesive layer was observed for groups with capsules.
Figure 4.

Fluorescence microscopy cross-sectional images (×40 magnification) of the resin-dentin interface after 24 h incubation. Open arrows indicate the presence of aggregates of sub-micron capsules. Close arrow illustrates isolated micron capsule located in the resin-adhesive interface. (A) control, (B) sub-micron, and (C) micron groups. Scale bar: 25 μm.
Images from micron group revealed the presence of isolated capsules between the resin-adhesive interface (Fig. 4C). SEM representative images from experimental groups after 2-year storage in SBF are shown in Figure 5. In the sub-micron group, small lines of fracture are observed within the adhesive layer indicating the removal of agglomerates of capsules. In the micron group, a higher magnification showed partial degradation of capsules surrounded by a gap in the interface adhesive-capsule and small cracks initiating from the gaps. The presence of isolated capsules adhered to the resin tags was evident in both sub-micron and micron-size capsules groups. Figure 6 depicts representative images of surfaces fractured at different time points. The debonding fracture pattern occurred most predominantly at the hybrid and adhesive layers. After 24 h the predominant fracture pattern for all groups was cohesive in the adhesive layer. Images of the sub-micron group showed capsules with an intact surface and out-of-plane fracture lines around the resin matrix, indicating crack propagation. However, micron group showed a fracture pattern at the top of the adhesive layer with some areas showing filler particles from the resin composite. Concave zones with different sizes represent the capsules imprints. After 1 year, differences were found between the groups containing capsules and control. More specifically, the fracture pattern for groups containing capsules was predominantly cohesive in the adhesive layer, whereas the control group showed fractures at the bottom of the hybrid layer, with areas of exposed collagen network partially covered with debris from the adhesive layer and fractured resin tags. After 2 years, control group showed fractures at the bottom of the hybrid layer with areas of cohesive fracture in the dentin. Capsule-containing groups showed similar pattern as observed after 1 year. Images revealed more imprints and empty vesicles than intact capsules in the adhesive layer.
Figure 5.

Representative scanning electron microscopy images of the resin-dentin interface after 2-year incubation at two magnifications, ×2,500 (left, scale bar: 10 μm) and ×5,000 (right, scale bar: 5 μm). Open arrows indicate the presence of capsules in the resin tags, observed in both sub-micron and micron groups. Closed arrows show small cracks initiating from the interface adhesive resin-capsules. Images in micron group evidenced degradation of the capsules’ shell wall and exposure of core. (A) control, (B) sub-micron, and (C) micron groups.
Figure 6.

Representative scanning electron microscopy images of the debonded surface following microtensile test after 24 hours, 1 and 2 years. All images at ×3,000 magnification (scale bar: 5 μm). Asterisks indicate the presence of fractured resin tags within the dentin tubules, open arrows show crack propagation lines between capsule sites, and closed arrows reflect imprints of capsules that detached or degraded. (A) control, (B) sub-micron, and (C) micron groups.
4. Discussion
This study explored an established and versatile drug delivery strategy by successfully encapsulating PACs in polylactide capsules for integration into a dental adhesive, allowing a controlled delivery of PACs for sustained enhancement of the resin-dentin interface. After 2 years, our results revealed that the presence of PACs-filled capsules did not have a negative effect on the microtensile adhesive strength, regardless of capsule size. Furthermore, micron-sized capsules reduced the micro-permeability, thus enhancing the stability of the adhesive interface. Therefore, the null hypothesis was partially rejected.
The stability of the resin-dentin interface is key for the success of adhesive-based restoration. Long-term in vitro bond strength consistently shows a reduction between 33 to 40% up to 2.5 years [22,23] and 48% after 4 years of water immersion [24]. Among the different strategies to reinforce the adhesive interface, biomodification with plant-derived PACs gained interest due to their potent interactions with dentin extracellular matrix [25], an integral component of dentin-resin interfaces. Herein, the bonding substrate for all groups consisted of dentin biomodified by application of a primer containing oligomeric-enriched PACs (VVe). Evidence of strong and stable bioadhesion (up to 1 year) was previously shown using the same oligomeric PACs used in this study [5]. Our results demonstrate that the enhanced stability is further maintained after two years.
