Abstract
Propionic acidemia (PA) is a rare autosomal-recessive metabolic disease that arises from mutations in propionyl-CoA (C3-CoA) carboxylase. Reduced enzyme activity slows C3-CoA metabolism leading to an elevated plasma C3:C2-carnitine ratio, the hallmark biomarker of PA. The metabolic imbalances experienced in PA are however poorly defined. Here we used a hypomorphic PA mouse model to demonstrate that C3-CoA accumulation in liver reduced non-esterified CoA (CoASH) and acetyl-CoA (C2-CoA). Tricarboxylic acid (TCA) cycle intermediates that are normally metabolized accumulated in the urine, providing direct evidence for compromised mitochondrial function in PA. Pantothenate kinase (PanK) is known to catalyze the rate-controlling step in CoA biosynthesis and its inhibition by C3-CoA prevents an increase in CoA biosynthesis toalleviate CoASH sequestration. PZ-3022 is an allosteric PanK activator that counteracts C3-CoA inhibition. PZ-3022 therapy increased hepatic CoASH and C2-CoA and decreased C3-CoA in the PA mouse model, leading to improved intracellular C3:C2-CoA and plasma C3:C2-carnitine ratios. Elevated urinary malate is a major component of the metabolic signature for TCA cycle dysfunction in the PA mouse and the 80% reduction in urine malate by PZ-3022 therapy indicates the restoration of mitochondrial function. Thus, CoASH sequestration in PA leads to reduced TCA cycle activity that is relieved following PZ-3022, providing preclinical proof of concept for PanK activators as a therapy to attenuate the underlying mitochondrial defect in PA.
One-sentence Summary:
Therapeutic relief of CoA sequestration improves mitochondrial dysfunction in a propionic acidemia mouse model.
Propionic acidemia (PA, OMIM 606054) is a devastating inborn error of metabolism with substantial morbidity and mortality (1). PA arises from missense mutations in one of the mitochondrial propionyl-CoA carboxylase (PCC) (E.C. 6.4.1.3) genes (PCCA or PCCB) that produce proteins with diminished catalytic activity, resulting in compromised catabolism of propionyl-CoA (C3-CoA) and disrupted metabolism (2-6). The CoA-dependent TCA cycle activity is dysfunctional in patients with PA, leading to TCA cycle intermediates being excreted in urine (7, 8). Reduced PCC activity is thought to lead to the accumulation of intracellular C3-CoA, which is converted to propionyl-carnitine (C3-carnitine) that is released into the plasma and urine (2). The severity of PA varies depending on the impact of the mutations on PCC catalytic activity (3-6, 9) and the plasma C3:C2-carnitine ratio is a key biomarker used to screen newborns (10). C3-carnitine is not considered a toxic metabolite but rather a mechanism to release non-esterified CoA (CoASH) and eliminate excess propionate from the body (2). Methylcitrate formed by the condensation of C3-CoA with oxaloacetate is another mechanism to release CoASH from C3-CoA. Methylcitrate is not a substrate for ATP:citrate lyase or aconitase (7, 11) and its elimination in urine removes propionate from the body. There are no approved therapies to treat PA, and patients with this disease are placed on low-protein or synthetic diets to reduce C3-CoA formation (1, 12). Human liver transplantation lowers the circulating concentrations of PA biomarkers and improves quality of life, although patients are still at risk of developing complications (13-19). Gene or RNA therapy are approaches being explored that reduce the concentrations of PA biomarkers in mouse models (20-23).
CoASH functions as a cofactor in numerous reactions in intermediary metabolism and is a critical substrate for two key steps in mitochondrial energy metabolism: pyruvate and α-ketoglutarate dehydrogenases. The concept of CoASH sequestration or trapping as a metabolic basis for disease was advanced in the 1990s to explain the cellular toxicity of xenobiotic carboxylic acids (24) such as valproate (25, 26), pivalate (27, 28), and benzoic acid (29). These compounds are converted to CoA thioesters leading to a reduction in CoASH needed to support intermediary metabolism and TCA cycle function (26). CoASH sequestration was proposed as an underlying cause for metabolic dysfunction in acidemia diseases (24, 30) based on the inhibition of pyruvate oxidation (31, 32), fatty acid oxidation (32, 33), ureagenesis (34, 35), and gluconeogenesis (34-36) in ex-vivo cultures treated with propionate. Treatment of hepatocytes with propionate decreases CoASH and C2-CoA concomitant with the rise in C3-CoA (37).
The cellular CoA concentration is controlled by the activity of pantothenate kinase (PanK), the first and rate-controlling step in CoASH biosynthesis (38). PanK activity is potently inhibited by CoA thioesters (39-41). The PanK•acetyl-CoA complex crystal structure shows CoA thioesters stabilize a PanK conformation that cannot interact with ATP (39, 42). Both active sites in each protomer simultaneously switch between the inactive, acyl-CoA-bound conformation to the active, ATP:Mg2+-bound state (39). PanK inhibition in liver leads to mitochondrial dysfunction highlighted by reduced fatty acid β-oxidation capacity and gluconeogenesis (43). Analysis of Pank1−/− mice shows that reduced liver CoASH prevents normal fuel switching to mitochondrial fatty acid oxidation during fasting (44). Pank1−/−Pank2−/− and Pank1−/−Syn-Pank2−/− mice are more severely compromised and succumb to a postnatal metabolic crisis (45, 46).
Here, we use a metabolomics approach to measure liver total CoA, CoASH, C2-CoA and C2-carnitine to determine if CoASH sequestration occurs in the Pcca−/−PCCA(A138T)tg/0 (PA) mouse model. The function of the TCA cycle was investigated by measuring the levels of TCA cycle intermediates in plasma and urine of PA mice. Pantazines are drug-like small molecules that elevate CoASH in cells by rendering PanK refractory to feedback inhibition by CoA thioesters (47). We determined whether the treatment of PA mice with an optimized pantazine, PZ-3022, leads to the normalization of hepatic total CoA, CoASH, C2-CoA and C2-carnitine. The impact of PZ-3022 therapy on the plasma C3:C2-carnitine ratio and the excretion of TCA cycle intermediates into the urine were also measured to determine if PanK activation by PZ-3022 relieves the metabolic stress on the TCA cycle that arises from trapping CoASH as C3-CoA in PA.
Results
Pcca−/−PCCA(A138T)tg/0 (PA) mice
Pcca−/− mice die shortly after birth and exhibit massive elevation in serum C3-carnitine and methylcitrate (20, 48). These animals are not suitable as PA models, therefore a hypomorphic mouse was created by complementing Pcca−/− mice with a transgene expressing a mutant human PCCA(A138T) allele (49). The transgene expresses a protein with ~9.4% of wild-type PCC activity (3, 4). Although the mutant PCCA allele is not expressed in its native genetic context, this mouse has been used extensively to model PA disease (20, 22, 23, 50-53). Human PA arises from many different mutations in PCC and the metabolic phenotypes vary from mild to severe depending on the nature of the mutation (3, 4, 6), meaning any knock-in or transgenic mouse would model only a subset of human PA. We used a breeding program to generate Pcca−/−PCCA(A138T)tg/0 mice that harbor a single copy of the transgene; however, control over the transgene copy number is not specified in many studies, suggesting the mice may be a mixture (20, 22, 50-53). One report used a mouse model with two copies of the transgene (23) that is anticipated to be less affected than an animal with a single transgene copy. Our first goal was to establish a baseline for the metabolic markers in the Pcca−/−PCCA(A138T)tg/0 mouse and validate the model, and we established the PA mouse liver as representative of human disease to study the metabolic imbalances caused by mutations that reduce liver PCC activity.
