Abstract
The present study analyzed the impact of 68Ga-labeled prostate-specific membrane antigen-HBED-CC (68Ga-PSMA-11) PET/CT on radiotherapeutic management in a large cohort of men with primary or recurrent prostate cancer. Methods: This study investigated 121 men with carcinoma of the prostate who underwent 68Ga-PSMA-11 PET/CT as well as conventional imaging. Fifty patients were treatment-naive, 11 had persistent prostate-specific antigen (PSA) soon after surgery, and 60 presented with recurrent PSA after definitive therapy. Changes in TNM classification of malignant tumor stage (TNM) and radiotherapeutic management after 68Ga-PSMA-11 imaging were compared with results achieved with conventional imaging. Results: In total, a change in TNM stage and radiotherapeutic management was observed for 49 patients (40.5%) and 62 patients (51.2%), respectively. In treatment-naive patients, a change in TNM stage and radiotherapeutic plan occurred in 26.0% and 44.0% of the cohort, respectively. For patients with PSA persistence or recurrence, TNM and radiotherapeutic management changed in 50.7% and 56.3%, respectively. Conclusion: 68Ga-PSMA-11 PET/CT may shortly become an indispensable tool for detecting prostate cancer lesions in treatment-naive patients as well as in men with recurrent disease or persistent PSA and seems to be helpful in personalizing radiotherapeutic management to the individual patients’ distribution of disease.
Keywords: prostate cancer, PSMA, PET/CT, radiotherapy, staging
Since the development of 68Ga-labeled prostate-specific membrane antigen-HBED-CC (68Ga-PSMA-11) PET/CT about 7 y ago, the ability to accurately stage prostate cancer has improved dramatically. 68Ga-PSMA-11 PET/CT enables a highly accurate identification of prostate cancer both within and outside the prostate. With a sensitivity and specificity for prostate cancer of up to 80% and 95%, respectively, in higher grade disease, the detection of lymph node metastases or bone lesions that would otherwise remain undetected using conventional imaging such as MRI or CT and bone scan is allowed. This is especially true for patients with high-risk disease but low prostate-specific antigen (PSA) levels (1–4). Eiber et al. demonstrated detection rates of 72.7% and 57.9% at PSA levels of 0.5–1 and 0.2–0.5 ng/mL, respectively, for a cohort of 248 patients after surgery (5). Detection rates with 68Ga-PSMA-11 are superior to other PET probes such as 18F-fluorocholine PET/CT, especially in patients with low PSA values (6). Therefore, as it becomes more available, 68Ga-PSMA-11 PET/CT is increasingly used to stage high- or intermediate-risk patients with newly diagnosed or recurrent prostate cancer (7).
The role of PSMA imaging in radiation oncology has not been widely discussed. Because histologic confirmation of PSMA imaging results in patients undergoing radiation therapy has trailed behind surgical series in which histologic validation is routine, there have been less data to report. However, data supporting the accuracy of PSMA imaging based on surgery have improved confidence in using the results to guide radiotherapy. Using 68Ga-PSMA-11 PET/CT to guide radiation could lead to a more individualized and precise delivery of radiotherapy, improving effectiveness while decreasing side effects. Although several studies have discussed the impact of 68Ga-PSMA-11 PET/CT on prostate cancer management, most of them are focused on recurrent disease (8–12) or have included only a small number of patients (13,14). Thus, the purpose of this study was to evaluate the role of 68Ga-PSMA-11 PET/CT in a large cohort of patients with treatment-naive or recurrent prostate cancer in altering TNM stage and radiotherapy planning.
MATERIALS AND METHODS
Study Design and Patient Characteristics
This retrospective single-center exploratory study was approved by the local institutional review board (S-636/2017) and conducted in agreement with the Declaration of Helsinki and its later amendments. Between July 2011 and August 2017, 68Ga-PSMA-11 PET/CT was performed in 2,186 patients with prostate carcinoma at initial diagnosis or with PSA persistence/recurrence after primary treatment. Of this cohort, 2,065 patients were excluded because no comparable conventional imaging or insufficient clinical data were available for evaluation. In the remaining 121 men, conventional imaging for staging was available at a maximum of 4 mo before or after 68Ga-PSMA-11 PET/CT (median, 28 d; range, 0–124 d). In this study, conventional imaging was performed either by MRI or CT according to national guidelines. One hundred ten patients (90.9%) underwent CT and 44 (36.4%) MRI scanning. Bone scanning was performed for all symptomatic patients and for men with 68Ga-PSMA-11 PET/CT–positive bone lesions without CT correlate. Eleven patients underwent only bone scanning due to the detection of multiple bone metastases.