The presence of capsules did not affect the adhesive strength. However, a significant reduction in bond strength was found in the sub-micron group after two-year aging. Fluorescence and SEM images revealed a distinct lack of dispersion of sub-micron capsules within the adhesive layer [26,27], differently than the adhesive containing micron-sized capsules (Fig. 4,6). Nanoparticles tend to cluster due to high surface area forming agglomerates [28]. Within an agglomerate of capsules, the spaces between capsules are filled with air, creating voids. Voids act as stress concentrators and when subjected to load, initiate cracks in the adjacent matrix and affect the mechanical properties [29,30]. Hence, the 25% reduction in bond strength observed in the sub-micron group can be associated with the presence of capsule agglomerates that increased the risk of crack initiation [31].
The presence of micron-sized capsules reduced the micro-permeability of the adhesive interface. Here, the protection of the dentin substrate by PAC biomodification was further enhanced by the release of PACs from the capsules. Regardless of their size, capsules penetrated the dentinal tubules, as found in SEM analysis (Fig. 5B,C). The penetration of capsules in dentin tubules was previously reported when associated with an etch-and-rinse adhesive system that favors the formation of resin tags. [26]. Capsules exposed to dentinal fluids most likely had the PLA shell wall degraded, resulting in an initial release of PACs and subsequent biomodification of the surrounding dentin. Although capsules are not able to diffuse into interfibrillar spaces [32], released PACs could have diffused into the surrounding dentin and stabilized the exposed dentin matrix by crosslinking collagen fibrils [21]. PACs released from capsules found in dentin tubules exemplify the sustained reinforcement and represent the first line of protection offered by the capsules. The second line of protection corresponds to the release from capsules embedded in the adhesive layer closest to the underlying dentin.
The capsule size influenced the interfacial micro-permeability, sub-micron capsules did not elicit a significant reduction of micro-permeability as the micron sized capsules. In fact, the bioavailability of VVe was lower in sub-micron capsules (15% core loading) when compared to micron capsules (21%). Differences in drug loading, drug release, bioavailability, and dosage are directly related to the size and distribution of capsules [27]. A possible explanation is that the lower loading of PACs in sub-micron capsules reduced their bioavailability in susceptible dentin, which consequently led to less protection against enzymatic degradation at the hybrid layer.
The bioavailability of encapsulated PACs was confirmed in a previous report. Encapsulated extracts from Vitis vinifera were bioactive even after 12 months-storage [12]. A time-dependent cumulative release profile of PAC-filled PLA capsules to determine sustained bioactivity beyond 2 years was not pursued, representing a limitation of this study. However, the data indicates that micron-sized capsules had a protective effect on the biostability of the hybrid layer due to bioactive release.
The assessment of the micro-permeability and fracture pattern at the adhesive interface allows the compilation of important information concerning the interface sealing and indicate the reason for adhesive failure [21]. Here, PAC released from the micron-sized capsules contributed to superior protection against hydrolytic degradation of the hybrid layer. After 24 months, fracture surfaces revealed that debonding failures were concentrated in the adhesive layer, while the dentin at the bottom of the hybrid layer remained preserved, as observed in a previous report using PACs [10]. Water sorption accelerates the degradation of resin and the exposed collagen fibrils [24]. When the unsupported demineralized dentin at the bottom of the hybrid layer undergoes strain, it becomes a weak point for breakage [6,23,33]. We attribute the mechanical endurance to the biomodification derived from the controlled release of PACs. PAC-mediated crosslinks decrease bound water due to the reduction of the surface hydrophilicity [34] and the intrinsic water dynamics [35]. It is worth mention that similar characteristics were identified at the fractured surfaces from both groups containing PAC-filled capsules. Conversely, the control group showed distinct features after 24 months. Debonded surfaces from control group presented mostly denuded tubules or fractured resin tags, however some remnants of adhesive layer found on the intertubular dentin indicate a strong dentin-adhesive interaction.
The encapsulation of reactive components to provide a responsive functional material has shown promising results in the dental field [36,37]. Besides hydrolytic or enzymatic environmental stimuli, other design parameters can control the degradation of the PLA capsule shell wall, thus, offering tunning of time-dependent release of core material. However, long-term studies using dental adhesives or resin composites containing capsules are scarce, limiting a clear understanding of the lifespan of such materials. For instance, voids left after capsules are fully degraded could create a pathway for hydrolytic degradation and crack initiation. This study found no negative effects of incorporated capsules after a 2-year follow-up. This promising outcome represents the first record on the longevity assessment for such system in this specific dental application.
