Abstract
There is an urgent need for development of new serodiagnostic strategies for leptospirosis, an emerging zoonosis with worldwide distribution. We have evaluated the diagnostic utility of five recombinant antigens in enzyme-linked immunosorbent assays (ELISAs) for serodiagnosis of leptospirosis. Sera from 50 healthy residents of a high-incidence region were used to determine cutoff values for 96% specificity. In paired sera from 50 cases of leptospirosis confirmed by the microscopic agglutination test, immunoglobulin G (IgG) but not IgM reacted with the recombinant leptospiral proteins. The recombinant LipL32 IgG ELISA had the highest sensitivities in the acute (56%) and convalescent (94%) phases of leptospirosis. ELISAs based on recombinant OmpL1, LipL41, and Hsp58 had sensitivities of 16, 24, and 18% during the acute phase and 72, 44, and 32% during convalescence, respectively. Compared to sera from healthy individuals, patient sera did not react significantly with recombinant LipL36 (P > 0.05). Recombinant LipL32 IgG ELISA demonstrated 95% specificity among 100 healthy individuals, and specificities ranging from 90 to 97% among 30 dengue patients, 30 hepatitis patients, and 16 patients with diseases initially thought to be leptospirosis. Among 39 Venereal Disease Research Laboratory test-positive individuals and 30 Lyme disease patients, 13 and 23% of sera, respectively, reacted positively with the rLipL32 antigen. These findings indicate that rLipL32 may be an useful antigen for the serodiagnosis of leptospirosis.
Leptospirosis is a zoonosis caused by pathogenic spirochetes of the genus Leptospira. Although traditionally considered an occupational risk among persons exposed to contaminated water or infected animal urine (15, 16), leptospirosis is becoming recognized as a common cause of febrile illness in tropical environments worldwide (5, 14, 38). Awareness of its public health importance has increased following recent reports of outbreaks associated with recreational exposures (7–9) and emerging epidemics of leptospirosis-associated severe pulmonary hemorrhage syndrome (28, 33, 35, 40, 49). Leptospirosis has now expanded to affect urban populations throughout Latin America and the Caribbean. Epidemics associated with high case fatality (greater than 15%) break out annually during seasonal periods of heavy rainfall in poor urban areas that lack basic sanitation infrastructure (23, 26). During these outbreaks, confusion between the broad spectrum of clinical presentations associated with leptospirosis and classic dengue fever (24, 37) complicates the early diagnosis required for the timely administration of antibiotic therapy. The need for rapid and appropriate diagnostic tests has become ever more urgent to aid clinical case identification and to facilitate the implementation of rapid outbreak investigations.
The standard serologic test, the microscopic agglutination test (MAT), is inadequate for rapid case identification since it can only be performed in a few reference laboratories and requires analyses of paired sera to achieve sufficient sensitivity (12, 15). Dependence upon the MAT results in delays in establishing the cause of outbreaks, as seen in several investigations (7, 47). Enzyme-linked immunosorbent assays (ELISAs) (12, 46, 48), and other rapid serologic tests based on whole-cell leptospiral antigen preparations (43, 44, 50) have been developed for use as an alternative method to screen for leptospiral infection, although the MAT is still required for case confirmation (12, 15). Recombinant-antigen-based serologic tests are widely used in screening for spirochetal infections such as Lyme disease and syphilis (17, 22, 27, 39), but the use of recombinant proteins for serodiagnosis of leptospirosis has not been widely investigated. Recently, a recombinant flagellar antigen immunocapture assay was described for serodiagnosis of bovine leptospirosis (6). A recombinant heat shock protein, Hsp58, showed a high degree of ELISA reactivity with serum samples from a small number of human cases (32). However, the utility of recombinant antigens for the serodiagnosis of human leptospirosis has not been investigated in large validation studies.
In a previous study, we identified leptospiral antigens that were serodiagnostic markers of infection in immunoblot analyses (18). A 32-kDa protein was identified to be an immunodominant antigen with the best serodiagnostic utility: 38 and 85% of cases of leptospirosis had detectable antibodies during the acute and convalescent phases, respectively, of their illness. Anti-32-kDa protein reactivity was detected in less than 5% of sera from control individuals. This antigen was identified as LipL32, a major leptospiral outer membrane protein whose expression is restricted to pathogenic Leptospira species (20). Hsp58, a member of the GroEL family of heat shock proteins, was also identified in immunoblots with human sera and was the antigen recognized most frequently by patient antibodies in the acute phase of the illness. Two membrane proteins, the porin OmpL1 (19) and lipoprotein LipL41 (41), were identified as immunoreactive proteins, but the frequency of seroreactivity could not be determined due to the limited resolution of the native proteins on sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels. The characterization of these leptospiral proteins makes it possible to express and purify recombinant fusion proteins in a form suitable for diagnostic formats such as recombinant-antigen ELISA. The present study was conducted to evaluate the utility of these recombinant proteins from Leptospira as antigens in ELISAs.
MATERIALS AND METHODS
Patient and control individuals.
Fifty paired sera were randomly selected from a bank of samples from more than 300 laboratory-confirmed cases identified during active hospital-based surveillance from March 1996 to October 1997 for urban epidemics of leptospirosis in the city of Salvador, Brazil. The MAT and culture isolation were performed for laboratory confirmation of leptospirosis as previously described (23). The first, “acute-phase” serum sample from these patients was collected during hospital admission, a median of 7 days (range. 2 to 23 days) after reported onset of symptoms. A second, “convalescent-phase” serum sample was collected following discharge, a median of 29.5 days (range, 17 to 113 days) after the reported onset of symptoms. The case definition for MAT confirmation was a fourfold rise in MAT titer between paired sera or a single serum MAT titer of >1:800. The highest agglutinating titers for 49 (98%) of the 50 patients were to reference strains of the serogroup Icterohaemorrhagiae. In addition to MAT, culture isolation and whole-Leptospira immunoglobulin M (IgM) ELISA were performed with patient samples. Among 14 patients who had a blood culture performed upon hospital admission, Leptospira interrogans serovar copenhageni was isolated from 7 (50%). The IgM whole-Leptospira ELISA was performed as described by Adler et al. (1) with sonicated antigen from a clinical isolate of L. interrogans serovar copenhageni (23). A positive reaction in IgM ELISA was defined as an absorbance value greater than that of the cutoff reference sample, which corresponded to the 98th percentile absorbance value among sera of healthy control individuals from an endemic region.