68Ga-PSMA-11 PET/CT Imaging
Fifty-three patients underwent imaging on a Biograph mCT Flow scanner (Siemens) using the following parameters: PET in 3-dimensional mode (matrix, 200 × 200) was acquired using FlowMotion. For the emission data, correction for randoms, scatter, and decay was performed. Images were reconstructed with an ordered-subset expectation maximization algorithm with 2 iterations/21 subsets and Gauss-filtered to a transaxial resolution of 5 mm in full width at half maximum. An unenhanced low-dose CT reconstructed to a slice thickness of 5 mm with an increment of 3–4 mm was used for attenuation correction.
The remaining 68 patients underwent imaging on a Biograph 6 PET/CT scanner (Siemens), and examinations were performed using the following parameters: a whole-body PET image in 3-dimensional mode (matrix, 168 × 168) was acquired. A 4-min acquisition time with a 15.5-cm field of view was used for each bed position (16.2 cm, 4.2 cm overlapping scale). For the emission data, correction for randoms, scatter, and decay was performed. The images were reconstructed with an ordered-subset expectation maximization algorithm with 2 iterations/8 subsets and Gauss-filtered to a transaxial resolution of 5 mm in full width at half maximum. An unenhanced low-dose CT reconstructed to a slice thickness of 5 mm with an increment of 1.5 mm was used for attenuation correction.
PET imaging was acquired 63 ± 9 min after injection of a median activity of 231 MBq (range, 77–361 MBq) of 68Ga-PSMA-11. 68Ga-PSMA-11 was synthesized according to sterile methods as previously described (15–17).
Image Evaluation
Images were analyzed using Syngo TrueD (Siemens) and an appropriate workstation. 68Ga-PSMA-11 PET/CT scans were evaluated retrospectively by 2 board-certified nuclear medicine physicians and 1 board-certified radiation oncologist. Any tracer accumulation that was not related to physiologic tracer-uptake with relevant difference to the background was considered tumor-positive. All findings on 68Ga-PSMA-11 PET/CT were interpreted in consensus. Conventional imaging was evaluated by 2 board-certified radiologists in consensus without knowledge of 68Ga-PSMA-11 PET/CT results, thus establishing the pre–68Ga-PSMA-11 PET/CT TNM classification. Lymph nodes were considered tumor-positive on CT and MRI when they had a short-axis diameter 8 mm or greater. On MRI, focal contrast-enhancement in a lesion or suggestive findings on diffusion-weighted imaging were considered positive for tumor. On the bone scan, activity not related to degenerative or traumatic processes was considered tumor-positive. According to clinical routine, physicians were not masked to patient history. Stage and radiotherapeutic management were documented before and after 68Ga-PSMA-11 PET/CT imaging by 2 nuclear medicine physicians and 1 radiation oncologist.
Statistical Analysis
For statistical analysis Microsoft Excel for Mac, version 15.41 (Microsoft Corp.) and SPSS Statistics, version 24 (IBM Corp.) were used. Descriptive analyses were performed for patients and their tumor characteristics. Normality was tested using the Kolmogorov–Smirnov test, and mean and SD are given where normality was observed. In all other cases, median and range are used. The correlation of TNM change was determined using the McNemar test. A P value of less than 0.05 was considered statistically significant.
RESULTS
In total, 121 men (median age, 71 y; range, 50–84 y) with prostate carcinoma underwent 68Ga-PSMA-11 PET/CT. Fifty patients (41.3%) were scanned at initial diagnosis, 11 patients (9.1%) presented with PSA persistence after surgery and were scanned soon thereafter, and 60 men (49.6%) presented with recurrent disease at variable times after initial definitive therapy. Overall, 100 patients (87.6%) had high-risk disease at diagnosis according to D’Amico risk classification (18) (Table 1).
TABLE 1.