In summary, the addition of PLA capsules did not negatively affect the adhesive bond strength after 2-year storage in SBF. Specifically for the micron-sized capsules, we further observed increased stability of the dentin-resin interface due to a decrease in the interfacial micro-permeability, most likely due to PACs release from the capsules located at the resin tags. The release of bioactive material will occur upon hydrolysis of the dental resin that allows fluid infiltration into the adhesive matrix to reach and degrade the capsule wall triggering release of PACs. The elicited effect is likely due to a boost in the reinforcement of PACs at the underlying dentin, in areas where the adhesive system underwent hydrolytic degradation exposing the PLA capsules. The findings support the use of PLA microcapsules as a supplementary strategy to on-demand PAC delivery.
Highlights.
PACs encapsulation involves emulsification and solvent evaporation techniques.
The addition of capsules did not affect the bond strength, regardless of their size.
Capsule penetration in dentinal tubules trigger release of PACs.
Acknowledgements
This work was supported by the National Institutes of Health [DE21040]. The authors would like to acknowledge the late Prof. Scott R. White for his insights and guidance on this work. The authors thank Bisco Inc. for providing the experimental resin adhesive. We thank Ms. Gresa Mustafa and Dr. Malissa Arroyo for helping with the microtensile bond strength experiment.
Abbreviations
- PAC
proanthocyanidin
- ECM
extracellular matrix
- SBF
simulated body fluid
- VV
Vitis vinifera
- MeOH
methanol
- DCM
dichloromethane
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
In memory of Scott White
References
- [1].Demarco FF, Corrêa MB, Cenci MS, Moraes RR, Opdam NJM. Longevity of posterior composite restorations: not only a matter of materials. Dent Mater 2012;28:87–101. 10.1016/j.dental.2011.09.003. [DOI] [PubMed] [Google Scholar]
- [2].Ástvaldsdóttir Á, Dagerhamn J, van Dijken JWV, Naimi-Akbar A, Sandborgh-Englund G, Tranæus S, Nilsson M. Longevity of posterior resin composite restorations in adults – A systematic review. J Dent 2015;43:934–54. 10.1016/j.jdent.2015.05.001. [DOI] [PubMed] [Google Scholar]
- [3].Maas MS, Alania Y, Natale LC, Rodrigues MC, Watts DC, Braga RR. Trends in restorative composites research: what is in the future? Braz Oral Res 2017;31:e55. 10.1590/1807-3107BOR-2017.vol31.0055. [DOI] [PubMed] [Google Scholar]
- [4].Zhou W, Liu S, Zhou X, Hannig M, Rupf S, Feng J, Peng X, Cheng L. Modifying adhesive materials to improve the longevity of resinous restorations. Int J Mol Sci 2019;20:723. 10.3390/ijms20030723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Leme-Kraus AA, Aydin B, Vidal CMP, Phansalkar RM, Nam JW, McAlpine J, Pauli GF, Chen S, Bedran-Russo AK. Biostability of the proanthocyanidins-dentin complex and adhesion studies. J Dent Res 2017;96:406–12. 10.1177/0022034516680586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Ferracane JL, Berge HX, Condon JR. In vitro aging of dental composites in water--effect of degree of conversion, filler volume, and filler/matrix coupling. J Biomed Mater Res 1998;42:465–72. 10.1002/(sici)1097-4636(19981205)42:3<465::aid-jbm17>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- [7].Bourbia M, Finer Y. Biochemical stability and interactions of dental resin composites and adhesives with host and bacteria in the oral cavity: a review. J Can Dent Assoc 2018;84:i1. [PubMed] [Google Scholar]
- [8].Pashley DH, Tay FR, Yiu C, Hashimoto M, Breschi L, Carvalho RM, Ito S. Collagen degradation by host-derived enzymes during aging. J Dent Res 2004;83:216–21. 10.1177/154405910408300306. [DOI] [PubMed] [Google Scholar]