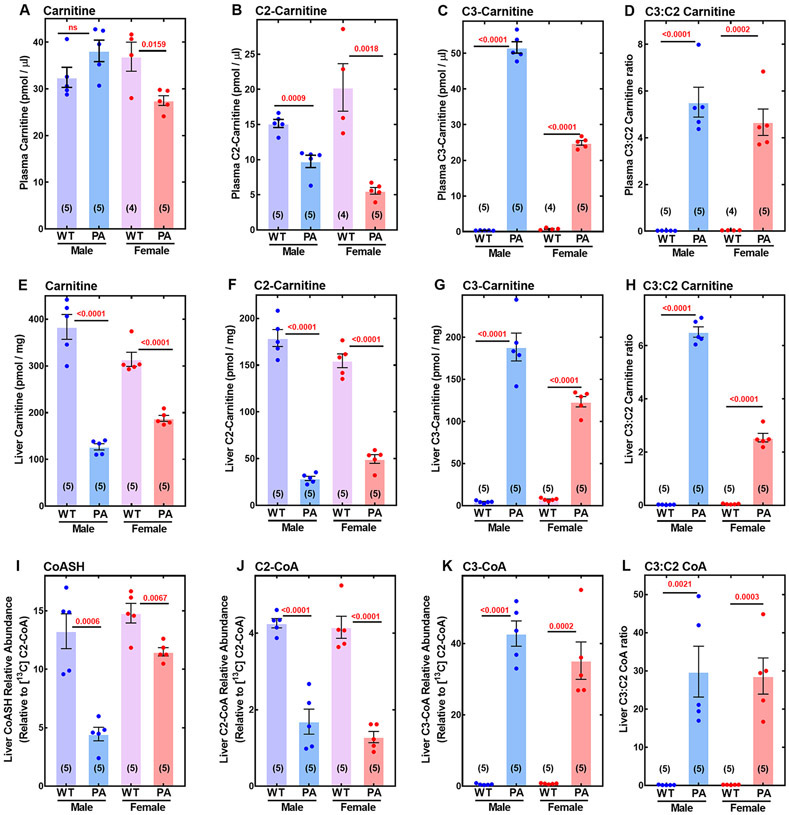
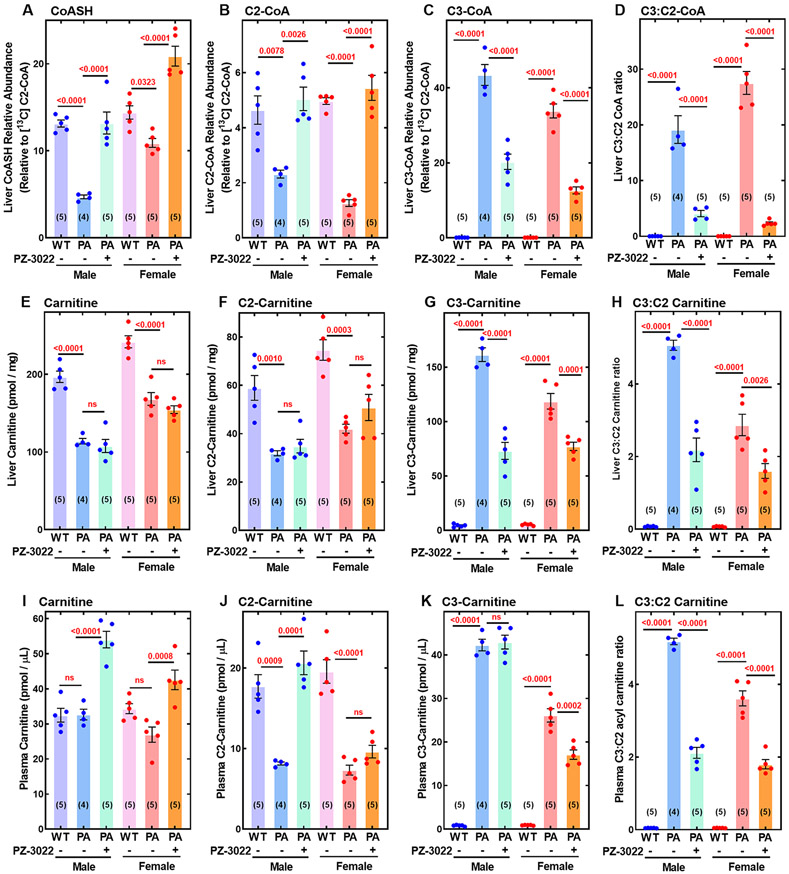
Acyl-carnitines in PA mice were quantified by mass spectrometry using [d9]carnitine, [d3]C2-carnitine and [d3]C3-carnitine as internal standards. Other carnitine species were present in low abundance, did not change in PA mice and were not analyzed further (fig. S1). Free carnitine (Fig. 1A) and C2-carnitine (Fig. 1B) were the major species in plasma of wild-type animals, whereas C3-carnitine (Fig. 1C) was in low abundance. The PA mice exhibited a massive elevation in plasma C3-carnitine that is the hallmark biomarker of human PA (Fig. 1C). Male PA mice had higher plasma C2- and C3-carnitines compared to the female PA mice (Fig. 1B & 1C), but the plasma C3:C2-carnitine ratios in both the male and female PA mice were similar (Fig. 1D). The clinical assay for the C3:C2 carnitine ratio is performed using MALDI mass spectrometry (49), which evaluates the relative ratios of the two metabolites. Our quantitative results show that a decrease in circulating C2-carnitine (Fig. 1B) in the PA mice contributed to the elevated C3:C2-carnitine ratio (Fig. 1D) in addition to increased C3-carnitine (Fig. 1C). In liver, carnitine (Fig. 1E) and C2-carntitine (Fig. 1F) were the major species in wild-type mice, and although C3-carnitine was detected, it was a minor component (Fig. 1G). PA mice had higher hepatic C3-carnitine coupled with decreased carnitine and C2-carntine. C3-carnitine was lower, and C2-carnitine and carnitine were higher in female compared to male liver resulting in an elevated liver C3:C2-carnitine ratio in males compared to females (Fig. 1H). Sexual dimorphism in acyl-carnitines is recognized in rodents (54, 55) and was evident in PA mice. Together, these data show that PA mice have the hallmark metabolic signature of PA.
Fig. 1. CoASH sequestration in PA mice.
Wild-type (WT) and Pcca−/−PCCA(A138T)tg/0 (PA) mice were maintained on a standard rodent chow and samples were harvested at day 68-71. (A) Plasma carnitine. (B) Plasma C2-carnitine. (C) Plasma C3-carnitine. (D) Plasma C3:C2-carnitine ratio. (E) Liver carnitine. (F) Liver C2-carnitine. (G) Liver C3-carnitine. (H) Liver C3:C2-carnitine ratio. (I) Liver CoASH (non-esterified CoA). (J) Liver C2-CoA. (K) Liver C3-CoA. (L) Liver C3:C2-CoA ratio. Male mice are in blue and female mice are in red. [13C]C2-CoA was used as the internal standard (fig. S2). The p values are in red. ns indicates p > 05. Sample sizes of animals are in parentheses.
CoASH sequestration in PA
The CoASH sequestration hypothesis posits that the accumulation of C3-CoA leads to reduced CoASH and C2-CoA. The amounts of liver CoASH, C2-CoA, and C3-CoA were determined by mass spectrometry relative to a [13C]acetyl-CoA internal standard in wild-type and PA liver. The detector response to C3-CoA was similar to C2-CoA, but CoASH was detected at about 2.4-fold lower efficiency (fig. S2) meaning that CoASH is about 2.4-fold more abundant than it appears in this analysis. There was no difference in liver CoASH (Fig. 1I) or C2-CoA (Fig. 1J) in male and female wild-type mice, and C3-CoA was a minor component (Fig. 1K). In PA mice, C3-CoA was increased relative to wild-type (Fig. 1K) and was associated with a decrease in CoASH (Fig. 1I) and C2-CoA (Fig. 1J). The C3:C2-CoA ratio ranged from 17 to 49 (Fig. 1L) which is 5-10 times higher than the C3:C2-carnitine ratios in either the liver or the plasma of affected animals. These data show that CoA is trapped as C3-CoA in PA liver leading to the depression of CoASH and C2-CoA.
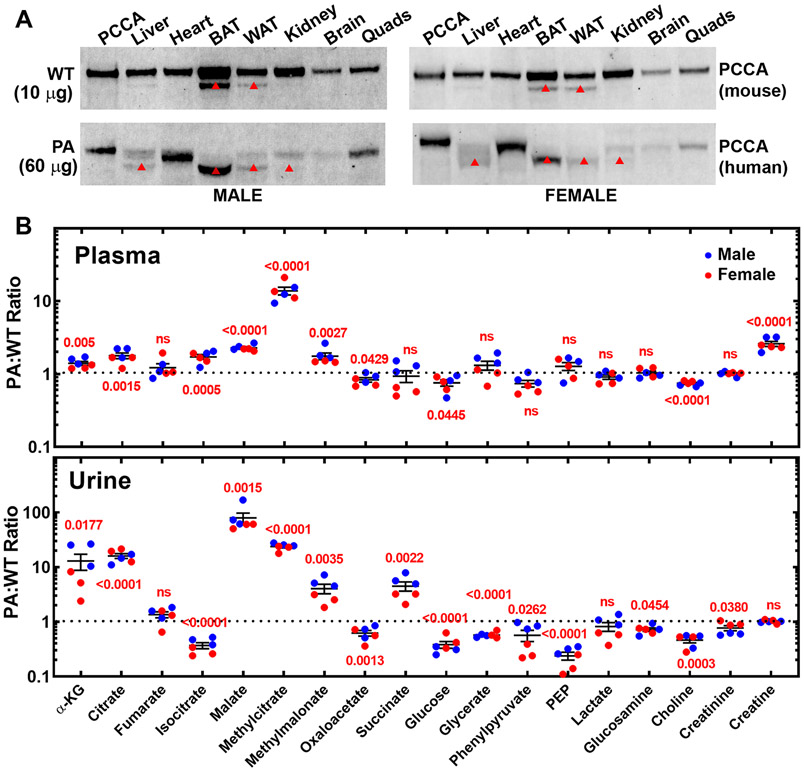
Liver had the highest amount of cellular C3-CoA compared to heart, quadriceps muscle, and brain (fig. S3A). Heart complications arise in human PA (56) and cardiac dysfunction was noted in 58% of 8-month-old PA mice in a previous study (53). Therefore, we examined heart CoA pool composition in more detail. The CoASH (fig. S3B) and C2-CoA were not altered (fig. S3C). Although C3-CoA was elevated in heart (fig. S3D), the magnitude of C3-CoA accumulation was 40-fold lower than in liver. There was no decrease in C2-CoA in PA mouse heart illustrating that CoA sequestration did not occur in this tissue. There were no alterations in electrocardiograms between 70-day old wild-type and PA mice (table S1). The 16% reduction in ejection fraction observed in the 8-month PA mice was not evident in the 70-day old animals. We did observe an increase in the heart:body weight ratio as previously reported (49) (fig. S12B). The high PCCA transgene expression in heart (Fig. 2A; fig. S4) accounts for the modest increase in C3-CoA (fig. S3) and identifies a confounding issue with this mouse model.
Fig. 2. Transgenic PCCA expression and TCA cycle metabolites in PA mice.
(A) Western blot analysis of PCCA(A138T) transgene expression in WT and PA mice. This blot illustrates the tissue-specific distribution of murine PCCA in WT mice (10 μg/lane) compared to the expressed human transgene PCCA(A138T) protein in PA mice (60 μg/lane). Western blots for each tissue were obtained from triplicate mice (fig. S3), and a fourth blot was performed for this figure. The red triangle indicates a non-specific band. (B) A metabolomics screen of TCA cycle metabolites in plasma (upper panel) and urine (lower panel) of three male and three female PA mice. The p values are in red.