Patient Characteristics
| Characteristic | All patients | Initial diagnosis | PSA persistence after surgery | PSA recurrence |
| No. of patients | 121 | 50 | 11 | 60 |
| Median age (y) | 71 (50–84) | 72 (50–84) | 67 (53–75) | 69 (50–84) |
| Gleason score (n) | ||||
| ≤6 | 6 (5.0%) | 4 (8.0%) | — | 2 (3.3%) |
| 7 | 59 (48.8%) | 18 (36.0%) | 2 (18.2%) | 39 (65.0%) |
| ≥8 | 55 (45.5%) | 28 (56.0%) | 9 (81.8%) | 18 (30.0%) |
| Unknown | 1 (0.8%) | — | — | 1 (1.7%) |
| Median iPSA (ng/mL) | 9.95 (1.40–520.00) | 11.9 (3.50–313.13) | 18.58 (5.20–87.55) | 9.14 (1.40–520.00) |
| Nadir PSA (ng/mL) | 0.09 (<0.01–9.70) | — | 1.36 (0.53–9.70) | 0.07 (<0.01–3.00) |
| PSA at PET (ng/mL) | 3.06 (0.03–41.24) | 9.76 (0.06–37.10) | 1.27 (0.03–9.70) | 1.10 (0.10–41–24) |
| Risk group according to D’Amico (n) | ||||
| Low | 3 (2.5%) | 3 (6.0%) | — | — |
| Intermediate | 12 (9.9%) | 9 (18.0%) | — | 3 (5.0%) |
| High | 106 (87.6%) | 38 (76.0%) | 11 (100.0%) | 57 (95.0%) |
| Ongoing ADT (n) | ||||
| Yes | 26 (21.5%) | 12 (24.0%) | 5 (45.5%) | 9 (15.0%) |
| No | 95 (78.5%) | 38 (76.0%) | 6 (54.5%) | 51 (85.0%) |
| Prior RP only | 34 (28.1%) | — | 11 (100.0%) | 23 (38.3%) |
| Prior RT only | 5 (4.1%) | — | — | 5 (8.3%) |
| Prior RP and RT | 32 (26.4%) | — | — | 32 (53.3%) |
| Surgery margin | ||||
| R0 | 40 (33.1%) | — | 2 (18.2%) | 38 (69.1%) |
| R1 | 24 (19.8%) | — | 9 (81.8%) | 15 (27.3%) |
| Rx | 2 (1.7%) | — | — | 2 (3.6%) |
iPSA = initial prostate-specific antigen serum level; ADT = androgen deprivation therapy; RP = radical prostatectomy; RT = radiotherapy.
Data in parentheses are ranges unless otherwise indicated.
Conventional imaging (CT or MRI ± bone scan) was performed in all patients. Using conventional imaging, lymph node metastases were diagnosed in 5 of 50 (10.0%) treatment-naive patients and 10 of 71 men (13.2%) after definitive treatment. Three patients (6.0%) at initial diagnosis and 23 men (32.4%) with persistence or recurrence had distant metastases (defined as extrapelvic lymph nodes, bone metastases, or soft-tissue metastases) on conventional imaging. 68Ga-PSMA-11 PET/CT detected lymph node metastases in 39 of 121 patients (32.2%) including 16.0% of the treatment-naive group and 43.7% of the persistence/recurrence group. Five patients (10.0%) at initial diagnosis and 36 men (50.7%) with persistence or recurrence had distant metastases on 68Ga-PSMA-11 PET/CT imaging. For men with recurrent or persistent PSA, CT or MRI led to inconclusive findings in 46.5% regarding local relapse, whereas 68Ga-PSMA-11 PET/CT was able to exclude recurrent disease in the prostate bed in 74.6% (Table 2). Overall, 68Ga-PSMA-11 PET/CT resulted in a change in TNM staging in 49 patients (40.5%), including 26.0% in the treatment-naive group and 50.7% in the persistence/recurrence group. In addition to the previously described changes in nodal and distant metastases status, T classification also changed in 14.9% of the entire cohort (treatment-naive, 8.0%; persistence/recurrence, 19.7%) after 68Ga-PSMA-11 PET/CT imaging. By focusing on patients with a TNM change, 11 of 13 (84.6%) treatment-naive patients and 36 of 36 patients (100%) with persistence/recurrence were upstaged after 68Ga-PSMA-11 PET/CT. The changes in TNM classification often led to a different radiotherapeutic approach. Comparing radiotherapeutic management with and without consideration of 68Ga-PSMA-11 PET/CT data in total, a change after 68Ga-PSMA-11 PET/CT occurred in 62 out of 121 men (51.2%) (Fig. 1).