Control serum samples were obtained from 100 healthy individuals and 136 patients with diseases other than leptospirosis. Serum samples from healthy individuals (n = 50) in Salvador, Brazil, a region of high leptospirosis incidence, were randomly selected from a serum bank collected during a city-wide serosurvey for infectious diseases performed in 1998. In addition, serum samples from healthy individuals (n = 50) from Northern California (gift of Michael Hendry, California State Department of Health, Berkeley, Calif.) were obtained as control sera from a region of low leptospirosis incidence. Patient sera were analyzed from the following control groups: cases of Lyme disease (n = 30) from the United States(gift of Martin Schriefer, Centers for Disease Control and Prevention, Fort Collins, Colo.) and four patient groups from Salvador, Brazil: Venereal Disease Research Laboratory (VDRL) test-positive individuals (n = 30; samples obtained from the Central Laboratory for the State of Bahia, Brazil [LACEN/BA]), laboratory-confirmed (dengue IgM ELISA) cases of dengue (n = 30; obtained from LACEN/BA), patients with acute hepatitis B virus infection (hepatitis B S-antigen positive; n = 30; obtained from LACEN/BA), and patients who were hospitalized with an initial clinical suspicion of leptospirosis and subsequently diagnosed as having another illness based on laboratory or radiological evidence (n = 16). Serum samples were aliquoted after collection and stored at −20°C.
Recombinant Leptospira antigens.
The pRSET plasmid (Invitrogen) constructs containing portions of genes encoding the leptospiral outer membrane proteins LipL32, OmpL1, and LipL41 were prepared as described previously (19, 20, 41). The previously described recombinant LipL36 (21) was included in the analysis as a control antigen because the expression of lipoprotein LipL36 is downregulated during infection, and therefore reactivity of patient sera is expected to be minimal. The PCR-amplified genes were ligated into the pRSET plasmid for expression as recombinant His6 fusion proteins. The 1,641-bp DNA fragment of the hsp58 coding region was obtained from PCR amplification of genomic DNA of an L. interrogans serovar copenhageni clinical isolate (23) with primers corresponding to the N-terminal and C-terminal sequences of a published sequence (GenBank accession number L14682) (2). The hsp58 fragment was inserted into the pQE30 expression vector (Qiagen) and electroporated into Escherichia coli M15 pREP4 cells (Qiagen).
Isopropyl-β-d-thiogalactopyranoside (IPTG; 2 mM final concentration; Life Technologies) was added to log-phase cultures of E. coli BLR(DE3)pLysS (Novagen) transformed with pRSET plasmids encoding leptospiral genes for expression of His6 fusion proteins rLipL32, rOmpL1, rLipL41, rLipL36, and rHsp58. For outer membrane proteins, 6 M guanidine was used to solubilize culture pellets. His6 fusion proteins were purified by affinity chromatography with Ni2+-nitrilotriacetic acid-agarose (Qiagen). The purity of eluted His6 fusion proteins was assessed by gel electrophoresis and staining with Coomassie brilliant blue. Proteins were dialyzed overnight against phosphate-buffered saline (PBS)–10% (vol/vol) glycerol–0.025% (wt/vol) sodium azide–0.1 to 0.3% (vol/vol) Triton X-100. After dialysis, the protein concentration was determined with bicinchoninic acid (42). Recombinant outer membrane proteins were then diluted 10- to 15-fold in buffer without Triton X-100. The culture pellet containing rHsp58 was suspended in 50 mM sodium phosphate buffer, cells were lysed with sonification, and rHsp58 was purified under native conditions with Ni2+-nitrilotriacetic acid-agarose (Qiagen). After dialysis against 0.1 M PBS, the concentrations of fusion proteins were determined using the DC Protein Assay (Bio-Rad).
Recombinant-antigen ELISAs.
Flat-bottomed polystyrene microtiter plates (Corning) were coated at 4°C overnight with His6 fusion proteins, 0.5 to 100 ng/well, suspended in 0.05 M sodium carbonate (pH 9.6). The plates were washed twice with distilled water and three times with PBS–0.05% (vol/vol) Tween 20 (PBST). Plates were incubated with blocking solution (PBST with 1% [wt/vol] bovine serum albumin) for 2 h at room temperature and, after four washes with PBST, stored at −20°C until use. Wells were incubated with 50 μl of sera, diluted 25- to 200-fold in blocking solution, for 1 h at room temperature with agitation. After four washes with PBST, wells were incubated with 50 μl of 5,000- to 20,000-fold dilutions of antihuman μ- or γ-chain goat antibodies conjugated to horseradish peroxidase (Sigma) for 1 h at room temperature with agitation. Afterwards, plates were washed twice with PBST and three times with PBS and incubated with 50 μl/well of 0.01% (wt/vol) 3,3′,5,5′-tetramethylbenzidine in substrate buffer (0.03% [vol/vol] hydrogen peroxide, 25 mM citric acid, 50 mM Na2HPO4 [pH 5.0]) for 20 min in the dark at room temperature. The color reaction was stopped by adding 25 μl of 2 N H2SO4, and the absorbance at 450 nm was measured in an Emax microplate reader (Molecular Devices, Sunnyvale, Calif.).
Initial assays were performed to determine the antigen concentration that best discriminated between ELISA reactions of serum samples from confirmed leptospirosis cases with reciprocal MAT titers greater than 800 (n = 8) and healthy individuals from a nonendemic area for leptospirosis in the United States (n = 4). Checkerboard titrations were performed with 25-, 50-, 100-, or 200-fold serum dilutions and antigen concentrations per well of 5, 25, 50, and 100 ng. In subsequent assays to determine sensitivity and specificity, plates were coated with the predetermined antigen concentrations. Incubations were performed with 50- and 20,000-fold dilutions of primary sera and secondary antibody conjugate, respectively. Individual serum samples were tested in duplicate, and the means of the two measurements were calculated for analysis. Paired measurements that differed by greater than 10% were retested. One positive control serum sample which reacted with all recombinant antigens and one negative control serum sample were included, in duplicate, on each plate as a quality control measure.
Statistical analyses.
Data were analyzed in Graph Pad Prism software (version 3.0; San Diego, Calif.). A cutoff value for each recombinant-antigen ELISA was defined as the 96th percentile of absorbance values among serum samples (n = 50) from Brazilian control individuals who reside in regions endemic for leptospirosis. Sensitivity was defined as the percentage of laboratory-confirmed cases of leptospirosis whose serum samples had mean absorbance of duplicate samples greater than the cutoff value. Specificity was calculated as the percentage of control individuals whose samples had mean absorbance below the cutoff value. The Mann-Whitney test was used to compare the median absorbance values for serum samples from leptospirosis cases with those from healthy individuals. Among confirmed cases of leptospirosis, the Wilcoxon ranked sign test for matched pairs was used to determine whether median optical density (OD) values of convalescent-phase sera were significantly different from those of acute-phase sera. The χ2 test with Yates' correction was used to compare proportions of positive ELISA reactions.
RESULTS
Standardization of recombinant-antigen ELISAs.