PCCA(A138T) expression
The hybrid cytomegalovirus early enhancer/chicken β-actin/rabbit β-globin (CAG) promoter was used to drive expression of the PCCA(A138T) transgene. We compared the expression of the transgene-derived human PCCA protein to the wild-type mouse PCCA protein in a series of tissues (Fig. 2A; fig. S4). Mouse PCCA exhibited a tissue-specific abundance pattern in mice that was highest in adipose tissue followed by liver, kidney, and heart. Human PCCA protein expressed by the PCCA(A138T) transgene was higher in skeletal muscle and heart compared to other tissues, similar to that reported in transgenic rodents expressing green fluorescent protein, human ATP7A or PANK2 driven by the same CAG promoter (57-60). Transgene-encoded human PCCA did not have the same tissue-specific abundance as endogenous mouse PCCA. For example, liver contained far less of the transgene-expressed human protein than was present in wild-type mice, and mouse adipose tissue, which expresses the highest amounts of mouse PCCA, was devoid of human PCCA protein expression. In contrast, heart and quadriceps muscles contained the same amount of transgene-derived PCCA as murine PCCA. Thus, heart is not as compromised in PCCA protein content as liver. PA mice had altered liver total fatty acids (fig. S5). Odd-chain fatty acids, which arise from C3-CoA, were below detection in wild-type mice but collectively rose to 8.8% of total fatty acids in male and female PA mice. Thus, the tissue distribution of PCCA(A138T) transgene expression identifies the liver of the PA mouse as most representative of severe human disease.
TCA metabolites
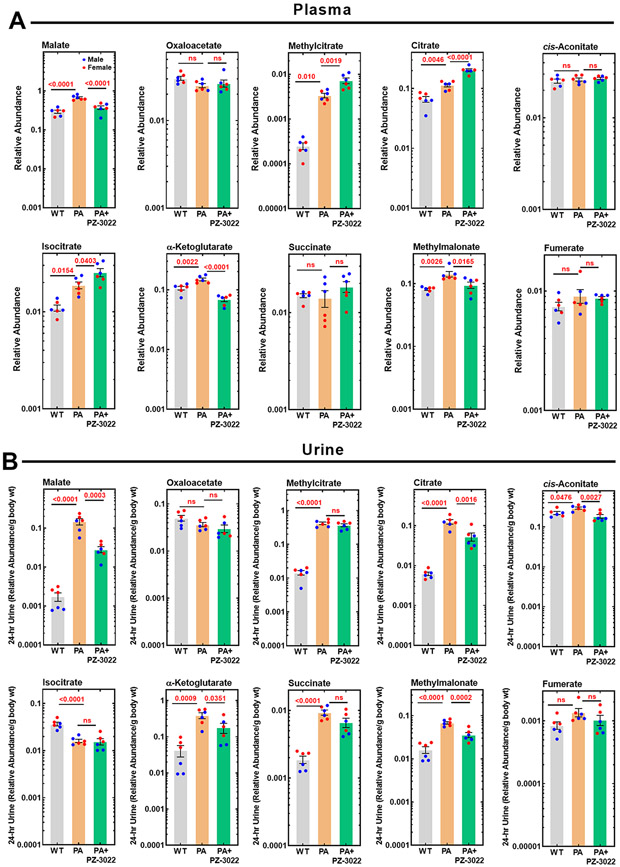
Dysfunctional mitochondrial metabolism is a characteristic of PA (24, 30-37), and the release of TCA cycle intermediates from cells and tissues signals mitochondrial malfunction (61-63). We hypothesized that reduced mitochondrial production of C2-CoA via PDH is a consequence of CoA sequestration and low CoASH leading to alternate fates for pyruvate and TCA cycle intermediates. The profiling of liver metabolites following propionate stress shows that when C3-CoA becomes the dominant CoA species, malate increases 6.6-fold along with other TCA cycle metabolites (64). Urinary TCA intermediates are not used as human PA biomarkers, but there is evidence for elevated of TCA cycle intermediates in PA urine (7, 8). We used a metabolomics screen to identify changes in TCA cycle metabolites, selected amino acids, and other metabolites in the PA mouse. Individual amino acid concentrations in plasma were not greatly altered in the PA mouse, but there were modest decreases in some urinary amino acids, such as tyrosine, proline and methionine (fig. S6). Glycine is elevated 2-10 fold in human PA depending on the patient (65-67), but there was only a modest elevation (50%) in plasma glycine in the PA mouse model (49). We measured glycine as its benzoyl derivative to increase detection sensitivity (68, 69) in PA mice and confirmed plasma glycine was elevated 40% in plasma but was unchanged in urine (fig. S6). The major alterations observed were in TCA cycle intermediates (Fig. 2B). Methylcitrate, formed from C3-CoA and oxaloacetate by citrate synthase and an established marker of PA disease, was elevated in plasma and urine (Fig. 2B). Malate, methylmalonate, isocitrate, citrate and α-ketoglutarate were all elevated in plasma, but TCA metabolites in urine were the clearest markers of mitochondrial dysfunction in PA mice. Malate (85-fold), succinate (5-fold), α-ketoglutarate (9-fold), methylmalonate (4-fold) and citrate (20-fold) were all elevated in urine. Malate was the most elevated biomarker in urine, and the smaller increase in malate in the plasma compared to urine was consistent with the saturation of malate resorption by kidney (70). These data suggest that pyruvate-derived TCA cycle intermediates normally used for energy production or intermediary metabolism are eliminated in the urine, providing a collective indicator of mitochondrial dysfunction in PA mice.
Properties of PZ-3022
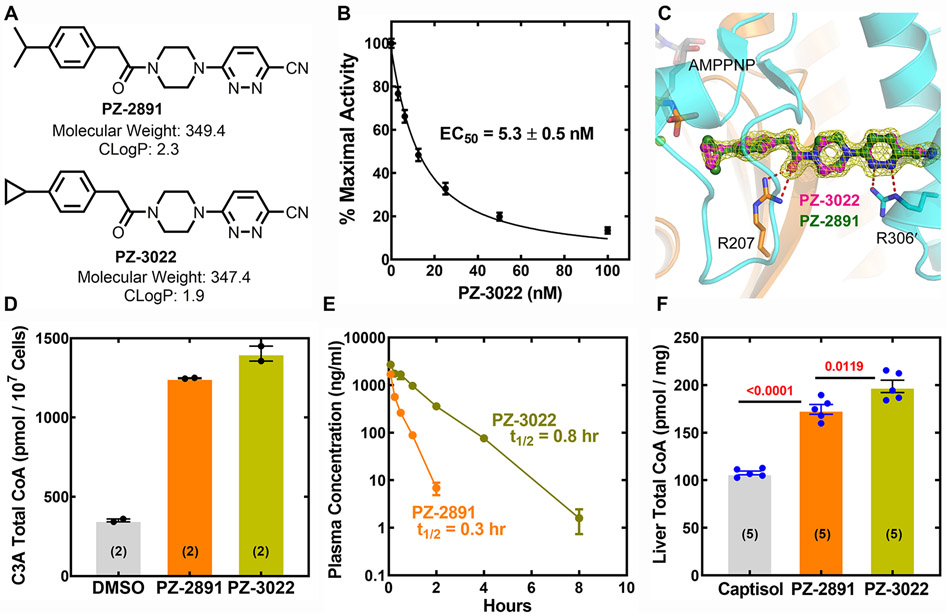
We previously developed a series of drug-like, small molecule allosteric activators of PanK called pantazines that render the enzyme resistant to feedback inhibition by CoA thioesters (47). The first generation pantazine, PZ-2891, effectively elevated intracellular CoA in cell cultures, and in the tissues of treated mice (47). A drawback to PZ-2891 was its short half-life in circulation (47). Therefore, PZ-3022 was developed to circumvent this problem by replacing the isopropyl group with a cyclopropyl moiety (Fig. 3A; fig. S7; fig. S8). PZ-3022 had an EC50 of 5.3 nM against PanK3 (Fig. 3B) and had a lower affinity for each of the PanK isoforms compared to PZ-2891 (table S2). The crystal structure shows PZ-3022 bound to the PanK dimer in the same location and orientation as PZ-2891 (Fig. 3C; table S3). The cyclopropyl group of PZ-3022 binds where the isopropyl group of PZ-2891 resides and PZ-3022 extends across the dimer interface to affect the active site on the opposite protomer. PZ-3022 was just as effective at raising total CoA in cultured C3A cells as PZ-2891 (Fig. 3D). The major impact of introducing the cyclopropyl group was the decreased rate of PZ-3022 clearance compared to PZ-2891 (table S4) due to a longer half-life (Fig. 3E). The key metabolite arises from hydroxylation of the isopropyl sidechain of PZ-2891 that, in turn, triggers further metabolism to a variety of hydroxylated products (fig. S9A). PZ-3022 was refractory to this hydroxylation event leading to reduced drug metabolism and higher circulating PZ-3022 (fig. S9B). These properties combined with the high oral bioavailability of PZ-3022 (table S4) resulted in higher concentrations of circulating (fig. S9C) and liver (fig. S9D) PZ-3022 compared to PZ-2891 after the same oral drug dosage. Like PZ-2891, PZ-3022 rendered all three PanK isoforms refractory to inhibition by C3-CoA (fig. S10). A direct comparison of the ability of PZ-3022 and PZ-2891 to elevate hepatic total CoA in C57BL/6J mice showed that PZ-3022 was slightly more potent in elevating CoA than PZ-2891 (Fig. 3F). Thus, the enhanced metabolic stability and bioavailability of PZ-3022 improves the overall efficacy of pantazine dosing in mice.
Fig. 3. Properties of PZ-3022 and its impact on CoA in PA mouse liver.
(A) Chemical structures and relevant properties of PZ-2891 and PZ-3022. Purity and NMR spectra of PZ-3022 are shown in figs. S8 & S9. (B) Inhibition of PANK3 by PZ-3022. Data were fit to the Morrison equation (line) and EC50 was calculated. (C) Crystal structure of the PanK3•AMPPNP•Mg2+•PZ-3022 complex (PDB ID: 6PE6) overlaid on the PZ-2891 structure (PDB ID: 6B3V). The two PANK3 protomers are colored gold and cyan. The Fo-Fc simulated annealing omit map is contoured at 3 σ (yellow mesh). (D) Elevation of total cellular CoA in C3A cells treated with 10 μM of either PZ-2891 or PZ-3022. (E) Half-life of PZ-2891 compared to PZ-3022 in mice. Complete pharmacokinetic profiles are in table S4. (F) Male C57BL/6 mice were orally gavaged every 24 h with Captisol containing 10 mg/kg of either PZ-2891 or PZ-3022 for three days, and liver total CoA was determined 4 h after the last dose using a fluorescent derivatization assay. The p values are in red. Number of biological replicates are in parentheses.