TABLE 2.
Comparison of Conventional and 68Ga-PSMA-11 PET/CT Imaging
| Characteristic | Conventional imaging | 68Ga-PSMA-11 PET/CT | Change | P |
| Local relapse (n) | ||||
| PSA recurrence/persistence | 30/71 (42.3%) | <0.001 | ||
| Total | ||||
| rcTx | 33/71 (46.5%) | 8/71 (11.3%) | ||
| rcT0 | 33/71 (46.5%) | 53/71 (74.6%) | ||
| rcT+ | 5/71 (7.0%) | 10/71 (14.1%) | ||
| Lymph node spread (n) | ||||
| All patients | 15/121 (12.4%) | 39/121 (32.2%) | 25/121 (20.7%)* | <0.001 |
| Initial diagnosis | 5/50 (10.0%) | 8/50 (16.0%) | 5/50 (10.0%) | |
| Intern iliac vessels | — | 2/50 (4.0%)† | ||
| Extern iliac vessels | 5/50 (10.0%)† | 5/50 (10.0%)† | ||
| Presacral | 1/50 (2.0%)† | 2/50 (4.0%)† | ||
| Obturatoric vessels | — | 2/50 (4.0%)† | ||
| Other | — | 1/50 (2.0%)† | ||
| PSA recurrence/persistence | 10/71 (14.1%) | 31/71 (43.7%) | 20/71 (28.2%) | |
| Intern iliac vessels | 3/71 (4.2%)† | 10/71 (14.1%)† | ||
| Extern iliac vessels | 3/71 (4.2%)† | 13/71 (18.3%)† | ||
| Presacral | 3/71 (4.2%)† | 12/71 (16.9%)† | ||
| Obturatoric vessels | — | 2/71 (2.8%)* | ||
| Other | 2/71 (2.8%)† | 8/71 (11.3%)† | ||
| Distant metastases (n) | ||||
| All patients | 26/121 (21.5%) | 41/121 (33.9%) | 23/121 (19.0%)* | <0.001 |
| Initial diagnosis | 3/50 (6.0%) | 5/50 (10.0%) | 7/50 (14.0%) | |
| Lymph nodes | 2/50 (4.0%) | 3/50 (6.0%)† | ||
| Bone | 1/50 (2.0%) | 3/50 (6.0%)† | ||
| Other | — | — | ||
| PSA recurrence/persistence | 23/71 (32.4%) | 36/71 (50.7%) | 16/71 (22.5%) | |
| Lymph nodes | 2/71 (2.8%) | 8/71 (11.3%)† | ||
| Bone | 18/71 (25.4%) | 27/71 (38.0%)† | ||
| Other | 3/71 (4.2%) | 3/71 (4.2%)† |
Also, some patients with change in T staging.
Some patients with multiple forms of lymphatic/distant spread.
FIGURE 1.
Impact of 68Ga-PSMA-11 PET/CT imaging on radiotherapeutic management. RT = radiation therapy.
In subgroup analyses, there was an alteration in the treatment plan in 44.0% of treatment-naive patients and 56.3% of patients with persistence/recurrence (Table 3). In general terms, 68Ga-PSMA-11 PET/CT data resulted in a reduction of the target volume (de-escalation of radiotherapy) in the treatment-naive group (in 68.2%) whereas de-escalation occurred in only 2.5% of the persistence/recurrence group. However, 68Ga-PSMA-11 PET/CT also resulted in substantive changes in the treatment plan without de-escalation in 10.0% of treatment-naive patients and 52.0% of persistence/recurrence patients (Fig. 1; Table 4).
TABLE 3.
Overview of Changes in Radiotherapeutic Management According to 68Ga-PSMA-11 PET/CT
| Characteristic | All patients | Initial diagnosis | PSA persistence/recurrence |
| Individual RT concept (n) | 5/22 (22.7%) | 37/40 (92.5%) | |
| SIB lymph node | 26/62 (41.9%) | 4/22 (18.2%) | 22/40 (55.0%) |
| SBRT lymph node | 5/62 (8.1%) | 1/22 (4.6%) | 4/40 (10.0%) |
| SBRT bone lesion | 6/62 (9.7%) | — | 6/40 (15.0%) |
| Other | 5/62 (8.1%) | — | 5/40 (12.5%) |
| RT de-escalation (n) | 16/62 (25.8%) | 15/22 (68.2%) | 1/40 (2.5%) |
| Palliative treatment (n) | 4/62 (6.5%) | 2/22 (9.1%) | 2/40 (5.0%) |
RT = radiotherapy; SIB = simultaneous integrated boost; SBRT = stereotactic body irradiation.