- [9].Mazzoni A, Tjäderhane L, Checchi V, Di Lenarda R, Salo T, Tay FR, Pashley DH, Breschi L. Role of dentin MMPs in caries progression and bond stability. J Dent Res 2015;94:241–51. 10.1177/0022034514562833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Leme-Kraus AA, Phansalkar RS, Dos Reis MC, Aydin B, Sousa ABS, Alania Y, McAlpine J, Chen SN, Pauli GF, Bedran-Russo AK. Dimeric proanthocyanidins on the stability of dentin and adhesive biointerfaces. J Dent Res 2020;99:175–81. 10.1177/0022034519892959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Trevelin LT, Alania Y, Mathew M, Phansalkar R, Chen S-N, Pauli GF, Bedran-Russo AK. Effect of dentin biomodification delivered by experimental acidic and neutral primers on resin adhesion. J Dent 2020;99:103354. 10.1016/j.jdent.2020.103354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Yourdkhani M, Leme-Kraus AA, Aydin B, Bedran-Russo AK, White SR. Encapsulation of grape seed extract in polylactide microcapsules for sustained bioactivity and time-dependent release in dental material applications. Dent Mater 2017;33:630–6. 10.1016/j.dental.2017.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Ma G Microencapsulation of protein drugs for drug delivery: Strategy, preparation, and applications. J Control Release 2014;193:324–40. 10.1016/j.jconrel.2014.09.003. [DOI] [PubMed] [Google Scholar]
- [14].Ahmed J, Varshney SK. Polylactides—Chemistry, properties and green packaging technology: a review. Int J Food Prop 2011;14:37–58. 10.1080/10942910903125284. [DOI] [Google Scholar]
- [15].Jain RA. The manufacturing techniques of various drug loaded biodegradable poly(lactide-co-glycolide) (PLGA) devices. Biomaterials 2000;21:2475–90. 10.1016/S0142-9612(00)00115-0. [DOI] [PubMed] [Google Scholar]
- [16].Anderson JM, Shive MS. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv Drug Deliv Rev 2012;64:72–82. 10.1016/j.addr.2012.09.004. [DOI] [PubMed] [Google Scholar]
- [17].Ouyang X, Huang X, Pan Q, Zuo C, Huang C, Yang X, Zhao Y. Synthesis and characterization of triethylene glycol dimethacrylate nanocapsules used in a self-healing bonding resin. J Dent 2011;39:825–33. 10.1016/j.jdent.2011.09.001. [DOI] [PubMed] [Google Scholar]
- [18].Genari B, Leitune VCB, Jornada DS, Camassola M, Pohlmann AR, Guterres SS, Samuel SMW, Collares FM. Effect of indomethacin-loaded nanocapsules incorporation in a dentin adhesive resin. Clin Oral Investig 2017;21:437–46. 10.1007/s00784-016-1810-7. [DOI] [PubMed] [Google Scholar]
- [19].Choi H-W, Um S-H, Rhee S-H. Preparation of fluoride-loaded microcapsules for anticariogenic bacterial growth using a coaxial ultrasonic atomizer. J Biomed Mater Res B Appl Biomater 2018;106:31–9. 10.1002/jbm.b.33810. [DOI] [PubMed] [Google Scholar]
- [20].Tezvergil-Mutluay A, Agee KA, Hoshika T, Carrilho M, Breschi L, Tjäderhane L, Nishitani Y, Carvalho RM, Looney S, Tay FR, Pashley DH. The requirement of zinc and calcium ions for functional MMP activity in demineralized dentin matrices. Dent Mater 2010;26:1059–67. 10.1016/j.dental.2010.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Aydin B, Hassan LS, Viana G, Bedran-Russo AK. Assessing collagen and micro-permeability at the proanthocyanidin-treated resin-dentin interface. J Adhes Dent 2016;18:529–34. 10.3290/j.jad.a37359. [DOI] [PubMed] [Google Scholar]
- [22].Stape THS, Mutluay MM, Tjäderhane L, Uurasjärvi E, Koistinen A, Tezvergil-Mutluay A. The pursuit of resin-dentin bond durability: Simultaneous enhancement of collagen structure and polymer network formation in hybrid layers. Dent Mater 2021;37:1083–95. 10.1016/j.dental.2021.03.010. [DOI] [PubMed] [Google Scholar]