PZ-3022 relieves CoASH sequestration
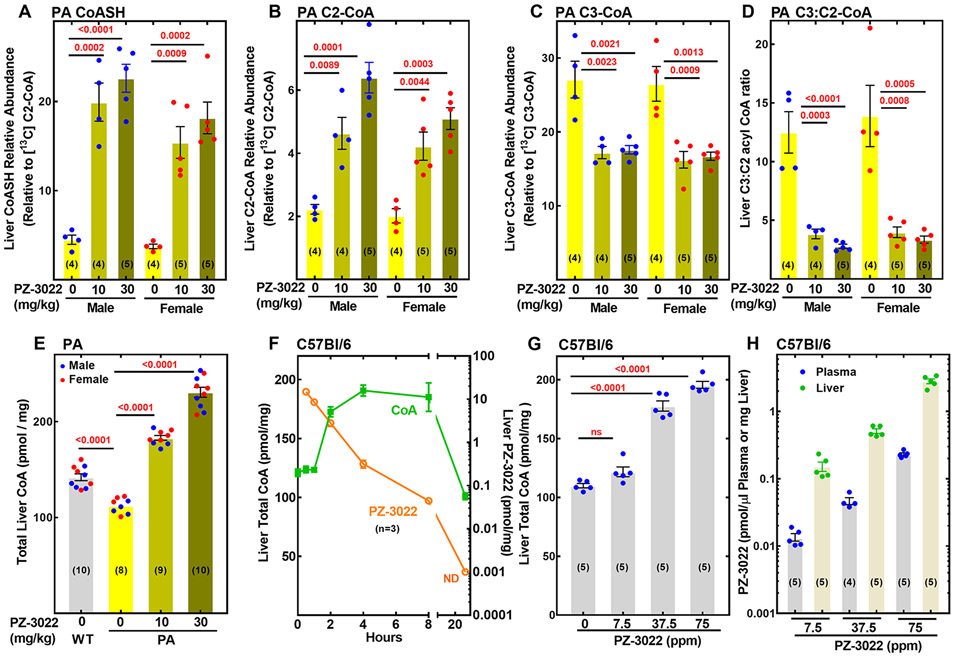
We evaluated the impact of treating PA mice by oral gavage with 10 or 30 mg/kg of PZ-3022 on hepatic CoA species distribution and plasma acyl-carnitines. Pantothenate potentiates the effect of pantazines (47), therefore a 50 mg/kg dose of pantothenate was included with PZ-3022, and a dose of 50 mg/kg of pantothenate served as the control. Groups of 4-5 male or female animals were orally gavaged with 3 doses delivered at 24 h intervals, and samples were collected four hours after the last dose. Both 10 and 30 mg/kg doses of PZ-3022 had an impact on liver CoA thioesters. Along with elevated CoASH (Fig. 4A), C2-CoA increased (Fig. 4B) and C3-CoA decreased but was not eliminated (Fig. 4C). These metabolic changes led to a marked improvement in the C3:C2-CoA ratio (Fig. 4D). Plasma and liver carnitine species were modestly altered by short-term PZ-3022 therapy resulting in a reduction at the 30 mg/kg dose in the C3:C2-carnitine ratio in males but not females (fig. S11). These data show that PZ-3022 treatment relieved CoASH sequestration in liver by increasing hepatic CoASH and C2-CoA and decreasing C3-CoA, consistent with the stimulation of TCA cycle flux. The total amount of CoA was measured using a fluorescent derivatization and liquid chromatography method (71, 72) that quantified CoASH together with acyl-CoA species. Total CoA measurements revealed a reduced total CoA in PA liver and a dose-dependent elevation in total CoA in the livers of PZ-3022-treated PA mice (Fig. 4E). Male and female total CoA amounts responded similarly to PZ-3022.
Fig. 4. Acute treatment with PZ-3022 relieves CoASH sequestration in PA mouse liver.
Mice were orally gavaged every 24 h with either 10 or 30 mg/kg PZ-3022 plus 50 mg/kg pantothenate. Control animals received 50 mg/kg pantothenate. Four hours after the third dose, the impact of short-term PZ-3022 treatment on liver CoA pools was determined using mass spectrometry. (A) CoASH. (B) C2-CoA. (C) C3-CoA. (D) C3:C2-CoA ratio. Male mice are in blue and female mice are in red. (E) Elevation of total hepatic CoA in PA mice dosed with PZ-3022. (F) C57BL/6J male mice were administered PZ-3022 by gavage and at the indicated times samples were taken (3 mice per point) to determine plasma PZ-3022 and liver total CoA. (G) Envigo chow containing 1000 ppm pantothenate was formulated with 7.5, 37.5 and 75 ppm PZ-3022. C57BL/6J male mice were maintained on the diets for 1 week and total liver CoA was determined. (H) PZ-3022 in plasma and liver as a function of PZ-3022 in the diet. The p values are in red. ns indicates p > 0.05. Mouse sample sizes are in parentheses.
We next determined the relationship between liver CoA elevation and drug exposure. These data show that in C57BL/6J mice the elevation of liver CoA began at 2 h after PZ-3022 administration and was maintained up to 8 h (Fig. 4F). However, circulating PZ-3022 was not detected at 24 h and the hepatic CoA had returned to the pre-dose concentrations, meaning that frequent dosing of PZ-3022 would be needed to maintain elevated liver CoA (Fig. 4F). This was accomplished by formulating PZ-3022 in the diet. We maintained wild-type C57BL/6J mice for one week on three concentrations of PZ-3022 in the chow and determined the impact on hepatic CoA (Fig. 4G). There was a dose-response relationship between total liver CoA and concentration of PZ-3022 in chow. Measurement of the plasma and liver PZ-3022 showed a dose-dependent increase of the compound (Fig. 4H). Based on the amount of CoA elevation exhibited by liver, the 75-ppm dose of PZ-3022 was selected to evaluate PZ-3022 therapy.
PZ-3022 therapy
PZ-3022 was tested as a PA therapeutic by introducing the compound into the mouse chow to provide a daily dose of the compound over the course of weeks. Mice (n = 5 per group) were maintained on chow formulated with 75 ppm PZ-3022 and 1000 ppm pantothenate to determine if the alterations in liver CoA thioesters induced by short-term PZ-3022 therapy were durable. Animals were placed on the diets at weaning (21 days) and when the mice reached 70 days of age, tissue and plasma samples were analyzed (Fig. 5). PZ-3022 therapy restored liver CoASH (Fig. 5A) and liver C2-CoA (Fig. 5B) to wild-type amounts in males and females (Fig. 5A). There was also a drop in hepatic C3-CoA (Fig. 5C) leading to a substantial decrease in the C3:C2-CoA ratio (Fig. 5D). PZ-3022 therapy thus alleviates the impact of C3-CoA accumulation on CoASH and C2-CoA in liver.
Fig. 5. Metabolic parameters in mice treated with PZ-3022 for 70 days.
Animals were maintained on a defined diet supplemented with 1000 ppm pantothenate either with or without 75 ppm PZ-3022 beginning at weaning on day 21. On day 68-70, liver CoAs and carnitines were determined by mass spectrometry in male (blue) and female (red) mice. (A-H) Liver: CoASH (A), C2-CoA (B), C3-CoA (C), C3:C2-CoA ratio (D), carnitine (E), C2-carnitine (F), C3-carnitine (G), and C3:C2-carnitine ratio (H). (I-L) Plasma: carnitine (I), C2-carnitine (J), C3-carnitine (K), and C3:C2-carnitine ratio (L). The p values are in red. ns indicates p > 0.05. Mouse sample sizes are in parentheses.
Liver carnitine profiles were also improved by PZ-3022 therapy. Carnitine (Fig. 5E) and C2-carnitine (Fig. 5F) did not increase after PZ-3022 treatment; however, liver C3-carnitine formation decreased in both male and female mice (Fig. 5G) leading to a corresponding decrease in the C3:C2-carnitine ratio in liver (Fig. 5H). An increase in plasma carnitine was observed in both male and female mice following PZ-3022 therapy (Fig. 5I). A key result was that PZ-3022 therapy increased C2-carnitine in the plasma of male PA mice (Fig. 5J) and lowered the C3-carnitine in female PA mice (Fig. 5K). We lack a mechanistic explanation for the differences in the male and female responses to PA and therapy, but sexual dimorphism is also observed in the PA mouse response to gene therapy (21, 23). PZ-3022 therapy substantially reduced the plasma C3:C2-carnitine ratio in both sexes (Fig. 5L). These data show that PZ-3022 therapy relieved hepatic CoA sequestration and elevated CoASH and C2-CoA. These alterations in hepatic CoA metabolism resulted in lower C3:C2-CoA ratio in liver and a reduced C3:C2-carnitine ratio in circulation, indicating an improvement in the metabolic state in PZ-3022-treated PA mice.
We examined a spectrum of urinary TCA cycle intermediates and related metabolites in plasma and urine to determine if mitochondrial utilization of these metabolites was improved by PZ-3022 therapy. Plasma malate, citrate, isocitrate, methylcitrate, α-ketoglutarate, and methylmalonate were elevated in plasma from PA mice indicating the leakage of TCA cycle intermediates out of the tissues (Fig. 6A). However, plasma metabolite concentrations are capped by the saturation of the kidney resorption mechanisms, and the most sensitive indicator of the amount of TCA cycle dysfunction was the accumulation of intermediates in urine (Fig. 6B). Urine malate, citrate, cis-aconitate, α-ketoglutarate and methylmalonate were reduced by PZ-3022 therapy. Malate was the most perturbed indicator of TCA cycle dysfunction, and PZ-3022 therapy reduced the excretion of malate by 80%. Urinary TCA cycle intermediates are a robust indicator of mitochondrial function and a potential indicator of the severity of TCA cycle dysfunction in PA disease. These data show that the relief of CoA sequestration by PZ-3022 therapy substantially improves compromised mitochondrial function based on the reduction in malate and other TCA cycle intermediates in urine.