TABLE 4.
Changes in Radiotherapeutic Management After Additional 68Ga-PSMA-11 PET/CT Imaging in Detail [n, (%)]
| RT after 68Ga-PSMA-11 PET/CT imaging |
||||||||
| Planned RT before 68Ga-PSMA-11 PET/CT imaging | RT prostate only | RT prostate and pLN | RT prostate, pLN and SIB | SBRT only | ADT | Other, individual RT concept | RT pLN and SIB | Systemic chemotherapy |
| RT prostate only | X | — | 4/62 (6.5%) | 2/62 (3.2%) | 1/62 (1.6%) | 1/62 (1.6%) | — | — |
| RT prostate and pLN | 15/62 (24.2%) | X | 8/62 (12.9%) | — | — | 1/62 (1.6%) | — | — |
| RT prostate, pLN, and SIB | — | 1/62 (1.6%) | X | 1/62 (1.6%) | 2/62 (3.2%) | 3/62 (4.8%) | — | — |
| SBRT only | — | — | — | X | — | 3/62 (4.8%) | 1/62 (1.6%) | 1/62 (1.6%) |
| ADT | — | — | — | 5/62 (8.1%) | X | 4/62 (6.5%) | 6/62 (9.7%) | — |
| Individual RT concept | — | — | — | — | — | X | 2/62 (3.2%) | — |
| BSC | — | — | — | 1/62 (1.6%) | — | — | — | — |
RT = radiotherapy; pLN = pelvic lymph nodes; SIB = simultaneous integrated boost; SBRT = stereotactic body radiotherapy, ADT = androgen deprivation therapy; BSC = best supportive care.
DISCUSSION
Radiation therapy usually benefits from more accurate depiction of the anatomic distribution of disease; for a fixed radiation dose, therapy can be directed to areas of higher risk, sparing noninvolved tissues, thus improving the therapeutic index. Because of its high sensitivity for prostate cancer, 68Ga-PSMA-11 PET/CT is helpful in individualizing treatment plans, thereby overcoming the limitations of existing conventional imaging, which is quite insensitive for extraprostatic and recurrent disease. Our study showed that TNM stage changed in 40.5% of patients after 68Ga-PSMA-11 PET/CT. This is comparable to the findings of other studies such as a study of 57 patients in which 68Ga-PSMA-11 PET/CT resulted in a change in TNM classification in 50.8% (14). Schiller et al. reported that 68Ga-PSMA-11 PET/CT changed TNM stage in 45.2% of patients with recurrent disease (19). 68Ga-PSMA-11 PET/CT seems to produce TNM changes in fewer treatment-naive patients. In our cohort, 26.0% of such patients had a TNM stage change after 68Ga-PSMA-11 PET/CT. Confirmatory studies in this population are scarce, however, one prospective study of 431 patients, of whom 108 were treatment-naive and the remainder were recurrent, confirmed that the rate of TNM change was much higher in the latter group (20). In contrast, in a smaller study of 15 treatment-naive prostate cancer patients with patient characteristics comparable to those in our study, TNM change was reported in 53.3% of treatment-naive patients after 68Ga-PSMA-11 PET/CT (13). This wide range of TNM stage alteration in treatment-naive patients may be explained by differences in the aggregate risk of each patient population and by the quality and thoroughness of conventional imaging at different institutions.