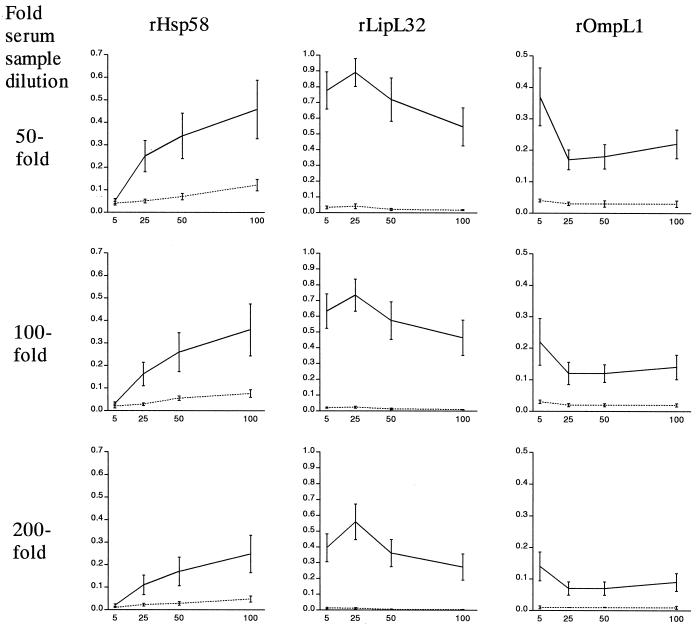
Five leptospiral antigens were expressed as His6 fusion proteins and purified by affinity chromatography. Prior to use in ELISAs, recombinant antigens were tested in immunoblots: pooled sera from leptospirosis cases had strong reactions to rLipL32 and rHsp58 and moderate reactions to OmpL1 and LipL41 and did not react with rLipL36 (data not shown). In initial ELISAs, the mean absorbance values among selected case sera were significantly higher than those of a sample of healthy U.S. individuals (P < 0.05), demonstrating IgG antibody reactivity to all five recombinant leptospiral proteins. IgM antibody reactivity was not detected in case or control samples to the three antigens, rLipL32, rHsp58, and rOmpL1, for which this response was evaluated. The performance of three representative recombinant IgG ELISAs at different antigen concentrations and serum dilutions is shown (Fig. 1). Sample absorbance in the rHsp58 IgG ELISA was positively correlated with increasing antigen concentration from 5 to 100 ng/well in a dose-dependent manner (Fig. 1). In contrast, maximum absorbance values were observed at a concentration of 25 ng/well in the rLipL32 ELISA, while absorbance decreased with increasing antigen concentrations above 5 ng/well in the rOmpL1 ELISA. A similar phenomenon was observed for recombinant membrane-associated proteins rLipL41 and rLipL36 and appeared to be associated with the concentration of Triton X-100 used to solubilize these recombinant proteins. Antigen preparations with reduced detergent concentrations had increased mean absorbances for case and control samples at all antigen concentration points (data not shown). At the lowest concentration of Triton X-100 used to solubilize recombinant proteins other than rHsp58, maximum absorbance values could be observed at the antigen concentrations of 25 ng/well or greater. In subsequent assays to determine sensitivity and specificity, a concentration of 5 ng/well was used for rLipL32, rLipL41, and rLipL36 ELISAs, while 25 ng/well was used for rHsp58 and rOmpL1 ELISAs. The ratio of mean absorbance values between case and control individual samples was not significantly different at 50-, 100-, and 200-fold dilutions of primary antisera, and therefore a 50-fold dilution was chosen for subsequent assays.
FIG. 1.
Performance of IgG ELISA at different serum sample dilutions and recombinant protein concentrations. Results are shown for assays with three selected recombinant leptospiral proteins, rHsp58 (left), rLipL32 (center), and rOmpL1 (right). For each point, a mean absorbance value (OD450 on the vertical axis) and standard deviation were plotted for serum samples from eight patients with confirmed leptospirosis (solid line) or four healthy control individuals (dashed line). Reactions for wells coated with 5, 25, 50, or 100 ng of recombinant antigen are represented on the horizontal axis. Plates were incubated with sera that were diluted 50- (first row), 100- (second row), or 200- (third row) fold.
Determination of cutoff values.
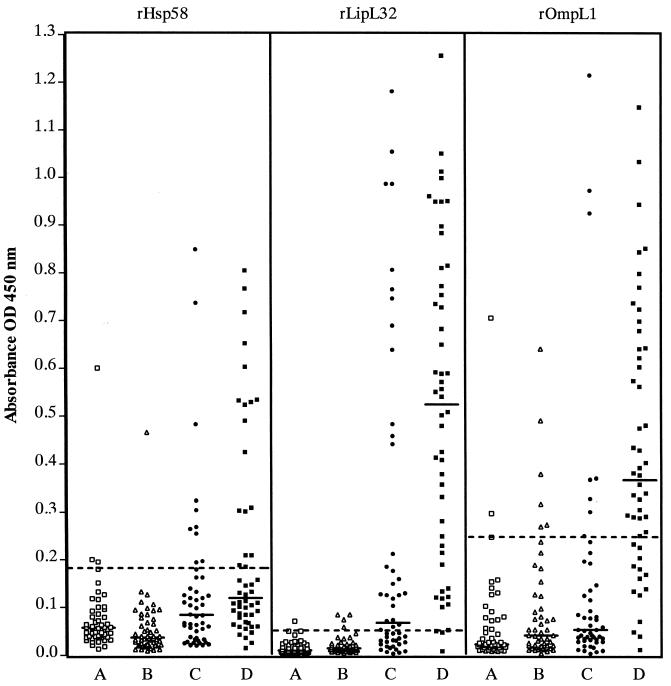
Serum samples (n = 50) from Brazilian control individuals who reside in a region endemic for leptospirosis were tested in parallel with samples from confirmed cases. The 96th percentile of absorbance values among samples from endemic region control individuals was defined as the cutoff value to achieve a diagnostic specificity of 96% (Fig. 2). Absorbance values among these control samples varied depending on the recombinant-antigen ELISA. The range of OD450 values for the rOmpL1 ELISA (0.008 to 0.705) was 10 times greater than the range of values observed for the rLipL32 ELISA (0.002 to 0.071). The defined cutoff value was five times higher for the rOmpL1 ELISA (OD450 = 0.247) than for the rLipL32 ELISA (OD450 = 0.050) (Table 1). Cutoff values for recombinant-antigen ELISAs are shown in Table 1. For four of the recombinant-protein ELISAs, median absorbance values of control samples from healthy U.S. individuals from a nonendemic region did not differ significantly from those of healthy Brazilian individuals from a high-incidence region. The one exception was the rHsp58 ELISA, in which the median value for endemic-area sera was 1.6 times higher than the median for nonendemic control sera (P < 0.01).
FIG. 2.
IgG antibody responses in leptospirosis patients and control groups to recombinant leptospiral proteins as determined by ELISA. Results are shown for three selected recombinant antigens, at concentrations of 25 ng of antigen per well (rHsp58 and rOmpL1) or 5 ng of antigen per well (rLipL32). Absorbance (OD450) values are shown for reactions with serum samples, diluted 50-fold, from healthy individuals in an endemic region for leptospirosis in Salvador, Brazil (group A, □), or a nonendemic U.S. region (group B, Δ) compared to paired serum samples from 50 patients with confirmed leptospirosis from Salvador, Brazil (group C, acute-phase ● or group D, convalescent-phase ▪). Fifty samples were tested in each group. Median absorbance values for each group of samples are shown as solid lines. The cutoff value, defined as the absorbance value for the 96th percentile of the group of 50 serum samples from healthy control individuals from Salvador, Brazil, are represented as dashed lines.