FIG. 6.
TCA cycle intermediates in plasma and urine of treated PA mice.(A) The impact of PZ-3022 therapy on the relative abundance of plasma TCA cycle intermediates (Fig. 2). (B) Effect of PZ-3022 therapy on TCA cycle intermediates in urine. Urinary TCA cycle metabolites eliminated over 24 hours were quantified by mass spectrometry and normalized to mouse body weight. The two-tailed Student’s t test was used to compare two indicated groups, and P values <0.05 are reported in red. ns means P > 0.05. Three male (blue) and three female (red) mice were used.
Heart total CoA increased after PZ-3022 therapy (fig. S12A), but there was no deficiency in C2-CoA to correct (fig. S3C). The increase in the heart:body weight ratio in the PA mouse (49) (fig. S12B) was normalized after PZ-3022 treatment (fig. S12B), suggesting a beneficial effect of therapy. Liver CoASH and acetyl-CoA increased as expected (47) in PZ-3022-treated wide-type mice (fig. S13A & S13B). C3-CoA (fig. S13C) and the C3:C2 ratios (fig. S13D) were not altered by PZ-3022. Concentrations of plasma and urine amino acids in treated wild-type mice were not changed except for lower histidine in plasma and higher histidine in the urine of PZ-3022-treated mice (fig. S13E). There were small increases in isocitrate, α-ketoglutarate and succinate in plasma (fig. S14A), but not in urine (fig. S14B) of PZ-3022-treated wild-type mice.
Discussion
Our study demonstrates that CoASH sequestration contributes to metabolic imbalances in PA disease. Normally, C3-CoA arising from catabolism enters the TCA cycle for disposition following its conversion to succinyl-CoA by the reversible methylmalonyl-CoA mutase (fig. S15). In PA, this pathway is constricted at the PCC step leading to the accumulation of C3-CoA. C2-CoA is usually the most abundant feedback regulator of PanK, but in PA, C3-CoA amounts are 10-fold higher than C2-CoA. PanK inhibition by the massive accumulation of C3-CoA blocks CoASH biosynthesis thereby exacerbating the CoASH deficiency (fig. S15). Reduced hepatic CoASH impairs mitochondrial function at two key steps (pyruvate and α-ketoglutarate dehydrogenases) leading to reduced C2-CoA in PA liver and the accumulation of TCA cycle intermediates in plasma and urine. Metabolism responds using several pathways to convert C3-CoA to CoASH and eliminate propionate from the body. Major pathways are the conversion of C3-CoA to either C3-carnitine or methylcitrate, which both liberate CoASH. C3-carnitine and methylcitrate exit tissues into the plasma and are eliminated from the body in the urine. This report identifies methylmalonyl-CoA hydrolysis as another pathway to liberate CoASH and excess propionate is removed from tissues by eliminating methylmalonate in the urine. These measures work together to counteract CoASH sequestration, but in the PA mouse liver they are insufficient to maintain CoASH and C2-CoA. PZ-3022 allosterically activates PanK to counteract its inhibition by C3-CoA and normalizes the intracellular concentrations of total CoA, CoASH, and C2-CoA, and reduces C3-CoA in PA liver. Together, these changes result in an improved plasma C3:C2-carnitine ratio, the hallmark biomarker of human PA.
Metabolomics profiling establishes a dysfunctional TCA cycle as an underlying metabolic consequence of PA that arises from CoASH sequestration (fig. S15). Animal models of CoA deficiency indicate that compromised mitochondrial metabolism is a major consequence of insufficient cellular CoASH (43-46, 73) suggesting that reduced CoASH is at the root of mitochondrial dysfunction in PA. CoASH is a key substrate for two irreversible steps in the TCA cycle, pyruvate and α-ketoglutarate dehydrogenases (fig. S15). CoASH deficiency slows these steps, reducing the ability of mitochondria to metabolize pyruvate and TCA cycle intermediates. Instead, these intermediates diffuse out of tissues into the plasma and accumulate in urine. Elevated TCA cycle intermediates in PA mouse urine provide direct evidence for a dysfunctional TCA cycle and corroborate the similar observations in human PA (7, 8). TCA cycle intermediates are not routinely assessed in plasma or urine of PA patients but future work will determine if they may serve as biomarkers for the severity of mitochondrial impairment or to assess treatment efficacy. Urinary malate is a sensitive indicator of TCA cycle function in the PA model that has not been previously appreciated. Plasma malate increased 3-fold, but malate was 85-fold higher in urine in PA mice. This relationship between plasma and urinary malate is consistent with the established limit on malate reabsorption by the kidney (70), a process that explains why many TCA intermediates are higher in urine than in plasma. PZ-3022 therapy of PA mice resulted in an 80% reduction in urinary malate, providing direct evidence that PZ-3022 therapy enhances mitochondrial function that is compromised by PA. TCA cycle dysfunction is so compromised by CoASH sequestration in PA that the methylmalonyl-CoA produced by the limited amount of PCC activity present in PA liver cannot be metabolized by the TCA cycle. Instead, methylmalonyl-CoA is cleaved to methylmalonate to release CoASH and is eliminated in the urine. After PZ-3022 therapy, succinyl-CoA arising from C3-CoA is utilized by the TCA cycle leading to a 50% reduction in C3-CoA in liver and a 47% reduction in urinary methylmalonate. Gene or RNA therapy to correct PCC activity in PA liver is another current focus for therapeutics development (20-23). These therapies restore liver PCC activity and reduce circulating C3-carnitine and methylcitrate biomarkers. However, CoASH biosynthesis is cell autonomous and correcting liver metabolism would not alleviate CoASH sequestration and TCA cycle malfunction in other tissues. PZ-3022 stabilizes intermediary metabolism and TCA cycle function in the PA mouse model providing translational proof-of-concept that pantazine therapy may improve mitochondrial function in human PA disease.
Pantazines are first in class allosteric PanK activators with the potential to be disease-modifying drugs in other conditions involving CoASH sequestration or mitochondrial dysfunction. CoASH is the major organic acid carrier in biology and several organic acidemias and β-oxidation inborn errors of metabolism are predicted to trigger the accumulation of a particular CoA thioester (74, 75) that sequesters CoASH and inhibits CoASH biosynthesis. Pantazines may treat toxicities arising from exposure to xenobiotic carboxylic acids (24, 27-29). Valproate is a widely used antiepileptic drug, but hepatotoxicity remains a dose-limiting side effect (76). Impaired mitochondrial function in valproate toxicity (76, 77) may arise from CoASH sequestered as thioesters of valproate and its degradation products (25, 26, 76, 78, 79). The ability of PZ-3022 to rapidly (in 2 h) alleviate liver CoASH sequestration suggests that pantazines may be useful in the treatment of valproate toxicity. Many age-related and degenerative diseases are associated with mitochondrial dysfunction (80) and pantazines may benefit these diseases by increasing CoASH availability to support TCA cycle function. Depressed CoASH and C2-CoA coupled with elevated TCA cycle intermediates may disrupt intermediary metabolism at several steps in light of their multiple roles in metabolism (81), metabolic regulation (82), epigenetic control (83, 84) and protein interaction networks (85). Examining the impact of all these disruptions is beyond the scope of this study, but the normalization of metabolites by PZ-3022 therapy would dampen any negative effect of their accumulation on intermediary metabolism.
Human diseases are difficult to precisely model in mice, and the widely used PA mouse model does have drawbacks. The tissue-selective pattern of transgene expression means that not all tissues are equally deficient in PCC activity as would occur in human PA. A knock-in mouse model would be an improvement but would also not provide insight into the entire spectrum of PA disease because the range of mutations that give rise to PA impact PCC activity to different extents. Nonetheless, the PA mouse model is useful to probe the underlying metabolic imbalances that arise from excess C3-CoA formation. While mutations in PCC cause the accumulation of C3-CoA, the hypothesis that TCA cycle dysfunction due to CoASH sequestration exacerbates C3-CoA and methylmalonate accumulation and contributes to the metabolic imbalances in PA are important concepts that deserves additional experimental validation.
Materials and Methods
Study design
An established PA mouse model was used to interrogate the metabolic imbalances related to CoA sequestration. Pcca−/−PCCA(A138T)tg/0 (PA) mice were obtained from the Mayo Clinic, re-animated and generated at St. Jude Children’s Research Hospital. PA mice were treated with PZ-3022, a member of a class of allosteric PanK activators to determine how the elevation of tissue CoA impacts metabolism in the PA mouse model. PZ-3022 was synthesized and developed at St. Jude Children’s Research Hospital. We used numbers of male and female biological replicates to detect sexual dimorphism and large enough to produce statistically meaningful results. The number of biological replicates for each experiment is specified in the figures or legends. Experimental techniques were developed to measure CoASH sequestration and established methods were used to evaluate other biochemical disease markers in wild type and PA mice. Multiple experimenters performed biochemical analyses and were blinded during data acquisition. Male and female PA mice were randomized into treatment groups without exclusions. All procedures were performed according to protocols 323 and 556 as approved by the St. Jude Children's Research Hospital Institutional Animal Care and Use Committee.
Statistical analysis
Statistical tests were performed using GraphPad Prism software v9.1.2 (http://graphpad.com/scientific-software/prism/). An unpaired parametric t test was used when comparing two sample populations. When comparing 3 or more groups, the 1-way ANOVA was used with Turkey post-hoc analysis. The sample sizes, the means and the p values are reported in the figures or legends.