The degree to which TNM stage is altered influences the degree of changes in radiotherapeutic management. Lower rates of TNM change have commensurately lower rates of treatment plan changes. In the current study, the radiotherapeutic management was altered in 44.0% of all treatment-naive patients. This contrasts with the results of Dewes et al. (33.3% treatment plan change) and Roach et al. (21.0% treatment plan change) (13,20). In recurrent or persistent disease, 68Ga-PSMA-11 PET/CT led to a change in the radiotherapy plan in 56.3% in this study, which is in accordance with the results of a study of 100 men with recurrent disease in which 68Ga-PSMA-11 PET/CT resulted in a change of radiotherapy planning in 59.0% of cases (21). Schmidt-Hegemann et al. reported 129 men with recurrent prostate carcinoma scanned with 68Ga-PSMA-11 PET/CT and showed changes in the radiotherapy plan in 56.6% (22). These consistent results are also supported by numerous other studies supporting the role of 68Ga-PSMA-11 PET/CT in this setting (11,12,23,24). 68Ga-PSMA-11 PET/CT imaging identification of isolated bone metastases has led to an increase in the use of stereotactic body radiation therapy, and the detection of small nodes often leads to increased use of simultaneous integrated boost to affected areas (Fig. 2). These treatment modifications can be achieved without increased acute toxicity. For instance, Zschaeck et al. reported a low rate of acute toxicity in 21 men treated with 68Ga-PSMA-11 PET/CT–guided radiotherapy. Other studies in patients with recurrent disease confirm these findings and demonstrate superior PSA response rates (25). Similarly, for treatment-naive patients 68Ga-PSMA-11 PET/CT offers the ability to provide an individualized radiotherapeutic treatment approach that boosts radiation to the dominant intraprostatic lesion, with hope resulting in better local control and clinical outcome (26). Interestingly, in as many as 30.0% of the patients in our cohort, 68Ga-PSMA-11 PET/CT results allowed de-escalation of radiotherapy in treatment-naive patients by, for instance, reducing the target volume (usually in pelvic lymph nodes), thus reducing side effects, particularly grade 3+ gastrointestinal adverse events (Fig. 3) (27).
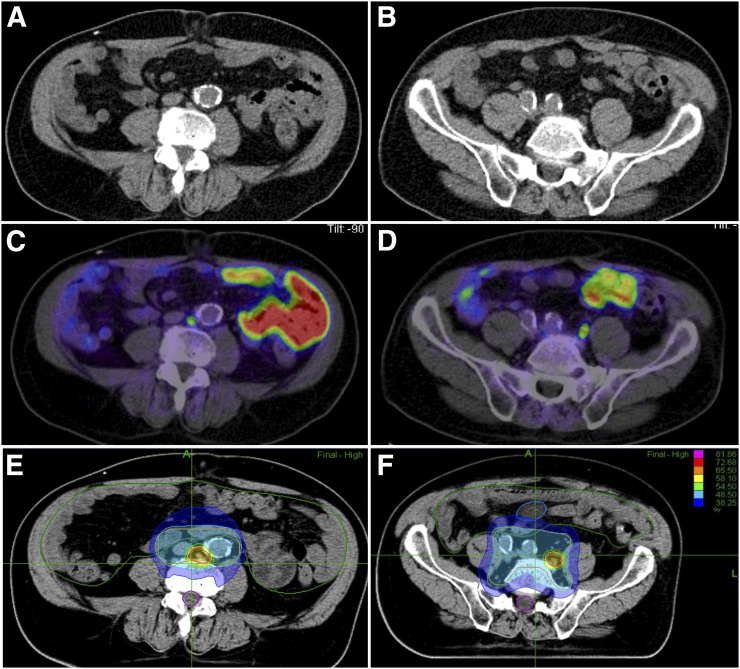
FIGURE 2.
68Ga-PSMA-11 PET/CT (C and D) guided radiotherapy (E and F) with simultaneous integrated boost of 2 lymph node metastases for a high-risk prostate cancer patient with negative CT scan (A and B).
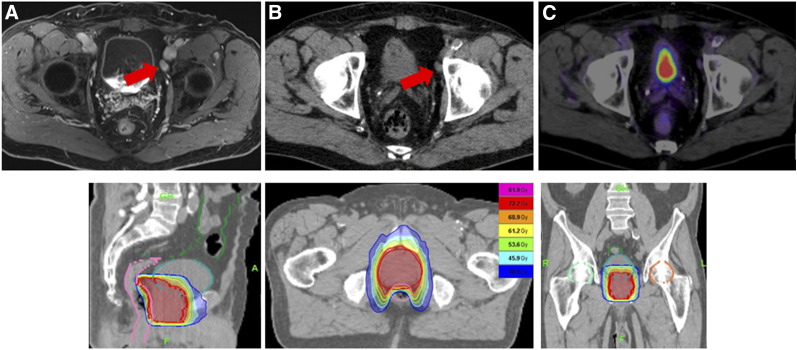
FIGURE 3.