TABLE 1.
IgG antibody reactivity of serum samples from patients with leptospirosis and control groups against five leptospiral His6 fusion proteins as determined by ELISA
| Study group | Total no. of serum samples | % of samples reactivea with recombinant leptospiral antigen (cutoff value, OD450):
|
||||
|---|---|---|---|---|---|---|
| rLipL32 (0.050) | rOmpL1 (0.247) | rLipL41 (0.099) | rHsp58 (0.195) | rLipL36 (0.123) | ||
| Confirmed cases of leptospirosis | ||||||
| Acute-phase samples | 50 | 56 | 16 | 24 | 18 | 12 |
| Convalescent-phase samples | 50 | 94 | 72 | 44 | 32 | 18 |
| Community-based control groups | ||||||
| Endemic | 50 | 2 | 4 | 4 | 4 | 4 |
| Nonendemic | 50 | 8 | 12 | 10 | 2 | 2 |
| Patient control groups | ||||||
| Dengue | 30 | 3 | 0 | 10 | 3 | ND |
| Hepatitis | 30 | 10 | 7 | 30 | 0 | ND |
| Initial suspicion of leptospirosisb | 16 | 6 | 6 | 13 | 13 | 19 |
| Lyme disease | 30 | 23 | 0 | 20 | 3 | ND |
| VDRL test positive | 30 | 13 | 10 | 33 | 0 | ND |
Proportion of samples with absorbance values greater than the cut off values for each recombinant-antigen ELISA. Cutoff values were defined as the 96th percentile of absorbance values of 50 serum samples from healthy individuals in Salvador, Brazil, a region of high leptospirosis incidence. ND, not determined.
Patients were hospitalized with suspected leptospirosis and subsequently diagnosed with other diseases based on clinical, radiological, and laboratory evidence. Final diagnoses included acute hepatitis B infection, bacterial pneumonia, and sepsis (two cases each) and meningitis, endocarditis, acute pancreatitis, upper gastrointestinal tract hemorrhage, arthritis, visceral leishmaniasis, typhoid fever, choledocholithiasis, parotiditis, and chronic hepatic disease (one case each).
Sensitivity of recombinant-antigen IgG ELISAs.
The rLipL32 ELISA demonstrated the highest sensitivity in acute- and convalescent-phase serum samples (56 and 94%, respectively) from confirmed cases of leptospirosis (Table 1 and Fig. 2). Other recombinant-antigen ELISAs demonstrated less than 30% sensitivity to detect leptospiral infection in the acute phase of illness. In convalescent-phase serum samples, the sensitivity of the rOmpL1 ELISA increased to 72%, while the rLipL41 and rHsp58 assays had sensitivities of 44 and 32%, respectively. Less than 20% of case serum samples had detectable antibody against rLipL36, a lipoprotein that is produced at low levels during infection, although the median serum absorbance values did increase significantly between the acute and convalescent phase of disease (median OD450, 0.044 versus 0.071; P < 0.05).
Combining results of the rHsp58 and rLipL32 ELISAs increased sensitivity from 56 to 64% in acute-phase sera, with a corresponding decrease in diagnostic specificity from 95 to 92%. Combinations of results from other recombinant-antigen ELISAs did not further increase the sensitivity for detecting leptospiral infection in acute-phase samples.
Specificity of recombinant-antigen IgG ELISAs in control patient groups.
Reactivities of antibodies from dengue, hepatitis, Lyme, and VDRL test-positive patients were not determined for rLipL36 since overall sensitivity was less than 20%. Percent reactivity among patient control groups did not differ significantly from community-based control groups from Brazil or the U.S. in ELISAs with rLipL32, rHsp58, rOmpL1, and rLipL41 (Table 1). Exceptions included Lyme disease patients in the rLipL32 assay, of whom seven (23%) had serum absorbance values above the cutoff (versus five [5%] healthy individuals), and hepatitis patients and VDRL test-positive individuals in the rLipL41 assay, of whom 10 (30%) and 11 (33%), respectively, had serum absorbance above the cutoff (versus 6 [7%] healthy individuals). Among patients with diseases initially suspected as being leptospirosis, 2 of the 16 patients in the first group were responsible for outlying values (greater than twice the cutoff value) in the rLipL32 ELISA; both of these patients had an invasive bacterial infection (sepsis or pneumonia).
rLipL32 IgG ELISA versus standard diagnostic tests.
The positivity of the rLipL32 IgG ELISA in acute-phase samples increased from 36% (10 of 28) during the first week (≤7 days) of symptoms to 86% (18 of 21) after the first week of symptoms (8 to 23 days) (Table 2). LipL32 seropositivity was not detected in five samples obtained in the first 4 days of illness.
TABLE 2.
Comparison of rLipL32 IgG ELISA with standard diagnostic tests for leptospirosis
| Days after initiation of symptomsa | No. of samples | Standard diagnostic tests
|
rLipL32 IgG ELISA
|
||||
|---|---|---|---|---|---|---|---|
| Maximum reciprocal MAT titer, median (range) | No. (%) of samples
|
Whole-Leptospira IgM ELISA positiveb | OD450 median (range) | No. (%) giving positive reactionsc | |||
| Reciprocal MAT, titer ≥ 100 | Culture isolation | ||||||
| Acute-phase samples (n = 50)d | |||||||
| 2–4 | 5 | 0 (0–200) | 1 (20) | 1 (100)e | 1 (20) | 0.008 (0.003–0.039) | 0 (0) |
| 5–7 | 23 | 0 (0–800) | 7 (29) | 6 (67)e | 9 (41) | 0.045 (0.005–1.181) | 10 (43) |
| 8–23 | 21 | 200 (0–3,200) | 12 (60) | 0 (0)e | 16 (80) | 0.176 (0.023–1.054) | 18 (86) |
| Convalescent-phase samples (n = 50) | |||||||
| 17–28 | 23 | 1,600 (200–6,400) | 23 (100) | NA | 21 (100) | 0.501 (0.052–0.998) | 23 (100) |
| 29–35 | 19 | 1,600 (400–6,400) | 19 (100) | NA | 18 (95) | 0.591 (0.008–1.050) | 16 (84) |
| 36–113 | 8 | 800 (200–800) | 8 (100) | NA | 8 (100) | 0.344 (0.100–1.256) | 8 (100) |
Days between reported onset of illness and serum collection; one was missing. NA, not applicable.
Results of the whole-Leptospira IgM ELISA were not available for two individuals' paired serum samples. Percentages shown are for the number of samples tested.
Positive reactions were defined as absorbance greater than the 96th percentile among 50 serum samples from healthy individuals.
Acute-phase serum samples were collected upon hospital admission.
A total of 14 blood cultures were performed for 50 serum samples collected during the acute phase of illness; 1, 9, and 5 were performed for samples collected in the first 4, 5 to 7, and >7 days of illness, respectively.