Supplementary Material
Acknowledgements:
We thank Dr. Michael Barry for providing the Pcca−/−PCCA(A138T)tg/0 founder mice, and Karen Miller, Laura Creed, Lois Richmond, Jina Wang, and Ruobing Zhou for their excellent technical assistance with the experiments. Small animal echocardiography was performed by Melissa Johnson and Thomas Confer in the SJCRH Center for In Vivo Imaging and Therapeutics.
Funding:
Research was supported by CoA Therapeutics (S.J.), NIGMS grant GM034496 (C.O.R.), Cancer Center Support Grant CA021765, and the American and Syrian Associated Charites. Animal imaging was performed by the Center for In Vivo Imaging and Therapeutics, which is supported in part by NIH grants P01 CA096832 and R50 CA211481. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
Competing interests: C.S., M.-K.Y., R.E.L., C.O.R., and S.J. are co-inventors of the patent “Small molecule modulators of pantothenate kinase” (#US2021/0061788) and S.J., C.O.R, R.E.L, A.V.E, C.S, R.T, and M.-K. Y. are co-inventors on the pending patent “Methods of treating disorders associated with CASTOR” (#WO2019/133632). S. J., C.O.R. and M.W.F. are co-inventors on pending patent “Methods and systems for assessing biomarkers” (filed 3/22/2021; 63/164,484). S.J. is a member of the scientific advisory board of CoA Therapeutics and a former member of the Scientific and Medical Advisory Board of the NBIA Disorders Association.
Data and materials availability. All data associated with this study are present in the paper or Supplementary Materials. The crystal structure of the PanK3•AMPPNP•Mg2+•PZ-3022 complex is deposited in the Protein Data Bank (PDB ID: 6PE6). Raw data from figures are reported in data file S1.
References and Notes
- 1.Baumgartner MR, Horster F, Dionisi-Vici C, Haliloglu G, Karall D, Chapman KA, Huemer M, Hochuli M, Assoun M, Ballhausen D, Burlina A, Fowler B, Grunert SC, Grunewald S, Honzik T, Merinero B, Perez-Cerda C, Scholl-Burgi S, Skovby F, Wijburg F, MacDonald A, Martinelli D, Sass JO, Valayannopoulos V, Chakrapani A, Proposed guidelines for the diagnosis and management of methylmalonic and propionic acidemia. Orphanet. J. Rare Dis 9, 130 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wongkittichote P, Ah Mew N, Chapman KA, Propionyl-CoA carboxylase - A review. Mol. Genet. Metab 122, 145–152 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Clavero S, Martinez MA, Perez B, Perez-Cerda C, Ugarte M, Desviat LR, Functional characterization of PCCA mutations causing propionic acidemia. Biochim. Biophys. Acta 1588, 119–125 (2002). [DOI] [PubMed] [Google Scholar]
- 4.Desviat LR, Perez B, Perez-Cerda C, Rodriguez-Pombo P, Clavero S, Ugarte M, Propionic acidemia: mutation update and functional and structural effects of the variant alleles. Mol. Genet. Metab 83, 28–37 (2004). [DOI] [PubMed] [Google Scholar]
- 5.Al-Hamed MH, Imtiaz F, Al-Hassnan Z, Al-Owain M, Al-Zaidan H, Alamoudi MS, Faqeih E, Alfadhel M, Al-Asmari A, Saleh MM, Almutairi F, Moghrabi N, AlSayed M, Spectrum of mutations underlying propionic acidemia and further insight into a genotype-phenotype correlation for the common mutation in Saudi Arabia. Mol. Genet. Metab. Rep 18, 22–29 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rivera-Barahona A, Navarrete R, Garcia-Rodriguez R, Richard E, Ugarte M, Perez-Cerda C, Perez B, Gamez A, Desviat LR, Identification of 34 novel mutations in propionic acidemia: Functional characterization of missense variants and phenotype associations. Mol. Genet. Metab 125, 266–275 (2018). [DOI] [PubMed] [Google Scholar]
- 7.Ando T, Rasmussen K, Wright JM, Nyhan WL, Isolation and identification of methylcitrate, a major metabolic product of propionate in patients with propionic acidemia. J. Biol. Chem 247, 2200–2204 (1972). [PubMed] [Google Scholar]
- 8.Bergstrom T, Greter J, Levin AH, Steen G, Tryding N, Wass U, Propionyl-CoA carboxylase deficiency: case report, effect of low-protein diet and identification of 3-oxo-2-methylvaleric acid 3-hydroxy-2-methylvaleric acid, and maleic acid in urine. Scand. J. Clin. Lab. Invest 41, 117–126 (1981). [DOI] [PubMed] [Google Scholar]
- 9.Perez B, Desviat LR, Rodriguez-Pombo P, Clavero S, Navarrete R, Perez-Cerda C, Ugarte M, Propionic acidemia: identification of twenty-four novel mutations in Europe and North America. Mol. Genet. Metab 78, 59–67 (2003). [DOI] [PubMed] [Google Scholar]
- 10.Turgeon CT, Magera MJ, Cuthbert CD, Loken PR, Gavrilov DK, Tortorelli S, Raymond KM, Oglesbee D, Rinaldo P, Matern D, Determination of total homocysteine, methylmalonic acid, and 2-methylcitric acid in dried blood spots by tandem mass spectrometry. Clin. Chem 56, 1686–1695 (2010). [DOI] [PubMed] [Google Scholar]
- 11.Cheema-Dhadli S, Leznoff CC, Halperin ML, Effect of 2-methylcitrate on citrate metabolism: implications for the management of patients with propionic acidemia and methylmalonic aciduria. Pediatr. Res 9, 905–908 (1975). [DOI] [PubMed] [Google Scholar]
- 12.Jurecki E, Ueda K, Frazier D, Rohr F, Thompson A, Hussa C, Obernolte L, Reineking B, Roberts AM, Yannicelli S, Osara Y, Stembridge A, Splett P, Singh RH, Nutrition management guideline for propionic acidemia: An evidence- and consensus-based approach. Mol. Genet. Metab 126, 341–354 (2019). [DOI] [PubMed] [Google Scholar]
- 13.Barshes NR, Vanatta JM, Patel AJ, Carter BA, O'Mahony CA, Karpen SJ, Goss JA, Evaluation and management of patients with propionic acidemia undergoing liver transplantation: a comprehensive review. Pediatr. Transplant 10, 773–781 (2006). [DOI] [PubMed] [Google Scholar]
- 14.Quintero J, Molera C, Juamperez J, Redecillas S, Meavilla S, Nunez R, Garcia C, Del Toro M, Garcia A, Ortega J, Segarra O, de Carpi JM, Bilbao I, Charco R, The Role of liver transplantation in propionic acidemia. Liver Transpl. 24, 1736–1745 (2018). [DOI] [PubMed] [Google Scholar]
- 15.Rela M, Battula N, Madanur M, Mieli-Vergani G, Dhawan A, Champion M, Raiman J, Heaton N, Auxiliary liver transplantation for propionic acidemia: a 10-year follow-up. Am. J. Transplant 7, 2200–2203 (2007). [DOI] [PubMed] [Google Scholar]
- 16.Kasahara M, Sakamoto S, Kanazawa H, Karaki C, Kakiuchi T, Shigeta T, Fukuda A, Kosaki R, Nakazawa A, Ishige M, Nagao M, Shigematsu Y, Yorifuji T, Naiki Y, Horikawa R, Living-donor liver transplantation for propionic acidemia. Pediatr. Transplant 16, 230–234 (2012). [DOI] [PubMed] [Google Scholar]
- 17.Vara R, Turner C, Mundy H, Heaton ND, Rela M, Mieli-Vergani G, Champion M, Hadzic N, Liver transplantation for propionic acidemia in children. Liver Transpl. 17, 661–667 (2011). [DOI] [PubMed] [Google Scholar]
- 18.Nagao M, Tanaka T, Morii M, Wakai S, Horikawa R, Kasahara M, Improved neurologic prognosis for a patient with propionic acidemia who received early living donor liver transplantation. Mol. Genet. Metab 108, 25–29 (2013). [DOI] [PubMed] [Google Scholar]
- 19.Charbit-Henrion F, Lacaille F, McKiernan P, Girard M, de Lonlay P, Valayannopoulos V, Ottolenghi C, Chakrapani A, Preece M, Sharif K, Chardot C, Hubert P, Dupic L, Early and late complications after liver transplantation for propionic acidemia in children: a two centers study. Am. J. Transplant 15, 786–791 (2015). [DOI] [PubMed] [Google Scholar]
- 20.Hofherr SE, Senac JS, Chen CY, Palmer DJ, Ng P, Barry MA, Short-term rescue of neonatal lethality in a mouse model of propionic acidemia by gene therapy. Hum. Gene Ther 20, 169–180 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Guenzel AJ, Collard R, Kraus JP, Matern D, Barry MA, Long-term sex-biased correction of circulating propionic acidemia disease markers by adeno-associated virus vectors. Hum. Gene Ther 26, 153–160 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Collard R, Majtan T, Park I, Kraus JP, Import of TAT-conjugated propionyl coenzyme A carboxylase using models of propionic acidemia. Mol. Cell. Biol 38, e00491–00417 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jiang L, Park JS, Yin L, Laureano R, Jacquinet E, Yang J, Liang S, Frassetto A, Zhuo J, Yan X, Zhu X, Fortucci S, Hoar K, Mihai C, Tunkey C, Presnyak V, Benenato KE, Lukacs CM, Martini PGV, Guey LT, Dual mRNA therapy restores metabolic function in long-term studies in mice with propionic acidemia. Nat. Commun 11, 5339 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brass EP, Overview of coenzyme A metabolism and its role in cellular toxicity. Chem. Biol. Interact 90, 203–214 (1994). [DOI] [PubMed] [Google Scholar]
- 25.Becker CM, Harris RA, Influence of valproic acid on hepatic carbohydrate and lipid metabolism. Arch Biochem. Biophys 223, 381–392 (1983). [DOI] [PubMed] [Google Scholar]