68Ga-PSMA-11 PET/CT–guided radiotherapy de-escalated after 68Ga-PSMA-11 PET/CT. Whereas MRI and CT demonstrated suspicious nodes (A and B), 68Ga-PSMA-11 PET/CT showed no increased activity in nodes (C). Thus, treatment could be planned for prostate itself with reduced doses to pelvic side walls.
Although this is one of the largest studies exploring the effect of 68Ga-PSMA-11 PET/CT on radiation treatment planning, it has several limitations. The major limitation is the retrospective nature of the study, which makes it prone to patient-selection biases. This can be overcome only by prospective randomized trials in which one arm uses 68Ga-PSMA-11 PET/CT to guide therapy and the other arm does not. As 68Ga-PSMA-11 imaging becomes more widely available, such trials will likely be conducted. Also, there was a relatively small number of patients with PSA persistence, although persistence may simply be considered as part of the spectrum of recurrent disease and it may not be necessary to subclassify this group.
CONCLUSION
This study confirms that 68Ga-PSMA-11 PET/CT is well suited to detect intra- and extraprostatic prostate cancer in men with high-risk disease, both at initial diagnosis and at the time of PSA persistence/recurrence. In clinical terms, the use of 68Ga-PSMA-11 PET/CT often results in a change in TNM staging and therefore, radiotherapeutic management. Even when considering that the impact might be greater for men with recurrent disease, this innovative new technology can be seen as a first step toward the realization of individualized radiation oncology for patients with advanced prostate carcinoma.
DISCLOSURE
No other potential conflict of interest relevant to this article was reported.
REFERENCES
- 1.Afshar-Oromieh A, Avtzi E, Giesel FL, et al. The diagnostic value of PET/CT imaging with the 68Ga-labelled PSMA ligand HBED-CC in the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2015;42:197–209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hijazi S, Meller B, Leitsmann C, et al. Pelvic lymph node dissection for nodal oligometastatic prostate cancer detected by 68Ga-PSMA-positron emission tomography/computerized tomography. Prostate. 2015;75:1934–1940. [DOI] [PubMed] [Google Scholar]
- 3.Giesel FL, Sterzing F, Schlemmer HP, et al. Intra-individual comparison of 68Ga-PSMA-11-PET/CT and multi-parametric MR for imaging of primary prostate cancer. Eur J Nucl Med Mol Imaging. 2016;43:1400–1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Perera M, Papa N, Christidis D, et al. Sensitivity, specificity, and predictors of positive 68Ga-prostate-specific membrane antigen positron emission tomography in advanced prostate cancer: a systematic review and meta-analysis. Eur Urol. 2016;70:926–937. [DOI] [PubMed] [Google Scholar]
- 5.Bluemel C, Krebs M, Polat B, et al. 68Ga-PSMA-PET/CT in patients with biochemical prostate cancer recurrence and negative 18F-choline-PET/CT. Clin Nucl Med. 2016;41:515–521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eiber M, Maurer T, Souvatzoglou M, et al. Evaluation of hybrid 68Ga-PSMA ligand PET/CT in 248 patients with biochemical recurrence after radical prostatectomy. J Nucl Med. 2015;56:668–674. [DOI] [PubMed] [Google Scholar]
- 7.Calais J, Cao M, Nickols NG. The utility of PET/CT in the planning of external radiation therapy for prostate cancer. J Nucl Med. 2018;59:557–567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bluemel C, Linke F, Herrmann K, et al. Impact of 68Ga-PSMA PET/CT on salvage radiotherapy planning in patients with prostate cancer and persisting PSA values or biochemical relapse after prostatectomy. EJNMMI Res. 2016;6:78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Verburg FA, Pfister D, Heidenreich A, et al. Extent of disease in recurrent prostate cancer determined by [68Ga]PSMA-HBED-CC PET/CT in relation to PSA levels, PSA doubling time and Gleason score. Eur J Nucl Med Mol Imaging. 2016;43:397–403. [DOI] [PubMed] [Google Scholar]