The results of the rLipL32 IgG ELISA were compared with those of the MAT for the 50 confirmed cases of leptospirosis (Table 2). At the time of hospital admission, the rLipL32 IgG ELISA had greater sensitivity (56 versus 42%) than MAT positivity at a 100-fold serum dilution, a commonly used criterion for screening for leptospiral infection. Among acute-phase samples, 12 (24%) that tested positive in the rLipL32 ELISA had undetectable agglutination reactions in the MAT, while 5 (10%) that had reciprocal MAT titers greater than or equal to 100 had negative rLipL32 ELISA results. For samples collected from MAT-confirmed leptospirosis cases in the convalescent phase of illness following discharge from the hospital, 47 (94%) of 50 had positive absorbance values in the rLipL32 IgG ELISA. A positive reaction in the rLipL32 assay was observed in one sample obtained from a patient 113 days after the onset of disease.
For acute-phase samples, culture isolation was negatively associated with rLipL32 ELISA absorbance value (P = 0.011). None of the seven samples from culture-positive cases were seroreactive in the rLipL32 assay, whereas six of seven (86%) from culture-negative cases were seroreactive. All culture-positive patient samples had undetectable MAT agglutination titers in the acute phase of the disease.
In the whole-Leptospira IgM ELISA, 26 (54%) acute-phase samples and 47 (98%) convalescent-phase samples were positive among the 48 patients tested. The whole-Leptospira IgM and IgG rLipL32 ELISAs demonstrated similar kinetics of reactivity in the first 2 weeks after the onset of illness (Table 2). Both IgM and rLipL32 IgG ELISAs were positive in 19 (40%) of 48 patients at the time of hospital admission, while 7 (15%) were only IgM positive, and 8 (17%) were only rLipL32 IgG positive.
DISCUSSION
Improved diagnostic tests for leptospirosis are urgently needed to aid clinical diagnosis in the initial phase of the disease and for rapid case confirmation during outbreak surveillance. Doxycycline therapy initiated soon after the onset of symptoms can prevent severe complications and deaths (15, 31). However, diagnosis during this critical period requires a high index of suspicion, as early symptoms are often indistinguishable from those of dengue fever and other febrile illnesses (24, 37). Efforts to develop new diagnostic tests to achieve high sensitivity in the acute phase have focused primarily on detecting IgM binding to whole-cell antigen preparations. The immunodominant moiety in whole-cell preparations appears to be a broadly reactive antigen (15, 46) that is a disaccharide epitope present in nonpathogenic leptospires as well as a diverse group of nonleptospiral species (29, 30). IgM ELISAs (12, 48), dipstick assays (25, 43, 44, 50), and simple agglutination assays (25, 45) appear to have variable sensitivities of 35 to 85% for serum samples collected in the first 10 days of illness from cases subsequently confirmed by MAT, in evaluations performed in different sites (Table 3) (43, 44).
TABLE 3.
Comparison of results of serodiagnostic tests for leptospirosis from selected validation studies with the rLipL32 ELISA
| Assay | Evaluation site | No. of samplesa | % Sensitivityb
|
% Specificityc | Reference | |
|---|---|---|---|---|---|---|
| Acute phase | Convalescent phase | |||||
| LipL32 IgG ELISA | Brazil | 50 | 56 | 94 | 95 | |
| IgM ELISA | Barbados | 92 | 52d | 93 | 95–98 | 12 |
| Australia | 41 | 42e | 100 | 98 | 48 | |
| Seychelles | 90 | 38 | 68 | 97 | 50 | |
| IgM dipstick | Barbados | 51 | 71 | 98 | 91 | 25 |
| Seychelles | 90 | 39 | 68 | 96 | 50 | |
| Multicenter | 485 | 35–86f | 69–100f | 88–99f | 43 | |
| Multicenter | 329 | 85 | 94 | 88–94 | 44 | |
| Latex agglutination | Netherlands | 254 | 54 | 94 | 94 | 45 |
| Multicenter | 137 | 38 | 88 | 95 | 45 | |
Laboratory case-confirmation includes MAT, culture, and PCR evaluation.
Acute-phase samples were collected from patients during the first 10 days of illness unless otherwise noted. Convalescent-phase samples were collected from patients following hospitalization.
Specificities are shown for groups of healthy individuals or nonleptospirosis patients.
Samples collected at the time of hospital admission (median = 5 days) after onset of symptoms.
For samples collected 0 to 8 days after the onset of symptoms.
Mean sensitivity was 60% for acute-phase and 87% for convalescent-phase samples; mean specificity was 93%.
Recombinant protein-based serologic tests may achieve high sensitivity and specificity because of the high concentration of immunoreactive antigens which can be used in assays and the lack of nonspecific moieties present in whole-cell preparations. An ideal antigen would be a principal target of the host immune response, expressed only in pathogenic Leptospira spp. and conserved among the more than 200 serovars associated with human disease in different geographic regions and epidemiological situations (15). An ideal test will need to discriminate between leptospirosis and a broad spectrum of diseases that cause acute febrile illnesses and have overlapping clinical presentations. Because the burden of leptospirosis is greatest in developing countries (15), there is a need to develop a test which can be produced at low cost and easily standardized for use in field settings.
In the present study, five recombinant leptospiral proteins were evaluated as antigens in ELISAs. The rLipL32 ELISA demonstrated the highest sensitivity: it detected IgG antibodies in 56 and 94% of the MAT-confirmed cases of leptospirosis during the acute and convalescent phases of illness, respectively. LipL32, the most prominent protein in the SDS-PAGE total protein profile (20) and the major outer membrane protein (51), was also the most frequently recognized antigen in immunoblots with patient sera (18). Whereas anti-LipL32 IgG reactivity was detected in samples collected from patients within the first 8 days of illness and this response correlated with the duration of symptoms, no anti-LipL32 IgM reactivity was detected in any acute- or convalescent-phase samples.
The diagnostic specificity of the rLipL32 IgG ELISA was 95% for healthy individuals living in regions of high and low leptospirosis incidence. The low background reactivity may be due, in part, to the restricted expression of LipL32 in pathogenic leptospires (20) and not saprophytic forms that are ubiquitous in the environment. The rLipL32 ELISA appears to differentiate patients with leptospirosis from those with other important causes of acute jaundice and febrile illnesses, such as dengue and hepatitis, since specificity in these groups was greater than 90%. Higher antibody reactivities in VDRL-positive and Lyme disease patients (13 and 23% reactivity, respectively) suggests that there may be cross-reactive epitopes in other spirochetes, including Borrelia and Treponema spp., which were not detected in BLAST homology searches (20).