- 26.Ponchaut S, van Hoof F, Veitch K, In vitro effects of valproate and valproate metabolites on mitochondrial oxidations. Relevance of CoA sequestration to the observed inhibitions. Biochem. Pharmacol 43, 2435–2442 (1992). [DOI] [PubMed] [Google Scholar]
- 27.Melegh B, Kerner J, Bieber LL, Pivampicillin-promoted excretion of pivaloylcarnitine in humans. Biochem. Pharmacol 36, 3405–3409 (1987). [DOI] [PubMed] [Google Scholar]
- 28.Konishi M, Hashimoto H, Determination of pivaloylcarnitine in human plasma and urine by high-performance liquid chromatography with fluorescence detection. J. Pharm. Sci 81, 1038–1041 (1992). [DOI] [PubMed] [Google Scholar]
- 29.McCune SA, Durant PJ, Flanders LE, Harris RA, Inhibition of hepatic gluconeogenesis and lipogenesis by benzoic acid, p-tert.-butylbenzoic acid, and a structurally related hypolipidemic agent SC-33459. Arch Biochem. Biophys 214, 124–133 (1982). [DOI] [PubMed] [Google Scholar]
- 30.Mitchell GA, Gauthier N, Lesimple A, Wang SP, Mamer O, Qureshi I, Hereditary and acquired diseases of acyl-coenzyme A metabolism. Mol. Genet. Metab 94, 4–15 (2008). [DOI] [PubMed] [Google Scholar]
- 31.Patel TB, DeBuysere MS, Olson MS, The effect of propionate on the regulation of the pyruvate dehydrogenase complex in the rat liver. Arch Biochem. Biophys 220, 405–414 (1983). [DOI] [PubMed] [Google Scholar]
- 32.Brass EP, Fennessey PV, Miller LV, Inhibition of oxidative metabolism by propionic acid and its reversal by carnitine in isolated rat hepatocytes. Biochem. J 236, 131–136 (1986). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang Y, Christopher BA, Wilson KA, Muoio D, McGarrah RW, Brunengraber H, Zhang GF, Propionate-induced changes in cardiac metabolism, notably CoA trapping, are not altered by L-carnitine. Am. J. Physiol. Endocrinol. Metab 315, E622–E633 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Walajtys-Rode E, Coll KE, Williamson JR, Effects of branched chain alpha-ketoacids on the metabolism of isolated rat liver cells. II. Interactions with gluconeogenesis and urea synthesis. J. Biol. Chem 254, 11521–11529 (1979). [PubMed] [Google Scholar]
- 35.Martin-Requero A, Corkey BE, Cerdan S, Walajtys-Rode E, Parrilla RL, Williamson JR, Interactions between α-ketoisovalerate metabolism and the pathways of gluconeogenesis and urea synthesis in isolated hepatocytes. J. Biol. Chem 258, 3673–3681 (1983). [PubMed] [Google Scholar]
- 36.Brass EP, Effect of α-ketobutyrate on palmitic acid and pyruvate metabolism in isolated rat hepatocytes. Biochim. Biophys. Acta 888, 18–24 (1986). [DOI] [PubMed] [Google Scholar]
- 37.Brass EP, Interaction of carnitine and propionate with pyruvate oxidation by hepatocytes from clofibrate-treated rats: importance of coenzyme A availability. J. Nutr 122, 234–240 (1992). [DOI] [PubMed] [Google Scholar]
- 38.Leonardi R, Zhang Y-M, Rock CO, Jackowski S, Coenzyme A: Back in action. Prog. Lipid Res 44, 125–153 (2005). [DOI] [PubMed] [Google Scholar]
- 39.Subramanian C, Yun MK, Yao J, Sharma LK, Lee RE, White SW, Jackowski S, Rock CO, Allosteric regulation of mammalian pantothenate kinase. J. Biol. Chem 291, 22302–22314 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang Y-M, Rock CO, Jackowski S, Feedback regulation of murine pantothenate kinase 3 by coenzyme A and coenzyme A thioesters. J. Biol. Chem 280, 32594–32601 (2005). [DOI] [PubMed] [Google Scholar]
- 41.Rock CO, Calder RB, Karim MA, Jackowski S, Pantothenate kinase regulation of the intracellular concentration of coenzyme A. J. Biol. Chem 275, 1377–1383 (2000). [DOI] [PubMed] [Google Scholar]
- 42.Hong BS, Senisterra G, Rabeh WM, Vedadi M, Leonardi R, Zhang YM, Rock CO, Jackowski S, Park HW, Crystal structures of human pantothenate kinases. Insights into allosteric regulation and mutations linked to a neurodegeneration disorder. J. Biol. Chem 282, 27984–27993 (2007). [DOI] [PubMed] [Google Scholar]
- 43.Zhang YM, Chohnan S, Virga KG, Stevens RD, Ilkayeva OR, Wenner BR, Bain JR, Newgard CB, Lee RE, Rock CO, Jackowski S, Chemical knockout of pantothenate kinase reveals the metabolic and genetic program responsible for hepatic coenzyme A homeostasis. Chem. Biol 14, 291–302 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Leonardi R, Rehg JE, Rock CO, Jackowski S, Pantothenate kinase 1 is required to support the metabolic transition from the fed to the fasted state. PLoS ONE 5, e11107 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Garcia M, Leonardi R, Zhang YM, Rehg JE, Jackowski S, Germline deletion of pantothenate kinases 1 and 2 reveals the key roles for CoA in postnatal metabolism. PLoS One 7, e40871 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Subramanian C, Yao J, Frank MW, Rock CO, Jackowski S, A pantothenate kinase-deficient mouse model reveals a gene expression program associated with brain coenzyme a reduction. Biochim. Biophys. Acta Mol. Basis Dis 1866, 165663 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sharma LK, Subramanian C, Yun MK, Frank MW, White SW, Rock CO, Lee RE, Jackowski S, A therapeutic approach to pantothenate kinase associated neurodegeneration. Nat. Commun 9, 4399 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Miyazaki T, Ohura T, Kobayashi M, Shigematsu Y, Yamaguchi S, Suzuki Y, Hata I, Aoki Y, Yang X, Minjares C, Haruta I, Uto H, Ito Y, Muller U, Fatal propionic acidemia in mice lacking propionyl-CoA carboxylase and its rescue by postnatal, liver-specific supplementation via a transgene. J. Biol. Chem 276, 35995–35999 (2001). [DOI] [PubMed] [Google Scholar]
- 49.Guenzel AJ, Hofherr SE, Hillestad M, Barry M, Weaver E, Venezia S, Kraus JP, Matern D, Barry MA, Generation of a hypomorphic model of propionic acidemia amenable to gene therapy testing. Mol. Ther 21, 1316–1323 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gallego-Villar L, Rivera-Barahona A, Cuevas-Martin C, Guenzel A, Perez B, Barry MA, Murphy MP, Logan A, Gonzalez-Quintana A, Martin MA, Medina S, Gil-Izquierdo A, Cuezva JM, Richard E, Desviat LR, In vivo evidence of mitochondrial dysfunction and altered redox homeostasis in a genetic mouse model of propionic acidemia: Implications for the pathophysiology of this disorder. Free Radic. Biol. Med 96, 1–12 (2016). [DOI] [PubMed] [Google Scholar]
- 51.Rivera-Barahona A, Alonso-Barroso E, Perez B, Murphy MP, Richard E, Desviat LR, Treatment with antioxidants ameliorates oxidative damage in a mouse model of propionic acidemia. Mol. Genet. Metab 122, 43–50 (2017). [DOI] [PubMed] [Google Scholar]
- 52.Rivera-Barahona A, Fulgencio-Covian A, Perez-Cerda C, Ramos R, Barry MA, Ugarte M, Perez B, Richard E, Desviat LR, Dysregulated miRNAs and their pathogenic implications for the neurometabolic disease propionic acidemia. Sci. Rep 7, 5727 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tamayo M, Fulgencio-Covian A, Navarro-Garcia JA, Val-Blasco A, Ruiz-Hurtado G, Gil-Fernandez M, Martin-Nunes L, Lopez JA, Desviat LR, Delgado C, Richard E, Fernandez-Velasco M, Intracellular calcium mishandling leads to cardiac dysfunction and ventricular arrhythmias in a mouse model of propionic acidemia. Biochim. Biophys. Acta Mol. Basis Dis 1866, 165586 (2020). [DOI] [PubMed] [Google Scholar]
- 54.Ruoppolo M, Caterino M, Albano L, Pecce R, Di Girolamo MG, Crisci D, Costanzo M, Milella L, Franconi F, Campesi I, Targeted metabolomic profiling in rat tissues reveals sex differences. Sci. Rep 8, 4663 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Devanathan S, Whitehead TD, Fettig N, Gropler RJ, Nemanich S, Shoghi KI, Sexual dimorphism in myocardial acylcarnitine and triglyceride metabolism. Biol. Sex. Differ 7, 25 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Park KC, Krywawych S, Richard E, Desviat LR, Swietach P, Cardiac complications of propionic and other inherited organic acidemias. Front. Cardiovasc. Med 7, 617451 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Corbin DR, Rehg JE, Shepherd DL, Stoilov P, Percifield RJ, Horner L, Frase S, Zhang YM, Rock CO, Hollander JM, Jackowski S, Leonardi R, Excess coenzyme A reduces skeletal muscle performance and strength in mice overexpressing human PANK2. Mol. Genet. Metab 120, 350–362 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Okabe M, Ikawa M, Kominami K, Nakanishi T, Nishimune Y, 'Green mice' as a source of ubiquitous green cells. FEBS Lett. 407, 313–319 (1997). [DOI] [PubMed] [Google Scholar]
- 59.Takeuchi K, Sereemaspun A, Inagaki T, Hakamata Y, Kaneko T, Murakami T, Takahashi M, Kobayashi E, Ookawara S, Morphologic characterization of green fluorescent protein in embryonic, neonatal, and adult transgenic rats. Anat. Rec. A Discov. Mol. Cell Evol. Biol 274, 883–886 (2003). [DOI] [PubMed] [Google Scholar]