- 10.Calais J, Czernin J, Cao M, et al. 68Ga-PSMA-11 PET/CT mapping of prostate cancer biochemical recurrence after radical prostatectomy in 270 patients with a PSA level of less than 1.0 ng/mL: impact on salvage radiotherapy planning. J Nucl Med. 2018;59:230–237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Afaq A, Alahmed S, Chen SH, et al. Impact of 68Ga-prostate-specific membrane antigen PET/CT on prostate cancer management. J Nucl Med. 2018;59:89–92. [DOI] [PubMed] [Google Scholar]
- 12.Grubmüller B, Baltzer P, D’Andrea D, et al. 68Ga-PSMA 11 ligand PET imaging in patients with biochemical recurrence after radical prostatectomy: diagnostic performance and impact on therapeutic decision-making. Eur J Nucl Med Mol Imaging. 2018;45:235–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dewes S, Schiller K, Sauter K, et al. Integration of 68Ga-PSMA-PET imaging in planning of primary definitive radiotherapy in prostate cancer: a retrospective study. Radiat Oncol. 2016;11:73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sterzing F, Kratochwil C, Fiedler H, et al. 68Ga-PSMA-11 PET/CT: a new technique with high potential for the radiotherapeutic management of prostate cancer patients. Eur J Nucl Med Mol Imaging. 2016;43:34–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Eder M, Schafer M, Bauder-Wust U, et al. 68Ga-complex lipophilicity and the targeting property of a urea-based PSMA inhibitor for PET imaging. Bioconjug Chem. 2012;23:688–697. [DOI] [PubMed] [Google Scholar]
- 16.Schäfer M, Bauder-Wust U, Leotta K, et al. A dimerized urea-based inhibitor of the prostate-specific membrane antigen for 68Ga-PET imaging of prostate cancer. EJNMMI Res. 2012;2:23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Eder M, Neels O, Muller M, et al. Novel Preclinical and radiopharmaceutical aspects of [68Ga]Ga-PSMA-HBED-CC: a new PET tracer for imaging of prostate cancer. Pharmaceuticals (Basel). 2014;7:779–796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.D’Amico AV, Whittington R, Malkowicz SB, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280:969–974. [DOI] [PubMed] [Google Scholar]
- 19.Schiller K, Sauter K, Dewes S, et al. Patterns of failure after radical prostatectomy in prostate cancer: implications for radiation therapy planning after 68Ga-PSMA-PET imaging. Eur J Nucl Med Mol Imaging. 2017;44:1656–1662. [DOI] [PubMed] [Google Scholar]
- 20.Roach PJ, Francis R, Emmett L, et al. The impact of 68Ga-PSMA PET/CT on management intent in prostate cancer: results of an Australian prospective multicenter study. J Nucl Med. 2018;59:82–88. [DOI] [PubMed] [Google Scholar]
- 21.Habl G, Sauter K, Schiller K, et al. 68 Ga-PSMA-PET for radiation treatment planning in prostate cancer recurrences after surgery: individualized medicine or new standard in salvage treatment. Prostate. 2017;77:920–927. [DOI] [PubMed] [Google Scholar]
- 22.Schmidt-Hegemann NS, Fendler WP, Buchner A, et al. Detection level and pattern of positive lesions using PSMA PET/CT for staging prior to radiation therapy. Radiat Oncol. 2017;12:176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gupta SK, Watson T, Denham J, et al. Prostate-specific membrane antigen positron emission tomography-computed tomography for prostate cancer: distribution of disease and implications for radiation therapy planning. Int J Radiat Oncol Biol Phys. 2017;99:701–709. [DOI] [PubMed] [Google Scholar]
- 24.Kabasakal L, Demirci E, Nematyazar J, et al. The role of PSMA PET/CT imaging in restaging of prostate cancer patients with low prostate-specific antigen levels. Nucl Med Commun. 2017;38:149–155. [DOI] [PubMed] [Google Scholar]
- 25.Zschaeck S, Wust P, Beck M, et al. Intermediate-term outcome after PSMA-PET guided high-dose radiotherapy of recurrent high-risk prostate cancer patients. Radiat Oncol. 2017;12:140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Koerber SA, Utzinger MT, Kratochwil C, et al. 68Ga-PSMA-11 PET/CT in newly diagnosed carcinoma of the prostate: correlation of intraprostatic PSMA uptake with several clinical parameters. J Nucl Med. 2017;58:1943–1948. [DOI] [PubMed] [Google Scholar]
- 27.Roach M, 3rd, DeSilvio M, Valicenti R, et al. Whole-pelvis, “mini-pelvis,” or prostate-only external beam radiotherapy after neoadjuvant and concurrent hormonal therapy in patients treated in the Radiation Therapy Oncology Group 9413 trial. Int J Radiat Oncol Biol Phys. 2006;66:647–653. [DOI] [PubMed] [Google Scholar]