After the rLipL32 ELISA, the two assays with the highest sensitivities were those based on rOmpL1 and rLipL41. Like LipL32, OmpL1 and LipL41 are found only in pathogenic Leptospira (19, 41) and are expressed by the pathogen during experimental infection (3). Triton X-100, used to maintain the solubility of these membrane-associated proteins, appeared to interfere with the binding of these antigens, as with LipL32, to ELISA plates, and reducing its concentration increased the sensitivity for these assays. In the present evaluation, the recombinant Hsp58 assay showed lower IgG reactivity than reported previously (32 versus 82%) (32). A high cutoff threshold was necessary to obtain greater than 95% specificity for the rHsp58 IgG ELISA. The dominant epitope in leptospiral Hsp58 is a 20-amino-acid sequence highly conserved among prokaryotic GroEL homologues (32). The observed background reactivity to recombinant Hsp58 may have contributed to the high cutoff value for this assay and lower sensitivity. In the present study, combinations of the results of other assays with those of the rLipL32 assay did not increase the sensitivity of ELISA for MAT-confirmed cases.
The robust IgG and undetectable IgM response to recombinant leptospiral proteins during early illness is surprising. However, there does exist precedence for this phenomenon in leptospirosis: several studies have documented IgG immunoblot reactivity to leptospiral proteins during acute-phase illness in the absence of specific IgM antibodies (10, 11, 18). Traditionally, IgM antibodies, directed primarily against carbohydrate epitopes (15), have been believed to be the predominant humoral response during acute-phase infection (1, 46). In this study, such a response was detected in whole-Leptospira ELISAs. Remarkably, the IgG response to rLipL32 and other recombinant antigens was found to have kinetics comparable to that of the IgM response to whole-antigen preparations. The rapid rise in IgG antibody may have been due to a memory response in individuals with prior exposure to leptospires. Alternatively, this could represent a rapid IgM-to-IgG class-switch phenomenon, since it appears unlikely that the relatively large proportion of patients who seroconverted in both whole-Leptospira IgM and rLipL32 IgG ELISAs were previously exposed. Together, these findings suggest that early host immune response to Leptospira infection is characterized by both IgM and IgG antibodies specific for different moieties, as observed in the early response to Borrelia (13, 27) and Treponema infection (39).
Our findings indicate that recombinant LipL32 may be an appropriate antigen for serodiagnosis in field settings. The rLipL32 ELISA demonstrated sensitivity comparable to currently available rapid screening tests in the acute phase and high sensitivity for MAT-confirmed cases in the convalescent phase (Table 3). Although whole Leptospira antigen can be used in serodiagnostic tests to detect either IgM or IgG antibodies (1, 46), extensive quality control measures are necessary to monitor batch-to-batch variability in antigen composition inherent in growing large cultures of Leptospira (15). Recombinant-antigen-based assays may circumvent this problem and furthermore may be produced at lower cost, an important consideration for implementation in developing countries.
One potential limitation to the use of this test in case confirmation is the persistence the anti-LipL32 IgG response after acute infection. In this study, seropositivity was detected in samples obtained up to 113 days after the onset of symptoms. The duration of seropositivity in ELISAs will need to be evaluated further in longitudinal studies of the host IgG response. However, the criterion for positivity used in the recombinant-antigen ELISA was a cutoff absorbance value which gave 98% specificity in community control groups from the same epidemic region as cases. Furthermore, the specificity of this cutoff was >90% for patient control groups from the same region. Therefore, the response to LipL32 may not be prolonged enough to interfere with the application of this test in epidemiologic and clinical situations associated with recurrent urban epidemics.
The conserved nature and high level of expression of LipL32 among pathogenic Leptospira spp. (20) suggest that the rLipL32 ELISA may exhibit similar performance regardless of the locally predominant serovar agent. Sera used in the present evaluation were from patients residing in regions in which the most prevalent Leptospira serovar is copenhageni, the etiologic agent of urban epidemics throughout Brazil (4, 23, 34, 36). Validation studies in geographical regions with a spectrum of etiologic serovars will be helpful in assessing whether the rLipL32 ELISA can be widely applied. Furthermore, recombinant LipL32 can be incorporated in rapid formats, such as dipstick, to facilitate its use in serodiagnosis in developing countries.
ACKNOWLEDGMENTS
This work was supported by grants from Biomanguinhos, Oswaldo Cruz Foundation, Brazilian Ministry of Health (09224-7), the Brazilian National Research Council (521.229/98-7, 300.861/96-6; 350.052/95-6, and FINEP 4196086200), VA Medical Research Funds, and the National Institutes of Health (AI-01605, AI-34431, TW-00905, and TW-00919).
We thank Mary Mazel and Tracy Young (Division of Infectious Diseases, West Los Angeles Veterans Affairs Medical Center) for preparation of the recombinant membrane-associated leptospiral proteins; and Fernanda Carvalho Pinheiro, Patrícia Guimarães Oliveira, Suzana Ramos Ferrer (Gonçalo Moniz Research Center, Oswaldo Cruz Foundation), and Kátia Salgado (Couto Maia Hospital, Secretary of Health for the State of Bahia) for technical assistance in collection of sera from patients and laboratory confirmation of leptospirosis.
REFERENCES
- 1.Adler B, Murphy A M, Locarnini S A, Faine S. Detection of specific antileptospiral immunoglobulins M and G in human serum by solid-phase enzyme-linked immunosorbent assay. J Clin Microbiol. 1980;11:452–457. doi: 10.1128/jcm.11.5.452-457.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ballard S A, Segers R P, Bleumink-Pluym N, Fyfe J, Faine S, Adler B. Molecular analysis of the hsp (groE) operon of Leptospira interrogans serovar copenhageni. Mol Microbiol. 1993;8:739–751. doi: 10.1111/j.1365-2958.1993.tb01617.x. [DOI] [PubMed] [Google Scholar]