- 60.Ke BX, Llanos RM, Wright M, Deal Y, Mercer JF, Alteration of copper physiology in mice overexpressing the human Menkes protein ATP7A. Am. J. Physiol. Regul. Integr. Comp. Physiol 290, R1460–1467 (2006). [DOI] [PubMed] [Google Scholar]
- 61.Shaham O, Slate NG, Goldberger O, Xu Q, Ramanathan A, Souza AL, Clish CB, Sims KB, Mootha VK, A plasma signature of human mitochondrial disease revealed through metabolic profiling of spent media from cultured muscle cells. Proc. Natl. Acad. Sci. U. S. A 107, 1571–1575 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Barshop BA, Metabolomic approaches to mitochondrial disease: correlation of urine organic acids. Mitochondrion 4, 521–527 (2004). [DOI] [PubMed] [Google Scholar]
- 63.Thompson Legault J, Strittmatter L, Tardif J, Sharma R, Tremblay-Vaillancourt V, Aubut C, Boucher G, Clish CB, Cyr D, Daneault C, Waters PJ, Consortium L, Vachon L, Morin C, Laprise C, Rioux JD, Mootha VK, Des Rosiers C, A metabolic signature of mitochondrial dysfunction revealed through a monogenic form of Leigh syndrome. Cell Rep. 13, 981–989 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wilson KA, Han Y, Zhang M, Hess JP, Chapman KA, Cline GW, Tochtrop GP, Brunengraber H, Zhang GF, Inter-relations between 3-hydroxypropionate and propionate metabolism in rat liver: relevance to disorders of propionyl-CoA metabolism. Am. J. Physiol. Endocrinol. Metab 313, E413–E428 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Scholl-Burgi S, Sass JO, Heinz-Erian P, Amann E, Haberlandt E, Albrecht U, Ertl C, Sigl SB, Lagler F, Rostasy K, Karall D, Changes in plasma amino acid concentrations with increasing age in patients with propionic acidemia. Amino Acids 38, 1473–1481 (2010). [DOI] [PubMed] [Google Scholar]
- 66.Scholl-Burgi S, Sass JO, Zschocke J, Karall D, Amino acid metabolism in patients with propionic acidaemia. J. Inherit. Metab. Dis 35, 65–70 (2012). [DOI] [PubMed] [Google Scholar]
- 67.Riemersma M, Hazebroek MR, Helderman-van den Enden A, Salomons GS, Ferdinandusse S, Brouwers M, van der Ploeg L, Heymans S, Glatz JFC, van den Wijngaard A, Krapels IPC, Bierau J, Brunner HG, Propionic acidemia as a cause of adult-onset dilated cardiomyopathy. Eur. J. Hum. Genet 25, 1195–1201 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Song P, Mabrouk OS, Hershey ND, Kennedy RT, In vivo neurochemical monitoring using benzoyl chloride derivatization and liquid chromatography-mass spectrometry. Anal. Chem 84, 412–419 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wong JM, Malec PA, Mabrouk OS, Ro J, Dus M, Kennedy RT, Benzoyl chloride derivatization with liquid chromatography-mass spectrometry for targeted metabolomics of neurochemicals in biological samples. J. Chromatogr. A 1446, 78–90 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Vishwakarma P, Lotspeich WD, The excretion of I-malic acid in relation to the tricarboxylic acid cycle in the kidney. J. Clin. Invest 38, 414–423 (1959). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zano SP, Pate C, Frank M, Rock CO, Jackowski S, Correction of a genetic deficiency in pantothenate kinase 1 using phosphopantothenate replacement therapy. Mol. Genet. Metab 116, 281–288 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Frank MW, Subramanian C, Rock CO, Jackowski S, Quantification of Coenzyme A in Cells and Tissues. J. Vis. Exp 151, 10.3791/60182 (2019). [DOI] [PubMed] [Google Scholar]
- 73.Leonardi R, Rock CO, Jackowski S, Pank1 deletion in leptin-deficient mice reduces hyperglycaemia and hyperinsulinaemia and modifies global metabolism without affecting insulin resistance. Diabetologia 57, 1466–1475 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Yang H, Zhao C, Tang MC, Wang Y, Wang SP, Allard P, Furtos A, Mitchell GA, Inborn errors of mitochondrial acyl-Coenzyme A metabolism: acyl-CoA biology meets the clinic. Mol. Genet. Metab, (2019). [DOI] [PubMed] [Google Scholar]
- 75.Yang H, Zhao C, Wang Y, Wang SP, Mitchell GA, Hereditary diseases of coenzyme A thioester metabolism. Biochem. Soc. Trans 47, 149–155 (2019). [DOI] [PubMed] [Google Scholar]
- 76.Silva MF, Aires CC, Luis PB, Ruiter JP, L IJ, Duran M, Wanders RJ, de Almeida I. Tavares, Valproic acid metabolism and its effects on mitochondrial fatty acid oxidation: a review. J. Inherit. Metab. Dis 31, 205–216 (2008). [DOI] [PubMed] [Google Scholar]
- 77.Pessayre D, Mansouri A, Haouzi D, Fromenty B, Hepatotoxicity due to mitochondrial dysfunction. Cell. Biol. Toxicol 15, 367–373 (1999). [DOI] [PubMed] [Google Scholar]
- 78.Kesterson JW, Granneman GR, Machinist JM, The hepatotoxicity of valproic acid and its metabolites in rats. I. Toxicologic, biochemical and histopathologic studies. Hepatology 4, 1143–1152 (1984). [DOI] [PubMed] [Google Scholar]
- 79.Silva MF, Ruiter JP, L IJ, Allers P, ten Brink HJ, Jakobs C, Duran M, de Almeida I. Tavares, Wanders RJ, Synthesis and intramitochondrial levels of valproyl-coenzyme A metabolites. Anal. Biochem 290, 60–67 (2001). [DOI] [PubMed] [Google Scholar]
- 80.Area-Gomez E, Guardia-Laguarta C, Schon EA, Przedborski S, Mitochondria, OxPhos, and neurodegeneration: cells are not just running out of gas. J. Clin. Invest 129, 34–45 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Shi L, Tu BP, Acetyl-CoA and the regulation of metabolism: mechanisms and consequences. Curr. Opin. Cell Biol 33, 125–131 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Martinez-Reyes I, Chandel NS, Mitochondrial TCA cycle metabolites control physiology and disease. Nat. Commun 11, 102 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kaelin WG Jr., McKnight SL, Influence of metabolism on epigenetics and disease. Cell 153, 56–69 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Chisolm DA, Weinmann AS, Connections between metabolism and epigenetics in programming cellular differentiation. Annu. Rev. Immunol 36, 221–246 (2018). [DOI] [PubMed] [Google Scholar]
- 85.Levy MJ, Montgomery DC, Sardiu ME, Montano JL, Bergholtz SE, Nance KD, Thorpe AL, Fox SD, Lin Q, Andresson T, Florens L, Washburn MP, Meier JL, A systems chemoproteomic analysis of acyl-CoA/protein interaction networks. Cell Chem. Biol 27, 322–333 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Leonardi R, Zhang Y-M, Yun M-K, Zhou R, Zeng F-Y, Lin W, Cui J, Chen T, Rock CO, White SW, Jackowski S, Modulation of pantothenate kinase 3 activity by small molecules that interact with the substrate/allosteric regulatory domain. Chem. Biol. Drug Des 17, 892–902 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Otwinowski Z, Minor W, Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol 276, 307–326 (1997). [DOI] [PubMed] [Google Scholar]
- 88.McCoy AJ, Solving structures of protein complexes by molecular replacement with Phaser. Acta Crystallogr. D Biol. Crystallogr 63, 32–41 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Afonine PV, Grosse-Kunstleve RW, Echols N, Headd JJ, Moriarty NW, Mustyakimov M, Terwilliger TC, Urzhumtsev A, Zwart PH, Adams PD, Towards automated crystallographic structure refinement with phenix.refine. Acta Crystallogr. D Biol. Crystallogr 68, 352–367 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Emsley P, Lohkamp B, Scott WG, Cowtan K, Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr 66, 486–501 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Chen VB, Arendall WB 3rd, Headd JJ, Keedy DA, Immormino RM, Kapral GJ, Murray LW, Richardson JS, Richardson DC, MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr 66, 12–21 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Bradford MM, A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 72, 248–254 (1976). [DOI] [PubMed] [Google Scholar]
- 93.Ivanova PT, Milne SB, Byrne MO, Xiang Y, Brown HA, Glycerophospholipid identification and quantitation by electrospray ionization mass spectrometry. Methods Enzymol 432, 21–57 (2007). [DOI] [PubMed] [Google Scholar]
- 94.Bajad SU, Lu W, Kimball EH, Yuan J, Peterson C, Rabinowitz JD, Separation and quantitation of water soluble cellular metabolites by hydrophilic interaction chromatography-tandem mass spectrometry. J. Chromatogr. A 1125, 76–88 (2006). [DOI] [PubMed] [Google Scholar]
- 95.Wang X, Bathina M, Lynch J, Koss B, Calabrese C, Frase S, Schuetz JD, Rehg JE, Opferman JT, Deletion of MCL-1 causes lethal cardiac failure and mitochondrial dysfunction. Genes Dev. 27, 1351–1364 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Morrison JF, Kinetics of the reversible inhibition of enzyme-catalysed reactions by tight-binding inhibitors. Biochim. Biophys. Acta 185, 269–286 (1969). [DOI] [PubMed] [Google Scholar]
- 97.Leonardi R, Rock CO, Jackowski S, Zhang Y-M, Activation of human mitochondrial pantothenate kinase 2 by palmitoylcarnitine. Proc. Natl. Acad. Sci. U. S. A 104, 1494–1499 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.