- [23].Feitosa VP, Leme AA, Sauro S, Correr-Sobrinho L, Watson TF, Sinhoreti MA, Correr AB. Hydrolytic degradation of the resin-dentine interface induced by the simulated pulpal pressure, direct and indirect water ageing. J Dent 2012;40:1134–43. 10.1016/j.jdent.2012.09.011. [DOI] [PubMed] [Google Scholar]
- [24].Abdalla AI, Feilzer AJ. Four-year water degradation of a total-etch and two self-etching adhesives bonded to dentin. J Dent 2008;36:611–7. 10.1016/j.jdent.2008.04.011. [DOI] [PubMed] [Google Scholar]
- [25].Bedran-Russo AK, Pauli GF, Chen S-N, McAlpine J, Castellan CS, Phansalkar RS, Aguiar TR, Vidal CMP, Napotilano JG, Nam JW, Leme AA. Dentin biomodification: strategies, renewable resources and clinical applications. Dent Mater 2014;30:62–76. 10.1016/j.dental.2013.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Fawzy AS, Priyadarshini BM, Selvan ST, Lu TB, Neo J. Proanthocyanidins-loaded nanoparticles enhance dentin degradation resistance. J Dent Res 2017;96:780–9. 10.1177/0022034517691757. [DOI] [PubMed] [Google Scholar]
- [27].Kothamasu P, Kanumur H, Ravur N, Maddu C, Parasuramrajam R, Thangavel S. Nanocapsules: the weapons for novel drug delivery systems. Bioimpacts 2012;2:71–81. 10.5681/bi.2012.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Rennhofer H, Zanghellini B. Dispersion state and damage of carbon nanotubes and carbon nanofibers by ultrasonic dispersion: a review. Nanomaterials 2021;11:1469. 10.3390/nano11061469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Ashraf MA, Peng W, Zare Y, Rhee KY. Effects of size and aggregation/agglomeration of nanoparticles on the interfacial/interphase properties and tensile strength of polymer nanocomposites. Nanoscale Res Lett 2018;13:214. 10.1186/s11671-018-2624-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Michler GH, von Schmeling H-HK-B. The physics and micro-mechanics of nano-voids and nano-particles in polymer combinations. Polymer 2013;54:3131–44. 10.1016/j.polymer.2013.03.035. [DOI] [Google Scholar]
- [31].Alania Y, Chiari MDS, Rodrigues MC, Arana-Chavez VE, Bressiani AHA, Vichi FM, Braga RR. Bioactive composites containing TEGDMA-functionalized calcium phosphate particles: Degree of conversion, fracture strength and ion release evaluation. Dent Mater 2016;32:e374–81. 10.1016/j.dental.2016.09.021. [DOI] [PubMed] [Google Scholar]
- [32].Tay FR, Moulding KM, Pashley DH. Distribution of nanofillers from a simplified-step adhesive in acid-conditioned dentin. J Adhes Dent 1999;1:103–17. [PubMed] [Google Scholar]
- [33].Chiaraputt S, Mai S, Huffman BP, Kapur R, Agee KA, Yiu CKY, Chan DCN, Harnirattisai C, Arola DD, Rueggeberg FA, Pashley DH, Tay FR. Changes in resin-infiltrated dentin stiffness after water storage. J Dent Res 2008;87:655–60. 10.1177/154405910808700704. [DOI] [PubMed] [Google Scholar]
- [34].He L, Mu C, Shi J, Zhang Q, Shi B, Lin W. Modification of collagen with a natural cross-linker, procyanidin. Int J Biol Macromol 2011;48:354–9. 10.1016/j.ijbiomac.2010.12.012. [DOI] [PubMed] [Google Scholar]
- [35].Fathima NN, Baias M, Blumich B, Ramasami T. Structure and dynamics of water in native and tanned collagen fibers: Effect of crosslinking. Int J Biol Macromol 2010;47:590–6. 10.1016/j.ijbiomac.2010.08.003. [DOI] [PubMed] [Google Scholar]
- [36].Althaqafi KA, Satterthwaite J, Silikas N. A review and current state of autonomic self-healing microcapsules-based dental resin composites. Dent Mater 2020;36:329–42. 10.1016/j.dental.2019.12.005. [DOI] [PubMed] [Google Scholar]
- [37].Wu J, Zhou C, Ruan J, Weir MD, Tay F, Sun J, Melo MAS, Oates TW, Chang X, Xu HHK. Self-healing adhesive with antibacterial activity in water-aging for 12 months. Dent Mater 2019;35:1104–16. 10.1016/j.dental.2019.05.004. [DOI] [PubMed] [Google Scholar]