- 3.Barnett J K, Barnett D, Bolin C A, Summers T A, Wagar E A, Cheville N F, Hartskeerl R A, Haake D A. Expression and distribution of leptospiral outer membrane components during renal infection of hamsters. Infect Immun. 1999;67:853–861. doi: 10.1128/iai.67.2.853-861.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Barocchi M A, Ko A I, Ferrer S R, Faria M T, Reis M G, Riley L W. Identification of new repetitive element in Leptospira interrogans serovar copenhageni and its application to PCR-based differentiation of Leptospira serogroups. J Clin Microbiol. 2001;39:191–195. doi: 10.1128/JCM.39.1.191-195.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bounlu K, Insisiengmay S, Vanthanouvong K, Saykham, Widjaja S, Iinuma K, Matsubayashi K, Laras K, Putri M, Endy T, Vaughn D, Raengsakulrach B, Hyams K, Hayden M, Scheffel C, Corwin A. Acute jaundice in Vientiane, Lao People's Democratic Republic. Clin Infect Dis. 1998;27:717–721. doi: 10.1086/514948. [DOI] [PubMed] [Google Scholar]
- 6.Bughio N I, Lin M, Surujballi O P. Use of recombinant flagellin protein as a tracer antigen in a fluorescence polarization assay for diagnosis of leptospirosis. Clin Diagn Lab Immunol. 1999;6:599–605. doi: 10.1128/cdli.6.4.599-605.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Centers for Disease Control. Outbreak of acute febrile illness among athletes participating in triathlons—Wisconsin and Illinois, 1998. Morb Mortal Wkly Rep. 1998;47:585–588. [PubMed] [Google Scholar]
- 8.Centers for Disease Control. Outbreak of acute febrile illness among participants in EcoChallenge Sabah 2000—Malaysia, 2000. Morb Mortal Wkly Rep. 2000;49:816–817. [Google Scholar]
- 9.Centers for Disease Control. Outbreak of leptospirosis among white-water rafters–Costa Rica, 1996. Morb Mortal Wkly Rep. 1997;46:577–579. [PubMed] [Google Scholar]
- 10.Chapman A J, Adler B, Faine S. Antigens recognised by the human immune response to infection with Leptospira interrogans serovar hardjo. J Med Microbiol. 1988;25:269–278. doi: 10.1099/00222615-25-4-269. [DOI] [PubMed] [Google Scholar]
- 11.Chapman A J, Everard C O, Faine S, Adler B. Antigens recognized by the human immune response to severe leptospirosis in Barbados. Epidemiol Infect. 1991;107:143–155. doi: 10.1017/s0950268800048779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cumberland P, Everard C O, Levett P N. Assessment of the efficacy of an IgM-elisa and microscopic agglutination test (MAT) in the diagnosis of acute leptospirosis. Am J Trop Med Hyg. 1999;61:731–734. doi: 10.4269/ajtmh.1999.61.731. [DOI] [PubMed] [Google Scholar]
- 13.Engstrom S M, Shoop E, Johnson R C. Immunoblot interpretation criteria for serodiagnosis of early lyme disease. J Clin Microbiol. 1995;33:419–427. doi: 10.1128/jcm.33.2.419-427.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Everard C O, Edwards C N, Everard J D, Carrington D G. A twelve-year study of leptospirosis on Barbados. Eur J Epidemiol. 1995;11:311–320. doi: 10.1007/BF01719436. [DOI] [PubMed] [Google Scholar]
- 15.Faine S, Adler B, Perolat P, Bolin C A. Leptospira and leptospirosis. 2nd ed. Melbourne, Australia: MediSci; 1999. [Google Scholar]
- 16.Farr R W. Leptospirosis. Clin Infect Dis. 1995;21:1–6. doi: 10.1093/clinids/21.1.1. [DOI] [PubMed] [Google Scholar]
- 17.Goossens H A T, van der Bogaard A E, Nohlmans M K E. Evaluation of fifteen commercially available serologic tests for diagnosis of lyme borreliosis. Eur J Clin Microbiol Infect Dis. 1999;18:551–560. doi: 10.1007/s100960050347. [DOI] [PubMed] [Google Scholar]
- 18.Guerreiro H, Croda J, Flannery B, Mazel M, Matsunaga J, Reis M G, Levett P N, Ko A I, Haake D A. Leptospiral proteins recognized during the humoral immune response to leptospirosis in humans. Infect Immun. 2001;69:4958–4968. doi: 10.1128/IAI.69.8.4958-4968.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Haake D A, Champion C I, Martinich C, Shang E S, Blanco D R, Miller J N, Lovett M A. Molecular cloning and sequence analysis of the gene encoding OmpL1, a transmembrane outer membrane protein of pathogenic Leptospira spp. J Bacteriol. 1993;175:4225–4234. doi: 10.1128/jb.175.13.4225-4234.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Haake D A, Chao G, Zuerner R L, Barnett J K, Barnett D, Mazel M, Matsunaga J, Levett P N, Bolin C A. The leptospiral major outer membrane protein LipL32 is a lipoprotein expressed during mammalian infection. Infect Immun. 2000;68:2276–2285. doi: 10.1128/iai.68.4.2276-2285.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Haake D A, Martinich C, Summers T A, Shang E S, Pruetz J D, McCoy A M, Mazel M K, Bolin C A. Characterization of leptospiral outer membrane lipoprotein LipL36: downregulation associated with late-log-phase growth and mammalian infection. Infect Immun. 1998;66:1579–1587. doi: 10.1128/iai.66.4.1579-1587.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hauser U, Wilske B. Enzyme-linked immunosorbent assays with recombinant internal flagellin fragments derived from different species of Borrelia burgdorferi sensu lato for the serodiagnosis of Lyme neuroborreliosis. Med Microbiol Immunol. 1997;186:145–151. doi: 10.1007/s004300050057. [DOI] [PubMed] [Google Scholar]
- 23.Ko A I, Galvao Reis M, Ribeiro Dourado C M, Johnson W D, Jr, Riley L W the Salvador Leptospirosis Study Group. Urban epidemic of severe leptospirosis in Brazil. Lancet. 1999;354:820–825. doi: 10.1016/s0140-6736(99)80012-9. [DOI] [PubMed] [Google Scholar]
- 24.Levett P N, Branch S L, Edwards C N. Detection of dengue infection in patients investigated for leptospirosis in Barbados. Am J Trop Med Hyg. 2000;62:112–114. doi: 10.4269/ajtmh.2000.62.112. [DOI] [PubMed] [Google Scholar]
- 25.Levett P N, Branch S L, Whittington C U, Edwards C N, Paxton H. Two methods for rapid serological diagnosis of acute leptospirosis. Clin Diagn Lab Immunol. 2001;8:349–351. doi: 10.1128/CDLI.8.2.349-351.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lomar A, Diament D, Torres J. Leptospirosis in Latin America. Infect Dis Clin North Am. 2000;14:23–39. doi: 10.1016/s0891-5520(05)70216-6. [DOI] [PubMed] [Google Scholar]
- 27.Magnarelli L A, Ijdo J W, Padula S J, Flavelli R A, Fikrig E. Serologic diagnosis of Lyme borreliosis by using enzyme-linked immunosorbent assays with recombinant antigens. J Clin Microbiol. 2000;38:1735–1739. doi: 10.1128/jcm.38.5.1735-1739.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Marotto P C, Nascimento C M, Eluf-Neto J, Marotto M S, Andrade L, Sztajnbok J, Seguro A C. Acute lung injury in leptospirosis: clinical and laboratory features, outcome, and factors associated with mortality. Clin Infect Dis. 1999;29:1561–1563. doi: 10.1086/313501. [DOI] [PubMed] [Google Scholar]
- 29.Matsuo K, Isogai E, Araki Y. Occurrence of [→3)-β-D-Manp-(1–4)-β-D-Manp-(1→]n units in the antigenic polysaccharides from Leptospira biflexa serovar patoc strain Patoc I. Carbohydr Res. 2000;328:517–524. doi: 10.1016/s0008-6215(00)00143-9. [DOI] [PubMed] [Google Scholar]
- 30.Matsuo K, Isogai E, Araki Y. Utilization of exocellular mannan from Rhodotorula glutinis as an immunoreactive antigen in diagnosis of leptospirosis. J Clin Microbiol. 2000;38:3750–3754. doi: 10.1128/jcm.38.10.3750-3754.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.McClain J B, Ballou W R, Harrison S M, Steinweg D L. Doxycycline therapy for leptospirosis. Ann Intern Med. 1984;100:696–698. doi: 10.7326/0003-4819-100-5-696. [DOI] [PubMed] [Google Scholar]
- 32.Park S H, Ahn B Y, Kim M J. Expression and immunologic characterization of recombinant heat shock protein 58 of Leptospira species: a major target antigen of the humoral immune response. DNA Cell Biol. 1999;18:903–910. doi: 10.1089/104454999314764. [DOI] [PubMed] [Google Scholar]
- 33.Park S K, Lee S H, Rhee Y K, Kang S K, Kim K J, Kim M C, Kim K W, Chang W H. Leptospirosis in Chonbuk Province of Korea in 1987: a study of 93 patients. Am J Trop Med Hyg. 1989;41:345–351. [PubMed] [Google Scholar]
- 34.Pereira M M, Matsuo M G, Bauab A R, Vasconcelos S A, Moraes Z M, Baranton G, Saint Girons I. A clonal subpopulation of Leptospira interrogans sensu stricto is the major cause of leptospirosis outbreaks in Brazil. J Clin Microbiol. 2000;38:450–452. doi: 10.1128/jcm.38.1.450-452.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rios-Goncalves A J, Capone D, Paz N A, Paulo R V V, Dias T B C M, Lago V C C, Carvalho L M A. Leptospirosis: observations on changes in clinical manifestations in Rio de Janeiro during a large epidemic in 1988. Arq Bras Med. 1990;64:389–397. [Google Scholar]
- 36.Sakata E E, Yasuda P H, Romero E C, Silva M V, Lomar A V. The serovars of Leptospira interrogans isolated from cases of human leptospirosis in Sao Paulo, Brazil. Rev Inst Med Trop Sao Paulo. 1992;34:217–221. [PubMed] [Google Scholar]
- 37.Sanders E J, Rigau-Perez J G, Smits H L, Deseda C C, Vorndam V A, Aye T, Spiegel R A, Weyant R S, Bragg S L. Increase of leptospirosis in dengue-negative patients after a hurricane in Puerto Rico in 1966. Am J TropMed Hyg. 1999;61:399–404. doi: 10.4269/ajtmh.1999.61.399. [DOI] [PubMed] [Google Scholar]
- 38.Sasaki D M, Pang L, Minette H P, Wakida C K, Fujimoto W J, Manea S J, Kunioka R, Middleton C R. Active surveillance and risk factors for leptospirosis in Hawaii. Am J Trop Med Hyg. 1993;48:35–43. doi: 10.4269/ajtmh.1993.48.35. [DOI] [PubMed] [Google Scholar]
- 39.Schmidt B L, Edjlalipour M, Luger A. Comparitive evaluation of nine different enzyme-linked immunosorbent assays for determination of antibodies against Treponema pallidum in patients with primary syphilis. J Clin Microbiol. 2000;38:1279–1282. doi: 10.1128/jcm.38.3.1279-1282.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sehgal S C, Murhekar M V, Sugunan A P. Outbreak of leptospirosis with pulmonary involvement in north Andaman. Indian J Med Res. 1995;102:9–12. [PubMed] [Google Scholar]
- 41.Shang E S, Summers T A, Haake D A. Molecular cloning and sequence analysis of the gene encoding LipL41, a surface-exposed lipoprotein of pathogenic Leptospira species. Infect Immun. 1996;64:2322–2330. doi: 10.1128/iai.64.6.2322-2330.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Smith P K, Krohn R I, Hermanson G T, Mallia A K, Gartner F H, Provenzano M D, Fujimoto E K, Goeke N M, Olson B J, Klenk D C. Measurement of protein using bicinchoninic acid. Anal Biochem. 1985;150:76–85. doi: 10.1016/0003-2697(85)90442-7. [DOI] [PubMed] [Google Scholar]
- 43.Smits H L, Ananyina Y V, Chereshsky A, Dancel L, Lai A F R F, Chee H D, Levett P N, Masuzawa T, Yanagihara Y, Muthusethupathi M A, Sanders E J, Sasaki D M, Domen H, Yersin C, Aye T, Bragg S L, Gussenhoven G C, Goris M G, Terpstra W J, Hartskeerl R A. International multicenter evaluation of the clinical utility of a dipstick assay for detection of Leptospira-specific immunoglobulin M antibodies in human serum specimens. J Clin Microbiol. 1999;37:2904–2909. doi: 10.1128/jcm.37.9.2904-2909.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Smits H L, Hartskeerl R A, Terpstra W J. International multi-centre evaluation of a dipstick assay for human leptospirosis. Trop Med Int Health. 2000;5:124–128. doi: 10.1046/j.1365-3156.2000.00525.x. [DOI] [PubMed] [Google Scholar]
- 45.Smits H L, van der Hoorn M A, Goris M G, Gussenhoven G C, Yersin C, Sasaki D M, Terpstra W J, Hartskeerl R A. Simple latex agglutination assay for rapid serodiagnosis of human leptospirosis. J Clin Microbiol. 2000;38:1272–1275. doi: 10.1128/jcm.38.3.1272-1275.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Terpstra W J, Ligthart G S, Schoone G J. ELISA for the detection of specific IgM and IgG in human leptospirosis. J Gen Microbiol. 1985;131:377–385. doi: 10.1099/00221287-131-2-377. [DOI] [PubMed] [Google Scholar]
- 47.Trevejo R T, Rigau-Perez J G, Ashford D A, McClure E M, Jarquin-Gonzalez C, Amador J J J, de los Reyes O, Gonzalez A, Zaki S R, Shieh W J, McLean R G, Nasci R S, Weyant R S, Bolin C A, Bragg S L, Perkins B A, Spiegel R A. Epidemic leptospirosis associated with pulmonary hemorrhage-Nicaragua, 1995. J Infect Dis. 1998;178:1457–1463. doi: 10.1086/314424. [DOI] [PubMed] [Google Scholar]
- 48.Winslow W E, Merry D J, Pirc M L, Devine P L. Evaluation of a commercial enzyme-linked immunosorbent assay for detection of immunoglobulin M antibody in diagnosis of human leptospiral infection. J Clin Microbiol. 1997;35:1938–1942. doi: 10.1128/jcm.35.8.1938-1942.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yersin C, Bovet P, Merien F, Clement J, Laille M, Van Ranst M, Perolat P. Pulmonary haemorrhage as a predominant cause of death in leptospirosis in Seychelles. Trans R Soc Trop Med Hyg. 2000;94:71–76. doi: 10.1016/s0035-9203(00)90445-0. [DOI] [PubMed] [Google Scholar]
- 50.Yersin C, Bovet P, Smits H L, Perolat P. Field evaluation of a one-step dipstick assay for the diagnosis of human leptospirosis in the Seychelles. Trop Med Int Health. 1999;4:38–45. doi: 10.1046/j.1365-3156.1999.00352.x. [DOI] [PubMed] [Google Scholar]
- 51.Zuerner R L, Knudtson W, Bolin C A, Trueba G. Characterization of outer membrane and secreted proteins of Leptospira interrogans serovar pomona. Microb Pathog. 1991;10:311–322. doi: 10.1016/0882-4010(91)90014-2. [DOI] [PubMed] [Google Scholar]